Abstract
Diabetic foot ulcers (DFUs) are among the most severe and costly complications of diabetes, affecting millions of individuals worldwide. This narrative review summarizes major advances in regenerative medicine relevant to the management of DFUs and discusses how these approaches contribute to faster and more effective wound healing. Stem cell-based therapies, particularly those using adipose-derived mesenchymal stem cells (AD-MSCs), have demonstrated promising clinical outcomes through their ability to modulate inflammation, promote angiogenesis, and support skin and soft tissue regeneration. Platelet-rich plasma (PRP), an accessible autologous therapy, delivers concentrated growth factors that accelerate wound closure, enhance neovascularization, and shorten healing time compared with standard care. In addition, decellularized extracellular matrix (dECM) scaffolds provide a biologically active structural framework that supports cell adhesion, tissue remodeling, and granulation tissue formation. Collectively, these regenerative strategies offer new perspectives for improving functional recovery and quality of life in patients with DFUs, transforming chronic non-healing wounds into opportunities for effective tissue repair.
1. Introduction
Diabetic foot ulcers (DFUs) represent one of the most serious and costly complications of diabetes mellitus and remain a major global public health challenge [1]. The lifetime risk of developing a DFU among individuals with diabetes is estimated to range between 19% and 34%, with substantial geographic variability in prevalence and clinical outcomes [1,2,3]. DFUs are the leading cause of non-traumatic lower-extremity amputations worldwide and are associated with markedly increased morbidity, mortality, and healthcare expenditure [3,4]. Beyond the economic burden imposed by hospitalization, surgical interventions, rehabilitation, and long-term care, DFUs profoundly impair patients’ quality of life, contributing to physical disability, loss of independence, and high rates of depression and anxiety [4,5,6].
The pathogenesis of DFUs is multifactorial and reflects the convergence of peripheral neuropathy, vascular insufficiency, impaired immune responses, and chronic inflammation [7]. Sustained hyperglycemia drives endothelial dysfunction, oxidative stress, reduced nitric oxide bioavailability, and defective angiogenesis, all of which disrupt the tightly regulated wound-healing cascade [7,8]. As a result, the ulcer microenvironment is characterized by persistent inflammation, inadequate tissue perfusion and oxygenation, impaired cellular migration and proliferation, and excessive matrix degradation [8]. These pathophysiological disturbances render conventional wound care approaches such as debridement, offloading, infection control, and revascularization frequently insufficient, particularly in chronic or complex ulcers [7,8].
In this context, regenerative medicine has emerged as a paradigm-shifting strategy aimed at restoring tissue structure and function rather than merely controlling infection or preventing progression [9]. Regenerative approaches seek to actively modulate the wound microenvironment by enhancing angiogenesis, regulating inflammation, stimulating extracellular matrix remodeling, and promoting epithelial regeneration [9]. Key modalities under investigation and clinical application include stem-cell-based therapies, platelet-rich plasma (PRP), and biomaterial scaffolds such as decellularized extracellular matrix (dECM) [9]. Collectively, these strategies represent an effort to re-activate endogenous repair mechanisms that are impaired in the diabetic milieu.
Among cell-based therapies, mesenchymal stem cells (MSCs), particularly adipose-derived MSCs (AD-MSCs), have gained prominence due to their immunomodulatory, pro-angiogenic, and paracrine signaling properties [9,10]. PRP, an autologous and relatively accessible therapy, provides a concentrated source of growth factors that can enhance granulation tissue formation and wound closure [10]. Complementarily, dECM scaffolds offer a biologically active framework that supports cell adhesion, migration, and tissue remodeling, bridging the gap between biological signaling and structural repair [10]. While these regenerative strategies have demonstrated encouraging results, their clinical translation is challenged by variability in protocols, heterogeneous study designs, and evolving regulatory frameworks.
Importantly, successful management of DFUs extends beyond biological mechanisms alone [9]. Sociocultural determinants including health literacy, patient beliefs, cultural practices, and barriers to healthcare access play a decisive role in ulcer development, progression, and treatment adherence [9,10]. Thus, regenerative medicine should be viewed not as a standalone solution, but as part of a holistic and patient-centered approach to DFU care.
In this review, we examine the current landscape of regenerative therapies for DFUs, integrating experimental, translational, and clinical evidence. We discuss underlying mechanisms of action, therapeutic potential, limitations of existing evidence, and key challenges to clinical implementation, with the aim of identifying realistic pathways toward improved outcomes and reduced amputation risk. To achieve these objectives, a structured narrative review approach was adopted, allowing for the integration of mechanistic insights, preclinical findings, and clinical evidence across diverse regenerative modalities. Given the rapidly evolving nature of regenerative medicine and the heterogeneity of available studies, this approach was chosen to provide a clinically relevant and conceptually coherent synthesis rather than an exhaustive systematic analysis. The methodological framework used to identify, select, and evaluate the literature is described below.
2. Materials and Methods
This narrative review was designed to provide a clinically oriented and mechanistically informed overview of regenerative medicine strategies for the management of DFUs. In line with current recommendations for evidence-informed narrative reviews, the objective was to integrate experimental, translational, and clinical evidence, while explicitly acknowledging heterogeneity in study design and levels of evidence. A structured literature search was conducted in PubMed, Scopus, and Web of Science databases for studies published between January 2005 and October 2025, using combinations of the terms “diabetic foot,” “regenerative medicine,” “mesenchymal stem cells,” “adipose-derived stem cells,” “platelet-rich plasma,” and “decellularized extracellular matrix.” Reference lists of key articles and recent reviews were manually screened to identify additional relevant studies. To enhance transparency, a PRISMA-like screening approach was applied. Approximately 2100 records were initially identified. After removal of duplicates, titles and abstracts were screened for relevance to DFU pathophysiology and regenerative interventions. Full-text articles were assessed for scientific relevance and methodological clarity.
