Abstract
Purpose: Survivors of pediatric brain tumor experience substantial post-treatment challenges, facing greater cognitive impairment and lower Health-Related Quality of Life (HRQoL) compared to survivors of other childhood cancers. Early studies report methylphenidate to benefit cognition and HRQoL in paediatric survivors of cancer via improved processing speed and attention. Parental opinion is an important element of treatment decision making, however parents’ views on methylphenidate treatment have not been reported. This study explored parental perceptions of their child’s treatment with methylphenidate. Materials and methods: An evaluation of 10 parents of children with previous brain tumour receiving methylphenidate was performed. Data were gathered by qualitative parental-report HRQoL questionnaire (PedsQL parent form) and by a questionnaire developed for use in this population. Results: Preliminary data suggest that parents are positive about the use of methylphenidate as an interventional measure, and believed this to have a beneficial impact on their child’s HRQoL. Conclusion: Early data from a small sample of parents suggest that methylphenidate may be effective in supporting HRQoL within this population. Parental information supports an increasing evidence base for the benefits of methylphenidate as an intervention for the management of long-term difficulties associated with paediatric cancer.
1. Introduction
The term ‘Health Related Quality of Life’ (HRQoL) describes the impact of physical or mental health upon one’s perceived quality of life. Low HRQoL is reported in survivors of paediatric brain tumour (PBT). This is related to the high treatment burden associated with ongoing cancer treatment and to the consequent long-term effects following tumour and treatment [1]. Clinical factors such as younger age at diagnosis, relapse, higher intensity treatment including cranial radiotherapy, and the presence of late effects such as cognitive difficulties are associated with low reported HRQoL [2]. Assessment and intervention of low HRQoL is therefore a clinical priority in the management of survivors of PBT [3].
1.1. Post Brain Tumour Late Effects
Over a quarter of paediatric cancer patients in the UK have a brain or central nervous system tumour diagnosis. Whilst advances in treatment have yielded improved survival rates across brain tumours as a group over the last 20-year period, treatment-related cognitive effects are reported in a significant proportion of children. A large body of evidence has shown that survivors of PBT are more susceptible to psychological, physical and neurocognitive sequelae as a result of permanent damage to the developing brain [4,5]. Many survivors show persistent deficits in working memory, attention, and processing speed [6] with associated decline in IQ and academic attainment [7]. Treatment-related effects are associated with academic difficulties, high rates of unemployment and a reduced quality of life [8]. In addition to cognitive effects, a high proportion of survivors report debilitating fatigue. Cancer-related fatigue is a significant problem that affects patients at all stages of treatment, and it is cited as the most debilitating symptom impeding on patient’s ability to carry out physical tasks and take part in social activities [9].
1.2. Pharmacological Intervention
Methylphenidate is a psycho-stimulant medication that has successfully been employed to manage attentional difficulties in children with ADHD or traumatic brain injuries [10,11,12,13]. An emerging evidence base for intervention with methylphenidate in survivors of PBT has offered promising results in improving cognitive impairment [14,15,16,17]. Improvement in attention and processing speed when using methylphenidate is reported in the paediatric cancer population, with increased reaction time and reduction in mental fatigue found in a meta-analysis of studies of adult cancer survivors [18].
1.3. Evaluation Rationale
The Paediatric Oncology Department in the Great North Children’s Hospital (GNCH) is the first service in the UK to use methylphenidate to manage the late-cognitive effects of survivors of PBT as standard treatment. Patients showing a clinically significant loss of processing speed and attention are eligible for a trial of methylphenidate if they have no medical contra-indications as per the British National Formulary (Child). Full details of eligibility criteria for our clinic are provided as Appendix A. Doses are based on NICE Guideline for Attention Deficit/Hyperactivity Disorder [19].
Patient opinion on the benefit of methylphenidate to HRQoL is very positive [20]. Parental views on methylphenidate treatment and treatment delivery in paediatric neuro-oncology, however, have not previously been presented. Parental opinion is a vital facet of paediatric treatment, affecting both direct outcome factors such as treatment adherence but also indirectly influencing upon children’s perceptions of treatment efficacy. Parents-as-experts are often, appropriately, the conduit between clinicians and their patient, advising clinicians of the difficulties experienced by their child and contributing to treatment decision-making. Depending upon the age and ability level of the patient, assessment of children’s HRQoL is often reliant on parental proxy reports [21]. Parental satisfaction with medical and neuropsychological intervention is therefore critical for the effective management of problems related to paediatric survivorship.
1.4. Evaluation Aims
The current study presents service evaluation data from parents of survivorship clinic patients in the initial months of the clinic’s running. We sought to:
- Gather preliminary data on parental perception of the impact of methylphenidate on HRQoL in this population.
- Gather preliminary data on the parental acceptability of methylphenidate as an interventional medium.
2. Materials and Methods
2.1. Design
A pilot project exploring the feasibility of methylphenidate intervention in survivors of PBT was established to gather data in support of a future grant application. The current study uses data from this pilot. A mixed-method approach of data collection was employed including use of a standardised psychometric parent-report questionnaire and a semi-structured questionnaire used within clinical interview.
