Abstract
Background: Diabetic foot infections (DFIs) are a major cause of hospitalization, limb loss, and mortality among patients with diabetic foot ulcers (DFUs). This study evaluated the risk of developing DFIs among patients with newly diagnosed DFUs across insurance categories. Methods: Adults ≥18 years with a new DFU diagnosis were identified in the PearlDiver insurance claims database (2010–2020) using validated ICD-9/10 codes. Insurance status at the index DFU was categorized as Medicaid, Medicare, commercial, or self-pay. Propensity score matching (1:3) based on age, sex, Charlson Comorbidity Index, and major comorbidities was used to compare Medicaid vs. non-Medicaid patients. Results: Among 258,122 patients with new DFUs, 20,638 (8.0%) were Medicaid beneficiaries. Medicaid patients were younger (50.1 ± 10.2 vs. 60.6 ± 12.1 years, p < 0.001) but had similar comorbidity burden compared with commercially insured and Medicare patients. In matched analysis post-matching, Medicaid insurance was independently associated with higher odds of DFI-related hospitalization within 12 months (aOR 1.18, 95% CI 1.14–1.24) and major amputation at 3 years (aOR 1.72, 95% CI 1.39–2.13). Higher CCI, chronic kidney disease, congestive heart failure, COPD, and peripheral vascular disease also predicted adverse outcomes. Conclusions: Medicaid insurance was independently associated with increased risks of DFI and major amputation among patients with newly diagnosed DFUs. These findings highlight infection as a potentially modifiable pathway driving limb loss and emphasize the need to improve early ulcer evaluation and infection management for Medicaid beneficiaries.
1. Background
Diabetic foot ulcers (DFUs) remain among the most devastating complications of diabetes mellitus, leading to significant morbidity, healthcare costs, and mortality [1]. Once ulceration occurs, the risk of infection rises in the presence of deep or chronic wounds, recurrent or traumatic ulcers, poor vascular supply, renal dysfunction, and diabetes-related immune compromise, particularly impaired neutrophil chemotaxis and phagocytic activity [2,3,4]. Peripheral neuropathy and PAD not only predispose to ulcer formation but also increase the likelihood of infection, delayed healing, and progression to limb loss [2,3,4].
The burden of infection-related outcomes is substantial, with approximately 50–60% of DFUs becoming infected, and 15–20% of moderate-to-severe infections leading to major amputation [1,5,6]. DFIs account for the majority of diabetes-related hospitalizations and are associated with a 5-year mortality approaching 30%, rising to over 70% following major amputation [1,5]. The coexistence of infection and PAD is particularly detrimental, with markedly higher rates of nonhealing, amputation, and death [1,2].
Beyond biological risk, recent evidence emphasizes the importance of system-level and preventive factors that modify outcomes in patients with PAD and diabetes [7,8]. In a national cohort of more than 214,000 adults with PAD, Howell et al. found that interruption of insurance coverage was independently associated with significantly higher risks of both major and minor amputations and reduced amputation-free survival, underscoring the impact of continuous access to longitudinal vascular, podiatric, and wound care on limb preservation [7].
Despite the substantial burden of infected diabetic foot ulcers, little is known about how insurance type affects clinical outcomes in this population.
Medicaid is the largest public health insurance program in the United States, jointly funded by federal and state governments to provide medical coverage for low-income individuals, including children, pregnant women, older adults, and people with disabilities [9]. Unlike universal health systems used in many countries, Medicaid eligibility, benefits, reimbursement rates, and provider participation vary substantially by state, resulting in marked heterogeneity in access to care [10,11]. While the federal government establishes minimum eligibility and coverage standards, states retain broad discretion over program design, including optional benefits, enrollment thresholds, and payment policies [10,11]. Historically, Medicaid reimbursement rates have been lower than those of Medicare or commercial insurance, which has been associated with reduced provider participation and limited access to specialty care [12,13]. As a result, Medicaid beneficiaries often experience delayed access to preventive services, fragmented care pathways, and higher rates of hospitalization and adverse health outcomes compared with individuals covered by Medicare or commercial insurance, particularly for chronic and resource-intensive conditions [14,15]. This study examines outcomes among Medicaid patients with infected DFUs compared with commercially insured and Medicare-insured patients.
2. Methods
2.1. Dataset Background
This study used the PearlDiver database, a national insurance claims repository widely utilized in health economics and outcomes research. The database contains de-identified patient-level information from multiple payor sources, including commercial plans, Medicaid, Medicare, other government payors, and cash-pay claims (www.pearldiverinc.com). As of November 2021, PearlDiver included longitudinal claims for more than 91 million unique patients across all 50 U.S. states and territories from 2010 to 2020. Patients are tracked across payor transitions and geographic moves. All analyses were performed using the platform’s integrated statistical tools.
