Experimental Study on Inhibition Characteristics of Imidazolium-Ionic-Liquid-Loaded Sepiolite Composite Inhibitor
Abstract
1. Introduction
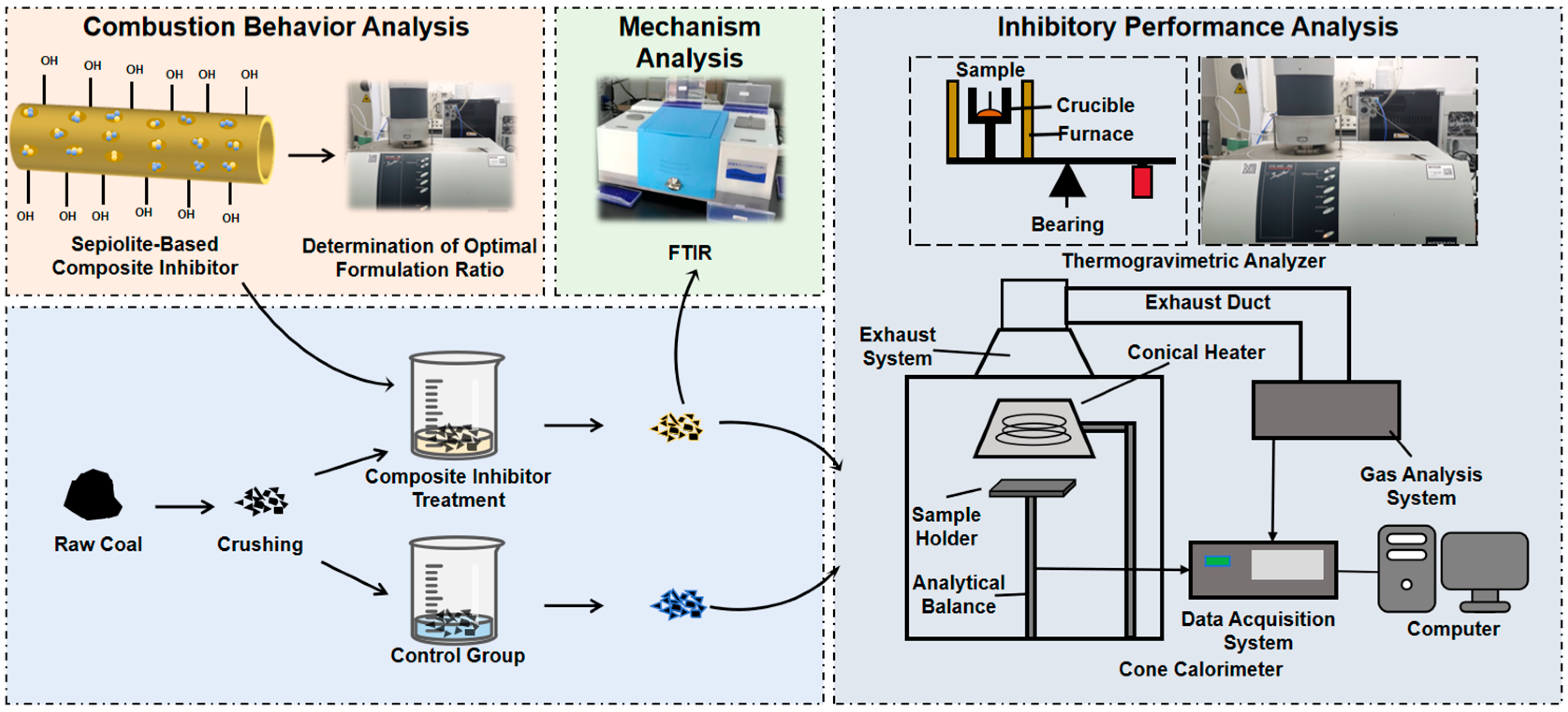
2. Experimental
2.1. Material Preparation
2.1.1. Preparation of Sepiolite-Based Composite Inhibitor
2.1.2. Preparation of Inhibited Coal Samples
2.2. Experimental Procedure
2.2.1. TG-DTG Experiment
2.2.2. FTIR Experiment
2.2.3. Cone Calorimeter Experiment
3. Analysis and Discussion
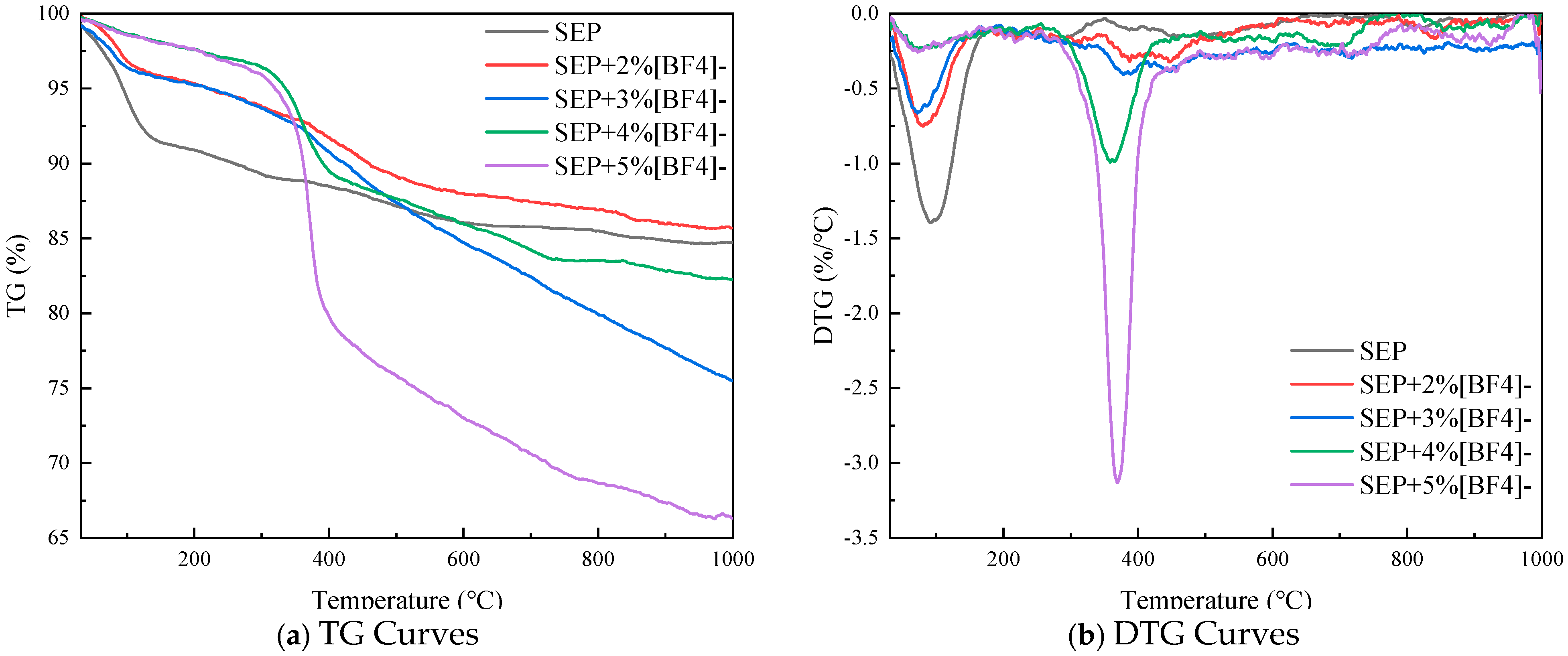
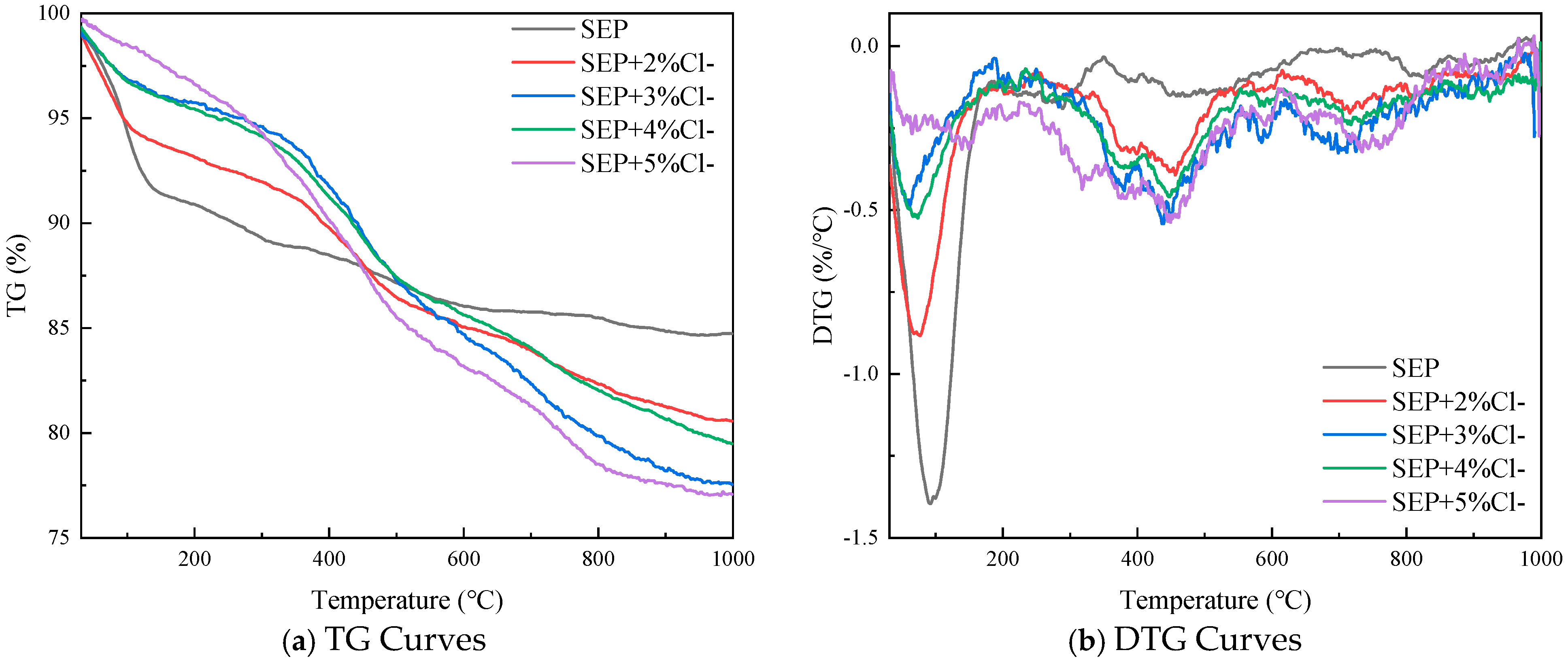
