Entomological Survey and Leishmania (Leishmania) mexicana Prevalence in Sand Fly Species during an Outbreak of Cutaneous Leishmaniasis in Quintana Roo State, Mexico
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Al-Kamel, M.A.N. Leishmaniasis and malignancy: A review and perspective. Clin. Skin Cancer 2017, 2, 54–58. [Google Scholar] [CrossRef][Green Version]
- Costa, W.A.; Souza, N.A. Sand flies: Medical importance. In Brazilian Sand Flies: Biology, Taxonomy, Medical Importance and Control; Springer: Cham, Switzerland, 2018; pp. 1–8. [Google Scholar]
- Alvar, J.; Vélez, I.D.; Bern, C.; Herrero, M.; Desjeux, P.; Cano, J.; Jannin, J.; den Boer, M.; Who Leishmaniasis Control the WHO Leishmaniasis Control Team. Leishmaniasis worldwide and global estimates of its incidence. PLoS ONE 2012, 7, e35671. [Google Scholar] [CrossRef] [PubMed]
- Pan American Health Organization/World Health Organization. Washington D.C Report No.: 7. 2019. Available online: http://iris.paho.org/xmlui/bitstream/handle/123456789/50505/Leishreport2019_eng.pdf?ua=1 (accessed on 25 July 2023).
- Herrera, G.; Barragán, N.; Luna, N.; Martínez, D.; De Martino, F.; Medina, J.; Niño, S.; Páez, L.; Ramírez, A.; Vega, L.; et al. An interactive database of Leishmania species distribution in the Americas. Sci. Data 2020, 7, 110. [Google Scholar] [CrossRef] [PubMed]
- Mikery-Pacheco, O.A.; Castillo-Vera, A. Leishmaniasis. In La Frontera Sur de México, ¿Una Salud en Crisis? López, H.O.D., Ed.; Academia Nacional de Medicina de México: Mexico City, Mexico, 2008; pp. 163–173. [Google Scholar]
- Centro Nacional de Programas Preventivos y Control de Enfermedades (CENAPRECE). Anuarios de Morbilidad Durante el Periodo 1995–2021. In Anuarios de Morbilidad, 1984–2021. 2023. Available online: http://www.epidemiologia.salud.gob.mx/anuario/html/anuarios.html (accessed on 8 August 2023).
- Pech-May, A.; Escobedo-Ortegón, F.J.; Berzunza-Cruz, M.; Rebollar-Téllez, E.A. Incrimination of four sandfly species previously unrecognized as vectors of Leishmania parasites in Mexico. Med. Vet. Entomol. 2010, 24, 150–161. [Google Scholar] [CrossRef]
- Pech-May, A.; Peraza-Herrera, G.; Moo-Llanes, D.A.; Escobedo-Ortegon, J.; Berzunza-Cruz, M.; Becker-Fauser, I.; Montes de Oca-Aguilar, A.; Rebollar-Téllez, E.A. Assessing the importance of four sandfly species (Diptera: Psychodidae) as vectors of Leishmania. Med. Vet. Entomol. 2016, 30, 310–320. [Google Scholar] [PubMed]
- Health Services of the State of Quintana Roo. Boletín Epidemiológico Estatal. Available online: https://qroo.gob.mx/sesa/dss/sve (accessed on 8 August 2023).
