Abstract
Five different plant-based coagulants were used with the objective of removing methylene blue (MB) color aqueous solution. The plant-based coagulants were characterized by Fourier transform infrared spectroscopy (FTIR), scanning electron microscope (SEM) and Brunauer–Emmett–Teller (BET) which showed that these materials had high porosity and could adsorb the contaminant MB from the aqueous solution. To increase the efficiency of the plant-based coagulants, bentonite was applied as a flocculant. Results showed MB removal of 90.9%, 91.9%, 91.4%, 86.9% and 88.9%, respectively, for seeds of Chelidonium majus L., Dactylis glomerata L., Festuca ampla Hack., Tanacetum vulgare L. and rachises of Vitis vinifera L. In conclusion, plant-based coagulants mixed with bentonite are a biologic, sustainable and cheap alternative for MB removal.
1. Introduction
Textile finishing industries are chemical industries in which dyes and pigments are used in very large quantities with a great volume of water. The presence of these textile dyes in wastewaters discharged in aquatic environments, such as wadis, rivers, seas and oceans, and their lack of biodegradability under normal ecological conditions can destroy the vital conditions of these different environments while preventing the penetration of light to the depths of aquatic environments [1]. In order to treat these wastewaters, a coagulation–flocculation–decantation process (CFD) can be applied to remove the textile dye. Traditionally, coagulants such as Poly Ferric Sulfate (PFS), Poly Aluminum Chloride (PAC) and Poly Aluminum Ferric Chloride (PAFC) have been used for textile dye removal. However, these types of materials release iron and aluminum to the wastewater causing problems for the environment [2]. Natural macromolecular coagulants extracted from plants have been identified as promising materials for wastewater treatment and have attracted a lot of attention due to advantages such as abundancy, low toxicity, being multi-purpose and biodegradable [3]. In this work, the application of seeds from Chelidonium majus L., Dactylis glomerata L., Festuca ampla Hack., Tanacetum vulgare L. and rachises of Vitis vinifera L. was tested for removal of a textile dye contaminant from an aqueous solution. The aim of this work is to (1) produce and characterize the plant-based coagulants, (2) optimize the CFD process, and (3) evaluate the application of bentonite as a flocculant agent.
2. Materials and Methods
2.1. Reagents
The activated sodium bentonite (Na-Mt) was supplied by Angelo Coimbra & Ca., Lda, Maia, Portugal, and the methylene blue (MB) was acquired from VWR Chemicals, Amadora, Portugal. NaOH and H2SO4 (95%) were both obtained from Analar Normapur. Deionized water was used to prepare the respective solutions.
2.2. Analytic Techniques
The maximum absorbance wavelength (λmax) of MB was found at 665 nm, and the concentration of the residual dye in solution was calculated using the Beer–Lambert law. Dye discoloration was analyzed as follows (Equation (1)):
where and are the concentrations of dye at reaction time t and 0, respectively.
2.3. Plant-Based Coagulant Preparation
All the plants used in this work were collected from the district of Vila Real (Portugal), and transported to the Environmental Engineering Laboratory of the University of Trás-os-Montes and Alto Douro, Vila Real, where they were stored until used. In Table 1, the plants sub species, part collected for this study and the herbarium number attributed by UTAD for the plant’s identification are shown. All the plant-based coagulants were prepared in accordance with Martins et al. [4].

Table 1.
Plant identification with description of specie, sub specie, part collected and herbarium number.
2.4. Characterization of Plant-Based Coagulants
The chemical composition of the plant-based coagulants was evaluated by FTIR analysis (Shimadzu, Kyoto, Japan) and the microstructural characterization was carried out with a scanning electron microscope (FEI QUANTA 400 SEM/ESEM, Fei Quanta, Hillsboro, WA, USA). The textural parameters of samples were obtained from N2 adsorption–desorption isotherms at 77 K using a Micromeritics ASAP 2020 apparatus (TriStar II Plus, Micromeritics Instrument Corporation, Norcross, GA, USA).
