Abstract
Sideritis scardica Griseb, also known as “mountain tea” and “Olympus tea” (Lamiaceae family) is an endemic plant from the mountainous regions of the Balkan Peninsula. In this study, we focused on an in-depth phytochemical analysis of S. scardica infusion using ultra-high-performance liquid chromatography hyphenated with high-resolution mass spectrometry (UHPLC–HRMS). Quantitative determination of the main secondary metabolites was carried out by UHPLC–HRMS analyses using the external standard method. The results revealed more than 100 metabolites, including five sugar acids and saccharides, 21 carboxylic, hydroxybenzoic, hydroxycinnamic acids, and derivatives, 15 acylquinic acids, 10 phenylpropanoid glycosides, four iridoid glycosides, 28 flavonoids, seven fatty acids, and four organosulfur compounds. Furthermore, a dereplication and fragmentation patterns of five caffeic acids oligomers and four acylhexaric acids was performed for the first time in S. scardica. Regarding the quantitative analysis, the phenylethanoid verbascoside (53) (151.54 ± 10.86 mg/g lyophilized infusion, li), the glycosides of isoscutellarein (78) (151.70 ± 14.78 mg/g li), methylisoscutelarein (82) (107.4 ± 9.07 mg/g li), and hypolaetin (79) (78.33 ± 3.29 mg/g li), as well as caffeic acid (20) (87.25 ± 6.54 mg/g li), were found to be the major compounds in S. scardica infusion. The performed state-of-the-art phytochemical analysis of S. scardica provides additional knowledge for the chemical constituents and usage of this valuable medicinal plant.
1. Introduction
Sideritis scardica Griseb (Lamiaceae family) is an endemic plant of the mountainous regions of the Balkan Peninsula [1,2]. It is often referred to as “mountain tea”, “ironwort”, “Olympus tea”, and “Pirin tea” [3]. Mountain tea is a perennial herbaceous plant with a well-developed root system, the stem is 15–40 cm and woody at the base, the leaves are opposite with gray hairs, the flowers are clustered in a dense spike, the middle bracts are 12–20 mm long, i.e., longer than the flowers, the corolla is lemon yellow with glandules, and the calyx is tubular-campanulate [2,3]. Usually, Sideritis plants are applied in traditional medicine, mostly as an aromatic herbal tea [4,5,6,7]. The tea is made from the aerial parts of the plant by infusion or decoction [8]. Historically, S. scardica has been used to treat inflammation, common colds, asthma, bronchitis, and gastrointestinal disorders. It is supposed to relieve pain, including rheumatic pain, as well as reducing stress and anxiety. The plant name comes from the Greek word “sideros”, meaning “iron”, as it was used in ancient times to heal wounds from iron weapons [4]. Regular consumption of mountain tea by rats has been shown to lead to weight loss and prevent insulin resistance by lowering blood glucose and triglyceride levels and increasing liver glycogen content [8]. Additionally, antioxidant properties and positive effects on memory and cognitive abilities have also been observed [7,9,10]. Sideritis species have also been used topically on the skin and as an antiseptic solution to sooth the pain of tooth extraction [3].
The traditional medicinal usage of the species is based on the phytochemical constituents, including phenolic acids (chlorogenic acid, 3-caffeoylquinic acid, feruloylquinic acid, and others), flavonoids and their derivatives (hypolaetin, isoscutellarein, and others), phenylethanoid glycosides (lavandulifolioside, verbascoside, echinacoside, allysonoside, and others), and terpenoids (mostly iridoid glycosides) [6,8,11,12]. These chemical compounds have been explored in phytochemical studies, operating with various extraction techniques such as hydrodistillation and solvent and supercritical extractions [8,12,13,14,15]. Precisely, the most abundant secondary metabolites of S. scardica water extracts (i.e., when making infusion or decoction) are flavonoids, hydoxycinnamic acid derivatives, and phenylethanoid glycosides [7,14,16,17]. Identification of closely related species of the genus Sideritis is based on the dominant 5-caffeoylquinic acid, lavandulifolioside, verbascoside, isoscutellarein 7-O-allosyl(1→2)glucoside, hypolaetin 7-O-[6‴O-acetyl]-allosyl(1→2)glucoside, isoscutellarein 7-O-[6‴-O-acetyl]-allosyl(1→2) glucoside, 3′-O-methylhypolaetin 7-O-[6‴-O-acetyl]-allosyl(1→2)glucoside, and 4′-O-methylhypolaetin 7-O-[6‴-O-acetyl]-allosyl-(1→2)-[6″-O-acetyl]-glucoside. Thus, these compounds have been used as chemotaxonomical markers [6].
Based on the literature available on Sideritis scardica, there is no detailed metabolite profiling of the species, something which seems important in light of its health benefits. An in-depth UHPLC–HRMS analysis of the main metabolites of S. scardica, together with quantitative determination, was conducted. More than 100 secondary metabolites were identified/tentatively elucidated in a lyophilized infusion of mountain tea. The performed phytochemical analysis of S. scardica will provide additional knowledge of the chemical constituents and usage of this valuable medicinal plant for the future.
2. Results and Discussion
2.1. Metabolite Profiling of S. scardica Lyophilized Infusion
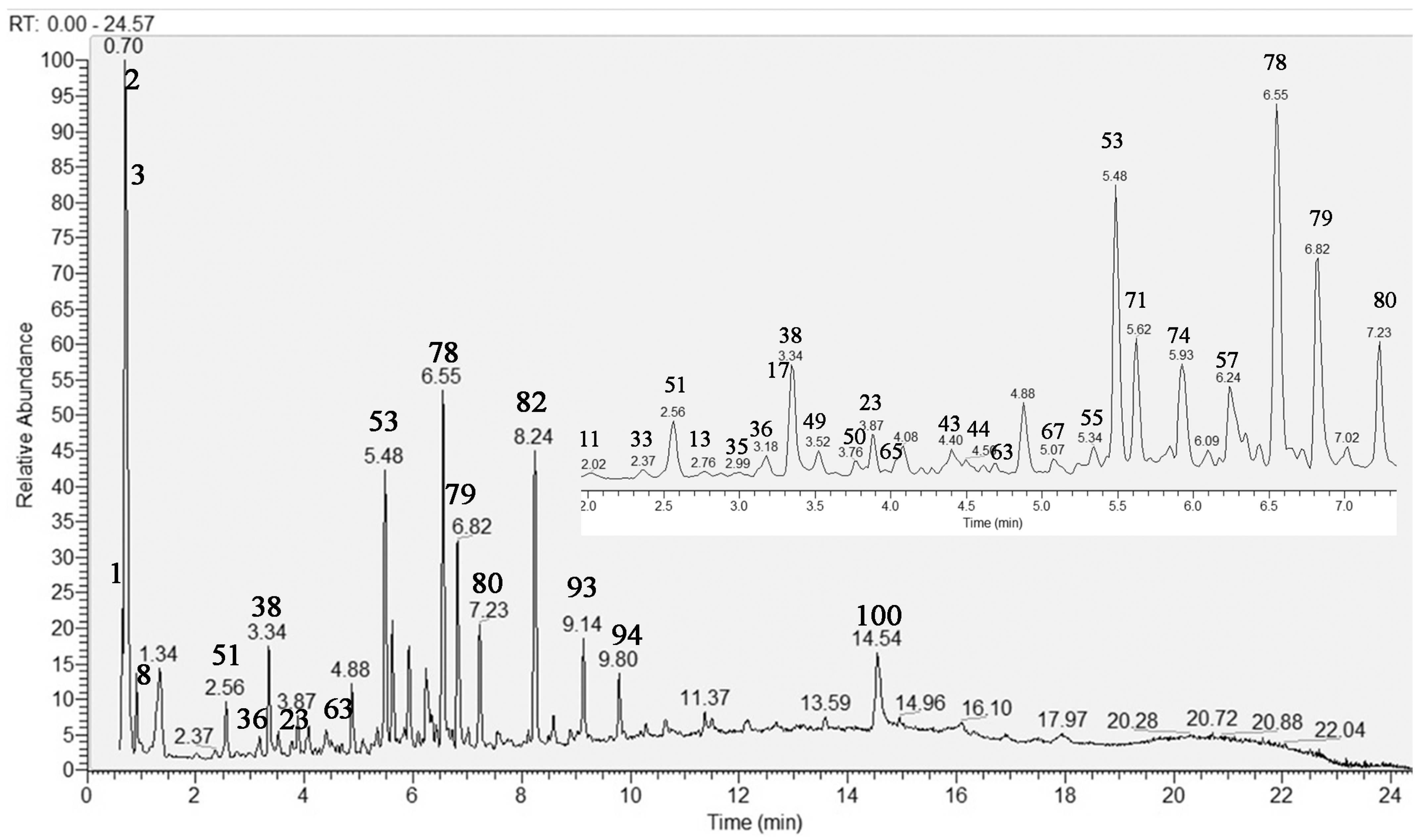
Herein, an in-depth UHPLC–HRMS analysis of S. scardica infusion was conducted by allowing the dereplication/annotation of 103 metabolites, including five sugar acids and saccharides, 21 carboxylic, hydroxybenzoic, hydroxycinnamic acids, and derivatives, five caffeic acids oligomers, 15 acylquinic acids, four acylhexaric acids, 10 phenylpropanoid glycosides, four iridoid glycosides, 28 flavonoids, seven fatty acids, and four organosulfur compounds (Table 1). This study allowed the identification of caffeic acids oligomers, acylquinic, acylhexaric acids, and flavonoids not previously reported in the taxon. The total ion chromatogram (TIC) in negative ion mode of the studied extract is presented in Figure 1.
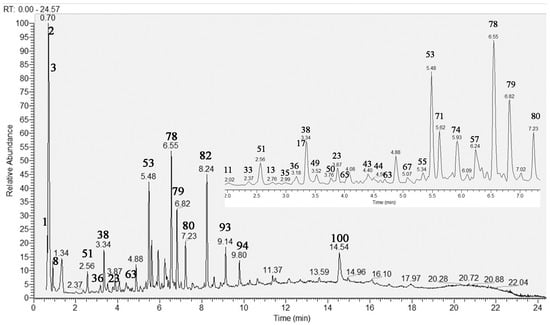
Figure 1.
Total ion chromatogram (TIC) in negative ion mode of Sideritis scardica extract; the same chromatogram 2–7 min. For compound numbering see Table 1.
2.1.1. Sugar Acids and Saccharides
Compound 1 ([M−H]− at m/z 165.041) gave fragment ions at m/z 147.02 [M−H−H2O]−, 129.018 [M−H−2H2O]−, 111.01 [M−H−3H2O]−, and 101.023 [M−H−3H2O−CO]−, as well as ions corresponding to the loss of 60 Da [M−H−C2H4O2]− and 90 Da [M−H−C3H6O3]−, respectively. Thus, compound 1 was annotated as xylonic acid. Analogously, 4 was related to pentose with a base peak at m/z 75.00 (C2H2O3). Moreover, 2 (with an additional CH2 group compared to 4) and 3 (with additional CH2O) were identified as hexose and gluconic acid, respectively (Table 1). The identity of asystoside (5) was suggested by the transitions 583.261→421.209→289.166→161.445, resulting from the losses of hexosyl (162.053 Da), pentosyl (132.043 Da), and oct-1-en-3-ol units (C8H16O, 128.122 Da) (Table 1). All above-mentioned compounds were previously identified in the species [12].
