Abattoir-Based Serological Surveillance and Spatial Risk Analysis of Foot-and-Mouth Disease, Brucellosis, and Q Fever in Lao PDR Large Ruminants
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Sample Collection
2.2. Serological Diagnostic Testing
2.3. Statistical, Spatial Correlation, and Relative Risk Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO, Food and Agriculture Organization of the United Nations. Chapter 1 Socio-Economic Consequences for Poor Livestock Farmers of Animal Diseases and VPH Problems; Animal Production and Health Division: Rome, Italy, 2002. [Google Scholar]
- FAO, Food and Agriculture Organisation of the United Nations. FAO and China Enter Agreement to Limit Animal Disease in Southeast Asia. 2018. Available online: http://www.fao.org/resilience/news-events/detail/en/c/1150117/ (accessed on 11 February 2021).
- Windsor, P. Perspectives on Australian animal health aid projects in South-East Asia. Transbound. Emerg. Dis. 2011, 58, 375–386. [Google Scholar] [CrossRef]
- Nampanya, S.; Khounsy, S.; Rast, L.; Windsor, P.A. Promoting transboundary animal disease risk management via a multiple health and husbandry intervention strategies in upland Lao PDR. Trop. Anim. Health Prod. 2014, 46, 439–446. [Google Scholar] [CrossRef]
- WPF, World Food Program. Lao PDR Country Strategic Plan (2017–2021); Via Cesare Giulio Viola: Rome, Italy, 2017. [Google Scholar]
- Siengsanan-Lamont, J.; Douangngeun, B.; Theppangna, W.; Khounsy, S.; Phommachanh, P.; Selleck, P.W.; Matsumoto, N.; Gleeson, L.J.; Blacksell, S.D. The development of an abattoir-based surveillance system in Lao PDR for the detection of zoonoses in large ruminants: Q fever and brucellosis seroepidemiology as a pilot study. Animals 2021, 11, 742. [Google Scholar] [CrossRef]
- OIE, World Organisation for Animal Health. Q Fever. OIE Listed 2021. Available online: https://www.oie.int/en/disease/q-fever/ (accessed on 8 March 2022).
- Bamaiyi, P.; Hassan, L.; Khairani-Bejo, S.; Zainal Abidin, M. Updates on brucellosis in Malaysia and Southeast Asia. Malays. J. Vet. Res. 2014, 5, 71–82. [Google Scholar]
- OIE, World Organisation for Animal Health. Brucellosis. OIE Listed 2021. Available online: https://www.oie.int/en/disease/brucellosis/ (accessed on 8 March 2022).
- Burns, R.J.; Douangngeun, B.; Theppangna, W.; Khounsy, S.; Mukaka, M.; Selleck, P.W.; Hansson, E.; Wegner, M.D.; Windsor, P.A.; Blacksell, S.D. Serosurveillance of Coxiellosis (Q-fever) and Brucellosis in goats in selected provinces of Lao People’s Democratic Republic. PLoS Negl. Trop. Dis. 2018, 12, e0006411. [Google Scholar] [CrossRef] [Green Version]
- Suputtamongkol, Y.; Rolain, J.-M.; Losuwanaruk, K.; Niwatayakul, K.; Suthinont, C.; Chierakul, W.; Pimda, K.; Raoult, D. Q fever in Thailand. Emerg. Infect. Dis. 2003, 9, 1186. [Google Scholar] [CrossRef]
- Te-Chaniyom, T.; Geater, A.F.; Kongkaew, W.; Chethanond, U.; Chongsuvivatwong, V. Goat farm management and Brucella serological test among goat keepers and livestock officers, 2011–2012, Nakhon Si Thammarat Province, southern Thailand. One Health 2016, 2, 126–130. [Google Scholar] [CrossRef] [Green Version]
- Peck, M.E.; Jenpanich, C.; Amonsin, A.; Bunpapong, N.; Chanachai, K.; Somrongthong, R.; Alexander, B.H.; Bender, J.B. Knowledge, attitudes and practices associated with brucellosis among small-scale goat farmers in Thailand. J. Agromedicine 2019, 24, 56–63. [Google Scholar] [CrossRef]
- Deka, R.P.; Magnusson, U.; Grace, D.; Shome, R.; Lindahl, J.F. Knowledge and practices of dairy farmers relating to brucellosis in urban, peri-urban and rural areas of Assam and Bihar, India. Infect. Ecol. Epidemiol. 2020, 10, 1769531. [Google Scholar] [CrossRef]
- Blacksell, S.; Conlan, J.V.; Gleeson, L.J.; Westbury, H.A.; Colling, A.; Paton, D.J.; Knowles, N.J.; Ferris, N.P.; Blacksell, S.D. Foot and mouth disease in the Lao People’s Democratic Republic: II. Seroprevalence estimates, using structured surveillance and surveys of abattoirs. Rev. Sci. Tech. 2008, 27, 851. [Google Scholar] [CrossRef]
- OIE, World Organisation for Animal Health. SEAFMD Campaign. 2021. Available online: https://rr-asia.oie.int/en/projects/foot-and-mouth-disease-fmd/seacfmd-campaign/ (accessed on 19 June 2020).
- ID.VET. ID SoftTM 2014. ID.VET. Available online: https://www.id-vet.com/ (accessed on 7 May 2021).