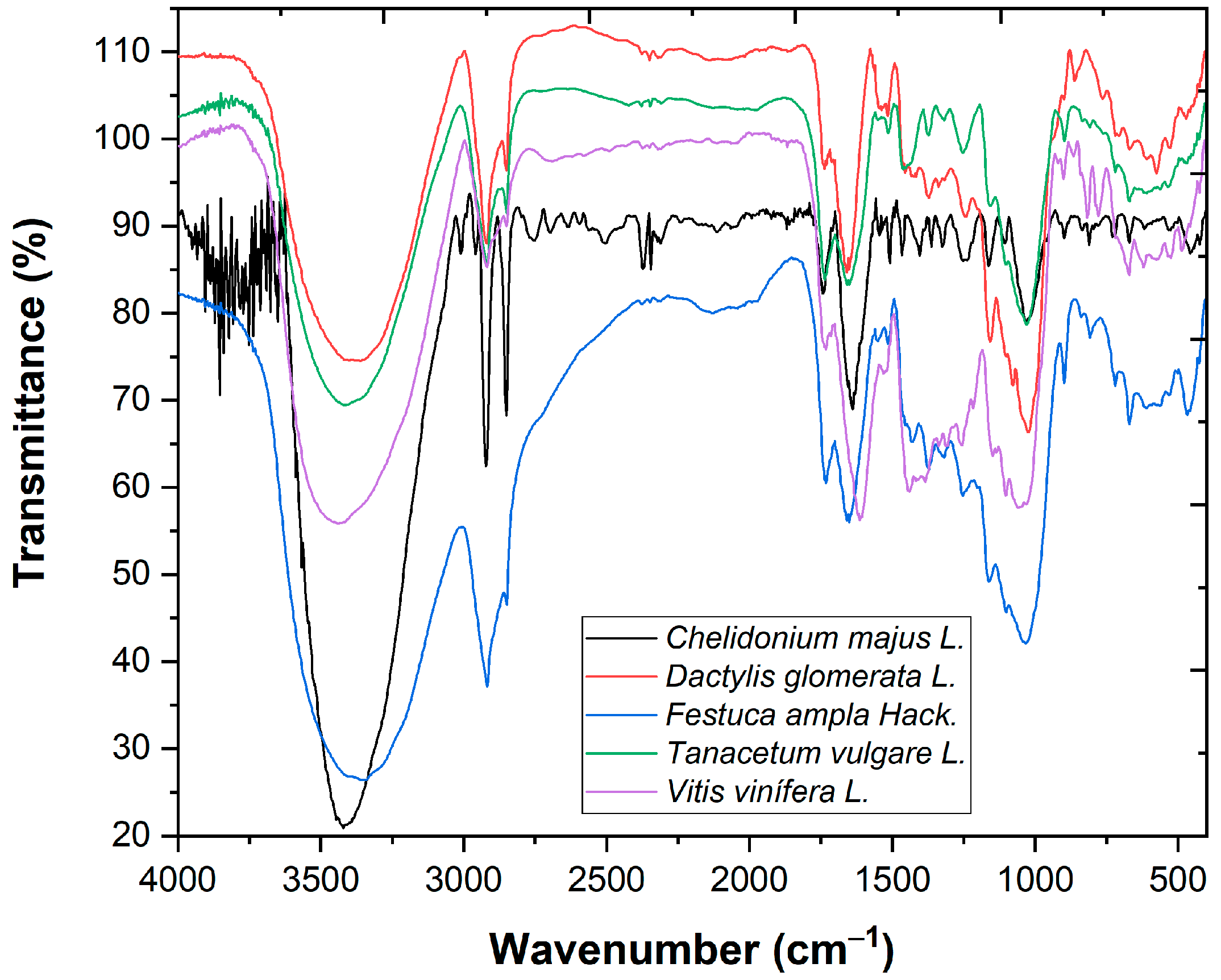
The FTIR analysis (Figure 1) presents vibration bands at 3421.72 cm−1 (stretching vibrations of OH groups (from water, alcohols, phenols, carbohydrates [5])), at 2920.23 and 2850.79 cm−1 (C-H stretching vibrations specific to CH3 and CH2 from lipids, metoxy derivatives, and C-H (aldehydes) including cis double bonds) and at 898.83–1253.73 cm−1 (C-O-C, C-C, and C-O stretching vibrations from carbohydrates [6]).
