Sample Entropy and Traditional Measures of Heart Rate Dynamics Reveal Different Modes of Cardiovascular Control During Low Intensity Exercise
Abstract
: Nonlinear parameters of heart rate variability (HRV) have proven their prognostic value in clinical settings, but their physiological background is not very well established. We assessed the effects of low intensity isometric (ISO) and dynamic (DYN) exercise of the lower limbs on heart rate matched intensity on traditional and entropy measures of HRV. Due to changes of afferent feedback under DYN and ISO a distinct autonomic response, mirrored by HRV measures, was hypothesized. Five-minute inter-beat interval measurements of 43 healthy males (26.0 ± 3.1 years) were performed during rest, DYN and ISO in a randomized order. Blood pressures and rate pressure product were higher during ISO vs. DYN (p < 0.001). HRV indicators SDNN as well as low and high frequency power were significantly higher during ISO (p < 0.001 for all measures). Compared to DYN, sample entropy (SampEn) was lower during ISO (p < 0.001). Concluding, contraction mode itself is a significant modulator of the autonomic cardiovascular response to exercise. Compared to DYN, ISO evokes a stronger blood pressure response and an enhanced interplay between both autonomic branches. Non-linear HRV measures indicate a more regular behavior under ISO. Results support the view of the reciprocal antagonism being only one of many modes of autonomic heart rate control. Under different conditions; the identical “end product” heart rate might be achieved by other modes such as sympathovagal co-activation as well.1. Introduction
Biological time series like the normal heartbeat-to-heartbeat fluctuation demonstrate complex dynamics [1–4]. Based on their potential to give additional information beyond traditional heart rate variability (HRV) indices [1], nonlinear parameters have been applied for investigating short and long term effects of exercise on heart rate (HR) control [5–8]. However, despite their diagnosticity and their clinical significance [9–16], the physiological background of their behavior is not very well established. It is assumed that complexity and regularity measures are fundamentally different from traditional HRV indices [17] and show no correlation to these measures [12,18]. However, many researchers found at least modest correlations for some nonlinear measures and traditional HRV indices under different conditions [5,9,19,20]. It has also been shown that complexity of short-term HRV is under control of the autonomic nervous system [21,22]. Currently, there are only few studies available that compared the cardiovascular response pattern to different exercise modes at similar HR. Lindquist et al. found a stronger increase of systolic (SBP) and diastolic arterial blood pressure (DBP) during isometric handgrip compared to cycling at comparable HR of 90 bpm [23]. Leicht and his associates compared the cardiovascular response to dynamic muscular activity of different muscle groups at 50% maximum HR (HRmax) and 65% HRmax, respectively. They have found greater HRV despite lower oxygen consumption during upper body dynamic exercise compared to lower or whole body dynamic exercise at similar HR and concluded that greater HRV may represent increased vagal or dual autonomic modulation [24]. Cottin et al. compared HRV indices during a judo randori vs. ergometer cycling eliciting the same HR level [25]. Assumptions drawn by these authors were, that steady-state dynamic exercise or conversely exercise made of both isometric and irregular dynamic efforts can be distinguished by HRV spectral analysis. They further concluded that autonomic control of the heart during exercise depends rather on HR level than on the mode of exercise [25]. However, due to the intense exercise in this study, with an average HR above 180 bpm, conclusions regarding the autonomic mode of HR control based on spectral analyses of HRV are strongly limited. HRV at greater HR-levels is often almost negligible and the remaining variance, especially within the high frequency band (HF, 0.15–0.4 Hz), is probably due to non-neural mechanisms [26–28]. Furthermore, as the location and size of the active muscles during cycling and judo exercises are different, the influence of contraction mode on HR control remains to be investigated. Princi et al. found different autonomic modes of cardiac control based on HRV-analysis when comparing sailing and cycling at similar HR. The authors found a different sympatho-vagal modulation of cardiac function under different exercise modes [8]. The generalizability of their finding is questionable, because only one athlete was investigated, and the muscle groups engaged were not similar during both exercises. The aim of this study was to test the diagnostic potential of the HRV sample entropy (SampEn) in distinguishing different types of exercise. Due to a different ergoreceptor feedback from the working muscle, we hypothesized a qualitatively different autonomic cardiovascular control. This difference should be mirrored by a different regularity of the heartbeat series, which might not be revealed by traditional HRV indices. Further, the correlation of SampEn with traditional HRV indices was tested to prove its additional value in the exercise related cardiovascular analysis.
2. Methods
This study was performed in compliance with the Declaration of Helsinki. Approval of the local ethics committee at the University of Rostock was obtained. Forty three healthy males were recruited by personal invitation and gave their informed written consent to take part in this study. Table 1 shows age and anthropometric characteristics of the participants. All volunteers were physically active and healthy and none of them took medication. They abstained from any exhaustive exercise and alcohol for 24 h prior to the experiment. Further, the consumption of caffeine or nicotine was not allowed during the night and on the morning of the experiment [29].
