The Importance of Considering the Service Environment When Studying and Predicting the Performance of Corrodible Structures
Abstract
1. Introduction
2. The Repository Environment
3. Examples of Injudicious Testing Conditions and Other Issues
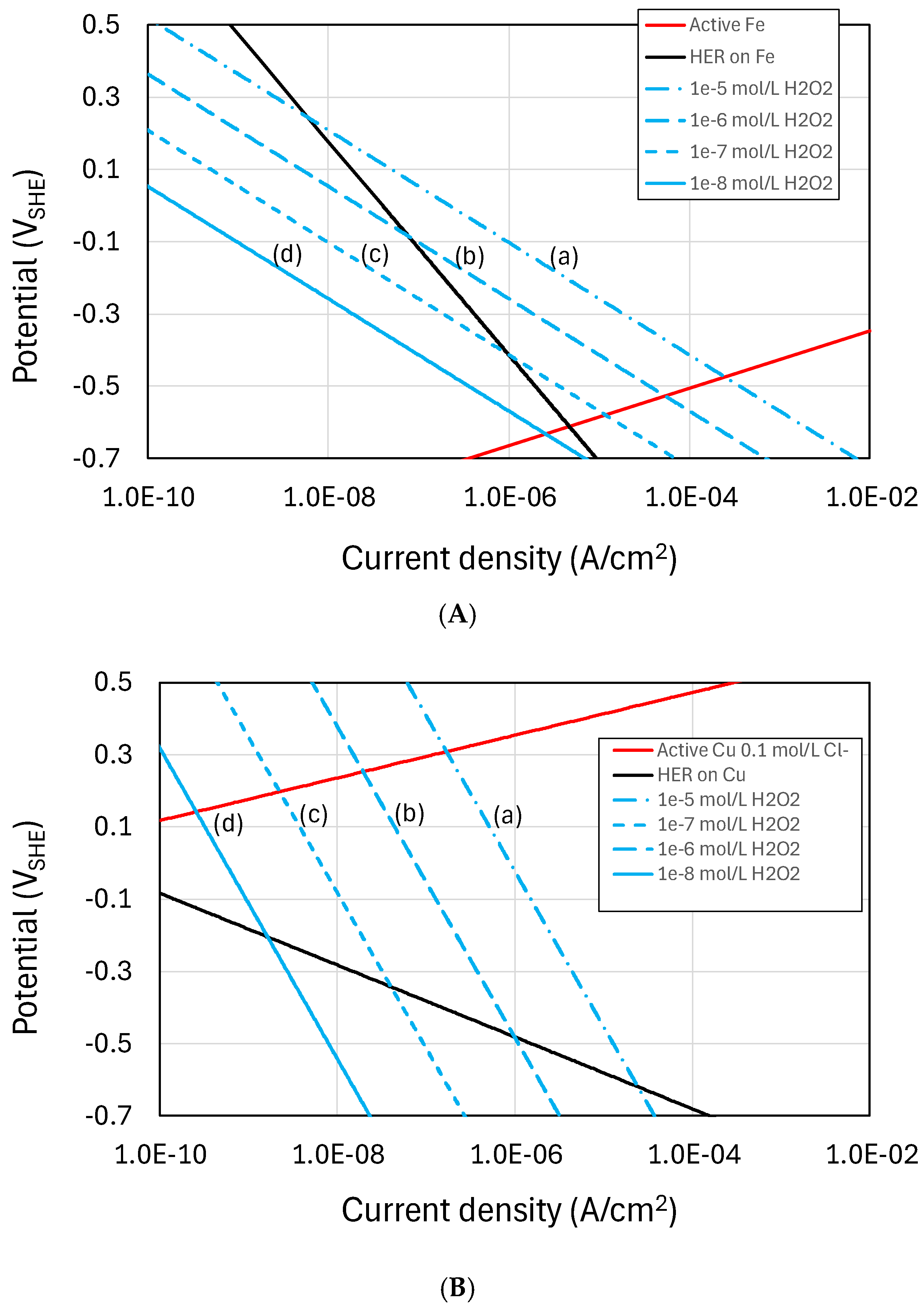
3.1. Corrosion of Copper in Sulfide Environments
- The likelihood of localized corrosion since there is no driving force for the transport of HS− ahead of the uniform corrosion front if the interfacial concentration is zero. Therefore, localized forms of corrosion requiring the presence of sulfide within the pit are not possible for sulfide fluxes lower than the transport limit.
