Identification of Patients with Sarcopenia Using Gait Parameters Based on Inertial Sensors
Abstract
1. Introduction
2. Related Work
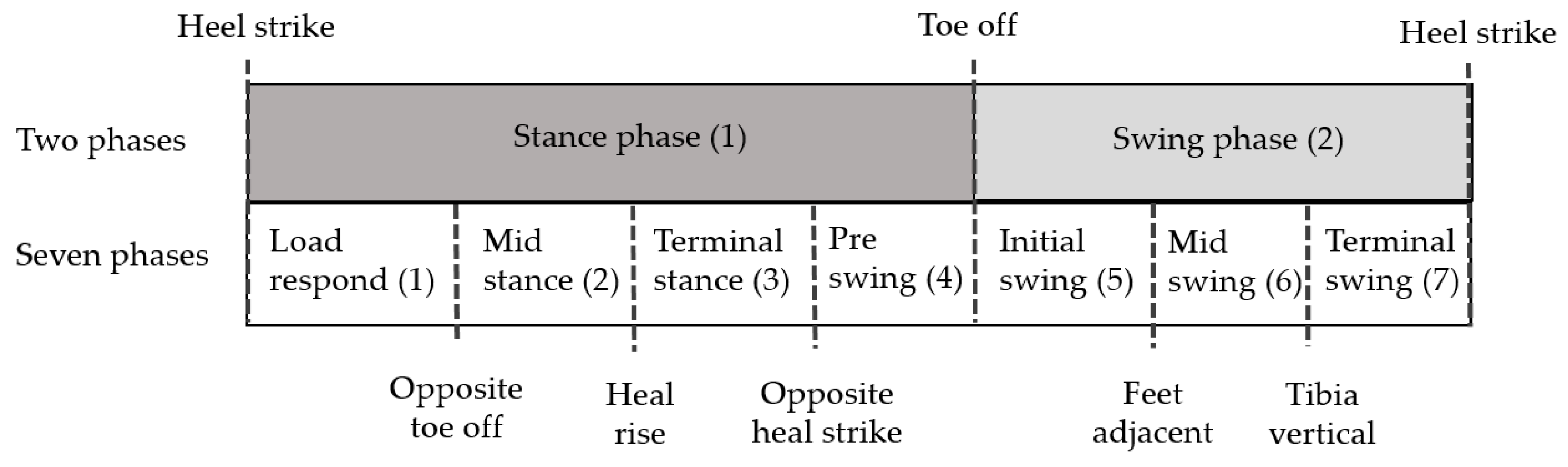
2.1. Gait Phases
2.2. Patient Identification Using IMU
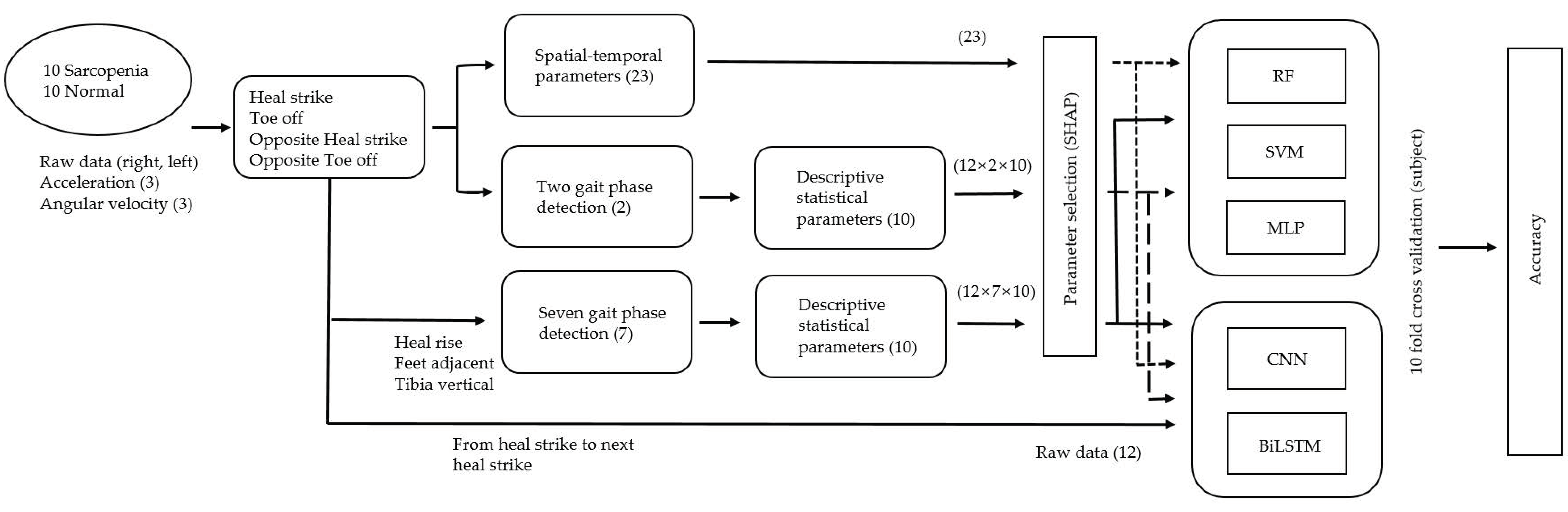
3. Methods
3.1. Subject, Equipment, and Data Collection
3.2. Extraction of Gait Parameters
3.3. Extraction of Gait Phase
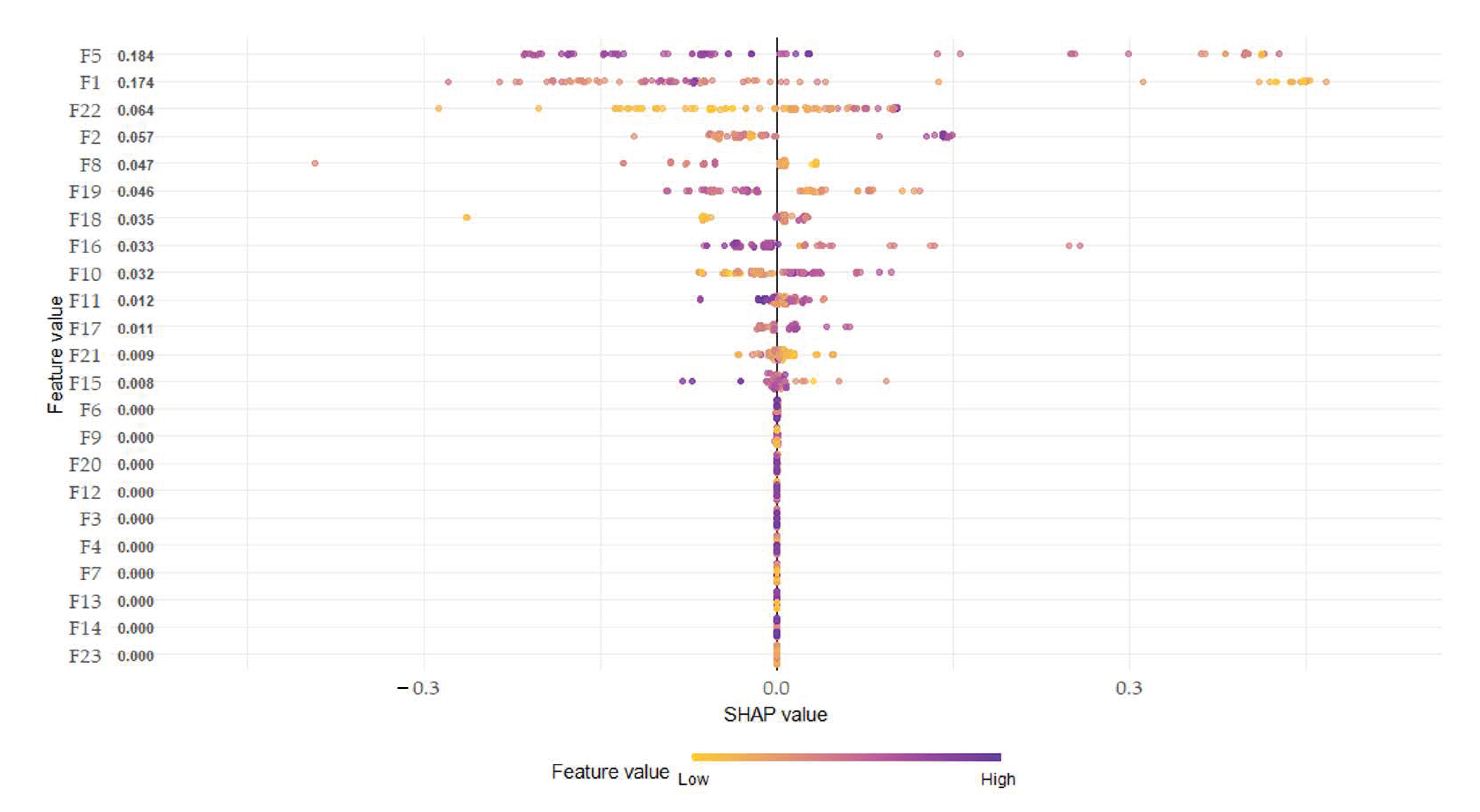
3.4. Feature Selection
3.5. Machine Learning
3.6. Proposed Data Pipeline
4. Results
4.1. Gait Parameters
- Spatial-temporal parameter (top 20): 5, 1, 22, 2, 8, 19, 18, 16, 10, 11, 17, 21, 15, 6, 9, 20, 12, 3, 4, and 7.
- Descriptive statistical parameters for two phases (top 20): 52, 126, 37, 97, 8, 51, 144, 211, 24, 3, 232, 115, 116, 31, 57, 50, 109, 43, 100, and 4.
- Descriptive statistical parameters for seven phases (top 20): 196, 524, 504, 97, 3, 231, 526, 507, 430, 187, 380, 8, 130, 57, 51, 200, 828, 283, 523, and 9.
4.2. Identification of Sarcopenia
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Number | Subject | Normal | Sarcopenia | Normal | Sarcopenia | T-Test (Mean) | |
|---|---|---|---|---|---|---|---|
| Parameter | Mean | Mean | STD | STD | p-Value | Statistic | |
| 1 | Stance phase_right (%) | 60.263 | 60.432 | 0.683 | 0.925 | 0.697 | −0.358 |
| 2 | Stance phase_left (%) | 59.870 | 60.808 | 0.772 | 1.042 | 0.014 | −2.250 |
| 3 | Swing phase_right (%) | 39.736 | 39.567 | 0.683 | 0.925 | 0.697 | 0.358 |
| 4 | Swing phase_left (%) | 40.129 | 39.191 | 0.772 | 1.042 | 0.014 | 2.250 |
| 5 | Single support phase_right (%) | 40.182 | 39.141 | 0.926 | 1.202 | 0.006 | 2.486 |
| 6 | Single support phase_left (%) | 39.826 | 39.633 | 0.901 | 1.169 | 0.657 | 0.439 |
| 7 | Double support phase (%) | 19.975 | 21.101 | 2.149 | 4.038 | 0.142 | −1.347 |
| 8 | Stance time_right (s) | 0.604 | 0.615 | 0.013 | 0.017 | 0.323 | −0.615 |
| 9 | Stance time_left (s) | 0.600 | 0.618 | 0.013 | 0.017 | 0.091 | −1.250 |
| 10 | Swing time_right (s) (stride time) | 0.415 | 0.417 | 0.009 | 0.012 | 0.588 | 0.180 |
| 11 | Swing time_left (s) | 0.418 | 0.414 | 0.009 | 0.012 | 0.250 | 1.713 |
| 12 | Single support_left (s) | 0.401 | 0.391 | 0.006 | 0.008 | 0.013 | 2.300 |
| 13 | Double support (s) | 0.200 | 0.210 | 0.018 | 0.034 | 0.186 | −1.216 |
| 14 | Single support_right (s) | 0.397 | 0.395 | 0.006 | 0.007 | 0.642 | 0.459 |
| 15 | Stride length (m) | 0.954 | 0.915 | 0.132 | 0.148 | 0.149 | 1.297 |
| 16 | Step length_right (m) | 0.483 | 0.467 | 0.141 | 0.147 | 0.226 | 1.050 |
| 17 | Step length_left (m) | 0.471 | 0.448 | 0.123 | 0.148 | 0.107 | 1.482 |
| 18 | Step time_right (s) | 0.515 | 0.523 | 0.009 | 0.011 | 0.262 | −0.697 |
| 19 | Step time_left (s) | 0.501 | 0.506 | 0.009 | 0.011 | 0.531 | −0.114 |
| 20 | Cadence (steps/min) | 123.331 | 121.978 | 0 | 0 | 0.399 | 0.251 |
| 21 | Stance phase dRL | 1.224 | 1.812 | 0.007 | 0.011 | <0.001 | −3.973 |
| 22 | Stance time dRL | 0.014 | 0.021 | 1.025 | 1.405 | <0.001 | −3.846 |
| 23 | Swing time dRL | 0.012 | 0.019 | 0.901 | 1.319 | <0.001 | −4.032 |
| Right | Left | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Parameter | Max | Min | STD | AbSum | RMS | Ku | Ske | MMgr | DMM | Mdif | Max | Min | STD | AbSum | RMS | Ku | Ske | MMgr | DMM | Mdif | |
| Stance phase | AccX | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 121 | 122 | 123 | 124 | 125 | 126 | 127 | 128 | 129 | 130 |
| AccY | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 131 | 132 | 133 | 134 | 135 | 136 | 137 | 138 | 139 | 140 | |