- Siengsanan-Lamont, J.; Douangngeun, B.; Theppangna, W.; Khounsy, S.; Phommachanh, P.; Kamolsiripichaiporn, S.; Udon, R.; Seeyo, K.B.; Selleck, P.W.; Matsumoto, N.; et al. Seroepidemiology of Foot and Mouth Disease using passive surveillance techniques in selected provinces of Lao PDR. Trop. Anim. Health Prod. 2021, 53, 303. [Google Scholar] [CrossRef]
- OIE, World Organization for Animal Health. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. In Chapter 3.01.04: Brucellosis; OIE: Paris, France, 2018; pp. 368–370. [Google Scholar]
- RStudio Team. RStudio: Integrated Development for R; RStudio, Inc.: Boston, MA, USA, 2015. [Google Scholar]
- Graul, C. leafletR: Interactive Web-Maps Based on the Leaflet JavaScript Library. R Package Version 0.4-0. 2016. Available online: https://rdrr.io/cran/leafletR/ (accessed on 17 May 2021).
- Stevenson, M. epiR: Tools for the Analysis of Epidemiological Data. R Package Version 1.0-14. 2020. Available online: https://CRAN.R-project.org/package=epiR (accessed on 17 May 2021).
- Reiczigel, J.; Földi, J.; Ózsvári, L. Exact confidence limits for prevalence of a disease with an imperfect diagnostic test. Epidemiol. Infect. 2010, 138, 1674–1678. [Google Scholar] [CrossRef]
- Harrison, E.; Pius, R. 9.2 Binary logistic regression. In R for Health Data Science; Chapman & Hall/C.R.C.: Boca Raton, FL, USA, 2021. [Google Scholar]
- Moraga, P. Chapter 5 Areal data. In Geospatial Health Data: Modeling and Visualisation with R-INLA and Shiny; Biostatistics Series; Chapman & Hall/C.R.C.: Boca Raton, FL, USA, 2019. [Google Scholar]
- Lindgren, F.; Rue, H.; Lindström, J. An explicit link between Gaussian fields and Gaussian Markov random fields: The stochastic partial differential equation approach. J. R. Stat. Soc. Ser. B Stat. Methodol. 2011, 73, 423–498. [Google Scholar] [CrossRef] [Green Version]
- Shira, D. Laos Increases Minimum Monthly Wage for the Third Time in Eight Years. 2018. Available online: https://www.aseanbriefing.com/news/laos-increases-minimum-monthly-wage-third-time-eight-years/ (accessed on 3 February 2022).
- Stür, W.W.; Gray, D.; Bastin, G. Review of the Livestock Sector in the Lao People’s Democratic Republic. 2002. Available online: https://cgspace.cgiar.org/bitstream/handle/10568/21136/adb_livestock_review.pdf?sequence=2&isAllowed=y (accessed on 8 March 2022).
- Lao Statistics Bureau. Results of Population and Housing Census 2015. Available online: https://www.familyplanning2020.org/sites/default/files/Lao-Population-census.pdf (accessed on 3 February 2022).
- Siengsanan-Lamont, J.; Tum, S.; Kong, L.; Selleck, P.W.; Gleeson, L.J.; Blacksell, S.D. Abattoir-Based Serological Surveillance for Transboundary and Zoonotic Diseases in Cattle and Swine in Cambodia; Mahidol-Oxford Tropical Medicine Research Unit, Mahidol University: Bangkok, Thailand, 2021. [Google Scholar] [CrossRef]
- FAO, Food and Agriculture Organisation of the United Nations. FAOSTAT: Livestock Primary. 2021. Available online: https://www.fao.org/faostat/en/#home (accessed on 21 June 2021).
- Douangngeun, B.; Theppangna, W.; Soukvilay, V.; Senaphanh, C.; Phithacthep, K.; Phomhaksa, S.; Yingst, S.; Lombardini, E.; Hansson, E.; Selleck, P.W.; et al. Seroprevalence of Q fever, brucellosis, and bluetongue in selected provinces in Lao People’s Democratic Republic. Am. J. Trop. Med. Hyg. 2016, 95, 558–561. [Google Scholar] [CrossRef] [Green Version]
- Holt, H.R.; Inthavong, P.; Blaszak, K.; Keokamphe, C.; Phongmany, A.; Blacksell, S.D.; Durr, P.A.; Graham, K.; Allen, J.; Donnelly, B.; et al. Production diseases in smallholder pig systems in rural Lao PDR. Prev. Vet. Med. 2019, 162, 110–116. [Google Scholar] [CrossRef]
- Phongmany, S.; Rolain, J.-M.; Phetsouvanh, R.; Blacksell, S.D.; Soukkhaseum, V.; Rasachack, B.; Phiasakha, K.; Soukkhaseum, S.; Frichithavong, K.; Chu, V.; et al. Rickettsial infections and fever, Vientiane, Laos. Emerg. Infect. Dis. 2006, 12, 256. [Google Scholar] [CrossRef]
- Greiner, A.L.; Gregory, C.J.; Bhengsri, S.; Edouard, S.; Parola, P.; Raoult, D.; Million, M.; Thamthitiwat, S.; Clarke, K.; Kersh, G.J. Acute Q fever case detection among acute febrile illness patients, Thailand, 2002–2005. Am. J. Trop. Med. Hyg. 2018, 98, 252–257. [Google Scholar] [CrossRef] [Green Version]
- CDNA, Communicable Diseases Network Australia. Q Fever, Department of Health. 2018. Available online: https://www1.health.gov.au/internet/main/publishing.nsf/Content/56DFBAB23468BF71CA2583520001F02F/$File/Q-fever-SoNG2018.pdf (accessed on 11 February 2022).
