Biological Networks Entropies: Examples in Neural Memory Networks, Genetic Regulation Networks and Social Epidemic Networks
Abstract
:1. Introduction
2. Material and Methods
2.1. The Dynamical Tools
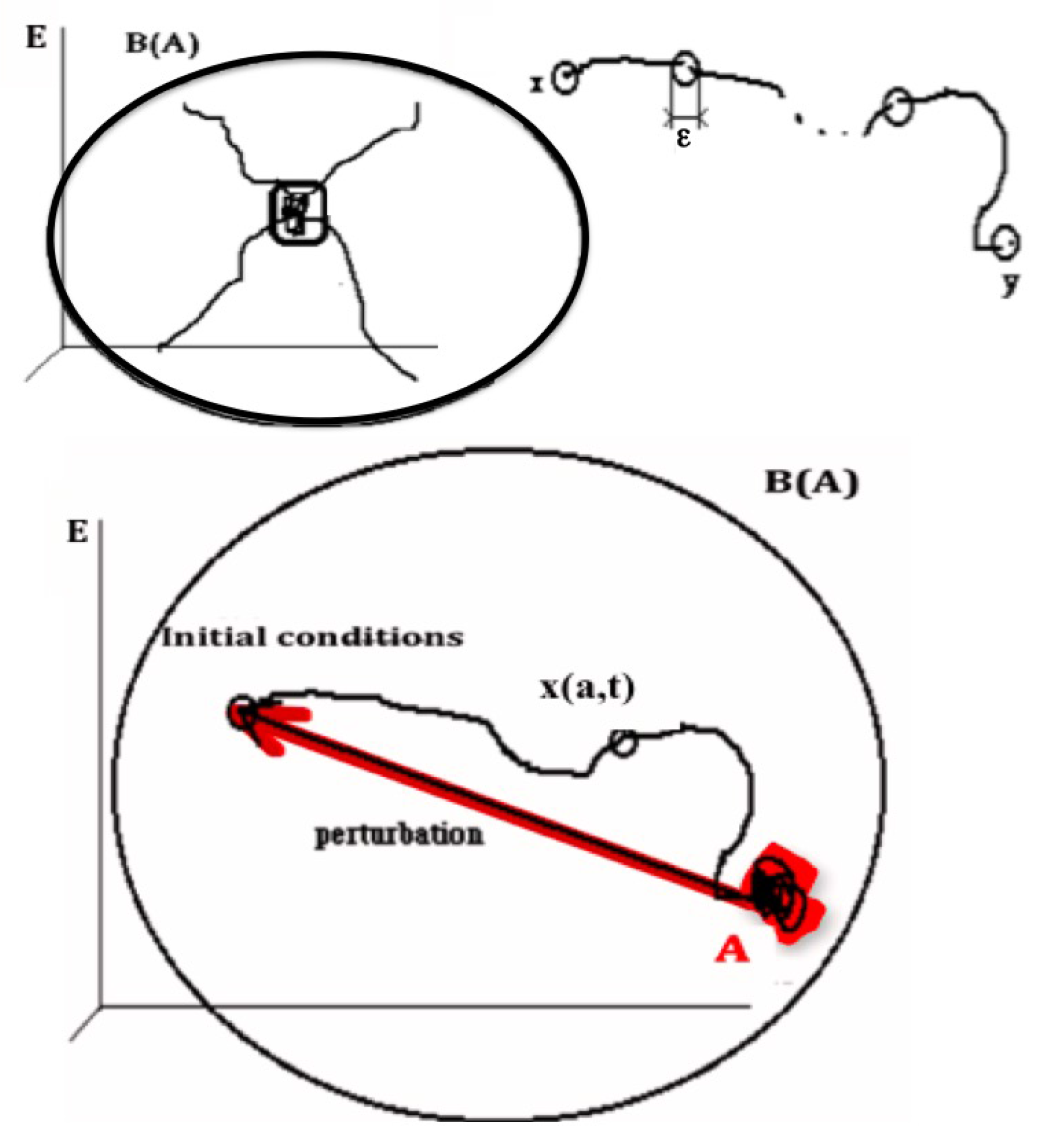
2.1.1. Definition of the Notion of Attractor
- (i)
- , where the composed operator is obtained by applying the basin operator B and then the limit operator L to the set A
- (ii)
- There is no set A’ containing strictly A, shadow-connected to A and verifying (i)
- (iii)
- There is no set A” strictly contained in A verifying (i) and (ii)
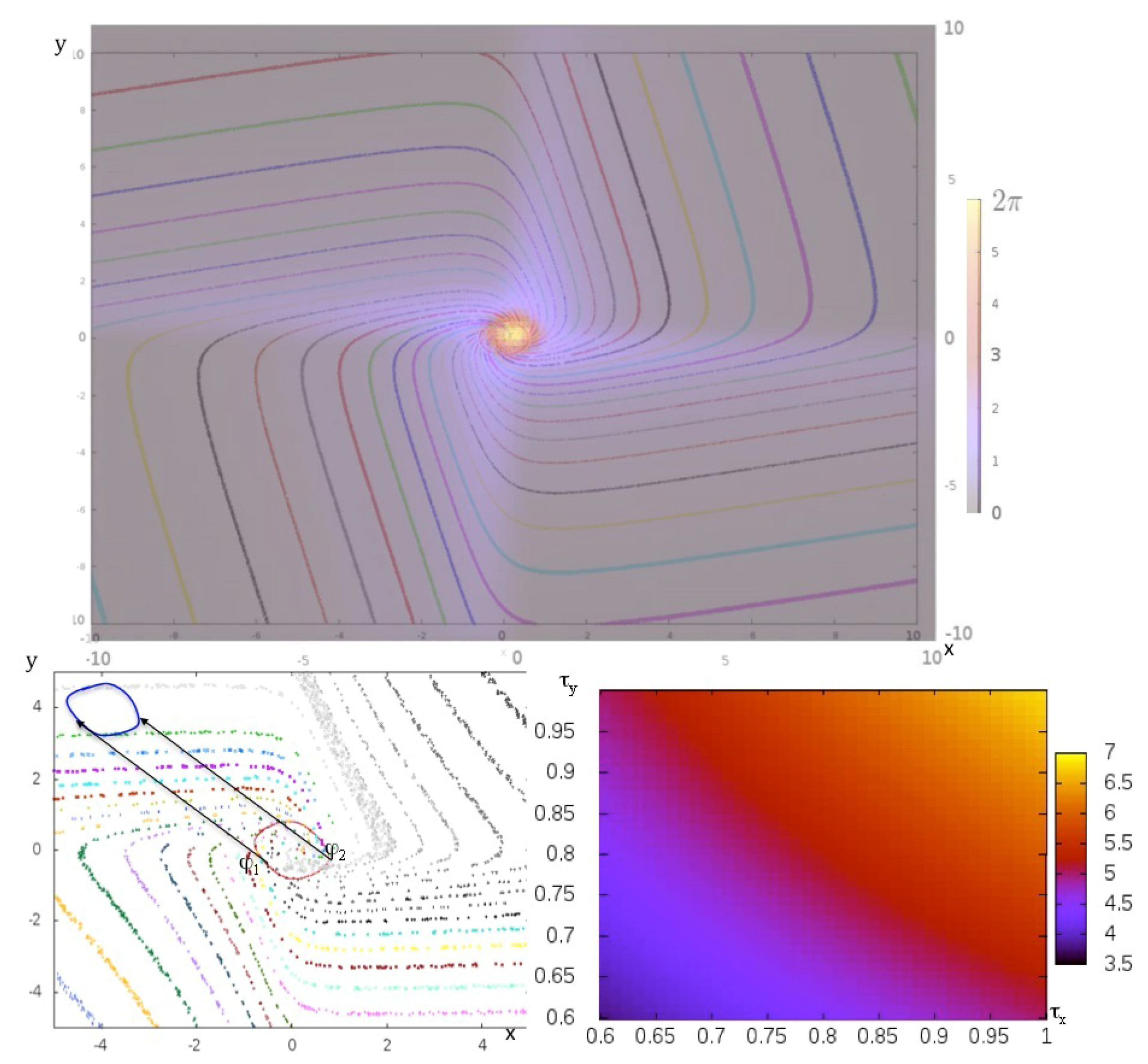
2.1.2. Potential Dynamics
2.1.3. Hamiltonian Dynamics
2.1.4. Potential-Hamiltonian Dynamics
2.2. Examples of Dissipative Energy
2.2.1. Discretization of the Continuous Potential System with Block-Parallel Updating
- interaction matrix W: represents the action of the gene j on gene i. W plays the role of a discrete Jacobian matrix. We can consider the associated signed matrix S, with , if , , if and , if ,
- updating matrix U: , if j is updated before or with i, else ,
- trajectory matrix F: , where b and c are two states of E, if and only if , else .
- in the case of W, the most frequent dependence is called the Hebbian dynamics: if the vectors and have a correlation coefficient , then , with , corresponding to a reinforcement of the absolute value of the interactions having succeeded in inhibiting or activating their target gene i: in case where, for , remained equal to one, that leads to increase the ’s, if the ’s were positive, and conversely to decrease the ’s , if the ’s were negative,
- in the case of U, we can have an autonomous (in time) clock based on the behaviour of r chromatin clock genes having indices , with three possible behaviours:
- if , then the rule (1) is available
- if and , where is the last time between and t, where
- if and , then (by exhaustion of the pool of expressed genes).
- (i)
- there are two chromatin clock genes involved in a regulon, i.e., the minimal network having only one negative circuit and one positive circuit reduced to an autocatalytic loop
- (ii)
- there are three morphogens corresponding to the apex and to axillary buds involved as nodes of a 3-switch network fully connected, with all interactions negative except three autocatalytic loops at each vertex
- (iii)