Inclusion criteria comprised preclinical studies addressing mechanisms of impaired diabetic wound healing, clinical studies evaluating regenerative therapies for DFUs or DFU-associated critical limb ischemia, and high-quality reviews or meta-analyses. Studies focusing exclusively on non-regenerative therapies, lacking methodological transparency, or providing redundant or outdated data were excluded. Given the heterogeneity of study designs, no formal quantitative synthesis was performed. Instead, evidence was qualitatively appraised based on study design, clinical relevance, and position along the translational continuum (preclinical to clinical). Conclusions were framed accordingly to avoid overinterpretation of lower-level evidence.
3. Stem Cell Therapy in Diabetic Foot: Mechanisms, Clinical Evidence, and Translational Challenges
Diabetes is a growing global public health concern, with diabetic foot infections representing one of the leading causes of lower-extremity amputations [9]. Standard management typically relies on a combination of systemic antibiotics, local wound care, and surgical debridement [9]. Despite these interventions, many patients with DFUs experience delayed, incomplete, or recurrent wound healing [9,10]. This limited therapeutic success reflects the complex pathophysiology of DFUs, which involves ischemia, peripheral neuropathy, chronic inflammation, and impaired cellular responses [10]. Consequently, conventional therapies often fail to restore tissue integrity, highlighting the need for advanced regenerative strategies [10]. Stem cells play a central role in regenerative medicine approaches aimed at enhancing wound repair. Clinically, stem cells can be administered through several routes, including topical application, incorporation into biomaterial scaffolds, wound gels or sprays, and intradermal or subcutaneous injections [11]. These approaches are frequently combined with supportive biomaterials or PRP to improve cell retention and biological activity at the wound site [11,12].
Multiple stem cell sources have been investigated for DFU treatment, including cells derived from peripheral blood, adipose tissue, bone marrow, and umbilical cord [12]. While stem cell therapy has shown promise in promoting wound healing in patients with DFUs, therapeutic efficacy varies substantially depending on the cellular source and preparation methods [13]. In current clinical practice, MSCs are the most employed cell type for DFU treatment [13]. MSCs can be obtained from bone marrow, adipose tissue, peripheral blood, and umbilical cord tissue, or as part of mononuclear cell populations [13,14]. These preparations represent heterogeneous cell mixtures that include hematopoietic stem cells, endothelial progenitor cells, lymphocytes, monocytes, and a smaller fraction of true MSCs [15]. Bone marrow-derived mononuclear cells exert therapeutic effects primarily through pro-angiogenic and paracrine mechanisms, but their regenerative potential is generally less specific and potent than that of isolated MSCs populations [16]. Randomized clinical trials comparing these approaches have demonstrated superior ulcer-healing outcomes with MSC-based therapies [16].
Although umbilical cord-derived MSCs have shown favorable safety profiles and therapeutic efficacy largely through immune modulation and secretion of growth factors clinical and translational evidence increasingly supports the preferential use of AD-MSCs [14]. AD-MSCs offer several practical advantages, including higher cell yield, less invasive harvesting procedures, and consistent regenerative outcomes across preclinical and clinical studies [14,17]. AD-MSCs are typically obtained through minimally invasive liposuction procedures performed under local anesthesia, most commonly from the lower abdomen [18]. Approximately 100–200 mL of adipose tissue is aspirated using a blunt cannula connected to a syringe containing saline supplemented with low-dose heparin [18,19]. The lipoaspirate is subsequently processed to isolate the stromal vascular fraction (SVF), a heterogeneous cellular compartment enriched in MSCs [20,21]. Isolation is commonly achieved via enzymatic digestion with collagenase type I (0.075%) at 37 °C for 30–45 min, followed by centrifugation to separate the SVF pellet [18,19]. After washing and resuspension in sterile saline or culture medium, cells may be used immediately or expanded in vitro under good manufacturing practice conditions [21]. For DFU treatment, AD-MSCs are administered either by local injections around the wound margins (intradermal or subcutaneous) or directly into the wound bed [18,20].
Several studies have demonstrated that SVF alone can effectively promote DFU healing, with the added advantage of a simpler and faster preparation process compared to expanded AD-MSCs [20,21]. Both SVF and AD-MSCs contribute to wound repair. However, important differences exist in their biological mechanisms and clinical outcomes [22]. SVF, owing to its heterogeneous cellular composition, exhibits strong chemotactic and epithelialization properties, accelerating wound closure and promoting re-epithelialization and angiogenesis, particularly in preclinical models and early-phase human studies [22,23,24]. In contrast, AD-MSCs display more pronounced immunomodulatory capacity, sustained secretion of pro-angiogenic factors, and stronger clinical evidence in chronic and complex wounds, with consistent improvements in pain reduction, healing time, and tissue regeneration [25,26]. Comparative analyses suggest that AD-MSC based therapies may provide superior long-term regenerative benefits relative to SVF alone [27].
At the mechanistic level, MSCs-mediated modulation of inflammation is central to their therapeutic efficacy in DFUs [25]. Under diabetic conditions, resident tissue stem cells exhibit aberrant activation of the nuclear factor kappa B (NF-κB) pathway, which perpetuates chronic inflammation and disrupts normal resolution of the wound-healing process [26]. This dysregulation impairs the polarization of macrophages toward the anti-inflammatory M2 phenotype, a critical transition required for tissue repair and remodeling. Restoration of MSC function either through pharmacological inhibition of NF-κB signaling or supplementation with immunomodulatory factors such as transforming growth factor-β1 (TGF-β1) has been shown to promote M2 macrophage polarization (Figure 1) and facilitate inflammation resolution [28].