- Vongxay, K.; Conlan, J.V.; Khounsy, S.; Dorny, P.; Fenwick, S.; Thompson, R.C.A.; Blacksell, S.D. Seroprevalence of major bovine-associated zoonotic infectious diseases in the Lao People’s Democratic Republic. Vector-Borne Zoonotic Dis. 2012, 12, 861–866. [Google Scholar] [CrossRef] [Green Version]
- Burniston, S.; Okello, A.L.; Khamlome, B.; Inthavong, P.; Gilbert, J.; Blacksell, S.D.; Allen, J.; Welburn, S.C. Cultural drivers and health-seeking behaviours that impact on the transmission of pig-associated zoonoses in Lao People’s Democratic Republic. Infect. Dis. Poverty 2015, 4, 11. [Google Scholar] [CrossRef] [Green Version]
- Jittapalapong, S.; Inpankaew, T.; Sangwaranond, A.; Phasuk, C.; Pinyopanuwat, N.; Chimnoi, W.; Kengradomkij, C.; Sununta, C.; Arunwipat, P. Current status of brucellosis in dairy cows of Chiang Rai Province, Thailand. Agric. Nat. Resour. 2008, 42, 67–70. [Google Scholar]
- Sagarasaeranee, O.; Kaewkalong, S.; Sujit, K.; Chanachai, K. Seroprevalence of brucellosis in small ruminants in Thailand, 2013. OSIR J. 2017, 9. [Google Scholar]
- Peck, M.E.; Chanachai, K.; Jenpanich, C.; Amonsin, A.; Alexander, B.H.; Bender, J.B. Seroprevalence of brucellosis in goats and sheep in Thailand: Results from the Thai National Brucellosis Surveillance System from 2013 to 2015. Transbound. Emerg. Dis. 2018, 65, 799–805. [Google Scholar] [CrossRef] [PubMed]
- McCAUGHEY, C.; Murray, L.J.; McKENNA, J.P.; Menzies, F.D.; McCULLOUGH, S.J.; O’Neill, H.J.; Wyatt, D.E.; Cardwell, C.R.; Coyle, P.V. Coxiella burnetii (Q fever) seroprevalence in cattle. Epidemiol. Infect. 2010, 138, 21–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- MacPhillamy, I.; Young, J.; Earp, F.; Khounsy, S.; Windsor, P.; Toribio, J.; Bush, R. Foot-and-mouth disease seroprevalence and reporting behaviours in nine northern provinces in Lao PDR: The current situation and challenges for control. Transbound. Emerg. Dis. 2021, 69, 645–659. [Google Scholar] [CrossRef]
- Xaydalasouk, K.; Innoula, N.; Putthana, V.; Chanthavongsa, K.; Snoeck, C.J.; Hübschen, J.M.; Oudomphone, P.; Chan, B.; Muller, C.P.; Black, A.P.; et al. High seroprevalence of Foot and Mouth Disease in Laos: Call for nationwide vaccination campaigns and disease surveillance. Transbound. Emerg. Dis. 2020, 68, 2345–2352. [Google Scholar] [CrossRef]
- Ferrari, G.; Paton, D.; Duffy, S.; Bartels, C. Foot and Mouth Disease Vaccination and Post-Vaccination Monitoring; FAO/OIE: Rome, Italy, 2016. [Google Scholar]
- Doel, T. Natural and vaccine-induced immunity to foot and mouth disease: The prospects for improved vaccines. Rev. Sci. Tech. Int. Off. Epizoot. 1996, 15, 883–911. [Google Scholar] [CrossRef]
- Nampanya, S.; Khounsy, S.; Abila, R.; Windsor, P.A. Implementing large Foot and Mouth Disease vaccination programmes for smallholder farmers: Lessons from Lao PDR. Epidemiol. Infect. 2018, 146, 2086–2095. [Google Scholar] [CrossRef] [Green Version]
- Souriya, V.; Piamsomboon, P.; Ajariyakhajorn, K.; Damrongwatanapokin, T.; Inchaisri, C. Risk factors of foot and mouth disease in an endemic area on low vaccination rate in Xayaboury province of Lao People’s Democratic Republic (Lao PDR). Trop. Anim. Health Prod. 2019, 52, 1103–1114. [Google Scholar] [CrossRef]
- Kerr, J.; Sieng, S.; Scoize, A. Working with traders to understand livestock movements and spread of animal disease in Cambodia and Lao PDR. In Animal Biosecurity in the Mekong: Future Directions for Research and Development; ACIAR Proceeding: Siem Reap, Cambodia, 2012; Volume 137, pp. 59–64. [Google Scholar]
- Sieng, S.; Patrick, I.W.; Walkden-Brown, S.; Sar, C. A cost-benefit analysis of foot and mouth disease control program for smallholder cattle farmers in Cambodia. Authorea Prepr. 2021. [Google Scholar] [CrossRef]
- Smith, P.; Luthi, N.B.; Huachun, L.; Oo, K.N. Movement Pathways and Market Chains of Large Ruminants in the Greater Mekong Sub-Region. 2015. Available online: https://core.ac.uk/download/pdf/231205375.pdf (accessed on 9 March 2022).