2.1.2. Carboxylic, Hydroxybenzoic, Hydroxycinnamic Acids and Their Derivatives
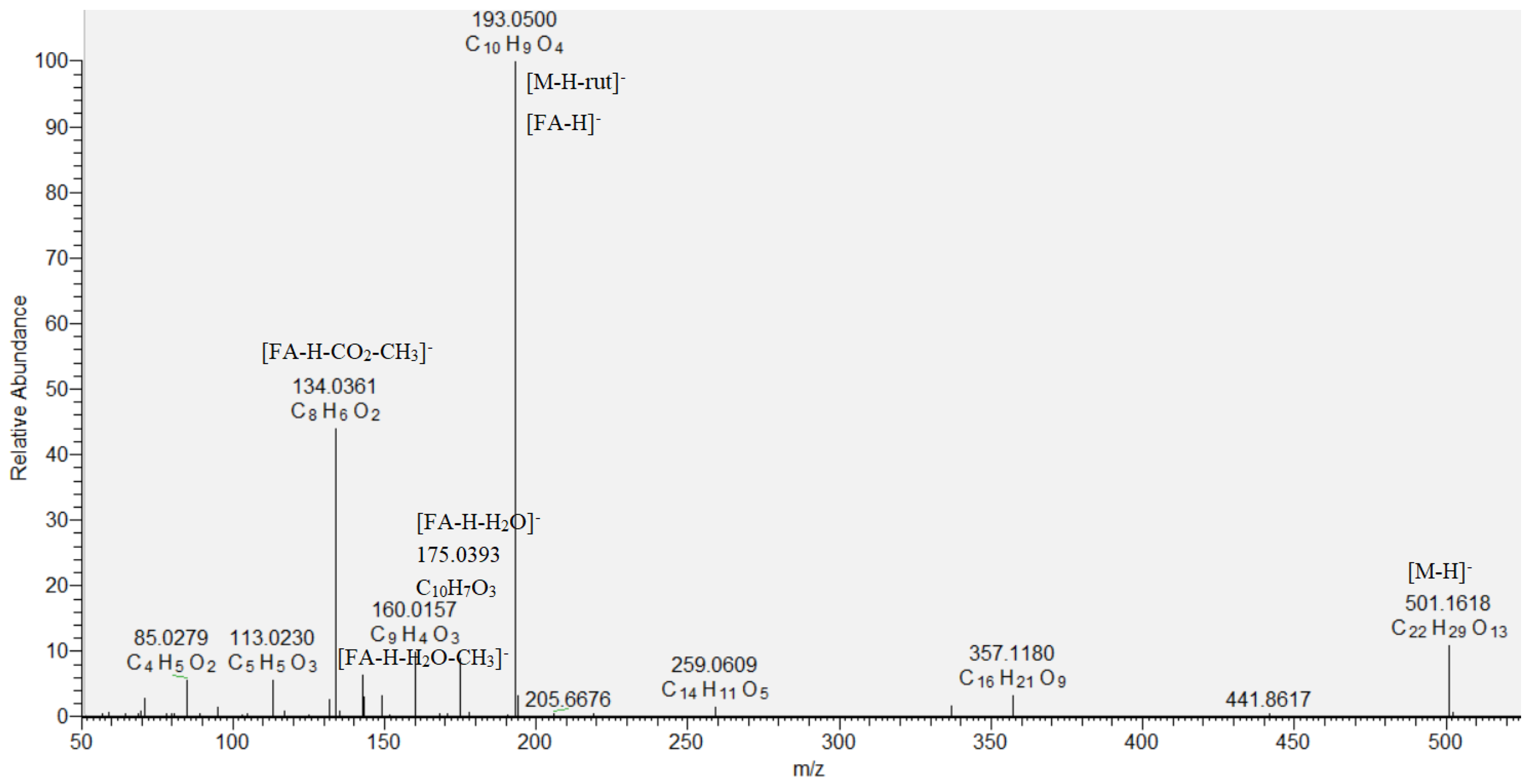
Eight hydroxybenzoic acids (9, 12, 15, 16, 18, 19, 21, and 24), four hydroxycinnamic acids (17, 20, 25, and 26) and their glycosides (10, 11, 13, 14, 22, and 23), together with quinic (6), oxaloglutaric (7), and citric acid (8), were identified based on the comparison with reference standards and literature data in the assayed extract (Table 1) [12,17]. Compound 8 ([M−H]− at m/z 191.018) showed fragment ions at m/z 173.008 [M−H−H2O]−, 147.028 [M−H−CO2]−, a base peak at m/z 111.007 [M−H−CO2−2H2O]−, and was related to citric acid [18]. A key step in the dereplication of phenolic acid glycosides was the neutral losses of 162.05, 132.04, and 308.11 Da, corresponding to hexose, pentose, and rutinose, respectively, together with the base peaks of the respective phenolic acid deprotonated molecule. Thus, pentosylhexosides of hydroxybenzoic acid (10) and vanillic acid (11), dihexoside of caffeic acid (14), and hexoside of coumaric acid (22) were annotated. Compounds 13 ([M−H]− at m/z 487.146) and 23 ([M−H]− at m/z 501.161) gave base peaks at m/z 179.034 [caffeic acid−H]− and 193.050 [ferulic acid−H]−, corresponding to the loss of rutinose ([M−H−rutinose]−), and were identified as caffeic acid O-rutinoside and ferulic acid O-rutinoside, respectively (Table 1, Figure 2). Compounds 10, 11, 12, 14, 15, 16, 17, 18, 19, 22, 23, 24, and 25 are reported for the first time in S. scardica.
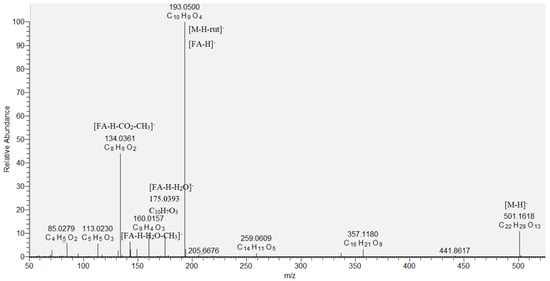
Figure 2.
MS/MS spectrum of ferulic acid O-rutinoside (23).
2.1.3. Caffeic Acids Oligomers
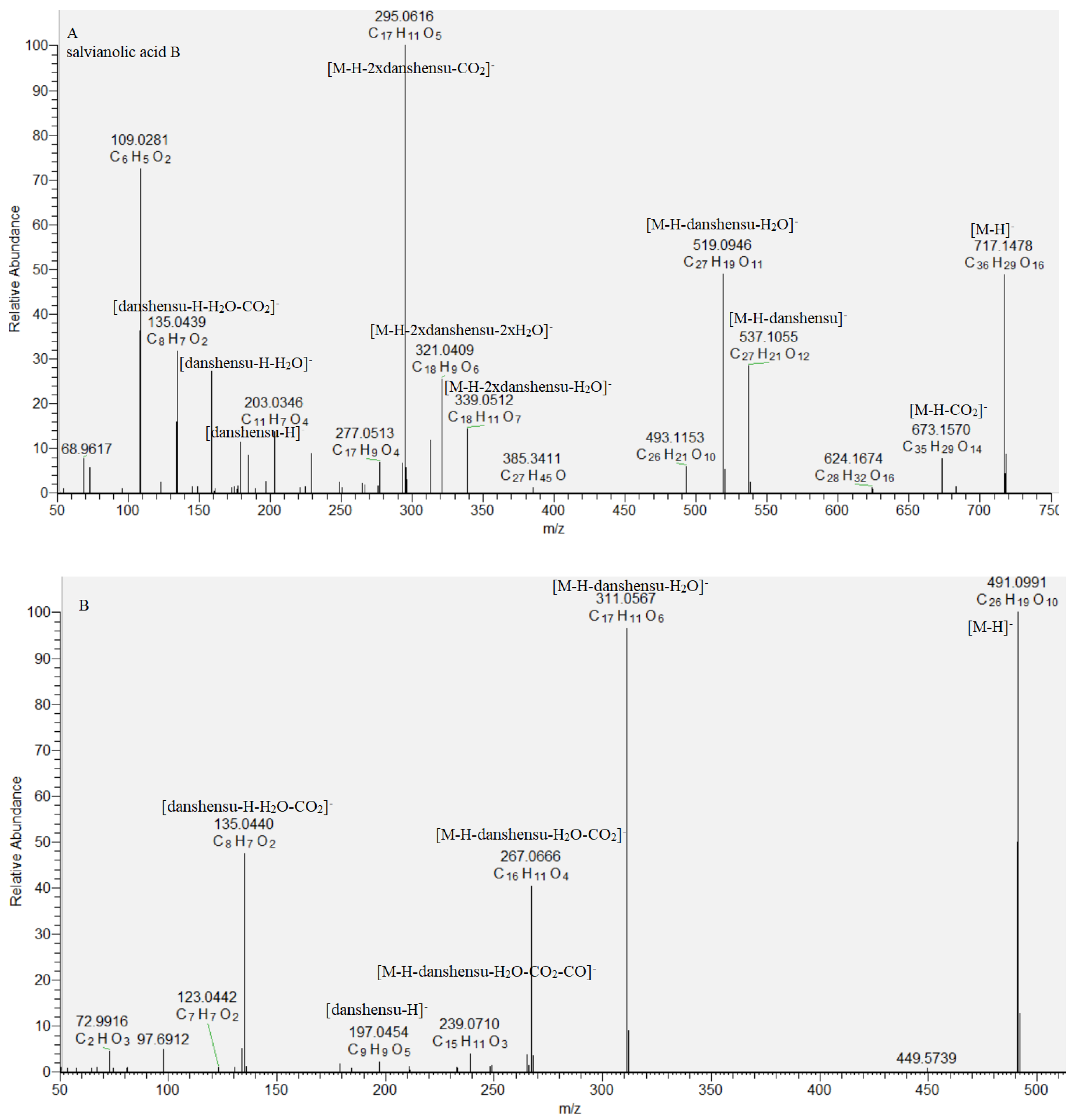
Caffeic acid oligomers consist of ester-bonded monomers such as danshensu, caffeic acid, and others and are present in Lamiaceae species [19,20,21]. Based on the accurate masses, MS/MS data, and literature data, a dimer rosmarinic acid (29), two trimers (27 and 30), and two tetramers (28 and 31) were dereplicated in the studied S. scardica extract. The fragmentation pattern and retention time of rosmarinic acid (29) were compared with reference standard. Key points in the caffeic acid oligomers annotation were the indicative fragment ions derived from the cleavage of a and b ester bonds with loss of danshensu [M−H−198.05]−, danshensoyl [M−H−180.04]−, and caffeoyl residue [M−H−162.03]−, respectively [19]. Compound 30 ([M−H]− at m/z 491.099) afforded a base peak at m/z 311.056, corresponding to the easier loss of danshensu, due to the dibenzooxepin structure, restraining the cleavage of the a bond. Based on a comparison with literature data, 30 was tentatively identified as isosalvianolic acid C [19] (Table 1, Figure 3). With respect to 27, an abundant fragment ion at m/z 493.114 [M−H−CO2]− and a base peak at m/z 339.059 [M−H−198.05]− were indicative of the presence of CO2 group attached to the benzofuran ring and danshensu residue. This allowed us to deduce the structure of lithospermic acid [19,20]. The fragmentation pathway of 28 included prominent ions at m/z 673.157 [M−H−CO2]−, 537.105 [M−H−180.04]−, 519.095 [M−H−198.05]−, 493.115 [M−H−180.04−CO2]−, 339.051 [M−H−2 × 198.05]−, and a base peak at m/z 321.041 [M−H−198.05−180.04−CO2]−, indicating, consequently, losses of two danshensu residues and carboxyl groups. Moreover, diagnostic ions at m/z 537.093, corresponding to deprotonated lythospermic acid, as well as the lack of loss of caffeoyl residue, suggested a terminal danshensu residue linked to lithospermic acid. Thus, compound 28 was dereplicated as salvianolic acid B (Table 1, Figure 3). Similarly, 31 was related to didehydrosalvianolic acid B (Table 1, Figure 3) [19]. Compounds 27, 28, 30, and 31 are reported for the first time in S. scardica.
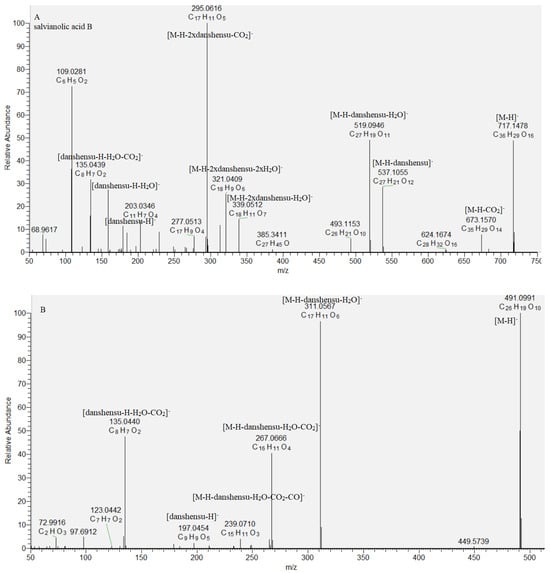
Figure 3.
MS/MS spectrum of (A) salvianolic acid B (28) and (B) isosalvianolic acid C (30).
2.1.4. Acylquinic Acid
Overall, three caffeoylquinic (33, 36, and 38), four p-coumaroylquinic (35, 41, 42, and 44), two syringoylquinic (37 and 40), three feruloylquinic (39, 43, and 45), together with two hexosides (32 and 34), and a syringoyl–caffeoylquinic acid (46) were dereplicated or annotated in the studied extract (Table 1). The acylquinic acids (AQAs) annotation was based on the fragment ions and their relative abundances corresponding to each subclass AQAs [22,23,24]. Three isobars shared the same deprotonated molecule [M−H]− at m/z 353.086. Compound 36 was identified as chlorogenic acid (5-caffeylquinic acid) due to the base peak at m/z 191.055 [quinic acid−H]−. The positional isomer neochlorogenic acid (3-caffeylquinic acid) (33) was apparent by the higher relative abundances of the fragment ions at m/z 179.033 (61.2%) and 135.043 (48.7%) than those of 36. Compounds 33 and 36 were unambiguously identified by comparison with reference standards. In the MS/MS spectrum of 38, 40, and 42, a base peak at m/z 173.044 [quinic acid−H−H2O]− was detected, indicating caffeoyl, syringoyl, and p-coumaroyl residues at position 4 of the quinic acid. Thus, 38, 40, and 42 were annotated as 4-caffeylquinic, 4-syringoyl, and 4-p-coumaroyl acids, respectively [24]. The compounds 3-p-coumaroylquinic acid (3-p-CoQA) (35) and 3-feruloylquinic acid (3-FQA) (39) were identified from the base peaks at m/z 163.039 [p-CoA−H]− and 193.050 [FA-H]− (Table 1). Compounds 37, 41, and 43 showed a base peak at m/z 191.055 and fragment ions at m/z 197.045 [syringic acid−H]−, 163.038 [p-CoA−H]−, and 193.050 [FA−H]−, respectively, and were identified as 5-syringoylquinic, 5-p-coumaroylquinic, and 5-feruloylquinic (FQA) acids [22,23,24] (Table 1).
