Abstract
Background/Objectives: Stevens–Johnson Syndrome (SJS) and toxic epidermal necrolysis (TEN) are rare, potentially fatal immunological conditions that affect cutaneous and mucosal surfaces and have the potential to involve the genitourinary tract. While genital involvement is common, urological manifestations are under-recognised clinically and there is a paucity of clear, evidence-based management pathways specific to urological manifestations of SJS/TEN. To map the spectrum of urological manifestations of SJS/TEN, to describe the short- and long-term outcomes of these manifestations, and to synthesise management and prevention strategies to inform clinical practice. Methods: This was a scoping review conducted in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) guideline. Data sources: Medline and PubMed articles published in English with publication date up to December 2025. Study selection: Eligible studies included case reports, case series, observational studies, clinical guidelines, and review articles describing urological manifestations, outcomes, management, or prevention strategies for patients with SJS/TEN. Articles limited to renal or isolated gynaecological involvement were excluded. Data extraction and synthesis: Articles were screened independently by two reviewers using a pre-defined data extraction template covering four domains: urological manifestations, outcomes and sequelae, management strategies, and prevention strategies. This criterion was refined after a pilot of 20 studies. Discrepancies were resolved by consensus with a third reviewer. Formal risk-of-bias assessment was not performed, consistent with scoping review methodology. Results: One hundred and four studies published between 1987 and 2025 were included in this review. Selected articles included case reports (n = 63), retrospective cohort studies (n = 23), prospective studies (n = 2), guidelines (n = 5), and summary articles (n = 11). Reported urological involvement ranged from genital cutaneous and mucosal disease including erosions, adhesions, and balanitis to urethral manifestations such as urethritis, stenosis, and strictures, as well as scarce upper urinary tract involvement including ureteric stricture and ureteric mucosal sloughing. While some manifestations resolved with supportive care, others progressed to chronic sequelae including persistent urethral strictures, voiding dysfunction, sexual dysfunction, recurrent infection, and in rare cases, obstructive uropathy. A multidisciplinary approach was recommended for all patients with SJS/TEN. Urological management centred around early and repeated urogenital examination, manual lysis of adhesions, urinary catheterisation, and timely intervention for urethral or ureteric obstruction. Long-term urological follow-up of 12 months was recommended for patients with significant urogenital involvement. Conclusions: Urological manifestations of SJS/TEN are diverse, clinically significant, and frequently under-recognised. Early urological involvement, systematic genital and urinary tract assessment, and proactive preventative measures may reduce long-term morbidity. This review provides a comprehensive synthesis of knowledge and recommendations to support urologists’ role in multidisciplinary care of patients with this pathology. This review also highlights the need for prospective research to guide further evidence-based management of urological complications of SJS/TEN.
1. Introduction
Stevens–Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) are rare, but potentially fatal, immunological conditions affecting the skin and mucosa [1]. They affect 1–7 people per million annually worldwide [2]. SJS/TEN are type IV hypersensitivity reactions initiated by infection or new drug exposure [2]. They commonly present with a fever and flu-like symptoms followed by cutaneous and mucosal erosion [2]. SJS/TEN is diagnosed clinically with thorough history and examination. Diagnosis can be supported by tissue biopsy.
SJS/TEN is a spectrum of disease categorised by percentage of total body surface area (%TBSA) involvement [3]. SJS describes disease states involving less than 10% TBSA, whilst TEN involves greater than 30% TBSA. SJS/TEN overlap syndrome involves 10–30% TBSA [3]. As the epidermis necroses, patients are at risk of dehydration, sepsis, and organ failure [4]. The risk of mortality correlates with TBSA involvement: 1–5% mortality with SJS, and up to 30% mortality with TEN [4]. Management of SJS/TEN predominantly takes the form of supportive care [5]. Culprit agents are removed, and judicious rehydration, analgesia, antibiotics, and corticosteroid therapy are provided [5], often in a critical care environment.
SJS/TEN most commonly affects mucosal surfaces of the mouth, eyes, and nose [4]. While gynaecological involvement is well described [6], urological manifestations of SJS/TEN are perceived as rare. Assessment of the urogenital tract is recommended for every patient with SJS/TEN, though this is often not performed [7]. Furthermore, preventative measures and treatment strategies for urological manifestations of SJS/TEN are not well described [8]. There is a paucity of clear evidence-based guidelines to guide acute and long-term management of urological manifestations of SJS/TEN [7]. The purpose of this review is to first identify and describe the spectrum of urological manifestations of SJS/TEN, and to then discuss strategies to manage these manifestations. This review will thereby serve as a resource for urologists and other healthcare workers to inform care practices in the management of urological manifestations of SJS/TEN.
2. Methods
This study follows the protocol entitled ‘Urological manifestations of SJS/TEN and their management: a scoping review protocol’ available on Open Science Framework (Supplementary Materials S1). The review follows the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) Extension for Scoping Review guidelines [9].
Patient and public involvement: There was no involvement of patients or the public in this study.
Search strategy: MEDLINE and PubMed were searched in December 2025. Key words and MeSH terms relevant to SJS/TEN and urological manifestations (such as ‘genital’, ‘penis’, ‘urethra’, ‘ureter’, ‘scrotum’, ‘phimosis’, and ‘balanitis’) were included in the search. After reviewing the results of a pilot search, terms relating to renal rather than urological manifestations (such as ‘renal’) were excluded from the search (Supplementary Materials S2). Search filters provided no restriction on publication type but limited the search to English-language publications.
Study selection: Publication titles and abstracts were screened independently by two reviewers (ZW and ADC). A third reviewer (AD) was available to address any discrepancies in article selection. Review articles, clinical guidelines, case reports, case series, and comparative study designs were considered in the search (Figure 1).
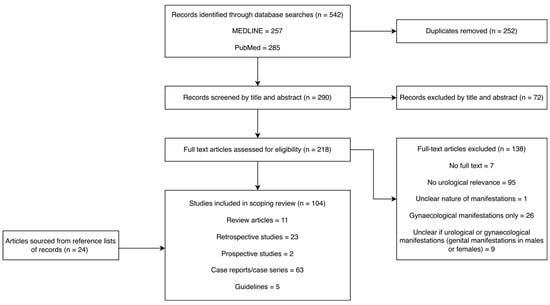
Figure 1.
Screening and eligibility assessment of articles generated by MEDLINE and PubMed searches of the literature.
Articles were included if they discussed the following:
Urological manifestations of SJS/TEN;
Outcomes of urological manifestations of SJS/TEN;
Management of urological manifestations of SJS/TEN;
Measures to prevent urological manifestations of SJS/TEN.
Reference lists of included articles were manually reviewed for eligibility under the same review criteria. Pilot study selection was performed with 20 articles, which yielded additional selection criteria: articles discussing solely renal or gynaecological manifestations of SJS/TEN were excluded. Articles discussing ‘genital manifestations’ in a mixed-gender group of patients, where urological manifestations could not be distinguished from gynaecological manifestations, were excluded. Articles discussing the management of gynaecological manifestations of SJS/TEN were excluded if they did not refer to urological manifestations of SJS/TEN and/or would be managed independently by a gynaecologist without urology input. Conference extracts, commentaries, and articles not available in English were excluded.
Data management software: Endnote 20 (Clarivate Analytics, Inc.) managed references generated by the search and excluded duplicates.
Data extraction: Extracted data were managed independently by the two reviewers. A bespoke data extraction template was used based on the four content domains listed above. This data extraction template was modified after pilot data extraction of 20 articles. A risk of bias assessment was not conducted in keeping with the methodology of scoping reviews. Two reviewers compared data at the conclusion of the data extraction process. Data were discussed and emerging themes were identified. Points of clarification were discussed with a third reviewer. The inclusion/exclusion criterion was revisited and remained unchanged.