Inter-beat (R-R) intervals of the participants were measured at rest (REST) and during two exercise sessions of five minutes: dynamic contractions (DYN) and isometric contraction (ISO) of the lower limbs. Data presented here, were pooled from two single experiments. The first experiment consisted of two-legged DYN and ISO exercise. The experimental setup is described elsewhere [7]. The second experiment consisted of one-legged DYN and ISO of the right M. quadriceps femoris using a CYBEX NORM dynamometer (Computer Sports Medicine®, Inc., Stoughton, MA, USA). DYN and ISO were performed in a randomized order. Between the respective exercise sessions a recovery phase not less than 10 min served as a washout period to prevent carryover effects. Exercise intensity of the first exercise treatment (DYN or ISO, respectively) was regulated to reach a significant, but moderately increased HR steady state. Intensity of the following exercise session was regulated to match a similar HR level. Forreal-time HR monitoring and the measurement of R-R intervals a Polar® HR monitor with an accuracy of one millisecond [30,31] was used. All experimental periods included the measurement of SBP and DBP using the automatic blood pressure (BP) measuring device Bosotron 2, (Boso Inc., Jungingen, Germany) [32]. Mean arterial pressure (MAP) was calculated by (SBP + 2 × DBP)/3; rate pressure product (RPP), a measure of myocardial oxygen consumption, was calculated by SBP × HR. To ensure steady state conditions only the last three minutes of each session were analyzed [33]. HR (beat-by-beat) was averaged for the three minutes. BP was measured in the last minute of each exercise session. A short term HRV-analysis was performed for three-minute beat-to-beat (R-R) interval segments during steady-state conditions. R-R series were processed using the free software Kubios HRV 2.1 (University of Kuopio, Kuopio, Finland). All analyzed R-R time series exhibited low noise (rate of erroneous R-R intervals below 5%). Before the computation, R-R time series were corrected for artifacts, using adaptive filtering, and detrended (detrending method: smoothn priors, λ = 500). Time domain measures SDNN and RMSSD, spectral power in the low (0.04–0.15 Hz, LFP) and high (0.15–0.4 Hz, HFP) frequency range and their relative values (% HF = HFP/TP (total spectral power) and % LF = LFP/TP). HFP is predominantly modulated by vagal outflow to the heart whereas LFP is supposed to be modulated by both autonomic branches. % LF is thought to reflect the sympathetic drive to the heart [34–36]. SampEn was used to assess the complexity of the HR “signal” under the different conditions [37–39]. SampEn measures the likelihood that runs of patterns that are close to each other will remain close in the next incremental comparisons [38,39]. Its calculation relies on counts of m-long templates matching within a tolerance r that also match at the next point. For SampEn calculation the value of m was selected to be m = 2, for tolerance r a fraction of the standard deviation of the R-R data (r = 0.2 * SDNN) was chosen [38]. Low entropy values arise from extremely regular time series, higher values reflect more complexity, and highest values are typical for stochastic data sets [40–42].
Analysis of variance (ANOVA) for repeated measures was used to test for a significant effect of the condition (REST, DYN and ISO) on BP, RPP, HR, SampEn and traditional HRV indices. If data violated the assumption of sphericity, Greenhouse-Geisser corrected significance levels and respective degrees of freedom were reported. If data sets violated the assumption of normal distribution, values were natural log-transformed before the statistical analysis. Significance levels of the post-hoc pair wise comparisons were adjusted using Bonferroni’s procedure.
3. Results
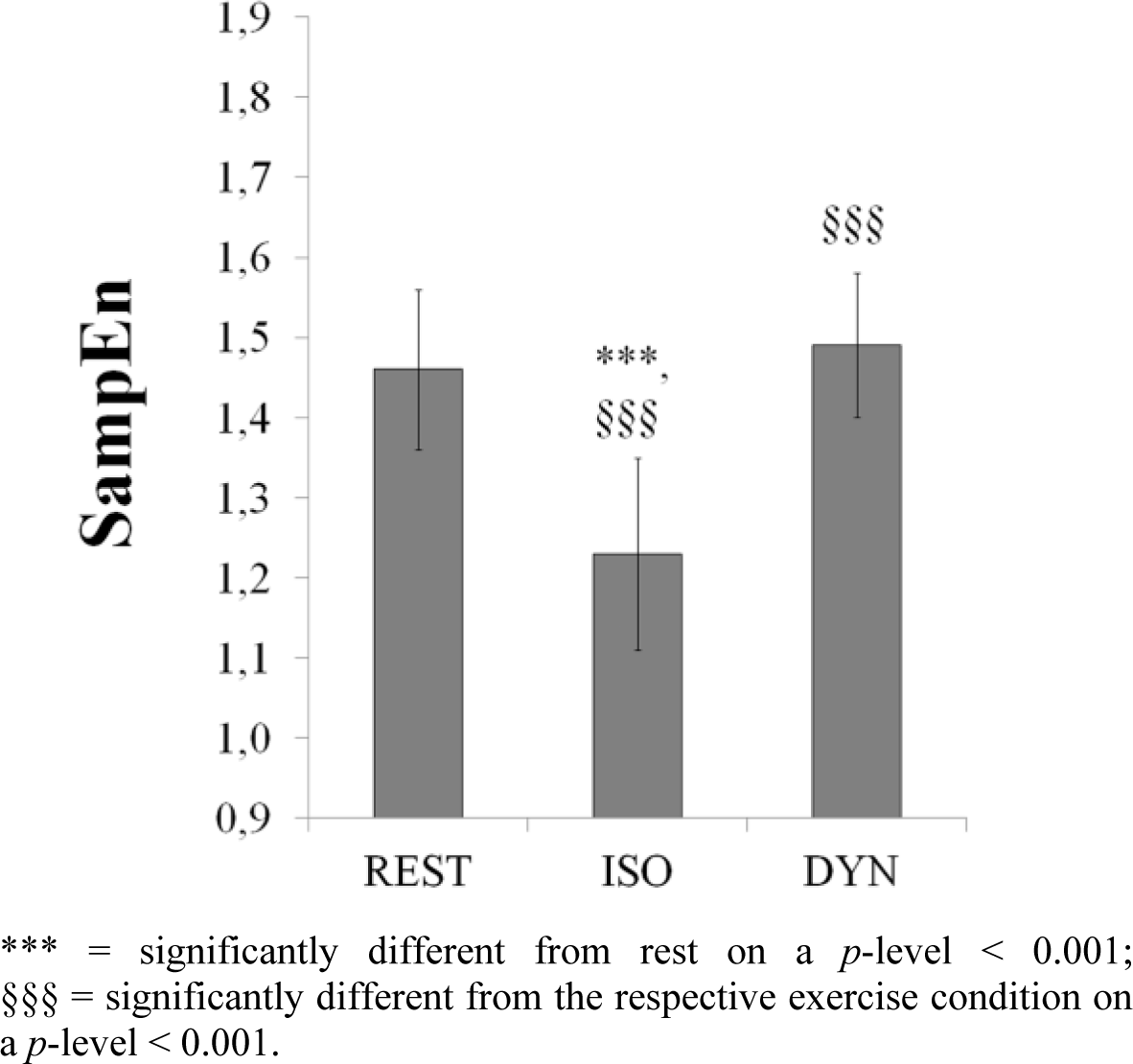
ANOVA revealed a significant effect of experimental condition on all cardiovascular measures and autonomic indices (Table 2). Average HR raised moderately from 65 ± 9 bpm at baseline to 85 ± 9 bpm during both types of exercise. HR during the first exercise perfectly matched HR of the subsequent exercise; average difference was only 0.3 ± 1.5 bpm (range: −2.6 to 4.3 bpm). Accordingly, HR and average R-R interval did not differ between DYN and ISO. The traditional vagal modulation HRV measure RMSSD was also not affected by the exercise mode, whereas SDNN was. Natural log-transformed HRV spectral indices HFP and LFP, the normalized powers LF n. u. and HF n. u. as well SampEn (Figure 1) were significantly different between DYN and ISO. Interestingly, SampEn did not differ between REST and DYN. There was no difference of the LF/HF ratio between REST and ISO, whereas comparison of REST vs. DYN showed a statistical trend (p = 0.077). Further, there was a small effect of condition on the HF peak frequency (F(2; 84) = 4.959, p < 0.01, η2 = 0.106). While HF peak significantly shifted from 0.22 ± 0.07 Hz during REST to 0.26 ± 0.09 Hz during DYN (p < 0.05), no difference was found between REST and ISO (0.23 ± 0. 07 Hz). Post-hoc pair wise comparison between DYN and ISO showed a statistical trend for the HF peak shift (p = 0.063). SBP and RPP were moderately, DBP and MAP largely affected by the type of exercise. In comparison to DYN, myocardial oxygen consumption, reflected by RPP, was about 5% higher under ISO [43,44]. Correlation analysis revealed only modest associations between traditional HRV indices and entropy measures during the different experimental conditions (Table 3). Consistent correlation coefficients across all conditions were found for SampEn and R-R length only.