- there are three inhibitory interactions from the auto-catalysed node of the regulon on the nodes of the 3-switch, as indicated on Figure 2 Top left.
2.2.2. Discrete Lyapunov Function (Neural Network)
3. Results
3.1. Attractor Entropy and Neural Networks
3.2. Dynamic Entropy and Genetic Networks
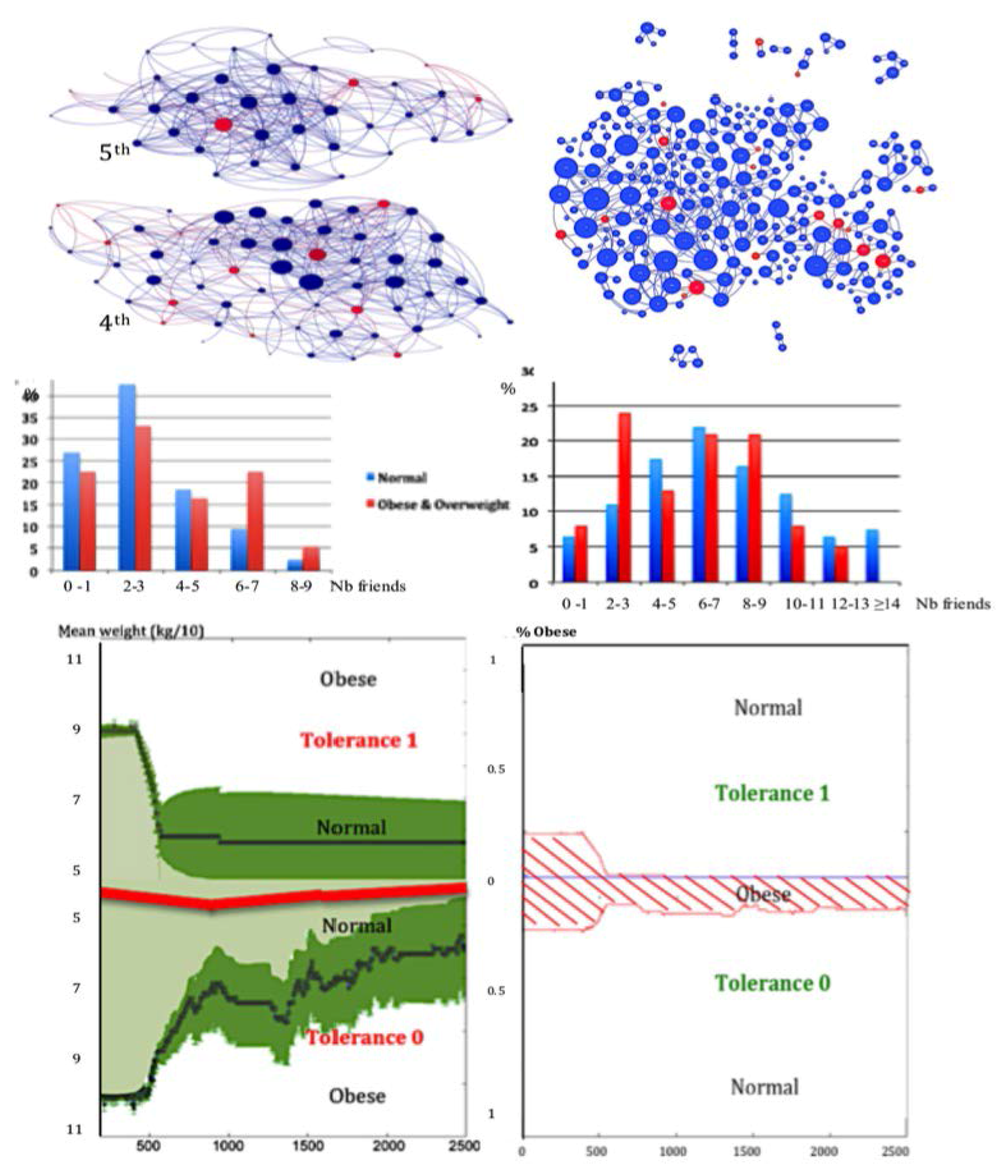
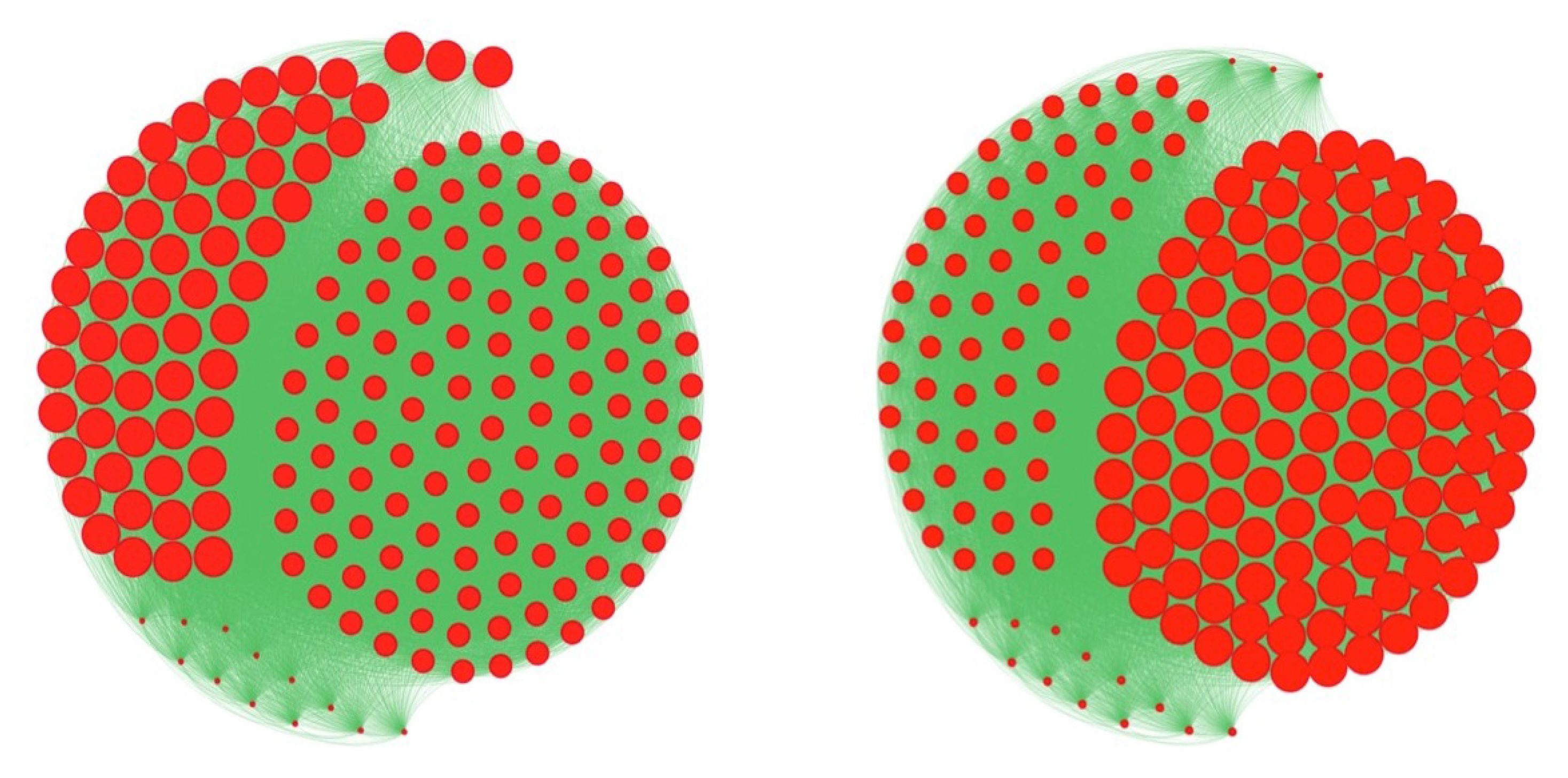
3.3. Centrality of Nodes and Social Networks
4. Discussion
4.1. The Notion of Entropy Centrality
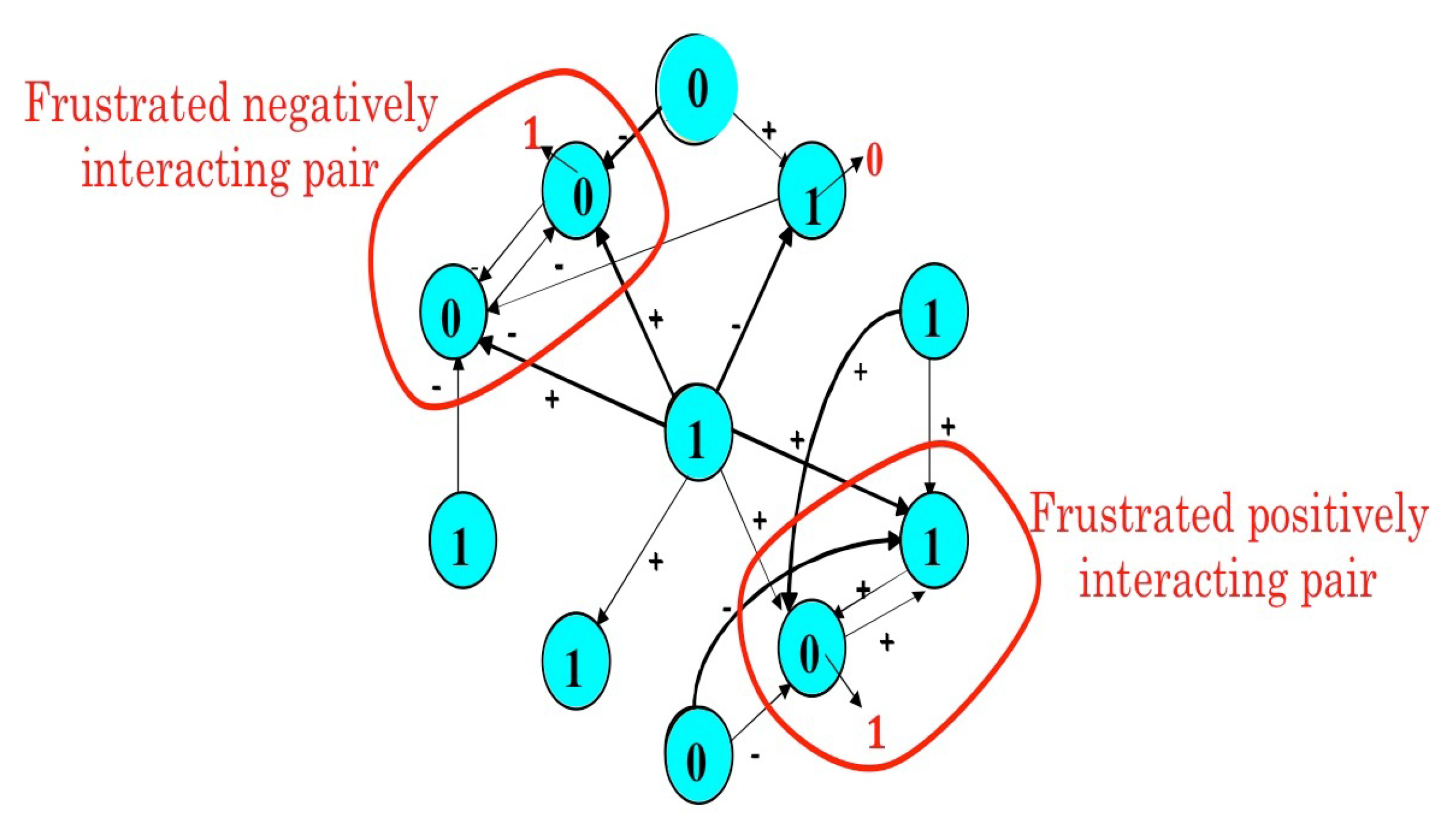
4.2. The Mathematical Problem of Robustness and the Notion of Global Frustration
5. Conclusions and Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mason, O.; Verwoerd, M. Graph theory and networks in biology. IET Syst. Biol. 2007, 1, 89–119. [Google Scholar] [CrossRef] [PubMed]
- Gosak, M.; Markovič, R.; Dolenšek, J.; Rupnik, M.S.; Marhl, M.; Stožer, A.; Perc, M. Network Science of Biological Systems at Different Scales: A Review. Phys. Life Rev. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kurz, F.T.; Kembro, J.M.; Flesia, A.G.; Armoundas, A.A.; Cortassa, S.; Aon, M.A.; Lloyd, D. Network dynamics: Quantitative analysis of complex behavior in metabolism, organelles, and cells, from experiments to models and back. Wiley Interdiscip. Rev. Syst. Biol. Med. 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Christakis, N.A.; Fowler, J.H. The spread of obesity in a large social network over 32 years. N. Engl. J. Med. 2007, 2007, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Demongeot, J.; Jelassi, M.; Taramasco, C. From susceptibility to frailty in social networks: The case of obesity. Math. Popul. Stud. 2017, 24, 219–245. [Google Scholar] [CrossRef]
- Federer, C.; Zylberberg, J. A self-organizing memory network. BioRxiv 2017. [Google Scholar] [CrossRef]
- Grillner, S. Biological pattern generation: the cellular and computational logic of networks in motion. Neuron 2006, 52, 751–766. [Google Scholar] [CrossRef] [PubMed]
- Demongeot, J.; Benaouda, D.; Jezequel, C. “Dynamical confinement” in neural networks and cell cycle. Chaos Interdiscip. J. Nonlinear Sci. 1995, 5, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Erdös, P.; Rényi, A. On random graphs. Pub. Math. 1959, 6, 290–297. [Google Scholar]
- Barabási, A.L.; Albert, R. Emergence of scaling in random networks. Science 1959, 286, 509–512. [Google Scholar]