- The formation of crack-like features under tensile loading for the same reason as for localized corrosion described above [34].
3.2. Effect of γ-Irradiation
3.3. Microbiologically Influenced Corrosion
3.4. Environmentally Assisted Cracking
4. Summary and Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Macdonald, D.D. The role of determinism in the prediction of corrosion damage. Corros. Mater. Degrad. 2023, 4, 212–273. [Google Scholar] [CrossRef]
- King, F.; Kolàř, M.; Briggs, S.; Behazin, M.; Keech, P.; Diomidis, N. Review of the modelling of corrosion processes and lifetime prediction for HLW/SF containers—Part 1: Process models. Corros. Mater. Degrad. 2024, 5, 124–199. [Google Scholar] [CrossRef]
- Anderko, A. Modeling of Aqueous corrosion. In Shreir’s Corrosion, 4th ed.; Cottis, R.A., Graham, M.J., Lindsay, R., Lyon, S.B., Richardson, J.A., Scantlebury, J.D., Stott, F.H., Eds.; Elsevier: Amsterdam, The Netherlands, 2010; Volume 2, pp. 1585–1629. [Google Scholar]
- Shoesmith, D.W. Assessing the corrosion performance of high-level nuclear waste containers. Corrosion 2006, 62, 703–722. [Google Scholar] [CrossRef]
- King, F.; Hall, D.S.; Keech, P.G. Nature of the nearfield environment in a deep geological repository and the implications for the corrosion behaviour of the container. Corros. Eng. Sci. Technol. 2017, 52, 25–30. [Google Scholar] [CrossRef]
- Villar, M.V.; Cuevas, J.F.; Idiart, A.; Coene, E.; Zabala, A.B.; Ruiz, A.I.; Ortega, A.; Iglesias, R.; Melón, A.M.; Heino, V. Five-year thermos-hydro-mechanical and chemical evolution of compacted bentonite: Physical and mineralogical analysis. Appl. Clay Sci. 2025, 276, 107931. [Google Scholar] [CrossRef]
- Wersin, P.; Curti, E.; Appelo, C.A.J. Modelling bentonite-water interactions at high solid/liquid ratios: Swelling and diffuse double layer effects. Appl. Clay Sci. 2004, 26, 249–257. [Google Scholar] [CrossRef]
- Muurinen, A.; Karnland, O.; Lehikoinen, J. Ion concentration caused by an external solution into the porewater of compacted bentonite. Phys. Chem. Earth 2004, 29, 119–127. [Google Scholar] [CrossRef]
- Ochs, M.; Lothenbach, B.; Shibata, M.; Yui, M. Thermodynamic modeling and sensitivity analysis of porewater chemistry in compacted bentonite. Phys. Chem. Earth 2004, 29, 129–136. [Google Scholar] [CrossRef]
- Bradbury, M.H.; Baeyens, B. Porewater chemistry in compacted re-saturated MX-80 bentonite. J. Contam. Hydrol. 2003, 61, 329–338. [Google Scholar] [CrossRef]
- King, F.; Litke, C.D.; Ryan, S.R. A mechanistic study of the uniform corrosion of copper in compacted Na-montmorillonite/sand mixtures. Corros. Sci. 1992, 33, 1979–1995. [Google Scholar] [CrossRef]
- Swedish Corrosion Institute. Copper as a Canister Material for Unreprocessed Nuclear Waste—Evaluation with Respect to Corrosion; Swedish Nuclear Fuel Supply Co Report, KBS-TR-90; Swedish Nuclear Fuel and Waste Management Co: Solna, Sweden, 1978. [Google Scholar]
- Bourg, I.C.; Bourg, A.C.M.; Sposito, G. Modeling diffusion and adsorption in compacted bentonite: A critical review. J. Contam. Hydrol. 2003, 61, 293–302. [Google Scholar] [CrossRef]
- Macdonald, D.D.; Engelhardt, G.R. A critical review of radiolysis issues in water-cooled fission and fusion reactors: Part II, Prediction of corrosion damage in operating reactors. Corros. Mater. Degrad. 2022, 3, 694–758. [Google Scholar] [CrossRef]
- Fukaya, Y.; Watanabe, Y. Charcterization and prediction of carbon steel corrosion in diluted seawater containing pentaborate. J. Nucl. Mater. 2018, 498, 159–168. [Google Scholar] [CrossRef]
- Bennet, D.G.; Gens, R. Overview of European concepts for high-level waste and spent fuel disposal with special reference waste container corrosion. J. Nucl. Mater. 2008, 379, 1–8. [Google Scholar] [CrossRef]