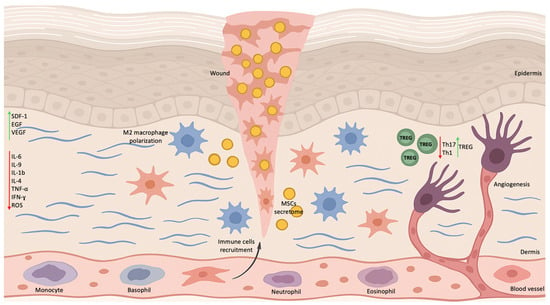
Figure 1.
Alterations in the Microenvironment of DFUs Following Stem Cell Therapy. Stem cell application modulates the wound microenvironment by promoting polarization of macrophages toward the M2 phenotype and increasing the secretion of anti-inflammatory cytokines. Stem cells enhance the expression of PDGFA, HGF, NGF, and FGF while reducing the levels of matrix metalloproteinases (MMP-2 and MMP-9) and pro-inflammatory cytokines. In addition, stem cells stimulate fibroblast activity, leading to increased collagen deposition and improved extracellular matrix remodeling. Elevated VEGF expression promotes the recruitment of endothelial progenitor cells and upregulation of PDGF and FGF-2 within the wound area, supporting angiogenesis. EGF enhances keratinocyte proliferation, whereas TGF-β contributes to local immune regulation through the induction of regulatory T cells.
Beyond immunomodulation, MSCs enhance DFU healing through the paracrine release of growth factors, including epidermal growth factor (EGF), vascular endothelial growth factor (VEGF), and stromal cell-derived factor-1 (SDF-1) [29]. These mediators stimulate angiogenesis, regulate cell migration, and optimize the wound microenvironment. MSCs further promote epithelialization by supporting fibroblast and keratinocyte proliferation and differentiation, enhancing regulatory T cell (Treg) activation while suppressing pro-inflammatory Th1 and Th17 responses (Figure 1), and reducing oxidative stress and pro-inflammatory cytokine activity [29].
A recent meta-analysis encompassing 24 studies and 1321 patients demonstrated that AD-MSC therapies significantly increased DFU healing rates compared with standard care [13]. AD-MSC based interventions have generally exhibited favorable short-term safety profiles, with no serious treatment-related adverse events reported in studies with follow-up periods of up to 12 months [30]. In a double-blind randomized trial involving 59 patients, 73% of DFUs treated with stem cells achieved complete healing within eight weeks, compared with 47% in the control group [31]. Despite these encouraging outcomes, the clinical translation of stem cell therapies for DFUs remains constrained by several critical limitations. First, autologous stem cells often exhibit reduced viability, impaired function, and poor retention within the hostile diabetic wound microenvironment [32]. Second, delivery methods and biomaterial support vary widely across studies, contributing to inconsistent outcomes and elevated costs. Third, manufacturing complexity and evolving regulatory frameworks pose significant barriers to widespread clinical adoption [33].
Moreover, the existing clinical evidence base is limited by small sample sizes, heterogeneous cell sources, non-standardized manufacturing protocols, and variable follow-up durations, which collectively restrict the generalizability and reproducibility of reported findings. Negative or neutral clinical trials are likely underreported, and direct comparative studies evaluating different cell types, dosages, and delivery strategies remain scarce. Although short-term safety appears favorable, long-term risks including aberrant angiogenesis, fibrosis, immune dysregulation, or theoretical tumorigenicity have not been fully elucidated.
From a regulatory standpoint, agencies such as U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) classify most stem cell-based interventions as advanced therapy medicinal products, imposing stringent manufacturing, safety, and clinical trial requirements. These regulatory constraints significantly limit routine clinical implementation outside of controlled research settings. Moving forward, progress in this field will depend on the establishment of standardized cell-product definitions, harmonized manufacturing protocols, and well-designed multicenter randomized clinical trials with long-term follow-up, alongside bioengineering strategies aimed at enhancing cell survival, function, and local retention.
4. Use of Platelet-Rich Plasma as a Regenerative Therapy in DFUs
PRP has emerged as an effective adjuvant therapy for the treatment of DFUs, demonstrating improvements in complete healing rates, acceleration of wound closure, and reductions in infection and amputation rates compared with conventional care [10]. Compared with stem cell-based therapies, PRP preparation is relatively simple and minimally invasive. The process typically involves venous blood collection (~30 mL) with the addition of an anticoagulant, such as citrate dextrose A, to prevent premature platelet activation [10]. This is followed by a two-step centrifugation protocol: the first centrifugation separates plasma from red blood cells, and the second concentrates platelets, yielding a plasma fraction enriched in platelets and growth factors. After centrifugation, the lower one-third of the plasma corresponds to PRP, while the upper two-thirds consist of platelet-poor plasma [34]. Also, depending on the clinical protocol and wound characteristics, PRP may be applied in liquid, gel, or injectable formulations [35].
Despite its clinical promise, a major limitation of PRP therapy is the lack of international standardization in preparation and application protocols, which hampers cross-study comparisons and reproducibility [10,34]. Variability in platelet concentration, activation methods, centrifugation parameters, and application frequency contributes to inconsistent clinical outcomes and complicates the establishment of universally accepted clinical guidelines. Clinical studies report that complete wound healing with PRP therapy generally occurs within 6 to 12 weeks, depending on ulcer size and depth, representing an average reduction in healing time of approximately 21 days compared with conventional treatment [36]. PRP is commonly administered weekly or biweekly, with treatment continued until complete epithelialization or for a maximum of 12 weeks, based on clinical response [37].