2.2. Participants
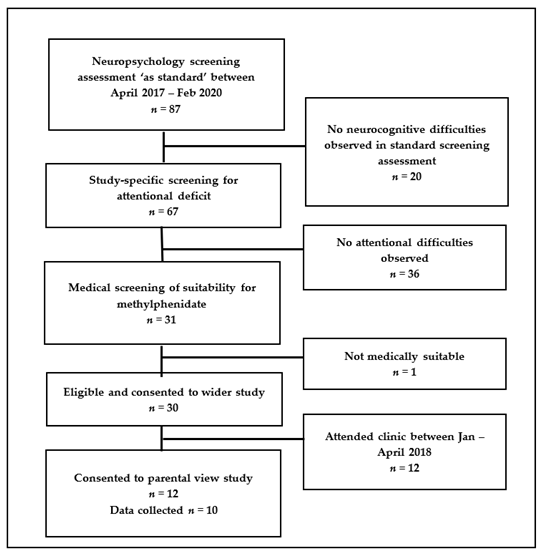
Eligible parents had a child in receipt of methylphenidate treatment for post-treatment attentional impairment. Each child had received methylphenidate for six months (± 6 weeks). All children had a brain tumour and had either previous hydrocephalus or cranial radiotherapy treatment. All parents attending clinic between January–April 2018 were approached (see Appendix B). Ten parents of the twenty-one attending clinic during this time period consented to the study (48%). Reasons for non-inclusion included non-return of questionnaire forms, presence of younger siblings at appointment, or parental lack of time to participate. There was no difference in methylphenidate efficacy between participants and non-participants. Demographic details of the patient group are presented as Figure 1 and Table 1.
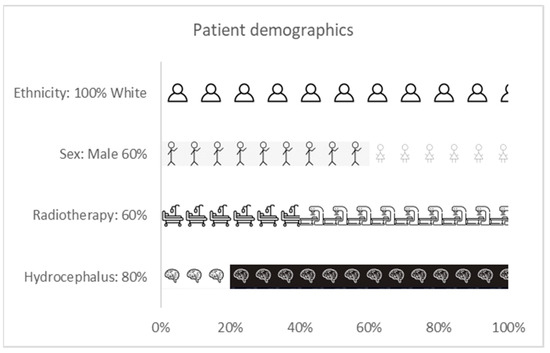
Figure 1.
Patient demographic data—Ethnicity, sex, radiotherapy, and Cerebro Spinal Fluid (CSF) status at diagnosis.

Table 1.
Patient demographic data—Diagnosis, age at treatment, and methylphenidate use.
2.3. Materials
- Pediatric Quality of Life Parent Report form (PedsQL). The PedsQL is a 23-item scale that explores four dimensions; school, emotional, physical, and social functioning. This reliable and valid multidimensional tool has frequently been used in both healthy populations and children and adolescents with cancer [22,23]. The PEDS-QL was completed by parents at baseline, at six weeks, and again at six months (± 6 weeks). Data from baseline and six months are presented in the current study.
- Experiences of Methylphenidate Treatment Questionnaire—Parent Form (EMTQ-parent form) (Appendix C). A bespoke questionnaire was designed for the study that explored parents’ views on the impact that the methylphenidate treatment had on their child’s HRQoL. Questionnaire development was informed by the Experiences of Methylphenidate Treatment Questionnaire developed for use with children. A focus group and individual consultations were held with parents prior to the development of this questionnaire. The questionnaire required parents to retrospectively reflect on their experiences of the service pre and post treatment delivery, and to evaluate the effect parents perceived that methylphenidate had on their children. Parents were provided the questionnaire at six months from the date of their child starting methylphenidate (±6 weeks).
Procedure
All participants were provided with HRQoL parent-report questionnaires during a routine clinical appointment. Adherence to the treatment regime was confirmed by clinical discussion. Participants filled in the PedsQL and EMTQ questionnaire independently, or received help from a clinical psychologist if required. Participants were asked to expand upon their responses to the EMTQ by clinical interview.
2.4. Data Analysis
2.4.1. Quantitative Data
Paired samples t-tests were performed upon the dimensions of the PedsQL parent form questionnaire, comparing baseline (pre methylphenidate) and post treatment conditions.
2.4.2. Qualitative Data
Qualitative data were analysed using thematic analysis [24]. A three-stage analytic process was conducted to enhance credibility, reliability and generalisability. Initial data coding was conducted by the authors using manual open coding techniques. Data were peer coded and coding decisions assessed for inter-rater agreement. Saturation was determined and higher-level theme stability provisionally confirmed. A member check was carried out with two participants to confirm validity of analyses.
2.5. Ethical Considerations
Ethical approval was obtained for the evaluation by the Newcastle University Ethics Committee. The evaluation was registered with Newcastle Upon Tyne Hospitals NHS Foundation Trust (Reg. No. 9049). Participants provided consent for data to be collected.