- Wu, M.; Behazin, M.; Nam, J.; Keech, P. Internal Corrosion of Used Fuel Container; Nuclear Waste Management Organization Technical Report, NWMO-TR-2019-02; Nuclear Waste Management Organization (NWMO): Toronto, ON, Canada, 2019. [Google Scholar]
- Morco, R.P.; Joseph, J.M.; Hall, D.S.; Medri, C.; Shoesmith, D.W.; Wren, J.C. Modelling of radiolytic production of HNO3 relevant to corrosion of a used fuel container in deep geologic repository environments. Corros. Eng. Sci. Technol. 2017, 52, 141–147. [Google Scholar] [CrossRef]
- Stroes-Gascoyne, S.; Hamon, C.J.; Maak, P.; Russell, S. The effects of the physical properties of highly compacted smectite clay (bentonite) on the culturability of indigenous microorganisms. Appl. Clay Sci. 2010, 47, 155–162. [Google Scholar] [CrossRef]
- Haynes, H.M.; Bailey, M.T.; Lloyd, J.R. Bentonite barrier materials and the control of microbial processes: Safety case implications for the geological disposal of radioactive waste. Chem. Geol. 2021, 581, 120353. [Google Scholar] [CrossRef]
- Taborowski, T.; Bengtsson, A.; Chukharkina, A.; Blom, A.; Pedersen, K. Bacterial Presence and Activity in Compacted Bentonites. MIND (Microbiology In Nuclear waste Disposal) Report, Deliverable 2.4, Version 2, Issued 25 April 2019. Available online: https://ec.europa.eu/research/participants/documents/downloadPublic?documentIds=080166e5c38ed173&appId=PPGMS (accessed on 9 April 2020).
- King, F.; Kolàř, M.; Briggs, S.; Behazin, M.; Keech, P.; Diomidis, N. Review of the modelling of corrosion processes and lifetime prediction for HLW/SF containers—Part 2: Performance assessment models. Corros. Mater. Degrad. 2024, 5, 289–339. [Google Scholar] [CrossRef]
- Giroud, N.; Tomonaga, Y.; Wersin, P.; Briggs, S.; King, F.; Vogt, T.; Diomidis, N. On the fate of oxygen in a spent fuel emplacement drift in Opalinus Clay. Appl. Geochem 2018, 97, 370–378. [Google Scholar] [CrossRef]
- Hultquist, G. Hydrogen evolution in corrosion of copper in pure water. Corros. Sci. 1986, 26, 173–177. [Google Scholar] [CrossRef]
- Szakálos, P.; Hultquist, G.; Wikmark, G. Corrosion of copper by water. Electrochem. Solid-State Lett. 2007, 10, C63–C67. [Google Scholar] [CrossRef]
- Hedin, A.; Johansson, A.J.; Lilja, C.; Boman, M.; Berastegui, P.; Berger, R.; Ottosson, M. Corrosion of copper in pure O2-free water? Corros. Sci. 2018, 137, 1–12. [Google Scholar] [CrossRef]
- Strömberg, B.; Calota, E.; Liu, J.; Egan, M. Assessment of canister degradation for the encapsulation of spent nuclear fuel: Key research issues encountered in recent regulatory reviews and government decision making in Sweden. Adv. Geosci. 2023, 62, 31–39. [Google Scholar] [CrossRef]
- Ma, J.; Pekala, M.; Alt-Epping, P.; Pastina, B.; Maanoja, S.; Wersin, P. 3D modelling of long-term sulfide corrosion of copper canisters in a spent nuclear fuel repository. Appl. Geochem 2022, 146, 105439. [Google Scholar] [CrossRef]
- King, F.; Kolàř, M.; Puigdomenech, I.; Pitkänen, P.; Lilja, C. Modeling microbial sulfate reduction and the consequences for corrosion of copper canisters. Mater. Corros. 2021, 72, 339–347. [Google Scholar] [CrossRef]
- King, F.; Chen, J.; Qin, Z.; Shoesmith, D.; Lilja, C. Sulphide-transport control of the corrosion of copper canisters. Corros. Eng. Sci. Technol. 2017, 52, 210–216. [Google Scholar] [CrossRef]
- Koho, P.; King, F.; Prihti, T.; Salonen, T.; Koskinen, L.; Pastina, B. Treatment of canister corrosion in Posiva’s safety case for the operating licence application. Mater. Corros. 2023, 74, 1567–1579. [Google Scholar] [CrossRef]
- Chen, J.; Qin, Z.; Wu, L.; Noël, J.J.; Shoesmith, D.W. The influence of sulphide transport on the growth and properties of copper sulphide films on copper. Corros. Sci. 2014, 87, 233–238. [Google Scholar] [CrossRef]
