Abstract
Using additive manufacturing (AM) techniques like SLM and EBM provides a valuable opportunity for manufacturing biomedical devices with precise porous structures that can mitigate adverse implant complications. Gyroid sheet network structures exhibit an excellent performance among porous structures due to their bioinspired morphology and mechanical properties. This study investigates the mechanical behavior of gyroid sheet networks with different morphological parameters suitable for biomedical implants. The results show that gyroid sheet networks with 1 to 2.5 mm unit cell sizes and porosities between 50% and 85% are ideal for biomedical implants. Additionally, porous implants made of gyroid sheet networks and mentioned morphologies can be produced using SLM with a layer thickness of 30 µm, spot size of 90 µm, and powder size of around 50 µm.
1. Introduction
The stiffness mismatch between solid implants and bone tissue can result in stress shielding and cortical hypertrophy, causing various patient problems [1]. To mitigate this issue, porous structures have been suggested as a practical solution, with previous studies demonstrating their ability to reduce stress shielding significantly [2]. Porous structures can be fabricated in different morphologies, such as strut-based (e.g., BCC and FCC) [3], TPMSs (e.g., gyroid and diamond) [4], or stochastic [5]. Among the different morphologies of porous structures, TPMS structures, particularly gyroid sheet networks, have been found to exhibit a superior performance due to their bioinspired morphology and identical mechanical properties to bone tissues [6]. Therefore, they are considered viable candidates for biomedical implants. To optimize the performance of these structures, pore size