Review questions:
What are the most common urological manifestations of SJS/TEN reported in the literature?
How are urological manifestations of SJS/TEN managed?
What are the outcomes of urological manifestations of SJS/TEN?
What measures are taken to prevent urological manifestations of SJS/TEN?
3. Results
One hundred and four studies were included in this review. Publication dates ranged from 1987 to 2025. Selected articles included case reports (n = 63), retrospective cohort studies (n = 23), prospective studies (n = 2), guidelines (n = 5), and summary articles (n = 11).
The range of urological manifestations of SJS/TEN are outlined in Figure 2. Urological manifestations range from penile and scrotal cutaneous lesions to mucositis, urethral strictures, and ureteric strictures.
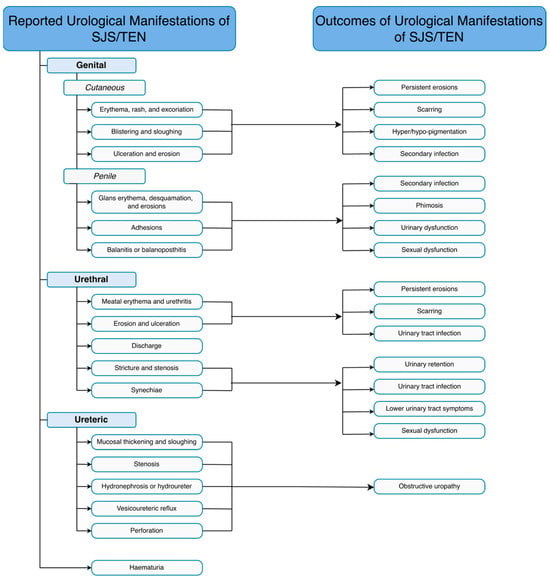
Figure 2.
Reported urological manifestations of SJS/TEN and outcomes of these manifestations. SJS: Stevens–Johnson Syndrome; Toxic epidermal necrolysis (TEN)3.1. Outcomes of Urological Manifestations of SJS/TEN.
Male genital involvement is common in SJS/TEN, occurring in up to two thirds of patients with SJS/TEN [10,11]. Genital involvement usually occurs concurrently with cutaneous and oral mucosal manifestations [11,12,13,14,15,16,17,18,19]. Isolated male genital manifestations of SJS/TEN are uncommon [14]. Male genital cutaneous manifestations range from erythema or purpura [20,21,22,23,24,25,26] to blistering [27,28,29,30] and ulceration/erosion with or without mucopurulent discharge [10,20,23,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46]. These genital cutaneous manifestations in males occur in 19–33% of patients with urogenital manifestations of SJS/TEN. Male genital mucosal manifestations include mucositis [26,28,47,48,49,50,51], erosion/ulceration [10,22,27,31,44,47,48,49,52,53,54,55,56,57,58,59], and secondary infection [49]. Penile sequelae including balanitis or balanoposthitis [4,10,14] and adhesions between the glans and the prepuce [60,61,62] are reported with variable frequency.
Urethral manifestations range from urethritis to erosion, ulceration, and necrosis, with significant exudate reported in up to 39% of patients [4,42,60,63,64]. Urethral sequelae of strictures [4,49,65,66], stenosis [60,67], synechiae [67,68], and discharge [37] have also been described in up to 5% of patients. In a retrospective study of 1167 patients with SJS/TEN in Korea, urethral sequelae were present in 5.7% of patients with SJS and 9.6% of patients with TEN [69]. ‘Urethral involvement’ was reported to be the second most common manifestation in this cohort.
Bladder and upper urinary tract manifestations of SJS/TEN are scarcely reported. Isolated case reports describe haematuria [70,71], ureteric mucosal damage [72,73], ureteric stenosis [72], hydronephrosis or hydroureter [74,75], and vesicoureteric reflux [76]. There is one report of multifocal nephrogenic adenoma of the bladder with concurrent mucosal impairment of the urethra and ureters in a 10-year-old boy 5 months post SJS [73].
3.1. Outcomes of Urological Manifestations of SJS/TEN
The progression of urological manifestations of SJS/TEN ranges from complete resolution with supportive care [27,32,60,77,78] to life-threatening outcomes. The range of outcomes of urological manifestations of SJS/TEN are outlined in Figure 2.
Cutaneous and mucosal urological lesions can be self-limiting [77]. Full epithelialisation of cutaneous and mucosal urological lesions has been described [32,44,64,72,78]. Hyper- or hypopigmentation of the glans penis at sites of previous lesions may also persist [33,45,60]. Urogenital lesions often demonstrate protracted healing and result in scarring [61,62]. Genital mucosal erosions in particular may persist for weeks to months before scarring [49,61]. Such persisting lesions have been described in up to 5/30 patients in one study [14]. Secondary infection may also result, commonly from bacteria or candida, and less frequently from reactivated herpes simplex virus [49]. Adhesions between the glans penis and the prepuce self-resolved in one case [60], while others progressed to phimosis [10,14,49,79,80,81].
Periurethral scarring, stricture, or stenosis can contribute to voiding lower urinary tract symptoms and urinary retention [6,7,8,49,82,83,84]. Dysuria is also frequently reported in patients with SJS/TEN [46,64,85,86,87,88]. Urinary tract infection has been identified in 5–70% of patients [48,89,90,91,92,93,94]. The relationship between urinary tract infection and mortality in the setting of SJS/TEN is unclear as a meta-analysis of 11 studies showed substantial heterogeneity [94]. Long-term morbidity may ensue from urethral strictures or stenosis secondary to fibrosis in SJS/TEN implicating both urinary and sexual function [7,46]. In one study, urethral strictures were persistent on follow-up in 2/32 patients [95]. Chronic balanitis has also been reported as longer-term urological sequela of SJS/TEN [8,96,97]. Chronic pain and psychological sequelae secondary to genital cutaneous changes or sexual dysfunction have also been reported [8].
Urological manifestations of SJS/TEN can occur in life-threatening cases of SJS/TEN and require prompt management. Bilateral ureteric obstruction secondary to ureteric complications of SJS/TEN required bilateral double-J ureteric stenting in one study, which was ultimately fatal secondary to anuric renal failure despite renal decompression [74]. Here, ureteric obstruction likely contributed to and exacerbated existing renal dysfunction caused by pre-renal or intrinsic kidney injury [74,82,98]. Septic shock and acute renal failure have also been described, at times secondary to common uropathogens [56,99]. Some of these cases required haemodialysis and some were fatal [24,56,71]. While genital mucosal damage is not independently correlated with mortality, concurrent involvement of oral, ocular, and external genitalia increases the risk of death in SJS/TEN [100,101].
3.2. Management and Prevention of Urological Manifestations of SJS/TEN
All papers echoed similar principles for the management of SJS/TEN. These principles centred around ceasing the causative agent, initiating high-dose immunosuppressive therapy, plasmapheresis, and supportive treatment, including fluid resuscitation, skin cares, and monitoring for infection [46]. The management of urological manifestations of SJS/TEN is largely supportive [61], though procedural intervention is occasionally required.
Management should be conducted by a multidisciplinary team in a tertiary centre with a specialised burns or plastics unit [8,43,45,46,66,97,102]. The multidisciplinary team should be led by a dermatologist with consultation from a urologist for management of urological manifestations [66]. Initial assessment of a patient with SJS/TEN should include urogenital examination by a urologist in male patients and a gynaecologist in female patients within 48 h of presentation [8,61]. Urologists should be involved if there is any concern for urethral involvement in female patients. Examination should be repeated daily during the acute phase of the illness and continued regularly even as cutaneous manifestations begin to resolve [8,61]. Wounds and body fluids should be sampled for secondary infection if appropriate [61].
