Abstract
Bacterial communities in diverse environmental niches respond to various external stimuli for survival. A primary means of communication between bacterial cells involves one-component (OC) and two-component signal transduction systems (TCSs). These systems are key for sensing environmental changes and regulating bacterial physiology. TCSs, which are the more complex of the two, consist of a sensor histidine kinase for receiving an external input and a response regulator to convey changes in bacterial cell physiology. For numerous reasons, TCSs have emerged as significant targets for antibacterial drug design due to their role in regulating expression level, bacterial viability, growth, and virulence. Diverse studies have shown the molecular mechanisms by which TCSs regulate virulence and antibiotic resistance in pathogenic bacteria. In this study, we performed a thorough analysis of the data from multiple public databases to assemble a comprehensive catalog of the principal detection systems present in both the non-pathogenic Pseudomonas putida KT2440 and the pathogenic Pseudomonas aeruginosa PAO1 strains. Additionally, we conducted a sequence analysis of regulatory elements associated with transcriptional proteins. These were classified into regulatory families based on Helix-turn-Helix (HTH) protein domain information, a common structural motif for DNA-binding proteins. Moreover, we highlight the function of bacterial TCSs and their involvement in functions essential for bacterial survival and virulence. This comparison aims to identify novel targets that can be exploited for the development of advanced biotherapeutic strategies, potentially leading to new treatments for bacterial infections.
1. Introduction
Bacteria are highly adaptable organisms, capable of quickly adjusting to environmental changes by regulating their metabolism and gene expression [1]. To this end, they have evolved several features, including signal transduction systems that enable intracellular regulation and facilitate crosstalk between the intracellular and extracellular environments. However, this same adaptability has also contributed to the recent emergence of bacterial strains with multidrug resistance (MDR) phenotypes, which has become a serious health problem worldwide, constituting one of the main causes of human mortality [2].
Pseudomonas is a Gram-negative bacterial genus commonly found in soil, water, and plants, with species distributed globally [3]. They are widespread in the environment due to their exceptional adaptive capacity [4]. While some Pseudomonas species are generally harmless, others can cause infections in humans, animals, and plants [5], especially, in patients with compromised immune systems or those with medical devices, like catheters or prosthetic joints. Pseudomonas infections can range from mild skin to severe respiratory infections due to the genus’s antibiotic resistance [6,7]. On the other hand, certain strains within this genus have been successfully used in bioremediation control and industrial applications, showcasing their ability to degrade a wide range of organic compounds [8,9,10,11]. For example, Pseudomonas putida KT2440 is known for its beneficial effects on plants, due to its plant growth-promoting capabilities and its ability to efficiently colonize plant roots [12,13]. In contrast, other species, such as Pseudomonas aeruginosa, are versatile and opportunistic nosocomial pathogens that can infect both animals and plants [3,14].
Pseudomonas aeruginosa is a common cause of hospital-acquired infections, including ventilator-associated pneumonia [15] and catheter infections, particularly in immunocompromised patients and those suffering from cystic fibrosis (CF) [16] and severe burns [17,18]. Cystic fibrosis is a genetic disorder caused by mutations in the cystic fibrosis transmembrane conductance regulator gene (CFTR), which plays a crucial role in epithelial cell function and the regulation of epithelial fluid transport in the airways and other organs. The defective CFTR impairs normal airway function and disrupts the epithelium’s ability to interact effectively with pathogens. This dysfunction activates Toll-like receptors (TLRs), triggering downstream signaling via the MyD88-dependent pathway and subsequent activation of the NF-κB pathway, leading to the production of inflammatory cytokines, such as IL-6 and IL-8. This inflammatory response, illustrated in Figure 1, is a critical component of the body’s defense against bacterial infections, including those caused by P. aeruginosa, which frequently colonizes the lungs of cystic fibrosis patients. In addition to its direct effects on airway epithelial cells, CFTR dysfunction also contributes to systemic issues, such as intestinal barrier dysfunction [19,20], and alterations in the intestinal microbiota (dysbiosis). These changes are exacerbated by factors such as prolonged antibiotic use, obesity, diabetes, and fatty liver, promoting P. aeruginosa colonization in the intestinal ecosystem [21,22]. The persistence of P. aeruginosa infections in cystic fibrosis patients can be attributed to the bacterium’s adaptive capabilities acquired through mutation [23,24] and horizontal gene transfer [24,25], as well as its proficiency in biofilm formation [26,27,28] and the production of virulence factors, like Exotoxin A (ToxA) [29,30]. ToxA, found in over 90% of clinical isolates, disrupts protein synthesis by ADP ribosylation of elongation factor 2 (EF2), leading to cell death [31,32,33,34]. Furthermore, P. aeruginosa utilizes various secretion systems to deliver virulence factors either extracellularly or directly into host cells, enhancing bacterial survival and replication in the hostile environment of cystic fibrosis-affected tissues. These secretion systems range from type I to type VI secretion systems (T1SS to T6SS), enabling the bacterium to evade immune detection and maintain chronic infections in cystic fibrosis patients [35,36,37].
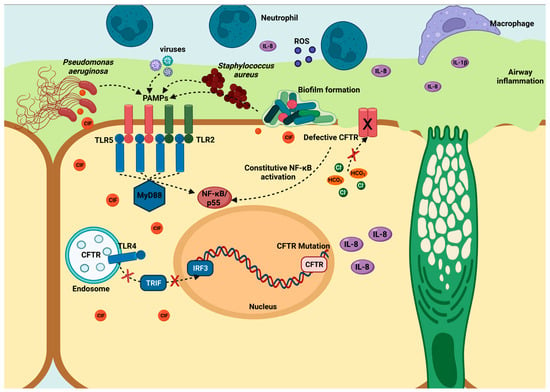
Figure 1.
Airway epithelial adaptation of pathogens in cystic fibrosis and chronic obstructive pulmonary disease. TLRs’ activation (TLR2, TLR4, and TLR5) triggers MyD88-dependent and -independent responses. The MyD88-dependent pathway involves TIRAP and leads to NF-κB activation, resulting in cytokine production (IL-8, TNF-α). In contrast, the MyD88-independent pathway, mediated by TRIF, activates IRF3 and also contributes to immune responses. Defective CFTR in cystic fibrosis disrupts ion transport, leading to constitutive NF-κB activation, chronic inflammation, and cytokine production, contributing to disease pathogenesis.
Nowadays, most antibiotics are discovered through screening processes designed to identify substances that inhibit bacterial growth [38]. However, these processes typically target a limited range of cellular activities. Bacteria develop resistance primarily through efflux pumps, which expel antibiotics and enzymes, such as beta-lactamases, which modify the drugs [39]. Additionally, strains like P. aeruginosa PAO1 and P. putida KT2440 have evolved complex signal transduction systems that detect and respond to environmental signals, enabling them to alter their cellular makeup in response to changing conditions.