2.2. Study Variables
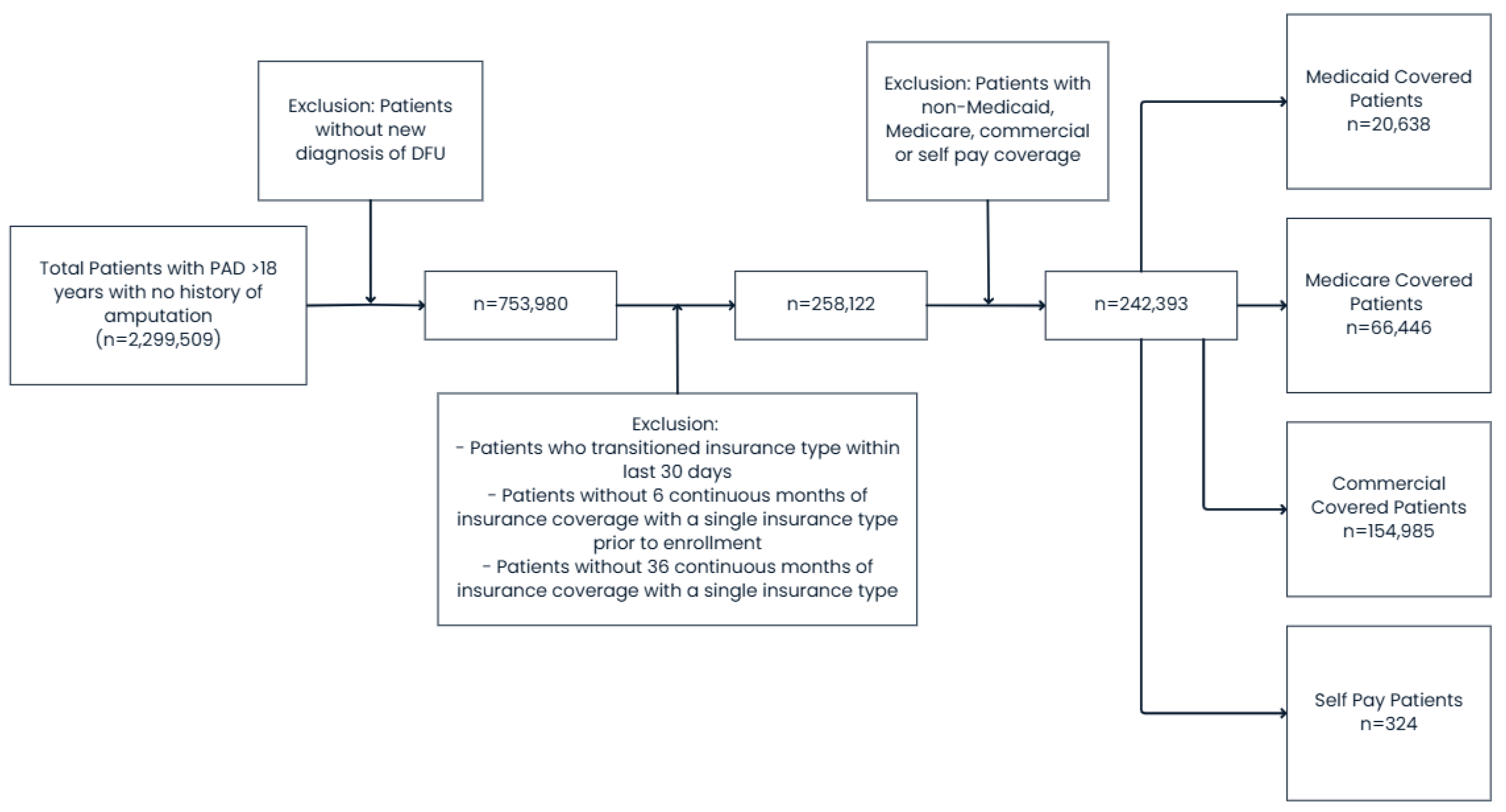
Patients ≥18 years old with a new diagnosis of DFU were identified using validated International Classification of Diseases, Ninth and Tenth Revision (ICD-9/10) diagnosis codes (Supplementary Table S1). The database records the first date on which the diagnosis appears, which was designated as the index date. Patients with a history of prior major lower-extremity amputation were excluded. After applying inclusion and exclusion criteria, 258,122 unique patients with infected DFUs were identified across all payor groups: Medicaid, Medicare, commercial insurance, and self-pay (Figure 1).
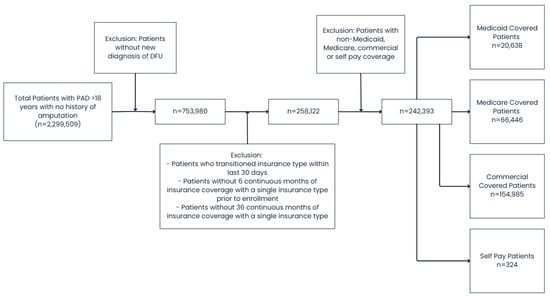
Figure 1.
Flowsheet detailing patient inclusion/exclusion process.
Insurance type at the index event was categorized as Medicaid, Medicare, commercial insurance, or self-pay. Patients who transitioned insurance type within 30 days before or after the index diagnosis were excluded to ensure group assignment validity. We then excluded patients without ≥6 months of continuous enrollment before and ≥36 months after the index diagnosis to ensure adequate longitudinal follow-up.
The primary outcome was diabetic foot ulcer infection, and the secondary outcome was major amputation, identified via ICD-9 procedure codes 84.10, 84.13–84.17, 84.3, and Current Procedural Terminology (CPT) codes 27590-92, 27594, 27596, 27598, 27880-82, 27884.
2.3. Covariates and Variables
Baseline demographics and comorbidities were abstracted from diagnostic and procedural coding data, including age, sex, Charlson Comorbidity Index (CCI), and the presence of cerebrovascular disease, chronic kidney disease, congestive heart failure, coronary artery disease, hypertension, peripheral vascular disease, and renal failure. Comorbid conditions were identified using validated ICD-9 and ICD-10 code groupings (Supplementary Table S1).
