Abstract
Twenty-six 5-azacytidine analogues have been synthesized, including 4-amino-6-alkyl-1-pyranosyl/ribofuranosyl-1,3,5-triazin-2(1H)-ones 1a-j, 6-amino-4-alkyl/aryl-1-pyranosyl/ribofuranosyl-1,3,5-triazin-2(1H)-ones 2a-f and 4-amino-6-alkyl-1,3,5-triazin-2-yl-1-thio-pyranosides/ribofuranosides 3a-j. The antiproliferative activities of these synthetic analogues were investigated in human leukemia HL-60 cells. Ribofuranosyl S-nucleoside 3a, a bioisostere of 5-azacytidine, had a similar antiproliferative ability as that of the latter. Introduction of a methyl at the 6 position of 5-azacytidine and/or replacement of the ribofuranosyl moiety with pyranosyl sugars or disaccharides significantly decreased the antiproliferative activities of the 5-azacytidine derivatives. Several compounds with the replacement of pyranosyl sugars enhanced all-trans retinoic acid-induced differentiation ability in human leukemia HL-60 cells.
Introduction
5-Azacytidine (5-aza-CR) and 5-aza-2’-deoxycytidine (5-aza-CdR, Decitabine) (Figure 1) are known DNA methyltransferase inhibitors and have been approved for the treatment of myelodysplastic syndrome (MDS) and chronic myelomonocytic leukemia (CMML) [1]. Although both agents are cytotoxic at high concentrations, the therapeutic effects of both compounds in MDS have been thought to be mediated through inhibition of the DNA methyltransferase at low concentrations [1]. Aberrant DNA methylation in the promoter region of genes can silence their expression [2]. Some tumor suppressor genes have been found to be silenced due to DNA hypermethylation and these genes can be reactivated by DNA demethylation [3]. Treatments of malignant cells with 5-aza-CR or 5-aza-CdR have been found to be associated with reversal of specific gene suppression [1,4]. Zebularine (Figure 1), a derivative of 5-aza-CR with the increased stability, has been reported to have little cytoxicity but to have maintained the ability of inhibiting DNA methytransferase activity [5,6]. Although 5-fluoro-deoxycytidine (FDAC) has both cytotoxic effects to malignant cells and inhibitory effects on DNA methyltransferase activity, 5-fluorouracil (5-FU) does not have the inhibitory effects on DNA methyltransferase activity [4]. These observations suggest that the riboside moiety of these nucleoside inhibitors is required for the inhibition of DNA methyltransferase activity [7]. Since the cytotoxic effects of these compounds are due to their incorporation into DNA or RNA [8,9], it seems that replacement of the riboside with other types of sugars will decrease the cytotoxic effects and will keep the abilities of DNA methyltransferase inhibition.
We have synthesized a series of 5-aza-CR derivatives with introduction of a methyl or an ethyl group at the 6 position of 5-aza-CR, and/or with a replacement of O by S, or with a replacement of ribofuranosyl moiety by pyranosyl sugars or disaccharides. Addition of a methyl or an ethyl group at the 6 position of 5-aza-CR could block the attack of water and will improve the chemical stability of 5-aza-CR. The replacement of ribofuranosyl moiety with pyranosyl sugars or disaccharides is estimated to reduce cytotoxicity of 5-aza-CR. The antiproliferative activities as well as the differentiation induction of these derivatives alone and in combination with all-trans retinoic acid (ATRA) were investigated in human leukemia HL-60 cells.
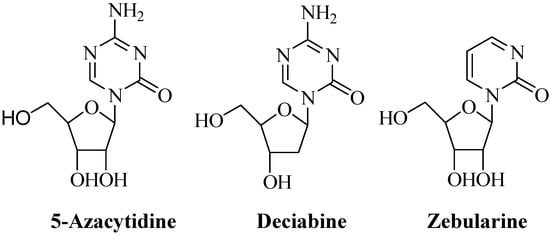
Figure 1.
Structures of 5-Azacytidine, Decitabine and Zebularine
Figure 1.
Structures of 5-Azacytidine, Decitabine and Zebularine
Results and Discussion
Chemistry
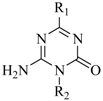
As illustrated in Scheme 1, the target compounds 1-3 were synthesized from guanylurea or guanylthiourea through the intermediate compounds 4-8. 4-Amino-6-alkyl/aryl-1,3,5-triazin-2(1H)-ones 4a-f, obtained in moderate yields from the reactions of guanylurea or guanylthiourea with orthoesters, reacted with hexamethyldisilazane (HMDS) to produce N,O-bistrimethylsilylated 1,3,5-triazinones 5a-f. All reactions were carried out according to the procedures described in the literature [10,11,12,13,14]. Vorbrüggen coupling of 5a-f and acylated sugars in anhydrous acetonitrile in the presence of a Lewis acid catalyst gave the acylated nucleosides in moderate yields. Purification of raw products by flash column chromatography gave acylated N1-nucleosides 6, acylated N3-nucleosides 7 or acylated S-nucleosides 8 as main products. 4-Amino-6-alkyl-1-pyranosyl/ribofuranosyl-1,3,5-triazin-2(1H)-ones 1a-j, 6-amino-4-alkyl/aryl-1-pyranosyl/ribofuranosyl-1,3,5-triazin-2(1H)-ones 2a-f and 4-amino-6-alkyl-1,3,5-triazin-2-yl-1-thio-pyranosides/ribofuranosides 3a-j were obtained after deprotection of acylated nucleosides 6-8 in a saturated methanol solution of ammonia at room temperature. The structures of target compounds are shown in Table 1.
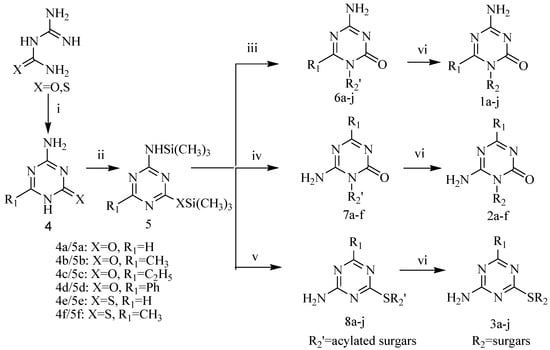
Scheme 1.
Synthetic route to the target compounds.
Scheme 1.
Synthetic route to the target compounds.
