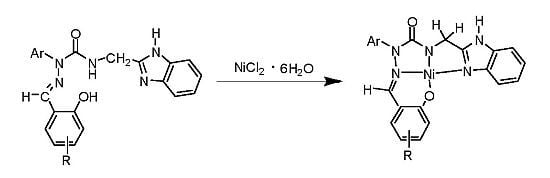
Syntheses of Nickel (II) Complexes from Novel Semicarbazone Ligands with Chloroformylarylhydrazine, Benzimidazole and Salicylaldehyde Moieties
Abstract
:1. Introduction
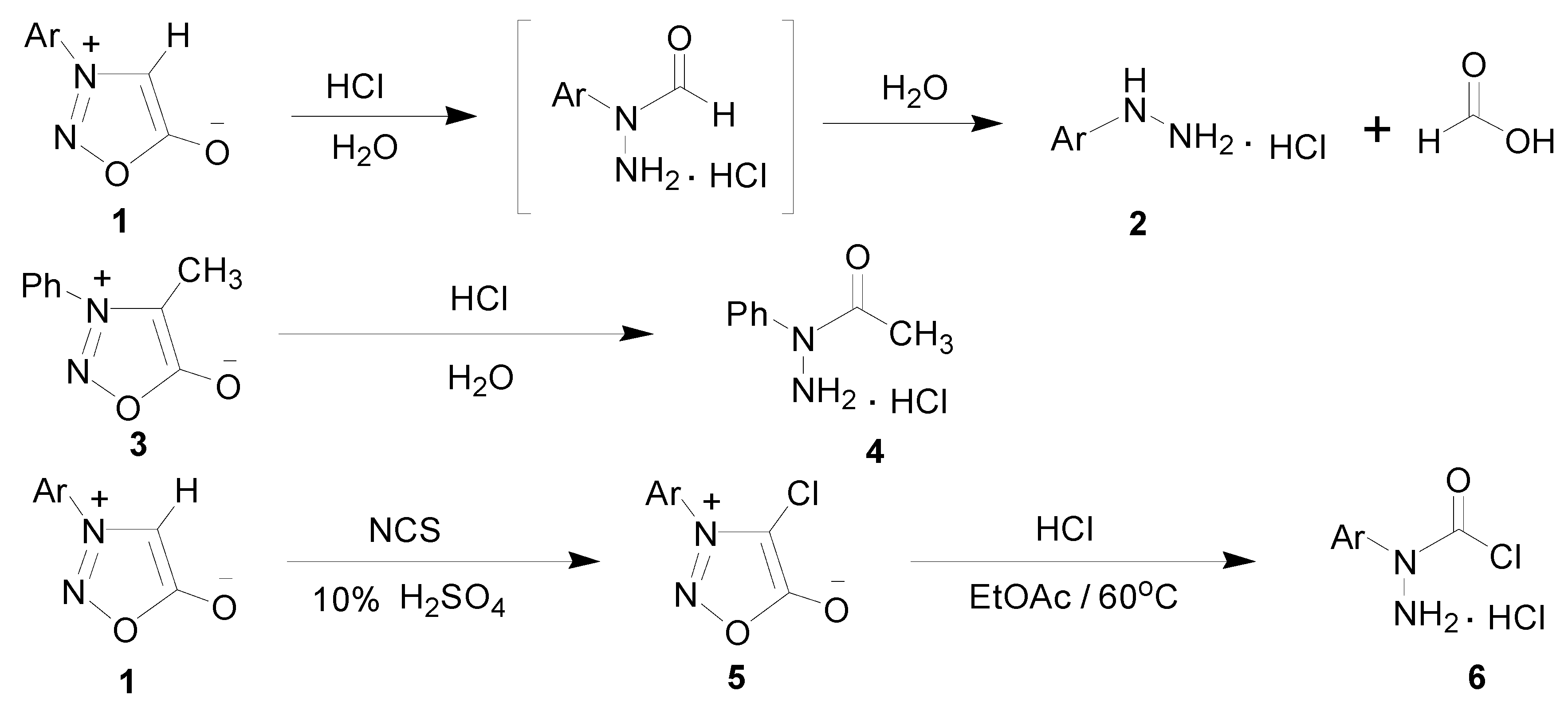
2. Results and Discussion
2.1. Synthetic Chemistry
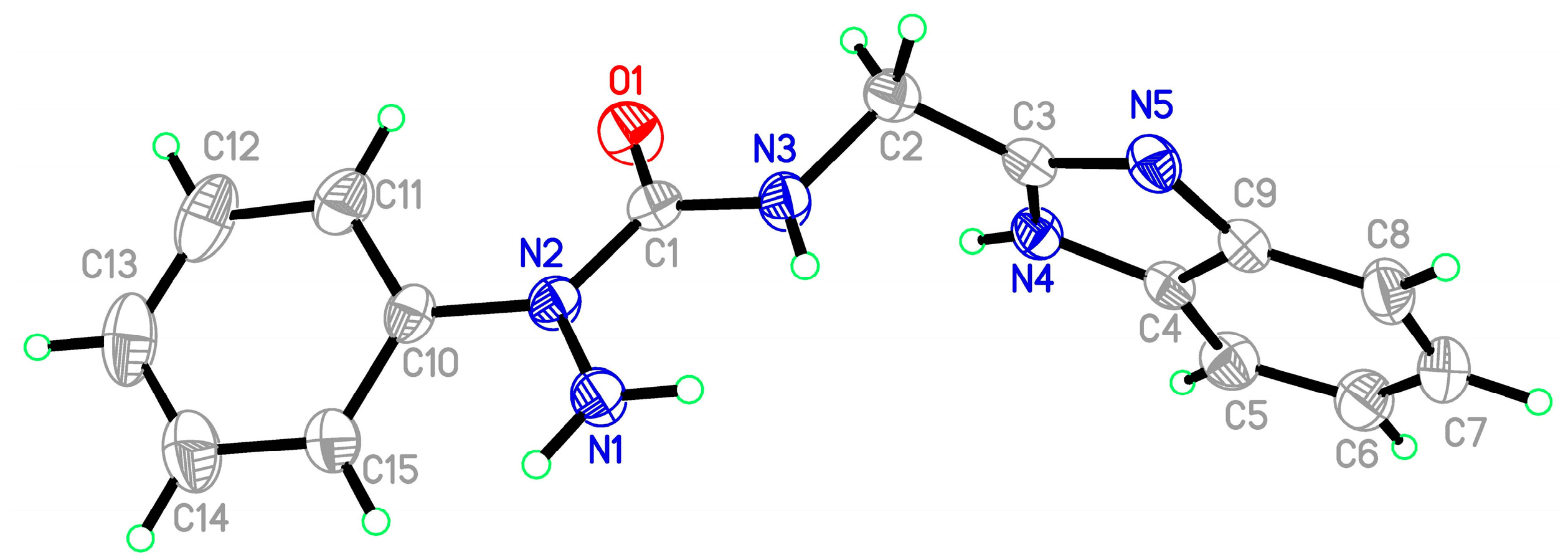

2.2. Spectroscopy Studies of Ligands and Metal Complexes
2.2.1. IR and NMR Studies
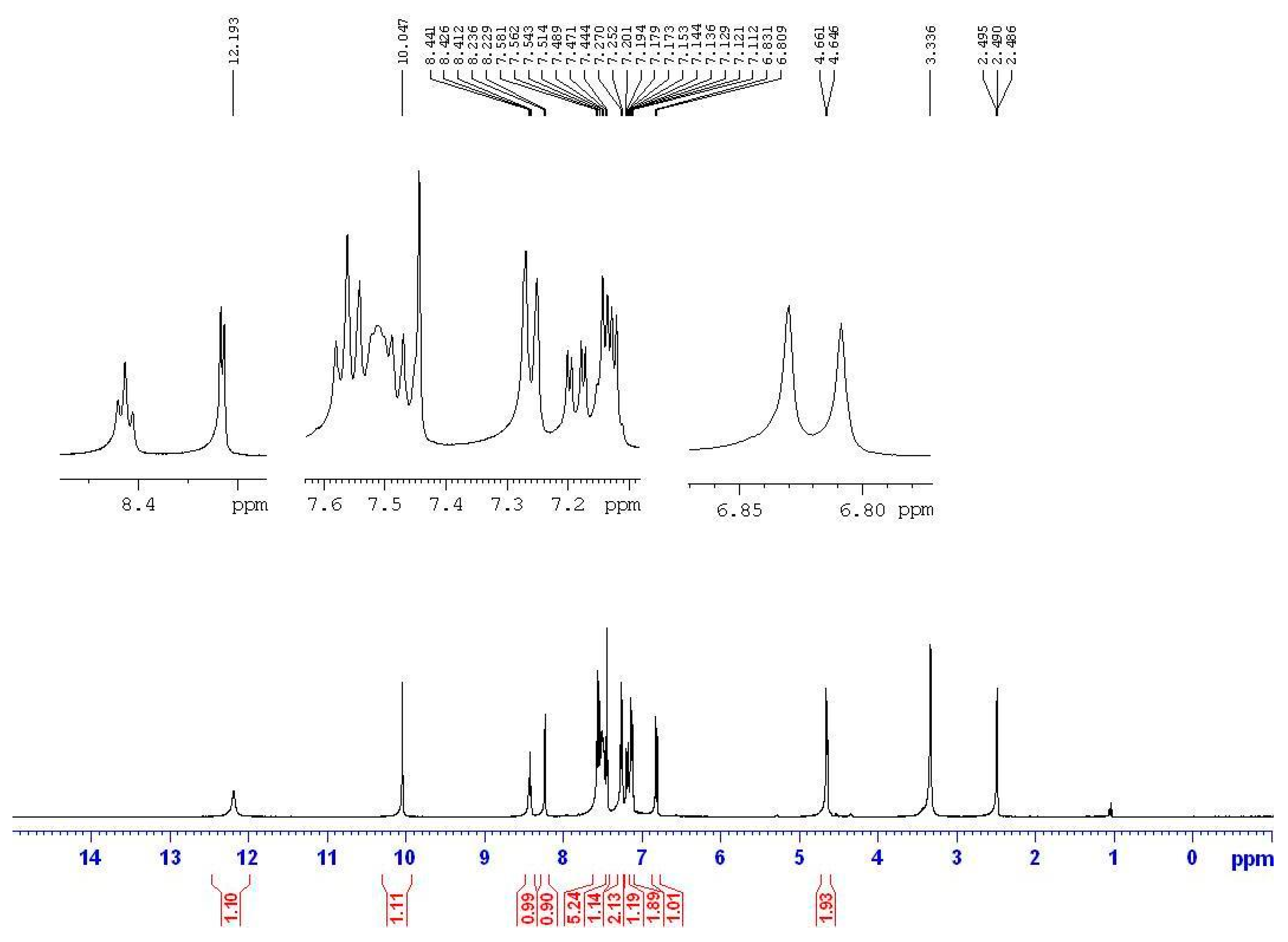
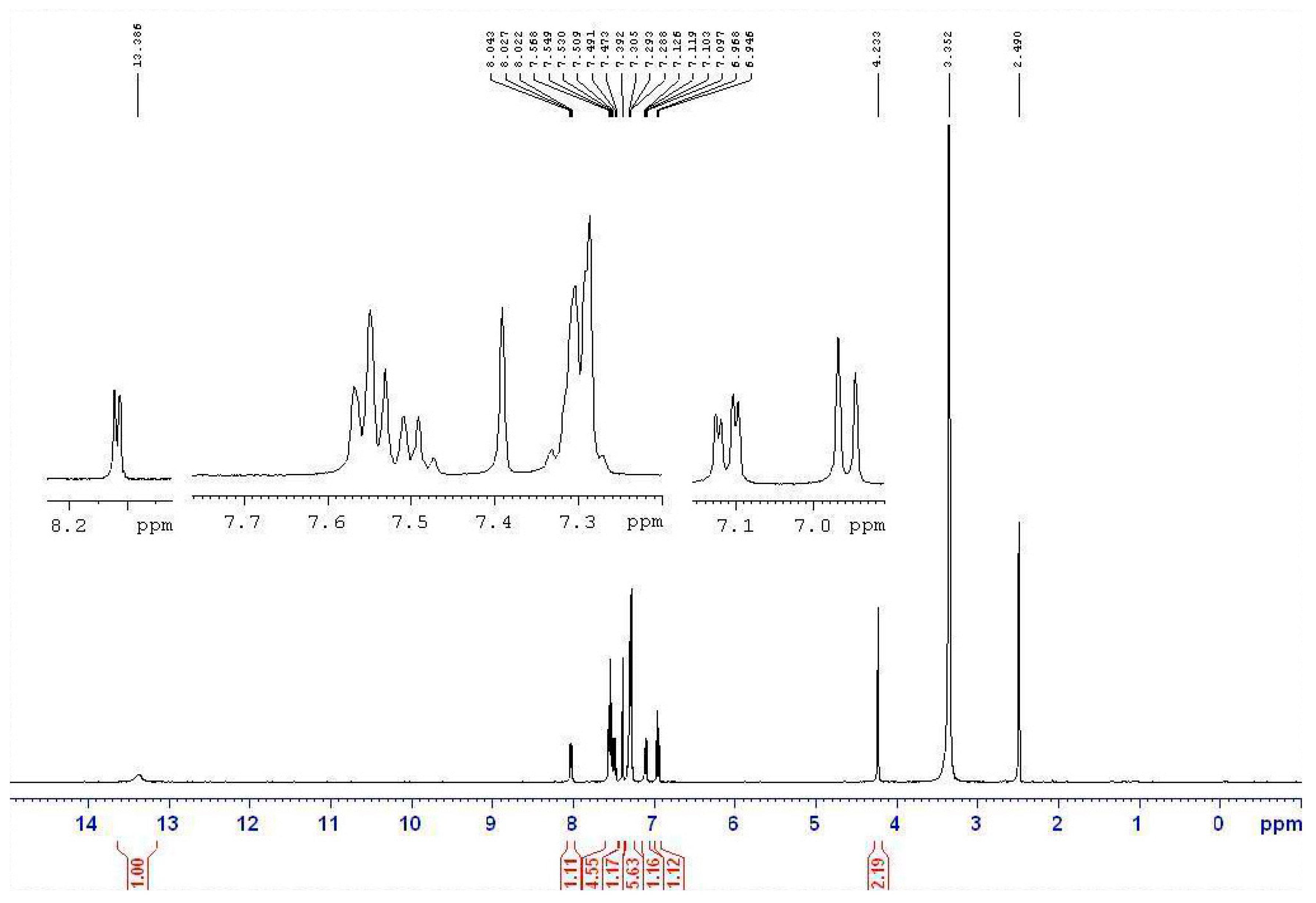
2.2.2. MS Study
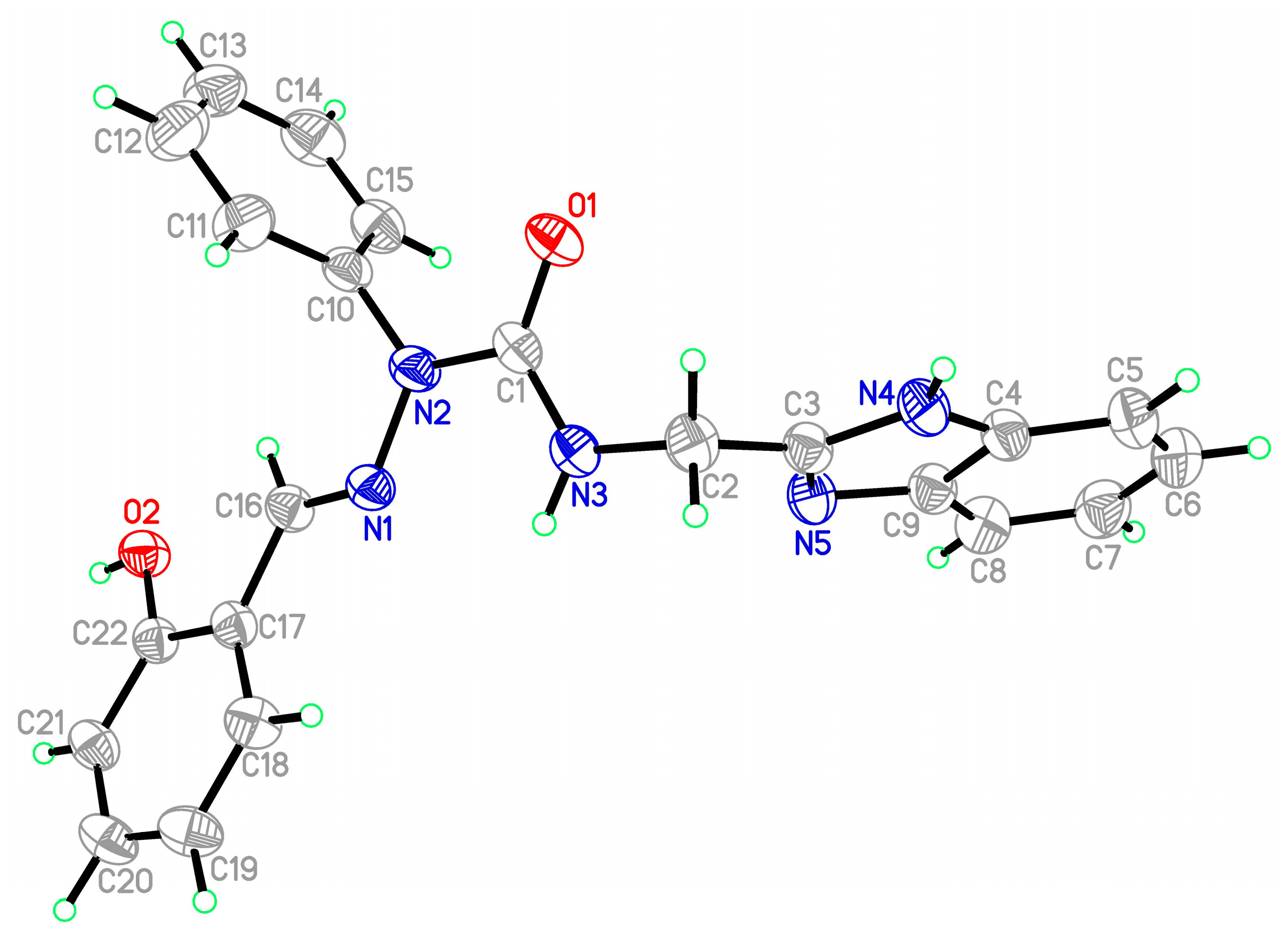
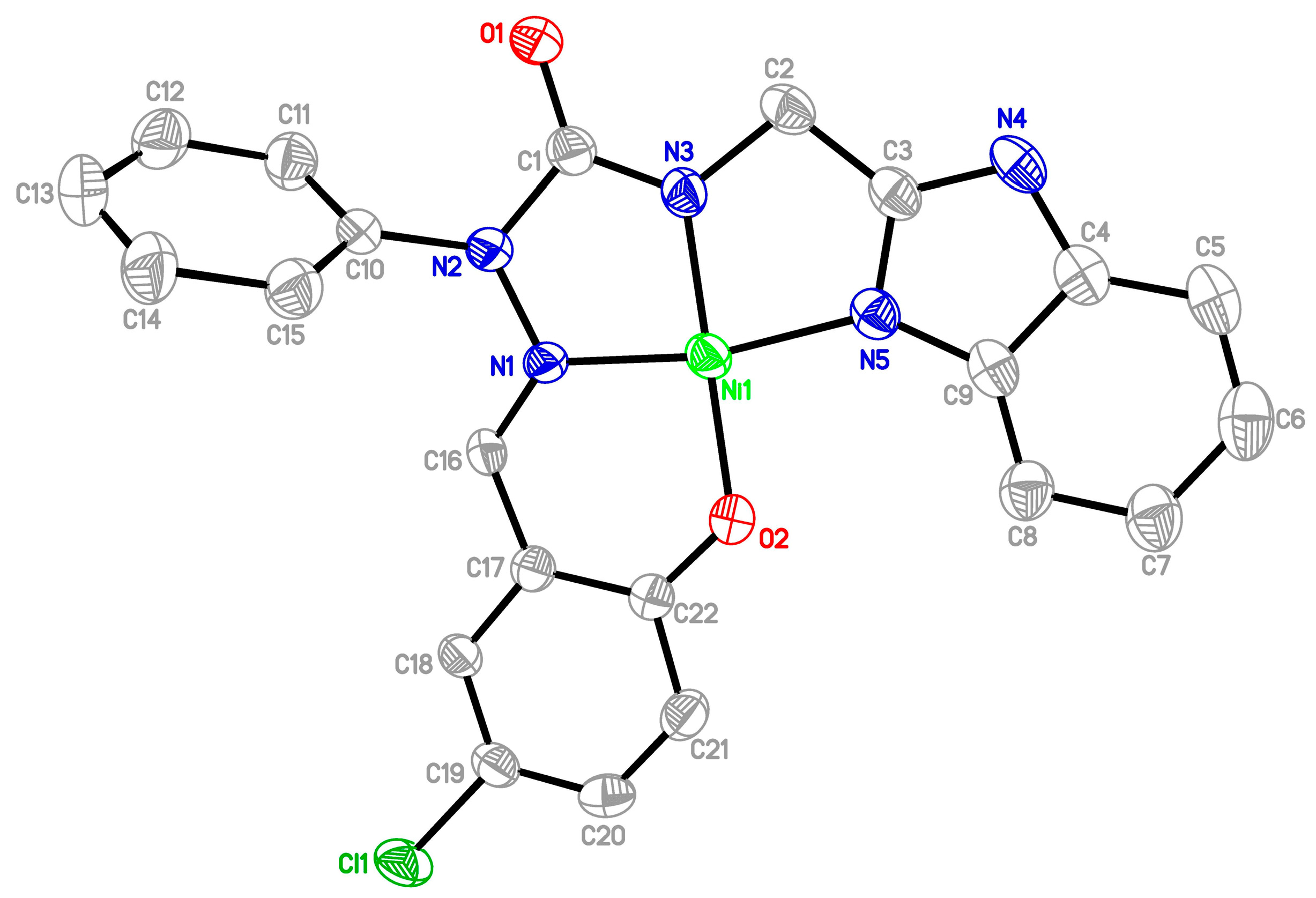
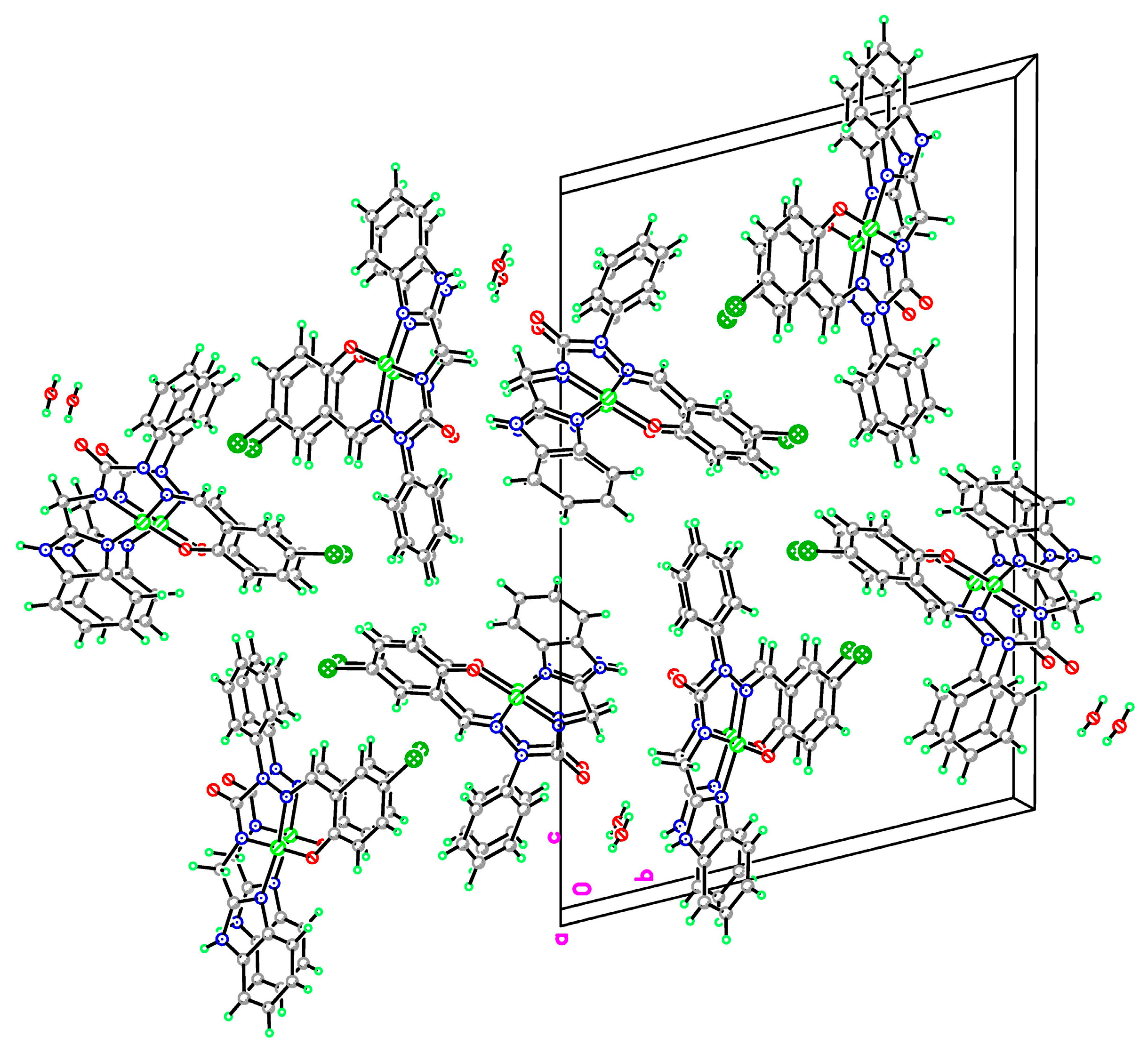
2.2.3. X-ray Study of Ligands and Complexes
| Compounds | 8a | 8b |
|---|---|---|
| Diffractometer | Nonius Kappa CCD | Nonius Kappa CCD |
| Formula | C15H15N5O | C16H17N5O·H2O |
| Formula weight | 281.32 | 313.36 |
| Crystal system | Monoclinic | Monoclinic |
| Space group | P2(1)/c | P2(1)/c |
| a/Å | 14.0940(3) | 15.3794(7) |
| b/Å | 10.0209(2) | 8.2222(5) |
| c/Å | 10.1067(2) | 13.8159(7) |
| α/° | 90.00 | 90 |
| β/° | 104.088(2) | 104.331(5) |
| γ/° | 90.00 | 90 |
| V/Å3 | 1384.48(5) | 1692.69(15) |
| Z | 4 | 4 |
| Dcalc (g·cm−3) | 1.350 | 1.230 |
| F000 | 592 | 664 |
| Absorption coefficient (mm−1) | 0.090 | 0.085 |
| Crystal size/mm | 0.25 × 0.20 × 0.15 | 0.25 × 0.25 × 0.15 |
| Temperature (K) | 295(2) | 295(2) |
| θrange, deg | 1.49–27.49 | 2.83–27.50 |
| Reflections collected | 18599 | 19743 |
