Does Cannabidiol (CBD) in Food Supplements Pose a Serious Health Risk? Consequences of the European Food Safety Authority (EFSA) Clock Stop Regarding Novel Food Authorisation
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
- Continuing with the current form of regulatory prohibition (i.e., proceeding with the current form of an unregulated market). This is probably the worst option as the prohibition approach has not worked in the past in similar areas of drug policy [31]. It is not expected that enforcement could be strengthened to the extent that CBD products could be completely removed from the market. Prohibition will only reduce consumer protection as the products will mostly drift into areas of completely unregulated and uncontrolled non-food products (such as CBD air fresheners), and effective enforcement of the novel food regulation also seems generally impossible in other areas [32].
- Approving low-dose CBD food supplements (up to 10 mg/day and less than 300 mg/package) on an intermediary basis (see also the post-Brexit UK approach), including warning labels about potential toxic effects. This option may give consumers easy access to low-dose CBD supplements, despite the uncertain risk. However, warning labels about potential toxic effects and maximum daily doses may deter some consumers from using the products and increase the safety margin. Additionally, the lack of strict regulation may lead to variations in the quality and safety of CBD products between manufacturers.
- Regulating low-dose CBD products as over-the-counter medicinal products available only in pharmacies, as an additional category to the already available prescription-based high-dose CBD medicinal products (see the suggestion by Health Canada [33]). This option may ensure that consumers have access to safe and high-quality CBD products. However, limiting the availability of low-dose CBD products to pharmacies may reduce their accessibility to consumers, and hence still allow for unregulated markets specifically on the internet.
- Regulating CBD products outside the scope of foods or medicines in a separate framework, e.g., within the currently planned controlled distribution of cannabis to adults for recreational use in licensed stores in Germany. This option may ensure that CBD products are subject to strict regulation, labelling, and quality control standards. Additionally, it may provide consumers with accessibility to CBD products only through licensed stores. However, the potential for more or less unrestricted use of CBD products for recreational purposes may arise, leading to public health concerns.
5. Conclusions
- The currently available data for CBD do not allow a conclusive assessment of its safety as a novel food.
- Benchmark dose (BMD) response modelling of several bioassays provides an alternative POD for toxicological risk assessment.
- The authors suggest that certain CBD products still available on the EU market as food supplements despite the lack of authorisation must be considered “unsafe”: Products exceeding a health-based guidance value of 10 mg/day must be considered “unfit for consumption”, while those exceeding the human LOAEL must be considered “injurious to health”.
- This risk assessment could only be superseded if further dose–response data become available, such as those expected from the novel food applicants.
- By encouraging risk managers to take a more active role in communicating about CBD-containing products, public health outcomes could be improved and the potential for harm reduced.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lachenmeier, D.W.; Rajcic de Rezende, T.; Schwarz, S. An update on sustainable valorization of coffee by-products as novel foods within the European Union. Biol. Life Sci. Forum 2021, 6, 37. [Google Scholar] [CrossRef]
- European Parliament and the Council of the European Union. Regulation (EU) 2015/2283 of the European Parliament and of the Council of 25 November 2015 on novel foods, amending Regulation (EU) No 1169/2011 of the European Parliament and of the Council and repealing Regulation (EC) No 258/97 of the European Parliament and of the Council and Commission Regulation (EC) No 1852/2001. Off. J. Eur. Union 2015, L327, 1–22. [Google Scholar]
- Lachenmeier, D.W.; Rajcic de Rezende, T.; Habel, S.; Bock, V.; Sproll, C.; Walch, S.G. Current case law confirms novel food classification of hemp extracts and cannabidiol (CBD) in foods—Narcotic classification of cannabis foods remains unclear. Dtsch. Lebensm. Rundsch. 2020, 116, 111–119. [Google Scholar] [CrossRef]
- Dräger, H.; Barthlott, I.; Golombek, P.; Walch, S.G.; Lachenmeier, D.W. Time trends of tetrahydrocannabinol (THC) in a 2008–2021 German national survey of hemp food products. Foods 2022, 11, 486. [Google Scholar] [CrossRef]
- PontEurope. The Budding CBD Industry in Europe. 2022. Available online: https://www.ponteurope.com/gb/news-gb/the-budding-cbd-industry-in-europe/ (accessed on 29 July 2022).
