Use of H2O2 to Cause Oxidative Stress, the Catalase Issue
Abstract
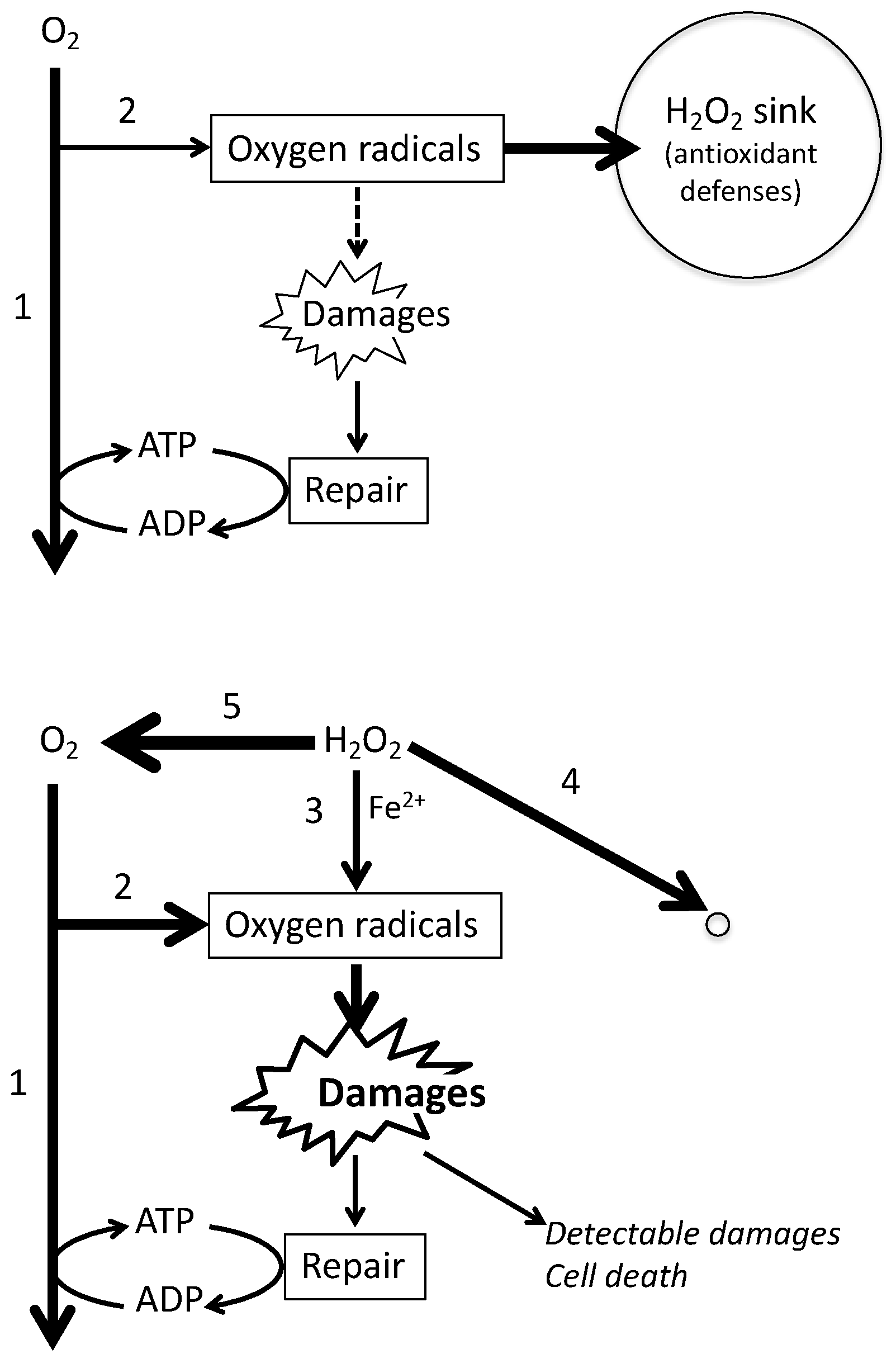
1. Introduction
2. Results
2.1. Values for H2O2 Concentration in the Literature
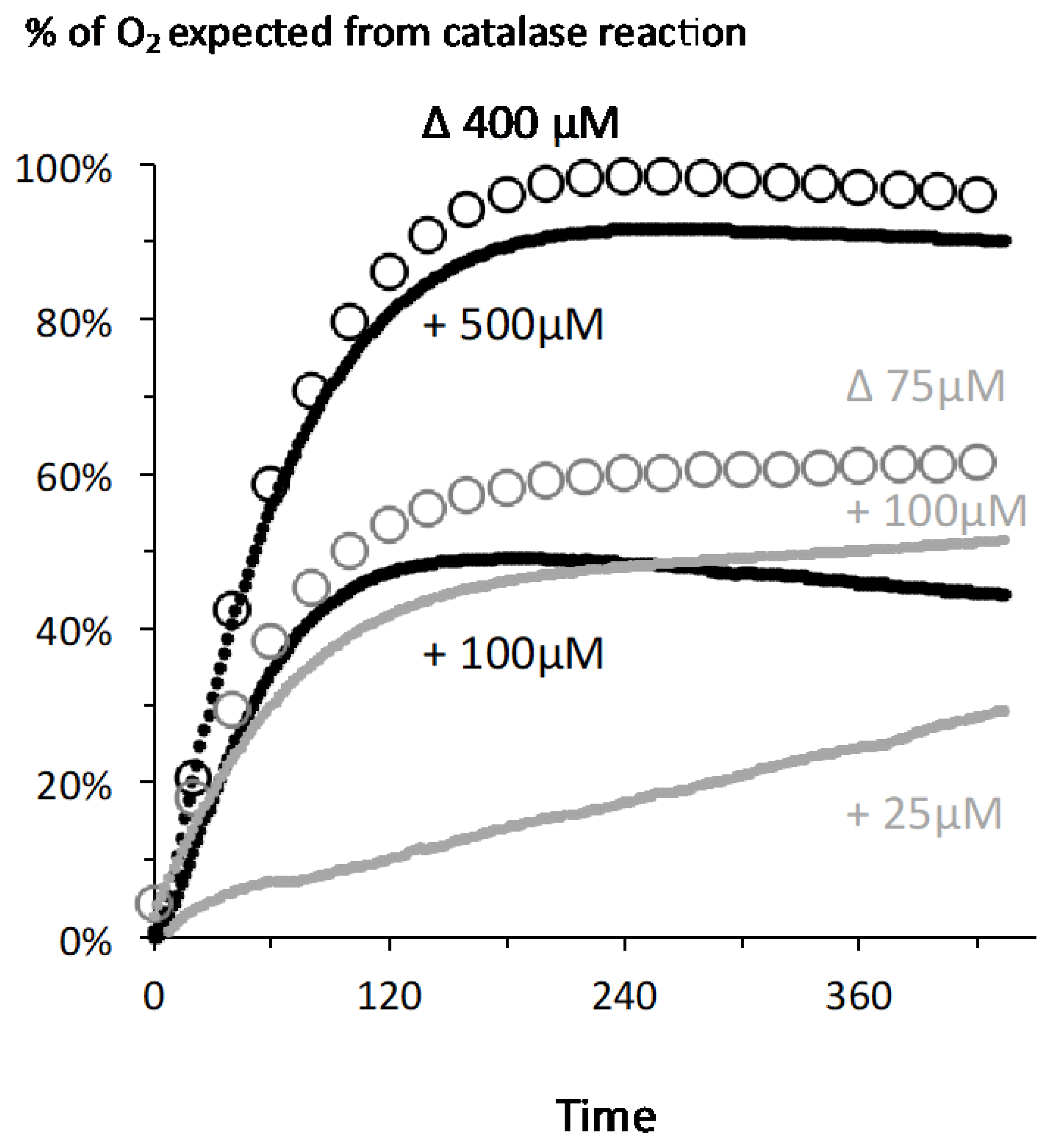
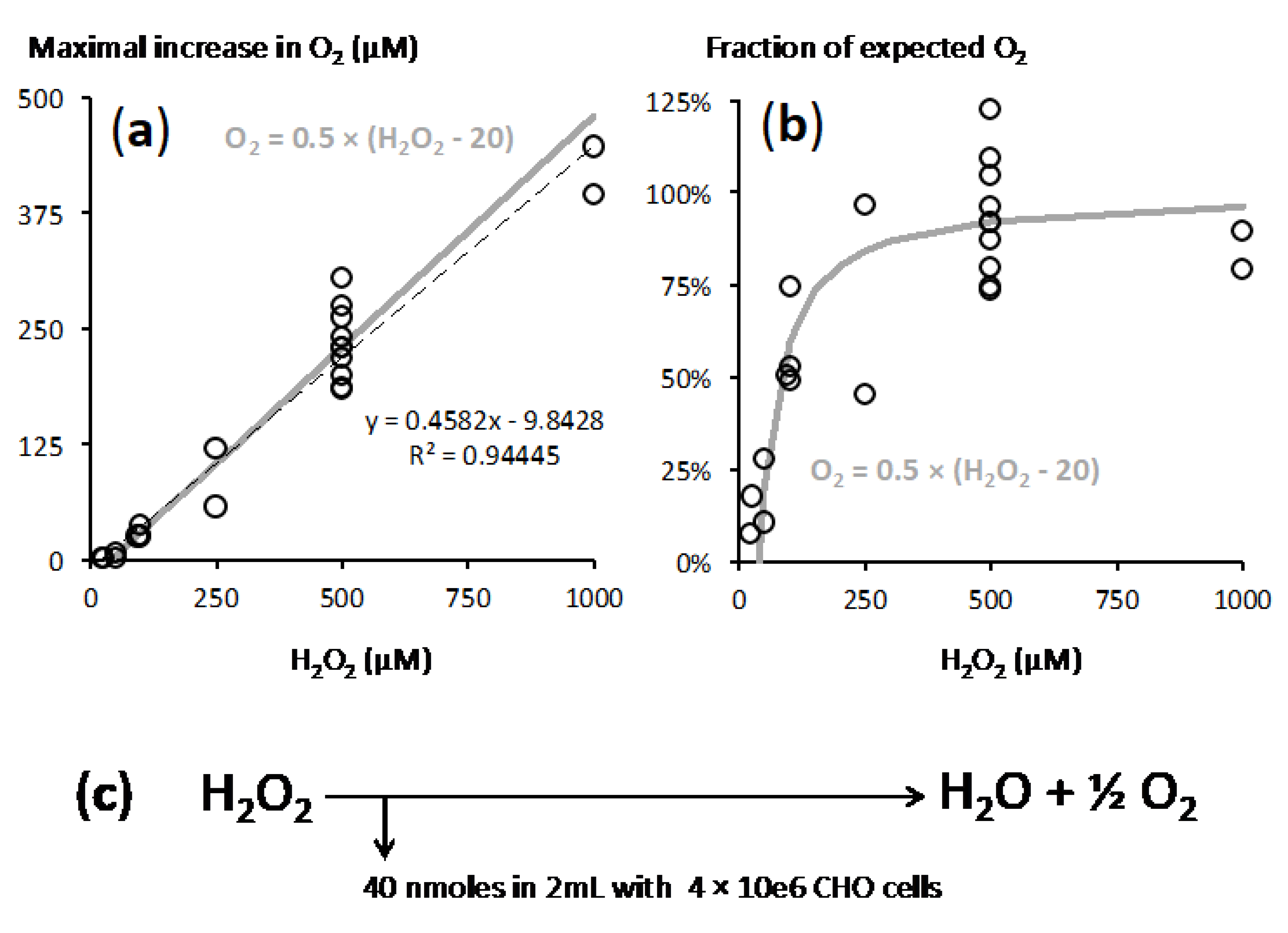
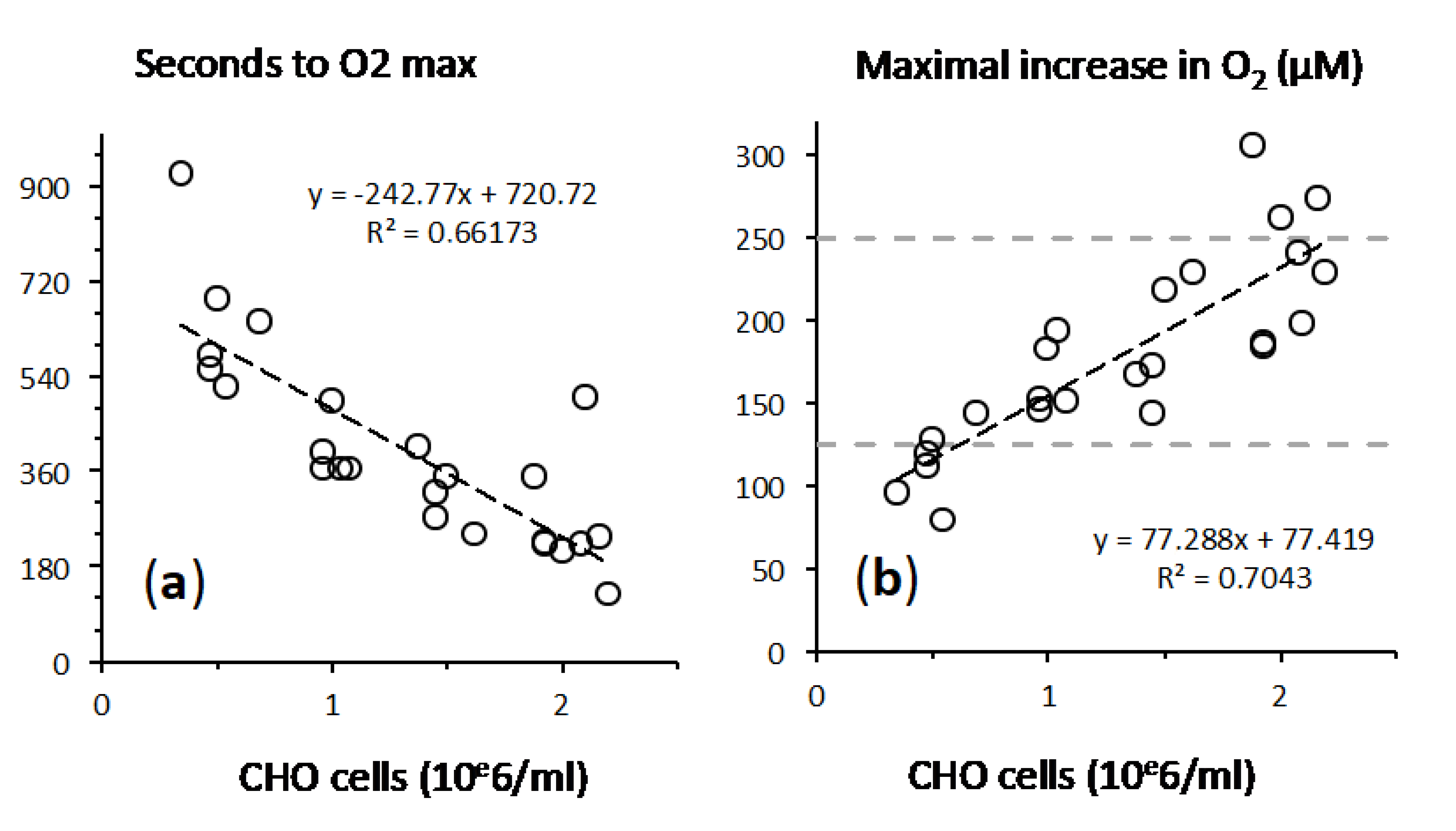
2.2. A Proportionate Increase in Dioxygen Follows High Micromolar H2O2 Addition
2.3. Fast Rates of Catalase Reaction
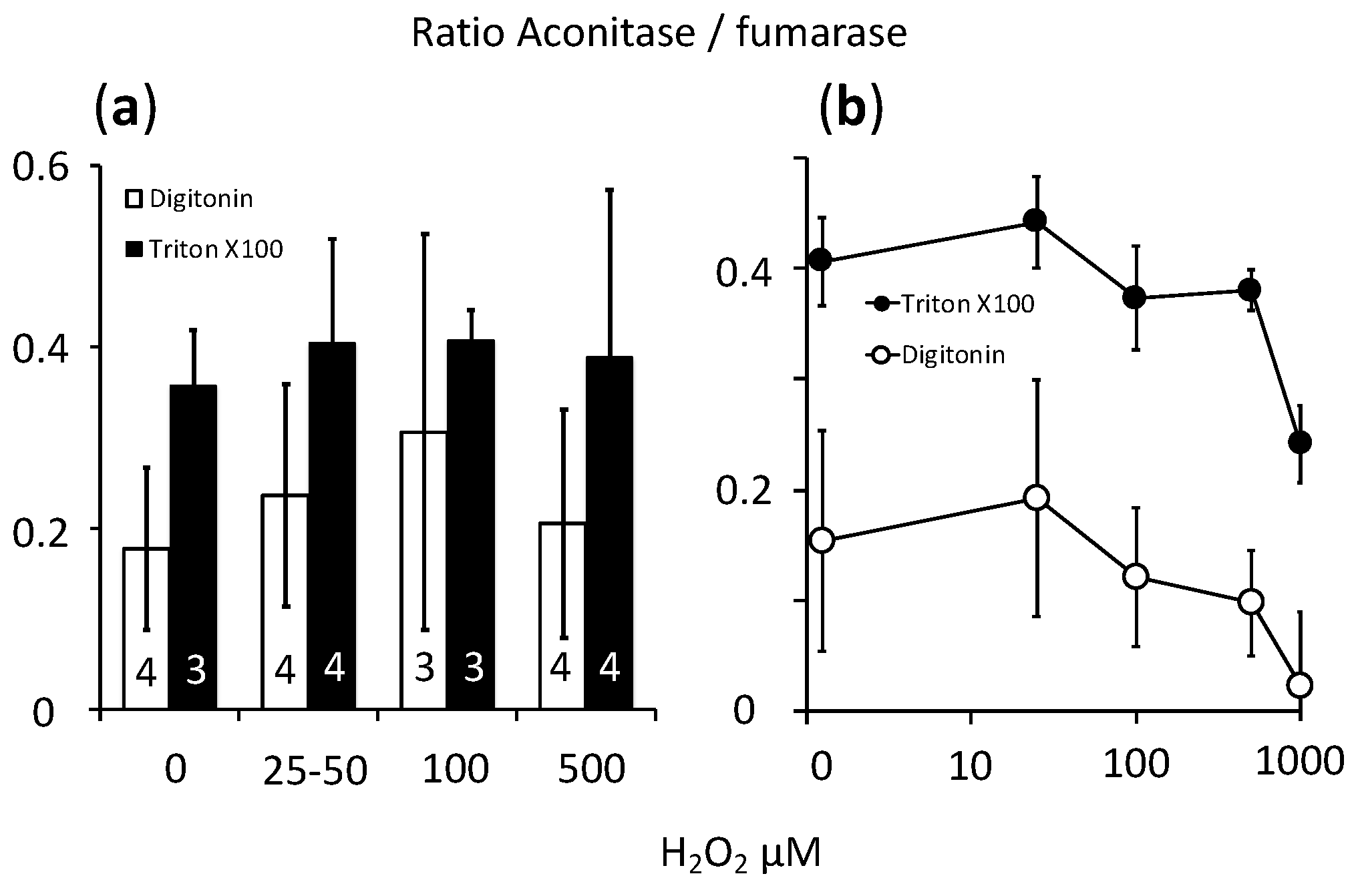
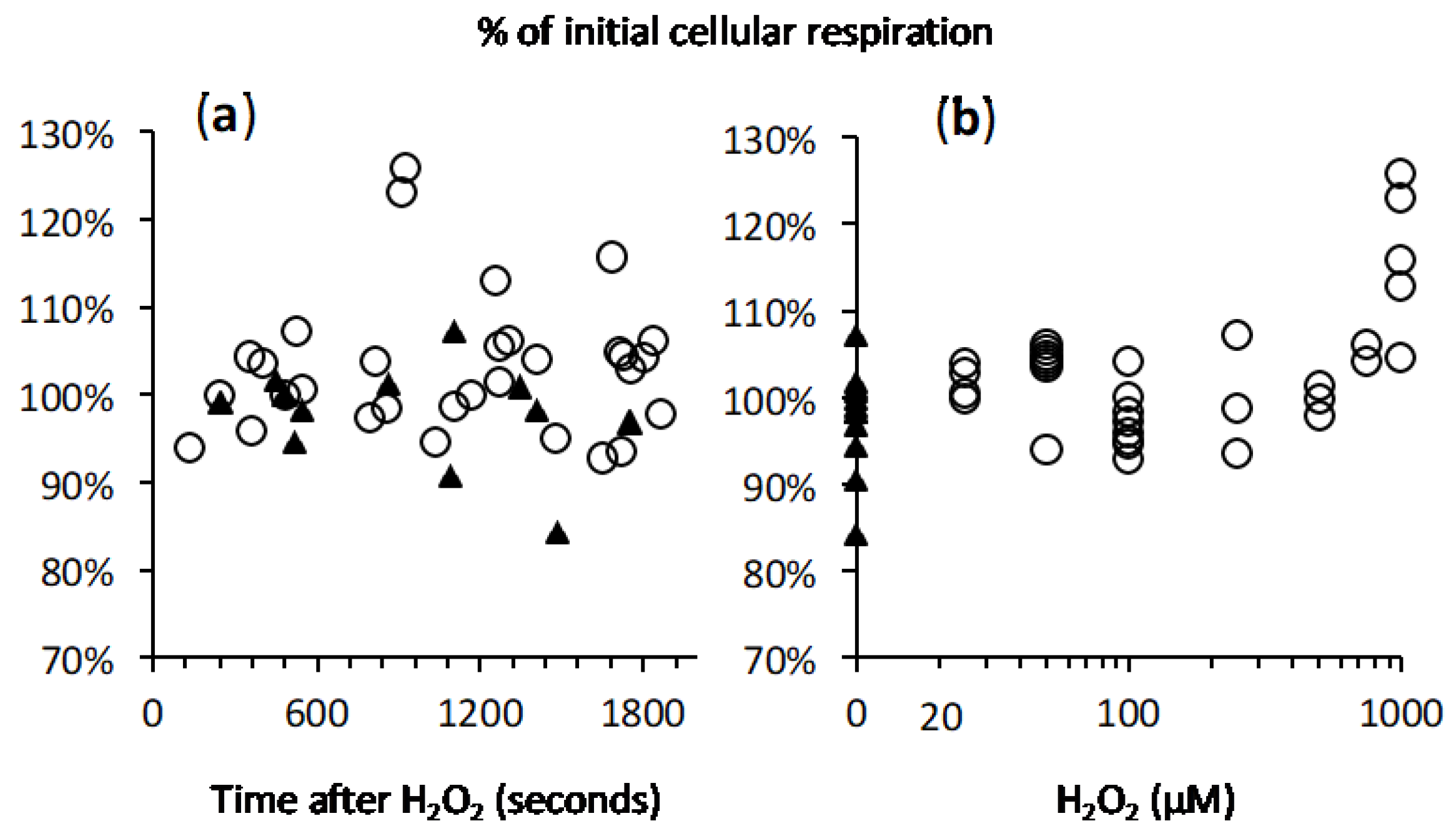
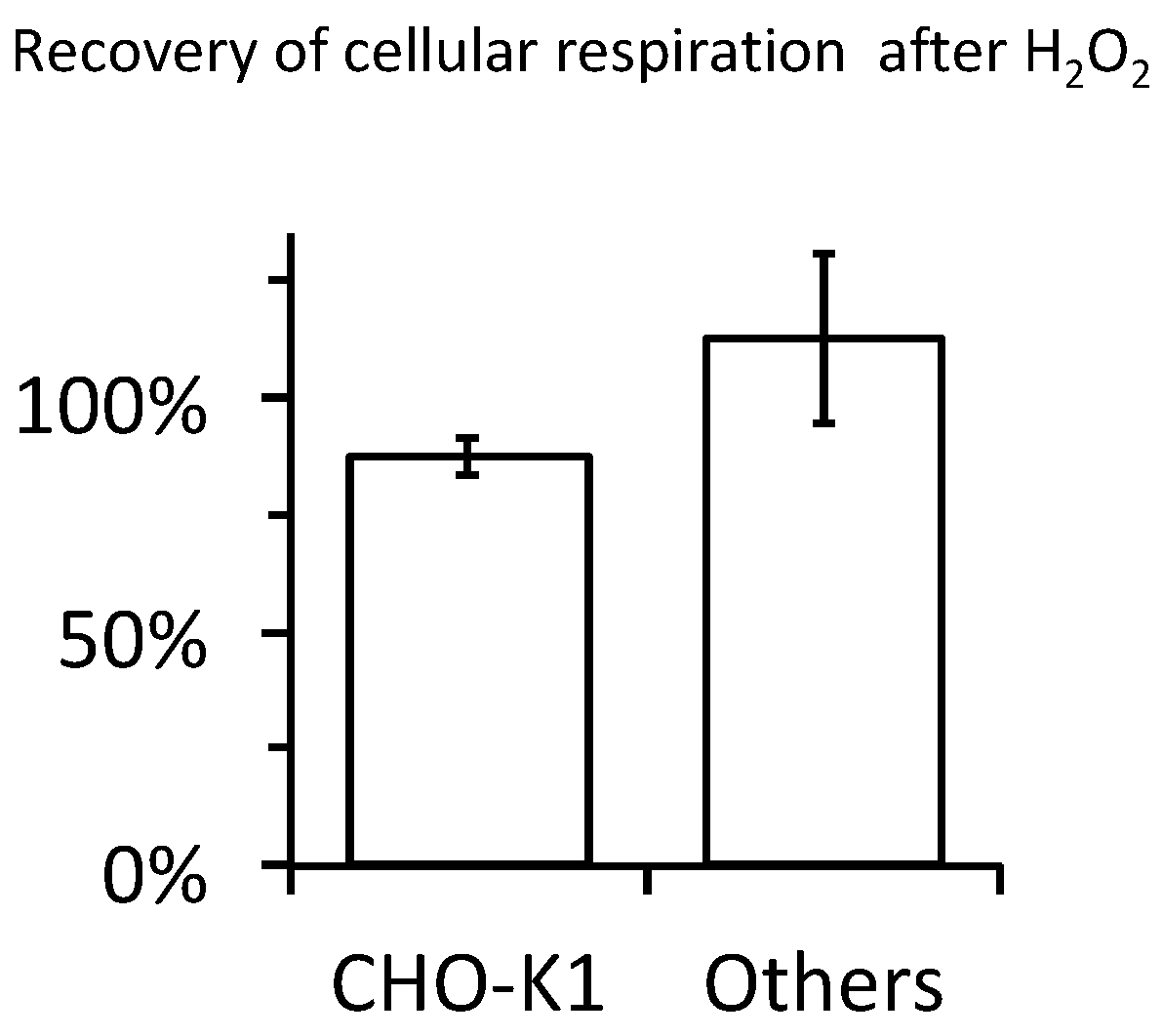
2.4. Cellular Respiration and Aconitase Activity Resistant to H2O2 in the High Micromolar Range
3. Discussion
4. Materials and Methods
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CHO | Chinese Hamster Ovary |
| ROS | Reactive Oxygen Species |
| MnSOD | Manganese SuperOxide Dismutase (mitochondrial for of SOD) |
| EDTA | Ethylene Diamine Tetra-acetic Acid |
| OD | Optical Density |
Appendix A
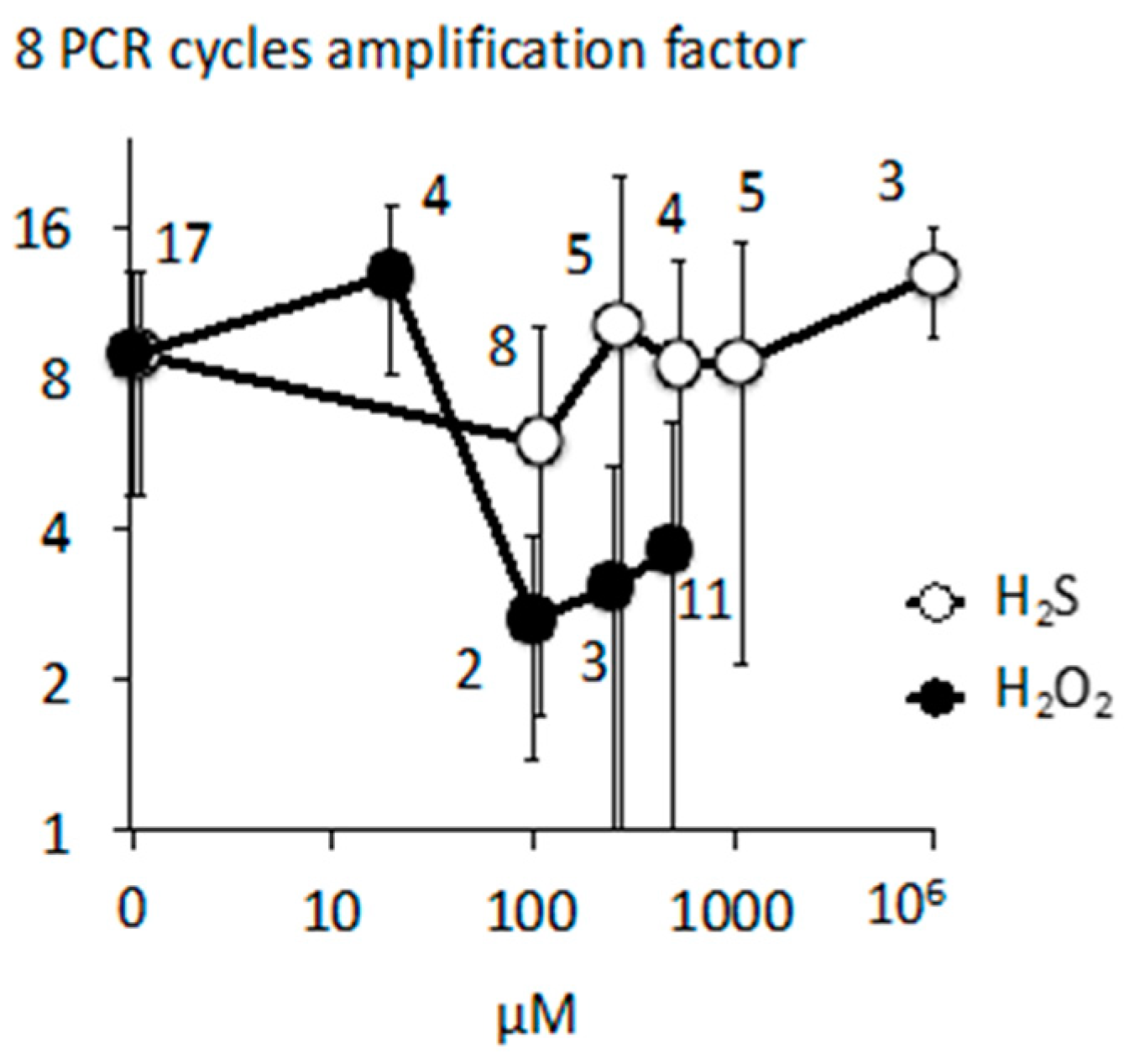