| Independent reflections | 3161[R(int) = 0.0522] | 3879 [R(int) = 0.0412] |
| Refinement method | Full-matrix least-squares on F2 | Full-matrix least-squares on F2 |
| Final R indices [I > 2.00σ(I)] | R1 = 0.0595, wR2 = 0.1625 | R1 = 0.0828, wR2 = 0.2736 |
| R indices (all data) | R1 = 0.0947, wR2 = 0.1888 | R1 = 0.1071, wR2 = 0.2861 |
| GoF | 1.263 | 1.051 |
3. Experimental Section
3.1. General Information
| Compounds | 12a | 16a |
|---|---|---|
| Diffractometer | Nonius Kappa CCD | Nonius Kappa CCD |
| Formula | C22H19N5O2·C2H5OH | [Ni2(C22H16N5O2Cl)2]·H2O |
| Formula weight | 431.49 | 971.13 |
| Crystal system | Triclinic | Triclinic |
| Space group | P-1 | P-1 |
| a/Å | 10.3571(7) | 5.2360(4) |
| b/Å | 10.3772(8) | 16.6535(9) |
| c/Å | 10.7518(7) | 25.6203(16) |
| α/° | 100.618(6) | 75.822(5) |
| β/° | 96.497(5) | 88.784(6) |
| γ/° | 90.357(6) | 87.121(5) |
| V/Å3 | 1128.06(14) | 2163.2(2) |
| Z | 2 | 2 |
| Dcalc (g·cm−3) | 1.270 | 1.491 |
| F000 | 456 | 996 |
| Absorption coefficient (mm−1) | 0.700 | 1.052 |
| Crystal size/mm | 0.25 × 0.15 × 0.10 | 0.2 × 0.1 × 0.02 |
| Temperature (K) | 295(2) | 295(2) |
| θrange, deg | 4.30–67.93 | 3.03–27.50 |
| Reflections collected | 10692 | 17218 |
| Independent reflections | 4069 [R(int) = 0.0287] | 9910 [R(int) = 0.0584] |
| Refinement method | Full-matrix least-squares on F2 | Full-matrix least-squares on F2 |
| Final R indices [I > 2.00σ(I)] | R1 = 0.0626, wR2 = 0.1871 | R1 = 0.0898, wR2 = 0.2252 |
