Abstract
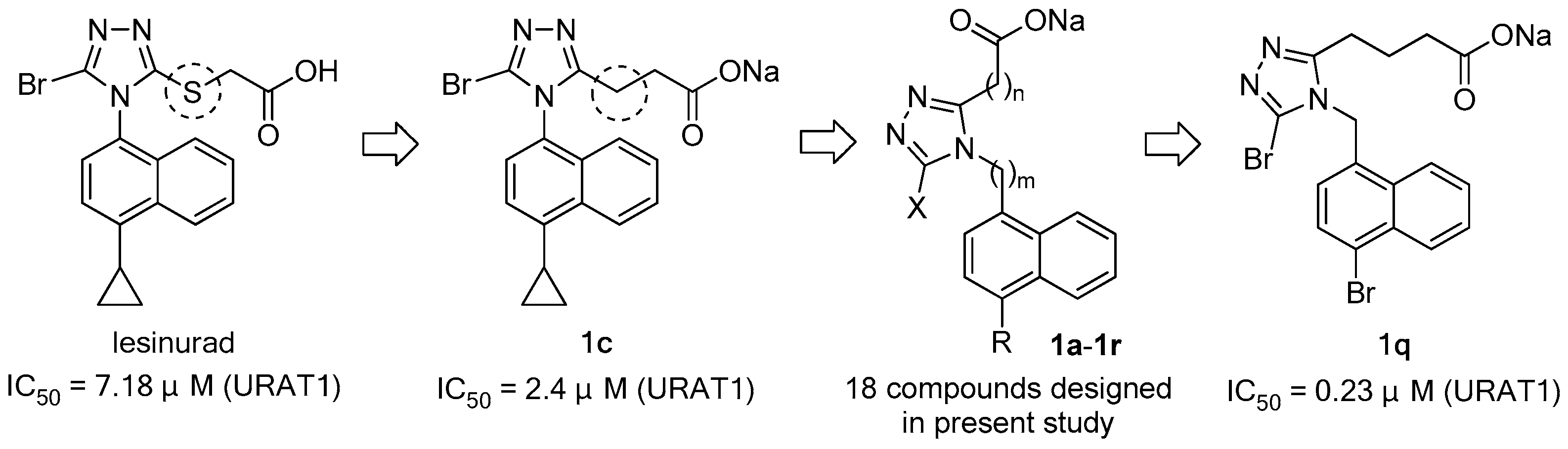
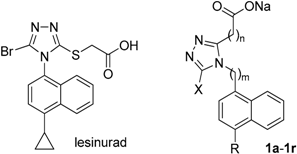
In order to systematically explore and understand the structure–activity relationship (SAR) of a lesinurad-based hit (1c) derived from the replacement of the S atom in lesinurad with CH2, 18 compounds (1a–1r) were designed, synthesized and subjected to in vitro URAT1 inhibitory assay. The SAR exploration led to the discovery of a highly potent flexible URAT1 inhibitor, 1q, which was 31-fold more potent than parent lesinurad (IC50 = 0.23 μM against human URAT1 for 1q vs 7.18 μM for lesinurad). The present study discovered a flexible molecular scaffold, as represented by 1q, which might serve as a promising prototype scaffold for further development of potent URAT1 inhibitors, and also demonstrated that the S atom in lesinurad was not indispensable for its URAT1 inhibitory activity.
1. Introduction
Gout, which is characterized by recurrent joint swelling and pain, is the most prevalent form of inflammatory arthritis. It is caused by the deposition of monosodium urate (MSU) in joints and soft tissues [1]. If left untreated or inadequately managed, gout will lead to permanent joint destruction, bone erosion, and kidney impairment, dramatically affecting patients’ quality of life and even threatening their lives [2]. More recently, a deeper understanding of the gout pathophysiology has led to the appreciation that gout impacts patients with consequences well beyond the episodes of acute inflammatory arthritis, and multiple lines of evidence have been accumulated to demonstrate that hyperuricemia was the independent risk factor for hypertension, hyperlipidemia, diabetes, cardiovascular diseases, etc. [3,4]. Persisted hyperuricemia is the prerequisite of MSU formation and deposition, which is defined as the elevation of serum uric acid (sUA) levels above the saturation point of MSU in the fluid at physiological pH and temperature, i.e., 6.8 mg/dL (404 μmol/L) [1]. Approximately 98% of the uric acid (pH = 5.75) is in the ionized form, i.e., urate anion, in the extracellular compartment (pH = 7.4), and due to the existence of a high concentration of sodium ion in the extracellular compartment, urate largely presents as MSU [2,5]. Gout is one of the earliest diseases recognized by physicians in history [6]. The last few decades have witnessed the rising prevalence of gout and hyperuricemia in both developed and developing countries [7,8,9], which presents significant burdens on the individual and community in many respects [10,11].
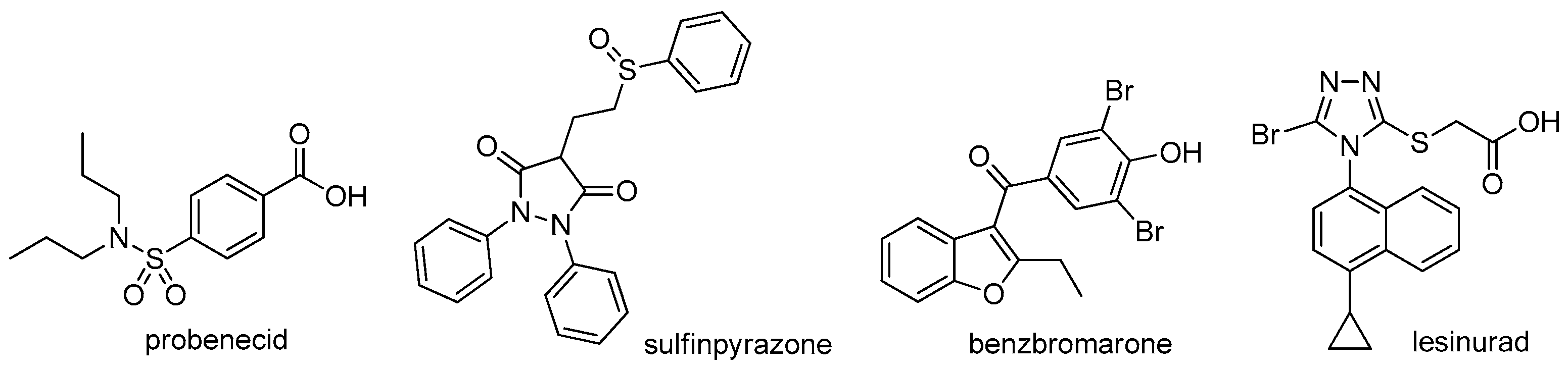
Acute gout attacks have been traditionally treated with colchicine, nonsteroidal anti-inflammatory drugs (NSAIDs), and corticosteroids, agents that often reduce gout flares but that are also associated with many adverse effects [12]. However, the fundamental of treatment of gout is to dissolve MSU crystals by urate-lowering therapy (ULT) with xanthine oxidase inhibitors (XOIs) such as allopurinol and febuxostat, uricosuric agents such as probenecid, sulfinpyrazone, and benzbromarone, and uricase such as pegloticase [13,14]. The target of ULT is to maintain sUA < 6 mg/dL (360 μmol/L) and even <5 mg/dL (300 μmol/L) for severe gout, as the guidelines recommend [1,2]. XOIs are usually used as first-line ULT, albeit with numerous severe adverse effects and often with low response rates, and when patients are refractory to or contraindicated for XOIs, uricosuric agents are used as second-line therapy [2,15]. Uricases are only indicated for patients with severe gout, such as those with tophaceous deformities and complications, which, however, are associated with infusion reactions and loss of urate-lowering efficacy within a few months of treatment due to the development of antibodies [15].
Unlike in other species, in humans and in most non-human primates, uric acid is the final metabolite of dietary and endogenous purine metabolism. Humans lack uricase, an enzyme capable of converting uric acid to allantoin, which is 5–10 times more water-soluble than uric acid and is therefore much more readily eliminated [2,16]. Hyperuricemia is caused by over-production and/or under-excretion of urate, and among the patients with hyperuricemia, 90% of them are urate under-excretors, while 10% are over-producers [1]. Approximately one-third of urate is excreted via the gastrointestinal tract, and the remaining two-thirds via kidney [2]. Most of the urate filtered in kidney is reabsorbed, and this process is mainly mediated by uric acid transporter 1 (URAT1), also known as urate-anion exchanger 1, which is prominently expressed in epithelial cells of proximal tubules in the renal cortex [17]. Given the fact that 90% of the patients with hyperuricemia are urate under-excretors, URAT1 inhibitors, which lower sUA by inducing uricosuria, were believed to be a very promising class of uricosuric agents for the treatment of hyperuricemia and gout [18]. A number of uricosuric agents that had already been approved were later found to be UART1 inhibitors after the identification of URAT1 in 2002 (Figure 1) [17]; however, these agents are associated with a variety of severe adverse effects or drawbacks, such as severe hepatotoxicity of benzbromarone, drug interactions of probenecid with NSAIDs, penicillin, heparin, etc., and limited efficacy of probenecid and sulfinpyrazone [15,17]. Besides, some of them are not available in specific countries. For example, benzbromarone was withdrawn in Europe in 2003 and has never been approved in the US because of severe hepatotoxicity. Lesinurad, a novel URAT1 inhibitor developed by Ardea Biosciences and AstraZeneca, was approved by the FDA at the end of 2015 for the treatment of hyperuricemia associated with gout in combination with an XOI [19]. In a study on lesinurad derivatives as URAT1 inhibitors in our laboratories, we were surprised to find that compound 1c, the sodium salt of 10b derived from the replacement of the S atom in lesinurad with CH2, was three-fold more potent than parent lesinurad in in vitro URAT1 inhibitory assay (Figure 2). This preliminary finding suggested that the S atom might not be indispensable and its replacement with CH2 could even increase the URAT1 inhibitory activity. Encouraged by this preliminary finding and promising hypothesis, we carried out a systematic structure–activity relationship (SAR) exploration of this interesting molecular scaffold, with the expectation of a better understanding of its SAR. Fortunately, the systematic SAR exploration led to the discovery of 1q as a highly potent URAT1 inhibitor, which was 31-fold more active than parent lesinurad in in vitro URAT1 inhibitory assay (IC50 = 0.23 μM against human URAT1 for 1q vs. 7.18 μM for lesinurad).
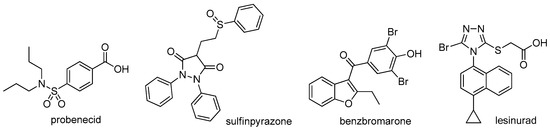
Figure 1.
Structures of approved URAT1 inhibitors.
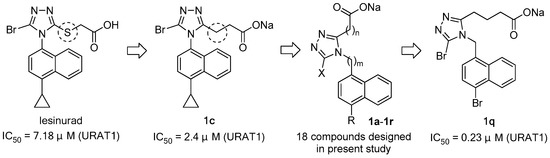
Figure 2.
Design of URAT1 inhibitors in present study and discovery of 1q.
2. Results and Discussion
2.1. Chemistry
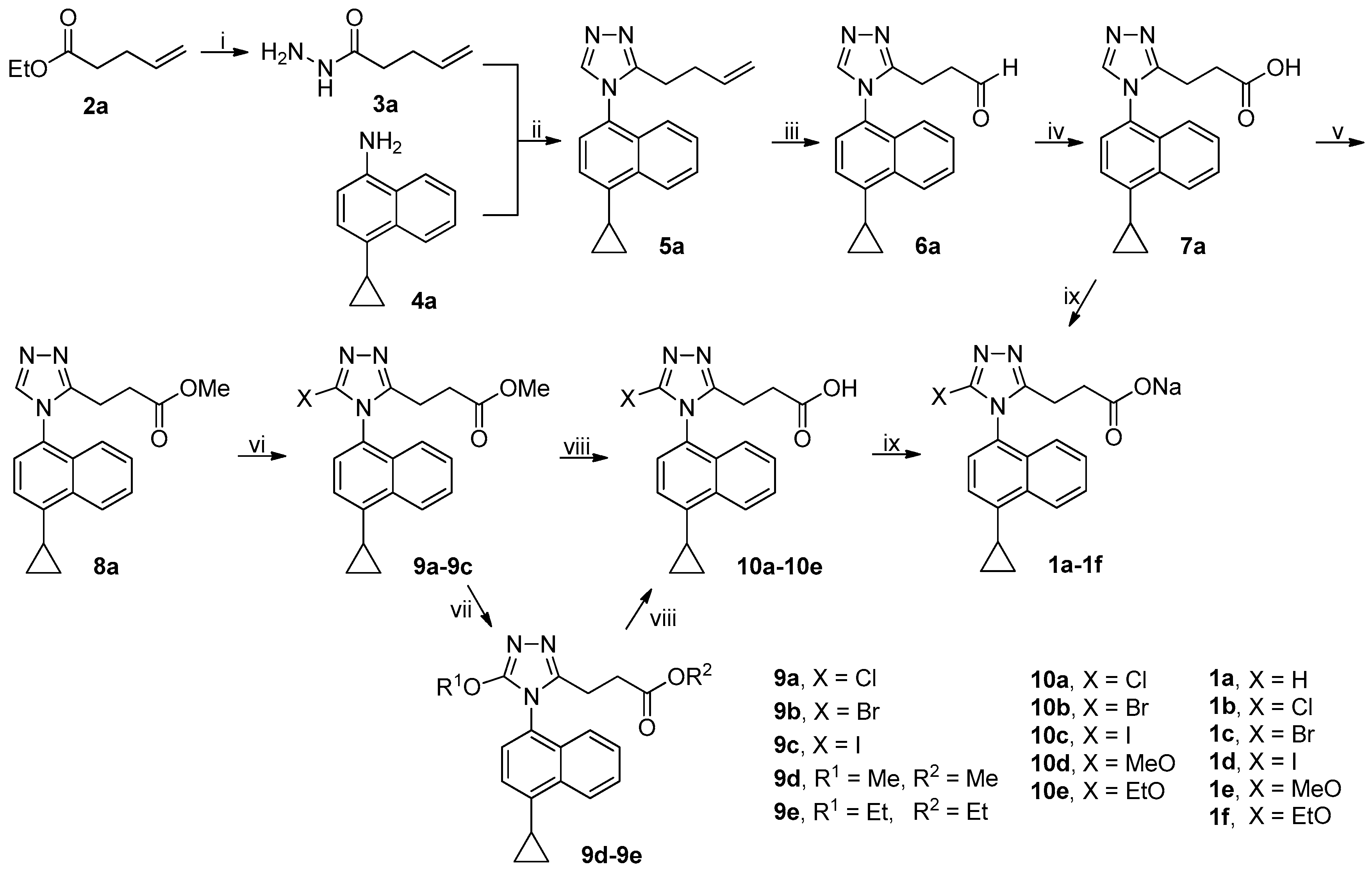
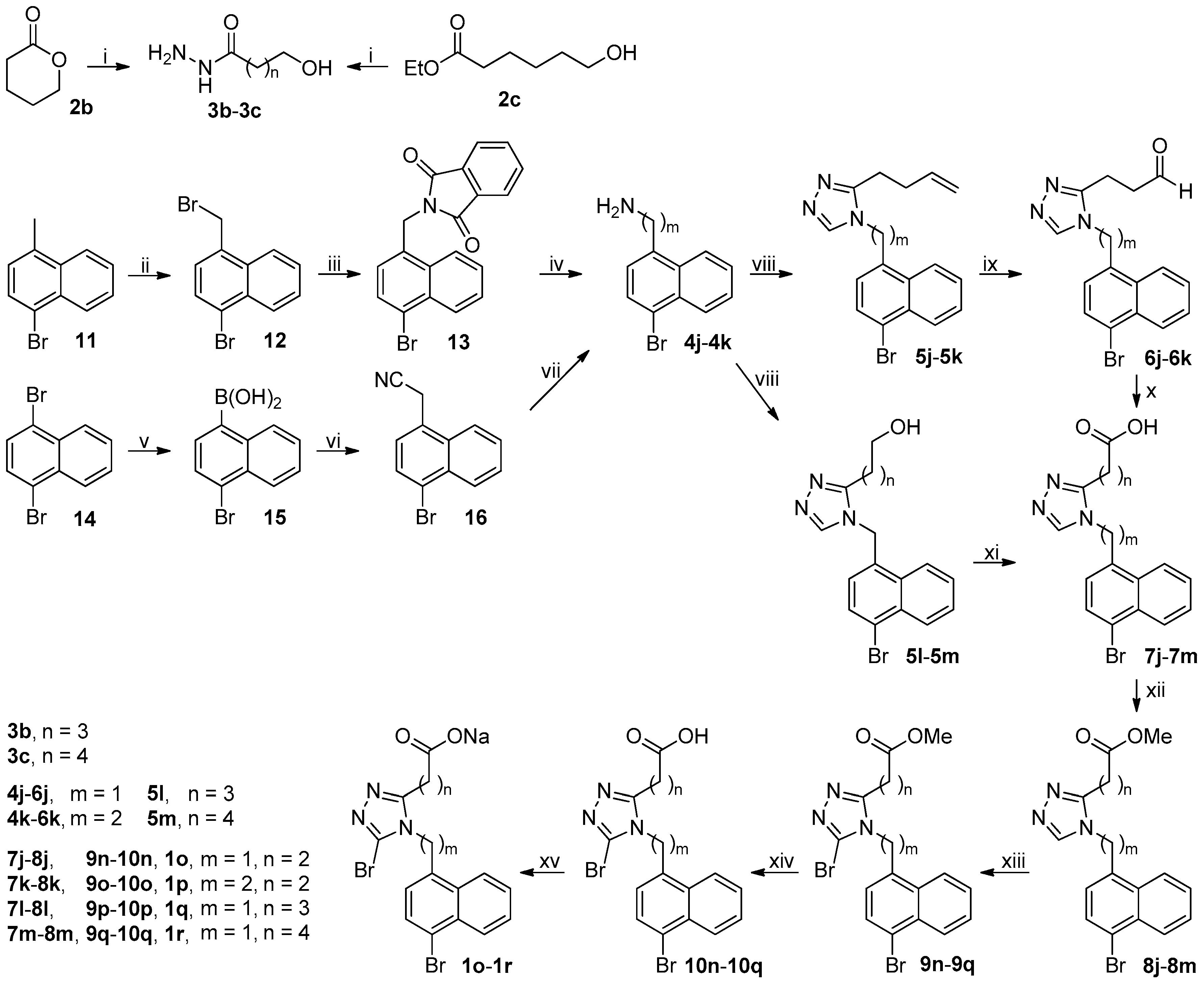
The synthetic route to target compounds 1a–1f was shown in Scheme 1. Ester 2a was treated with 80% aqueous hydrazine hydrate in methanol at room temperature to smoothly give acyl hydrazide 3a. An efficient one-pot, three-component synthetic approach was employed to construct the desired 1,2,4-triazole core, which involved the initial condensation of 3a and N,N-dimethylformamide dimethyl acetal (DMFDMA) in acetonitrile at 50 °C in an open vessel to produce an adduct that in turn reacted with amine 4a added subsequently in refluxing acetic acid to furnish 1,2,4-triazole 5a via ring closing [20]. Dihydroxylation of olefin 5a by treatment of 5a with osmium tetroxide and N-methylmorpholine N-oxide (NMMO) in THF/H2O (4/1) at room temperature afforded the corresponding diol, which, after isolation and purification, was further treated with sodium periodate in THF/H2O (4/1) at room temperature to yield aldehyde 6a [21]. Pinnick oxidation of aldehyde 6a with NaClO2 in the presence of NaH2PO4 and 2-methyl-2-butene in t-BuOH/H2O (4/1) at room temperature cleanly produced the corresponding carboxylic acid 7a [22]. Esterification of 7a with methanol in the presence of 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDCI) and 4-dimethylaminopyridine (DMAP) in dichloromethane at room temperature smoothly produced ester 8a. However, an initial attempt to achieve this esterification with 1,3-dicyclohexylcarbodiimide (DCC) in replacement of EDCI was unsatisfactory because an appreciable amount of intermediate resulting from the adduction of 7a and DCC was observed even in refluxing tetrahydrofuran, which eventually resulted in a quite low yield of 8a. Halogenation at 5-position of 1,2,4-triazole ring in 8a was achieved by treatment of 8a with N-halosuccinimides (NCS, NBS and NIS) in acetonitrile at room temperature (NBS) or at reflux (NCS and NIS), leading to the formation of 9a–9c. Aromatic nucleophilic substitution of the Br atom in 9b by treatment of 9b with MeONa and EtONa in corresponding alcohols at reflux successfully produced 9d and 9e, respectively, albeit in quite low yields (14% for 9d and 13% for 9e). It should be noted that methyl/ethyl transesterification occurred in the case of 9e. Alkaline hydrolysis of esters 9a–9e with aqueous LiOH in methanol at room temperature afforded corresponding carboxylic acids 10a–10e. Finally, acids 7a and 10a–10e were converted to sodium salts thereof 1a–1f with aqueous NaOH in methanol for the sake of improving the solubility in in vitro URAT1 inhibitory assay.
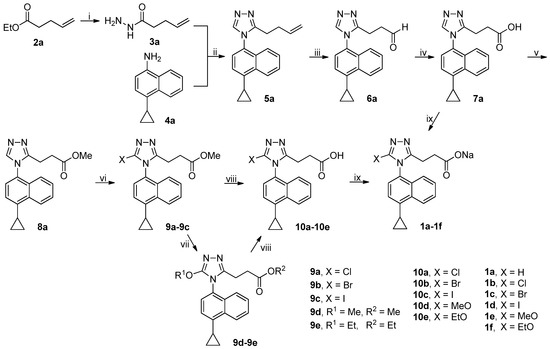
Scheme 1.
Synthetic route to 1a–1f. Reagents and conditions: (i) 80% N2H4·H2O, MeOH, r.t.; (ii) (a) DMFDMA, 3a, MeCN, 50 °C, open vessel; (b) 4a, AcOH, reflux; (iii) (a) NMMO, OsO4, THF/H2O (4/1), r.t.; (b) NaIO4, THF/H2O (4/1), r.t.; (iv) NaClO2, NaH2PO4, 2-methyl-2-butene, t-BuOH/H2O (4/1), 0 °C-rt; (v) MeOH, EDCI, DMAP, CH2Cl2, 0 °C-rt, N2; (vi) NCS (9a) or NIS (9c), MeCN, reflux; NBS (9b), MeCN, r.t.; (vii) for 9d, MeONa, MeOH, reflux, N2; for 9e, EtONa, EtOH, reflux, N2; (viii) aq LiOH, EtOH, r.t.; (ix) aq NaOH, MeOH, r.t.