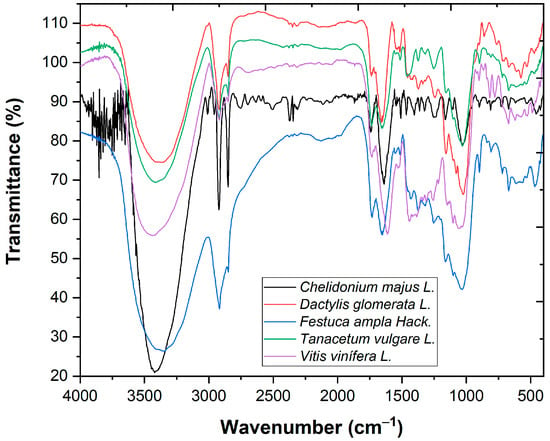
Figure 1.
FTIR analysis of plant-based coagulants.
In Figure 2, it was observed that plant-based coagulants exhibited a heterogeneous and relatively porous morphology. The spaces available (represented in dark) may facilitate adsorption of the MB contaminant [7].

Figure 2.
SEM images of (a) C. majus and (b) D. glomerata.
The BET analysis (Table 2) showed that all plant-based coagulants had a low BET surface area. The shape of its N2 adsorption–desorption isotherm was that of a type I isotherm, typical of microporous solids having relatively small external surfaces as defined by the International Union of Pure and Applied Chemistry (IUPAC) classification [8].

Table 2.
B.E.T. analysis of the plant-based coagulants.
2.5. Coagulation–Flocculation–Decantation Experimental Setup
The CFD process was performed in a jar-test device (ISCO JF-4) using 500 mL of MB aqueous solution in 1000 mL beakers. The equipment was provided by a set of 4 mechanic agitators powered by a regulated speed engine. In the CFD process, 5 plant-based coagulants (C. majus, D. glomerata, F. ampla, T. vulgare and V. vinifera) were tested and the process was optimized as follows:
- (1)
- The pH was varied (3.0, 5.0, 7.0, 9.0 and 11.0) under the operational conditions: [MB] = 50 mg/L, [coagulant] = 1.0 g/L, fast mix 150 rpm/3 min, slow mix 20 rpm/20 min, T = 298 K, V = 250 mL, sedimentation time = 30 min;
- (2)
- After determining the best pH in (1), the dosage of coagulant was varied (0.1, 0.5, 1.0 and 2.0 g/L) under the operational conditions: [MB] = 50 mg/L, fast mix 150 rpm/3 min, slow mix 20 rpm/20 min, T = 298 K, V = 250 mL, sedimentation time = 30 min;
- (3)
- With the best pH (1) and coagulant dosage (2), the mixing conditions were tested by varying the fast and slow mix under the operational conditions: [MB] = 50 mg/L, T = 298 K, V = 250 mL, sedimentation time = 30 min;
- (4)
- Under pre-determinate values of pH, coagulant dosage and mixing conditions obtained in (1)–(3), the concentration of activated sodium bentonite (0.1, 0.5, 1.0 and 2.0 g/L) as a flocculant was varied under the operational conditions: [MB] = 50 mg/L, T = 298 K, V = 250 mL, sedimentation time = 30 min.
All the experiments were performed in triplicate and analysis of variance (ANOVA) was performed using OriginLab 2019 software (Northampton, MA, USA). Tukey’s test was used for the comparison of means, which were considered to be significantly different when p < 0.05. The data are presented as mean and standard deviation (mean ± SD).
3. Results and Discussion
Coagulation–Flocculation–Decantation Experiments
In this work, five different plant-based coagulants were applied to an aqueous solution contaminated with a textile dye (MB) with the purpose of removing this contaminant.