- Nyland, C.; Moyer, D.C. Regulating for safety: Cannabidiol dose in food. J. Food Prot. 2022, 85, 1355–1369. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA); Turck, D.; Bohn, T.; Castenmiller, J.; De Henauw, S.; Hirsch-Ernst, K.I.; Maciuk, A.; Mangelsdorf, I.; McArdle, H.J.; Naska, A.; et al. Statement on safety of cannabidiol as a novel food: Data gaps and uncertainties. EFSA J. 2022, 20, 7322. [Google Scholar] [CrossRef]
- Watkins, P.B.; Church, R.J.; Li, J.; Knappertz, V. Cannabidiol and abnormal liver chemistries in healthy adults: Results of a phase I clinical trial. Clin. Pharmacol. Ther. 2021, 109, 1224–1231. [Google Scholar] [CrossRef] [PubMed]
- Dziwenka, M.; Dolan, L.; Mitchell, J. Toxicological safety of VOHO hemp oil; a supercritical fluid extract from the aerial parts of hemp. PLoS ONE 2021, 16, e0261900. [Google Scholar] [CrossRef] [PubMed]
- Hindelang, P.; Scharinger, A.; Richling, E.; Walch, S.G.; Lachenmeier, D.W. Using the BMD approach to derive acceptable daily intakes of cannabidiol (CBD) and tetrahydrocannabinol (THC) relevant to electronic cigarette liquids. Front. Biosci. 2022, 27, 228. [Google Scholar] [CrossRef]
- Davis, J.A.; Gift, J.S.; Zhao, Q.J. Introduction to benchmark dose methods and U.S. EPA’s benchmark dose software (BMDS) version 2.1.1. Toxicol. Appl. Pharmacol. 2011, 254, 181–191. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. About Benchmark Dose Software (BMDS)—Version 3.2. 2022. Available online: https://www.epa.gov/bmds/about-benchmark-dose-software-bmds-version-3201 (accessed on 24 June 2022).
- FDA. Drug Approval Package: Epidiolex (Cannabidiol). Company: GW Research Ltd. Application Number: 210365 Orig 1. FDA Application Review Files. Pharmacology Review(s). 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2018/210365Orig1s000OtherR.pdf (accessed on 29 July 2022).
- Marx, T.K.; Reddeman, R.; Clewell, A.E.; Endres, J.R.; Beres, E.; Vertesi, A.; Glavits, R.; Hirka, G.; Szakonyine, I.P. An assessment of the genotoxicity and subchronic toxicity of a supercritical fluid extract of the aerial parts of hemp. J. Toxicol. 2018, 2018, 8143582. [Google Scholar] [CrossRef] [PubMed]
- Dziwenka, M.; Coppock, R.; Alexander, M.; Palumbo, E.; Ramirez, C.; Lermer, S. Safety assessment of a hemp extract using genotoxicity and oral repeat-dose toxicity studies in Sprague-Dawley rats. Toxicol. Rep. 2020, 7, 376–385. [Google Scholar] [CrossRef]
- EFSA. Information Session for Applicants on “The Safety of Cannabidiol as a Novel Food: Data Gaps and Uncertainties”. 2022. Available online: https://www.efsa.europa.eu/sites/default/files/2022-07/Infosessio-CBD-slides.pdf (accessed on 29 July 2022).
- Crippa, J.A.S.; Zuardi, A.W.; Guimaraes, F.S.; Campos, A.C.; de Lima, O.F.; Loureiro, S.R.; Dos Santos, R.G.; Souza, J.D.S.; Ushirohira, J.M.; Pacheco, J.C.; et al. Efficacy and safety of cannabidiol plus standard care vs standard care alone for the treatment of emotional exhaustion and burnout among frontline health care workers during the COVID-19 pandemic: A randomized clinical trial. JAMA Netw. Open 2021, 4, e2120603. [Google Scholar] [CrossRef] [PubMed]
- EFSA Scientific Committee. Guidance on selected default values to be used by the EFSA Scientific Committee, Scientific Panels and Units in the absence of actual measured data. EFSA J. 2012, 10, 2579. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Scientific opinion on the risks for human health related to the presence of tetrahydrocannabinol (THC) in milk and other food of animal origin. EFSA J. 2015, 13, 4141. [Google Scholar] [CrossRef]
- Lachenmeier, D.W.; Habel, S.; Fischer, B.; Herbi, F.; Zerbe, Y.; Bock, V.; Rajcic de Rezende, T.; Walch, S.G.; Sproll, C. Are adverse effects of cannabidiol (CBD) products caused by tetrahydrocannabinol (THC) contamination? F1000Research 2021, 8, 1394. [Google Scholar] [CrossRef]
- Kruse, D.; Beitzke, B. Comment on Lachenmeier et al. (2020) “Are side effects of cannabidiol (CBD) products caused by tetrahydrocannabinol (THC) contamination?”: Disputation on various points in the publication. F1000Research 2020, 9, 900. [Google Scholar] [CrossRef]
- Federal Food Safety and Veterinary Office (FSVO). Briefing Letter Cannabidiol (CBD) in Foods and the Effects on the Liver. 2021. Available online: https://www.blv.admin.ch/dam/blv/en/dokumente/lebensmittel-und-ernaehrung/publikationen-forschung/briefing-letter-lebensmittel-lebereffekte.pdf.download.pdf/Briefing%20Letter%20Cannabidiol%20in%20Lebensmitteln%20und%20Lebereffekte%20DE.pdf (accessed on 29 July 2022). (In German).