- Tenny, S.; Hoffman, M.R. Relative Risk; StatPearls Publishing: Treasure Island, FL, USA, 2017. [Google Scholar]
- Subharat, S.; Wada, M.; Sutar, A.; Abila, R.; Khounsy, S.; Heuer, C. Livestock movement patterns in the main livestock production provinces of Lao PDR. Transbound. Emerg. Dis. 2021. [Google Scholar] [CrossRef]
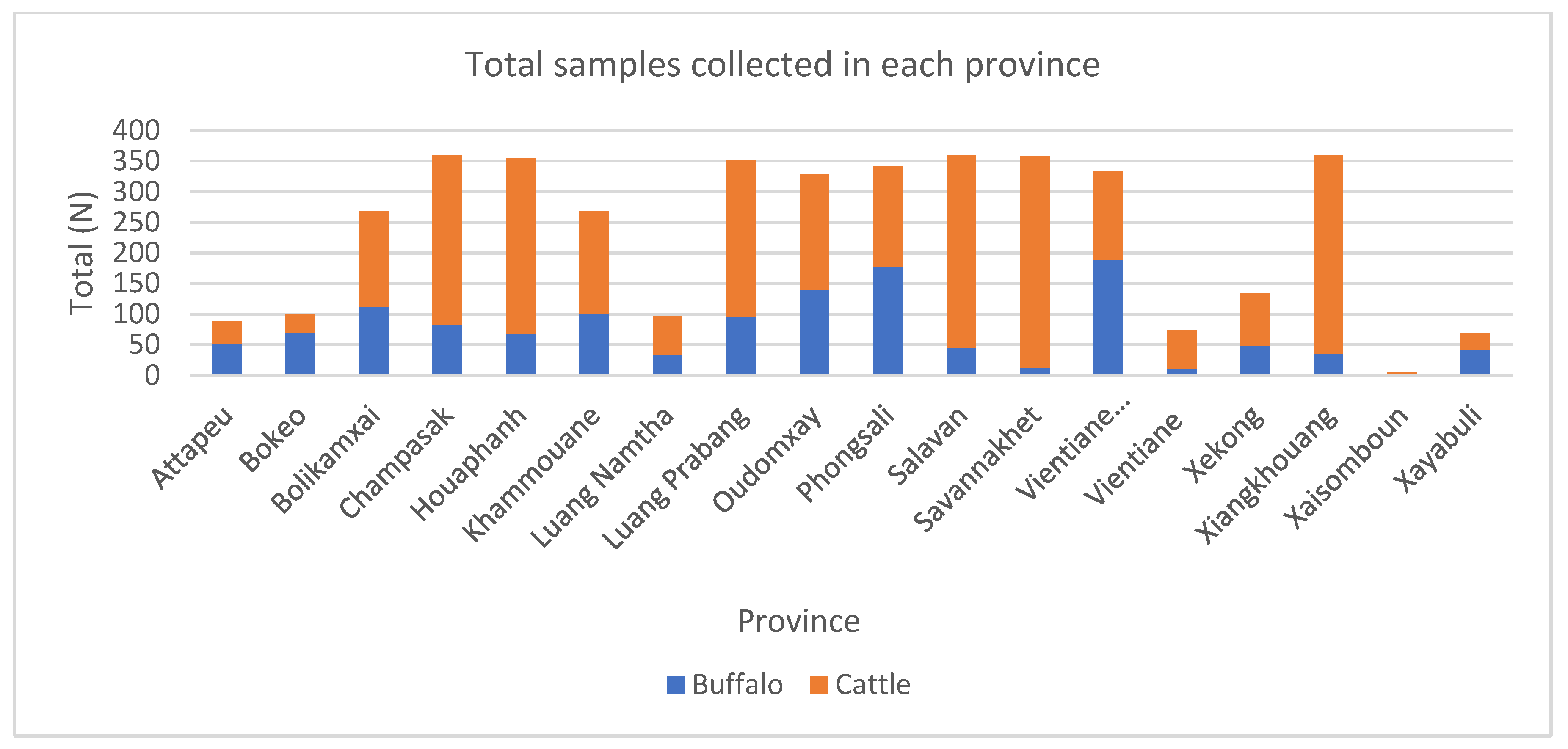
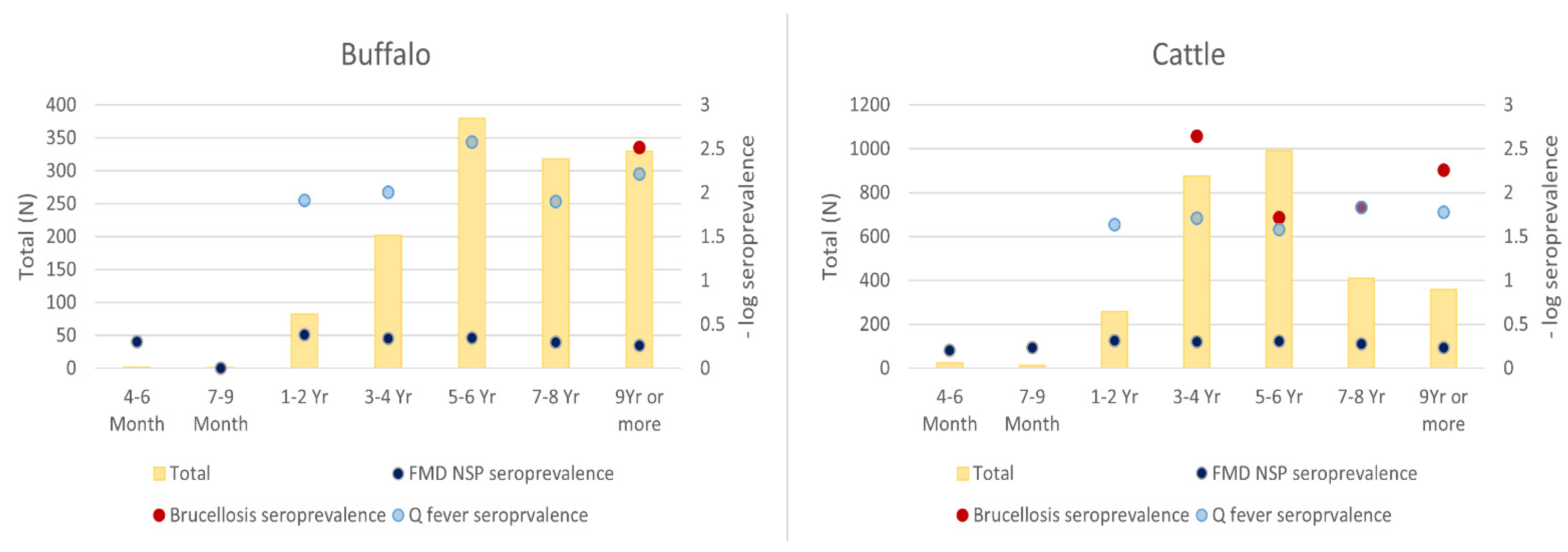

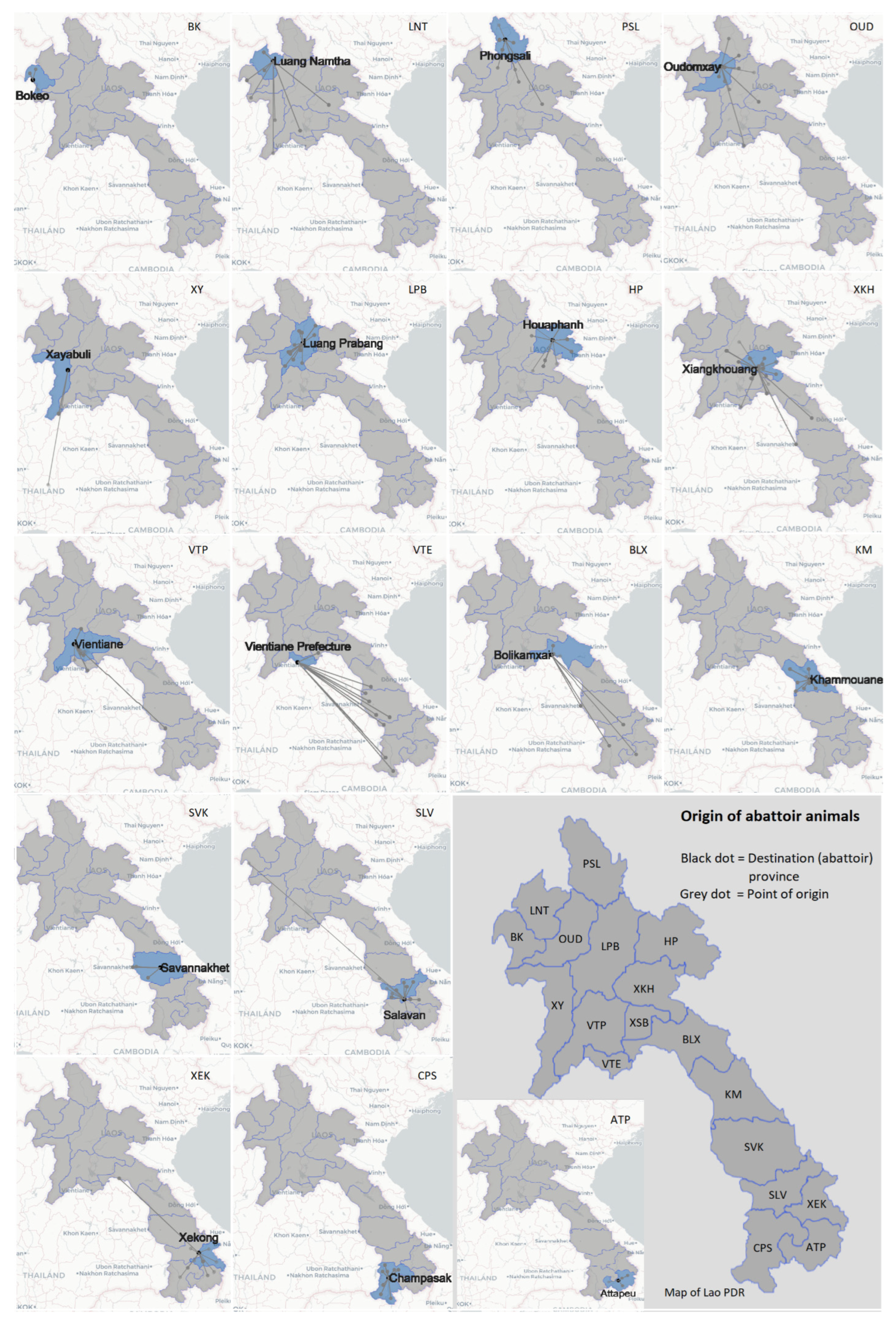
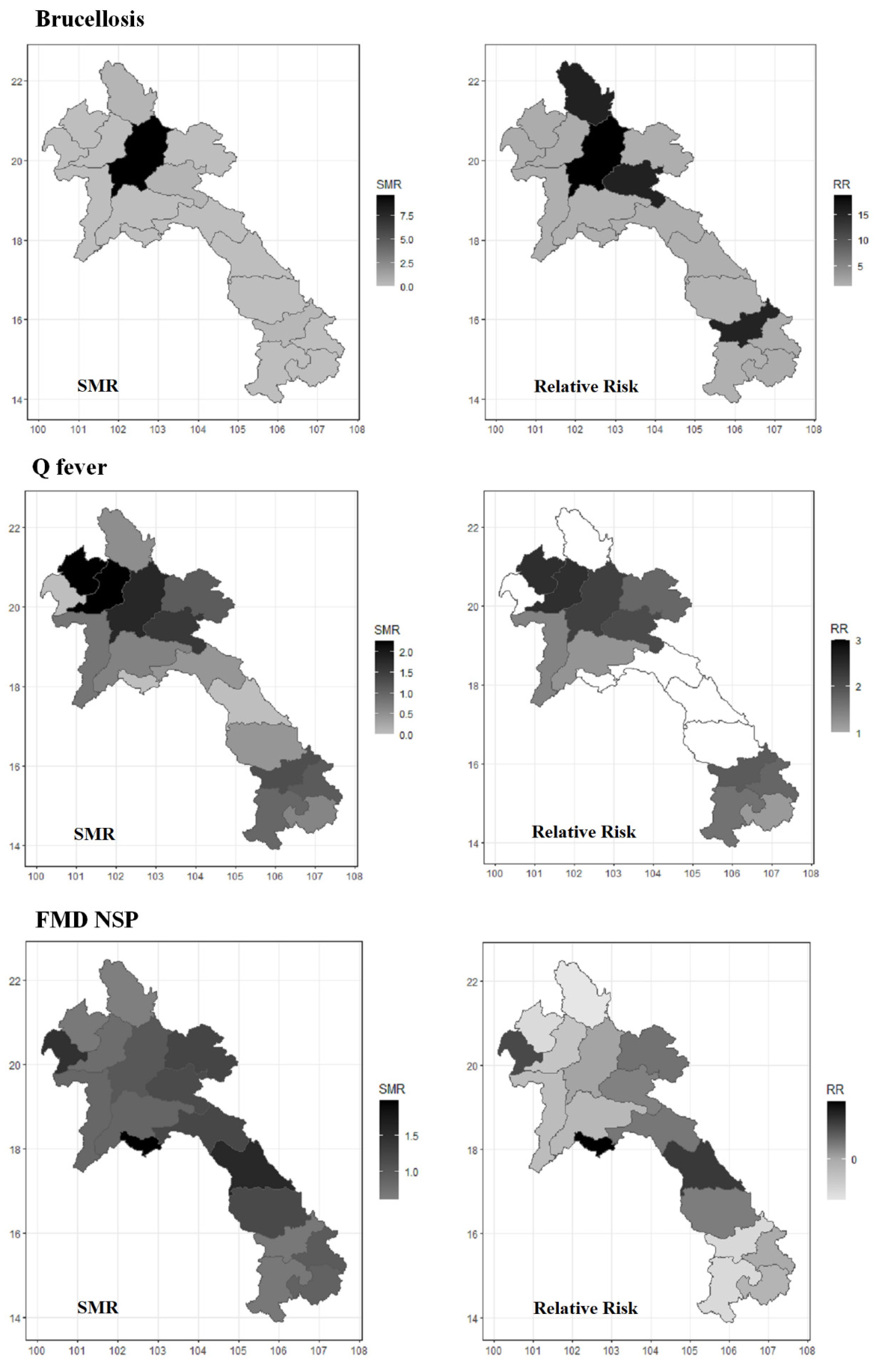
| Tested Animals (n = 4247) | Positive | % Apparent Seroprevalence (95% CI) | % True Seroprevalence (95% CI) |
|---|---|---|---|
| FMD NSP * | 2144 | 50.5 (49.0, 52.0) | 54.6 (53.0, 56.2) |
| Brucellosis | 30 | 0.7 (0.5, 1.0) | 0.4 (0.2, 0.7) |
| Q fever | 71 | 1.7 (1.3, 2.1) | 1.7 (1.3, 2.1) |
| Province of Origin | Date Collected | Type | Positive |
|---|---|---|---|
| Salavan | 28 May 2020 | Cattle | 1 |
| Xiangkhouang | 24 August 2020 | Cattle | 1 |
| Phongsali | 6 September 2020 | Buffalo | 1 |
| Luang Prabang | 6 September 2020 | Cattle | 3 |
| 7 September 2020 | Cattle | 8 | |
| 8 September 2020 | Cattle | 3 | |
| 9 September 2020 | Cattle | 4 | |
| 26 September 2020 | Cattle | 3 | |
| 27 September 2020 | Cattle | 3 | |
| 28 September 2020 | Cattle | 3 | |
| Total | 30 | ||
| Province of Origin | Type | Total (n) | FMD NSP * | Brucellosis | Q Fever | |||
|---|---|---|---|---|---|---|---|---|
| Positive | Sero Prevalence | Positive | Sero Prevalence | Positive | Sero Prevalence | |||
| Attapeu (ATP) | Buffalo | 57 | 30 | 52.6% | 0 | 0.0% | 1 | 1.8% |
| Cattle | 39 | 14 | 35.9% | 0 | 0.0% | 0 | 0.0% | |