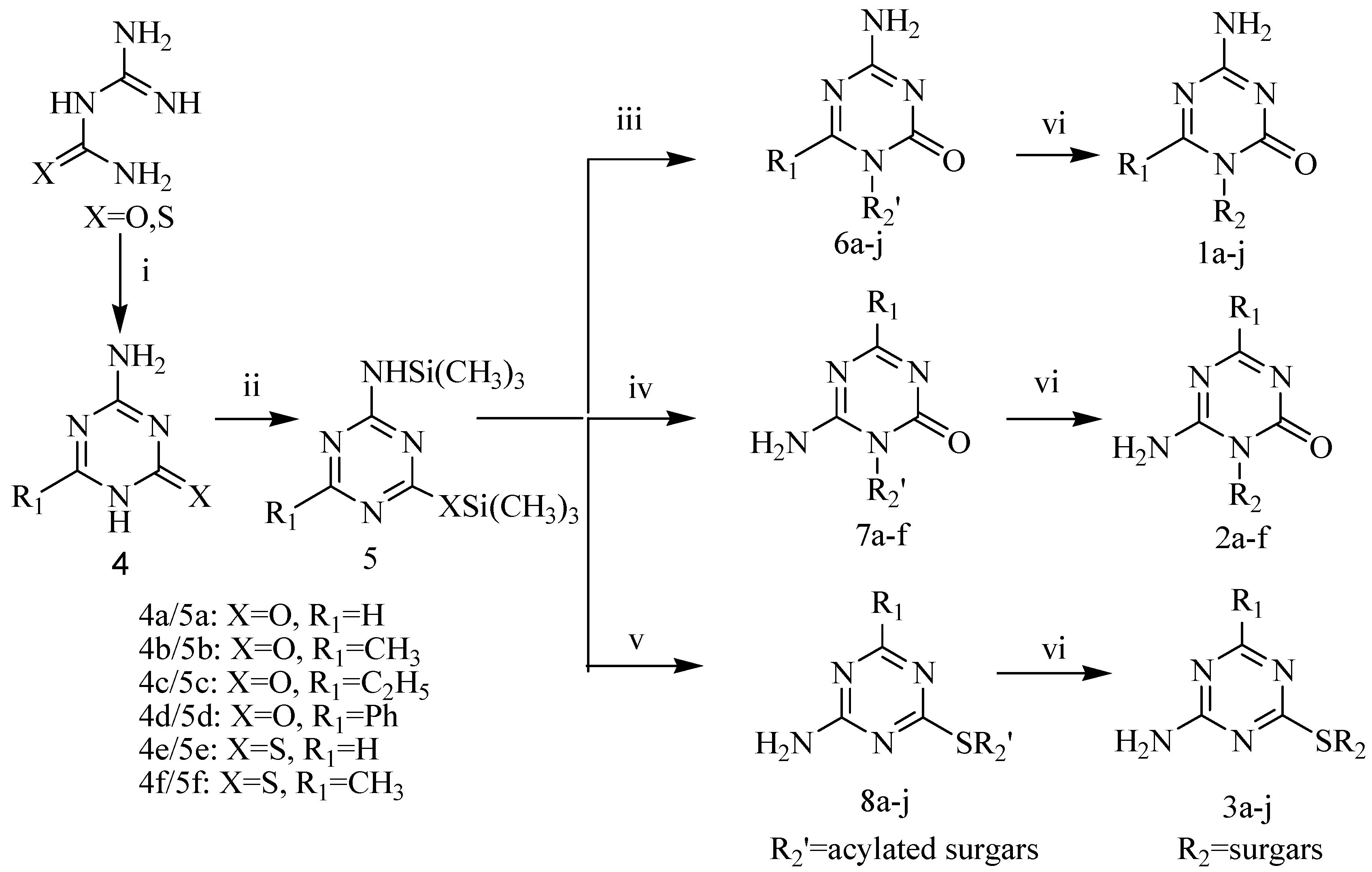
Reagents and conditions: (i)orthoesters/DMF, 130 oC, 80%-85%; (ii) HMDS/(NH4)2SO4, 92%-95%; (iii) acylated sugars/SnCl4 orTMSOTf/CH3CN, 40%-45%; (iv) Acylated ribose(pyranose)/SnCl4/ CH3CN, 24%-32%; (v) acylated ribose(pyranose)/TMSOTf/CH3CN, 30%-37%; vi: NH3/CH3OH, 50%-65%.
Vorbrüggen coupling of 5a with acylated sugars and of 5b with 1-O-acetyl-2,3,5-tri-O-benzoyl-β-d-ribose in anhydrous acetonitrile with SnCl4 or trimethylsilyltriflate (TMSOTf) [15] catalysis gave acylated nucleosides 6a-g. However, when the coupling procedure was applied to 5b and acetylated pyranoses in the presence of SnCl4 (2 equiv.), the acylated N3-isomers 7a-d were obtained as the main products. The ratios of N3/N1 glucopyranoside, xylopyranoside, manopyranoside and maltopyranoside were 6.5:1, 5.5:1, 3.5:1, 8.2:1, respectively. It was observed that the N1-isomers 6h-j were the predominant products when a weaker Lewis acid (TMSOTf, 2 equiv.) was used as the catalyst. The N1/N3 ratios of glucopyranoside, xylopyranoside and manopyranoside were 4:1, 5.2:1, 3.8:1, respectively. N3-Nucleosides 7e-f were formed during couplings of 5c and 5d with 1-O-acetyl-2,3,5-tri-O-benzoyl-β-d-ribose under the same reaction conditions. The regioselectivity of nucleoside synthesis depended both on the substrates and catalysts. Basicity and steric hindrance of N1 in the N,O-bistrimethylsilylated 1,3,5-triazinones 5a-f increased when a methyl, an ethyl or a phenyl was introduced to the 6 position. When the substitute was a methyl, a small electrophilic group, the basicity of N1 was the major factor affecting N1/N3 regioselectivity. Since TMSOTf is a weaker Lewis acid than SnCl4, less σ-complex was formed between TMSOTf and the N1 of 5b and therefore more free N1 was present to form acylated N1-nucleosides 6h-j [16]. When an ethyl or a phenyl was introduced to the 6 position the steric hindrance turned to be a major factor determining the regioselectivity. The N3-nucleosides 7e-f were predominant products, even when TMSOTf was used as a catalyst. The acylated S-nucleosides 8a-j, which are kinetically controlled products, were the predominant products during couplings of 5e and 5f with acylated sugars in anhydrous acetonitrile under SnCl4 or TMSOTf catalysis. Thus was presumably due to the stronger nucleophilicity of a sulphur compared to an oxygen.
The structures of all compounds were determined by application of IR, 1H-NMR and MS spectral data. The structure assignments for compound 1, 2 and 3 were supported by comparing the 1H-NMR and HMBC spectral data with each other. The H-1’ peak was found to be correlated with C2 (C=O) and C6 (C-R1) in HMBC spectrum in N1-nucleosides 1. There was a single peak for the two hydrogen signal of the amino group in 1H-NMR. However, the H-1’ peak was correlated with C2 (C=O) and C6 (C-NH2) in HMBC spectrum in N3-nucleosides 2. There were two single peaks at about 8.31-8.46 and 6.94-7.87 ppm in 1H-NMR spectra corresponding to the two hydrogen signals of the amino group. This non-equivalence of the amino protons was caused by the pyranosyl sugar, which enhances the rotational barrier of the amino group. The S-nucleosides 3 were identified by HMBC spectra analysis. The H-1’ peak was found to be correlated with C2 (C-S) in HMBC spectrum. The H-1’ peak of N3-nucleosides was observed at about 5.85 ppm and these values slightly shifted to low field comparing to that of the N1-nucleosides and S-nucleosides (5.35 ppm). The configuration of the nucleosides was deduced from the trans-diaxial coupling between H-1’ and H-2’ of pyranose in the 1H-NMR spectra. 1c, 1d, 1j, 2c, 3d, and 3i were α-manopyranosyl or α-rhamnopyranosyl and β-configuration nucleosides.