| AccZ | 21 | 22 | 23 | 24 | 25 | 26 | 27 | 28 | 29 | 30 | 141 | 142 | 143 | 144 | 145 | 146 | 147 | 148 | 149 | 150 | |
| GyroX | 31 | 32 | 33 | 34 | 35 | 36 | 37 | 38 | 39 | 40 | 151 | 152 | 153 | 154 | 155 | 156 | 157 | 158 | 159 | 160 | |
| GyroY | 41 | 42 | 43 | 44 | 45 | 46 | 47 | 48 | 49 | 50 | 161 | 162 | 163 | 164 | 165 | 166 | 167 | 168 | 169 | 170 | |
| GyroZ | 51 | 52 | 53 | 54 | 55 | 56 | 57 | 58 | 59 | 60 | 171 | 172 | 173 | 174 | 175 | 176 | 177 | 178 | 179 | 180 | |
| Swing phase | AccX | 61 | 62 | 63 | 64 | 65 | 66 | 67 | 68 | 69 | 70 | 181 | 182 | 183 | 184 | 185 | 186 | 187 | 188 | 189 | 190 |
| AccY | 71 | 72 | 73 | 74 | 75 | 76 | 77 | 78 | 79 | 80 | 191 | 192 | 193 | 194 | 195 | 196 | 197 | 198 | 199 | 200 | |
| AccZ | 81 | 82 | 83 | 84 | 85 | 86 | 87 | 88 | 89 | 90 | 201 | 202 | 203 | 204 | 205 | 206 | 207 | 208 | 209 | 210 | |
| GyroX | 91 | 92 | 93 | 94 | 95 | 96 | 97 | 98 | 99 | 100 | 211 | 212 | 213 | 214 | 215 | 216 | 217 | 218 | 219 | 220 | |
| GyroY | 101 | 102 | 103 | 104 | 105 | 106 | 107 | 108 | 109 | 110 | 221 | 222 | 223 | 224 | 225 | 226 | 227 | 228 | 229 | 230 | |
| GyroZ | 111 | 112 | 113 | 114 | 115 | 116 | 117 | 118 | 119 | 120 | 231 | 232 | 233 | 234 | 235 | 236 | 237 | 238 | 239 | 240 | |
| Right | Left | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Parameter | Max | Min | STD | AbSum | RMS | Ku | Ske | MMgr | DMM | Mdif | Max | Min | STD | AbSum | RMS | Ku | Ske | MMgr | DMM | Mdif | |
| Loading response | AccX | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 421 | 422 | 423 | 424 | 425 | 426 | 427 | 428 | 429 | 430 |
| AccY | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 431 | 432 | 433 | 434 | 435 | 436 | 437 | 438 | 439 | 440 | |
| AccZ | 21 | 22 | 23 | 24 | 25 | 26 | 27 | 28 | 29 | 30 | 441 | 442 | 443 | 444 | 445 | 446 | 447 | 448 | 449 | 450 | |
| GyroX | 31 | 32 | 33 | 34 | 35 | 36 | 37 | 38 | 39 | 40 | 451 | 452 | 453 | 454 | 455 | 456 | 457 | 458 | 459 | 460 | |
| GyroY | 41 | 42 | 43 | 44 | 45 | 46 | 47 | 48 | 49 | 50 | 461 | 462 | 463 | 464 | 465 | 466 | 467 | 468 | 469 | 470 | |
| GyroZ | 51 | 52 | 53 | 54 | 55 | 56 | 57 | 58 | 59 | 60 | 471 | 472 | 473 | 474 | 475 | 476 | 477 | 478 | 479 | 480 | |
| Mid stance | AccX | 61 | 62 | 63 | 64 | 65 | 66 | 67 | 68 | 69 | 70 | 481 | 482 | 483 | 484 | 485 | 486 | 487 | 488 | 489 | 490 |
| AccY | 71 | 72 | 73 | 74 | 75 | 76 | 77 | 78 | 79 | 80 | 491 | 492 | 493 | 494 | 495 | 496 | 497 | 498 | 499 | 500 | |
| AccZ | 81 | 82 | 83 | 84 | 85 | 86 | 87 | 88 | 89 | 90 | 501 | 502 | 503 | 504 | 505 | 506 | 507 | 508 | 509 | 510 | |
| GyroX | 91 | 92 | 93 | 94 | 95 | 96 | 97 | 98 | 99 | 100 | 511 | 512 | 513 | 514 | 515 | 516 | 517 | 518 | 519 | 520 | |
| GyroY | 101 | 102 | 103 | 104 | 105 | 106 | 107 | 108 | 109 | 110 | 521 | 522 | 523 | 524 | 525 | 526 | 527 | 528 | 529 | 530 | |
| GyroZ | 111 | 112 | 113 | 114 | 115 | 116 | 117 | 118 | 119 | 120 | 531 | 532 | 533 | 534 | 535 | 536 | 537 | 538 | 539 | 540 | |
| Terminal stance | AccX | 121 | 122 | 123 | 124 | 125 | 126 | 127 | 128 | 129 | 130 | 541 | 542 | 543 | 544 | 545 | 546 | 547 | 548 | 549 | 550 |
| AccY | 131 | 132 | 133 | 134 | 135 | 136 | 137 | 138 | 139 | 140 | 551 | 552 | 553 | 554 | 555 | 556 | 557 | 558 | 559 | 560 | |
| AccZ | 141 | 142 | 143 | 144 | 145 | 146 | 147 | 148 | 149 | 150 | 561 | 562 | 563 | 564 | 565 | 566 | 567 | 568 | 569 | 570 | |
| GyroX | 151 | 152 | 153 | 154 | 155 | 156 | 157 | 158 | 159 | 160 | 571 | 572 | 573 | 574 | 575 | 576 | 577 | 578 | 579 | 580 | |
