Abstract
Background: The efficacy of pharmacotherapy for attention-deficit/hyperactivity disorder (ADHD) has been confirmed in numerous controlled studies. However, in clinical practice, pharmacological treatment is heavily dependent upon the parents or guardians of patients. Parental attitudes are shaped not only by medical knowledge but also by parental beliefs about the use of pharmacotherapy and psychoactive substances, including ADHD pharmacotherapy. Parental beliefs about the safety and possible side effects of pharmacotherapy significantly influence their decision to accept or reject pharmacotherapy. This study aimed to explore parental beliefs and attitudes toward ADHD pharmacotherapy and their association with parental treatment acceptance and treatment-related decision-making. Methods: The cross-sectional online survey included 506 parents of children diagnosed with ADHD, recruited through closed social media groups. Parental treatment acceptance and decisions regarding initiation of pharmacotherapy were examined. Results: Parents of children with experience of pharmacotherapy more frequently considered pharmacotherapy safe (83.4% vs. 39.7%, p < 0.001) and expressed readiness to start treatment immediately (73.8% vs. 32.5%, p < 0.001). In this group, 72.6% of parents indicated that the benefits of pharmacotherapy outweigh potential risks. However, concerns about addiction were similar in both groups (49.4% vs. 45.3%, p = 0.400). In a gendered analysis, fathers were more likely than mothers to consider pharmacotherapy unnecessary for treating ADHD (35.3–22.4%; p = 0.002; V = 0.142) or disbelieve in ADHD treatment (25.7–15.1%; p = 0.005; V = 0.132). Furthermore, fathers were more likely than mothers to support limiting the use of pharmacotherapy for treating ADHD to those over 18 (41.3% vs. 26.5%; p < 0.001; V = 0.156), and to report parental opposition (28% vs. 8.1%; p < 0.001; V = 0.264), with a minimal to moderate effect size. Conclusions: Parental treatment acceptance decisions were associated with more favorable parental beliefs, although subjective concerns about addiction remained. Given the study’s cross-sectional and exploratory nature, causal interpretations should be avoided. Parental gender was associated with differences in beliefs and attitudes toward the use of pharmacotherapy for treating children diagnosed with ADHD, especially regarding necessity, consent and age-specificity. In our surveyed sample, fathers tended to be more restrictive or cautious than mothers. The findings also highlight the importance of psychoeducation and partnership-based communication between clinicians and families as key factors that may help support treatment acceptance-related decision-making in ADHD.
1. Introduction
Attention-deficit/hyperactivity disorder (ADHD) is one of the most frequently diagnosed neurodevelopmental disorders in children and adolescents. It is characterized by persistent inattention, hyperactivity, and impulsivity, leading to significant difficulties in family, academic, occupational, and social functioning—both during childhood and adulthood [1,2]. ADHD affects approximately 5–7% of children worldwide, while in the United States, its prevalence is about 11% [3]. In Poland, epidemiological data are limited, but available analyses indicate rates similar to those of other European Union countries, ranging from 4.4% to 6.2% [4]. For many individuals, ADHD symptoms persist into adolescence and adulthood, and without treatment, they are associated with serious consequences in multiple areas of life. The disorder contributes to educational difficulties, interpersonal problems, chronic family stress, and reduced quality of life [5]. Moreover, research indicates that individuals with ADHD have a shorter life expectancy compared to the general population, underscoring the importance of early diagnosis and intervention [6]. Despite strong evidence supporting the effectiveness of pharmacological treatment, discrepancies between guidelines and actual clinical practice remain. These discrepancies include delayed initiation or omission of pharmacotherapy despite clear clinical indications. Stimulants show high efficacy in reducing ADHD symptoms, with therapeutic response rates of up to 70–80% [7], while non-stimulant pharmacotherapy, though effective, generally shows lower potency [5]. According to current clinical guidelines, pharmacotherapy is the first-line treatment for children and adolescents with severe ADHD symptoms; in moderate cases, it should be used when non-pharmacological interventions prove insufficient [8,9,10]. However, despite the availability of effective treatment options, the application of pharmacotherapy in clinical practice is often delayed or inconsistent. Both international and national guidelines emphasize that treatment decisions should consider patients’ beliefs, concerns, and level of knowledge, as well as the role of their families or caregivers [11]. Since children do not independently decide to follow prescribed treatment, parental attitudes play a crucial role in initiating and maintaining therapy [12]. The Theory of Planned Behavior (TPB) can be used as a helpful concept for comprehending the process of parental decision-making during the treatment of ADHD. According to the TPB, attitudes toward the behavior, subjective norms, and perceived behavioral control jointly predict behavioral intentions, while intentions and perceived behavioral control directly influence behavior [13]. The framework can be used to clarify differences in parental help-seeking behaviors and treatment acceptance across cultures. For evaluating the application of TPB to decisions concerning ADHD treatment, the manner in which subjective norms, experience of pharmacotherapy, and experience of control are correlated with each other in influencing parental choices has been studied [14]. In one exploratory study, mothers attributed inattentive and impulsive child behavioral symptoms to more global and stable causes than fathers. Fathers were more negative about ADHD symptoms with an internal origin than mothers. Fathers believed more in psychological explanations and ADHD treatments, whereas mothers had more trust in behavior management [15]. Data from the Caregiver Perspective on Paediatric ADHD (CAPPA) survey, a large European caregiver study, showed that female 79 caregivers—primarily mothers—were the predominant respondents (approximately 66%), suggesting a gendered distribution of caregiving responsibilities and treatment decision-making in pediatric ADHD [16]. Comparable data addressing these dynamics in the Polish context are currently scarce. The literature indicates that parental gender played a role in accessing and accepting treatment for ADHD. For instance, Wright et al. (2015) [17] performed a systematic review that suggested that decision-making may be affected by the knowledge and attitudes of parents, teachers, and professionals. The review showed that parental decision-making influences access to services, while stigma negatively affects families, their willingness to engage in care, and the effectiveness of school-based interventions for children and adolescents. In a separate study, the authors aimed to identify features of ADHD stimulant medications that influence treatment choices among parents of children with ADHD across six European Union (EU) countries. In total, 220 parents of teenagers and 380 parents of younger children participated. The study found that the degree of symptom control was the most critical feature, with parents of adolescents having an OR of 4.85 [95% CI = 4.28–5.49], preferring ‘very substantially improved symptoms’ over ‘minimally 94 improved’, and parents of children having an OR of 6.37 (95% CI = 5.79–7.01). Inter-country differences showed that parents prioritized symptom control over other factors. The study concluded that the differences among parents in all countries who prioritized treatment duration and degree of symptom control were due to cultural disparities in attributes [17]. For understanding parental attitudes and belief differences, there is a need to understand cultural and ethnic factors. Shehadeh Sheeny, A. et al. (2023) [18] found that cultural factors influence parents’ beliefs and attitudes, making social and cultural elements crucial to understanding ADHD diagnosis and treatment patterns. The authors identified differences in two ethnic groups due to their cultural variabilities. Arab mothers and instructors saw ADHD as a form of childhood social behavior, while Jewish mothers and teachers saw it as a medical issue. Arab mothers felt guilty about ADHD, whereas Jewish mothers sympathized with their children. The social milieu influenced Arab mothers more than Jewish mothers. Arab mothers alone reported stigmatization [18]. Thus, some families associate social stigma with a child’s behavior or an ADHD diagnosis. Consequently, caregivers may be reluctant to pursue diagnosis and treatment due to social stigma [19]. These barriers can make it challenging to navigate health and receive timely access to diagnosis, treatment, and medical support. Therefore, it is essential to understand the cultural and social aspects of the Polish population, which differ from those of other EU or non-EU countries. Despite a substantial body of research conducted in European Union (EU) countries, parental beliefs about ADHD and its treatment vary considerably due to social, cultural, and ethnic factors [13,20]. However, studies examining parents’ beliefs and perspectives in Poland remain limited. This gap in the literature provided the rationale for conducting the present survey among Polish parents, focusing on factors influencing their decision-making regarding ADHD treatment in children. Moreover, previous research has primarily focused on the efficacy and safety of ADHD pharmacotherapy, while psychosocial determinants of parental decision-making have received less attention [13]. Understanding parental beliefs and attitudes may help to identify barriers and facilitators of treatment acceptance, particularly in countries in Central and Eastern Europe, such as Poland. This study examined parental attitudes toward ADHD pharmacotherapy in relation to parental experience of the child’s pharmacotherapy and explored whether parental gender was associated with differences in these beliefs and attitudes.