2.4. Outcomes and Statistical Analysis
Baseline characteristics were compared across insurance groups using descriptive statistics. To mitigate confounding, Medicaid and self-insured patients were propensity score matched (1:3) to commercially insured and Medicare patients (i.e., non-Medicaid) using age, sex, CCI, and major comorbidities through nearest-neighbor matching without replacement. Covariate balance was assessed using standardized mean differences (SMDs), with SMD < 0.1 indicating adequate balance. Logistic regression at prespecified fixed time points was selected as the primary analytic approach to estimate the association between insurance status and clinically and policy-relevant outcomes. Kaplan–Meier survival curves were constructed to compare major and minor amputation-free survival between groups in both unmatched and matched cohorts for descriptive purposes. Log-rank tests assessed between-group differences. Univariate Cox proportional hazards models were first used to identify potential predictors of major amputation. Variables with p < 0.05 in univariate analysis were entered into multivariable Cox models to estimate adjusted hazard ratios (HRs) with 95% confidence intervals. All tests were two-sided, and p < 0.05 was considered statistically significant.
3. Results
3.1. Cohort Characteristics
Of 258,122 eligible patients, 20,638 (8.0%) were insured through Medicaid, 66,446 (25.7%) through Medicare, 154,985 (60.0%) through commercial payors, and 324 (0.1%) were self-pay (Table 1). Medicaid patients were significantly younger (50.1 ± 10.2 years vs. 60.6 ± 12.1 years, p < 0.001) with a comparable comorbidity burden (CCI 2.3 ± 1.1 vs. 2.3 ± 1.0, p = 0.42) (Table 2). Before matching, Medicaid enrollees had slightly lower rates of coronary artery disease (42.7% vs. 44.8%, p = 0.002), cerebrovascular disease (33.9% vs. 35.4%, p = 0.01), and peripheral vascular disease (46.4% vs. 46.8%, p = 0.64), but higher rates of depression (59.7% vs. 45.6%, p < 0.001).

Table 1.
Comorbidities breakdown by insurance type.

Table 2.
Demographic information comparison between Medicare/commercial insurance patients (Control) and Medicaid patients (Treatment) before matching.
3.2. Propensity-Matched Cohort
Propensity score matching produced well-balanced cohorts with a 1:3 treated-to-control ratio, with a total of 20,638 in the Medicaid cohort and 61,914 in the non-Medicaid cohort. All covariates achieved excellent post-match balance (all SMD < 0.05). After matching, the mean age was identical between Medicaid and non-Medicaid patients (50.1 ± 10.2 vs. 50.0 ± 10.4 years, p = 0.78). Comorbidity prevalence was similar across groups: cerebrovascular disease (33.9% vs. 34.0%, p = 0.94), chronic kidney disease (38.6% vs. 37.7%, p = 0.42), congestive heart failure (23.9% vs. 22.6%, p = 0.31), coronary artery disease (42.7% vs. 43.1%, p = 0.66), hypertension (90.9% vs. 91.0%, p = 0.89), peripheral vascular disease (46.4% vs. 46.2%, p = 0.83), and renal failure (24.6% vs. 24.0%, p = 0.54). None of the differences reached statistical significance, confirming excellent covariate balance between treatment and control groups.
3.3. DFI Admission Within 12 Months
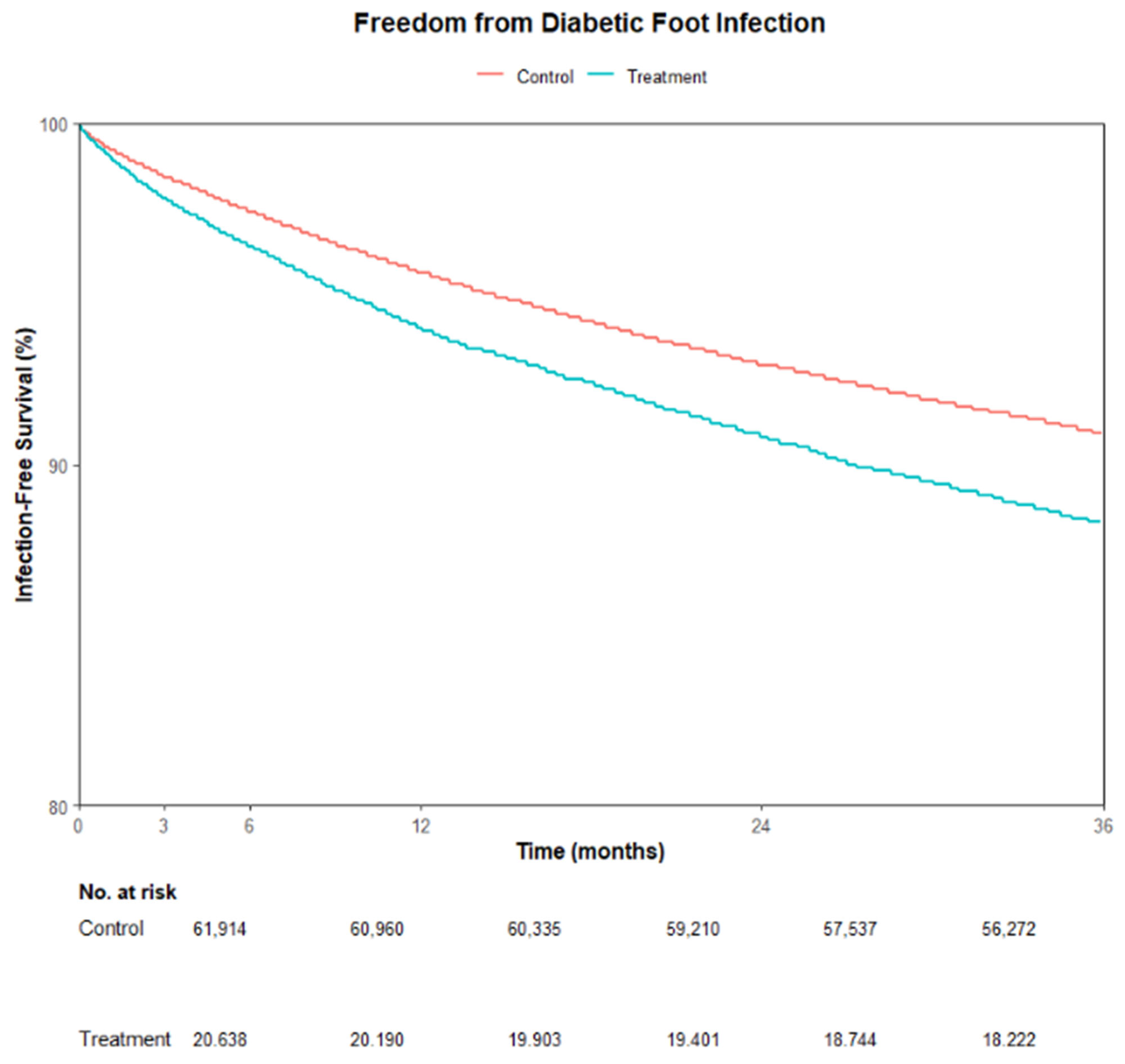
Kaplan–Meier analysis demonstrated significant differences in DFI-free survival between groups (log-rank χ2 = 121, df = 1, p < 0.001) (Figure 2). The Medicaid cohort experienced more admissions than expected (Observed = 2418 vs. Expected = 1992), whereas the non-Medicaid cohort had fewer (Observed = 5644 vs. Expected = 6070), indicating worse DFI-free survival in the Medicaid beneficiaries with DFUs.