3.3. Genital Cutaneous Lesions
Inflamed, non-eroded urogenital surfaces can be treated with [26,50,103] topical emollients such as white soft paraffin ointment or petroleum gel in the acute phase to maintain barrier protection, prevent adhesions, and reduce pain [8,49,60,61,102]. Reapplication is recommended multiple times daily [8,60,61]. High-potency topical corticosteroids can be considered for inflamed, non-eroded areas [8,26,49,61,103]. The recommended limit for the duration of topical corticosteroid use is 5 days [61]. Doses should be tapered when disease progression halts or clinical improvement is noted [8,61]. Human epidermal growth factor gel [64] and topical antibiotics such as mupirocin ointment [64] or polymyxin B ointment [45] can also be applied to complete medical management [65]. Non-adherent, silver-impregnated, antibacterial dressings with a second overlying dressing to absorb exudate are recommended until complete re-epithelialisation is achieved [8,44,61,62]. Necrotic tissue should not be debrided in the setting of SJS/TEN [61]. Even in the absence of genital cutaneous lesions, the urogenital area should be cleaned with 0.9% sodium chloride or antiseptic solutions at the time of dressing changes [61].
3.4. Urogenital Adhesions
Manual severing of visible adhesions and synechiae without damaging mucosa is recommended for the management of genital adhesions [4,60,61,104]. For men with adhesions between the glans penis and the prepuce, this can be achieved by gentle preputial mobilisation [12] to prevent phimosis [12,105]. For women, this would involve severing synechiae between the labia minora and majora to prevent urinary obstruction, which could be performed by urology or gynaecology [12]. Assessment of preputial retraction should form part of the initial urological assessment for all males with SJS/TEN to risk stratify adhesional phimosis [49]. Regular preputial retraction should be performed in uncircumcised males, including those without observed adhesions at the glans penis, to prevent phimosis [8,49,61]. Regular preputial retraction should not be performed in children or in adult men with established phimosis [61].
3.5. Urethral Strictures
Visible adhesions at the external urethral orifice should be managed with manual separation to manage lower urinary tract symptoms and prevent stricture formation [4,60,104]. Urethral catheterisation is recommended to prevent labial synechiae and more proximal urethral stricture [4,8,39,43,49,61,103,104,105,106]. Further management strategies for labial synechiae are implemented by gynaecology and are outside the scope of this review. In a study of 31 paediatric patients with SJS/TEN, the incidence of urethral stricture was low, and no adverse outcomes or long-term sequelae of urethral catheterisation were described [60,102]. Only one study cautioned against urethral catheterisation due to concern for risk of urinary tract infection [83]. In the acute phase, and particularly if urethral mucosal irritation is known or suspected, urethral catheterisation and catheter changes are recommended to be performed with adequate analgesia, with or without sedation [61].
3.6. Other Urological Manifestations
Three studies describe haematuria in patients with SJS/TEN [8,70,71]. Continuous bladder irrigation via a three-way urethral catheter alongside regular supportive care was successful in managing haematuria and preventing clot formation [71]. Dysuria can be managed symptomatically with phenazopyridine [102] or with urethral catheterisation in the setting of urethral mucositis or erosion [8,61]. One study describes bilateral ureteric obstruction secondary to right ureteric stricture and left ureteric mucosal sloughing and forniceal rupture of the left lower pole calyx [72]. Here, bilateral retrograde ureteric stenting was successful in managing ureteric obstruction and perforation [72]. A further study describes bilateral ureteric obstruction secondary to mucosal debris in a 10-year-old male, which was also managed with bilateral double-J ureteric stents [73]. In this case, the ureteric stents were removed 2 months later secondary to stent blockage by mucosal debris. Serial ultrasonography was performed for one year and did not reveal further obstruction [73].
3.7. Long-Term Follow-Up
Since urological manifestations can occur for up to one year after the initial episode of SJS/TEN, regular longer-term urological follow-up is recommended to monitor for longer-term sequelae and ensure complete recovery [44,60,73,107]. Children should be included in this long-term follow-up despite the rarity of genitourinary complications of SJS/TEN in this population [60,66]. Close urology follow-up should be conducted at 1–2 months post discharge in cases of severe urogenital manifestations of SJS/TEN or those at high risk of developing urethral stricture of synechiae [66]. Follow-up is recommended for at least 1 year following the acute episode [73].
4. Discussion/Recommendations
While SJS/TEN is rare, genital involvement is common and urological manifestations can be expected. Here, we provide a comprehensive summary of the spectrum of urological manifestations of SJS/TEN, their potential sequelae, and strategies for the management and prevention of these complications.
SJS/TEN can affect the skin and mucous membranes of urological structures including the penis, scrotum, urethra, and ureters. Urological involvement ranges from cutaneous lesions of the penis and scrotum to urethral stricture and upper urinary tract obstruction. Management centres around supportive care and repeated systems-based assessments. Urologists perform regular examinations to assess urogenital involvement and facilitate procedural management including manual lysis of urogenital adhesions, urinary catheterisation for the prevention of urethral strictures and adhesions, continuous bladder irrigation for clot retention, and renal decompression in the setting of ureteric obstruction. Where cutaneous and mucosal manifestations can be self-limiting, secondary infection can occur. Persisting manifestations and their sequelae also contribute to urinary obstruction and sexual dysfunction and have psychological implications. While isolated genital involvement does not increase the risk of death from SJS/TEN [100], concurrent involvement of genital, ocular, and oral mucosa is associated with increased mortality [101]. It is unclear whether this association is independent of the known association of rising mortality in SJS/TEN with rising TBSA involvement.
The incidence of urological manifestations of SJS/TEN is difficult to quantify. Acute-phase genitourinary involvement has been reported to affect up to 70% of males with SJS/TEN [8,45,60,98,102]. The largest cohort study yielded by our search was performed by Yang et al. in Korea [69]. In their retrospective study of 1167 patients with SJS/TEN, urethral sequelae were present in 5.7% of patients with SJS and 9.6% of patients with TEN [69]. ‘Urethral sequelae’ were reportedly the second most common manifestations in their cohort; however, the nature of these manifestations was not described. We sought clarification of the nature of these sequelae but did not receive any response.
Multidisciplinary management is key to the management of any patient with SJS/TEN. Urology and/or gynaecology involvement is recommended for all patients with SJS/TEN, even for patients without initially identified urogenital involvement [8]. In an audit of 147 adult patients with SJS/TEN, 37% did not have a urogenital tract assessment, and documentation of urethral involvement was identified in only 12% of cases [7]. Daily assessments of the urinary tract and genital skin occurred in only 10% of patients with SJS/TEN [7]. Whether the absence of documentation of these examinations is secondary to negative examination findings or substandard urogenital examination is unclear. Multidisciplinary involvement is likely to yield more thorough and regular multi-system assessments of patients with SJS/TEN to allow earlier identification and management of urogenital manifestations. An absence of multidisciplinary involvement can increase the risk of unidentified urogenital issues in patients with SJS/TEN, which may worsen long-term outcomes [102].
The rarity of urological manifestations of SJS/TEN makes the management and prevention of sequelae difficult to study. The centralisation of care to specialised burns centres has improved recognition of urological sequelae and overall morbidity and mortality of SJS/TEN patients [5]. However, a number of questions regarding management principles remain. While topical steroid management appears to be a mainstay in the management of SJS/TEN, it is unclear if steroids increase the risk of severe secondary infection or reduce mortality [26,61]. Similarly, while several studies advocate for the use of urinary catheters for the management of dysuria [8], haematuria [71], or the prevention of urethral stricture or adhesions [4,39,49,104,105,106], others caution against the potential for catheter-associated urinary tract infection in an already compromised patient [4,83]. Given that most SJS/TEN patients require care in intensive care units, the benefit of urinary catheterisation, at least for fluid status monitoring, is likely to be more beneficial than harmful, regardless of urological considerations [8,12,60,106,108]. The utility of urinary catheterisation in preventing urological sequelae in stable patients without urogenital involvement remains unanswered. Furthermore, variations in the frequency of catheter changes from the standard 4–6 weeks require exploration, as does the optimal duration of catheterisation. Following our review, we posit that urethral catheters should be removed upon evidence of urethral healing [8].