- FDA. Comment from GW Pharmaceuticals. Posted by the Food and Drug Administration on 19 July 2019. GW Pharmaceuticals’ Submission on Scientific Data and Information about Products Containing Cannabis or Cannabis-Derived Compounds. Document ID FDA-2019-N-1482-4257. 2019. Available online: https://downloads.regulations.gov/FDA-2019-N-1482-4257/attachment_1.pdf (accessed on 29 July 2022).
- Maronpot, R.R. NTP Nonneoplastic Lesion Atlas. Liver, Hepatocyte—Hypertrophy. National Toxicology Program, U.S. Department of Health and Human Services, Research Triangle Park, NC, USA. Available online: https://ntp.niehs.nih.gov/nnl/hepatobiliary/liver/hhypertr/liver,_hepatocyte-hypertrophy_508.pdf (accessed on 8 December 2022).
- Giannini, E.G.; Testa, R.; Savarino, V. Liver enzyme alteration: A guide for clinicians. CMAJ 2005, 172, 367–379. [Google Scholar] [CrossRef]
- European Parliament and Council. Regulation (EC) No. 178/2002 of the European Parliament and of the council of 28 January 2002 laying down the general principles and requirements of food law, establishing the European Food Safety Authority and laying down procedures in matters of food safety. Off. J. Eur. Communities 2002, L31, 1–24. [Google Scholar]
- Dietz, T.; Gerstenlauer, J.; Sproll, C.; Walch, S.G.; Lachenmeier, D.W. Cannabidiol (CBD) in Lebensmitteln—Beurteilung der Sicherheit: Gesundheitsschädlich bei über 300 mg/Tag. Dtsch. Lebensm. Rundsch. 2022, 118, 446–453. [Google Scholar] [CrossRef]
- Schweikle, S.; Golombek, P.; Sproll, C.; Walch, S.G.; Lachenmeier, D.W. The challenge of risk assessment of tetrahydrocannabinol (THC) in cannabidiol (CBD) oils and food supplements: An approach for deriving maximum limits. Challenges 2022, 13, 32. [Google Scholar] [CrossRef]
- Lachenmeier, D.W.; Golombek, P.; Walch, S.G. Hanfhaltige Lebensmittel. Weiteres Update zur Verkehrsfähigkeit nach Entscheidungen des EuGH und BGH. Dtsch. Lebensm. Rundsch. 2021, 117, 481–487. [Google Scholar] [CrossRef]