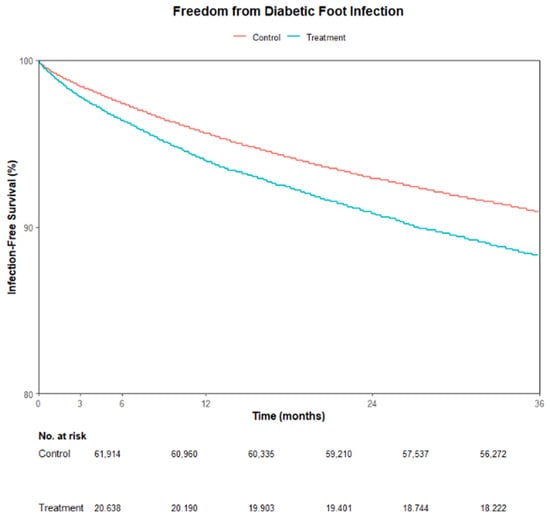
Figure 2.
Kaplan–Meier curve showing infection-free survival for Medicare/commercial insurance patients (Control) versus Medicaid patients (Treatment) post propensity matching.
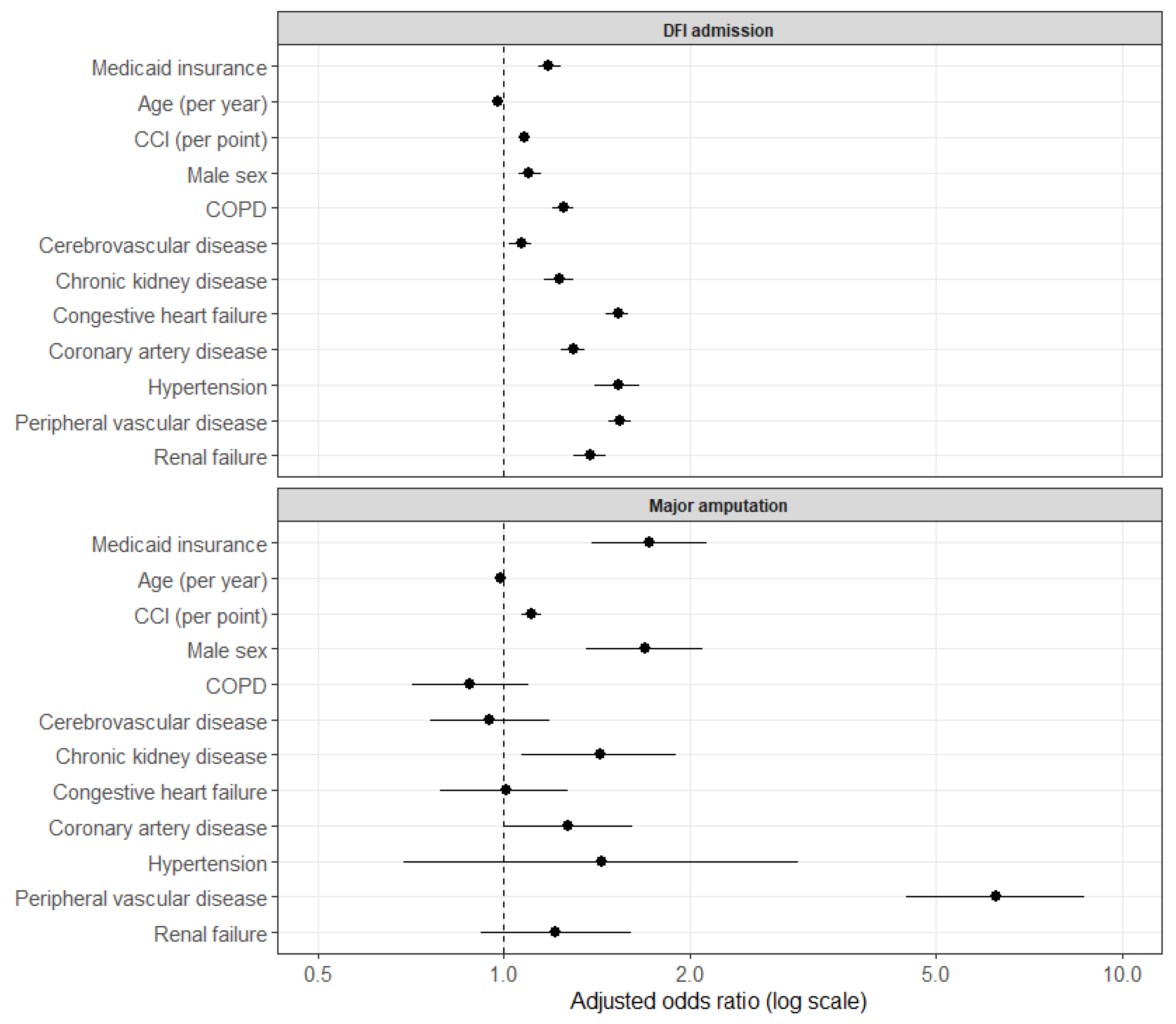
In multivariable logistic regression, Medicaid beneficiaries with DFUs had higher odds of DFI hospitalization within 12 months (aOR 1.18, 95% CI 1.14–1.24, p < 0.001) (Table 3). Other independent predictors included higher CCI (aOR 1.08 per point, 95% CI 1.07–1.09, p < 0.001), male sex (aOR 1.10, 95% CI 1.06–1.15, p < 0.001), COPD (aOR 1.25, 95% CI 1.20–1.30, p < 0.001), chronic kidney disease (aOR 1.23, 95% CI 1.16–1.30, p < 0.001), congestive heart failure (aOR 1.53, 95% CI 1.46–1.59, p < 0.001), coronary artery disease (aOR 1.30, 95% CI 1.24–1.35, p < 0.001), hypertension (aOR 1.53, 95% CI 1.40–1.66, p < 0.001), peripheral vascular disease (aOR 1.54, 95% CI 1.48–1.61, p < 0.001), and renal failure (aOR 1.38, 95% CI 1.30–1.46, p < 0.001) (Figure 3). Older age was associated with lower odds of DFI hospitalization (aOR 0.98 per year, 95% CI 0.98–0.98, p < 0.001).

Table 3.
Multivariable logistic regression for 12-month DFI hospitalization of Medicaid patients compared to matched non-Medicaid patients.
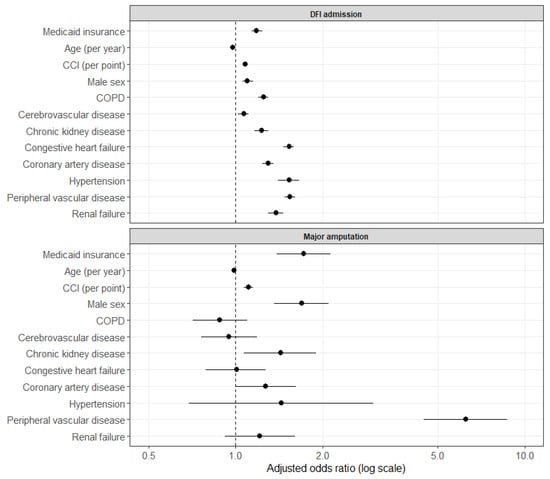
Figure 3.
Forest plot of adjusted odds ratios from a logistic regression model for 12-month DFI admission, with matched non-Medicaid patients as the reference group.
3.4. Major Amputation Within 3 Years
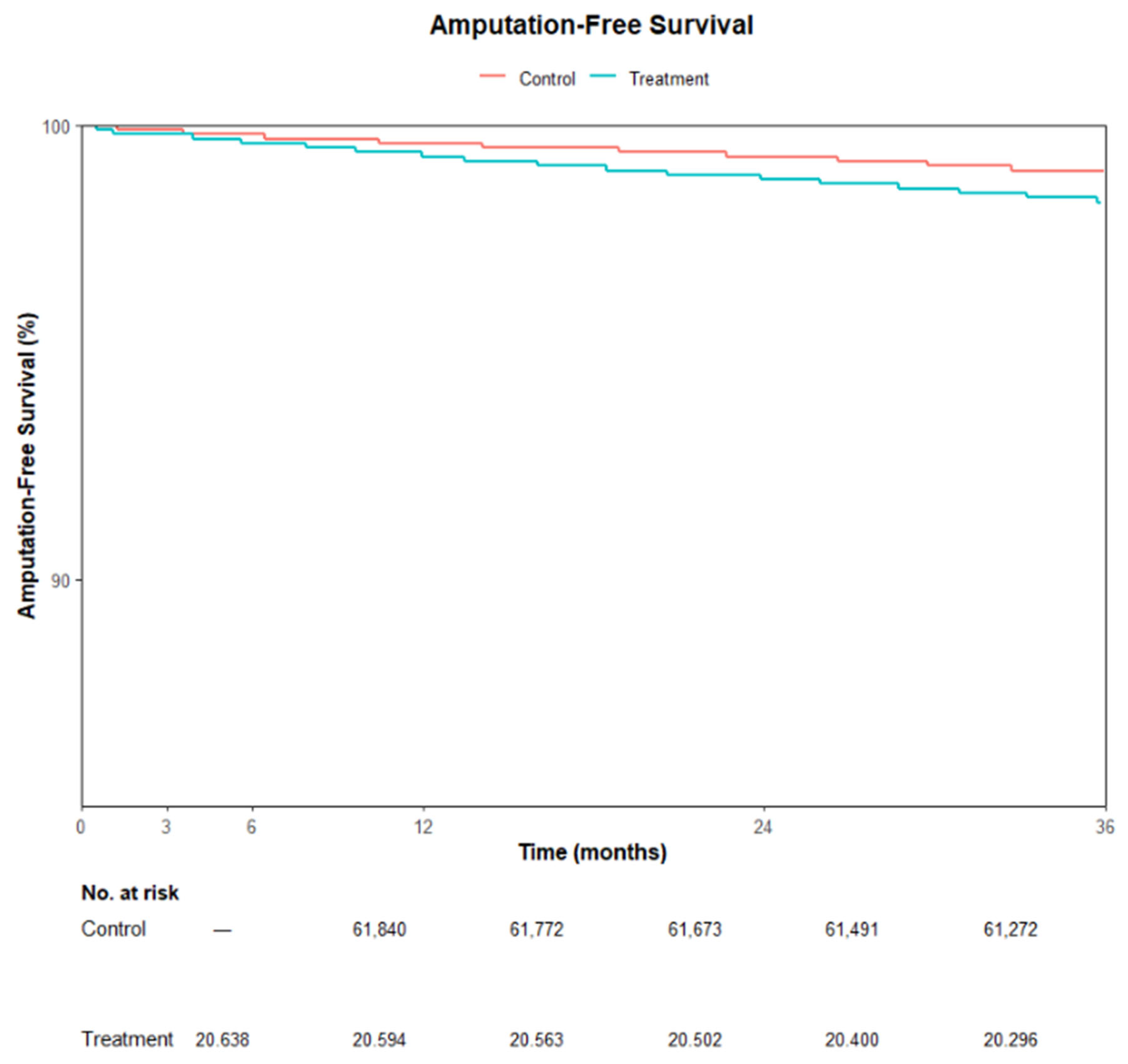