| GyroY | 161 | 162 | 163 | 164 | 165 | 166 | 167 | 168 | 169 | 170 | 581 | 582 | 583 | 584 | 585 | 586 | 587 | 588 | 589 | 590 | |
| GyroZ | 171 | 172 | 173 | 174 | 175 | 176 | 177 | 178 | 179 | 180 | 591 | 592 | 593 | 594 | 595 | 596 | 597 | 598 | 599 | 600 | |
| Pre swing | AccX | 181 | 182 | 183 | 184 | 185 | 186 | 187 | 188 | 189 | 190 | 601 | 602 | 603 | 604 | 605 | 606 | 607 | 608 | 609 | 610 |
| AccY | 191 | 192 | 193 | 194 | 195 | 196 | 197 | 198 | 199 | 200 | 611 | 612 | 613 | 614 | 615 | 616 | 617 | 618 | 619 | 620 | |
| AccZ | 201 | 202 | 203 | 204 | 205 | 206 | 207 | 208 | 209 | 210 | 621 | 622 | 623 | 624 | 625 | 626 | 627 | 628 | 629 | 630 | |
| GyroX | 211 | 212 | 213 | 214 | 215 | 216 | 217 | 218 | 219 | 220 | 631 | 632 | 633 | 634 | 635 | 636 | 637 | 638 | 639 | 640 | |
| GyroY | 221 | 222 | 223 | 224 | 225 | 226 | 227 | 228 | 229 | 230 | 641 | 642 | 643 | 644 | 645 | 646 | 647 | 648 | 649 | 650 | |
| GyroZ | 231 | 232 | 233 | 234 | 235 | 236 | 237 | 238 | 239 | 240 | 651 | 652 | 653 | 654 | 655 | 656 | 657 | 658 | 659 | 660 | |
| Initial swing | AccX | 241 | 242 | 243 | 244 | 245 | 246 | 247 | 248 | 249 | 250 | 661 | 662 | 663 | 664 | 665 | 666 | 667 | 668 | 669 | 670 |
| AccY | 251 | 252 | 253 | 254 | 255 | 256 | 257 | 258 | 259 | 260 | 671 | 672 | 673 | 674 | 675 | 676 | 677 | 678 | 679 | 680 | |
| AccZ | 261 | 262 | 263 | 264 | 265 | 266 | 267 | 268 | 269 | 270 | 681 | 682 | 683 | 684 | 685 | 686 | 687 | 688 | 689 | 690 | |
| GyroX | 271 | 272 | 273 | 274 | 275 | 276 | 277 | 278 | 279 | 280 | 691 | 692 | 693 | 694 | 695 | 696 | 697 | 698 | 699 | 700 | |
| GyroY | 281 | 282 | 283 | 284 | 285 | 286 | 287 | 288 | 289 | 290 | 701 | 702 | 703 | 704 | 705 | 706 | 707 | 708 | 709 | 710 | |
| GyroZ | 291 | 292 | 293 | 294 | 295 | 296 | 297 | 298 | 299 | 300 | 711 | 712 | 713 | 714 | 715 | 716 | 717 | 718 | 719 | 720 | |
| Mid swing | AccX | 301 | 30 | 303 | 304 | 305 | 306 | 307 | 308 | 309 | 310 | 721 | 722 | 723 | 724 | 725 | 726 | 727 | 728 | 729 | 730 |
| AccY | 311 | 312 | 313 | 314 | 315 | 316 | 317 | 318 | 319 | 320 | 731 | 732 | 733 | 734 | 735 | 736 | 737 | 738 | 739 | 740 | |
| AccZ | 321 | 322 | 323 | 324 | 325 | 326 | 327 | 328 | 329 | 330 | 741 | 742 | 743 | 744 | 745 | 746 | 747 | 748 | 749 | 750 | |
| GyroX | 331 | 332 | 333 | 334 | 335 | 336 | 337 | 338 | 339 | 340 | 751 | 752 | 753 | 754 | 755 | 756 | 757 | 758 | 759 | 760 | |
| GyroY | 341 | 342 | 343 | 344 | 345 | 346 | 347 | 348 | 349 | 350 | 761 | 762 | 763 | 764 | 765 | 766 | 767 | 768 | 769 | 770 | |
| GyroZ | 351 | 352 | 353 | 354 | 355 | 356 | 357 | 358 | 359 | 360 | 771 | 772 | 773 | 774 | 775 | 776 | 777 | 778 | 779 | 780 | |
| Terminal swing | AccX | 361 | 362 | 363 | 364 | 365 | 366 | 367 | 368 | 369 | 370 | 781 | 782 | 783 | 784 | 785 | 786 | 787 | 788 | 789 | 790 |
| AccY | 371 | 372 | 373 | 374 | 375 | 376 | 377 | 378 | 379 | 380 | 791 | 792 | 793 | 794 | 795 | 796 | 797 | 798 | 799 | 800 | |
| AccZ | 381 | 382 | 383 | 384 | 385 | 386 | 387 | 388 | 389 | 390 | 801 | 802 | 803 | 804 | 805 | 806 | 807 | 808 | 809 | 810 | |
| GyroX | 391 | 392 | 393 | 394 | 395 | 396 | 397 | 398 | 399 | 400 | 811 | 812 | 813 | 814 | 815 | 816 | 817 | 818 | 819 | 820 | |
| GyroY | 401 | 402 | 403 | 404 | 405 | 406 | 407 | 408 | 409 | 410 | 821 | 822 | 823 | 824 | 825 | 826 | 827 | 828 | 829 | 830 | |
| GyroZ | 411 | 412 | 413 | 414 | 415 | 416 | 417 | 418 | 419 | 420 | 831 | 832 | 833 | 834 | 835 | 836 | 837 | 838 | 839 | 840 | |
References