4. Discussion
As expected, HR increased and traditional HRV indices decreased from baseline rest to exercise. However, HRV SampEn did not differ between REST and DYN but reached its minimum during ISO. Despite exercise eliciting the same HR level, SBP, DBP, MAP, RPP and HRV differed significantly between the two exercise modes. Compared to DYN, ISO was characterized by higher SBP and DBP, larger HRV HF- and LF-power and lower HRV complexity. Metabolite accumulation as well as mechanical stimuli in the isometrically working muscle (muscle metaboreflex) can lead to an enhanced vascular response [45–61]. This metaboreflex likely overrides the baroreflex, leading to a stronger sympathetic efferent activity to the vessels during ISO [46,47,53,62–66]. Beside the BP response, that seems to evidence increased sympathetic efferent drive to the vessels during ISO, there was a significant increase of the log-power in the LF and HF range, whereas the normalized spectral power values were similar between the exercise modes. Analysis of the LF/HF ratio showed that—in contrast to ISO—DYN led to a change in the sympathovagal balance in favor of the sympathetic branch if compared to REST. In addition to the significant differences of absolute spectral powers the increase of SDNN also demonstrates the distinct autonomic HR modulation under the different contraction modes. Together, these results speak for an increased dual autonomic cardiac modulation under ISO and supports evidence from recent studies [7,24,67]. Complementary to traditional HRV measures, entropy was significantly decreased during ISO, pointing towards an enhanced regularity of the heart beat series [37,40–42]. Porta and coworkers have found a significant reduction of SampEn during head-up tilting. Results indicated that a change of sympatho-vagal balance towards a sympathetic dominance increases regularity and thus reduces entropy values. However, from this experiment it cannot not be concluded whether vagal withdrawal and/or sympathetic drive to the heart is the main contributor of HR complexity. After one of the first attempts to elucidate the autonomic influence on HR entropy during autonomic blocking and exercise, Tulppo et al. concluded that sympathetic rather than vagal efferent activity modulates HR complexity [68]. In a recent experiment the group around Porta used refined methods to estimate HR complexity and an autonomic blocking protocol to elucidate the underlying autonomic mechanisms [22]. From their results the authors concluded that vagal efferent activity is the main contributor, because high dose of atropine significantly reduced complexity compared to baseline conditions, while sympathetic blocking with propranolol did not have any effect on HR complexity. However, the finding that the reduction in complexity after dual autonomic blocking was not that strong as after vagal blocking alone, might give some evidence for a sympathetic contribution to HR regularity as well. It can be speculated, that the lack of an additional increase of HR entropy after propranolol administration might be due to a saturation of the vagal HR modulation under supine rest [69]. Early work indicated that vagal withdrawal is the dominant mechanism during low intensity dynamic exercise [70]. Thus, it is not clear why SampEn in our study was similar between REST and DYN, if SampEn reflects changes in vagal HR modulation only. Further, we cautiously conclude from the results of the HRV frequency and BP analysis, that—beyond a decrease of vagal efferent activity (compared to REST)—also an increased sympathetic cardiac drive contributes to the reduction in HR complexity seen under ISO. Recent work also points to a sympathetic HR modulation even at the onset of exercise [71–73]. Porta et al. also found a reduction of HR complexity during static handgrip and suggested an increase of sympathetic efferent activity as a possible cause [74].
Further, low correlations with traditional HRV indices as well as the distinct behavior of SampEn across the three experimental conditions suggest that beyond autonomic influences other mechanisms might be involved in the modulation of HR complexity measures [6,68].
Summarizing, it appears likely that a change of mechanoreceptor afferent activity is a main contributor of the distinct autonomic circulatory responses to the different contraction modes. In an experiment, applying sustained passive stretching (comparable with ISO) and rhythmic passive stretching (comparable with DYN) of the right triceps surae muscle, Gladwell and Coote found distinct cardiovascular responses to these stretching modes. Continuous stretching activated type III and IV afferents and led to significant BP and HR increases, whereas rapid rhythmic stretch—activating large group I and II muscle afferents—did not [75]. Also other work has shown, that type III and IV mechanosensitive afferents contribute to the cardiovascular response to ISO, while type I and II fibers do not or only little [58,76]. As all participants reached HR steady states during both types of exercise, we conclude that there was no compromise in muscle blood flow during ISO as well. Thus, a different stimulation of mechanoreceptors rather than an increased chemoreceptor feedback from the working muscle might be involved in the distinct cardiovascular response pattern under low intensity exercise. An alternative or additional explanation for the increase in HRV spectral power and regularity under ISO might be an elevated sensitivity of the baroreflex [77–81]. The shift of the respiration-related HF peak from a higher frequency during DYN to a lower frequency during ISO indicates a reduction of the respiratory rate under ISO. This slowing of breathing in turn can contribute to an increased baroreflex sensitivity, leading to a more regular HR pattern and an elevation of baroreceptor mediated HRV measures [82,83]. However, as this shift amounts to an average of only 0.03 Hz, a significant effect of breathing seems to be not very likely. The lack of direct measurements of respiration is a limitation of this study, because it is well known that especially slow breathing can exert strong effects on HRV. However, participants were instructed to breathe normal and to avoid valsalva maneuvers and slow breathing during the experiments. Further, valsalva maneuvers are unlikely during low workload conditions such as in our study. Further, investigators did not register any irregular breathing pattern during the experiments as well. In addition to the traditional HRV power calculation, we analyzed the HRV spectral peaks to get an estimation of breathing rate.