- Chen, J.; Qin, Z.; Shoesmith, D.W. Key parameters determining structure and properties of sulphide films formed on copper corroding in anoxic sulphide solutions. Corros. Eng. Sci. Technol. 2014, 49, 415–419. [Google Scholar] [CrossRef]
- Salonen, T.; Lamminmäki, T.; King, F.; Pastina, B. Status report of the Finnish spent fuel geologic repository programme and ongoing corrosion studies. Mater. Corros. 2021, 72, 14–24. [Google Scholar] [CrossRef]
- Posiva. Canister Evolution; Working Report WR-2021-06; Posiva Oy: Eurajoki, Finland, 2021. [Google Scholar]
- Mao, F.; Dong, C.; Sharifi-Asl, S.; Lu, P.; Macdonald, D.D. Passivity breakdown on copper: Influence of chloride ion. Electrochim. Acta 2014, 144, 391–399. [Google Scholar] [CrossRef]
- Kong, D.; Xu, A.; Dong, C.; Mao, F.; Xiao, K.; Li, X.; Macdonald, D.D. Electrochemical investigation and ab initio computation of passive film properties on copper in anaerobic sulphide solutions. Corros. Sci. 2017, 116, 34–43. [Google Scholar] [CrossRef]
- Huttunen-Saarivirta, E.; Ghanbari, E.; Mao, F.; Rajala, P.; Carpén, L.; Macdonald, D.D. Kinetic properties of the passive film on copper in the presence of sulfate-reducing bacteria. J. Electrochem. Soc. 2018, 165, C450–C460. [Google Scholar] [CrossRef]
- Martino, T.; Smith, J.; Chen, J.; Qin, Z.; Noël, J.J.; Shoesmith, D.W. The properties of electrochemically-grown copper sulfide films. J. Electrochem. Soc. 2019, 166, C9–C18. [Google Scholar] [CrossRef]
- Martino, T.; Chen, J.; Guo, M.; Ramamurthy, S.; Shoesmith, D.W.; Noël, J.J. Comments on E. Huttunen-Saarivirta et al., “Kinetic Properties of the Passive Film on Copper in the Presence of Sulfate-Reducing Bacteria”, J. Electrochem. Soc. 165(9) (2018) C450–C460. J. Electrochem. Soc. 2019, 166, Y13–Y16. [Google Scholar] [CrossRef]
- Guo, M.; Daub, K.; Dong, Q.; Long, F.; Binns, W.J.; Daymond, M.R.; Shoesmith, D.W.; Noël, J.J.; Persaud, S.Y. The early-stage corrosion of copper materials in chloride and sulfide solutions: Nanoscale characterization and the effect of microstructure. J. Electrochem. Soc. 2022, 169, 031509. [Google Scholar] [CrossRef]
- Taniguchi, N.; Kawasaki, M. Influence of sulfide concentration on the corrosion behaviour of pure copper in synthetic seawater. J. Nucl. Mater. 2008, 379, 154–161. [Google Scholar] [CrossRef]
- Chen, J.; Qin, Z.; Martino, T.; Shoesmith, D.W. Non-uniform film growth and micro-macro-galvanic corrosion of copper in aqueous sulphide solutions containing chloride. Corros. Sci. 2017, 114, 72–78. [Google Scholar] [CrossRef]
- Chen, J.; Qin, Z.; Martino, T.; Guo, M.; Shoesmith, D.W. Copper transport and sulphide sequestration during copper corrosion in anaerobic aqueous sulphide solutions. Corros. Sci. 2018, 131, 245–251. [Google Scholar] [CrossRef]
- Chen, J.; Pan, X.; Nie, H.Y.; Kobe, B.; Bergendal, E.; Lilja, C.; Behazin, M.; Shoesmith, D.W.; Noël, J.J. Topographical and statistical studies of the corrosion damage underneath a sulfide film formed on a Cu surface. Corros. Sci. 2025, 248, 112801. [Google Scholar] [CrossRef]
- Yue, X.; Malmberg, P.; Isotahdon, E.; Ratia-Hanby, V.; Huttunen-Saarivirta, E.; Leygraf, C.; Pan, J. Penetration of corrosive species into copper exposed to simulated O2-free groundwater by time-of-flight secondary ion mass spectrometry (ToF-SIMS). Corros. Sci. 2023, 210, 110833. [Google Scholar] [CrossRef]
- Behazin, M.; Briggs, S.; King, F. Radiation-induced corrosion model for copper-coated used fuel containers. Part 1. Validation of the bulk radiolysis submodel. Mater. Corros. 2023, 74, 1834–1847. [Google Scholar] [CrossRef]
- Nagra. Design and Performance Assessment of HLW Disposal Canisters. National Cooperative for the Disposal of Radioactive Waste, Nagra Technical Report NTB 24-20. 2024. Available online: https://drbg.ch/rbg-gtl/referenzberichte/2768-ntb-24-20 (accessed on 22 June 2025).