These systems allow the bacteria to sense external stimuli, including the presence of signaling molecules in the environment. Upon detection, the systems efficiently relay these signals into the cell, initiating a cellular response that allows the bacteria to modify their cellular composition appropriately.
A prior analysis encompassing over 1000 genomes of diverse bacterial species revealed that signal transduction in bacteria is primarily mediated by proteins from two major superfamilies: one-component regulatory systems (OCSs) and two-component regulatory systems (TCSs) (Figure 2) [40,41,42,43,44,45,46]. In the context of TCSs, a transcriptional regulator generally consists of a histidine kinase (HK) protein or sensor, and a cognate response regulatory protein (R), which together modulate diverse signal transduction pathways (Figure 2). Sensor HKs possess multiple domains, including a periplasmic sensor domain for recognizing specific signals, a signal transduction domain, a cytoplasmic sensor domain, an adenosine triphosphate (ATP) catalytic domain, and a dimerization histidine phosphotransfer domain (D). The distribution of sensor proteins has been shown to vary, even among closely related microorganisms [43].
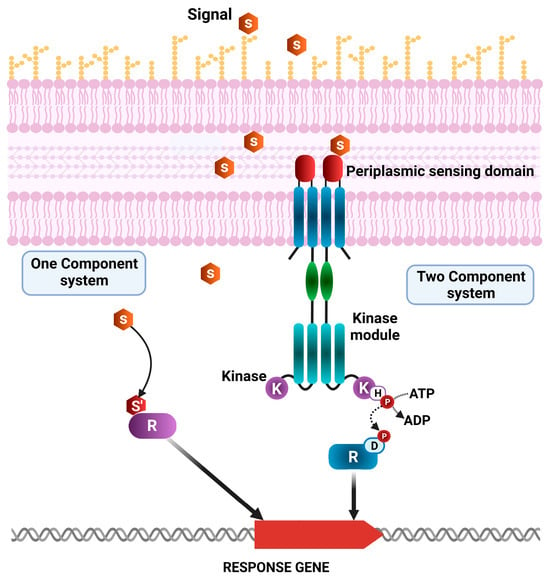
Figure 2.
Schematic representation of one-component system (OCS) and two-component system (TCS) classes of P. putida KT2440 and P. aeruginosa PAO1 transcriptional regulators. R: Response regulatory protein; E: ligand-binding domain; S: signal molecules; K: histidine kinase, involved in phosphorylation/dephosphorylation events that activate the response regulator.
Upon the detection of a stimulus through the periplasmic or cytoplasmic detection domains, the HK sensor initiates the autophosphorylation of the conserved histidine residue within the HK domain. The phosphate group is then transferred to the aspartate residue of the R protein, which modulates the expression of genes involved in cellular responses. The effector domain of the R protein undergoes a conformational change upon phosphorylation, enabling it to bind or release DNA, thereby initiating changes in gene expression [44,45,46,47,48,49]. The responses elicited by these signal transduction systems can be highly diverse. In addition to regulating gene expression, certain response domains also mediate interactions with RNA, small ligands, other proteins, or exhibit enzymatic or transporter activities. However, one of the greatest challenges in understanding the complex bacterial signal transduction networks lies in identifying the signaling molecules that bind to sensor domains, initiating the signaling cascade [50].
In contrast, OCSs consist of proteins encompassing both a sensory and a DNA-binding domain but lacking the histidine kinase domains and extracellular receptors (Figure 2). Therefore, in these systems, a single protein performs both the detection of the intracellular signal and the initiation of the cellular response. Despite their simpler structure, these systems are more ancient and broadly distributed than TCSs. Both types of systems display a similar range of input and output domains. Hence, besides their structural differences, they may be able to detect similar stimuli and activate comparable cellular responses.
In this study, we analyzed the data from various public databases to compile an updated and detailed catalog of the main detection systems found in both the non-pathogenic P. putida KT2440 and the pathogenic P. aeruginosa PAO1 strains. Our primary goal was to delineate the significant distinctions in the protein components of signal transduction systems between these pathogenic and non-pathogenic bacteria. This comparison aims to elucidate the biological mechanisms that underpin the development of pathogenic traits.
Furthermore, we identified and cataloged several transcriptional regulators exclusive to the genome of P. aeruginosa, highlighting their pivotal roles in promoting bacterial virulence. This paper explores the unique attributes of these regulators and their potential as targets for therapeutic intervention. By linking the genomic characteristics of these transcriptional regulators to pathogenic traits, our research provides valuable insights for the development of novel therapeutic strategies.
2. Results and Discussion
2.1. Understanding the Genomic Sequences of P. aeruginosa PAO1 and P. putida KT2440: An Overview
The strain P. putida KT2440 (GCA_000007565.2) has a genome size of 6.2 Mbp with 5693 predicted genes (Figure 3A) [51,52], and P. aeruginosa PAO1 (GCA_000006765.1) boasts a genome spanning over 6.3 Mbp housing a predicted set of 5697 genes (Figure 3B) [53] Notably, P. aeruginosa PAO1 showcases a predicted array of 381 proteins falling under the transcription regulator classification, from which 44 identified as extra-cytoplasmic function (ECF) sigma factors [54]. The transcriptional activity has great relevance in this organism because many TCSs that respond to environmental conditions confer upon this bacterium the status of an infectious agent. The complex regulatory interplays within P. aeruginosa can be effectively visualized in the form of a transcriptional regulatory network.
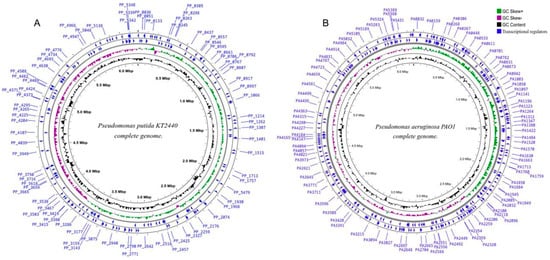
Figure 3.
Circular genome analysis of (A) Pseudomonas putida KT2440 and (B) Pseudomonas aeruginosa PAO1 using the CGView tool (http://genocat.tools/tools/cgview_server.html) (Version not specified). The blue circus ring represents the distribution of genes identified as transcriptional regulatory.
2.2. The Set of DNA-Binding Transcriptional Regulators in the Non-Pathogenic P. putida KT2440 and Pathogenic P. aeruginosa PAO1 Strains
Understanding and linking gene expression to regulatory and physiological properties in bacterial strains becomes more achievable by identifying and characterizing the set of DNA-binding transcriptional regulators. Therefore, the genome sequences of both the environmental strain P. putida KT2440 and the opportunistic pathogen P. aeruginosa PAO1 were examined to detect genes encoding transcriptional regulators. Our data collection analysis revealed a total of 319 and 381 genes from transcriptional regulators (Figure 4) in the genomes of P. putida KT2440 and P. aeruginosa PAO1 (Tables S1 and S2), respectively.