- Miller, O.S.; Elder, E.J., Jr.; Jones, K.J.; Gidal, B.E. Analysis of cannabidiol (CBD) and THC in nonprescription consumer products: Implications for patients and practitioners. Epilepsy Behav. 2022, 127, 108514. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P.; Berridge, V.; Conrod, P.; Dudley, R.; Hellman, M.; Lachenmeier, D.; Lingford-Hughes, A.; Miller, D.; Rehm, J.; Room, R.; et al. Reframing the science and policy of nicotine, illegal drugs and alcohol—Conclusions of the ALICE RAP Project. F1000Research 2017, 6, 289. [Google Scholar] [CrossRef]
- Lachenmeier, D.W.; Walch, S.G. Essay for Innovative Food Law Writing Competition (2nd edition): Do we need urgent revision of the novel food law in the European Union to ensure food security in times of global supply shortage? Lebensmittelchemie 2023, 77, 22–26. [Google Scholar] [CrossRef]
- Health Canada. Review of Cannabidiol: Report of the Science Advisory Committee on Health Products Containing Cannabis; Health Canada: Ottawa, ON, Canada, 2022. [Google Scholar]
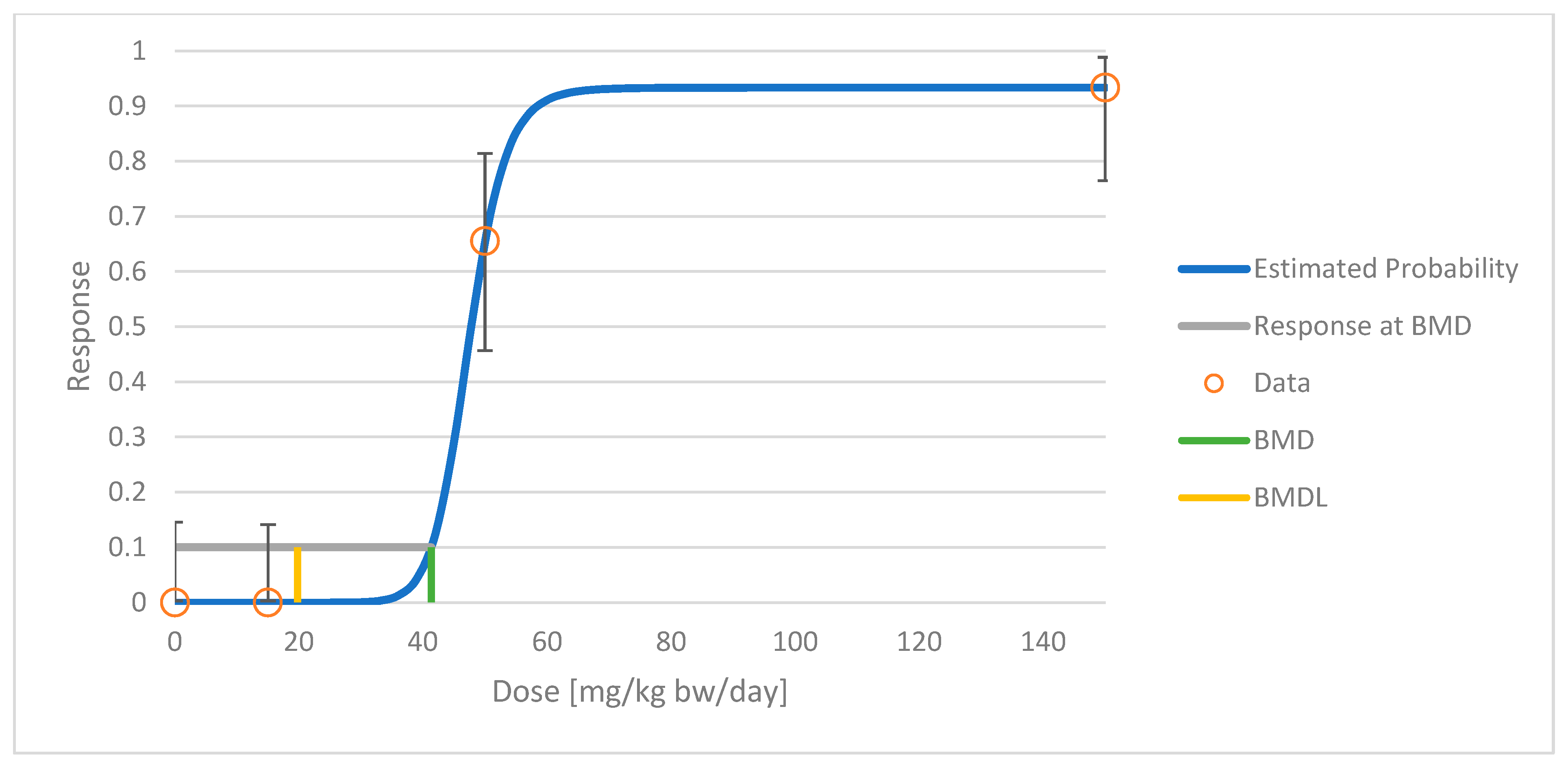
| Study, Animal Model | Study Design, CBD Doses | Endpoint | Sex | Model a | p-Value b | BMD c (mg/kg bw/day) | BMDL d (mg/kg bw/day) |
|---|---|---|---|---|---|---|---|
| GWTX1412 [13], rats | 26-week oral at doses of 0, 15, 50, and 150 mg/kg bw/day (n = 15/sex/group) | Liver, centrilobular hypertrophy e | Males + females combined f | Dichotomous Hill | 0.9989 | 41 | 20 |
| GWTX1413 [13], dogs | 39-week oral at doses of 0, 10, 50, and 100 mg/kg bw/day (n = 4/sex/group) | Liver, hepatocyte hypertrophy e | Males + females combined f | Log-Probit | 0.5771 | (3) g | (2) g |
| Marx et al. [14], rats | 90-day oral at doses of 0, 25, 90, and 180 mg/kg bw/day (n = 10/sex/group) h | Liver weight | Males i | Exponential 2 | 0.5235 | (52) j | (43) j |
| Females i | Polynomial 3 | 0.9771 | (52) j | (34) j | |||
| Dziwenka et al. [9], rats | 90-day oral at doses of 0, 6.3, 22.7 and 81.6 mg/kg bw/day (n = 10/sex/group) k | Relative liver weight | Females | Exponential 2 | 0.1941 | (39) j | (26) j |
| CBD | Animal Data | Human Data |
|---|---|---|
| Type of point of departure (POD) | BMDL, see Table 1 | LOAEL [7,17] |