The therapeutic effects of PRP are primarily mediated by the local release of platelet-derived growth factors and cytokines, including platelet-derived growth factor (PDGF), transforming growth factor-β (TGF-β), vascular endothelial growth factor (VEGF), epidermal growth factor (EGF), and insulin-like growth factor-1 (IGF-1) [38]. These bioactive molecules promote fibroblast and keratinocyte proliferation and migration, stimulate angiogenesis, enhance extracellular matrix deposition, and accelerate granulation tissue formation, collectively facilitating wound closure [39].
Taken together, PRP represents a promising regenerative strategy for the management of DFUs, particularly as an accessible and autologous therapy suitable for outpatient wound care settings. While current evidence supports its ability to improve healing outcomes and reduce amputation rates, further well-designed, standardized clinical trials are needed to optimize PRP preparation protocols and treatment regimens. Future directions include combining PRP with bioengineered scaffolds or stem cell-based therapies to enhance regenerative efficacy and to establish robust, evidence-based clinical guidelines for its optimal use in diabetic wound management.
5. dECM as a Biomaterial Scaffold for DFU Regeneration
Tissue engineering integrates stem cells, biomaterial scaffolds, and biochemical cues to guide the cellular behaviors required for tissue repair and regeneration [40]. Among available biomaterials, dECM has emerged as a particularly promising natural scaffold. dECM is derived from tissues such as skin and intestine and undergoes chemical and physical processing to remove cellular components while preserving the three-dimensional architecture and essential bioactive molecules of the native extracellular matrix [40]. These components include collagen, elastin, proteoglycans, and growth factors, all of which are critical for supporting cell adhesion, proliferation, and differentiation [41].
Sources of dECM include bovine, porcine, and human tissues, and bovine-derived matrices are commonly used in orthopedic and cardiovascular applications, whereas porcine matrices are frequently employed in skin, abdominal wall, and cardiovascular reconstruction [42]. Human-derived matrices, obtained from tissues such as dermis, placenta, or blood vessels, are often preferred in clinical contexts requiring reduced immunological risk [42,43]. The decellularization process typically involves a combination of physical (e.g., agitation, pressure, freeze–thaw cycles), chemical (e.g., detergents such as sodium dodecyl sulfate or Triton X-100, saline solutions, alcohols), and enzymatic (e.g., nucleases, trypsin) treatments to achieve effective cell lysis and removal of nuclear, lipid, and cytoplasmic residues, followed by extensive washing to eliminate residual agents [44,45]. Regardless of tissue origin, dECM functions as a bioactive scaffold that supports cell integration and functional tissue repair, including in the healing of DFUs [43,46].
The principal structural and functional components of dECM include collagen, laminin, fibronectin, and glycosaminoglycans [47,48,49,50]. Collagen, the most abundant structural protein, provides mechanical support, promotes fibroblast migration and proliferation, and facilitates new matrix deposition, all of which are essential for granulation tissue formation and re-epithelialization in DFUs [47]. Laminin supports adhesion, migration, and differentiation of epithelial and endothelial cells, thereby enhancing angiogenesis and maintaining skin barrier integrity [48]. Fibronectin plays a central role in extracellular matrix organization, promoting cell adhesion, chemotaxis, and granulation tissue formation; notably, fibronectin deficiency in diabetic wounds has been associated with impaired healing [49]. Glycosaminoglycans, including hyaluronic acid, contribute to wound bed hydration, regulation of inflammation, promotion of angiogenesis, and remodeling [50].
Through the combined action of these components, dECM creates a bioactive microenvironment that stimulates tissue regeneration, modulates inflammatory responses, enhances angiogenesis, and accelerates DFU healing, often outperforming conventional wound dressings [47,48,49,50]. In terms of clinical outcomes, randomized studies and meta-analyses have demonstrated that dECM, when used as an adjunct to standard care, is associated with higher wound closure rates at 12 weeks (48–69%) compared with standard dressings alone (27–57%) and with some cellular products, alongside faster reductions in wound area and shorter time to complete healing [51,52].
From a cost-effectiveness perspective, dECM is generally more economical than cellular therapies or autologous grafts, offering comparable or superior clinical outcomes with lower total costs per patient and fewer applications required [53]. Nevertheless, several barriers limit the widespread adoption of dECM-based therapies. These include variability and lack of standardization in manufacturing processes, challenges related to large-scale industrial production, and the scarcity of robust long-term safety and durability data [54]. Moreover, many clinical studies evaluating dECM in DFUs are constrained by small sample sizes and short follow-up periods, underscoring the need for well-designed, long-term comparative trials to establish durability, safety, and cost-effectiveness across diverse patient populations.
6. Discussion and Conclusions
Despite the growing enthusiasm surrounding regenerative medicine for the management of DFUs, it is essential to critically appraise the limitations, risks, and translational barriers associated with these therapies. Although stem cell-based therapies, PRP, and dECM products have demonstrated encouraging outcomes, the current body of evidence remains heterogeneous and, in many cases, methodologically constrained [54]. A substantial proportion of available data originates from single-center studies, pilot trials, or non-randomized designs, which limits external validity and generalizability. Furthermore, follow-up durations are frequently short, typically ranging from 8 to 24 weeks, restricting conclusions regarding long-term wound durability, ulcer recurrence, limb salvage, and cost-effectiveness.
For stem cell-based interventions, additional variability arises from differences in cell source (adipose tissue, bone marrow, or umbilical cord), isolation and processing techniques, cell dose, delivery route, and frequency of administration. The lack of standardized manufacturing protocols, particularly under Good Manufacturing Practice conditions, complicates cross-study comparisons and undermines reproducibility. Importantly, autologous MSCs harvested from patients with diabetes may exhibit reduced viability, impaired paracrine signaling, and diminished regenerative potential due to the underlying metabolic and inflammatory milieu, potentially limiting therapeutic efficacy.