5. Conclusions
Findings of this study suggest that—despite the same net effect on HR—autonomic control of cardiovascular responses to ISO and DYN is different. They further support the model of an autonomic space, where equivalent HR can be seen as an “end product”, which is achieved by different autonomic modes, such as reciprocal behavior or a sympatho-vagal coactivation [84]. The traditional view of the interplay between both autonomic branches as a reciprocal antagonism, with a reduction of vagal efferent activity at the beginning or during low intensity exercise to a minimum at HRs around 100 bpm and a subsequent rise in sympathetic activity during higher exercise intensities, is currently challenged [72,73]. Our results suggest that, depending on the mode of exercise, HR at low intensities can be achieved by a concomitant increase of sympathetic and vagal outflow to the heart as well. It can be concluded, that the contraction mode itself is a significant modulator of the cardiovascular response to exercise with ISO leading to an increased dual HR modulation if compared to DYN.
Acknowledgments
Part of this work was funded by the German Federal Ministry of Education and Research (BMBF), Grant Number: 03Z1KN11. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Part of this work was technically supported by the Institute for Preventive Medicine, Rostock University Medical Center. During the conduction of the study, the first author was partly employed at Rostock University Medical Center, Institute for Preventive Medicine.
List of Abbreviations
| ANOVA | analysis of variance |
| DBP | diastolic arterial blood pressure |
| DYN | dynamic leg exercise |
| HF | high frequency (0.15–0.4 Hz) range of HRV spectrum |
| HR | heart rate |
| HRV | heart rate variability |
| ISO | isometric leg exercise |
| LF | low frequency (0.04–0.15 Hz) range of HRV spectrum |
| R-R interval | inter beat interval, time between two consecutive R-waves |
| REST | resting condition |
| RMSSD | root mean square of successive R-R differences |
| RPP | rate pressure product |
| SampEn | sample entropy |
| SBP | systolic arterial blood pressure |
| SD | standard deviation |
| SDNN | SD of R-R intervals |
Author Contributions
Matthias Weippert: conception and design of the study, acquisition and analysis of the data, interpretation of the data, drafting the article. Martin Behrens: acquisition and analysis of the data, interpretation of the data, critical revision of the manuscript. Annika Rieger: critical revision of the manuscript. Kristin Behrens: analysis of the data, interpretation of the data, critical revision of the manuscript. All authors have read and approved the final manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Peng, C.K.; Havlin, S.; Stanley, H.E.; Goldberger, A.L. Quantification of scaling exponents and crossover phenomena in nonstationary heartbeat time series. Chaos 1995, 5, 82–87. [Google Scholar]
- Iyengar, N.; Peng, C.K.; Morin, R.; Goldberger, A.L.; Lipsitz, L.A. Age-related alterations in the fractal scaling of cardiac interbeat interval dynamics. Am. J. Physiol. 1996, 271, 1078–1084. [Google Scholar]
- Acharya, R.U.; Lim, C.M.; Joseph, P. Heart rate variability analysis using correlation dimension and detrended fluctuation analysis. Rev. Eur. Technol. Biomed. (ITBM-RBM) 2002, 23, 333–339. [Google Scholar]
- Goldberger, A.L. Fractal mechanisms in the electrophysiology of the heart. IEEE Eng. Med. Biol. Mag. 1992, 11, 47–52. [Google Scholar]
- Tulppo, M.P.; Hautala, A.J.; Makikallio, T.H.; Laukkanen, R.T.; Nissila, S.; Hughson, R.L.; Huikuri, H.V. Effects of aerobic training on heart rate dynamics in sedentary subjects. J. Appl. Physiol. 2003, 95, 364–372. [Google Scholar]
- Tulppo, M.P.; Hughson, R.L.; Mäkikallio, T.H.; Airaksinen, K.E.J.; Seppänen, T.; Huikuri, H.V. Effects of exercise and passive head-up tilt on fractal and complexity properties of heart rate dynamics. Am. J. Physiol.-Heart Circ. Physiol. 2001, 280, 1081–1087. [Google Scholar]
- Weippert, M.; Behrens, K.; Rieger, A.; Stoll, R.; Kreuzfeld, S. Heart rate variability and blood pressure during dynamic and static exercise at similar heart rate levels. PloS One 2013, 8. [Google Scholar] [CrossRef]
- Princi, T.; Accardo, A.; Peterec, D. Linear and non-linear parameters of heart rate variability during static and dynamic exercise in a high-performance dinghy sailor. Biomed. Sci. Instrum. 2004, 40, 311–316. [Google Scholar]
- Bigger, J.T.; Steinman, R.C.; Rolnitzky, L.M.; Fleiss, J.L.; Albrecht, P.; Cohen, R.J. Power law behavior of RR-interval variability in healthy middle-aged persons, patients with recent acute myocardial infarction, and patients with heart transplants. Circulation 1996, 93, 2142–2151. [Google Scholar]
- Huikuri, H.V.; Mäkikallio, T.H.; Peng, C.K.; Goldberger, A.L.; Hintze, U.; Møller, M. Fractal correlation properties of R-R interval dynamics and mortality in patients with depressed left ventricular function after an acute myocardial infarction. Circulation 2000, 101, 47–53. [Google Scholar]