With respect to compound 46, the base peak at 197.045 [syringic acid-H]−, together with a diagnostic fragment ion at m/z 335.077 [CQA−H−H2O]− indicated syringoyl–caffeoylquinic acid. Additionally, two hexosides of neochlorogenic (32) and chlorogenic acid (34) were also dereplicated (Table 1).
2.1.5. Acylhexaric Acids
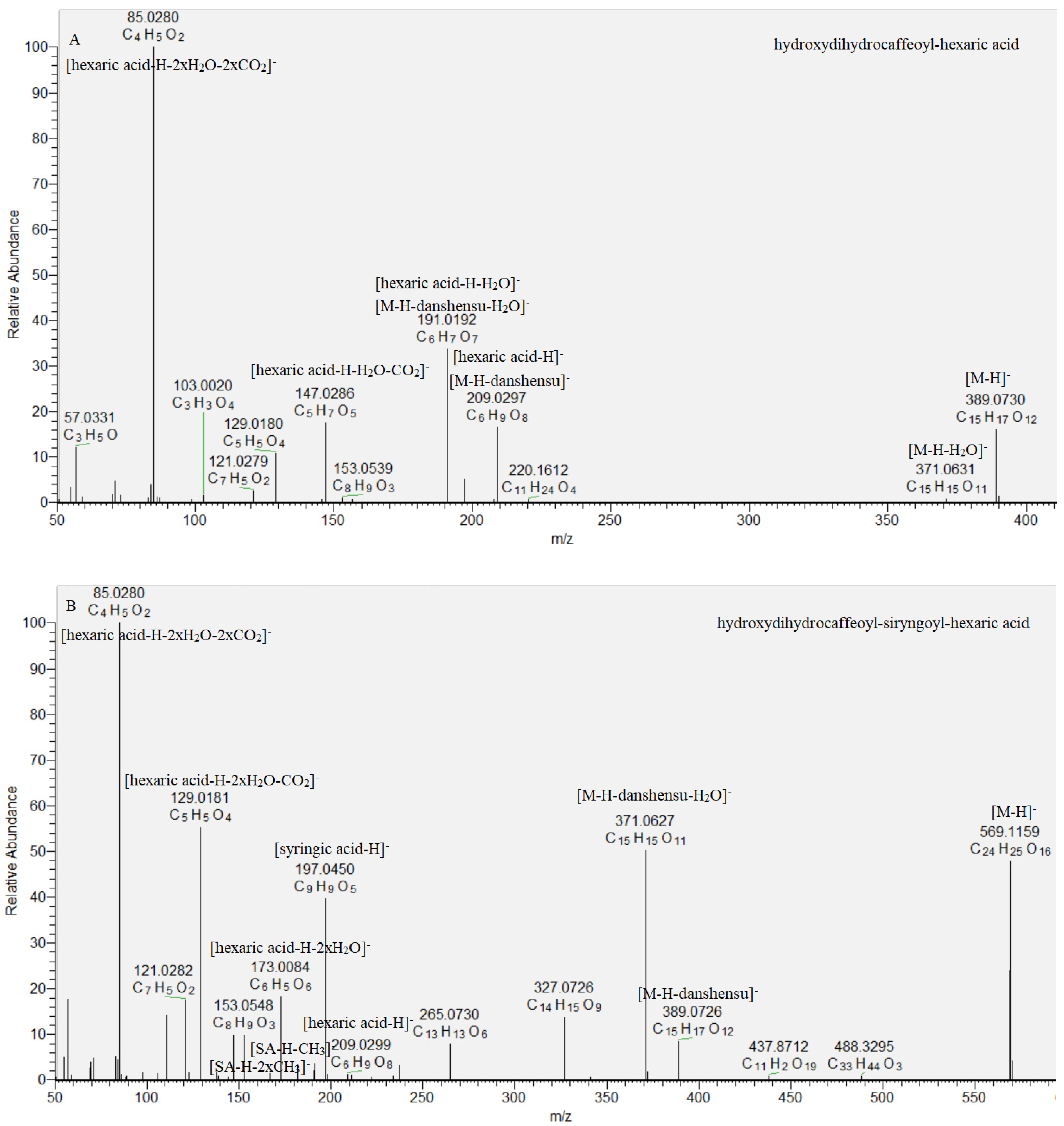
Key steps in the acylhexaric acids annotation were the subsequent losses of one hydroxydihydrocaffeoyl (47, 48) and two (49, 50) hydroxydihydrocaffeoyl and syringoyl residues (Table 1, Figure 4). Thus, the base peak in the MS/MS spectra was consistent with [hexaric acid (HA)−H]− at m/z 209.030 (C6H9O8) supported by the series of indicative ions at m/z 191.019 [HA-H-H2O]−, 147.029 [HA−H−H2O−CO2]−, 129.018 [HA−H−2H2O−CO2]−, 111.007 [HA−H−3H2O−CO2]−, and 85.028 [HA−H−2H2O−2CO2]− (Table 1) [25]. Compounds 49 and 50 shared the same [M−H]− at m/z 569.116. They formed the prominent fragment ions at m/z 389.073 [M−H−180.04]−, 371.063 [M−H−198.05]−, and 209.030 [M−2 × 180.04]−, resulting from the concomitant loss of hydroxydihydrocaffeoyl and syringoyl residues. Syringoyl moiety was suggested by the fragment ions at m/z 197.045 [syringic acid (SA)−H]−, 182.021 [(SA−H)−CH3]•− and 153.055 [(SA−H)−CO2]−. Compounds 49 and 50 were identified as isomeric hydroxydihydrocaffeoyl–syringoyl–hexaric acids (Table 1, Figure 4). Acylhexaric acids are reported for the first time in S. scardica.
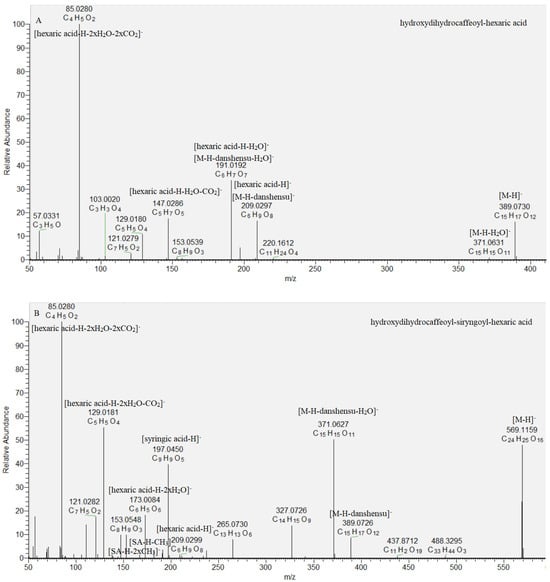
Figure 4.
MS/MS spectrum of (A) hydroxydihydrocaffeoyl–hexaric acid (47) and (B) hydroxydihydrocaffeoyl–siryngoyl–hexaric acid (49).
2.1.6. Phenylethanoid Glycosides
A class of secondary metabolites distinctive for Sideritis species were phenylethanoid glycosides [12]. The typical fragmentation pattern revealed the loss of 162.05, 146.05, 179.03, 18.01 Da—corresponding to glucosyl and rhamnosyl moieties—deprotonated caffeic acid, and H2O, respectively. Detailed discussion on the MS/MS fragmentation has been previously provided [12]. Based on a comparison with literature data, 10 phenylethanoid glycosides were dereplicated in the studied S. scardica extract (Table 1).
2.1.7. Iridoid Glycosides
The characteristic loss of hexose (−162.05 Da) and 7-(hydroxymethyl)-4,5-dihydrocyclopentapyran-4,5-diol (−182.06 Da, C9H10O4) indicated the presence of iridoid glycosides [12]. Compound 61 with deprotonated molecules at m/z 523.166 was dereplicated as melittoside [12]. Fragment ions at m/z 163.039, 179.034, and 193.050 corresponding to the deprotonated coumaric, caffeic, and ferulic acids, led to the identification of 62, 63, and 64 as p-coumaroylmelittoside, caffeoylmelitoside, and feruloylmelitosside, respectively. Compounds 63 and 64 were found for the first time in S. scardica (Table 1). Previously, 63 has been isolated from S. lanata [26], while 64 has been found in S. trojana [27].
2.1.8. Flavonoids
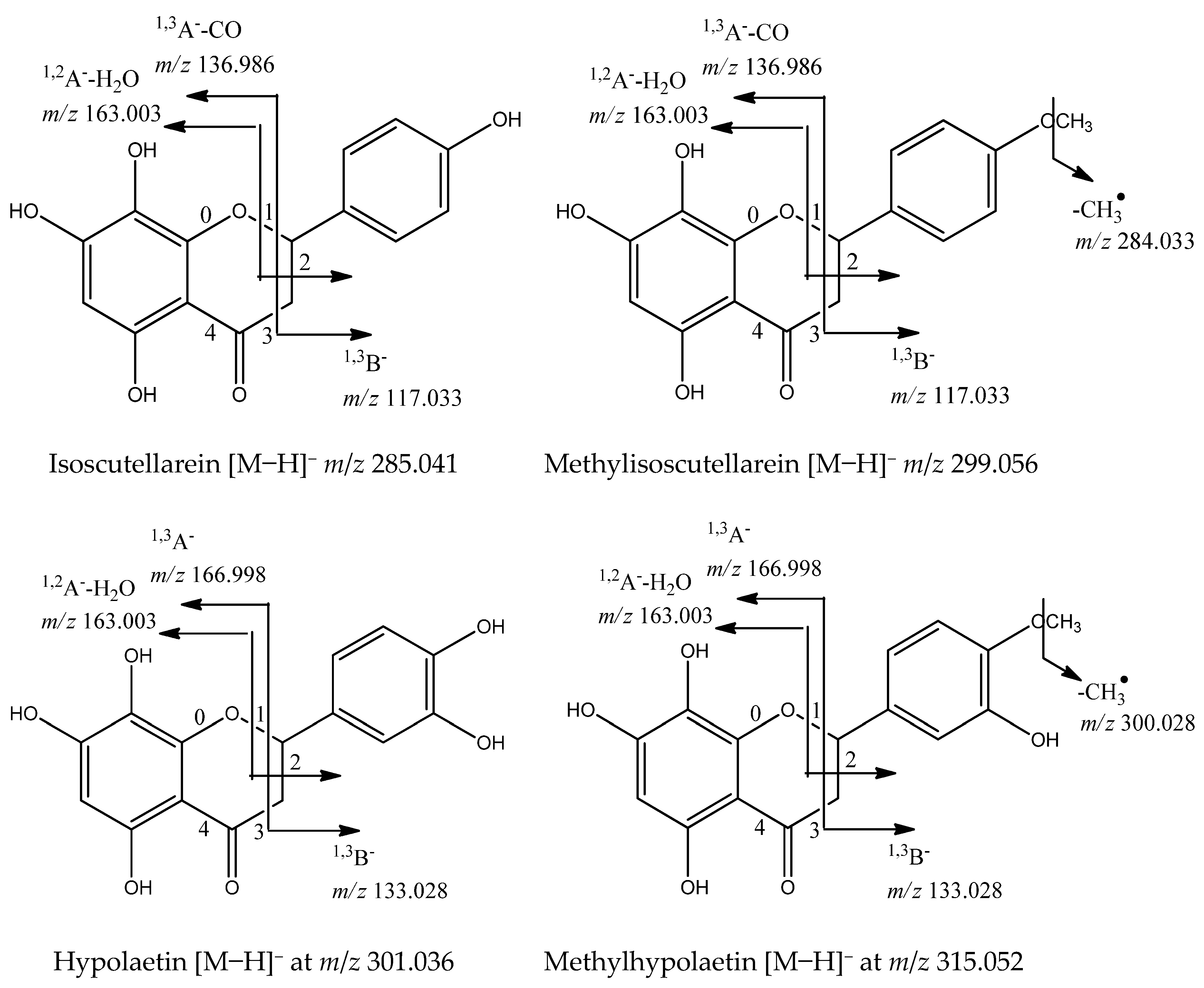
Flavonoids are the dominant secondary metabolites in Sideritis species [12]. The main flavonoids in the studied species were O-glycosides of the flavones isoscutellarein ([Agl−H]− at m/z 285.041), methylisoscutellarein ([Agl−H]− at m/z 299.056), hypolaetin ([Agl−H]− at m/z 301.036), methylhypolaetin ([Agl−H]− at m/z 315.052), and apigenin ([Agl−H]− at m/z 285.041). The MS/MS fragmentation patterns of the glycosides have been described previously [12] and are based on the loss of 162.054, 324.106, and 42.016 Da, corresponding to O-hexose/dihexose/acetyl group, respectively. The occurrence of a 180 Da loss [M−H-hex-H2O]− was indicative of 1→2 glycosylation between two sugars [16]. Significant fragments in the flavone aglycone annotation were a series of neutral losses of CO (−28 Da), CO2 (−44 Da), CH2O (−30 Da), and H2O (−18 Da), supported by the retro-Diels–Alder (rDA) cleavages 0,4A−, 1,2A−, 1,3A−, 1,2B−, and 1,3B− [22,23]. In general, five glycosides of isoscutellarein (IS) (70, 71, 72, 78, 83), three of methylisoscutellarein (MIS) (80, 82, 88), two of hypolaetin (HL) (67, 73), four of methylhypolaetin (MHL) (74, 76, 79, and 84), five of apigenin (Api) (65, 69, 75, 77, 86), and one of luteolin (Lu) (66) were dereplicated in the S. scardica extract (Table 1). Apigenin and luteolin were evidenced by the rDA ions at m/z 151.002 (1,3A−), 107.012 (0,4A−), 117.033 (1,3B−) (Api), and 133.028 (1,3B−) (Lu). The aglycones isoscutellarein and hypolaetin were deduced from the rDA cleavages 1,2A−-H2O at m/z 163.003, 1,3A−-CO at m/z 136.986, as well as 1,3B− at m/z 117.033 (IS) and 1,3B- at m/z 133.028 (HL). Their methoxylated derivatives revealed a fragment ion [Agl−H−CH3]•− at m/z 284.033 (MIS) and 300.028 (MHL). Compound 70 ([M−H]− at m/z 579.136) gave a base peak at m/z 285.041 [M−H−pent-hex]−. Thus, 70 was related to isoscutellarein O-pentosylhexoside. Illustrations of the fragmentation pathways of glycosides of the four abovementioned flavones aglycons are presented in Figure 5. The flavanone naringenin [M−H]− at m/z 271.061 (85) was identified based on the RDA fragments at m/z 151.002 (1,3A−), 107.012 (0,4A−), and 119.049 (1,3B−). In addition, its dihexoside (68) and coumaroylhexoside (87) were annotated based on the neutral loss of two hexoses and coumaroylhexose, respectively. Compounds 68, 70, 85, and 87 were annotated for the first time.