| R indices (all data) | R1 = 0.0778, wR2 = 0.2127 | R1 = 0.1419, wR2 = 0.2457 |
| GoF | 1.013 | 1.306 |
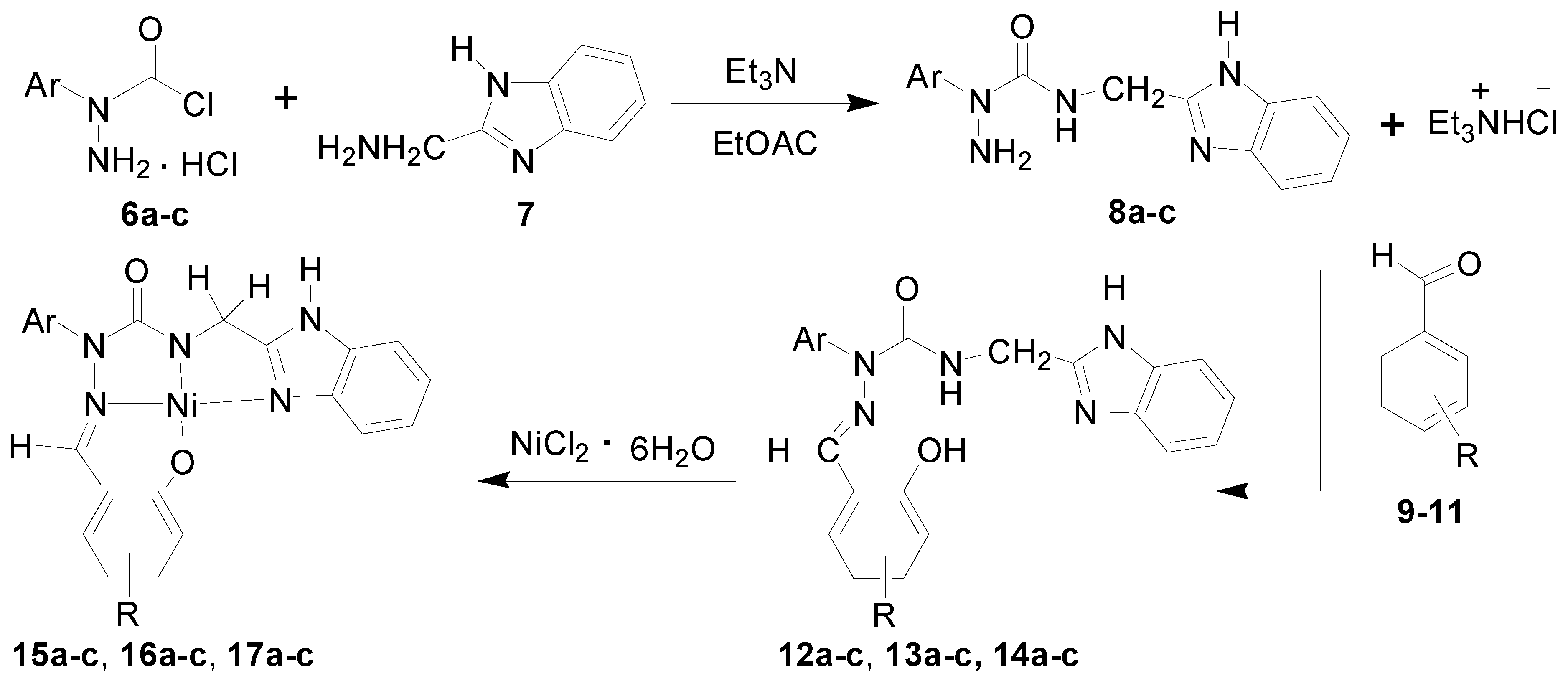
3.2. Syntheses of 2-aryl-4-[(1H-benzo[d]imidazol-2-yl)methyl]semicarbazides 8a–c
3.3. Syntheses of Substituted-Salicylaldehyde 2-Aryl-4-[(1H-benzo[d]imidazol-2-yl)methyl] semicarbazones 12a–14c
3.4. Syntheses of Ni Complexes of Substituted-salicylaldehyde 2-Aryl-4-[(1H-benzo[d]imidazol-2-yl)methyl] Semicarbazones 15a–17c
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ho, J.; Lee, W.Y.; Koh, K.J.T.; Lee, P.P.F.; Yan, Y.K. Rhenium(I) tricarbonyl complexes of salicylaldehyde semicarbazones: Synthesis, crystal structures and cytotoxicity. J. Inorg. Biochem. 2013, 19, 10–20. [Google Scholar] [CrossRef]
- Garbelini, E.R.; Martin, M.G.M.B.; Back, D.F.; Evans, D.J.; Müller-Santos, M.; Ribeiro, R.R.; Lang, E.S.; Nunes, F.S. Synthesis, characterization and chemical properties of 1-((E)-2-pyridinylmethylidene) semicarbazone manganese(II) and iron(II) complexes. J. Mol. Struct. 2012, 1008, 35–41. [Google Scholar] [CrossRef]
- Shih, M.H.; Chen, J.C.; Ling, G.L.; Lin, T.T.; Sun, M.H. Novel synthesis of palladium (II) complexes derived from 3-arylsydnone-4-carbaldehyde N(4)-phenylthiosemicarbazones and biological activity. J. Pharm. Pharmacol. 2014, 66, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Aziz, S.; Cockerill, A.F.; Tillett, J.G. Nucleophilic catalysis. Part III. Acid-catalysed hydrolyses of 3-phenylsydnone and 3-m-nitrophenylsydnone. J. Chem. Soc. (B) 1970, 416–420. [Google Scholar]