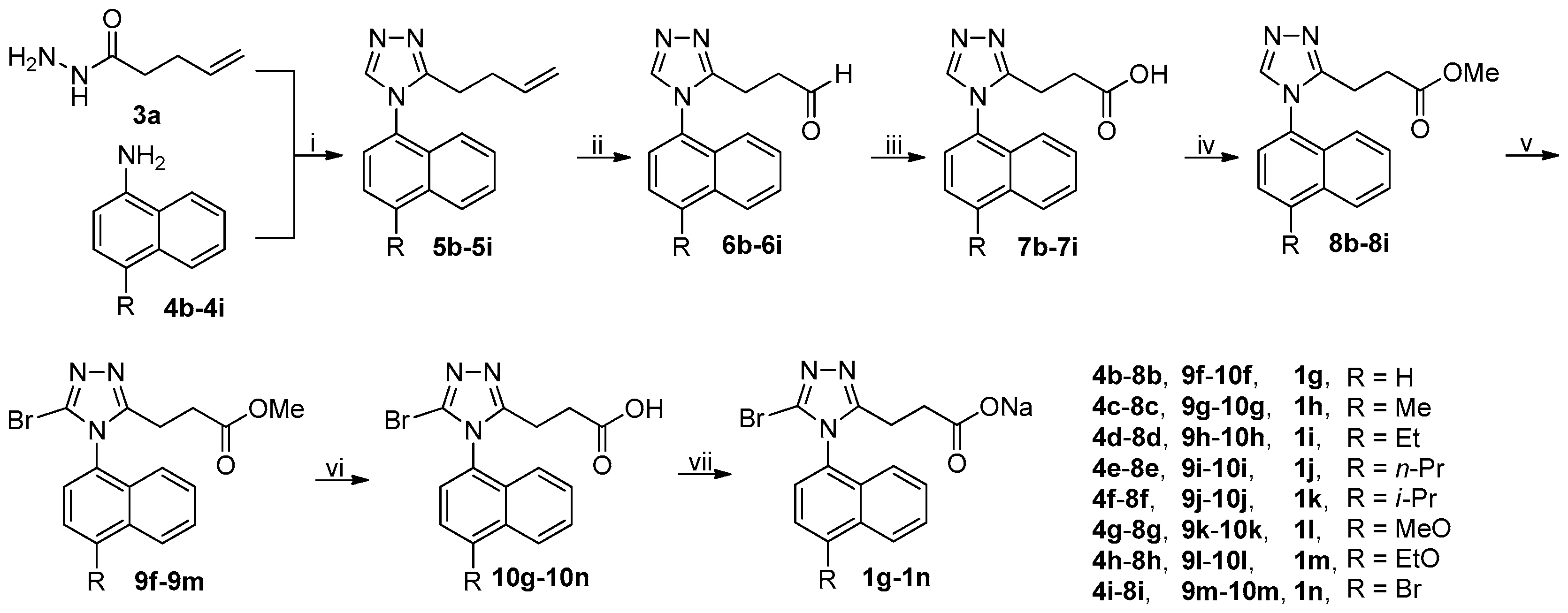
The synthetic route to target compounds 1g–1n was summarized in Scheme 2. Following the identical procedure used for the synthesis of 1c starting from 3a and 4a, target compounds 1g–1n were successfully prepared from 3a and 4b–4i. It should be noted that brominations of 8g and 8h with NBS at room temperature led to the formation of large amounts of byproducts (byproducts/9k or 9l = 55/45) that were preliminarily identified by 1H-NMR to result from the bromination at the naphthalene ring instead of the desired 5-position of 1,2,4-triazole ring. This phenomenon is consistent with the theoretical prediction that the naphthalene ring activated by strongly electron-donating alkoxy groups readily undergoes electrophilic substitution; on the contrary, the bromination of 8i needed elevated temperature (60 °C) to reach a reasonable reaction rate due to the presence of a weakly electron-withdrawing Br atom at 4-position of the naphthalene ring. In an unsuccessful attempt, it was experimentally found that a counterpart of 8i with R being NO2, a strongly electron-withdrawing group, could not even be brominated with NBS even in refluxing acetonitrile.
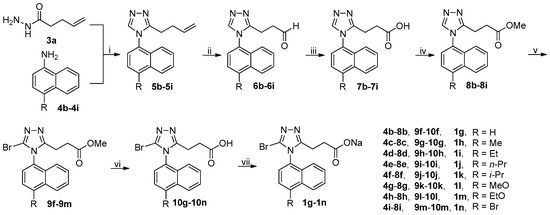
Scheme 2.
Synthetic route to 1g–1n. Reagents and conditions: (i) DMFDMA, 3a, MeCN, 50 °C, open vessel; (b) then 4b–4i, AcOH, reflux; (ii) (a) NMMO, OsO4, THF/H2O (4/1), r.t.; (b) NaIO4, THF/H2O (4/1), r.t.; (iii) NaClO2, NaH2PO4, 2-methyl-2-butene, t-BuOH/H2O (4/1), 0 °C-rt; (iv) MeOH, EDCI, DMAP, CH2Cl2, 0 °C-rt, N2; (v) NBS, MeCN, r.t. (for 8b–8h) or 60 °C (for 8i); (vi) aq LiOH, EtOH, r.t.; (vii) aq NaOH, MeOH, r.t.
The synthetic route to target compounds 1o–1r was depicted in Scheme 3. Esters 2b and 2c were hydrazinolyzed to acyl hydrazides 3b and 3c, respectively, following the procedure used for the synthesis of 3a from 2a except that the hydrazinolysis of 2c needed to be carried out at reflux. Compound 11 was treated with NBS in the presence of benzoyl peroxide (BPO) in refluxing n-hexane to furnish 12, which was in turn transformed to corresponding amine 4j via Gabriel synthesis involving the reaction of 12 and potassium naphthalimide in DMF at 100 °C to give 13 and subsequent hydrazinolysis of 13 with 80% aqueous hydrazine hydrate in refluxing ethanol to yield 4j [23]. Transformation of bisbromide 14 to mono-boronic acid 15 was achieved by treatment of 14 with 1 eq of n-BuLi at −78 °C followed by quenching with triisopropyl borate. Aryl boronic acid 15 was coupled with aminoacetonitrile to afford aryl acetonitrile 16 according to an efficient deaminative coupling approach which involved the reaction of 15 and aminoacetonitrile hydrochloride in the presence of sodium nitrite in toluene/H2O (20/1) at 50 °C [24]. An initial attempt to construct aryl acetonitrile 16 by Suzuki coupling, which involved the reaction of 15 and chloroacetonitrile under a well-established Pd-catalyzed condition [25,26], was unsuccessful in that a complex mixture of unidentified products was formed presumably due to the competitive reactions resulting from the presence of two cross-coupling acceptors (aryl bromide 15 and chloroacetonitrile) in this case. Reduction of aryl acetonitrile 16 with lithium aluminum hydride in dried THF at room temperature smoothly afforded desired amine 4k. Amines 4j–4k and hydrazide 3a were transformed to 7j–7k following the same procedure used for the synthesis of 7a from 4a and 3a. On the other hand, amine 4j and hydrazides 3b–3c were subjected to the same procedure used for the synthesis of 5a from 4a and 3a to produce 5l–5m. TEMPO/NaClO-catalyzed oxidation of 5l–5m with sodium chlorite in acetonitrile/phosphate buffer (pH = 6.7) at 35 °C smoothly produced corresponding carboxylic acids 7l–7m, respectively [27]. Carboxylic acids 7j–7m were transformed to target compounds 1o–1r following the procedure for the synthesis of 1c from 7a.
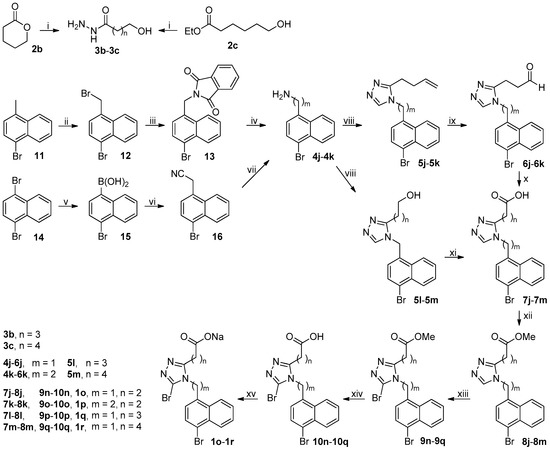
Scheme 3.
Synthetic route to 1o–1r. Reagents and conditions: (i) 80% N2H4·H2O, MeOH, r.t. (2b) or reflux (2c); (ii) NBS, BPO, n-hexane, reflux, N2; (iii) potassium phthalimide, DMF, 100°C, N2; (iv) 80% N2H4·H2O, EtOH, reflux; (v) (a) n-BuLi (1.0 eq), THF, −78 °C; (b) B(i-PrO)3, −78 °C-rt; (vi) CNCH2NH2·HCl, NaNO2, PhMe/H2O (20/1), 50 °C; (vii) LiAlH4, THF, 0 °C-rt, N2; (viii) (a) DMFDMA, 3a–3c, MeCN, 50 °C, open vessel; (b) 4j–4k, AcOH, reflux; (ix) (a) NMMO, OsO4, THF/H2O (4/1), r.t.; (b) NaIO4, THF/H2O (4/1), r.t.; (x) NaClO2, NaH2PO4, 2-methyl-2-butene, t-BuOH/H2O (4/1), 0 °C-rt; (xi) NaClO2, TEMPO, NaClO, MeCN, phosphate buffer (pH = 6.7), 35 °C; (xii) MeOH, EDCI, DMAP, CH2Cl2, 0 °C-rt, N2; (xiii) NBS, MeCN, r.t.; (xiv) aq LiOH, EtOH, r.t.; (xv) aq NaOH, MeOH, r.t.
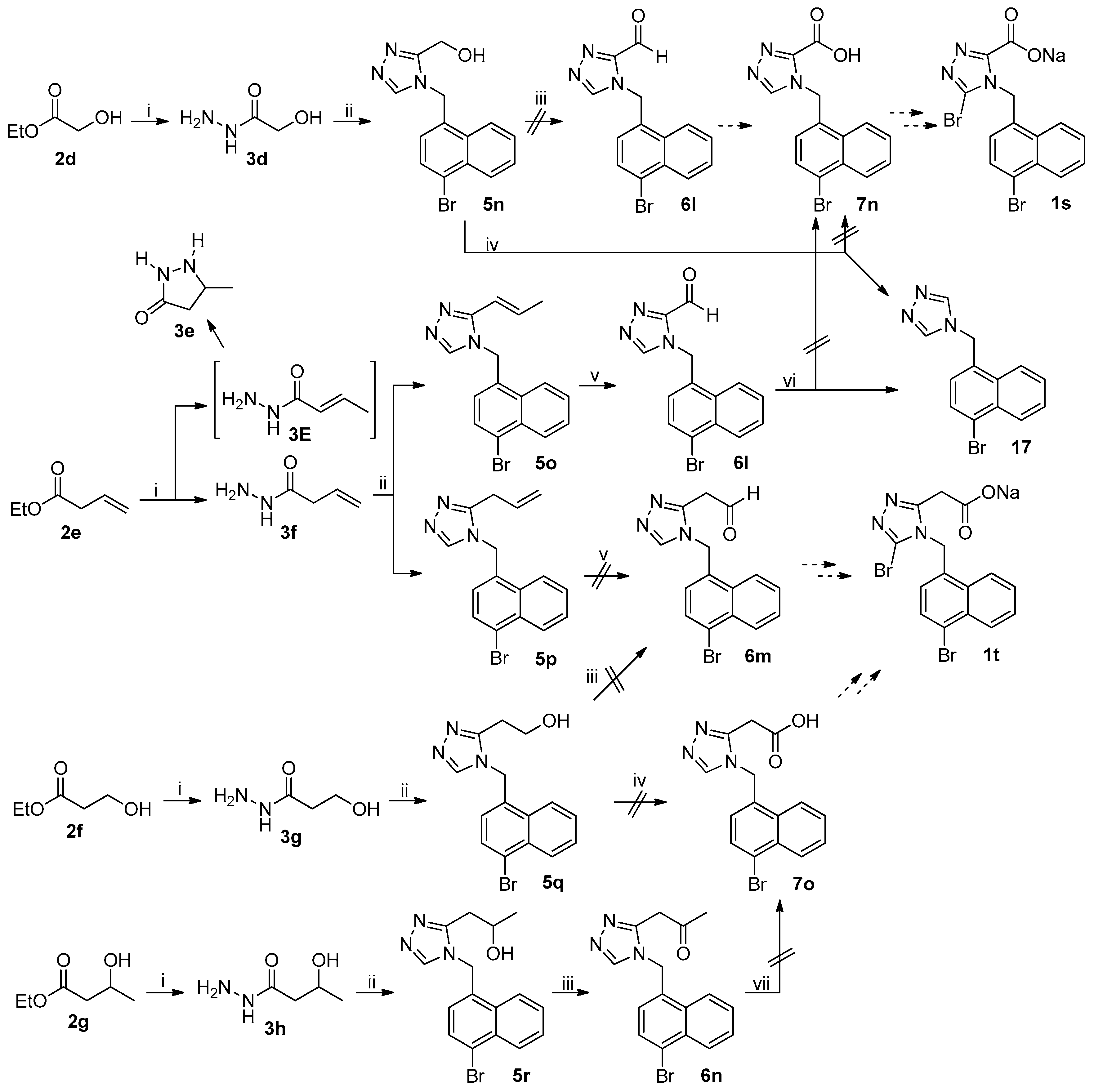
The unsuccessful synthetic routes to anticipated target compounds 1s and 1t were depicted in Scheme 4. Ester 2d was transformed to 5n according to the procedure used for the synthesis of 5l–5m from 2b–2c described above. Swern oxidation of triazolylmethanol 5n to corresponding triazolylaldehyde 6l involving the treatment of 5n with (COCl)2/DMSO/Et3N [28] was unsuccessful in that a complex mixture of unidentified products was formed. Aldehyde 6l was initially expected to be transformed to anticipated target compound 1s by the procedure used for the synthesis of 1c from 6a. Direct oxidation of primary alcohol 5n to corresponding carboxylic acid 7n with TEMPO/NaClO-catalyzed oxidation, aforementioned, to avoid the involvement of aldehyde 6l was also unsuccessful because 17 was isolated as the major product that clearly resulted from the decarboxylation of 7n, strongly indicating that 7n was probably an unstable compound. Hydrazinolysis of 2e under the reaction conditions described above led to the formation of 3e and 3f in a ratio of approximately 1/9; the isolation of 3e unambiguously suggested the formation of 3E during the hydrazinolysis of 2e, an isomer of 3f resulting from the C=C double bond migration [29]. 1,2,4-Triazole formation from 3f and 4j under the reaction conditions as described above led to the formation of 5o and 5p in a ratio of approximately 1/5, suggesting that the C=C double bond migration also occurred to a notable extent in this reaction. Transformation of olefin 5o to aldehyde 6l according to the procedure used for the transformation of 5a to 6a described above was indeed successful, but further oxidation of aldehyde 6l to carboxylic acid 7n by Pinnick oxidation, aforementioned, failed due to the isolation of decarboxylated product 17 once again, further confirming that triazolylcarboxylic acid 7n was not a stable compound (but the corresponding triazolylaldehyde 6l was). On the other hand, the cleavage of olefin 5p with anticipation to furnish acetaldehyde 6m as described above failed due to the formation of a complex mixture of unidentified products; the latter was initially expected to be used for the synthesis of anticipated target compounds 1t. Ethyl alcohol 5q was prepared from 2f by the procedure used for the synthesis of 5l–5m from 2b–2c. Oxidation of 5q to acetaldehyde 6m by Swern oxidation described above also failed, strongly suggesting that triazolylacetaldehyde 6m was unstable. Direct oxidation of 5q to corresponding acetic acid 7o also failed, leading to a complex mixture of unidentified products. Secondary alcohol 5r was prepared from 2g following the procedure described above. Swern oxidation of 5r under the reaction conditions described above successfully furnished corresponding acetone 6n, but further transformation of methyl ketone 6n to acetic acid 7o by haloform reaction [30] also failed, indicating that triazolylacetic acid 7o was probably unstable.
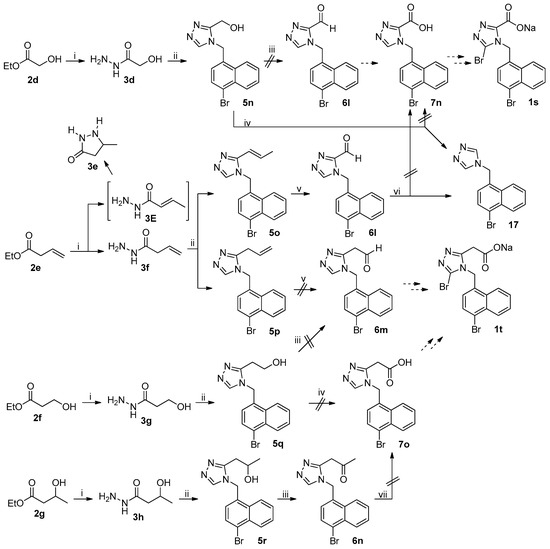
Scheme 4.
Unsuccessful synthetic route to 1s–1t. Reagents and conditions: (i) 80% N2H4·H2O, MeOH, r.t.; (ii) (a) DMFDMA, 3d–3h, MeCN, 50 °C, open vessel; (b) 4j, AcOH, reflux; (iii) (a) (COCl)2, DMSO, 5n, 5q, or 5r, CH2Cl2, −60 °C; (b) Et3N, −60 °C to r.t.; (iv) NaClO2, TEMPO, NaClO, MeCN, phosphate buffer (pH = 6.7), 35 °C; (v) (a) NMMO, OsO4, THF/H2O (4/1), r.t.; (b) NaIO4, THF/H2O (4/1), r.t.; (vi) NaClO2, NaH2PO4, 2-methyl-2-butene, t-BuOH/H2O (4/1), 0 °C-rt; (vii) I2 or Br2, NaOH, 1,4-dioxane/H2O, rt-reflux.
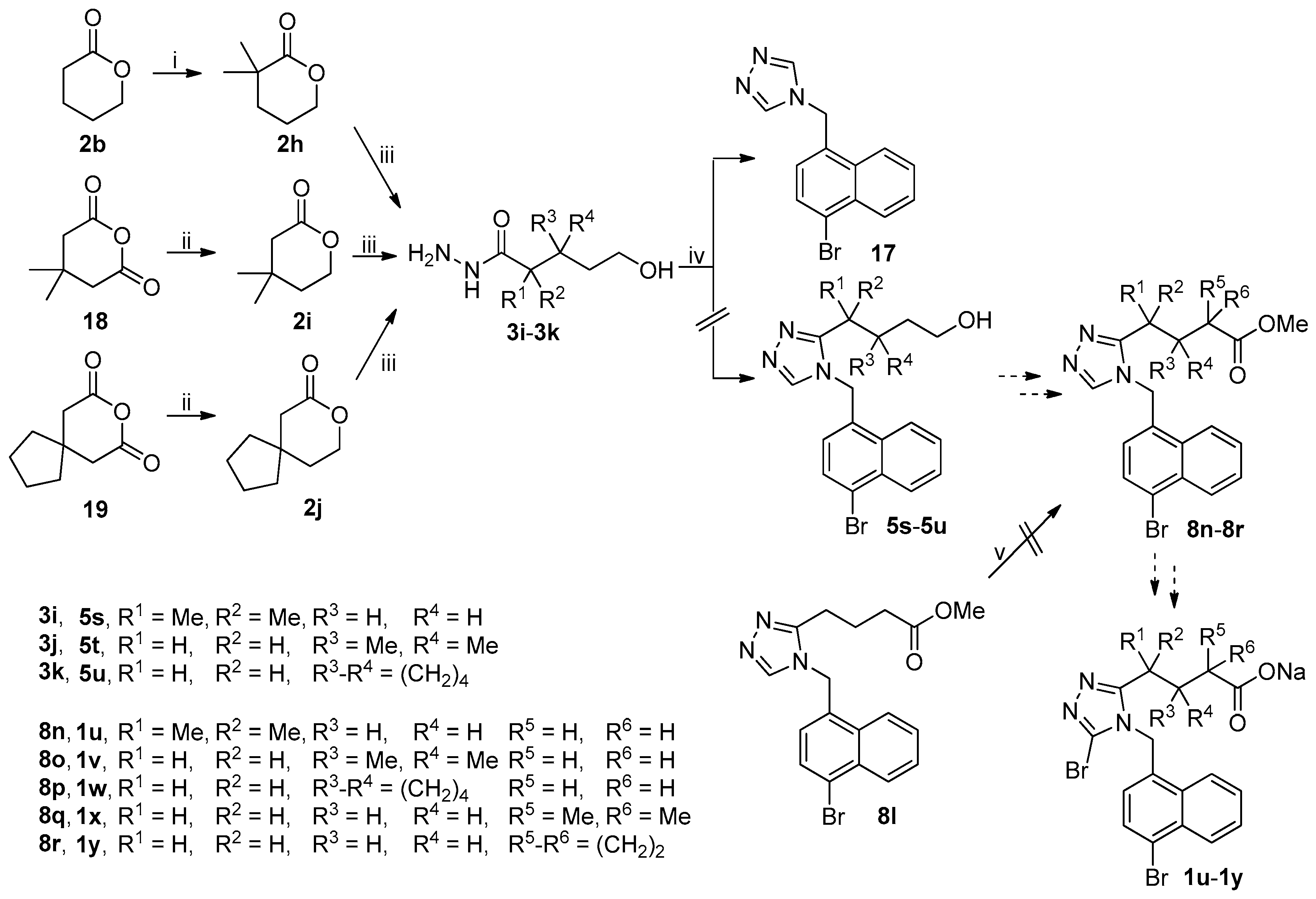
The unsuccessful synthetic routes to anticipated target compounds 1u–1y were depicted in Scheme 5. Ester 2b was treated with lithium diisopropylamide (LDA) generated in situ from n-BuLi and diisopropylamine in dried THF at −78 °C and then quenched by methyl iodide and 1,3-dimethyltetrahydropyrimidin-2(1H)-one (DMPU) at −78 °C to room temperature and this process was repeated twice to give rise to 2h with the gem-dimethyl group at the α-position of carboxylate [31,32]. Reduction of anhydrides 18 and 19 with sodium borohydride in THF at 0 °C to room temperature followed by treatment with 6 M hydrochloric acid afforded esters 2i and 2j, respectively [33]. Hydrazinolysis of 2h–2j under the reaction conditions described above gave acyl hydrazides 3i–3k. 1,2,4-Triazole ring formation with acyl hydrazides 3i–3k and amine 4j under the reaction conditions described above failed because all three reactions uniformly yielded 17 as the major product instead of the desired 5s–5u which were initially expected to be employed for the synthesis of anticipated target compounds 1u–1w. Attempts to introduce a gem-dimethyl group via the approach for the synthesis of 2h from 2b described above and a cyclopropyl group via sequential deprotonation with LDA and quenching with 1,3,2-dioxathiolane-2,2-dioxide [34] at the α-position of carboxylate in 8l to prepare 8q and 8r, respectively, were unsuccessful because a complex mixture of unidentified products was formed in both cases. Esters 8q and 8r were initially expected to be used for the synthesis of the anticipated target compounds 1x and 1y, respectively.