Bioactivity
The antiproliferative activities of these synthetic compounds were determined in human leukemia HL-60 cells. 5-aza-CR is a potent growth inhibitor with a GI50 value of 0.29 μM. By using a trypan blue exclusion assay, we found that 5-aza-CR killed half of cells at a concentration of 1.0 μM. Among all the listed compounds, only compound 3a has a lower GI50 value of 1.7 μM. Compound 1g has a GI50 value of 18.5 μM. The GI50 values of other compounds can not be obtained since these compounds do not inhibit 50% cell growth at concentrations less than 50 μM. Compound 3a is the bioisostere of 5-aza-CR, with a ribofuranosyl S-nucleoside. Compound 1g only has an introduction of a methyl group at the 6 position of 5-aza-CR without replacement of the ribofuranosyl group. Comparing the structures of 1a-f with 5-aza-CR it was found that replacement of the β-d-ribofuranosyl with a β-d-glucopyranosyl, a β-d-xylopyranosyl, a α-d-mannopyranosyl, a α-l-rhamnopyranosyl, a β-d-maltopyranosyl or a β-d-lactopyranosyl significantly decreased the antiproliferative activities. Comparison of the antiproliferative activities of 5-aza-CR with compound 1g revealed that replacement of the H with a CH3 (1g) slightly decreased the antiproliferative activities. However, replacement of the H (3a) with a CH3 (3f), where both are ribofuranosyl S-nucleosides, evidently decreased the antiproliferative activity. The antiproliferative activity of 5-aza-CR is due to its incorporation into DNA and/or RNA [8], it seems that replacement of the β-d-ribofuranosyl in these nucleosides with other sugar moieties would not incorporate into DNA or RNA, that may explain their non-toxic effect to HL-60 cells. It has been shown that ATRA induced differentiation of human leukemia cells with induction of RARβ2 that has been found to be hypermethylated [17]. We have investigated the differentiation activities of these compounds alone and in combination with ATRA in HL-60 cells using NBT reduction assay as a differentiation marker. ATRA treatment at 0.1 μM induced 25.9% of HL-60 cells undergoing differentiation after 5 days. All of these compounds alone did not induce NBT reduction but some compounds such as 1c, 1d and 3i enhanced ATRA-induced differentiation. The enhancement on ATRA differentiation induction by these compounds may be due to the enhanced RARβ2 expression through inhibition of DNA methyltransferase that needs to be further investigated. Since non-nucleoside compounds such as (-)-epigallocatechin-3-gallate, hydralazine and procainamide inhibit DNA methyltransferase activity [18,19], we prospect that some of these synthetic compounds will keep the ability of inhibiting DNA methyltransferase activity without incorporating into DNA. The inhibitory abilities of these compounds on DNA methyltransferase activity are under investigation.

Table 1.
Chemical structures, antiproliferative and differentiation induction abilities of target compounds in human leukemia HL-60 cells.
| Compound | Structure | Growth Inhibitiona (%) | Differentiationb Induction (%) | |
|---|---|---|---|---|
| R1 | R2 | |||
 | ||||
| 1a | H | β-D-glucopyranosyl | 14.4±8.4 | 21.2±1.2 |
| 1b* | H | β-D-xylopyranosyl | 24.2±6.7 | 22.7±1.9 |
| 1c* | H | α-D-mannopyranosyl | 24.0±5.2 | 34.8±3.7# |
| 1d* | H | α-L-rhamnopyranosyl | 24.5±12.0 | 34.4±1.7## |
| 1e* | H | β-D-maltopyranosyl | 36.1±7.3 | 25.0±1.2 |
| 1f* | H | β-D-lactopyranosyl | 25.4±6.1 | 23.9±2.2 |
| 1g | CH3 | β-D-ribofuranosyl | (18.5±3.9μM) | 22.8±1.4(10μM) |
| 1h* | CH3 | β-D-glucopyranosyl | 28.3±5.2 | 22.5±1.6 |
| 1i* | CH3 | β-D-xylopyranosyl | 40.7±9.9 | 21.3±1.1 |
| 1j* | CH3 | α-D-mannopyranosyl | ND | ND |
 | ||||
| 2a* | CH3 | β-D-glucopyranosyl | 23.7±6.0 | 26.6±1.6 |
| 2b* | CH3 | β-D-xylopyranosyl | 22.0±5.3 | 22.6±2.1 |
| 2c* | CH3 | α-D-mannopyranosyl | 5.7±4.3 | 22.8±2.4 |
| 2d* | CH3 | β-D-maltopyranosyl | 25.9±8.8 | 20.8±1.5 |
| 2e* | C2H5 | β-D-ribofuranosyl | 3.3±1.8 | 24.8±2.4 |
| 2f | C6H5 | β-D-ribofuranosyl | 3.8±3.3 | 20.3±2.9 |
 | ||||
| 3a* | H | β-D-ribofuranosyl | (1.7±0.1μM) | 19.8±2.2(0.85μM) |
| 3b* | H | β-D-glucopyranosyl | 12.9±3.4 | 24.0±1.7 |
| 3c* | H | β-D-xylopyranosyl | 32.1±9.3 | 21.3±1.5 |
| 3d* | H | α-D-mannopyranosyl | 26.5±8.9 | 19.4±1.6 |
| 3e* | H | β-D-maltopyranosyl | 2.1±2.1 | 23.5±1.9 |
| 3f* | CH3 | β-D-ribofuranosyl | 15.4±4.4 | 23.2±2.4 |
| 3g* | CH3 | β-D-glucopyranosyl | 2.6±2.6 | 20.6±1.9 |
| 3h* | CH3 | β-D-xylopyranosyl | 15.4±3.2 | 24.2±2.3 |
| 3i* | CH3 | α-D-mannopyranosyl | 36.0±11.9 | 31.4±2.3# |
| 3j* | CH3 | β-D-maltopyranosyl | 20.0±5.1 | 21.7±1.3 |
| 5-azaCR | (0.29±0.015μM) | 24.0±1.3(0.15μM) | ||
| ATRA | 25.9±1.4 | |||
Notes: asterisked compounds are novel compounds. ND, not determined.a. Data shown are growth inhibition rates in HL-60 cells after treatment with the listed compounds at 50 μM for 72 h. The GI50 values of 1g, 3a and 5-aza-CR are listed in parenthesis. Data shown are Mean±SD of three independent experiments. b. Data shown are percentage of NBT positive cells in HL-60 cells after treatment with the listed compounds at 50 μM or with 1g, 3a or 5-aza-CR at the concentrations listed in parenthesis in combination with ATRA at 0.1μM for 120 h. Data shown are Mean ± SD of three independent experiments. # P<0.05; ## P<0.01 compared to cells treated with ATRA alone.
In summary, our data indicate that 1) changing 5-aza-CR into a bioisosteric ribofuranosyl S-nucleoside (3a) does not influence the antiproliferative ability; 2) replacement of the ribofuranosyl moiety with pyranosyl sugars or disaccharides significantly decreases the antiproliferative efficacies; 3) compounds with the ribofuranosyl moiety replaced by the pyranosyl sugars (1c, 1d and 3i) enhance ATRA differentiation induction ability.
Experimental
General
The melting points were determined on an electrically heated X4 digital visual melting point apparatus and were uncorrected.. IR spectra were recorded on Bruker IFS 55 (KBr). 1H-NMR spectra were recorded on a Bruker ARX-300 spectrometer at 300MHz with DMSO-d6 as solvent and TMS as an internal standard. Mass spectra were taken in ESI mode on Agilent 1100 LC-MS. Elemental analysis was determined on a Carlo-Erba 1106 Elemental analysis instrument. All solvents and reagents were commercially available.
