1. Introduction
Stainless steels represent one of the most versatile and sustainable classes of engineering materials, widely utilized in energy, transportation, chemical processing, and emerging hydrogen and additive manufacturing sectors. Their outstanding corrosion resistance is due to chromium rich oxide film that protects the underlying metal even in aggressive environments. However, this corrosion reliability is not absolute as it depends on the microstructural stability of the alloy. Thermodynamically stable but corrosion-sensitive secondary phases, such as sigma (σ), chi (χ), Laves, or chromium-rich carbides and nitrides, frequently precipitate in stainless steels during fabrication or long-term service at intermediate temperatures (500–950 °C). These phases cause electrochemical heterogeneity by reducing chromium and molybdenum from nearby areas, ultimately affecting the quality of the passive film, i.e., corrosion performance of these alloys, along with increased susceptibility towards hydrogen-induced cracking. Diffusion kinetics and thermodynamic driving forces interact to control the formation of secondary phases. The σ phase is a Cr and Mo-rich tetragonal intermetallic, which forms between 600 °C and 950 °C in duplex and super-duplex grades. As it is mainly formed in the ferrite phase, the surrounding areas are reduced in Cr content, which ultimately degrades the corrosion and embrittlement resistance as reported elsewhere, not only for sigma but also for Cr rich α’ phase. While Laves-type Fe
2(Mo, Nb, W) phases form in Mo-bearing steels under comparable thermal exposures, the χ phase, which is frequently seen as a metastable precursor to σ, is rich in Mo and Nb. In austenitic stainless steels, chromium-depleted zones, prone to intergranular corrosion, are created when M
23C
6 carbides and Cr
2N nitrides preferentially nucleate along grain boundaries in the sensitization temperature range of 500–850 °C [
1,
2,
3,
4]. The chemistry of the alloy, particularly the concentrations of Cr, Mo, N, W, and Si, as well as previous thermomechanical history, has a significant influence on the formation of these phases. CALPHAD and DICTRA-based simulations, which include diffusion data and Gibbs free-energy functions for complicated multicomponent systems, can be used to accurately estimate the thermodynamic stability and transformation kinetics of these phases [
5,
6,
7].
The precise effect of secondary phases on corrosion resistance is still being studied in austenitic, duplex, and lean stainless steel alloys [
8,
9,
10]. While molybdenum partitioning further modifies the protective qualities of the passive film, chromium depletion at the phase matrix interface can lower the local chromium level below 12 wt.% required to maintain passivity in these alloys. Micro-galvanic cells that promote localized corrosion are created at the nanoscale due to the electrochemical potential difference between intermetallics and the matrix. The cathodic nature of the σ or χ phases and their function in pitting initiation is reported to be due to a potential difference of up to 200 mV, when measured using scanning Kelvin probe force microscopy (SKPFM) [
11]. This galvanic coupling due to variation in potential explains why there is a significant decrease in pitting potential (E
pit) and repassivation ability, even by small fractions (<1 vol. %) of σ phase. Furthermore, as these intermetallic phases behave in a brittle manner, they can contribute in environmental induced cracking, such as hydrogen embrittlement. Therefore, from a corrosion perspective, secondary phases are considered undesirable as they compromise the localized corrosion resistance due to the combined effects of compositional depletion, galvanic interactions, and local stress concentration.
Over the recent years, due to advancements in characterization tools, the understanding regarding the precipitation of these phases and subsequent issues has evolved significantly. With the advent of three-dimensional compositional mapping with near-atomic precision using atom probe tomography (APT), nanoscale Cr/Mo enrichment within the σ and χ phases and associated depletion zones in the nearby austenite can be accurately determined [
12]. The validation of thermodynamic and kinetic predictions is made possible by real-time insights into phase nucleation and growth during thermal exposure provided by transmission electron microscopy (TEM) and in situ synchrotron X-ray diffraction. The long-standing discrepancy between equilibrium phase diagrams and real microstructures created under industrial processing settings is addressed by these experimental developments. Direct correlation between nanoscale phase chemistry and localized corrosion susceptibility is now achievable due to the combination of APT, high-resolution TEM, and correlative electron backscatter diffraction (EBSD), which was previously impossible with conventional microscopy [
13].
Alongside these analytical advances, alloy design has been transformed by computational materials science. Precipitation sequences, phase fractions, and composition gradients may now be predicted during material processing using phase-field models and simulations based on CALPHAD. The integration of machine learning with CALPHAD datasets has greatly accelerated σ-phase susceptibility predictive modeling and pitting-resistance equivalent number (PREN) optimization. Data-driven frameworks enable multi-objective optimization by balancing mechanical strength, corrosion resistance, and processability across wide compositional spaces. These computerized tools guide the creation of next-generation stainless steels that simultaneously meet sustainability and performance standards.
With all of these developments and sustainability considerations in mind, attempts are being made to create affordable and lightweight stainless steels because traditional Ni and Mo-rich alloys are expensive and energy-intensive. Thus, the goal of many ongoing research efforts is to develop lean Ni/Mo or Ni-free duplex and austenitic grades using intelligent microalloying and thermal management without sacrificing the corrosion resistance of the alloys [
14,
15,
16,
17,
18]. For instance, it has been demonstrated that nitrogen is an excellent austenite stabilizer, increases PREN, and also prevents the formation of the σ-phase, so it can be used to lower the Ni content of the new alloys. Similarly, Fe–Mn–Al–C lightweight stainless steels have attracted attention due to their superior strength and ductility. By replacing σ-phase precipitation with κ-carbides (Fe
3AlC), these new alloys address chromium depletion issues and so improve localized corrosion/embrittlement resistance.
The secondary phase precipitation is undesirable from a sustainability perspective because it compromises stainless steel life-cycle performance. Due to the relationship between phase stability, corrosion reliability, and environmental impact, the development of new stainless steels aligns with global sustainability goals. After post-build heat treatment, segregation-induced secondary phases may emerge as a result of the nonequilibrium microstructures produced by the additive manufacturing (AM) methods, due to extremely rapid cooling rates and sharp temperature gradients. Therefore, computationally guided alloy chemistries and optimized thermal cycles are required to manage these changes and improve the corrosion performance of AM stainless steels.
Over the past few years, the science of analyzing and managing secondary phases in stainless steels has evolved from descriptive to predictive, and ongoing research endeavors integrate data-driven optimization, real-time characterization, and thermodynamic modeling to tackle associated issues. Understanding and controlling secondary phase precipitation is essential for designing stainless steels that not only resist corrosion in challenging environments but also contribute to the global transition to low-carbon, resource-efficient alloy systems.
2. Classification and Formation of Secondary Phases
Any crystalline component that is different from the basic matrix phases, austenite (γ, FCC), ferrite (α, BCC), or martensite (α′, BCT), is referred to as the secondary phase in stainless steel. When the solid solution becomes thermodynamically or kinetically unstable, these secondary constituents may develop during solidification or later during thermal or mechanical exposure. When the Gibbs free-energy decrease linked to phase separation surpasses the interfacial-energy, precipitation or segregation takes place. Typical transformation pathways include “continuous spinodal decomposition within ferrite (α → α + α′), discontinuous precipitation of carbides or nitrides along grain boundaries, and nucleation of ordered intermetallic compounds such as σ or χ phases”, respectively [
19,
20]. These processes collectively govern microstructural stability and, consequently, corrosion behavior.
