Chitosan-Based Nanoparticles and Biomaterials for Pulp Capping and Regeneration: A Systematic Review with Quantitative and Evidence-Mapping Synthesis
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol and Registration
2.2. Research Question
2.3. Objectives
- To systematically identify and appraise preclinical (in vitro and animal) studies evaluating chitosan-based nanoparticles or biomaterials for pulp capping or regeneration.
- To quantitatively synthesize comparable outcomes using meta-analysis where feasible, and to use direction-of-effect synthesis and evidence mapping where meta-analysis is not appropriate.
2.4. Eligibility Criteria
- Controlled in vitro or in vivo studies evaluating chitosan-based nanoparticles or biomaterials (scaffolds, hydrogels, composites) for dental pulp capping or regeneration.
- Studies reporting quantitative or digitizable outcomes (mean ± SD/SE, p-values).
- Comparators: calcium hydroxide, MTA, Biodentine, or other pulp-capping materials.
- Reviews, editorials, letters, or conference abstracts.
- Material characterization studies without biological outcomes.
- Case reports, single-arm experiments, or studies lacking control groups.
2.5. PICO Framework
2.6. Search Strategy
2.7. Study Selection
2.8. Data Extraction and Management
2.9. Risk of Bias Assessment
2.9.1. In Vivo Studies
2.9.2. In Vitro Studies
2.10. Quantitative and Semi-Quantitative Synthesis
2.10.1. Meta-Analysis Feasibility and Approach
2.10.2. Direction-of-Effect and Weighting
- mean ± SD (weight = 2)
- p-value only (weight = 1)
- qualitative (weight = 0.5)
2.10.3. Certainty of Evidence
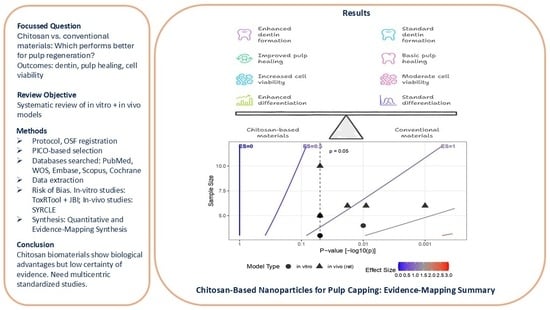
3. Results
3.1. Overview
3.1.1. In Vivo Findings
3.1.2. In Vitro Findings
3.1.3. Integrated Summary
3.2. Risk of Bias
3.2.1. In Vivo Studies (SYRCLE Assessment)
3.2.2. In Vitro Studies (ToxRTool/JBI Assessment)
3.3. Quantitative Findings
3.3.1. Meta-Analysis Feasibility
3.3.2. Direction-of-Effect Synthesis
3.4. Certainty of Evidence (GRADE-Preclinical Assessment) [20]
4. Discussion
Strength and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ahmed, B.; Ragab, M.H.; Galhom, R.A.; Hassan, H.Y. Evaluation of dental pulp stem cells behavior after odontogenic differentiation induction by three different bioactive materials on two different scaffolds. BMC Oral Health 2023, 23, 252. [Google Scholar] [CrossRef]
- Hiremath, G.; Pramanik, S.; Horatti, P.; Anil. Comparative evaluation of bioactivity of MTA plus and MTA plus chitosan conjugate in phosphate buffer saline an invitro study. Saudi Dent. J. 2024, 36, 1097–1104. [Google Scholar] [CrossRef]
- Zhu, N.; Chatzistavrou, X.; Papagerakis, P.; Ge, L.; Qin, M.; Wang, Y. Silver-Doped Bioactive Glass/Chitosan Hydrogel with Potential Application in Dental Pulp Repair. ACS Biomater. Sci. Eng. 2019, 5, 4624–4633. [Google Scholar] [CrossRef]
- Hashemi-Beni, B.; Khoroushi, M.; Foroughi, M.R.; Karbasi, S.; Khademi, A.A. Cytotoxicity assessment of polyhydroxybutyrate/chitosan/nano-bioglass nanofiber scaffolds by stem cells from human exfoliated deciduous teeth stem cells from dental pulp of exfoliated deciduous tooth. Dent. Res. J. 2018, 15, 136–145. [Google Scholar]
- Huang, Z.Y.; Hermosa, G.C.; Wu, J.S.; Wu, T.L.; Chien, C.C.; Liao, C.S.; Huang, Y.T.; Wang, H.D.; Sun, A.A. Chitosan-Based Antibacterial Bioceramic Materials for Dental Pulp Capping. ACS Biomater. Sci. Eng. 2025, 11, 6119–6133. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, T.; Yanagiguchi, K.; Yamada, S.; Ohara, N.; Ikeda, T.; Hayashi, Y. Chitosan monomer promotes tissue regeneration on dental pulp wounds. J. Biomed. Mater. Res. A 2006, 76, 711–720. [Google Scholar] [CrossRef]