There is a paucity of data regarding appropriate follow-up regimes for patients with urogenital manifestations of SJS/TEN. For patients with established urogenital manifestations of SJS/TEN, regular follow-up is recommended for up to one year to ensure complete recovery and monitor for long-term sequelae [44,60,73,107]. Close monitoring every 1–2 months should be performed for those with severe urological manifestations. We reviewed one case of a 10-year-old patient who presented with ureteric obstruction secondary to significant mucosal sloughing 5 months after an acute episode of SJS/TEN [72]. Interestingly, multifocal nephrogenic adenoma of the bladder was also identified in this patient and has not been reported in any other cases of SJS/TEN [72]. In this case, presentation with bilateral flank pain 5 months post SJS/TEN led to ultrasonography and subsequent cystoscopic and ureteroscopic examination, which identified these urological manifestations [72]. The timing of these manifestations is peculiar and highlights the potential for late urological manifestations of SJS/TEN requiring surgical intervention. Despite this report of late-onset urological manifestations without documented acute-phase urogenital involvement, this is an isolated case. There is unlikely to be significant benefit in regular urogenital examinations and serial imaging in patients who have never demonstrated urogenital involvement. Based on our review, we recommend close monitoring for one year, initially every 1–2 months, and then as per practitioners’ judgement, for patients with urogenital manifestations of SJS/TEN. However, we cannot make a strong evidence-based recommendation for regular urological follow-up for patients who have not demonstrated urological manifestations of SJS/TEN.
Following our review, we outline our the recommendations for management and prevention of urological manifestations of SJS/TEN in Figure 3.

Figure 3.
Recommended management of urological manifestations of SJS/TEN. SJS: Stevens–Johnson Syndrome; Toxic epidermal necrolysis (TEN).
Strengths and Limitations
This study provides a scoping review of the literature with rigorous adherence to the study methodology. This study answers the four review questions according to the level of detail available in the literature.
There are a number of limitations of this study. In keeping with the nature of scoping reviews, the quality of evidence was not assessed. Only a few, large retrospective studies exist; the remainder of the evidence is sourced from single case studies or small volume case series, as well as from several reviews and guidelines (Figure 1).
A number of studies comment on ‘genital manifestations’ in grouped male and female patients. Given the difficulties in determining the gynaecological or urological nature of these genital manifestations, these studies were excluded, which reduced the availability of frequency data. These data are helpful in predicting sequelae and initiating early involvement of urological and gynaecological teams. Furthermore, there is an overlap of gynaecological and genitourinary manifestations of SJS/TEN [102]. Our exclusion of studies reporting gynaecological manifestations of SJS/TEN could impact our reporting of urological manifestations at this point of overlap.
Another limitation of this study is the large number of studies excluded due to imprecise descriptions of the nature and location of urological manifestations, which prevented their inclusion in this review. Most notably, descriptions of urological strictures were often of unspecified location in the urinary tract or described as ‘urethral’ strictures without specification of meatal or more proximal location. Descriptions of ureteric manifestations of SJS/TEN are also limited. The pathophysiological association of these manifestations is unclear, and these manifestations may arise from renal manifestations of SJS/TEN causing papillary necrosis and other injuries. While descriptions of ureteric manifestations of SJS/TEN were included in this review, their aetiology should be interpreted with caution.
Most studies described acute-phase urological manifestations of SJS/TEN with little data on long-term outcomes, impairing strong recommendations for long-term management. Our suggestion for 3-, 6-, and 12-monthly reviews is in acknowledgement of urological manifestations being reported to persist for up to one year.
5. Conclusions
Urological manifestations of SJS/TEN are diverse, clinically significant, and frequently under-recognised. Here, we described the range and progression of urological manifestations of SJS/TEN and synthesise recommended management strategies. The review serves as a comprehensive resource to support urologists’ role in the multidisciplinary care of patients with SJS/TEN. This review also highlights the need for prospective research to guide further evidence-based management of urological complications of SJS/TEN.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/siuj7010019/s1, Supplementary Materials S1: Study Protocol; Supplementary Materials S2: Search Strategy.
Author Contributions
Z.W.—protocol drafting, database search strategy development and execution, study screening and assessment of study eligibility, data extraction, manuscript drafting. P.K.—data synthesis: manuscript drafting, manuscript review. A.D.C.—study screening and assessment of study eligibility, data extraction. J.K.—protocol development, manuscript review. A.S.—protocol development, manuscript review. A.D.—protocol development, study selection (third review as tiebreaker), manuscript review. A.S.J.C.—protocol development, manuscript review. All authors have read and agreed to the published version of the manuscript.
Funding
A.D.C. received funding from Common Good Trust of Royal North Shore Hospital. The other authors did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sectors for this research.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analysed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
A.S.J.C. is a proctor for Boston Scientific, Coloplast and Medtronic. A.S.J.C. is an advisory board member for Coloplast. The other authors declare no competing interests.
References
- Lissia, M.; Mulas, P.; Bulla, A.; Rubino, C. Toxic epidermal necrolysis (Lyell’s disease). Burns 2010, 36, 152–163. [Google Scholar] [CrossRef]