Similarly, the PRP literature is characterized by substantial methodological heterogeneity, including differences in centrifugation protocols, platelet concentration, leukocyte content, activation strategies, and application frequency. These inconsistencies contribute to variable clinical outcomes and hinder the establishment of optimal treatment parameters. Although several meta-analyses report improved healing rates with PRP, others highlight considerable inter-study variability and emphasize the need for standardized classification systems and reporting guidelines. For dECM-based therapies, randomized trials and meta-analyses suggest superior wound closure rates compared with standard care; however, variability in tissue source, decellularization methods, crosslinking processes, and residual bioactive content may influence both efficacy and immunogenicity. Long-term comparative data between dECM and cellular therapies remain limited. Available evidence suggests that clinical benefit may be highly dependent on appropriate patient selection, wound characteristics, and integration with comprehensive multidisciplinary care. Although dECM products are generally associated with low immunogenicity, incomplete decellularization or residual processing agents may provoke inflammatory responses in some cases.
Regulatory oversight represents an additional and significant barrier to widespread clinical implementation. In the United States, the FDA classifies most stem cell-based interventions as biological drugs requiring Investigational New Drug approval, while in Europe, the EMA regulates many of these approaches as Advanced Therapy Medicinal Products. Although these regulatory frameworks are essential for patient safety, they impose substantial financial, logistical, and temporal constraints that limit large-scale clinical adoption. PRP occupies a regulatory gray zone, often classified as minimally manipulated and autologous. However, the absence of harmonized international guidelines contributes to inconsistent clinical practice. dECM products, while more clearly regulated as medical devices or biologics, still face challenges related to manufacturing scalability, quality control, and post-market surveillance.
Recent qualitative and ethnographic studies emphasize that advanced therapeutic innovations must be contextualized within patients’ social and cultural environments to achieve meaningful and sustained clinical outcomes [8,9,10,55]. DFUs remain a major clinical and socioeconomic burden, associated with high risks of infection, limb loss, and reduced quality of life [56]. Regenerative therapies have emerged as promising adjuncts to standard care by targeting impaired inflammation resolution, angiogenesis, and tissue repair. Taken together, current evidence supports the potential role of regenerative approaches in DFU management but does not yet justify their universal adoption as standard therapy. These interventions should be viewed as complementary strategies, most effective when integrated into multidisciplinary treatment frameworks that include infection control, vascular assessment, offloading, metabolic optimization, and patient engagement.
Future progress will depend on well-designed, multicenter randomized controlled trials; standardized manufacturing and reporting protocols; longer follow-up periods; and robust health-economic evaluations. Advances in bioengineering such as combining stem cells with dECM scaffolds, controlled-release growth factor systems, or gene-modified cells may help overcome current limitations and enhance therapeutic consistency. By acknowledging both promise and limitations, a balanced interpretation underscores that regenerative medicine is not a replacement for established DFU care, but rather a rapidly evolving field with the potential to substantially improve outcomes once scientific, regulatory, and logistical challenges are systematically addressed.
Author Contributions
Conceptualization, D.C.M. and T.F.M.A.; methodology, D.C.M. and T.F.M.A.; validation, G.H.S.B.; writing—original draft preparation, D.C.M.; writing—review and editing, T.F.M.A. and G.H.S.B.; supervision, G.H.S.B.; project administration, D.C.M. and G.H.S.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| DFUs | Diabetic foot ulcers |
| AD-MSCs | Adipose-derived mesenchymal stem cells |
| PRP | platelet-rich plasma |
| dECM | decellularized extracellular matrix |
| MSCs | mesenchymal stem cells |
| SVF | stromal vascular fraction |
| PDGFA | platelet-derived growth factor A |
| HGF | hepatocyte growth factor |
| NGF | nerve growth factor |
| bFGF | basic fibroblast growth factor |
| MMP-2 | matrix metalloproteinase-2 |
| MMP-9 | matrix metalloproteinase-9 |
| FGF-2 | fibroblast growth factor-2 |