- Biagi, F.F.; de Biagi, A.M.; Beltrán, F.H. Phlebotomus flaviscutellatus, transmisor natural de Leishmania mexicana. Prensa Med. Mex. 1965, 30, 267–272. [Google Scholar]
- Sánchez-García, L.; Berzunza-Cruz, M.; Becker-Fauser, I.; Rebollar-Téllez, E.A. Sand flies naturally infected by Leishmania (L.) mexicana in the peri-urban area of Chetumal city, Quintana Roo, México. Trans. R. Soc. Trop. Med. Hyg. 2010, 104, 406–411. [Google Scholar] [CrossRef]
- Castrejón, O.V.; Sánchez, B.R.; Saldaña, A.M.; Hobart, O. Leishmaniasis cutánea de perros en México. Enferm. Infecc. Microbiol. Clin. 2009, 29, 135–140. [Google Scholar]
- Longoni, S.S.; Marín, C.; Sauri-Arceo, C.H.; López-Cespedes, A.; Rodríguez-Vivas, R.I.; Villegas, N.; Sánchez-Moreno, M. An iron-superoxide dismutase antigen-based serological screening of dogs indicates their potential role in the transmission of cutaneous leishmaniasis and trypanosomiasis in Yucatan, Mexico. Vector Borne Zoonotic Dis. 2011, 11, 815–821. [Google Scholar] [CrossRef]
- Zarza, H.; Arias-Alzate, A.; González-Maya, J.F.; Chávez, C.; Ceballos, G. First record of Leishmaniasis in wild Jaguars (Panthera onca) from Mexico. Mammal. Notes 2015, 2, 11–12. [Google Scholar] [CrossRef]
- Salazar Grosskelwing, E.; Bolio González, M.E.; Rodríguez Vivas, R.I.; Miró Corrales, G.; Escobedo Ortegón, F.J.; Ruíz Piña, H.A.; Loría-Cervera, E.N. Frecuencia serológica y molecular de Leishmania spp y Trypanosoma cruzi y factores asociados en perros con signos sugestivos de infección por tripanosomátidos en Quintana Roo, México. Rev. Investig. Vet. Perú 2020, 31, e17336. [Google Scholar] [CrossRef]
- May-Uc, E.; Hernández-Arana, H.A.; Rebollar-Téllez, E. Distribución de Flebotominos (Dipetra Psychodidae) en Quintana Roo, Mexico. Acta Zool. Mex. 2011, 27, 273–289. [Google Scholar] [CrossRef][Green Version]
- Rodríguez-Rojas, J.J.; Rebollar-Téllez, E.A. Effect of trapping methods on the estimation of alpha diversity of a phlebotomine sandfly assemblage in southern Mexico. Med. Vet. Entomol. 2017, 31, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez-Bernal, S.; Rebollar-Téllez, E. Estado actual del conocimiento de los hábitos hematófagos de los Phlebotominae (Diptera: Psychodidae) de México y sus implicaciones zoonóticas. Rev. Biomed. 2022, 34, 32–43. [Google Scholar] [CrossRef]