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.-P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef]
- Bahat, G.; Tufan, A.; Tufan, F.; Kilic, C.; Akpinar, T.S.; Kose, M.; Erten, N.; Karan, M.A.; Cruz-Jentoft, A.J. Cut-off points to identify sarcopenia according to European Working Group on Sarcopenia in Older People (EWGSOP) definition. Clin. Nutr. 2016, 35, 1557–1563. [Google Scholar] [CrossRef]
- Larsson, L.; Degens, H.; Li, M.; Salviati, L.; Lee, Y.I.; Thompson, W.; Kirkland, J.L.; Sandri, M. Sarcopenia: Aging-Related Loss of Muscle Mass and Function. Physiol. Rev. 2019, 99, 427–511. [Google Scholar] [CrossRef]
- Perez-Sousa, M.A.; Venegas-Sanabria, L.C.; Chavarro-Carvajal, D.A.; Cano-Gutierrez, C.A.; Izquierdo, M.; Correa-Bautista, J.E.; Ramírez-Vélez, R. Gait speed as a mediator of the effect of sarcopenia on dependency in activities of daily living. J. Cachexia Sarcopenia Muscle 2019, 10, 1009–1015. [Google Scholar] [CrossRef]
- De Miguel, K.; Brunete, A.; Hernando, M.; Gambao, E. Home Camera-Based Fall Detection System for the Elderly. Sensors 2017, 17, 2864. [Google Scholar] [CrossRef]
- Zhao, G.; Liu, G.; Li, H.; Pietikainen, M. 3D Gait Recognition Using Multiple Cameras. In Proceedings of the 7th International Conference on Automatic Face and Gesture Recognition (FGR06), Southampton, UK, 10–12 April 2006; pp. 529–534. [Google Scholar]
- Rashid, U.; Kumari, N.; Taylor, D.; David, T.; Signal, N. Gait Event Anomaly Detection and Correction During a Split-Belt Treadmill Task. IEEE Access 2019, 7, 68469–68478. [Google Scholar] [CrossRef]
- Seel, T.; Raisch, J.; Schauer, T. IMU-Based Joint Angle Measurement for Gait Analysis. Sensors 2014, 14, 6891–6909. [Google Scholar] [CrossRef]
- Panahandeh, G.; Mohammadiha, N.; Leijon, A.; Händel, P. Continuous Hidden Markov Model for Pedestrian Activity Clas-sification and Gait Analysis. IEEE Trans. Instrum. Meas. 2013, 62, 1073–1083. [Google Scholar] [CrossRef]
- Wang, Z.; Ji, R. Estimate spatial-temporal parameters of human gait using inertial sensors. In Proceedings of the 2015 IEEE International Conference on Cyber Technology in Automation, Control, and Intelligent Systems (CYBER), Shenyang, China, 8–12 June 2015; pp. 1883–1888. [Google Scholar]
- Kyritsis, K.; Tatli, C.L.; Diou, C.; Delopoulos, A. Automated analysis of in meal eating behavior using a commercial wristband IMU sensor. In Proceedings of the 2017 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Jeju, Korea, 11–15 July 2017; pp. 2843–2846. [Google Scholar]
- Dindorf, C.; Teufl, W.; Taetz, B.; Bleser, G.; Fröhlich, M. Interpretability of Input Representations for Gait Classification in Patients after Total Hip Arthroplasty. Sensors 2020, 20, 4385. [Google Scholar] [CrossRef]
- Taborri, J.; Palermo, E.; Rossi, S.; Cappa, P. Gait Partitioning Methods: A Systematic Review. Sensors 2016, 16, 66. [Google Scholar] [CrossRef]
- Levine, D.; Richards, J.; Whittle, M.W. Whittle’s Gait Analysis-E-Book, 5th ed.; Churchill Livingstone: London, UK, 2012. [Google Scholar]
- Misu, S.; Asai, T.; Ono, R.; Sawa, R.; Tsutsumimoto, K.; Ando, H.; Doi, T. Development and validity of methods for the esti-mation of temporal gait parameters from heel-attached inertial sensors in younger and older adults. Gait Posture 2017, 57, 295–298. [Google Scholar] [CrossRef]
- Mo, S.; Chow, D.H. Accuracy of three methods in gait event detection during overground running. Gait Posture 2018, 59, 93–98. [Google Scholar] [CrossRef]