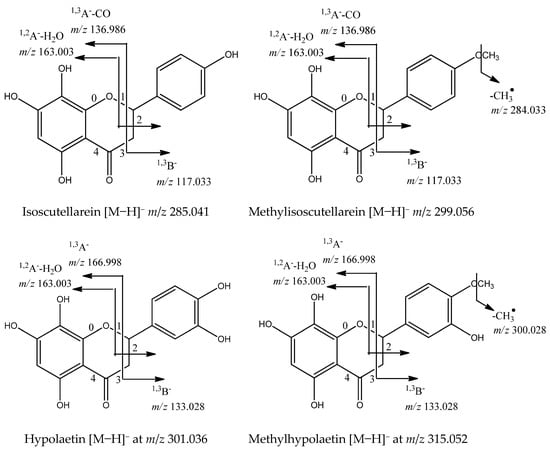
Figure 5.
Fragmentation pathways for flavones aglycons caused by cleavage of C-ring bonds in negative ion mode.
2.1.9. Fatty Acids and Organosulfur Compounds
A saturated (98), two monounsaturated (94 and 97), and four polyunsaturated (93, 95, 96, and 99) fatty acids were tentatively identified in S. scardica extract. Among them, the main fatty acids were trihydroxyoctadecadienoic acid (93) and trihydroxyoctadecenoic acid (94), previously reported for the species [12]. The dereplication and fragmentation pathway of dihydroxy fatty acids have been previously described [28]. In addition, four organosulfur compounds (100–103) were dereplicated based on fragment ions at m/z 96.959 [HO4S]− and 79.956 [O3S]−. These compounds have been previously described for S. scardica [12].

Table 1.
UHPLC–HRMS metabolite profiling of Sideritis scardica infusion with content (mg/g lyophilized infusion) of compounds assayed.
Table 1.
UHPLC–HRMS metabolite profiling of Sideritis scardica infusion with content (mg/g lyophilized infusion) of compounds assayed.
| No | Identified/Tentatively Annotated Compound | Molecular Formula | Exact Mass [M−H]− | Fragmentation Pattern in (-) ESI-MS/MS | tR (min) | Δ ppm | Level of Confidence [29] | Content [mg/g li] Mean ± SD |
|---|---|---|---|---|---|---|---|---|
| Sugar acids and saccharides | ||||||||
| 1. | Xylonic acid | C5H10O6 | 165.0405 | 165.0395 (60.5), 147.0288 (8.8), 129.0181 (12.3), 111.0073 (0.3), 105.0179 (33.1), 101.0228 (2.7), 87.0073 (43.1), 75.0072 (100) | 0.68 | −6.006 | 2 | - |
| 2. | Hexose | C6H12O6 | 179.0561 | 179.0541 (68.6), 161.0444 (11.1), 143.0338 (10.1), 125.0231 (1.53), 99.0437 (1.71), 81.0331 (4.8), 75.0072 (100) | 0.70 | −4.975 | 2 | - |
| 3. | Gluconic acid | C6H12O7 | 195.0510 | 195.0503 (96.1), 177.0396 (18.6), 159.0288 (14.7), 147.0287 (15.5), 141.0184 (5.9), 129.0180 (48.3), 111.0073 (5.3), 105.0179 (332.8), 75.0072 (100) | 0.72 | −3.619 | 2 | - |
| 4. | Pentose | C5H10O5 | 149.0456 | 149.0444 (21.6), 131.0334 (5.2), 101.0224 (0.8), 89.0229 (17.8), 75.0072 (100) | 0.74 | −7.358 | 2 | - |
| 5. | Asystoside | C25H44O15 | 583.2607 | 583.2618 (100), 421.2090 (4.9), 289.1662 (15.4), 161.0445 (15.8), 101.0230 (21.3), 71.0123 (30.6) | 6.34 | 1.880 | 2 | - |
| Carboxylic acids | ||||||||
| 6. | Quinic acid | C7H12O6 | 191.0561 | 191.0553 (100), 173.0446 (1.8), 155.0340 (0.2), 137.022 (0.2), 127.0387 (3.3), 99.0438 (0.6), 93.0331 (5.6), 85.0279 (18.2), 71.0123 (1.6), 59.0123 (1.37) | 0.69 | −4.194 | 2 | - |
| 7. | Oxaloglutaric acid | C7H8O7 | 203.0197 | 203.0191 (100), 159.0292 (2.3), 141.0181 (27.8), 115.0022 (11.7), 97.0279 (97.1), 95.0123 (14.2), 79.0174 (11.2), 72.9915 (7.1), 71.0123 (50.6), 69.0330 (66.2) | 0.88 | −2.984 | 2 | - |
| 8. | Citric acid | C6H8O7 | 191.0197 | 191.0191 (2.6), 173.0084 (1.43), 154.9979 (0.7), 147.0286 (0.4), 129.0181 (6.1), 111.0074 (100), 101.0231 (0.7), 87.0073 (43.8), 85.0280 (27.0) | 0.90 | −3.119 | 2 | - |
| Hydroxybenzoic, hydroxycinnamic acids, and their derivatives | ||||||||
| 9. | Gallic acid | C7H6O5 | 169.0143 | 169.0133 (37.5), 125.0231 (100), 97.0280 (3.8), 69.0330 (4.9) | 1.13 | −5.660 | 1 | 3.45 ± 0.51 |
| 10. | Hydroxybenzoic acid O-pentosylhexoside a | C18H24O12 | 431.1194 | 431.1200 (69.3), 299.0776 (2.3), 137.0232 (100), 93.0331 (73.0) | 1.78 | 1.231 | 2 | 1.32 ± 0.13 |
| 11. | Vanillic acid O-pentosylhexoside a | C19H26O13 | 461.1301 | 461.1310 (76.2), 329.0879 (1.3), 167.0340 (100), 152.0104 (52.4), 123.0438 (11.3), 108.0203 (27.4) | 1.99 | 2.052 | 2 | 2.27 ± 0.35 |
| 12. | Protocatechuic acid a | C7H6O4 | 153.0193 | 153.0182 (15.9), 123.0439 (0.1), 109.0281 (100), 81.0330 (1.4), 65.0380 (0.38) | 2.01 | −7.397 | 1 | 7.98 ± 0.54 |
| 13. | Caffeic acid O-rutinoside (swertiamacroside) | C21H28O13 | 487.1457 | 487.1465 (28.9), 179.0342 (100), 161.0234 (14.4), 135.0439 (45.5), 113.0232 (2.9) | 2.80 | 1.572 | 2 | 4.68 ± 0.45 |
| 14. | Caffeic acid O-dihexoside a | C21H28O14 | 503.1406 | 503.1414 (34.7), 341.0878 (42.9), 179.0341 (100), 135.0438 (77.1) | 2.82 | 1.513 | 2 | - |
| 15. | 2,3-Dihydroxybenzoic acid a | C7H6O4 | 153.0193 | 153.0182 (51.9), 123.0074 (26.7), 108.0203 (100), 95.0124 (32.1), 85.0280 (33.1) | 2.95 | −7.267 | 2 | - |
| 16. | p-Hydroxybenzoic acid a | C7H6O3 | 137.0244 | 137.0231 (35.2), 108.0200 (3.5), 93.0331 (100) | 2.97 | −9.614 | 1 | - |
| 17. | Dihydrocaffeic acid a | C9H10O4 | 181.0506 | 181.0499 (52.1), 163.0377 (0.4), 137.0596 (100), 135.0439 (19.6), 123.0436 (58.0), 121.0282 (24.9), 119.0489 (15.1), 109.0281 (27.6), 93.0332 (2.5), 59.0124 (86.3) | 3.33 | −4.154 | 2 | - |
| 18. | 2,4-Dihydroxybenzoic acid a | C7H6O4 | 153.0193 | 153.0182 (76.8), 135.0075 (29.7), 123.0439 (0.26), 109.0281 (100), 108.0201 (0.2), 91.0174 (5.6), 81.0333 (0.3), 65.0381 (14.5) | 3.47 | −7397 | 1 | 4.77 ± 0.74 |
| 19. | p-Hydroxyphenyl acetic acid a | C8H8O3 | 151.0400 | 151.0389 (100), 136.0156 (20.1), 123.0075 (4.6), 109.0283 (11.5), 107.0489 (2.3) | 3.49 | −7.133 | 1 | - |
| 20. | Caffeic acid | C9H8O4 | 179.0350 | 179.0341 (21.3), 135.0439 (100), 117.0335 (0.7), 107.0487 (1.35), 91.0537 (0.5) | 3.54 | −4.759 | 1 | 87.25 ± 6.54 |
| 21. | Gentisic acid | C7H6O4 | 153.0193 | 153.0182 (45.9), 109.0281 (100), 91.0175 (1.1), 108.0203 (8.7), 81.0330 (1.9), 65.0382 (0.1) | 3.65 | −7.397 | 1 | 0.42 ± 0.05 |
| 22. | Ferulic acid O-rutinoside a,b | C22H30O13 | 501.1614 | 501.1618 (10.2), 193.0500 (100), 175.0393 (8.4), 160.0157 (9.7), 134.0361 (43.4), 113.0230 (5.5) | 3.78 | 3.125 | 2 | - |
| 23. | p-Coumaric acid O-hexoside a | C15H18O8 | 325.0928 | 163.0391 (100), 145.0288 (4.14), 119.0488 (35.7) | 3.81 | 3.197 | 2 | - |
| 24. | Syringic acid a | C9H10O5 | 197.0455 | 197.0449 (40.0), 182.0213 (48.9), 153.0547 (100), 138.0311 (11.5), 123.0075 (26.7), 121.0281 (85.6), 106.0046 (9.67), 95.0123 (8.1), 89.0018 (14.97) | 4.34 | −3.231 | 2 | 1.69 ± 0.26 |
| 25. | O-coumaric acid a | C9H8O3 | 163.0401 | 163.0390 (9.7), 135.0075 (0.2), 119.0489 (100) | 4.55 | −6.363 | 1 | - |
| 26. | Ferulic acid | C10H10O4 | 193.0506 | 193.0499 (100), 178.0260 (14.1), 165.0545 (13.5), 149.0600 (21.4), 134.0358 (12.3), 123.0438 (92.0), 79.0538 (4.1) | 5.17 | −3.637 | 1 | - |
| Caffeic acid oligomers | ||||||||
| 27. | Lithospermic acid a | C27H22O12 | 537.1038 | 537.1035 (7.1), 493.1142 (30.4), 339.0515 (100), 313.0726 (8.8), 295.0613 (23.6), 267.0671 (10.9), 179.0345 (12.4), 135.0440 (49.3) | 4.97 | −0.743 | 2 | 0.51 ± 0.05 |
| 28. | Salvianolic acid B a | C36H30O16 | 717.1460 | 717.1478 (49.4), 673.1570 (7.7), 537.1055 (28.0), 519.0946 (47.9), 493.1153 (5.9), 339.0512 (13.0), 321.0409 (26.0), 313.0717 (11.5), 295.0616 (100), 277.0513 (6.7), 229.0141 (8.7), 203.0346 (13.9), 197.0447 (2.5), 179.0340 (11.3), 161.0237 (1.1), 135.0439 (32.2), 109.0281 (71.9) | 6.06 | 2.401 | 2 | 1.42 ± 0.01 |
| 29. | Rosmarinic acid | C18H16O8 | 359.0772 | 359.0782 (14.1), 197.0449 (26.0), 179.0342 (11.3), 161.0233 (100), 135.0439 (14.64), 133.0282 (19.8), 109.0275 (0.4) | 6.33 | 2.56 | 1 | 6.07 ± 0.46 |
| 30. | Isosalvianolic acid C a | C26H20O10 | 491.0983 | 491.0991 (100), 311.0567 (98.4), 267.0666 (39.3), 265.0508 (3.9), 249.0559 (1.5), 197.0454 (2.2), 179.0339 (1.8), 135.0440 (48.7) | 7.29 | 1.466 | 2 | 0.42 ± 0.03 |
| 31. | Didehydrosalvianolic acid B a | C36H28O16 | 715.1305 | 715.1324 (52.4), 535.0894 (20.2), 517.0786 (5.4), 337.0357 (8.1), 319.0241 (12.4), 311.0575 (7.6), 293.0461 (100), 265.0503 (7.1), 197.0446 (8.9), 135.0438 (10.5), 109.0279 (5.5) | 7.76 | 2.786 | 2 | 0.19 ± 0.03 |
| Acylquinic acids | ||||||||
| 32. | (Neo)chlorogenic acid O-hexoside a | C22H28O14 | 515.1406 | 515.1414 (52.2), 353.0883 (5.6), 191.0553 (100), 179.0351 (3.4), 135.0441 (4.0), 93.0332 (11.0) | 2.13 | 1.536 | 2 | - |
| 33. | Neochlorogenic acid | C16H18O9 | 353.0877 | 353.0883 (40.7), 191.0555 (100), 179.0341 (61.2), 173.0451 (3.1), 161.0235 (3.5), 135.0439 (48.7), 93.0331 (4.5) | 2.36 | 0.575 | 1 | - |
| 34. | Chlorogenic acid O-hexoside a | C22H28O14 | 515.1406 | 515.1414 (100), 323.0767(51.9), 191.0554 (94.8), 179.0327 (4.5), 161.0238 (33.5), 135.0434 (6.4), 111.0435 (4.0) | 2.85 | 2.487 | 1 | - |