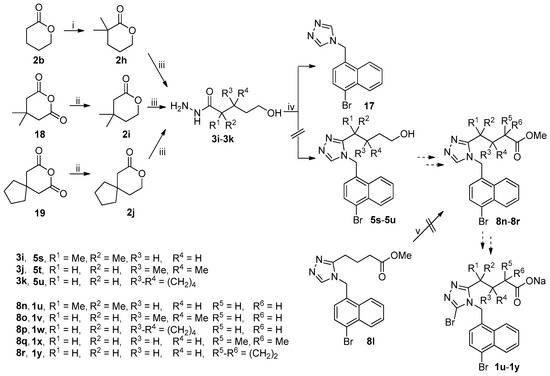
Scheme 5.
Unsuccessful synthetic route to 1u–1y. Reagents and conditions: (i) LDA, THF, −78 °C, then MeI, DMPU, −78 °C-rt (two cycles); (ii) NaBH4, THF, 0 °C-rt, then 6 M HCl; (iii) 80% N2H4·H2O, MeOH, r.t.; (iv) (a) DMFDMA, 3i–3k, MeCN, 50 °C, open vessel; (b) 4j, AcOH, reflux; (v) for 8q: LDA, THF, −78 °C, then MeI, DMPU, −78 °C-rt (two cycles); for 8r: LDA, THF, −78 °C, then 1,3,2-dioxathiolane-2,2-dioxide, DMPU, −78 °C-rt.
2.2. In Vitro URAT1 Inhibitory Activity
Table 1 summarized the results for the in vitro inhibitory activity of 1a–1r as well as lesinurad as a positive control against human URAT1. Replacement of the S atom in lesinurad with CH2 led to the increase of in vitro URAT1 inhibitory activity by three fold (1c vs. lesinurad), suggesting that the S atom was probably not indispensable and its replacement with CH2 could even increase the URAT1 inhibitory activity. In the series of designed compounds 1a–1r in the present study, the SAR exploration commenced with the screening of the substituents at the 5-position of the 1,2,4-triazole ring (1a–1f), and it turned out that H and OMe led to the complete loss of bioactivity, while the three halogen atoms, Cl, Br, and I, as well as OEt, maintained the IC50 at the same order of magnitude, with Br being the most favorable substituent. In the second round of SAR exploration, we turned to screen the substituents at the 4-position of the naphthalene ring while fixing the Br atom at 5-position of the triazole ring (1c and 1g–1n). It was very obvious that the bioactivity essentially increased as the steric volume of the alkyl substituent increased (1c and 1h–1k), with i-Pr being the most favorable one. OMe was detrimental to the bioactivity as demonstrated by the dramatic drop of the bioactivity of 1l. The most potent bioactivity in this round was observed from 1n, with Br emerging as the optimal substituent at this position. With Br being discovered to be the optimal substituents for both positions aforementioned, we went on to explore the lengths of the two linkers connecting the naphthalene and triazole rings as well as the triazole and carboxylate functionality, respectively (1n–1p for “m”; 1o, 1q and 1r for “n”). The linker length “m” was screened first (1n–1p), and it was very clear that the bioactivity first increased slightly and then decreased dramatically when the “m” increased from 0 to 2, with m = 1 being the most favorable linker length. By fixing m = 1, we subsequently screened the linker length “n” (1o, 1q and 1r). A similar trend to that for “m” was observed, and when n = 3 the bioactivity was most potent, with 1q emerging as the most potent URAT1 inhibitor among all the designed target compounds 1a–1r, which was 31-fold more potent than lesinurad.

Table 1.
In vitro inhibitory activity of 1a–1r as well as lesinurad against human URAT1 (IC50).
Actually, as shown in Scheme 4 and Scheme 5, besides the compounds 1a–1r, we originally designed 1s–1t and 1u–1y with anticipation to further explore the SAR of the linker length “n” and the substituents at the side chain at the 3-position of triazole in 1q, respectively. However, as discussed above, none of these designed compounds were synthetically accessible although considerable efforts have been made.
3. Experimental Section
3.1. General
Melting points were measured with an RY-2 microscopic melting point apparatus and are uncorrected. 1H-NMR and 13C-NMR spectra were recorded on a Bruker AV400 NMR spectrometer (Bruker BioSpin AG, Faellanden, Switzerland), using DMSO-d6, CDCl3 or MeOH-d4 as solvent and known chemical shifts of residual proton signals of deuterated solvents (for 1H-NMR) or carbon signals of deuterated solvents (for 13C-NMR) as the internal standard. High-resolution mass spectra (HRMS) were determined with an Agilent Q-TOF 6510 mass spectrometer (Agilent Technologies, Santa Clara, CA, USA) using the direct injection method, and electrospray ionization (ESI) was used as an ionization technique in negative mode (ESI−).
Esters 2a–2g and amines 4a–4i were commercially available. All the dried solvents were prepared by standard methods.
3.2. Chemistry
3.2.1. Synthesis of 2,2-Dimethyl-δ-valerolactone (2h)
To a magnetically stirred solution of diisopropylamine (23.27 g, 230 mmol) in dried THF (250 mL) cooled at −78 °C under N2 was added dropwise 1.6 M n-BuLi in n-hexane (131 mL, 210 mmol) via syringe. After addition, the resulting mixture was stirred at this temperature for 10 min, followed by dropwise addition of δ-valerolactone 2b (10.01 g, 100 mmol). The stirring was continued at this temperature for another 0.5 h, followed by successive additions of MeI (28.39 g, 200 mmol) and DMPU (14.10 g, 110 mmol) in a dropwise manner via syringe. After addition, the reaction mixture was stirred at room temperature overnight.
The reaction mixture was concentrated on a rotary evaporator to about 100 mL and then poured into ice-water (500 mL). The resulting aqueous mixture was extracted with CH2Cl2 (100 mL × 3), and the combined extracts were washed successively with 1 M hydrochloric acid (100 mL × 3) and 5% brine (100 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography to yield a colorless oil. The colorless oil was subjected to the identical procedure described above once again to yield 2h after column chromatography. Colorless oil, 9.10 g (71%). 1H-NMR (DMSO-d6, 400 MHz) δ: 4.27 (t, J = 5.8 Hz, 2H), 1.78–1.84 (m, 2H), 1.68–1.71 (m, 2H), 1.17 (s, 6H). The 1H-NMR data were in good agreement with those reported [35].
3.2.2. General Procedure for the Synthesis of Lactones 2i and 2j
To a stirred suspension of NaBH4 (5.67 g, 150 mmol) in dried THF (50 mL) cooled in an ice-water bath was added dropwise a solution of 18 or 19 (100 mmol) in dried THF (50 mL). The resulting mixture was stirred at room temperature for 5 h and then re-cooled in an ice-water bath, followed by addition of 6 M hydrochloric acid (50 mL). The mixture thus obtained was stirred for another 5 min and poured into ice-water (300 mL). The resulting mixture was extracted with CH2Cl2 (100 mL × 3), and the combined extracts were washed with 5% brine (100 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography to yield 2i or 2j.
3,3-Dimethyl-δ-valerolactone (2i): Colorless oil; 9.36 g (73%). 1H-NMR (DMSO-d6, 400 MHz) δ: 4.28 (t, J = 6.2 Hz, 2H), 2.27 (s, 2H), 1.62 (t, J = 6.0 Hz, 2H), 0.99 (s, 6H). The 1H-NMR data were in good agreement with those reported [36].
8-Oxaspiro[4,5]decan-7-one (2j): Colorless oil; 11.41 g (74%). 1H-NMR (DMSO-d6, 400 MHz) δ: 4.28 (t, J = 6.0 Hz, 2H), 2.37 (s, 2H), 1.70 (t, J = 6.2 Hz, 2H), 1.45–1.62 (m, 4H), 1.37–1.42 (m, 2H).
3.2.3. General Procedure for the Synthesis of Acyl Hydrazides 3a–3k
To a stirred solution of esters 2a–2j (70 mmol) in MeOH (30 mL) cooled in an ice-water bath was added dropwise 80% aqueous hydrazine hydrate (6.26 g, 100 mmol). The resulting solution was stirred at room temperature (2a–2b or 2d–2j), or reflux (2c), until the completion of reaction as indicated by TLC analysis (typically within 5 h).
The reaction mixture was evaporated on a rotary evaporator to give a residue, which was purified by column chromatography through a short silica gel column to yield 3a–3k after trituration with n-hexane if possible.
4-Pentenoyl hydrazide (3a): White solid; 6.47 g (81%); m.p. 44.5–45.5 °C (literature value, 45 °C [37]). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.92 (brs, 1H), 5.72–5.82 (m, 1H), 4.98–5.03 (m, 1H), 4.92–4.95 (m, 1H), 4.13 (brs, 2H), 2.20–2.25 (m, 2H), 2.08 (t, J = 7.6 Hz, 2H).
5-Hydroxypentanoyl hydrazide (3b): White solid; 7.59 g (82%); m.p. 108–109.5 °C (literature value, 107–108 °C [38]). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.89 (brs, 1H), 4.34 (brs, 1H), 4.12 (brs, 2H), 3.35 (t, J = 6.4 Hz, 2H), 1.99 (t, J = 7.4 Hz, 2H), 1.45–1.53 (m, 2H), 1.33–1.40 (m, 2H).
6-Hydroxyhexanoyl hydrazide (3c): White solid; 8.39 g (82%); m.p. 116.5–118 °C (literature value, 114.5–116 °C [39]). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.88 (brs, 1H), 4.31 (t, J = 5.0 Hz, 1H), 4.11 (brs, 2H), 3.35 (q, J = 6.0 Hz, 2H), 1.98 (t, J = 7.6 Hz, 2H), 1.42–1.50 (m, 2H), 1.35–1.40 (m, 2H), 1.19–1.26 (m, 2H).
2-Hydroxyacetyl hydrazide (3d): White solid; 5.36 g (85%); m.p. 91.5–93 °C (literature value, 93 °C [40]). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.81 (brs, 1H), 5.28 (brs, 1H), 4.20 (brs, 2H), 3.82 (s, 2H).
5-Methylpyrazolidin-3-one (3e): Colorless thick oil; 0.56 g (8%); 1H-NMR (DMSO-d6, 400 MHz) δ: 8.89 (brs, 1H), 5.06 (brs, 1H), 3.46-3.51 (m, 1H), 2.29 (dd, J = 7.0 Hz and 15.4 Hz, 1H), 1.91 (dd, J = 8.2 Hz and 15.8 Hz, 1H), 1.09 (d, J = 6.4 Hz, 3H). The 1H-NMR data were in good agreement with those reported [29].
3-Butenoyl hydrazide (3f): White solid; 5.19 g (74%); m.p. 47.5–48.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.97 (brs, 1H), 5.79–5.89 (m, 1H), 5.03–5.10 (m, 2H), 4.15 (brs, 2H), 2.81 (d, J = 6.8 Hz, 2H). The 1H-NMR data were in good agreement with those reported [41].
3-Hydroxypropionyl hydrazide (3g): White solid; 5.76 g (79%); m.p. 102–103.5 °C (literature value, 103–104 °C [42]). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.93 (brs, 1H), 4.57 (brs, 1H), 4.15 (brs, 2H), 3.58 (t, J = 6.6 Hz, 2H), 2.16 (t, J = 6.6 Hz, 2H).
3-Hydroxybutanoyl hydrazide (3h): White solid; 6.86 g (83%); m.p. 124–126 °C (literature value, 126–128 °C [43]). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.90 (brs, 1H), 4.58 (brs, 1H), 4.12 (brs, 2H), 3.91–3.98 (m, 1H), 2.14 (dd, J = 7.0 Hz and 13.8 Hz, 1H), 2.03 (dd, J = 6.0 Hz and 13.6 Hz, 1H), 1.03 (d, J = 6.0 Hz, 3H).
5-Hydroxy-2,2-dimethylpentanoyl hydrazide (3i): Colorless oil; 9.76 g (87%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.71 (brs, 1H), 4.32 (t, J = 5.2 Hz, 1H), 4.13 (brs, 2H), 3.28–3.33 (m, 2H), 1.37–1.41 (m, 2H), 1.23–1.33 (m, 2H), 1.03 (s, 6H).
5-Hydroxy-3,3-dimethylpentanoyl hydrazide (3j): White solid; 9.20 g (82%); m.p. 57.5–59 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.87 (brs, 1H), 4.33 (brs, 1H), 4.16 (brs, 2H), 3.44–3.47 (m, 2H), 1.91 (s, 2H), 1.44 (t, J = 7.4 Hz, 2H), 0.91 (s, 6H).
2-(1-(2-Hydroxyethyl)cyclopentyl)acetyl hydrazide (3k): Colorless oil; 11.21 g (86%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.90 (brs, 1H), 4.38 (brs, 1H), 4.16 (brs, 2H), 3.47 (t, J = 7.2 Hz, 2H), 1.99 (s, 2H), 1.49–1.57 (m, 8H), 1.30–1.37 (m, 2H).
3.2.4. Synthesis of (4-Bromonaphth-1-yl)methylamine 4j
A suspension of 11 (35.37 g, 160 mmol), BPO (0.78 g, 3.2 mmol), and NBS (34.17 g, 192 mmol) in n-hexane (400 mL) was refluxed under N2 until the completion of reaction as indicated by TLC analysis (typically 36 h; once the reaction commenced, 0.78 g of BPO was added every 8 h until the reaction completed). The reaction mixture was cooled to room temperature while stirring, and the precipitates were collected via vacuum filtration. The precipitates were triturated successively with saturated aqueous NaHCO3 (500 mL × 2), water (800 mL × 2), and n-hexane (800 mL) to give rise to (4-bromonaphth-1-yl)methyl bromide 12. White solid; 35.04 g (73%); m.p. 104.5–106 °C (literature value, 102–104 °C [44]). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.20–8.26 (m, 2H), 7.85 (d, J = 7.6 Hz, 1H), 7.71–7.77 (m, 2H), 7.62 (d, J = 7.6 Hz, 1H), 5.21 (s, 2H).
A mixture of 12 (33.00 g, 110 mmol) and potassium phthalimide (20.37 g, 110 mmol) in DMF (200 mL) was stirred at 100 °C under N2 until the completion of reaction as indicated by TLC analysis (typically within 12 h). On cooling to room temperature, the reaction mixture was poured into ice-water (600 mL), and the aqueous mixture thus obtained was extracted with CH2Cl2 (150 mL × 3). The combined extracts were washed with 5% brine (100 mL × 5), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was recrystallized from ethanol to produce N-((4-bromonaphth-1-yl)methyl)phthalimide 13. White solid; 36.66 g (91%); m.p. 168.5–170 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.30–8.33 (m, 1H), 8.17–8.21 (m, 1H), 7.83–7.92 (m, 4H), 7.80 (d, J = 8.0 Hz, 1H), 7.69–7.74 (m, 2H), 7.32 (d, J = 7.6 Hz, 1H), 5.23 (s, 2H).
A mixture of 13 (32.96 g, 90 mmol) and 80% aqueous hydrazine hydrate (11.26 g, 180 mmol) in ethanol (600 mL) was refluxed until the completion of reaction as indicated by TLC analysis (typically within 12 h), when a white slurry was formed. On slight cooling, 1 M aqueous NaOH (300 mL) was added to the reaction mixture, which turned to a clear solution and was concentrated on a rotary evaporator to half its original volume. The residue was poured into ice-water (350 mL). The aqueous mixture thus obtained was extracted with CH2Cl2 (100 mL × 3). The combined extracts were washed successively with 5% aqueous NaOH (100 mL × 2) and 5% brine (100 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography through a short silica gel column to produce 4j. Colorless oil; 18.27 g (86%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.15–8.18 (m, 2H), 7.83 (d, J = 7.6 Hz, 1H), 7.61–7.70 (m, 2H), 7.49 (d, J = 7.6 Hz, 1H), 4.17 (s, 2H), 1.94 (brs, 2H).
3.2.5. Synthesis of 2-(4-Bromonaphth-1-yl)ethylamine 4k
To a magnetically stirred solution of 1,4-dibromonaphthalene 14 (57.19 g, 200 mmol) in dried THF (600 mL) cooled at −78 °C under N2 was added dropwise 1.6 M n-BuLi in n-hexane (125 mL, 200 mmol) via syringe. After addition, the resulting mixture was stirred at this temperature for another 0.5 h, followed by addition of B(i-PrO)3 (75.23 g, 400 mmol) in a dropwise manner via syringe. The reaction mixture was slowly warmed to room temperature and stirred at room temperature for another 1 h. The reaction mixture was slowly poured into ice-water (600 mL) with concentrated hydrochloric acid (10 mL) while stirring. The precipitates formed were collected via vacuum filtration, washed with cooled water, and triturated with EtOAc/n-hexane to yield 4-bromonaphthalene-1-boronic acid 15. White solid; 46.16 g (92%). A varying amount of boronic anhydride was found to exist in the sample of 15 and therefore 15 was directly used in the next step without further structural characterization.
A mixture of 15 (45.16 g, 180 mmol), aminoacetonitrile hydrochloride (33.31 g, 360 mmol) and NaNO2 (31.05 g, 450 mmol) in toluene (600 mL)/water (30 mL) was stirred at 50 °C until the completion of reaction as indicated by TLC analysis (typically within 12 h). On cooling to room temperature, the reaction mixture was poured into ice-water (300 mL) and the organic phase was separated. The aqueous phase was back-extracted with toluene (100 mL). The combined extracts were washed with 5% brine, dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography to produce 2-(4-bromonaphth-1-yl)acetonitrile 16. White solid; 35.88 g (81%); m.p. 74.5–75.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.21–8.24 (m, 1H), 8.08–8.11 (m, 1H), 7.92 (d, J = 7.6 Hz, 1H), 7.74–7.79 (m, 2H), 7.53 (d, J = 7.6 Hz, 1H), 4.50 (s, 2H).
To a stirred solution of 16 (34.45 g, 140 mmol) in dried THF (300 mL) cooled in an ice-water bath was added portionwise LiAlH4 (10.63 g, 280 mmol). The resulting mixture was stirred at room temperature under N2 until the completion of reaction as indicated by TLC analysis (typically within 12 h). An appropriate amount of water was carefully added to the stirred reaction mixture to decompose the excess LiAlH4, and the mixture thus obtained was filtered off through celite. The filtrate was poured into ice-water (600 mL), and the resulting mixture was extracted with CH2Cl2 (200 mL × 3). The combined extracts were washed with 5% brine (100 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography through a short silica gel column to produce 4k. Colorless oil; 25.56 g (73%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.15–8.19 (m, 2H), 7.79 (d, J = 7.6 Hz, 1H), 7.64–7.69 (m, 2H), 7.29 (d, J = 7.6 Hz, 1H), 3.11 (t, J = 7.4 Hz, 2H), 2.84 (t, J = 7.4 Hz, 2H), 1.73 (brs, 2H).
3.2.6. General Procedure for the Synthesis of 5a–5r and 17
A mixture of 3a–3d or 3f–3k (75 mmol) and DMFDMA (8.94 g, 75 mmol) in MeCN (100 mL) was stirred at 50 °C in an open vessel in a well-ventilated hood until the completion of reaction as indicated by TLC analysis (typically within 2 h). The reaction mixture was evaporated on a rotary evaporator to almost dryness and the residue thus obtained was dissolved in glacial acetic acid (100 mL), followed by addition of 4a–4k (75 mmol). The resulting mixture was refluxed until the completion of reaction as indicated by TLC analysis (typically within 12 h).
On slight cooling, the reaction mixture was concentrated on a rotary evaporator to approximately 50 mL and poured into ice-water (600 mL) while stirring, and the resulting mixture was extracted with CH2Cl2 (200 mL × 3). The combined extracts were washed successively with 1 M hydrochloric acid (100 mL), saturated aqueous NaHCO3 (until the aqueous pH > 7 persistently), and 5% brine (100 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography to produce 5a–5r or 17 after trituration with EtOAc/n-hexane if possible.
3-(3-Buten-1-yl)-4-(4-cyclopropylnaphth-1-yl)-4H-1,2,4-triazole (5a): Colorless oil; 16.93 g (78%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.68 (s, 1H), 8.55 (d, J = 8.4 Hz, 1H), 7.69–7.74 (m, 1H), 7.61–7.65 (m, 1H), 7.55 (dd, J = 3.4 Hz and 7.4 Hz, 1H), 7.39 (d, J = 7.6 Hz, 1H), 7.12 (d, J = 8.4 Hz, 1H), 5.61–5.71 (m, 1H), 4.83–4.88 (m, 2H), 2.49–2.55 (m, 1H), 2.20-2.26 (m, 2H), 1.08–1.17 (m, 2H), 0.84–0.89 (m, 2H), 0.76–0.78 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 153.66, 144.97, 141.67, 136.81, 133.32, 129.10, 128.11, 127.78, 127.04, 125.22, 124.91, 122.51, 121.80, 115.60, 30.54, 23.40, 12.81, 7.26, 6.79.