- Schwartz, R.A.; McDonough, P.H.; Lee, B.W. Toxic epidermal necrolysis: Part I. Introduction, history, classification, clinical features, systemic manifestations, etiology, and immunopathogenesis. J. Am. Acad. Dermatol. 2013, 69, 173.e1–173.e13. [Google Scholar] [CrossRef]
- Bastuji-Garin, S.; Rzany, B.; Stern, R.S.; Shear, N.H.; Naldi, L.; Roujeau, J.-C. Clinical classification of cases of toxic epidermal necrolysis, Stevens-Johnson syndrome, and erythema multiforme. Arch. Dermatol. 1993, 129, 92–96. [Google Scholar] [CrossRef]
- Schwartz, R.A.; McDonough, P.H.; Lee, B.W. Toxic epidermal necrolysis: Part II. Prognosis, sequelae, diagnosis, differential diagnosis, prevention, and treatment. J. Am. Acad. Dermatol. 2013, 69, 187.e1–187.e16. [Google Scholar] [CrossRef]
- Dalli, R.L.; Kumar, R.; Kennedy, P.; Maitz, P.; Lee, S.; Johnson, R. Toxic epidermal necrolysis/Stevens–Johnson syndrome: Current trends in management. ANZ J. Surg. 2007, 77, 671–676. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, K.F.; Bradley, S.E.; Mitchell, C.M.; Cardis, M.A.; Mauskar, M.M.; Pasieka, H.B. Vulvovaginal manifestations in Stevens-Johnson syndrome and toxic epidermal necrolysis: Prevention and treatment. J. Am. Acad. Dermatol. 2021, 85, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Tasker, F.; Smith, S.P.; Mustapa, M.F.M.; de Berker, D.A.R. British Association of Dermatologists national clinical audit on the management of Stevens–Johnson syndrome/toxic epidermal necrolysis in adults. Clin. Exp. Dermatol. 2024, 49, 810–816. [Google Scholar] [CrossRef] [PubMed]
- Seminario-Vidal, L.; Kroshinsky, D.; Malachowski, S.J.; Sun, J.; Markova, A.; Beachkofsky, T.M.; Kaffenberger, B.H.; Ergen, E.N.; Mauskar, M.; Bridges, A.; et al. Society of Dermatology Hospitalists supportive care guidelines for the management of Stevens-Johnson syndrome/toxic epidermal necrolysis in adults. J. Am. Acad. Dermatol. 2020, 82, 1553–1567. [Google Scholar] [CrossRef] [PubMed]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Gulanikar, A.; Abrol, A.; Sagar, S. Study of genital manifestations of Stevens Johnson syndrome/Toxic epidermal necrolysis. Indian J. Sex. Transm. Dis. AIDS 2022, 43, 39–42. [Google Scholar] [CrossRef] [PubMed]
- Sauteur, P.M.M.; Goetschel, P.; Lautenschlager, S. Mycoplasma pneumoniae and mucositis—Part of the Stevens-Johnson syndrome spectrum. J. Der Dtsch. Dermatol. Ges. 2012, 10, 740–745. [Google Scholar] [CrossRef]
- Charlton, O.A.; Harris, V.; Phan, K.; Mewton, E.; Jackson, C.; Cooper, A. Toxic epidermal necrolysis and Stevens–Johnson syndrome: A comprehensive review. Adv. Wound Care 2020, 9, 426–439. [Google Scholar] [CrossRef] [PubMed]
- Chhipa, S.A.; Masood, S.; Salarzai, Y.; Khan, Q.A.; Ahmad, K.; Sajid, S. Ocular manifestation, complications and aetiological factors in Stevens-Johnson syndrome/toxic epidermal necrolysis. J. Pak. Med. Assoc. 2015, 65, 62–64. [Google Scholar]
- Revuz, J.; Penso, D.; Roujeau, J.-C.; Guillaume, J.-C.; Payne, C.R.; Wechsler, J.; Touraine, R. Toxic epidermal necrolysis. Clinical findings and prognosis factors in 87 patients. Arch. Dermatol. 1987, 123, 1160–1165. [Google Scholar] [CrossRef] [PubMed]
- Chatproedprai, S.; Wutticharoenwong, V.; Tempark, T.; Wananukul, S. Clinical features and treatment outcomes among children with Stevens-Johnson syndrome and toxic epidermal necrolysis: A 20-year study in a tertiary referral hospital. Dermatol. Res. Pract. 2018, 2018, 1–9. [Google Scholar] [CrossRef]
- Sevketoglu, E.; Hatipoglu, S.; Akman, M.; Bicer, S. Toxic Epidermal necrolysis in a child after carbamazepine dosage increment. Pediatr. Emerg. Care 2009, 25, 93–95. [Google Scholar] [CrossRef]
- Saitoh, A.; Ohya, T.; Yoshida, S.; Hosoya, R.; Nishimura, K. A case report of Stevens-Johnson syndrome with Mycoplasma pneumoniae infection. Pediatr. Int. 1995, 37, 113–115. [Google Scholar] [CrossRef]
- Canter, N.; Smith, L. Incomplete Stevens-Johnson syndrome caused by sulfonamide antimicrobial exposure. Clin. Pract. Cases Emerg. Med. 2019, 3, 240–242. [Google Scholar] [CrossRef]
- Abuhammad, A.; Ayyad, M.; Albandak, M.; Natsheh, M.; Attawna, S.I.Y. Levofloxacin-induced fuchs syndrome: A rare atypical Stevens–Johnson syndrome variant with a comprehensive literature review. Case Rep. Med. 2025, 2025, 9944674. [Google Scholar] [CrossRef]
- Mermershtain, W.; Cohen, A.; Lazarev, I.; Grunwald, M.; Ariad, S. Toxic epidermal necrolysis associated with gemcitabine therapy in a patient with metastatic transitional cell carcinoma of the bladder. J. Chemother. 2003, 15, 510–511. [Google Scholar] [CrossRef]
- Bannaga, A.; Rahama, O.; Barlow, G. Nevirapine-induced Stevens-Johnson syndrome following HIV postexposure prophylaxis. BMJ Case Rep. 2013, 2013, bcr2013009453. [Google Scholar] [CrossRef]
- Sendi, P.; Graber, P.; Lepère, F.; Schiller, P.; Zimmerli, W. Mycoplasma pneumoniae infection complicated by severe mucocutaneous lesions. Lancet Infect. Dis. 2008, 8, 268. [Google Scholar] [CrossRef]
- Viscuse, P.V.; Marques-Piubelli, M.L.; Heberton, M.M.; Parra, E.R.; Shah, A.Y.; Siefker-Radtke, A.; Gao, J.; Goswami, S.; Ivan, D.; Curry, J.L.; et al. Case report: Enfortumab vedotin for metastatic urothelial carcinoma: A case series on the clinical and histopathologic spectrum of adverse cutaneous reactions from fatal Stevens-Johnson syndrome/toxic epidermal necrolysis to dermal hypersensitivity reaction. Front. Oncol. 2021, 11, 621591. [Google Scholar] [CrossRef]
- Karthikeyan, K.; Sameera, K.V.; Shaji, S.; Swetha, M.A.C.; Madhu, C.S. Capecitabine induced Steven–Johnson syndrome: A rare case report. J. Oncol. Pharm. Pract. 2021, 28, 250–254. [Google Scholar] [CrossRef]
- Cocca, S.; Viviano, M. Stevens-Johnson syndrome and abuse of anabolic steroids. J. Korean Assoc. Oral Maxillofac. Surg. 2017, 43, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Buganu, A.; Atta, M.; Solomon, M.; Banerjee, P.R.; Ganti, L. Stevens Johnson syndrome initiated by an adverse reaction to trimethoprim-sulfamethoxazole. Cureus 2020, 12, e10023. [Google Scholar] [CrossRef] [PubMed]
- Alves, D.D.; Lourenço, N.; Franco, D.; Calmeiro, E.; Silva, R. Toxic epidermal necrolyisis and sepsis. Eur. J. Case Rep. Intern. Med. 2016, 3, 000502. [Google Scholar] [CrossRef] [PubMed]
- Grafanaki, K.; Lygeros, S.; Vryzaki, E.M.; Georgiou, S. Rare vardenafil-associated Stevens-Johnson syndrome: Toxic epidermal necrolysis. BMJ Case Rep. 2022, 15, e249013. [Google Scholar] [CrossRef]