- Khandelwal, S.; Wickström, N. Gait event detection in real-world environment for long-term applications: Incorporating domain knowledge into time-frequency analysis. IEEE Trans. Neural Syst. Rehabil. Eng. 2016, 24, 1363–1372. [Google Scholar] [CrossRef]
- Kim, J.; Bae, M.-N.; Lee, K.B.; Hong, S.G. Gait event detection algorithm based on smart insoles. ETRI J. 2019, 42, 46–53. [Google Scholar] [CrossRef]
- Perez-Ibarra, J.C.; Siqueira, A.A.G.; Krebs, H.I. Real-Time Identification of Gait Events in Impaired Subjects Using a Single-IMU Foot-Mounted Device. IEEE Sens. J. 2019, 20, 2616–2624. [Google Scholar] [CrossRef]
- Zhao, H.; Wang, Z.; Qiu, S.; Wang, J.; Xu, F.; Wang, Z.; Shen, Y. Adaptive gait detection based on foot-mounted inertial sensors and multi-sensor fusion. Inf. Fusion 2019, 52, 157–166. [Google Scholar] [CrossRef]
- Seel, T.; Werner, C.; Raisch, J.; Schauer, T. Iterative learning control of a drop foot neuroprosthesis—Generating physiological foot motion in paretic gait by automatic feedback control. Control Eng. Pract. 2016, 48, 87–97. [Google Scholar] [CrossRef]
- Gouwanda, D.; Gopalai, A.A.; Khoo, B.H. A Low Cost Alternative to Monitor Human Gait Temporal Parameters–Wearable Wireless Gyroscope. IEEE Sens. J. 2016, 16, 9029–9035. [Google Scholar] [CrossRef]
- Rueterbories, J.; Spaich, E.G.; Andersen, O.K. Gait event detection for use in FES rehabilitation by radial and tangential foot accelerations. Med. Eng. Phys. 2014, 36, 502–508. [Google Scholar] [CrossRef]
- Mariani, B.; Rouhani, H.; Crevoisier, X.; Aminian, K. Quantitative estimation of foot-flat and stance phase of gait using foot-worn inertial sensors. Gait Posture 2013, 37, 229–234. [Google Scholar] [CrossRef]
- Sabatini, A.M.; Martelloni, C.; Scapellato, S.; Cavallo, F. Assessment of Walking Features from Foot Inertial Sensing. IEEE Trans. Biomed. Eng. 2005, 52, 486–494. [Google Scholar] [CrossRef]
- Kang, D.W.; Choi, J.S.; Kim, H.S.; Oh, H.S.; Seo, J.W.; Lee, J.W.; Tack, G.R. Wireless gait event detection system based on single gyroscope. In Proceedings of the 6th International Conference on Ubiquitous Information Management and Communication, Kuala Lumpur, Malaysia, 20–22 February 2012; p. 88. [Google Scholar]
- Mannini, A.; Genovese, V.; Sabatini, A.M. Online Decoding of Hidden Markov Models for Gait Event Detection Using Foot-Mounted Gyroscopes. IEEE J. Biomed. Health Inform. 2013, 18, 1122–1130. [Google Scholar] [CrossRef]
- Abaid, N.; Cappa, P.; Palermo, E.; Petrarca, M.; Porfiri, M. Gait detection in children with and without hemiplegia using sin-gle-axis wearable gyroscopes. PLoS ONE 2013, 8, e73152. [Google Scholar] [CrossRef]
- Teufl, W.; Taetz, B.; Miezal, M.; Lorenz, M.; Pietschmann, J.; Jöllenbeck, T.; Fröhlich, M.; Bleser, G. Towards an Inertial Sen-sor-Based Wearable Feedback System for Patients after Total Hip Arthroplasty: Validity and Applicability for Gait Classification with Gait Kinematics-Based Features. Sensors 2019, 19, 5006. [Google Scholar] [CrossRef]
- Caramia, C.; Torricelli, D.; Schmid, M.; Muñoz-Gonzalez, A.; Gonzalez-Vargas, J.; Grandas, F.; Pons, J.L. IMU-based classi-fication of Parkinson’s disease from gait: A sensitivity analysis on sensor location and feature selection. IEEE J. Biomed. Health Inform. 2018, 22, 1765–1774. [Google Scholar] [CrossRef]
- Howcroft, J.; Kofman, J.; Lemaire, E.D. Prospective Fall-Risk Prediction Models for Older Adults Based on Wearable Sensors. IEEE Trans. Neural Syst. Rehabil. Eng. 2017, 25, 1812–1820. [Google Scholar] [CrossRef]