General procedure for preparation target nucleosides
A mixture of guanylurea or guanylthiourea, orthoesters and dimethylformamide was refluxed for 90 min and allowed to stand at room temperature, the precipitate were recrystallized from the solution, then filtered off to give 4-amino-6-alkyl/aryl-1,3,5-triazin-2(1H)-ones 4a-f. The structures were identified by MS [10,11,12]. Then a mixture of 4, HMDS and (NH4)2SO4 or pyrimidine as catalyst was refluxed for 12-16 h. After evaporation under vacuum, the silylated bases 5a-f were obtained [13,14]. The crude product was used in glycosylation without further purification. Silylated base (1 equiv) was dissolved in anhydrous acetonitrile, to this solution were added 1-O-acetyl-2,3,5-tri-O-benzoyl-β-d-ribose (1 equiv.) or acetylated pyranosyl sugar (1 equiv.) and SnCl4 (2 equiv.) or TMSOTf (2 equiv.) [15] in acetonitrile. The mixture was left standing at room temperature under stirring for 10-18 h, then diluted in chloroform and neutralized with a saturated solution of sodium bicarbonate. The organic layer was separated, dried and evaporated. The residue was purified by flash column chromatography (CHCl3-CH3OH). Acetylated nucleosides 6-8 were obtained as white solids which were identified by 1H-NMR and MS spectra. Then the acetylated nucleosides and saturated NH3-CH3OH solution were stirred at room temperature for 5 h and evaporated to dryness. The residue was purified by flash column chromatography (CHCl3-CH3OH), and recrystallized from water/methanol to give target nucleosides 1-3 as whte crystals.
4-Amino-1-β-d-glucopyranosyl-1,3,5-triazin-2(1H)-one (1a). Yield: 21.6%; mp: 258-260°C (dec.) [20] (water/methanol); IR ν/cm-1: 3350.5, 1645.0, 1383.6, 1074.1, 902.4, 849.9, 796.5; 1H-NMR δ/ppm:3.18-3.19 (2H, m, H-6’, H-6”), 3.21-3.27 (2H, m, H-4’, H-5’), 3.56-3.57 (1H, m, H-3’),3.67-3.71 (1H, m, H-2’), 4.58 (1H, s, OH), 5.12 (1H, s, OH), 5.26 (1H, s, OH), 5.34 (1H, d, J = 9.4 Hz, H-1’), 5.38 (1H, s, OH), 7.59 (2H, s, NH2), 8.37 (1H, s, H-6). ESI-MS: 275.4[M+H]+; Anal. Calcd. for C9H14N4O6: C 39.42, H 5.15, N 20.43. Found: C 39.38, H 5.12, N 20.45.
4-Amino-1-β-d-xylopyranosyl-1,3,5-triazin-2(1H)-one (1b). Yield: 21.6%; mp: 191-193°C (water/ methanol); IR ν/cm-1: 3352.8, 1640.8, 1481.6, 1383.0, 1173.7, 1094.2, 1056.2, 897.9, 864.2, 797.8; 1H-NMR δ/ppm: 3.17-3.21 (3H, m, H-4’, H-5’, H-5”), 3.62-3.63 (1H, m, H-3’), 3.78-3.79 (1H, m, H-2’), 5.13 (1H, m, OH), 5.25-5.29 (2H, m, OH), 5.39 (1H, d, J = 4.2 Hz, H-1’), 7.61 (2H, s, NH2), 8.38 (1H, s, H-6). ESI-MS: 245.1 [M+H]+; Anal. Calcd. for C8H12N4O5: C 39.35, H 4.95, N 22.94. Found: C 39.42, H 4.90, N 22.98.
4-Amino-1-α-d-mannopyranosyl-1,3,5-triazin-2(1H)-one (1c). Yield: 16.8%; mp: 158-160°C (water/ methanol); IR ν/cm-1: 3396.0, 1695.1, 1383.4, 1098.7, 1051.4, 979.9; 1H-NMR δ/ppm: 3.60-3.64 (3H, m, H-4’, H-6’, H-6”), 3.79 (1H, s, H-5’), 3.87 (1H, s, H-3’), 4.11(1H, s, H-2’), 4.56 (1H, s, OH), 5.12-5.24 (3H, m, OH), 5.74-5.77 (1H, d, J = 9.0 Hz, H-1’), 7.55 (2H, s, NH2), 8.34 (1H, s, H-6); ESI-MS: 273.3 [M-H]+; Anal. Calcd. for C9H14N4O6: C 39.42, H 5.15, N 20.43. Found: C 39.44, H 5.08, N 20.39.
4-Amino-1-(6'-deoxy-α-l-mannopyranosyl)-1,3,5-triazin-2(1H)-one (1d). Yield: 12.4 %; mp: 126-128°C (water/methanol); IR ν/cm-1: 3391.4, 2922.3, 1639.8, 1511.5, 1383.5, 1054.1, 932.3, 798.4; 1H-NMR δ/ppm: 1.32-1.37 (3H, d, J = 6.9 Hz, CH3), 3.87 (2H, s, H-4’, H-3’), 3.95-3.98 (1H, d, J = 6.9 Hz, H-5’), 4.07 (1H, s, H-2’), 5.10-5.12 (1H, d, OH), 5.19 (2H, s, OH), 5.80-5.83 (1H, d, J = 9.6 Hz, H-1’), 7.50-7.53 (2H, d, NH2), 8.32 (1H, s, H-6); ESI-MS: 259.2 [M+H]+; Anal. Calcd. For C9H14N4O5: C 41.86, H 5.46, N 21.70. Found: C 41.92, H 5.49, N 21.76.
4-Amino-1-(4'-O-α-d-glucopyranosyl-β-d-glucopyranosyl)-1,3,5-triazin-2(1H)-one (1e). Yield: 19.6%; mp: 127-129 °C (water/methanol); IR ν/cm-1: 3369.2, 1697.4, 1382.4, 1169.6, 1088.4, 1038.6, 893.1, 780.2; 1H-NMR δ/ppm: 3.08-3.09 (1H, m), 3.25-3.26 (1H, m), 3.39-3.51 (5H, m), 3.54-3.59 (2H, m), 3.61-3.67 (2H, m), 3.70-3.72 (1H, m), 4.53-4.56 (2H, m, OH), 4.94 (2H, s, OH), 5.03-5.04 (1H, d, J = 3.4 Hz, H-1”), 5.39-5.41 (1H, d, J = 9.4 Hz, H-1’), 5.43-5.44 (1H, d, OH), 5.52 (1H, s, OH), 5.71 (1H, s, OH), 7.58-7.60 (2H, d, NH2), 8.39 (1H, s, H-6); ESI-MS: 437.4 [M+H]+; Anal. Calcd. for C15H24N4O11: C 41.29, H 5.54, N 12.84. Found C 41.36, H 5.58, N 12.80.
