Acute Effect of Caffeine on the Synthesis of Pro-Inflammatory Cytokines in the Hypothalamus and Choroid Plexus during Endotoxin-Induced Inflammation in a Female Sheep Model
Abstract
:1. Introduction
2. Results
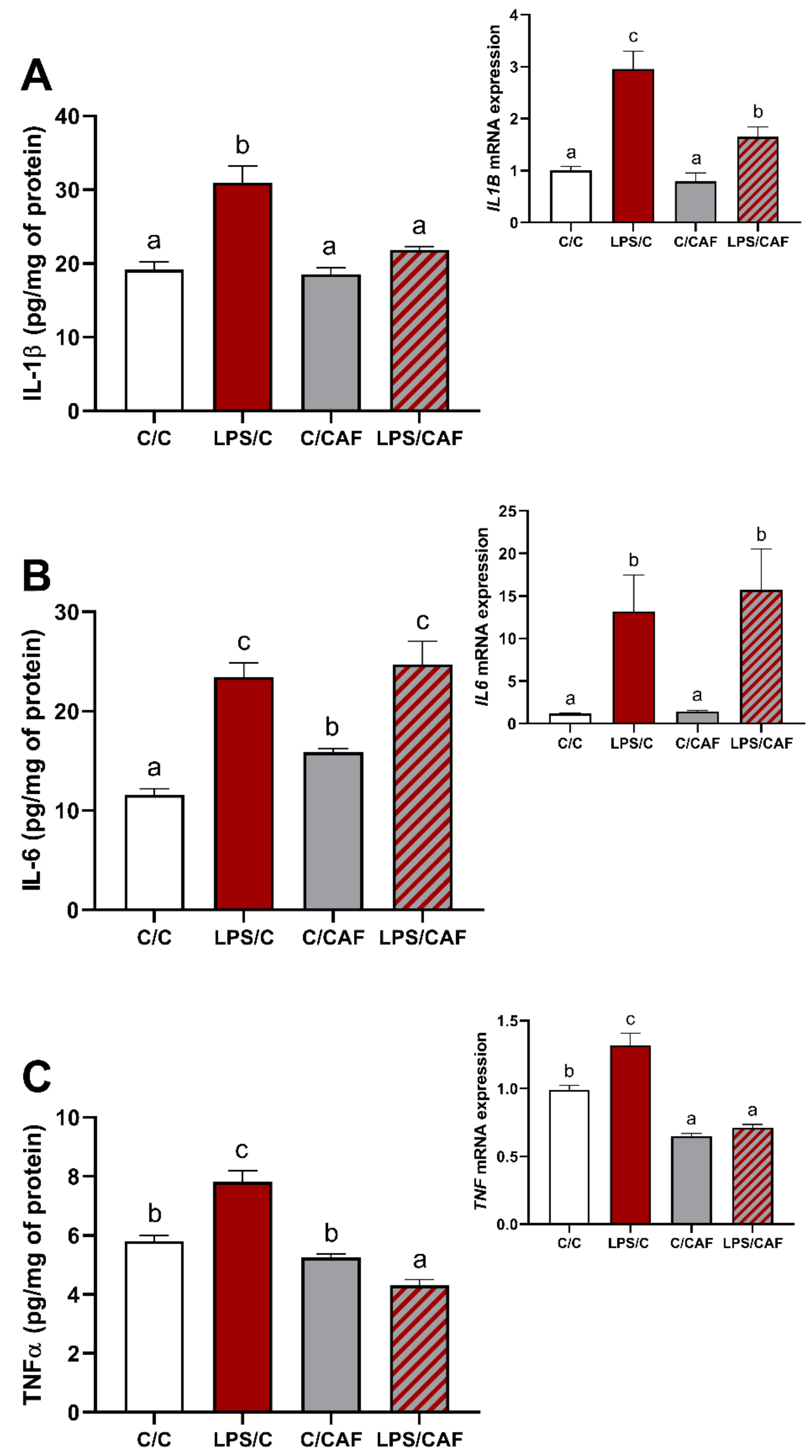
2.1. The Effect of Caffeine on Gene Expression and Concentration of Pro-Inflammatory Cytokines in the Hypothalamus under Basal and LPS-Challenge Conditions
2.2. The Effect of Caffeine on Pro-Inflammatory Cytokines Receptors Gene Expression in the Ovine Hypothalamus under Basal and LPS-Challenge Conditions
2.3. The Effect of Caffeine on Gene Expression of Pro-Inflammatory Cytokines and Their Receptors in the Choroid Plexus under Basal and LPS-Challenge Conditions
3. Discussion
4. Materials and Methods
4.1. Animals and Experimental Design
4.2. Inflammatory Cytokines and Cortisol Concentration Assessment
4.3. Relative Gene Expression Determination
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Kluger, M.J. Fever: Role of pyrogens and cryogens. Physiol. Rev. 1991, 71, 93–127. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, D.F.; Bowen, J.M.; Krasa, H.B.; Thrun, L.A.; Viguie, C.; Karsch, F.J. Endotoxin inhibits the reproductive neuroendocrine axis while stimulating adrenal steroids: A simultaneous view from hypophyseal portal and peripheral blood. Endocrinology 1997, 138, 4273–4281. [Google Scholar] [CrossRef]
- Lawrence, C.B.; Rothwell, N.J. Anorexic but not pyrogenic actions of interleukin-1 are modulated by central melanocortin-3/4 receptors in the rat. J. Neuroendocrinol. 2001, 13, 490–495. [Google Scholar] [CrossRef]
- Palomba, M.; Bentivoglio, M. Chronic inflammation affects the photic response of the suprachiasmatic nucleus. J. Neuroimmunol. 2008, 193, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, C.; Yoshioka, K.; Iwamura, S.; Hirosa, H. Endotoxin induces delayed ovulation following endocrine aberration during the proestrous phase In Holstein heifers. Dom. Anim. Endocrinol. 2001, 20, 267–278. [Google Scholar] [CrossRef]
- Daniel, J.A.; Abrams, M.S.; De Souza, L.; Wagner, C.G.; Whitlock, B.K.; Sartin, J.L. Endotoxin inhibition of luteinizing hormone in sheep. Domest. Anim. Endocrinol. 2003, 25, 13–19. [Google Scholar] [CrossRef]
- Jana, B.; Kucharski, J.; Zięcik, A. Effect of intrauterine infusion of Escherichia coli on hormonal patterns in gilts during the estrous cycle. Reprod. Nutr. Dev. 2004, 44, 37–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Refojo, D.; Arias, P.; Moguilevsky, J.A.; Felder, C. Effect of bacterial endotoxin on in vivo pulstile gonadotropin secretion in adult male rats. Neuroendocrinology 1998, 67, 275–281. [Google Scholar] [CrossRef]