- Mäkikallio, T.H.; Tapanainen, J.M.; Tulppo, M.P.; Huikuri, H.V. Clinical applicability of heart rate variability analysis by methods based on nonlinear dynamics. Card. Electrophysiol. Rev. 2002, 6, 250–255. [Google Scholar]
- Mäkikallio, T.H.; Seppänen, T.; Niemelä, M.; Airaksinen, K.E.; Tulppo, M.; Huikuri, H.V. Abnormalities in beat to beat complexity of heart rate dynamics in patients with a previous myocardial infarction. J. Am. Coll. Cardiol. 1996, 28, 1005–1011. [Google Scholar]
- Mäkikallio, T.H.; Huikuri, H.V.; Hintze, U.; Videbaek, J.; Mitrani, R.D.; Castellanos, A.; Myerburg, R.J.; Moller, M. Fractal analysis and time- and frequency-domain measures of heart rate variability as predictors of mortality in patients with heart failure. Am. J. Cardiol. 2001, 87, 178–182. [Google Scholar]
- Vikman, S.; Makikallio, T.H.; Yli-Mayry, S.; Pikkujamsa, S.; Koivisto, A.M.; Reinikainen, P.; Airaksinen, K.E.J.; Huikuri, H.V. Altered complexity and correlation properties of R-R interval dynamics before the spontaneous onset of paroxysmal atrial fibrillation. Circulation 1999, 100, 2079–2084. [Google Scholar]
- Raab, C.; Kurths, J.; Schirdewan, A.; Wessel, N. Normalized correlation dimension for heart rate variability analysis. Biomed. Tech. 2006, 51, 229–232. [Google Scholar]
- Javorka, M.; Trunkvalterova, Z.; Tonhajzerova, I.; Javorkova, J.; Javorka, K.; Baumert, M. Short-term heart rate complexity is reduced in patients with type 1 diabetes mellitus. Clin. Neurophysiol. 2008, 119, 1071–1081. [Google Scholar]
- Ho, K.K.; Moody, G.B.; Peng, C.K.; Mietus, J.E.; Larson, M.G.; Levy, D.; Goldberger, A.L. Predicting survival in heart failure case and control subjects by use of fully automated methods for deriving nonlinear and conventional indices of heart rate dynamics. Circulation 1997, 96, 842–848. [Google Scholar]
- Schmidt, G.; Morfill, G.E. Nonlinear methods for heart rate variability assessment. In Heart Rate Variability; Malik, M., Camm, A.J., Eds.; Futura: Armonk, NY, USA, 1995; pp. 87–98. [Google Scholar]
- Tulppo, M.P.; Mäkikallio, T.H.; Seppänen, T.; Shoemaker, K.; Tutungi, E.; Hughson, R.L.; Huikuri, H.V. Effects of pharmacological adrenergic and vagal modulation on fractal heart rate dynamics. Clin. Physiol. 2001, 21, 515–523. [Google Scholar]
- Perkiomaki, J.S.; Zareba, W.; Badilini, F.; Moss, A.J. Influence of atropine on fractal and complexity measures of heart rate variability. Ann. Noninvasive Electrocardiol. 2002, 7, 326–331. [Google Scholar]
- Porta, A.; Gnecchi-Ruscone, T.; Tobaldini, E.; Guzzetti, S.; Furlan, R.; Montano, N. Progressive decrease of heart period variability entropy-based complexity during graded head-up tilt. J. Appl. Physiol. 2007, 103, 1143–1149. [Google Scholar]
- Porta, A.; Castiglioni, P.; Bari, V.; Bassani, T.; Marchi, A.; Cividjian, A.; Quintin, L.; di Rienzo, M. K-nearest-neighbor conditional entropy approach for the assessment of the short-term complexity of cardiovascular control. Physiol. Meas. 2013, 34, 17–33. [Google Scholar]
- Lindquist, V.A.; Spangler, R.D.; Blount, S.G. A comparison between the effects of dynamic and isometric exercise as evaluated by the systolic time intervals in normal man. Am. Heart J. 1973, 85, 227–236. [Google Scholar]
- Leicht, A.S.; Sinclair, W.H.; Spinks, W.L. Effect of exercise mode on heart rate variability during steady state exercise. Eur. J. Appl. Physiol. 2008, 102, 195–204. [Google Scholar]
- Cottin, F.; Durbin, F.; Papelier, Y. Heart rate variability during cycloergometric exercise or judo wrestling eliciting the same heart rate level. Eur. J. Appl. Physiol. 2004, 91, 177–184. [Google Scholar]
- Casadei, B.; Moon, J.; Johnston, J.; Caiazza, A.; Sleight, P. Is respiratory sinus arrhythmia a good index of cardiac vagal tone in exercise? J. Appl. Physiol. 1996, 81, 556–564. [Google Scholar]
- Casadei, B.; Cochrane, S.; Johnston, J.; Conway, J.; Sleight, P. Pitfalls in the interpretation of spectral analysis of the heart rate variability during exercise in humans. Acta Physiol. Scand. 1995, 153, 125–131. [Google Scholar]
- Perini, R.; Veicsteinas, A. Heart rate variability and autonomic activity at rest and during exercise in various physiological conditions. Eur. J. Appl. Physiol. 2003, 90, 317–325. [Google Scholar]
- Yeragani, V.K.; Krishnan, S.; Engels, H.J.; Gretebeck, R. Effects of caffeine on linear and nonlinear measures of heart rate variability before and after exercise. Depress. Anxiety 2005, 21, 130–134. [Google Scholar]
- Kingsley, M.; Lewis, M.J.; Marson, R.E. Comparison of Polar 810s and an ambulatory ECG system for RR interval measurement during progressive exercise. Int. J. Sports Med. 2005, 26, 39–44. [Google Scholar]