| 35. | 3-p-coumaroylquinic acid a | C16H18O8 | 337.0928 | 337.0932 (6.9), 191.0555 (6.9), 173.0449 (3.5), 163.0390 (100), 135.0437 (0.5), 119.0488 (26.5), 111.0438 (0.7), 93.0332 (0.8) | 2.99 | 0.918 | 2 | - |
| 36. | Chlorogenic acid | C16H18O9 | 353.0877 | 353.0879 (4.6), 191.0554 (100), 179.0380 (0.9), 161.0235 (1.9), 111.0437 (0.8), 93.0331 (2.7), 85.0280 (7.5) | 3.17 | 0.325 | 1 | 5.22 ± 0.21 |
| 37. | 5-Syringoylquinic acid a | C16H20O10 | 371.0983 | 371.0987 (36.1), 197.0448 (4.9), 191.0554 (100), 173.0443 (14.6), 153.0538 (2.3), 121.0279 (9.4), 111.0435 (2.8), 93.0331 (31.6), 85.0279 (3.7) | 3.30 | 0.862 | 3 | - |
| 38. | 4-Caffeoylquinic acid a | C16H18O9 | 353.0877 | 353.0883 (32.7), 191.0555 (39.50), 179.0342 (75.3), 173.0447 (100), 135.0439 (52.1), 111.0436 (3.3), 93.0331 (21.3) | 3.36 | 0.515 | 2 | 7.65 ± 0.96 |
| 39. | 3-Feruloylquinic acid | C17H20O9 | 367.1034 | 367.1036 (19.5), 193.0500 (100), 173.0447 (4.7), 137.0226 (3.4), 134.0361 (56.7) | 3.42 | 0.339 | 2 | 0.63 ± 0.04 |
| 40. | 4-Syringoylquinic acid a | C16H20O10 | 371.0983 | 371.0978 (9.8), 197.0456 (11.5), 191.0554 (100), 173.0443 (14.6), 153.0538 (2.3), 121.0283 (4.9), 111.0435 (2.8), 93.0331 (14.5) | 3.43 | −1.509 | 3 | - |
| 41. | 5-p-Coumaroylquinic acid a | C16H18O8 | 337.0928 | 337.0940 (8.9), 191.0554 (100), 173.0446 (6.6), 163.0390 (5.31), 119.0488 (4.8), 111.0437 (2.0), 93.0331 (16.7) | 3.96 | 3.172 | 2 | 1.85 ± 0.03 |
| 42. | 4-p-Coumaroylquinic acid a | C16H18O8 | 337.0928 | 337.0941 (8.6), 191.0555 (2.7), 173.0445 (100), 163.0390 (18.5), 119.0488 (9.0), 111.0436 (3.1), 93.0330 (22.2) | 4.02 | 3.558 | 2 | 1.82 ± 0.05 |
| 43. | 5-Feruloylquinic acid | C17H20O9 | 367.1034 | 367.1038 (23.7), 193.0500 (16.9), 191.0554 (100), 173.0446 (81.4), 155.0340 (3.8), 134.0361 (19.2), 111.0438 (4.9), 93.0331 (39.7) | 4.38 | 0.912 | 2 | 5.13 ± 0.08 |
| 44. | 1-p-Coumaroylquinic acid a | C16H18O8 | 337.0928 | 337.0932 (7.2), 191.0554 (100), 173.0445 (100), 135.0447 (2.3), 163.0393 (0.5), 119.0487 (1.1), 111.0437 (0.5), 93.0331 (5.3) | 4.60 | 1.007 | 2 | 0.56 ± 0.04 |
| 45. | 1-Feruloylquinic acid | C17H20O9 | 367.1034 | 367.1039 (10.2), 191.0554 (100), 179.0340 (0.5), 173.0448 (2.3), 161.0239 (0.3), 134.0360 (3.1), 111.0440 (1.41), 93.0331 (5.15) | 4.90 | −2.996 | 2 | 1.12 ± 0.16 |
| 46. | Syringoyl–caffeoylquinic acid a | C25H26O13 | 533.1300 | 533.1305 (14.5), 335.0777 (44.6), 291.0875 (20.7), 197.0450 (100), 153.0546 (12.6), 137.0232 (38.4), 123.0073 (17.0), 111.0439 (10.4), 93.0331 (49.0) | 6.90 | 0.799 | 2 | 0.81 ± 0.08 |
| Acylhexaric acids | ||||||||
| 47. | Hydroxydihydrocaffeoyl–hexaric acid a,b | C15H18O12 | 389.0725 | 389.0730 (16.8), 371.0631 (0.8), 209.0297 (16.8), 191.0192 (34.2), 153.0539 (1.0), 147.0286 (17.9), 129.0180 (10.8), 85.0280 (100) | 1.65 | 1.236 | 3 | 1.12 ± 0.16 |
| 48. | Hydroxydihydrocaffeoyl–hexaric acid isomer a,b | C15H18O12 | 389.0725 | 389.0728 (16.8), 371.0621 (2.7), 209.0294 (17.2), 197.0442 (3.7), 191.0191 (45.3), 173.0087 (3.1), 147.0286 (20.6), 129.0180 (13.5), 111.0079 (4.5), 85.0280 (100) | 2.60 | 0.773 | 3 | 0.81 ± 0.08 |
| 49. | Hydroxydihydrocaffeoyl–siryngoyl–hexaric acid a,b | C24H26O16 | 569.1148 | 569.1159 (49.1), 389.0726 (8.8), 371.0627 (52.7), 327.0726 (13.8), 209.0299 (1.4), 197.0450 (39.1), 191.0186 (3.7), 182.0211 (3.3), 173.0084 (18.4), 166.9975 (1.6), 147.0285 (10.0), 138.0309 (1.7), 129.0181 (54.5), 123.0072 (1.6), 121.0282 (17.5), 111.0073 (14.3), 97.6908 (1.7), 85.0280 (100) | 3.53 | 1.937 | 3 | 1.77 ± 0.15 |
| 50. | Hydroxydihydrocaffeoyl–siryngoyl–hexaric acid isomer a,b | C24H26O16 | 569.1148 | 569.1158 (43.9), 389.0731 (7.6), 371.0625 (42.8), 327.0728 (12.2), 209.0295 (1.13), 197.0450 (40.8), 191.0186 (3.8), 182.0214 (4.3), 173.0084 (15.4), 166.9978 (1.0), 153.0548 (10.2), 147.0288 (8.0), 138.0315 (2.2), 129.0181 (59.1), 123.0074 (3.6), 121.0281 (16.5), 111.0073 (12.5), 85.0280 (100) | 3.77 | 2.148 | 3 | 3.53 ± 0.237 |
| Phenylethanoid glycosides | ||||||||
| 51. | Decaffeoyl aceteoside/verbasoside | C20H30O12 | 461.1664 | 461.1673 (100), 315.1085 (5.2), 297.0984 (2.6), 135.0439 (29.4), 113.0230 (46.0), 85.0280 (20.6), 71. 0123 (20.7) | 2.57 | 0.881 | 2 | 35.09 ± 2.46 |
| 52. | Hydroxyverbascoside | C29H36O16 | 639.1931 | 639.1946 (86.8), 621.1832 (5.3), 459.1533 (1.9), 179.0342 (36.0), 161.0232 (5.3), 135.0440 (22.3), 133.0283 (37.6), 113.0231 (8.6) | 4.49 | 2.381 | 2 | 13.45 ± 0.47 |
| 53. | Verbascoside | C29H36O15 | 623.1987 | 623.1996 (60.3), 461.1674 (9.3), 315.1078 (1.80), 179.0340 (2.4), 161.0234 (100), 135.0440 (8.7), 133.0283 (23.9) | 5.48 | 2.305 | 2 | 151.54 ± 10.86 |
| 54. | Echinacoside | C35H46O20 | 785.2509 | 785.2529 (72.5), 623.2175 (7.7), 461.1666 (6.3), 179.0349 (3.6), 161.0234 (100), 135.0440 (24.9), 133.0282 (47.0) | 5.23 | 2.424 | 2 | 0.74 ± 0.005 |
| 55. | Forsythoside B/samioside/lavandulifolioside | C34H44O19 | 755.2403 | 755.2424 (80.5), 593.2080 (8.2), 461.1675 (8.7), 267.1614 (1.5), 179.0341 (6.8), 161.0234 (100), 135.0439 (23.5), 133.0282 (45.8), 113.0231 (9.4) | 5.38 | 2.606 | 2 | 6.57 ± 0.103 |
| 56. | Alyssonoside | C35H46O19 | 769.2560 | 769.2578 (100), 593.2076 (8.5), 461.1658 (8.9), 193.05 (20.9), 175.0393 (44.1), 161.0236 (10.2), 160.0156 (44.4), 135.0442 (15.4), 134.0362 (20.9), 132.0206 (14.9), 123.0439 (7.4), 113.023 (15.1), 85.0281 (9.3), 71.0124 (10.0) | 6.07 | 22.207 | 2 | 2.16 ± 0.14 |
| 57. | Leucoseptoside A | C30H38O15 | 637.2138 | 637.2154 (100), 461.1669 (16.2), 315.1091 (3.8), 193.0501 (13.00), 175.0392 (67.8), 161.0233 (109), 160.0155 (60.1), 113.0230 (18.9) | 6.25 | 2.505 | 2 | 22.80 ± 0.82 |
| 58. | Leontoside B/stachyoside D | C36H48O19 | 783.2716 | 783.2734 (100), 193.0501 (34.7), 175.0392 (92.4), 167.0700 (1.5), 160.0156 (73.8), 132.0205 (26.8) | 6.97 | 2.142 | 2 | 0.90 ± 0.05 |
| 59. | Acetylverbascoside | C31H38O16 | 665.2087 | 665.2103 (65.0), 503.1778 (3.3), 461.1676 (3.1), 161.0234 (100), 179.0339 (2.9), 135.0440 (10.4), 133.0283 (38.4), 113.0229 (1.9) | 6.99 | 2.348 | 2 | 0.90 ± 0.01 |
| 60. | Martynoside | C31H40O15 | 651.2294 | 651.2317 (95.8), 475.1836 (1.2), 329.1252 (1.1), 193.0500 (13.6), 175.0392 (100), 160.0156 (69.1), 132.0204 (25.1), 113.0230 (14.0) | 7.21 | 3.450 | 2 | 11.35 ± 0.75 |
| Iridoid glycosides | ||||||||
| 61. | Melittoside | C21H32O15 | 523.1668 | 523.1676 (10.9), 361.1139 (4.5), 343.1040 (5.4), 325.0919 (1.3), 313.9523 (0.4), 283.0820 (0.5), 253.0722 (1.3), 223.0613 (1.7), 205.0506 (1.6), 179.0553 (100), 161.0447 (16.8), 119.0337 (34.9), 101.0230 (30.8), 89.0229 (80.9) | 1.33 | 1.446 | 2 | 13.22 ± 1.36 |
| 62. | p-Coumaroylmelittoside | C30H38O17 | 669.2035 | 669.2049 (100), 489.1423 (5.4), 325.0932 (28.3), 307.0823 (5.0), 265.0729 (3.2), 235.0605 (3.4), 205.0499 (16.9), 163.0390 (71.9), 145.0283 (84.4), 119.0488 (46.4), 93.0330 (4.9), 89.0230 (6.4) | 3.98 | 1.909 | 2 | 3.71 ± 0.06 |
| 63. | Caffeoylmelittoside a,b | C30H38O18 | 685.1974 | 685.1998 (100), 649.1036 (1.3), 523.1473 (1.9), 187.0396 (1.1), 181.0498 (24.9), 179.0342 (61.7), 163.0391 (39.9), 161.0235 (12.7), 135.0440 (78.4), 93.0331 (2.4), 89.0227 (2.3) | 4.79 | 1.799 | 2 | 0.70 ± 0.01 |
| 64. | Feruloylmelittoside a,b | C31H40O18 | 699.2142 | 699.2161 (75.9), 519.1519 (8.4), 357.0992 (54.0), 193.0500 (100), 163.0389 (30.6), 135.0435 (16.10), 134.0361 (70.0) | 5.56 | 2.778 | 2 | - |
| Flavonoids | ||||||||
| 65. | Apigenin 6,8-C-hexosyl hexoside | C27H30O15 | 593.1522 | 593.1522 (100), 503.1212 (4.8), 473.1094 (14.5), 413.0891 (2.0), 395.0770 (0.8), 383.0776 (18.0), 353.0672 (30.1), 325.0729 (2.8), 297.0770 (10.7), 161.0235 (2.2), 117.0333 (3.3) | 4.05 | 1.630 | 2 | 1.59 ± 0.18 |
| 66. | Luteolin 7-O-dihexoside | C27H30O16 | 609.1461 | 609.1473 (100), 447.0949 (4.7), 285.0408 (61.9), 284.0331 (18.1), 256.0380 (1.0), 151.0025 (3.8), 133.0281 (3.4), 107.0121 (2.1) | 4.94 | 2.023 | 2 | 0.58 ± 0.01 |
| 67. | Hypolaetin 7-O-hexosyl (1→2)-hexoside | C27H30O17 | 625.1410 | 625.1422 (100), 463.0883 (6.6), 445.0775 (6.5), 301.0356 (88.8), 300.0279 (30.4), 283.0244 (0.7), 255.0292 (3.6), 227.0350 (2.3), 166.9975 (3.3), 163.0029 (1.1), 137.0232 (4.0), 133.0280 (7.5) | 5.08 | 1.900 | 1 | 5.30 ± 0.09 |