- Arjona-Jiménez, G.; Villegas, N.; López-Céspedes, Á.; Marín, C.; Longoni, S.S.; Bolio-González, M.E.; Rodríguez-Vivas, R.I.; Sauri-Arceo, C.H.; Sánchez-Moreno, M. Prevalence of antibodies against three species of Leishmania (L. mexicana, L. braziliensis, L. infantum) and possible associated factors in dogs from Mérida, Yucatán, Mexico. Trans. R. Soc. Trop. Med. Hyg. 2012, 106, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Shaw, J.J. New World leishmaniasis: The ecology of leishmaniasis and the diversity of leishmanial species in Central and South America. In World Class Parasites: Leishmania; Farrell, J., Ed.; Kluwer Academic Publishers: London, UK; Boston, MA, USA, 2002; pp. 11–31. [Google Scholar]
- Pérez, J.E.; Villaseca, P.; Llanos-Cuentas, A.; Campos, M.; Guerra, H. Técnicas para colectar “titiras” (Lutzomyia spp., Diptera: Psychodidae) en ambientes alto andinos peruanos. Rev. Peru Entomol. 1987, 30, 77–80. [Google Scholar]
- Ibáñez-Bernal, S. Phlebotominae (Diptera: Psychodidae) de México. VI. Clave ilustrada para la identificación de las hembras de Lutzomyia Franca. Folia Entomol. Mex. 2005, 44, 195–212. [Google Scholar]
- Lozano-Sardaneta, Y.N.; Marina, C.F.; Torres-Monzón, J.A.; Sánchez-Cordero, V.; Becker, I. Molecular detection of Wolbachia and Bartonella as part of the microbiome of phlebotomine sand flies from Chiapas, Mexico. Parasitol. Res 2023, 122, 1293–1301. [Google Scholar] [CrossRef]
- Young, D.G.; Duncan, M.A. Guide to the identification and geographic distribution of Lutzomyia sand flies in Mexico, the West Indies, Central and South America (Diptera: Psychodidae). Mem. Am. Entomol. Inst. 1994, 54, 881. [Google Scholar]
- Ibáñez-Bernal, S. Phlebotominae (Diptera: Psychodidae) de México. V. Clave ilustrada para la identificación de los machos de Lutzomyia Franca. Folia Entomol. Mex. 2005, 44, 49–66. [Google Scholar]