- Eskofier, B.M.; Lee, S.I.; Daneault, J.-F.; Golabchi, F.N.; Ferreira-Carvalho, G.; Vergara-Diaz, G.; Sapienza, S.; Costante, G.; Klucken, J.; Kautz, T.; et al. Recent machine learning advancements in sensor-based mobility analysis: Deep learning for Parkinson’s disease assessment. In Proceedings of the 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 16–20 August 2016; pp. 655–658. [Google Scholar]
- Tunca, C.; Salur, G.; Ersoy, C. Deep learning for fall risk assessment with inertial sensors: Utilizing domain knowledge in spa-tio-temporal gait parameters. IEEE J. Biomed. Health Inform. 2019, 24, 1994–2005. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Romijnders, R.; Hansen, C.; Van Campen, J.; Maetzler, W.; Hortobágyi, T.; Lamoth, C.J.C. The detection of age groups by dynamic gait outcomes using machine learning approaches. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Rantalainen, T.; Pirkola, H.; Karavirta, L.; Rantanen, T.; Linnamo, V. Reliability and concurrent validity of spatiotemporal stride characteristics measured with an ankle-worn sensor among older individuals. Gait Posture 2019, 74, 33–39. [Google Scholar] [CrossRef]
- Cho, Y.-S.; Jang, S.-H.; Cho, J.-S.; Kim, M.-J.; Lee, H.D.; Lee, S.Y.; Moon, S.-B. Evaluation of Validity and Reliability of Inertial Measurement Unit-Based Gait Analysis Systems. Ann. Rehabil. Med. 2018, 42, 872–883. [Google Scholar] [CrossRef] [PubMed]
- Washabaugh, E.P.; Kalyanaraman, T.; Adamczyk, P.G.; Claflin, E.S.; Krishnan, C. Validity and repeatability of inertial measurement units for measuring gait parameters. Gait Posture 2017, 55, 87–93. [Google Scholar] [CrossRef]
- Nilsson, J.-O.; Gupta, A.K.; Handel, P. Foot-mounted inertial navigation made easy. In Proceedings of the 2014 International Conference on Indoor Positioning and Indoor Navigation (IPIN), Busan, Korea, 27–30 October 2014; pp. 24–29. [Google Scholar]
- Skog, I.; Nilsson, J.-O.; Handel, P. Pedestrian tracking using an IMU array. In Proceedings of the 2014 IEEE International Conference on Electronics, Computing and Communication Technologies (CONECCT), Bangalore, India, 6–7 January 2014; pp. 1–4. [Google Scholar]
- Adadi, A.; Berrada, M. Peeking Inside the Black-Box: A Survey on Explainable Artificial Intelligence (XAI). IEEE Access 2018, 6, 52138–52160. [Google Scholar] [CrossRef]
- Lundberg, S.M.; Erion, G.; Chen, H.; DeGrave, A.; Prutkin, J.M.; Nair, B.; Katz, R.; Himmelfarb, J.; Bansal, N.; Lee, S.I. Ex-plainable AI for trees: From local explanations to global understanding. arXiv 2019, arXiv:1905.04610. [Google Scholar]
- Chen, T.; Guestrin, C. Xgboost: A scalable tree boosting system. In Proceedings of the 22nd acm sigkdd international conference on knowledge discovery and data mining, San Francisco, CA, USA, 13–17 August 2016; pp. 785–794. [Google Scholar]
- Friedman, J.H. Greedy function approximation: A gradient boosting machine. Ann. Stat. 2001, 29, 1189–1232. [Google Scholar] [CrossRef]
- Chen, C.; Liaw, A.; Breiman, L. Using Random Forest to Learn Imbalanced Data. University of California, Berkeley. 2004. Available online: https://statistics.berkeley.edu/sites/default/files/tech-reports/666.pdf (accessed on 3 March 2021).
- Suykens, J.A.K.; Vandewalle, J. Least Squares Support Vector Machine Classifiers. Neural Process. Lett. 1999, 9, 293–300. [Google Scholar] [CrossRef]
- Gardner, M.W.; Dorling, S.R. Artificial neural networks (the multilayer perceptron)—A review of applications in the atmos-pheric sciences. Atmos. Environ. 1998, 32, 2627–2636. [Google Scholar] [CrossRef]