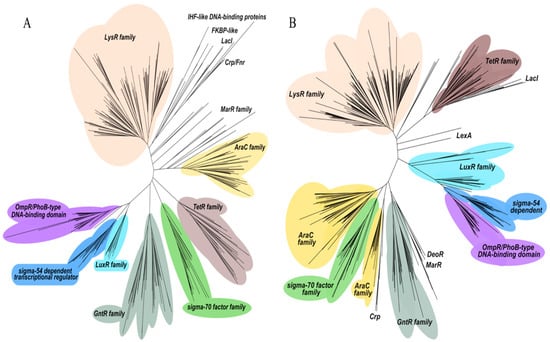
Figure 4.
Pseudomonas putida KT2440 (A) and Pseudomonas aeruginosa PAO1 (B) phylogenetic tree representation of transcriptional regulators of the Helix-turn-Helix family. The colored area corresponds to sequences with an HTH family domain.
Notably, a total of 27 and 44 genes of P. putida KT2440 and P. aeruginosa PAO1, respectively, were annotated as sigma factors. However, only 285 genes in P. putida KT2440 (Figure 4A and Table S1) and 331 genes in P. aeruginosa PAO1 (Figure 4B and Table S2) were classified within the HTH family (Helix-Turn-Helix). Interestingly, while the strain KT2440 contains 44 genes associated with TCSs, strain PAO1 harbors 81 genes associated with these systems.
2.3. Categorization of Transcriptional Regulators in P. putida KT2440 and P. aeruginosa PAO1 into Regulatory Protein Families
Transcriptional regulators can be grouped into evolutionary regulatory protein families based on their amino acid sequence similarities [55]. Using amino acid sequence alignments obtained from the KEGG database, we categorized the complete collection of transcriptional regulators in P. putida and P. aeruginosa into 16 HTH-characterized protein families (Table S3). This included 120 regulators from KT2440, 142 from PAO1, and 18 from another unclassified HTH family (Figure 5 and Table S3). The majority of the identified regulatory protein families show a uniform size distribution among their assigned members, indicating a high degree of amino acid sequence similarity and suggesting a shared evolutionary origin. The number of identified regulatory protein families varies significantly, with the LysR family being the largest family with up to 20 and 26 members in P. putida KT2440 and P. aeruginosa PAO1, respectively (Table S3). In contrast, some protein families, like GntR, DeoR, LuxR, MerR, FuR, IcIR, CRp/FNR, RrF2, CopR, and ArsR, have as few as one-to-three members (Figure 5).
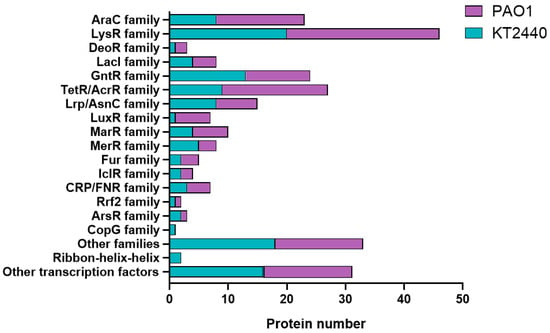
Figure 5.
Categorization of DNA-binding transcriptional regulators in P. putida KT2440 and P. aeruginosa PAO1 into distinct regulatory protein families. The names of the identified regulatory protein families are shown. These families are labeled based on designations from the Pfam database.
The LysR and GntR families of transcriptional regulators are extensively found in prokaryotes, including P. putida and P. aeruginosa strains. These families share a conserved structure comprising two functional domains: a conserved N-terminal DNA-binding Helix-Turn-Helix (HTH) motif and a C-terminal coinducer-binding or oligomerization domain. Typically, they exhibit negative autoregulation and are involved in the activation of a single divergently transcribed gene, influencing various cellular processes, like cell motility, glucose metabolism, bacterial resistance, pathogenesis, and virulence [56,57]. Significant distinctions in the transcriptional regulatory repertoire between pathogenic P. aeruginosa and non-pathogenic P. putida KT2440 are evident when comparing the number of proteins categorized into the TetR/AcrR, AraC, and LuxR families (Figure 5 and Table S3). The TetR/AcrR protein families are significantly prevalent in both Pseudomonas bacterial genomes. These families constitute a large group of OCS proteins that play a crucial role in regulating various processes, including efflux regulation, cell division, and stress responses processes critical for antimicrobial resistance [58,59].
Additionally, TetR/AcrR belongs to a transcriptional regulator family made up of two-domain proteins: an N-terminal HTH DNA-binding motif and a C-terminal ligand recognition domain. This design enables this regulator to recognize chemical sensors, to monitor various aspects of cellular environmental dynamics, such as antibiotics production, carbohydrate metabolism, osmotic stress, efflux pumps function, multidrug resistance, cellular virulence, and biofilm formation [58,60]. This diversity in DNA-binding transcriptional regulators certainly makes sense, given their role as chemical sensors or responding to environmental fluctuations. The prevalence of proteins in the GntR family may provide these bacteria the ability to thrive on various carbon sources and promptly adapt their gene expression in response to environmental shifts. Furthermore, this might indicate that pathogenic bacteria require a less versatile carbohydrate metabolism, as their natural habitats provide limited access to a diverse range of carbon sources.
It is noteworthy that alterations in the number of TetR/AcrR chemical regulatory sensors might correlate to variations in environmental conditions. For instance, pathogenic bacteria often tend to import the compound directly from the cell instead of relying on external sources. In particular, the number of regulatory proteins belonging to the TetR family is notably higher in the pathogenic species (Figure 5 and Table S3). This finding suggests that the TetR repressor family can serve as a universal switch for governing gene expression in P. aeruginosa, crucial for its more complex lifestyle and the need to regulate a wider array of genes associated with pathogenicity and resistance.
2.4. A Differential Repertoire Transcriptional Regulator Protein Is Evident in Pathogenic and Non-Pathogenic Pseudomonas Strains
As evidenced in Table S4, both pathogenic and non-pathogenic strains exhibit a similar number of genes encoding regulatory proteins, with 103 in KT2440 and 106 genes in PAO1. Additionally, our analysis identified a collection of 69 TCSs transcriptional regulators genes in KT2440, while the chromosomal sequences of PAO1 revealed 90 TCSs transcriptional regulator genes (Table 1). These findings suggest that the presence of TCSs is generally linked to a greater complexity in the regulation of gene expression, a phenomenon likely shaped by bacterial evolution.

Table 1.
Categorization list of two-component system genes found in P. putida KT2440 and P. aeruginosa PAO1 and their corresponding families. nd: Not identified.