Thermodynamic calculations using the CALPHAD methodology have enabled detailed mapping of equilibrium phase fields in multicomponent Fe–Cr–Ni–Mo–N systems. At the atomic scale, chromium acts as the principal ferrite stabilizer, while nickel and nitrogen favor austenite. Increasing chromium and molybdenum contents expands the σ-phase stability region between roughly 600 and 950 °C; tungsten and silicon have similar effects, whereas nitrogen retards σ formation by enlarging the γ-phase domain. Molybdenum, though indispensable for pitting resistance, increases the driving force for σ and χ precipitation. Hence, alloy design inevitably involves balancing corrosion resistance and phase stability [
21].
Spinodal decomposition of ferrite is particularly relevant in lean duplex and ferritic stainless steels exposed to 400–550 °C. In this range, the solid solution separates into Cr-rich α′ and Fe-rich α domains without the need for nucleation. The resulting nanometric compositional modulation (≈10 nm wavelength) leads to 475 °C embrittlement and elevated corrosion rates due to chromium clustering [
22]. CALPHAD-based thermodynamic databases have successfully reproduced such miscibility gaps, validating the predicted spinodal boundaries against atom-probe tomography (APT) and small-angle neutron-scattering data. In contrast, the discontinuous precipitation of carbides (M
23C
6, M
6C) or nitrides (Cr
2N) along grain boundaries is nucleation-controlled and highly sensitive to local carbon, nitrogen, and chromium activities. These reactions occur preferentially in austenitic grades during thermal exposures between 500 and 850 °C, the classical sensitization window [
23,
24]. The principal compositional ranges and phase-stability features of representative stainless-steel families are summarized in
Table 1.
2.1. Key Secondary Phases and Their Characteristics
Among all secondary phases, the σ phase has received the greatest attention because of its pronounced effects on both mechanical and corrosion properties. It is a Cr and Mo-rich intermetallic compound with a complex tetragonal structure (space group P42/mnm), typically containing ≈40–50 wt % Cr, 4–10 wt % Mo, and the remainder Fe. The σ phase forms primarily at α/γ interfaces in duplex stainless steels and within ferrite grains in high Cr alloys during prolonged exposure at 600–950 °C. Its precipitation consumes chromium and molybdenum from the matrix, thereby reducing the local pitting-resistance equivalent number (PREN) and degrading toughness. Even short-term exposure (such as 1 h at 850 °C) can decrease the impact energy of SAF 2507 by >80%. Because σ is thermodynamically stable, its complete dissolution requires solution annealing above 1050–1100 °C followed by rapid quenching.
The χ phase is a cubic intermetallic enriched in Mo and Nb, usually appearing as a metastable precursor to σ. It precipitates at intermediate temperatures (600–900 °C) and transforms to σ upon prolonged aging through Cr/Mo diffusion. Despite being less common, χ can effectively absorb molybdenum and encourage galvanic reactions with the surrounding matrix [
25]. In Mo or Nb-bearing steels, Laves-type Fe
2(Mo, Nb, W) phases usually occur between 550 and 750 °C and have hexagonal structures (C14 or C36) [
26]. Despite often having a modest volume fraction, they help pit initiation in chloride medium and introduce potent micro-galvanic effects. Complex degradation morphologies can be produced by the coexistence of the σ, χ, and Laves phases in high-temperature service environments like desalination units or petrochemical reformers.
Chromium-rich carbides (M
23C
6) are among the earliest identified secondary phases in austenitic stainless steels. They nucleate on grain boundaries or dislocations between 500 and 850 °C, producing a Cr-depleted zone of ≈2–5 µm adjacent to the carbide/matrix interface. Once the local chromium concentration falls below ≈12 wt %, the passive film loses stability, and intergranular corrosion (IGC) proceeds rapidly. M
6C carbides, rich in molybdenum and tungsten, occur in high-alloyed austenitic or super austenitic grades during long-term aging. Similarly, Cr
2N precipitates appear readily in nitrogen-alloyed steels when the nitrogen solubility in ferrite decreases, such as during rapid cooling from the solution-annealing temperature or within the heat-affected zone (HAZ) of welds [
27]. These nitrides compromise ductility and corrosion resistance, particularly in duplex and lean duplex grades. Hosseini et al. [
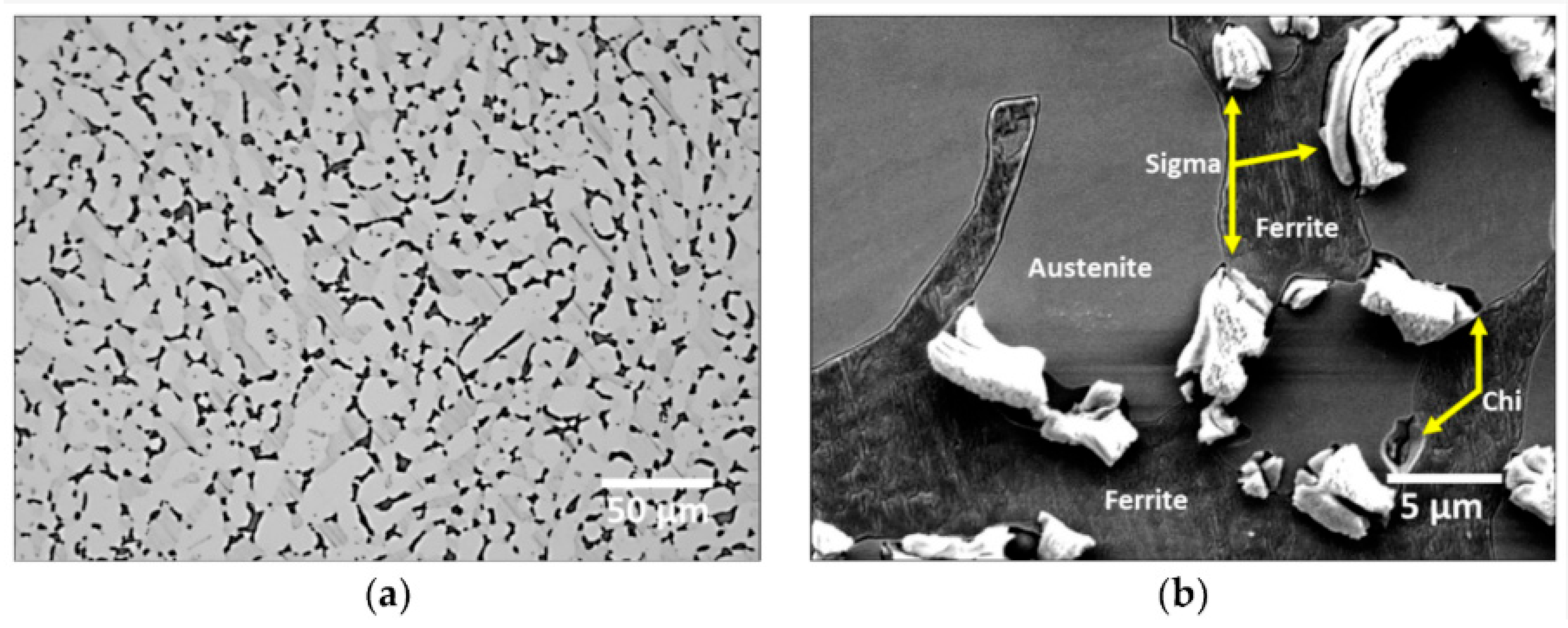
28] reported the distribution and shape of the sigma phase precipitation in a functionally graded microstructure at temperatures ranging from 630 to 1010 degrees Celsius under different agitation settings (
Figure 1). The figure clearly shows the presence of several phases, including δ, main γ, σ, and χ, as well as nitrides and the R phase.