- Xiao, E.; Xia-Zhang, L.; Barth, A.; Zhu, J.; Ferin, M. Stress and the menstrual cycle: Relevance of cycle quality in the short- and long-term response to a 5-day endotoxin challenge during the follicular phase in the rhesus monkey. J. Clin. Endocrinol. Metab. 1998, 83, 2454–2460. [Google Scholar] [CrossRef]
- Rapone, B.; Ferrara, E.; Montemurro, N.; Ilaria Converti, I.; Loverro, M.; Loverro, M.T.; Gnoni, A.; Scacco, S.; Siculella, L.; Corsalini, M.; et al. Oral Microbiome and Preterm Birth: Correlation or Coincidence? A Narrative Review. Open Access Maced J. Med. Sci. 2020, 8, 123–132. [Google Scholar] [CrossRef]
- Tomaszewska-Zaremba, D.; Herman, A. The role of immunological system in the regulation of gonadoliberin and gonadotropin secretion. Reprod. Biol. 2009, 9, 11–23. [Google Scholar] [CrossRef]
- Creasey, A.A.; Stevens, P.; Kenney, J.; Allison, A.C.; Warren, K.; Catlett, R.; Hinshaw, L.; Taylor, F.B., Jr. Endotoxin and cytokine profile in plasma of baboons challenged with lethal and sublethal Escherichia coli. Circ. Shock. 1991, 33, 84–91. [Google Scholar] [PubMed]
- Watanobe, H.; Hayakawa, Y. Hypothalamic interleukin-1β and tumor necrosis factor-α, but not interleukin-6, mediate the endotoxin-induced suppression of the reproductive axis in rats. Endocrinology 2003, 144, 4868–4875. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erickson, M.A.; Banks, W.A. Cytokine and chemokine responses in serum and brain after single and repeated injections of lipopolysaccharide: Multiplex quantification with path analysis. Brain Behav. Immun. 2011, 25, 1637–1648. [Google Scholar] [CrossRef] [Green Version]
- Skipor, J.; Kowalewska, M.; Szczepkowska, A.; Majewska, A.; Misztal, T.; Jalynski, M.; Herman, A.P.; Zabek, K. Plasma and cerebrospinal fluid interleukin-1β during lipopolysaccharide-induced systemic inflammation in ewes implanted or not with slow-release melatonin. J. Anim. Sci. Biotechnol. 2017, 8, 76. [Google Scholar] [CrossRef] [Green Version]
- Rivest, S.; Lee, S.; Attardi, B.; Rivier, C. The chronic intracerebroventricular infusion of interleukin-1b alters the activity of the hypothalamic-pituitary-gonadal axis of cycling rats. I. Effect on LHRH and gonadotropin biosynthesis and secretion. Endocrinology 1993, 133, 2424–2430. [Google Scholar] [CrossRef]
- Herman, A.P.; Tomaszewska-Zaremba, D. Effect of endotoxin on the expression of GnRH and GnRHR genes in the hypothalamus and anterior pituitary gland of anestrous ewes. Anim. Reprod. Sci. 2010, 120, 105–111. [Google Scholar] [CrossRef]
