Abstract
Background: Cardiovascular complications are a leading cause of death in patients with type 2 diabetes (T2D). The GLP-1 receptor agonist (GLP-1RA) semaglutide has shown cardiometabolic benefits in individual studies, but a comprehensive analysis of its effects in both oral and subcutaneous formulations is lacking. Objective: This study aimed to systematically evaluate the impact of semaglutide, in oral and subcutaneous formulations, on major adverse cardiovascular events (MACE) in patients with T2D. Methods: This review adhered to the PRISMA guidelines and included a comprehensive search of PubMed, MEDLINE, and Google Scholar from November 2016 to June 2025. High-quality randomized controlled trials (RCTs) comparing semaglutide with placebo in patients with T2D were included. The primary endpoint was MACE, defined as cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke. Hazard ratio (HR) and 95% confidence intervals (CIs) were pooled using a random-effects model. Results: Of the 127 articles screened, 3 trials involving 16,130 participants met the inclusion criteria. The pooled HR for MACE across the SOUL, SUSTAIN-6, and PIONEER-6 trials was 0.83 (95% CI: 0.76–0.92; I2 = 25%), indicating a 17% relative risk reduction with low heterogeneity. Adverse event profiles were comparable between the semaglutide and placebo groups. Conclusions: Semaglutide use was associated with a significant and consistent reduction in MACE in patients with T2D, supporting its role as a valuable therapeutic option for combined glycemic control and cardiovascular risk reduction.
1. Introduction
Cardiovascular disease is the leading cause of morbidity and mortality in patients with type 2 diabetes mellitus (T2D), and the risk of major adverse cardiovascular events (MACE: cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke) is significantly elevated in this population. The underlying mechanisms include chronic hyperglycemia, insulin resistance, dyslipidemia, inflammation, and endothelial dysfunction, all of which accelerate atherosclerosis and increase the likelihood of myocardial infarction, stroke, and heart failure [1,2,3].
Glucagon-like peptide-1 receptor agonists (GLP-1RAs) effectively reduce cardiovascular risk in patients with T2D and established cardiovascular disease or high cardiovascular risk. They reduce the incidence of MACE by approximately 14% compared with placebo, with similar efficacy observed for both injectable and oral formulations. In addition to improving glycemic control and promoting weight reduction, GLP-1RAs improve blood pressure, lipid profile, inflammatory markers, and endothelial function, contributing to their cardioprotective effects [1,2,3,4,5].
Semaglutide, a GLP-1RA, was approved by the FDA in 2024 for the treatment of T2D and for reducing the risk of major cardiovascular events in patients with T2D and established cardiovascular disease. Large randomized clinical trials (RCTs) have demonstrated that semaglutide reduces the risk of MACE by 14–20% in patients with T2D and/or obesity, with benefits independent of the degree of weight loss. Semaglutide also improves other cardiometabolic risk factors (including blood pressure, lipid profile, and inflammatory markers) and slows the progression of chronic kidney disease [1,3,4,6,7].
GLP-1RAs, and semaglutide in particular, are recommended as therapy for patients with type 2 diabetes and high cardiovascular risk [2]. Important questions remain regarding whether cardiovascular benefits differ by route of administration (oral versus subcutaneous) and whether effects are consistent across diverse patient populations. Given the growing clinical use of semaglutide and the availability of new trial data, a focused synthesis of cardiovascular outcomes exclusively for this agent is timely and clinically relevant. By pooling data from all available high-quality RCTs, this study aims to provide a definitive assessment of semaglutide’s cardiovascular efficacy and safety in T2D, including the impact of administration route and patient characteristics on clinical outcomes.
1.1. Evidence Before This Study
Semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist available in both subcutaneous and oral formulations, has demonstrated favorable cardiometabolic effects in individuals with T2D. To characterize the existing evidence regarding its cardiovascular efficacy, we conducted a systematic literature search of PubMed, MEDLINE, and Google Scholar for randomized, placebo-controlled trials reporting major adverse cardiovascular events (MACE)—defined as the composite of cardiovascular death, non-fatal myocardial infarction, and non-fatal stroke. The search encompassed publications from November 2016 to June 2025 using the following search terms: “semaglutide”, “oral semaglutide”, “subcutaneous semaglutide”, “cardiovascular outcomes”, “myocardial infarction”, “stroke”, “heart failure”, and “randomized controlled trial”. Prior meta-analyses have broadly evaluated cardiovascular outcomes for the GLP-1 receptor agonist class but lacked agent-specific analyses for semaglutide and did not incorporate data from the SOUL trial (2025), which represents the largest cardiovascular outcomes trial of oral semaglutide to date and substantially augments the evidence base for this formulation.
1.2. Added Value of This Study
This systematic review and meta-analysis represent an agent-specific synthesis of cardiovascular outcome data for semaglutide, integrating findings from three pivotal cardiovascular outcomes trials: SUSTAIN-6, PIONEER-6, and SOUL. Through pooled analysis of 16,130 participants across these high-quality randomized controlled trials, we provide the most comprehensive contemporary evaluation of semaglutide’s cardiovascular effects. Distinct from previous class-wide analyses, our study incorporates the SOUL trial (n = 9650), thereby enabling robust assessment of oral semaglutide efficacy and permitting direct comparison of cardiovascular benefits across both oral and subcutaneous formulations. We assessed MACE as the primary endpoint, along with between-study heterogeneity, renal outcomes, and safety profiles, providing a comprehensive evaluation of semaglutide’s cardioprotective potential. The inclusion of formulation-specific subgroup analyses and rigorous sensitivity testing enhances the clinical applicability of our findings for evidence-based therapeutic decision-making in patients with T2D and elevated cardiovascular risk.
2. Methods
We systematically searched PubMed, MEDLINE, and Google Scholar from November 2016 to June 2025 to identify randomised, placebo-controlled trials that enrolled individuals with T2D and evaluated semaglutide (oral or subcutaneous). Trials were eligible if they included more than 500 participants, reported MACE as a primary or major secondary endpoint, and provided hazard ratios with confidence intervals. Studies were screened and extracted independently by two reviewers following PRISMA guidelines. Risk of bias was evaluated using the Cochrane Risk of Bias 2 tool. Hazard ratios were pooled using a random-effects model.