- Albert, R.; Jeong, H.; Barabási, A.L. Error and attack tolerance of complex networks. Nature 2000, 406, 378–382. [Google Scholar]
- Demetrius, L. Thermodynamics and evolution. J. Theor. Biol. 2000, 206, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.; Mason, S.P.; Barabási, A.L.; Oltvai, Z.N. Lethality and centrality in protein networks. Nature 2001, 411, 41–42. [Google Scholar] [CrossRef] [PubMed]
- Demetrius, L.; Manke, T. Robustness and network evolution—An entropic principle. Phys. A Stat. Mech. Appl. 2005, 346, 682–696. [Google Scholar] [CrossRef]
- Manke, T.; Demetrius, L.; Vingron, M. An entropic characterization of protein interaction networks and cellular robustness. J. R. Soc. Interface 2006, 3, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Gardeñes, J.; Latora, V. Entropy rate of diffusion processes on complex networks. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2008, 78, 065102. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Bing-Hong, W.; Wen-Xu, W.; Tao, Z. Network entropy based on topology configuration and its computation to random networks. Chin. Phys. Lett. 2008, 25, 4177–4180. [Google Scholar] [CrossRef]
- Teschendorff, A.E.; Severini, S. Increased entropy of signal transduction in the cancer metastasis phenotype. BMC Syst. Biol. 2010, 4, 104. [Google Scholar] [CrossRef] [PubMed]
- West, J.; Bianconi, G.; Severini, S.; Teschendorff, A. Differential network entropy reveals cancer system hallmarks. Sci. Rep. 2012, 2. [Google Scholar] [CrossRef] [PubMed]
- Banerji, C.R.; Miranda-Saavedra, D.; Severini, S.; Widschwendter, M.; Enver, T.; Zhou, J.X.; Teschendorff, A.E. Cellular network entropy as the energy potential in Waddington’s differentiation landscape. Sci. Rep. 2013, 3, 3039. [Google Scholar] [CrossRef] [PubMed]
- Teschendorff, A.E.; Sollich, P.; Kuehn, R. Signalling entropy: A novel network-theoretical framework for systems analysis and interpretation of functional omic data. Methods 2014, 67, 282–293. [Google Scholar] [CrossRef] [PubMed]
- Teschendorff, A.E.; Banerji, C.R.S.; Severini, S.; Kuehn, R.; Sollich, P. Increased signaling entropy in cancer requires the scale-free property of protein interaction networks. Sci. Rep. 2015, 2, 802. [Google Scholar] [CrossRef] [PubMed]
- Toulouse, G. Theory of the frustration effect in spin glasses: I. Commun. Phys. 1977, 2, 115–119. [Google Scholar]