- Weippert, M.; Kumar, M.; Kreuzfeld, S.; Arndt, D.; Rieger, A.; Stoll, R. Comparison of three mobile devices for measuring R-R intervals and heart rate variability: Polar S810i, Suunto t6 and an ambulatory ECG system. Eur. J. Appl. Physiol. 2010, 109, 779–786. [Google Scholar]
- Weber, F.; Lindemann, M.; Erbel, R.; Philipp, T. Indirect and direct simultaneous, comparative blood pressure measurements with the Bosotron 2 device. Kidney Blood Press. Res. 1999, 22, 166–171. [Google Scholar]
- Magagnin, V.; Bassani, T.; Bari, V.; Turiel, M.; Maestri, R.; Pinna, G.D.; Porta, A. Non-stationarities significantly distort short-term spectral, symbolic and entropy heart rate variability indices. Physiol. Meas. 2011, 32, 1775–1786. [Google Scholar]
- Malliani, A.; Pagani, M.; Lombardi, F.; Cerutti, S. Cardiovascular neural regulation explored in the frequency domain. Circulation 1991, 84, 482–492. [Google Scholar]
- Pagani, M.; Lombardi, F.; Guzzetti, S.; Rimoldi, O.; Furlan, R.; Pizzinelli, P.; Sandrone, G.; Malfatto, G.; Dell’Orto, S.; Piccaluga, E.; et al. Power spectral analysis of heart rate and arterial pressure variabilities as a marker of sympatho-vagal interaction in man and conscious dog. Circ. Res. 1986, 59, 178–193. [Google Scholar]
- Lombardi, F.; Malliani, A. Task force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart rate variability: Standards of measurement, physiological interpretation and clinical use. Circulation 1996, 93, 1043–1065. [Google Scholar]
- Richman, J.S.; Moorman, J.R. Physiological time-series analysis using approximate entropy and sample entropy. Am. J. Physiol.-Heart Circ. Physiol. 2000, 278, 2039–2049. [Google Scholar]
- Pincus, S.M. Approximate entropy as a measure of system complexity. Proc. Natl. Acad. Sci. 1991, 88, 2297–2301. [Google Scholar]
- Rickards, C.A.; Ryan, K.L.; Convertino, V.A. Characterization of common measures of heart period variability in healthy human subjects: implications for patient monitoring. J. Clin. Monit. Comput. 2010, 24, 61–70. [Google Scholar]
- Lake, D.E.; Moorman, J.R. Accurate estimation of entropy in very short physiological time series: The problem of atrial fibrillation detection in implanted ventricular devices. Am. J. Physiol.-Heart Circ. Physiol. 2011, 300, 319–325. [Google Scholar]
- Lake, D.E.; Richman, J.S.; Griffin, M.P.; Moorman, J.R. Sample entropy analysis of neonatal heart rate variability. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2002, 283, 789–797. [Google Scholar]
- Pincus, S.M.; Viscarello, R.R. Approximate entropy: A regularity measure for fetal heart rate analysis. Obstet. Gynecol. 1992, 79, 249–255. [Google Scholar]
- Nelson, R.R.; Gobel, F.L.; Jorgensen, C.R.; Wang, K.; Wang, Y.; Taylor, H.L. Hemodynamic predictors of myocardial oxygen consumption during static and dynamic exercise. Circulation 1974, 50, 1179–1189. [Google Scholar]
- Gobel, F.L.; Norstrom, L.A.; Nelson, R.R.; Jorgensen, C.R.; Wang, Y. The rate-pressure product as an index of myocardial oxygen consumption during exercise in patients with angina pectoris. Circulation 1978, 57, 549–556. [Google Scholar]
- Williams, C.A.; Mudd, J.G.; Lind, A.R. Sympathetic control of the forearm blood flow in man during brief isometric contractions. Eur. J. Appl. Physiol. Occup. Physiol. 1985, 54, 156–162. [Google Scholar]
- Rowell, L.B.; O’Leary, D.S. Reflex control of the circulation during exercise: Chemoreflexes and mechanoreflexes. J. Appl. Physiol. 1990, 69, 407–418. [Google Scholar]
- Mitchell, J.H. Cardiovascular control during exercise: Central and reflex neural mechanisms. Am. J. Cardiol. 1985, 55, 34–41. [Google Scholar]
- Iellamo, F. Neural mechanisms of cardiovascular regulation during exercise. Auton. Neurosci. 2001, 90, 66–75. [Google Scholar]
- Augustyniak, R.A.; Collins, H.L.; Ansorge, E.J.; Rossi, N.F.; O’Leary, D.S. Severe exercise alters the strength of the muscle metaboreflex. Am. J. Physiol.-Heart Circ. Physiol. 2001, 280, 1645–1652. [Google Scholar]
- Nobrega, A.C.; Williamson, J.W.; Garcia, J.A.; Mitchell, J.H. Mechanisms for increasing stroke volume during static exercise with fixed heart rate in humans. J. Appl. Physiol. 1997, 83, 712–717. [Google Scholar]
- Crisafulli, A.; Scott, A.C.; Wensel, R.; Davos, C.H.; Francis, D.P.; Pagliaro, P.; Coats, A.J.; Concu, A.; Piepoli, M.F. Muscle metaboreflex-induced increases in stroke volume. Med. Sci. Sports Exerc. 2003, 35, 221–228. [Google Scholar]
- Elstad, M.; Nadland, I.H.; Toska, K.; Walloe, L. Stroke volume decreases during mild dynamic and static exercise in supine humans. Acta Physiol. 2009, 195, 289–300. [Google Scholar]
- Abboud, F.M. Integration of reflex responses in the control of blood pressure and vascular resistance. Am. J. Cardiol. 1979, 44, 903–911. [Google Scholar]
- Casadei, B. Vagal control of myocardial contractility in humans. Exp. Physiol. 2001, 86, 817–823. [Google Scholar]
- Rotto, D.M.; Kaufman, M.P. Effect of metabolic products of muscular contraction on discharge of group III and IV afferents. J. Appl. Physiol. 1988, 64, 2306–2313. [Google Scholar]