- Herman, A.P.; Misztal, T.; Romanowicz, K.; Tomaszewska-Zaremba, D. Central injection of exogenous IL-1β in the control activities of hypothalamic-pituitary-gonadal axis in anestrous ewes. Reprod. Domest. Anim. 2012, 47, 44–52. [Google Scholar] [CrossRef]
- Herman, A.P.; Skipor, J.; Krawczyńska, A.; Bochenek, J.; Wojtulewicz, K.; Pawlina, B.; Antushevich, H.; Herman, A.; Tomaszewska-Zaremba, D. Effect of central injection of neostigmine on the bacterial endotoxin induced suppression of GnRH/LH secretion in ewes during the follicular phase of the estrous cycle. Int. J. Mol. Sci. 2019, 20, 4598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banks, W.A.; Kastin, A.J.; Broadwell, R.D. Passage of cytokines across the blood-brain barier. Neuroimmunomodulation 1995, 2, 241–248. [Google Scholar] [CrossRef]
- Pan, W.; Stone, K.P.; Hsuchou, H.; Manda, V.K.; Zhang, Y.; Kastin, A.J. Cytokine signaling modulates blood-brain barrier function. Curr. Pharm. Des. 2011, 17, 3729–3740. [Google Scholar] [CrossRef] [Green Version]
- Vitkovic, L.; Konsman, J.P.; Bockaert, J.; Dantzer, R.; Homburger, V.; Jacque, C. Cytokine signals propagate through the brain. Mol. Psychiatry 2000, 5, 604–615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kovac, A.; Erickson, M.A.; Banks, W.A. Brain microvascular pericytes are immunoactive in culture: Cytokine, chemokine, nitric oxide, and LRP-1 expression in response to lipopolysaccharide. J. Neuroinflamm. 2011, 8, 139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kowalewska, M.; Herman, A.P.; Szczepkowska, A.; Skipor, J. The effect of melatonin from slow-release implants on basic and TLR-4-mediated gene expression of inflammatory cytokines and their receptors in the choroid plexus in ewes. Res. Vet. Sci. 2017, 113, 50–55. [Google Scholar] [CrossRef]
- Ghersi-Egea, J.F.; Strazielle, N.; Catala, M.; Silva-Vargas, V.; Doetsch, F.; Engelhardt, B. Molecular anatomy and functions of the choroidal blood-cerebrospinal fuid barrier in health and disease. Acta Neuropathol. 2018, 135, 337–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meeker, R.B.; Williams, K.; Killebrew, D.A.; Hudson, L.C. Cell trafficking through the choroid plexus. Cell Adhes. Migr. 2012, 6, 390–396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, G.Q.; Chen, Y.Y.; Wang, X.S.; Wu, S.Z.; Yang, H.M.; Xu, H.Q.; He, J.C.; Wang, X.T.; Chen, J.F.; Zheng, R.Y. Chronic caffeine treatment attenuates experimental autoimmune encephalomyelitis induced by guinea pig spinal cord homogenates in Wistar rats. Brain Res. 2010, 1309, 116–125. [Google Scholar] [CrossRef]