| 68. | Naringenin 7-O-dihexoside | C27H32O15 | 595.1668 | 595.1658 (100), 475.1172 (4.6), 355.0674 (5.6), 271.0619 (54.8), 270.0215 (1.8), 269.0457 (38.2), 151.0027 (43.4), 119.0488 (21.5), 107.0123 (18.3) | 5.45 | −1.703 | 2 | - |
| 69. | Apigenin 7-O-allosyl (1→2) glucoside | C27H30O15 | 593.1512 | 593.1522 (64.7), 431.0986 (3.0), 269.0459 (100), 225.0546 (0.7), 151.0027 (1.4), 161.0232 (1.7), 117.0333 (3.3), 107.0125 (1.8) | 5.50 | 1.731 | 2 | 1.93 ± 0.04 |
| 70. | Isoscutellarein 7-O-pentosyl–hexoside a,b | C26H28O15 | 579.1356 | 579.1366 (38.9), 461.0086 (0.4), 285.0408 (100), 257.0451 (1.7), 241.0506 (1.6), 229.0497 (1.5), 213.0554 (5.1), 187.0393 (4.4), 136.9867 (0.9), 117.0330 (0.5) | 5.51 | 1.825 | 2 | 1.01 ± 0.05 |
| 71. | Isoscutellarein 7-O-hexosyl (1→2)-hexoside | C27H30O16 | 609.1461 | 609.1472 (87.2), 447.0912 (0.4), 429.0833(6.8), 285.0409 (100), 284.0328 (9.5), 255.0303 (2.2), 167.0496 (0.5), 163.0028 (2.1), 136.9868 (1.7), 117.0333 (1.8) | 5.62 | 1.826 | 2 | 47.47 ± 0.95 |
| 72. | Isoscutellarein 7-O-hexoside | C21H20O11 | 447.0933 | 447.0941 (26.4), 285.0408 (100), 229.0509 (1.0), 136.9868 (2.3), 117.0332 (1.1) | 5.81 | 1.801 | 2 | 0.58 ± 0.01 |
| 73. | Hypolaetin 7-O-acetylhexosiyl–hexoside | C29H32O18 | 667.1516 | 625.1406 (3.2), 463.0898 (3.2), 445.079 (6.6), 301.0357 (76.0), 300.0278 (30.8), 283.0285 (1.1), 255.0298 (1.4), 227.0347 (1.0), 166.9973 (1.9), 163.0020 (1.0), 137.0232 (4.0), 133.0284 (8.3), 109.0282 (1.0) | 5.93 | 3.152 | 2 | 19.17 ± 1.07 |
| 74. | Methylhypolaetin 7-O-dihexoside | C28H32O17 | 639.1567 | 639.1579 (75.3), 315.0516 (100), 300.0279 (40.7), 271.0264 (1.2), 243.0296 (2.5), 165.9901 (0.7), 136.9871 (6.2), 117.1944 (0.5), 133.0283 (2.5) | 5.96 | 1.920 | 2 | 17.33 ± 0.25 |
| 75. | Apigenin 7-O-glucoside | C21H20O10 | 431.0984 | 431.0988 (100), 269.0453 (24.9), 268.0381 (54.3), 211.0395 (1.6), 151.0025 (3.2), 117.0330 (1.8), 170.0124 (2.0) | 6.06 | 1.044 | 1 | 0.46 ± 0.02 |
| 76. | Methylhypolaetin 7-O-hexoside | C22H22O12 | 477.1039 | 477.1045 (30.5), 315.0515 (100), 300.0278 (32.0), 227.0350 (1.6), 136.9870 (6.2) | 6.13 | 1.406 | 2 | 0.26 ± 0.01 |
| 77. | Apigenin 7-O-[6‴-O-acetyl]-hexosyl(1→2)-hexoside | C29H32O16 | 635.1618 | 635.1629 (60.6), 593.1563 (1.1), 431.0981 (2.3), 269.0458 (100), 225.0560 (12.4), 151.0024 (1.7), 117.0332 (5.1), 107.0126 (2.6) | 6.44 | 1.798 | 2 | 2.18 ± 0.003 |
| 78. | Isoscutellarein 7-O-hexosyl-(1→2)-[6″-O-acetyl]-hexoside | C29H32O17 | 651.1567 | 651.1581 (69.3), 429.0831 (8.9), 285.0408 (100), 255.0285 (1.0), 239.0344 (1.1), 163.0026 (1.5), 136.9863 (0.8), 117.0334 (1.1) | 6.58 | 2.161 | 2 | 151.70 ± 14.79 |
| 79. | 4′-Methylhypolaetin 7-O-acetyl–hexosyl–hexoside | C30H34O18 | 681.1672 | 681.1688 (89.6), 639.1533 (1.2), 357.0594 (0.9), 315.0516 (100), 300.0279 (44.2), 271.0248 (1.6), 243.0291 (1.4), 136.9868 (7.9), 133.0283 (4.1) | 6.83 | 2.236 | 2 | 78.33 ± 3.29 |
| 80. | 4′-Methylisoscutellarein 7-O-dihexoside | C28H32O16 | 623.1618 | 623.1630 (100), 461.1117 (0.6), 299.0565 (83.6), 284.0330 (39.2), 255.0299 (3.0), 117.0330 (0.7) | 7.23 | 1.929 | 2 | 24.20 ± 0.98 |
| 81. | Tremasperin | C30H34O16 | 649.1774 | 649.1786 (5.4), 607.1672 (2.6), 283.0616 (100), 268.0381 (55.5), 284.0649 (5.8), 240.0431 (2.2), 151.0024 (0.5) | 8.21 | 1.913 | 2 | 0.55 ± 0.04 |
| 82. | 4′-O-methylisoscutellarein 7-O-[6‴-O-acetyl]hexosyl-(1→2)hexoside | C30H34O17 | 665.1723 | 665.1740 (84.8), 299.0565 (100), 284.0330 (30.6), 255.0293 (2.5), 240.0429 (2.5), 227.0343 (2.5), 163.0025 (1.1), 136.9867 (9.2), 117.0338 (1.9) | 8.24 | 2.447 | 2 | 107.44 ± 9.07 |
| 83. | Isoscutellarein 7-O-acetylhexosyl-O-acetylhexoside | C31H34O18 | 693.1672 | 693.1663 (85.8), 471.0903 (7.4), 285.0407 (100), 213.0551 (6.0), 163.0022 (4.5), 136.9864 (1.9), 117.0331 (3.4) | 8.26 | −1.323 | 2 | - |
| 84. | Methylhypolaetin 7-O-acetylhexosyl-O-acetylhexoside | C32H36O19 | 723.1778 | 723.1794 (89.7), 315.0515 (100), 300.0280 (44.9), 271.0255 (1.1), 243.0298 (1.4), 199.0390 (4.6), 136.9866 (9.5), 133.0284 (5.9) | 8.47 | 2.182 | 2 | 0.31 ± 0.02 |
| 85. | Naringenin | C15H12O5 | 271.0612 | 271.0615 (100), 227.0701 (0.7), 165.0180 (2.5), 151.0025 (65.7), 125.0228 (1.3), 119.0489 (52.3), 107.0124 (15.5), 93.0331 (11.9) | 8.60 | 1.193 | 2 | - |
| 86. | Apigenin 7-O-p-coumaroyl-O-hexoside | C30H26O12 | 577.1352 | 577.1360 (100), 431.0990 (13.4), 413.0890 (7.9), 269.0459 (77.0), 145.0283 (83.4), 163.0391 (4.4), 117.0332 (38.3), 107.0121 (1.1), 151.0026 (2.1) | 9.06 | 1.457 | 2 | 0.75 ± 0.01 |
| 87. | Naringenin 7-O-coumaroylhexoside a | C30H28O12 | 579.1508 | 579.1514 (100), 415.1033 (3.9), 307.0829 (12.0), 271.0616 (79.4), 151.0026 (40.9), 163.0391 (15.7), 145.0283 (57.4), 119.0489 (40.9), 107.0125 (15.2), 117.0332 (21.9) | 9.15 | 0.985 | 2 | 1.09 ± 0.09 |
| 88. | 4′-Methylisoscutellarein 7-O-(6‴-acetyl)-hexosyl(1→2)-[6′-O-acetyl]hexoside | C32H36O18 | 707.1829 | 707.1844 (12.1), 299.0565 (100), 284.0330 (31.9), 300.0598 (6.8), 298.0496 (8.2), 255.0292 (4.7), 240.0424 (3.2), 227.0341 (1.7), 163.0023 (3.5), 136.9867 (10.7), 117.0332 (1.5) | 9.89 | 2.125 | 2 | 0.09 ± 0.003 |
| 89. | Pectolinarigenin | C17H14O6 | 313.0718 | 313.0721 (100), 298.0485 (53.9), 283.0251 (52.0), 269.0468 (2.4), 255.0302 (14.4), 227.0342 (2.17), 211.0386 (1.3), 183.0446 (2.1), 178.9918 (3.0), 163.0031 (11.8), 135.0075 (2.8), 117.0331 (13.1) | 10.36 | 0.922 | 1 | 3.11 ± 0.50 |
| 90. | Eupatilin | C18H16O7 | 343.0823 | 343.0826 (85.2), 328.0594 (100), 313.0360 (54.6), 298.0125 (13.3), 285.0412 (2.0), 270.0174 (42.6), 257.0095 (2.6), 133.0282 (3.7), 123.0439 (4.6) | 11.05 | 0.915 | 2 | 0.47 ± 0.07 |
| 91. | 8-Methoxycirsilineol | C18H16O7 | 343.0823 | 343.0826 (100), 328.0594 (48.7), 313.0360 (71.8), 299.0952 (0.7), 298.0124 (209), 270.0175 (10.6), 242.0220 (4.2), 161.0233 (0.8), 117.0333 (8.9) | 11.24 | 0.828 | 2 | 14.15 ± 2.30 |
| 92. | Genkwanin | C16H12O5 | 283.0611 | 283.0615 (100), 268.0379 (67.4), 240.0428 (6.2), 239.0352 (1.8), 178.9915 (1.2), 151.0025 (4.1), 107.0125 (3.3) | 11.42 | 1.036 | 2 | - |
| Fatty acids | ||||||||
| 93. | Trihydroxyoctadecadienoic acid | C18H32O5 | 327.2177 | 327.2166 (100), 309.2069 (0.8), 291.1971 (3.5), 229.1443 (12.5), 211.1334 (16.2), 183.1383 (1.6), 171.1015 (6.0), 85.0280 (2.5), 57.0329 (0.9) | 9.15 | 0.986 | 2 | - |
| 94. | Trihydroxyoctadecenoic acid | C18H34O5 | 329.2334 | 329.2338 (100), 311.2232 (1.4), 293.2119 (0.4), 229.1442 (17.2), 211.1335 (23.2), 183.1381 (2.7), 171.1020 (4.4), 127.1115 (1.6) | 9.80 | 1.466 | 2 | - |
| 95. | Dihydroxyoctadecatrienoic acid | C18H30O4 | 309.2071 | 309.2076 (100), 291.1972 (54.3), 247.2075 (1.0), 185.1179 (5.4), 137.0959 (17.9), 97.0645 (4.1) | 10.90 | 1.641 | 2 | - |
| 96. | Dihydroxyoctadecadienoic acid | C18H32O4 | 311.2228 | 311.2233 (100), 293.2132 (7.2), 275.2029 (6.4), 201.1128 (61.0), 183.1387 (1.6), 171.1015 (11.2), 127.1114 (4.3)12.62 | 12.62 | 1.662 | 2 | - |
| 97. | Dihydroxyoctadecenoic acid | C18H34O4 | 313.2384 | 313.2389 (100), 295.2266 (5.8), 277.2166 (4.9), 201.1126 (42.8), 171.1012 (5.11), 127.1116 (4.8), 125.0960 (3.3) | 13.75 | 1.460 | 2 | - |
| 98. | Dihydroxyoctadecanoic acid | C18H36O4 | 315.2541 | 315.2544 (100), 297.2450 (4.3), 287.2241 (4.3), 171.1380 (0.6), 141.1272 (3.2), 127.1116 (0.6), 89.0230 (0.5) | 14.87 | 1.101 | 2 | - |
| 99. | Hydroxylinoleic acid | C18H32O3 | 295.2279 | 295.2282 (100), 277.2175 (14.6), 195.1384 (17.5), 113.0960 (1.1) | 15.98 | 1.056 | 2 | - |
| Organosulfur compounds | ||||||||
| 100. | Dodecyl sulfate | C12H26O4S | 265.1479 | 265.1482 (100), 96.9586 (66.4), 79.9558 (1.6) | 14.55 | 1.081 | 2 | - |
| 101. | Lauryl ether sulfate | C14H30O5S | 309.1741 | 309.1746 (100), 122.9746 (1.9), 104.9527 (0.2), 96.9586 (54.3), 79.9558 (6.6) | 16.10 | 1.624 | 2 | - |
| 102. | 4-Dodecylbenenesulfonic acid | C18H30O3S | 325.1842 | 325.1846 (100), 216.0095 (0.2), 183.0113 (46.5), 197.0272 (0.8), 184.0147 (1.9) | 17.44 | 0.957 | 2 | - |
| 103. | Myristyl sulfate | C14H30O4S | 293.1792 | 293.1796 (100), 96.9586 (73.6), 79.9558 (2.2) | 17.89 | 1.455 | 2 | - |
a—reported for the first time in the studied species; b—undescribed in the literature; level of confidence: 1—compound identified by comparison with reference standard; 2—putatively annotated compound; 3—putatively characterized compound classes.