3. Results
3.1. Parental Perception of Effect of Methylphenidate on HRQoL
3.1.1. Quantitative Data
Health-Related Quality of Life was measured using the PEDS-QL parent-report questionnaire. Group mean results are shown as Table 2. Individual parent-report scores are presented as Figure 2. Higher scores on the PEDS-QL indicate a greater number/greater severity of problems. Decreased scores therefore indicate benefit to HRQoL associated with use of methylphenidate.

Table 2.
Pre to post intervention scores of HRQoL function.
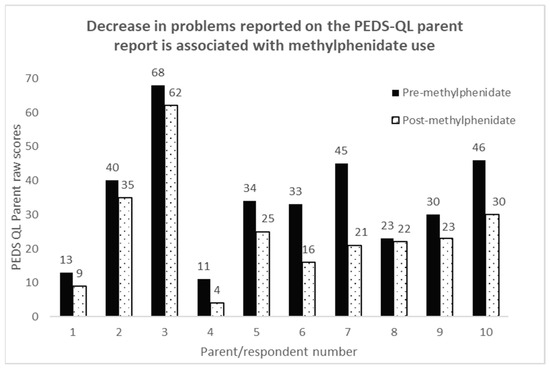
Figure 2.
Total Quality of Life scores prior to and following use of methylphenidate as measured by parent-report tool PEDS-QL.
Differences between scores at baseline (prior to methylphenidate treatment) and following treatment were significant on parent report for the domains of school function (t (9) = −3.19, p =< 0.01), emotional functioning (t (9) = −2.64, p < 0.02), and social functioning (t (9) = −2.375, p < 0.04). The direction of differences showed a perceived positive impact of methylphenidate on these domains, such that difficulties in all domains of HRQoL were reduced following methylphenidate treatment. Pre and Post scores were not significant for physical function (t (9) = −1.8, p = 0.11)
3.1.2. Qualitative Data
Data gained via the qualitative questionnaire and follow-up were analysed using thematic analysis. Parental opinion of the benefit of methylphenidate were grouped into five key themes: physical impact, academic improvement, emotional impact, neuropsychological effects, and social impact. Each theme is illustrated with a quote from individual parents (Figure 3).
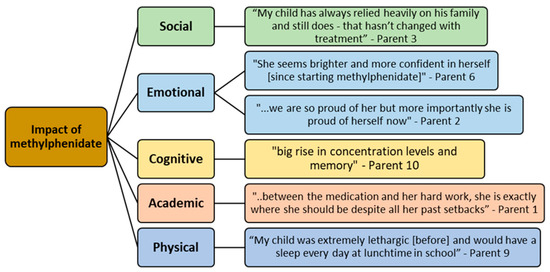
Figure 3.
Thematic analytic ‘themes’ on benefit of methylphenidate to HRQoL as reported by parents.
3.1.3. Physical Effects
The physical impact of methylphenidate was highlighted by parents. Fatigue was a common narrative throughout all participants’ responses and the impact it had on their children’s daily functioning. The fatigue-reduction aspect of treatment benefit was described as ‘life-changing’ by one family within clinic and was rated as the side-effect of methylphenidate that was most valued by families. Following participation in the methylphenidate trial, seven out of ten (70%) participants reported significant improvements in energy levels and the positive effect this then had on their physical activity levels.
3.1.4. Emotional Impact
Seven (70%) participants reported an increase in their child’s self-esteem and confidence in themselves and their abilities following methylphenidate treatment. Reports of the impact of methylphenidate on self-esteem included phrases such as ‘proud of (his/her) self’. Parents also reported a perceived positive impact on mood of methylphenidate. Increase in mood was associated by parents with an apparent increase in social activity and a reported increase in willingness to try new things.
3.1.5. Social Impact
There was relatively little perceived impact of methylphenidate upon the child’s social function, with most participants not reporting any direct benefit in this area.
3.1.6. Cognitive and Academic Impact
Parents reported a perceived positive effect of methylphenidate on their child’s academic and cognitive functioning, commenting upon the quantity and quality of their child’s academic function as observed via their child’s performance of homework and enjoyment of school. Increase in academic performance was reported by nine out of the ten (90%) participants. At baseline assessment (prior to methylphenidate treatment) all participants reported their child had experienced treatment-related cognitive difficulties that impacted upon academic performance, including memory difficulties, slowing of processing speed, and lowered concentration. Nine out of the ten (90%) participants also reported improvement in their child’s cognitive functions. Parental responses attributed perceived academic improvement to improvement in their child’s attention, concentration, and memory.
3.2. Parental Views on Treatment Acceptability
Data gained via the qualitative questionnaire and follow-up were analysed using thematic analysis. Three higher-order themes were identified: alleviation of concerns about starting methylphenidate, informed decision-making, and post-methylphenidate concerns. Within these higher-order themes, sub-themes were further identified. These are presented as a thematic diagram (Figure 4).