The function of carbides and nitrides in the nucleation of the χ-phase and σ-phase, respectively, was covered by Llorca-Isern et al. [
29]. Although the σ-phase and χ-phase grow concurrently, the amount of χ-phase is typically higher at the beginning. Some alloying elements, such as Cr, Si, and Mo, speed up the creation of these phases. The challenge of sigma phase nucleation at ferrite/ferrite borders is the primary cause of this. According to the authors, the χ-phase changes to the σ-phase with longer annealing times, although the early production of Cr
23C
6 aids in the nucleation of the new intermetallic phases.
Other less common but technologically relevant phases include the G-phase (Ni
16Si
7Nb
6), R-phase (Fe–Mo-rich hexagonal phase), and α′ martensite formed through spinodal decomposition. The G-phase precipitates during long-term aging of ferritic and precipitation-hardening grades, while the R-phase occurs between 550 and 650 °C in Mo-bearing alloys and often transforms to σ or Laves with continued exposure. Spinodal α′ domains, enriched in chromium, induce 475 °C embrittlement and act as anodic sites during corrosion testing. The collective presence of these phases explains the wide variety of microstructural degradation pathways observed in different stainless-steel families. The major secondary phases, their structural features, and corrosion implications are listed in
Table 2.
Typical transformation behavior is conveniently summarized using time–temperature–transformation (TTT) and time–temperature–composition (TTC) diagrams [
30]. Such diagrams, derived from isothermal aging and thermodynamic assessments, reveal that relatively small changes in Mo or N contents can shift σ-phase start temperatures by ±50 °C. These datasets are being used to optimize solution-annealing or post-weld heat-treatment regimens in industrial process simulations. For instance, duplex steels with around 25% Cr and 4% Mo must not be exposed to temperatures between 700 and 950 °C, as this is when σ and χ precipitate most quickly. On the other hand, the increased austenite field in nitrogen-enriched lean duplex alloys (22–23 weight percent Cr, 0.2–0.3 weight percent N) results in noticeably slower kinetics.
2.2. Precipitation Kinetics and Modeling
The nucleation-and-growth hypothesis, which describes the precipitation behavior of secondary phases classically, states that the diffusion coefficients of solute components, interfacial energy, and thermodynamic driving force all affect transformation rates. While austenite serves as a reservoir for Ni and N, σ-phase growth in duplex stainless steels is diffusion-regulated and primarily controlled by Cr and Mo transport in ferrite. CALPHAD thermodynamics is extended by contemporary computational tools like DICTRA and TC-PRISMA to model the time-dependent evolution of precipitate fractions, particle sizes, and morphologies. These kinetic simulations anticipate σ-phase fractions within ±10% of measured values, accurately reproducing experimental data from transmission electron microscopy and in situ X-ray diffraction experiments.
When kept in solid solution, alloying metals such as molybdenum and chromium improves overall corrosion resistance while simultaneously increasing the driving force for σ formation. By decreasing ferrite supersaturation, nitrogen stabilizes austenite and considerably delays σ nucleation. While tungsten replaces molybdenum and favors Laves-phase stability, silicon enhances chromium partitioning into ferrite, which promotes σ production. Carbon and nitrogen can be trapped by trace additions of Nb or Ti, resulting in the formation of tiny carbides or nitrides that decrease M23C6 and Cr2N precipitation. Thus, the primary lever for phase management is provided by the combined control of composition and heat treatment.
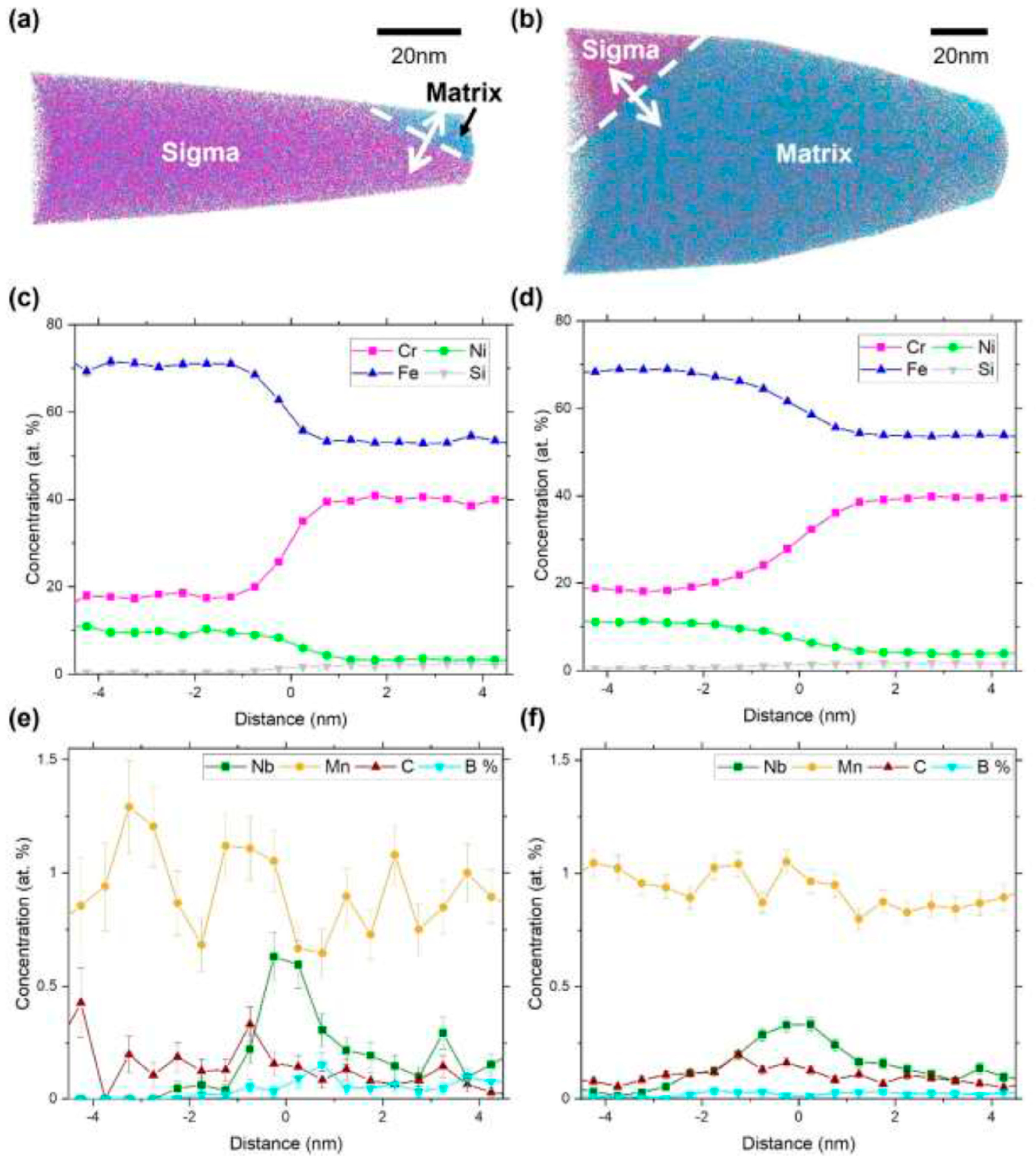
After heat treatment at 1000 °C, Shin et al. [
31] reported the crystallization of secondary phases in SAF2507 at austenite/ferrite boundaries (
Figure 2a). The phase precipitation followed the grain boundaries, and, at higher magnifications, they also observed ferrite and austenite at the top (
Figure 2b). They verified the existence of a low corrosion resistance (Cr deficient Chi phase) and a greater corrosion resistance sigma phase, which was Cr rich.