- Calvo, E.J.; Schiffrin, D.J. The reduction of hydrogen peroxide on passive iron in alkaline solutions. J. Electroanal. Chem. 1984, 163, 257–275. [Google Scholar] [CrossRef]
- Vazquez, M.V.; de Sanchez, S.R.; Calvo, E.J.; Schiffrin, D.J. The electrochemical reduction of hydrogen peroxide on polycrystalline copper in borax buffer. J. Electroanal. Chem. 1994, 374, 179–187. [Google Scholar] [CrossRef]
- Van Ede, M.C.; Angst, U. Tafel sloped and exchange current densities of oxygen reduction and hydrogen on steel. Corros. Eng. Sci. Technol. 2024, 59, 39–55. [Google Scholar] [CrossRef]
- Sharifi-Asl, S.; Macdonald, D.D. Investigation of the kinetics and mechanism of the hydrogen evolution reaction on copper. J. Electrochem. Soc. 2013, 160, H382–H391. [Google Scholar] [CrossRef]
- Asakura, S.; Nobe, K. Electrodissolution kinetics of iron in chloride solutions. Part I. Neutral solutions. J. Electrochem. Soc. 1971, 118, 13–18. [Google Scholar] [CrossRef]
- King, F.; Litke, C.D.; Quinn, M.J.; LeNeveu, D.M. The measurement and prediction of the corrosion potential of copper in chloride solutions as a function of oxygen concentration and mass-transfer coefficient. Corros. Sci. 1995, 37, 833–851. [Google Scholar] [CrossRef]
- Briggs, S.; Behazin, M.; King, F. Validation of water radiolysis models against experimental data in support of the prediction of the radiation-induced corrosion of copper-coated used fuel containers. Corros. Mater. Degrad. 2025, 6, 14. [Google Scholar] [CrossRef]
- Enning, D.; Garrelfs, J. Corrosion of iron by sulfate-reducing bacteria: New views of an old problem. Appl. Environ. Microbiol. 2014, 80, 1226–1236. [Google Scholar] [CrossRef]
- Li, Y.; Xu, D.; Chen, C.; Li, X.; Jia, R.; Zhang, D.; Sand, W.; Wang, F.; Gu, T. Anaerobic microbiologically influenced corrosion mechanisms interpreted using bio energetics and bioelectrochemistry: A review. J. Mater. Sci. Tech. 2018, 34, 1713–1718. [Google Scholar] [CrossRef]
- Marciales, A.; Peralta, Y.; Haile, T.; Crosby, T.; Wolodko, J. Mechanistic microbiologically influenced corrosion modeling—A review. Corros. Sci. 2019, 146, 99–111. [Google Scholar] [CrossRef]
- Little, B.J.; Blackwood, D.J.; Hinks, J.; Lauro, F.M.; Marsili, E.; Okamoto, A.; Rice, S.A.; Wade, S.A.; Flemming, H.-C. Microbially influenced corrosion—Any progress? Corros. Sci. 2020, 170, 108641. [Google Scholar] [CrossRef]
- King, F. Microbiologically influenced corrosion of nuclear waste containers. Corrosion 2009, 65, 233–251. [Google Scholar] [CrossRef]
- Mijnendonckx, K.; Monsieurs, P.; Černá, K.; Hlaváčová, V.; Steinová, J.; Burzan, N.; Bernier-Latmani, R.; Boothman, C.; Miettinen, H.; Kluge, S.; et al. Molecular techniques for understanding microbial abundance and activity in clay barriers used for geodisposal. In The Microbiology of Nuclear Waste Disposal; Lloyd, J.R., Cherkouk, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 71–96. [Google Scholar]
- Huttunen-Saarivirta, E.; Rajala, P.; Bomberg, M.; Carpén, L. Corrosion of copper in oxygen-deficient groundwater with and without deep bedrock micro-organisms: Characterisation of microbial communities and surface processes. Appl. Surf. Sci. 2017, 396, 1044–1057. [Google Scholar] [CrossRef]