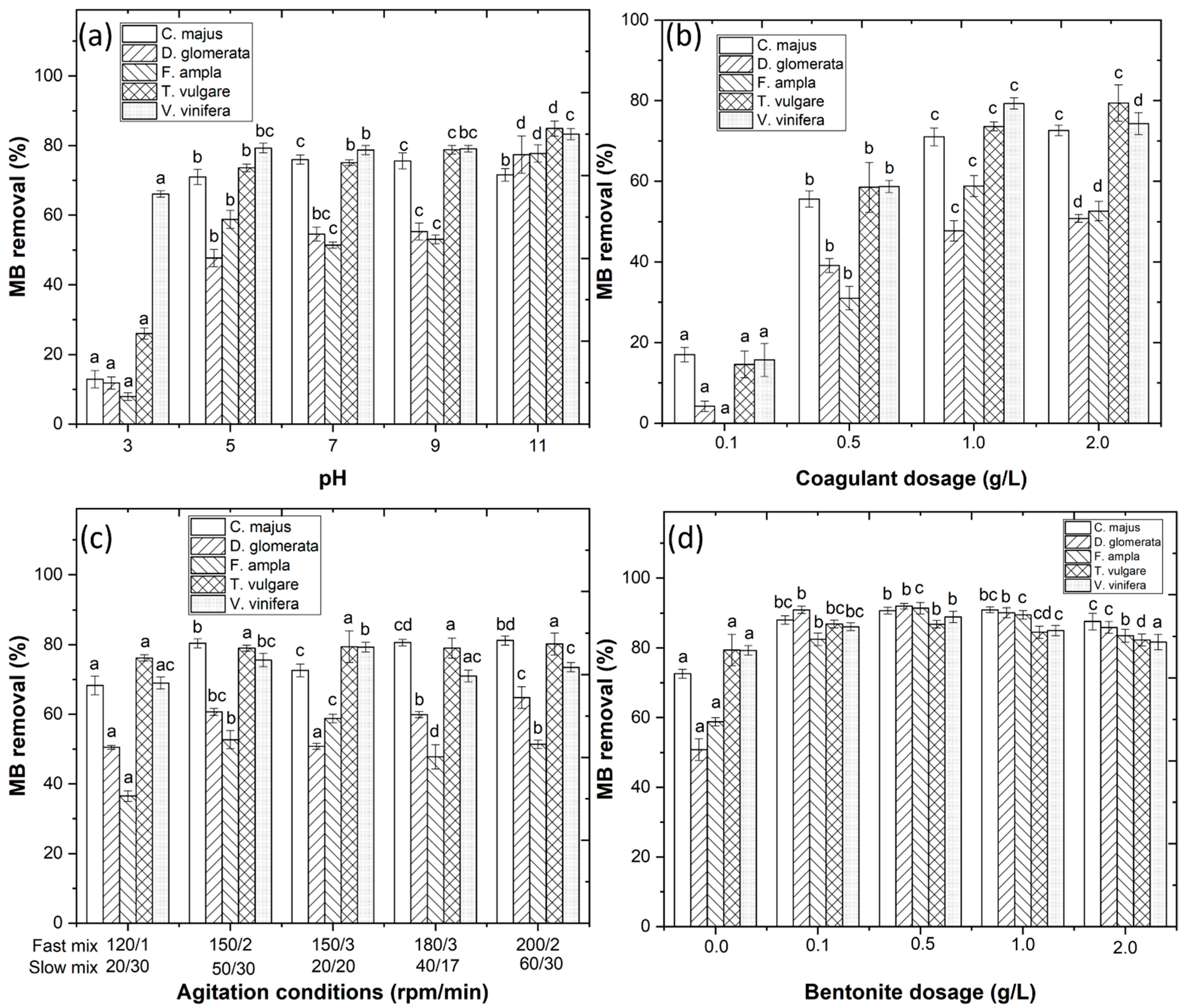
The pH of the wastewater plays an important role in the CFD process by affecting the nature of the functional groups of dye molecules and existing forms of coagulants [3]. Initially, wastewater pH was varied from 3.0 to 11.0 and results showed MB removal of 71.0%, 47.7%, 58.8%, 73.6% and 79.3%, respectively, for C. majus, D. glomerata, F. ampla, T. vulgare and V. vinifera (Figure 3a) at pH 5.0. The coagulant dosage was varied (0.1–2.0 g/L) and results showed MB removal of 72.6%, 50.8%, 58.8%, 79.4% and 79.3% (Figure 3b) with 2.0, 2.0, 1.0, 2.0 and 1.0 g/L coagulant, respectively. These results were in agreement with Beltrán-Heredia et al. [9] who observed that Carmine Indigo dye concentration decreased as the Moringa seed extract concentration increased. The effect of mixing conditions was also considered in this work and results showed MB removal of 81.2%, 64.8%, 58.8%, 79.4% and 79.3%, respectively (Figure 3c). These results are in agreement with Sánchez-Martín et al. [10] who observed that an increase in stirring speed improved the clarification of real surface water with the application of a plant-based extract. Finally, to improve the efficiency of the CFD process, bentonite was added as a flocculant aid and results showed MB removal of 90.9%, 91.9%, 91.4%, 86.9% and 88.9% (Figure 3d) for 1.0, 0.5, 0.5, 1.0 and 0.5 g/L bentonite, respectively. These results are in agreement with Sanghi and Bhattacharya [11] and Jorge et al. [12] who demonstrated that the combination of a coagulant with bentonite increased the removal of textile dyes and organic matter, respectively, from the wastewater.