- García-González, L.A.; Rodrigo-Tapia, J.P.; Sánchez-Lazo, P.; Ramos, S.; Suárez-Nieto, C. DNA extraction using Chelex resin for oncogenic amplification analysis in head and neck tumours. Acta Otorrinolaringol. Esp. 2004, 55, 139–144. [Google Scholar] [PubMed]
- El Tai, N.O.; El Fari, M.; Mauricio, I.; Miles, M.A.; Oskam, L.; El Safi, S.H.; Presber, W.H.; Schönian, G. Leishmania donovani: Intraspecific polymorphisms of Sudanese isolates revealed by PCR-based analyses and DNA sequencing. Exp. Parasitol. 2001, 97, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Librado, P.; Rozas, J. DnaSP v5: A software for comprehensive analysis of DNA polymorphism data. Bioinformatics 2009, 25, 1451–1452. [Google Scholar] [CrossRef]
- Canto-Lara, S.B.; Bote-Sánchez, M.D.; Rebollar-Téllez, E.A.; Andrade-Narváez, F.J. Detection and identification of Leishmania kDNA in Lutzomyia olmeca olmeca and Lutzomyia cruciata (Diptera: Psychodidae) by polymerase chain reaction in southern Mexico. Entomol. News 2007, 118, 217–222. [Google Scholar]
- Pérez-Blas, L.G.; Chiyean-Acosta, G.; Canché-Pool, E.B.; Tello-Martín, R.; Torres-Castro, J.R.; Ruiz-Piña, H.A.; Flores-Mejía, R.; Cortés, O.R.; Reyes-Novelo, E. Molecular Detection of Leishmania (Leishmania) mexicana in Sandflies from the State of Yucatan, Mexico. Vector Borne Zoonotic Dis. 2022, 22, 589–595. [Google Scholar] [CrossRef] [PubMed]
- González, C.; Rebollar-Téllez, E.A.; Ibáñez-Bernal, S.; Becker-Fauser, I.; Martínez-Meyer, E.; Peterson, A.T.; Sánchez-Cordero, V. Current knowledge of Leishmania vectors in Mexico: How geographic distributions of species relate to transmission areas. Am. J. Trop. Med. Hyg. 2011, 85, 839–846. [Google Scholar] [CrossRef] [PubMed]
- Moo-Llanes, D.; Ibarra-Cerdeña, C.N.; Rebollar-Téllez, E.A.; Ibañez-Bernal, S.; González, C.; Ramsey, J.M. Current and future niche of North and Central American sand flies (Diptera: Psychodidae) in climate change scenarios. PLoS Negl. Trop. Dis. 2013, 7, e2421. [Google Scholar] [CrossRef]