- Robert, F. Discrete Iterations: A Metric Study; Springer: Berlin, Germany, 1986; Volume 6. [Google Scholar]
- Cosnard, M.; Demongeot, J. Attracteurs: Une approche déterministe. C. R. Acad. Sci. Ser. I Math. 1985, 300, 551–556. [Google Scholar]
- Cosnard, M.; Demongeot, J. On the definitions of attractors. Lect. Notes Comput. Sci. 1985, 1163, 23–31. [Google Scholar]
- Szabó, K.G.; Tél, T. Thermodynamics of attractor enlargement. Phys. Rev. E 1994, 50, 1070–1082. [Google Scholar] [CrossRef]
- Wang, J.; Xu, L.; Wang, E. Potential landscape and flux framework of nonequilibrium networks: Robustness, dissipation, and coherence of biochemical oscillations. Proc. Natl. Acad. Sci. USA 2008, 105, 12271–12276. [Google Scholar] [CrossRef] [PubMed]
- Menck, P.J.; Heitzig, J.; Marwan, N.; Kurths, J. How basin stability complements the linear-stability paradigm. Nat. Phys. 2013, 9, 89–92. [Google Scholar] [CrossRef]
- Bowen, R. ω-limit sets for axiom A diffeomorphisms. J. Differ. Equ. 1975, 18, 333–339. [Google Scholar] [CrossRef]
- Williams, R. Expanding attractors. Publ. Math. l’Inst. Hautes Études Sci. 1974, 43, 169–203. [Google Scholar] [CrossRef]
- Ruelle, D. Small random perturbations of dynamical systems and the definition of attractors. Commun. Math. Phys. 1981, 82, 137–151. [Google Scholar] [CrossRef]
- Ruelle, D. Small random perturbations and the definition of attractors. Geom. Dyn. 1983, 82, 663–676. [Google Scholar]
- Haraux, A. Attractors of asymptotically compact processes and applications to nonlinear partial differential equations. Commun. Part. Differ. Equ. 1988, 13, 1383–1414. [Google Scholar] [CrossRef]
- Hale, J. Asymptotic behavior of dissipative systems. Bull. Am. Math. Soc. 1990, 22, 175–183. [Google Scholar]
- Audin, M. Hamiltonian Systems and Their Integrability; American Mathematical Society: Providence, RI, USA, 2008. [Google Scholar]
- Demongeot, J.; Glade, N.; Forest, L. Liénard systems and potential-Hamiltonian decomposition I–methodology. C. R. Math. 2007, 344, 121–126. [Google Scholar] [CrossRef]
- Waddington, C. Organizers and Genes; Cambridge University Press: Cambridge, UK, 1940. [Google Scholar]
- Demongeot, J.; Thomas, R.; Thellier, M. A mathematical model for storage and recall functions in plants. C. R. Acad. Sci. Ser. III 2000, 323, 93–97. [Google Scholar] [CrossRef]