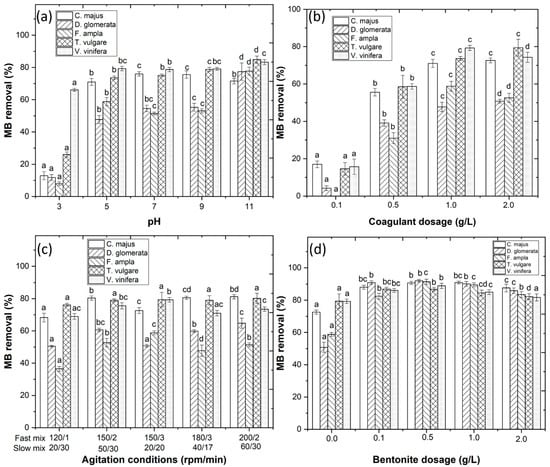
Figure 3.
Optimization of the CFD process with variations of (a) pH (3.0–11.0); (b) coagulant dosage (0.1–2.0 g/L); (c) agitation conditions; and (d) bentonite dosage (0.0–2.0 g/L). Means in bars with different letters represent differences (p < 0.05) within each coagulant (C. majus, D. glomerata, F. ampla, T. vulgare and V. vinifera) by comparing wastewaters.
4. Conclusions
The application of plant-based coagulants in combination with bentonite presents excellent results for MB removal from aqueous solution. Considering the results, the following is concluded:
- (1)
- The plant-based coagulants are carbon-based materials with porous structures that can adsorb the contaminants;
- (2)
- Under the best operational conditions, the plant-based coagulants achieve a high removal of MB from aqueous solution;
- (3)
- The addition of bentonite significantly increases the efficiency of the CFD process.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ECP2022-12659/s1.
Author Contributions
Conceptualization, N.J., A.R.T. and L.M.; methodology, N.J.; software, N.J.; validation, N.J., P.B.T., M.S.L. and J.A.P.; formal analysis, N.J.; investigation, N.J., A.R.T. and L.M.; resources, N.J.; data curation, N.J.; writing—original draft preparation, N.J., A.R.T. and L.M.; writing—review and editing, N.J. and J.A.P.; visualization, N.J., P.B.T., M.S.L. and J.A.P.; supervision, M.S.L. and J.A.P.; project administration, J.A.P.; funding acquisition, J.A.P. All authors have read and agreed to the published version of the manuscript.