- Huttunen-Saarivirta, E.; Rajala, P.; Carpén, L. Corrosion behaviour of copper under biotic and abiotic conditions in anoxic ground water: Electrochemical study. Electrochim. Acta 2016, 203, 350–365. [Google Scholar] [CrossRef]
- Sushko, V.; Dressler, M.; Wei, S.T.-S.; Neubert, T.; Kühn, L.; Cherkouk, A.; Stumpf, T.; Matschiavelli, N. No signs of microbial-influenced corrosion of cast iron and copper in bentonite microcosms after 400 days. Chemosphere 2024, 363, 143007. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, R.; Černoušek, T.; Stoulil, J.; Kovářová, H.; Sihelská, K.; Špánek, R.; Ševců, A.; Steinová, J. Anaerobic microbial corrosion of carbon steel under conditions relevant for deep geological repository of nuclear waste. Sci. Total Environ. 2021, 800, 149539. [Google Scholar] [CrossRef]
- Grigoryan, A.A.; Jalique, D.R.; Medihala, P.; Stroes-Gascoyne, S.; Wolfaardt, G.M.; McKelvie, J.; Korber, D.R. Bacterial diversity and production of sulfide in microcosms containing uncompacted bentonites. Heliyon 2018, 4, e00722. [Google Scholar] [CrossRef]
- Černoušek, T.; Ševců, A.; Shrestha, R.; Steinová, J.; Kokinda, J.; Vizelková, K. Microbially influenced corrosion of container material. In The Microbiology of Nuclear Waste Disposal; Lloyd, J.R., Cherkouk, A., Eds.; Elsevier: Amsterdam; The Netherlands, 2021; pp. 119–136. [Google Scholar]
- Engel, K.; Ford, S.E.; Binns, W.J.; Diomidis, N.; Slayer, G.F.; Neufeld, J.D. Stable microbial community in compacted bentonite after 5 years of exposure to natural granitic groundwater. mSphere 2023, 8, e00048-23. [Google Scholar] [CrossRef]
- Maanoja, S.; Lakaniemi, A.-M.; Lehtinen, L.; Salminen, L.; Auvinen, H.; Kokko, M.; Palmroth, M.; Muuri, E.; Rintala, J. Compacted bentonite as a source of substrates for sulfate-reducing microorganisms in a simulated excavation-damaged zone of a spent nuclear fuel repository. Appl. Clay Sci. 2020, 196, 105746. [Google Scholar] [CrossRef]
- Taxén, C.; Núñez, A.M.; Lilja, C. Stress corrosion of copper in sulfide solutions: Variation on pH-buffer, strain rate, and temperature. Mater. Corros. 2023, 74, 1632–1644. [Google Scholar] [CrossRef]


| Medium | Effective Diffusivity (cm2/s) | Typical Diffusion Layer Thickness δ (cm) | Steady-State Mass-Transfer Coefficient D/δ (cm/s) |
|---|---|---|---|
| Compacted bentonite | 10−7 | 30 | 3 × 10−9 |
| Stagnant laboratory solution | 10−5 | 0.01–0.1 | 10−4–10−3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
King, F. The Importance of Considering the Service Environment When Studying and Predicting the Performance of Corrodible Structures. Corros. Mater. Degrad. 2026, 7, 8. https://doi.org/10.3390/cmd7010008
King F. The Importance of Considering the Service Environment When Studying and Predicting the Performance of Corrodible Structures. Corrosion and Materials Degradation. 2026; 7(1):8. https://doi.org/10.3390/cmd7010008
Chicago/Turabian StyleKing, Fraser. 2026. "The Importance of Considering the Service Environment When Studying and Predicting the Performance of Corrodible Structures" Corrosion and Materials Degradation 7, no. 1: 8. https://doi.org/10.3390/cmd7010008
APA StyleKing, F. (2026). The Importance of Considering the Service Environment When Studying and Predicting the Performance of Corrodible Structures. Corrosion and Materials Degradation, 7(1), 8. https://doi.org/10.3390/cmd7010008