- Kenner, J.; Mackay, K. α-Acyl hydrazines. Nature 1947, 160, 465–466. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.N.; Wu, M.H.; Chen, S.P.; Li, T.P.; Hung, C.Y.; Yeh, M.Y. Syntheses of α-haloformylaryl-hydrazines and their self-dimerizations. J. Chin. Chem. Soc. 1994, 41, 849–856. [Google Scholar]
- Kuo, W.F.; Lee, C.Y.; Yeh, M.Y. Syntheses of 4-(5-oxo-1,2,4-triazol-3-yl)-sydnones and 4-(4-arylamino-5-oxo-1,2,4-triazol-3-yl)-sydnones from sydnone derivatives and their fragments. J. Chin. Chem. Soc. 2000, 47, 227–240. [Google Scholar]
- Shih, M.H. Studies on the syntheses of heterocycles from 3-arylsydnone-4-carbohydroximic acid chlorides with N-arylmaleimides, [1,4]naphthoquinone and aromatic amines. Tetrahedron 2002, 58, 10437–10445. [Google Scholar] [CrossRef]
- Shih, M.H.; Yeh, M.Y. Access to the syntheses of sydnonyl-substituted α,β-unsaturated ketones and 1,3-dihydro-indol-2-ones by modified Knoevenagel reaction. Tetrahedron 2003, 59, 4103–4111. [Google Scholar] [CrossRef]
- Shih, M.H. A concise synthetic method for sydnonyl-substituted pyrazoline derivatives. Synthesis 2004, 1, 26–32. [Google Scholar] [CrossRef]
- Shih, M.H.; Ke, F.Y. Syntheses and evaluation of antioxidant activity of sydnonyl substituted thiazolidinone and thiazoline derivatives. Bioorg. Med. Chem. 2004, 12, 4633–4643. [Google Scholar] [CrossRef] [PubMed]