Kaplan–Meier analysis of 3-year major amputation-free survival also showed a significant difference between cohorts (log-rank χ2 = 51.1, df = 1, p < 0.001) (Figure 4). The Medicaid group experienced more major amputations than expected (Observed = 343 vs. Expected = 246), while the Medicare/commercial insurance group had fewer (Observed = 643 vs. Expected = 740), consistent with worse long-term limb salvage among treated patients.
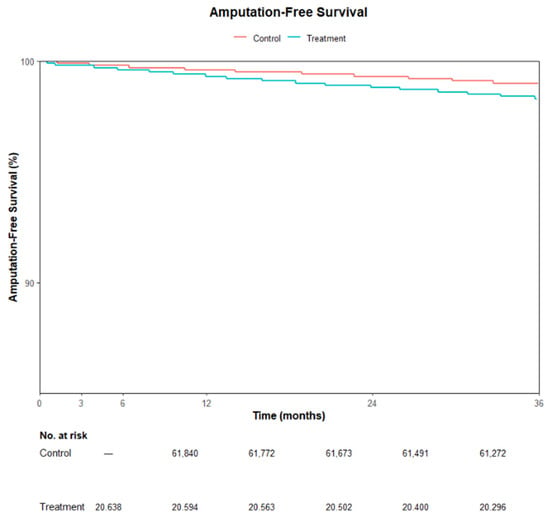
Figure 4.
Kaplan–Meier curve showing amputation-free survival for Medicare/commercial insurance patients (Control) versus Medicaid patients (Treatment) post propensity matching.
In multivariable analysis, Medicaid insurance type was associated with increased odds of major amputation (aOR 1.72, 95% CI 1.39–2.13, p < 0.001). Peripheral vascular disease was the strongest independent predictor (aOR 6.25, 95% CI 4.53–8.83, p < 0.001). Additional predictors included higher CCI (aOR 1.11 per point, p < 0.001), male sex (aOR 1.69, p < 0.001), and chronic kidney disease (aOR 1.43, p = 0.015).
4. Discussion
In this large national cohort of adults with newly diagnosed DFUs, Medicaid insurance status was strongly associated with significantly worse limb-related outcomes, including higher rates of DFI hospitalization within 12 months and substantially increased odds of major amputation. These findings demonstrate a clinically meaningful escalation along the DFU disease continuum, from ulcer to infection requiring inpatient care, and ultimately to limb loss. Although Medicaid beneficiaries were younger, their comorbidity burden was similar to that of commercially insured and Medicare patients, suggesting that insurance status might be an important factor influencing the pattern of access, care utilization, and disease progression once a DFU develops. The consistent association between Medicaid coverage and adverse limb events highlights a persistent disparity affecting this high-risk population.
Patients with low socioeconomic status (SES), including those insured through Medicaid, experience disproportionate burdens of CLTI [16]. Such patients are more likely to present with advanced ischemia, gangrene, or foot sepsis rather than earlier, more treatable stages of disease, and are less likely to receive preventive foot care, timely vascular evaluation, or guideline-recommended medical therapy [16,17,18,19]. Prior studies have shown that Medicaid patients receive fewer revascularization procedures, present with more advanced disease, receive lower quality PAD-related care, and experience decreased amputation-free survival than commercially insured patients [20,21,22]. They also exhibit higher rates of emergency department utilization and hospital readmissions for CLTI, reflecting inadequate access to coordinated outpatient and specialty care. For example, patients in distressed communities demonstrate higher ED visit rates and fewer vascular specialist encounters compared with those in more affluent areas [22]. These systemic inequities likely contribute to the excess burden of DFI hospitalization and amputations demonstrated in this study, which are consistent with the broader literature.