3-(3-Buten-1-yl)-4-(naphth-1-yl)-4H-1,2,4-triazole (5b): Colorless oil; 14.02 g (75%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.73 (s, 1H), 8.16–8.20 (m, 1H), 8.11–8.13 (m, 1H), 7.60–7.71 (m, 4H), 7.15 (d, J = 8.0 Hz, 1H), 5.61–5.71 (m, 1H), 4.86–4.87 (m, 1H), 4.83 (d, J = 0.8 Hz, 1H), 2.54 (t, J = 7.6 Hz, 2H), 2.20–2.25 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 153.59, 144.89, 136.78, 133.66, 130.12, 129.85, 129.11, 128.49, 128.14, 127.13, 125.62, 125.60, 121.25, 115.62, 30.55, 23.44.
3-(3-Buten-1-yl)-4-(4-methylnaphth-1-yl)-4H-1,2,4-triazole (5c): Colorless oil; 15.01 g (76%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.69 (s, 1H), 8.18 (d, J = 8.4 Hz, 1H), 7.69 (t, J = 7.6 Hz, 1H), 7.62 (t, J = 7.6 Hz, 1H), 7.52–7.58 (m, 2H), 7.11 (d, J = 8.4 Hz, 1H), 5.63–5.70 (m, 1H), 4.83–4.87 (m, 2H), 2.75 (s, 3H), 2.52–2.53 (m, 2H), 2.20–2.25 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 153.68, 144.99, 136.93, 136.82, 132.52, 129.15, 128.17, 127.76, 127.02, 126.03, 125.19, 124.98, 121.75, 115.62, 30.57, 23.39, 19.01.
3-(3-Buten-1-yl)-4-(4-ethylnaphth-1-yl)-4H-1,2,4-triazole (5d): Colorless oil; 14.77 g (71%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.69 (s, 1H), 8.24 (d, J = 8.8 Hz, 1H), 7.66–7.70 (m, 1H), 7.58–7.63 (m, 2H), 7.54 (d, J = 7.2 Hz, 1H), 7.12 (d, J = 8.0 Hz, 1H), 5.62–5.72 (m, 1H), 4.86–4.88 (m, 1H), 4.83 (d, J = 1.2 Hz, 1H), 3.11–3.23 (m, 2H), 2.49–2.53 (m, 2H), 2.20–2.26 (m, 2H), 1.34 (t, J = 7.4 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 153.69, 145.00, 142.66, 136.81, 131.74, 129.39, 128.18, 127.64, 127.04, 125.32, 124.50, 124.46, 121.94, 115.59, 30.55, 25.16, 23.42, 14.87.
3-(3-Buten-1-yl)-4-(4-n-propylnaphth-1-yl)-4H-1,2,4-triazole (5e): White solid; 16.61 g (76%); m.p. 92.5–94 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.70 (s, 1H), 8.24 (d, J = 8.4 Hz, 1H), 7.65–7.69 (m, 1H), 7.58–7.62 (m, 2H), 7.52 (d, J = 7.2 Hz, 1H), 7.11 (d, J = 8.0 Hz, 1H), 5.63–5.70 (m, 1H), 4.86–4.87 (m, 1H), 4.83 (d, J = 1.6 Hz, 1H), 3.06–3.18 (m, 2H), 2.49–2.53 (m, 2H), 2.19–2.25 (m, 2H), 1.71–1.77 (m, 2H), 1.00 (t, J = 7.2 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 153.68, 145.02, 141.13, 136.84, 131.90, 129.44, 128.21, 127.63, 126.99, 125.51, 125.15, 124.70, 121.92, 115.63, 34.15, 30.56, 23.53, 23.43, 13.91.
3-(3-Buten-1-yl)-4-(4-isopropylnaphth-1-yl)-4H-1,2,4-triazole (5f): Colorless oil; 16.39 g (75%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.70 (s, 1H), 8.34 (d, J = 8.8 Hz, 1H), 7.56–7.72 (m, 4H), 7.12 (d, J = 8.0 Hz, 1H), 5.63–5.71 (m, 1H), 4.87–4.89 (m, 1H), 4.84 (d, J = 1.2 Hz, 1H), 3.82-3.88 (m, 1H), 2.52–2.54 (m, 2H), 2.22–2.27 (m, 2H), 1.41 (d, J = 6.8 Hz, 3H), 1.36 (d, J = 6.8 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 154.39, 147.65, 145.74, 137.55, 132.05, 130.14, 128.75, 128.23, 127.75, 126.03, 124.75, 122.72, 122.22, 116.33, 31.24, 28.76, 24.30, 24.14, 23.66.
3-(3-Buten-1-yl)-4-(4-methoxynaphth-1-yl)-4H-1,2,4-triazole (5g): White solid; 15.29 g (73%); m.p. 92.5–94 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.66 (s, 1H), 8.27–8.30 (m, 1H), 7.60–7.65 (m, 3H), 7.11 (d, J = 8.4 Hz, 1H), 7.03–7.06 (m, 1H), 5.62–5.72 (m, 1H), 4.86–4.88 (m, 1H), 4.84 (d, J = 1.2 Hz, 1H), 4.05 (s, 3H), 2.48–2.53 (m, 2H), 2.20–2.26 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 155.93, 153.89, 145.19, 136.86, 130.00, 128.48, 126.44, 126.25, 125.02, 122.26, 122.22, 121.22, 115.60, 103.81, 56.06, 30.60, 23.37.
3-(3-Buten-1-yl)-4-(4-ethoxynaphth-1-yl)-4H-1,2,4-triazole (5h): White solid; 16.28 g (74%); m.p. 72.5–73.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.65 (s, 1H), 8.29–8.31 (m, 1H), 7.57–7.64 (m, 3H), 7.09 (d, J = 8 Hz, 1H), 7.02–7.05 (m, 1H), 5.61–5.72 (m, 1H), 4.86–4.88 (m, 1H), 4.83 (s, 1H), 4.30 (q, J = 6.9 Hz, 2H), 2.51–2.53 (m, 2H), 2.20–2.25 (m, 2H), 1.49 (t, J = 7.0 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz): 155.17, 153.88, 145.15, 136.82, 130.05, 128.39, 126.29, 126.24, 125.12, 122.30, 122.06, 121.16, 115.52, 104.36, 64.02, 30.58, 23.37, 14.42.
4-(4-Bromonaphth-1-yl)-3-(3-buten-1-yl)-4H-1,2,4-triazole (5i): White solid; 19.20 g (78%); m.p. 97.5–99 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.74 (s, 1H), 8.30 (d, J = 8.4 Hz, 1H), 8.09 (d, J = 8.0 Hz, 1H), 7.81–7.85 (m, 1H), 7.73 (t, J = 7.6 Hz, 1H), 7.65 (d, J = 7.6 Hz, 1H), 7.18 (d, J = 8.4 Hz, 1H), 5.62–5.72 (m, 1H), 4.86–4.88 (m, 1H), 4.84 (s, 1H), 2.52–2.56 (m, 2H), 2.21–2.27 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 153.61, 144.86, 136.75, 131.65, 130.34, 130.00, 129.75, 129.18, 129.03, 127.21, 126.39, 123.84, 122.29, 115.69, 30.52, 23.36.
4-((4-Bromonaphth-1-yl)methyl)-3-(3-buten-1-yl)-4H-1,2,4-triazole (5j): White solid; 19.76 g (77%); m.p. 120.5–122.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.44 (s, 1H), 8.22–8.24 (m, 1H), 8.13–8.16 (m, 1H), 7.86 (d, J = 7.6 Hz, 1H), 7.71–7.78 (m, 2H), 6.77 (d, J = 8.0 Hz, 1H), 5.74–5.85 (m, 3H), 4.90–4.99 (m, 2H), 2.72 (t, J = 7.6 Hz, 2H), 2.32–2.38 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 153.21, 144.43, 137.12, 132.41, 131.26, 131.12, 129.66, 128.06, 127.69, 127.17, 125.00, 123.81, 122.13, 115.52, 44.39, 30.45, 23.18.
4-(2-(4-Bromonaphth-1-yl)ethyl)-3-(3-buten-1-yl)-4H-1,2,4-triazole (5k): Colorless oil; 19.77 g (74%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.40 (s, 1H), 8.17-8.19 (m, 2H), 7.76 (d, J = 7.6 Hz, 1H), 7.65–7.73 (m, 2H), 7.12 (d, J = 7.6 Hz, 1H), 5.61–5.72 (m, 1H), 4.89 (s, 1H), 4.85–4.86 (m, 1H), 4.25 (t, J = 7.0 Hz, 2H), 3.49 (t, J = 7.0 Hz, 2H), 2.33–2.37 (m, 2H), 2.14–2.21 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 152.51, 143.66, 137.06, 134.32, 132.61, 131.23, 129.53, 127.75, 127.57, 127.30, 127.12, 124.28, 121.02, 115.27, 43.68, 32.89, 30.30, 22.71.
4-((4-Bromonaphth-1-yl)methyl)-3-(4-hydroxybutyl)-4H-1,2,4-triazole (5l): White solid; 19.72 g (73%); m.p. 126.5–128 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.43 (s, 1H), 8.21–8.24 (m, 1H), 8.13-8.15 (m, 1H), 7.85 (d, J = 7.6 Hz, 1H), 7.71–7.78 (m, 2H), 6.74 (d, J = 8.0 Hz, 1H), 5.74 (s, 2H), 4.35 (t, J = 5.0 Hz, 1H), 3.31–3.35 (m, 2H), 2.64 (t, J = 7.6 Hz, 2H), 1.58–1.66 (m, 2H), 1.38–1.45 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 153.86, 144.34, 132.49, 131.25, 131.13, 129.67, 128.06, 127.70, 127.19, 124.88, 123.78, 122.13, 60.23, 44.40, 31.84, 23.45, 23.17.
4-((4-Bromonaphth-1-yl)methyl)-3-(5-hydroxypentyl)-4H-1,2,4-triazole (5m): White solid; 20.77 g (74%); m.p. 88.5–91 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.43 (s, 1H), 8.22–8.24 (m, 1H), 8.14 (d, J = 8.8 Hz, 1H), 7.86 (d, J = 8.0 Hz, 1H), 7.71–7.78 (m, 2H), 6.75 (d, J = 8.0 Hz, 1H), 5.74 (s, 2H), 4.30 (t, J = 5.0 Hz, 1H), 3.29–3.33 (m, 2H), 2.62 (t, J = 7.4 Hz, 2H), 1.52–1.60 (m, 2H), 1.22–1.37 (m, 4H). 13C-NMR (DMSO-d6, 100 MHz) δ: 153.82, 144.35, 132.53, 131.26, 131.13, 129.69, 128.10, 127.72, 127.20, 124.92, 123.81, 122.10, 60.50, 44.39, 32.07, 26.45, 25.07, 23.65.
4-((4-Bromonaphth-1-yl)methyl)-3-(hydroxymethyl)-4H-1,2,4-triazole (5n): White solid; 18.85 g (79%); m.p. 197–200 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.40 (s, 1H), 8.22 (d, J = 8.4 Hz, 1H), 8.17 (d, J = 8.0 Hz, 1H), 7.86 (d, J = 7.6 Hz, 1H), 7.69–7.77 (m, 2H), 6.93 (d, J = 7.6 Hz, 1H), 5.82 (s, 2H), 5.75 (t, J = 5.6 Hz, 1H), 4.59 (d, J = 5.6 Hz, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 154.07, 145.58, 132.97, 132.11, 131.84, 130.43, 128.75, 128.49, 127.89, 126.44, 124.53, 122.92, 54.32, 45.53.
4-((4-Bromonaphth-1-yl)methyl)-3-((E)-1-propen-1-yl)-4H-1,2,4-triazole (5o): White solid; 2.95 g (12%); m.p. 157–159 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.54 (s, 1H), 8.21–8.23 (m, 1H), 8.14–8.16 (m, 1H), 7.84 (d, J = 7.6 Hz, 1H), 7.71–7.78 (m, 2H), 6.67 (d, J = 7.6 Hz, 1H), 6.26 (dd, J = 1.6 Hz and 11.6 Hz, 1H), 6.06–6.14 (m, 1H), 5.80 (s, 2H), 2.11 (dd, J = 1.6 Hz and 7.2 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 150.79, 143.82, 135.12, 132.61, 131.13, 131.06, 129.66, 128.07, 127.69, 127.15, 124.59, 123.72, 122.02, 112.61, 44.39, 15.69.
3-Allyl-4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazole (5p): White solid; 18.85 g (60%); m.p. 146.5–148 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.43 (s, 1H), 8.22–8.24 (m, 1H), 8.10–8.12 (m, 1H), 7.86 (d, J = 7.6 Hz, 1H), 7.71–7.78 (m, 2H), 6.79 (d, J = 7.6 Hz, 1H), 5.85–5.95 (m, 1H), 5.72 (s, 2H), 5.04 (s, 1H), 5.00–5.02 (m, 1H), 3.51 (d, J = 6.4 Hz, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 151.87, 144.57, 132.61, 132.19, 131.27, 131.12, 129.68, 128.05, 127.72, 127.19, 125.20, 123.74, 122.17, 117.42, 44.54, 28.30.
4-((4-Bromonaphth-1-yl)methyl)-3-(2-hydroxyethyl)-4H-1,2,4-triazole (5q): White solid; 19.18 g (77%); m.p. 179–182 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.40 (s, 1H), 8.22–8.24 (m, 1H), 8.15 (d, J = 8.4 Hz, 1H), 7.85 (d, J = 7.6 Hz, 1H), 7.71–7.78 (m, 2H), 6.76 (d, J = 7.6 Hz, 1H), 5.78 (s, 2H), 4.83 (t, J = 5.4 Hz, 1H), 3.68–3.72 (m, 2H), 2.82 (t, J = 6.8 Hz, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 152.36, 144.28, 132.45, 131.28, 131.14, 129.74, 128.12, 127.77, 127.20, 125.02, 123.83, 122.11, 59.11, 44.56, 27.65.
4-((4-Bromonaphth-1-yl)methyl)-3-(2-hydroxypropyl)-4H-1,2,4-triazole (5r): White solid; 18.70 g (72%); m.p. 155.5–157 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.36 (s, 1H), 8.22–8.24 (m, 1H), 8.13–8.15 (m, 1H), 7.85 (d, J = 8.0 Hz, 1H), 7.71–7.78 (m, 2H), 6.75 (d, J = 7.6 Hz, 1H), 5.80 (d, J = 17.6 Hz, 1H), 5.76 (d, J = 17.6 Hz, 1H), 4.86 (d, J = 4.4 Hz, 1H), 3.97–4.03 (m, 1H), 2.77 (dd, J = 5.6 Hz and 14.8 Hz, 1H), 2.70 (dd, J = 6.8 Hz and 14.8 Hz, 1H), 1.09 (d, J = 6.4 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 152.34, 144.16, 132.47, 131.28, 131.12, 129.69, 128.06, 127.72, 127.17, 125.05, 123.83, 122.10, 65.16, 44.63, 33.52, 23.17.
4-((4-Bromonaphth-1-yl)methyl)-4H-1,2,4-triazole (17): White solid; 13.83 g (64%); m.p. 201–204 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.61 (s, 2H), 8.22 (d, J = 8.0 Hz, 2H), 7.90 (d, J = 7.6 Hz, 1H), 7.69–7.76 (m, 2H), 7.20 (d, J = 8.0 Hz, 1H), 5.79 (s, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 143.42, 132.61, 131.47, 131.21, 129.77, 128.03, 127.83, 127.23, 126.87, 123.80, 122.57, 45.13.
3.2.7. General Procedure for the Synthesis of 6a–6l
To a stirred solution of 5a–5k or 5o (50 mmol) in THF/H2O (4/1 by v/v, 100 mL in total) at room temperature were added commercially available 50% solution of NMMO in water (23.43 g, 100 mmol) and a 0.16 M stock solution of OsO4 in t-BuOH/H2O (4/1 by v/v; 31.25 mL, 5 mmol). The resulting mixture was stirred at room temperature until the completion of reaction as indicated by TLC analysis (typically within 24 h). The reaction mixture was filtered off through celite via vacuum filtration and the filtrate was poured into ice-water (300 mL). The resulting mixture was extracted with CH2Cl2 (100 mL × 3). The combined extracts were washed successively with 1 M aqueous Na2S2O3 (100 mL) and 5% brine (100 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography followed by trituration with EtOAc/n-hexane to produce the pure diol intermediates. These diol intermediates were not structurally characterized and were directly used in the next step.
The diol intermediates (deemed to be 50 mmol) were dissolved in THF/H2O (4/1 by v/v, 200 mL in total), followed by addition of NaIO4 (32.08 g, 150 mmol). The resulting mixture was stirred at room temperature until the completion of reaction as indicated by TLC analysis (typically within 2 h). The reaction mixture was poured into ice-water (400 mL) and the resulting mixture was extracted with CH2Cl2 (100 mL × 3). The combined extracts were washed with 5% brine (100 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography to produce 6a–6l after trituration with EtOAc/n-hexane if possible.
3-(4-(4-Cyclopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionaldehyde (6a): White solid; 11.36 g (78%); m.p. 126.5–128.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 9.63 (s, 1H), 8.70 (s, 1H), 8.56 (d, J = 8.4 Hz, 1H), 7.71–7.75 (m, 1H), 7.62–7.66 (m, 1H), 7.58 (d, J = 8.0 Hz, 1H), 7.40 (d, J = 7.6 Hz, 1H), 7.15 (d, J = 8.4 Hz, 1H), 2.86 (t, J = 7.0 Hz, 2H), 2.63–2.71 (m, 1H), 2.51–2.59 (m, 2H), 1.11–1.14 (m, 2H), 0.84–0.90 (m, 1H), 0.79–0.80 (m, 1H). 13C-NMR (CDCl3, 100 MHz) δ: 199.79, 154.05, 144.70, 142.62, 134.17, 129.47, 127.92, 127.89, 127.12, 125.23, 124.74, 123.07, 121.80, 40.17, 17.15, 13.33, 6.80, 6.74.
3-(4-(Naphth-1-yl)-4H-1,2,4-triazol-3-yl)propionaldehyde (6b): White solid; 9.93 g (79%); m.p. 112–114 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 9.63 (s, 1H), 8.77 (s, 1H), 8.17–8.20 (m, 1H), 8.13 (d, J = 7.6 Hz, 1H), 7.61–7.71 (m, 4H), 7.18 (d, J = 8.4 Hz, 1H), 2.87 (t, J = 7.0 Hz, 2H), 2.66–2.73 (m, 1H), 2.49–2.61 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 201.58, 153.32, 145.03, 133.69, 130.17, 129.72, 129.05, 128.50, 128.20, 127.15, 125.64, 121.29, 39.38, 16.94.
3-(4-(4-Methylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionaldehyde (6c): White solid; 10.08 g (76%); m.p. 101.5–103.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 9.62 (s, 1H), 8.71 (s, 1H), 8.18 (d, J = 8.4 Hz, 1H), 7.68–7.71 (m, 1H), 7.63 (t, J = 7.2 Hz, 1H), 7.53–7.59 (m, 2H), 7.15 (d, J = 8.0 Hz, 1H), 2.85 (t, J = 7.0 Hz, 2H), 2.75 (s, 3H), 2.64–2.73 (m, 1H), 2.52–2.59 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 201.59, 153.43, 145.14, 137.00, 132.57, 129.10, 128.06, 127.83, 127.04, 126.06, 125.23, 125.00, 121.79, 39.40, 19.02, 16.93.
3-(4-(4-Ethylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionaldehyde (6d): White solid; 10.75 g (77%); m.p. 114–115.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 9.63 (s, 1H), 8.71 (s, 1H), 8.25 (d, J = 8.4 Hz, 1H), 7.69 (t, J = 7.6 Hz, 1H), 7.60–7.63 (m, 2H), 7.55 (d, J = 7.2 Hz, 1H), 7.16 (d, J = 8.4 Hz, 1H), 3.12–3.23 (m, 2H), 2.86 (t, J = 7.0 Hz, 2H), 2.64–2.72 (m, 1H), 2.52–2.59 (m, 1H), 1.35 (t, J = 7.6 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 201.63, 153.43, 145.16, 142.74, 131.76, 129.33, 128.06, 127.72, 127.07, 125.36, 124.52, 121.97, 39.38, 25.18, 16.94, 14.93.
3-(4-(4-n-Propylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionaldehyde (6e): White solid; 11.00 g (75%); m.p. 97.5–99.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 9.62 (s, 1H), 8.72 (s, 1H), 8.25 (d, J = 8.4 Hz, 1H), 7.66–7.70 (m, 1H), 7.59–7.63 (m, 2H), 7.53 (d, J = 7.6 Hz, 1H), 7.15 (d, J = 8.4 Hz, 1H), 3.05–3.20 (m, 2H), 2.85 (t, J = 7.0 Hz, 2H), 2.64–2.71 (m, 1H), 2.50–2.59 (m, 1H), 1.70-1.78 (m, 2H), 1.01 (t, J = 7.4 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 201.62, 153.44, 145.16, 141.20, 131.95, 129.39, 128.09, 127.69, 127.01, 125.54, 125.19, 124.71, 121.95, 39.38, 34.18, 23.56, 16.94, 13.95.
3-(4-(4-Isopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionaldehyde (6f): Colorless oil; 11.44 g (78%). 1H-NMR (DMSO-d6, 400 MHz) δ: 9.63 (s, 1H), 8.72 (s, 1H), 8.35 (d, J = 8.8 Hz, 1H), 7.60–7.71 (m, 4H), 7.16 (d, J = 8.0 Hz, 1H), 3.82–3.89 (m, 1H), 2.87 (t, J = 7.0 Hz, 2H), 2.64–2.72 (m, 1H), 2.53–2.59 (m, 1H), 1.41 (d, J = 6.8 Hz, 3H), 1.37 (d, J = 6.8 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 201.57, 153.44, 147.00, 145.15, 131.39, 129.39, 127.94, 127.58, 127.05, 125.36, 124.05, 122.06, 121.57, 39.37, 28.08, 23.54, 22.99, 16.97.
3-(4-(4-Methoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionaldehyde (6g): White solid; 10.27 g (73%); m.p. 149–150.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 9.63 (s, 1H), 8.68 (s, 1H), 8.28–8.30 (m, 1H), 7.61–7.65 (m, 3H), 7.13 (d, J = 8.4 Hz, 1H), 7.07–7.10 (m, 1H), 4.06 (s, 3H), 2.85 (t, J = 7.0 Hz, 2H), 2.64–2.72 (m, 1H), 2.54–2.59 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 201.62, 155.98, 153.62, 145.33, 129.94, 128.53, 126.45, 126.28, 125.06, 122.23, 122.14, 121.25, 103.86, 56.08, 39.42, 16.90.