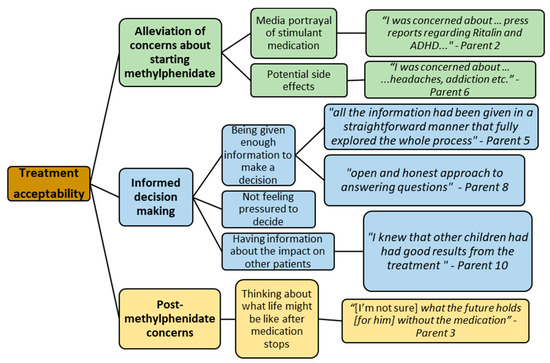
Figure 4.
Parental data on acceptability of methylphenidate suggest that provision of information and the opportunity to discuss concerns is paramount to confidence in treatment.
Most participants (nine participants—90%) reported some treatment related concerns prior to their children taking methylphenidate. Responses within this theme included comments about potential side effects, and the parents’ pre-existing ‘knowledge’ about methylphenidate taken from social media or the mainstream press. For many participants (eight participants—80%), the prospect of their children beginning methylphenidate prompted concerns about potential physical and psychological consequences of taking a stimulant drug. All parents reported that concerns were alleviated by consultation with clinic staff.
Parents particularly recalled being advised of potential side effects, and that the medication may not have a positive effect for their child. For many participants (eight participants—80%), the prospect of their children beginning methylphenidate prompted concerns about potential physical and psychological consequences of taking a stimulant drug. All participants (ten participants—100%) reported feeling that they had been provided with sufficient information about the treatment to make an informed decision about their child’s treatment. Some participants commented that they had spoken to parents whose children were already receiving this treatment in the service. Parents reported that they were given sufficient time to make a joint decision about treatment, and that this was important to them.
Approximately one-third (three participants—30%) of participants reported concern related to how their children would now cope if they were no longer prescribed methylphenidate. These parents reported a high level of current satisfaction with the medication, and uncertainty as to their children’s future if methylphenidate were not continued.
4. Discussion
Methylphenidate is emerging as a potential treatment option for the management of post-treatment challenges faced by survivors of paediatric brain tumour. This study aimed to:
- Gather preliminary data on parental perception of the impact of methylphenidate on HRQoL in this population.
- Gather preliminary data on the parental acceptability of methylphenidate as an interventional medium.
4.1. Parental Perception of the Impact on HRQoL
Analyses of preliminary PedsQL parent questionnaire data show perceived improvement in academic, social, and emotional functioning following use of methylphenidate. This supports qualitative data gathered in our small sample, suggesting that parental perception of methylphenidate is positive and that child HRQoL was improved following use of methylphenidate.
4.2. Physical Impact of Methylphenidate
Fatigue is reported to be the most frequently experienced symptom of paediatric oncology survivorship, both in treatment and later survivorship [25]. Cancer-related fatigue has been described as a persistent, distressing sense of emotional, physical and/or cognitive tiredness. Despite parental report of improvement in activity levels and consistent reduction in fatigue referenced within semi-structured questionnaire responses, no significant difference was found in quantitative (PedsQL) measures of physical functioning in this sample cohort. This contrasts with a previous study by our team that gathered data on children’s perceptions of the effect of methylphenidate [20]. This study found that child-report PedsQL data showed a strong positive impact of methylphenidate on perceived fatigue reduction by the children themselves. It is possible that, whilst children reported ‘feeling better’ due to the reduction of fatigue effected by psychostimulant treatment, parents focused upon the remaining level of physical disability faced by their child. Despite non-significant findings in respect of fatigue alleviation on the parental PedsQL measure, parental reports of methylphenidate-related changes gathered by questionnaire frequently referred to the perceived reduction of fatigue in children. Discordance between quantitative and qualitative data in the current study may be due to the use of the standard PedsQL measure for gathering of physical data, rather than use of the research specific PedsQL Multi-Dimensional Fatigue Scale (MDFS). Whilst not capturing wider physical function, the fatigue-specific nature of the specialist measure is indicated as a possible appropriate tool for measurement of methylphenidate-related change in this group. Study with a larger, more generalizable, cohort would be supported by using the MDFS.
4.3. Emotional and Social Function
Our preliminary data suggest a possible relationship between emotional wellbeing, self-esteem, social function, and fatigue/physical energy levels. It is known that children with brain tumour score lower on measures of each of these areas. Low self-esteem is a common difficulty in paediatric oncology patients, being attributed to factors including disrupted friendship groups associated with treatment and reduced opportunity for social interactions associated with illness and fatigue [26,27]. Children with brain tumour typically experience higher levels of social isolation than peers and siblings. They are perceived by their peer group as being sick, easily fatigued, and frequently absent from school [28]. Schulte et al. [29] reported that paediatric survivors of brain tumours frequently experience social adjustment deficits that can worsen over time and negatively impact survivors’ long-term quality of life. Our parent data show that, whilst some parents reported an increase in social activity that they associated with alleviated fatigue following methylphenidate use, other parents report a continued low social activity level of their child associated with a remaining level of disability. Parents associated reduced social activity level secondary to fatigue and disability to lower self-esteem and reduced friendship opportunities. Mediational models used on data from a larger sample are needed to explore the relationship between these factors. In particular, we must establish the direct and indirect associations and direction of relationship between fatigue, self-esteem, social activity level, and overall HRQoL.