- Cruz-Ruíz, A.L.; García-Rejón, J.; Manrique-Saide, P.; Pérez-Motul, P. Taxonomical identification of anthropophilic species of Lutzomyia in Quintana Roo, Peninsula of Yucatan, Mexico. Rev. Biomed. 1994, 5, 127–131. [Google Scholar]
- Kocher, A.; Cornuault, J.; Gantier, J.; Manzi, S.; Chavy, A.; Girod, R.; Dusfour, I.; Forget, P.; Ginouves, M.; Prévot, G.; et al. Biodiversity and vector-borne diseases: Host dilution and vector amplification occur simultaneously for Amazonian leishmaniases. Mol. Ecol. 2023, 32, 1817–1831. [Google Scholar] [CrossRef]
- Williams, P. On the vertical distribution of phlebotomine sandflies (Dipt., Psychodidae) in British Honduras (Belize). Bull. Entomol. Res. 1970, 59, 637–646. [Google Scholar] [CrossRef]
- Christensen, H.A.; Fairchild, G.B.; Herrer, A.; Johnson, C.M.; Young, D.G.; Vásquez, A.M. The ecology of cutaneous leishmaniasis in the Republic of Panama. J. Med. Entomol. 1983, 20, 463–484. [Google Scholar] [CrossRef] [PubMed]
- Rebollar-Téllez, E.A.; Tun-Ku, E.; Manrique-Saide, P.C.; Andrade-Narvaez, F.J. Relative abundances of sandfly species (Diptera: Phlebotominae) in two villages in the same area of Campeche, in southern Mexico. Ann. Trop. Med. Parasitol. 2005, 99, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Tesh, R.B.; Chaniotis, B.N.; Aronson, M.D.; Johnson, K.M. Natural host preferences of Panamanian phlebotomine sandflies as determined by precipitin test. Am. J. Trop. Med. Hyg. 1971, 20, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Tesh, R.B.; Chaniotis, B.N.; Carrera, B.R.; Johnson, K.M. Further studies on the natural host preferences of Panamanian phlebotomine sandflies. Am. J. Epidemiol. 1972, 95, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Moo-Llanes, D.A.; Baak-Baak, C.M.; Cigarroa-Toledo, N.; Tzuc-Dzul, J.C.; Panti-May, J.A.; García-Rejón, J.E. Impacto Del Cambio Climático En La Distribución de Tres Roedores Endémicos de La Península de Yucatán: Implicaciones Para La Conservación de Otonyctomys hatti. Rev. Mex. Mastozool. Nueva Época 2021, 11, 1–14. [Google Scholar]
