Abstract
Marine isopods are the ancestors of the harmless herbivorous woodlouse species often found in piles of leaves. In contrast to woodlice, marine isopods of the family Cirolanidae (cirolanid isopods) are scavengers and predatory carnivores that form swarms and can cause damage to fishing and aquaculture industries. Furthermore, these animals are known to bite swimming and diving humans and therefore may have negative effects on recreational activities in areas where swarms form. One cirolanid isopod species, Cirolana harfordi, displays social behaviour, an attribute that may facilitate the formation of swarms. This species gives live birth, a highly unusual mode of reproduction for an invertebrate and isopod. The rate of viviparous reproduction in C. harfordi is sped up by warmer conditions, indicating the threat that cirolanid isopods pose to ocean resources may intensify with global warming.
1. Anatomy and Predatory Capabilities
The family Cirolanidae contains 68 genera and over 590 species of isopods, most of whom live in marine or estuarine environments [1]. The family was named after its type genus Cirolana and members of the Cirolanidae family are often referred to as cirolanid isopods or cirolanids [2,3]. Cirolanids are voracious scavengers and hunters [2] and have evolved anatomical features that make them extremely capable of finding and consuming food. They are equipped with two pairs of antennae that contain receptors that can detect olfactory molecules from food and prey, and the paddle-shaped appendages on the ventral portion of their pleon allow them to be strong swimmers. For example, the cirolanid species Natatolana borealis can swim at speeds in the range 0.3 to 11.2 m per minute and can sense food 190 m away [4].
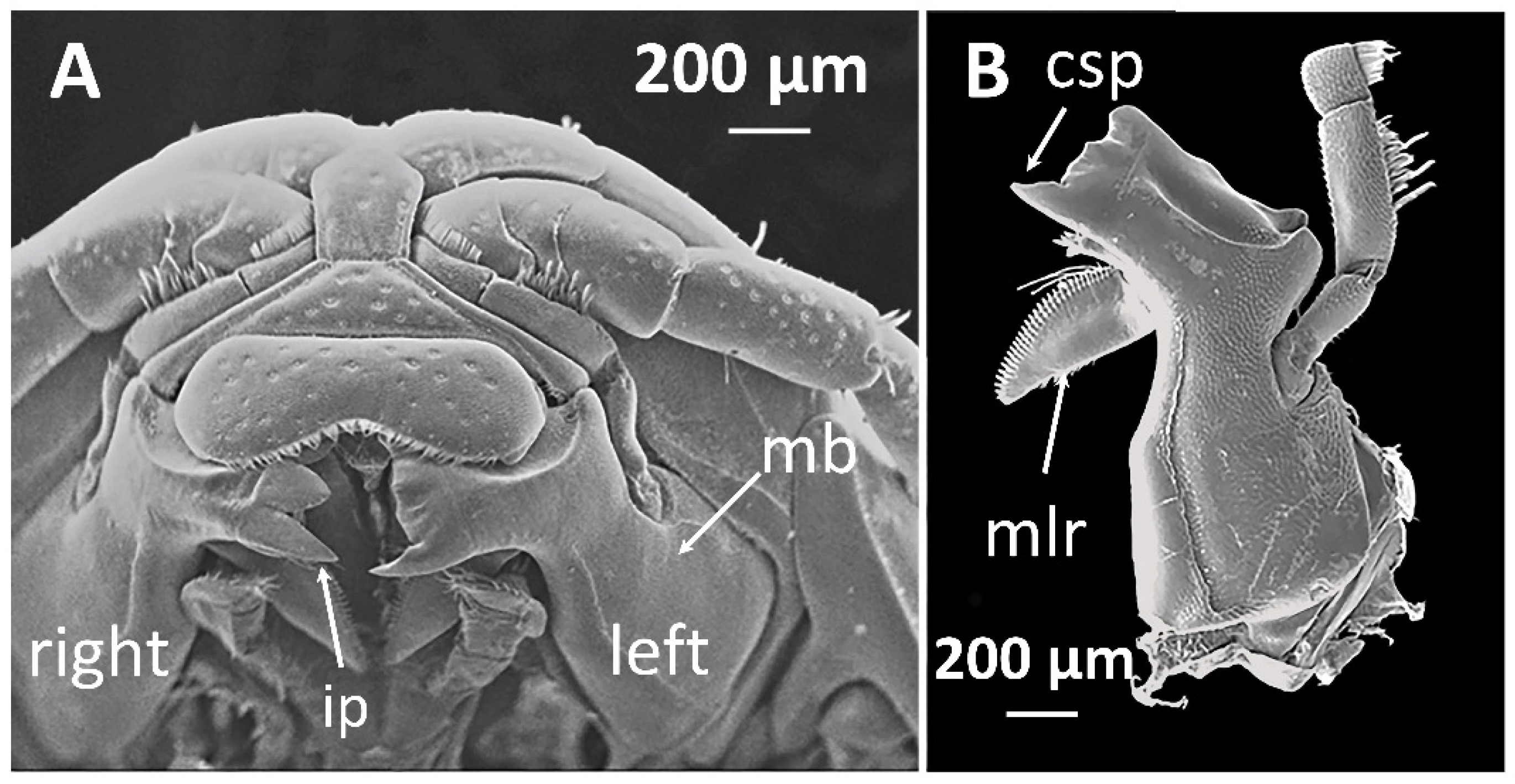
Like most isopods, cirolanids have a dorso-ventrally flattened body which is divided into the cephalon (head), pereon (thorax) and pleon (tail), see Figure 1. The pereon in adults houses seven pairs of walking legs, named pereopods. The pereopods terminate in a sharp claw-like spine called a dactylus, and isopods use their dactyli to grasp their prey in a strong grip while attacking and feeding. The underside of the pleon contains five pairs of paddle-shaped pleopods which are used for swimming and to propel water over the respiratory tissues. The mouthparts of cirolanids are adapted for carnivorous feeding. The innermost mouthparts are mandibles, and each of the pair of mandibles is equipped with a flesh-shearing blade-like cutting edge and a grinding and ripping protrusion called the molar process (Figure 2). Other mouthparts are equipped with long spine-like teeth for grasping and the outermost mouthparts are spoon-shaped so that they can push food into the mouth [5].

Figure 1.
A sample of the species Bathynomus pelor which was caught in a baited trap from an oil rig on the NW Shelf of Australia [5]: (A) lateral view, (B) ventral view, (C) dorsal view, (D) ventral view.
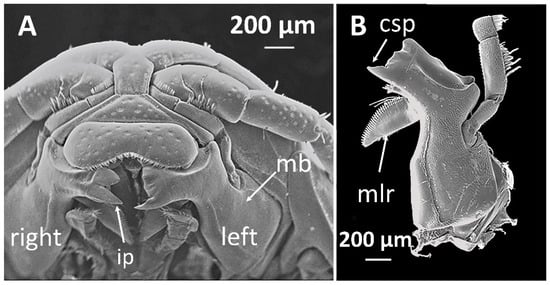
Figure 2.
The mandibles of Cirolana harfordi specimens: (A) mandibles in situ, the mouthparts superficial to the mandible have been removed, and (B) left mandible, ventral view. csp, cusp; ip, incisor process; mb, mandible; mlr, molar process. Reproduced with permission, from [6].
In their natural environment cirolanids are known to eat a large variety of foods, including fish, crustaceans, and other invertebrates such as polychaetes and nematodes, as well as sometimes eating plant material [7,8]. The deep-sea cirolanid Bathynomus pelor has even shown a voracious taste for cooked bacon, which demonstrates this species’ willingness to devour unfamiliar food [8]. Several cirolanid species are known to attack large live animals such as fish, including sharks, by first clasping on to their prey, then entering the body via the operculum openings over the gills, the eye sockets or the anus, and then eating the soft tissue [9,10,11]. When these animals attack in swarms it is often only the skeleton and skin of the fish that they leave behind. There are several reports of cirolanid isopods scavenging on dead humans which describe a common technique used by these isopods: they chew a hole in the skin and after entry into the subcutaneous layer eat the soft subcutaneous tissue, leaving behind the skin (except for the entry holes that resemble shotgun wounds) and skeleton [12,13,14,15,16].