2.5. Two-Component System Function in Pathogenic and Non-Pathogenic Pseudomonas Strains
The ability of bacterial strains to respond to external stimuli is mediated by a specialized signal transduction mechanism, which relies on TCSs [47,48]. When activated, the sensor protein within the TCS catalyzes the autophosphorylation of a conserved histidine residue using adenosine triphosphate (ATP). The phosphoryl group is then transferred to a conserved aspartate residue in the regulatory protein (R), which may alter its ability to bind DNA sequences [45,47]. In recent years, a number of techniques have been developed to study two-component signal transduction systems [61], enabling the identification of stimuli-responsive TCSs. It has also been reported that some TCSs regulate gene clusters that contribute to cell growth, biofilm formation, and virulence in pathogenic bacteria [50,62,63,64]. However, in several cases, the role of TCSs in bacterial pathogenicity remains poorly understood. For instance, while TCS mutant strains often display attenuated virulence, the precise mechanisms underlying this effect are not yet fully elucidated. Functional analyses are essential for definitively determining the role of these sensors in the early stages of the infection process. Our findings suggest that these mechanisms and associated genes could serve as indicators of the diverse behaviors exhibited by different strains (Table S5 of KT2440 and Table S6 of PAO1). The results presented offer insights into the categorization and distribution of TCS transcriptional regulators in P. putida KT2440 and P. aeruginosa (Figure 6) based on PANTHER classifications.
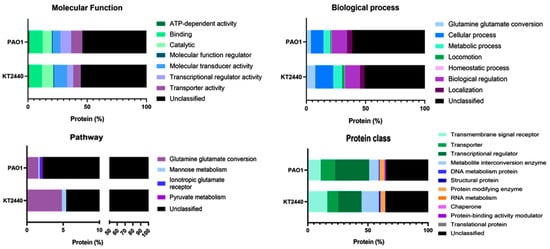
Figure 6.
Functional comparison of genes from TCSs in P. putida KT2440 and P. aeruginosa PAO1. Functions are categorized into four groups: molecular function, biological process, pathway, and protein class. The primary functional Panther-GO categories are represented as a percentage of transcriptional regulators compared to the total number of HTH regulatory genes.
Molecular Function: The comparative analysis of the molecular functions between P. putida KT2440 (Table S5) and P. aeruginosa PAO1 (Table S6) strains reveals both similarities and intriguing differences (Figure 6). In both strains, there were relatively few genes associated with “Transcription Regulator Activity”; 5.24% and 8.93% in KT2440 and PAO1, suggesting that specific genes play a role in modulating transcriptional processes. A significant proportion of genes in both strains is associated with “Binding Activity”, which facilitates molecular interactions and binding with other molecules. In KT2440 and PAO1 (Figure 6), 10.47% and 11.17% of genes, respectively, are involved in “Binding Activity”. Furthermore, “Catalytic Activity” accounts for 9.95% of genes in KT2440, compared to 7.90% in PAO1, indicating similar functional requirements in both strains. Notably, genes in P. aeruginosa PAO1 linked to “Molecular Transducer Activity” (6.70%) and “Transporter Activity” (9.45%) displayed a distinct profile compared to P. putida KT2440 (10.99% and 6.28%, respectively). Interestingly, only a few genes were involved in “ATP-Dependent Activity” (1.05% in KT240 and 1.20% in PAO1), implying a limited dependence on ATP for certain molecular functions. It should be noted that, in the strain KT2440, a significant proportion of genes (55.50%) compared to (54.12%) in PAO1 remain “Non-Characterized”, indicating a need for further investigation to shed light on the functional significance and potential applications of these unique molecular functions in these strains.
Biological process: Surprisingly, the total number of genes involved in biological processes differs significantly between non-pathogenic P. putida KT2440 and P. aeruginosa PAO1. In the KT2440 strain, this number stands at 212 genes, while in P. aeruginosa it is three-times higher (594 genes), indicating a more complex transcriptional regulation system (Figure 6 and Table S6). Specifically, the “Cellular Process” category accounts for 15.09% in KT2440 and 10.61% in PAO1 of the TCSs and encompasses fundamental cellular activities. These regulators are likely pivotal in controlling essential cell functions. Additionally, involvement in cellular localization processes is marked by 3.77% in KT2440 and 4.55% in PAO1, suggesting that a subset of transcriptional regulators plays a role determining the location of cellular components, which could be crucial for cell organization and structure. However, a notable proportion of these regulators, 50.94% in KT2440 and 61.11% in PAO1 (Figure 6), fall into no specific category. This lack of categorization highlights a substantial gap in our understanding of the roles and classifications of these transcriptional regulators. Consequently, further research and more detailed categorization are crucial for gaining a comprehensive understanding of their functions (Figure 6).
Pathway: The comparative analysis using PANTHER Pathway distributions (Figure 6) between P. putida KT2440 (Table S5) and P. aeruginosa PAO1 (Table S6) reveals notable differences in the types and proportions of genes associated with specific pathways. In this analysis, 166 genes were identified in the KT2440 strain and 512 in PAO1. A significant majority of these genes, 94.58% in KT2440 and 97.85% in PAO1, were categorized as “NO PANTHER category assigned” suggesting a considerable gap in our current understanding of the functional categorization of these genes. Specific pathways, such as “Glutamine glutamate conversion” and “Mannose metabolism”, were represented by less than 3.27% and 0.4% of the genes in KT2440 and PAO1, respectively. In contrast, both the “Ionotropic glutamate receptor” and “Pyruvate metabolism” pathway showed an exclusive representation in PAO1 at 0.20%, compared to KT2440 (Table S5 of KT2440 and Table S6 of PAO1). These slight variations in pathway representation may reflect distinct biological characteristics and functional preferences unique to each bacterial strain.
Protein class: In P. putida KT2440, genes categorized under “Transmembrane Signal Receptor” represent a significant 16.27% result, suggesting their vital role in cellular signaling processes. “Transporter” genes, accounting for 9.04%, are integral in the transportation of molecules across cellular membranes. Genes identified as “Gene-Specific Transcriptional Regulator” (19.28%) play a pivotal role in regulating gene expression and transcriptional processes. “Metabolite Interconversion Enzyme” genes, making up to 14.46%, underscore the importance of metabolic transformations in this strain. However, the mere 0.6% of “DNA Metabolism Protein” genes suggests a minor role in DNA metabolism. “Structural Protein” genes (0.6%) hint at their involvement in cellular structure, while “ProteinModifying Enzyme” genes (4.22%) are likely involved in modifying the structure or function of other proteins. A substantial portion of genes (35.54%) remains “Unclassified”, highlighting a gap in our understanding of their functions (Figure 6, Table S5 of KT2440 and Table S6 of PAO1).
Contrastingly, in P. aeruginosa PAO1, the “Transmembrane Signal Receptor” genes comprise 10.81%, indicating a significant but reduced role in cellular signaling compared to P. putida KT2440. “Transporter” genes at 11.98% are crucial for molecular transportation across cellular compartments. The substantial presence of “Gene-Specific Transcriptional Regulation” genes (28.49%) points to an active involvement in gene regulation processes. “Metabolite Interconversion Enzyme” genes (8.06%) parallel the role seen in KT2440 (Figure 6). The 0.39% of “DNA Metabolism Protein” genes suggests a less prominent role in DNA metabolism than in P. putida KT2440. “Structural Protein” genes (0.2%) and “Protein Modifying Enzyme” genes (3.73%) have roles in structural support and protein modification, respectively. “Protein Binding Activity Modulator” genes (0.39%) are involved in modulating protein-binding activities. However, a significant 35.36% of genes remain “Unclassified”, calling for further investigation of these genes.