4-Amino-1-(4'-O-β-d-galactopyranosyl-β-d-glucopyranosyl)-1,3,5-triazin-2(1H)-one (1f). Yield: 20.8%; mp: 181-182 °C (water/methanol); IR ν/cm-1: 3385.3, 1689.5, 1085.2, 896.3, 782.6; 1H-NMR δ/ppm: 3.35-3.60 (7H, m), 3.63-3.72 (3H, m), 3.75-3.76 (2H, m), 4.22 (1H, s, OH), 4.56-4.60 (2H, d, OH), 4.69 (1H, s, OH), 4.82 (2H, s, H-1”, OH), 5.15(1H, s, OH), 5.41-5.44 (2H, d, J = 9.2 Hz, H-1’), 5.46 (1H, s, OH), 7.59 (2H, s, NH2), 8.38(1H, s, H-6); ESI-MS: 437.3 [M+H]+; Anal. Calcd. For C15H24N4O11: C 41.29, H 5.54, N 12.84. Found: C 41.32, H 5.62, N 12.86.
4-Amino-6-methyl-1-β-d-ribofuranosyl-1,3,5-triazin-2(1H)-one (1g). Yield: 23.5%; mp: 142-145 °C [13] (water/methanol); IR ν/cm-1: 3359.6, 1650.8, 1594.7, 1105.4, 895.1, 797.8; 1H-NMR δ/ppm: 2.43 (3H, s, CH3), 3.45-3.47 (2H, m, H-5’, H-5”), 3.59-3.63 (1H, m, H-4’), 3.75-3.76 (1H, d, H-3’), 4.56 (1H, s, H-2’), 4.75 (1H, s, OH), 4.97 (1H, s, OH), 5.20 (1H, s, OH), 5.59-5.61 (1H, d, H-1’), 7.46-7.47 (2H, s, NH2); ESI-MS: 259.2 [M+H]+; Anal. Calcd. for C9H14N4O5: C 41.86, H 5.46, N 21.70. Found C 41.89, H 5.43, N 21.75.
4-Amino-β-d-glucopyranosyl-6-methyl-1,3,5-triazin-2(1H)-one (1h). Yield: 16.8%; mp: 168-169 °C (water/methanol); IR ν/cm-1: 3373.2, 2926.8, 1636.9, 1526.2, 1383.8, 1190.4, 1077.8, 893.3, 797.7; 1H-NMR δ/ppm: 2.44( 3H, m, CH3), 3.22-3.25 (2H, m, H-6’, H-6”), 3.45-3.47 (2H, m, H-4’, H-5’), 3.67-3.71 (2H, d, H-3’), 4.21-4.52 (5H, br, OH, H-2’), 5.26-5.27 (1H, d, J = 5.2 Hz, H-1’); 7.32 (2H, s, NH2); ESI-MS: 287.4 [M-H]-; Anal. Calcd. for C10H16N4O6: C 41.67, H 5.59, N 19.44. Found C 41.56, H 5.64, N 19.49.
4-Amino-6-methyl-1-β-d-xylopyranosyl-1,3,5-triazin-2(1H)-one (1i). Yyield 19.7%; mp: 156-158 °C (water/methanol); IR ν/cm-1: 3406.9, 2923.4, 1669.1, 1301.2, 1048.0, 978.0, 943.5, 896.2; 1H-NMR δ/ppm: 2.41 (3H, s, CH3), 3.16-3.20 (4H, m, H-3’, H-4’, H-5’, H-5”), 3.81-3.82 (1H, m,H-2’), 5.08-5.13 (4H, m, br, OH), 5.35-5.36 (1H, d, J = 8.5 Hz, H-1’), 7.30 (2H, s, NH2); ESI-MS: 257.3 [M-H]+; Anal. Calcd. for C9H14N4O5: C 41.86, H 5.46, N 21.70. Found C 41.80, H5.38, N 21.72.
4-Amino-1-α-d-mannopyranosyl-6-methyl-1,3,5-triazin-2(1H)-one (1j). Yield: 14.7 %; mp: 173-175 °C (water/methanol); IR ν/cm-1: 3383.8, 2926.3, 1633.7, 1529.3, 1384.2, 1113.7, 1055.0, 922.2, 865.0, 797.9; 1H-NMR δ/ppm: 2.47 (3H, s, CH3), 3.56 (1H, m, H-6”), 3.63-3.70 (2H, m, H-4’, H-5’), 3.84-3.85 (1H, m, H-3’), 3.91-3.94 (1H, m, H-6’), 4.52-4.56 (2H, m, OH, H-2’), 4.95-4.97 (1H, d, OH), 5.04-5.08 (2H, OH), 5.93-5.96 (1H, d, J = 8.9 Hz, H-1’), 7.25 (2H, s, NH2); ESI-MS: 287.2 [M-H]-; Anal. Calcd. for C10H16N4O6: C 41.67, H 5.59, N 19.44. Found C 41.58, H 5.54, N 19.35.
6-Amino-1-β-d-glucopyranosyl-4-methyl-1,3,5-triazin-2(1H)-one (2a). Yield: 10.6%; mp: 181-182 °C (water/methanol); IR ν/cm-1: 3394.1, 1688.5, 1564.4, 1069.4, 899.9, 795.4; 1H-NMR δ/ppm: 2.06 (3H,s,CH3), 3.23-3.36 (2H, m, H-6’, H-6”), 3.49-3.62 (3H, m, H-3’, H-4’, H-5’), 3.73 (1H, s, H-2’), 4.73-4.77 (1H, t, OH), 5.16-5.18 (1H, d, OH), 5.25-5.26 (1H, d, OH), 5.40-5.42 (1H, d, OH), 5.80 (1H, s, H-1’), 6.94 (1H, s, NH), 8.35 (1H, s, NH); ESI-MS: 287.3 [M-H]+; Anal. Calcd. for C10H16N4O6: C 41.67, H 5.59, N 19.44. Found C 41.58, H 5.57, N 19.38.
6-Amino-4-methyl-1-β-d-xylopyranosyl-1,3,5-triazin-2(1H)-one (2b). Yield: 12.1%; mp: 170-172 °C (water/methanol); IR ν/cm-1: 3378.7, 1692.2, 1565.2, 1384.1, 1091.9, 1038.3, 895.4, 846.6, 797.5; 1H-NMR δ/ppm: 2.08 (3H, s, CH3), 3.18 (2H, s, H-5’, H-5”), 3.66-3.83 (3H, m, H-2’, H-3’, H-4’), 5.04-5.06 (1H, d, OH), 5.19(1H, s, OH), 5.36-5.38 (1H, d, OH), 5.75(1H, s, br, H-1’), 7.04 (1H, s, br, NH), 8.31 (1H, s, br, NH); ESI-MS: 258.7 [M+H]+; Anal. Calcd. for C9H14N4O5: C 41.86, H 5.46, N 21.70. Found C 41.80, H 5.40, N 21.63.
6-Amino-1-α-d-mannopyranosyl-4-methyl-1,3,5-triazin-2(1H)-one (2c). Yield: 8.9%; mp: 162-164 °C (water/methanol); IR ν/cm-1: 3400.5, 1687.0, 1558.7, 1481.7, 1384.6, 1050.0, 1012.3, 921.4, 866.8, 796.4; 1H-NMR δ/ppm: 2.09 (3H, s, CH3), 3.54-3.57 (1H, m, H-6”), 3.73 (1H, s, H-3’), 3.89 (3H, s, H-4’, H-5’, H-6’), 4.28-4.30 (1H, m, H-2’), 4.73 (1H, s, OH), 5.05-5.07 (1H, d, OH), 5.21-5.22 (1H, d, OH), 5.51 (1H, s, OH), 6.27-6.30 (1H, d, J = 9.7 Hz, H-1’), 6.98 (1H, s, br, NH), 8.46 (1H, s, br, NH); ESI-MS: 287 [M-H]+; Anal. Calcd. for C10H16N4O6: C 41.67, H 5.59, N 19.44. Found C 41.56, H 5.64, N 19.49.