Our findings demonstrate that semaglutide therapy significantly reduces the risk of MACE in individuals with T2D, with consistent benefits observed across formulations. The pooled hazard ratio of 0.83 (95% CI: 0.76–0.92) indicates a robust cardioprotective effect, corresponding to a 17% relative risk reduction, supported by low heterogeneity across studies (I2 = 25%). Semaglutide showed a favourable safety profile comparable to placebo, with no unexpected adverse events. No significant renal benefit was observed in the pooled analysis, aside from modest effects in individual trials. Given the consistent reduction in cardiovascular risk, semaglutide represents a valuable therapeutic option for patients with T2D requiring both glycemic and cardiovascular risk management.
2.1. Study Design and Registration
This systematic review and meta-analysis were conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The study protocol was prospectively registered with PROSPERO (registration number: CRD420251074309) on 15 June 2025.
2.2. Search Strategy
A comprehensive literature search was conducted in PubMed, MEDLINE, and Google Scholar from database inception through June 2025. The search strategy combined the following terms: “semaglutide”, “oral semaglutide”, “subcutaneous semaglutide”, “cardiovascular outcomes”, “MACE”, “major adverse cardiovascular events”, “type 2 diabetes”, and “randomized controlled trial”. Reference lists of relevant articles were manually screened to identify additional eligible studies.
2.3. Eligibility Criteria
Studies were eligible for inclusion if they met the following criteria:
- Study design: Randomized, placebo-controlled trials;
- Population: Adult patients (≥18 years) with T2D;
- Intervention: semaglutide (oral or subcutaneous formulation) versus placebo;
- Outcomes: Reported MACE as a primary or major secondary endpoint, defined as the composite of cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke;
- Sample size: ≥500 participants;
- Sufficient follow-up duration to capture cardiovascular endpoints (≥12 months).
2.4. Exclusion Criteria
The following types of studies were excluded:
- Non-randomized studies, observational studies, or narrative reviews;
- Studies in non-diabetic populations;
- Trials with incomplete or unreported cardiovascular outcome data;
- Conference abstracts without full-text publication;
- Duplicate publications from the same trial cohort.
2.5. Study Selection and Data Extraction
Two independent reviewers (G.A. and V.M.) screened titles and abstracts, followed by a full-text review of potentially eligible articles. Disagreements were resolved through consensus or consultation with a third reviewer. Data were extracted independently by both reviewers using a standardized form. The following information was extracted from each trial:
- Study characteristics: first author, publication year, trial name, study design;
- Population characteristics: sample size, baseline demographics, cardiovascular risk profile;
- Intervention details: semaglutide formulation, dose, duration of treatment;
- Outcomes: number of MACE events, hazard ratios (HRs) with 95% confidence intervals (CIs), secondary cardiovascular outcomes;
- Follow-up duration and loss to follow-up rates.
2.6. Risk of Bias Assessment
Risk of bias for each included randomized controlled trial was assessed independently by two reviewers using the Cochrane Risk of Bias 2 (RoB 2) tool. The assessment covered five domains:
- Bias arising from the randomization process;
- Bias due to deviations from intended interventions;
- Bias due to missing outcome data;
- Bias in the measurement of outcomes;
- Bias in the selection of the reported result.
Each domain was rated as “low risk,” “some concerns,” or “high risk”. Each study’s risk of bias was evaluated.
All three included trials demonstrated low risk of bias across all assessed domains. Randomization procedures were appropriate with adequate allocation concealment. Outcome ascertainment was performed by blinded adjudication committees using standardized definitions. Missing outcome data were minimal (<5% in all studies), and all prespecified outcomes were reported. Overall, the methodological quality of included studies was high.
2.7. Publication Bias
Due to the small number of included studies (n = 3), formal assessment of publication bias using funnel plot asymmetry or Egger’s test was not feasible. A leave-one-out sensitivity analysis was performed to assess the robustness of the pooled estimates. Each study was sequentially excluded, and the remaining two studies were re-analysed to determine whether any single study exerted a disproportionate influence on the overall effect estimate. Additionally, the analysis was repeated using a fixed-effects model to evaluate whether the choice of statistical model substantially altered the pooled results. However, all included trials were large, prospective, and registered before initiation, reducing the likelihood of publication bias.
2.8. Statistical Analysis
Hazard ratios and 95% confidence intervals for MACE were pooled across studies using a random-effects model based on the DerSimonian–Laird method, which accounts for both within-study and between-study variability. Statistical heterogeneity was quantified using the I2 statistic, with values of 25%, 50%, and 75% representing low, moderate, and high heterogeneity, respectively.
Prespecified subgroup analyses were conducted to explore potential effect modification by route of administration (oral vs. subcutaneous). Sensitivity analyses using a leave-one-out approach were performed to assess the robustness of the pooled estimate. All statistical analyses were performed using Review Manager (RevMan) version 5.4 and R software (version 4.3.0) with the meta package. A two-sided p-value < 0.05 was considered statistically significant.
2.9. Patient and Public Involvement
No patients or members of the public were involved in the design, conduct, reporting, or dissemination of this research. It represents a secondary analysis of publicly available data from previously published randomized controlled trials.