4.4. Cognitive and Academic Function
Our preliminary data show an association between a perceived increase in academic function/cognitive ability with the child’s enjoyment of school and willingness to attend. It must be noted that the association made by parents in this study of educational improvement with attendance/willingness to attend may not reflect the actual mechanism of change in this relationship. No study to date has evidenced improvement in academic attainment associated with methylphenidate use in the paediatric brain tumour population. Nonetheless, it is important to acknowledge the importance of enjoyment of school in impacting upon children’s own perceived quality of life. Future research with a more representative sample would benefit from a longer-term follow up study of the effect of methylphenidate on academic performance measured over a two year plus timescale.
4.5. Acceptability of Treatment
Whilst based on a small cohort, preliminary parental data on the acceptability of methylphenidate as an interventional medium were very positive. Questionnaire data suggest that the level of psycho-education provided to families in advance of treatment commencement has the potential to have a positive impact on families’ willingness to trial and maintain treatment. Given the extent to which familial ‘buy in’ to treatment affects treatment adherence in this age range, this was clearly an important factor in the patient’s access to methylphenidate.
Data suggest that pre-treatment psychoeducation was most valued in relation to negative preconceived beliefs about methylphenidate held by families. Methylphenidate is a drug that is distributed under several different trade names. Families were most familiar with the brand ‘Ritalin’, citing this frequently as a drug that they had heard of, and read about in the mainstream press. Families also raised awareness of the treatment as being a psychostimulant, associating this in many cases with so-called ‘recreational’ drugs such as cocaine. The most frequently reported pre-treatment concern related to mainstream press stories of children with Attention Deficit/Hyperactivity Disorder (ADD/ADHD), in which parents raised concern that their child might be over-medicated to the point of sedation.
Feared negative consequences of treatment in clinic were associated by families with higher pre-treatment beliefs about Ritalin. This is consistent with the literature regarding parental perceptions of methylphenidate, which has identified common recurrent beliefs that the medication could be addictive, dull cognition and stigmatize a child [30,31]. However, research has shown that paediatric cancer survivors tolerate methylphenidate well [32], and given the significant benefits that methylphenidate can have, further consideration into addressing misperceptions is required.
All participants reported that, following individual consultation appointments, their initial concerns had been alleviated. Participants reflected on how having space in consultations to discuss their concerns and fears was very helpful and enabled them to feel empowered and validated. Participants also reinforced the fact that not being overloaded with information was important, and they valued honest information regarding potential benefits and side effects, and the supporting evidence base. Parents reported a high level of satisfaction with service delivery, describing it as person-centred, informative and thorough throughout all stages of their child’s treatment.
4.6. Concerns about Treatment
A small number of parents discussed concerns about treatment end. The main concerns expressed were fears about the possible loss of treatment gains, uncertainty about their child’s future, and how they might cope emotionally and cognitively without medication post-education. Uncertainly about the long-term use of this medication in this population was also raised, including concerns about the potential for any unforeseen side-effects of long-term methylphenidate use following brain tumour. Given the early stage of research on the utility of methylphenidate in the paediatric Neuro-Oncology population, no protocols for treatment-length currently exist. These preliminary data highlight unaddressed parental concern about the potential impact of treatment termination using this intervention.
4.7. Strengths, Limitations and Future Directions
The current study is the first to present parental opinion on methylphenidate in this population and the treatment acceptability to families of the same. We are pleased to include the perspectives of Neuro-Oncology parents and to demonstrate our commitment to participant-inclusive research. Parental support of paediatric treatment is a critical component of effective clinical intervention. The suggestion that methylphenidate is perceived to be beneficial by parents is a welcome addition to an emerging knowledge base about the use of this medication.
Despite the promising nature of our data, the study is subject to significant methodological limitation. Generalisability of findings is constrained by the very small sample size (n = 10). As no demographic data on the parent respondents were collected, we would particularly note that responses presented here may reflect that of particular demographics rather than of most parents. Whilst no difference in treatment efficacy was seen between children whose parents responded versus children whose parents did not respond, it is recognised that all patients in this centre prescribed methylphenidate are White, UK-born, and speakers of English as a native language. Our data potentially obscure the experiences of families from some more marginalised demographics, privileging and legitimising data from a narrow group of participants. A no-treatment control group was not provided. It was initially intended that treatment-refusers might offer a potential control, however, all initial families offered methylphenidate treatment consented to treatment leaving no appropriate control. Furthermore, our qualitative data were gathered at the six-month time point. The retrospective nature of the parent-report qualitative data risks inaccuracy of parental report. Research is required to investigate parental perception of benefit in the medium to long term, potentially by diary method. Finally, our data show a correlation between decrease in HRQoL difficulties and use of methylphenidate. Whilst the temporal relationship between use of methylphenidate and the improvement to children’s lives in a range of domains is promising, we do not claim to evidence causality.