In summary, compared to P. putida KT2440, P. aeruginosa PAO1 exhibits a similar percentage (16.27%) of genes in the “NO Panther Category” and a higher percentage in the “Gene-specific transcriptional regulation” and “Transporter” classes (Figure 6, Table S5 of KT2440 and Table S6 of PAO1) compared to KT2440. Conversely, P. putida KT2440 has a greater proportion of genes related to “Transmembrane signal receptor” and “Metabolite interconversion enzyme”. These variations highlight the distinct genetic composition and functional capacities inherent to these strains.
2.6. Forecasting and Choice of Transcriptional Regulators Associated with Pathogenicity
The exploration of regulatory mechanisms controlling the expression of virulence factors in bacterial pathogens reveals promising avenues for therapeutic intervention. In this context, two-component systems (TCSs) have emerged as compelling targets for the development of novel antibacterial agents [63]. Unlike traditional antibiotics that typically inhibit essential bacterial proteins, targeting TCSs offers a strategy to disrupt upstream regulatory networks, thereby impairing a pathogen’s ability to adapt and express virulence determinants. For example, in P. aeruginosa PAO1, TCSs such as PhoPQ, GacSA, and PmrAB regulate a suite of virulence-associated genes, including toxA (exotoxin A), exoS, exoT, and the components of the type III secretion system, like pscC and popB. These regulators also influence secondary metabolite production, including pyocyanin and rhamnolipids.
By contrast, P. putida KT2440 harbors TCSs, such as GacSA and FleSR, which are mainly involved in environmental sensing, motility, and type VI secretion system (T6SS) regulation, but it lacks several key effectors associated with pathogenicity, reflecting its reduced virulence potential [65,66]. The absence of genes like toxA and exoU further underscores its classification as a non-pathogenic, environmentally adapted species.
These genomic distinctions align with their divergent ecological roles: P. aeruginosa is a recognized opportunistic pathogen capable of forming biofilms, evading host immunity, and thriving in polymicrobial settings (Table 2), whereas P. putida is widely regarded as a chassis for industrial and biotechnological applications. As indicated in Table 2, genes are grouped according to the system involved, such as motility, secretion systems, immune modulation, biofilm, quorum sensing, metabolites, virulence factors, and others.

Table 2.
Categorization of virulence genes in P. aeruginosa PAO1 and P. putida KT2440. nd: Not identified.
Because TCSs regulate antibiotic resistance determinants, such as mexXY (controlled by ParRS protein) and arnBCADTEF (regulated by PhoPQ and PmrAB proteins) in P. aeruginosa [67], therapeutic strategies combining TCS inhibitors with conventional antibiotics could enhance efficacy and mitigate resistance development.
Importantly, the reliance of bacterial TCSs on histidine phosphorylation, a mechanism absent in mammals, suggests that specific inhibitors would have minimal off-target toxicity. Moreover, the conserved architecture of histidine kinase and response regulator domains raises the possibility of designing broad-spectrum inhibitors capable of targeting multiple TCSs simultaneously, thus reducing the likelihood of chromosomal resistance emergence.
3. Material and Methods
3.1. Genomic Analysis
The general method used to identify DNA-binding transcriptional regulators in sequenced Pseudomonas genomes involved exploring combination of diverse bioinformatics databases. Putative DNA-binding proteins were initially searched for in the complete genome sequences of Pseudomonas aeruginosa PAO1 (RefSeq: GCA_000006765.1) and Pseudomonas putida KT2440 (RefSeq: GCF_000007565.2) using keywords, sequence similarity techniques, and also the databases PROSITE-Expasy (Swiss Institute of Bioinformatics, Lausanne, Switzerland; https://prosite.expasy.org/) (accessed on 6 December 2024), KEGG (Kyoto Encyclopedia of Genes and Genomes, Kyoto University Bioinformatics Center, Kyoto, Japan; https://www.genome.jp/kegg/) (accessed on 6 December 2024), and Pfam (European Bioinformatics Institute, Hinxton, Cambridgeshire, UK; http://pfam-legacy.xfam.org/) (accessed on 6 December 2024). Subsequently, defined collections of putative transcriptional regulators were manually curated for the selected strains’ genomes (Tables S1 and S2) and plotted using the CGView Server (version not specified), (Genome Context Tools, Bogotá, Colombia; http://genocat.tools/tools/cgview_server.html) (accessed on 6 December 2024). Moreover, to identify the common set of DNA-binding transcriptional regulators, comparative genomic analyses were performed.
3.2. Distribution of DNA-Binding Proteins
The search for Pseudomonas DNA-binding proteins was performed by means of the genome assignment server superfAMILY (https://supfam.org/SUPERFAMILY/) (University of Cambridge, United Kingdom), (accessed on 6 December 2024) that contains a library of hidden Markov models (HMMs) of the Pfam database based on the sequences of protein domains. To identify among the DNA-binding proteins those potentially representing transcriptional regulators, different HMM profiles of bacterial protein families with a known function in the transcriptional regulation of gene expression were downloaded from the Pfam database and used for searches against the predicted Pseudomonas proteins using hidden Markov model profiles. The HTH recognition tool designed by Dodd and Egan was used to scan the putative DNA-binding transcriptional regulators for the presence and position of HTH motifs by using PROSITE-Expasy, KEGG, and Pfam databases. Finally, the putative DNA-binding transcriptional regulators were grouped into regulatory protein families using the PANTHER knowledgebase (https://www.pantherdb.org/) (Stanford University, California, United States), (accessed on 6 December 2024).
3.3. Phylogenetic Analysis
Protein sequences with the same HTH domain were used to carry out a phylogenetic tree representation. All sequences were aligned by MUSCLE v5.2 software (https://www.ebi.ac.uk/jdispatcher/msa/muscle5?stype=protein) (accessed on 6 December 2024) and aligned proteins were used as inputs for the FastTree program (version 2.1.11) to build a phylogenetic tree using the JTT+CAT model. The resulting tree was plotted using iTool (iTOL v6) and annotated manually according to the individual protein annotation.
3.4. Classification Analysis
The functional classification of genes from the PAO1 and KT2440 strains was conducted by employing the Panther-GO plotting tool from PANTHER Classification System, version 17.0, based on PANTHER GO-slims and Gene Ontology terms (https://www.geneontology.org/), (accessed on 6 December 2024).