The analyses focused on the following aims:
- To examine how parents’ beliefs and safety-related concerns regarding ADHD pharmacotherapy relate to treatment initiation and parental acceptance decisions.
- To identify factors, including parental experience of the child’s pharmacotherapy, associated with differences in parental beliefs regarding ADHD pharmacotherapy.
- To examine whether parental gender is associated with differences in beliefs and attitudes relevant to parental treatment acceptance.
2. Materials and Methods
2.1. Study Design
This cross-sectional online survey was descriptive, comparative, and exploratory. It was conducted using an anonymous online questionnaire. Due to the naturalistic and descriptive character of the project, no prior research hypothesis was formulated, and analyses were conducted using an exploratory framework. Data collection was carried out between October 2024 and March 2025. The STROBE guidelines were followed while reporting and writing this cross-sectional study [21].
2.2. Participants
All participants were parents or legal guardians of at least one child diagnosed with ADHD. The sampling procedure was purposive. Participants were recruited exclusively through closed thematic Facebook groups focused on ADHD-related topics, primarily targeting parents of children with a confirmed diagnosis of this disorder. These participants were mainly peer network support groups, as parents and legal guardians could share experiences, consult other members, and provide information regarding coping with ADHD. These groups were not aimed at a particular advocacy or biomedical point of view but were instead places to address numerous treatment modalities, including both pharmacotherapy and psychotherapy. Thus, the recruitment strategy enabled effective access to the intended target population.
2.3. Eligibility Criteria
Inclusion criteria:
- Parents or legal guardians of children with confirmed diagnoses of ADHD;
- Parents/legal guardians residing in Poland;
- Parents who provided electronic informed consent were allowed to proceed to complete an anonymous online survey.
Exclusion criteria:
- Parents or legal guardians who did not provide a confirmed diagnosis of ADHD;
- Participants who provide incomplete responses to key sections of the questionnaire.
2.4. Procedure
The survey was conducted online in Poland using the professional research platform Ariadna. To ensure data integrity and that only eligible participants completed the survey, the following response control strategies were applied: Duplicate submissions by participants were avoided using standard, automated, platform-level technical controls. These independent working controls identified participants without collecting personal identifiers, ensuring anonymity. Response validation was ensured by including only complete responses with unique identifiers. A complete survey response was ensured by only allowing participants to move on to the next section of questions when the previous section was completed. This reduced inconsistency and minimized incomplete responses. The questionnaire link was distributed solely within closed Facebook groups and was not publicly available for access control. Participation in this study was entirely voluntary. The questionnaire was fully anonymous—no personal or identifying data (such as names, email addresses, or IP addresses) were collected to ensure data security. Before beginning the survey, respondents received detailed information about its purpose, along with assurances regarding confidentiality and data security. Consent to participate was obtained electronically by selecting an option confirming that the participant had read the information and agreed to the terms of participation. Only after providing consent could respondents proceed to the main part of the questionnaire. Due to the anonymous nature of the study and the impossibility of identifying participants, the project did not require approval from a bioethics committee.
Due to the anonymous nature of the study and the impossibility of identifying participants, the project did not require approval from a bioethics committee. The study was conducted in accordance with current national and institutional regulations as well as the principles of the Declaration of Helsinki.
2.5. Questionnaire
The questionnaire was developed by the authors with the consultation of experts and by reviewing the peer-reviewed literature and tailored to the objectives of this study. It comprised three sections, each addressing a distinct thematic area. However, no incentive was provided to the participants to enroll in this cross-sectional anonymous online survey study. The first (sociodemographic) section included items on respondents’ basic characteristics (gender, age, marital status, place of residence, education, employment status, and number of children) and their relationship to the child diagnosed with ADHD. Additional questions concerned the child’s characteristics (gender, age, education level, and employment status if applicable) and their age at ADHD diagnosis. The second (beliefs and attitudes) section, administered to all participants, covered the perceived safety of ADHD pharmacotherapy (mental and physical health), concerns about adverse effects (including the risk of addiction), perceived effectiveness, and readiness to initiate pharmacotherapy after diagnosis. Importantly, two distinct items referred to pharmacotherapy. The first asked whether the child had ever taken or was currently taking ADHD pharmacotherapy (“Has your child ever taken or currently taken pharmacotherapy for ADHD?”); responses to this item were used to define the comparison groups in the analysis (ever vs. never). The second was a multiple-choice question that asked which treatment methods had been used so far in the child’s ADHD management (e.g., psychotherapy, pharmacotherapy, combined, or other). Multiple responses were allowed, and categories were not mutually exclusive. The third (treatment history among pharmacotherapy-exposed families) section was intended for respondents whose children were currently taking or had previously taken ADHD pharmacotherapy. It included questions on the name, dose, and frequency of the administration of pharmacotherapy and parents’ subjective evaluation of treatment outcomes across cognitive, academic, emotional, social, and family domains, as well as items on the parental supervision of pharmacotherapy and discussions with the child about treatment.