- Shih, M.H.; Yeh, M.Y.; Lee, M.J.; Su, Y.S. Efficient syntheses of 3-(3-arylsydnon-4-yl)triazole derivatives. Synthesis 2004, 17, 2877–2885. [Google Scholar] [CrossRef]
- Shih, M.H.; Wu, C.L. Efficient syntheses of thiadiazoline and thiadiazole derivatives by the cyclization of 3-aryl-4-formylsydnone thiosemicarbazones with acetic anhydride and ferric chloride. Tetrahedron 2005, 61, 10917–10925. [Google Scholar] [CrossRef]
- Shih, M.H.; Tsai, C.H.; Wang, Y.C.; Shieh, M.Y.; Lin, G.L.; Wei, C.Y. Microwave-assisted synthesis of sydnonyl-substituted imidazoles. Tetrahedron 2007, 63, 2990–2999. [Google Scholar] [CrossRef]
- Shih, M.H.; Su, Y.S.; Wu, C.L. Syntheses of aromatic substituted hydrazino-thiazole derivatives to clarify structural characterization and antioxidant activity between 3-arylsydnonyl and aryl substituted hydrazino-thiazoles. Chem. Pharm. Bull. 2007, 58, 1126–1135. [Google Scholar] [CrossRef]
- Tonelli, M.; Simone, M.; Tasso, B.; Novelli, F.; Boido, V.; Sparatore, F.; Paglietti, G.; Pricl, S.; Giliberti, G.; Blois, S.; et al. Antiviral activity of benzimidazole derivatives. II. Antiviral activity of 2-phenylbenzimidazole derivatives. Bioorg. Med. Chem. 2010, 18, 2937–2953. [Google Scholar] [CrossRef] [PubMed]
- Dettmann, S.; Szymanowitz, K.; Wellner, A.; Schiedel, A.; Müller, C.E.; Gust, R. 2-Phenyl-1-[4-(2-piperidine-1-yl-ethoxy)benzyl]-1H-benzimidazoles as ligands for the estrogen receptor: Synthesis and pharmacological evaluation. Bioorg. Med. Chem. 2010, 18, 4905–4916. [Google Scholar] [CrossRef] [PubMed]