Steep temperature gradients and brief thermal cycles during welding produce nonequilibrium solidification pathways that encourage heterogeneous σ, χ, and Cr2N nucleation in the HAZ. These phases are frequently irregular and may partially dissolve when heated, but if they are left in place, they significantly impair corrosion resistance and toughness. Because of their incredibly fast rates of heating and cooling, laser and electron-beam welds display exceptionally fine secondary-phase networks. The dynamic nature of these transitions has been demonstrated by transitory σ-phase production and dissolution within milliseconds by phase-field simulations in conjunction with high-speed thermal photography. Submicron segregation patterns of Cr and Mo are produced at cooling rates exceeding 103–106 K s−1 in additive manufacturing (AM) methods like selective-laser melting (SLM) or directed-energy deposition (DED). These patterns can develop into transitory σ or Cr2N precipitates after subsequent heat treatment. It usually takes solution annealing at temperatures from 1050 to 1150 °C to homogenize these AM microstructures.
Predictive modeling of phase formation under arbitrary temperature histories is now possible because of the integration of machine learning (ML) with the CALPHAD and DICTRA databases. Incubation periods and σ-phase start temperatures can be estimated in a matter of seconds using neural-network and Gaussian-process models that have been trained on thermodynamic datasets. In order to reduce undesired precipitation during production, these computerized frameworks provide process-parameter optimization, including scan speed, layer thickness, and cooling rate. Data-driven thermokinetic modeling provides previously unheard-of precision for predicting phase stability in complicated alloys when paired with experimental confirmation via APT and synchrotron research [
28]. In the end, the way secondary phases are categorized and formed in stainless steels demonstrates the fine balance that exists between chemistry, processing, and performance. Researchers may now create microstructures that optimize corrosion resistance while preserving mechanical integrity and sustainability by fusing thermodynamic predictions, kinetic modeling, and sophisticated characterization.
4. Effect of Secondary Phases on Corrosion Performance
Electrochemical and microstructural studies have demonstrated that σ-phase precipitation significantly degrades the corrosion resistance of duplex stainless steels by promoting anodic dissolution of the surrounding ferritic matrix. Polarization measurements show that increasing σ-phase volume fraction lowers the corrosion potential and increases the corrosion rate, while post-corrosion XRD and EPMA analyses reveal preferential dissolution of the α phase adjacent to σ precipitates. This behavior is attributed to the formation of localized Cr- and Mo-depleted zones around σ phase, which destabilize passivity and act as initiation sites for corrosion [
3]. Electrochemical studies on UNS S32205 duplex stainless steel have demonstrated that σ-phase precipitation during continuous cooling markedly degrades corrosion resistance, as evidenced by reduced corrosion and pitting potentials and increased corrosion rates. The formation of σ phase enriches Cr and Mo within the intermetallic while creating adjacent chromium-depleted ferritic regions, which act as anodic sites and promote localized corrosion and pit initiation in chloride-containing environments. Furthermore, impedance spectroscopy reveals that increasing σ-phase fraction disrupts the integrity of the passive film, leading to lower charge-transfer resistance and enhanced susceptibility to pitting corrosion [
45]. Combined in situ mechanical testing, digital image correlation, and atomistic modelling studies have shown that hydrogen-induced degradation in duplex stainless steels is strongly governed by phase and interphase boundary interactions. Hydrogen preferentially accumulates and diffuses along ferrite–austenite boundaries, leading to intense strain localization and accelerated embrittlement through phase-dependent hardening and softening responses. These findings highlight that secondary-phase networks and interphase boundaries act as critical pathways for environmentally assisted damage, linking microstructural stability directly to corrosion- and hydrogen-related failure mechanisms [
46].
The magnitude of the Volta potential difference between the σ phase and the surrounding matrix depends on alloy chemistry, particularly molybdenum content, and environmental exposure, and typically lies in the range of ~150–250 mV. Correlative studies combining SKPFM with finite-element simulations of ionic transport have demonstrated that pit-initiation probability is governed by steep potential gradients and local chromium-depleted halos adjacent to σ precipitates, rather than the σ phase itself. Complementary transmission electron microscopy and in situ XPS analyses of passive films reveal pronounced chemical heterogeneity in these regions, with an increased fraction of Fe- and Mo-rich hydroxides and a discontinuous, locally depleted Cr
2O
3 layer, leading to enhanced point-defect transport and reduced film protectiveness. Time-resolved electrochemical impedance spectroscopy further shows that increasing σ-phase fraction is accompanied by a significant rise in passive-film capacitance, indicative of thinner and more defective oxide layers, thereby promoting localized corrosion in the matrix regions surrounding intermetallic precipitates [
47,
48].
Table 4 summarizes the relationship between particular secondary phases and the corrosion modes that correspond to them. Potentiodynamic polarization combined with metastable pit analysis has been widely used to quantify the effect of σ-phase precipitation on pitting resistance in duplex stainless steels. For alloys aged near 850 °C, even small σ-phase fractions (≈1–3 vol %) cause a pronounced decrease in pitting potential (E
pit) on the order of 200–300 mV and a marked increase in metastable pit frequency, reflecting the formation of chromium- and molybdenum-depleted zones adjacent to σ precipitates. Localized electrochemical measurements using scanning droplet and microcapillary techniques further demonstrate that pit initiation occurs preferentially in austenitic regions adjacent to σ phase, where the local PREN drops well below that of the bulk alloy, confirming that pitting susceptibility is governed by local chemistry rather than nominal composition [
49,
50].
Thermal aging of super duplex stainless steel at intermediate temperatures promotes nanoscale phase separation within ferrite, leading to chromium-rich and chromium-depleted regions that markedly degrade pitting corrosion resistance. Potentiodynamic polarization measurements show a pronounced decrease in pitting potential and passivation range with increasing extent of phase separation, which correlates closely with the growth of Cr-depleted zones quantified by small-angle neutron scattering. These findings demonstrate that corrosion susceptibility in aged duplex stainless steels is governed by nanostructure-induced chemical heterogeneity rather than by the nominal bulk composition alone [
4]. Liu et al. [
51] investigated the effect of aging on the corrosion behavior of DSS and reported that, up to 24 h of aging, corrosion resistance was decreased; however, it increased with aging from 48 to 96 h. Though this did not change the pitting potential, passivity had almost disappeared, and this deterioration was linked with the presence of sigma phase precipitation. A quantitative relation between carbide coverage and grain-boundary corrosion rate has been established for sensitized austenitic steels. When the areal fraction of continuous M
23C
6 films exceeds ≈ 0.6, intergranular penetration increases exponentially with potential above the corrosion threshold. Transmission electron microscopy studies have shown that chromium-depleted zones extending several micrometers from carbide interfaces can develop in stainless steels, creating electrochemical gradients sufficient to sustain localized anodic dissolution even under mildly oxidizing conditions. In duplex stainless steels, a similar degradation mechanism occurs at Cr
2N-decorated ferrite grain and phase boundaries, although its severity is strongly influenced by nitrogen solubility in ferrite; solution heat treatments above ~1050 °C followed by rapid quenching effectively dissolve Cr
2N precipitates and restore chromium homogeneity in the surrounding matrix [
52]. Isothermal aging of duplex stainless steels promotes the precipitation of secondary phases at ferrite–austenite interfaces, leading to the formation and progressive widening of chromium-depleted zones that strongly reduce pitting resistance. Electrochemical measurements reveal a pronounced decrease in pitting potential and critical pitting temperature with increasing aging time, accompanied by a transition from metastable pit formation at phase boundaries to stable pit growth within chromium-depleted regions. These observations confirm that localized chemical heterogeneity induced by secondary-phase precipitation governs pitting initiation and its evolution from localized to more generalized corrosion [
53].