6-Amino-1-(4'-O-α-d-glucopyranosyl-β-d-glucopyranosyl)-4-methyl-1,3,5-triazin-2(1H)-one (2d). Yield: 14.6%; mp: 175-177 °C (water/methanol); IR ν/cm-1: 3418.6, 2923.2, 1692.2, 1379.9, 1059.2, 918.6, 893.2, 869.0, 840.9, 798.3; 1H-NMR δ/ppm: 2.11 (3H, s, CH3), 3.10-3.12 (1H, m), 3.28 (2H, s), 3.60-3.83 (8H, m), 4.47 (1H, s, OH), 4.56 (1H, s, OH), 4.78 (1H, s, OH), 4.97 (2H, s, OH, H-2’), 5.02-5.03 (1H, s, H-1”), 5.54 (1H, s, OH), 5.58-5.60 (1H, d, OH), 5.74 (1H, s, OH), 5.89 (1H, s, H-1’), 7.02 (1H, s, br, NH), 8.40 (1H, s, br, NH); ESI-MS: 449.4 [M-H]+; Anal. Calcd. for C16H26N4O11: C 42.67, H 5.82, N 12.44. Found C 42.55, H 5.93, N 12.49.
6-Amino-4-ethyl-1-β-d-ribofuranosyl-1,3,5-triazin-2(1H)-one (2e). Yield: 10.3%; mp: 180-182 °C (water/methanol); IR ν/cm-1: 3314.9, 1682.0, 1655.5, 1552.7, 1115.2, 1085.0, 900.3, 876.1, 854.0, 806.1; 1H-NMR δ/ppm: 1.11-1.16 (3H, t, J = 7.5 Hz, CH3), 2.32-2.40 (2H, q, J = 7.5 Hz, CH2), 3.57-3.67 (2H, m, H-5’, H-5”), 3.94 (1H, s, H-3’), 4.03 (1H, s, H-2’), 4.34-4.41 (1H, m, H-4’), 5.08-5.09 (1H, d, OH), 5.24-5.26 (1H, d, OH), 5.72 (1H, s, H-1’), 6.31-6.34 (1H, d, OH), 7.87(1H, s, br, NH), 8.38 (1H, s, br, NH); ESI-MS: 273.2 [M+H]+; Anal. Calcd. for C10H16N4O5: C 44.12, H 5.92, N 20.58. Found C 44.32, H 5.96, N 20.54.
6-Amino-4-phenyl-1-β-d-ribofuranosyl-1,3,5-triazin-2(1H)-one (2f). Yield: 17.8%; mp: 200-202 °C (dec.) [12] (water/methanol); IR ν/cm-1: 3383.5, 1680.8, 1478.4, 1109.5, 1077.4, 902.9, 786.0; 1H-NMR δ/ppm: 3.63-3.69 (2H, m, H-5’, H-5”), 3.97 (1H, s, H-4’), 4.06 (1H, s, H-3’), 4.42-4.42-4.45 (1H, m, H-2’), 5.14-5.15 (1H, d, OH), 5.33-5.36 (1H, d, OH), 5.81 (1H, s, OH), 6.39-6.41 (1H, d, OH), 7.47-7.57 (3H, m, aromatic H ), 8.07 (1H, s, br, NH), 8.25-8.31(2H, m, aromatic H), 8.59 (1H, s, br, NH); ESI-MS: 321.1[M+H]+; Anal. Calcd. for C14H16N4O5: C 52.50, H 5.03, N 17.49. Found C 52.48, H 5.08, N 17.52.
4-Amino-1,3,5-triazin-2-yl-1-thio-β-d-ribofuranoside (3a). Yield: 15.9%; mp: 117-118 °C (water/ methanol); IR ν/cm-1: 3278.4, 1682.1, 1569.6, 1212.3, 1066.0, 1047.9, 985.6, 935.3, 915.7, 831.7, 756.7; 1H-NMR δ/ppm: 3.40-3.45 (2H, m, H-5’, H-5”), 3.78-3.79 (1H, d, H-4’), 3.90-3.92 (1H, d, H-3’), 4.00-4.02 (1H, d, J = 4.5 Hz, H-2’), 4.78 (1H, s, OH), 5.01-5.02 (1H, d, OH), 5.38-5.40 (1H, d, OH), 5.86-5.87 (1H, d, J = 4.5 Hz, H-1’), 7.62 (2H, s, NH2), 8.24 (1H, s, H-6); ESI-MS: 261.1 [M+H]+; Anal. Calcd. for C8H12N4O4S: C 36.92, H 4.65, N 21.53. Found C 36.81, H 4.69, N 21.59.
4-Amino-1,3,5-triazin-2-yl-1-thio-β-d-glucopyranoside (3b). Yield: 10.7%; mp: 134-135 °C (water/ methanol); IR ν/cm-1: 3334.0, 2918.7, 1647.5, 1564.8, 1384.8, 1195.1, 1044.3, 942.2, 877.3, 808.4, 759.3; 1H-NMR δ/ppm: 3.16 (4H, s, H-4’, H-5’, H-6’, H-6”), 3.43-3.46(1H, m, H-2’), 3.61-3.62 (1H, m, H-3’), 4.48 (1H, s, OH), 5.00 (1H, s, OH), 5.14-5.15 (1H, d, OH), 5.34-5.38 (1H,d, J = 9.9 Hz, H-1’), 5.40-5.42 (1H, d, OH), 7.56-7.59 (2H, d, NH2), 8.25 (1H, s, H-6); ESI-MS: 291.3 [M+H]+; Anal. Calcd. for C9H14N4O5S: C 37.24, H 4.86, N 19.30. Found C 37.34, H 4.89, N 19.38.
4-Amino-1,3,5-triazin-2-yl-1-thio-β-d-xylopyranoside (3c). Yield: 14.6%; mp: 150-152 °C (water/ methanol); IR ν/cm-1: 3288.0, 1691.9, 1575.4, 1382.2, 110.7, 1068.6, 900.9, 833.4, 804.1, 757.7; 1H-NMR δ/ppm: 3.12-3.23 (4H,m, H-2’, H-3’, H-4’, H-5”), 3.77-3.83 (1H, m, H-5’), 5.06-5.08 (1H, d, OH), 5.22-5.24 (1H, d, OH), 5.38-5.41 (1H, d, J = 8.7 Hz, H-1’), 5.42-5.44 (1H, OH), 7.59 (2H, s, NH2), 8.25(1H, s, H-6); ESI-MS: 259.2 [M-H]+; Anal. Calcd. for C8H12N4O4S: C 36.92, H 4.65, N 21.53. Found C 36.98, H 4.58, N 21.50.