| Bokeo (BK) | Buffalo | 74 | 53 | 71.6% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 36 | 28 | 77.8% | 0 | 0.0% | 0 | 0.0% | |
| Bolikamxai (BLX) | Buffalo | 106 | 69 | 65.1% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 170 | 99 | 58.2% | 0 | 0.0% | 2 | 1.2% | |
| Champasak (CPS) | Buffalo | 121 | 49 | 40.5% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 308 | 98 | 31.8% | 0 | 0.0% | 7 | 2.3% | |
| Houaphanh (HP) | Buffalo | 66 | 40 | 60.6% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 256 | 160 | 62.5% | 0 | 0.0% | 6 | 2.3% | |
| Khammouane (KM) | Buffalo | 114 | 85 | 74.6% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 181 | 148 | 81.8% | 0 | 0.0% | 0 | 0.0% | |
| Luang Namtha (LNT) | Buffalo | 11 | 3 | 27.3% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 42 | 15 | 35.7% | 0 | 0.0% | 2 | 4.8% | |
| Luang Prabang (LBP) | Buffalo | 117 | 51 | 43.6% | 0 | 0.0% | 1 | 0.9% |
| Cattle | 282 | 154 | 54.6% | 27 | 9.6% | 11 | 3.9% | |
| Oudomxay (OUD) | Buffalo | 114 | 38 | 33.3% | 0 | 0.0% | 6 | 5.3% |
| Cattle | 178 | 80 | 44.9% | 0 | 0.0% | 5 | 2.8% | |
| Phongsali (PSL) | Buffalo | 181 | 57 | 31.5% | 1 | 0.6% | 0 | 0.0% |
| Cattle | 159 | 48 | 30.2% | 0 | 0.0% | 3 | 1.9% | |
| Salavan (SLV) | Buffalo | 94 | 39 | 41.5% | 0 | 0.0% | 2 | 2.1% |
| Cattle | 326 | 106 | 32.5% | 1 | 0.3% | 7 | 2.1% | |
| Savannakhet (SVK) | Buffalo | 110 | 42 | 38.2% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 437 | 284 | 65.0% | 0 | 0.0% | 4 | 0.9% | |
| Vientiane prefecture (VTE) | Cattle | 1 | 1 | 100.0% | 0 | 0.0% | 0 | 0.0% |