3-(4-(4-Ethoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionaldehyde (6h): White solid; 11.08 g (75%); m.p. 138.5–140 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 9.62 (s, 1H), 8.67 (s, 1H), 8.29–8.32 (m, 1H), 7.60–7.64 (m, 3H), 7.07–7.11 (m, 2H), 4.31 (q, J = 6.9 Hz, 2H), 2.84 (t, J = 7.0 Hz, 2H), 2.64–2.72 (m, 1H), 2.53–2.59 (m, 1H), 1.50 (t, J = 6.8 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 201.61, 155.22, 153.62, 145.32, 129.99, 128.49, 126.35, 126.30, 125.16, 122.33, 121.94, 121.20, 104.47, 64.07, 39.42, 16.89, 14.46.
3-(4-(4-Bromonaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionaldehyde (6i): White solid; 12.88 g (78%); m.p. 139–140.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 9.62 (s, 1H), 8.76 (s, 1H), 8.31 (d, J = 8.4 Hz, 1H), 8.10 (d, J = 8.0 Hz, 1H), 7.82–7.86 (m, 1H), 7.72–7.76 (m, 1H), 7.66 (d, J = 7.6 Hz, 1H), 7.23 (d, J = 8.4 Hz, 1H), 2.86 (t, J = 7.0 Hz, 2H), 2.67–2.74 (m, 1H), 2.56–2.62 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 201.54, 153.35, 144.97, 131.68, 130.30, 129.92, 129.77, 129.21, 129.02, 127.21, 126.41, 123.89, 122.34, 39.39, 16.91.
3-(4-((4-Bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)propionaldehyde (6j): White solid; 13.25 g (77%); m.p. 131.5–133 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 9.69 (s, 1H), 8.46 (s, 1H), 8.22–8.24 (m, 1H), 8.15–8.17 (m, 1H), 7.86 (d, J = 7.6 Hz, 1H), 7.72–7.79 (m, 2H), 6.78 (d, J = 7.6 Hz, 1H), 5.77 (s, 2H), 2.85–2.93 (m, 4H). 13C-NMR (DMSO-d6, 100 MHz) δ: 201.77, 152.91, 144.65, 132.19, 131.28, 131.15, 129.73, 128.11, 127.75, 127.20, 125.02, 123.83, 122.16, 44.41, 39.32, 16.88.
3-(4-(2-(4-Bromonaphth-1-yl)ethyl)-4H-1,2,4-triazol-3-yl)propionaldehyde (6k): White solid; 12.90 g (72%); m.p. 42.5–44 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 9.62 (s, 1H), 8.33 (s, 1H), 8.18–8.21 (m, 2H), 7.78 (d, J = 8.0 Hz, 1H), 7.65–7.73 (m, 2H), 7.18 (d, J = 7.6 Hz, 1H), 4.28 (t, J = 7.2 Hz, 2H), 3.50 (t, J = 7.4 Hz, 2H), 2.77 (t, J = 6.8 Hz, 2H), 2.67–2.71 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 201.74, 152.16, 143.94, 134.40, 132.64, 131.22, 129.60, 127.75, 127.66, 127.38, 127.11, 124.38, 120.97, 43.72, 39.39, 32.60, 16.46.
(4-((4-Bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)carboxaldehyde (6l): White solid; 12.01 g (76%); m.p. 211–213 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 10.06 (s, 1H), 8.84 (s, 1H), 8.22–8.25 (m, 1H), 8.13–8.15 (m, 1H), 7.83 (d, J = 7.6 Hz, 1H), 7.72–7.79 (m, 2H), 6.80 (d, J = 7.6 Hz, 1H), 6.04 (s, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 182.52, 150.19, 147.54, 131.96, 131.20, 131.07, 129.69, 128.10, 127.90, 127.23, 125.09, 123.57, 122.29, 46.13.
3.2.8. Synthesis of 1-(4-((4-Bromonaphth-1-yl)methyl)-4H-1,2,4-Triazol-3-yl)acetone 6n
To a stirred solution of DMSO (2.34 g, 30 mmol) in dried CH2Cl2 (40 mL) cooled at −78 °C under N2 was added dropwise a solution of (COCl)2 (1.90 g, 15 mmol) in dried CH2Cl2 (10 mL) via syringe, and the resulting solution was stirred at this temperature for 0.5 h, followed by addition of a solution of 5r (3.46 g, 10 mmol) in dried CH2Cl2 (10 mL) in a dropwise manner via syringe. After addition, the stirring was continued at this temperature for 1 h, and Et3N (6.07 g, 60 mmol) was added in a dropwise manner via syringe. The reaction mixture thus obtained was stirred at room temperature until the completion of reaction as indicated by TLC analysis (typically within 3 h).
The reaction mixture was poured into ice-water (200 mL). The resulting mixture was extracted with CH2Cl2 (100 mL × 3). The combined extracts were washed successively with saturated aqueous NaHCO3 (50 mL), 1 M hydrochloric acid (50 mL), and 5% brine (100 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography to produce 6n. White solid; 2.44 g (71%); m.p. 187–190 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.37 (s, 1H), 8.22 (d, J = 8.0 Hz, 1H), 8.08 (d, J = 7.6 Hz, 1H), 7.87 (d, J = 7.6 Hz, 1H), 7.69–7.77 (m, 2H), 6.90 (d, J = 7.6 Hz, 1H), 5.63 (s, 2H), 4.11 (s, 2H), 2.15 (s, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 203.07, 148.91, 144.52, 131.92, 131.43, 131.16, 129.69, 128.05, 127.73, 127.17, 125.90, 123.92, 122.35, 44.89, 39.04, 29.47.
3.2.9. General Procedure for the Synthesis of 7a–7k and 17
To a stirred solution of 6a–6l (35 mmol) in t-BuOH (200 mL) and 2-methyl-2-butene (73.64 g, 1.05 mol) cooled with an ice-water bath was added a suspension of NaClO2 (80%; 11.87 g, 105 mmol) and NaH2PO4 (25.20 g, 210 mmol) in water (50 mL). The resulting mixture was stirred at room temperature until the completion of reaction as indicated by TLC analysis (typically within 6 h).
The reaction mixture was poured into ice-water (500 mL), and the mixture thus obtained was acidified (pH = 1–2) with concentrated hydrochloric acid and extracted with CH2Cl2 (100 mL × 3). The combined extracts were washed with water (100 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography to produce 7a–7k or 17 after trituration with EtOAc/n-hexane.
3-(4-(4-Cyclopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (7a): White solid; 9.04 g (84%); m.p. 213.5–215 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.22 (brs, 1H), 8.71 (s, 1H), 8.56 (d, J = 8.8 Hz, 1H), 7.70–7.74 (m, 1H), 7.60–7.64 (m, 1H), 7.56 (d, J = 7.6 Hz, 1H), 7.41 (d, J = 7.6 Hz, 1H), 7.16 (d, J = 8.4 Hz, 1H), 2.58–2.67 (m, 3H), 2.45–2.55 (m, 2H), 1.08–1.17 (m, 2H), 0.73-0.91 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.01, 153.49, 145.04, 141.72, 133.37, 129.07, 128.05, 127.81, 127.07, 125.25, 124.92, 122.60, 121.93, 30.44, 19.51, 12.86, 7.18, 6.88.
3-(4-(Naphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (7b): White solid; 7.95 g (85%); m.p. 225–226.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.18 (brs, 1H), 8.74 (s, 1H), 8.18 (dd, J = 2.0 Hz and 7.2 Hz, 1H), 8.13 (d, J = 7.6 Hz, 1H), 7.59–7.72 (m, 4H), 7.19 (d, J = 8.0 Hz, 1H), 2.52–2.67 (m, 4H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.70, 154.11, 145.65, 134.39, 130.85, 130.47, 129.78, 129.18, 128.85, 127.85, 126.34, 122.09, 31.15, 20.22.
3-(4-(4-Methylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (7c): White solid; 8.66 g (88%); m.p. 225 °C (dec). 1H-NMR (DMSO-d6, 400 MHz) δ: 12.17 (brs, 1H), 8.70 (s, 1H), 8.18 (d, J = 8.4 Hz, 1H), 7.69 (t, J = 7.6 Hz, 1H), 7.61 (t, J = 7.4 Hz, 1H), 7.53–7.57 (m, 2H), 7.15 (d, J = 8.4 Hz, 1H), 2.75 (s, 3H), 2.58–2.65 (m, 3H), 2.51–2.56 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.01, 153.54, 145.07, 137.02, 132.59, 129.13, 128.10, 127.81, 127.07, 126.08, 125.25, 125.01, 121.89, 30.48, 19.52, 19.05.
3-(4-(4-Ethylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (7d): White solid; 8.48 g (82%); m.p. 206.5 °C (dec). 1H-NMR (DMSO-d6, 400 MHz) δ: 12.17 (brs, 1H), 8.70 (s, 1H), 8.25 (d, J = 8.4 Hz, 1H), 7.67–7.71 (m, 1H), 7.58–7.62 (m, 2H), 7.54 (d, J = 7.6 Hz, 1H), 7.16 (d, J = 8.0 Hz, 1H), 3.12–3.23 (m, 2H), 2.59–2.67 (m, 3H), 2.52–2.56 (m, 1H), 1.35 (t, J = 7.4 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.70, 154.20, 145.76, 143.42, 132.46, 130.04, 128.79, 128.36, 127.76, 126.04, 125.20, 122.76, 31.12, 25.88, 20.20, 15.62.
3-(4-(4-n-Propylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (7e): White solid; 9.10 g (84%); m.p. 171–173.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.15 (brs, 1H), 8.71 (s, 1H), 8.24 (d, J = 8.4 Hz, 1H), 7.68 (t, J = 7.6 Hz, 1H), 7.52–7.61 (m, 3H), 7.15 (d, J = 8.4 Hz, 1H), 3.04–3.19 (m, 2H), 2.60–2.64 (m, 2H), 2.49–2.56 (m, 2H), 1.74–1.76 (m, 2H), 1.01 (t, J = 7.2 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.13, 153.53, 145.06, 141.18, 131.94, 129.40, 128.14, 127.64, 127.01, 125.53, 125.18, 124.69, 122.04, 34.19, 30.54, 23.57, 19.55, 13.97.
3-(4-(4-Isopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (7f): White solid; 8.88 g (82%); m.p. 178–180.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.16 (brs, 1H), 8.71 (s, 1H), 8.34 (d, J = 8.8 Hz, 1H), 7.69 (t, J = 7.6 Hz, 1H), 7.58–7.64 (m, 3H), 7.16 (d, J = 8.4 Hz, 1H), 3.82–3.89 (m, 1H), 2.59–2.68 (m, 3H), 2.51–2.56 (m, 1H), 1.41 (d, J = 6.8 Hz, 3H), 1.37 (d, J = 6.8 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.03, 153.52, 146.99, 145.08, 131.38, 129.40, 127.97, 127.54, 127.06, 125.36, 124.04, 122.15, 121.57, 30.41, 28.09, 23.57, 23.02, 19.53.
3-(4-(4-Methoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (7g): White solid; 8.74 g (84%); m.p. 243–245 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.15 (brs, 1H), 8.67 (s, 1H), 8.28–8.30 (m, 1H), 7.60–7.65 (m, 3H), 7.07–7.13 (m, 2H), 4.06 (s, 3H), 2.59–2.65 (m, 3H), 2.52–2.57 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.00, 155.98, 153.69, 145.24, 129.96, 128.49, 126.46, 126.28, 125.05, 122.22, 122.18, 121.33, 103.87, 56.09, 30.49, 19.47.
3-(4-(4-Ethoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (7h): White solid; 8.94 g (82%); m.p. 231–233 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.23 (brs, 1H), 8.66 (s, 1H), 8.29–8.32 (m, 1H), 7.57–7.64 (m, 3H), 7.07–7.11 (m, 2H), 4.30 (q, J = 6.9 Hz, 2H), 2.49–2.65 (m, 4H), 1.50 (t, J = 7.0 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.10, 155.21, 153.72, 145.22, 130.01, 128.44, 126.35, 126.28, 125.14, 122.30, 121.99, 121.28, 104.47, 64.07, 30.59, 19.51, 14.47.
3-(4-(4-Bromonaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (7i): White solid; 10.78 g (89%); m.p. 237–240 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.09 (brs, 1H), 8.75 (s, 1H), 8.30 (d, J = 8.4 Hz, 1H), 8.10 (d, J = 8.0 Hz, 1H), 7.83 (t, J = 7.6 Hz, 1H), 7.72 (t, J = 7.6 Hz, 1H), 7.64 (d, J = 8.0 Hz, 1H), 7.23 (d, J = 8.4 Hz, 1H), 2.52-2.69 (m, 4H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.01, 153.46, 144.89, 131.68, 130.32, 129.97, 129.78, 129.17, 129.04, 127.21, 126.41, 123.87, 122.44, 30.49, 19.48.
3-(4-((4-Bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)propionic acid (7j): White solid; 10.59 g (84%); m.p. 225–227 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.18 (brs, 1H), 8.43 (s, 1H), 8.23 (d, J = 7.6 Hz, 1H), 8.14–8.16 (m, 1H), 7.85 (d, J = 7.6 Hz, 1H), 7.72–7.75 (m, 2H), 6.78 (d, J = 7.6 Hz, 1H), 5.76 (s, 2H), 2.81–2.85 (m, 2H), 2.68–2.72 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.22, 152.99, 144.50, 132.23, 131.29, 131.14, 129.72, 128.12, 127.76, 127.20, 125.04, 123.85, 122.12, 44.36, 30.45, 19.35.
3-(4-(2-(4-Bromonaphth-1-yl)ethyl)-4H-1,2,4-triazol-3-yl)propionic acid (7k): White solid; 10.61 g (81%); m.p. 210–213 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.18 (brs, 1H), 8.31 (s, 1H), 8.18–8.21 (m, 2H), 7.78 (d, J = 7.6 Hz, 1H), 7.65–7.73 (m, 2H), 7.18 (d, J = 7.6 Hz, 1H), 4.28 (t, J = 7.2 Hz, 2H), 3.49 (t, J = 7.2 Hz, 2H), 2.69 (t, J = 6.6 Hz, 2H), 2.61 (t, J = 6.4 Hz, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.25, 152.34, 143.82, 134.41, 132.66, 131.24, 129.60, 127.74, 127.65, 127.38, 127.13, 124.39, 120.99, 43.72, 32.59, 30.64, 18.96.
4-((4-Bromonaphth-1-yl)methyl)-4H-1,2,4-triazole (17): 4.13 g (41%). The physical properties of the sample 17 obtained by this procedure were in good agreement with those for the sample obtained from 3i–3k described above.
3.2.10. General Procedure for the Synthesis of 7l, 7m and 17
To a stirred solution of 5l–5n (35 mmol) and (2,2,6,6-tetramethylpiperidin-1-yl)oxyl (TEMPO; 0.55 g, 3.5 mmol) in MeCN (200 mL) and phosphate buffer (150 mL, pH = 6.7; prepared by mixing aqueous 0.67 M Na2HPO4 and 0.67 M NaH2PO4 in a ratio of 1/1) at 35 °C were added aqueous NaClO2 and aqueous NaClO simultaneously over 2 h. The aqueous NaClO2 solution was prepared by dissolving 80% solid NaClO2 (80%; 7.91 g, 70 mmol) in water (40 mL), while the aqueous NaClO solution was prepared by diluting a commercially available aqueous NaClO solution (21% by w/w; 0.35 mL, 1 mmol) with water (20 mL). After addition, the reaction mixture was stirred at 35 °C until the completion of reaction as indicated by TLC analysis (typically within 10 h).
The reaction mixture was poured into ice-water (200 mL), and the aqueous mixture thus obtained was acidified (pH = 1–2) with concentrated hydrochloric acid and extracted with CH2Cl2 (100 mL × 3). The combined extracts were washed successively with 1% aqueous Na2S2O3 (100 mL) and water (100 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography through a short silica gel column to produce 7l, 7m or 17 after trituration with EtOAc/n-hexane.
4-(4-((4-Bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)butanoic acid (7l): White solid; 10.74 g (82%); m.p. 175–177.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.03 (brs, 1H), 8.44 (s, 1H), 8.22–8.24 (m, 1H), 8.13–8.15 (m, 1H), 7.85 (d, J = 7.6 Hz, 1H), 7.72–7.78 (m, 2H), 6.72 (d, J = 7.6 Hz, 1H), 5.74 (s, 2H), 2.68 (t, J = 7.6 Hz, 2H), 2.29 (t, J = 7.2 Hz, 2H), 1.81–1.89 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 174.02, 153.43, 144.45, 132.46, 131.24, 131.14, 129.74, 128.11, 127.74, 127.21, 124.74, 123.80, 122.11, 44.41, 32.72, 22.93, 21.93.
5-(4-((4-Bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)pentanoic acid (7m): White solid; 11.01 g (81%); m.p. 179.5–181 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 11.99 (brs, 1H), 8.42 (s, 1H), 8.21–8.24 (m, 1H), 8.13–8.15 (m, 1H), 7.85 (d, J = 8.0 Hz, 1H), 7.71–7.78 (m, 2H), 6.76 (d, J = 7.6 Hz, 1H), 5.74 (s, 2H), 2.65 (t, J = 7.4 Hz, 2H), 2.16 (t, J = 7.2 Hz, 2H), 1.58–1.65 (m, 2H), 1.47–1.54 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 174.29, 153.65, 144.38, 132.46, 131.30, 131.17, 129.72, 128.10, 127.74, 127.23, 124.99, 123.82, 122.18, 44.44, 33.28, 25.96, 24.01, 23.36.
4-((4-Bromonaphth-1-yl)methyl)-4H-1,2,4-triazole (17): 3.43 g (34%). The physical properties of sample 17 obtained by this procedure were in good agreement with those for the sample obtained from 3i–3k described above.
3.2.11. General Procedure for the Synthesis of 8a–8m
To a stirred mixture of 7a–7m (28 mmol) in dried CH2Cl2 (100 mL) cooled in an ice-water bath were added EDCI (8.05 g, 42 mmol), DMAP (1.71 g, 14 mmol) and MeOH (8.97 g, 280 mmol), and the resulting mixture was stirred at room temperature under N2 until the completion of reaction as indicated by TLC analysis (typically within 6 h).
The reaction mixture was diluted with CH2Cl2 (200 mL) and washed successively with 1 M hydrochloric acid (100 mL), saturated aqueous Na2CO3 (100 mL), and 5% brine (100 mL). The organic phase was dried over anhydrous Na2SO4 and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography to produce 8a–8m after trituration with EtOAc/n-hexane if possible.
Methyl 3-(4-(4-cyclopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (8a): White solid; 6.93 g (77%); m.p. 88.5–89.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.70 (s, 1H), 8.56 (d, J = 8.4 Hz, 1H), 7.72 (t, J = 7.6 Hz, 1H), 7.64 (t, J = 7.4 Hz, 1H), 7.56 (d, J = 7.6 Hz, 1H), 7.41 (d, J = 7.6 Hz, 1H), 7.15 (d, J = 8.4 Hz, 1H), 3.51 (s, 3H), 2.63–2.75 (m, 3H), 2.51–2.59 (m, 2H), 1.09–1.16 (m, 2H), 0.85–0.88 (m, 1H), 0.76–0.79 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.99, 153.26, 145.09, 141.76, 133.36, 129.05, 127.97, 127.84, 127.09, 125.27, 124.94, 122.59, 121.87, 51.37, 30.11, 19.39, 12.85, 7.20, 6.89.
Methyl 3-(4-(naphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (8b): White solid; 5.99 g (76%); m.p. 129–130.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.75 (s, 1H), 8.17–8.20 (m, 1H), 8.13 (d, J = 7.6 Hz, 1H), 7.61–7.72 (m, 4H), 7.18 (d, J = 8.0 Hz, 1H), 3.51 (s, 3H), 2.65–2.75 (m, 3H), 2.54–2.63 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.97, 153.16, 145.00, 133.69, 130.18, 129.69, 129.05, 128.49, 128.18, 127.16, 125.65, 125.63, 121.32, 51.36, 30.12, 19.40.
Methyl 3-(4-(4-methylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (8c): White solid; 6.37 g (77%); m.p. 138.5–140.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.70 (s, 1H), 8.19 (d, J = 8.0 Hz, 1H), 7.70 (dt, J = 1.2 Hz and 8.4 Hz, 1H), 7.63 (dt, J = 1.2 Hz and 6.8 Hz, 1H), 7.53–7.58 (m, 2H), 7.15 (d, J = 8.0 Hz, 1H), 3.51 (s, 3H), 2.75 (s, 3H), 2.70–2.74 (m, 2H), 2.63–2.69 (m, 1H), 2.52–2.59 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.96, 153.26, 145.10, 137.01, 132.55, 129.09, 128.02, 127.80, 127.04, 126.05, 125.22, 124.99, 121.81, 51.36, 30.13, 19.38, 19.02.
Methyl 3-(4-(4-ethylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (8d): White solid; 6.84 g (79%); m.p. 116.5–118 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.71 (s, 1H), 8.25 (d, J = 8.4 Hz, 1H), 7.67–7.71 (m, 1H), 7.59–7.63 (m, 2H), 7.55 (d, J = 7.2 Hz, 1H), 7.15 (d, J = 8.0 Hz, 1H), 3.51 (s, 3H), 3.14–3.21 (m, 2H), 2.70–2.75 (m, 2H), 2.64–2.67 (m, 1H), 2.54–2.59 (m, 1H), 1.35 (t, J = 7.6 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.97, 153.26, 145.10, 142.74, 131.75, 129.32, 128.03, 127.68, 127.06, 125.34, 124.51, 122.00, 51.35, 30.11, 25.17, 19.39, 14.91.
Methyl 3-(4-(4-n-propylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (8e): White solid; 6.88 g (76%); m.p. 116.5–118 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.72 (s, 1H), 8.25 (d, J = 8.4 Hz, 1H), 7.68 (t, J = 7.6 Hz, 1H), 7.58–7.63 (m, 2H), 7.53 (d, J = 7.6 Hz, 1H), 7.15 (d, J = 8.0 Hz, 1H), 3.51 (s, 3H), 3.06–3.17 (m, 2H), 2.69–2.74 (m, 2H), 2.63–2.67 (m, 1H), 2.54–2.59 (m, 1H), 1.71–1.78 (m, 2H), 1.01 (t, J = 7.4 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.96, 153.25, 145.09, 141.19, 131.93, 129.37, 128.06, 127.64, 126.99, 125.52, 125.16, 124.68, 121.97, 51.34, 34.18, 30.10, 23.55, 19.39, 13.93.