2. Effects on Fishing and Aquaculture Industries
Several species of Cirolanidae demonstrate diel vertical migration, a term given to zooplankton that bury or hide in the substrate during the day and emerge at night to move higher in the water to hunt [17]. This pattern of behaviour explains why some fishing industries have suffered from nocturnal attacks by cirolanids. In 1982 a study set out to determine whether cage-reared fishes close to the shore would be attacked by crustaceans. The researchers placed juvenile and adult kelpfish of the species Heferostichus rosfratu in cages made out of PVC piping and 1.3 cm mesh, which were weighted down to the seabed close to a kelp forest near Santa Catalina Island, California [18]. Juveniles placed in this environment survived for numerous days; however, adults were usually killed by the cirolanid Cirolana diminuta, a species that grows to a maximum of 10.5 mm long [18,19]. This species displays diel vertical migration behaviour with its characteristic nocturnal hunting pattern [18]. In the attacks on the adult kelpfish, C. diminuta individuals entered the fishes’ gill area and damaged those, and in many cases they entered the body via the anus and ate the gonads and liver. SCUBA divers observed the net cages at night to gain more information on the pattern of the attacks and discovered that the fish were first attacked by swarms of the small (1 mm long) ostracod species Vargula tsujii and then by swarms of C. dimunata. The authors proposed that the smaller bites by the ostracods released odours into the sea that attracted the cirolanids which inflicted the fatal injuries [18].
Large numbers of Natatolana woodjonesi have been observed in experiments that deployed video cameras baited with sardines close to Blue Fin Tuna aquaculture nets close to Port Lincon in South Australia [20]. Sardines are a major component of the food provided to cultured Blue Fin Tuna and uneaten food will fall to the benthos. While scavengers such as N. woodjonesi are reducing the harmful effects of rotting food on the cultured fish and environment, care should be taken to minimise surplus cultured fish food lying on the seabed as it may attract cirolanid isopods which may attack the farmed fish [20].
During a 6-month period in 2007, 45% of the Sea Bass (Lates calcerifer) juveniles (fingerlings) that were being reared in an aquaculture cage farm on the South West Coast of India were killed and extensively eaten by Cirolana fluviatilis [21]. Sanil and colleagues noted that the nets were fouled with organic matter and this appeared to provide an attractive substrate for numerous C. fluviatilis individuals to take up residence and effectively use the cages of young fish as their pantry [21].
In addition to being a risk to the economic income of net aquaculture fisheries, cirolanids are also known to attack the harvest of set-net and line fishing industries. In set-net fishing a net is suspended in the water with floats and anchored to the sea floor, often by weights, to form a vertical curtain of net that catches fish as they attempt to swim through it. This technique is also called stationary gillnetting. The holes in the nets are big enough to let the heads of many fish through but not their bodies. As the fish try to back out of the net, the strands get caught behind the gill covers (gill operculum). The fish hanging from the net are captured prey for isopod swarms, who often eat out visible sections of the fish that prevent it being able to be sold commercially. In a study on the effects of marine isopods on line and net fishing in the Southern Aegean Sea, Turkey, the fishers consulted unanimously reported that losses due to isopods feeding on the catch were higher at night than during the day [22]. The amount of fish spoiled by isopods increased over the period 2014–2018 [22]. Natatolana neglecta individuals attacked the outside of the fishes’ bodies as well as entering the fishes’ bodies through the eyes and the gill openings at the edge of the opercula and feeding on the flesh. It is not unusual for these cirolanids to largely skeletonise a fish if they prey on it [10]. Set nets are often left out overnight, but to try and avoid strikes by N. neglecta, some fishermen have changed to setting nets at sunset and only leaving them out for 2 h, a strategy that will reduce the catch [10]. It has been proposed that aquaculture in the region of Güllük Bay, on the Turkish coast of the southern Aegean Sea, has been responsible for attracting more isopods to the nearby waters [10]. In 2018 the estimated annual economic loss to each set-net fisher in the Aegean sea due to spoilage by marine isopods (predominantly of the species N. neglecta) was 10,357 ± 5799 Turkish Lira [22]. In addition to the danger posed by cirolanids to teleost fishing, cirolanid isopods in swarms frequently attack sharks. Two shark fishing companies were forced into closure due to cirolanid isopods attacking and rendering their catch unsaleable; the first instance happened to a South Australian fishery in the 1930s and the second a Florida fishery in the 1970s [23].
There is also a risk from cirolanids posed to the invertebrate harvesting industries, both from cirolanids eating the harvest and by eating the bait needed to catch the harvest. Cirolana fluviatilis is found in estuaries in South Africa [14] and Southern India [21] and it burrows into muddy sediment [14]. Individuals of this species have been collected from the baits (prawn and fish meat) attached to crab nets. C. fluviatilis is a pest to the crab fishing industry in Southern India as populations of this cirolanid can completely eat all of the bait attached to the nets [24]. In addition to fish and crustaceans, C. fluviatilis has been reported to eat human bodies floating in water [14]. This is another cirolanid species that forms attacking swarms and can quickly strip the flesh off their prey [14].
3. Cirolanids and Deep-Sea Fishing
Most cirolanid isopods species do not normally venture lower than 200 m [2,19]; however, two genera, Natatolana and Bathynomus, contain some species which have been found at depths of thousands of metres [2]. This means that in addition to being a pest to people fishing in shallow waters, cirolanid isopods can also spoil the catch from deep-sea fishing. N. borealis is unusual in that it inhabits such a wide range of depths, from 20 to 2500 m [25,26,27]. This animal has a maximum length of 28 mm and is known to be an enthusiastic feeder on caught fish [11,28]. The species has a reputation for attacking and eating live and dead fish (including sharks) caught on lines, traps or in nets [11].
Fishing depths have increased over time and the average depth of fishing increased between 1950 and 2012 to reach approximately 350 m [29]. As well as an increase in average depth, the maximum depth of fishing ventures has increased to up to 1600 m [30] even though there are concerns that fishing at these depths significantly damages the environment and is unsustainable [31]. This is because many deep-sea species display slow growth, and their population sizes are slow to recuperate after devastating reductions by deep-sea bottom trawling. Deep-sea trawling has a scraping effect on the benthos and damages the sea floor’s ability to support life [32]. Nonetheless, the increase in fishing depth has brought the fishing industry into the territory of cirolanids in the genus Bathynomus which are substantially larger than most of the shallow-water cirolanids. For example, the common shallow-water isopod Cirolana harfordi grows to a maximum length of 18 mm, whereas Bathynomus doederleini has a maximum length of 138 mm and Bathynomus giganteus of 363 mm [2,33]. These Bathynomus species have been found at depths of 300–520 m and 349–1800 m, respectively [33,34,35,36,37]. Because individuals of the Bathynomus genus are so much bigger and have much larger and more powerful cutting mouthparts than most other cirolanid species [5], they may pose a greater potential for spoiling fishery catches as it is possible they can cause damage more quickly, and as individuals rather than swarms. B. giganteus populations appear capable of reaching plentiful proportions as a study that used baited traps to catch deep-sea crabs of the genus Chaceon over two month-long periods in the northcentral Gulf of Mexico caught almost as many B. giganteus individuals (1064) as bycatch, compared to Chaceon crabs (1321) [38].
B. doederleini individuals have been found in a deep-water shark Dalatius licha that was snagged in a gill net near Kumano on the Pacific side of Central Japan, at a depth of 520 m. The cirolanids of lengths 4 to 12 cm had chewed away the flesh around the shark’s anus to gain entry into the abdominal cavity [36].
Peer-reviewed literature has not yet confirmed that B. giganteus attacks fish caught in gill nets or traps, although there have been videos uploaded to the YouTube website (titled “Giant Isopod Kills Shark” and “Scavengers gather to enjoy a dead tuna”) showing a B. giganteus individual attacking a dogfish shark in a net and individuals feeding on a dead tuna. Scientific video taken from a remotely operated underwater vehicle of an individual of the closely related species B. pelor has shown the high speed in which this animal can lock onto food, slice it with its mandibles and devour it [5], but there have not been equivalent peer-reviewed studies on B. giganteus. There is one scientific report of B. giganteus being caught in a fishing trawler’s net; however, this report included a description of the animal’s stomach being dissected open to find that the animal’s stomach was empty [39]. More research using remotely operated vehicles that can take baited video cameras down to depths inhabited by B. giganteus is needed to determine whether members of this species in their natural environment regularly eat fish on a daily basis or intermittently, e.g., has a once a month eating urge. This would indicate whether B. giganteus, like B. doederleini, poses a danger to deep-sea fishing enterprises. There are two reports of a new species of Bathynomus where samples have been obtained from commercial fisheries. Specimens of Bathynomus paracelensis were caught near the Paracel Islands [40], South China Sea, and Bathynomus vaderi were caught in Vietnam [41]; however, for both species the stomach contents of these specimens were not analysed and the fishing catch obtained with the Bathynomus specimens was not examined for attack by the giant isopods.