4. Conclusions
The bacterial TCSs play a key role in signaling, enabling the survival and colonization of pathogenic bacteria within their host. Given the urgent need for novel antimicrobial drugs, the regulatory function of TCSs makes them highly promising targets for the development of innovative therapeutics against bacterial infections. Due to their widespread presence and functional versatility, evaluating multiple compounds that target TCSs is essential. Numerous studies have documented both natural and synthetic compounds that exhibit a strong affinity for TCSs, demonstrating effective antimicrobial action against pathogenic bacteria. Therefore, a deeper understanding of the interactions between OCS and TCS and their targeted compound is critical. Such knowledge can contribute to refining the molecular structures of these compounds, thereby enhancing their specificity and effectiveness as ligands. Consequently, further research is needed to comprehensively elucidate the precise mechanisms of action of these drugs.
In this paper, we present a comprehensive analysis of transcriptional regulatory systems in both the non-pathogenic P. putida KT2440 and the pathogenic P. aeruginosa PAO1 strains. Additionally, we categorize and highlight the significance of TCSs in pathways related to virulence, resistance, and metabolism. Given that TCS-encoding genes are present in all Gram-positive and Gram-negative bacterial genomes, the development of a pharmacological TCS inhibitor that acts broadly and achieves the desired therapeutic effect would be exceptionally valuable in the fight against antibiotic resistance.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms26104677/s1.
Author Contributions
Z.U.: Methodology, Investigation and Reviewing. K.A.S.: Methodology and Investigation. A.R.M.B.: Methodology and Investigation. M.T.-G.: Methodology and Investigation. J.C.M.: Methodology and Reviewing. A.D.: Conceptualization, Methodology, Supervision, Writing—Reviewing and Editing. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Wani, A.K.; Akhtar, N.; Sher, F.; Navarrete, A.A.; Américo-Pinheiro, J.H.P. Microbial adaptation to different environmental conditions: Molecular perspective of evolved genetic and cellular systems. Arch. Microbiol. 2022, 204, 144. [Google Scholar] [CrossRef]
- Narciso-da-Rocha, C.; Manaia, C.M. Multidrug resistance phenotypes are widespread over different bacterial taxonomic groups thriving in surface water. Sci. Total Environ. 2016, 563–564, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Abram, K.Z.; Jun, S.-R.; Udaondo, Z. Pseudomonas aeruginosa Pangenome: Core and Accessory Genes of a Highly Resourceful Opportunistic Pathogen. Adv. Exp. Med. Biol. 2022, 1386, 3–28. [Google Scholar] [CrossRef]
- Goldberg, J.B. Why is Pseudomonas aeruginosa a pathogen? F1000 Biol. Rep. 2010, 2, 29. [Google Scholar] [CrossRef]
- Daddaoua, A.; Fillet, S.; Fernández, M.; Udaondo, Z.; Krell, T.; Ramos, J.L. Genes for carbon metabolism and the ToxA virulence factor in Pseudomonas aeruginosa are regulated through molecular interactions of PtxR and PtxS. PLoS ONE 2012, 7, e39390. [Google Scholar] [CrossRef] [PubMed]
- Horcajada, J.P.; Montero, M.; Oliver, A.; Sorlí, L.; Luque, S.; Gómez-Zorrilla, S.; Benito, N.; Grau, S. Epidemiology and Treatment of Multidrug-Resistant and Extensively Drug-Resistant Pseudomonas aeruginosa Infections. Clin. Microbiol. Rev. 2019, 32, e00031-19. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Gómez, J.M.; Santiago, C.M.; Udaondo, Z.; Garitaonaindia, M.T.; Krell, T.; Ramos, J.-L.; Daddaoua, A. Full Transcriptomic Response of Pseudomonas aeruginosa to an Inulin-Derived Fructooligosaccharide. Front. Microbiol. 2020, 11, 202. [Google Scholar] [CrossRef]
- Udaondo, Z.; Molina, L.; Daniels, C.; Gómez, M.J.; Molina-Henares, M.A.; Matilla, M.A.; Roca, A.; Fernández, M.; Duque, E.; Segura, A.; et al. Metabolic potential of the organic-solvent tolerant Pseudomonas putida DOT-T1E deduced from its annotated genome. Microb. Biotechnol. 2013, 6, 598–611. [Google Scholar] [CrossRef]
- Molina-Santiago, C.; Udaondo, Z.; Gómez-Lozano, M.; Molin, S.; Ramos, J.-L. Global transcriptional response of solvent-sensitive and solvent-tolerant Pseudomonas putida strains exposed to toluene. Environ. Microbiol. 2017, 19, 645–658. [Google Scholar] [CrossRef]
- Abram, K.Z.; Udaondo, Z. Towards a better metabolic engineering reference: The microbial chassis. Microb. Biotechnol. 2020, 13, 17–18. [Google Scholar] [CrossRef]
- Duque, E.; Udaondo, Z.; Molina, L.; de la Torre, J.; Godoy, P.; Ramos, J.L. Providing octane degradation capability to Pseudomonas putida KT2440 through the horizontal acquisition of oct genes located on an integrative and conjugative element. Environ. Microbiol. Rep. 2022, 14, 934–946. [Google Scholar] [CrossRef]
- Pizarro-Tobías, P.; Fernández, M.; Niqui, J.L.; Solano, J.; Duque, E.; Ramos, J.; Roca, A. Restoration of a Mediterranean forest after a fire: Bioremediation and rhizoremediation field-scale trial. Microb. Biotechnol. 2014, 8, 77–92. [Google Scholar] [CrossRef] [PubMed]
- Molina, L.; Segura, A.; Duque, E.; Ramos, J.-L. The versatility of Pseudomonas putida in the rhizosphere environment. Adv. Appl. Microbiol. 2020, 110, 149–180. [Google Scholar] [CrossRef]
- Mikkelsen, H.; McMullan, R.; Filloux, A. The Pseudomonas aeruginosa reference strain PA14 displays increased virulence due to a mutation in ladS. PLoS ONE 2011, 6, e29113. [Google Scholar] [CrossRef]
- Chastre, J.; Fagon, J.-Y. Ventilator-associated pneumonia. Am. J. Respir. Crit. Care Med. 2002, 165, 867–903. [Google Scholar] [CrossRef]
- Hassett, D.J.; Korfhagen, T.R.; Irvin, R.T.; Schurr, M.J.; Sauer, K.; Lau, G.W.; Sutton, M.D.; Yu, H.; Hoiby, N. Pseudomonas aeruginosa biofilm infections in cystic fibrosis: Insights into pathogenic processes and treatment strategies. Expert Opin. Ther. Targets 2010, 14, 117–130. [Google Scholar] [CrossRef] [PubMed]