3. Results
3.1. Study Selection
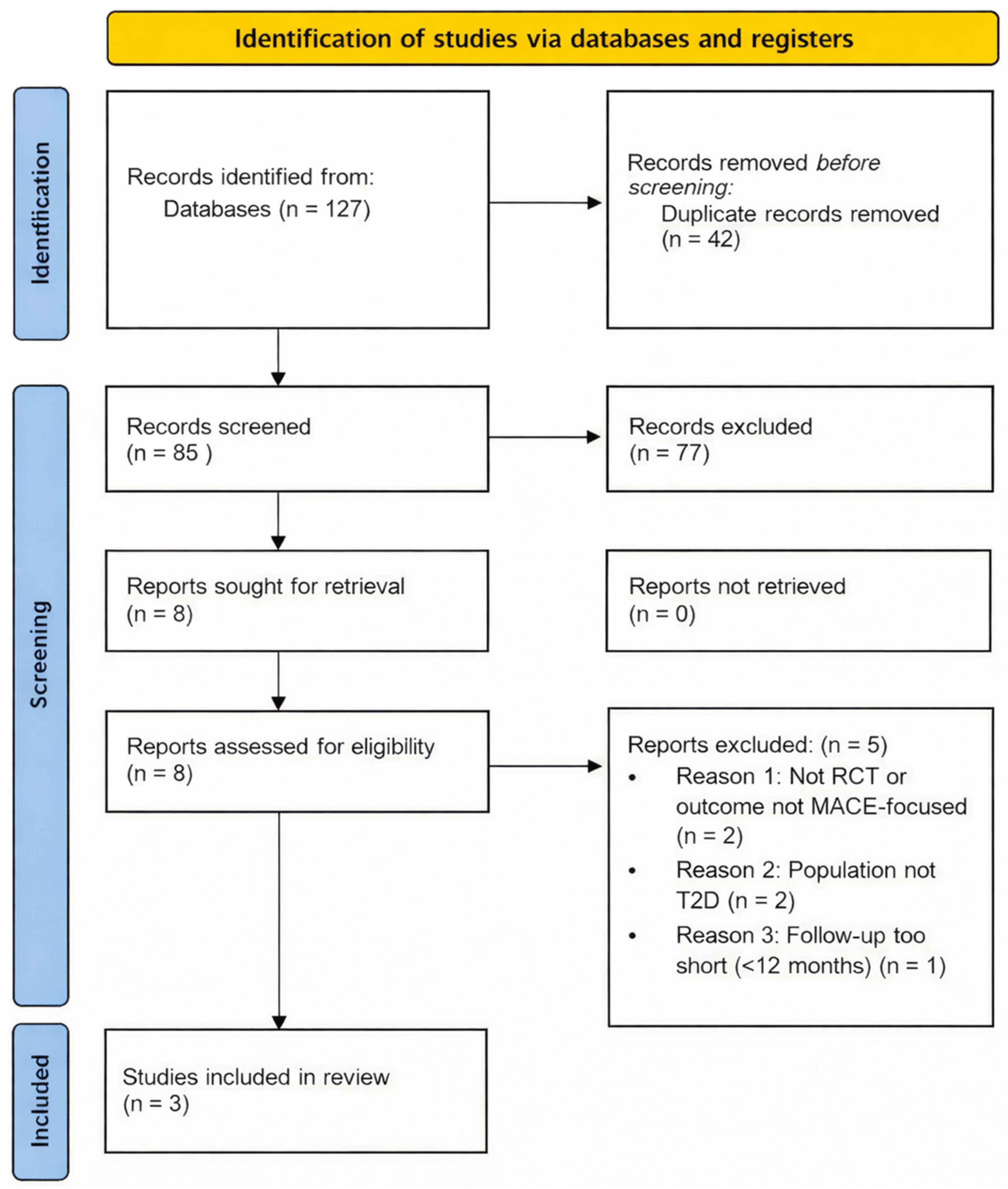
The initial search identified 127 potentially relevant articles. After removal of duplicates (n = 42), 85 records remained for title and abstract screening. Of these, 77 articles were excluded due to non-semaglutide interventions, lack of cardiovascular outcomes, non-randomized design, non–type 2 diabetes populations, or non-clinical studies. 8 full-text articles were assessed for eligibility. Of these, 5 were excluded due to the absence of a placebo control (n = 2), insufficient sample size (n = 1), non-cardiovascular primary outcome (n = 1), and duplicate publication (n = 1). Ultimately, 3 randomized controlled trials met all inclusion criteria and were included in the quantitative synthesis (Figure 1).
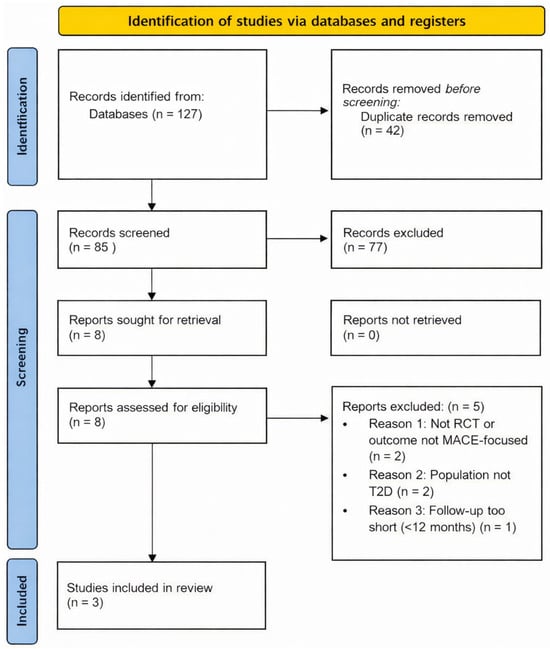
Figure 1.
PRISMA flow diagram for the selection of randomised clinical trials, included in this review. Note: MACE—major adverse cardiovascular events; T2D—type 2 diabetes; PRISMA—preferred reporting items for systematic reviews and meta-analyses; RCT—randomised clinical trial.
3.2. Characteristics of Included Studies
Three large-scale cardiovascular outcome trials enrolling a total of 16,130 participants were included in the meta-analysis (Table 1).

Table 1.
Selected RCTs and extracted data for the major adverse cardiovascular events (MACE) as a primary endpoint.
All three trials enrolled patients with type 2 diabetes at high cardiovascular risk. SOUL was the largest trial (n = 9650, representing 60% of the pooled sample) and had the longest median follow-up duration of 49.5 months, compared with 25 months in SUSTAIN-6 and 15.9 months in PIONEER-6. Table 2 presents the dose and route of administration of semaglutide, the baseline characteristics of the patients, and the medication history.

Table 2.
The baseline characteristics of the patients, included in the SUSTAIN 6, PIONEER 6, and SOUL trials [8,9,10].