| Vientiane (VTP) | Buffalo | 13 | 5 | 38.5% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 53 | 25 | 47.2% | 0 | 0.0% | 0 | 0.0% | |
| Xekong (XEK) | Buffalo | 44 | 26 | 59.1% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 115 | 53 | 46.1% | 0 | 0.0% | 3 | 2.6% | |
| Xiangkhouang (XKH) | Buffalo | 46 | 33 | 71.7% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 297 | 168 | 56.6% | 1 | 0.3% | 9 | 3.0% | |
| Xaisomboun (XSB) | Buffalo | 4 | 2 | 50.0% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 20 | 8 | 40.0% | 0 | 0.0% | 1 | 5.0% | |
| Xayabuli (XY) | Buffalo | 43 | 19 | 44.2% | 0 | 0.0% | 0 | 0.0% |
| Cattle | 31 | 13 | 41.9% | 0 | 0.0% | 1 | 3.2% | |
| Thailand | Buffalo | 1 | 1 | 100.0% | 0 | 0.0% | 0 | 0.0% |
| Total | 4247 | 2144 | 50.5% | 30 | 0.7% | 71 | 1.7% | |
| Variable (Reference Group) | p-Value ^,** | Category ** | Odds Ratio (95% CI) |
|---|---|---|---|
| FMD NSP * | |||
| Destination province (Phongsali) | <0.00001 | Attapeu | 1.8 (1.1, 2.8) |
| Bokeo | 6.0 (3.7, 9.9) | ||
| Bolikamxai | 3.7 (2.7, 5.2) | ||
| Houaphanh | 3.6 (2.6, 4.9) | ||
| Khammouane | 10.1 (6.9,14.8) | ||
| Luang Namtha | 2.2 (1.4, 3.5) | ||
| Luang Prabang | 2.5 (1.8, 3.4) | ||
| Oudomxay | 1.5 (1.1, 2.1) | ||
| Savannakhet | 4.6 (3.4, 6.4) | ||
| Vientiane prefecture | 1.9 (1.4, 2.7) | ||
| Xekong | 3.0 (2.0, 4.6) | ||
| Xiangkhouang | 3.2 (2.4, 4.4) | ||
| Xayabuli | 1.9 (1.1, 3.2) | ||
| Province of origin (Phongsali) | <0.00001 | Attapeu | 1.9 (1.2, 3.0) |
| Bokeo | 6.3 (3.9, 10.1) | ||
| Bolikamxai | 3.5 (2.5, 4.9) | ||
| Houaphanh | 3.7 (2.7, 5.1) | ||