- Sosa-Bibiano, E.I.; Sánchez-Martínez, L.A.; López-Ávila, K.B.; Chablé-Santos, J.B.; Torres-Castro, J.R.; Fernández-Figueroa, E.A.; Rangel-Escareño, C.; Loría-Cervera, E.N. Leishmania (Leishmania) mexicana Infection in Wild Rodents from an Emergent Focus of Cutaneous Leishmaniasis in Yucatan, Mexico. J. Trop. Med. 2022, 2022, 8392005. [Google Scholar] [CrossRef]
- Rebollar-Téllez, E.A.; Moo-Llanes, D.A. Diversidad alfa, beta y co-ocurrencia de especies de flebotomineos (Diptera: Psychodidae) en Calakmul, Campeche, México. Rev. Chil. Entomol. 2020, 46, 221–235. [Google Scholar]
- Navarrete-Carballo, J.; Huerta-Jiménez, H.; Loría-Cervera, E.N.; Manrique-Saide, P.; Sosa-Bibiano, E.I. Phlebotomine sand flies (Diptera: Psychodidae) from an emergent focus of localized cutaneous leishmaniasis in Yucatan, Southeast Mexico. J. Vector Ecol. 2022, 47, 9–18. [Google Scholar] [CrossRef]
- de Oca-Aguilar, A.C.M.; Rebollar-Téllez, E.A.; Sosa-Bibiano, E.I.; López-Avila, K.B.; Torres-Castro, J.R.; Loría-Cervera, E.N. Effect of land use change on the phlebotomine sand fly assemblages in an emergent focus of cutaneous leishmaniasis in Yucatan, Mexico. Acta Trop. 2022, 235, 106628. [Google Scholar] [CrossRef]
- Pérez, J.; Virgen, A.; Rojas, J.C.; Rebollar-Téllez, E.A.; Alfredo, C.; Infante, F.; Mikery, O.; Marina, C.F.; Ibáñez-Bernal, S. Species composition and seasonal abundance of sandflies (Diptera: Psychodidae: Phlebotominae) in coffee agroecosystems. Mem. Inst. Oswaldo Cruz 2014, 109, 80–86. [Google Scholar] [CrossRef]
- Lozano-Sardaneta, Y.N.; Colunga-Salas, P.; Sánchez-Montes, S.; Cáceres, A.G.; Becker, I. First report of Bartonella sp. in sand flies (Diptera: Psychodidae: Phlebotominae) from southern Mexico. J. Am. Mosq. Control Assoc. 2019, 35, 224–227. [Google Scholar] [CrossRef] [PubMed]
- Lozano-Sardaneta, Y.N.; Paternina, L.E.; Sánchez-Montes, S.; Quintero, A.; Ibáñez-Bernal, S.; Sánchez-Cordero, V.; Bejarano, E.E.; Becker, I. DNA barcoding and fauna of phlebotomine sand flies (Diptera: Psychodidae: Phlebotominae) from Los Tuxtlas, Veracruz, Mexico. Acta Trop. 2020, 201, 105220. [Google Scholar] [CrossRef] [PubMed]