| EGF | epidermal growth factor |
| IL-10 | interleukin-10 |
| TNF-α | tumor necrosis factor-α |
| Tregs | regulatory T cells |
| IFN-γ | interferon-γ |
| TGF-β | transforming growth factor-β |
References
- Zhang, P.; Lu, J.; Jing, Y.; Tang, S.; Zhu, D.; Bi, Y. Global epidemiology of diabetic foot ulceration: A systematic review and meta-analysis. Ann. Med. 2017, 49, 106–116. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Tan, T.; Boulton, A.J.M.; Bus, S.A. Diabetic Foot Ulcers: A Review. JAMA 2023, 330, 62–75. [Google Scholar] [CrossRef]
- Gallagher, K.A.; Mills, J.L.; Armstrong, D.G.; Conte, M.S.; Kirsner, R.S.; Minc, S.D.; Plutzky, J.; Southerland, K.W.; Tomic-Canic, M.; American Heart Association Council on Peripheral Vascular Disease; et al. Current Status and Principles for the Treatment and Prevention of Diabetic Foot Ulcers in the Cardiovascular Patient Population: A Scientific Statement from the American Heart Association. Circulation 2024, 149, e232–e253. [Google Scholar] [CrossRef]
- Creager, M.A.; Matsushita, K.; Arya, S.; Beckman, J.A.; Duval, S.; Goodney, P.P.; Gutierrez, J.A.T.; Kaufman, J.A.; Joynt Maddox, K.E.; Pollak, A.W.; et al. Reducing Nontraumatic Lower-Extremity Amputations by 20% by 2030: Time to Get to Our Feet: A Policy Statement From the American Heart Association. Circulation 2021, 143, e875–e891. [Google Scholar] [CrossRef]
- Boutoille, D.; Féraille, A.; Maulaz, D.; Krempf, M. Quality of life with diabetes-associated foot complications: Comparison between lower-limb amputation and chronic foot ulceration. Foot Ankle Int. 2008, 29, 1074–1078. [Google Scholar] [CrossRef]
- McDonald, S.; Sharpe, L.; Blaszczynski, A. The psychosocial impact associated with diabetes-related amputation. Diabet. Med. 2014, 31, 1424–1430. [Google Scholar] [CrossRef] [PubMed]
- Falanga, V. Wound healing and its impairment in the diabetic foot. Lancet 2005, 366, 1736–1743. [Google Scholar] [CrossRef]
- Brem, H.; Tomic-Canic, M. Cellular and molecular basis of wound healing in diabetes. J. Clin. Investig. 2007, 117, 1219–1222. [Google Scholar] [CrossRef]
- Castellino, L.M.; Crisologo, P.A.; Chhabra, A.; Öz, O.K. Diabetic Foot Infections. Infect. Dis. Clin. N. Am. 2025, 39, 465–482. [Google Scholar] [CrossRef] [PubMed]
- OuYang, H.; Yang, J.; Wan, H.; Huang, J.; Yin, Y. Effects of different treatment measures on the efficacy of diabetic foot ulcers: A network meta-analysis. Front. Endocrinol. 2024, 15, 1452192. [Google Scholar] [CrossRef] [PubMed]
- Farabi, B.; Roster, K.; Hirani, R.; Tepper, K.; Atak, M.F.; Safai, B. The Efficacy of Stem Cells in Wound Healing: A Systematic Review. Int. J. Mol. Sci. 2024, 25, 3006. [Google Scholar] [CrossRef]
- Kucharzewski, M.; Rojczyk, E.; Wilemska-Kucharzewska, K.; Wilk, R.; Hudecki, J.; Los, M.J. Novel trends in application of stem cells in skin wound healing. Eur. J. Pharmacol. 2019, 843, 307–315. [Google Scholar] [CrossRef]
- Tong, L.; Tang, L.; Tang, B.; Zhang, J. Impacts of stem cells from different sources on wound healing rate in diabetic foot ulcers: A systematic review and meta-analysis. Front. Genet. 2025, 15, 1541992. [Google Scholar] [CrossRef] [PubMed]
- Mrozikiewicz-Rakowska, B.; Szabłowska-Gadomska, I.; Cysewski, D.; Rudziński, S.; Płoski, R.; Gasperowicz, P.; Konarzewska, M.; Zieliński, J.; Mieczkowski, M.; Sieńko, D.; et al. Allogenic Adipose-Derived Stem Cells in Diabetic Foot Ulcer Treatment: Clinical Effectiveness, Safety, Survival in the Wound Site, and Proteomic Impact. Int. J. Mol. Sci. 2023, 24, 1472. [Google Scholar] [CrossRef]
- Krasilnikova, O.A.; Baranovskii, D.S.; Lyundup, A.V.; Shegay, P.V.; Kaprin, A.D.; Klabukov, I.D. Stem and Somatic Cell Monotherapy for the Treatment of Diabetic Foot Ulcers: Review of Clinical Studies and Mechanisms of Action. Stem Cell Rev. Rep. 2022, 18, 1974–1985. [Google Scholar] [CrossRef]
- Lu, D.; Chen, B.; Liang, Z.; Deng, W.; Jiang, Y.; Li, S.; Xu, J.; Wu, Q.; Zhang, Z.; Xie, B.; et al. Comparison of bone marrow mesenchymal stem cells with bone marrow-derived mononuclear cells for treatment of diabetic critical limb ischemia and foot ulcer: A double-blind, randomized, controlled trial. Diabetes Res. Clin. Pract. 2011, 92, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Gu, Y.; Zhang, K.; Tu, Y.; Ouyang, C. Roles and mechanisms of umbilical cord mesenchymal stem cells in the treatment of diabetic foot: A review of preclinical and clinical studies. J. Diabetes Complicat. 2024, 38, 108671. [Google Scholar] [CrossRef]
- Bura, A.; Planat-Benard, V.; Bourin, P.; Silvestre, J.S.; Gross, F.; Grolleau, J.L.; Saint-Lebese, B.; Peyrafitte, J.A.; Fleury, S.; Gadelorge, M.; et al. Phase I trial: The use of autologous cultured adipose-derived stroma/stem cells to treat patients with non-revascularizable critical limb ischemia. Cytotherapy 2014, 16, 245–257. [Google Scholar] [CrossRef]