4-Amino-1,3,5-triazin-2-yl-1-thio-α-d-mannopyranoside (3d). Yield: 11.7%; mp: 116-118 °C (water/ methanol); IR ν/cm-1: 3401.9, 1652.3, 1564.4, 1384.1, 1105.1, 880.7, 753.7; 1H-NMR δ/ppm: 3.17-3.44 (4H, m, H-4’, H-5’, H-6’, H-6”), 3.62 (1H, s, H-3’), 3.84 (1H, s, H-2’), 4.50 (1H, s, OH), 4.89 (2H, s, OH), 5.24 (1H, s, OH), 6.30 (1H, s, H-1’), 7.66 (2H, s, NH2), 8.27 (1H, s, H-6); ESI-MS: 291.1 [M+H]+; Anal. Calcd. for C9H14N4O5S: C 37.24, H 4.86, N 19.30. Found C 37.16, H 4.85, N 19.37.
4-Amino-4’-O-α-d-glucopyranosyl-1,3,5-triazin-2-yl-1-thio-β-d-glucopyranoside (3e). Yield: 14.3%; mp: 191-193 °C (water/methanol); IR ν/cm-1: 3404.3, 2923.2, 1642.0, 1566.0, 1518.8, 1384.2, 1032.7, 810.5, 759.3; 1H-NMR δ/ppm: 3.06-3.31 (3H, m), 3.40-3.46 (6H, m), 3.59-3.62 (3H, m, H-2’, H-2”, H-3’), 4.48-4.50 (2H, m, OH), 4.88-4.93 (2H, OH), 5.03-5.04 (1H, d, J = 3.6 Hz, H-1”), 5.36-5.40 (1H, d, J = 10.5 Hz, H-1’), 5.42-5.44 (1H, d, OH), 5.51-5.53 (1H, d, OH), 5.67-5.68 (1H, d, OH), 7.55-7.61 (2H, d, NH2), 8.25 (1H, s, H-6); ESI-MS: 453.2 [M+H]+; Anal. Calcd. for C15H24N4O10S: C 39.82, H 5.35, N 12.38. Found C 39.76, H 5.28, N 12.34.
4-Amino-6-methyl-1,3,5-triazin-2-yl-1-thio-β-d-ribofuranoside (3f). Yield: 16.8%; mp: 117-118 °C (water/methanol); IR ν/cm-1: 3406.9, 3180.9, 1662.1, 1557.3, 1043.3, 801.9; 1H-NMR δ/ppm: 2.20 (3H, s, CH3), 3.39-3.41 (1H, m, H-5”), 3.45-3.46 (1H, m, H-5’), 3.78-3.79 (1H, m, H-4’), 3.89-3.91 (1H, m, H-3’), 3.98-4.01 (1H, m, H-2’), 4.76-4.78 (1H, m, OH), 5.01-5.03 (1H, d, OH), 5.38-5.40 (1H, d, OH), 5.88-5.89 (1H, d, J = 4.5 Hz, H-1’), 7.47 (2H, s, NH2); ESI-MS: 275.1 [M+H]+; Anal Calcd. for C9H14N4O4S: C 39.41, H 5.14, N 20.43. Found C 39.52, H 5.18, N 20.45.
4-Amino-6-methyl-1,3,5-triazin-2-yl-1-thio-β-d-glucopyranoside (3g). Yield: 15.9%; mp: 117-119 °C (water/methanol); IR ν/cm-1: 3340.1, 1644.8, 1545.3, 1384.3, 1283.5, 1049.1, 879.0, 820.7; 1H-NMR δ/ppm: 2.21 (3H, s, CH3), 3.12-3.17 (3H, m, H-5’, H-6’, H-6”), 3.21-3.22 (1H, m, H-4’), 3.44-3.45 (1H, m, H-2’), 3.60-3.61 (1H, m, H-3’), 4.46-4.48 (1H, t, OH), 5.00-5.01 (1H, d, OH), 5.14-5.15 (1H, d, OH), 5.37-5.38 (1H, d, J = 10.4 Hz, H-1’), 5.39-5.40 (1H, d, OH), 7.40 (1H, s, NH), 7.45 (1H, s, NH); ESI-MS: 303.3 [M-H]+; Anal. Calcd. for C10H16N4O5S: C 39.47, H 5.30, N 18.41. Found C 39.53, H 5.32, N 18.52.
4-Amino-6-methyl-1,3,5-triazin-2-yl-1-thio-β-d-xylopyranoside (3h). White crystal; yield: 13.8%; mp: 150-152 °C (water/methanol); IR ν/cm-1: 3280.5, 1684.9, 1582.4, 1379.2, 1068.6, 902.5, 836.6, 759.2; 1H-NMR δ/ppm: 2.22 (3H, s, CH3), 3.12-3.23 (3H, m, H-3’, H-4’, H-5”), 3.39 (1H, m, H-2’), 3.78-3.82 (1H, m, H-5’), 5.05-5.07 (1H, d, OH), 5.21-5.22 (1H, s, OH), 5.40-5.44 (2H, m, OH, H-1’), 7.45 (2H, d, NH2). ESI-MS: 273.4 [M-H]+; Anal. Calcd. for C9H14N4O4S: C 39.41, H 5.14, N 20.43. Found C 39.50, H 5.32, N 20.46.
4-Amino-6-methyl-1,3,5-triazin-2-yl-1-thio-α-d-mannopyranoside (3i). Yield: 12.4%; mp: 199-201 °C (water/methanol); IR ν/cm-1: 3339.3, 1654.6, 1553.5, 1281.2, 1212.6, 1071.6, 973.5, 899.8, 844.5, 804.3, 766.0; 1H-NMR δ/ppm: 2.23 (3H, s, CH3), 3.44-3.48 (4H, m, H-3’, H-4’, H-5’, H-6’), 3.61-3.65 (1H, d, H-6”), 3.83 (1H, s, H-2’), 4.49 (1H, s, OH), 4.88 (2H, s, OH), 5.21 (1H, s, OH), 6.34 (1H, s, H-1’), 7.49-7.51 (2H, d, NH2); ESI-MS: 303.4 [M-H]+; Anal. Calcd. for C10H16N4O5S: C 39.47, H 5.30, N 18.41. Found C 39.52, H 5.36, N 18.44.
4-Amino-4’-O-α-d-glucopyranosyl-6-methyl-1,3,5-triazin-2-yl-1-thio-β-d-glucopyranoside (3j). Yield: 16.8%; mp: 194-196 °C (water/methanol); IR ν/cm-1: 3384.5, 1645.0, 1556.8, 1281.5, 1074.0, 895.2, 805.0; 1H-NMR δ/ppm: 2.24 (3H, s, CH3), 3.23-3.26 (2H, m), 3.34-3.52 (2H, m), 3.54-3.55 (3H, m), 3.56-3.58 (5H, m), 3.65-3.72 (3H, m), 4.24-4.27 (1H, d, OH), 4.28-5.32 (5H, br, OH), 5.41-5.45 (1H, d, J = 10.3 Hz, H-1’), 7.44-7.49 (2H, d, NH2); ESI-MS: 465.4 [M-H]+; Anal. Calcd. for C16H26N4O10S: C 41.20, H 5.62, N 12.01. Found C 41.32, H 5.68, N 12.05.
Biological activity assay
Cell culture
Human leukemia HL-60 cells obtained from ATCC were cultured in RPMI 1640. The media were supplemented with 100 units/mL penicillin, 100 μg/mL streptomycin, 0.2% NaHCO3, and 10% (v/v) heat-inactivated fetal bovine serum (FBS).