2.2. Quantitative Determination
The quantitative determination of the main compounds in the profile of S. scardica lyophilized infusion was based on a common approach, where the HPLC analysis of the analytes was performed with a mobile phase composed of formic acid acetonitrile and water [22]. The content of the assayed compounds is revealed in Table 1. The main compounds in the tested lyophilized infusion were isoscutellarein-7-O-hexosyl-(1→2)-[6″-O-acetyl]-hexoside (78) and verbascoside (53), followed by 4′-O-methylisoscutellarein-7-O-[6‴-O-acetyl]hexosyl-(1→2)hexoside (82). Other dominant phenolic compounds include caffeic acid (20), 4′-methylhypolaetin-7-O-acetyl–hexosyl–hexoside (79), and isoscutellarein 7-O-hexosyl (1→2)-hexoside (71) (Table 1). Moreover, the data reveled moderate quantity of the phenylethanoid glycosides leucoseptoside A (57) and martynoside (60), as well as iridoid glycoside melittoside (61) (Table 1). The content of phenylethanoid glycosides ranged from 0.74 mg/g (54) to 151.54 mg/g lyophilized infusion (li) (53). With respect to caffeic acid oligomers, their quantities were found to range from 0.19 ± 0.033 mg/g li (31) to 6.07 ± 0.46 mg/g li (29), while caffeoylhexaric acids ranged from 0.81 ± 0.075 (48) to 3.53 ± 0.237 (50) (Table 1). However, this is the first attempt to quantify the secondary metabolites of the above classes in Sideritis species.
4-Caffeoylquinic acid (38) was found to be the dominant acylquinic acid (7.65 ± 0.96 mg/g li), followed by chlorogenic (36) and 5-feruloylquinic acid (43). Earlier quantitative research on the genus Sideritis showed that 5-caffeoylquinic acid has been found in all studied species as the most abundant hydroxycinnamic acid. In addition, the dominant phenolic compounds were the isoscutellarein derivatives isoscutellarein 7-O-[6‴-O-acetyl]-allosyl-(1→2)glucoside and 4′-O-methylisoscutellarein 7-O-allosyl-(1→2)-[6″-O-acetyl]-glucoside. Recently, eight compounds were detected in different Sideritis species: 5-caffeoylquinic acid, lavandulifolioside, verbascoside, isoscutellarein 7-O-allosyl(1→2)glucoside, hypolaetin 7-O-[6″-O-acetyl]-allosyl(1→2)glucoside, isoscutellarein 7-O-[6″-O-acetyl]-allosyl(1→2) glucoside, 3′-O-methylhypolaetin 7-O-[6″-O-acetyl]-allosyl(1→2)glucoside, 4′-O-methylhypolaetin, and 7-O-[6″-O-acetyl]-allosyl-(1→2)-[6″-O-acetyl]-glucoside). They represent 50% to 80% of the total phenolic content in S. scardica, S. raeseri, S. syriaca, and S. Taurica, and up to 90% in S. lanata [6]. The most abundant compounds present in the analyzed Siderits samples belonged to the group of phenylethanoid glycosides. The content of phenylethanoid glycosides ranged from 1.22 mg/g dry herb for S. lanata to 108.3 mg/g dry herb for S. scardica from Rhodopi Mountain, Bulgaria. The contribution of phenylethanoid glycosides to total phenolic content was around 50% for all samples, except for S. lanata where it accounted only for around 7% [6]. Eleven acetylated glycosides of isoscutellarein, hypolaetin, methylhypolaetin and methylisoscutellarein were previously isolated from 80% EtOH extract [30]. The differences between previous studies and our results can be ascribed to the different extraction methods and solvents.
2.3. Study Strength, Limitation and Future Direction
The study strength is that the presented extraction method of infusion is similar to the approach used in traditional medicine to process S. scardica tea. Therefore, this provides an insight into the phytochemical composition of common tea used in the traditional medicine and in-home remedies. A notable contribution of this study is the first-time dereplication and fragmentation patterns of five caffeic acids oligomers and four acylhexaric acids in S. scardica, expanding the current understanding of its chemical profile. The quantitative analysis identified major compounds in S. scardica infusion, with phenylethanoid verbascoside, glycosides of isoscutellarein, methylisoscutelarein, hypolaetin, and caffeic acid standing out as significant constituents. The reported concentrations add quantitative depth to the qualitative richness of the chemical composition. However, there are some limitations to the proposed method. In the quantitative assessment, a semi-quantitation was conducted, multiple detected substances were quantified based on a standard with a similar, yet different, chemical structure, as detailed above. Hence, a variation in the ionization between a standard and analytes may be a limitation. Future quantification based on the individual isolated secondary metabolites is recommended. In addition, isolation and accurate identification of the newly annotated caffeic acid oligomers and caffeoylhexaric acids will strengthen the validity of the present work.
3. Materials and Methods
3.1. Chemicals
Acetonitrile (hypergrade for LC–MS), formic acid (for LC–MS), and methanol (analytical grade) were purchased from Chromasolv (Sofia, Bulgaria). The reference standards used for compound identification were obtained from Extrasynthese (Genay, France) for protocatechuic, gentisic acids, and apigenin. Chlorogenic, caffeic, rosmarinic, cichoric acid, pectolinarigenin, and scutellarein were supplied from Phytolab (Vesten-bergsgreuth, Bavaria, Germany).
3.2. Plant Material
S. scardica seedlings were bought from a certified greenhouse “Mursalski-biogroup” (Bulgaria) and subsequently bred on alluvial soil with sunny exposure in an herbal garden (Rayanovtsi village, Vidin region) in Bulgaria at 349 m a.s.l. (43.7023° N 22.5206° E). The plant was identified by one of the authors (D.Z.) according to Assenov (1989) [31]. The plant material (aerial parts of 4-year-old plants) was collected during the flowering stage in July 2022 and dried for one week in the shade at room temperature. Then it was comminuted with a grinder (Rohnson, R-942, 220–240 V, 50/60 Hz, 200 W, Prague, Czech Republic) and stored in a dry and cool place until further analysis. The fresh/dried mass ratio is 4:1.
3.3. Sample Extraction
Air-dried aerial parts (100 g) were infused twice with boiled water (1:20 w/v) and extracted for 15 min at room temperature. The herbal infusion was lyophilized (lyophilizer Biobase BK-FD10P, BIOBASE, Jinan, China) to yield crude extracts of 12.5 g.
3.4. UHPLC–HRMS Dereplication/Annotation
The UHPLC–HRMS analyses were performed, as described previously [22], using a Q Exactive Plus mass spectrometer (Thermo Fisher Scientific, Inc., Waltham, MA, USA) equipped with a heated electrospray ionization (HESI-II) probe (Thermo Scientific). The equipment was operated in negative ion mode within the m/z range from 130 to 2000 at a resolution of 70,000. Other instrument parameters for full MS mode were set as follows: automatic gain control (AGC) target 3 × 106, maximum injection time (IT) 100 ms, number of scan ranges 1. For the DD-MS2 mode, the instrument parameters were as follows: microscans 1, resolution 17,500, AGC target 1 × 105, maximum IT 50 ms, MSX count 1, Top5, isolation window 2.0 m/z, stepped normalized collision energy (NCE) 10, 20, 60 eV. The chromatographic separation was achieved on a reversed phase column Kromasil EternityXT C18 (1.8 µm, 2.1 × 100 mm) at 40 °C. The UHPLC analyses were run with a mobile phase containing 0.1% formic acid in water (A) and 0.1% formic acid in acetonitrile (B). The run time was 33 min. The gradient elution program was used as follows: 0–1 min, 0–5% B; 1–20 min, 5–30% B; 20–25 min, 30–50% B; 25–30 min, 50–70% B; 30–33 min, 70–95%; 33–34 min 95–5%B. The equilibration time was 4 min. The injection volume and the flow rate were set to 1 µL and 300 µL/min, respectively. Data acquisition was performed using Xcalibur 4.2 (Thermo Scientific, Waltham, MA, USA) instrument control/data handling software.
3.5. UHPLC–HRMS Quantification
The UHPLC–HRMS quantification was conducted using the external standard method. Standard calibrations of protocatechuic (12), gentisic (21), caffeic (20), rosmarinic (27), chlorogenic (36), cichoric acids, apigenin, and scutellarein were established at five data points covering the concentration range of each analyte according to the level expected in the plant samples. Working solutions containing 0.05, 0.025, 0.012, 0.006, and 0.003 mg/mL of the assayed analytes were prepared from a stock solution in methanol containing 0.1 mg/mL. Based on the similar structure, the quantity of 9, 10, 14, and 21 was determined based on the calibration curve of gentisic acid; 11, 12, 18, and 24 as protocatechuic acid; 13, 14, 20, 51–56, and 63 as caffeic acid; 27–31 as rosmarinic acid; 36, 38, 39, 41–44 as chlorogenic acid; 65, 66, 69, 75, 77, 86–91 as apigenin; while 70–74, 76, 78–84, and 88 as scutellarein. Regression equations were as follows: gentisic acid y = 1,977,866,070x + 2460.9184 (R2 = 0.9951) protocatechuic acid y = 1,416,556,589.51x + 1581.8936 (R2 = 0.9965); caffeic acid y = 1,133,573,161x − 410.2916 (R2 = 0.9928); rosmarinic acid y = 2,190,130,610x + 22,248.9144 (R2 = 0.9950); chlorogenic acid y = 4,602,799,047.3258x − 921.0750 (R2 = 0.9926); cichoric acid y = 4,589,524,443.3458x + 85.8499 (R2 = 0.9999); apigenin y = 4,594,027,463.2779x + 21,088.0103 (R2 = 0.9983); scutellarein y = 1,976,006,256x − 41,744.9522 (R2 = 0.9420). The peak areas were calculated by integrating the Area Under the Curve (AUC) of the full-scan intensity scans for the corresponding molecular ion. These scans were also filtered for the presence of the characteristic base peak. MZmine 2.53 software was applied to the UHPLC–HRMS raw files of the studied S. scardica lyophilized infusion to obtain the peak area in the quantitative analysis. Results are expressed as mg/g lyophilized infusion.