3.1. Combustion Characteristics Analysis of Sepiolite-Based Composite Inhibitor
3.2. Analysis of the Effect of Sepiolite-Based Composite Inhibitor on Coal Oxidation Characteristics
3.3. Cone Calorimeter Experiments on the Influence of Composite Inhibitors on Coal Combustion Characteristics
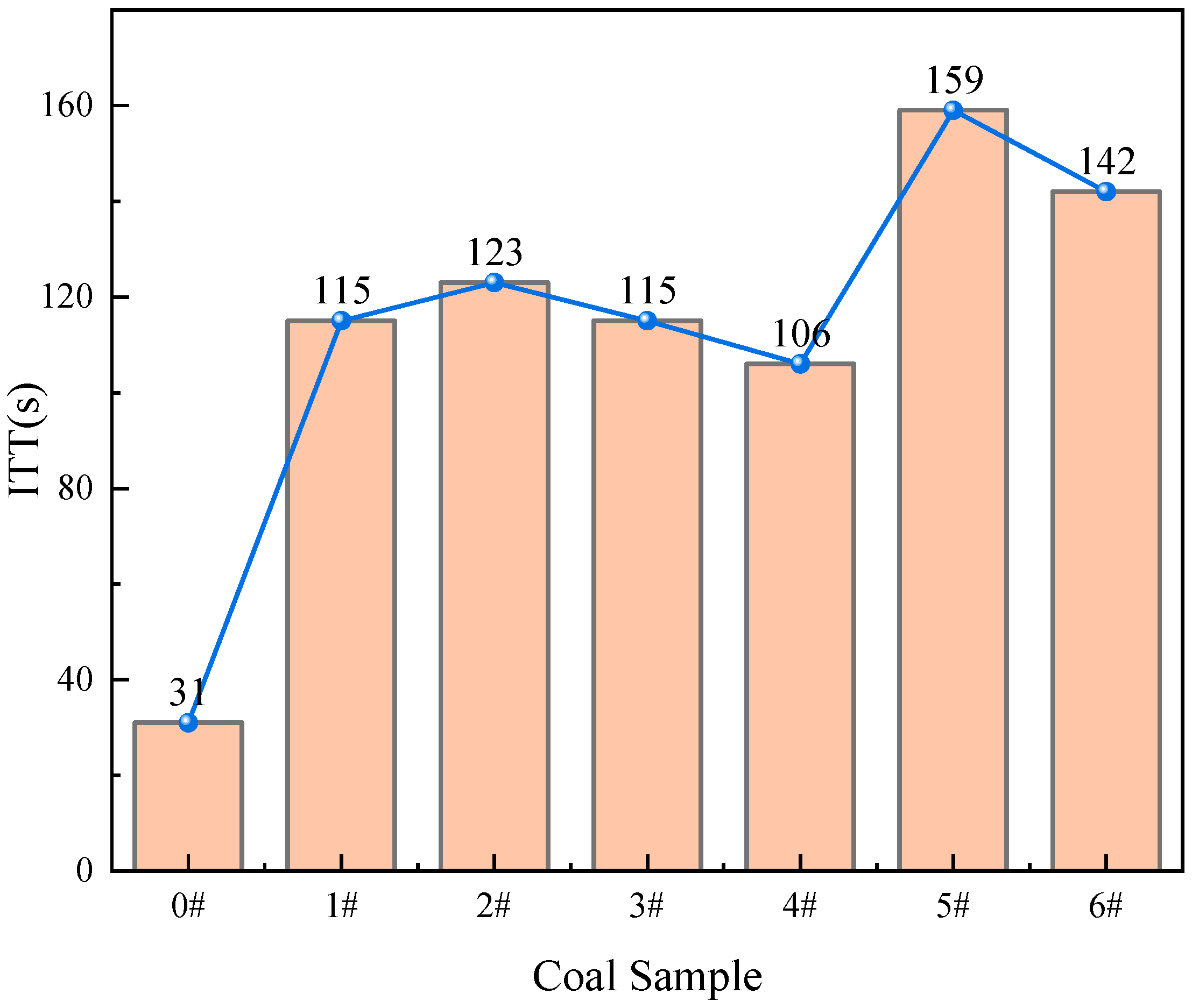
3.3.1. Ignition Time Analysis
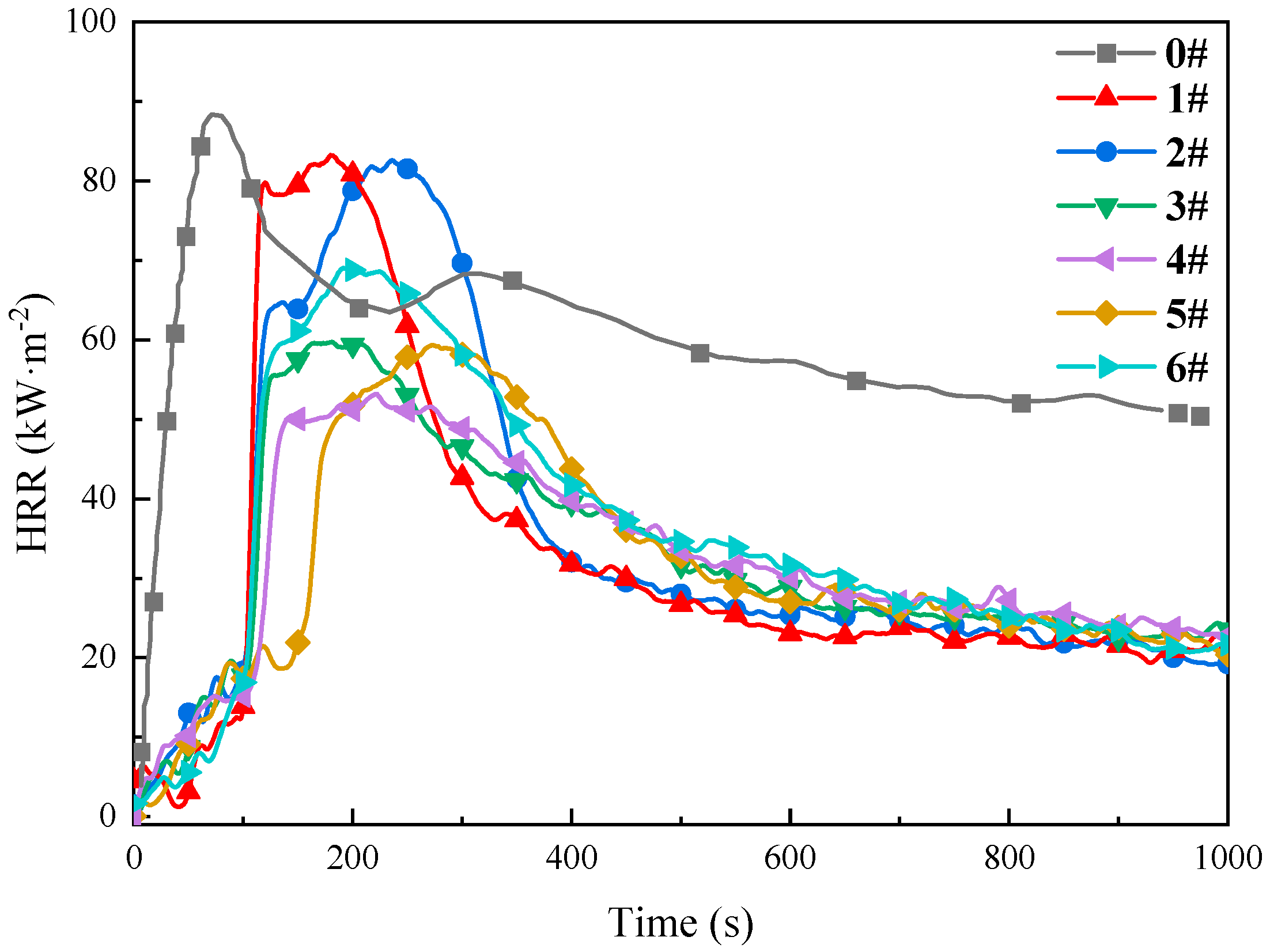
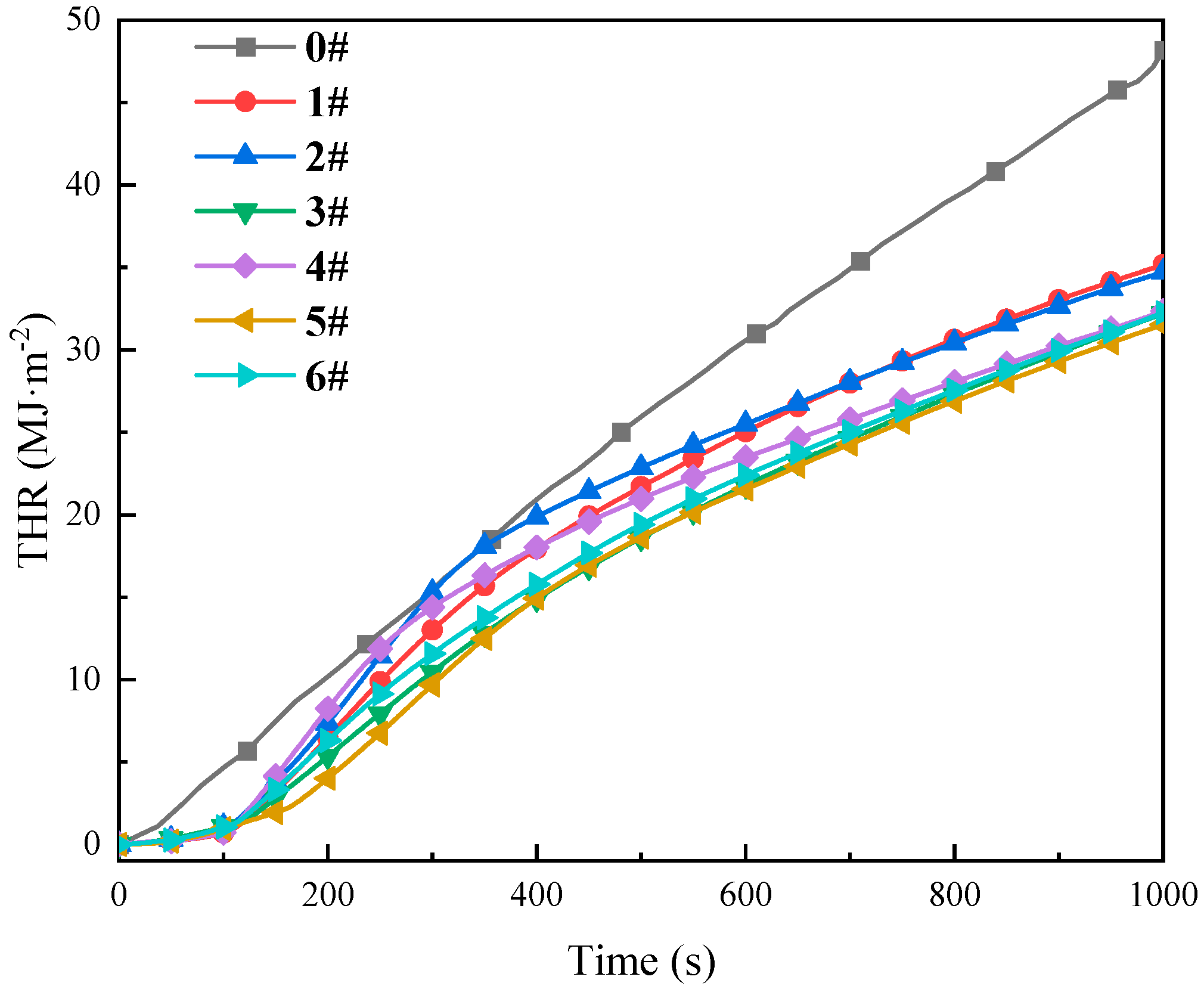
3.3.2. Analysis of Heat Release Characteristics
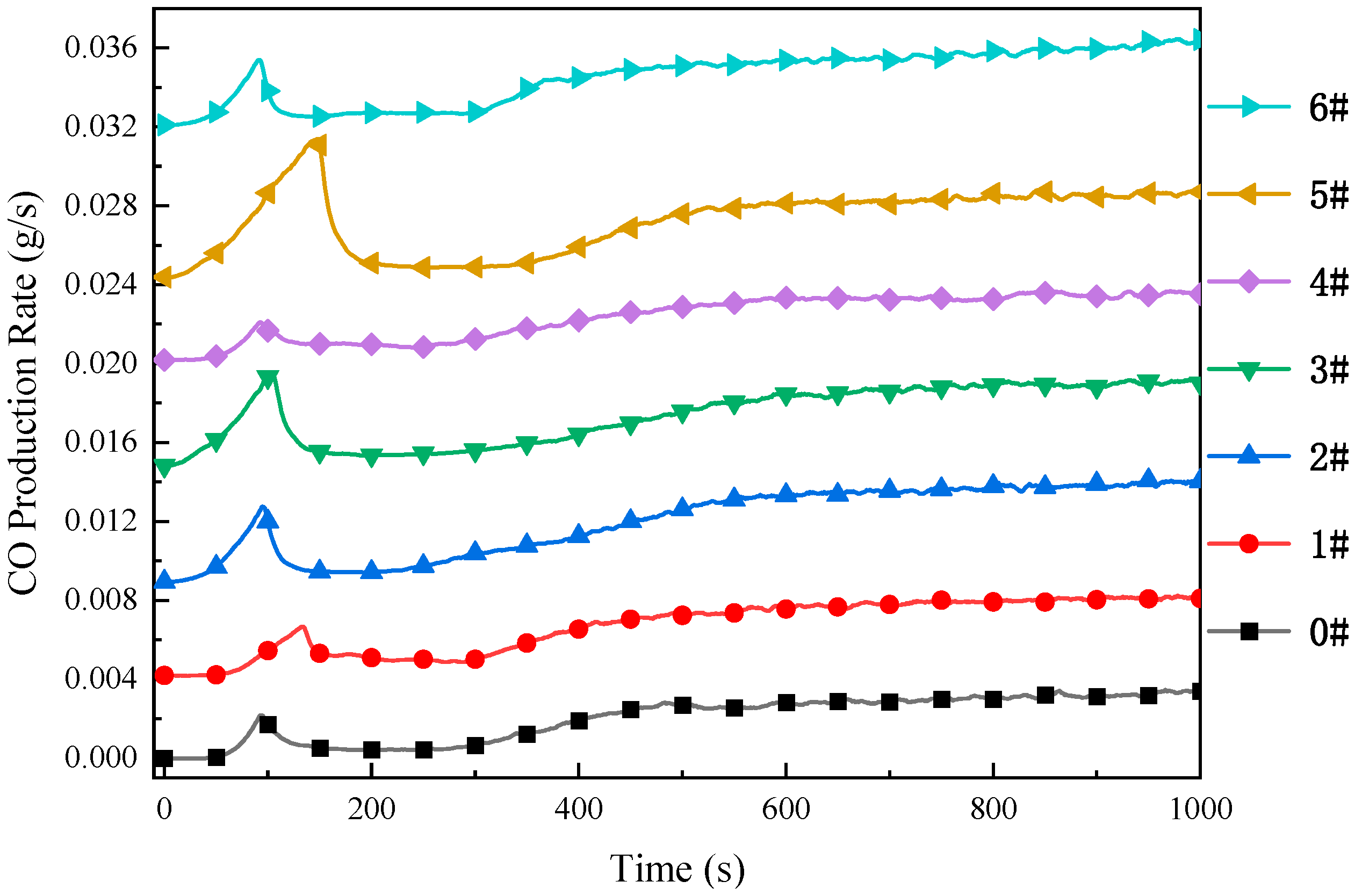
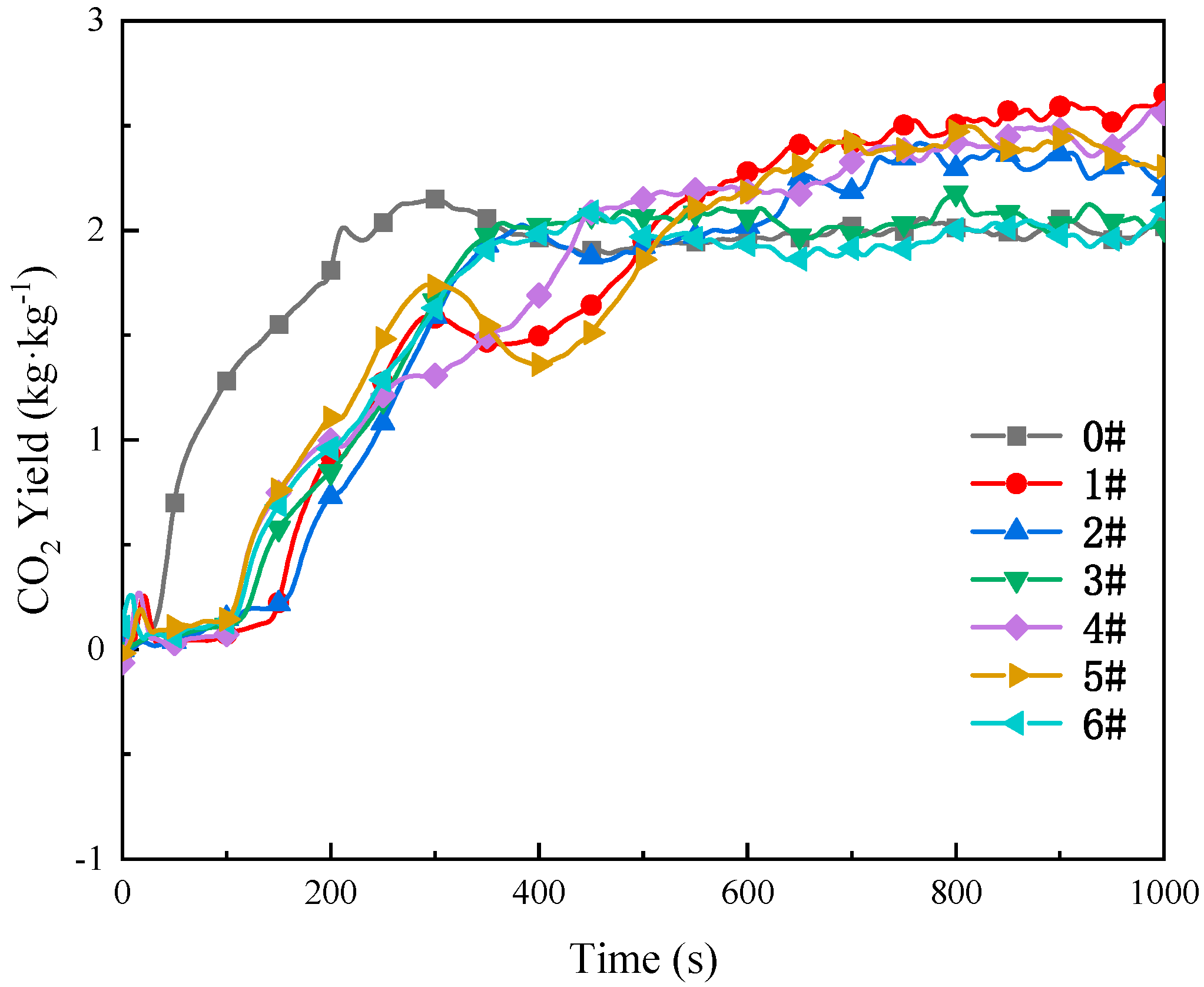