- Loría-Cervera, E.N.; Sosa-Bibiano, E.I.; Van Wynsberghe, N.R.; Torres-Castro, J.R.; Andrade-Narváez, F.J. Preliminary epidemiological findings of Leishmania infection in the municipality of Tinum, Yucatan State, Mexico. Parasite Epidemiol. Control 2019, 4, e00088. [Google Scholar] [CrossRef] [PubMed]
- Canché-Pool, E.B.; Canto-Hau, D.M.; Vargas-Meléndez, M.A.; Tello-Martín, R.; Reyes-Novelo, E.; Escobedo-Ortegón, F.J.; Ruiz-Piña, H.A.; Cambranes-Puc, L.H.; Torres-Castro, J.R.; Palacio-Vargas, J.A.; et al. Report of autochthonous cases of localized cutaneous leishmaniasis caused by Leishmania (Leishmania) mexicana in vulnerable, susceptible areas of Southeastern Mexico. Rev. Inst. Med. Trop. 2022, 64, e35. [Google Scholar] [CrossRef]
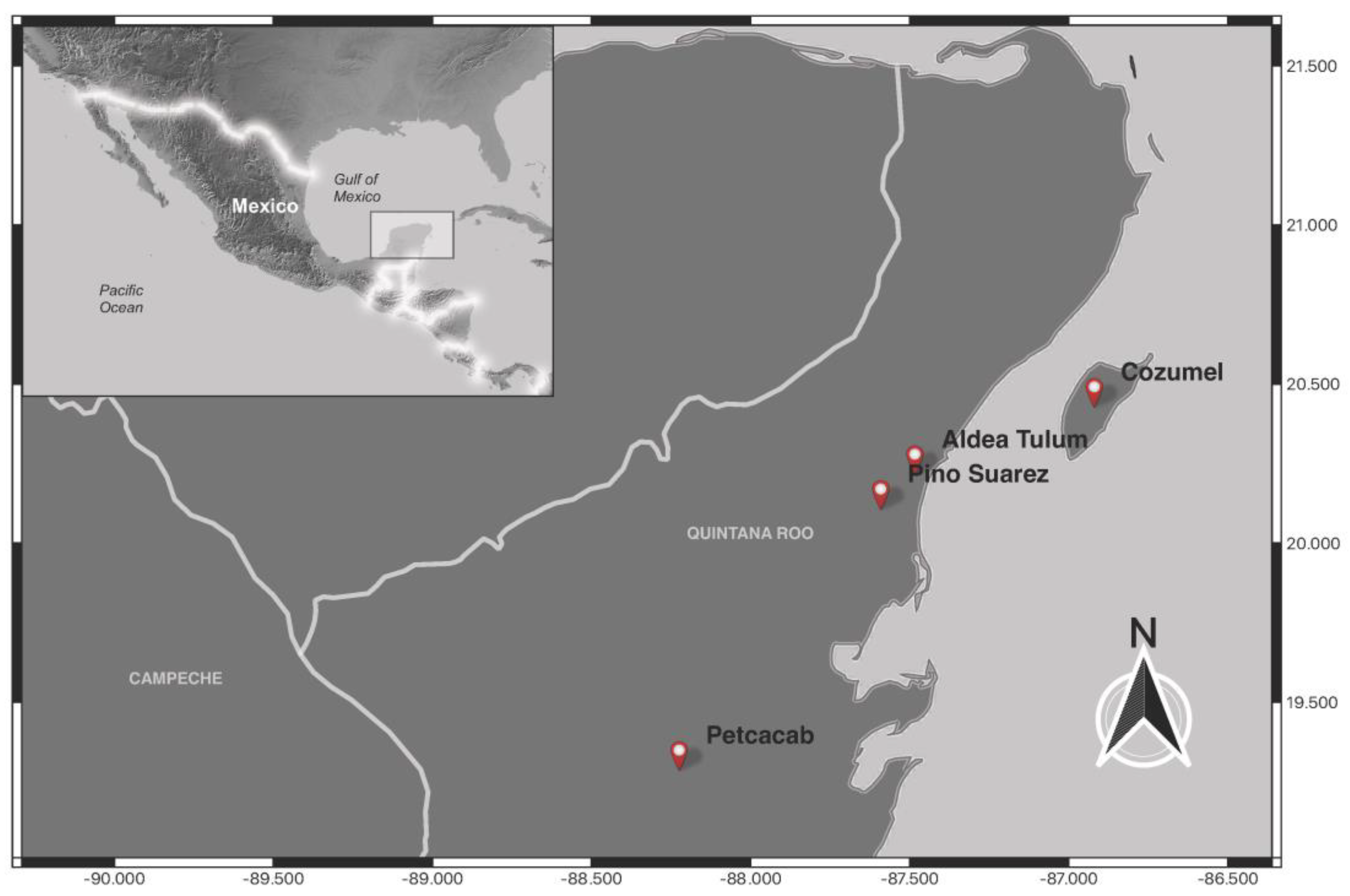
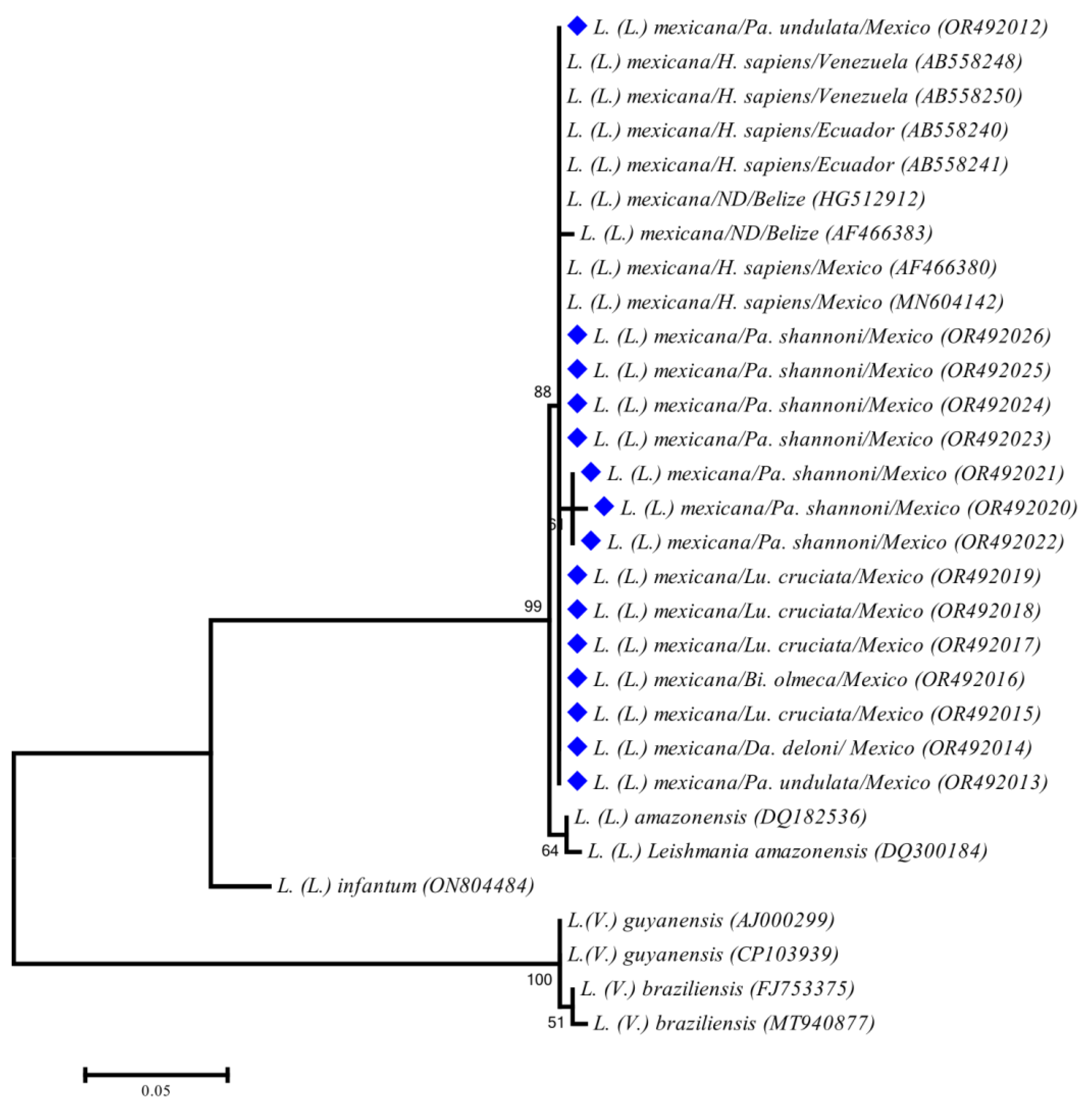
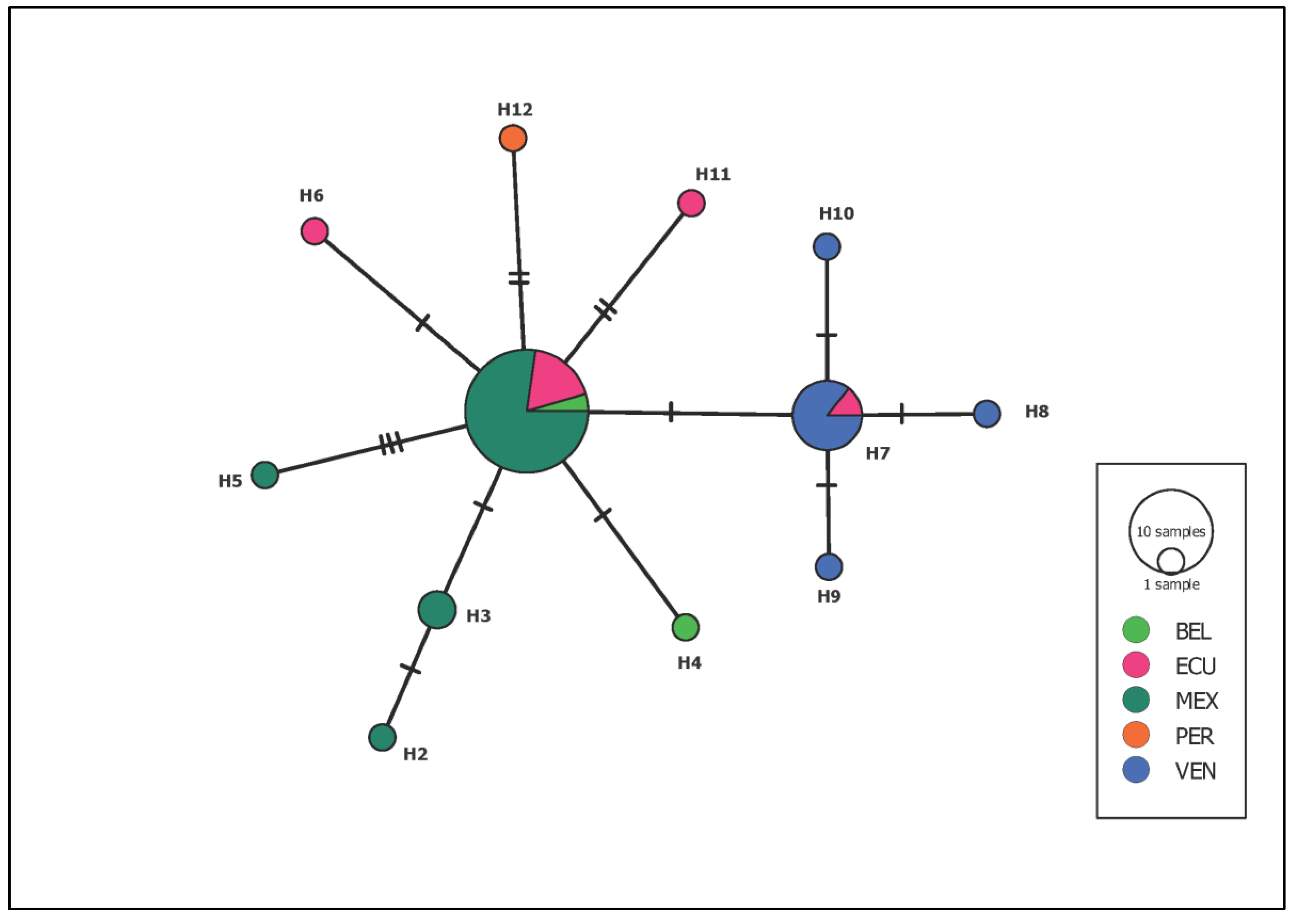
| Locality | Latitude | Longitude | Date | Species | n | SH | MLT | AES | Feeding | + | % |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Aldea Tulum | 20°13′11.246″ N | 87°29′2.018″ O | 12–13 April 2023 | Bichromomyia olmeca | 3 | 3 | 0 | 0 | 0 | 0 | 0 |
| Lutzomyia cruciata | 5 | 3 | 2 | 0 | 0 | 0 | 0 | ||||
| Psathyromyia shannoni | 5 | 3 | 2 | 0 | 0 | 0 | 0 | ||||
| Psathyromyia undulata | 1 | 1 | 0 | 0 | 0 | 0 | 0 | ||||
| Cozumel | 20°25′54.331″ N | 86°55′12.988″ O | 20 November 2023 | Psathyromyia undulata | 23 | 0 | 0 | 23 | 7 | 2 | 7.41 |
| Petcacab | 19°17′24.215″ N | 19°17′24.215″ O | 21–22 March 2023 | Dampfomyia deloni | 7 | 1 | 6 | 0 | 0 | 1 | 14.29 |
| Bichromomyia olmeca | 3 | 3 | 0 | 0 | 1 | 1 | 33.33 | ||||
| Lutzomyia cruciata | 22 | 11 | 11 | 0 | 0 | 4 | 18.18 | ||||
| Pintomyia serrana | 1 | 1 | 0 | 0 | 0 | 0 | 0 | ||||
| Psthyromyia shannoni | 152 | 84 | 68 | 0 | 8 | 17 | 11.18 | ||||
| Psathyromyia undulata | 1 | 0 | 1 | 0 | 1 | 0 | 0 | ||||
| Psychodopygus panamensis | 1 | 1 | 0 | 0 | 0 | 0 | 0 | ||||
| Pino Suárez | 20°6′37.427″ N | 87°35′28.346″ O | 13 April 2023 | Lutzomyia cruciata | 18 | 0 | 18 | 0 | 0 | 0 | 0 |