Fractographic analyses of hydrogen-charged duplex stainless steels show that σ-phase clusters within ferrite act as preferential sites for quasi-cleavage fracture, consistent with brittle cracking under hydrogen embrittlement conditions. Thermal desorption spectroscopy demonstrates that σ-containing microstructures absorb up to several times more diffusible hydrogen than σ-free counterparts, with characteristic activation energies of ~28–32 kJ·mol
−1, indicative of reversible trapping at σ/matrix interfaces. These interfaces act as combined lattice and dislocation trapping sites, while residual tensile stresses surrounding rigid σ precipitates further enhance local hydrogen accumulation through stress-assisted diffusion, whereas increasing austenite fraction or introducing traps such as TiN can mitigate hydrogen embrittlement by redistributing hydrogen away from critical interfaces [
54]. Wang et al. [
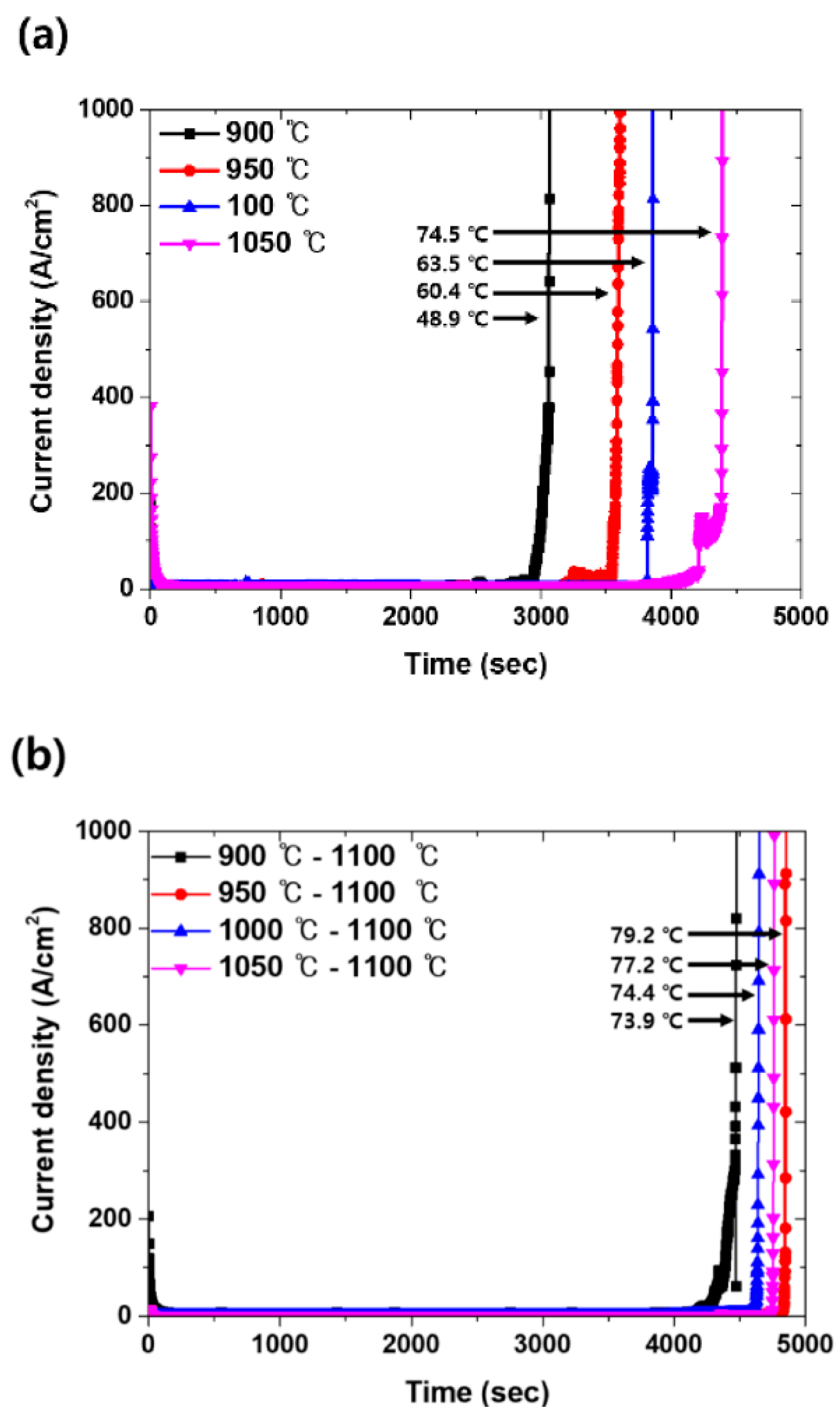
55] investigated the effect of secondary phase precipitation at different solution annealing temperatures on corrosion resistance and reported the best corrosion performance at 1100 °C, as shown in
Figure 6a,b. Before and after this temp., the corrosion current density was high, and it was related to the precipitation of secondary phases. Due to the formation of Cr and Mo-rich phases (σ phase and Cr
2N), passive film quality was significantly degraded, and so the corrosion rate was increased. Solution annealing at 1100 °C helped to dissolve such phases and homogenize the alloying concentration, which improved the passive film quality.
The influence of secondary phases on the oxidation behavior of duplex stainless steels can be rationalized by considering local chemical redistribution and its impact on the formation and adherence of protective oxide scales. The precipitation of σ phase leads to chromium and molybdenum enrichment within the intermetallic and concomitant depletion in the surrounding matrix, which hinders the development of a continuous, adherent chromium-rich oxide layer. As a result, σ-containing microstructures exhibit accelerated oxidation and poorer scale integrity compared with solution-annealed conditions, where a more uniform chemistry promotes the formation of stable, protective oxides. These observations highlight the importance of controlling σ-phase precipitation through appropriate heat treatment for components intended for elevated-temperature service [
56]. The oxidation behavior of duplex stainless steels is strongly influenced by secondary intermetallic phases through their impact on local chemistry and oxide-scale integrity. While solution-annealed microstructures tend to form adherent, chromium-rich protective scales, the precipitation of σ and other refractory-element-rich intermetallics disrupts chromium availability and promotes non-uniform oxide growth, leading to accelerated oxidation and scale spallation during elevated-temperature exposure, thereby necessitating post-aging homogenization for high-temperature service [
56].