- Cechella, J.L.; Leite, M.R.; Dobrachinski, F.; da Rocha, J.T.; Carvalho, N.R.; Duarte, M.M.; Soares, F.A.; Bresciani, G.; Royes, L.F.; Zeni, G. Moderate swimming exercise and caffeine supplementation reduce the levels of inflammatory cytokines without causing oxidative stress in tissues of middle-aged rats. Amino Acids 2014, 46, 1187–1195. [Google Scholar] [CrossRef] [PubMed]
- Iris, M.; Tsoua, P.S.; Sawalha, A.H. Caffeine inhibits STAT1 signaling and downregulates inflammatory pathways involved in autoimmunity. Clinical. Immunol. 2018, 192, 68–77. [Google Scholar] [CrossRef]
- Chavez-Valdez, R.; Wills-Karp, M.; Ahlawat, R.; Cristofalo, E.A.; Nathan, A.; Gauda, E.B. Caffeine modulates TNF-alpha production by cord blood monocytes: The role of adenosine receptors. Pediatr. Res. 2009, 65, 203–208. [Google Scholar] [CrossRef] [Green Version]
- Lv, X.; Chen, Z.; Li, J.; Zhang, L.; Liu, H.; Huang, C.; Zhu, P. Caffeine protects against alcoholic liver injury by attenuating inflammatory response and oxidative stress. Inflamm. Res. 2010, 59, 635–645. [Google Scholar] [CrossRef]
- Fredholm, B.B.; IJzerman, A.P.; Jacobson, K.A.; Klotz, K.N.; Linden, J. International Union of Pharmacology. XXV. Nomenclature and classification of adenosine receptors. Pharmacol. Rev. 2001, 53, 527–552. [Google Scholar]
- Chavez-Valdez, R.; Ahlawat, R.; Wills-Karp, M.; Gauda, E.B. Mechanisms of modulation of cytokine release by human cord blood monocytes exposed to high concentrations of caffeine. Pediatr. Res. 2016, 80, 101–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheth, S.; Brito, R.; Mukherjea, D.; Rybak, L.P.; Ramkumar, V. Adenosine receptors: Expression, function and regulation. Int. J. Mol. Sci. 2014, 15, 2024–2052. [Google Scholar] [CrossRef] [Green Version]
- Kalaria, R.N.; Harik, S.I. Adenosine receptors of cerebral microvessels and choroid plexus. J. Cereb. Blood Flow Metab. 1986, 6, 463–470. [Google Scholar] [CrossRef] [Green Version]
- Mills, J.H.; Thompson, L.F.; Mueller, C.; Waickman, A.T.; Jalkanen, S.; Niemela, J.; Airas, L.; Bynoe, M.S. CD73 is required for efficient entry of lymphocytes into the central nervous system during experimental autoimmune encephalomyelitis. Proc. Natl. Acad. Sci. USA 2008, 105, 9325–9330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eltzschig, H.K.; Sitkovsky, M.V.; Robson, S.C. Purinergic signaling during inflammation. N. Engl. J. Med. 2012, 367, 2322–2333. [Google Scholar] [CrossRef] [Green Version]
- Available online: https://maayanlab.cloud/Harmonizome/gene_set/choroid+plexus+of+the+lateral+ventricle/Allen+Brain+Atlas+Adult+Human+Brain+Tissue+Gene+Expression+Profiles (accessed on 12 May 2021).