- Refaat, H.M. Synthesis and anticancer activity of some novel 2-substituted benzimidazole derivatives. Eur. J. Med. Chem. 2010, 45, 2949–2956. [Google Scholar] [CrossRef] [PubMed]
- Shaharyar, M.; Abdullah, M.M.; Bakht, M.A.; Majeed, J. Pyrazoline bearing benzimidazoles: Search for anticancer agent. Eur. J. Med. Chem. 2010, 45, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Hranjec, M.; Starčević, K.; Pavelić, S.K.; Lučin, P.; Pavelić, K.; Zamola, G.K. Synthesis, spectroscopic characterization and antiproliferative evaluation in vitro of novel Schiff bases related to benzimidazoles. Eur. J. Med. Chem. 2011, 46, 2274–2279. [Google Scholar] [CrossRef] [PubMed]
- Małecki, J.G.; Kruszynski, R.; Mazurak, Z. Synthesis, spectroscopic and structural characterizations of two new complexes of ruthenium with 2-(hydroxymethyl)benzimidazole and 1,10-phenanthroline ligands. Polyhedron 2009, 28, 3891–3898. [Google Scholar] [CrossRef]
- Lumb, I.; Hundal, M.S; Mathur, P.; Corbella, M.; Aliaga-Alcalde, N.; Hundal, G. First report on a dinuclear Cu(II) complex based on pyridine dicarboxamido ligand having benzimidazole moieties in the amide side arms: Synthesis, structure and magnetic properties of [Cu(GBPA)]2·4H2O, GBPA=N,N'-bis (2-methylbenzimidazolyl)-pyridine-1,3-dicarboxamide. Polyhedron 2012, 36, 85–91. [Google Scholar] [CrossRef]