and porosity are crucial parameters, with most studies suggesting that pore sizes between 300 and 800 µm and a porosity of more than 50% are suitable for enhanced osseointegration and cell ingrowth [7]. This study evaluates the mechanical properties of gyroid sheet network structures within the appropriate pore size, unit cell size, and porosity ranges for biomedical implants.
2. Materials and Methods
The methodology involves using finite element method (FEM) analysis to model gyroid sheet network structures with unit cell sizes 1, 1.5, 2, and 2.5 mm and porosities between 50 and 85% (32 models in total). Elasticity modulus (quasi-elastic gradient) and yield strength (compressive offset stress) were determined through numerical analysis based on ISO 13314 [8] testing conditions using the Johnson–Cook strength model for Ti6Al4V [9]. Compression testing was conducted by applying vertical displacements with a constant strain rate of 0.01 s−1 and measuring the corresponding reaction force and displacement to calculate the elasticity modulus and yield strength. The models were evaluated based on mesh sensitivity and number of unit cell sensitivity analyses.
3. Results and Discussion
The following results have been obtained from the analyses:
3.1. Pore Size, Unit Cell Size, and Porosity Relationship
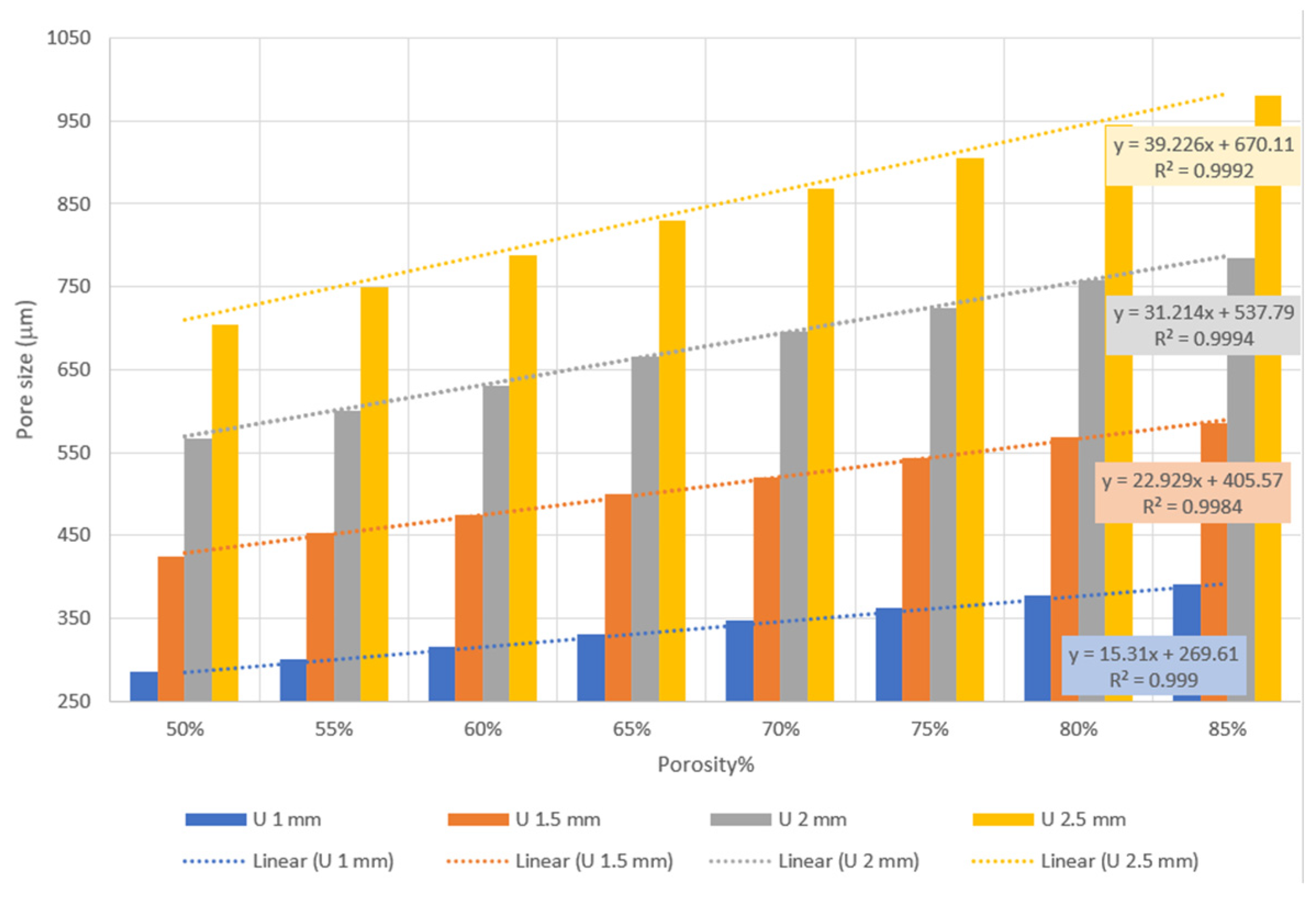
The results provided in Figure 1 reveal that pore sizes between 285 and 980 µm can be covered by altering unit cell size and porosity from 1 mm and 50% to 2.5 mm and 85%, respectively. Additionally, the results show a strong linear correlation between porosity and pore size with an R2 value near 1.
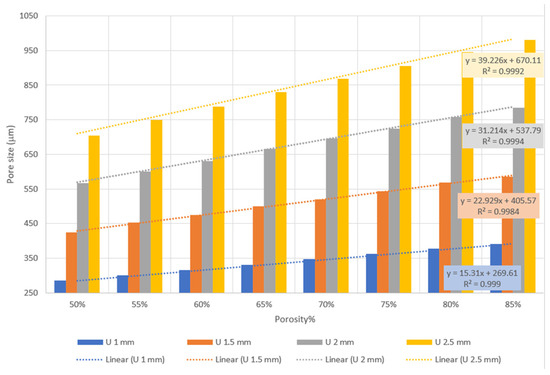
Figure 1.
Relationship between pore size, unit cell size, and porosity.