3.3. Primary Outcome: Major Adverse Cardiovascular Events (MACE)
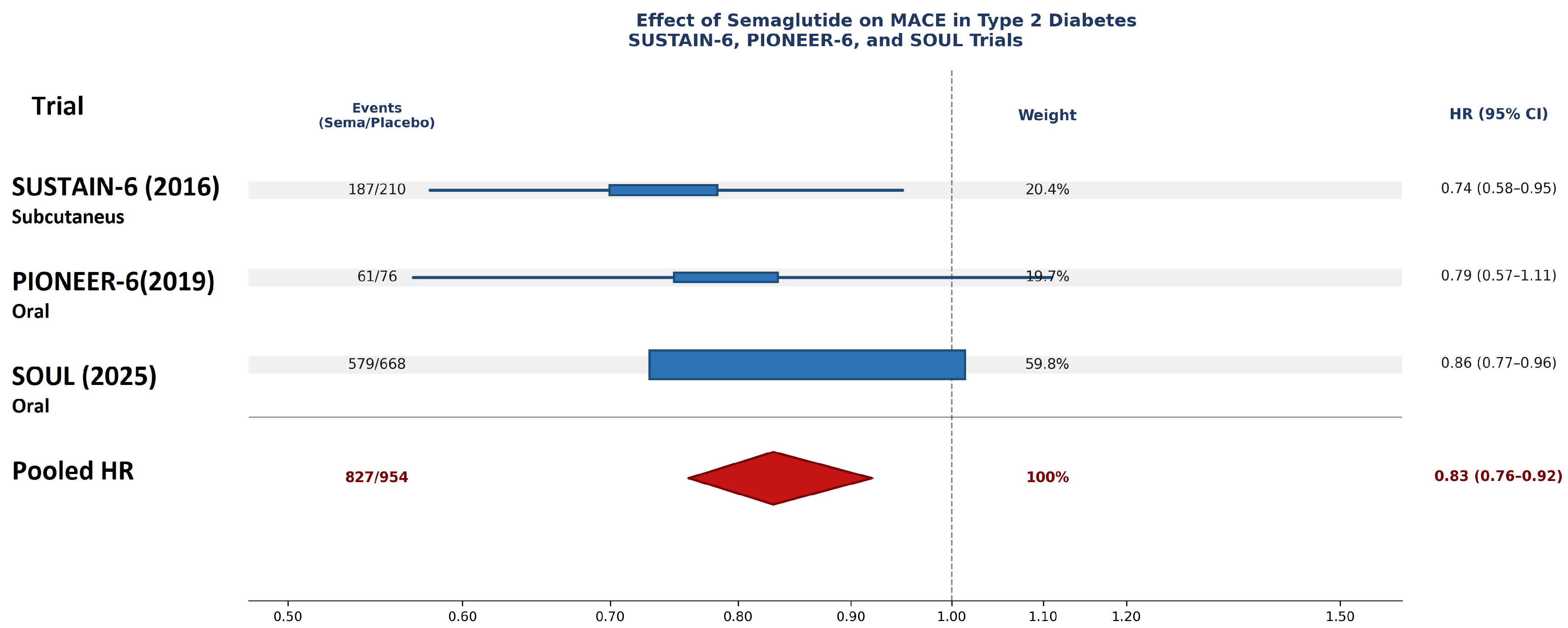
The pooled hazard ratio for MACE across the three trials was 0.83 (95% CI: 0.76–0.92; p < 0.001), corresponding to a statistically significant 17% relative risk reduction with semaglutide compared with placebo (Figure 2). The I2 statistic was 25%, indicating low statistical heterogeneity among the included trials. However, given the limited number of studies and the dominance of a single large trial, these findings should be interpreted with caution. The numbers of MACE events in the included trials are presented in Table 3. Based on pooled event rates, the estimated number needed to treat (NNT) is approximately 64 patients with T2D at high cardiovascular risk treated with semaglutide for about 3 years to prevent one major adverse cardiovascular event. This estimate represents an average effect across trials with differing follow-up durations and is largely influenced by the SOUL trial. This estimate represents an average effect across trials with substantially differing follow-up durations and should be interpreted accordingly, as NNT is inherently time-dependent.
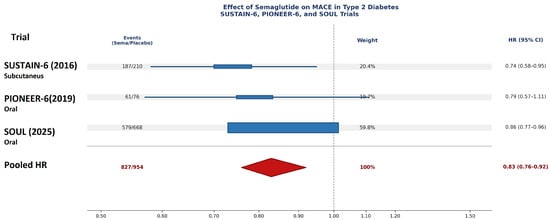
Figure 2.
Hazard ratios for MACE from individual trials [1,8,9] and pooled estimate for semaglutide in T2D (random-effects model).

Table 3.
Major Adverse Cardiovascular Events in the SUSTAIN 6, PIONEER 6, and SOUL trials [8,9,10].
3.4. Subgroup Analysis by Route of Administration
A prespecified subgroup analysis compared cardiovascular efficacy by route of administration:
Oral semaglutide (PIONEER-6 and SOUL):
- Pooled HR = 0.84 (95% CI: 0.76–0.93; I2 = 0%);
- 2 trials, n = 12,833 participants.
Subcutaneous semaglutide (SUSTAIN-6):
- HR = 0.74 (95% CI: 0.58–0.95);
- 1 trial, n = 3297 participants.
No statistically significant subgroup difference was observed (p-interaction = 0.42), suggesting that cardiovascular benefit is consistent regardless of administration route. It should be noted, however, that the subcutaneous subgroup comprises a single trial, precluding heterogeneity assessment and limiting the interpretability of the between-formulation comparison. The overlapping confidence intervals and low heterogeneity within the oral semaglutide subgroup support the internal consistency of the cardioprotective effect across oral formulations, while the directional concordance with SUSTAIN-6 provides qualitative support for a comparable benefit with subcutaneous administration.
3.5. Sensitivity Analysis
Leave-one-out sensitivity analyses were conducted to assess the robustness of the primary findings. When each trial was sequentially excluded, the pooled hazard ratio for MACE ranged from 0.76 to 0.85, with all estimates remaining statistically significant (p < 0.05):
- Excluding SUSTAIN-6: HR = 0.85 (95% CI: 0.77–0.93);
- Excluding PIONEER-6: HR = 0.83 (95% CI: 0.76–0.92);
- Excluding SOUL: HR = 0.76 (95% CI: 0.61–0.95).
These results confirm that the overall treatment effect is consistent and not unduly influenced by any single study, including the largest trial (SOUL).
3.6. Secondary Cardiovascular Outcomes
Individual components of the MACE composite and all-cause mortality were analyzed separately (Figure 2):
- Cardiovascular death: HR = 0.88 (95% CI: 0.76–1.02; I2 = 0%)
- Nonfatal myocardial infarction: HR = 0.74 (95% CI: 0.63–0.87; I2 = 0%)
- Nonfatal stroke: HR = 0.61 (95% CI: 0.47–0.80; I2 = 23%)
- All-cause mortality: HR = 0.85 (95% CI: 0.75–0.96; I2 = 0%)
Semaglutide demonstrated statistically significant reductions in nonfatal myocardial infarction, nonfatal stroke, and all-cause mortality, with a non-significant trend toward reduced cardiovascular death.