Previous Pharmacotherapy Experience Operationalization:
Previous experience with ADHD pharmacotherapy was one of the variables used in this study and it was applied as follows: Current vs. Past Users: The participants were requested to answer a question on whether or not their child had ever taken any or was currently taking any ADHD pharmacotherapy. The answer choices were yes (to mean that one is currently being or has been used) or no (to mean that they have never experienced pharmacotherapy use). According to these answers, the participants were divided into two sets: Group A: Parents that had used pharmacotherapy (both current and former users); Group B: Parents who had not utilized pharmacotherapy in their children. For respondents whose children had current or prior experience with pharmacotherapy, additional information was collected regarding the type of medication (stimulant or non- stimulant), the frequency of administration (e.g., number of days per week), and parents’ subjective evaluation of treatment effects across cognitive, academic, emotional, social, and family domains. Although information on medication type was collected, subgroup analyses comparing stimulant and non-stimulant treatments were not conducted due to the small size of the non-stimulant subgroup, which limited statistical power. These data were used to characterize treatment experience descriptively. Treatment duration was not assessed as a separate variable, as the primary aim of this study was to examine parental beliefs and attitudes in relation to the presence versus absence of pharmacotherapy experience, rather than to model treatment trajectories or time-dependent effects.
Operational Definition of Treatment Acceptance in this Study
In this study, treatment acceptance was operationalized as parental willingness to initiate or consider pharmacotherapy for their child, based on self-reported attitudes and decisions, rather than objective measures of medication-taking behavior (e.g., dose compliance or persistence). This operationalization reflects the central role of parents or legal guardians in treatment decision-making for children with ADHD and is consistent with the cross-sectional, exploratory design of this study. Most items were closed-ended (Yes/No). Several open-ended items were included in the third section (e.g., medication name and dose). Although no standardized psychometric scales were employed, the instrument enabled a detailed assessment of the cognitive–emotional aspects of parental attitudes toward ADHD pharmacotherapy. The survey explicitly instructed that the third section should be completed only by respondents whose child had prior or current experience with ADHD pharmacotherapy. Furthermore, the internal consistency of the self-designed questionnaire was evaluated using Cronbach’s alpha. The overall instrument demonstrated high reliability, with a Cronbach alpha coefficient of 0.88, indicating strong internal consistency among key attitude-related items, including perceived safety, perceived effectiveness, necessity of pharmacotherapy, readiness to initiate treatment, and restrictive or cautious beliefs regarding medication use.
2.6. Statistical Analysis
We first conducted descriptive statistics for all variables. Continuous variables are presented with the number of observations, mean, median, standard deviation (SD), first and third quartiles (Q1, Q3), and range (minimum–maximum). We used box plots to visualize distributions and check for skewness or outliers (with medians, means, and individual outlier points indicated). Categorical variables are summarized as frequencies and percentages. Between-group differences (e.g., Group A vs. Group B, mothers vs. fathers) were examined using non-parametric tests since many variables were not normally distributed (Shapiro–Wilk tests indicated non-normality). For comparisons of continuous or ordinal data between two independent groups, we used the Mann–Whitney U test. For categorical data, we used the chi-square test; if expected cell counts were small, Fisher’s exact test was applied. When a difference was statistically significant, we reported Cramér’s V as a measure of effect size for categorical comparisons. All statistical tests were two-tailed and used a significance level of α = 0.05. In addition to bivariate analyses, we performed a multivariable linear regression to identify independent predictors of parental acceptance attitudes. The dependent variable was a composite acceptance score constructed from key attitude items (with higher scores reflecting more accepting/favorable attitudes toward pharmacotherapy). The primary predictors entered into the model were parental gender (mother vs. father) and prior pharmacotherapy experience (child ever medicated vs. never medicated). We also included parents’ age and child’s age (operationalized as year of birth) as covariates to adjust for potential confounding by generational or life-stage differences, and we adjusted for child’s gender as well. By including these covariates, we aimed to estimate the independent association of parental gender and medication experience with the acceptance attitude score. Regression coefficients (β) with standard errors, standardized betas, and 95% confidence intervals were calculated. All statistical analyses were conducted using R software (version 4.4).
3. Results
3.1. Sample Characteristics
This study included 506 parents of children diagnosed with ADHD (53.8% mothers, 43.1% fathers, and 3.2% legal guardians). The largest age groups were 35–44 years (32.6%) and 45–54 years (24.7%). Participants represented various regions of Poland, with the highest proportions being from the Mazowieckie (22.7%) and S’laskie (11.5%) provinces. Most respondents had one child (39.7%) or two children (41.1%). The mean age at ADHD diagnosis was 8.36 years (SD = 5.67; median = 7 years). When asked what methods were used in their child’s ADHD treatment, the following results were reported:
- A total of 56.9% of parents indicated psychotherapy use;
- A total of 30.0% reported pharmacotherapy use;
- A total of 22.9% stated that their child received both psychotherapy and pharmacotherapy.
In a separate question regarding whether they had ever used pharmacotherapy in ADHD treatment, 53.7% of parents confirmed that their child had at some point received pharmacological treatment for ADHD, whereas 46.3% reported no previous pharmacotherapy use. These results are consistent, as the first question allowed for multiple selections, while the second referred specifically to any history of pharmacotherapy. In terms of parental perceptions, 63.2% indicated that they regarded ADHD pharmacotherapy as safe for mental health, 61.7% as safe for physical health, and 65.4% believed it could exert a positive long-term influence. Conversely, 28.1% questioned the necessity of pharmacotherapy, and 20.0% expressed disbelief in ADHD as a genuine disorder (Table 1).

Table 1.
Demographic characteristics of the participants.
3.2. Reliability Analysis of the Questionnaire
The self-designed questionnaire demonstrated good internal consistency, with an overall Cronbach’s alpha of 0.88. Reliability coefficients for individual domains ranged from 0.75 to 0.86, indicating acceptable to good internal consistency across all thematic areas. These findings affirm the reliability of the instrument for conducting exploratory assessment of parental attitudes toward ADHD pharmacotherapy (Table 2).

Table 2.
The internal consistency reliability of the self-designed questionnaire.
3.3. Pharmacotherapy Experience and Parental Attitudes
Following data collection, an exploratory comparative analysis was conducted to examine whether prior experience with ADHD pharmacotherapy was associated with differences in parental beliefs and attitudes. The analysis was not based on a pre-specified hypothesis but emerged from the descriptive phase of this study, aimed at exploring potential patterns within the data. To examine whether experience with ADHD pharmacotherapy is associated with parental beliefs, respondents were divided into two groups:
- Group A: parents whose children had at some point received ADHD pharmacotherapy (n = 271; 53.7%);
- Group B: parents whose children had never received ADHD pharmacotherapy (n = 234; 46.3%).