- Popov, D.; Pavlov, G. Sepsis models in experimental animals. Trakia J.Ournal Sci. 2013, 1, 13–23. [Google Scholar]
- Piccione, G.; Giannetto, C.; Casella, S.; Caola, G. Circadian activity rhythm in sheep and goats housed in stable conditions. Folia Biol. 2008, 56, 133–137. [Google Scholar] [CrossRef] [Green Version]
- Skipor, J.; Szczepkowska, A.; Kowalewska, M.; Domżalska, M.; Herman, A.P.; Krawczyńska, A. Photoperiod alters the choroid plexus response to LPS-induced acute inflammation in ewes. Ann. Anim. Sci. 2021, 21, 497–512. [Google Scholar] [CrossRef]
- Mallik, S.B.; Mudgal, J.; Susan Hall, S.; Kinra, M.; Grant, G.D.; Nampoothiri, M.; Anoopkumar-Dukie, S.; Arora, D. Remedial effects of caffeine against depressive-like behaviour in mice by modulation of neuroinflammation and BDNF. Nutr. Neurosci. 2021, 5, 1–9. [Google Scholar] [CrossRef]
- Banks, W.A.; Robinson, S.M. Minimal penetration of lipopolysaccharide across the murine blood-brain barrier. Brain Behav. Immun. 2010, 24, 102–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagyőszi, P.; Wilhelm, I.; Farkas, A.E.; Fazakas, C.; Dung, N.T.; Hasko, J.; Krizbai, I.A. Expression and regulation of toll-like receptors in cerebral endothelial cells. Neurochem. Int. 2010, 57, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Skipor, J.; Szczepkowska, A.; Kowalewska, M.; Herman, A.P.; Lisiewski, P. Profile of toll-like receptor mRNA expression in the choroid plexus in adult ewes. Acta Vet. Hung. 2015, 63, 69–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lopez-Castejon, G.; Brough, D. Understanding the mechanism of IL-1β secretion. Cytokine Growth Factor Rev. 2011, 22, 189–195. [Google Scholar] [CrossRef]
- Halle, A.; Hornung, V.; Petzold, G.C.; Stewart, C.R.; Monks, B.G.; Reinheckel, T.; Fitzgerald, K.A.; Latz, E.; Moore, K.J.; Golenbock, D.T. The NALP3 inflammasome is involved in the innate immune response to amyloid-beta. Nat. Immunol. 2008, 9, 857–865. [Google Scholar] [CrossRef] [Green Version]
- Jha, S.; Srivastava, S.Y.; Brickey, W.J.; Iocca, H.; Toews, A.; Morrison, J.P.; Chen, V.S.; Gris, D.; Matsushima, G.K.; Ting, J.P. The inflammasome sensor, NLRP3, regulates CNS inflammation and demyelination via caspase-1 and interleukin-18. J. Neurosci. 2010, 30, 15811–15820. [Google Scholar] [CrossRef] [PubMed]
- Nagyőszi, P.; Nyúl-Tóth, Á.; Fazakas, C.; Wilhelm, I.; Kozma, M.; Molnár, J.; Haskó, J.; Krizbai, I.A. Regulation of NOD-like receptors and inflammasome activation in cerebral endothelial cells. J. Neurochem. 2015, 135, 551–564. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.Q.; Song, K.Y.; Feng, J.Z.; Huang, S.Y.; Guo, X.M.; Zhang, L.; Zhang, G.; Huo, Y.C.; Zhang, R.R.; Ma, Y.; et al. Caffeine Inhibits Activation of the NLRP3 Inflammasome via Autophagy to Attenuate Microglia-Mediated Neuroinflammation in Experimental Autoimmune Encephalomyelitis. J. Mol. Neurosci. 2021. [Google Scholar] [CrossRef] [PubMed]
- Shimada, A.; Hasegawa-Ishii, S. Increased cytokine expression in the choroid plexus stroma and epithelium in response to endotoxin-induced systemic inflammation in mice. Toxicol. Rep. 2021, 8, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Atri, C.; Guerfali, F.Z.; Laouini, D. Role of Human Macrophage Polarization in Inflammation during Infectious Diseases. Int. J. Mol. Sci. 2018, 19, 1801. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ivan, D.C.; Walthert, S.; Locatelli, G. Central Nervous System Barriers Impact Distribution and Expression of iNOS and Arginase-1 in Infiltrating Macrophages During Neuroinflammation. Front. Immunol. 2021, 12, 666961. [Google Scholar] [CrossRef]