Although hospitalization for heart failure (HHF) is not a component of the 3-point MACE composite endpoint, it represents a clinically meaningful cardiovascular outcome in patients with type 2 diabetes. Data from SUSTAIN-6 and PIONEER-6 indicate a numerically neutral effect of semaglutide on HHF. In SUSTAIN-6, the rate of heart failure-related events was 2.4 versus 2.1 events per 100 patient-years in the semaglutide and placebo groups, respectively, whereas in PIONEER-6, rates were 1.3 versus 1.3 events per 100 patient-years, with no statistically significant difference between groups in either trial. A pooled post hoc analysis of both trials similarly demonstrated no significant effect of semaglutide on HHF (HR 1.03; 95% CI 0.75–1.40) [14]. In the SOUL trial, the composite endpoint included hospitalization or emergency department visit for heart failure and cardiovascular death, with events centrally adjudicated. In subgroup analyses (HFpEF, HFrEF, and unknown heart failure subtype), no statistically significant effect of semaglutide compared with placebo on HHF was observed [15].
Taken together, data across all three trials consistently indicate a neutral effect of semaglutide on HHF. These findings stand in contrast to SGLT-2 inhibitors, which have demonstrated consistent and clinically significant reductions in HHF across multiple large cardiovascular outcome trials. Accordingly, the neutral effect of semaglutide on HHF should be taken into consideration when selecting therapy in patients with established heart failure. Furthermore, the absence of HHF as a pre-specified, independently adjudicated primary or secondary endpoint in the available analyses represents a notable limitation that warrants consideration when interpreting these data.
3.7. Renal Outcomes
Pooled analysis showed no significant effect of semaglutide on composite renal endpoints (HR = 0.94; 95% CI: 0.85–1.05; I2 = 18%). SUSTAIN-6 demonstrated modest renal benefits, including delayed albuminuria progression, while PIONEER-6 and SOUL showed no significant renal effects. These inconsistent findings likely reflect the fact that none of these trials were designed or powered for renal outcomes, with varying follow-up durations and endpoint definitions. The subsequent dedicated renal outcomes trial FLOW (ClinicalTrials.gov: NCT03819153) has since confirmed clinically significant renal protection with semaglutide in patients with type 2 diabetes and chronic kidney disease (CKD), supporting a broader cardiorenal protective profile [16]. Regarding study population, FLOW was specifically designed to enroll patients with established CKD, defined by an eGFR of 25–75 mL/min/1.73 m2 and significant albuminuria, representing a population at substantially elevated cardio-renal risk [16]. In contrast, SUSTAIN-6 and PIONEER-6 enrolled predominantly high cardiovascular risk patients without a mandatory requirement for CKD at baseline. Notably, in post hoc analyses of SUSTAIN-6 and PIONEER-6, the effect of semaglutide on eGFR slope was more pronounced in patients with a baseline eGFR <60 mL/min/1.73 m2 (estimated treatment difference 1.06 mL/min/1.73 m2/year), suggesting a potentially greater nephroprotective benefit in those with pre-existing renal impairment; however, these analyses were not pre-specified and should be interpreted with caution.
According to endpoint design, FLOW incorporated a pre-specified composite renal primary endpoint comprising CKD, a sustained 50% reduction in eGFR from baseline, or death from kidney-related or cardiovascular causes, whereas renal outcomes in SUSTAIN-6 and PIONEER-6 were captured only as secondary or post hoc endpoints, limiting their inferential weight. FLOW was designed and powered to detect renal events, whereas the earlier trials were not. Regarding follow-up duration and statistical power, FLOW achieved a median follow-up of 3.4 years with sufficient event accumulation to support pre-specified renal endpoint analysis, whereas SUSTAIN-6 and PIONEER-6 had shorter follow-up periods and accrued fewer renal events, further precluding robust renal efficacy conclusions from those trials.
3.8. Safety and Adverse Events
Serious adverse events occurred at similar rates in the semaglutide and placebo groups across all trials (pooled risk ratio = 1.02; 95% CI: 0.96–1.08). Gastrointestinal adverse events, including nausea, vomiting, and diarrhea, were more common with semaglutide (approximately 15–25% vs. 5–10% with placebo), consistent with the known side effect profile of GLP-1 receptor agonists. Treatment discontinuation rates due to adverse events were modestly higher with semaglutide (7–11%) compared to placebo (3–5%), primarily driven by gastrointestinal intolerance. No unexpected safety signals were identified.
4. Discussion
This systematic review and meta-analysis demonstrate that semaglutide significantly reduces major adverse cardiovascular events in patients with T2D at high cardiovascular risk. The pooled hazard ratio of 0.83 (95% CI: 0.76–0.92) represents a 17% relative risk reduction in MACE, with low heterogeneity (I2 = 25%) supporting consistency across trials and formulations.
4.1. Interpretation and Comparison with Existing Evidence
Our findings extend previous class-wide meta-analyses of GLP-1 receptor agonists, which reported pooled MACE reductions of approximately 12–14% [17,18]. The cardiovascular benefit of semaglutide appears comparable to that of SGLT2 inhibitors (pooled HR ~0.89–0.93) [19,20], though the latter demonstrate greater efficacy for heart failure and renal outcomes. Analysis of individual MACE components revealed particularly robust reductions in nonfatal myocardial infarction (HR 0.74) and nonfatal stroke (HR 0.61), suggesting that semaglutide’s primary mechanism involves atherosclerosis stabilization rather than direct cardiac effects.
The cardiovascular protection likely reflects multiple mechanisms beyond glycemic control, including improved endothelial function, reduced inflammation, favorable lipid modulation, blood pressure reduction, and substantial weight loss (typically 5–15% of body weight) [21,22,23,24]. Importantly, subgroup analyses from individual trials showed consistent MACE reduction across baseline HbA1c levels, indicating glucose-independent cardioprotective effects.
4.2. Formulation and Safety Considerations
Our subgroup analysis found no significant difference between oral and subcutaneous formulations (p-interaction = 0.42), with overlapping confidence intervals supporting equivalent cardiovascular efficacy. This provides clinical flexibility based on patient preference, as many individuals prefer oral over injectable therapy [25]. The favorable benefit-risk profile, with a significant MACE reduction and manageable gastrointestinal side effects, supports broader utilization in high-risk populations.
Regarding safety, early concerns about diabetic retinopathy complications observed in SUSTAIN-6 [26] were not replicated in PIONEER-6 and SOUL, with post hoc analyses suggesting this may have reflected rapid glycemic improvement in patients with pre-existing severe retinopathy rather than a direct drug toxic effect [27], with diabetic retinopathy progression appearing low when advanced cases are excluded [28]. Ongoing clinical trials, such as FOCUS (NCT03811561), are examining the long-term ocular safety of semaglutide in patients with type 2 diabetes [28]. Our pooled analysis found no significant renal benefit (HR 0.94; 95% CI: 0.85–1.05), contrasting with SGLT2 inhibitors but not indicating harm.