- Zhu, M.; Heydarkhan-Hagvall, S.; Hedrick, M.; Benhaim, P.; Zuk, P. Manual isolation of adipose-derived stem cells from human lipoaspirates. J. Vis. Exp. 2013, 79, e50585. [Google Scholar]
- Xu, Y.F.; Wu, Y.X.; Wang, H.M.; Gao, C.H.; Xu, Y.Y.; Yan, Y. Bone marrow-derived mesenchymal stem cell-conditioned medium ameliorates diabetic foot ulcers in rats. Clinics 2023, 78, 100181. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Yan, J.; Dong, Z.; Wang, J.; Jiang, X.; Cui, T.; Huang, Y.; Liu, H. Adipose-derived Mesenchymal Stem Cells are Ideal for the Cell-based Treatment of Refractory Wounds: Strong Potential for Angiogenesis. Stem Cell Rev. Rep. 2024, 20, 313–328. [Google Scholar] [CrossRef] [PubMed]
- Tanios, E.; Ahmed, T.M.; Shafik, E.A.; Sherif, M.F.; Sayed, D.; Gaber, N.; Hassan, Y. Efficacy of adipose-derived stromal vascular fraction cells in the management of chronic ulcers: A randomized clinical trial. Regen. Med. 2021, 16, 975–988. [Google Scholar] [CrossRef] [PubMed]
- Karagergou, E.; Dionyssopoulos, A.; Karayannopoulou, M.; Psalla, D.; Theodoridis, A.; Demiri, E.; Koliakos, G. Adipose-derived stromal vascular fraction aids epithelialisation and angiogenesis in an animal model. J. Wound Care 2018, 27, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Wang, L.; Feng, J.; Lu, F. Treatment of human chronic wounds with autologous extracellular matrix/stromal vascular fraction gel: A STROBE-compliant study. Medicine 2018, 97, e11667. [Google Scholar] [CrossRef]
- Huang, Y.Z.; Gou, M.; Da, L.C.; Zhang, W.Q.; Xie, H.Q. Mesenchymal Stem Cells for Chronic Wound Healing: Current Status of Preclinical and Clinical Studies. Tissue Eng. Part B Rev. 2020, 26, 555–570. [Google Scholar] [CrossRef]
- Gao, M.; Guo, H.; Dong, X.; Wang, Z.; Yang, Z.; Shang, Q.; Wang, Q. Regulation of inflammation during wound healing: The function of mesenchymal stem cells and strategies for therapeutic enhancement. Front. Pharmacol. 2024, 15, 1345779. [Google Scholar] [CrossRef]
- Domergue, S.; Bony, C.; Maumus, M.; Toupet, K.; Frouin, E.; Rigau, V.; Vozenin, M.C.; Magalon, G.; Jorgensen, C.; Noël, D. Comparison between Stromal Vascular Fraction and Adipose Mesenchymal Stem Cells in Remodeling Hypertrophic Scars. PLoS ONE 2016, 11, e0156161. [Google Scholar] [CrossRef]
- Ko, K.I.; Syverson, A.L.; Kralik, R.M.; Choi, J.; DerGarabedian, B.P.; Chen, C.; Graves, D.T. Diabetes-Induced NF-κB Dysregulation in Skeletal Stem Cells Prevents Resolution of Inflammation. Diabetes 2019, 68, 2095–2106. [Google Scholar] [CrossRef]
- Hetta, H.F.; Elsaghir, A.; Sijercic, V.C.; Akhtar, M.S.; Gad, S.A.; Moses, A.; Zeleke, M.S.; Alanazi, F.E.; Ahmed, A.K.; Ramadan, Y.N. Mesenchymal stem cell therapy in diabetic foot ulcer: An updated comprehensive review. Health Sci. Rep. 2024, 7, e2036. [Google Scholar] [CrossRef]
- Carstens, M.H.; Quintana, F.J.; Calderwood, S.T.; Sevilla, J.P.; Ríos, A.B.; Rivera, C.M.; Calero, D.W.; Zelaya, M.L.; Garcia, N.; Bertram, K.A.; et al. Treatment of chronic diabetic foot ulcers with adipose-derived stromal vascular fraction cell injections: Safety and evidence of efficacy at 1 year. Stem Cells Transl. Med. 2021, 10, 1138–1147. [Google Scholar] [CrossRef]
- Moon, K.C.; Suh, H.S.; Kim, K.B.; Han, S.K.; Young, K.W.; Lee, J.W.; Kim, M.H. Potential of Allogeneic Adipose-Derived Stem Cell-Hydrogel Complex for Treating Diabetic Foot Ulcers. Diabetes 2019, 68, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Ariyanti, A.D.; Zhang, J.; Marcelina, O.; Nugrahaningrum, D.A.; Wang, G.; Kasim, V.; Wu, S. Salidroside-Pretreated Mesenchymal Stem Cells Enhance Diabetic Wound Healing by Promoting Paracrine Function and Survival of Mesenchymal Stem Cells Under Hyperglycemia. Stem Cells Transl. Med. 2019, 8, 404–414. [Google Scholar] [CrossRef]
- Yu, Q.; Qiao, G.H.; Wang, M.; Yu, L.; Sun, Y.; Shi, H.; Ma, T.L. Stem Cell-Based Therapy for Diabetic Foot Ulcers. Front. Cell Dev. Biol. 2022, 10, 812262. [Google Scholar] [CrossRef] [PubMed]
- Dhurat, R.; Sukesh, M. Principles and Methods of Preparation of Platelet-Rich Plasma: A Review and Author’s Perspective. J. Cutan. Aesthetic Surg. 2014, 7, 189–197. [Google Scholar] [CrossRef]
- Picard, F.; Hersant, B.; Bosc, R.; Meningaud, J.P. The growing evidence for the use of platelet-rich plasma on diabetic chronic wounds: A review and a proposal for a new standard care. Wound Repair Regen. 2015, 23, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.P.; Kumar, V.; Pandey, A.; Pandey, P.; Gupta, V.; Verma, R. Role of platelet-rich plasma in healing diabetic foot ulcers: A prospective study. J. Wound Care 2018, 27, 550–556. [Google Scholar] [CrossRef]