5. Conclusions
Survivors of PBT experience substantial post-treatment challenges, reporting greater difficulties and reduced HRQoL compared to survivors of other childhood cancers. An emerging evidence base for methylphenidate in children with PBT suggests potential gains to cognitive performance and to HRQoL. Parental opinion is a vital facet of paediatric treatment, affecting outcome factors such as treatment adherence and influencing children’s own perception of treatment efficacy. This study offers preliminary provisional data from a small group of participants on the effect of methylphenidate on HRQoL and the acceptability of methylphenidate as an interventional approach.
Author Contributions
Conceptualization: S.J.V.; Methodology: S.J.V.; Data collection: S.J.V.; Formal analysis and investigation: L.S.; Writing-original draft preparation: L.S.; Writing-review and editing: S.J.V.; Supervision: S.J.V. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no funding.
Institutional Review Board Statement
These data are part of a clinical audit registered with Newcastle Upon Tyne Hospitals NHS Foundation Trust-Clinical Register No. 9049, registered 29 January 2019. The study was conducted according to the guidelines of the Declaration of Helsinki.
Informed Consent Statement
All participants were over 18 years old at time of participation and consented to complete questionnaires throughout their child’s treatment for the purpose of clinical follow up and research.
Data Availability Statement
Raw data (transcripts) can be made available upon request, however, these will be subject to redaction to ensure the confidentiality of participants is preserved.
Acknowledgments
Uschi Hiermeier (Assistant Psychologist, Northumbria Healthcare NHS Foundation Trust) assisted with manuscript preparation. Families of the Great North Children’s Hospital assisted with the development of the EMTQ tool described in this paper (Cooper family, Angus family).
Conflicts of Interest
The authors have no conflict of interest to declare that are relevant to the content of this article.
Appendix A. Eligibility Criteria for Trial of Methylphenidate Treatment
Appendix A.1. Inclusion Criteria
- Participant has been diagnosed with a CNS tumour in the previous 10 years, counting from diagnosis date.
- Patient is aged between 6 years 0 months and 16 years 0 months at start of methylphenidate treatment.
- Has been off therapy/active treatment for CNS tumour for 12 months at the start of methylphenidate and has a likely life expectancy of > 5 years.
- No known signs of clinical or radiological tumour progression at screening for methylphenidate.
- English is the sole or primary language (enables provision of valid psychometric assessment).
- Patient and family have provided assent/consent for trial of methylphenidate.
- Neurocognitive impairment of processing speed or attention as assessed by:
- Clinically significant loss of previous function in processing speed and/or attention as shown by repeat psychometric assessment or estimate of premorbid ability.
- Discrepancy between the General Ability Index (GAI) and Cognitive Processing Index (CPI) (GAI > CPI) that is statistically significant at p = 0.05 for age.
Appendix A.2. Exclusion Criteria
- Contraindications to methylphenidate medication as per current BNF for Children (BNFC) and NICE Guideline 87:
- Family history of tic disorder
- Glaucoma (history in family from an early age)
- Current antidepressant or anxiolytic use
- Significant problems maintaining weight or previous eating disorder
- History of substance misuse
- Recent or current poorly controlled seizures.
- History of cardiac issues
- Pregnancy: whilst pregnancy and breastfeeding are not listed as contraindications in the BNFC, methylphenidate is listed as ‘Limited experience—avoid unless potential benefit outweighs risk’. Prospective patients known to be pregnant or breastfeeding at screening/registration will not be offered methylphenidate.
- Significant mental health difficulties (e.g., clinically severe depression, psychosis).
- Diagnosis of Attention Deficit/Hyperactivity Disorder or Autism Spectrum Disorder
- Full Scale Intelligence Quotient (FSIQ) of <50.
- Concerns about family ability to safely store or administer methylphenidate, or to report side effects appropriately/Concerns about familial substance misuse.
Appendix B
Flowchart of screening for eligibility
Appendix C. Experience of Methylphenidate Treatment Questionnaire
Thank you for taking the time to answer the following questions. We are interested in learning about your perception of the effect of methylphenidate on your child. Please aim to expand on your answers to provide as much information as possible.
We will then discuss with you your answers in your next clinic appointment. We might ask you to give us a bit more information about some of your answers, if you can.
- What were the biggest difficulties in your child’s life before taking methylphenidate?
- How did you know when the methylphenidate started working (if it did)?
- What were the first things you noticed?
- How did you notice these things?
- Has methylphenidate had an impact on your child’s social life, and if so how?
- Has methylphenidate had an impact on your child’s independence, and if so how?
- Has methylphenidate had an impact on your child’s mood, and if so, how?
- Has methylphenidate affected your child’s confidence, and if so how?
- Has methylphenidate impacted upon your child’s school life, and if so how?
- Has methylphenidate changed anything about how you think your child feels about themselves, and if so how?
- Has methylphenidate had an impact on your child’s relationships, and if so how?
- Has methylphenidate affected your child’s fatigue levels, and if so how?