- Light, A.R.; Hughen, R.W.; Zhang, J.; Rainier, J.; Liu, Z.; Lee, J. Dorsal root ganglion neurons innervating skeletal muscle respond to physiological combinations of protons, ATP, and lactate mediated by ASIC, P2X, and TRPV1. J. Neurophysiol. 2008, 100, 1184–1201. [Google Scholar]
- Secher, N.H.; Amann, M. Human investigations into the exercise pressor reflex. Exp. Physiol. 2012, 97, 59–69. [Google Scholar]
- Kaufman, M.P.; Longhurst, J.C.; Rybicki, K.J.; Wallach, J.H.; Mitchell, J.H. Effects of static muscular contraction on impulse activity of groups III and IV afferents in cats. J. Appl. Physiol. 1983, 55, 105–112. [Google Scholar]
- Carrington, C.A.; Fisher, W.J.; Davies, M.K.; White, M.J. Muscle afferent and central command contributions to the cardiovascular response to isometric exercise of postural muscle in patients with mild chronic heart failure. Clin. Sci. 2001, 100, 643–651. [Google Scholar]
- Goodwin, G.M.; McCloskey, D.I.; Mitchell, J.H. Cardiovascular and respiratory responses to changes in central command during isometric exercise at constant muscle tension. J. Physiol. 1972, 226, 173–190. [Google Scholar]
- Fisher, J.P.; Ogoh, S.; Dawson, E.A.; Fadel, P.J.; Secher, N.H.; Raven, P.B.; White, M.J. Cardiac and vasomotor components of the carotid baroreflex control of arterial blood pressure during isometric exercise in humans. J. Physiol. 2006, 572, 869–880. [Google Scholar]
- Mark, A.L.; Victor, R.G.; Nerhed, C.; Wallin, B.G. Microneurographic studies of the mechanisms of sympathetic-nerve responses to static exercise in humans. Circ. Res. 1985, 57, 461–469. [Google Scholar]
- Hartwich, D.; Dear, W.E.; Waterfall, J.L.; Fisher, J.P. Effect of muscle metaboreflex activation on spontaneous cardiac baroreflex sensitivity during exercise in humans. J. Physiol. 2011, 589, 6157–6171. [Google Scholar]
- Piepoli, M.; Clark, A.L.; Coats, A.J. Muscle metaboreceptors in hemodynamic, autonomic, and ventilatory responses to exercise in men. Am. J. Physiol. 1995, 269, 1428–1436. [Google Scholar]
- Ponikowski, P.; Chua, T.P.; Piepoli, M.; Ondusova, D.; Webb-Peploe, K.; Harrington, D.; Anker, S.D.; Volterrani, M.; Colombo, R.; Mazzuero, G.; et al. Augmented peripheral chemosensitivity as a potential input to baroreflex impairment and autonomic imbalance in chronic heart failure. Circulation 1997, 96, 2586–2594. [Google Scholar]
- Saito, M.; Mano, T. Exercise mode affects muscle sympathetic nerve responsiveness. Jpn. J. Physiol. 1991, 41, 143–151. [Google Scholar]
- Gonzalez-Camarena, R.; Carrasco-Sosa, S.; Roman-Ramos, R.; Gaitan-Gonzalez, M.J.; Medina-Banuelos, V.; Azpiroz-Leehan, J. Effect of static and dynamic exercise on heart rate and blood pressure variabilities. Med. Sci. Sports Exerc. 2000, 32, 1719–1728. [Google Scholar]
- Tulppo, M.P.; Mäkikallio, T.H.; Takala, T.E.; Seppänen, T.; Huikuri, H.V. Quantitative beat-to-beat analysis of heart rate dynamics during exercise. Am. J. Physiol.-Heart Circ. Physiol. 1996, 271, 244–252. [Google Scholar]
- Kiviniemi, A.M.; Hautala, A.J.; Seppanen, T.; Makikallio, T.H.; Huikuri, H.V.; Tulppo, M.P. Saturation of high-frequency oscillations of R-R intervals in healthy subjects and patients after acute myocardial infarction during ambulatory conditions. Am. J. Physiol.-Heart Circ. Physiol. 2004, 287, 1921–1927. [Google Scholar]
- Robinson, B.F.; Epstein, S.E.; Beiser, G.D.; Braunwald, E. Control of heart rate by the autonomic nervous system. Studies in man on the interrelation between baroreceptor mechanisms and exercise. Circ. Res. 1966, 19, 400–411. [Google Scholar]
- Matsukawa, K. Central command: Control of cardiac sympathetic and vagal efferent nerve activity and the arterial baroreflex during spontaneous motor behaviour in animals. Exp. Physiol. 2012, 97, 20–28. [Google Scholar]
- Fisher, J.P. Autonomic control of the heart during exercise in humans: Role of skeletal muscle afferents. Exp. Physiol. 2014, 99, 300–305. [Google Scholar]
- White, D.W.; Raven, P.B. Autonomic neural control of heart rate during dynamic exercise: Revisited. J. Physiol. 2014, 592, 2491–2500. [Google Scholar]
- Porta, A.; Guzzetti, S.; Furlan, R.; Gnecchi-Ruscone, T.; Montano, N.; Malliani, A. Complexity and nonlinearity in short-term heart period variability: Comparison of methods based on local nonlinear prediction. IEEE Trans. Biomed. Eng. 2007, 54, 94–106. [Google Scholar]
- Gladwell, V.F.; Coote, J.H. Heart rate at the onset of muscle contraction and during passive muscle stretch in humans: A role for mechanoreceptors. J. Physiol. 2002, 540, 1095–1102. [Google Scholar]
- Waldrop, T.G.; Rybicki, K.J.; Kaufman, M.P. Chemical activation of group I and II muscle afferents has no cardiorespiratory effects. J. Appl. Physiol. 1984, 56, 1223–1228. [Google Scholar]