4.3. Clinical Implications
According to cardiovascular efficacy, semaglutide demonstrates a significant reduction in the risk of MACE in patients with type 2 diabetes and established atherosclerotic cardiovascular disease, with an approximately 19% relative risk reduction versus placebo (HR 0.81; 95% CI 0.74–0.88) [3]. Comparable benefits have been observed with other GLP-1RAs, including liraglutide (LEADER: HR 0.87; 95% CI 0.78–0.97) and dulaglutide (REWIND: HR 0.88; 95% CI 0.79–0.99). Indirect comparisons and network meta-analyses suggest that semaglutide may be numerically superior to dulaglutide with respect to all-cause mortality, myocardial infarction, stroke, and HHF (HR for HHF 0.88; 95% CI 0.85–0.91) [29]. However, in the absence of direct head-to-head randomised trials, such comparisons should be interpreted with caution.
These findings support semaglutide as a preferred glucose-lowering agent for patients with T2D and established cardiovascular disease or multiple risk factors, aligning with current ADA and ESC guidelines [30,31]. The consistency of benefit across formulations allows individualized treatment selection. Healthcare providers should consider semaglutide for integrated glycemic and cardiovascular risk management in high-risk diabetic populations.
4.4. Strengths and Limitations
Key strengths: (1) extends prior semaglutide-specific meta-analyses by incorporating results from the SOUL trial; (2) all included trials were large, high-quality RCTs, with low risk of bias and independent event adjudication; (3) low statistical heterogeneity across trials, suggesting consistency of treatment effect; and (4) robust leave-one-out sensitivity analyses confirming directional concordance of findings across all iterations.
Limitations of our study include the following: (1) the small number of eligible trials (n = 3), which precludes formal assessment of publication bias via funnel plot asymmetry or Egger’s test—methods that require a minimum of ten studies for reliable interpretation; this limitation is partially mitigated by prospective trial registration and independent regulatory oversight; (2) enrollment of predominantly high-cardiovascular-risk patients across all three trials, which may limit the generalizability of findings to lower-risk populations; (3) industry sponsorship of all included trials, though rigorous regulatory standards, independent data monitoring committees, and pre-specified statistical analysis plans minimize the risk of systematic bias; (4) variability in follow-up duration (15.9–49.5 months), which may influence observed event rates and limit direct comparability of effect sizes across trials; (5) the absence of individual patient-level data, precluding detailed subgroup analyses by age, renal function, baseline cardiovascular risk, or concomitant therapy; and (6) as a quantitative synthesis of previously published trials, this analysis largely consolidates existing evidence rather than generating novel mechanistic insights, a limitation inherent to meta-analyses comprising a small number of mature trials.
Despite the consistency of direction across all leave-one-out iterations, the pooled hazard ratio shifted meaningfully upon exclusion of SOUL (HR 0.76; 95% CI 0.61–0.95), compared with exclusion of SUSTAIN-6 (HR 0.85) or PIONEER-6 (HR 0.83). Given that SOUL contributes approximately 60% of the total study population (9650 of 16,130 participants), this finding indicates that the pooled estimate is substantially driven by the largest trial, and that the overall effect size should be interpreted with caution. This moderating influence likely reflects SOUL’s more contemporary background therapy—including substantially higher SGLT2 inhibitor use (26.9%) compared with earlier trials—which may have attenuated the incremental cardiovascular benefit of semaglutide against an already optimised pharmacological background. This degree of statistical dependence on a single study is an inherent limitation of meta-analyses comprising a small number of trials (n = 3) and does not necessarily reflect true heterogeneity in treatment effect, but rather the disproportionate weight of the dominant study. Importantly, all three individual trials consistently demonstrated a directionally concordant reduction in MACE, and no trial reported a neutral or harmful effect, which provides qualitative support for the robustness of the overall conclusion despite quantitative instability in the pooled estimate.
4.5. Future Directions
Important questions remain for future investigation: (1) head-to-head trials comparing semaglutide with SGLT2 inhibitors or evaluating combination therapy; (2) studies in primary prevention populations with lower baseline cardiovascular risk; (3) longer-term follow-up to assess durability of benefits and late safety signals; (4) mechanistic studies using advanced imaging to elucidate atherosclerotic plaque stabilization effects; and (5) real-world effectiveness studies in diverse clinical practice settings.
5. Conclusions
Semaglutide significantly reduces major adverse cardiovascular events by 17% in patients with T2D at high cardiovascular risk, with consistent benefit across oral and subcutaneous formulations and low heterogeneity supporting reliability. The favorable safety profile and robust evidence base support semaglutide as a preferred therapeutic option for integrated glycemic and cardiovascular risk management in high-risk diabetic populations. Healthcare providers should consider semaglutide in either formulation for patients with T2D and established cardiovascular disease or multiple cardiovascular risk factors, with treatment decisions guided by patient preferences and clinical context.
Author Contributions
G.A.: Investigation, Project administration, Formal analysis, Conceptualization. V.M.: Investigation, Validation. B.D.: Investigation, Supervision. G.S.: Writing—review and editing, Supervision. All authors have read and agreed to the published version of the manuscript.
Funding
This study is financed by the European Union-NextGenerationEU, through the National Recovery and Resilience Plan of the Republic of Bulgaria, project № BG-RRP-2.004-0003.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
During the preparation of this work, the authors used Open AI GPT-4 series-to complete the grammar and spell checks. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Conflicts of Interest
The authors declare no commercial or financial relationships that could be construed as a potential conflict of interest.