4. Attacks on Humans and Human Infrastructure
There have been a number of instances when beachgoers in the United States of America have been bitten by swarms of shallow-water cirolanids, which has made the day painful and unpleasant for them [42,43]. In these situations, as long as swimmers are close to the shore and can come out of water and brush the isopods off their body, serious injury is unlikely. In Australia, however, a young man standing in a rock pool encountered numerous bites on his feet and ankles that he did not initially feel due to the coldness of the water [44]. The injuries bled profusely and required hospital treatment. The next day, specimens of the cirolanid species C. harfordi and specimens of the amphipod family Lysianassidae were collected from the rock pool in which the injury occurred and either of these species or both of them in combination could have been responsible [44]. Cirolanids can in rare instances pose an occupational safety hazard to professional divers, for example, divers cleaning jetty pilings in Sydney Harbour had their face bitten by cirolanids in areas of the face not covered by the mask or wetsuit hood [23]. There has been a case of a Japanese fisherman who fell overboard and had so much of his flesh eaten by a swarm of C. harfordi individuals that his head and upper trunk were described as skeletonised [45]. This demonstrates the importance of a swimmer that encounters a cirolanid swarm being able to get out of the water quickly.
Could unpleasant interactions with cirolanids pose an increasing risk to recreational swimming and tourist industries if cirolanid populations around beaches were to rise in number in future warmer waters? As a basis for this hypothesis there is some evidence that the presence of something undesirable at a beach can reduce the appeal of the area for beachgoers and affect the economy of the region. A study on the effects of marine debris on beach appeal and the economy concluded that marine debris has a detrimental economic impact. For example, the study’s projected outcome for a doubling of marine debris in Orange County, California, was a decrease of $414 million in tourism spending [46,47]. Likewise, there have been concerns raised about the effects of the presence of bluebottle jellyfish of the genus Physalia on the experience of beachgoers [48]. Somewhat analogous to the situation with intertidal cirolanids, bluebottle jellyfish can cause pain to swimmers (via stinging) but rarely cause serious injuries or fatality. There are concerns that the presence of bluebottle jellyfish reduces beach attendance and these animals have been described as a threat, or potential threat, to recreational economies and tourism [48,49,50,51]. More research is required on the potential of biting cirolanids to reduce the number of beachgoers in affected areas.
As well as posing a hazard to humans directly, one cirolanid species has proven its ability to damage human infrastructure. Bathynomus giganteus individuals chewed through underwater cables that ran to seabed cameras which were making observations of benthic fauna, including Cuban Dogfish and lobsters at The Bahamas’ Cape Eleuthera Institute, Bahamas Exuma Sound. The damage was so extensive it disabled the cables. A contributing factor to the incident was that the cables were covered with a spongey rubber casing that the huge cirolanids found attractive. To protect the camera cables from future shredding by B. giganteus, the scientists installed a heavy duty plastic guard around the submarine wires. [52]. This is a great example of people finding a practical and effective solution to a problem caused by a cirolanid isopod species.
5. Viviparous Reproduction in Some Cirolanid Species
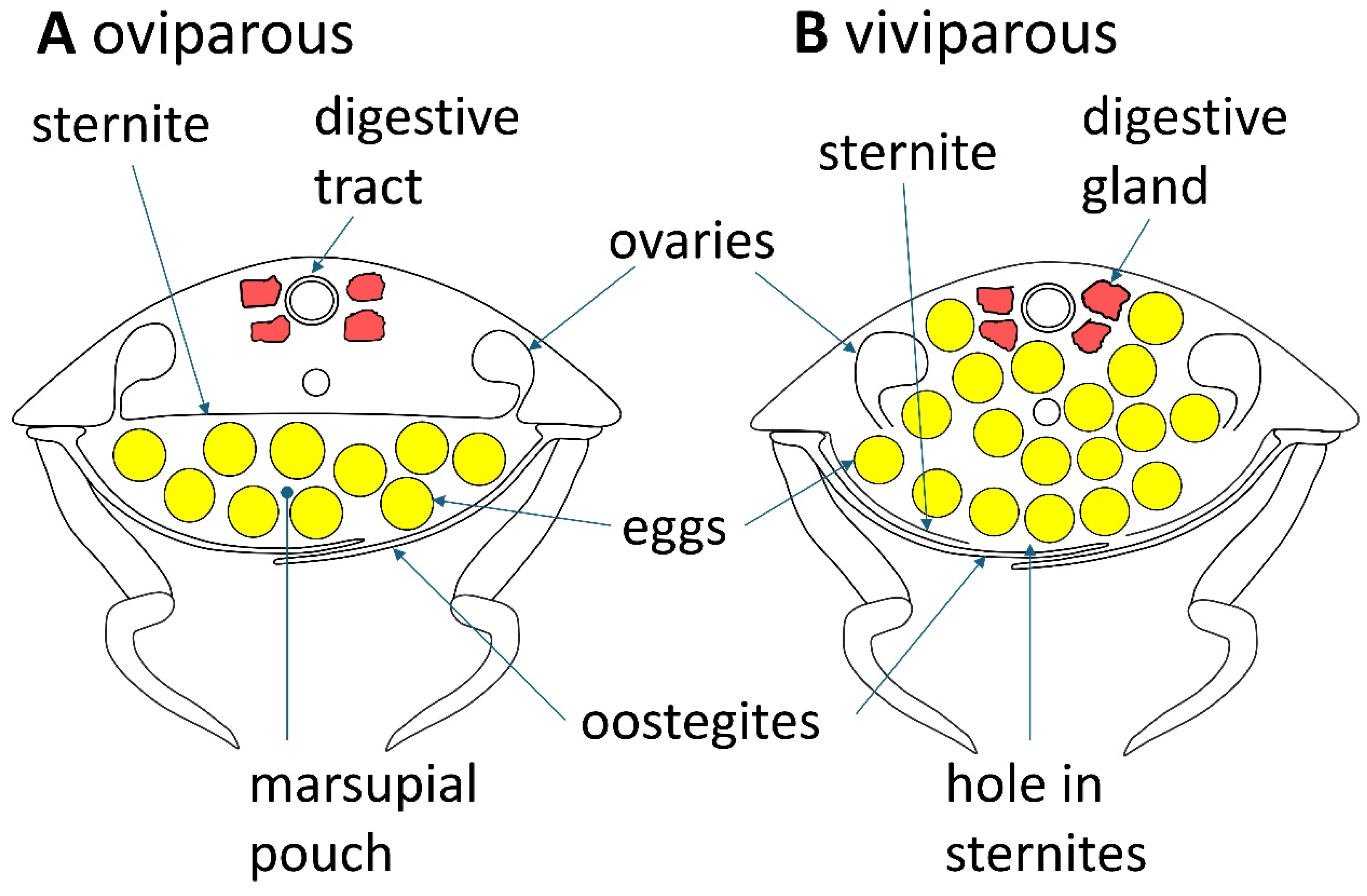
Some cirolanid species display live birth and this viviparity may serve to better enclose and protect the developing young, an advantage that may help form larger population sizes [53,54]. The females in most of the over 10,000 species of isopods extrude eggs from their pereon into a marsupial pouch which is formed when leaf-shaped cuticular structures called oostegites develop from the base of the pereopods to form the outer portion of the marsupial pouch [55,56,57,58,59,60] (see Figure 3 and Figure 4). The eggs develop in the marsupium through embryonic stages to reach a stage which resembles a miniature adult, with the exception that the juvenile has six pairs of legs instead of the seven seen in adults. At this stage the juveniles are called mancas [61]. In the development of the marsupium, long thin strings of mainly epithelial tissue called cotyledons grow from the sternites down into the marsupial pouch [62]. Cotyledons are somewhat analogous to the placenta in that they secrete marsupial fluid that contains nutrients and oxygen to nourish and sustain the young [57,62,63]. The cotyledons also contain mucous which has been suggested serves as a food source for the mancas when they have developed enough to eat [55,64]. One of the main differences between the mammalian placenta and isopod cotyledons is that the cotyledons are not attached to the developing young.

Figure 3.