One respondent had missing data for the ‘Child has taken/is taking medication’ item; therefore, group comparisons are based on N = 505. Parents with pharmacotherapy experience demonstrated significantly more positive attitudes toward treatment than those without such experience. The proportion of parents of the medicated group who viewed ADHD pharmacotherapy as safe in regard to mental health (83.4% vs. 39.7%; p < 0.001; V = 0.451) and physical health (79.0% vs. 41.5%; p < 0.001; V = 0.384) was far higher, and more parents from the medicated group thought that pharmacotherapy could have a positive impact on the child (80.4% vs. 47.9%; p < 0.001). Parents in the medicated group were also more willing to allow their child to start pharmacotherapy as soon as they received a diagnosis (73.8% vs. 32.5%; p < 0.001; V = 0.414) and were likely to recommend that their child visit a psychiatrist to consider pharmacotherapy (72.3% vs. 43.2%; p < 0.001; V = 0.295). Equally, a higher percentage of these parents were of the view that all the diagnosed children should be treated with ADHD pharmacotherapy (56.1% vs. 21.8; p < 0.001; V = 0.348) and that the advantages of pharmacotherapy are greater than the risks. However, fears about the addictive nature of the treatment for ADHD patients were similar in both groups (49.4% vs. 45.3; p = 0.400; V = 0.041), and even parents who had positive attitudes towards the treatment did not overlook the possible negative effects. Other variables, such as a willingness to consent despite the recommendation of the doctor, use of pharmacotherapy only in patients older than 18, and willingness to accept ADHD treatment without pharmacotherapy, had smaller or non-significant differences between groups (p > 0.05; V < 0.1). On the whole, such results indicate that parental experience with pharmacotherapy is strongly associated with more positive beliefs or attitudes toward pharmacotherapy and greater readiness to think about using pharmacological treatment for their children (Table 3).

Table 3.
Parental responses to statements regarding ADHD pharmacotherapy stratified by child pharmacotherapy status.
3.4. Parents’ Beliefs of Pharmacotherapy Effectiveness
To provide a more detailed understanding of parental beliefs regarding pharmacological treatment, an additional analysis was conducted within the subgroup of parents whose children had received pharmacotherapy for ADHD. An exploratory study assessed whether parental gender was related to attitudes to ADHD pharmacotherapy differences. It analyzed mothers and fathers. On the whole, mothers exhibited slightly more positive attitudes to ADHD pharmacotherapy in multiple areas, but only some of them were statistically significant. A larger percentage of mothers, compared to fathers, thought that ADHD drug treatment may benefit the future of their child (69.9% vs. 61%; p = 0.050; V = 0.093). In contrast, fathers, compared to mothers, were more willing to support perspectives that doubted the necessity of pharmacological treatment for ADHD (35.3–22.4%; p = 0.002; V = 0.142) or disbelieved in ADHD (25.7–15.1%; p = 0.005; V = 0.132). Moreover, fathers, compared to mothers, were more inclined to state that the use of ADHD pharmacotherapy should be restricted to people above 18 (41.3% vs. 26.5%; p < 0.001; V = 0.156) and that they would not grant consent even if doctor had prescribed it (38.1% vs. 26.1%; p = 0.006; V = 0.128). Equally, fathers reported more cases when the child did not take pharmacotherapy due to either a lack of parental consent (35.8% vs. 21.7%; p < 0.001; V = 0.156) or parental opposition (28% vs. 8.1%; p < 0.001; V = 0.264).
Beliefs about pharmacotherapy’s safety for mental or physical health, readiness to initiate pharmacotherapy on diagnosis, or consent for ADHD treatment in the absence of pharmacotherapy did not show any significant differences between mothers and fathers (p > 0.05). Overall, these findings indicate that parental gender is associated with differences in attitudes toward ADHD pharmacotherapy, particularly regarding the perceived necessity of treatment, consent, and age-related restrictions on use, with fathers generally expressing more restrictive or cautious views than mothers (Table 4).

Table 4.
The proportion of parents responding Yes and No to statements regarding ADHD pharmacotherapy by parental gender (Mother N = 272; Father N = 218). EF stands for Effect size, * indicates p < 0.05, ** p < 0.001.
Figure 1 shows that the vast majority of parents observed a positive effect from pharmacotherapy on their child’s everyday functioning. A total of 79.7% of respondents agreed that pharmacological treatment improved their child’s academic performance, and 75.9% reported that pharmacotherapy increased the amount of time their child spent studying. Additionally, 74.7% of parents stated that their child received fewer negative remarks from teachers or lecturers while on pharmacotherapy, and 75.1% agreed that tasks usually performed with ease became significantly more difficult without pharmacotherapy.
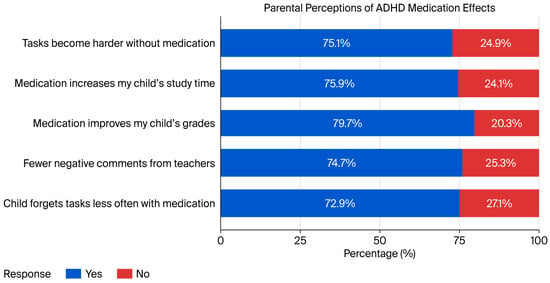
Figure 1.
Parental beliefs regarding the effects of ADHD pharmacotherapy.
Furthermore, 72.9% of respondents observed that their child forgot responsibilities less frequently since starting pharmacotherapy. Overall, these findings suggest that parents perceive pharmacological treatment for ADHD as substantially improving their child’s academic and behavioral functioning, as well as their ability to manage everyday responsibilities.
3.5. The Child Takes Pharmacotherapy Despite My Lack of Consent
To further explore the complexity of parental attitudes toward ADHD pharmacotherapy, this study also examined situations in which children received pharmacotherapy despite a lack of consent from their parent or caregiver. Among parents whose children had received pharmacotherapy for ADHD, 24.7% (n = 67) reported having experienced a situation in which their child took prescribed ADHD pharmacotherapy despite the parent’s lack of consent. These findings suggest intra-family disagreements regarding the appropriateness of ADHD pharmacotherapy. The majority of parents (75.3%; n = 204) did not report such incidents, indicating that, in most cases, the initiation and continuation of pharmacotherapy were made collaboratively within families (Table 5).

Table 5.
The proportion of parents responding ‘Yes’ and ‘No’ to the statement regarding the child taking pharmacotherapy despite lack of consent.