- Abdel Ghani, N.T.; Mansour, A.M. Novel palladium(II) and platinum(II) complexes with 1H-benzimidazol-2-ylmethyl-N-(4-bromo-phenyl)-amine: Structural studies and anticancer activity. Eur. J. Med. Chem. 2012, 47, 399–411. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.; Kar, P.; Ianelli, S.; Ghosh, A. Isolation of a novel intermediate during unsymmetrical to symmetrical rearrangement of a tetradentate Schiff base ligand in a manganese(III) complex: Catalytic activity of the rearranged product towards alkene epoxidation. Inorg. Chim. Acta 2011, 365, 318–324. [Google Scholar] [CrossRef]
- Shit, S.; Rosair, G.; Mitra, S. A new tetranuclear copper(II) Schiff base complex containing Cu4O4 cubane core: Structural and spectral characterizations. J. Mol. Struct. 2011, 991, 79–83. [Google Scholar] [CrossRef]
- Hong, M.; Yin, H.D.; Chen, S.W.; Wang, D.Q. Synthesis and structural characterization of organotin(IV) compounds derived from the self-assembly of hydrazone Schiff base series and various alkyltin salts. J. Organomet. Chem. 2010, 695, 653–662. [Google Scholar] [CrossRef]
- Mathew, N.; Kurup, M.R.P. Synthesis and characterization of Mo(VI) complexes derived from ONO donor acylhydrazones. Spectrochim. Acta Part A-Mol. Biomol. Spectrosc. 2011, 78, 1424–1428. [Google Scholar] [CrossRef]
- Ahmadi, M.; Mague, J.T.; Akbari, A.; Takjoo, R. Dianion N1,N4-bis(salicylidene)-S-allyl-thiosemicarbazide complexes: Synthesis, structure, spectroscopy and thermal behavior. Polyhedron 2012, 42, 128–134. [Google Scholar] [CrossRef]