(A) A living Cirolana harfordi individual emerging from under a rock to feed on a piece of pink salmon. (B) A scanning electron micrograph of the ventral side of a pregnant Cirolana harfordi female, the pereopods have been removed.
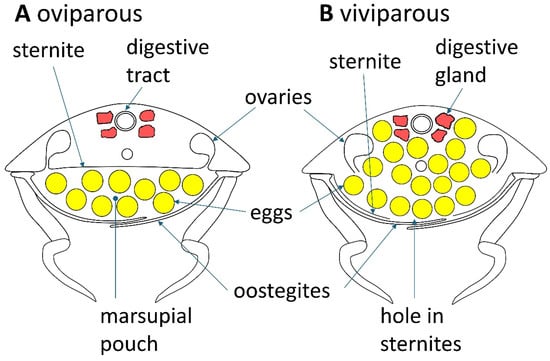
Figure 4.
The anatomy of the viviparous reproduction of Cirolana harfordi as compared to the oviparous form of reproduction displayed by most of the 10,000 plus species of isopods.
In 1970 Klapow and colleagues reported that the cirolanid isopod Excirolana chiltoni showed a different reproductive mode from most isopod species, with embryos and then mancas incubating in an internal pair of uteri which develop at the distal ends of the oviducts [53]. When it is time for birth, pressure from coelomic fluid causes the uteri to evert and expel the mancas through a hole in the uteri that forms around the area of the base of the mother’s fifth and sixth pereopods, and a hole in the ventral pereon that either forms or is ripped open by the exiting mancas who emerge from their mothers body backwards and in single file. The observed mancas did not appear to be using their own pereopods to locomote their birth, rather they were passively being propelled out by pressure. The mothers still developed oostegites that the authors propose serve to support the ventral sternites that were thinly stretched by the added bulk of the mancas within [53]. While the authors do not describe the mancas being pushed out between the oostegite plates, this would have to happen for the mancas to leave their mother’s body. Samples of gravid mothers from the U.S. National Museum from six additional species of the genus Excirolana also contained embryos in twin uteri inside the pereon, demonstrating that the viviparous mode is present in multiple species within the genus [53]. Over forty years later, viviparity was discovered in Australian specimens of C. harfordi; however, this species shows a somewhat different mode of reproduction than E. chiltoni [54]. Dissected samples of C. harfordi mothers that were pregnant with eggs showed that the eggs were wholly within the pereon but were not gathered in uteri; they were unrestrained in the thoracic cavity. When it is time for birth, C. harfordi mancas take an independent approach to their birth and self-locomote out of their mother’s pereon. An oval-shaped hole in the sternites of the mother’s ventral pereon develops before birth to allow for the exit of mancas. Most of the ventral pereon is covered by oostegites which protect the mother’s ruptured sternites and keeps the developing young and the mother’s internal organs in place.
In a study that investigated the effects of ocean warming and acidification on reproduction in C. harfordi, the speed in which juveniles developed in their mother’s body was observed by restraining pregnant C. harfordi individuals upside down in a sandwich of microscope slides and using a stereo microscope to look at the juveniles through the mothers transparent ventral pereon [6]. Indeed, C. harfordi’s transparent “window” to the mother’s internal young makes this animal one of the very few where viviparous development can be studied non-destructively and in real time. In several instances during this project, mancas were seen moving around inside the mother’s pereon cavity and then actively scrambling out to freedom by pushing headfirst through the overlapping oostegites [6]. It has been proposed that incubating developing young inside the pereon of intertidal viviparous cirolanids gives the species an evolutionary advantage because the young will be better protected from strong buffeting by waves, and the viviparous anatomy may help prevention of the expulsion of young more effectively than a marsupial pouch [53]. Additionally, the addition of the sternites in the barrier between the environment and the developing young may be more effective at protecting the embryos/mancas from sand incursion [53].
6. What Effects Will Warmer and More Acidic Oceans Have on Viviparous Cirolanid Population Sizes?
Increased temperature has been shown to have positive effects on reproduction in the viviparous cirolanid species C. harfordi and there is interest in determining if population sizes will increase in future warmer oceans as this situation could increase the negative effects of this animal on ocean resources [6,65]. Increasing carbon dioxide (CO2) in the atmosphere is driving up air and ocean temperatures via the greenhouse effect and depressing pH values as the excess CO2 dissolved in seawater reacts with water to form carbonic acid [66,67]. The extra protons in seawater combine with carbonate ions to produce bicarbonate which lowers the amount of carbonate that marine invertebrate animals need to produce calcium carbonate which in turn is needed to develop and maintain their shells or exterior cuticle [68]. Prior to the Industrial Revolution, oceanic pH was 8.2 but has dropped since that time to reach 8.1, and it is possible that in another 75 years it could fall to 7.7 [67]. Experiments on a wide range of organisms have indicated that some species will be able to survive or even be positively affected and thrive in warmer and more acidic seas while others will experience negative effects to their ability to survive and reproduce [69,70,71,72,73]. Because cirolanid isopods in moderate population sizes have important positive effects on the benthic environment but can cause damage to ocean resources when population rises lead to swarm formation, it is important to determine how these animals will be affected by ocean warming and acidification. Prior to 2002, however, marine isopods in general were overlooked in experiments studying the effects of ocean warming and acidification on the physiology and reproduction of ocean dwellers [74].
To determine if the viviparous reproduction in Cirolana harfordi is affected by different temperatures, specimens were held for fourteen weeks in flow-through tanks with natural sea water at three different temperatures, 18 °C, 20 °C and 22 °C [65]. The higher temperatures of 20 °C and 22 °C resulted in a faster development of embryos and mancas and onset of parturition compared to 18 °C. Consequently, there was a significantly higher amount of free-swimming manca offspring in the higher temperature tanks at 14 weeks [65]. A later project tested the effects of raised temperature in combination with lowered pH and tracked the effects on C. harfordi for the longer time of 22 weeks [6]. C. harfordi specimens were held in four different combinations of temperature and pH as follows: 18 °C/pH 8.1, 24 °C/pH 7.7, 18 °C/pH 7.7 and 24 °C/pH 8.1. The higher temperature resulted in faster development of the embryos/mancas with a shorter gestation time, and at 14 weeks there were significantly more swimming mancas in the tanks held at 24 °C. At 22 weeks, however, the amount of free-swimming manca offspring in the 18 °C tanks had caught up to the 24 °C tanks and therefore there was no significant effect of the higher temperature on offspring number at 22 weeks. In contrast, the average size of the free-swimming mancas was significantly larger in the warmer tanks.
The lowered pH had no effect on developmental time or offspring number, and the free-swimming mancas born in the low pH seawater were all normally developed. Interestingly, the females in the tanks held at 18 °C had one pregnancy during the 22 weeks, whereas some females in the 24 °C started a second period of pregnancy, indicating that future higher ocean temperatures could increase C. harfordi population sizes by multiplying the number of pregnancies that these animals can have per year. It appears that in future warmer and more acidic oceans, C. harfordi and possibly other cirolanid isopods will at least thrive and there is a possibility they could proliferate, thereby increasing the negative effects they can pose on ocean resources.