References
- Pogorzala, M.; Styczynski, J.; Kurylak, A.; Debski, R.; Wojtkiewicz, M.; Wysocki, M. Health-related quality of life among paediatric survivors of primary brain tumours and acute leukaemia. Qual. Life Res. 2010, 19, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Macartney, G.; Harrison, M.B.; VanDenKerkhof, E.; Stacey, D.; McCarthy, P. Quality of Life and Symptoms in Pediatric Brain Tumor Survivors: A Systematic Review. J. Pediatr. Oncol. Nurs. 2014, 31, 65–77. [Google Scholar] [CrossRef]
- Zebrack, B.J.; Gurney, J.G.; Oeffinger, K.; Whitton, J.; Packer, R.J.; Mertens, A.; Turk, N.; Castleberry, R.; Dreyer, Z.; Robison, L.L.; et al. Psychological Outcomes in Long-Term Survivors of Childhood Brain Cancer: A Report from the Childhood Cancer Survivor Study. J. Clin. Oncol. 2004, 22, 999–1006. [Google Scholar] [CrossRef] [PubMed]
- Karschnia, P.; Parsons, M.W.; Dietrich, J. Pharmacologic management of cognitive impairment induced by cancer therapy. Lancet Oncol. 2019, 20, e92–e102. [Google Scholar] [CrossRef]
- Shah, S.S.; Dellarole, A.; Peterson, E.C.; Bregy, A.; Komotar, R.; Harvey, P.D.; Elhammady, M.S. Long-term psychiatric outcomes in pediatric brain tumor survivors. Child’s Nerv. Syst. 2015, 31, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Kahalley, L.S.; Conklin, H.M.; Tyc, V.L.; Hudson, M.M.; Wilson, S.J.; Wu, S.; Xiong, X.; Hinds, P.S. Slower processing speed after treatment for pediatric brain tumor and acute lymphoblastic leukemia. Psycho-Oncology 2013, 22, 1979–1986. [Google Scholar] [CrossRef]
- Iyer, N.S.; Balsamo, L.M.; Bracken, M.B.; Kadan-Lottick, N.S. Chemotherapy-only treatment effects on long-term neurocognitive functioning in childhood ALL survivors: A review and meta-analysis. Blood 2015, 126, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Zeltzer, L.K.; Recklitis, C.; Buchbinder, D.; Zebrack, B.; Casillas, J.; Tsao, J.C.; Lu, Q.; Krull, K. Psychological Status in Childhood Cancer Survivors: A Report From the Childhood Cancer Survivor Study. J. Clin. Oncol. 2009, 27, 2396–2404. [Google Scholar] [CrossRef] [PubMed]
- Gong, S.; Sheng, P.; Jin, H.; He, H.; Qi, E.; Chen, W.; Dong, Y.; Hou, L. Effect of Methylphenidate in Patients with Cancer-Related Fatigue: A Systematic Review and Meta-Analysis. PLoS ONE 2014, 9, e84391. [Google Scholar] [CrossRef]
- Bolea-Alamañac, B.; Nutt, D.J.; Adamou, M.; Asherson, P.; Bazire, S.; Coghill, D.; Heal, D.; Müller, U.; Nash, J.; Santosh, P.; et al. Evidence-based guidelines for the pharmacological management of attention deficit hyperactivity disorder: Update on recommendations from the British Association for Psychopharmacology. J. Psychopharmacol. 2014, 28, 179–203. [Google Scholar] [CrossRef] [PubMed]
- Johansson, B.; Andréll, P.; Rönnbäck, L.; Mannheimer, C. Follow-up after 5.5 years of treatment with methylphenidate for mental fatigue and cognitive function after a mild traumatic brain injury. Brain Inj. 2019, 34, 229–235. [Google Scholar] [CrossRef]
- Johansson, B.; Wentzel, A.-P.; Andréll, P.; Rönnbäck, L.; Mannheimer, C. Long-term treatment with methylphenidate for fatigue after traumatic brain injury. Acta Neurol. Scand. 2016, 135, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Johansson, B.; Wentzel, A.-P.; Andréll, P.; Mannheimer, C.; Rönnbäck, L. Methylphenidate reduces mental fatigue and improves processing speed in persons suffered a traumatic brain injury. Brain Inj. 2015, 29, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Mulhern, R.K.; Khan, R.B.; Kaplan, S.; Helton, S.; Christensen, R.; Bonner, M.; Brown, R.; Xiong, X.; Wu, S.; Gururangan, S.; et al. Short-Term Efficacy of Methylphenidate: A Randomized, Double-Blind, Placebo-Controlled Trial Among Survivors of Childhood Cancer. J. Clin. Oncol. 2004, 22, 4795–4803. [Google Scholar] [CrossRef] [PubMed]
- Conklin, H.M.; Khan, R.B.; Reddick, W.E.; Helton, S.; Brown, R.; Howard, S.C.; Bonner, M.; Christensen, R.; Wu, S.; Xiong, X.; et al. Acute Neurocognitive Response to Methylphenidate Among Survivors of Childhood Cancer: A Randomized, Double-Blind, Cross-Over Trial. J. Pediatr. Psychol. 2007, 32, 1127–1139. [Google Scholar] [CrossRef]
- Conklin, H.M.; Helton, S.; Ashford, J.; Mulhern, R.K.; Reddick, W.E.; Brown, R.; Bonner, M.; Jasper, B.W.; Wu, S.; Xiong, X.; et al. Predicting Methylphenidate Response in Long-Term Survivors of Childhood Cancer: A Randomized, Double-Blind, Placebo-Controlled, Crossover Trial. J. Pediatr. Psychol. 2010, 35, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Conklin, H.M.; Reddick, W.E.; Ashford, J.; Ogg, S.; Howard, S.C.; Morris, E.B.; Brown, R.; Bonner, M.; Christensen, R.; Wu, S.; et al. Long-Term Efficacy of Methylphenidate in Enhancing Attention Regulation, Social Skills, and Academic Abilities of Childhood Cancer Survivors. J. Clin. Oncol. 2010, 28, 4465–4472. [Google Scholar] [CrossRef]
- Minton, O.; Richardson, A.; Sharpe, M.; Hotopf, M.; Stone, P.C. Psychostimulants for the Management of Cancer-Related Fatigue: A Systematic Review and Meta-Analysis. J. Pain Symptom Manag. 2011, 41, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Attention Deficit Hyperactivity Disorder: Diagnosis and Management NICE Guideline. 2018. Available online: www.nice.org.uk/guidance/ng87 (accessed on 23 February 2022).