- Ahmad, J.U.; Räisänen, M.T.; Nieger, M.; Leskelä, M.; Repo, T. A facile synthesis of mixed ligand Cu(II) complexes with salicylaldehyde and salicylaldimine ligands and their X-ray structural characterization. Inorg. Chim. Acta 2012, 384, 275–280. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 13a–b, 15a–c, 16b, 17a, 17b are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shih, M.-H.; Xu, Y.-Y.; Yang, Y.-S.; Lin, T.-T. Syntheses of Nickel (II) Complexes from Novel Semicarbazone Ligands with Chloroformylarylhydrazine, Benzimidazole and Salicylaldehyde Moieties. Molecules 2015, 20, 5184-5201. https://doi.org/10.3390/molecules20035184
Shih M-H, Xu Y-Y, Yang Y-S, Lin T-T. Syntheses of Nickel (II) Complexes from Novel Semicarbazone Ligands with Chloroformylarylhydrazine, Benzimidazole and Salicylaldehyde Moieties. Molecules. 2015; 20(3):5184-5201. https://doi.org/10.3390/molecules20035184
Chicago/Turabian StyleShih, Mei-Hsiu, Yu-Yuan Xu, Yu-Sheng Yang, and Tzu-Ting Lin. 2015. "Syntheses of Nickel (II) Complexes from Novel Semicarbazone Ligands with Chloroformylarylhydrazine, Benzimidazole and Salicylaldehyde Moieties" Molecules 20, no. 3: 5184-5201. https://doi.org/10.3390/molecules20035184
APA StyleShih, M.-H., Xu, Y.-Y., Yang, Y.-S., & Lin, T.-T. (2015). Syntheses of Nickel (II) Complexes from Novel Semicarbazone Ligands with Chloroformylarylhydrazine, Benzimidazole and Salicylaldehyde Moieties. Molecules, 20(3), 5184-5201. https://doi.org/10.3390/molecules20035184