7. Social Behaviour
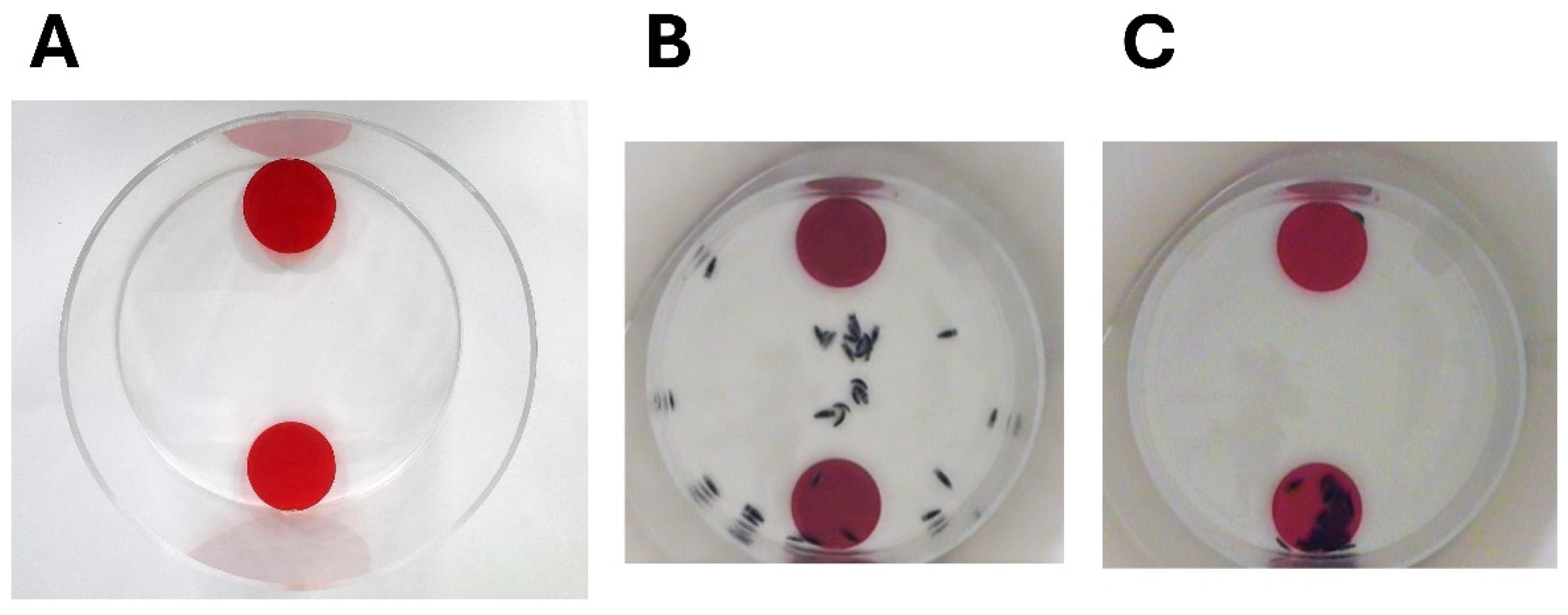
Members of one species of cirolanid, C. harfordi, have been shown to be social animals and this social behaviour may help them form efficient predatory swarms [75,76]. Since 1976 it has been recognised that members of this species form aggregates in their natural environment [77,78]. For some years, however, the question remained, were these animals aggregating in common areas due primarily to an attraction to available resources such as food and water [79] and simply tolerating each other’s proximity? Or did they actively seek out each other’s company for reasons other than mating? These questions were answered using a binary choice arena (see Figure 5), a piece of equipment that had been developed for studying social behaviour in terrestrial isopods and which consisted of a 200 mm diameter acrylic cylinder open at the top with two shelters at opposite sides of the arena [75,80,81,82,83]. The shelters were made of red-coloured acrylic discs of 50 mm diameter that were attached so that there was a 6 mm space underneath them to allow isopods to take shelter there. Specimens of C. harfordi were initially placed at the centre of the arena in a temporary holding container made from a plastic cylinder. The temporary holding container was removed at the start of the experiment releasing the animals and their movements were filmed from overhead for 60 min. In most of these experiments the animals, as a group, picked one of the shelters at random and preferentially aggregated there [75]. These results demonstrated the animals’ desire to seek out the company of conspecifics, thus showing social behaviour. The shelter preferred by the group of specimens was referred to as the “winning shelter” and the other termed the “losing shelter” [75,84].
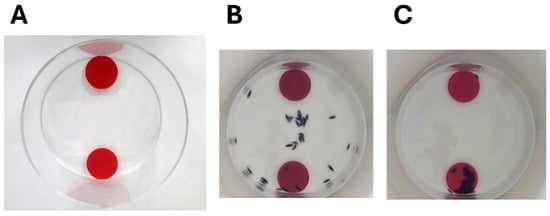
Figure 5.
Social behaviour in isopods tested in a binary choice arena. (A) The binary choice arena. (B) A video capture taken 5 s after 32 specimens of Cirolana harfordi were released from the centre of the arena. (C) As (B), except the video capture was taken 3 min 42 s after release of the specimens.
The steps taken by C. harfordi groups to pick the winning shelter show an insight into this animal’s cognitive abilities. When released from the holding container the animals usually walk or run to the outer part of the container and then ambulate along the edge of the vertical wall until they reach the underside of a shelter where they stop. After a while, individuals often leave a shelter and visit the other shelter, often remaining there or returning to the original shelter. After this to-and-fro behaviour, a winning shelter is selected by the group and the to-and-fro behaviour diminishes. The fact that a winning shelter is selected by the group indicates that the animals are more likely to stay under a shelter if the group there is larger than under the other shelter, and that the animal can gauge the size of the groups under each shelter so as to settle with the largest group [75,76].
Social aggregative behaviour in animals is thought to evolve in response to harnessing advantages in relation to the ecological landscape in which the animals live, and their reproductive biology [85,86,87]. Social and aggregative behaviour including the formation of groups or swarms may have conferred evolutionary advantages to cirolanid species, including cooperative defence. For example, a fish approaching a single cirolanid with a view to eat it would be in little to no danger, but a fish approaching a large group of cirolanids that could easily overwhelm that fish and strip its flesh off the bones presents a much higher level of danger and deterrent to would-be predators [76]. Likewise, the ability of aggregative cirolanid swarms to turn these animals into fearsome hunters confers a significant advantage to cirolanids in obtaining food. Additionally, using group decision making to find suitable habitats and resting places such as crevices in rocks and shaded areas assists the animals in finding high-quality and safe habitats [76]. The need to find protective shelter is particularly important for intertidal cirolanids like C. harfordi who often live in mussel and oyster beds or under rocks on sand, as without shelter they can be picked off by birds at low tide and fish at high tide [77,78]. C. harfordi often aggregate in their environment when they are resting and not hunting or scavenging, and groups of over 1000 individuals are common under a single rock on sand in the intertidal zone [78].
The cirolanid, ecological niche, modes of food acquisition and reproductive biology offer good explanations as to why cirolanid isopods have not developed a social system that includes division of labour and reproductive roles as well as group care of offspring. This group of behavioural patterns is often referred to as eusocial behaviour [88].
Eusocial animals often build or develop their group dwelling, whereas cirolanids do not need to do this as they often burrow into the mud or sand of the benthos or take refuge under rocks. Because cirolanids do not need to build or maintain a nest or hive, there has been no evolutionary pressure to develop dwelling-related division of labour. This, however, has not prevented all cirolanids from inhabiting a shared dwelling structure. As discussed previously, C. fluviatis have shown the ability to opportunistically move into a ready-formed community shelter by taking up residence in aquaculture nets covered in mud. Additionally, after most isopods developed highly efficient reproductive patterns in both oviparous and viviparous modes there was no need to develop group social care of neonates as isopod mothers keep their eggs and then developing young, either close to the body, or inside it, until the young are fully developed and can feed themselves. Some animals that did not evolve to possess these kinds of efficient reproductive strategies may have evolved to show eusocial behaviour that could compensate. As an example, bees lay eggs into honeycomb cells, in which the larvae need care and feeding, and this care and feeding drove the evolution of specialised nurse bees that showed eusocial behaviour. Interestingly, some isopods species are an exception to these general trends in isopod social behaviour. In the desert-dwelling isopod species Hemilepistus reaumuri, for example, males and females mate for life, divide nest maintenance between mother and father on gender principles and provide post hatch care to their offspring [86,89,90]. This demonstrates isopods are able to develop forms of eusocial behaviour, but this behaviour was not needed by cirolanids. Therefore, while cirolanid isopods may be immune to eusocial behaviour, the major genetic drivers for cirolanid social evolution appear to have been to help these animals locate food and shelter. Indeed, not engaging in eusocial behaviour has allowed cirolanids more time for pack hunting and resting in places and numbers that offer protection from predators. As discussed, while their propensities in pack hunting are beneficial to cirolanids, they can have negative effects on ocean resources.
8. Possible Solutions to Protect Against Cirolanid Swarms
What kind of strategies can be taken to protect fishing harvests and swimming humans from cirolanid swarms? As mentioned, some fishermen have adjusted the times when they place their nets to avoid the diel nighttime migration that includes cirolanid isopods. The cleaning of net aquaculture cages from mud and organic material buildup may help prevent cirolanids using these structures as a substrate or housing to rest conveniently close to a captured food source. Keeping food provided to fish in aquaculture, to an absolute minimum, may prevent excess food on the benthos attracting cirolanids.