Appendix B
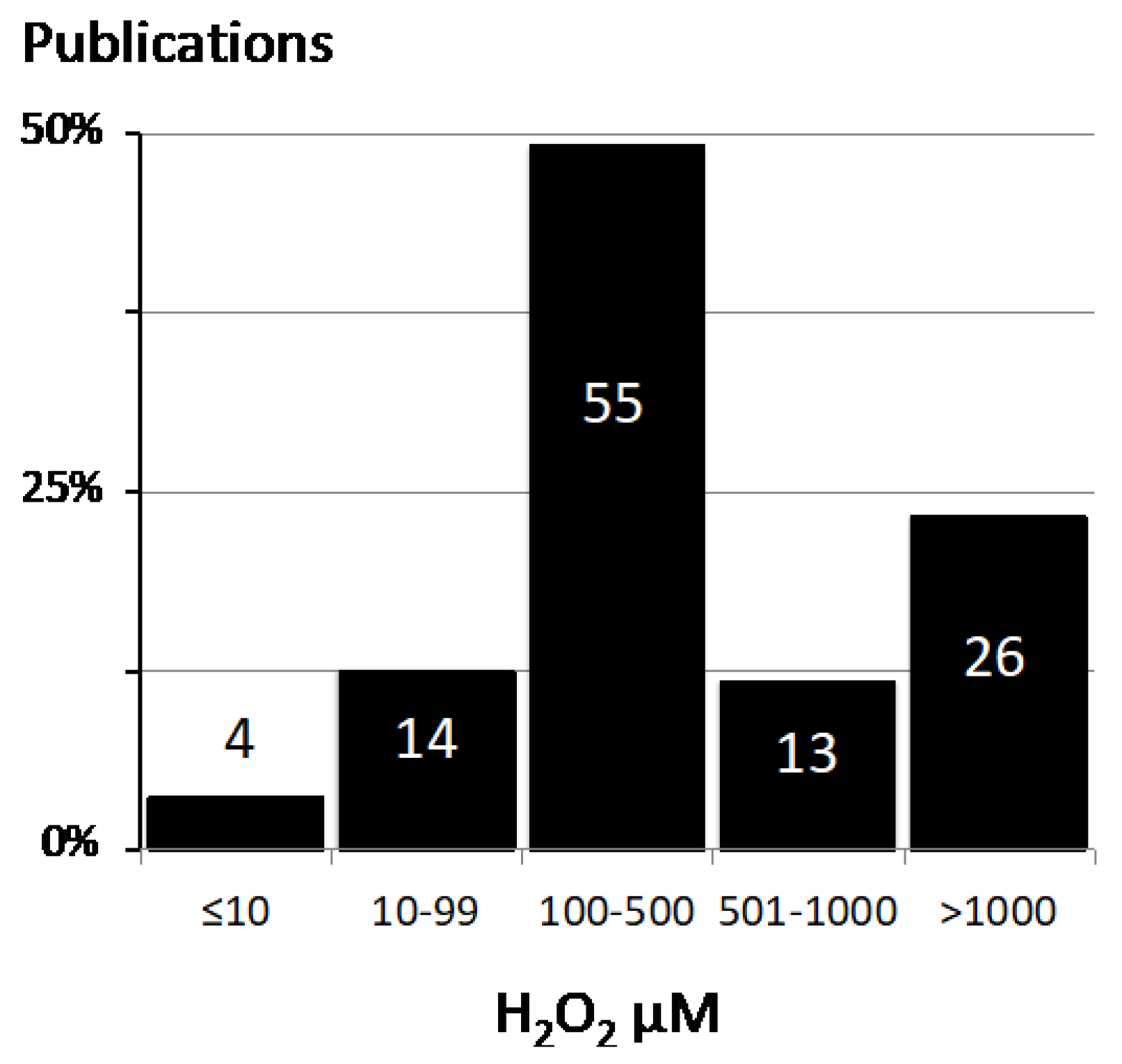
| H2O2 ≤ 10 µM |
| 20572791 |
| 29550333 |
| 23044977 |
| 9188618 |
| H2O2 range 10–99 µM |
| 17925005 |
| 19840190 |
| 14981287 |
| 12673362 |
| 12832847 |
| 15830107 |
| 17451824 |
| 29470430 |
| 29208995 |
| 10082315 |
| 16331539 |
| 18463796 |
| 21293477 |
| 26310930 |
| H2O2 range 100–500 µM |
| 21284033 |
| 12195838 |
| 25383141 |
| 31465957 |
| 20072135 |
| 28188272 |
| 22885031 |
| 25279550 |
| 19151733 |
| 12067712 |
| 23484085 |
| 14578137 |
| 26861269 |
| 27105496 |
| 31562333 |
| 26764141 |
| 9012815 |
| 22766155 |
| 28204825 |
| 29983082 |
| 18501193 |
| 23326372 |
| 27582624 |
| 31353362 |
| 31683554 |
| 17879239 |
| 26225431 |
| 29121706 |
| 29386875 |
| 18497977 |
| 20847286 |
| 23519197 |
| 9551742 |
| 22914987 |
| 26991551 |
| 29165341 |
| 28977633 |
| 20559505 |
| 11905987 |
| 18520066 |
| 10409635 |
| 14627202 |
| 28122344 |
| 26898812 |
| 29431851 |
| 27479053 |
| 18003976 |