- Verity, S.J.; Bell, L.; Ryles, J.; Hill, R.M. “I Feel Happy Again”: Methylphenidate Supports Health-Related Quality of Life in Survivors of Pediatric Brain Tumor. Children 2022, 9, 1058. [Google Scholar] [CrossRef] [PubMed]
- Qadeer, R.A.; Ferro, M.A. Child–parent agreement on health-related quality of life in children with newly diagnosed chronic health conditions: A longitudinal study. Int. J. Adolesc. Youth 2018, 23, 99–108. [Google Scholar] [CrossRef] [Green Version]
- Chaudhry, Z.; Siddiqui, S. Health related quality of life assessment in Pakistani paediatric cancer patients using PedsQLTM 4.0 generic core scale and PedsQL™ cancer module. Health Qual. Life Outcomes 2012, 10, 52. [Google Scholar] [CrossRef] [PubMed]
- Felder-Puig, R.; Frey, E.; Proksch, K.; Varni, J.; Gadner, H.; Topf, R. Validation of the German version of the Pediatric Quality of Life InventoryTM(PedsQLTM) in childhood cancer patients off treatment and children with epilepsy. Qual. Life Res. 2004, 13, 223–234. [Google Scholar] [CrossRef]
- Braun, V.; Clarke, V. Using thematic analysis in psychology. Qual. Res. Psychol. 2006, 3, 77–101. [Google Scholar] [CrossRef]
- Wood, J.; Verity, S.J. Exploring Evidence of Fatigue in Survivors of Pediatric Brain Tumors: A Systematic Review. Cancer Care Research Online 2021, 1, e012. [Google Scholar] [CrossRef]
- Castellino, S.M.; Ullrich, N.J.; Whelen, M.J.; Lange, B.J. Developing Interventions for Cancer-Related Cognitive Dysfunction in Childhood Cancer Survivors. JNCI J. Natl. Cancer Inst. 2014, 106, dju186. [Google Scholar] [CrossRef] [PubMed]
- Langeveld, N.E.; Grootenhuis, M.A.; Voûte, P.A.; De Haan, R.J.; Van Den Bos, C. Quality of life, self-esteem and worries in young adult survivors of childhood cancer. Psycho-Oncology 2004, 13, 867–881. [Google Scholar] [CrossRef] [PubMed]
- Vannatta, K.; Gartstein, M.; Short, A.; Noll, R.B. A Controlled Study of Peer Relationships of Children Surviving Brain Tumors: Teacher, Peer, and Self Ratings. J. Pediatr. Psychol. 1998, 23, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Schulte, F.; Kunin-Batson, A.S.; Olson-Bullis, B.A.; Banerjee, P.; Hocking, M.C.; Janzen, L.; Kahalley, L.S.; Wroot, H.; Forbes, C.; Krull, K.R. Social attainment in survivors of pediatric central nervous system tumors: A systematic review and meta-analysis from the Children’s Oncology Group. J. Cancer Surviv. 2019, 13, 921–931. [Google Scholar] [CrossRef]
- Bussing, R.; Gary, F.A. Practice Guidelines and Parental ADHD Treatment Evaluations: Friends or Foes? Harv. Rev. Psychiatry 2001, 9, 223–233. [Google Scholar] [CrossRef]
- Charach, A.; Skyba, A.; Cook, L.; Antle, B.J. Using Stimulant Medication for Children with ADHD: What Do Parents Say? A Brief Report. J. Can. Acad. Child Adolesc. Psychiatry 2006, 15, 75. [Google Scholar] [PubMed]
- Conklin, H.M.; Lawford, J.; Jasper, B.W.; Morris, E.B.; Howard, S.C.; Ogg, S.W.; Wu, S.; Xiong, X.; Khan, R.B. Side Effects of Methylphenidate in Childhood Cancer Survivors: A Randomized Placebo-Controlled Trial. Pediatrics 2009, 124, 226–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).