Another approach may be to deploy large-scale baited traps similar in design to the traps used in scientific studies to catch cirolanids [91,92]. These traps are made of a piece of pipe with an inverted funnel at one end and a piece of stainless steel mesh at the other, to allow water to flow through. The traps are baited with pieces of fish and weighted, so they rest on the sea floor. Cirolanids enter through the inverted funnel but cannot find their way out and are trapped. These kinds of traps could be deployed and used intermittently to prevent population levels rising to hazardous numbers, thus preventing swarm formation in areas used for aquaculture or recreational swimming.
Divers in cirolanid-infested waters may opt to cover all areas of their skin to guard against cirolanid bites.
9. Conclusions
The threat of cirolanid swarms have long been known to fishermen, and in many areas around the world these animals have a significantly negative effect on the harvest of fish, as well as acting as pests to recreational swimmers. Further research is needed to determine whether cirolanid swarms will increase in the warmer, more acidic oceans of the future, and to develop techniques and protocols to effectively limit the growth of cirolanid populations where and when necessary.
Funding
This research received no external funding.
Data Availability Statement
No new data were created or analysed in this study.
Acknowledgments
The author has reviewed and edited the output and takes full responsibility for the content of this publication. The author acknowledges the technical and scientific assistance of Sydney Microscopy & Microanalysis, the University of Sydney node of Microscopy Australia.
Conflicts of Interest
The author declares no conflicts of interest.
References
- Bruce, N.L.; Ton, H.M.L.; Poore, G.C.B. Family CIROLANIDAE Dana, 1852. Available online: https://biodiversity.org.au/afd/taxa/Cirolanidae#:~:text=Introduction,of%20all%20marine%20cirolanid%20species (accessed on 16 January 2026).
- Bruce, N.L. Cirolanidae (Crustacea: Isopoda) of Australia. Rec. Aust. Mus. Suppl. 1986, 6, 1–239. [Google Scholar] [CrossRef]
- Sidabalok, C.M.; Bruce, N.L. Review of the Cirolana ‘pleonastica-group’ (Crustacea: Isopoda: Cirolanidae) with description of four new species from the Indo-Malaysian region. Raffles Bull. Zool. 2018, 66, 177–207. [Google Scholar]
- Johansen, P.-O. Bait attraction studies on the scavenging deepwater isopod Natatolana borealis (Crustacea, Isopoda). Ophelia 2000, 53, 27–35. [Google Scholar] [CrossRef]
- Thomson, M. Mouthparts of the deep-sea isopod Bathynomus pelor (Isopoda, Cirolanidae). Crustaceana 2010, 83, 1483–1506. [Google Scholar] [CrossRef]
- Thomson, M.; Harianto, J.; Byrne, M. Reproduction of the viviparous marine isopod Cirolana harfordi held in seawater with raised temperature and lowered pH. Mar. Ecol. Prog. Ser. 2025, 775, 87–98. [Google Scholar] [CrossRef]
- Briones-Fourzán, P.; Lozano-Alvarez, E. Aspects of the biology of the giant isopod Bathynomus giganteus A. Milne Edwards, 1879 (Flabellifera, Cirolanidae), off the Yucatan Peninsula. J. Crustac. Biol. 1991, 11, 375–385. [Google Scholar] [CrossRef]
- Thomson, M.; Robertson, K.; Pile, A. Microscopic structure of the antennulae and antennae on the deep-sea isopod Bathynomus pelor. J. Crustac. Biol. 2009, 29, 302–316. [Google Scholar] [CrossRef]
- Berrow, S. Fish predation by the marine crustaceans Orchomene nana (Kroyer) and Natatolana borealis (Lilljeborg). Ir. Nat. J. 1994, 24, 514. [Google Scholar]
- Kirkim, F.; Horton, T.; Akyol, O.; Ceyhan, T. Natatolana neglecta (Isopoda, Cirolanidae): An increasing threat for artisanal fishing in the Turkish Aegean Sea. Crustaceana 2019, 92, 881–887. [Google Scholar] [CrossRef]
- Youssef, F.; Mansour, L.; Quilichini, Y.; Zouari Tlig, S.; Benmansour, B. Morphology and scavenging behaviour of two species of the genus Natatolana (Crustacea: Isopoda: Cirolanidae) attacking elasmobranchs from the Tunisian coast. Cahiers Biol. Mar. 2020, 61, 395–404. [Google Scholar] [CrossRef]
- Byard, R.W.; James, R.A.; Gilbert, J.D. Diagnostic problems associated with cadaveric trauma from animal activity. Am. J. Forensic Med. Pathol. 2002, 23, 238–244. [Google Scholar] [CrossRef]
- Erkol, Z.; Hösükle, E. Postmortem animal attacks on human corpses. In Post Mortem Examination and Autopsy; Dogan, K.H., Ed.; IntechOpen: London, UK, 2018. [Google Scholar]
- Newman, B.K.; Wooldridge, T.H.; Cockcroft, A.C. Aspects of the biology and ecology of the estuarine cirolanid isopod, Cirolana fluviatilis. Afr. Zool. 2007, 42, 12–22. [Google Scholar] [CrossRef]
- Tiemensma, M.; Bruce, N.L.; Willan, R.C. Post-mortem human cadaver scavenging by marine crustaceans (Isopoda: Cirolanidae) in tropical waters. Forensic Sci. Med. Pathol. 2017, 13, 515–517. [Google Scholar] [CrossRef] [PubMed]
- Tsokos, M. (Ed.) Postmortem changes and artifacts occurring during the early postmortem interval. In Forensic Pathology Reviews; Humana Press: Totowa, NJ, USA, 2005; pp. 183–238. [Google Scholar]
- Alldredge, A.L.; King, J.M. Distribution, abundance, and substrate preferences of demersal reef zooplankton at Lizard Island Lagoon, Great Barrier Reef. Mar. Biol. 1977, 41, 317–333. [Google Scholar] [CrossRef]
- Stepien, C.A.; Brusca, R.C. Nocturnal attacks on nearshore fishes in Southern California by crustacean zooplankton. Mar. Ecol. Prog. Ser. 1985, 25, 91–105. [Google Scholar] [CrossRef]
- Stebbins, T.D.; Wetzer, R. Review and guide to the isopods (Crustacea, Isopoda) of littoral and sublittoral marine habitats in the Southern California Bight. Zookeys 2023, 1162, 1–167. [Google Scholar] [CrossRef]
- Svane, I.; Barnett, J. The occurrence of benthic scavengers and their consumption at tuna farms off Port Lincoln, South Australia. J. Exp. Mar. Biol. Ecol. 2008, 363, 110–117. [Google Scholar] [CrossRef]
- Sanil, N.K.; Vikas, P.A.; Ratheesh, T.B.; George, K.C.; Vijayan, K.K. Mortalities caused by the crustacean isopod, Cirolana fluviatilis, in tropical, cage-cultured Asian seabass, Lates calcarifer: A case study from the southwest coast of India. Aquac. Res. 2009, 40, 1626–1633. [Google Scholar] [CrossRef]
- Öndes, F. Quantification of the problem caused by isopods in the small scale fishery in the Southern Aegean Sea, Turkey. Intern. J. Agric. Nat. Sci. 2019, 12, 20–22. [Google Scholar]
- Bruce, N.L. Marine isopod crustaceans in New Zealand. Water Atmos. 2001, 9, 12–13. [Google Scholar]
- Mathew, K.J.; Selvaraj, G.S.D.; Naomi, T.S.; Molly, V.; Sridhar, N.; Manpal, S.; Geetha, A.; Leela Bhai, K.S.; Anilkumar, R.; So-lomon, K. Studies on the infestation of an isopod crustacean, Cirolana fluviatilis in some parts of the Cochin Backwaters, Kerala. Mar. Fish. Inf. Serv. Tech. Ext. Ser. 1994, 131, 2–10. [Google Scholar]
- Johansen, P.-O.; Brattegard, T. Observations on behaviour and distribution of Natatolana borealis (Lilljeborg) (Crustacea, Isopoda). Sarsia 1998, 83, 347–360. [Google Scholar] [CrossRef]