3.3.3. Analysis of CO and CO2 Gas Emissions
3.3.4. Fire Risk Analysis of Coal Combustion Characteristics
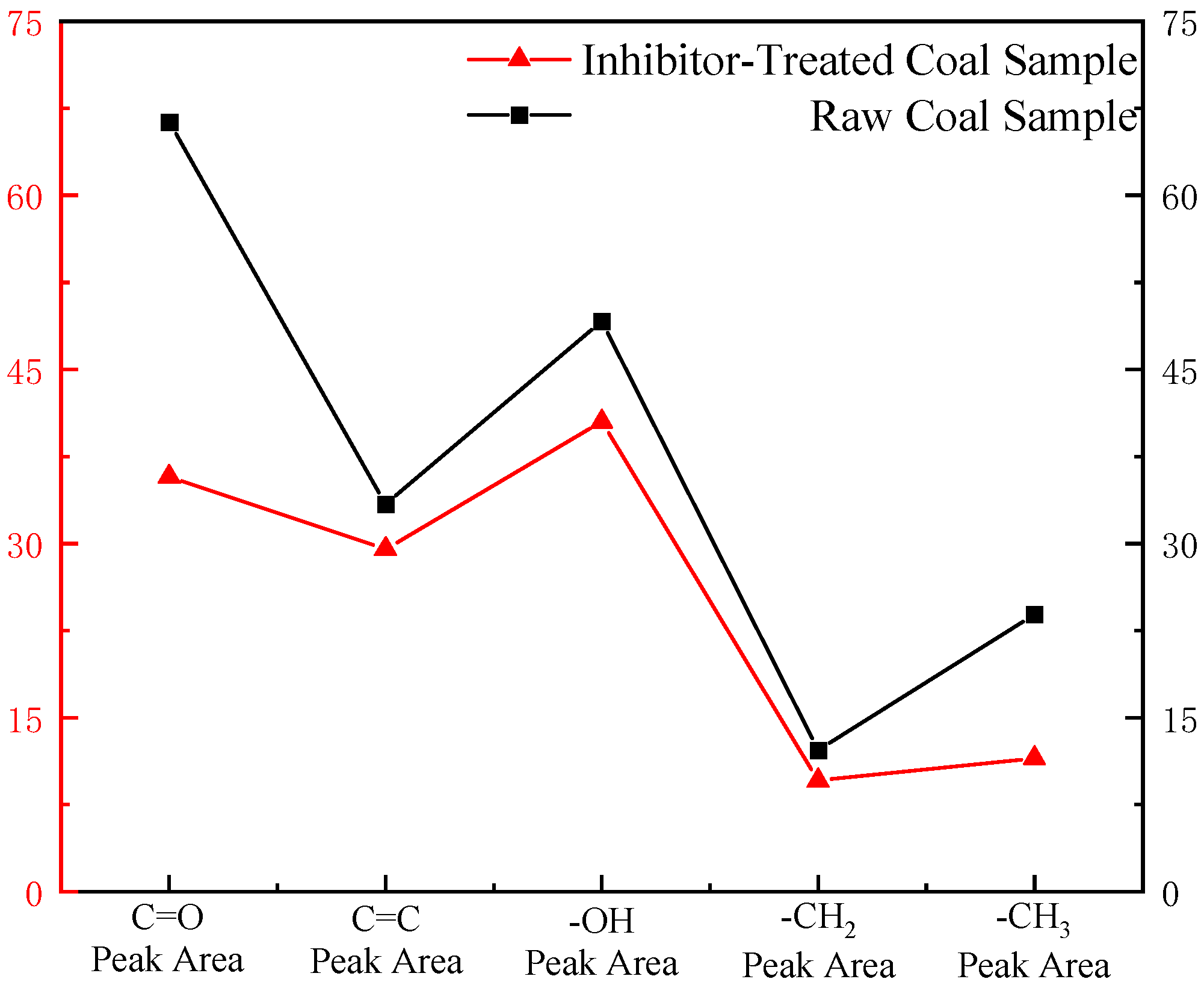
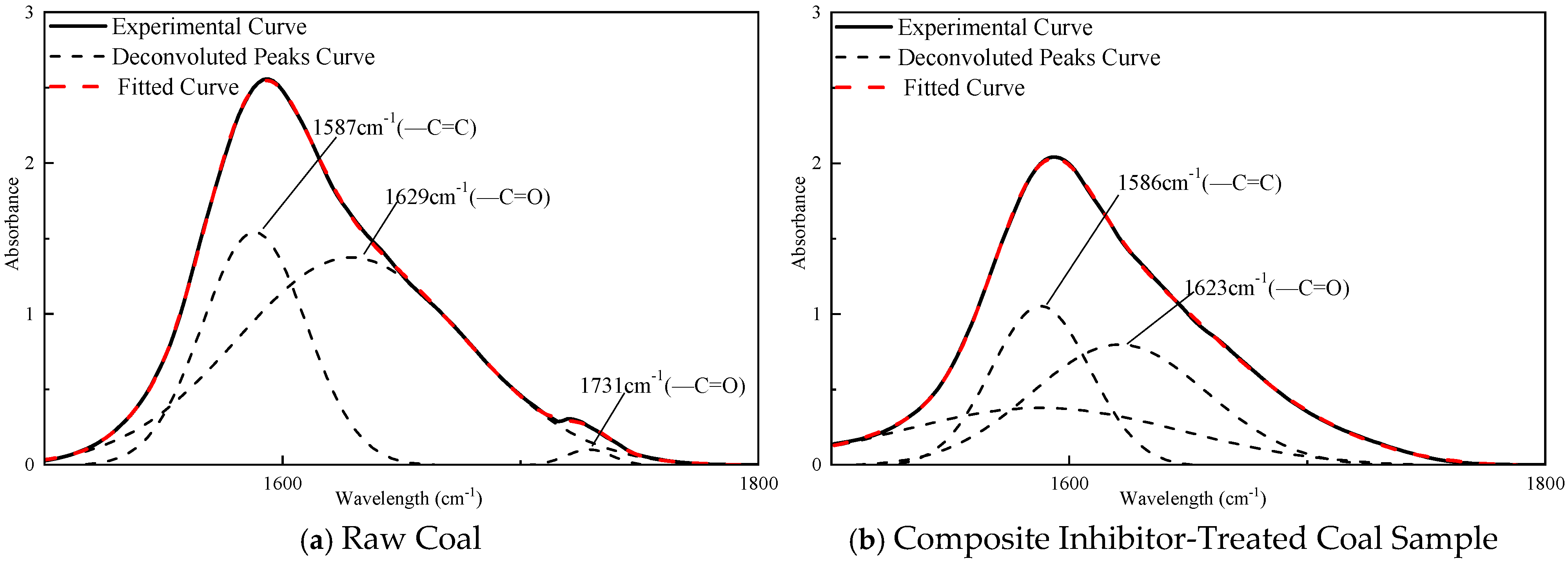
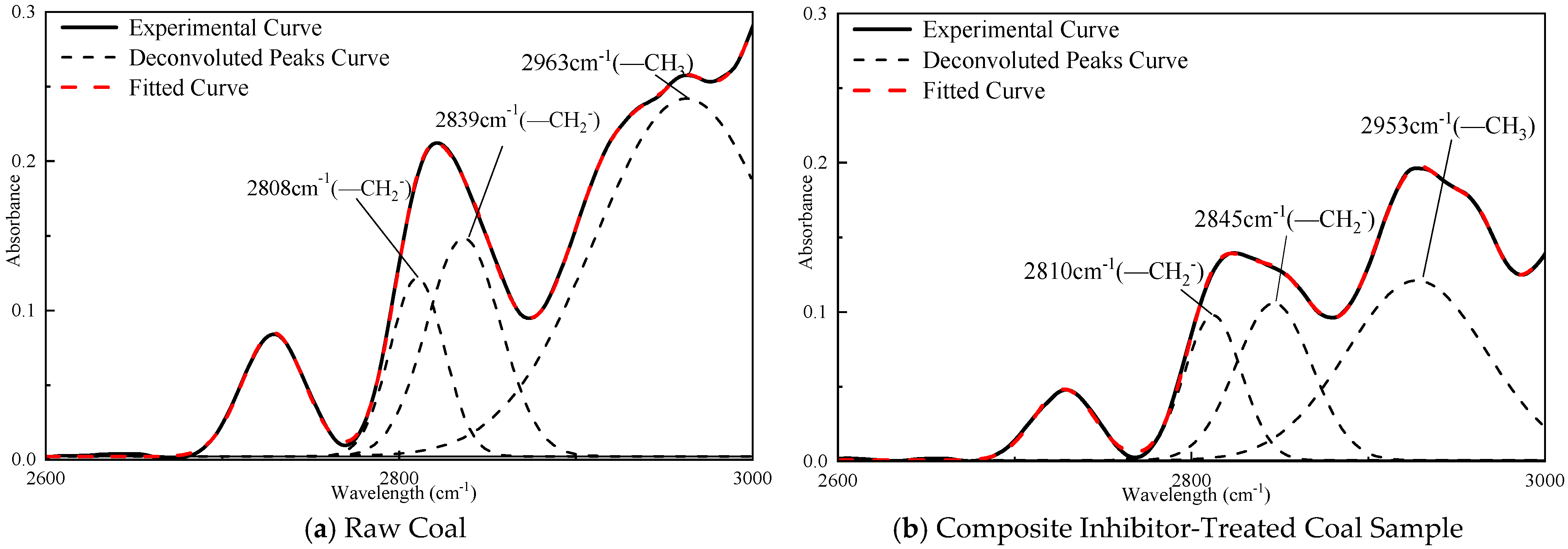
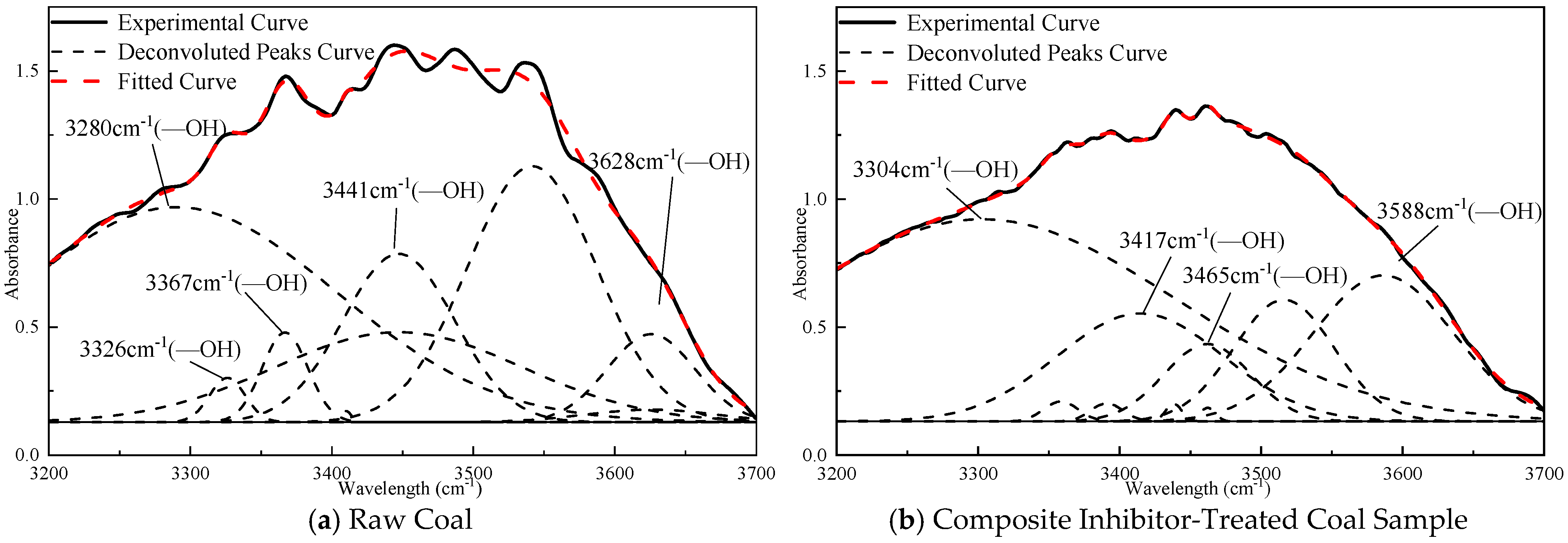
3.4. Quantitative Analysis of FTIR Spectra for Coal Samples
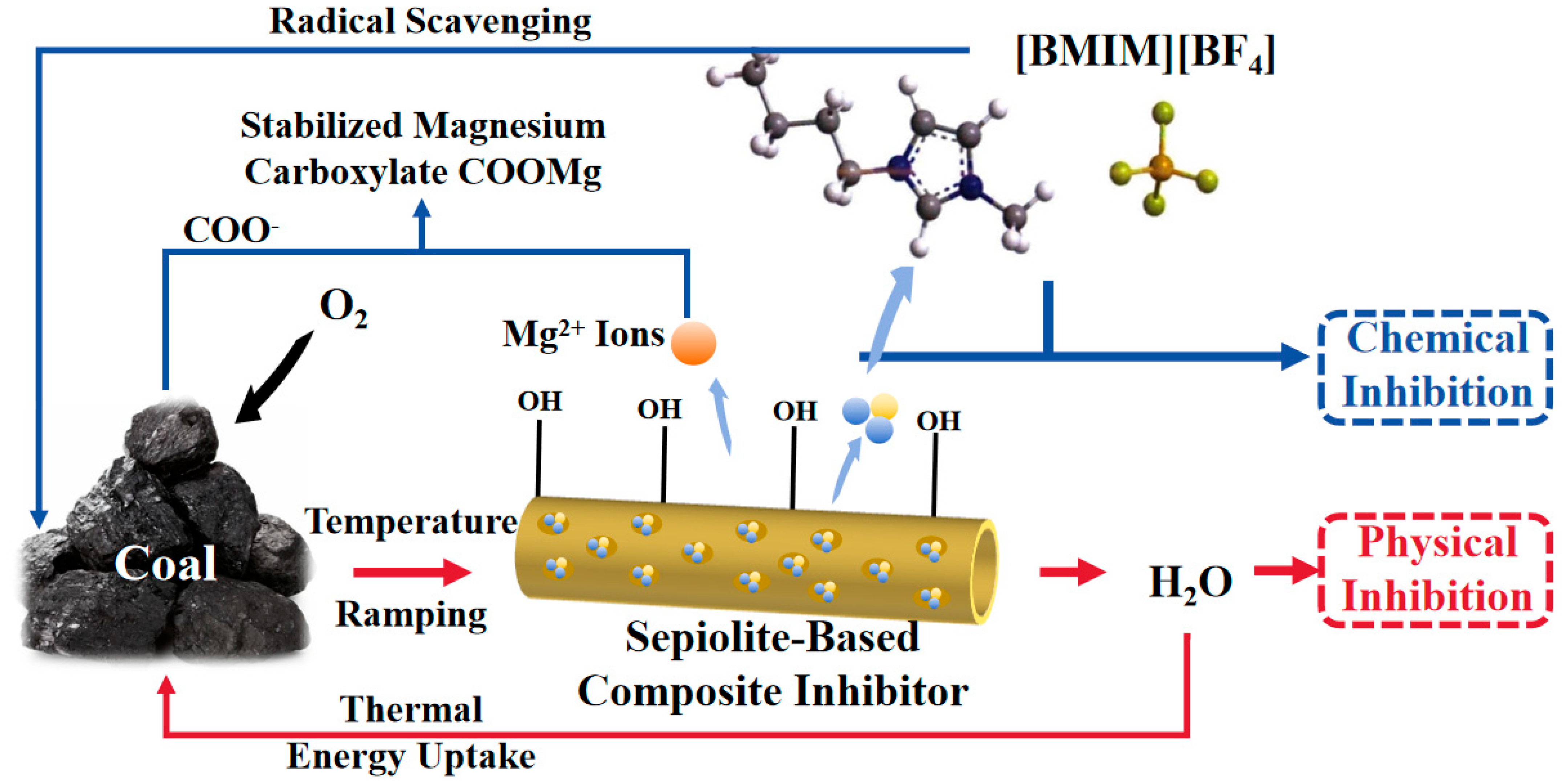
3.5. Mechanism of Sepiolite-Based Composite Inhibitor in Suppressing Coal Spontaneous Combustion
- 1.
- Physical Inhibition Mechanism: During the initial stage of coal spontaneous combustion, SEP adsorbs and retains substantial moisture on its surface due to its high specific surface area and structural characteristics. This moisture absorbs heat during temperature elevation, effectively reducing thermal accumulation on the coal surface. Additionally, SEP contains structural water within its framework, which desorbs at elevated temperatures, further lowering the coal matrix temperature. Physical inhibition suppresses the coal self-ignition reaction rate by altering the reaction conditions.
- 2.
- Chemical Inhibition Mechanism: In the early stage of coal spontaneous combustion, [BMIM][BF4] infiltrates coal pores through SEP-mediated loading, effectively neutralizing surface-active groups to mitigate self-ignition. Concurrently, Mg2+ ions in SEP chemically interact with carboxylic acid groups in coal via complexation reactions, forming stable COO-Mg structures that reduce the reactivity of C=O bonds in carboxyl groups. Chemical inhibition suppresses the coal self-ignition reaction rate by decreasing the population of surface-active groups and interrupting subsequent chain reactions.