| 31112588 |
| 24695490 |
| 11558568 |
| 25381633 |
| 28067784 |
| 22796327 |
| 15060142 |
| 29386875 |
| H2O2 range 501–1000 µM |
| 23349856 |
| 25236744 |
| 24642881 |
| 9453543 |
| 24634295 |
| 20224152 |
| 6292103 |
| 30662338 |
| 30662323 |
| 15138265 |
| 22580126 |
| 16150148 |
| 18954526 |
| H2O2 range > 1000 µM |
| 10846797 |
| 22302021 |
| 8176261 |
| 31783479 |
| 30513827 |
| 30366994 |
| 16101127 |
| 22542292 |
| 23357941 |
| 9422344 |
| 8628669 |
| 9735602 |
| 26555819 |
| 25528699 |
| 21429293 |
| 17085975 |
| 20824890 |
| 29351805 |
| 22208537 |
| 29661935 |
| 14688260 |
| 20566850 |
| 16194237 |
| 16936020 |
| 21176282 |
| 30634966 |
References
- Ward, J.F.; Evans, J.W.; Limoli, C.L.; Calabro-Jones, P.M. Radiation and hydrogen peroxide induced free radical damage to DNA. Br. J. Cancer Suppl. 1987, 8, 105–112. [Google Scholar] [PubMed]