- Dai, J.; Jiang, C.; Sun, Y.; Chen, H. Autologous platelet-rich plasma treatment for patients with diabetic foot ulcers: A meta-analysis of randomized studies. J. Diabetes Complicat. 2020, 34, 107611. [Google Scholar] [CrossRef]
- Zhou, D.; Liang, Q.; Ge, X.; Xu, J. Allogeneic platelet-rich plasma inhibits ferroptosis in promoting wound repair of type 2 diabetic ulcers. Free Radic. Biol. Med. 2024, 215, 37–47. [Google Scholar] [CrossRef]
- Peng, Y.; Wang, J.; Liu, X.; Zhou, Y.; Jia, S.; Xu, J.; Zheng, C. Efficacy of Platelet-Rich Plasma in the Treatment of Diabetic Foot Ulcers: A Systematic Review and Meta-Analysis. Ann. Vasc. Surg. 2024, 98, 365–373. [Google Scholar] [CrossRef]
- Cho, H.; Blatchley, M.R.; Duh, E.J.; Gerecht, S. Acellular and cellular approaches to improve diabetic wound healing. Adv. Drug Deliv. Rev. 2019, 146, 267–288. [Google Scholar] [CrossRef]
- Solarte David, V.A.; Güiza-Argüello, V.R.; Arango-Rodríguez, M.L.; Sossa, C.L.; Becerra-Bayona, S.M. Decellularized Tissues for Wound Healing: Towards Closing the Gap Between Scaffold Design and Effective Extracellular Matrix Remodeling. Front. Bioeng. Biotechnol. 2022, 10, 821852. [Google Scholar] [CrossRef]
- Keane, T.J.; Swinehart, I.T.; Badylak, S.F. Methods of tissue decellularization used for preparation of biologic scaffolds and in vivo relevance. Methods 2015, 84, 25–34. [Google Scholar] [CrossRef]
- Brown, M.; Li, J.; Moraes, C.; Tabrizian, M.; Li-Jessen, N.Y.K. Decellularized extracellular matrix: New promising and challenging biomaterials for regenerative medicine. Biomaterials 2022, 289, 121786. [Google Scholar] [CrossRef]
- Crapo, P.M.; Gilbert, T.W.; Badylak, S.F. An overview of tissue and whole organ decellularization processes. Biomaterials 2011, 32, 3233–3243. [Google Scholar] [CrossRef]
- Ventura, R.D.; Padalhin, A.R.; Park, C.M.; Lee, B.T. Enhanced decellularization technique of porcine dermal ECM for tissue engineering applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 104, 109841. [Google Scholar] [CrossRef]
- Lee, J.S.; Choi, Y.S.; Cho, S.W. Decellularized Tissue Matrix for Stem Cell and Tissue Engineering. Adv. Exp. Med. Biol. 2018, 1064, 161–180. [Google Scholar] [PubMed]
- McGrath, M.; Zimkowska, K.; Genoud, K.J.; Maughan, J.; Gutierrez Gonzalez, J.; Browne, S.; O’Brien, F.J. A Biomimetic, Bilayered Antimicrobial Collagen-Based Scaffold for Enhanced Healing of Complex Wound Conditions. ACS Appl. Mater. Interfaces 2023, 15, 17444–17458. [Google Scholar] [CrossRef] [PubMed]
- Tyeb, S.; Shiekh, P.A.; Verma, V.; Kumar, A. Adipose-Derived Stem Cells (ADSCs) Loaded Gelatin-Sericin-Laminin Cryogels for Tissue Regeneration in Diabetic Wounds. Biomacromolecules 2020, 21, 294–304. [Google Scholar] [CrossRef]
- Kanta, J.; Zavadakova, A.; Sticova, E.; Dubsky, M. Fibronectin in hyperglycaemia and its potential use in the treatment of diabetic foot ulcers: A review. Int. Wound J. 2023, 20, 1750–1761. [Google Scholar] [CrossRef]
- Deng, T.; Li, J.; Li, X.; Yang, X.; Tao, M.; Wang, Y.; Wang, X.; Sun, L.; Li, H.; Wu, M. A snail glycosaminoglycan-derived patch inspired by extracellular matrix accelerates diabetic wound healing via promoting re-epithelization. Carbohydr. Polym. 2025, 368, 124168. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Chen, Y.; Wang, N.; Yin, G.; Wei, C.; Xu, W. The Efficacy and Safety of Acellular Matrix Therapy for Diabetic Foot Ulcers: A Meta-Analysis of Randomized Clinical Trials. J. Diabetes Res. 2020, 2020, 6245758. [Google Scholar] [CrossRef]
- Shawa, H.; Kaur, R.; Tchanque-Fossuo, C.; Lev-Tov, H.; West, K.; Lim, P.S.; Yang, N.T.; Dafinone, M.; Lyle, R.E.; Li, C.S.; et al. Cellular Versus Acellular Matrix Products for Diabetic Foot Ulcer Treatment: The Dermagraft and Oasis Longitudinal Comparative Efficacy Study (DOLCE)—A Randomized Clinical Trial. Diabetes Care 2025, 48, 966–973. [Google Scholar] [CrossRef]
- Gilligan, A.M.; Waycaster, C.R.; Landsman, A.L. Wound closure in patients with DFU: A cost-effectiveness analysis of two cellular/tissue-derived products. J. Wound Care 2015, 24, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.R.; Lai, X.S.; Cheong, H.F.; Gui, D.K.; Zhao, Y.H.; Xu, Y.H. Advances in biomaterials and regenerative medicine for diabetic foot ulcer therapy. Ageing Res. Rev. 2025, 109, 102779. [Google Scholar] [CrossRef] [PubMed]
- Costa, D.; Gallelli, G.; Scalise, E.; Ielapi, N.; Bracale, U.M.; Serra, R. Socio-Cultural Aspects of Diabetic Foot: An Ethnographic Study and an Integrated Model Proposal. Societies 2024, 14, 240. [Google Scholar] [CrossRef]
- Metwaly, A.; Ismail, S.; Nagy, Y.M.; Abdallah, A.; Hassanin, S.M.; Ramadan, K.; Nady, A.; Melad, L.; Mahmoud, M.; Abdullah, M.U.; et al. Limb Salvage: A review of stem cell and growth factor therapies for diabetic foot ulcers. Diabetes Res. Clin. Pract. 2025, 231, 113036. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.