Methyl 3-(4-(4-isopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (8f): White solid; 6.88 g (76%); m.p. 80–81.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.71 (s, 1H), 8.34 (d, J = 8.4 Hz, 1H), 7.69 (t, J = 7.6 Hz, 1H), 7.60–7.64 (m, 3H), 7.15 (d, J = 8.4 Hz, 1H), 3.82–3.88 (m, 1H), 3.51 (s, 3H), 2.71–2.76 (m, 2H), 2.64–2.70 (m, 1H), 2.53–2.59 (m, 1H), 1.41 (d, J = 6.8 Hz, 3H), 1.37 (d, J = 6.4 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.97, 153.26, 146.99, 145.10, 131.36, 129.36, 127.89, 127.54, 127.04, 125.34, 124.03, 122.07, 121.55, 51.33, 30.07, 28.05, 23.54, 22.99, 19.39.
Methyl 3-(4-(4-methoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (8g): White solid; 6.80 g (78%); m.p. 134.5–135.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.67 (s, 1H), 8.28–8.30 (m, 1H), 7.60–7.65 (m, 3H), 7.13 (d, J = 8.4 Hz, 1H), 7.06–7.10 (m, 1H), 4.06 (s, 3H), 3.51 (s, 3H), 2.52–2.74 (m, 4H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.99, 156.00, 153.46, 145.29, 129.95, 128.51, 126.45, 126.28, 125.05, 122.23, 122.11, 121.28, 103.86, 56.08, 51.36, 30.15, 19.36.
Methyl 3-(4-(4-ethoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (8h): White solid; 6.92 g (76%); m.p. 117–118.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.67 (s, 1H), 8.29–8.32 (m, 1H), 7.61–7.65 (m, 2H), 7.59 (d, J = 8.0 Hz, 1H), 7.11 (d, J = 8.0 Hz, 1H), 7.06–7.09 (m, 1H), 4.31 (q, J = 6.9 Hz, 2H), 3.51 (s, 3H), 2.64–2.74 (m, 3H), 2.52–2.61 (m, 1H), 1.50 (t, J = 7.0 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.96, 155.22, 153.43, 145.27, 129.98, 128.45, 126.34, 126.27, 125.13, 122.30, 121.90, 121.21, 104.45, 64.06, 51.33, 30.14, 19.34, 14.45.
Methyl 3-(4-(4-bromonaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (8i): White solid; 8.07 g (80%); m.p. 126–127.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.76 (s, 1H), 8.31 (d, J = 8.4 Hz, 1H), 8.10 (d, J = 7.6 Hz, 1H), 7.82–7.86 (m, 1H), 7.72–7.76 (m, 1H), 7.65 (d, J = 8.0 Hz, 1H), 7.22 (d, J = 8.4 Hz, 1H), 3.51 (s, 3H), 2.68–2.75 (m, 3H), 2.53–2.63 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.94, 153.19, 144.93, 131.67, 130.29, 129.88, 129.77, 129.18, 129.02, 127.20, 126.39, 123.89, 122.35, 51.36, 30.12, 19.35.
Methyl 3-(4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)propionate (8j): White solid; 8.07 g (77%); m.p. 100–101.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.45 (s, 1H), 8.22–8.25 (m, 1H), 8.14–8.17 (m, 1H), 7.86 (d, J = 8.0 Hz, 1H), 7.72–7.79 (m, 2H), 6.77 (d, J = 8.0 Hz, 1H), 5.77 (s, 2H), 3.56 (s, 3H), 2.85–2.88 (m, 2H), 2.75–2.79 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.20, 152.74, 144.60, 132.22, 131.26, 131.13, 129.71, 128.12, 127.75, 127.19, 124.96, 123.84, 122.12, 51.37, 44.35, 30.07, 19.26.
Methyl 3-(4-(2-(4-bromonaphth-1-yl)ethyl)-4H-1,2,4-triazol-3-yl)propionate (8k): White solid; 8.15 g (75%); m.p. 112.5–114 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.33 (s, 1H), 8.18–8.20 (m, 2H), 7.78 (d, J = 7.6 Hz, 1H), 7.65–7.73 (m, 2H), 7.16 (d, J = 7.6 Hz, 1H), 4.28 (t, J = 7.2 Hz, 2H), 3.56 (s, 3H), 3.49 (t, J = 7.2 Hz, 2H), 2.60–2.68 (m, 4H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.17, 152.03, 143.88, 134.38, 132.62, 131.23, 129.58, 127.74, 127.63, 127.36, 127.12, 124.35, 121.00, 51.37, 43.70, 32.64, 30.24, 18.83.
Methyl 4-(4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)butanoate (8l): white foam; 8.59 g (79%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.44 (s, 1H), 8.22–8.24 (m, 1H), 8.13–8.15 (m, 1H), 7.85 (d, J = 7.6 Hz, 1H), 7.72–7.79 (m, 2H), 6.71 (d, J = 7.6 Hz, 1H), 5.73 (s, 2H), 3.51 (s, 3H), 2.67 (t, J = 7.4 Hz, 2H), 2.37 (t, J = 7.2 Hz, 2H), 1.83–1.90 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.88, 153.27, 144.46, 132.48, 131.23, 131.13, 129.74, 128.15, 127.76, 127.20, 124.76, 123.82, 122.07, 51.18, 44.37, 32.36, 22.82, 21.81.
Methyl 5-(4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)pentanoate (8m): Colorless oil; 9.01 g (80%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.43 (s, 1H), 8.22–8.24 (m, 1H), 8.13–8.15 (m, 1H), 7.85 (d, J = 7.6 Hz, 1H), 7.72–7.78 (m, 2H), 6.74 (d, J = 7.6 Hz, 1H), 5.73 (s, 2H), 3.53 (s, 3H), 2.64 (t, J = 7.2 Hz, 2H), 2.24 (t, J = 7.2 Hz, 2H), 1.50–1.61 (m, 4H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.09, 153.54, 144.38, 132.52, 131.26, 131.13, 129.71, 128.13, 127.73, 127.20, 124.90, 123.83, 122.08, 51.11, 44.36, 32.81, 25.83, 23.86, 23.22.
3.2.12. General Procedure for the Synthesis of 9a–9c and 9f–9q
A mixture of 8a–8m (6 mmol) and N-halosuccinimide (NCS, NBS or NIS; 7.2 mmol) in MeCN (30 mL) was stirred at room temperature (9b, 9d–9l or 9n–9q), 60 °C (for 9m), or reflux (9a and 9c), until the completion of reaction as indicated by TLC analysis (typically within 24 h).
The reaction mixture was poured into ice-water (100 mL) and the aqueous mixture thus obtained was extracted with CH2Cl2 (50 mL × 3). The combined extracts were washed successively with 5% aqueous Na2S2O3 (50 mL), saturated aqueous Na2CO3 (50 mL × 3), and 5% brine (50 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography to produce 9a–9c or 9f–9q after trituration with EtOAc/n-hexane if possible.
Methyl 3-(5-chloro-4-(4-cyclopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (9a): White solid; 1.26 g (59%); m.p. 104–105.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.58 (d, J = 8.4 Hz, 1H), 7.74 (t, J = 7.6 Hz, 1H), 7.64–7.68 (m, 2H), 7.44 (d, J = 7.6 Hz, 1H), 7.15 (d, J = 8.0 Hz, 1H), 3.52 (s, 3H), 2.62–2.73 (m, 3H), 2.51–2.57 (m, 2H), 1.12–1.16 (m, 2H), 0.84–0.87 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.76, 155.96, 142.81, 141.45, 133.44, 128.71, 128.17, 127.21, 126.48, 126.14, 125.13, 122.67, 121.42, 51.39, 29.38, 20.45, 12.83, 7.23, 7.14.
Methyl 3-(5-bromo-4-(4-cyclopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (9b): White solid; 1.61 g (67%); m.p. 122.5–123.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.58 (d, J = 8.4 Hz, 1H), 7.73 (t, J = 7.6 Hz, 1H), 7.62–7.67 (m, 2H), 7.44 (d, J = 7.6 Hz, 1H), 7.10 (d, J = 8.4 Hz, 1H), 3.51 (s, 3H), 2.62–2.72 (m, 3H), 2.51–2.56 (m, 2H), 1.12–1.16 (m, 2H), 0.84–0.87 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.78, 156.41, 142.66, 133.42, 130.48, 128.82, 128.09, 127.18, 127.01, 126.53, 125.11, 122.68, 121.58, 51.40, 29.51, 20.42, 12.84, 7.23, 7.16.
Methyl 3-(4-(4-cyclopropylnaphth-1-yl)-5-iodo-4H-1,2,4-triazol-3-yl)propionate (9c): White solid; 1.37 g (51%); m.p. 138.5–140 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.58 (d, J = 8.4 Hz, 1H), 7.73 (t, J = 7.6 Hz, 1H), 7.64 (t, J = 7.2 Hz, 1H), 7.55 (d, J = 7.6 Hz, 1H), 7.43 (d, J = 7.6 Hz, 1H), 7.02 (d, J = 8.4 Hz, 1H), 3.51 (s, 3H), 2.62–2.71 (m, 3H), 2.52–2.57 (m, 2H), 1.12–1.17 (m, 2H), 0.85–0.88 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.83, 156.29, 142.38, 133.41, 129.07, 128.42, 127.91, 127.12, 126.62, 125.06, 122.65, 121.89, 106.22, 51.38, 29.82, 20.33, 12.85, 7.26, 7.18.
Methyl 3-(5-bromo-4-(naphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (9f): White solid; 1.34 g (62%); m.p. 152–154 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.22–8.25 (m, 1H), 8.14–8.16 (m, 1H), 7.72–7.76 (m, 2H), 7.63–7.70 (m, 2H), 7.13 (d, J = 8.0 Hz, 1H), 3.52 (s, 3H), 2.65–2.73 (m, 3H), 2.51–2.58 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.78, 156.34, 133.77, 131.01, 130.29, 128.89, 128.78, 128.68, 128.48, 127.30, 126.92, 125.82, 121.04, 51.42, 29.53, 20.43.
Methyl 3-(5-bromo-4-(4-methylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (9g): White solid; 1.37 g (61%); m.p. 127–128.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.20 (d, J = 8.4 Hz, 1H), 7.68–7.72 (m, 1H), 7.57–7.66 (m, 3H), 7.10 (d, J = 8.4 Hz, 1H), 3.51 (s, 3H), 2.76 (s, 3H), 2.63–2.72 (m, 3H), 2.51–2.56 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.78, 156.43, 138.01, 132.68, 130.52, 128.89, 128.09, 127.17, 127.12, 126.49, 126.28, 125.21, 121.54, 51.42, 29.56, 20.44, 19.08.
Methyl 3-(5-bromo-4-(4-ethylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (9h): White solid; 1.47 g (63%); m.p. 107.5–109 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.27 (d, J = 8.4 Hz, 1H), 7.58–7.72 (m, 4H), 7.10 (d, J = 8.4 Hz, 1H), 3.52 (s, 3H), 3.19 (q, J = 7.6 Hz, 2H), 2.63–2.72 (m, 3H), 2.52–2.57 (m, 1H), 1.37 (t, J = 7.4 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.80, 156.44, 143.57, 131.91, 130.52, 129.09, 127.97, 127.20, 127.12, 126.60, 124.71, 124.61, 121.71, 51.41, 29.53, 25.16, 20.45, 14.68.
Methyl 3-(5-bromo-4-(4-n-propylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (9i): White solid; 1.57 g (65%); m.p. 104.5–106 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.27 (d, J = 8.4 Hz, 1H), 7.57–7.71 (m, 4H), 7.10 (d, J = 8.4 Hz, 1H), 3.51 (s, 3H), 3.13 (t, J = 7.6 Hz, 2H), 2.63–2.72 (m, 3H), 2.52–2.57 (m, 1H), 1.74–1.80 (m, 2H), 1.02 (t, J = 7.4 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.80, 156.43, 142.09, 132.06, 130.52, 129.15, 127.93, 127.14, 126.42, 125.65, 124.88, 121.69, 51.41, 34.19, 29.51, 23.41, 20.44, 13.97.
Methyl 3-(5-bromo-4-(4-isopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (9j): White solid; 1.50 g (62%); m.p. 108.5–110 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.36 (d, J = 8.4 Hz, 1H), 7.61–7.72 (m, 4H), 7.11 (d, J = 8.4 Hz, 1H), 3.83–3.90 (m, 1H), 3.52 (s, 3H), 2.69–2.74 (m, 2H), 2.63–2.66 (m, 1H), 2.47–2.54 (m, 1H), 1.40 (d, J = 6.8 Hz, 6H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.81, 156.44, 147.83, 131.48, 130.52, 129.14, 127.82, 127.17, 126.98, 126.59, 124.27, 121.78, 51.40, 29.47, 28.16, 23.28, 23.22, 20.45.
Methyl 3-(5-bromo-4-(4-methoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (9k): White solid; 0.70 g (30%); m.p. 124.5–126.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.29–8.31 (m, 1H), 7.68 (d, J = 8.4 Hz, 1H), 7.62–7.66 (m, 2H), 7.17 (d, J = 8.4 Hz, 1H), 7.03–7.05 (m, 1H), 4.08 (s, 3H), 3.52 (s, 3H), 2.66–2.72 (m, 3H), 2.51–2.58 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.80, 156.61, 156.52, 130.90, 129.75, 128.77, 127.66, 126.57, 125.15, 122.41, 121.12, 121.01, 104.11, 56.13, 51.41, 29.59, 20.44.
Methyl 3-(5-bromo-4-(4-ethoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (9l): White solid; 0.75 g (31%); m.p. 120–122.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.31–8.33 (m, 1H), 7.63–7.66 (m, 3H), 7.14 (d, J = 8.4 Hz, 1H), 7.02–7.04 (m, 1H), 4.33 (q, J = 6.9 Hz, 2H), 3.52 (s, 3H), 2.66–2.72 (m, 3H), 2.51–2.58 (m, 1H), 1.51 (t, J = 7.0 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.78, 156.60, 155.77, 130.90, 129.79, 128.72, 127.66, 126.47, 125.22, 122.48, 120.96, 120.90, 104.66, 64.14, 51.40, 29.58, 20.43, 14.46.
Methyl 3-(5-bromo-4-(4-bromonaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (9m): White solid; 1.61 g (61%); m.p. 162–163.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.32 (d, J = 8.4 Hz, 1H), 8.16 (d, J = 8.0 Hz, 1H), 7.85 (t, J = 7.6 Hz, 1H), 7.72–7.78 (m, 2H), 7.19 (d, J = 8.4 Hz, 1H), 3.52 (s, 3H), 2.66–2.73 (m, 3H), 2.52–2.60 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.75, 156.37, 131.82, 130.18, 130.09, 130.02, 129.52, 129.21, 128.94, 127.70, 127.42, 124.94, 122.08, 51.42, 29.57, 20.40.
Methyl 3-(5-bromo-4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)propionate (9n): White solid; 1.71 g (63%); m.p. 90.5–91.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.22–8.26 (m, 2H), 7.78–7.83 (m, 3H), 6.36 (d, J = 8.0 Hz, 1H), 5.78 (s, 2H), 3.55 (s, 3H), 2.90 (t, J = 7.0 Hz, 2H), 2.77 (t, J = 7.0 Hz, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.96, 156.06, 131.25, 131.04, 130.82, 130.10, 129.68, 128.29, 127.75, 127.17, 123.78, 122.46, 121.80, 51.42, 44.94, 29.59, 20.13.
Methyl 3-(5-bromo-4-(2-(4-bromonaphth-1-yl)ethyl)-4H-1,2,4-triazol-3-yl)propionate (9o): White foam; 1.82 g (65%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.18–8.21 (m, 1H), 8.14–8.16 (m, 1H), 7.77 (d, J = 8.0 Hz, 1H), 7.66–7.74 (m, 2H), 7.09 (d, J = 7.6 Hz, 1H), 4.26 (t, J = 7.0 Hz, 2H), 3.57 (s, 3H), 3.46 (t, J = 7.0 Hz, 2H), 2.60–2.68 (m, 4H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.00, 155.20, 133.79, 132.74, 131.28, 129.59, 128.89, 127.99, 127.69, 127.47, 127.21, 124.05, 121.28, 51.44, 44.66, 31.64, 29.62, 19.78.
Methyl 4-(5-bromo-4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)butanoate (9p): White solid; 1.77 g (63%); m.p. 154–155.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.21–8.25 (m, 2H), 7.78–7.82 (m, 3H), 6.31 (d, J = 8.0 Hz, 1H), 5.74 (s, 2H), 3.49 (s, 3H), 2.71 (t, J = 7.4 Hz, 2H), 2.36 (t, J = 7.2 Hz, 2H), 1.81–1.89 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.76, 156.64, 131.43, 131.04, 130.78, 129.98, 129.70, 128.28, 127.73, 127.17, 123.72, 122.31, 121.76, 51.16, 44.99, 32.15, 23.72, 21.52.
Methyl 5-(5-bromo-4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)pentanoate (9q): Colorless oil; 1.76 g (61%). 1H-NMR (DMSO-d6, 400 MHz) δ: 8.21–8.25 (m, 2H), 7.77–7.82 (m, 3H), 6.31 (d, J = 8.0 Hz, 1H), 5.74 (s, 2H), 3.50 (s, 3H), 2.68 (t, J = 7.2 Hz, 2H), 2.22 (t, J = 7.2 Hz, 2H), 1.49–1.60 (m, 4H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.04, 156.96, 131.53, 131.06, 130.81, 129.87, 129.69, 128.30, 127.75, 127.18, 123.76, 122.38, 121.76, 51.10, 44.98, 32.73, 25.48, 24.11, 23.68.
3.2.13. General Procedure for the Synthesis of 9d and 9e
To a stirred solution of MeONa (150 mmol) in dried MeOH (for 9d) or EtONa (150 mmol) in dried EtOH (for 9e) prepared by dissolving Na (3.45 g, 150 mmol) in dried MeOH (100 mL) or EtOH (100 mL) was added 9b (12.01 g, 30 mmol). The resulting mixture was stirred at reflux under N2 until the completion of reaction as indicated by TLC analysis (typically within 24 h).
On cooling to room temperature, the reaction mixture was poured into ice-water (200 mL) and the aqueous mixture thus obtained was extracted immediately with CH2Cl2 (50 mL × 3). The combined extracts were washed with 5% brine (50 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography to produce 9d or 9e after trituration with EtOAc/n-hexane.
Methyl 3-(4-(4-cyclopropylnaphth-1-yl)-5-methoxy-4H-1,2,4-triazol-3-yl)propionate (9d): White solid; 1.48 g (14%); m.p. 115–116.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.54 (d, J = 8.4 Hz, 1H), 7.71 (t, J = 7.6 Hz, 1H), 7.63 (t, J = 7.6 Hz, 1H), 7.54 (d, J = 7.2 Hz, 1H), 7.38 (d, J = 7.6 Hz, 1H), 7.23 (d, J = 8.4 Hz, 1H), 3.91 (s, 3H), 3.51 (s, 3H), 2.64 (t, J = 7.0 Hz, 2H), 2.51–2.60 (m, 2H), 2.38–2.47 (m, 1H), 1.10–1.13 (m, 2H), 0.77–0.87 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.95, 159.33, 150.97, 141.82, 133.45, 129.14, 127.74, 126.96, 126.39, 126.10, 124.98, 122.73, 121.83, 57.47, 51.34, 29.47, 20.19, 12.82, 7.17, 6.88.
Ethyl 3-(4-(4-cyclopropylnaphth-1-yl)-5-ethoxy-4H-1,2,4-triazol-3-yl)propionate (9e): White solid; 1.48 g (13%); m.p. 94.5–96 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 8.54 (d, J = 8.4 Hz, 1H), 7.71 (t, J = 7.0 Hz, 1H), 7.63 (t, J = 7.2 Hz, 1H), 7.53 (d, J = 7.6 Hz, 1H), 7.38 (d, J = 7.6 Hz, 1H), 7.23 (d, J = 8.4 Hz, 1H), 4.31–4.37 (m, 2H), 3.96 (q, J = 7.1 Hz, 2H), 2.56–2.62 (m, 2H), 2.49–2.54 (m, 2H), 2.38–2.44 (m, 1H), 1.08–1.18 (m, 8H), 0.81–0.84 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 171.48, 158.63, 150.70, 141.72, 133.45, 129.14, 127.65, 126.95, 126.51, 126.03, 124.97, 122.75, 121.87, 66.42, 59.91, 29.63, 20.21, 14.22, 13.95, 12.81, 7.11, 6.95.
3.2.14. General Procedure for the Synthesis of 10a–10q
To a stirred solution of 9a–9q (1.5 mmol) in EtOH (15 mL) was added an aqueous solution of LiOH prepared by dissolving LiOH·H2O (0.19 g, 4.5 mmol) in water (1 mL), and the resulting mixture was stirred at room temperature until the completion of reaction as indicated by TLC analysis (typically within 3 h).
The reaction mixture was poured into ice-water (100 mL) and the mixture thus obtained was acidified (pH = 1–2) by concentrated hydrochloric acid and extracted with CH2Cl2 (50 mL × 3). The combined extracts were washed with water (50 mL), dried over anhydrous Na2SO4, and evaporated on a rotary evaporator to afford a residue, which was purified by column chromatography to produce 10a–10q after trituration with EtOAc/n-hexane.
3-(5-Chloro-4-(4-cyclopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (10a): White solid; 0.45 g (88%); m.p. 146–147.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.21 (brs, 1H), 8.58 (d, J = 8.4 Hz, 1H), 7.72–7.76 (m, 1H), 7.63–7.67 (m, 2H), 7.44 (d, J = 7.6 Hz, 1H), 7.16 (d, J = 8.0 Hz, 1H), 2.44–2.65 (m, 5H), 1.13–1.16 (m, 2H), 0.86–0.87 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.81, 156.22, 142.79, 141.38, 133.45, 128.73, 128.16, 127.22, 126.49, 126.21, 125.15, 122.70, 121.50, 29.69, 20.58, 12.86, 7.23, 7.16.
3-(5-Bromo-4-(4-cyclopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (10b): White solid; 0.52 g (89%); m.p. 132.5–134.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.21 (brs, 1H), 8.58 (d, J = 8.4 Hz, 1H), 7.73 (t, J = 7.6 Hz, 1H), 7.61–7.66 (m, 2H), 7.44 (d, J = 7.6 Hz, 1H), 7.11 (d, J = 8.4 Hz, 1H), 2.42–2.65 (m, 5H), 1.12–1.16 (m, 2H), 0.84-0.88 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.85, 156.70, 142.66, 133.46, 130.42, 128.87, 128.09, 127.20, 127.11, 126.55, 125.13, 122.72, 121.68, 29.84, 20.58, 12.89, 7.24, 7.19.