- Hirai, D.M.; Colburn, T.D.; Craig, J.C.; Hotta, K.; Kano, Y.; Musch, T.I.; Poole, D.C. Skeletal muscle interstitial O2 pressures: Bridging the gap between the capillary and myocyte. Microcirculation 2019, 26, e12497. [Google Scholar] [CrossRef] [PubMed]
- Gnaiger, E. Oxygen Conformance of Cellular Respiration: A perspective of mitochondrial physiology. In Hypoxia; Roach, R.C., Wagner, P.D., Hackett, P.H., Eds.; Advances in Experimental Medicine and Biology; Springer: Boston, MA, USA, 2003; Volume 543, pp. 39–55. ISBN 978-1-4613-4753-8. [Google Scholar]
- Gnaiger, E.; Lassnig, B.; Kuznetsov, A.V.; Margreiter, R. Mitochondrial respiration in the low oxygen environment of the cell effect of ADP on oxygen kinetics. Biochim. Biophys. Acta BBA Bioenergy 1998, 1365, 249–254. [Google Scholar] [CrossRef]
- Benamar, A.; Rolletschek, H.; Borisjuk, L.; Avelange-Macherel, M.-H.; Curien, G.; Mostefai, H.A.; Andriantsitohaina, R.; Macherel, D. Nitrite-nitric oxide control of mitochondrial respiration at the frontier of anoxia. Biochim. Biophys. Acta 2008, 1777, 1268–1275. [Google Scholar] [CrossRef] [PubMed]