3.6. Multivariable Analysis of Parental Acceptance of ADHD Pharmacotherapy
The multivariable linear regression analysis was performed to identify factors related to parental acceptance of ADHD pharmacotherapy, using the composite acceptance score as the dependent variable. Parental gender and prior experience of ADHD pharmacotherapy were the main predictors of interest. Parental age (in years) and child year of birth (used as a proxy of child age) were taken as covariates in the model to explain the possibility of confounding. The regression model also included the child’s gender as an adjustment variable, allowing estimation of the independent association between parental gender and the acceptance score while adjusting for the child’s gender. Parental gender was also found to be a significant predictor of acceptance after all covariates were adjusted for, with mothers showing much lower acceptance scores than fathers (B = −0.403, SE = 0.096, β = −0.166, p < 0.001; 95% CI: −0.592 to −0.215).
Conversely, past experience with ADHD pharmacotherapy was positively correlated with parental acceptance, with a higher level of acceptance among parents whose child already took medicine (B = 0.799, SE = 0.095, β = 0.331, p < 0.001; 95% CI: 0.612 to 0.986). The age of parents (B =−0.044, p = 0.713) and the year of birth of the child (B = 0.0001, p = 0.994) did not show statistically significant association with acceptance scores after adjustment. On the whole, these results indicate that gender-specific attitudes and previous experience of treatment have a stronger association with parental attitude towards pharmacotherapy in ADHD management than demographic factors (Table 6).

Table 6.
Regression Analysis of Parental and Child Factors on Treatment Acceptance.
Although item-level comparisons (Table 4) indicated that fathers were more likely to express restrictive attitudes on specific dimensions—such as consent or perceived necessity—the composite acceptance score reflects a broader construct. Fathers may have evaluated other components more positively, such as treatment safety or long-term effectiveness, which contributed to a higher overall score.
4. Discussion
Our study observed an association between prior exposure to ADHD pharmacotherapy and more positive parental attitudes toward treatment, though the evidence is limited to exploratory due to convenience sampling and self-report nature of the data.
The most pronounced differences were observed in beliefs about the safety of pharmacotherapy and parents’ readiness to initiate therapy immediately after diagnosis, particularly among parents who are actively engaged and have positive attitude toward treatment acceptance-related decision-making. At the same time, concerns regarding addiction persisted at similar levels in both groups of parents.
Hence, parental beliefs about the safety of pharmacotherapy seem to play an important role in treatment-related decision-making, even among parents who otherwise report positive attitudes toward pharmacotherapy. Future researchers must explore the influence of these beliefs in broader populations (including lower literature or non-socially active parents) to diversify the evidence. Moreover, these findings can be interpreted in light of the TPB framework introduced earlier, which emphasizes the role of attitudes and perceived social norms in shaping treatment-related intentions and behaviors. Within this framework, parental attitudes are conceptualized as dynamic constructs that may evolve over time in response to experience, information, and social context. The association observed between parental beliefs and treatment-related decisions is consistent with TPB framework. Thus, it should be cautiously interpreted given the limitations of the study. The observed discrepancy between item-level and multivariable findings may initially seem counterintuitive. While fathers expressed more skepticism on specific questions, such as consent and treatment necessity, they also rated other dimensions—like perceived safety and effectiveness—more favorably. These positive assessments likely contributed to their higher overall acceptance score in the regression model. This highlights the importance of understanding composite constructs and how divergent item-level responses can coexist within an aggregated measure.
Our study’s findings are also in line with those of other international studies on the significance of parental attitudes in the initiation and maintenance of ADHD pharmacotherapy. Charach and Fernandez observed that caregivers’ willingness to continue ADHD pharmacotherapy is closely linked to their beliefs regarding treatment effectiveness and perceived improvements in their child’s behavior and functioning [22]. Meta-analyses conducted by Faraone et al. (2006) [23] confirmed the high efficacy of psychostimulant pharmacotherapy and indicated that the perceived benefits of therapy are a key factor shaping positive parental attitudes. Previous research has focused primarily on the efficacy and safety of ADHD pharmacotherapy [23], while parental attitudes and beliefs have received considerably less attention. However, it is precisely the parents who decide whether the child begins treatment, and their acceptance is, therefore, a necessary condition for the application of therapy. As an example, the Multimodal Treatment Study of ADHD (MTA) proved that parents who saw positive changes in the functioning of their child would be more inclined to continue the treatment and experience greater satisfaction with ADHD medication [24].
4.1. Parental Concerns: Addiction and Perceived Risks
A very intriguing conclusion of this research is that the issue of addiction and permanent damage is still relevant despite the fact that some parents who experienced the beneficial outcomes of pharmacotherapy for ADHD have been observed. Such long-lasting concerns may be explained by the risk perception theory, which explains that people tend to exaggerate risks when it comes to dealing with unknown or stigmatized interventions, including psychotropic drugs in children [25,26]. However, the generalizability of these concerns may vary depending on the social and cultural context. Additionally, skepticism at the pharmacotherapy stage could be viewed from the perspective of a lack of trust in the pharmaceutical industries, especially in Eastern European nations. Studies have shown that disbelief in psychotropic medications due to concerns related to addiction and side effects may also shape the psychological mindset of parents in certain societies, including Poland and other Eastern European countries [27].
Moreover, the studies from Central Europe and East Asia that we analyzed indicate that parental worries regarding the safety of drugs, such as concerns about addiction and long-term damage, are still common, even in parents who have positive experiences with medication [28]. Our results agree with these studies, where parental suspicion regarding the efficacy of pharmacotherapy in the long term prevails in light of apparent positive outcomes. Nevertheless, our study also reported that fear of addiction was not significantly different between the medicated and non-medicated groups, and this is in line with a study that reported adverse effects and stigma as the most prevalent reasons for discontinuing pharmacotherapy in child patients with ADHD [29].
4.2. Gender Differences in Parental Attitudes
Another difference also evident in our study was that between mothers and fathers in their attitude towards ADHD medication. Mothers viewed ADHD medication as valuable for the future of the child (69.9% vs. 61%), whereas fathers showed doubt about the need to use pharmacological treatment (35.3% vs. 22.4%). Such results are consistent with the results of research that found that mothers more frequently worry about medication safety, while fathers prefer to emphasize functional outcomes and less often ask questions regarding the need for medication [15,18].
This gendered decision-making approach can be seen as informed by the traditional gendered roles of caregiving, where mothers are often expected to be more directly involved in the health management of their children. This tendency can be traced in the global context, as mothers tend to have many more questions about side effects or addiction, and parents tend to be more in favor of using drugs as treatment [26,29].