- Kovács, E.G.; Alatshan, A.; Budai, M.M.; Czimmerer, Z.; Bíró, E.; Benkő, S. Caffeine Has Different Immunomodulatory Effect on the Cytokine Expression and NLRP3 Inflammasome Function in Various Human Macrophage Subpopulations. Nutrients 2021, 13, 2409. [Google Scholar] [CrossRef]
- Skinner, R.A.; Gibson, R.M.; Rothwell, N.J.; Pinteaux, E.; Penny, J.I. Transport of interleukin-1 across cerebromicrovascular endothelial cells. Br. J. Pharmacol. 2009, 156, 1115–1123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carman, A.J.; Mills, J.H.; Krenz, A.; Kim, D.G.; Bynoe, M.S. Adenosine receptor signaling modulates permeability of the blood-brain barrier. J. Neurosci. 2011, 31, 13272–13280. [Google Scholar] [CrossRef] [PubMed]
- Bynoe, M.S.; Viret, C.; Yan, A.; Kim, D.G. Adenosine receptor signaling: A key to opening the blood-brain door. Fluids Barriers CNS 2015, 12, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horrigan, L.A.; Kelly, J.P.; Connor, T.J. Caffeine suppresses TNF-alpha production via activation of the cyclic AMP/protein kinase A pathway. Int. Immunopharmacol. 2004, 4, 1409–1417. [Google Scholar] [CrossRef]
- Idriss, H.T.; Naismith, J.H. TNF alpha and the TNF receptor superfamily: Structure-function relationship(s). Microsc. Res. Tech. 2000, 50, 184–195. [Google Scholar] [CrossRef]
- Hwang, J.H.; Kim, K.J.; Ryu, S.J.; Lee, B.Y. Caffeine prevents LPS-induced inflammatory responses in RAW264.7 cells and zebrafish. Chem. Biol. Interact. 2016, 248, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Tauler, P.; Martínez, S.; Moreno, C.; Monjo, M.; Martínez, P.; Aguiló, A. Effects of caffeine on the inflammatory response induced by a 15-km run competition. Med. Sci. Sports Exerc. 2013, 45, 1269–1276. [Google Scholar] [CrossRef]
- Rodas, L.; Martinez, S.; Aguilo, A.; Tauler, P. Caffeine supplementation induces higher IL-6 and IL-10 plasma levels in response to a treadmill exercise test. J. Int. Soc. Sports Nutr. 2020, 17, 47. [Google Scholar] [CrossRef]
- Fang, C.; Cai, X.; Hayashi, S.; Hao, S.; Sakiyama, H.; Wang, X.; Yang, Q.; Akira, S.; Nishiguchi, S.; Fujiwara, N.; et al. Caffeine-stimulated muscle IL-6 mediates alleviation of non-alcoholic fatty liver disease. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2019, 1864, 271–280. [Google Scholar] [CrossRef]
- Bustamante, A.; Sobrino, T.; Giralt, D.; García-Berrocoso, T.; Llombart, V.; Ugarriza, I.; Espadaler, M.; Rodríguez, N.; Sudlow, C.; Castellanos, M.; et al. Prognostic value of blood interleukin-6 in the prediction of functional outcome after stroke: A systematic review and meta-analysis. J. Neuroimmunol. 2014, 274, 215–224. [Google Scholar] [CrossRef]
- Pasquini, S.; Contri, C.; Borea, P.A.; Vincenzi, F.; Varani, K. Adenosine and Inflammation: Here, There and Everywhere. Int. J. Mol. Sci. 2021, 22, 7685. [Google Scholar] [CrossRef]