- Maciejewska, J.; Jankowski, M.; Zegarska, B.; Czajkowski, R. Stevens-Johnson syndrome/toxic epidermal necrolysis presumably induced by norfloxacin. Adv. Dermatol. Allergol. 2014, 3, 194–196. [Google Scholar] [CrossRef]
- Taqi, S.A.; Zaki, S.A.; Nilofer, A.R.; Sami, L.B. Trimethoprim-sulfamethoxazole-induced Stevens Johnson syndrome in an HIV-infected patient. Indian J. Pharmacol. 2012, 44, 533–535. [Google Scholar] [CrossRef]
- Barman, K.D.; Verma, K.K.; Agrawal, S.; Agarwalla, A.; Rijal, A. Stevens-Johnson Syndrome with idiopathic thrombocytopenic purpura treated with dexamethasone pulse therapy. J. Dermatol. 2003, 30, 54–58. [Google Scholar] [CrossRef]
- Carrera, D.; Ulloa, J.G. Iodinated contrast-induced Stevens-Johnson syndrome: A report of a rare complication for a common imaging agent. J. Vasc. Surg. Cases Innov. Tech. 2022, 8, 455–457. [Google Scholar] [CrossRef] [PubMed]
- Case, R.A.; Patel, V.; Mejia-Otero, C.; Siroy, A.E.; Patel, D.C. Anti-MDA-5 dermatomyositis with development of drug-mediated necrolytic skin lesions. J. Investig. Med. High Impact Case Rep. 2020, 8, 2324709620940496. [Google Scholar] [CrossRef] [PubMed]
- Honma, M.; Tobisawa, S.; Iinuma, S.; Shibuya, T.; Komatsu, S.; Takahashi, I.; Ishida-Yamamoto, A.; Iizuka, H. Toxic epidermal necrolysis with prominent facial pustules: A case with reactivation of human herpesvirus 7. Dermatology 2010, 221, 306–308. [Google Scholar] [CrossRef]
- Ikeda, M.; Fujita, T.; Amoh, Y.; Mii, S.; Matsumoto, K.; Iwamura, M. Stevens-Johnson syndrome induced by sorafenib for metastatic renal cell carcinoma. Urol. Int. 2013, 91, 482–483. [Google Scholar] [CrossRef]
- Kong, J.; Cuevas-Castillo, F.; Nassar, M.; Lei, C.M.; Idrees, Z.; Fix, W.C.; Halverstam, C.; Mir, A.; Elbendary, A.; Mathew, A. Bullous drug eruption after second dose of mRNA-1273 (Moderna) COVID-19 vaccine: Case report. J. Infect. Public Health 2021, 14, 1392–1394. [Google Scholar] [CrossRef]
- Mazumdar, G.; Shome, K. Stevens-Johnson syndrome following use of metronidazole in a dental patient. Indian J. Pharmacol. 2014, 46, 121–122. [Google Scholar] [CrossRef] [PubMed]
- Mohammadzadeh, I.; Khaledi, M.; Rezaei, N. Stevens-Johnson syndrome: Report on a case with a strange complaint of allergy to bus. Acta. Clin. Croat. 2013, 52, 391–394. [Google Scholar]
- Sood, M.; O Oyibo, S.; Rajkanna, J. A case of toxic epidermal necrolysis caused by the use of trimethoprim alone. Cureus 2021, 13, e14783. [Google Scholar] [CrossRef]
- Umebayashi, Y.; Enomoto, H.; Ogasawara, M. Drug eruption due to peplomycin: An unusual form of Stevens-Johnson syndrome with pustules. J. Dermatol. 2004, 31, 802–805. [Google Scholar] [CrossRef]
- Wang, Y.-S.; Tay, Y.-K.; Kwok, C. Toxic epidermal necrolysis caused by alfuzosin, an α1-adrenoceptor antagonist. Arch. Dermatol. 2006, 142, 927–947. [Google Scholar] [CrossRef]
- Marlowe, E.; Palmer, R.; Rahrig, A.L.; Dinora, D.; Harrison, J.; Skiles, J.; Rahim, M.Q. Case report: Toxic epidermal necrolysis as a unique presentation of acute graft versus host disease in a pediatric patient. Front. Immunol. 2025, 15, 1452245. [Google Scholar] [CrossRef]
- Kwong, E.Y.L.; Kuok, M.C.I.; Lam, K.F.; Chan, W.K.Y. Case report: Multi-targeted therapy in the treatment of severe toxic epidermal necrolysis. Front. Pediatr. 2024, 12, 1460579. [Google Scholar] [CrossRef]
- Li, N.; Li, J. Coronavirus-disease-2019-associated Stevens–Johnsons syndrome in a 15-year-old boy: A case report and review of the literature. J. Med. Case Rep. 2024, 18, 493. [Google Scholar] [CrossRef]
- Samar, A.; Sahana, G.; Deepak, P.; Nagaral, J.V.; Saranyaa, M. Drug-induced Stevens–Johnson syndrome in post traumatic facial injury. Indian J. Pharmacol. 2024, 56, 58–60. [Google Scholar] [CrossRef]
- Heuer, R.; Paulmann, M.; Annecke, T.; Behr, B.; Boch, K.; Boos, A.M.; Brockow, K.; French, L.E.; Gille, J.; Gundlach, V.; et al. S3 guideline: Diagnosis and treatment of epidermal necrolysis (Stevens-Johnson syndrome and toxic epidermal necrolysis)—Part 1: Diagnosis, initial management, and immunomodulating systemic therapy. J. Der Dtsch. Dermatol. Ges. 2024, 22, 1448–1466. [Google Scholar] [CrossRef] [PubMed]
- Walicka, M.; Majsterek, M.; Rakowska, A.; Słowińska, M.; Sicińska, J.; Góralska, B.; Ptasińska, M.; Rudnicka, L.; Marcinowska-Suchowierska, E. Mycoplasma pneumoniae-induced pneumonia with Stevens-Johnson syndrome of acute atypical course. Pol. Arch. Intern. Med. 2008, 118, 449–453. [Google Scholar] [CrossRef]
- Lipozencic, J.; Milavec-Puretic, V.; Kotrulja, L.; Tomicic, H.; Buzina, D.Š. Toxic epidermal necrolysis due to cotrimoxazole. J. Eur. Acad. Dermatol. Venereol. 2002, 16, 182–183. [Google Scholar] [CrossRef] [PubMed]
- Creamer, D.; Walsh, S.A.; Dziewulski, P.; Exton, L.S.; Lee, H.Y.; Dart, J.K.G.; Setterfield, J.; Bunker, C.B.; Ardern-Jones, M.R.; Watson, K.M.T.; et al. U.K. guidelines for the management of Stevens–Johnson syndrome/toxic epidermal necrolysis in adults 2016. Br. J. Dermatol. 2016, 174, 1194–1227. [Google Scholar] [CrossRef] [PubMed]
- Rajaratnam, R.; Mann, C.; Balasubramaniam, P.; Marsden, J.R.; Taibjee, S.M.; Shah, F.; Lim, R.; Papini, R.; Moiemen, N.; Lewis, H. Toxic epidermal necrolysis: Retrospective analysis of 21 consecutive cases managed at a tertiary centre. Clin. Exp. Dermatol. 2010, 35, 853–862. [Google Scholar] [CrossRef]
- Buenrostro-Rubio, I.; Silva-Villaseñor, J.A.; Hatami-Blechner, A.W.; Salazar-Del Valle, J.J.; Vázquez-Cárdenas, N.A.; Bustamante-Montes, L.P.; González-Alvarez, R. Allopurinol-induced toxic epidermal necrolysis. Drug Saf.-Case Rep. 2019, 6, 8. [Google Scholar] [CrossRef]
- Mehta, H.; Mete, U.K.; Gupta, P.; Ranjan, K.R.; Saikia, U.N.; Mahajan, R. Toxic epidermal necrolysis-like presentation of toxic erythema of chemotherapy. Clin. Exp. Dermatol. 2022, 47, 1201–1203. [Google Scholar] [CrossRef]
- Stătescu, L.; Constantin, M.; Morariu, H.S.; Solovăstru, L.G. Toxic epidermal necrolysis—A case report. J. Crit. Care Med. 2017, 3, 29–33. [Google Scholar] [CrossRef]
- Bouziri, A.; Khaldi, A.; Hamdi, A.; Borgi, A.; Ghorbel, S.; Kharfi, M.; Hadj, S.B.; Menif, K.; Ben Jaballah, N. Toxic epidermal necrolysis complicated by small bowel intussusception: A case report. J. Pediatr. Surg. 2011, 46, e9–e11. [Google Scholar] [CrossRef]
- Yachoui, R.; Kolasinski, S.L.; Feinstein, D.E. Mycoplasma pneumoniae with atypical stevens-Johnson syndrome: A diagnostic challenge. Case Rep. Infect. Dis. 2013, 2013, 457161. [Google Scholar] [CrossRef] [PubMed]