| Khammouane | 8.4 (5.9, 12.1) | ||
| Luang Prabang | 2.4 (1.7, 3.2) | ||
| Oudomxay | 1.5 (1.1, 2.1) | ||
| Savannakhet | 3.3 (2.5, 4.4) | ||
| Vientiane | 1.9 (1.1, 3.2) | ||
| Xekong | 2.2 (1.5, 3.3) | ||
| Xiangkhouang | 3.2 (2.3, 4.3) | ||
| Xayabuli | 1.7 (1.0, 2.9) | ||
| Sex (Male) | 7.97 × 10−8 | Sex: Female | 1.4 (1.3, 1.6) |
| Age (1–2 Years) | 0.009269 | Age: 9 Years or more | 1.5 (1.1, 1.9) |
| Date collection (25 January 2020) | <0.00001 | Date collected 23 September 2020 | 10.5 (1.4, 78.1) |
| Date collected 6 November 2020 | 15.3 (1.9, 122.8) | ||
| Date collected 7 November 2020 | 9.7 (1.4, 65.4) | ||
| Date collected 8 November 2020 | 11.5 (1.6, 85.2) | ||
| Date collected 20 November 2020 | 8.0 (1.1, 56.8) | ||
| Collection month (March) | <0.00001 | Collection.month: August | 1.4 (1.0, 1.9) |
| Collection.month: September | 2.3 (1.7, 3.2) | ||
| Collection.month: October | 1.9 (1.3, 2.5) | ||
| Collection.month: November | 2.3 (1.7, 3.2) | ||
| Collection.month: December | 1.4 (1.1, 2.0) | ||
| Q fever | |||
| Destination province (Bokeo) | 0.005182 | - | - |
| Province of origin (Bokeo) | 0.005772 | - | - |
| Animal type (Buffalo) | 0.000855 | Animal type: Cattle | 2.8 (1.4, 5.4) |
| Collection month (February) | 3.40 × 10−6 | - | - |
| Brucellosis | |||
| Destination province (Bokeo) | <0.00001 | - | - |
| Province of origin (Bokeo) | <0.00001 | - | - |
| Body condition score (score 1) | 0.003206 | - | - |
| Age (1–2 Years) | 0.007222 | - | - |
| Animal type (Buffalo) | 9.87 × 10−5 | Animal type: Cattle | 13.1 (1.8, 96.5) |