Spatially resolved electrochemical techniques provide a quantitative link between σ-phase morphology and localized corrosion kinetics in duplex stainless steels. Local electrochemical impedance mapping combined with EBSD reveals that the low-frequency impedance modulus decreases markedly with increasing σ-phase connectivity, even at comparable overall σ fractions, indicating that corrosion propagation is governed by intermetallic network morphology rather than bulk phase content. Finite-element current-distribution modelling reproduces this behavior by showing that interconnected σ regions act as preferential cathodic pathways, accelerating anodic dissolution in the adjacent ferrite. In welded duplex stainless steels, micro-EIS and scanning Kelvin probe force microscopy further reveal pronounced spatial gradients in Volta potential across the heat-affected zone, associated with σ- and Cr
2N-rich precipitation bands formed during repeated thermal cycles; these gradients translate into locally reduced depassivation potentials, which can be mitigated by post-weld heat treatments that dissolve intermetallics and homogenize nitrogen distribution. Together, these findings demonstrate that σ-phase evolution can be quantitatively correlated with spatial variations in corrosion driving force and kinetics through combined experimental mapping and thermodynamic–electrochemical modelling approaches [
57,
58].
5. Mitigation and Control Strategies
From an alloy-design perspective, controlling the concentrations of chromium, molybdenum, nitrogen, and tungsten is critical to achieving high corrosion resistance without excessively expanding the σ- and χ-phase stability fields [
59]. In duplex and super-duplex stainless steels, the thermodynamic driving force for σ formation increases sharply as chromium and molybdenum contents rise beyond the typical ranges of ~22–27 wt.% Cr and ~2–4 wt.% Mo, owing to enhanced partitioning of these elements into ferrite. Nitrogen additions in the range of ~0.15–0.30 wt.% are particularly effective because they stabilize austenite, reduce chromium activity in ferrite, and shift σ-phase precipitation to lower kinetics and longer incubation times, thereby delaying σ nucleation during thermal exposure or welding [
5]. Tungsten must be carefully controlled, as moderate additions improve pitting resistance in a manner similar to molybdenum, whereas excessive tungsten contents promote the formation of Laves phases that further reduce microstructural stability [
59]. Consequently, increasing the pitting resistance equivalent number (PREN = %Cr + 3.3 × %Mo + 16 × %N) through excessive molybdenum enrichment may paradoxically enhance σ susceptibility, despite improving localized corrosion resistance. CALPHAD-based thermodynamic and kinetic calculations provide a robust framework for resolving this trade-off, indicating that an optimal balance between corrosion resistance and phase stability is typically achieved by maintaining PREN values in the range of ~40–45 and restricting the σ-phase stability window to approximately 650–950 °C [
60]. Such computational approaches are increasingly adopted in industrial alloy and welding design to define composition–processing windows and reduce reliance on extensive empirical testing.
In addition to compositional control, grain-boundary engineering represents a powerful strategy for mitigating σ-phase precipitation in duplex stainless steels. Increasing the fraction of low-Σ coincidence site lattice (CSL) boundaries reduces the density of high-energy grain boundary sites available for heterogeneous σ nucleation. Experimental studies have demonstrated that thermomechanical routes promoting recrystallization and grain boundary alignment, such as controlled hot rolling followed by short annealing treatments, can reduce σ nucleation density by up to ~50% compared with conventionally processed microstructures [
61]. A complementary approach relies on rapid solidification techniques to suppress elemental segregation and shorten the effective diffusion length for σ-forming solutes. In highly alloyed, high-Mo compositions, processes such as strip casting or laser surface melting produce fine cellular or sub-grain microstructures with enhanced chemical homogeneity, effectively suppressing σ precipitation even under conditions where it would normally be thermodynamically favored [
62]. Phase stability can be further improved through targeted micro-alloying additions. Elements such as Ti and Nb preferentially form stable carbides and nitrides, thereby scavenging carbon and nitrogen that would otherwise promote the formation of deleterious M
23C
6 carbides or Cr
2N nitrides. Rare-earth additions (e.g., Ce and La) act as strong oxygen and sulfur getters, refining inclusion populations and providing more uniform heterogeneous nucleation sites during solidification, which reduces macro-segregation and suppresses secondary-phase precipitation. Taken together, the combination of compositional design, grain-boundary engineering, and rapid-solidification processing offers an effective pathway to enhance intrinsic resistance to σ-phase instability without compromising corrosion performance. Different heat treatments (ranging from 900 to 1050 °C) were performed by Kim et al. [
63] in order to comprehend the microstructural alterations in super duplex stainless steels. They stated that secondary phase precipitation served as a phase boundary and that the austenite phase was present as islands in the ferrite matrix. They noted that there was a lot of secondary precipitation between 900 and 1000 °C, and that the chemical composition of Chi was poor in Cr and Mo, whereas that of Sigma was rich in both elements. As seen in
Figure 7, the volume fraction of ferrite and secondary phase changed significantly as a result of variations in the Cr content, but austenite showed no change.
Table 5 summarized the mitigation and control strategies.
5.1. Thermomechanical Processing and Post-Treatment
Optimizing heat treatment is still the mainstay of secondary-phase mitigation. Dissolving pre-existing intermetallics and avoiding reprecipitation during cooling is the general idea. By spreading Mo and N evenly across ferrite and austenite and dissolving σ, χ, and Cr
2N precipitates, solution annealing between 1050 °C and 1150 °C, followed by quick water or gas quenching, restores homogeneity [
62]. To prevent secondary σ production inside the crucial 700–950 °C range, the cooling rate must be greater than ≈30 °C s
−1. When compared to their older equivalents, duplex steels treated in this way show a recovery of pitting potential (Epit) of more than 200 mV. Homogenization at intermediate temperatures is especially important after welding or additive manufacturing, where rapid solidification produces strong compositional gradients. Such treatment reduces local δ-ferrite supersaturation and rebalances phase fractions to the nominal 50:50 ratio required for optimum mechanical and corrosion properties. Subcritical annealing below 900 °C must be avoided because it accelerates χ → σ transformation and triggers carbide precipitation. Thermomechanical processing provides further control by manipulating deformation and recrystallization kinetics. Controlled forging or rolling just below the δ/γ solvus refines grain size, enhances diffusion pathways for homogenization, and disrupts continuous σ networks formed during prior aging. Studies show that deformation strains of 10–20% followed by short annealing cycles (≈2–5 min at 1050 °C) yield fine, equiaxed duplex microstructures free of intermetallics [
63]. Warm deformation of austenitic steels within 700–800 °C combined with subsequent rapid quenching can also minimize M
23C
6 precipitation by decreasing boundary segregation.
Surface modification techniques are increasingly employed to complement bulk alloy design and heat treatment strategies for controlling deleterious secondary phases. Laser surface remelting (LSR) is particularly effective because it induces localized melting and rapid re-solidification, enabling the dissolution or removal of surface-connected σ phase, carbides, and other corrosion-active heterogeneities while simultaneously refining the near-surface microstructure. The extremely high cooling rates associated with LSR (typically exceeding 10
4 K s
−1) suppress micro-segregation and promote a more homogeneous redistribution of chromium and nitrogen, leading to the formation of thicker, chromium-rich passive films with improved electrochemical stability [
64]. As a result, duplex and austenitic stainless steels subjected to optimized LSR treatments exhibit significantly enhanced pitting resistance in chloride-containing environments, with reported increases in pitting potential of up to ~60% relative to untreated conditions [
64]. Nevertheless, chemical passivation remains an essential post-processing step to fully restore and stabilize the protective oxide film after mechanical, thermal, or laser-based treatments. Acidic passivation treatments based on nitric or citric acid dissolve residual intermetallics and selectively enrich the surface in chromium, thereby reinforcing the inner Cr
2O
3-dominated passive layer. In addition, plasma-assisted nitriding and oxidation treatments have been shown to generate nitrogen-enriched passive films that compensate for chromium-depleted zones associated with prior σ or nitride precipitation. When properly optimized, these hybrid surface-engineering approaches synergistically combine microstructural homogenization and surface chemical modification, resulting in improved long-term corrosion resistance for demanding service environments.