3-(4-(4-Cyclopropylnaphth-1-yl)-5-iodo-4H-1,2,4-triazol-3-yl)propionic acid (10c): White solid; 0.55 g (85%); m.p. 151 °C (dec). 1H-NMR (DMSO-d6, 400 MHz) δ: 12.18 (brs, 1H), 8.57 (d, J = 8.4 Hz, 1H), 7.72 (t, J = 7.4 Hz, 1H), 7.62 (t, J = 7.6 Hz, 1H), 7.54 (d, J = 7.6 Hz, 1H), 7.43 (d, J = 7.6 Hz, 1H), 7.03 (d, J = 8.4 Hz, 1H), 2.46–2.66 (m, 5H), 1.12–1.18 (m, 2H), 0.86–0.90 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.84, 156.52, 142.34, 133.41, 129.08, 128.49, 127.87, 127.11, 126.61, 125.04, 122.66, 121.94, 106.11, 30.13, 20.45, 12.86, 7.26, 7.15.
3-(4-(4-Cyclopropylnaphth-1-yl)-5-methoxy-4H-1,2,4-triazol-3-yl)propionic acid (10d): White solid; 0.46 g (90%); m.p. 167 °C (dec). 1H-NMR (DMSO-d6, 400 MHz) δ: 12.18 (brs, 1H), 8.54 (d, J = 8.4 Hz, 1H), 7.70 (t, J = 7.2 Hz, 1H), 7.62 (t, J = 7.4 Hz, 1H), 7.53 (d, J = 7.6 Hz, 1H), 7.38 (d, J = 7.6 Hz, 1H), 7.23 (d, J = 8.4 Hz, 1H), 3.90 (s, 3H), 2.47–2.57 (m, 4H), 2.34–2.43 (m, 1H), 1.08–1.16 (m, 2H), 0.79–0.87 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.02, 159.30, 151.22, 141.77, 133.45, 129.16, 127.71, 126.94, 126.47, 126.09, 124.97, 122.75, 121.88, 57.45, 29.87, 20.35, 12.83, 7.14, 6.88.
3-(4-(4-Cyclopropylnaphth-1-yl)-5-ethoxy-4H-1,2,4-triazol-3-yl)propionic acid (10e): White solid; 0.46 g (87%); m.p. 154.5–155.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.13 (brs, 1H), 8.54 (d, J = 8.4 Hz, 1H), 7.70 (t, J = 7.4 Hz, 1H), 7.62 (t, J = 7.6 Hz, 1H), 7.52 (d, J = 7.6 Hz, 1H), 7.38 (d, J = 7.6 Hz, 1H), 7.23 (d, J = 8.4 Hz, 1H), 4.30–4.38 (m, 2H), 2.46–2.57 (m, 4H), 2.32–2.41 (m, 1H), 1.11–1.18 (m, 5H), 0.82–0.87 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.00, 158.63, 150.92, 141.70, 133.46, 129.15, 127.64, 126.95, 126.57, 126.05, 124.97, 122.77, 121.93, 66.42, 29.77, 20.33, 14.23, 12.83, 7.10, 6.96.
3-(5-Bromo-4-(naphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (10f): White solid; 0.46 g (88%); m.p. 177.5–179 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.22 (brs, 1H), 8.24 (t, J = 4.6 Hz, 1H), 8.15 (d, J = 7.6 Hz, 1H), 7.61–7.74 (m, 4H), 7.14 (d, J = 8.0 Hz, 1H), 2.61–2.68 (m, 3H), 2.46–2.55 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.80, 156.60, 133.78, 130.98, 130.20, 128.92, 128.86, 128.67, 128.44, 127.29, 126.92, 125.82, 121.11, 29.84, 20.56.
3-(5-Bromo-4-(4-methylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (10g): White solid; 0.46 g (86%); m.p. 186 °C (dec). 1H-NMR (DMSO-d6, 400 MHz) δ: 12.24 (brs, 1H), 8.20 (d, J = 8.4 Hz, 1H), 7.68–7.72 (m, 1H), 7.57–7.65 (m, 3H), 7.11 (d, J = 8.4 Hz, 1H), 2.76 (s, 3H), 2.59–2.64 (m, 3H), 2.46–2.53 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.96, 156.75, 137.99, 132.68, 130.41, 128.92, 128.08, 127.21, 127.18, 126.50, 126.28, 125.22, 121.60, 30.07, 20.65, 19.10.
3-(5-Bromo-4-(4-ethylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (10h): White solid; 0.48 g (85%); m.p. 167.5–170 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.21 (brs, 1H), 8.27 (d, J = 8.4 Hz, 1H), 7.58–7.72 (m, 4H), 7.11 (d, J = 8.4 Hz, 1H), 3.19 (q, J = 7.5 Hz, 2H), 2.58–2.67 (m, 2H), 2.45–2.53 (m, 2H), 1.37 (t, J = 7.6 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.87, 156.73, 143.57, 131.95, 130.46, 129.15, 127.96, 127.22, 126.62, 124.72, 124.62, 121.82, 29.87, 25.20, 20.62, 14.69.
3-(5-Bromo-4-(4-n-propylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (10i): White solid; 0.52 g (89%); m.p. 139.5–141.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.20 (brs, 1H), 8.26 (d, J = 8.4 Hz, 1H), 7.56–7.71 (m, 4H), 7.10 (d, J = 8.0 Hz, 1H), 3.13 (t, J = 7.6 Hz, 2H), 2.58–2.64 (m, 2H), 2.45–2.52 (m, 2H), 1.72–1.80 (m, 2H), 1.02 (t, J = 7.4 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.52, 157.37, 142.76, 132.76, 131.13, 129.88, 128.61, 127.91, 127.83, 127.12, 126.34, 125.58, 122.46, 34.90, 30.52, 24.11, 21.27, 14.69.
3-(5-Bromo-4-(4-isopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (10j): White solid; 0.48 g (83%); m.p. 197 °C (dec). 1H-NMR (DMSO-d6, 400 MHz) δ: 12.21 (brs, 1H), 8.36 (d, J = 8.4 Hz, 1H), 7.60–7.72 (m, 4H), 7.11 (d, J = 8.0 Hz, 1H), 3.83–3.90 (m, 1H), 2.59–2.65 (m, 3H), 2.44–2.51 (m, 1H), 1.40 (d, J = 6.8 Hz, 6H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.85, 156.69, 147.82, 131.49, 130.45, 129.17, 127.81, 127.18, 127.06, 126.59, 124.27, 121.86, 121.78, 29.79, 28.17, 23.31, 23.25, 20.58.
3-(5-Bromo-4-(4-methoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (10k): White solid; 0.49 g (87%); m.p. 188 °C (dec). 1H-NMR (DMSO-d6, 400 MHz) δ: 12.20 (brs, 1H), 8.29–8.31 (m, 1H), 7.67 (d, J = 8.0 Hz, 1H), 7.62–7.65 (m, 2H), 7.17 (d, J = 8.4 Hz, 1H), 7.03–7.06 (m, 1H), 4.08 (s, 3H), 2.59–2.66 (m, 3H), 2.47–2.52 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.83, 156.86, 156.51, 130.82, 129.77, 128.75, 127.66, 126.58, 125.16, 122.40, 121.19, 121.08, 104.13, 56.15, 29.90, 20.56.
3-(5-Bromo-4-(4-ethoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (10l): White solid; 0.52 g (89%); m.p. 108–110.5 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.20 (brs, 1H), 8.30–8.34 (m, 1H), 7.60–7.65 (m, 3H), 7.14 (d, J = 8.0 Hz, 1H), 7.01–7.05 (m, 1H), 4.33 (q, J = 6.9 Hz, 2H), 2.59–2.67 (m, 3H), 2.47–2.55 (m, 1H), 1.51 (t, J = 7.0 Hz, 3H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.81, 156.84, 155.75, 130.82, 129.81, 128.70, 127.66, 126.47, 125.22, 122.47, 121.03, 120.97, 104.66, 64.14, 29.89, 20.55, 14.47.
3-(5-Bromo-4-(4-bromonaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionic acid (10m): White solid; 0.58 g (91%); m.p. 202 °C (dec). 1H-NMR (DMSO-d6, 400 MHz) δ: 12.21 (brs, 1H), 8.32 (d, J = 8.4 Hz, 1H), 8.15 (d, J = 8.0 Hz, 1H), 7.85 (t, J = 7.8 Hz, 1H), 7.75 (d, J = 7.6 Hz, 1H), 7.72 (d, J = 8.0 Hz, 1H), 7.19 (d, J = 8.4 Hz, 1H), 2.60–2.69 (m, 3H), 2.49–2.56 (m, 1H). 13C-NMR (DMSO-d6, 100 MHz) δ: 172.80, 156.63, 131.83, 130.11, 130.02, 129.50, 129.22, 129.02, 127.71, 127.42, 124.91, 122.17, 29.87, 20.52.
3-(5-Bromo-4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)propionic acid (10n): White solid; 0.56 g (85%); m.p. 173–176 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.22 (brs, 1H), 8.22–8.26 (m, 2H), 7.76–7.82 (m, 3H), 6.36 (d, J = 8.0 Hz, 1H), 5.77 (s, 2H), 2.85 (t, J = 7.0 Hz, 2H), 2.69 (t, J = 7.2 Hz, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.68, 157.03, 131.97, 131.74, 131.52, 130.70, 130.38, 128.99, 128.46, 127.88, 124.48, 123.21, 122.49, 45.63, 30.62, 20.93.
3-(5-Bromo-4-(2-(4-bromonaphth-1-yl)ethyl)-4H-1,2,4-triazol-3-yl)propionic acid (10o): White solid; 0.57 g (84%); m.p. 190–192 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.21 (brs, 1H), 8.15–8.20 (m, 2H), 7.76 (d, J = 7.6 Hz, 1H), 7.66–7.73 (m, 2H), 7.11 (d, J = 8.0 Hz, 1H), 4.26 (t, J = 7.0 Hz, 2H), 3.46 (t, J = 7.0 Hz, 2H), 2.71 (t, J = 6.6 Hz, 2H), 2.62 (t, J = 6.4 Hz, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.10, 155.49, 133.82, 132.78, 131.28, 129.58, 128.82, 127.98, 127.68, 127.47, 127.22, 124.07, 121.26, 44.63, 31.63, 29.96, 19.93.
4-(5-Bromo-4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)butanoic acid (10p): White solid; 0.56 g (83%); m.p. 177–179 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 12.02 (brs, 1H), 8.21–8.25 (m, 2H), 7.76–7.82 (m, 3H), 6.30 (d, J = 8.0 Hz, 1H), 5.74 (s, 2H), 2.71 (t, J = 7.4 Hz, 2H), 2.27 (t, J = 7.2 Hz, 2H), 1.78–1.86 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 173.91, 156.81, 131.47, 131.05, 130.79, 129.95, 129.71, 128.30, 127.75, 127.18, 123.74, 122.27, 121.75, 45.01, 32.49, 23.84, 21.67.
5-(5-Bromo-4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)pentanoic acid (10q): White solid; 0.57 g (82%); m.p. 166.5–168 °C. 1H-NMR (DMSO-d6, 400 MHz) δ: 11.95 (brs, 1H), 8.21–8.25 (m, 2H), 7.76–7.83 (m, 3H), 6.31 (d, J = 8.0 Hz, 1H), 5.74 (s, 2H), 2.68 (t, J = 7.4 Hz, 2H), 2.14 (t, J = 7.2 Hz, 2H), 1.58–1.64 (m, 2H), 1.45–1.53 (m, 2H). 13C-NMR (DMSO-d6, 100 MHz) δ: 174.21, 157.03, 131.54, 131.07, 130.83, 129.84, 129.70, 128.30, 127.76, 127.19, 123.77, 122.39, 121.78, 44.99, 33.15, 25.57, 24.19, 23.79.
3.2.15. General Procedure for the Synthesis of 1a–1r
To a stirred mixture of 7a or 10a–10q (1 mmol, accurately weighted to four decimal places) in MeOH (5 mL) was added an aqueous solution of NaOH (prepared by dissolving 0.0400 g, 1 mmol of NaOH in a minimal volume of water), and the resulting mixture was stirred at room temperature until a clear solution was obtained (typically within 1 h).
The reaction mixture was filtered off and the filtrate was evaporated on a rotary evaporator to give rise to a residue, which was co-evaporated with CH2Cl2 (10 mL × 3) on a rotary evaporator and further dried in vacuo at room temperature to yield 1a–1r.
Sodium 3-(4-(4-cyclopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (1a): White solid; 0.32 g (98%); m.p. 140 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.59 (d, J = 8.4 Hz, 1H), 8.56 (s, 1H), 7.68 (t, J = 7.6 Hz, 1H), 7.60 (t, J = 7.6 Hz, 1H), 7.55 (d, J = 7.6 Hz, 1H), 7.42 (d, J = 7.6 Hz, 1H), 7.19 (d, J = 7.6 Hz, 1H), 2.75–2.80 (m, 2H), 2.43–2.53 (m, 3H), 1.16–1.19 (m, 2H), 0.82–0.85 (m, 2H). 13C-NMR (MeOH-d4, 100 MHz) δ: 179.27, 157.30, 146.36, 144.16, 135.44, 130.90, 129.21, 129.11, 128.23, 126.56, 126.33, 124.00, 122.79, 35.53, 22.43, 14.13, 7.59, 7.56. ESI-HRMS [M − Na]−: (m/z) calcd. for C18H16N3O2: 306.1243, found: 306.1235.
Sodium 3-(5-chloro-4-(4-cyclopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (1b): White solid; 0.36 g (99%); m.p. 135 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.61 (d, J = 8.4 Hz, 1H), 7.67–7.72 (m, 1H), 7.60–7.64 (m, 1H), 7.57 (d, J = 7.6 Hz, 1H), 7.45 (dd, J = 0.6 Hz and 7.8 Hz, 1H), 7.17 (d, J = 8.4 Hz, 1H), 2.65–2.78 (m, 2H), 2.40–2.59 (m, 3H), 1.16 (m, 2H), 0.84–0.88 (m, 2H). 13C-NMR (MeOH-d4, 100 MHz) δ: 178.94, 159.53, 144.97, 143.92, 135.58, 130.72, 129.35, 128.33, 127.83, 127.67, 126.50, 124.12, 122.56, 34.80, 23.54, 14.16, 7.63. ESI-HRMS [M − Na]−: (m/z) calcd. for C18H15ClN3O2: 340.0853, found: 340.0854.
Sodium 3-(5-bromo-4-(4-cyclopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (1c): White solid; 0.41 g (100%); m.p. 162 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.61 (d, J = 8.8 Hz, 1H), 7.68 (t, J = 7.4 Hz, 1H), 7.60 (t, J = 7.4 Hz, 1H), 7.55 (d, J = 7.6 Hz, 1H), 7.44 (d, J = 7.6 Hz, 1H), 7.13 (d, J = 8.4 Hz, 1H), 2.68–2.77 (m, 2H), 2.38–2.57 (m, 3H), 1.14–1.20 (m, 2H), 0.83–0.89 (m, 2H). 13C-NMR (MeOH-d4, 100 MHz) δ: 178.07, 159.87, 144.91, 135.57, 130.78, 129.67, 129.28, 128.60, 128.32, 127.75, 126.47, 124.10, 122.72, 34.04, 23.17, 14.16, 7.65. ESI-HRMS [M − Na]−: (m/z) calcd. for C18H15BrN3O2: 384.0348 (79Br), found: 384.0339; calcd. for C18H15BrN3O2: 386.0327 (81Br), found: 386.0317.
Sodium 3-(4-(4-cyclopropylnaphth-1-yl)-5-iodo-4H-1,2,4-triazol-3-yl)propionate (1d): White solid; 0.44 g (97%); m.p. 158 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.60 (d, J = 8.8 Hz, 1H), 7.68 (td, J = 0.9 Hz and 7.6 Hz, 1H), 7.57–7.61 (m, 1H), 7.50 (d, J = 7.6 Hz, 1H), 7.44 (d, J = 7.6 Hz, 1H), 7.07 (d, J = 8.4 Hz, 1H), 2.71–2.78 (m, 2H), 2.47–2.56 (m, 2H), 2.37–2.46 (m, 1H), 1.15–1.21 (m, 2H), 0.83-0.90 (m, 2H). 13C-NMR (MeOH-d4, 100 MHz) δ: 179.28, 160.15, 144.70, 135.57, 131.00, 129.99, 129.14, 128.27, 127.95, 126.43, 124.11, 122.99, 105.86, 35.45, 23.54, 14.18, 7.71, 7.60. ESI-HRMS [M − Na]−: (m/z) calcd. for C18H15IN3O2: 432.0209, found: 432.0200.
Sodium 3-(4-(4-cyclopropylnaphth-1-yl)-5-methoxy-4H-1,2,4-triazol-3-yl)propionate (1e): White solid; 0.36 g (99%); m.p. 133 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.57 (d, J = 8.4 Hz, 1H), 7.66 (dt, J = 1.1 Hz and 7.7 Hz, 1H), 7.58 (dt, J = 1.2 Hz and 7.6 Hz, 1H), 7.48 (d, J = 7.6 Hz, 1H), 7.40 (d, J = 7.6 Hz, 1H), 7.25 (d, J = 8.4 Hz, 1H), 3.99 (s, 3H), 2.65 (t, J = 8.0 Hz, 2H), 2.43–2.51 (m, 2H), 2.31–2.39 (m, 1H), 1.14–1.18 (m, 2H), 0.81–0.84 (m, 2H). 13C-NMR (MeOH-d4, 100 MHz) δ: 179.46, 161.32, 155.00, 144.05, 135.55, 131.08, 128.87, 128.03, 127.80, 127.35, 126.32, 124.13, 122.86, 58.56, 35.19, 23.38, 14.12, 7.53, 7.44. ESI-HRMS [M − Na]−: (m/z) calcd. for C19H18N3O3: 336.1348, found: 336.1346.
Sodium 3-(4-(4-cyclopropylnaphth-1-yl)-5-ethoxy-4H-1,2,4-triazol-3-yl)propionate (1f): White solid; 0.37 g (98%); m.p. 101.5 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.57 (d, J = 8.4 Hz, 1H), 7.66 (t, J = 7.0 Hz, 1H), 7.59 (t, J = 7.6 Hz, 1H), 7.48 (d, J = 7.6 Hz, 1H), 7.41 (d, J = 7.6 Hz, 1H), 7.26 (d, J = 8.0 Hz, 1H), 4.39 (q, J = 7.1 Hz, 2H), 2.64 (t, J = 7.6 Hz, 2H), 2.44–2.49 (m, 2H), 2.37–2.38 (m, 1H), 1.22 (t, J = 7.0 Hz, 3H), 1.14–1.19 (m, 2H), 0.82–0.86 (m, 2H). 13C-NMR (MeOH-d4, 100 MHz) δ: 178.85, 160.64, 154.61, 143.99, 135.57, 131.09, 128.82, 128.03, 127.89, 127.33, 126.33, 124.18, 122.93, 68.47, 34.74, 23.20, 14.63, 14.14, 7.51, 7.44. ESI-HRMS [M − Na]−: (m/z) calcd. for C20H20N3O3: 350.1505, found: 350.1497.
Sodium 3-(5-bromo-4-(naphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (1g): White solid; 0.37 g (100%); m.p. 182–184 °C. 1H-NMR (MeOH-d4, 400 MHz) δ: 8.18 (d, J = 7.6 Hz, 1H), 8.06–8.09 (m, 1H), 7.59–7.72 (m, 4H), 7.17–7.19 (m, 1H), 2.68–2.81 (m, 2H), 2.55–2.63 (m, 1H), 2.44–2.52 (m, 1H). 13C-NMR (MeOH-d4, 100 MHz) δ: 178.50, 159.84, 135.80, 132.52, 132.01, 130.82, 130.27, 129.92, 129.67, 128.52, 128.10, 126.72, 122.16, 34.33, 23.29. ESI-HRMS [M − Na]−: (m/z) calcd. for C15H11BrN3O2: 344.0035 (79Br), found: 344.0030; calcd. for C15H11BrN3O2: 346.0014 (81Br), found: 346.0010.
Sodium 3-(5-bromo-4-(4-methylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (1h): White solid; 0.38 g (99%); m.p. 155 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.21 (d, J = 8.4 Hz, 1H), 7.65–7.69 (m, 1H), 7.59–7.63 (m, 1H), 7.54 (s, 2H), 7.13–7.15 (m, 1H), 2.80 (s, 3H), 2.71–2.78 (m, 2H), 2.50–2.58 (m, 1H), 2.38–2.49 (m, 1H). 13C-NMR (MeOH-d4, 100 MHz) δ: 179.26, 160.14, 139.95, 134.69, 132.22, 130.82, 129.24, 128.69, 128.36, 127.74, 127.29, 126.28, 122.72, 35.11, 23.60, 19.64. ESI-HRMS [M − Na]−: (m/z) calcd. for C16H13BrN3O2: 358.0190 (79Br), found: 358.0192; calcd. for C16H13BrN3O2: 360.0171 (81Br), found: 360.0171.
Sodium 3-(5-bromo-4-(4-ethylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (1i): White solid; 0.38 g (97%); m.p. 198 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.27 (d, J = 8.4 Hz, 1H), 7.64–7.69 (m, 2H), 7.57–7.61 (m, 2H), 7.14 (d, J = 8.0 Hz, 1H), 3.23 (q, J = 7.6 Hz, 2H), 2.71–2.76 (m, 2H), 2.52–2.59 (m, 1H), 2.42–2.48 (m, 1H), 1.43 (t, J = 7.4 Hz, 3H). 13C-NMR (MeOH-d4, 100 MHz) δ: 179.13, 160.07, 145.72, 133.80, 132.18, 131.00, 129.11, 128.59, 128.32, 127.81, 125.88, 125.71, 122.82, 34.91, 26.90, 23.52, 15.37. ESI-HRMS [M − Na]−: (m/z) calcd. for C17H15BrN3O2: 372.0348 (79Br), found: 372.0349; calcd. for C17H15BrN3O2: 374.0327 (81Br), found: 374.0333.