- Gallegos, J.A.O.; Amar, S. Stevens-Johnson syndrome/toxic epidermal necrolysis overlap caused by enzalutamide in patient with metastatic prostate cancer. BMJ Case Rep. 2021, 14, e242319. [Google Scholar] [CrossRef]
- Lyon, D.R.; Akinboboye, O.; Jain, P.V.; Jha, P. A Case of drug-induced Stevens-Johnson syndrome. Wis. Med. J. 2022, 121, E63–E65. [Google Scholar] [PubMed]
- Endo, Y.; Oka, A.; Uehara, A.; Toki, S.; Motegi, S.; Ishikawa, O.; Miyazawa, Y.; Suzuki, K. Fatal case of toxic epidermal necrolysis due to apalutamide used as a novel prostate cancer drug. J. Dermatol. 2020, 47, E359–E360. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Hama, N.; Nakamura, R.; Yamaguchi, S.; Suzuki, K.; Komeyama, T.; Yokoyama, A.; Abe, R. A fatal case of enfortumab vedotin-caused cutaneous adverse reaction mimicking toxic epidermal necrolysis with myelosuppression: Distinct clinical and histopathological features. J. Dermatol. 2025, 1–3. [Google Scholar] [CrossRef]
- Van Batavia, J.P.; Chu, D.I.; Long, C.J.; Jen, M.; Canning, D.A.; Weiss, D.A. Genitourinary involvement and management in children with Stevens–Johnson syndrome and toxic epidermal necrolysis. J. Pediatr. Urol. 2017, 13, 490.e1–490.e7. [Google Scholar] [CrossRef]
- Paulmann, M.; Heuer, R.; Annecke, T.; Behr, B.; Boch, K.; Boos, A.M.; Brockow, K.; French, L.E.; Gille, J.; Gundlach, V.; et al. S3 guideline: Diagnosis and treatment of epidermal necrolysis (Stevens-Johnson syndrome and toxic epidermal necrolysis)—Part 2: Supportive therapy of EN in the acute and post-acute stages. J. der Dtsch. Dermatol. Ges. 2024, 22, 1576–1593. [Google Scholar] [CrossRef]
- Lee, H.; Walsh, S.; Creamer, D. Long-term complications of Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN): The spectrum of chronic problems in patients who survive an episode of SJS/TEN necessitates multidisciplinary follow-up. Br. J. Dermatol. 2017, 177, 924–935. [Google Scholar] [CrossRef]
- Wang, Q.; Cao, H.; Zhang, X.; Wu, H.; Tang, Z. Case report: Apalutamide-induced severe lethal cutaneous adverse effects in China. Front. Immunol. 2024, 14, 1291564. [Google Scholar] [CrossRef] [PubMed]
- Kuang, H.; Huang, D.; Hu, C.; Gong, L.; Yu, Z.; Zhu, X.; Lan, H.; Huang, G. Stevens–Johnson syndrome induced by sintilimab: A case report and literature review. Front. Oncol. 2025, 15, 1568316. [Google Scholar] [CrossRef]
- Bang, D.; Shah, T.; Thakker, D.; Shah, Y.; Raval, A.D. Drug-induced Stevens–Johnson syndrome: Case series from tertiary care centre in Gujarat. Pharmacoepidemiol. Drug Saf. 2012, 21, 384–395. [Google Scholar] [CrossRef] [PubMed]
- Ingen-Housz-Oro, S.; Schmidt, V.; Ameri, M.M.; Abe, R.; Brassard, A.; Mostaghimi, A.; Paller, A.S.; Romano, A.; Didona, B.; Kaffenberger, B.H.; et al. Post-acute phase and sequelae management of epidermal necrolysis: An international, multidisciplinary DELPHI-based consensus. Orphanet J. Rare Dis. 2023, 18, 33. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, M.P.; Carvalho, C.H.C.; Santi, C.G.; Avancini, J. Stevens-Johnson syndrome and toxic epidermal necrolysis—retrospective review of cases in a high complexity hospital in Brazil. Int. J. Dermatol. 2020, 59, 191–196. [Google Scholar] [CrossRef]
- Boyraz, G.; Basaran, D.; Salman, M.C.; Ozgul, N.; Yuce, K. Vaginal reconstruction for vaginal obliteration secondary to stevens Johnson syndrome: A case report and review of literature. Oman Med. J. 2017, 32, 436–439. [Google Scholar] [CrossRef]
- Yang, M.-S.; Lee, J.Y.; Kim, J.; Kim, G.-W.; Kim, B.-K.; Kim, J.-Y.; Park, H.-W.; Cho, S.-H.; Min, K.-U.; Kang, H.-R. Incidence of Stevens-Johnson syndrome and toxic epidermal necrolysis: A nationwide population-based study using National Health Insurance Database in Korea. PLoS ONE 2016, 11, e0165933. [Google Scholar] [CrossRef]
- Blum, L.; Chosidow, O.; Rostoker, G.; Philippon, C.; Revuz, J.; Roujeau, J.-C. Renal involvement in toxic epidermal necrolysis. J. Am. Acad. Dermatol. 1996, 34, 1088–1090. [Google Scholar] [CrossRef]
- Hu, L.; Lu, X.; Xia, J.; Hou, M. A case of Stevens–Johnson syndrome with gross hematuria. Int. J. Women’s Dermatol. 2016, 2, 65–66. [Google Scholar] [CrossRef]
- Baccaro, L.M.; Sakharpe, A.; Miller, A.; Amani, H. The first reported case of ureteral perforation in a patient with severe toxic epidermal necrolysis syndrome. J. Burn. Care Res. 2014, 35, e265–e268. [Google Scholar] [CrossRef]
- Wang, C.; Huang, Y. Case Report: Hydronephrosis and nephrogenic adenoma secondary to Stevens-Johnson syndrome. Front. Pharmacol. 2025, 16, 1624432. [Google Scholar] [CrossRef]
- Bruce-Hickman, D.; Jiang, X.; Thia, J.J.-P.; Kansal, A. Stevens-Johnson syndrome complicated by obstructive uropathy, pneumothorax, and pneumomediastinum: A case report and literature review. Burn. Trauma 2019, 7, 14. [Google Scholar] [CrossRef]
- Niemeijer, I.C.; van Praag, M.C.G.; van Gemund, N. Relevance and consequences of erythema multiforme, Stevens-Johnson syndrome and toxic epidermal necrolysis in gynecology. Arch. Gynecol. Obstet. 2009, 280, 851–854. [Google Scholar] [CrossRef] [PubMed]
- Gleghorn, K.L.; Voigt, C.; Kelly, B. Toxic epidermal necrolysis and Stevens-Johnson syndrome/toxic epidermal necrolysis overlap in pediatric patients with a focus on newer antiepileptic drugs: A 25-year retrospective study at a single tertiary care center. Pediatr. Dermatol. 2021, 38, 812–818. [Google Scholar] [CrossRef]
- Donta-Bakoyianni, K.; Mitsea, A.G.; Deodoropoulou-Papadimitriou, K. Stevens-Johnson syndrome: Case presentation. J. Clin. Pediatr. Dent. 2002, 27, 71–76. [Google Scholar] [PubMed]
- Kumar, H.; Kumar, A.; Kumar, S. Severe cutaneous adverse reaction to piperacillin-tazobactam: A case of Stevens-Johnson syndrome. Cureus 2023, 15, e42839. [Google Scholar] [CrossRef] [PubMed]
- Baroni, A.; Ruocco, E. Lyell’s syndrome. Skinmed 2005, 4, 221–225. [Google Scholar] [CrossRef]
- Sanmarkan, A.D.; Sori, T.; Thappa, D.M.; Jaisankar, T.J. Retrospective analysis of Stevens-Johnson syndrome and toxic epidermal necrolysis over a period of 10 years. Indian J. Dermatol. 2011, 56, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Weisz, L.M.C.; Escuredo, I.M.; Soto, J.B.A.; Gutiérrez, J.J.G. Toxic epidermal necrolysis (TEN): Acute complications and long-term sequelae management in a multidisciplinary follow-up. J. Plast. Reconstr. Aesthet. Surg. 2020, 73, 319–327. [Google Scholar] [CrossRef]