Cell growth inhibition
Cells were seeded at 5 × 104 cells/mL and incubated with various concentrations of the indicated agents for three days. The total cell number in each group was determined with the aid of a hemocytometer and the drug concentration that inhibits half of cell growth (GI50) was calculated. The cell viability was estimated by trypan-blue exclusion assay.
NBT reduction assay
The nitroblue tetrazolium (NBT) reduction assay as a determination of cell differentiation was performed as reported previously [21] and the cells were treated with 0.15 μM 5-aza-CR, 0.85 μM 3a, 10 μM 1g or 50 μM each of other compounds alone and in combination with 0.1 μM ATRA for 5 days. Total 300 cells were counted and percentages of NBT positive cells were calculated.
Acknowledgements
This work was supported by Joint Research Fund for Overseas Chinese Young Scholars of National Natural Science Foundation of China (30328030).
References
- Griffiths, E. A.; Gore, S. D. DNA methyltransferase and histone deacetylase inhibitors in the treatment of myelodysplastic syndromes. Semin. Hematol. 2008, 45, 23–30. [Google Scholar] [CrossRef]
- Grønbaek, K.; Hother, C.; Jones, P. A. Epigenetic changes in cancer. APMIS 2007, 115, 1039–1059. [Google Scholar] [CrossRef]
- Egger, G.; Liang, G.; Aparicio, A.; Jones, P. A. Epigenetics in human disease and prospects for epigenetic therapy. Nature 2004, 429, 457–463. [Google Scholar] [CrossRef]
- Zelent, A.; Petrie, K.; Lotan, R.; Waxman, S.; Gore, S. D. Clinical translation of epigenetics in cancer: eN-CORe--a report on the second workshop. Mol. Cancer. Ther. 2005, 4, 810–819. [Google Scholar]
- Marquez, V. E.; Kelley, J. A.; Agbaria, R.; Ben-Kasus, T.; Cheng, J. C.; Yoo, C. B.; Jones, P. A. Zebularine: a unique molecule for an epigenetically based strategy in cancer chemotherapy. Ann. N. Y. Acad. Sci. 2005, 1058, 246–254. [Google Scholar]
- Cheng, J. C.; Matsen, C. B.; Gonzales, F. A.; Ye, W.; Greer, S.; Marquez, V. E.; Jones, P. A.; Selker, E. U. Inhibition of DNA methylation and reactivation of silenced genes by zebularine. J. Natl. Cancer. Inst. 2003, 95, 399–409. [Google Scholar] [CrossRef]
- Gowher, H.; Jeltsch, A. Mechanism of inhibition of DNA methyltransferases by cytidine analogs in cancer therapy. Cancer Biol. Ther. 2004, 3, 1062–1068. [Google Scholar] [CrossRef]
- Beumer, J. H.; Parise, R. A.; Newman, E. M.; Doroshow, J. H.; Synold, T. W.; Lenz, H. J.; Egorin, M. J. Concentrations of the DNA methyltransferase inhibitor 5-fluoro-2'-deoxycytidine (FdCyd) and its cytotoxic metabolites in plasma of patients treated with FdCyd and tetrahydrouridine (THU). Cancer. Chemother. Pharmacol. 2008, 62, 363–368. [Google Scholar] [CrossRef]
- Noordhuis, P.; Holwerda, U.; Van der Wilt, C. L.; Van Groeningen, C. J.; Smid, K.; Meijer, S.; Pinedo, H. M.; Peters, G. J. 5-Fluorouracil incorporation into RNA and DNA in relation to thymidylate synthase inhibition of human colorectal cancers. Ann. Oncol. 2004, 15, 1025–1032. [Google Scholar] [CrossRef]
- Hartenstein, R.; Fridivich, I. Amidinourea formate, a precursor of 2-amino-4-hydroxy-s-triazine. J. Org. Chem. 1967, 32, 1653–1654. [Google Scholar] [CrossRef]
- Piskala, A. Nucleic acid components and their analogues Cl.* synthesis of 5-azacytosine (4-amino-1,2-dihydro-1,3,5-triazin-2-one) and its methyl derivatives. Coll. Czech. Chem. Commun. 1967, 32, 3966–3976. [Google Scholar]
- Hanna, N. B.; Masojidkova, M.; Fiedler, P.; Piskala, A. Synthesis of some 6-substituted 5-azacytidine. Collect. Czech. Chem. Commun. 1998, 63, 222–230. [Google Scholar] [CrossRef]
- Hanna, N. B.; Zajicek, J.; Piskala, A. 6-Methyl-5-azacytidine-synthesis, conformational properties and biological activity. a comparison of molecular conformation with 5-azacytidine. Nucleos. Nucleot 1997, 16, 129–144. [Google Scholar] [CrossRef]
- Niedballa, U.; Vorbruggen, H. A general synthesis of N-glycoside. V. synthesis of 5-azacytidines. J. Org. Chem. 1974, 39, 3672–3674. [Google Scholar] [CrossRef]
- Gaubert, G.; Mathe, C.; Imbach, J. L.; Eriksson, S.; Vincenzetti, S.; Salvatori, D.; Vita, A.; Maury, G. Unnatural enantiomers of 5-azacytidine analogues: synthesis and enzymatic properties. Eur. J. Med. Chem. 2000, 35, 1011–1019. [Google Scholar]
- Vorbruggen, H.; Hofle, G. On the mechanism of nucleoside synthesis. Chem. Ber. 1981, 114, 1256–1268. [Google Scholar] [CrossRef]
- Di Croce, L.; Raker, V. A.; Corsaro, M.; Fazi, F.; Fanelli, M.; Faretta, M.; Fuks, F.; Lo Coco, F.; Kouzarides, T.; Nervi, C.; Minucci, S.; Pelicci, P. G. Methyltransferase recruitment and DNA hypermethylation of target promoters by an oncogenic transcription factor. Science 2002, 295, 1079–1082. [Google Scholar] [CrossRef]
- Stresemann, C.; Brueckner, B.; Musch, T.; Stopper, H.; Lyko, F. Functional diversity of DNA methyltransferase inhibitors in human cancer cell lines. Cancer Res. 2006, 66, 2794–2800. [Google Scholar]
- Chuang, J. C.; Yoo., C. B.; Kwan, J. M.; Li, T. W.; Liang, G.; Yang, A. S.; Jones, P. A. Comparison of biological effects of non-nucleoside DNA methylation inhibitors versus 5-aza-2'-deoxycytidine. Mol. Cancer. Ther. 2005, 4, 1515–20. [Google Scholar] [CrossRef]
- Piskala, A.; Sorm, F. Nucleic acids components and their analogues. LI.* synthesis of 1-glycosyl derivatives of 5-azauracil and 5-azacytosine. Collect. Czech. Chem. Commun. 1964, 29, 2060–2076. [Google Scholar] [CrossRef]
- Jing, Y.; Wang, L.; Xia, L.; Chen, G. Q.; Chen, Z.; Miller, W. H.; Waxman, S. Combined effect of all-trans retinoic acid and arsenic trioxide in acute promyelocytic leukemia cells in vitro and in vivo. Blood 2001, 97, 264–269. [Google Scholar] [CrossRef]
- Sample Availability: Contact the authors.
© 2008 by the authors. Licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