- Sepasi Tehrani, H.; Moosavi-Movahedi, A.A. Catalase and its mysteries. Prog. Biophys. Mol. Biol. 2018, 140, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Nandi, A.; Yan, L.-J.; Jana, C.K.; Das, N. Role of Catalase in Oxidative Stress- and Age-Associated Degenerative Diseases. Oxidative Med. Cell. Longev. 2019, 2019, 9613090. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.-K.; Cho, H.-W.; Song, S.-E.; Im, S.-S.; Bae, J.-H.; Song, D.-K. Oxidative stress resulting from the removal of endogenous catalase induces obesity by promoting hyperplasia and hypertrophy of white adipocytes. Redox Biol. 2020, 37, 101749. [Google Scholar] [CrossRef] [PubMed]
- Attene-Ramos, M.S.; Wagner, E.D.; Gaskins, H.R.; Plewa, M.J. Hydrogen Sulfide Induces Direct Radical-Associated DNA Damage. Mol. Cancer Res. 2007, 5, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Yakes, F.M.; Van Houten, B. Mitochondrial DNA damage is more extensive and persists longer than nuclear DNA damage in human cells following oxidative stress. Proc. Natl. Acad. Sci. USA 1997, 94, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Beaumont, M.; Andriamihaja, M.; Lan, A.; Khodorova, N.; Audebert, M.; Blouin, J.-M.; Grauso, M.; Lancha, L.; Benetti, P.-H.; Benamouzig, R.; et al. Detrimental effects for colonocytes of an increased exposure to luminal hydrogen sulfide: The adaptive response. Free Radic. Biol. Med. 2016, 93, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.-Y.; Pedersen, C.; Day, B.J.; Patel, M. Dependence of excitotoxic neurodegeneration on mitochondrial aconitase inactivation: Mitochondrial superoxide and excitotoxicity. J. Neurochem. 2001, 78, 746–755. [Google Scholar] [CrossRef]
- Lagoutte, E.; Mimoun, S.; Andriamihaja, M.; Chaumontet, C.; Blachier, F.; Bouillaud, F. Oxidation of hydrogen sulfide remains a priority in mammalian cells and causes reverse electron transfer in colonocytes. Biochim. Biophys. Acta 2010, 1797, 1500–1511. [Google Scholar] [CrossRef]