4. Conclusions
- 1.
- SEP achieves thermal performance optimization through the precise loading of functional components at specific ratios. Compared to SEP, the modified materials effectively suppress secondary thermal decomposition while maintaining thermal stability: quantitative DTG curve analysis reveals 53% and 61% reductions in peak intensities within the 100–200 °C low-temperature weight loss region for SEP + 3% Cl− and SEP + 5% Cl−, respectively, confirming the significant mitigation of organic component volatilization. The SEP + 2% [BF4]− system exhibits optimal comprehensive performance, demonstrating an 82.1% residue rate at 800 °C and the narrowest DTG peak half-width, indicative of the highly uniform dispersion of loaded components within the matrix, thereby avoiding stepwise thermal decomposition caused by localized agglomeration. Based on orthogonal experimental analysis, two optimized formulations of sepiolite-based composites SEP + 3% [BF4]− and SEP + 5% Cl− were selected as the primary systems for subsequent investigations.
- 2.
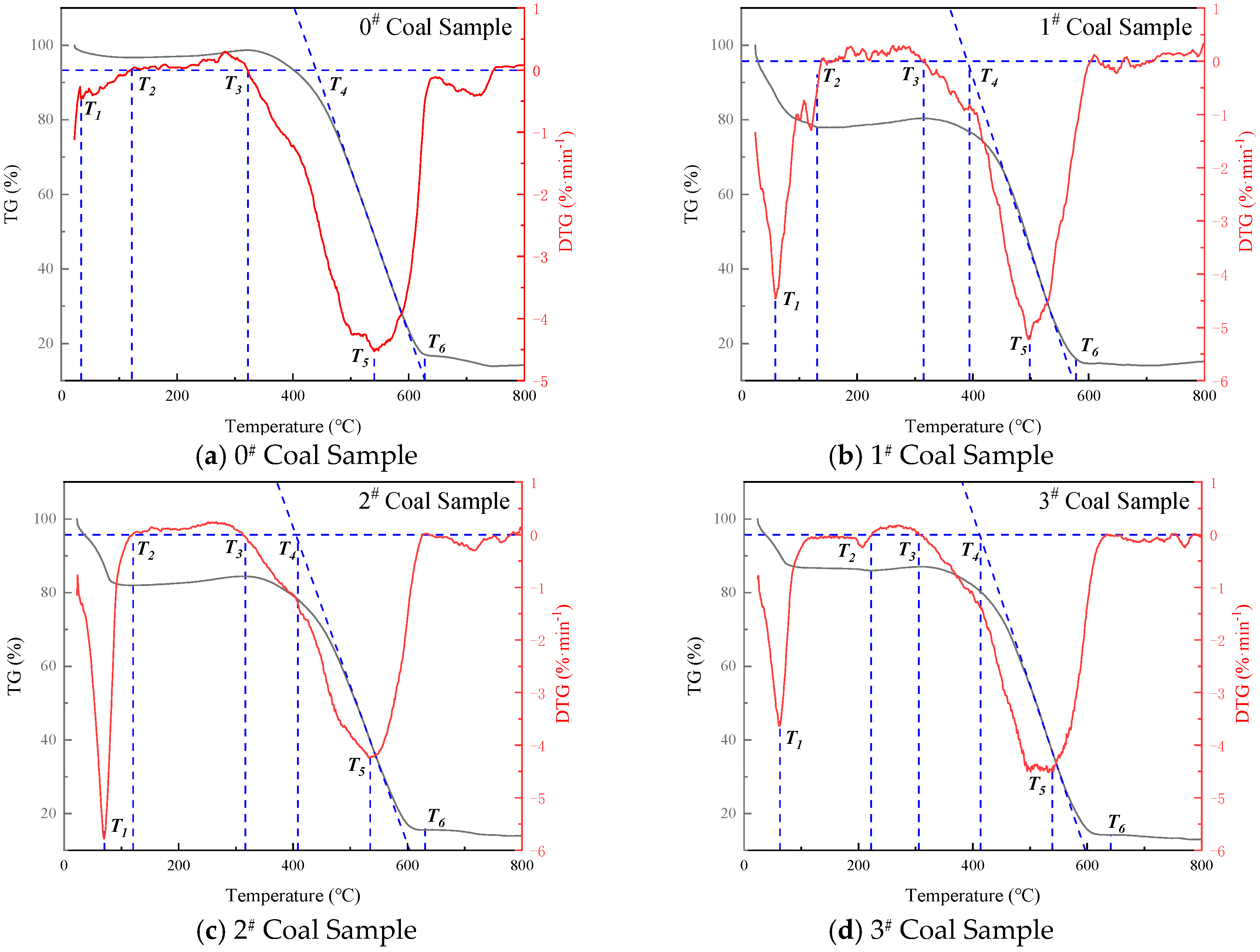
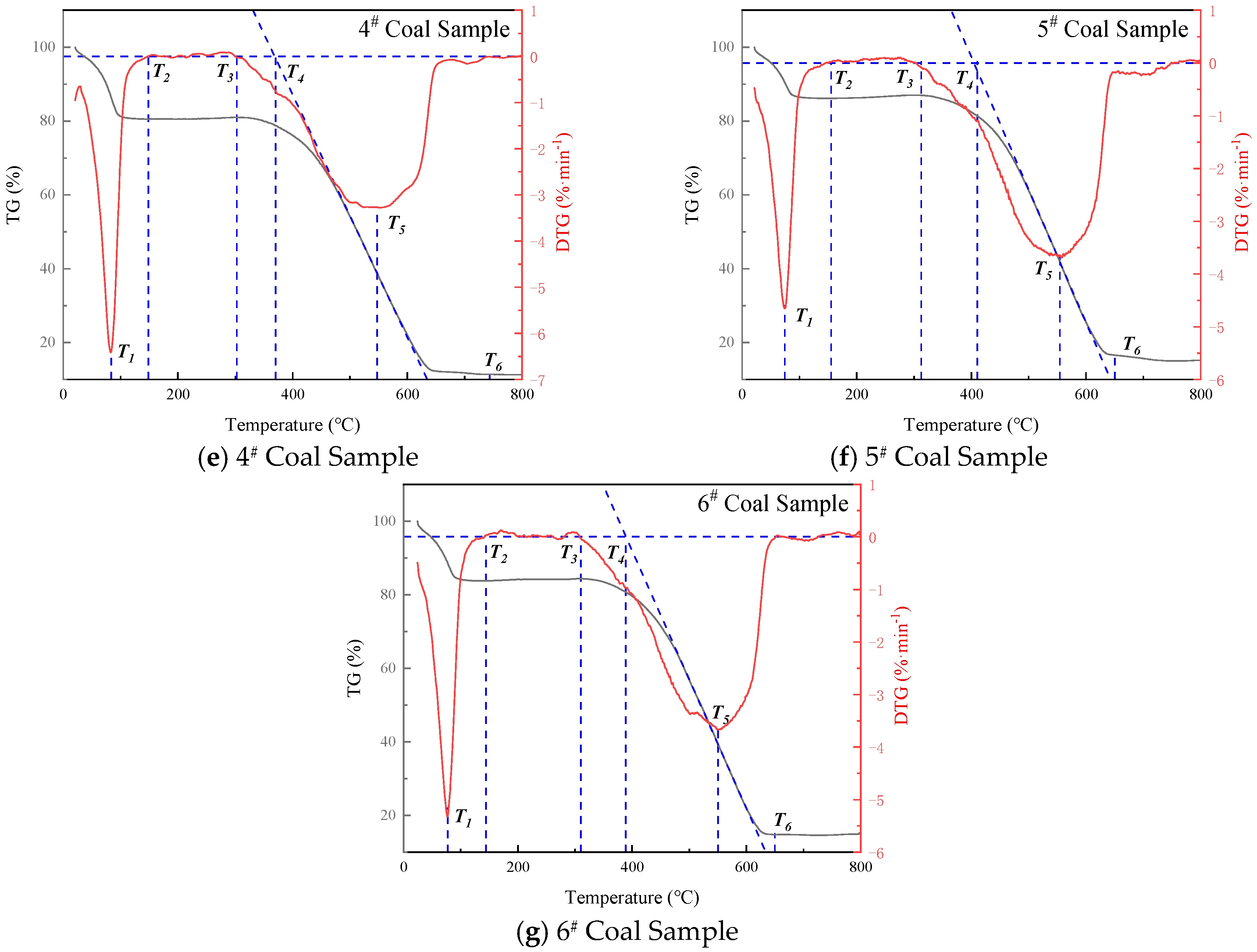
- TG-DTG analysis confirms the significant inhibitory effect of the composite inhibitor on coal spontaneous combustion. Inhibited Coal Sample 5# (SEP loaded with 3% [BMIM][BF4]) exhibits optimal inhibition performance, achieving an 84.8% mass retention rate and the maximum delay in characteristic temperatures. Cone calorimeter tests further reveal the synergistic flame-retardant mechanism. Compared to MgCl2-inhibited coal, the composite system delays ignition time by 8–44 s and significantly postpones the peak heat release rate. Critical parameters indicate that Sample 5# shows a 3.02-fold increase in CO emission peak intensity within the 0–200 s interval compared to raw coal, while reducing the CO2 production rate by 13.2% (to 2.64 kg·kg−1) at 1000 s relative to MgCl2-treated samples. The PPFI increases maximally to 2.33 m2·s·kW−1, and the FGFI reaches the lowest value (0.21 kW·m−2·s−1), conclusively validating the superior synergistic flame-retardant performance of the SEP-loaded 3% [BMIM][BF4] composite inhibitor.