The finding that Medicaid patients were younger yet experienced worse outcomes warrants particular consideration. This pattern suggests earlier disease onset and accelerated progression rather than greater biological frailty. The drivers of these disparities are multifactorial and rooted in both healthcare system limitations and broader social determinants of health. Reduced access to primary care, fewer specialty clinics in low-resource areas, limited transportation, fragmented care coordination, and insurance-related barriers all contribute to delayed recognition of PAD and progression to advanced CLTI before patients enter the vascular system [16,17,18,19]. In this context, Medicaid coverage appears to function primarily as a marker of socioeconomic and structural vulnerability rather than a proxy for poorer baseline health, a distinction supported by the comparable comorbidity burden observed after matching in the present study. Socioeconomic vulnerability further reduces engagement with preventive care due to competing life priorities, out-of-pocket costs, and inadequate coverage for routine foot surveillance or podiatric services, resulting in missed opportunities for early detection and intervention [5,16,19]. Together, these factors create a persistent cycle in which patients with Medicaid present later in the disease course, receive fewer preventive or guideline-directed therapies, and ultimately experience substantially higher rates of limb loss.
DFI is a critical inflection point in the trajectory of diabetic foot disease. Contemporary IWGDF/IDSA guidelines emphasize that DFIs substantially increase the risk of lower-extremity amputation and are associated with substantial morbidity and mortality [2]. Armstrong and colleagues similarly highlight that the development of a foot infection often represents the transition from a chronic ulcer to a limb-threatening condition, with infection being the leading precipitant of amputation in patients with diabetes [1]. A single episode of DFI increases the risk of major amputation several-fold, and 5-year mortality after diabetes-related amputation approaches 60–70%, exceeding survival rates for many cancers [1,23]. The present study reinforces these known associations: Medicaid patients not only had higher odds of DFI admission (aOR 1.18) but also experienced substantially higher odds of major amputation (aOR 1.72) compared with matched controls. Given the strong causal pathway linking infection, tissue loss, and limb loss, these findings highlight a dangerous amplification of risk in an already vulnerable population.
Limitations
This study has limitations inherent to claims-based analyses, such as a lack of granular details regarding ulcer severity, extent of infection, vascular status, wound characteristics, and anatomic details of revascularization. Coding variability across health systems may introduce misclassification bias, and differences in care-seeking behavior cannot be fully captured. Although robust matching and adjustment were performed, residual confounding factors related to unmeasured social, behavioral, or geographic factors remain possible. Despite these limitations, the large national cohort and consistent findings across multiple outcomes strengthen the validity of the observed associations.
5. Conclusions
This study demonstrates that Medicaid insurance is independently associated with higher rates of DFI hospitalization and major amputation, despite comparable comorbidity burden after adjustment. Importantly, these findings frame Medicaid coverage not as a surrogate for poorer baseline health, but as a marker of structural vulnerability embedded within the U.S. healthcare system. While Medicaid is specific to the United States, the underlying mechanisms identified, such as socioeconomic disadvantage, delayed access to specialty care, fragmented care pathways, and inequitable resource distribution, are highly generalizable to international health systems. Similar disparities in limb outcomes have been observed across countries with publicly funded or mixed insurance models, suggesting that socioeconomic vulnerability, rather than insurance structure alone, is a fundamental driver of preventable limb loss worldwide. Addressing these structural barriers, with such things as rapid access to specialists for evaluation and wound care, is essential not only for improving outcomes among Medicaid beneficiaries in the United States but also for reducing global disparities in diabetic foot–related morbidity and mortality. Future studies should integrate claims data with clinical, vascular, and granular wound-level data to better delineate how access to outpatient foot care, infection severity, and timeliness of multidisciplinary intervention mediate the progression from DFU to DFI hospitalization and limb loss.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/diabetology7030052/s1. Table S1. Relevant ICD-9 and ICD-10 codes.
Author Contributions
Conceptualization: D.G.A., T.-W.T. and E.M.; methodology: T.-W.T., D.G.A. and E.M.; validation: C.T., T.-W.T., D.G.A. and E.M.; formal analysis: C.-D.S. and T.-W.T.; data curation: T.-W.T.; writing—original draft preparation, K.S., C.T. and T.-W.T.; writing—review and editing: C.T., K.S., C.-D.S., L.S., E.M., D.G.A. and T.-W.T.; supervision: T.-W.T.; project administration: L.S., D.G.A. and T.-W.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Armstrong, D.G.; Boulton, A.J.M.; Bus, S.A. Diabetic foot ulcers and their recurrence. N. Engl. J. Med. 2017, 376, 2367–2375. [Google Scholar] [CrossRef]
- Senneville, É.; Albalawi, Z.; van Asten, S.A.; Abbas, Z.G.; Allison, G.; Aragón-Sánchez, J.; Embil, J.M.; A Lavery, L.; Alhasan, M.; Oz, O.; et al. IWGDF/IDSA guidelines on the diagnosis and treatment of diabetes-related foot infections (IWGDF/IDSA 2023). Clin. Infect. Dis. 2024, 79, 286. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Parker, C.N.; Parker, T.J.; Kinnear, E.M.; Derhy, P.H.; Alvarado, A.M.; Huygens, F.; Lazzarini, P.A. Incidence and risk factors for developing infection in patients presenting with uninfected diabetic foot ulcers. PLoS ONE 2017, 12, e0177916. [Google Scholar] [CrossRef]
- Matheson, E.M.; Bragg, S.W.; Blackwelder, R.S. Diabetes-related foot infections: Diagnosis and treatment. Am. Fam. Physician 2021, 104, 386–394. [Google Scholar] [PubMed]
- McDermott, K.; Fang, M.; Boulton, A.J.M.; Selvin, E.; Hicks, C.W. Etiology, epidemiology, and disparities in the burden of diabetic foot ulcers. Diabetes Care 2023, 46, 209–221. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.G.; Tan, T.W.; Boulton, A.J.M.; Bus, S.A. Diabetic Foot Ulcers A Review. JAMA 2023, 330, 62–75. [Google Scholar] [CrossRef]
- Howell, C.; Lane, A.; Weinkauf, C.; Armstrong, D.G.; Arias, J.C.; Tan, T.W. Interruption of insurance coverage and the risk of amputation in patients with preexisting commercial health insurance and peripheral artery disease. Ann. Vasc. Surg. 2023, 96, 284–291. [Google Scholar] [CrossRef]
- Tan, T.W.; Tolson, J.P.; Arias Aristizabal, J.C.; Urbina, D.J.; Fermawi, S.A.; Weinkauf, C.; Marrero, D.G.; Armstrong, D.G. Association of preulcerative foot care and outcomes of diabetic foot ulceration. J. Am. Podiatr. Med. Assoc. 2024, 114, 22–071. [Google Scholar] [CrossRef]
- Centers for Medicare & Medicaid Services. Medicaid Program Overview. Updated 2023. Available online: https://www.medicaid.gov (accessed on 3 October 2025).