- Tochigi, M.; Inoue, T.; Suzuki-Karasaki, M.; Ochiai, T.; Ra, C.; Suzuki-Karasaki, Y. Hydrogen peroxide induces cell death in human TRAIL-resistant melanoma through intracellular superoxide generation. Int. J. Oncol. 2013, 42, 863–872. [Google Scholar] [CrossRef] [PubMed]
- Biary, N.; Xie, C.; Kauffman, J.; Akar, F.G. Biophysical properties and functional consequences of reactive oxygen species (ROS)-induced ROS release in intact myocardium. J. Physiol. 2011, 589, 5167–5179. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.-H.; Ding, J.; Xie, X.-X.; Yang, X.-H.; Wu, X.-F.; Chen, Z.-X.; Guo, Q.-L.; Gao, W.-Y.; Wang, X.-Z.; Li, D. Hepatitis B virus X protein promotes liver cell pyroptosis under oxidative stress through NLRP3 inflammasome activation. Inflamm. Res. Off. J. Eur. Histamine Res. Soc. Al 2020, 69, 683–696. [Google Scholar] [CrossRef] [PubMed]
- Lismont, C.; Revenco, I.; Fransen, M. Peroxisomal Hydrogen Peroxide Metabolism and Signaling in Health and Disease. Int. J. Mol. Sci. 2019, 20, 3673. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ransy, C.; Vaz, C.; Lombès, A.; Bouillaud, F. Use of H2O2 to Cause Oxidative Stress, the Catalase Issue. Int. J. Mol. Sci. 2020, 21, 9149. https://doi.org/10.3390/ijms21239149
Ransy C, Vaz C, Lombès A, Bouillaud F. Use of H2O2 to Cause Oxidative Stress, the Catalase Issue. International Journal of Molecular Sciences. 2020; 21(23):9149. https://doi.org/10.3390/ijms21239149
Chicago/Turabian StyleRansy, Céline, Clément Vaz, Anne Lombès, and Frédéric Bouillaud. 2020. "Use of H2O2 to Cause Oxidative Stress, the Catalase Issue" International Journal of Molecular Sciences 21, no. 23: 9149. https://doi.org/10.3390/ijms21239149
APA StyleRansy, C., Vaz, C., Lombès, A., & Bouillaud, F. (2020). Use of H2O2 to Cause Oxidative Stress, the Catalase Issue. International Journal of Molecular Sciences, 21(23), 9149. https://doi.org/10.3390/ijms21239149