- Thellier, M.; Demongeot, J.; Norris, V.; Guespin, J.; Ripoll, C.; Thomas, R. A logical (discrete) formulation for the storage and recall of environmental signals in plants. Plant Biol. 2004, 6, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R. Boolean formalization of genetic control circuits. J. Theor. Biol. 1973, 42, 563–585. [Google Scholar] [CrossRef]
- Thomas, R. On the relation between the logical structure of systems and their ability to generate multiple steady states or sustained oscillations. Springer Ser. Synerg. 1981, 9, 180–193. [Google Scholar]
- Tonnelier, A.; Meignen, S.; Bosch, H.; Demongeot, J. Synchronization and desynchronization of neural oscillators. Neural Netw. 1999, 12, 1213–1228. [Google Scholar] [CrossRef]
- Demongeot, J.; Kaufman, M.; Thomas, R. Positive feedback circuits and memory. C. R. Acad. Sci. Ser. III 2000, 323, 69–79. [Google Scholar] [CrossRef]
- Ben Amor, H.; Glade, N.; Lobos, C.; Demongeot, J. The isochronal fibration: Characterization and implication in biology. Acta Biotheor. 2010, 58, 121–142. [Google Scholar] [CrossRef] [PubMed]
- Demongeot, J.; Hazgui, H.; Ben Amor, H.; Waku, J. Stability, complexity and robustness in population dynamics. Acta Biotheor. 2014, 62, 243–284. [Google Scholar] [CrossRef] [PubMed]
- Demongeot, J.; Jezequel, C.; Sené, S. Asymptotic behavior and phase transition in regulatory networks. I. Theoretical results. Neural Netw. 2008, 21, 962–970. [Google Scholar] [CrossRef] [PubMed]
- Demongeot, J.; Elena, A.; Sené, S. Robustness in neural and genetic networks. Acta Biotheor. 2008, 56, 27–49. [Google Scholar] [CrossRef] [PubMed]
- Demongeot, J.; Ben Amor, H.; Elena, A.; Gillois, P.; Noual, M.; Sené, S. Robustness in regulatory interaction networks. A generic approach with applications at different levels: physiologic, metabolic and genetic. Int. J. Mol. Sci. 2009, 10, 4437–4473. [Google Scholar] [CrossRef] [PubMed]
- Demongeot, J.; Elena, A.; Noual, M.; Sené, S.; Thuderoz, F. “Immunetworks”, intersecting circuits and dynamics. J. Theor. Biol. 2011, 280, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Demongeot, J.; Waku, J. Robustness in biological regulatory networks I: Mathematical approach. C. R. Math. 2012, 350, 221–224. [Google Scholar] [CrossRef]