- Kaïm-Malka, R.A. Biology and life cycle of Natatolana borealis Lilj.1851, a scavenging isopod from the continental slope of the Mediterranean. Deep Sea Res. 1 Oceanogr. Res. Pap. 1997, 44, 2045–2067. [Google Scholar] [CrossRef]
- Keable, S.J.; Bruce, N.L. Redescription of the North Atlantic and Mediterranean Species of Natatolana (Crustacea: Isopoda: Cirolanidae). J. Mar. Biol. Assoc. United Kingd. 2009, 77, 655–705. [Google Scholar] [CrossRef]
- Bird, P.M. The occurrence of Cirolana borealis (Isopoda) in the hearts of sharks from Atlantic coastal waters of Florida. Fish. Bull. 1981, 79, 376–383. [Google Scholar]
- Watson, R.A.; Morato, T. Fishing down the deep: Accounting for within-species changes in depth of fishing. Fish. Res. 2013, 140, 63–65. [Google Scholar] [CrossRef]
- Bailey, D.M.; Collins, M.A.; Gordon, J.D.M.; Zuur, A.F.; Priede, I.G. Long-term changes in deep-water fish populations in the northeast Atlantic: A deeper reaching effect of fisheries? Proc. R. Soc. B 2009, 276, 1965–1969. [Google Scholar] [CrossRef]
- Stephenson, F.; Zelli, E.; Bennion, M.; Rowden, A.; Anderson, O.; Clark, M.; Tablada, J.; Hiddink, J.; Kaikkonen, L.; Finucci, B.; et al. Large-scale assessments of bottom trawling effects on vulnerable marine ecosystems can significantly under-represent impacts. J. Environ. Manag. 2025, 395, 127672. [Google Scholar] [CrossRef]
- Good, E.; Holman, L.; Pusceddu, A.; Russo, T.; Rius, M.; Iacono, C. Detection of community-wide impacts of bottom trawl fishing on deep-sea assemblages using environmental DNA metabarcoding. Mar. Pollut. Bull. 2022, 183, 114062. [Google Scholar] [CrossRef]
- Barradas-Ortiz, C.; Briones-Fourzán, P.; Lozano-Alvarez, E. Seasonal reproduction and feeding ecology of giant isopods Bathynomus giganteus from the continental slope of the Yucatan peninsula. Deep Sea Res. I Oceanogr. Res. Pap. 2003, 50, 495–513. [Google Scholar] [CrossRef]
- Boos, H.; Scalco, A.; Araujo, P. Biological and ecological traits of Bathynomus giganteus and Bathynomus miyarei (Crustacea: Isopoda): Contribution to the conservation of deep-sea in Southern Brazil. Aquat. Conserv. Mar. Freshw. Ecosyst. 2021, 31, 2084–2094. [Google Scholar] [CrossRef]
- Holthuis, L.B.; Mikulka, W.R. Notes on deep-sea isopods of genus Bathynomus A. Milne Edwards, 1879. Bull. Mar. Sci. 1972, 22, 575–591. [Google Scholar]
- Sekiguchi, H.; Yamaguchi, Y.; Kobayashi, H. Bathynomus (Isopoda: Cirolanidae) attacking sharks caught in a gill-net. Bull. Fac. Fish. Mie Univ. 1981, 8, 11–17. [Google Scholar]
- Soong, K.; Mok, H.-K. Size and maturity stage observations of the deep-sea isopod Bathynomus doederleini Ortmann, 1894 (Flabellifera: Cirolanidae), in Eastern Taiwan. J. Crustac. Biol. 1994, 14, 72–79. [Google Scholar] [CrossRef]
- Perry, H.; Waller, R.; Trigg, C.; McBee, J.; Erdman, R.; Blake, N. Blake A note on bycatch associated with deepwater trapping of Chaceon in the northcentral Gulf of Mexico. Gulf Res. Rep. 1995, 9, 139–142. [Google Scholar]
- Nayak, T.H.; Dineshbabu, A.P.; Zacharia, P.D. A note on the capture of ‘giant isopod’, Bathynomus giganteus A. Milne edwards, 1879 off Mangalore Coast, India. J. Bombay Nat. Hist. Soc. 2007, 104, 369–370. [Google Scholar]
- Huang, M.-C.; Kawai, T. A new species of supergiant Bathynomus A. Milne-Edwards, 1879 (Isopoda: Cirolanidae) from the Paracel Islands, South China Sea. Biodivers. Data J. 2025, 13, e144238. [Google Scholar] [CrossRef]
- Ng, P.K.L.; Sidabalok, C.M.; Nguyen, T.S. A new species of supergiant Bathynomus A. Milne-Edwards, 1879 (Crustacea, Isopoda, Cirolanidae) from Vietnam, with notes on the taxonomy of Bathynomus jamesi Kou, Chen & Li, 2017. ZooKeys 2025, 1223, 289–310. [Google Scholar] [CrossRef]
- Carbone, C. ‘Small Piranhas’: Beachgoers in California Plagued by SWARMS of Tiny, Hungry Bugs That Bite Their Feet and Toes, Causing Pain and Drawing Blood. Daily Mail, 5 September 2022. [Google Scholar]
- Hawkins, J. These Hideous Little Bugs Are Invading California and Biting People’s Feet. BGR News, 3 September 2022. [Google Scholar]
- Sea Bug Attack: Why Was a Wading Teenager Left Covered in Blood? BBC News, 8 August 2017.
- Hirasawa, T.; Kida, M.; Yanase, M. A case report of encroached corpse skeletonized within 24 hours by the ravage of Cirolana harfordi japonica. Res. Pract. Forensic Med. 1999, 42, 221–223. [Google Scholar]
- Marine Debris Program, NOAA. The Economic Impacts of Marine Debris on Tourism-Dependent Communities. Available online: https://marinedebris.noaa.gov/research/economic-impacts-marine-debris-tourism-dependent-communities (accessed on 25 January 2016).
- English, E.; Wagner, C.; Holmes, J. The Effects of Marine Debris on Beach Recreation and Regional Economies in Four Coastal Communities: A Regional Pilot Study. Final Report, 30 September 2019. [Google Scholar]
- Rodrigues, T.; Domínguez-Pérez, D.; Almeida, D.; Matos, A.; Antunes, A. Medusozoans reported in Portugal and its ecological and economical relevance. Reg. Stud. Mar. Sci. 2020, 35, 101230. [Google Scholar] [CrossRef]
- Prieto, L.; Macías, D.; Peliz, A.; Ruiz, J. Portuguese Man-of-War (Physalia physalis) in the Mediterranean: A permanent invasion or a casual appearance? Sci. Rep. 2015, 5, 11545. [Google Scholar] [CrossRef] [PubMed]
- De Donno, A.; Idolo, A.; Bagordo, F.; Grassi, T.; Leomanni, A.; Serio, F.; Guido, M.; Canitano, M.; Zampardi, S.; Boero, F.; et al. Impact of stinging jellyfish proliferations along South Italian Coasts: Human health hazards, treatment and social costs. Int. J. Environ. Res. Public Health 2014, 11, 2488–2503. [Google Scholar] [CrossRef]
- Tiralongo, F.; Badalamenti, R.; Arizza, V.; Prieto, L.; Lo Brutto, S. The Portuguese Man-of-War has always entered the Mediterranean Sea—Strandings, sightings, and museum collections. Front. Mar. Sci. 2022, 9, 856979. [Google Scholar] [CrossRef]
- Tucker, A. The Sea Monster Bathynomus. Smithsonian Magazine, 7 June 2012. [Google Scholar]
- Klapow, L.A. Ovoviviparity in the genus Excirolana (Crustacea: Isopoda). J. Zool. 1970, 162, 359–369. [Google Scholar] [CrossRef]
- Thomson, M. Ovoviviparous reproduction in Australian specimens of the intertidal isopod Cirolana harfordi. Invertebr. Repr. Dev. 2014, 58, 218–225. [Google Scholar] [CrossRef]
- Csonka, D.; Halasy, K.; Hornung, E. Histological studies on the marsupium of two terrestrial isopods (Crustacea, Isopoda, Oniscidea). Zookeys 2015, 515, 81–92. [Google Scholar] [CrossRef]
- Hoese, B. The marsupium in terrestrial isopods. Symp. Zool. Soc. Lond 1984, 53, 65–76. [Google Scholar]
- Janssen, H.H.; Hoese, B. Marsupium morphology and brooding biology of the Antarctic giant isopod, Glyptonotus antarcticus Eights 1853 (Crustacea, Isopoda, Chaetiliidae). Polar Biol. 1993, 13, 145–149. [Google Scholar] [CrossRef]
- Warburg, M.R. Marsupial contents and losses due to putative intramarsupial cannibalism by the mancas in three Oniscid isopod species. J. Crustac. Biol. 1994, 14, 560–567. [Google Scholar] [CrossRef]