- Bernardi, L.; Gabutti, A.; Porta, C.; Spicuzza, L. Slow breathing reduces chemoreflex response to hypoxia and hypercapnia, and increases baroreflex sensitivity. J. Hypertens. 2001, 19, 2221–2229. [Google Scholar]
- Bernardi, L.; Porta, C.; Spicuzza, L.; Bellwon, J.; Spadacini, G.; Frey, A.W.; Yeung, L.Y.; Sanderson, J.E.; Pedretti, R.; Tramarin, R. Slow breathing increases arterial baroreflex sensitivity in patients with chronic heart failure. Circulation 2002, 105, 143–145. [Google Scholar]
- Joseph, C.N.; Porta, C.; Casucci, G.; Casiraghi, N.; Maffeis, M.; Rossi, M.; Bernardi, L. Slow breathing improves arterial baroreflex sensitivity and decreases blood pressure in essential hypertension. Hypertension 2005, 46, 714–718. [Google Scholar]
- Raupach, T.; Bahr, F.; Herrmann, P.; Luethje, L.; Heusser, K.; Hasenfuss, G.; Bernardi, L.; Andreas, S. Slow breathing reduces sympathoexcitation in COPD. Eur. Respir. J. 2008, 32, 387–392. [Google Scholar]
- Van De Borne, P.; Mezzetti, S.; Montano, N.; Narkiewicz, K.; Degaute, J.P.; Somers, V.K. Hyperventilation alters arterial baroreflex control of heart rate and muscle sympathetic nerve activity. Am. J. Physiol.-Heart Circ. Physiol. 2000, 279, 536–541. [Google Scholar]
- Eckberg, D.L. Human sinus arrhythmia as an index of vagal cardiac outflow. J. Appl. Physiol. 1983, 54, 961–966. [Google Scholar]
- Hirsch, J.A.; Bishop, B. Respiratory sinus arrhythmia in humans: How breathing pattern modulates heart rate. Am. J. Physiol. 1981, 241, 620–629. [Google Scholar]
- Berntson, G.G.; Cacioppo, J.T.; Quigley, K.S. Cardiac psychophysiology and autonomic space in humans: empirical perspectives and conceptual implications. Psychol. Bull. 1993, 114, 296–322. [Google Scholar]
| Mean ± SD | Range | |
|---|---|---|
| Age [years] | 26.0 ± 3.1 | 21.0–36.0 |
| Weight [kg] | 80.6 ± 8.3 | 62.9–100.0 |
| Height [m] | 183.7 ± 6.4 | 171.0–195.0 |
| BMI [kg/m2] | 23.9 ± 1.9 | 19.0–28.5 |
| Parameter | REST | ISO | DYN |
|---|---|---|---|
| Heart rate [bpm] | 65.3 ± 9.4 | 85.2 ± 9.2*** | 84.9 ± 9.1*** |
| SBP [mmHg] | 128.2 ± 9.2 | 157.6 ± 12.6***, §§§ | 144.6 ± 14.0***, §§§ |
| DBP [mmHg] | 77.2 ± 6.8 | 94.4 ± 8.0***, §§§ | 73.7 ± 10.3*, §§§ |
| MAP [mmHg] | 94.2 ± 5.2 | 115.5 ± 7.6***, §§§ | 97.4 ± 10.1*, §§§ |
| RPP [mmHg/min] | 8,374.6 ± 1,363.3 | 13,454.0 ± 1,961.5***, §§§ | 12,294.9 ± 1,844.6***, §§§ |
| R-R interval [ms] | 943.6 ± 137.3 | 714.4 ± 78.0*** | 715.9 ± 77.9*** |
| SDNN [ms] | 61.3 ± 30.7 | 35.3 ± 20.0***, §§§ | 27.9 ± 17.3***, §§§ |
| RMSSD [ms] | 53.3 ± 29.9 | 23.6 ± 16.5*** | 22.3 ± 17.1*** |
| lnHFP [ms2] | 6.5 ± 1.1 | 5.1 ± 1.3***, §§§ | 4.4 ± 1.4***, §§§ |
| lnLFP [ms2] | 7.0 ± 1.1 | 6.3 ± 0.9***, §§§ | 5.6 ± 0.9***, §§§ |
| HF n. u. | 41.3 ± 18.0 | 26.7 ± 15.1** | 27.4 ± 19.3** |
| LF n. u. | 58.7 ± 20.5 | 73.3 ± 15.1*** | 72.4 ± 19.4** |
| LF/HF | 3.0 ± 5.7 | 4.7 ± 4.6 | 5.1 ± 4.6# |
SBP = systolic blood pressure; DBP = diastolic blood pressure; MAP = mean arterial pressure; RPP = rate pressure product; R-R interval = time interval between two consecutive heart beats; SDNN = standard deviation of the R-R intervals of the record; RMSSD = square root of the mean sum of squares of the differences between adjacent R-R intervals in the record; HFP = high frequency power; LFP = low frequency power;*/**/***significantly different from rest on a p-level < 0.05/ < 0.01/ < 0.001;§/§§/§§§significantly different from the respective exercise condition on a p-level < 0.05/ < 0.01/ < 0.001;#different from REST on a p-level of 0.077.
| SampEn (REST) | SampEn (ISO) | SampEn (DYN) | |
|---|---|---|---|
| R-R interval | 0.460** | 0.601** | 0.506** |
| SDNN | −0.224 | 0.045 | 0.070 |
| RMSSD | 0.038 | 0.159 | 0.209 |
| lnLFP | −0.273 | −0.197 | 0.082 |
| lnHFP | 0.231 | 0.153 | 0.247 |
| % LF | −0.426** | −0.377* | −0.249 |
| % HF | 0.441** | 0.263 | 0.329* |
*p < 0.05;**p < 0.01.
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weippert, M.; Behrens, M.; Rieger, A.; Behrens, K. Sample Entropy and Traditional Measures of Heart Rate Dynamics Reveal Different Modes of Cardiovascular Control During Low Intensity Exercise. Entropy 2014, 16, 5698-5711. https://doi.org/10.3390/e16115698
Weippert M, Behrens M, Rieger A, Behrens K. Sample Entropy and Traditional Measures of Heart Rate Dynamics Reveal Different Modes of Cardiovascular Control During Low Intensity Exercise. Entropy. 2014; 16(11):5698-5711. https://doi.org/10.3390/e16115698
Chicago/Turabian StyleWeippert, Matthias, Martin Behrens, Annika Rieger, and Kristin Behrens. 2014. "Sample Entropy and Traditional Measures of Heart Rate Dynamics Reveal Different Modes of Cardiovascular Control During Low Intensity Exercise" Entropy 16, no. 11: 5698-5711. https://doi.org/10.3390/e16115698
APA StyleWeippert, M., Behrens, M., Rieger, A., & Behrens, K. (2014). Sample Entropy and Traditional Measures of Heart Rate Dynamics Reveal Different Modes of Cardiovascular Control During Low Intensity Exercise. Entropy, 16(11), 5698-5711. https://doi.org/10.3390/e16115698