- Demongeot, J.; Cohen, O.; Henrion-Caude, A. MicroRNAs and robustness in biological regulatory networks. A generic approach with applications at different levels: physiologic, metabolic, and genetic. In Systems Biology of Metabolic and Signaling Networks; Springer: Berlin, Germany, 2013; pp. 63–114. [Google Scholar]
- Demongeot, J.; Demetrius, L. Complexity and stability in biological systems. Int. J. Bifurc. Chaos 2015, 25, 1540013. [Google Scholar] [CrossRef]
- Demongeot, J.; Jacob, C. Confineurs: une approche stochastique des attracteurs. C. R. Acad. Sci. Ser. I Math. 1989, 309, 699–702. [Google Scholar]
- Demetrius, L. Statistical mechanics and population biology. J. Stat. Phys. 1983, 30, 709–753. [Google Scholar] [CrossRef]
- Demetrius, L.; Demongeot, J. A Thermodynamic approach in the modeling of the cellular-cycle. Biometrics 1984, 40, 259–260. [Google Scholar]
- Demetrius, L. Directionality principles in thermodynamics and evolution. Proc. Natl. Acad. Sci. USA 1997, 94, 3491–3498. [Google Scholar] [CrossRef] [PubMed]
- Demetrius, L.; Gundlach, V.; Ochs, G. Complexity and demographic stability in population models. Theor. Popul. Biol. 2004, 65, 211–225. [Google Scholar] [CrossRef] [PubMed]
- Demetrius, L.; Ziehe, M. Darwinian fitness. Theor. Popul. Biol. 2007, 72, 323–345. [Google Scholar] [CrossRef] [PubMed]
- Demongeot, J.; Sené, S. Asymptotic behavior and phase transition in regulatory networks. II Simulations. Neural Netw. 2008, 21, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Delbrück, M. Discussion au Cours du Colloque International sur les Unités Biologiques Douées de Continuité Génétique; CNRS: Paris, France, 1949; Volume 8, pp. 33–35. [Google Scholar]
- Cinquin, O.; Demongeot, J. Positive and negative feedback: Striking a balance between necessary antagonists. J. Theor. Biol. 2002, 216, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Barthelemy, M. Betweenness centrality in large complex networks. Eur. Phys. J. B Conden. Matter Complex Syst. 2004, 38, 163–168. [Google Scholar] [CrossRef]
- Myers, A.; Rosen, J.C. Obesity stigmatization and coping: Relation to mental health symptoms, body image, and self-esteem. Int. J. Obes. Related Metab.Disord. 1999, 23. [Google Scholar] [CrossRef]
- Cohen-Cole, E.; Fletcher, J.M. Is obesity contagious? Social networks vs. environmental factors in the obesity epidemic. J. Health Econ. 2008, 27, 1382–1387. [Google Scholar] [CrossRef] [PubMed]