Figure 8 illustrates how CPT changes when secondary phases emerge and how solution treatment helps stainless steels perform better against corrosion. These phases act as pitting initiation sites because of the depletion of Cr and Mo in the surrounding areas; CPT decreased as the volume fraction of such phases rose. Although the CPT was enhanced by solution annealing because secondary phases were dissolved in ferrite, the volume percentage of ferrite remained low because of compositional differences; therefore, the CPT was still below the acceptable level.
Heat-treatment studies on duplex and super duplex stainless steels show that solution annealing at elevated temperatures (≈1100–1200 °C), followed by appropriate cooling rates, can restore a near-balanced austenite–ferrite microstructure even after significant secondary-phase precipitation. In particular, when secondary phases have formed during prior thermal exposure, reheating to this temperature range promotes their dissolution and enables re-equilibration toward approximately equal phase fractions, provided that cooling is sufficiently rapid to suppress reprecipitation [
65,
66]. These findings highlight that phase balance is governed not only by peak temperature but also by the interplay between secondary-phase dissolution kinetics and post-annealing cooling rate.
5.2. Emerging Approaches
Secondary-phase formation control has seen both new opportunities and challenges as a result of additive manufacturing (AM). Element segregation and residual δ-ferrite are characteristics of nonequilibrium microstructures produced by the severe temperature gradients (103–106 K s−1) and cyclic reheating inherent in selective laser melting (SLM) or directed energy deposition (DED). While equilibrium σ production is suppressed by quick solidification, transient σ and Cr2N precipitation along sub-grain boundaries can be induced by subsequent thermal exposure, either during build or post-processing. In AM alloys, post-build heat treatment and process optimization are the main mitigation techniques. The solidification path and cooling pace are controlled by varying the hatch spacing, scan speed, and linear energy density. Predictive suppression of σ is now possible by machine-learning-assisted parameter optimization, which finds process windows that minimize the segregation of Mo and Cr. Hot-isostatic pressing (HIP) simultaneously eliminates porosity and encourages homogeneity, while post-build solution annealing at 1050–1150 °C and quick quenching dissolves transient precipitates. Under such circumstances, AM duplex steels with microstructural modifications show corrosion behavior similar to that of their wrought equivalents.
Predictive phase control has been transformed by combining kinetic and data-driven models with CALPHAD thermodynamics. Equilibrium and non-equilibrium phase fractions under various thermal histories can be computed simultaneously using modern databases. Time-temperature-precipitation (TTP) diagrams for σ, χ, and carbides can be predicted using DICTRA simulations in conjunction with TC-PRISMA precipitation models, offering quantitative direction for heat-treatment scheduling. These capabilities are further enhanced by machine learning (ML), which makes it possible to quickly explore the links between composition, processing, and microstructure. There is less need for in-depth experimental aging investigations because neural networks trained on computational and experimental datasets can forecast σ-phase start temperatures or incubation periods in a matter of seconds. Target qualities, including PREN, mechanical strength, and σ-fraction limit, are balanced via multi-objective optimization frameworks. For example, composition windows in Fe–Cr–Mo–Ni–N systems that retain PREN > 40 while guaranteeing σ start >1000 °C, beyond normal service exposures, have been discovered using ML-assisted CALPHAD design. These digital-metallurgy methods are currently developing into corrosion prediction digital twins. Researchers can model how small compositional changes translate into potential maps and pit-initiation probabilities by connecting thermodynamic outputs to electrochemical models. When these simulations are combined with in situ synchrotron data, closed-loop design systems with adaptive learning capabilities are produced. The long-term objective is an integrated computational materials engineering (ICME) framework that predicts corrosion kinetics, passive-film evolution, and σ-phase precipitation simultaneously.
By reducing reliance on critical raw materials like Ni and Mo, the latest mitigation techniques not only address σ-phase risk but also emphasize sustainability. High-Mn, high-Al austenitic steels and low-Ni duplex variants maintain corrosion resistance while keeping strength and ductility due to the formation of κ-carbide (Fe
3AlC) rather than σ precipitation [
67]. These alloys are representative of “phase-aware sustainability, and “green metallurgy”, by giving priority to closed-loop recyclability and energy-efficient procedures. Flash annealing, laser-assisted quenching, and controlled induction heating minimize thermal exposure and carbon footprint in comparison to conventional furnaces [
68]. These eco-friendly techniques can be combined with AI-assisted alloy design to produce stainless steels that meet durability and sustainability requirements. Last but not least, a new paradigm is developing in the form of hybrid mitigation concepts that integrate advanced thermodynamic modeling, corrosion testing, and characterization. Real-time feedback from APT or synchrotron experiments informs CALPHAD-ML simulations, then suggests the best treatments, which are confirmed by electrochemical mapping. This iterative technique accelerates discovery cycles and directly translates phase control into quantitative corrosion reliability [
7]. These concerted efforts will not only make stainless steels more corrosion-resistant in the future, but they will also align with the principles of responsible consumption and climate action.
6. Outlook and Future Perspectives
Over the past decade, the study of corrosion–microstructure relationships in stainless steels has evolved from predominantly empirical investigations toward integrated, data-driven frameworks. The formation and stability of secondary phases are now increasingly interpreted through multiscale approaches that combine thermodynamic modeling, diffusion kinetics, and localized electrochemical behavior. Recent advances in CALPHAD-based high-throughput calculations coupled with machine-learning and Bayesian optimization demonstrate the potential to translate phase stability metrics directly into predictive descriptors of corrosion susceptibility. The remaining challenge lies in consolidating these computational and experimental advances into unified predictive tools capable of mapping corrosion behavior directly from phase-field simulations, thermodynamic databases, and in situ observations [
6]
Table 6 presents a summary of future directions to develop stainless steel alloys with controlled precipitation of secondary phases and high corrosion resistance. Integrating multiscale modeling with high-resolution analytics offers the most promising route to such predictive capability. Integrating multiscale modeling with high-resolution experimental analytics represents one of the most promising routes toward truly predictive corrosion frameworks. At the macroscopic level, CALPHAD-based thermodynamic databases reliably delineate equilibrium and metastable phase fields, while kinetic tools such as DICTRA and TC-PRISMA extend these descriptions to time-dependent precipitation and dissolution processes. However, these approaches rely largely on mean-field assumptions and therefore cannot fully capture the effects of micro-segregation, defect networks, or local stress states that govern phase stability and corrosion initiation at smaller length scales. Bridging this gap increasingly requires coupling mesoscale phase-field simulations with real-space characterization techniques such as atom probe tomography (APT), electron backscatter diffraction (EBSD), and three-dimensional tomography. Recent studies demonstrate that combining experimentally measured Cr and Mo partitioning profiles from APT with diffusion coefficients derived from first-principles density-functional theory enables direct validation and calibration of kinetic parameters used in DICTRA, substantially reducing uncertainty in precipitation predictions [
69]. A parallel advance is occurring in corrosion modeling, where the long-standing assumption of uniform electrochemical behavior within a given phase is being replaced by spatially resolved descriptions. Finite-element corrosion models now increasingly incorporate local electrochemical potentials measured by scanning electrochemical cell techniques or scanning Kelvin probe force microscopy and map them directly onto EBSD-resolved microstructures. In this framework, pit initiation and propagation can be predicted as functions of σ-phase connectivity and interfacial area density rather than bulk phase fraction alone, and future extensions to three-dimensional microstructures using 3D-EBSD and synchrotron-based nano-tomography are expected to enable fully phase-aware corrosion forecasting under coupled thermal, chemical, and mechanical loading conditions [
70].