- Rossetto, I.M.U.; Cagnon, V.H.A.; Kido, L.A.; Lizarte Neto, F.S.; Tirapelli, L.F.; Tirapelli, D.P.D.C.; de Almeida Chuffa, L.G.; Martinez, F.E.; Martinez, M. Caffeine consumption attenuates ethanol-induced inflammation through the regulation of adenosinergic receptors in the UChB rats cerebellum. Toxicol. Res. 2021, 10, 835–849. [Google Scholar] [CrossRef]
- Rosas-Ballina, M.; Tracey, K.J. Cholinergic control of inflammation. J. Intern. Med. 2009, 265, 663–679. [Google Scholar] [CrossRef]
- Herman, A.P.; Tomaszewska-Zaremba, D.; Kowalewska, M.; Szczepkowska, A.; Oleszkiewicz, M.; Krawczyńska, A.; Wójcik, M.; Antushevich, H.; Skipor, J. Neostigmine Attenuates Proinflammatory Cytokine Expression in Preoptic Area but Not Choroid Plexus during Lipopolysaccharide-Induced Systemic Inflammation. Mediat. Inflamm. 2018, 2018, 9150207. [Google Scholar] [CrossRef]
- Pohanka, M.; Dobes, P. Caffeine inhibits acetylcholinesterase, but not butyrylcholinesterase. Int. J. Mol. Sci. 2013, 14, 9873–9882. [Google Scholar] [CrossRef] [PubMed]
- Pollard, I.; Williamson, S.; Downing, J.; Scaramuzzi, R. Pharmacokinetics of caffeine in the oestrogen-implanted ovariectomized ewe. J. Vet. Pharmacol. Ther. 1996, 19, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Kokot, F. Metody Radioimmunologiczne i Radiokompetycyjne Stosowane w Klinice; Państwowy Zakład Wydawnictw Lekarskich: Warsaw, Poland, 1985. [Google Scholar]
- Szczepkowska, A.; Kowalewska, M.; Krawczyńska, A.; Herman, A.P.; Skipor, J. Photoperiod Affects Leptin Action on the Choroid Plexus in Ewes Challenged with Lipopolysaccharide-Study on the mRNA Level. Int. J. Mol. Sci. 2020, 21, 7647. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, R. Quantification on the LightCycler. In Rapid Cycle Real-Time PCR Methods and Applications; Meuer, S., Wittwer, C., Nakagawara, K., Eds.; Springer: Berlin, Germany, 2001; pp. 21–34. [Google Scholar]
- Zhao, S.; Fernald, R.D. Comprehensive algorithm for quantitative real-time polymerase chain reaction. J. Comput. Biol. 2005, 12, 1047–1064. [Google Scholar] [CrossRef]
| Gene | C/C | LPS/C | C/CAF | LPS/CAF |
|---|---|---|---|---|
| IL1R1 interleukin 1 receptor 1 | 1 ± 0.1 A | 2.8 ± 0.4 B | 1.3 ± 0.1 A | 2.3 ± 0.3 B |
| IL1R2 interleukin 1 receptor 2 | 1 ± 0.1 A | 4.5 ± 0.7 B | 1.5 ± 0.1 A | 4.2 ± 0.6 B |
| IL6R interleukin 6 receptor | 1 ± 0.1 A | 1 ± 0.1 A | 1.1 ± 0.1 A | 1 ± 0.1 A |
| IL6ST interleukin 6 signal transducer | 1 ± 0.1 A | 1.4 ± 0.1 C | 1.2 ± 0.1 B | 1.3 ± 0.1 B |
| TNFRSF1A TNF receptor superfamily member 1A | 1 ± 0.1 A | 1.6 ± 0.2 B | 0.8 ± 0.1 A | 1 ± 0.1 A |
| TNFRSF1B TNF receptor superfamily member 1B | 1 ± 0.1 A | 1.9 ± 0.3 B | 1.1 ± 0.1 A | 1.8 ± 0.1 B |
| Gene | C/C | LPS/C | C/CAF | LPS/CAF |
|---|---|---|---|---|
| IL1B interleukin 1β; IL1R1 | 1 ± 0.1 A | 7.8 ± 2.1 B | 0.7 ± 0.1 A | 9.8 ± 3.1 B |
| IL1R1 interleukin 1 receptor 1 | 1 ± 0.1 A | 3.6 ± 0.8 B | 2.3 ± 0.3 A | 4.1 ± 0.4 B |
| IL1R2 interleukin 1 receptor 2 | 1 ± 0.2 A | 9.7 ± 1.6 C | 2.1 ± 0.3 A | 6.5 ± 0.3 B |
| IL-6 interleukin 6 | 1 ± 0.1 A | 587.4 ± 146.0 C | 1.9 ± 0.2 B | 489.7 ± 58.0 C |
| IL6R interleukin 6 receptor | 1 ± 0.1 AB | 0.8 ± 0.2 A | 1.4 ± 0.2 B | 0.8 ± 0.1 A |
| IL6ST interleukin 6 signal transducer | 1 ± 0.1 A | 2.8 ± 0.4 C | 1.9 ± 0.2 B | 2.9 ± 0.2 C |
| TNF tumor necrosis factor | 1 ± 0.1 AB | 2.1 ± 0.8 B | 0.7 ± 0.1 A | 0.8 ± 0.2 A |
| TNFRSF1A TNF receptor superfamily member 1A | 1 ± 0.1 A | 1.5 ± 0.2 B | 1.1 ± 0.1 A | 1.4 ± 0.1 B |