Abbreviations
| CKD | chronic kidney diseases |
| CVD | cardio-vascular diseases |
| eGFR | estimated glomerular filtration rate |
| GLP-1 RAs | Glucagon-like peptide-1 receptor agonists |
| HR MACE | Hazard Ratio for Major Adverse Cardiovascular Events |
| HHF | hospitalization for heart failure |
| HFpEF | heart failure with preserved ejection fraction |
| HFrEF | heart failure with reduced ejection fraction |
| MACE | major adverse cardiovascular events |
| MI | myocardial infarction |
| PRISMA | preferred reporting items for systematic reviews and meta-analyses |
| RCTs | randomized clinical trials |
| SGLT2-i | sodium-glucose co-transporter-2 inhibitors |
| T2D | type 2 diabetes |
References
- McGuire, D.K.; Marx, N.; Mulvagh, S.L.; Deanfield, J.E.; Inzucchi, S.E.; Pop-Busui, R.; Mann, J.F.E.; Emerson, S.S.; Poulter, N.R.; Engelmann, M.D.M.; et al. Oral Semaglutide and Cardiovascular Outcomes in High-Risk Type 2 Diabetes. N. Engl. J. Med. 2025, 392, 2001–2012. [Google Scholar] [CrossRef]
- Marx, N.; Deanfield, J.E.; Mann, J.F.E.; Arechavaleta, R.; Bain, S.C.; Bajaj, H.S.; Bayer Tanggaard, K.; Birkenfeld, A.L.; Buse, J.B.; Davicevic-Elez, Z.; et al. Oral Semaglutide and Cardiovascular Outcomes in People with Type 2 Diabetes, According to SGLT2i Use: Prespecified Analyses of the SOUL Randomized Trial. Circulation 2025, 151, 1639–1650. [Google Scholar] [CrossRef]
- Sadraei, S.; Aarabi, A.; Rajai Firouzabadi, S.; Alinejadfard, M.; Mohammadi, I.; Jolfayi, A.G. Cardiovascular Benefits of Semaglutide: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. BMC Cardiovasc. Disord. 2025, 25, 881. [Google Scholar] [CrossRef]
- Cleto, A.S.; Schirlo, J.M.; Beltrame, M.; Gomes, V.H.O.; Acras, I.H.; Neiverth, G.S.; Silva, B.B.; Juliatto, B.M.S.; Machozeki, J.; Martins, C.M. Semaglutide Effects on Safety and Cardiovascular Outcomes in Patients with Overweight or Obesity: A Systematic Review and Meta-Analysis. Int. J. Obes. 2025, 49, 21–30. [Google Scholar] [CrossRef]
- Rebelo, T.G.; De Araujo Paysano, M.L.B.; Matheus, G.T.F.U.; Ribeiro, D.M.; Said, T.B.; Kelly, F.A. Effects of Oral Semaglutide on Cardiovascular Outcomes: A Systematic Review and Meta-Analysis. Int. J. Cardiol. 2025, 440, 133683. [Google Scholar] [CrossRef]
- Sillassen, C.D.B.; Petersen, J.J.; Faltermeier, P.; Yucel, D.; Siddiqui, F.; Andersen, R.K.; Graever, L.; Bjerg, J.L.; Kamp, C.B.; Grand, J.; et al. The Adverse Effects Associated with Semaglutide Use in Patients at Increased Risk of Cardiovascular Events: A Systematic Review with Meta-Analysis and Trial Sequential Analysis. BMC Med. 2025, 23, 654. [Google Scholar] [CrossRef]
- Lincoff, A.M.; Brown-Frandsen, K.; Colhoun, H.M.; Deanfield, J.; Emerson, S.S.; Esbjerg, S.; Hardt-Lindberg, S.; Hovingh, G.K.; Kahn, S.E.; Kushner, R.F.; et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. N. Engl. J. Med. 2023, 389, 2221–2232. [Google Scholar] [CrossRef] [PubMed]
- Marso, S.P.; Bain, S.C.; Consoli, A.; Eliaschewitz, F.G.; Jódar, E.; Leiter, L.A.; Lingvay, I.; Rosenstock, J.; Seufert, J.; Warren, M.L.; et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 1834–1844. [Google Scholar] [CrossRef] [PubMed]
- Husain, M.; Birkenfeld, A.L.; Donsmark, M.; Dungan, K.; Eliaschewitz, F.G.; Franco, D.R.; Jeppesen, O.K.; Lingvay, I.; Mosenzon, O.; Pedersen, S.D.; et al. Oral Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N. Engl. J. Med. 2019, 381, 841–851. [Google Scholar] [CrossRef] [PubMed]
- McGuire, D.K.; Busui, R.P.; Deanfield, J.; Inzucchi, S.E.; Mann, J.F.E.; Marx, N.; Mulvagh, S.L.; Poulter, N.; Engelmann, M.D.M.; Hovingh, G.K.; et al. Effects of Oral Semaglutide on Cardiovascular Outcomes in Individuals with Type 2 Diabetes and Established Atherosclerotic Cardiovascular Disease and/or Chronic Kidney Disease: Design and Baseline Characteristics of SOUL, a Randomized Trial. Diabetes Obes. Metab. 2023, 25, 1932–1941. [Google Scholar] [CrossRef]
- Mellbin, L.G.; Bhatt, D.L.; David, J.-P.; Ekström, K.; Petrie, M.C.; Rasmussen, S.; Vilsbøll, T. Semaglutide and Cardiovascular Outcomes by Baseline HbA1c in Diabetes: The SUSTAIN 6 and PIONEER 6 Trials. Eur. Heart J. 2024, 45, 1371–1374. [Google Scholar] [CrossRef] [PubMed]
- Nauck, M.A.; Quast, D.R. Cardiovascular Safety and Benefits of Semaglutide in Patients with Type 2 Diabetes: Findings from SUSTAIN 6 and PIONEER 6. Front. Endocrinol. 2021, 12, 645566. [Google Scholar] [CrossRef] [PubMed]
- Bain, S.C.; Mosenzon, O.; Arechavaleta, R.; Bogdański, P.; Comlekci, A.; Consoli, A.; Deerochanawong, C.; Dungan, K.; Faingold, M.C.; Farkouh, M.E.; et al. Cardiovascular Safety of Oral Semaglutide in Patients with Type 2 Diabetes: Rationale, Design and Patient Baseline Characteristics for the PIONEER 6 Trial. Diabetes Obes. Metab. 2019, 21, 499–508. [Google Scholar] [CrossRef]