- 3.
- The quantitative FTIR analysis of coal samples before and after treatment with the sepiolite-based composite inhibitor reveals post-inhibition reductions of 22–51% in peak areas for -OH, -CH3, and -CH2- groups, demonstrating the composite inhibitor’s pronounced suppression of coal spontaneous combustion at the molecular level. The sepiolite-based composite inhibitor operates through dual inhibition mechanisms: Physical inhibition: SEP’s high surface area and structural water adsorption mitigate heat accumulation. Chemical inhibition: [BMIM][BF4] disrupts radical chain reactions, while Mg2+ ions form stable complexes with oxygen-containing functional groups, reducing their oxidative activity. This integrated physical–chemical mechanism effectively interrupts the self-accelerating exothermic processes underlying coal spontaneous combustion, providing a robust theoretical foundation for developing advanced fire prevention technologies in mining engineering.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| [BMIM][BF4] | 1-butyl-3-methylimidazolium tetrafluoroborate |
| [BMIM][Cl] | 1-butyl-3-methylimidazolium tetrafluorochlorate |
| FGI | Fire growth index |
| PFI | Fire performance index |
| FTIR | Fourier-transform infrared spectroscopy |
| HRR | Heat release rate |
| ITT | Ignition time threshold |
| ILs | Ionic liquids |
| PHRR | Peak heat release rate |
| SEP | Sepiolite |
| TG-DTG | Thermogravimetric–differential thermogravimetric analysis |
| THR | Total heat release |
References
- Wang, J.; Fan, F.; Xue, Y.; Xiao, J.; Ren, Z.; Zhang, X. Study on co migration characteristics after blasting in tunnel under different ventilation methods. Energy Sources Part A Recovery Util. Environ. Eff. 2024, 46, 16217–16230. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, R.; Wang, Z.; Zhang, X. Research on the fire extinguishing performance of new gel foam for preventing and controlling the spontaneous combustion of coal gangue. Environ. Sci. Pollut. Res. Int. 2023, 30, 88548–88562. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, Z.; Chen, W. Optimisation of synergistic ventilation between dust and gas in a gas tunnel. Sci. Rep. 2024, 14, 27582. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Sun, J.; Liu, C.; Sun, Q. Synthesis comparative evaluation of hydrophobic polymer/surfactant fracturing fluids on methane adsorption–desorption and pore structure modifications in coal. Int. J. Coal Sci. Technol. 2025. [Google Scholar] [CrossRef]
- Fu, B.Z.; Tong, Q.X.; Bo, B.S. Coexistence of gas and coal spontaneous combustion (II): New prevention and control technologies. J. China Coal Soc. 2013, 38, 353–360. [Google Scholar]
- Huang, Z.A.; Tian, Y.; Shao, Z.L.; Gao, Y.K.; Zhang, Y.H.; Li, J.Y.; Zhu, H.Y. Novel super-absorbent polymer-grafted tea polyphenol composite inhibitor for the prevention of coal spontaneous combustion. Fire Mater. 2020, 44, 975–988. [Google Scholar] [CrossRef]
- Qin, B.; Dou, G.; Wang, Y.; Xin, H.; Ma, L.; Wang, D. A superabsorbent hydrogel–ascorbic acid composite inhibitor for the suppression of coal oxidation. Fuel 2017, 190, 129–135. [Google Scholar] [CrossRef]
- Sun, W.; Liu, L.; Zhou, J.; Zhao, Y.; Shao, C.; Lyu, Y. Effect of inorganic salt composition on strength, microstructure and leaching toxicity of coal-based solid waste backfill materials. Process Saf. Environ. Prot. 2024, 192, 1110–1126. [Google Scholar] [CrossRef]
- Wu, M.; Li, H.; Wang, J.; Wang, L.; Li, S.; Wang, Y.; Yu, M.; Li, J.; Shu, C. Inhibitory performance and mechanism analysis of modified fly-ash inhibitor on the coal spontaneous combustion: A combined study of laboratory experiments and molecular dynamic simulation. Process Saf. Environ. Prot. 2025, 193, 313–326. [Google Scholar] [CrossRef]
- Tang, Y. Experimental investigation of applying mgcl2 and phosphates to synergistically inhibit the spontaneous combustion of coal. J. Energy Inst. 2018, 91, 639–645. [Google Scholar] [CrossRef]
- Li, L.; Li, Y.; Li, Z.; Wu, L.; Gou, J.; He, X.; Xu, C.; Xie, C.; Wu, W. Preparation and flame-retardant mechanism of mgalzn-based hydrotalcite-like coal spontaneous combustion inhibitor. Materials 2025, 18, 70. [Google Scholar] [CrossRef]
- Ji, B.; Jiang, B.; Yuan, L.; Zhou, Y.; Wang, H.; Tao, W.; Zhang, Y. Effect of side chain functional groups of ionic liquids on improving wettability of coal: Simulation and experimental discussion. Energy 2023, 285, 129453. [Google Scholar] [CrossRef]
- Jiang, B.; Ji, B.; Yuan, L.; Yu, C.; Tao, W.; Zhou, Y.; Wang, H.; Wang, X.; Liao, M. Experimental and molecular dynamics simulation study of the ionic liquids’ chain-length on wetting of bituminous coal. Energy 2023, 283, 128507. [Google Scholar] [CrossRef]
- Bai, Z.; Deng, J.; Wang, C.; Zhang, Y.; Kang, F.; Shu, C. Mechanism of [bmim][bf4] inhibiting coal groups activity from experiments and dft calculations. Fuel 2023, 353, 129175. [Google Scholar] [CrossRef]
- Lin, Z.; Chen, X.; Xu, C.; Wang, H.; Zhu, X. Effect of ionic liquid-surfactant compound solutions on the wettability and functional groups of coal. J. Mol. Liq. 2024, 401, 124650. [Google Scholar] [CrossRef]
- Hapuarachchi, T.D.; Peijs, T. Multiwalled carbon nanotubes and sepiolite nanoclays as flame retardants for polylactide and its natural fibre reinforced composites. Compos. Part A Appl. Sci. Manuf. 2010, 41, 954–963. [Google Scholar] [CrossRef]
- Jiang, Y.; Lin, F.; Guo, Z.; Liu, X.; Huang, Z.; Fan, J.; Min, X.; Qiao, J. Performance study of an environmentally friendly, flame-retardant, and sustainable energy storage composite phase change material based on sepiolite-gelatin-graphene aerogel. J. Energy Storage 2025, 117, 116207. [Google Scholar] [CrossRef]
- Pappalardo, S.; Russo, P.; Acierno, D.; Rabe, S.; Schartel, B. The synergistic effect of organically modified sepiolite in intumescent flame retardant polypropylene. Eur. Polym. J. 2016, 76, 196–207. [Google Scholar] [CrossRef]
- Geng, X.; Liu, Y.; Zhang, T.; Zheng, S.; Hu, Y.; Kang, W. Review of Flame Retardant Properties of Thermoset Polymer/Sepiolite Composites. China Plast. Ind. 2023, 51, 30–36. [Google Scholar]
- Meng, J.; Ma, Y.; Xin, Z.; Xia, L. Research progress in structure of sepiolite and its application in flame retardant polymer materials. China Synth. Rubber Ind. 2022, 45, 253–258. [Google Scholar]
- Zhan, Z.; Xu, M.; Li, B. Synergistic effects of sepiolite on the flame retardant properties and thermal degradation behaviors of polyamide 66/aluminum diethylphosphinate composites. Polym. Degrad. Stab. 2015, 117, 66–74. [Google Scholar] [CrossRef]
- Song, J.; Ren, X.; Hu, G.; Hu, X.; Cheng, W. Enhanced pms activation by mof-derived co3o4/sepiolite composite for norfloxacin degradation: Performance, mechanism and degradation pathway. Process Saf. Environ. Prot. 2023, 176, 140–154. [Google Scholar] [CrossRef]
- Zhou, K.; Qin, Q.; Duan, G.; Fu, Q.; Yang, H.; Han, X.; Zhang, C.; He, S.; Jiang, S. Preparation, modification and applications of sepiolite nanofibers: A review. Inorganica Chim. Acta 2025, 579, 122567. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, J.; Xue, Y.; Wang, X.; Chen, Y. Preparation of high thermal-stable antibacterial material with sepiolite droped ionic liquid. China Powder Sci. Technol. 2022, 28, 1–9. [Google Scholar]