Funding
The authors are grateful for the financial support of the Project AgriFood XXI, operation nº NORTE-01-0145-FEDER-000041, and to the Fundação para a Ciência e a Tecnologia (FCT) for the financial support provided to CQVR through UIDB/00616/2020. Ana R. Teixeira also thanks the FCT for the financial support provided through the doctoral scholarship UI/BD/150847/2020.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Liang, J.; Ning, X.; Kong, M.; Liu, D.; Wang, G.; Cai, H.; Sun, J.; Zhang, Y.; Lu, X.; Yuan, Y. Elimination and Ecotoxicity Evaluation of Phthalic Acid Esters from Textile-Dyeing Wastewater. Environ. Pollut. 2017, 231, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Jorge, N.; Teixeira, A.R.; Matos, C.C.; Lucas, M.S.; Peres, J.A. Combination of Coagulation–Flocculation–Decantation and Ozonation Processes for Winery Wastewater Treatment. Int. J. Environ. Res. Public Health 2021, 18, 8882. [Google Scholar] [CrossRef] [PubMed]
- Tie, J.; Jiang, M.; Li, H.; Zhang, S.; Zhang, X. A Comparison between Moringa Oleifera Seed Presscake Extract and Polyaluminum Chloride in the Removal of Direct Black 19 from Synthetic Wastewater. Ind. Crop. Prod. 2015, 74, 530–534. [Google Scholar] [CrossRef]
- Martins, R.B.; Jorge, N.; Lucas, M.S.; Raymundo, A.; Barros, A.I.; Peres, J.A. Food By-Product Valorization by Using Plant-Based Coagulants Combined with AOPs for Agro-Industrial Wastewater Treatment. Int. J. Environ. Res. Public Health 2022, 19, 4134. [Google Scholar] [CrossRef] [PubMed]
- Zavoi, S.; Fetea, F.; Ranga, F.; Pop, R.M.; Baciu, A.; Socaciu, C. Comparative Fingerprint and Extraction Yield of Medicinal Herb Phenolics with Hepatoprotective Potential, as Determined by UV-Vis and FT-MIR Spectroscopy. Not. Bot. Horti Agrobot. Cluj-Napoca 2011, 39, 82–89. [Google Scholar] [CrossRef] [Green Version]
- Zimmermann, B.; Bağcıoğlu, M.; Tafinstseva, V.; Kohler, A.; Ohlson, M.; Fjellheim, S. A High-Throughput FTIR Spectroscopy Approach to Assess Adaptive Variation in the Chemical Composition of Pollen. Ecol. Evol. 2017, 7, 10839–10849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vunain, E.; Mike, P.; Mpeketula, G.; Monjerezi, M.; Etale, A. Evaluation of Coagulating Efficiency and Water Borne Pathogens Reduction Capacity of Moringa Oleifera Seed Powder for Treatment of Domestic Wastewater from Zomba, Malawi. J. Environ. Chem. Eng. 2019, 7, 103118. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S. Physisorption of Gases, with Special Reference to the Evaluation of Surface Area and Pore Size Distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef] [Green Version]
- Beltrán-Heredia, J.; Sánchez-Martín, J.; Delgado-Regalado, A. Removal of Carmine Indigo Dye with Moringa Oleifera Seed Extract. Ind. Eng. Chem. Res. 2009, 48, 6512–6520. [Google Scholar] [CrossRef]
- Sánchez-Martín, J.; Beltrán-Heredia, J.; Peres, J.A. Improvement of the Flocculation Process in Water Treatment by Using Moringa Oleifera Seeds Extract. Braz. J. Chem. Eng. 2012, 29, 495–501. [Google Scholar] [CrossRef] [Green Version]
- Sanghi, R.; Bhattacharya, B. Adsorption-Coagulation for the Decolorisation of Textile Dye Solutions. Water Qual. Res. J. 2003, 38, 553–562. [Google Scholar] [CrossRef]
- Jorge, N.; Teixeira, A.R.; Guimarães, V.; Lucas, M.S.; Peres, J.A. Treatment of Winery Wastewater with a Combination of Adsorption and Thermocatalytic Processes. Processes 2022, 10, 75. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).