| TNFRSF1B TNF receptor superfamily member 1B | 1 ± 0.1 A | 3.8 ± 0.8 B | 1.7 ± 0.3 A | 4.0 ± 0.3 B |
| Group | No. of Animals | Experimental Treatment I | Dose [ng/kg] | Experimental Treatment II | Dose [mg/kg] |
|---|---|---|---|---|---|
| C/C | 6 | NaCl | 0 | NaCl | 0 |
| LPS/C | 6 | LPS | 400 | NaCl | 0 |
| C/CAF | 6 | NaCl | 0 | caffeine | 30 |
| LPS/CAF | 6 | LPS | 400 | caffeine | 30 |
| Gene | (Forward/Reverse) Sequence 5′ → 3′ | Amplicon Size (bp) | References/ Sources | |
|---|---|---|---|---|
| Genes under study | IL1B | F: CAGCCGTGCAGTCAGTAAAA R: GAAGCTCATGCAGAACACCA | 137 | [24,68] |
| IL1R1 | F: GGGAAGGGTCCACCTGTAAC R: ACAATGCTTTCCCCAACGTA | 124 | [24,68] | |
| IL1R2 | F: CGCCAGGCATACTCAGAAA R: GAGAACGTGGCAGCTTCTTT | 162 | [24,68] | |
| IL6 | F: GTTCAATCAGGCGATTTGCT R: CCTGCGATCTTTTCCTTCAG | 165 | [24,68] | |
| IL6R | F: TCAGCGACTCCGGAAACTAT R: CCGAGGACTCCACTCACAAT | 149 | [24,68] | |
| IL6ST | F: GGCTTGCCTCCTGAAAAACC R: ACTTCTCTGTTGCCCACTCAG | 139 | [24,68] | |
| TNF | F: CAAATAACAAGCCGGTAGCC R: AGATGAGGTAAAGCCCGTCA | 153 | [24,68] | |
| TNFRSF1A | F: AGGTGCCGGGATGAAATGTT R: CAGAGGCTGCAGTTCAGACA | 137 | [24,68] | |
| TNFSFR2A | F: ACCTTCTTCCTCCTCCCAAA R: AGAAGCAGACCCAATGCTGT | 122 | [24,68] | |
| Reference genes | GAPDH | F: AGAAGGCTGGGGCTCACT R: GGCATTGCTGACAATCTTGA | 134 | [68] hypothalamus |
| GAPDH | F: TGACCCCTTCATTGACCTTC R: GATCTCGCTCCTGGAAGATG | 143 | [72] ChP | |
| ACTB | F: CTTCCTTCCTGGGCATGG R: GGGCAGTGATCTCTTTCTGC | 168 | [68] hypothalamus | |
| ACTB | F: GCCAACCGTGAGAAGATGAC R: TCCATCACGATGCCAGTG | 122 | [72] ChP | |
| HDAC1 | F: CTGGGGACCTACGGGATATT R: GACATGACCGGCTTGAAAAT | 115 | [24,68] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szczepkowska, A.; Wójcik, M.; Tomaszewska-Zaremba, D.; Antushevich, H.; Krawczyńska, A.; Wiechetek, W.; Skipor, J.; Herman, A.P. Acute Effect of Caffeine on the Synthesis of Pro-Inflammatory Cytokines in the Hypothalamus and Choroid Plexus during Endotoxin-Induced Inflammation in a Female Sheep Model. Int. J. Mol. Sci. 2021, 22, 13237. https://doi.org/10.3390/ijms222413237
Szczepkowska A, Wójcik M, Tomaszewska-Zaremba D, Antushevich H, Krawczyńska A, Wiechetek W, Skipor J, Herman AP. Acute Effect of Caffeine on the Synthesis of Pro-Inflammatory Cytokines in the Hypothalamus and Choroid Plexus during Endotoxin-Induced Inflammation in a Female Sheep Model. International Journal of Molecular Sciences. 2021; 22(24):13237. https://doi.org/10.3390/ijms222413237
Chicago/Turabian StyleSzczepkowska, Aleksandra, Maciej Wójcik, Dorota Tomaszewska-Zaremba, Hanna Antushevich, Agata Krawczyńska, Wiktoria Wiechetek, Janina Skipor, and Andrzej Przemysław Herman. 2021. "Acute Effect of Caffeine on the Synthesis of Pro-Inflammatory Cytokines in the Hypothalamus and Choroid Plexus during Endotoxin-Induced Inflammation in a Female Sheep Model" International Journal of Molecular Sciences 22, no. 24: 13237. https://doi.org/10.3390/ijms222413237
APA StyleSzczepkowska, A., Wójcik, M., Tomaszewska-Zaremba, D., Antushevich, H., Krawczyńska, A., Wiechetek, W., Skipor, J., & Herman, A. P. (2021). Acute Effect of Caffeine on the Synthesis of Pro-Inflammatory Cytokines in the Hypothalamus and Choroid Plexus during Endotoxin-Induced Inflammation in a Female Sheep Model. International Journal of Molecular Sciences, 22(24), 13237. https://doi.org/10.3390/ijms222413237