- Topçu, M.A.; Rüşen, A. Simple and selective copper recovery from valuable industrial waste by imidazolium based ionic liquids with bf4- anions. Process Saf. Environ. Prot. 2023, 169, 788–796. [Google Scholar] [CrossRef]
- Xiao, Y.; Li, D.; Lu, H.; Deng, J. Research on imidazolium ionic liquid inhibiting coal oxidation thermo-kinetics parameters. J. China Coal Soc. 2019, 44 (Suppl. S1), 187–194. [Google Scholar]
- Pandey, J.; Mohalik, N.K.; Mishra, R.K.; Khalkho, A.; Kumar, D.; Singh, V.K. Investigation of the role of fire retardants in preventing spontaneous heating of coal and controlling coal mine fires. Fire Technol. 2015, 51, 227–245. [Google Scholar] [CrossRef]
- Zhao, W.; He, M.; Zhang, W.; Liu, Y.; Wang, J. Preparation and flame retardant properties of new mining fireproof gel. Case Stud. Therm. Eng. 2024, 61, 105073. [Google Scholar] [CrossRef]
- Wei, L.; Qian, T.H. Relation between the change rules of coal structures when being oxidized andspontaneous combustion process of coal. J. China Coal Soc. 2007, 9, 939–944. [Google Scholar]
- Sun, L.; Zhan, M.; Zhang, C.; Shi, Q.; Huang, Q.; Wang, W. Experimental study on prevention of spontaneous combustion of coal by ionic surfactant solution injection in coal seam. Energy 2022, 260, 125079. [Google Scholar] [CrossRef]
- Upadhyay, A.; Kumar, B.; Raj, R. Ionic liquid as a cosurfactant for critical heat flux enhancement during boiling with aqueous surfactant solutions. Appl. Therm. Eng. 2024, 246, 122962. [Google Scholar] [CrossRef]
- He, X.; Liu, X.; Nie, B.; Song, D. Ftir and raman spectroscopy characterization of functional groups in various rank coals. Fuel 2017, 206, 555–563. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, J.; Li, Y.; Gao, S.; Yang, C.; Shi, X. Oxidation characteristics of functional groups in relation to coal spontaneous combustion. ACS Omega 2021, 6, 7669–7679. [Google Scholar] [CrossRef]
- Zhou, C.; Zhang, Y.; Wang, J.; Xue, S.; Wu, J.; Chang, L. Study on the relationship between microscopic functional group and coal mass changes during low-temperature oxidation of coal. Int. J. Coal Geol. 2017, 171, 212–222. [Google Scholar] [CrossRef]
- Deng, J.; Lu, H.; Li, D.; Xiao, Y. Thermal behavior effects of 1-butyl-3-methylimidazole tetrafluoroborate on coals spontaneous combustion with different metamorphic levels. J. China Coal Soc. 2019, 44, 254–262. [Google Scholar]
- Zhang, Y.N.; Hou, Y.C.; Liu, B.; Deng, J.; Liu, C.H.; Yang, J.J.; Wen, X.Y. Mechanism and performance of coal spontaneous combustion with a halide carrier inorganic salt inhibitor. Chin. J. Eng. 2021, 43, 1295–1303. [Google Scholar]
- Song, C.; Liu, X.; Zhao, S.; Yang, Z. Flame retardancy of anions in imidazolium-basedionic liquids on Xinjiang lignite. J. China Coal Soc. 2020, 45 (Suppl. S1), 470–480. [Google Scholar]
- Zeng, F.; Xie, K. Theoretical system and methodology of coal structural chemistry. J. China Coal Soc. 2004, 4, 443–447. [Google Scholar]
- Onifade, M.; Genc, B. Spontaneous combustion of coals and coal-shales. Int. J. Min. Sci. Technol. 2018, 28, 933–940. [Google Scholar] [CrossRef]
- Yan, L.; Wen, H.; Liu, W.Y.; Jin, Y.F.; Liu, Y.; Li, C.S. Adiabatic spontaneous coal combustion period derived from the thermal effect of spontaneous combustion. Energy 2022, 239, 122101. [Google Scholar] [CrossRef]

| Composite Inhibitor Sample | Sample ID | Sepiolite, g | Ionic Liquid, g | Water, g | |
|---|---|---|---|---|---|
| Sepiolite | 0% | SEP | 20 | 0 | 200 |
| [BMIM][BF4]-tc | 2% | SEP + 2% [BF4]− | 20 | 4 | 196 |
| 3% | SEP + 3% [BF4]− | 20 | 6 | 194 | |
| 4% | SEP + 4% [BF4]− | 20 | 8 | 192 | |
| 5% | SEP + 5% [BF4]− | 20 | 10 | 190 | |
| [BMIM][CL]-tc | 2% | SEP + 2% Cl− | 20 | 4 | 196 |
| 3% | SEP + 3% Cl− | 20 | 6 | 194 | |
| 4% | SEP + 4% Cl− | 20 | 8 | 192 | |
| 5% | SEP + 5% Cl− | 20 | 10 | 190 | |
| Moisture, % | Volatile Matter, % | Ash, % | Fixed Carbon, % |
|---|---|---|---|
| 0.39 | 35.89 | 16.54 | 47.18 |
| Inhibitor Name | Sample ID | Solute Component, g | Water, g | Coal, g |
|---|---|---|---|---|
| Raw Coal | 0 | 0 | 0 | 80 |
| 20%-MgCl2 | 1 | 4 | 16 | |
| 10%-SEP | 2 | 2 | 18 | |
| 5%-[BMIM][BF4] | 3 | 1 | 19 | |
| 5%-[BMIM][Cl] | 4 | 1 | 19 | |
| 10%-SEP + 3% [BF4]− | 5 | 2 | 18 | |
| 10%-SEP + 5% [Cl]− | 6 | 2 | 18 |
| Inhibitor Name | Coal Sample ID | T1, °C | T2, °C | T3, °C | T5, °C | T6, °C | Mass Loss, % |
|---|---|---|---|---|---|---|---|
| Raw Coal | 0 | 35.5 | 121.5 | 310.3 | 540.4 | 628.3 | 85.9 |
| 20%-MgCl2 | 1 | 58.9 | 130.9 | 314.9 | 497.9 | 577.9 | 84.8 |
| 10%-SEP | 2 | 70.3 | 122.6 | 316.4 | 534.8 | 642.3 | 85.5 |
| 5%-[BMIM][BF4] | 3 | 62.8 | 222.3 | 315.7 | 538.9 | 640.8 | 85.7 |
| 5%-[BMIM][Cl] | 4 | 83.7 | 148.6 | 302.8 | 547.6 | 743.6 | 88.7 |
| 10%-SEP + 3% [BF4]− | 5 | 74.5 | 155.5 | 312.5 | 554.5 | 650.3 | 84.8 |
| 10%-SEP + 5% [Cl]− | 6 | 70.4 | 144.4 | 310.4 | 550.4 | 649.8 | 85.2 |
| Inhibitor Name | Coal Sample ID | Time to Peak Heat Release Rate, s | Peak Heat Release Rate, (kW·m−2) | Total Heat Release, (MJ·m−2) |
|---|---|---|---|---|
| Raw Coal | 0 | 71 | 88.17 | 48.17 |
| 20%-MgCl2 | 1 | 179 | 83.11 | 36.50 |
| 10%-SEP | 2 | 238 | 82.45 | 35.94 |
| 5%-[BMIM][BF4] | 3 | 196 | 59.05 | 33.60 |
| 5%-[BMIM][Cl] | 4 | 223 | 53.03 | 35.96 |
| 10%-SEP + 3% [BF4]− | 5 | 276 | 59.21 | 32.74 |
| 10%-SEP + 5% [Cl]− | 6 | 199 | 68.84 | 33.68 |
| Inhibitor Name | Coal Sample ID | Time to Ignition, s | Fire Performance Index, (m2·s·kW−1) | Fire Growth Index, (kW·m−2·s−1) |
|---|---|---|---|---|
| Raw Coal | 0 | 31 | 0.35 | 1.24 |
| 20%-MgCl2 | 1 | 115 | 1.38 | 0.46 |
| 10%-SEP | 2 | 123 | 1.49 | 0.35 |
| 5%-[BMIM][BF4] | 3 | 115 | 1.94 | 0.31 |
| 5%-[BMIM][Cl] | 4 | 106 | 1.99 | 0.24 |
| 10%-SEP + 3% [BF4]− | 5 | 159 | 2.68 | 0.21 |
| 10%-SEP + 5% [Cl]− | 6 | 142 | 2.06 | 0.34 |
| Vibration Modes | Wavenumber Range, cm−1 | Functional Groups | Spectral Band Assignment |
|---|---|---|---|
| Oxygen-Containing Functional Groups | 3590–3697 | -OH | Free Non-Associated Hydroxyl Groups |
| 3200–3500 | Intermolecular Hydrogen-Bonded Association | ||
| 1715–1790 | C=O | C=O Stretching Vibration | |
| Aliphatic Hydrocarbons | 2950–2975 | -CH3 | Asymmetric CH3 Vibration |
| 2850–2860 | Symmetric CH2 Stretching Vibration | ||
| 2915–2940 | -CH2- | Asymmetric CH2 Stretching Vibration | |
| Aromatic Hydrocarbons | 1490–1620 | C=C | Conjugated C=C In-Plane Ring Vibration |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Sun, J.; Li, W.; Zhang, Q. Experimental Study on Inhibition Characteristics of Imidazolium-Ionic-Liquid-Loaded Sepiolite Composite Inhibitor. Fire 2025, 8, 343. https://doi.org/10.3390/fire8090343
Zhang X, Sun J, Li W, Zhang Q. Experimental Study on Inhibition Characteristics of Imidazolium-Ionic-Liquid-Loaded Sepiolite Composite Inhibitor. Fire. 2025; 8(9):343. https://doi.org/10.3390/fire8090343
Chicago/Turabian StyleZhang, Xiaoqiang, Jinghong Sun, Wenlin Li, and Qin Zhang. 2025. "Experimental Study on Inhibition Characteristics of Imidazolium-Ionic-Liquid-Loaded Sepiolite Composite Inhibitor" Fire 8, no. 9: 343. https://doi.org/10.3390/fire8090343
APA StyleZhang, X., Sun, J., Li, W., & Zhang, Q. (2025). Experimental Study on Inhibition Characteristics of Imidazolium-Ionic-Liquid-Loaded Sepiolite Composite Inhibitor. Fire, 8(9), 343. https://doi.org/10.3390/fire8090343