| Total | 242 | 111 | 108 | 23 | 17 | 25 | 10.33 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cañeda-Guzmán, I.C.; Oca-Aguilar, A.C.M.d.; Miranda-Caballero, C.I.; Grostieta, E.; Correa-Morales, F.; Romero-Pérez, R.; Romero-Contreras, F.E.; Rodríguez-Atanacio, J.A.; Ruiz-Tovar, K.; Huerta, H.; et al. Entomological Survey and Leishmania (Leishmania) mexicana Prevalence in Sand Fly Species during an Outbreak of Cutaneous Leishmaniasis in Quintana Roo State, Mexico. Trop. Med. Infect. Dis. 2023, 8, 465. https://doi.org/10.3390/tropicalmed8100465
Cañeda-Guzmán IC, Oca-Aguilar ACMd, Miranda-Caballero CI, Grostieta E, Correa-Morales F, Romero-Pérez R, Romero-Contreras FE, Rodríguez-Atanacio JA, Ruiz-Tovar K, Huerta H, et al. Entomological Survey and Leishmania (Leishmania) mexicana Prevalence in Sand Fly Species during an Outbreak of Cutaneous Leishmaniasis in Quintana Roo State, Mexico. Tropical Medicine and Infectious Disease. 2023; 8(10):465. https://doi.org/10.3390/tropicalmed8100465
Chicago/Turabian StyleCañeda-Guzmán, Isabel C., Ana C. Montes de Oca-Aguilar, Carlos I. Miranda-Caballero, Estefania Grostieta, Fabián Correa-Morales, Raquel Romero-Pérez, Francisco E. Romero-Contreras, José A. Rodríguez-Atanacio, Karina Ruiz-Tovar, Herón Huerta, and et al. 2023. "Entomological Survey and Leishmania (Leishmania) mexicana Prevalence in Sand Fly Species during an Outbreak of Cutaneous Leishmaniasis in Quintana Roo State, Mexico" Tropical Medicine and Infectious Disease 8, no. 10: 465. https://doi.org/10.3390/tropicalmed8100465
APA StyleCañeda-Guzmán, I. C., Oca-Aguilar, A. C. M. d., Miranda-Caballero, C. I., Grostieta, E., Correa-Morales, F., Romero-Pérez, R., Romero-Contreras, F. E., Rodríguez-Atanacio, J. A., Ruiz-Tovar, K., Huerta, H., Mis-Avila, P. C., Quintanilla-Cedillo, M. R., Lammoglia-Villagómez, M. A., Blum-Domínguez, S., Tamay-Segovia, P., Rojas-Ronquillo, R., Sánchez-Montes, S., & Becker, I. (2023). Entomological Survey and Leishmania (Leishmania) mexicana Prevalence in Sand Fly Species during an Outbreak of Cutaneous Leishmaniasis in Quintana Roo State, Mexico. Tropical Medicine and Infectious Disease, 8(10), 465. https://doi.org/10.3390/tropicalmed8100465