- Montie, T.C.; Doyle-Huntzinger, D.; Craven, R.C.; Holder, I.A. Loss of virulence associated with absence of flagellum in an isogenic mutant of Pseudomonas aeruginosa in the burned-mouse model. Infect. Immun. 1982, 38, 1296–1298. [Google Scholar] [CrossRef] [PubMed]
- Montie, T.C.; Craven, R.C.; Holder, I.A. Flagellar preparations from Pseudomonas aeruginosa: Isolation and characterization. Infect. Immun. 1982, 35, 281–288. [Google Scholar] [CrossRef]
- Cano, P.G.; Santacruz, A.; Trejo, F.M.; Sanz, Y. BifidobacteriumCECT 7765 improves metabolic and immunological alterations associated with obesity in high-fat diet-fed mice. Obesity 2013, 21, 2310–2321. [Google Scholar] [CrossRef]
- Miura, K.; Ohnishi, H. Role of gut microbiota and Toll-like receptors in nonalcoholic fatty liver disease. World J. Gastroenterol. 2014, 20, 7381–7391. [Google Scholar] [CrossRef]
- Ohara, T.; Itoh, K. Significance of Pseudomonas aeruginosa colonization of the gastrointestinal tract. Intern. Med. 2003, 42, 1072–1076. [Google Scholar] [CrossRef] [PubMed]
- von Klitzing, E.; Bereswill, S.; Heimesaat, M.M. Multidrug-Resistant Pseudomonas aeruginosa Induce Systemic Pro-Inflammatory Immune Responses in Colonized Mice. Eur. J. Microbiol. Immunol. 2017, 7, 200–209. [Google Scholar] [CrossRef]
- Alvarez-Ortega, C.; Wiegand, I.; Olivares, J.; Hancock, R.E.W.; Martínez, J.L. The intrinsic resistome of Pseudomonas aeruginosa to β-lactams. Virulence 2011, 2, 144–146. [Google Scholar] [CrossRef]
- Pachori, P.; Gothalwal, R.; Gandhi, P. Emergence of antibiotic resistance Pseudomonas aeruginosa in intensive care unit; a critical review. Genes Dis. 2019, 6, 109–119. [Google Scholar] [CrossRef]
- Hall, J.P.J.; Brockhurst, M.A.; Harrison, E. Sampling the mobile gene pool: Innovation via horizontal gene transfer in bacteria. Philos. Trans. R. Soc. London. Ser. B Biol. Sci. 2017, 372, 20160424. [Google Scholar] [CrossRef]
- Hogardt, M.; Heesemann, J. Adaptation of Pseudomonas aeruginosa during persistence in the cystic fibrosis lung. Int. J. Med. Microbiol. 2010, 300, 557–562. [Google Scholar] [CrossRef] [PubMed]
- Mah, T.-F.; Pitts, B.; Pellock, B.; Walker, G.C.; Stewart, P.S.; O’Toole, G.A. A genetic basis for Pseudomonas aeruginosa biofilm antibiotic resistance. Nature 2003, 426, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.K.; Yeung, A.T.Y.; Hancock, R.E.W. Antibiotic resistance in Pseudomonas aeruginosa biofilms: Towards the development of novel anti-biofilm therapies. J. Biotechnol. 2014, 191, 121–130. [Google Scholar] [CrossRef]
- Iglewski, B.H.; Kabat, D. NAD-dependent inhibition of protein synthesis by Pseudomonas aeruginosa toxin. Proc. Natl. Acad. Sci. USA 1975, 72, 2284–2288. [Google Scholar] [CrossRef]
- Ortiz-Castro, R.; Pelagio-Flores, R.; Méndez-Bravo, A.; Ruiz-Herrera, L.F.; Campos-García, J.; López-Bucio, J. Pyocyanin, a virulence factor produced by Pseudomonas aeruginosa, alters root development through reactive oxygen species and ethylene signaling in Arabidopsis. Mol. Plant. Microbe. Interact. 2014, 27, 364–378. [Google Scholar] [CrossRef]
- Matar, G.M.; Ramlawi, F.; Hijazi, N.; Khneisser, I.; Abdelnoor, A.M. Transcription levels of Pseudomonas aeruginosa exotoxin a gene and severity of symptoms in patients with otitis externa. Curr. Microbiol. 2002, 45, 350–354. [Google Scholar] [CrossRef] [PubMed]
- McEwan, D.L.; Kirienko, N.V.; Ausubel, F.M. Host translational inhibition by Pseudomonas aeruginosa Exotoxin A Triggers an immune response in Caenorhabditis elegans. Cell Host Microbe 2012, 11, 364–374. [Google Scholar] [CrossRef]
- Yates, S.P.; Jørgensen, R.; Andersen, G.R.; Merrill, A.R. Stealth and mimicry by deadly bacterial toxins. Trends Biochem. Sci. 2006, 31, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Hamood, A.N.; Ohman, D.E.; West, S.E.; Iglewski, B.H. Isolation and characterization of toxin A excretion-deficient mutants of Pseudomonas aeruginosa PAO1. Infect. Immun. 1992, 60, 510–517. [Google Scholar] [CrossRef]
- Durand, E.; Verger, D.; Rêgo, A.T.; Chandran, V.; Meng, G.; Fronzes, R.; Waksman, G. Structural biology of bacterial secretion systems in gram-negative pathogens--potential for new drug targets. Infect. Disord. Drug Targets 2009, 9, 518–547. [Google Scholar] [CrossRef]
- Bleves, S.; Viarre, V.; Salacha, R.; Michel, G.P.F.; Filloux, A.; Voulhoux, R. Protein secretion systems in Pseudomonas aeruginosa: A wealth of pathogenic weapons. Int. J. Med. Microbiol. 2010, 300, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Allsopp, L.P.; Wood, T.E.; Howard, S.A.; Maggiorelli, F.; Nolan, L.M.; Wettstadt, S.; Filloux, A. RsmA and AmrZ orchestrate the assembly of all three type VI secretion systems in Pseudomonas aeruginosa. Proc. Natl. Acad. Sci. USA 2017, 114, 7707–7712. [Google Scholar] [CrossRef]
- Brown, E.D.; Wright, G.D. Antibacterial drug discovery in the resistance era. Nature 2016, 529, 336–343. [Google Scholar] [CrossRef]
- Munita, J.M.; Arias, C.A. Mechanisms of Antibiotic Resistance. Microbiol. Spectr. 2016, 4, 464–473. [Google Scholar] [CrossRef]
- Ulrich, L.E.; Koonin, E.V.; Zhulin, I.B. One-component systems dominate signal transduction in prokaryotes. Trends Microbiol. 2005, 13, 52–56. [Google Scholar] [CrossRef]
- Mascher, T. Intramembrane-sensing histidine kinases: A new family of cell envelope stress sensors in Firmicutes bacteria. FEMS Microbiol. Lett. 2006, 264, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Hazelbauer, G.L.; Falke, J.J.; Parkinson, J.S. Bacterial chemoreceptors: High-performance signaling in networked arrays. Trends Biochem. Sci. 2008, 33, 9–19. [Google Scholar] [CrossRef]