- Cauchi, D.; Gauci, D.; Calleja, N.; Marsh, T.; Webber, L. Application of the UK foresight obesity model in Malta: Health and economic consequences of projected obesity trend. Obes. Facts 2015, 8, 141. [Google Scholar]
- Jones, R.; Breda, J. Surveillance of Overweight including Obesity in Children Under 5: Opportunities and Challenges for the European Region. Obes. Facts 2015, 8, 124. [Google Scholar]
- Shaw, A.; Retat, L.; Brown, M.; Divajeva, D.; Webber, L. Beyond BMI: projecting the future burden of obesity in England using different measures of adiposity. Obes. Facts 2015, 8, 135–136. [Google Scholar]
- Demongeot, J.; Elena, A.; Taramasco, C. Social Networks and Obesity. Application to an interactive system for patient-centred therapeutic education. In Metabolic Syndrome: A Comprehensive Textbook; Ahima, R.S., Ed.; Springer: New York, NY, USA, 2015; pp. 287–307. [Google Scholar]
- Breda, J. WHO plans for action on primary prevention of obesity. Obes. Facts 2015, 8, 17. [Google Scholar]
- Hopfield, J. Neural networks and physical systems with emergent collective computational abilities. Proc. Natl. Acad. Sci. USA 1982, 79, 2554–2558. [Google Scholar] [CrossRef] [PubMed]
- Gardner, M.; Ashby, W. Connectance of large dynamic (cybernetic) systems: Critical values for stability. Nature 1970, 228, 784. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A. Robustness and evolvability in living systems. In Princeton Studies in Complexity; Princeton University Press: Princeton, NJ, USA, 2005. [Google Scholar]
- Gunawardena, S. The robustness of a biochemical network can be inferred mathematically from its architecture. Biol. Syst. Theory 2010, 328, 581–582. [Google Scholar]
- Demongeot, J.; Noual, M.; Sené, S. Combinatorics of Boolean automata circuits dynamics. Discret. Appl. Math. 2012, 160, 398–415. [Google Scholar] [CrossRef]
- Demongeot, J.; Demetrius, L. La derivé démographique et la selection naturelle: Étude empirique de la France (1850–1965). Population 1989, 2, 231–248. [Google Scholar]
- Demongeot, J.; Goles, E.; Morvan, M.; Noual, M.; Sené, S. Attraction basins as gauges of robustness against boundary conditions in biological complex systems. PLoS ONE 2010, 5, e11793. [Google Scholar] [CrossRef] [PubMed]
- Ventsell, A.; Freidlin, M. On small random perturbations of dynamical systems. Russ. Math. Surv. 1970, 25, 1–55. [Google Scholar] [CrossRef]