- Letko, E.; Papaliodis, D.N.; Papaliodis, G.N.; Daoud, Y.J.; Ahmed, A.R.; Foster, C.S. Stevens-Johnson syndrome and toxic epidermal necrolysis: A review of the literature. Ann. Allergy, Asthma Immunol. 2005, 94, 419–436. [Google Scholar] [CrossRef]
- Heimbach, D.M.; Engrav, L.H.; Marvin, J.A.; Harnar, T.J.; Grube, B.J. Toxic epidermal necrolysis. A step forward in treatment. JAMA 1987, 257, 2171–2175. [Google Scholar] [CrossRef] [PubMed]
- Belver, M.T.; Michavila, A.; Bobolea, I.; Feito, M.; Bellón, T.; Quirce, S. Severe delayed skin reactions related to drugs in the paediatric age group: A review of the subject by way of three cases (Stevens–Johnson syndrome, toxic epidermal necrolysis and DRESS). Allergol. et Immunopathol. 2016, 44, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Okan, G.; Yaylaci, S.; Peker, O.; Kaymakoglu, S.; Saruc, M. Vanishing bile duct and Stevens-Johnson syndrome associated with ciprofloxacin treated with tacrolimus. World J. Gastroenterol. 2008, 14, 4697–4700. [Google Scholar] [CrossRef]
- Shahraki, T.; Hassanpour, K.; Arabi, A.; Ansari, I.; Sadoughi, M.-M. Corona virus disease 2019-associated Stevens-Johnson syndrome: A case report. BMC Ophthalmol. 2021, 21, 274. [Google Scholar] [CrossRef]
- Hasbini, J.; Safawi, N.; Berjaoui, C.; Rajab, M.; Naous, A. Mycoplasma pneumonia complicated by Stevens-Johnson syndrome: A case report. Respir. Med. Case Rep. 2024, 51, 102081. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.R.; Jaiswal, H.; Rathod, M.M.; Vijayvargi, S. Ofloxacin, paracetamol and cefixime induced Stevens-Johnson syndrome—Toxic epidermal necrolysis in an adult female patient: A case report. J. Pharm. Health Care Sci. 2025, 11, 98. [Google Scholar] [CrossRef]
- Peters, W.; Zaidi, J.; Douglas, L. Toxic epidermal necrolysis: A burn-centre challenge. CMAJ. 1991, 144, 1477–1480. [Google Scholar]
- Patel, T.K.; Barvaliya, M.J.; Sharma, D.; Tripathi, C. A systematic review of the drug-induced Stevens-Johnson syndrome and toxic epidermal necrolysis in Indian population. Indian J. Dermatol. Venereol. Leprol. 2013, 79, 389–398. [Google Scholar] [CrossRef]
- Sharma, V.; Sethuraman, G.; Minz, A. Stevens Johnson syndrome, toxic epidermal necrolysis and SJS-TEN overlap: A retrospective study of causative drugs and clinical outcome. Indian J. Dermatol. Venereol. Leprol. 2008, 74, 238–240. [Google Scholar] [CrossRef]
- Lipový, B.; Holoubek, J.; Hanslianová, M.; Cvanová, M.; Klein, L.; Grossová, I.; Zajíček, R.; Bukovčan, P.; Koller, J.; Baran, M.; et al. Toxic epidermal necrolysis data from the CELESTE multinational registry. Part I: Epidemiology and general microbiological characteristics. Burns 2018, 44, 1551–1560. [Google Scholar] [CrossRef]
- Engelhardt, S.L.; Schurr, M.J.; Helgerson, R.B. Toxic epidermal necrolysis: An analysis of referral patterns and steroid usage. J. Burn Care Rehab. 1997, 18, 520–524. [Google Scholar] [CrossRef]
- Stewart, T.J.; Chan, C.-W.J.; Shah, H.; Frew, J. Infectious complications of Stevens-Johnson syndrome and toxic epidermal necrolysis: A systematic review and meta-analysis. Int. J. Dermatol. 2025, 64, 830–848. [Google Scholar] [CrossRef] [PubMed]
- Dore, J.; Salisbury, R.E. Morbidity and mortality of mucocutaneous diseases in the pediatric population at a tertiary care center. J. Burn. Care Res. 2007, 28, 865–870. [Google Scholar] [CrossRef]
- de Jesus, L.E.; Dekermacher, S.; Manhães, C.R.; Faria, L.M.; Barros, M.L. Acquired labial sinechiae and hydrocolpos secondary to Stevens-Johnson syndrome. Urology 2012, 80, 919–921. [Google Scholar] [CrossRef]
- Olteanu, C.; Shear, N.H.; Burnett, M.; Hashimoto, R.; Jeschke, M.G.; Ziv, M.; Dodiuk-Gad, R.P. Retrospective study of patients with SJS/TEN treated at a tertiary burn unit in Canada: Overview of 17 years of treatment. J. Cutan. Med. Surg. 2021, 25, 271–280. [Google Scholar] [CrossRef]
- Saeed, H.; Mantagos, I.S.; Chodosh, J. Complications of Stevens–Johnson syndrome beyond the eye and skin. Burns 2016, 42, 20–27. [Google Scholar] [CrossRef]
- Holm, S.; Thurfjell, V.; Lara-Valencia, P.; Huss, F. Purpura fulminans, TEN, and disseminated herpes simplex: An unexpected combination. Clin. Case Rep. 2022, 10, e05784. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Mei, X.-L. Retrospective Analysis of Stevens-Johnson syndrome and toxic epidermal necrolysis in 88 Chinese patients. Chin. Med. J. 2017, 130, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
- Yamane, Y.; Matsukura, S.; Watanabe, Y.; Yamaguchi, Y.; Nakamura, K.; Kambara, T.; Ikezawa, Z.; Aihara, M. Retrospective analysis of Stevens–Johnson syndrome and toxic epidermal necrolysis in 87 Japanese patients—Treatment and outcome. Allergol. Int. 2016, 65, 74–81. [Google Scholar] [CrossRef]
- Shanbhag, S.S.; Chodosh, J.; Fathy, C.; Goverman, J.; Mitchell, C.; Saeed, H.N. Multidisciplinary care in Stevens-Johnson syndrome. Ther. Adv. Chronic Dis. 2020, 11, 2040622319894469. [Google Scholar] [CrossRef]
- Theva, D.P.; Li, L.; Ng, L. Urologic presentation of Stevens-Johnson syndrome: A case report. Int. J. Exp. Clin. Res. 2017, 130. [Google Scholar] [CrossRef]
- Estrella-Alonso, A.; Aramburu, J.A.; González-Ruiz, M.Y.; Cachafeiro, L.; Sánchez, M.S.; Lorente, J.A. Toxic epidermal necrolysis: A paradigm of critical illness. Rev. Bras. De Ter. Intensiv. 2017, 29, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Lerma, V.; Macías, M.; Toro, R.; Moscoso, A.; Alonso, Y.; Hernández, O.; de Abajo, F. Care in patients with epidermal necrolysis in burn units. A nursing perspective. Burns 2018, 44, 1962–1972. [Google Scholar] [CrossRef]
- Magone, M.T.; Maiberger, M.; Clayton, J.; Pasieka, H. Vulvovaginal and ocular involvement and treatment in female patients with Stevens–Johnson syndrome and toxic epidermal necrolysis: A review. Int. J. Women’s Dermatol. 2021, 7, 520–528. [Google Scholar] [CrossRef]
- Petukhova, T.A.; Maverakis, E.; Ho, B.; Sharon, V.R. Urogynecologic complications in Stevens-Johnson syndrome and toxic epidermal necrolysis: Presentation of a case and recommendations for management. JAAD Case Rep. 2016, 2, 202–205. [Google Scholar] [CrossRef] [PubMed][Green Version]
- SBI Practice Guidelines Committee; Advisory Subcommittee; Steering Subcommittee. ISBI practice guidelines for burn care, Part 2. Burns 2018, 44, 1617–1706. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Published by MDPI on behalf of the Société Internationale d’Urologie. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.