3.2. Mechanical Properties
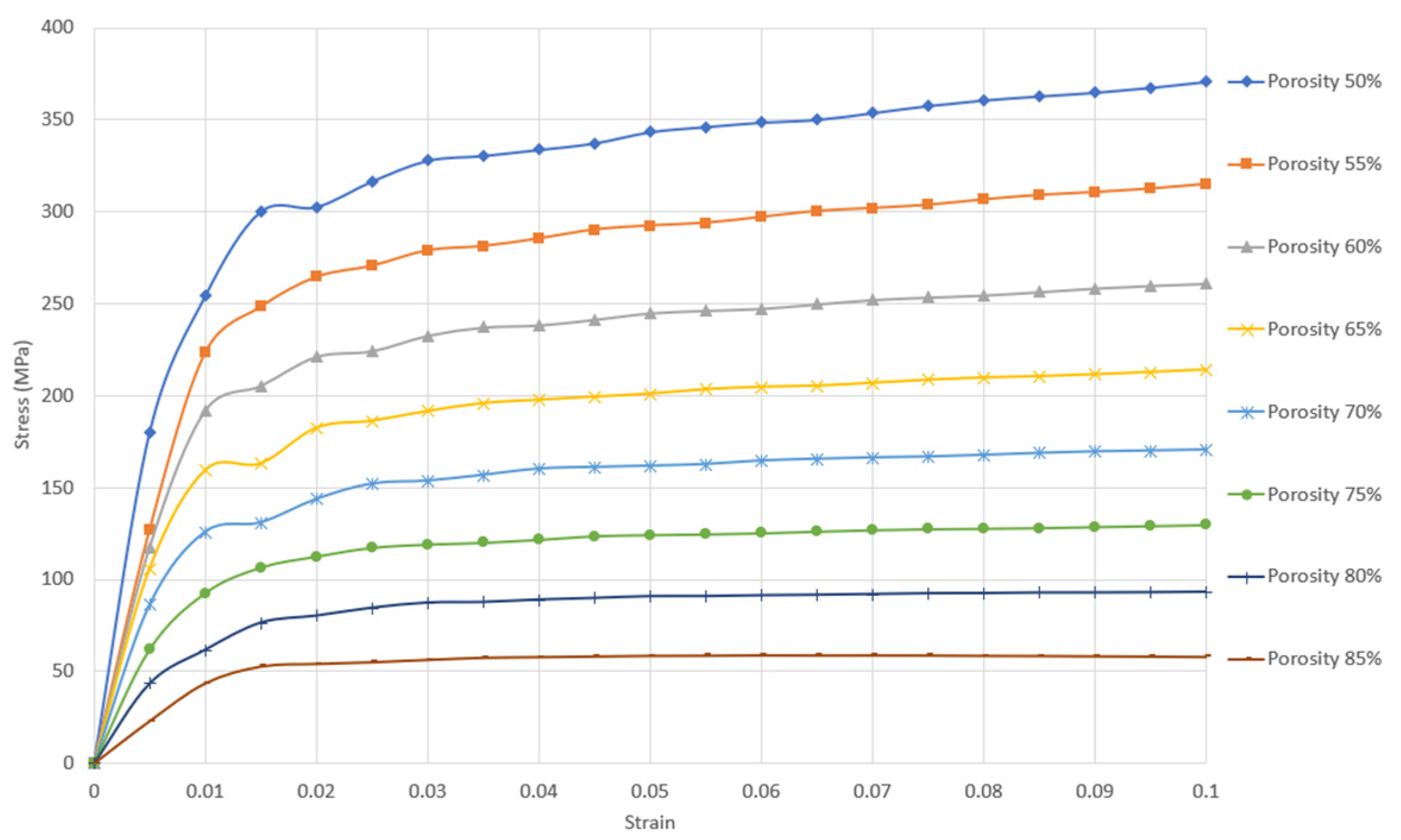
The elastic-plastic regions for a unit cell size of 1 mm are depicted in Figure 2. The quasi-elastic gradient and compressive offset stress for each porosity were calculated based on ISO 13314. The results are presented in Table 1.

Figure 2.
Stress–strain curve in the elastic-plastic region of gyroid sheet network with unit cell size 1 mm and porosity between 50 and 85%.

Table 1.
Elasticity Modulus/Yield strength for gyroid sheet network with different unit cell sizes (1, 1.5, 2, and 2.5 mm).
The present study employed the Gibson–Ashby model, as represented in Equations (1) and (2), to investigate the relationship between the relative elasticity modulus, relative yield strength, and relative density (RD) of the gyroid sheet network.
where E*, σ*, and ρ* denote the relative elasticity modulus, relative yield strength, and relative density, respectively.
The constants C1 and n were found to be 0.695 and 1.5046, respectively, with an R2 value of 0.9992. Notably, the value of n is between 1 and 2, suggesting that the gyroid sheet network exhibits a combination of stretching- and bending-dominated behavior, consistent with the findings of Abueidda et al. [10], who reported C1 and n values of 0.555 and 1.406, respectively, for higher porosities. Furthermore, the values of C2 and m for yield strength were found to be 0.6909 and 1.4564, respectively, with an R2 value of 0.999.