- Kalchbrenner, N.; Grefenstette, E.; Blunsom, P. A Convolutional Neural Network for Modelling Sentences. In Proceedings of the 52nd Annual Meeting of the Association for Computational Linguistics (Volume 1: Long Papers), Baltimore, MD, USA, 22–27 June 2014; pp. 655–665. [Google Scholar]
- Zaremba, W.; Sutskever, I.; Vinyals, O. Recurrent neural network regularization. arXiv 2014, arXiv:1409.2329. [Google Scholar]
- Hopfield, J.J. Hopfield network. Scholarpedia 2007, 2. [Google Scholar] [CrossRef]
- Hochreiter, S.; Schmidhuber, J. Long Short-Term Memory. Neural Comput. 1997, 9, 1735–1780. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Xu, R.; He, Y.; Wang, X. Improving sentiment analysis via sentence type classification using BiLSTM-CRF and CNN. Expert Syst. Appl. 2017, 72, 221–230. [Google Scholar] [CrossRef]
- Hida, T.; Harada, A. Fall risk and fracture. Diagnosing sarcopenia and sarcopenic leg to prevent fall and fracture: Its difficulty and pit falls. Clin. Calcium 2013, 23, 707–712. [Google Scholar]
| First Author, Year | Phase/Events | Signals | Algorithm Class | Position |
|---|---|---|---|---|
| Seel et al., 2016 [21] | 4/HS-ST-HO-SW | 3 Acc + 3 Gyro | Rule-based | Forefoot |
| Gouwanda et al., 2016 [22] | 3/HS-HO-MSw | 1 Gyro | Rule-based | Forefoot |
| Rueterbories et al., 2014 [23] | 4/HS-FF-HO-TO | 2 Acc | Rule-based | Forefoot |
| Mariani et al., 2013 [24] | 4/HS-TS-HO-TO | 3 Acc + 3 Gyro | Rule-based | Forefoot |
| Sabatini et al., 2005 [25] | 4/HS-ST-HO-SW | 1 Gyro | Rule-based | Forefoot |
| Kang et al., 2012 [26] | 4/HS-FF-HO-TO | 1 Gyro | Rule-based | Forefoot |
| Mannini et al., 2014 [27] | 4/HS-FF-HO-TO | 3 Gyro | HMM-based | Forefoot |
| Abaid et al., 2013 [28] | 4/HS-FF-HO-TO | 3 Gyro | HMM-based | Forefoot |
| Zhao et al., 2019 [20] | 4/HS-FF-HO-TO | 3 Acc + 3 Gyro | HMM-based | Hindfoot |
| Pérez-Ibarra et al., 2019 [19] | 4/HS-TS-HO-TO | 1 Gyro | Rule-based | Hindfoot |
| Ours | 7/HS-oTO-HR-oHS-TO-FA-TV | 1 Acc + 1 Gyro | Rule-based | Hindfoot |
| First Author, Year | Phase/Events | Signals | Position | Classification | Accuracy |
|---|---|---|---|---|---|
| Teufl et al., 2019 [29] | Stride length, stride time, cadence, speed, ROM (hip, pelvis) | THA | Hip, thigh, shank, foot | SVM | 97% |
| Caramia et al., 2018 [30] | Step length, step time, stride time, speed, Rom (hip, knee, ankle) | PD | R&L ankle, knee, hip, chest | LDA, NB, k-NN, SVM, SVM rbf, DT, majority of votes | 96.88% |
| Howcroft et al., 2017 [31] | Cadence, stride time maximum, mean, and standard deviation of acceleration | Faller | Head, pelvis, right left shank | NB, SVM, NN | 57% |
| Eskofier et al., 2016 [32] | Energy, maximum, minimum, mean, variance, skewness, kurtosis, fast Fourier transform | PD | Upper limbs | AdaBoost, PART, kNN, SVM, CNN | 90.9% |
| Tunca et al., 2019 [33] | Stride length, cycle time, stance time, swing time, clearance, stance ratio, cadence, speed | Faller | Both feet | SVM, RF, MLP, HMM, LSTM | 94.30% |
| Zhou et al., 2020 [34] | Root mean square, cross entropy, Lyapunov exponent, step regularity, gait speed | Age groups by dynamic gait outcomes | Trunk | SVM, RF, ANN | 90% |
| Dindorf et al., 2020 [12] | Various parameters | THA | Hip, knee, pelvis, ankle | RF, SVM, SVM rbf, MLP | 100% |
| Ours | Various parameters | Sarcopenia | Both feet | RF, SVM, MLP, CNN, BiLSTM | 95% |
| Parameter | Normal | Sarcopenia |
|---|---|---|
| Age (years) | 69.6 ± 3.0 | 71.1 ± 2.0 |
| Height (cm) | 153.5 ± 5.0 | 151.0 ± 4.8 |