4.3. Cultural Influences on Parental Attitudes
Based on international studies, this study also emphasized that the attitudes of parents towards ADHD and its treatment largely depend on cultural background. In Eastern Europe, parental beliefs regarding ADHD are usually influenced by skepticism and stigma towards psychiatric drugs, though these attitudes may differ based on the local culture of the society or region. All these elements of society make parents mistrustful of using pharmacotherapy for ADHD [24]. This argument is supported by our study because a great number of parents were unsure of whether pharmacological treatment was required or whether ADHD was a valid disorder. The same issues with non-medical interventions, including dietary changes or intensive physical exercise, were also observed in other countries, especially in those in which ADHD diagnosis and treatment are less culturally acceptable [30].
Parents in our study most often reported clear improvements in their children’s academic performance, study engagement, and daily functioning after starting pharmacotherapy. They also noticed fewer negative remarks from teachers and better task completion, indicating that the effects of pharmacotherapy are perceived as meaningful and observable in everyday life. The influence of caregivers’ beliefs and attitudes is further supported by findings from broader psychiatric research. The effectiveness of pharmacotherapy has been shown to depend not only on its pharmacodynamic properties but also on the patient’s subjective interpretation of treatment, including accompanying beliefs and the therapeutic relationship [31]. The literature emphasized that psychological and relational factors co- determine psychopharmacological outcomes [32]. This perspective has been reaffirmed in recent commentaries on ADHD treatment guidelines for adults, which underscore the relevance of individual experiences and belief systems in shaping treatment acceptance [33].
Furthermore, a study conducted in Saudi Arabia demonstrated that parental beliefs play a significant role in initiating ADHD pharmacotherapy in children [34]. Similarly, in ADHD, parental concerns, such as the fear of addiction, may influence treatment acceptance of therapeutic recommendations regardless of the actual effectiveness of pharmacotherapy. Indeed, a systematic review reported that adverse effects and social stigma are among the most common reasons for discontinuing ADHD pharmacotherapy, underscoring how negative experiences can hinder treatment acceptance [35]. In some families, treatment decisions may also involve internal disagreement or conflicting views between caregivers, indicating potential decisional conflict between families regarding treatment approach. This reflects the complexity of decision-making within families and highlights the need for transparent, collaborative communication between clinicians and caregivers.
As also shown in studies conducted in other countries, in our sample, some parents questioned the legitimacy of pharmacotherapy or even the existence of ADHD as a clinical entity. International research indicates that a substantial proportion of parents attribute ADHD symptoms to inadequate upbringing rather than a neurodevelopmental disorder and seek alternative treatment methods, such as dietary changes or intensive physical exercise [35]. The media represent one of the main sources of information about ADHD, which may both facilitate and hinder the treatment process—in some cases, media messages even discourage parents from administering pharmacotherapy [35].
4.4. Psychoeducation, Clinical Implications, and Future Directions
The findings of this research highlight that psychoeducational interventions targeting parental beliefs about ADHD pharmacotherapy may be beneficial. The previously conducted studies have also indicated that providing evidence-based information on ADHD and its treatment may help to address parents’ concerns and is associated with improvement in treatment acceptance [29,36].
Researchers encourage incorporating psychoeducation into clinical practice, particularly education provided by various professionals (e.g., child psychiatrists, pediatricians, school psychologists, ADHD nurses) involved in the treatment pathway. Psychoeducation may also be relevant at key stages of care, such as diagnostic evaluation, initiation of treatment, and follow-up appointments, when parental beliefs and concerns are likely to affect treatment decisions.
Moreover, psychoeducation may be delivered through various methods, such as one-on-one meetings, group sessions, and online and digital tools, which may ensure accessibility and address parental needs [37]. Hence, this study is an exploratory cross- sectional online survey; these findings are presented as important clinical implications rather than formal recommendations. However, these findings underscore the need for more diverse, population-based longitudinal studies to replicate and extend these findings. Future studies should employ multivariable designs to disentangle the independent effects of parental gender, treatment experience, and clinical severity.
This study has important clinical implications. The results suggest that parental acceptance is a prerequisite for the effective application of ADHD treatment. It is ultimately the parents’ beliefs that strongly influence whether a child begins pharmacotherapy or does not do so despite medical indications. In practice, this suggests that working with parents may be an important component of ADHD therapy, and psychoeducation could serve as a valuable component of the diagnostic and therapeutic process. Reducing unfounded fears, correcting misconceptions unsupported by scientific evidence, and building trust in pharmacological treatment are not merely complementary interventions but constitute key factors determining therapeutic effectiveness [7,10,18].
4.5. Limitations of the Study
This study has several limitations that should be considered when interpreting the findings. Importantly, key clinical context variables—such as ADHD symptom severity, level of functional impairment, time since diagnosis, and the strength of clinician treatment recommendations—were not assessed. Consequently, differences observed between parents of medicated and non-medicated children may partly reflect underlying differences in the child’s clinical status (confounding or reverse causation), rather than the effect of pharmacotherapy experience itself. Although a multivariable model was employed to adjust for selected demographic factors, we were unable to account for these potentially important confounders due to the lack of corresponding data.
Given the exploratory nature of the study, no formal correction for multiple comparisons was applied, which increases the risk of Type I errors and underscores the need for cautious interpretation of statistically significant results. The cross-sectional design precludes causal inference, and all data were based on parental self-report using a self- developed questionnaire that lacked formal psychometric validation, which may introduce reporting bias.
Recruitment via closed social media groups and the use of purposive sampling may have introduced selection bias and limited generalizability, as the sample may overrepresent parents who are more actively engaged in their child’s ADHD care and underrepresent parents who are less engaged or have limited access to online communities. No quotas or stratification were applied during recruitment; participation was entirely self-selected. Additionally, reports of pharmacotherapy use despite a lack of parental consent may reflect misclassification or differing interpretations of questionnaire items, highlighting potential decisional conflict within families rather than inappropriate clinical or legal practices.
Finally, the study did not collect detailed contextual information related to parental decision-making dynamics, such as legal guardianship arrangements, shared parental responsibility, or the child’s level of assent. Although data on medication type were collected, subgroup analyses comparing stimulant and non-stimulant treatments were not conducted due to insufficient sample size in the non-stimulant group.
Longitudinal studies are needed to assess how parents’ attitudes evolve over the course of treatment, and interventional studies are needed to evaluate the effectiveness of psychoeducational programs aimed at reducing barriers to pharmacotherapy. Future researchers are also encouraged to focus on underrepresented populations (e.g., those of lower socioeconomic status or less actively involved in ADHD treatment decisions) to enhance the generalizability of future findings.
5. Conclusions
Parents of children with prior experience of ADHD pharmacotherapy exhibited more positive attitudes toward treatment, but concerns about the risk of addiction remained common in both groups. These findings highlight that positive treatment experience does not necessarily eliminate safety-related worries, particularly those related to addiction, which appear to persist and may be influenced by cultural factors.