- Zhou, J.; Husain, M.; Li, Y.; Liu, W.; Shen, Z.; Vilsbøll, T.; Ge, J. Effect of semaglutide versus placebo on cardiorenal outcomes by prior cardiovascular disease and baseline body mass index: Pooled post hoc analysis of SUSTAIN 6 and PIONEER 6. Diabetes Obes. Metab. 2025, 27, 5706–5715. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pop-Busui, R.; Rasmussen, S.; Deanfield, J.E.; Buse, J.B.; Marx, N.; Mulvagh, S.L.; Inzucchi, S.E.; Mann, J.F.E.; Emerson, S.S.; Poulter, N.R.; et al. Oral Semaglutide and Heart Failure Outcomes in Persons with Type 2 Diabetes: A Secondary Analysis of the SOUL Randomized Clinical Trial. JAMA Intern. Med. 2026. Online ahead of print. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Perkovic, V.; Tuttle, K.R.; Rossing, P.; Mahaffey, K.W.; Mann, J.F.E.; Bakris, G.; Baeres, F.M.M.; Idorn, T.; Bosch-Traberg, H.; Lausvig, N.L.; et al. Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes. N. Engl. J. Med. 2024, 391, 109–121. [Google Scholar] [CrossRef]
- Kristensen, S.L.; Rørth, R.; Jhund, P.S.; Docherty, K.F.; Sattar, N.; Preiss, D.; Køber, L.; Petrie, M.C.; McMurray, J.J.V. Cardiovascular, Mortality, and Kidney Outcomes with GLP-1 Receptor Agonists in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis of Cardiovascular Outcome Trials. Lancet Diabetes Endocrinol. 2019, 7, 776–785. [Google Scholar] [CrossRef] [PubMed]
- Sattar, N.; Lee, M.M.Y.; Kristensen, S.L.; Branch, K.R.H.; Del Prato, S.; Khurmi, N.S.; Lam, C.S.P.; Lopes, R.D.; McMurray, J.J.V.; Pratley, R.E.; et al. Cardiovascular, Mortality, and Kidney Outcomes with GLP-1 Receptor Agonists in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis of Randomised Trials. Lancet Diabetes Endocrinol. 2021, 9, 653–662. [Google Scholar] [CrossRef]
- Zelniker, T.A.; Wiviott, S.D.; Raz, I.; Im, K.; Goodrich, E.L.; Bonaca, M.P.; Mosenzon, O.; Kato, E.T.; Cahn, A.; Furtado, R.H.M.; et al. SGLT2 Inhibitors for Primary and Secondary Prevention of Cardiovascular and Renal Outcomes in Type 2 Diabetes: A Systematic Review and Meta-Analysis of Cardiovascular Outcome Trials. Lancet 2019, 393, 31–39. [Google Scholar] [CrossRef]
- McGuire, D.K.; Shih, W.J.; Cosentino, F.; Charbonnel, B.; Cherney, D.Z.I.; Dagogo-Jack, S.; Pratley, R.; Greenberg, M.; Wang, S.; Huyck, S.; et al. Association of SGLT2 Inhibitors with Cardiovascular and Kidney Outcomes in Patients with Type 2 Diabetes: A Meta-Analysis. JAMA Cardiol. 2021, 6, 148. [Google Scholar] [CrossRef]
- Nauck, M.A.; Quast, D.R.; Wefers, J.; Meier, J.J. GLP-1 Receptor Agonists in the Treatment of Type 2 Diabetes—State-of-the-Art. Mol. Metab. 2021, 46, 101102. [Google Scholar] [CrossRef]
- Marso, S.P.; Baeres, F.M.M.; Bain, S.C.; Goldman, B.; Husain, M.; Nauck, M.A.; Poulter, N.R.; Pratley, R.E.; Thomsen, A.B.; Buse, J.B. Effects of Liraglutide on Cardiovascular Outcomes in Patients with Diabetes with or Without Heart Failure. J. Am. Coll. Cardiol. 2020, 75, 1128–1141. [Google Scholar] [CrossRef]
- Rakipovski, G.; Rolin, B.; Nøhr, J.; Klewe, I.; Frederiksen, K.S.; Augustin, R.; Hecksher-Sørensen, J.; Ingvorsen, C.; Polex-Wolf, J.; Knudsen, L.B. The GLP-1 Analogs Liraglutide and Semaglutide Reduce Atherosclerosis in ApoE−/− and LDLr−/− Mice by a Mechanism That Includes Inflammatory Pathways. JACC Basic Transl. Sci. 2018, 3, 844–857. [Google Scholar] [CrossRef]
- Wilding, J.P.H.; Batterham, R.L.; Calanna, S.; Davies, M.; Van Gaal, L.F.; Lingvay, I.; McGowan, B.M.; Rosenstock, J.; Tran, M.T.D.; Wadden, T.A.; et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N. Engl. J. Med. 2021, 384, 989–1002. [Google Scholar] [CrossRef] [PubMed]
- Thieu, V.T.; Robinson, S.; Kennedy-Martin, T.; Boye, K.S.; Garcia-Perez, L.-E. Patient Preferences for Glucagon-like Peptide-1 Receptor–Agonist Treatment Attributes. Patient Prefer. Adherence 2019, 13, 561–576. [Google Scholar] [CrossRef]
- Aroda, V.R.; Erhan, U.; Jelnes, P.; Meier, J.J.; Abildlund, M.T.; Pratley, R.; Vilsbøll, T.; Husain, M. Safety and Tolerability of Semaglutide across the SUSTAIN and PIONEER Phase IIIa Clinical Trial Programmes. Diabetes Obes. Metab. 2023, 25, 1385–1397. [Google Scholar] [CrossRef]
- Vilsbøll, T.; Bain, S.C.; Leiter, L.A.; Lingvay, I.; Matthews, D.; Simó, R.; Helmark, I.C.; Wijayasinghe, N.; Larsen, M. Semaglutide, Reduction in Glycated Haemoglobin and the Risk of Diabetic Retinopathy. Diabetes Obes. Metab. 2018, 20, 889–897. [Google Scholar] [CrossRef]
- Muzurović, E.; Zečević, K.; Maggio, V.; El Tanani, M.; Rizzo, M. Semaglutide and the Retina: Weighing Evidence against Concern. J. Diabetes Its Complicat. 2025, 39, 109148. [Google Scholar] [CrossRef]
- Derington, C.G.; Sarwal, A.; Wei, G.; Hartsell, S.E.; Throolin, M.; Singh, R.; Nevers, M.R.; Zhang, C.; Katkam, N.; Takyi, A.; et al. Liraglutide vs Semaglutide vs Dulaglutide in Veterans with Type 2 Diabetes. JAMA Netw. Open 2025, 8, e2537297. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cosentino, F.; Grant, P.J.; Aboyans, V.; Bailey, C.J.; Ceriello, A.; Delgado, V.; Federici, M.; Filippatos, G.; Grobbee, D.E.; Hansen, T.B.; et al. 2019 ESC Guidelines on Diabetes, Pre-Diabetes, and Cardiovascular Diseases Developed in Collaboration with the EASD. Eur. Heart J. 2020, 41, 255–323. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association Professional Practice Committee; ElSayed, N.A.; Aleppo, G.; Bannuru, R.R.; Bruemmer, D.; Collins, B.S.; Ekhlaspour, L.; Gaglia, J.L.; Hilliard, M.E.; Johnson, E.L.; et al. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Care in Diabetes—2024. Diabetes Care 2024, 47, S158–S178. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.