4. Conclusions
In conclusion, an in-depth phytochemical analysis of S. scardica infusion using UHPLC–HRMS was performed. More than 100 metabolites, including sugar acids and saccharides, carboxylic, hydroxybenzoic, hydroxycinnamic, acylquinic and acylhexaric acids, caffeic acids oligomers, phenylpropanoid and iridoid glycosides, flavonoids, fatty acids, and organosulfur compounds were dereplicated/annotated. In addition, 62 metabolites of S. scardica were quantified. The presented extraction method of infusion is similar to the approach used in traditional medicine to process S. scardica tea. Therefore, the performed state-of-the-art phytochemical analysis of S. scardica provide additional knowledge with respect to the chemical constituents of this valuable medicinal plant.
Author Contributions
Conceptualization, D.Z.-D. and R.G.; methodology, D.Z.-D. and Y.V.; software, D.Z.-D. and Y.V.; validation, D.Z.-D.; formal analysis, D.Z.-D.; investigation, D.Z.-D. and V.B.; resources, D.Z.-D.; data curation, D.Z.-D.; writing—original draft preparation, D.Z.-D.; writing—review and editing, D.Z.-D., V.B. and R.G.; visualization, D.Z.-D.; supervision, R.G.; project administration, D.Z.-D.; funding acquisition, D.Z.-D. All authors have read and agreed to the published version of the manuscript.
Funding
This study is financed by the European Union-NextGenerationEU, through the National Recovery and Resilience Plan of the Republic of Bulgaria, project No. BG-RRP-2.004-0004-C01.
Data Availability Statement
Data are contained within the article.
Acknowledgments
The authors are grateful to Stephan Mladenov, Faculty of Medicine, Medical University Sofia, for the supply of S. scardica plant material.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Petrova, A.; Vladimirov, V. Balkan Endemics in the Bulgarian Flora. Phytol. Balcan. 2010, 16, 293–311. [Google Scholar]
- Assenov, I.; Ganchev, G. Morphological Characteristics of Sideritis scardica Griseb. Pharmacia 1978, 2, 29–32. [Google Scholar]
- Evstatieva, L. Sideritis scardica Griseb. In Red Data Book of the Republic of Bulgaria; Peev, D., Ed.; Joint Edition of the Bulgarian Academy of Sciences & Ministry of Environment and Water of Bulgaria, Digital Edition; Bulgarian Academy of Sciences: Sofia, Bulgaria, 2011; Volume 1, Available online: http://e-ecodb.bas.bg/rdb/en/ (accessed on 1 December 2023).
- González-Burgos, E.; Carretero, M.E.; Gómez-Serranillos, M.P. Sideritis spp.: Uses, Chemical Composition and Pharmacological Activities—A Review. J. Ethnopharmacol. 2011, 135, 209–225. [Google Scholar] [CrossRef] [PubMed]
- Yaneva, I.; Balabanski, V. History of the Uses of Pirin Mountain Tea (Sideritis scardica Griseb.) in Bulgaria. Bulg. J. Public Health 2013, 5, 48–57. [Google Scholar]
- Stanoeva, J.P.; Stefova, M.; Stefkov, G.; Kulevanova, S.; Alipieva, K.; Bankova, V.; Aneva, I.; Evstatieva, L.N. Chemotaxonomic Contribution to the Sideritis species Dilemma on the Balkans. Biochem. Syst. Ecol. 2015, 61, 477–487. [Google Scholar] [CrossRef]
- Irakli, M.; Tsifodimou, K.; Sarrou, E.; Chatzopoulou, P. Optimization Infusions Conditions for Improving Phenolic Content and Antioxidant Activity in Sideritis scardica Tea Using Response Surface Methodology. J. Appl. Res. Med. Aromat. Plants 2018, 8, 67–74. [Google Scholar] [CrossRef]
- Todorova, M.; Trendafilova, A. Sideritis scardica Griseb.; An Endemic Species of Balkan Peninsula: Traditional Uses, Cultivation, Chemical Composition, Biological Activity. J. Ethnopharmacol. 2014, 152, 256–265. [Google Scholar] [CrossRef]
- Jeremic, I.; Petricevic, S.; Tadic, V.; Petrovic, D.; Tosic, J.; Stanojevic, Z.; Petronijevic, M.; Vidicevic, S.; Trajkovic, V.; Isakovic, A. Effects of Sideritis scardica Extract on Glucose Tolerance, Triglyceride Levels and Markers of Oxidative Stress in Ovariectomized Rats. Planta Med. 2019, 85, 465–472. [Google Scholar] [CrossRef]
- Wightman, E.; Jackson, P.A.; Khan, J.; Forster, J.; Heiner, F.; Feistel, B.; Suarez, C.G.; Pischel, I.; Kennedy, D.O. The Acute and Chronic Cognitive and Cerebral Blood Flow Effects of a Sideritis scardica (Greek Mountain Tea) Extract: A Double Blind, Randomized, Placebo Controlled, Parallel Groups Study in Healthy Humans. Nutrients 2018, 10, 955. [Google Scholar] [CrossRef]
- Żyżelewicz, D.; Kulbat-Warycha, K.; Oracz, J.; Żyżelewicz, K. Polyphenols and Other Bioactive Compounds of Sideritis Plants and Their Potential Biological Activity. Molecules 2020, 25, 3763. [Google Scholar] [CrossRef]
- Mróz, M.; Malinowska-Pańczyk, E.; Bartoszek, A.; Kusznierewicz, B. Comparative Study on Assisted Solvent Extraction Techniques for the Extraction of Biologically Active Compounds from Sideritis raeseri and Sideritis scardica. Molecules 2023, 28, 4207. [Google Scholar] [CrossRef] [PubMed]
- Kostadinova, E.; Nikolova, D.; Alipieva, K.; Stefova, M.; Stefkov, G.; Evstatieva, L.; Matevski, V.; Bankova, V. Chemical Constituents of the Essential Oils of Sideritis scardica Griseb. and Sideritis raeseri Boiss and Heldr. from Bulgaria and Macedonia. Nat. Prod. Res. 2007, 21, 819–823. [Google Scholar] [CrossRef] [PubMed]
- Petreska Stanoeva, J.; Stefova, M. Assay of Urinary Excretion of Polyphenols after Ingestion of a Cup of Mountain Tea (Sideritis scardica) Measured by HPLC-DAD-ESI-MS/MS. J. Agric. Food Chem. 2013, 61, 10488–10497. [Google Scholar] [CrossRef] [PubMed]
- Tadić, V.M.; Jeremic, I.; Dobric, S.; Isakovic, A.; Markovic, I.; Trajkovic, V.; Bojovic, D.; Arsic, I. Anti-inflammatory, Gastroprotective, and Cytotoxic Effects of Sideritis scardica extracts. Planta Med. 2012, 78, 415–427. [Google Scholar] [CrossRef] [PubMed]
- Petreska, J.; Stefova, M.; Ferreres, F.; Moreno, D.A.; Tomás-Barberán, F.A.; Stefkov, G.; Kulevanova, S.; Gil-Izquierdo, A. Potential Bioactive Phenolics of Macedonian Sideritis Species Used for Medicinal “Mountain Tea”. Food Chem. 2011, 125, 13–20. [Google Scholar] [CrossRef]
- Alipieva, K.; Petreska, J.; Gil-Izquierdo, Á.; Stefova, M.; Evstatieva, L.; Bankova, V. Influence of the Extraction Method on the Yield of Flavonoids and Phenolics from Sideritis spp. (Pirin Mountain Tea). Nat. Prod. Commun. 2010, 130, 51–54. [Google Scholar] [CrossRef]
- Al Kadhi, O.; Melchini, A.; Mithen, R.; Saha, S. Development of a LC-MS/MS Method for The Simultaneous Detection of Tricarboxylic Acid Cycle Intermediates in A Range of Biological Matrices. J. Anal. Methods Chem. 2017, 2017, 5391832. [Google Scholar] [CrossRef]
- Liu, A.H.; Guo, H.; Ye, M.; Lin, Y.H.; Sun, J.H.; Xu, M.; Guo, D.A. Detection, Characterization and Identification of Phenolic Acids in Danshen Using High-Performance Liquid Chromatography with Diode Array Detection and Electrospray Ionization Mass Spectrometry. J. Chromatogr. A 2007, 1161, 170–182. [Google Scholar] [CrossRef]
- Zheleva-Dimitrova, D.; Simeonova, R.; Gevrenova, R.; Savov, Y.; Balabanova, V.; Nasar-Eddin, G.; Bardarov, K.; Danchev, N. In vivo Toxicity Assessment of Clinopodium vulgare L. Water Extract Characterized by UHPLC-HRMS. Food Chem. Toxicol. 2019, 134, 110841. [Google Scholar] [CrossRef]
- Zhu, L.; Ma, S.; Li, K.; Xiong, P.; Qin, S.; Cai, W. Systematic Screening of Chemical Constituents in the Traditional Chinese Medicine Arnebiae Radix by UHPLC-Q-Exactive Orbitrap Mass Spectrometry. Molecules 2022, 27, 2631. [Google Scholar] [CrossRef]
- Gevrenova, R.; Zengin, G.; Sinan, K.I.; Zheleva-Dimitrova, D.; Balabanova, V.; Kolmayer, M.; Voynikov, Y.; Joubert, O. An In-Depth Study of Metabolite Profile and Biological Potential of Tanacetum balsamita L. (Costmary). Plants 2023, 12, 22. [Google Scholar] [CrossRef] [PubMed]
- Gevrenova, R.; Zheleva-Dimitrova, D.; Balabanova, V.; Voynikov, Y.; Sinan, K.I.; Mahomoodally, M.F.; Zengin, G. Integrated phytochemistry, bio-functional potential and multivariate analysis of Tanacetum macrophyllum (Waldst. & Kit.) Sch.Bip. and Telekia speciosa (Schreb.) Baumg. (Asteraceae). Ind. Crops Prod. 2020, 155, 112817. [Google Scholar]
- Clifford, M.N.; Knight, S.; Kuhnert, N. Discriminating between the six isomers of dicaffeoylquinic acid by LC-MS n. J. Agric. Food Chem. 2005, 53, 3821–3832. [Google Scholar] [CrossRef] [PubMed]
- Kleczek, N.; Malarz, J.; Gierlikowska, B.; Kiss, A.; Stojakowska, A. Constituents of Xerolekia speciosissima (L.) Anderb. (Inuleae), and Anti-Inflammatory Activity of 7,10-Diisobutyryloxy-8,9-epoxythymyl Isobutyrate. Molecules 2020, 25, 4913. [Google Scholar] [CrossRef] [PubMed]
- Alipieva, K.; Kostadinova, E.; Evstatieva, L.; Stefova, M.; Bankova, V. An iridoid and a flavonoid from Sideritis lanata L. Fitoterapia 2009, 80, 51–53. [Google Scholar] [CrossRef]
- Kirmizibekmez, H.; Ariburnu, E.; Masullo, M.; Festa, M.; Capasso, A.; Yesilada, E.; Piacente, S. Iridoid, phenylethanoid and flavonoid glycosides from Sideritis trojana. Fitoterapia 2012, 83, 130–136. [Google Scholar] [CrossRef]
- Sinan, K.I.; Zengin, G.; Zheleva-Dimitrova, D.; Etienne, O.K.; Mahomoodally, M.F.; Bouyahya, A.; Lobine, D.; Chiavaroli, A.; Ferrante, C.; Menghini, L.; et al. Qualitative Phytochemical Fingerprint and Network Pharmacology Investigation of Achyranthes aspera Linn. Extracts. Molecules 2020, 25, 1973. [Google Scholar] [CrossRef]
- Sumner, L.W.; Amberg, A.; Barrett, D.; Beale, M.H.; Beger, R.; Daykin, C.A.; Fan, T.W.-M.; Fiehn, O.; Goodacre, R.; Griffin, J.L. Proposed Minimum Reporting Standards for Chemical Analysis: Chemical Analysis Working Group (CAWG) Metabolomics Standards Initiative (MSI). Metabolomics 2007, 3, 211–221. [Google Scholar] [CrossRef]
- Moussavi, N.; Azizullah, H.; Malterud, K.; Inngjerdingen, K. Immunomodulating polyphenols from Sideritis scardica. J. Funct. Food 2022, 96, 105197. [Google Scholar] [CrossRef]
- Assenov, I. Sideritis L. In Flora of PR Bulgaria; Velchev, V., Ed.; Bulgarian Academy of Sciences Publishing House: Sofia, Bulgaria, 1989; Volume 9, pp. 369–374. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).