Psychoeducation, open communication, and a collaborative partnership with parents may support treatment acceptance-related decision-making regarding therapeutic recommendations. Parental attitudes and beliefs play an important role in the treatment process, in line with current scientific evidence and international guidelines, and may influence how pharmacotherapy is accepted and initiated in children with ADHD.
Overall, the findings underscore the importance of addressing parental beliefs and concerns as part of routine ADHD care. Interventions aimed at improving shared decision- making and providing evidence-based information tailored to parental concerns may help to reduce barriers to pharmacotherapy initiation and continuation, while still respecting the exploratory and cross-sectional nature of this study.
Author Contributions
Conceptualization, K.J. and S.M.; methodology, K.J.; formal analysis, K.J.; investigation, K.J.; resources, K.J. and S.M.; data curation, K.J.; writing—original draft preparation, K.J.; writing—review and editing, S.M.; supervision, S.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki. Ethical review and approval were waived for this study due to its anonymous, non-interventional design and the absence of identifiable personal data.
Informed Consent Statement
Informed consent was obtained electronically from all subjects involved in this study.
Data Availability Statement
Due to privacy restrictions, the dataset generated and analyzed during this study is not publicly available but can be obtained from the authors upon reasonable request in anonymized form.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ADHD | Attention-Deficit/hyperactivity disorder |
| CAPPA | Caregiver Perspective on Paediatric ADHD |
| TPB | Theory of Planned Behavior |
| STROBE | Strengthening the Reporting of Observational Studies in Epidemiology |
References
- Wanni Arachchige Dona, S.; Badloe, N.; Sciberras, E.; Gold, L.; Coghill, D.; Le, H. The impact 633 of childhood attention-deficit/hyperactivity disorder (ADHD) on children’s health-related 634 quality of life: A systematic review and meta-analysis. J. Atten. Disord. 2023, 27, 598–611. [Google Scholar] [CrossRef]
- Barnes, G.; Wretham, A.; Sedgwick, R.; Boon, G.; Cheesman, K.; Moghraby, O. Evaluation of a diagnostic ADHD pathway in a community child mental health service in South London. Ment. Health Rev. J. 2020, 25, 1–19. [Google Scholar] [CrossRef]
- Danielson, M.; Claussen, A.; Bitsko, R.; Katz, S.; Newsome, K.; Blumberg, S.; Kogan, M.; Ghandour, R. ADHD prevalence among US children and adolescents in 2022: Diagnosis, severity, co-occurring disorders, and treatment. J. Clin. Child Adolesc. Psychol. 2024, 53, 343–360. [Google Scholar] [CrossRef]
- Szaniawska, M.; Kokoszka, A. Prevalence of ADHD Among Nonpsychotic Patients of Day-Care Centers and Comparison of Psychiatric Comorbidities Among Persons with and Without ADHD: A Pilot Study. J. Clin. Med. 2025, 14, 1153. [Google Scholar] [CrossRef]
- Groom, M.; Cortese, S. Current Pharmacological Treatments for ADHD; Springer: Berlin, Germany, 2022; pp. 19–50. [Google Scholar] [CrossRef]
- O’nions, E.; El Baou, C.; John, A.; Lewer, D.; Mandy, W.; McKechnie, D.; Petersen, I.; Stott, J. Life expectancy and years of life lost for adults with diagnosed ADHD in the UK: Matched cohort study. Br. J. Psychiatry 2025, 226, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Van Vyve, L.; Dierckx, B.; Lim, C.; Danckaerts, M.; Koch, B.; Häge, A.; Banaschewski, T. Pharmacotherapy for ADHD in children and adolescents: A summary and overview of different European guidelines. Eur. J. Pediatr. 2024, 183, 1047–1056. [Google Scholar] [CrossRef]
- Catalá-López, F.; Hutton, B.; Núñez-Beltrán, A.; Mayhew, A.; Page, M.; Ridao, M.; Tobías, A.; Catalá, M.; Tabarés-Seisdedos, R.; Moher, D. The pharmacological and non-pharmacological treatment of attention deficit hyperactivity disorder in children and adolescents: Protocol for a systematic review and network meta-analysis of randomized controlled trials. Syst. Rev. 2015, 4, 19. [Google Scholar] [CrossRef]
- Sibley, M.; Flores, S.; Murphy, M.; Basu, H.; Stein, M.; Evans, S.; Zhao, X.; Manzano, M.; van Dreel, S. Research Review: Pharmacological and non-pharmacological treatments for adolescents with attention deficit/hyperactivity disorder–a systematic review of the literature. J. Child Psychol. Psychiatry 2025, 66, 132–149. [Google Scholar] [CrossRef] [PubMed]
- Ogundele, M.; Ayyash, H. ADHD in children and adolescents: Review of current practice of non-pharmacological and behavioural management. AIMS Public Health 2023, 10, 35. [Google Scholar] [CrossRef]
- Gondek, T.; Stramecki, F.; Cie’sla, M.; Ziegart-Sadowska, K.; Foryciarz, K.; Główczy’nski, P.; Krupa, A.; Krysta, K.; Malec, A.; Rewekant, A. Assessment and management of adults with ADHD. Guidelines of the Specialty Training Section of the Polish Psychiatric Association and a coalition of organizations for people with ADHD–update, 2025. Psychiatr. Spersonalizowana/Pers. Psychiatry 2025, 4, 80–115. [Google Scholar] [CrossRef]
- Taylor, L.; Antshel, K. Factors associated with parental treatment attitudes and information-673 seeking behaviors for childhood ADHD. J. Atten. Disord. 2021, 25, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Jhawar, N. Parental Help-Seeking Intentions for Childhood ADHD in Asian Indian American Parents: Application of the Theory of Planned Behavior. Ph.D. Thesis, Syracuse University, Syracuse, NY, USA, 2022. [Google Scholar]
- Champ, R.; Adamou, M.; Tolchard, B. The impact of psychological theory on the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in adults: A scoping review. PLoS ONE 2021, 16, e0261247. [Google Scholar] [CrossRef]
- Chen, M.; Seipp, C.; Johnston, C. Mothers’ and fathers’ attributions and beliefs in families of girls and boys with attention-deficit/hyperactivity disorder. Child Psychiatry Hum. Dev. 2008, 39, 85–99. [Google Scholar] [CrossRef]