Sodium 3-(5-bromo-4-(4-n-propylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (1j): White solid; 0.41 g (99%); m.p. 183 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.25 (d, J = 8.4 Hz, 1H), 7.64–7.68 (m, 1H), 7.53–7.61 (m, 3H), 7.14 (d, J = 8.0 Hz, 1H), 3.17 (t, J = 7.6 Hz, 2H), 2.67–2.76 (m, 2H), 2.54–2.62 (m, 1H), 2.43–2.51 (m, 1H), 1.79–1.88 (m, 2H), 1.08 (t, J = 7.4 Hz, 3H). 13C-NMR (MeOH-d4, 100 MHz) δ: 178.64, 159.97, 144.27, 134.04, 132.22, 131.11, 129.11, 128.65, 128.27, 127.64, 126.75, 126.07, 122.85, 36.06, 34.50, 25.06, 23.36, 14.47. ESI-HRMS [M − Na]−: (m/z) calcd. for C18H17BrN3O2: 386.0504 (79Br), found: 386.0509; calcd. for C18H17BrN3O2: 388.0484 (81Br), found: 388.0498.
Sodium 3-(5-bromo-4-(4-isopropylnaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (1k): White solid; 0.41 g (100%); m.p. 194 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.35 (d, J = 8.4 Hz, 1H), 7.57–7.69 (m, 4H), 7.14 (d, J = 8.4 Hz, 1H), 3.86-3.92 (m, 1H), 2.66-2.78 (m, 2H), 2.53–2.61 (m, 1H), 2.42–2.50 (m, 1H), 1.46 (d, J = 6.8 Hz, 3H), 1.46 (d, J = 6.8 Hz, 3H). 13C-NMR (MeOH-d4, 100 MHz) δ: 178.99, 160.08, 150.11, 133.54, 132.23, 131.13, 129.02, 128.48, 128.30, 127.84, 125.45, 122.95, 122.77, 34.82, 29.98, 23.83, 23.50. ESI-HRMS [M − Na]−: (m/z) calcd. for C18H17BrN3O2: 386.0504 (79Br), found: 386.0508; calcd. for C18H17BrN3O2: 388.0484 (81Br), found: 388.0494.
Sodium 3-(5-bromo-4-(4-methoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (1l): White solid; 0.39 g (98%); m.p. 211 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.36-8.38 (m, 1H), 7.57–7.62 (m, 3H), 7.09 (d, J = 8.4 Hz, 1H), 7.05–7.07 (m, 1H), 4.11 (s, 3H), 2.72–2.77 (m, 2H), 2.53–2.61 (m, 1H), 2.44–2.50 (m, 1H). 13C-NMR (MeOH-d4, 100 MHz) δ: 178.60, 160.20, 158.96, 132.70, 131.71, 129.92, 128.86, 127.60, 127.38, 124.02, 122.59, 122.01, 104.54, 56.63, 34.57, 23.37. ESI-HRMS [M − Na]−: (m/z) calcd. for C16H13BrN3O3: 374.0140 (79Br), found: 374.0137; calcd. for C16H13BrN3O3: 376.0120 (81Br), found: 376.0115.
Sodium 3-(5-bromo-4-(4-ethoxynaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (1m): White solid; 0.40 g (96%); m.p. 194 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.38–8.41 (m, 1H), 7.58–7.61 (m, 3H), 7.05–7.07 (m, 2H), 4.33 (q, J = 7.1 Hz, 2H), 2.67–2.77 (m, 2H), 2.47–2.64 (m, 2H), 1.58 (t, J = 7.0 Hz, 3H). 13C-NMR (MeOH-d4, 100 MHz) δ: 178.02, 160.05, 158.23, 132.75, 131.73, 129.87, 128.84, 127.51, 127.45, 124.08, 122.33, 121.98, 105.17, 65.59, 33.99, 23.13, 14.97. ESI-HRMS [M − Na]−: (m/z) calcd. for C17H15BrN3O3: 388.0297 (79Br), found: 388.0299; calcd. for C17H15BrN3O3: 390.0276 (81Br), found: 390.0283.
Sodium 3-(5-bromo-4-(4-bromonaphth-1-yl)-4H-1,2,4-triazol-3-yl)propionate (1n): White solid; 0.43 g (97%); m.p. 204 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.40 (d, J = 8.8 Hz, 1H), 8.05 (d, J = 7.6 Hz, 1H), 7.76–7.81 (m, 1H), 7.69–7.73 (m, 1H), 7.61 (d, J = 8.0 Hz, 1H), 7.22 (d, J = 8.4 Hz, 1H), 2.71–2.76 (m, 2H), 2.55–2.63 (m, 1H), 2.46–2.52 (m, 1H). 13C-NMR (MeOH-d4, 100 MHz) δ: 178.68, 159.89, 134.02, 131.96, 131.84, 131.03, 130.63, 130.38, 130.09, 129.09, 128.72, 127.02, 123.14, 34.54, 23.36. ESI-HRMS [M − Na]−: (m/z) calcd. for C15H10Br2N3O2: 423.9119, found: 423.9115.
Sodium 3-(5-bromo-4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)propionate (1o): White solid; 0.46 g (100%); m.p. 152.5 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.32–8.34 (m, 1H), 8.18–8.20 (m, 1H), 7.71–7.75 (m, 3H), 6.43 (d, J = 7.6 Hz, 1H), 5.84 (s, 2H), 2.94 (t, J = 7.4 Hz, 2H), 2.65 (t, J = 7.4 Hz, 2H). 13C-NMR (MeOH-d4, 100 MHz) δ: 178.62, 159.53, 133.22, 132.53, 131.92, 131.65, 130.71, 129.04, 128.94, 128.80, 124.18, 124.01, 123.78, 46.84, 34.65, 22.95. ESI-HRMS [M − Na]−: (m/z) calcd. for C16H12Br2N3O2: 437.9276, found: 437.9272.
Sodium 3-(5-bromo-4-(2-(4-bromonaphth-1-yl)ethyl)-4H-1,2,4-triazol-3-yl)propionate (1p): White solid; 0.47 g (99%); m.p. 167 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.24–8.28 (m, 1H), 8.07–8.11 (m, 1H), 7.68 (d, J = 7.6 Hz, 1H), 7.61–7.66 (m, 2H), 7.07 (d, J = 7.6 Hz, 1H), 4.42 (t, J = 7.0 Hz, 2H), 3.54 (t, J = 6.8 Hz, 2H), 2.69 (t, J = 7.0 Hz, 2H), 2.57 (t, J = 7.2 Hz, 2H). 13C-NMR (MeOH-d4, 100 MHz) δ: 178.88, 158.84, 134.75, 134.50, 133.41, 130.81, 130.53, 129.16, 128.92, 128.60, 128.48, 124.85, 123.29, 46.60, 34.83, 33.24, 22.69. ESI-HRMS [M − Na]−: (m/z) calcd. for C17H14Br2N3O2: 451.9432, found: 451.9426.
Sodium 4-(5-bromo-4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)butanoate (1q): White solid; 0.47 g (98%); m.p. 166 °C (dec). 1H-NMR (MeOH-d4, 400 MHz) δ: 8.30–8.33 (m, 1H), 8.16–8.19 (m, 1H), 7.71–7.75 (m, 3H), 6.39 (d, J = 8.0 Hz, 1H), 5.79 (s, 2H), 2.78 (t, J = 7.8 Hz, 2H), 2.21 (t, J = 7.2 Hz, 2H), 1.95–2.02 (m, 2H). 13C-NMR (MeOH-d4, 100 MHz) δ: 180.55, 159.43, 133.19, 132.49, 131.85, 131.78, 130.65, 129.05, 128.97, 128.91, 124.18, 124.04, 123.66, 46.83, 37.21, 25.94, 24.57. ESI-HRMS [M − Na]−: (m/z) calcd. for C17H14Br2N3O2: 451.9432, found: 451.9434.
Sodium 5-(5-bromo-4-((4-bromonaphth-1-yl)methyl)-4H-1,2,4-triazol-3-yl)pentanoate (1r): White solid; 0.48 g (98%); m.p. 146–149 °C. 1H-NMR (MeOH-d4, 400 MHz) δ: 8.32–8.34 (m, 1H), 8.16–8.19 (m, 1H), 7.71–7.76 (m, 3H), 6.42 (d, J = 8.0 Hz, 1H), 5.77 (s, 2H), 2.75 (t, J = 7.4 Hz, 2H), 2.11 (t, J = 7.4 Hz, 2H), 1.67–1.74 (m, 2H), 1.56–1.63 (m, 2H). 13C-NMR (MeOH-d4, 100 MHz) δ: 181.17, 159.48, 133.23, 132.52, 131.86, 131.78, 130.67, 129.09, 129.01, 128.98, 124.17, 124.13, 123.84, 46.83, 37.64, 27.66, 26.65, 26.05. ESI-HRMS [M-Na]−: (m/z) calcd. for C18H16Br2N3O2: 465.9589, found: 465.9588.
3.3. In Vitro URAT1 Inhibitory Activity
The in vitro URAT1 inhibitory activity of the compounds 1a–1r and lesinurad (in the form of sodium salt as well) was determined by the inhibition of URAT1-mediated [8-14C]uric acid uptake by human embryonic kidney 293 (HEK293) cells stably expressing human URAT1 (HEK293-URAT1 cells). Thus, HEK293-URAT1 cells were subcultured for 2–3 generations in DMEM (with 10% FBS, 100 IU/mL penicillin, and 50 μg/mL streptomycin at 37 °C, 5% CO2, and 95% air). After 48 h, when the HEK293-URAT1 cells were grown to confluence, the cells were treated by 0.25% trypsin for 1–2 min. Thereafter, the digested cells were diluted to a density of 1.5 × 105/mL and inoculated into the 24-well cell culture plates for about 48 h. The HEK293-URAT1 cells were pre-incubated for 10 min in HBSS (Cl− free) and then incubated in the uptake solution (PBS) containing 5 μM [8-14C]uric acid (2 μCi/mL) with or without the tested compounds (at a series of concentrations). The HEK293-URAT1 cells were then washed three times with ice-cold HBSS (Cl− free) and treated by 0.1 M aqueous NaOH solution. After that, 500 μL of cell lysis was transferred to scintillation vials and solubilized with scintillation solution. The radioactivity in the cells was measured by a liquid scintillation counter (PerkinElmer, Waltham, MA, USA). The concentrations of the compounds to inhibit 50% [8-14C]uric acid uptake by HEK293 cells (IC50) were calculated with GraphPad Prism software (San Diego, CA, USA).
4. Conclusions
Systematic SAR exploration of a lesinurad-based hit 1c led to the discovery of a highly potent URAT1 inhibitor, 1q, which was 31-fold more potent than the parent lesinurad (IC50 = 0.23 μM against human URAT1 for 1q vs. 7.18 μM for lesinurad). URAT1 inhibitor 1q was structurally flexible as compared with parent lesinurad and could probably serve as a promising prototype scaffold for the further design of URAT1 inhibitors. The SAR study also demonstrated that the S atom in lesinurad was not indispensable for its URAT1-inhibitory activity. We will go on to perform some studies on some drug-like properties such as logP and solubility, and further SAR exploration of the naphthalene ring.
Acknowledgments
The financial support from Key Projects of Tianjin Science and Technology Support Plan (16YFZCSY00910) was gratefully acknowledged.
Author Contributions
G.Z. and J.W. conceived and designed the experiments; H.T., W.L., Z.Z., Q.S., Y.L. and Y.X. performed the experiments; C.L., W.X. and L.T. contributed helpful discussions. G.Z. analyzed the data; G.Z. wrote the paper. All authors discussed and agreed with the results and conclusions in the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Punzi, L.; Scanu, A.; Ramonda, R.; Oliviero, F. Gout as autoinflammatory disease: New mechanisms for more appropriated treatment targets. Autoimmun. Rev. 2012, 12, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Richette, P.; Bardin, T. Gout. Lancet 2010, 375, 318–328. [Google Scholar] [CrossRef]
- Abeles, A.M. Hyperuricemia, gout, and cardiovascular disease: An update. Curr. Rheumatol. Rep. 2015, 17, 13. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, D.; Unwin, R. The pathophysiology of hyperuricaemia and its possible relationship to cardiovascular disease, morbidity and mortality. BMC Nephrol. 2013, 14, 164. [Google Scholar] [CrossRef] [PubMed]
- Liebman, S.E.; Taylor, J.G.; Bushinsky, D.A. Uric acid nephrolithiasis. Curr. Rheumatol. Rep. 2007, 9, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Nuki, G.; Simkin, P.A. A concise history of gout and hyperuricemia and their treatment. Arthritis Res. Ther. 2006, 8, S1. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kuo, C.-F.; Grainge, M.J.; Zhang, W.; Doherty, M. Global epidemiology of gout: Prevalence, incidence and risk factors. Nat. Rev. Rheumatol. 2015, 11, 649–662. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Pandya, B.J.; Choi, H.K. Prevalence of gout and hyperuricemia in the US general population: The national health and nutrition examination survey 2007–2008. Arthritis Rheum. 2011, 63, 3136–3141. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhang, X.-M.; Wang, L.-Y.; Liu, B.-C. Prevalence of hyperuricemia among Chinese adults: A national cross-sectional survey using multistage, stratified sampling. J. Nephrol. 2014, 27, 653–658. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.; Hoy, D.; Cross, M.; Merriman, T.R.; Vos, T.; Buchbinder, R.; Woolf, A.; March, L. The global burden of gout: Estimates from the global burden of disease 2010 study. Ann. Rheum. Dis. 2014, 73, 1470–1476. [Google Scholar] [CrossRef] [PubMed]
- Wertheimer, A.; Morlock, R.; Becker, M.A. A revised estimate of the burden of illness of gout. Curr. Ther. Res. 2013, 75, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Bernal, J.A.; Quilis, N.; Andrés, M.; Sivera, F.; Pascual, E. Gout: Optimizing treatment to achieve a disease cure. Ther. Adv. Chronic. Dis. 2016, 7, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Gliozzi, M.; Malara, N.; Muscoli, S.; Mollace, V. The treatment of hyperuricemia. Int. J. Cardiol. 2016, 213, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.A. Emerging therapies for gout. Expert Opin. Emerg. Drugs 2012, 17, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Shahid, H.; Singh, J.A. Investigational drugs for hyperuricemia. Expert Opin. Investig. Drugs 2015, 24, 1013–1030. [Google Scholar] [CrossRef] [PubMed]
- Burns, C.M.; Wortmann, R.L. Gout therapeutics: New drugs for an old disease. Lancet 2011, 377, 165–177. [Google Scholar] [CrossRef]
- Enomoto, A.; Kimura, H.; Chairoungdua, A.; Shigeta, Y.; Jutabha, P.; Cha, S.H.; Hosoyamada, M.; Takeda, M.; Sekine, T.; Igarashi, T.; et al. Molecular identification of a renal urate anion exchanger that regulates blood urate levels. Nature 2002, 417, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Hyndman, D.; Liu, S.; Miner, J.N. Urate handling in the human body. Curr. Rheumatol. Rep. 2016, 18, 34. [Google Scholar] [CrossRef] [PubMed]
- Hoy, S.M. Lesinurad: First global approval. Drugs 2016, 76, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Stocks, M.J.; Cheshire, D.R.; Reynolds, R. Efficient and regiospecific one-pot synthesis of substituted 1,2,4-triazoles. Org. Lett. 2004, 6, 2969–2971. [Google Scholar] [CrossRef] [PubMed]
- Wender, P.A.; Koehler, M.F.T.; Sendzik, M. A new synthetic approach to the C ring of known as well as novel bryostatin analogues. Org. Lett. 2003, 5, 4549–4552. [Google Scholar] [CrossRef] [PubMed]
- Bal, B.S.; Childers, W.E., Jr.; Pinnick, H.W. Oxidation of α,β-unsaturated aldehydes. Tetrahedron 1981, 37, 2091–2096. [Google Scholar] [CrossRef]
- Takahashi, B.; Funami, H.; Iwaki, T.; Maruoka, H.; Shibata, M.; Koyama, M.; Nagahira, A.; Kamiide, Y.; Kanki, S.; Igawa, Y.; et al. Orally active ghrelin receptor inverse agonists and their actions on a rat obesity model. Bioorg. Med. Chem. 2015, 23, 4792–4803. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Deng, Y.; Wu, C.; Zhang, Y.; Wang, J. Synthesis of α-aryl esters and nitriles: Deaminative coupling of α-aminoesters and α-aminoacetonitriles with arylboronic acids. Angew. Chem. Int. Ed. 2014, 53, 10510–10514. [Google Scholar] [CrossRef] [PubMed]
- Nambo, M.; Yar, M.; Smith, J.D.; Crudden, C.M. The concise synthesis of unsymmetric triarylacetonitriles via Pd-catalyzed sequential arylation: A new synthetic approach to tri- and tetraarylmethanes. Org. Lett. 2015, 17, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wu, J.; Liu, W.; Liu, Y.; Xie, Y.; Shang, Q.; Zhou, Z.; Xu, W.; Tang, L.; Wang, J.; et al. Discovery of flexible naphthyltriazolylmethane-based thioacetic acids as highly active uric acid transporter 1 (URAT1) inhibitors for the treatment of hyperuricemia of gout. Med. Chem. 2016. [Google Scholar] [CrossRef]
- Zhao, M.M.; Li, J.; Mano, E.; Song, Z.J.; Tschaen, D.M. Oxidation of primary alcohols to carboxylic acids with sodium chlorite catalyzed by tempo and bleach: 4-methoxyphenylacetic acid. Org. Synth. 2005, 81, 195–203. [Google Scholar]
- Sun, H.; Hu, W.; Wang, Z.-H.; Li, Y.-Z.; Zhao, G.-L.; Wang, J.-W. Synthesis of two modified intermediates in epothilone biosynthesis. Chin. J. Org. Chem. 2008, 28, 657–662. [Google Scholar]
- Gould, E.; Lebl, T.; Slawin, A.M.Z.; Reid, M.; Smith, A.D. Structural effects in yrazolidinone-mediated organocatalytic Dielse-Alder reactions. Tetrahedron 2010, 66, 8992–9008. [Google Scholar] [CrossRef]
- Hoffman, A. Beta-phenylisobutylmethyl ketone and its derivatives. The synthesis of alpha- and of beta-phenylisovalerianic acid. J. Am. Chem. Soc. 1929, 51, 2542–2547. [Google Scholar] [CrossRef]
- Morris, D.J.; Hayes, A.M.; Wills, M. The “reverse-tethered” Ruthenium (II) catalyst for asymmetric transfer hydrogenation: Further applications. J. Org. Chem. 2006, 71, 7035–7044. [Google Scholar] [CrossRef] [PubMed]
- Gallant, M.; Chauret, N.; Claveau, D.; Day, S.; Deschênes, D.; Dubé, D.; Huang, Z.; Lacombe, P.; Laliberté, F.; Lévesque, J.-F.; et al. Design, synthesis, and biological evaluation of 8-biarylquinolines: A novel class of PDE4 inhibitors. Bioorg. Med. Chem. Lett. 2008, 18, 1407–1412. [Google Scholar] [CrossRef] [PubMed]
- Beck, A.L.; Mascal, M.; Moody, C.J.; Coates, W.J. Synthesis of 3,4-bridged indoles by photocyclisation reactions. Part 2. Photocyclisation of halogenoacetyl tryptophol derivatives and α-chloro indol-3-ylalkanoate esters. J. Chem. Soc. Perkin Trans. 1 1992, 813–821. [Google Scholar] [CrossRef]
- Futatsugi, K.; Kung, D.W.; Orr, S.T.; Cabral, S.; Hepworth, D.; Aspnes, G.; Bader, S.; Bian, J.; Boehm, M.; Carpino, P.A.; et al. Discovery and optimization of imidazopyridine-based inhibitors of diacylglycerol acyltransferase 2 (DGAT2). J. Med. Chem. 2015, 58, 7173–7185. [Google Scholar] [CrossRef] [PubMed]
- Ishii, Y.; Yoshida, T.; Yamawaki, K.; Ogawa, M. Lactone synthesis by a,ω-diols with hydrogen peroxide catalyzed by heteropoly acids combined with cetylpyridinium chloride. J. Org. Chem. 1988, 53, 5549–5552. [Google Scholar] [CrossRef]
- Little, R.D.; Muller, G.W.; Venegas, M.G.; Carroll, G.L.; Bukhari, A.; Stone, K. 1,3-Diyl trapping reactions: Fundamental investigations with application to the synthesis of linearly fused tricyclopentanoids. Tetrahedron 1981, 37, 4371–4383. [Google Scholar] [CrossRef]
- Gilchrist, T.L.; Richards, P. Intramolecular cycloaddition reactions of in situ generated azoalkenes: Synthesis of pyrrolo[1,2-b]pyridazine derivatives from α-haloketone 4-pentenotlhydrazones. Synthesis 1983, 1983, 153–154. [Google Scholar] [CrossRef]
- McElvain, S.M.; McKay, G.R., Jr. Ketene acetals. XXXV. Cyclic ketene acetals and orthoesters from 2,2-dimethoxy-2,3-dihydropyran. J. Am. Chem. Soc. 1955, 77, 5601–5606. [Google Scholar] [CrossRef]
- Huisgen, R.; Ott, H. Die konfiguration der carbonestergruppe und die sondereigenschaften der lactone. Tetrahedron 1959, 6, 253–267. [Google Scholar] [CrossRef]
- Rink, M.; Krebber, D.; Fanslau, D.; Mehta, S. Synthese von durchhydrierten dipiperidazino-tetrazin, -tetrazepin- und -tetrazozinderivaten. Arch. Pharm. 1966, 299, 254–262. [Google Scholar] [CrossRef]
- Gilchrist, T.L.; Wasson, R.C. Intramolecular cycloaddition of azoalkenes derived from terminal alkenoic and alkynoic acids. J. Chem. Soc. Perkin Trans. 1 1987, 2511–2516. [Google Scholar] [CrossRef]
- Gresham, T.L.; Jansen, J.E.; Shaver, S.W.; Bankert, R.A.; Fiedorek, F.T. β-Propiolactone. XI. Reactions with ammonia and amines. J. Am. Chem. Soc. 1951, 73, 3168–3171. [Google Scholar] [CrossRef]
- Ottaway, J.H. A preparation of d(−)-β-hydroxybutyric acid. Biochem. J. 1962, 84, 11–12. [Google Scholar] [CrossRef] [PubMed]
- Boekelheide, V.; Goldman, M. A synthesis of 3-bromoperinaphthanol-7. J. Am. Chem. Soc. 1954, 76, 604–605. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1a–1r are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).