| Value of point of departure (POD) | 20 mg/kg bw/day (1400 mg/day a) | 4.3 mg/kg bw/day (300 mg/day a) |
| Uncertainty factor (UF) | 100 b | 30 c |
| Health-based guidance value (HBGV) | 0.20 mg/kg bw/day (14 mg/day a) | 0.14 mg/kg bw/day (10 mg/day a) |
| % CBD | Number of Samples | Intake Amount [g] to Exceed LOAEL | Number of Drops to Exceed LOAEL a |
|---|---|---|---|
| 70 | 1 | 0.4 | 15 |
| 48 | 1 | 0.6 | 22 |
| 30 | 3 | 1.0 | 36 |
| 27 | 3 | 1.1 | 40 |
| 25 | 5 | 1.2 | 43 |
| 24 | 2 | 1.3 | 45 |
| 20 | 12 | 1.5 | 54 |
| 18 | 3 | 1.7 | 60 |
| 15 | 15 | 2.0 | 72 |
| 12 | 1 | 2.5 | 90 |
| 10 | 49 | 3.0 | 108 |
| 9 | 2 | 3.3 | 120 |
| 8 | 1 | 3.8 | 135 |
| 6 | 1 | 5.0 | 180 |
| 5 | 33 | 6.0 | 216 |
| 4 | 2 | 7.5 | 270 |
| 3 | 3 | 10.0 | 360 |
| 2.75 | 3 | 10.9 | 392 |
| 2.5 | 2 | 12.0 | 431 |
| 2 | 2 | 15.0 | 539 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lachenmeier, D.W.; Sproll, C.; Walch, S.G. Does Cannabidiol (CBD) in Food Supplements Pose a Serious Health Risk? Consequences of the European Food Safety Authority (EFSA) Clock Stop Regarding Novel Food Authorisation. Psychoactives 2023, 2, 66-75. https://doi.org/10.3390/psychoactives2010005
Lachenmeier DW, Sproll C, Walch SG. Does Cannabidiol (CBD) in Food Supplements Pose a Serious Health Risk? Consequences of the European Food Safety Authority (EFSA) Clock Stop Regarding Novel Food Authorisation. Psychoactives. 2023; 2(1):66-75. https://doi.org/10.3390/psychoactives2010005
Chicago/Turabian StyleLachenmeier, Dirk W., Constanze Sproll, and Stephan G. Walch. 2023. "Does Cannabidiol (CBD) in Food Supplements Pose a Serious Health Risk? Consequences of the European Food Safety Authority (EFSA) Clock Stop Regarding Novel Food Authorisation" Psychoactives 2, no. 1: 66-75. https://doi.org/10.3390/psychoactives2010005
APA StyleLachenmeier, D. W., Sproll, C., & Walch, S. G. (2023). Does Cannabidiol (CBD) in Food Supplements Pose a Serious Health Risk? Consequences of the European Food Safety Authority (EFSA) Clock Stop Regarding Novel Food Authorisation. Psychoactives, 2(1), 66-75. https://doi.org/10.3390/psychoactives2010005