The integration of computational thermodynamics with machine-learning (ML) approaches is increasingly reshaping alloy optimization within a data-driven materials design framework. Recent studies demonstrate that ML models trained on large experimental and simulated datasets can efficiently identify compositional regimes that balance high pitting resistance equivalent number (PREN) with delayed σ-phase formation, thereby accelerating exploration of complex alloy spaces [
71]. When coupled with multi-objective optimization schemes, such approaches enable identification of Pareto-optimal solutions that balance corrosion resistance, mechanical performance, and processability. Building on these developments, the concept of digitally assisted microstructure evolution central to Integrated Computational Materials Engineering (ICME) is gaining traction, wherein thermodynamic and kinetic models are continuously refined using experimental feedback to improve predictability across processing and service conditions [
72]. In parallel, future alloy development is expected to rely increasingly on phase-stability engineering strategies that deliberately suppress deleterious σ and χ phases while preserving desirable phase balance. In duplex and super-duplex stainless steels, alloying elements such as nitrogen and manganese are particularly effective because they expand the austenite stability range, reduce ferrite supersaturation, and enhance repassivation kinetics, thereby improving resistance to σ-phase precipitation and localized corrosion under service conditions [
73].
Emerging Fe–Cr–Mn–N alloy systems demonstrate the ability to achieve pitting resistance equivalent numbers (PRENs) exceeding 40 while substantially reducing nickel and molybdenum contents, thereby offering corrosion performance comparable to conventional 2205 duplex stainless steel with improved cost and sustainability profiles [
7]. In parallel, lightweight Fe–Mn–Al–C steels have attracted growing interest, as their strengthening is governed by κ-carbide (Fe
3AlC) precipitation, which provides high strength through ordered nanoscale particles rather than chromium-depleting intermetallic phases. This precipitation-based strengthening mechanism enables favorable strength-to-weight ratios for energy and transportation applications while avoiding the corrosion penalties typically associated with σ-phase formation in Cr-rich stainless steels [
7].
Advances in rapid solidification and gradient heat treatment allow for localized phase control at the processing level. Techniques like laser surface melting, gradient annealing, and induction-based micro heat treatment provide thermal or compositional profiles that dissolve σ at the surface without compromising core strength. By combining these processes with in situ thermal simulation, microstructural repeatability is guaranteed. In the meantime, phase dispersion can be intentionally engineered rather than avoided thanks to additive manufacturing, which provides unprecedented control over local solidification paths. The goal has changed from merely eliminating secondary phases to altering their chemistry and structure for beneficial results, like regulated nanoscale nitrides that stop hydrogen diffusion or sub-micron carbides that boost mechanical strength without compromising corrosion.
Despite substantial progress in understanding σ-phase precipitation, significant data gaps continue to limit the predictive management of secondary-phase evolution in duplex stainless steels. While industrial components are exposed to complex, multiaxial stress states and non-isothermal thermal histories over service lifetimes spanning decades, most experimental studies remain confined to short-term laboratory exposures (typically <10
3 h) under simplified loading conditions [
74]. Quantitative models describing σ-phase nucleation and growth under cyclic thermal loading or stress-assisted conditions are still scarce, and the coupled effects of oxidation, σ coarsening, and chloride ingress during long-term service remain particularly challenging to characterize experimentally [
58,
74]. Addressing these limitations requires coordinated durability initiatives that integrate accelerated aging protocols with in situ characterization and systematic data assimilation. The development of shared, open databases containing compositional metadata, thermal and mechanical histories, and spatially resolved corrosion metrics would enable cross-validation of σ-phase prediction models across institutions and alloy systems. A parallel challenge lies in scaling atomic-level insights to engineering-scale design tools: although atom-probe tomography and first-principles calculations provide a detailed understanding of nanoscale segregation and diffusion, translating these results into effective transport parameters for continuum corrosion models remains non-trivial [
61]. Multiscale homogenization strategies that link atomic diffusivities to effective kinetic coefficients in finite-element frameworks are therefore essential for realistic lifetime prediction. Finally, phase-stability models must be extended to account for complex service environments such as supercritical water, CO
2-rich atmospheres, or hydrogen-containing media. Incorporating volatile-species thermodynamics and interfacial energy contributions into CALPHAD-based descriptions will broaden their applicability to high-pressure and mixed-environment corrosion scenarios Equally important is the evolution of the human–data interface: intuitive visualization tools that translate high-dimensional, three-dimensional microstructural data into actionable corrosion-risk maps are required for industrial deployment. When integrated with digital-twin frameworks, such tools would allow engineers to directly assess how modest compositional or microstructural changes, such as a 0.2 wt.% reduction in nitrogen or a 1 vol.% increase in σ phase, affect projected service life, thereby closing the loop between alloy design, processing, and in-service performance.
It is evident that research on stainless steel is trending toward ecologically friendly, data-driven design. Combining high-resolution experimental methods with multi-scale modeling and machine learning ushers in a new era where phase evolution and corrosion may be anticipated rather than only seen. Future research will focus on developing universal phase-corrosion models that have been verified for a variety of alloy systems, including lean and lightweight Fe–Mn–Al–C variants as well as traditional duplex grades. By integrating these models with digital-twin manufacturing and life-cycle monitoring, stainless steels will evolve from empirically optimal materials to self-informed systems that can anticipate and mitigate their own degradation. Such developments will not only increase corrosion reliability but also align stainless steel metallurgy with global resource efficiency and climate neutrality targets.
7. Conclusions
By combining data-driven alloy design, advanced characterization, and thermodynamic modeling, we have made great strides in our comprehension of how secondary phases impact stainless steel corrosion behavior. Local electrochemistry, phase thermodynamics, and diffusion kinetics all affect the controllable microstructural instability that causes corrosion deterioration rather than being an inherent restriction of these alloys.
The σ, χ, and Laves intermetallics, together with chromium-rich carbides and nitrides, continue to be the primary phases that determine corrosion reliability. By eliminating nitrogen, molybdenum, and chromium from the matrix, their precipitation weakens passive films and promotes galvanic coupling.
While tailored compositions and heat treatments can preserve long-term phase stability and corrosion resistance, even small σ or χ fractions can significantly reduce pitting potential and improve intergranular corrosion.
New developments in in situ synchrotron diffraction, high-resolution TEM, and atom-probe tomography have shown that phase nucleation is preceded by nanoscale segregation processes. These methods are now used to validate phase diagrams and transformation kinetics in conjunction with CALPHAD and DICTRA models. Electrochemical mapping, SKPFM, and correlative EBSD further connect microstructural characteristics to localized corrosion mechanisms.
From empirical treatments to predictive design, mitigation measures have changed over time. Rapid solution annealing, homogenization, balanced Cr–Mo–N compositions, and laser remelting eliminate harmful phases and restore passivity. Corrosion engineering is in line with sustainability objectives thanks to new additive manufacturing, thermomechanical, and CALPHAD machine-learning techniques that allow proactive control of phase stability while lowering dependency on Ni and Mo.
In the future, digital-twin simulations of microstructural evolution under service circumstances and predictive phase-corrosion mapping will be made possible by combining multi-scale modeling with high-resolution analytics. This change will be accelerated by the creation of open datasets on environmental impacts and long-term aging.