- Warburg, M.R. The oniscid isopod female reproductive system and gestation, with a partial review. Invertebr. Repr. Dev. 2012, 56, 87–110. [Google Scholar] [CrossRef]
- Suzuki, S.; Yamasaki, K. Ovarian control of oostegite formation in the terrestrial isopod, Armadillidium vulgare (malacostraca, crustacea). Gen. Comp. Endocrinol. 1991, 84, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Wolff, C. The embryonic development of the malacostracan crustacean Porcellio scaber (Isopoda, Oniscidea). Dev. Genes Evol. 2009, 219, 545–564. [Google Scholar] [CrossRef] [PubMed]
- Warburg, M.R.; Rosenberg, M. Brood-pouch structures in terrestrial isopods. Invertebr. Repr. Dev. 1996, 29, 213–222. [Google Scholar] [CrossRef]
- Lardies, M.A.; Cotoras, I.S.; Bozinovic, F. The energetics of reproduction and parental care in the terrestrial isopod Porcellio laevis. J. Insect Physiol. 2004, 50, 1127–1135. [Google Scholar] [CrossRef]
- Akahira, Y. The function of thoracic processes found in females of the common wood-louse, Porcellio scaber. J. Fac. Sci. Hokkaido Univ. 1956, 12, 493–498. [Google Scholar]
- Gavel, I.; Byrne, M.; Thomson, M. Effects of raised temperature on viviparous reproduction in the marine isopod Cirolana harfordi. J. Exp. Mar. Biol. Ecol. 2022, 546, 151648. [Google Scholar] [CrossRef]
- Orr, J.C.; Caldeira, K.; Fabry, V.; Gattuso, J.P.; Haugan, P.; Lehodey, P.; Pantoja, S.; Portner, H.O.; Riebesell, U.; Trull, T.; et al. Research priorities for understanding ocean acidification summary from the second symposium on the ocean in a high-CO2 world. Oceanography 2009, 22, 182–189. [Google Scholar] [CrossRef]
- Orr, J.C.; Fabry, V.J.; Aumont, O.; Bopp, L.; Doney, S.C.; Feely, R.A.; Gnanadesikan, A.; Gruber, N.; Ishida, A.; Joos, F.; et al. Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 2005, 437, 681–686. [Google Scholar] [CrossRef]
- Walther, K.; Sartoris, F.J.; Pörtner, H.O. Impacts of temperature and acidification on larval calcium incorporation of the spider crab Hyas araneus from different latitudes (54° vs. 79° N). Mar. Biol. 2011, 158, 2043–2053. [Google Scholar] [CrossRef]
- Asnicar, D.; Marin, M.G. Effects of Seawater Acidification on Echinoid Adult Stage: A Review. J. Mar. Sci. Eng. 2022, 10, 477. [Google Scholar] [CrossRef]
- Byrne, M.; Ross, P.M.; Dworjanyn, S.A.; Parker, L. Larval Ecology in the Face of Changing Climate—Impacts of Ocean Warming and Ocean Acidification. In Evolutionary Ecology of Marine Invertebrate Larvae; Carrier, T., Reitzel, A., Heyland, A., Eds.; Oxford University Press: Oxford, UK, 2017; pp. 251–272. [Google Scholar]
- Minuti, J.J.; Byrne, M.; Campbell, H.; Hemraj, D.A.; Russell, B.D. Live-fast-die-young: Carryover effects of heatwave-exposed adult urchins on the development of the next generation. Glob. Change Biol. 2022, 28, 5781–5792. [Google Scholar] [CrossRef] [PubMed]
- Padilla-Gamiño, J.L.; Alma, L.; Spencer, L.H.; Venkataraman, Y.R.; Wessler, L. Ocean acidification does not overlook sex: Review of understudied effects and implications of low pH on marine invertebrate sexual reproduction. Front. Mar. Sci. 2022, 9, 977754. [Google Scholar] [CrossRef]
- Venegas, R.M.; Acevedo, J.; Treml, E.A. Three decades of ocean warming impacts on marine ecosystems: A review and perspective. Deep-Sea Res. II Top. Stud. Oceanogr. 2023, 212, 105318. [Google Scholar] [CrossRef]
- Baag, S.; Mandal, S. Combined effects of ocean warming and acidification on marine fish and shellfish: A molecule to ecosystem perspective. Sci. Total Environ. 2022, 802, 149807. [Google Scholar] [CrossRef]
- Salma, U.; Thomson, M. Gregarious aggregative behavior in the marine isopod Cirolana harfordi. Invertebr. Biol. 2016, 135, 225–234. [Google Scholar] [CrossRef]
- Salma, U.; Thomson, M. Social aggregation of the marine isopod Cirolana harfordi does not rely on the availability of light-reducing shelters. Physiol. Entomol. 2018, 43, 60–68. [Google Scholar] [CrossRef]
- Bugnot, A.B.; Coleman, R.A.; Figueira, W.F.; Marzinelli, E.M. Patterns of the non-indigenous isopod Cirolana harfordi in Sydney Harbour. PLoS ONE 2014, 9, e86765. [Google Scholar] [CrossRef]
- Johnson, W.S. Biology and population dynamics of the intertidal isopod Cirolana harfordi. Mar. Biol. 1976, 36, 343–350. [Google Scholar] [CrossRef]
- Allee, W.C. Studies in animal aggregations: Causes and effects of bunching in land isopods. J. Exp. Zool. 1926, 45, 255–277. [Google Scholar] [CrossRef]
- Broly, P.; Deneubourg, J.L. Behavioural contagion explains group cohesion in a social crustacean. PLoS Comput. Biol. 2015, 11, e1004290. [Google Scholar] [CrossRef] [PubMed]
- Broly, P.; Devigne, L.; Deneubourg, J.L.; Devigne, C. Effects of group size on aggregation against desiccation in woodlice (Isopoda: Oniscidea). Physiol. Entomol. 2014, 39, 165–171. [Google Scholar] [CrossRef]
- Broly, P.; Mullier, R.; Deneubourg, J.-L.; Devigne, C. Aggregation in woodlice: Social interaction and density effects. ZooKeys 2012, 176, 133–144. [Google Scholar] [CrossRef]
- Broly, P.; Mullier, R.; Devigne, C.; Deneubourg, J.L. Evidence of self-organization in a gregarious land-dwelling crustacean (Isopoda: Oniscidea). Anim. Cogn. 2016, 19, 181–192. [Google Scholar] [CrossRef]
- Devigne, C.; Broly, P.; Deneubourg, J.L. Individual preferences and social interactions determine the aggregation of woodlice. PLoS ONE 2011, 6, e17389. [Google Scholar] [CrossRef]
- Korb, J. The ecology of social evolution in termites. In Ecology of Social Evolution; Korb, J., Heinze, J., Eds.; Springer: Heidelberg, Germany, 2008; pp. 151–174. [Google Scholar]
- Thiel, M. The evolution of sociality: Peracarid crustaceans as model organisms. In New Frontiers in Crustacean Biology; Asakura, A., Ed.; Brill: Leiden, The Netherlands, 2011; pp. 285–297. [Google Scholar]
- Whitehouse, M.E.A.; Lubin, Y. The functions of societies and the evolution of group living: Spider societies as a test case. Biol. Rev. 2005, 80, 347–361. [Google Scholar] [CrossRef]
- Friedman, D.A.; Johnson, B.R.; Linksvayer, T.A. Distributed physiology and the molecular basis of social life in eusocial insects. Horm. Behav. 2020, 122, 104757. [Google Scholar] [CrossRef]
- Linsenmair, K.E. Comparative studies on the social behaviour of the desert isopod Hemilepistus reaumuri and of a Porcellio species. Symp. Zool. Soc. Lond 1984, 53, 423–453. [Google Scholar]
- Linsenmair, K.E. Kin recognition in subsocial arthropods, in particular in the desert isopod Hemilepistus reaumuri. In Kin Recognition in Animals; Fletcher, D.J.C., Michener, C.D., Eds.; Wiley: New York, NY, USA, 1987; pp. 121–208. [Google Scholar]
- Keable, S.J. Structure of the marine invertebrate scavenging guild of a tropical reef ecosystem: Field studies at Lizard Island, Queensland, Australia. J. Nat. Hist. 1995, 29, 27–45. [Google Scholar] [CrossRef]
- Thomson, M. Mouthparts and their setae of the intertidal isopod Cirolana harfordi. J. Microsc. 2013, 252, 111–121. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.