- Rallo, M.S.; Berke, C.N.; Shaw, T.C.; Bunch, J.A.; Menger, R.P. State-to-state variation in Medicaid reimbursement for neurosurgical procedures: Implications for healthcare access in vulnerable populations. Neurosurgery 2024, 94, 1201–1210. [Google Scholar] [CrossRef]
- Donohue, J.M.; Cole, E.S.; James, C.V. The US Medicaid program: Coverage, financing, reforms, and implications for health equity. JAMA 2022, 328, 1085–1096. [Google Scholar] [CrossRef]
- Medicaid and CHIP Payment and Access Commission. MACStats: Medicaid and CHIP Data Book. December 2023. Available online: https://www.macpac.gov/wp-content/uploads/2023/12/MACSTATS_Dec2023_WEB-508.pdf (accessed on 3 October 2025).
- Decker, S.L. In 2011 nearly one-third of physicians said they would not accept new Medicaid patients, but rising fees may help. Health Aff. 2012, 31, 1673–1679. [Google Scholar] [CrossRef]
- Corbisiero, M.F.; Tolbert, B.; Sanches, M.; Shelden, N.; Hachicha, Y.; Dao, H.; Thillot, B.; Adkins, K.; Muffly, T.M. Medicaid coverage and access to obstetrics and gynecology subspecialists: Findings from a national mystery caller study in the United States. Am. J. Obstet. Gynecol. 2023, 228, 722.e1–722.e9. [Google Scholar] [CrossRef] [PubMed]
- Timbie, J.W.; Kranz, A.M.; Mahmud, A.; Damberg, C.L. Specialty care access for Medicaid enrollees in expansion states. Am. J. Manag. Care 2019, 25, e83–e87. [Google Scholar] [PubMed]
- Allison, M.A.; Armstrong, D.G.; Goodney, P.P.; Hamburg, N.M.; Kirksey, L.; Lancaster, K.J.; Mena-Hurtado, C.I.; Misra, S.; Treat-Jacobson, D.J.; Solaru, K.T.W.; et al. Health disparities in peripheral artery disease: A scientific statement from the American Heart Association. Circulation 2023, 148, 286–296. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, O.I.; Yang, L.; Shultz, K.; Genovese, E.; Damrauer, S.M.; Wang, G.J.; Secemsky, E.A.; Treat-Jacobson, D.J.; Womeodu, R.J.; Fakorede, F.A.; et al. Racial, socioeconomic, and geographic disparities in preamputation vascular care for patients with chronic limb-threatening ischemia. Circ. Cardiovasc. Qual. Outcomes 2025, 18, e010931. [Google Scholar] [CrossRef]
- de Jager, E.; Gunnarsson, R.; Ho, Y.H. Disparities in advanced peripheral arterial disease presentation by socioeconomic status. World J. Surg. 2022, 46, 1500–1507. [Google Scholar] [CrossRef]
- Criqui, M.H.; Matsushita, K.; Aboyans, V.; Hess, C.N.; Hicks, C.W.; Kwan, T.W.; McDermott, M.M.; Misra, S.; Ujueta, F. Lower extremity peripheral artery disease: Contemporary epidemiology, management gaps, and future directions: A scientific statement from the American Heart Association. Circulation 2021, 144, e171–e191. [Google Scholar] [CrossRef]
- Kim, L.K.; Swaminathan, R.V.; Minutello, R.M.; Gade, C.L.; Yang, D.C.; Charitakis, K.; Shah, A.; Kaple, R.; Bergman, G.; Singh, H.; et al. Trends in Hospital Treatments for Peripheral Arterial Disease in the United States and Association Between Payer Status and Quality of Care/Outcomes, 2007–2011. Catheter. Cardiovasc. Interv. 2015, 86, 864–872. [Google Scholar] [CrossRef]
- O’Brien-Irr, M.S.; Harris, L.M.; Dosluoglu, H.H.; Dryjski, M.L. Procedural Trends in the Treatment of Peripheral Arterial Disease by Insurer Status in New York State. J. Am. Coll. Surg. 2012, 215, 311–321.e1. [Google Scholar] [CrossRef]
- Dubosq-Lebaz, M.; Kim, J.M.; Li, S.; Medina, F.; Jouffroy, M.; Schermerhorn, M.L.; Gouëffic, Y.; Secemsky, E.A. Impact of socioeconomic wealth on CLTI revascularization outcomes among Medicare patients from 2016 to 2023. J. Vasc. Surg. 2025, 83, 234–245.e44. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Swerdlow, M.A.; Armstrong, A.A.; Conte, M.S.; Padula, W.V.; Bus, S.A. Five year mortality and direct costs of care for people with diabetic foot complications are comparable to cancer. J. Foot Ankle Res. 2020, 13, 16. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.