- Galperin, M.Y. A census of membrane-bound and intracellular signal transduction proteins in bacteria: Bacterial IQ, extroverts and introverts. BMC Microbiol. 2005, 5, 35. [Google Scholar] [CrossRef] [PubMed]
- Nixon, B.T.; Ronson, C.W.; Ausubel, F.M. Two-component regulatory systems responsive to environmental stimuli share strongly conserved domains with the nitrogen assimilation regulatory genes ntrB and ntrC. Proc. Natl. Acad. Sci. USA 1986, 83, 7850–7854. [Google Scholar] [CrossRef]
- Bijlsma, J.J.E.; Groisman, E.A. Making informed decisions: Regulatory interactions between two-component systems. Trends Microbiol. 2003, 11, 359–366. [Google Scholar] [CrossRef]
- Bourret, R.B.; Silversmith, R.E. Two-component signal transduction. Curr. Opin. Microbiol. 2010, 13, 113–115. [Google Scholar] [CrossRef]
- Cheung, J.; Hendrickson, W.A. Sensor domains of two-component regulatory systems. Curr. Opin. Microbiol. 2010, 13, 116–123. [Google Scholar] [CrossRef]
- Groisman, E.A. Feedback Control of Two-Component Regulatory Systems. Annu. Rev. Microbiol. 2016, 70, 103–124. [Google Scholar] [CrossRef] [PubMed]
- Gislason, A.S.; Choy, M.; Bloodworth, R.A.M.; Qu, W.; Stietz, M.S.; Li, X.; Zhang, C.; Cardona, S.T. Competitive Growth Enhances Conditional Growth Mutant Sensitivity to Antibiotics and Exposes a Two-Component System as an Emerging Antibacterial Target in Burkholderia cenocepacia. Antimicrob. Agents Chemother. 2017, 61, e00790-16. [Google Scholar] [CrossRef]
- Mitrophanov, A.Y.; Groisman, E.A. Signal integration in bacterial two-component regulatory systems. Genes Dev. 2008, 22, 2601–2611. [Google Scholar] [CrossRef]
- Nelson, K.E.; Weinel, C.; Paulsen, I.T.; Dodson, R.J.; Hilbert, H.; Martins dos Santos, V.A.P.; Fouts, D.E.; Gill, S.R.; Pop, M.; Holmes, M.; et al. Complete genome sequence and comparative analysis of the metabolically versatile Pseudomonas putida KT2440. Environ. Microbiol. 2002, 4, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Belda, E.; van Heck, R.G.A.; José Lopez-Sanchez, M.; Cruveiller, S.; Barbe, V.; Fraser, C.; Klenk, H.-P.; Petersen, J.; Morgat, A.; Nikel, P.I.; et al. The revisited genome of Pseudomonas putida KT2440 enlightens its value as a robust metabolic chassis. Environ. Microbiol. 2016, 18, 3403–3424. [Google Scholar] [CrossRef] [PubMed]
- Stover, C.K.; Pham, X.Q.; Erwin, A.L.; Mizoguchi, S.D.; Warrener, P.; Hickey, M.J.; Brinkman, F.S.; Hufnagle, W.O.; Kowalik, D.J.; Lagrou, M.; et al. Complete genome sequence of Pseudomonas aeruginosa PAO1, an opportunistic pathogen. Nature 2000, 406, 959–964. [Google Scholar] [CrossRef] [PubMed]
- Tettmann, B.; Dötsch, A.; Armant, O.; Fjell, C.D.; Overhage, J. Knockout of extracytoplasmic function sigma factor ECF-10 affects stress resistance and biofilm formation in Pseudomonas putida KT2440. Appl. Environ. Microbiol. 2014, 80, 4911–4919. [Google Scholar] [CrossRef] [PubMed]
- Brune, I.; Brinkrolf, K.; Kalinowski, J.; Pühler, A.; Tauch, A. The individual and common repertoire of DNA-binding transcriptional regulators of Corynebacterium glutamicum, Corynebacterium efficiens, Corynebacterium diphtheriae and Corynebacterium jeikeium deduced from the complete genome sequences. BMC Genomics 2005, 6, 86. [Google Scholar] [CrossRef]
- Lindquist, S.; Lindberg, F.; Normark, S. Binding of the Citrobacter freundii AmpR regulator to a single DNA site provides both autoregulation and activation of the inducible ampC beta-lactamase gene. J. Bacteriol. 1989, 171, 3746–3753. [Google Scholar] [CrossRef]
- Schell, M.A. Molecular biology of the LysR family of transcriptional regulators. Annu. Rev. Microbiol. 1993, 47, 597–626. [Google Scholar] [CrossRef]
- Deng, W.; Li, C.; Xie, J. The underling mechanism of bacterial TetR/AcrR family transcriptional repressors. Cell. Signal. 2013, 25, 1608–1613. [Google Scholar] [CrossRef]
- Colclough, A.L.; Scadden, J.; Blair, J.M.A. TetR-family transcription factors in Gram-negative bacteria: Conservation, variation and implications for efflux-mediated antimicrobial resistance. BMC Genomics 2019, 20, 731. [Google Scholar] [CrossRef]
- Routh, M.D.; Su, C.-C.; Zhang, Q.; Yu, E.W. Structures of AcrR and CmeR: Insight into the mechanisms of transcriptional repression and multi-drug recognition in the TetR family of regulators. Biochim. Biophys. Acta 2009, 1794, 844–851. [Google Scholar] [CrossRef]
- Scharf, B.E. Summary of useful methods for two-component system research. Curr. Opin. Microbiol. 2010, 13, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Eguchi, Y.; Utsumi, R. Introduction to bacterial signal transduction networks. In Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2008; Volume 631, pp. 1–6. [Google Scholar] [CrossRef]
- Gotoh, Y.; Eguchi, Y.; Watanabe, T.; Okamoto, S.; Doi, A.; Utsumi, R. Two-component signal transduction as potential drug targets in pathogenic bacteria. Curr. Opin. Microbiol. 2010, 13, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Schaefers, M.M.; Liao, T.L.; Boisvert, N.M.; Roux, D.; Yoder-Himes, D.; Priebe, G.P. An Oxygen-Sensing Two-Component System in the Burkholderia cepacia Complex Regulates Biofilm, Intracellular Invasion, and Pathogenicity. PLoS Pathog. 2017, 13, e1006116. [Google Scholar] [CrossRef]
- Poole, K. Bacterial stress responses as determinants of antimicrobial resistance. J. Antimicrob. Chemother. 2012, 67, 2069–2089. [Google Scholar] [CrossRef]
- Barrett, J.F.; Isaacson, R.E. Chapter 12. Bacterial Virulence as a Potential Target for Therapeutic Intervention. In Annual Reports in Medicinal Chemistry; Academic Press: Cambridge, MA, USA, 1995; Volume 30, pp. 111–118. [Google Scholar]
- Rodrigue, A.; Quentin, Y.; Lazdunski, A.; Méjean, V.; Foglino, M. Two-component regulatory systems in Pseudomonas aeruginosa: Why so many, and what are they doing? Microbiology 2000, 8, 498–504. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).