- Young, L. Some Large Deviation Results for Dynamical Systems. Trans. Am. Math. Soc. 1990, 318, 525–543. [Google Scholar] [CrossRef]
- Weaver, D.; Workman, C.; Stormo, G. Modeling regulatory networks with weight matrices. Proc. Pac. Symp. Biocomput. 1999, 4, 112–123. [Google Scholar]

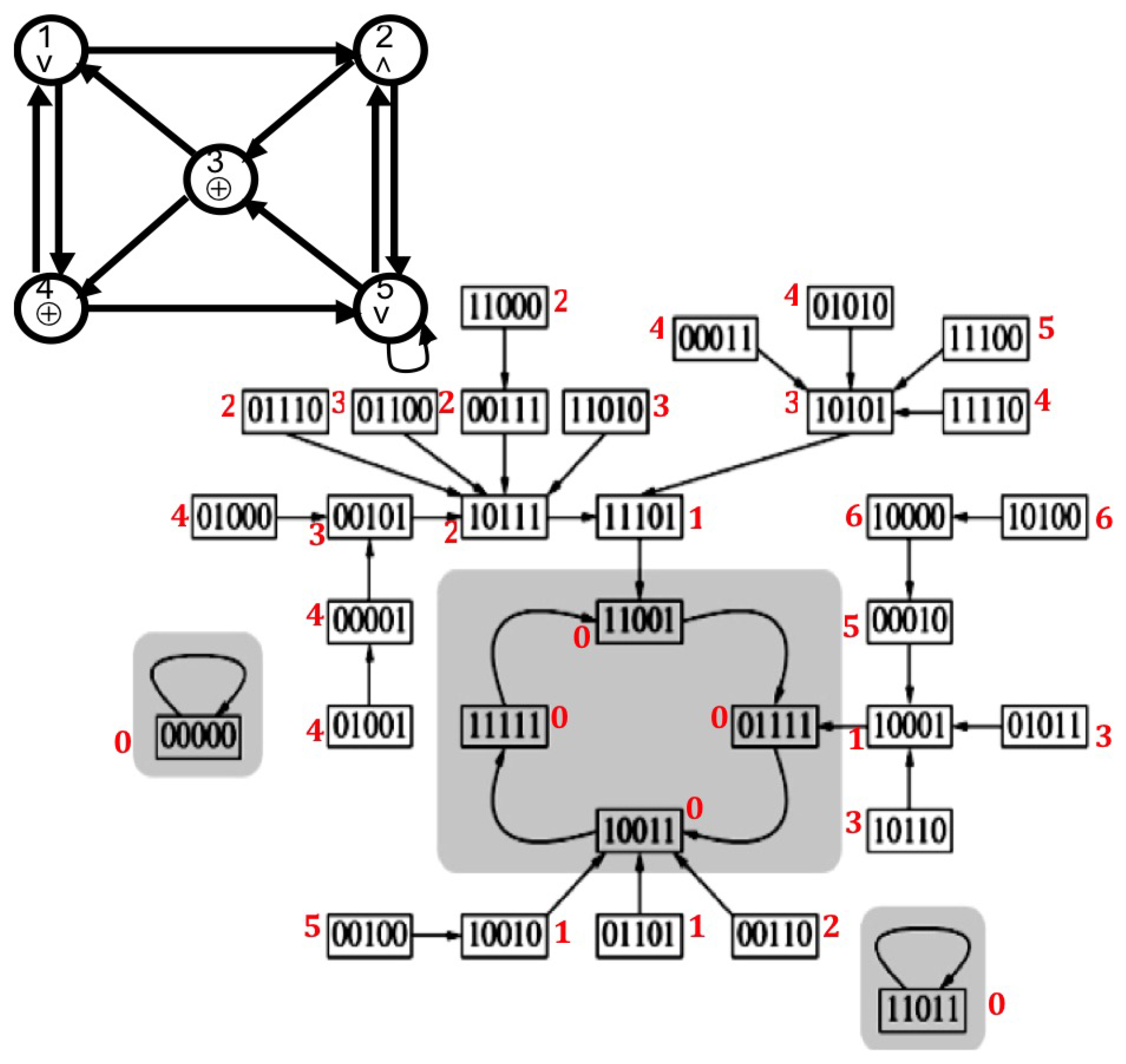
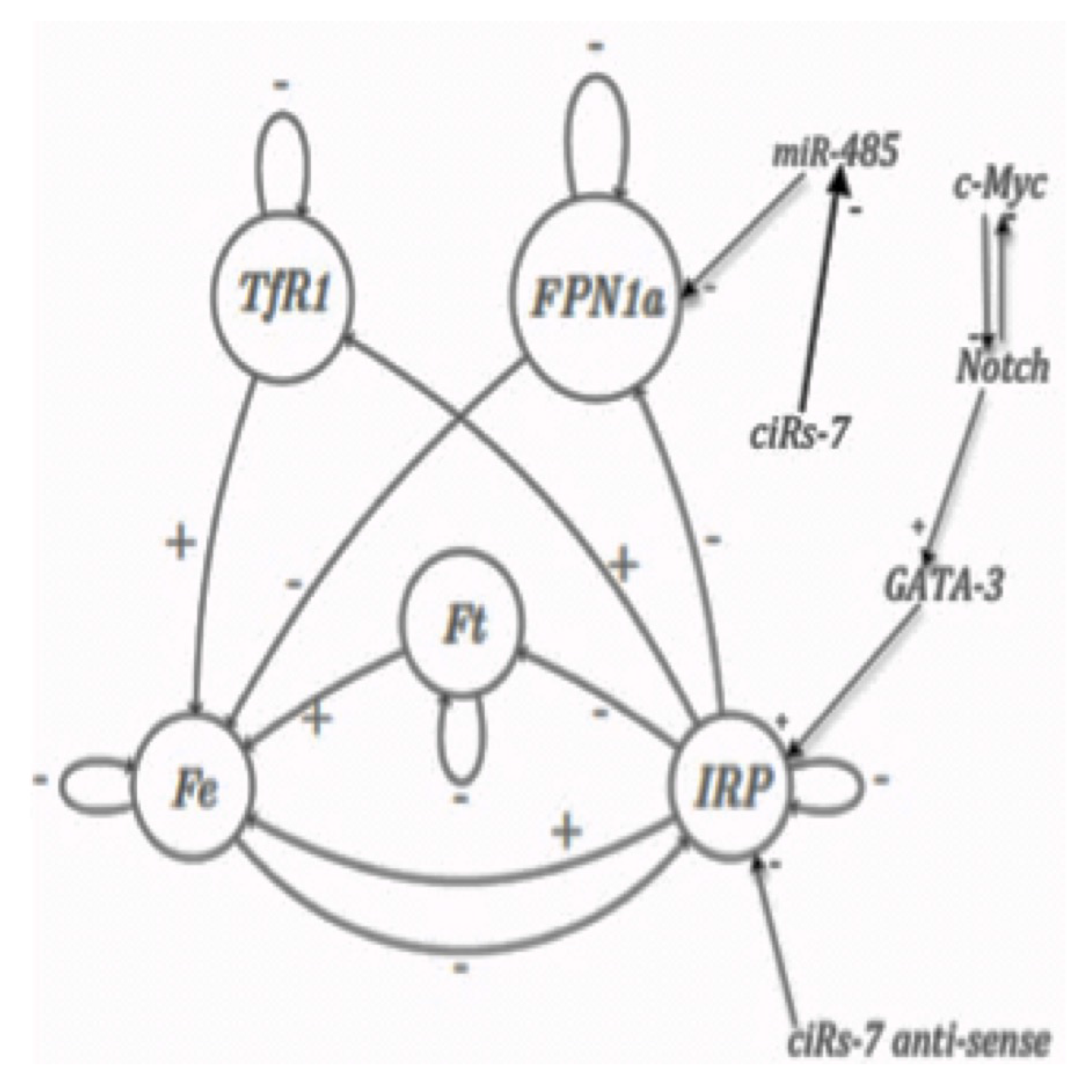


| Order | Gene | Fixed Point | Fixed Point 2 | Limit-Cycle 1 | Limit-Cycle 2 | |||
|---|---|---|---|---|---|---|---|---|
| 1 | TfR1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 |
| 2 | FPN1a | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 3 | C-Myc | 0 | 1 | 0 | 0 | 0 | 0 | 0 |
| 4 | Notch | 0 | 0 | 1 | 1 | 1 | 1 | 1 |
| 5 | GATA-3 | 0 | 0 | 1 | 1 | 1 | 1 | 1 |
| 6 | IRP | 0 | 0 | 0 | 1 | 0 | 0 | 1 |
| 7 | Ft | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 8 | Fe | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| 9 | MiR-485 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10 | CiRs-7 anti-sense | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Relative Attraction Basin Size | 512/1024 | 256/1024 | 216/1024 | 40/1024 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demongeot, J.; Jelassi, M.; Hazgui, H.; Ben Miled, S.; Bellamine Ben Saoud, N.; Taramasco, C. Biological Networks Entropies: Examples in Neural Memory Networks, Genetic Regulation Networks and Social Epidemic Networks. Entropy 2018, 20, 36. https://doi.org/10.3390/e20010036
Demongeot J, Jelassi M, Hazgui H, Ben Miled S, Bellamine Ben Saoud N, Taramasco C. Biological Networks Entropies: Examples in Neural Memory Networks, Genetic Regulation Networks and Social Epidemic Networks. Entropy. 2018; 20(1):36. https://doi.org/10.3390/e20010036
Chicago/Turabian StyleDemongeot, Jacques, Mariem Jelassi, Hana Hazgui, Slimane Ben Miled, Narjes Bellamine Ben Saoud, and Carla Taramasco. 2018. "Biological Networks Entropies: Examples in Neural Memory Networks, Genetic Regulation Networks and Social Epidemic Networks" Entropy 20, no. 1: 36. https://doi.org/10.3390/e20010036
APA StyleDemongeot, J., Jelassi, M., Hazgui, H., Ben Miled, S., Bellamine Ben Saoud, N., & Taramasco, C. (2018). Biological Networks Entropies: Examples in Neural Memory Networks, Genetic Regulation Networks and Social Epidemic Networks. Entropy, 20(1), 36. https://doi.org/10.3390/e20010036