| Weight (kg) | 60.8 ± 5.1 | 52.7 ± 5.0 |
| Feet size (mm) | 238.0 ± 5.1 | 232.0 ± 5.5 |
| Grasp power right (kg) | 22.5 ± 2.6 | 14.4 ± 3.5 |
| Grasp power left (kg) | 22.3 ± 2.8 | 14.2 ± 3.7 |
| ASM (kg) | 14.7 ± 1.6 | 11.3 ± 0.9 |
| LMI (kg/m2) | 6.3 ± 0.4 | 4.9 ± 0.2 |
| Gait Parameters | Definition |
|---|---|
| Spatial-temporal Parameters | |
| Cadence | Number of steps acquired per minute |
| Stance phase (time) | Percent (time) starting with HS and ending with TO of the same foot |
| Swing phase (time) | Percent (time) starting with TO and ending with HS of the same foot |
| Single support phase (time) | Percent (time) when only one foot is on the ground |
| Double support phase (time) | Percent (time) when both feet are on the ground |
| Step time (length) | Distance by which a foot moves in front of the other foot. The sum of two successive step lengths corresponds to stride length |
| Stride length | Distance starting with HS and ending with next HS of the same foot |
| Phase (time) dRL | Absolute values of the difference between the right and left sides of the stance and swing phases (time) |
| Speed | Stride length/cycle time |
| Descriptive Statistical Parameters | |
| Max | Greatest values |
| Min | Least or smallest values |
| Standard deviation (STD) | Standard deviation of values |
| AbSum | Absolute sum of values |
| Root-mean-square (RMS) | Arithmetic mean of the squares of a set of values |
| Kurtosis | Assesses whether the tails of a given distribution contain extreme values |
| Skewness | A measure of the asymmetry of the probability distribution of a real-valued random variable about its mean |
| MMgr | Gradient from maximum to minimum of values |
| DMM | Difference between max and min of values |
| Mdif | Maximum for the difference between two successive values |
| Parameters | SVM | RF | MLP |
|---|---|---|---|
| Spatial-temporal (23) | 65 (21.08) | 75 (24.47) | 77 (21.1) |
| Two phases descriptive statistics (2 × 12 × 10) | 50 (27.76) | 52.5 (24.47) | 50 (11.25) |
| Two phases descriptive statistics (top 20) | 80 (22.20) | 77.5 (25.81) | 77.5 (24.39) |
| Seven phases descriptive statistics (7 × 12 × 10) | 48.75 (3.95) | 60 (27.66) | 66.3 (17.5) |
| Seven phases descriptive statistics (top 20) | 95 (15.81) | 85 (20.28) | 90 (20) |
| Parameter | CNN | BiLSTM |
|---|---|---|
| Raw IMU data (100 × 12) | 54.2 (20.72) | 45.3 (17.50) |
| Spatial-temporal (23) | 74.76 (24.01) | 62.5 (30.33) |
| Two phases descriptive statistics (top 20) | 69.75 (22.53) | 56.25 (16.05) |
| Seven phases descriptive statistics (top 20) | 87.5 (19.36) | 86.26 (19.72) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.-K.; Bae, M.-N.; Lee, K.B.; Hong, S.G. Identification of Patients with Sarcopenia Using Gait Parameters Based on Inertial Sensors. Sensors 2021, 21, 1786. https://doi.org/10.3390/s21051786
Kim J-K, Bae M-N, Lee KB, Hong SG. Identification of Patients with Sarcopenia Using Gait Parameters Based on Inertial Sensors. Sensors. 2021; 21(5):1786. https://doi.org/10.3390/s21051786
Chicago/Turabian StyleKim, Jeong-Kyun, Myung-Nam Bae, Kang Bok Lee, and Sang Gi Hong. 2021. "Identification of Patients with Sarcopenia Using Gait Parameters Based on Inertial Sensors" Sensors 21, no. 5: 1786. https://doi.org/10.3390/s21051786
APA StyleKim, J.-K., Bae, M.-N., Lee, K. B., & Hong, S. G. (2021). Identification of Patients with Sarcopenia Using Gait Parameters Based on Inertial Sensors. Sensors, 21(5), 1786. https://doi.org/10.3390/s21051786