| Collection month (February) | <0.00001 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siengsanan-Lamont, J.; Theppangna, W.; Phommachanh, P.; Khounsy, S.; Selleck, P.W.; Matsumoto, N.; Gleeson, L.J.; Blacksell, S.D. Abattoir-Based Serological Surveillance and Spatial Risk Analysis of Foot-and-Mouth Disease, Brucellosis, and Q Fever in Lao PDR Large Ruminants. Trop. Med. Infect. Dis. 2022, 7, 78. https://doi.org/10.3390/tropicalmed7050078
Siengsanan-Lamont J, Theppangna W, Phommachanh P, Khounsy S, Selleck PW, Matsumoto N, Gleeson LJ, Blacksell SD. Abattoir-Based Serological Surveillance and Spatial Risk Analysis of Foot-and-Mouth Disease, Brucellosis, and Q Fever in Lao PDR Large Ruminants. Tropical Medicine and Infectious Disease. 2022; 7(5):78. https://doi.org/10.3390/tropicalmed7050078
Chicago/Turabian StyleSiengsanan-Lamont, Jarunee, Watthana Theppangna, Phouvong Phommachanh, Syseng Khounsy, Paul W. Selleck, Nina Matsumoto, Laurence J. Gleeson, and Stuart D. Blacksell. 2022. "Abattoir-Based Serological Surveillance and Spatial Risk Analysis of Foot-and-Mouth Disease, Brucellosis, and Q Fever in Lao PDR Large Ruminants" Tropical Medicine and Infectious Disease 7, no. 5: 78. https://doi.org/10.3390/tropicalmed7050078
APA StyleSiengsanan-Lamont, J., Theppangna, W., Phommachanh, P., Khounsy, S., Selleck, P. W., Matsumoto, N., Gleeson, L. J., & Blacksell, S. D. (2022). Abattoir-Based Serological Surveillance and Spatial Risk Analysis of Foot-and-Mouth Disease, Brucellosis, and Q Fever in Lao PDR Large Ruminants. Tropical Medicine and Infectious Disease, 7(5), 78. https://doi.org/10.3390/tropicalmed7050078