- Fridman, M.; Banaschewski, T.; Sikirica, V.; Quintero, J.; Erder, M.; Chen, K. Caregiver perspective on pediatric attention-deficit/hyperactivity disorder: Medication satisfaction and symptom 686 control. Neuropsychiatr. Dis. Treat. 2017, 13, 443–455. [Google Scholar] [CrossRef]
- Wright, N.; Moldavsky, M.; Schneider, J.; Chakrabarti, I.; Coates, J.; Daley, D.; Kochhar, P.; Mills, J.; Sorour, W.; Sayal, K. Practitioner review: Pathways to care for ADHD—A systematic review of barriers and facilitators. J. Child Psychol. Psychiatry 2015, 56, 598–617. [Google Scholar] [CrossRef]
- Shehadeh Sheeny, A.; Goldblatt, H.; Baron Epel, O. Mothers’ decisions regarding treating their children with attention deficit hyperactivity disorder. Eur. J. Public Health 2023, 33, ckad160.760. [Google Scholar] [CrossRef]
- Young, S.; Asherson, P.; Lloyd, T.; Absoud, M.; Arif, M.; Colley, W.; Cortese, S.; Cubbin, S.; Doyle, N.; Morua, S. Failure of healthcare provision for attention-deficit/hyperactivity disorder in the United Kingdom: A consensus statement. Front. Psychiatry 2021, 12, 649399. [Google Scholar] [CrossRef]
- Nafees, B.; Setyawan, J.; Lloyd, A.; Ali, S.; Hearn, S.; Sasane, R.; Sonuga-Barke, E.; Hodgkins, P. Parent preferences regarding stimulant therapies for ADHD: A comparison across six European countries. Eur. Child Adolesc. Psychiatry 2014, 23, 1189–1200. [Google Scholar] [CrossRef][Green Version]
- Cuschieri, S. The STROBE guidelines. Saudi J. Anaesth. 2019, 13, S31–S34. [Google Scholar] [CrossRef] [PubMed]
- Charach, A.; Fernandez, R. Enhancing ADHD medication adherence: Challenges and opportunities. Curr. Psychiatry Rep. 2013, 15, 371. [Google Scholar] [CrossRef] [PubMed]
- Faraone, S.; Biederman, J.; Spencer, T.; Aleardi, M. Comparing the efficacy of medications for ADHD using meta-analysis. Medscape Gen. Med. 2006, 8, 4. [Google Scholar]
- Hinshaw, S.; Scheffler, R.; Fulton, B.; Aase, H.; Banaschewski, T.; Cheng, W.; Mattos, P.; Holte, A.; Levy, F.; Sadeh, A. International variation in treatment procedures for ADHD: Social context and recent trends. Psychiatr. Serv. 2011, 62, 459–464. [Google Scholar] [CrossRef]
- Amiri, S.; Shafiee-Kandjani, A.; Noorazar, S.; Ivrigh, S.; Abdi, S. Knowledge and attitude of parents of children with attention deficit hyperactivity disorder towards the illness. Iran. J. Psychiatry Behav. Sci. 2016, 10, e122. [Google Scholar] [CrossRef]
- Slobodin, O. ADHD in culturally and linguistically diverse children. In Clinical Handbook of ADHDAssessment and Treatment Across the Lifespan; Springer: Berlin, Germany, 2023; pp. 1–15. [Google Scholar] [CrossRef]
- Fridman, M.; Banaschewski, T.; Sikirica, V.; Quintero, J.; Chen, K. Access to diagnosis, treatment, and supportive services among pharmacotherapy-treated children/adolescents with ADHD in Europe: Data from the Caregiver Perspective on Pediatric ADHD survey. Neuropsychiatr. Dis. Treat. 2017, 13, 947–958. [Google Scholar] [CrossRef]
- Hong, J.; Novick, D.; Treuer, T.; Montgomery, W.; Haynes, V.; Wu, S.; Haro, J. Patient characteristics associated with treatment initiation among paediatric patients with attention-deficit/hyperactivity disorder symptoms in a naturalistic setting in Central Europe and East Asia. BMC Psychiatry 2014, 14, 304. [Google Scholar] [CrossRef]
- Bussing, R.; Gary, F.; Mills, T.; Garvan, C. Cultural variations in parental health beliefs, knowledge, and information sources related to attention-deficit/hyperactivity disorder. J. Fam. Issues 2007, 28, 291–318. [Google Scholar] [CrossRef]
- Moldavsky, M.; Sayal, K. Knowledge and attitudes about attention-deficit/hyperactivity disorder (ADHD) and its treatment: The views of children, adolescents, parents, teachers and healthcare professionals. Curr. Psychiatry Rep. 2013, 15, 377. [Google Scholar] [CrossRef]
- Faraone, S.; Biederman, J.; Zimmerman, B. An analysis of patient adherence to treatment during a 1-year, open-label study of OROS® methylphenidate in children with ADHD. J. Atten. Disord. 2007, 11, 157–166. [Google Scholar] [CrossRef]
- Marrero, R.; Fumero, A.; deMiguel, A.; Penate, W. Psychologicalfactors involved in psychopharmacological medication adherence in mental health patients: A systematic review. Patient Educ. Couns. 2020, 103, 2116–2131. [Google Scholar] [CrossRef]
- Murawiec, S. Komentarz do rekomendacji: Diagnostyka i postepowanie terapeutyczne u dorosłych z ADHD. Rekomendacje Sekcji Kształcenia Specjalizacyjnego Polskiego Towarzystwa Psychiatrycznego i koalicji organizacji na rzecz osób z ADHD–2024 r. Psychiatr. Spersonalizowana/Pers. Psychiatry 2024, 3, 74–76. [Google Scholar] [CrossRef]
- Alrahili, N.; Al Harthi, M.; Ababtain, S.; Al Sharif, S.; Alnuwaysir0, M.; Al Huzaimi, R. Parental attitudes and beliefs toward attention deficit hyperactivity disorder in prince sultan military medical city, Riyadh city. Psych 2022, 4, 238–246. [Google Scholar] [CrossRef]
- Gajria, K.; Lu, M.; Sikirica, V.; Greven, P.; Zhong, Y.; Qin, P.; Xie, J. Adherence, persistence, and medication discontinuation in patients with attention-deficit/hyperactivity disorder—A systematic literature review. Neuropsychiatr. Dis. Treat. 2014, 10, 1543–1569. [Google Scholar] [CrossRef] [PubMed]
- Vitulano, L.; Mitchell, J.; Vitulano, M.; Leckman, J.; Saunders, D.; Davis, N.; Woodward, D.; Goodhue, B.; Artukoglu, B.; Kober, H. Parental perspectives on attention-deficit/hyperactivity disorder treatments for children. Clin. Child Psychol. Psychiatry 2022, 27, 1019–1032. [Google Scholar] [CrossRef]
- Dixon, J.; Akins, R.; Miller, E.; Breslau, J.; Gill, S.; Bisi, E.; Schweitzer, J. Changing parental knowledge and treatment acceptance for ADHD: A pilot study. Clin. Pediatr. 2023, 62, 301–308. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.