4. Conclusions
In conclusion, the results of this study demonstrate that the gyroid sheet network can be utilized to achieve the required pore size for various biomedical applications, with a unit cell size between 1 and 2.5 mm and a porosity range of 50% to 85%. Moreover, a linear relationship exists between pore size and porosity for all unit cells. The elasticity modulus was insensitive to unit cell size within the range of 1 mm to 2.5 mm but required further investigation for larger unit cell sizes. The Gibson–Ashby model for a gyroid sheet network with a unit cell size of 1 mm resulted in E* = 0.695(ρ*)1.5046 with R2 = 0.9992, and σ* = 0.6909(ρ*)1.4564 with R2 = 0.999. The elasticity moduli obtained from the gyroid sheet network ranged between 4.5 and 28 GPa, which falls within the range of cortical bone stiffness, making these lattice structures suitable for biomedical devices to reduce stress shielding. Finally, the study confirms that these lattice structures can be fabricated using SLM with a layer thickness of approximately 30 µm and a powder diameter for Ti-6Al-4V of around 50 µm.
Author Contributions
Conceptualization, W.S., X.V. and B.Z.; methodology, W.S., X.V. and B.Z.; software, B.Z.; validation, B.Z.; formal analysis, B.Z.; investigation, B.Z.; resources, B.Z.; data curation, B.Z.; writing—original draft preparation, B.Z.; writing—review and editing, W.S. and X.V.; supervision, W.S. and X.V. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in the study are included in the article and further inquiries can be directed to the corresponding author.
Acknowledgments
Atlantic Technological University supports this Research through the PRTP in Modelling and Computation for Health and Society (MOCHAS).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Nicoletti, D. Cortical Hypertrophy in Total Hip Arthroplasty with Cementless Stem. Available online: https://radiopaedia.org/cases/87298 (accessed on 20 August 2023).
- Naghavi, S.A.; Tamaddon, M.; Garcia-Souto, P.; Moazen, M.; Taylor, S.; Hua, J.; Liu, C. A novel hybrid design and modelling of a customised graded Ti-6Al-4V porous hip implant to reduce stress-shielding: An experimental and numerical analysis. Front. Bioeng. Biotechnol. 2023, 11, 1092361. [Google Scholar] [CrossRef]
- Müller, P.; Gembarski, P.C.; Lachmayer, R. Density-Based Topology Optimization for a Defined External State of Stress in Individualized Endoprosthesis. Proc. Des. Soc. 2022, 2, 533–542. [Google Scholar] [CrossRef]
- Cortis, G.; Mileti, I.; Nalli, F.; Palermo, E.; Cortese, L. Additive manufacturing structural redesign of hip prostheses for stress-shielding reduction and improved functionality and safety. Mech. Mater. 2021, 165, 104173. [Google Scholar] [CrossRef]
- Tan, N.; van Arkel, R.J. Topology Optimisation for Compliant Hip Implant Design and Reduced Strain Shielding. Materials 2021, 14, 7184. [Google Scholar] [CrossRef]
- Rezapourian, M.; Jasiuk, I.; Saarna, M.; Hussainova, I. Selective laser melted Ti6Al4V split-P TPMS lattices for bone tissue engineering. Int. J. Mech. Sci. 2023, 251, 108353. [Google Scholar] [CrossRef]
- Alkentar, R.; Kladovasilakis, N.; Tzetzis, D.; Mankovits, T. Effects of Pore Size Parameters of Titanium Additively Manufactured Lattice Structures on the Osseointegration Process in Orthopedic Applications: A Comprehensive Review. Crystals 2023, 13, 113. [Google Scholar] [CrossRef]
- ISO 13314; Mechanical Testing of Metals–Ductility Testing–Compression Test for Porous and Cellular Metals. International Organization for Standardization: Geneva, Switzerland, 2011.
- Liu, Z.; Gong, H.; Gao, J. Enhancement in the fatigue resistances of triply periodic surfaces-based scaffolds. Int. J. Mech. Sci. 2023, 245, 108119. [Google Scholar] [CrossRef]
- Abueidda, D.W.; Abu Al-Rub, R.K.; Dalaq, A.S.; Lee, D.-W.; Khan, K.A.; Jasiuk, I. Effective conductivities and elastic moduli of novel foams with triply periodic minimal surfaces. Mech. Mater. 2016, 95, 102–115. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).