- Sadeghinia, A.; Davaran, S.; Salehi, R.; Jamalpoor, Z. Nano-hydroxy apatite/chitosan/gelatin scaffolds enriched by a combination of platelet-rich plasma and fibrin glue enhance proliferation and differentiation of seeded human dental pulp stem cells. Biomed. Pharmacother. 2019, 109, 1924–1931. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.S.; Kritika, S.; Karthikeyan, N.S.; Sujatha, V.; Mahalaxmi, S.; Ravichandran, C. Development of cobalt-incorporated chitosan scaffold for regenerative potential in human dental pulp stem cells: An in vitro study. Int. J. Biol. Macromol. 2023, 253, 126574. [Google Scholar] [CrossRef] [PubMed]
- Bordini, E.A.F.; Cassiano, F.B.; Silva, I.S.P.; Usberti, F.R.; Anovazzi, G.; Pacheco, L.E.; Pansani, T.N.; Leite, M.L.; Hebling, J.; de Souza Costa, C.A. Synergistic potential of 1α, 25-dihydroxyvitamin D3 and calcium–aluminate–chitosan scaffolds with dental pulp cells. Clin. Oral Investig. 2020, 24, 663–674. [Google Scholar] [CrossRef]
- Gould, M.L.; Downes, N.J.; Woolley, A.G.; Hussaini, H.M.; Ratnayake, J.T.; Ali, M.A.; Friedlander, L.T.; Cooper, P.R. Harnessing the Regenerative Potential of Purified Bovine Dental Pulp and Dentin Extracellular Matrices in a Chitosan/Alginate Hydrogel. Macromol. Biosci. 2024, 24, e2400254. [Google Scholar] [CrossRef]
- Hoveizi, E.; Naddaf, H.; Ahmadianfar, S.; Gutmann, J.L. Encapsulation of human endometrial stem cells in chitosan hydrogel containing titanium oxide nanoparticles for dental pulp repair and tissue regeneration in male Wistar rats. J. Biosci. Bioeng. 2023, 135, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Zhou, Y.; Yu, Y.; Zhou, X.; Du, W.; Wan, M.; Fan, Y.; Zhou, X.; Xu, X.; Zheng, L. Evaluation of Chitosan Hydrogel for Sustained Delivery of VEGF for Odontogenic Differentiation of Dental Pulp Stem Cells. Stem Cells Int. 2019, 2019, 1515040. [Google Scholar] [CrossRef]
- Gurucharan, I.; Saravana Karthikeyan, B.; Mahalaxmi, S.; Baskar, K.; Rajkumar, G.; Dhivya, V.; Kishen, A.; Sankaranarayanan, S.; Gurucharan, N. Characterization of nano-hydroxyapatite incorporated carboxymethyl chitosan composite on human dental pulp stem cells. Int. Endod. J. 2023, 56, 486–501. [Google Scholar] [CrossRef]
- Sularsih, S.; Fransiska, W.; Salsabila, S.; Rahmitasari, F.; Soesilo, D.; Prananingrum, W. Potency of the Combination of Chitosan and Hydroxyapatite on Angiogenesis and Fibroblast Cell Proliferation in Direct Pulp Capping of Rattus norvegicus. Eur. J. Dent. 2024, 18, 1135–1141. [Google Scholar] [CrossRef]
- Widyastuti, N.H.; Prayitno, A.; Cilmiaty, R.; Wasita, B. Effect of chitosan nanoparticles from red snapper scales (Lutjanus sp.) on pulp repair: In Vivo study. J. Pharm. Pharmacogn. Res. 2024, 12, S69–S74. [Google Scholar] [CrossRef]
- Sornamalar, M.; Dhanavel, C.; Balavaishnavi, G.; Shajahan, S.; Baskar, K.; Raja, S.V. Nano Phosphorylated Pullulan-Integrated Carboxymethyl Chitosan Scaffold for Regeneration of the Pulp-Dentin Complex: An In vitro Study. J. Pharm. Bioallied Sci. 2024, 16, S4563–S4567. [Google Scholar] [CrossRef] [PubMed]
- Hooijmans, C.R.; Rovers, M.M.; De Vries, R.B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 43. [Google Scholar] [CrossRef] [PubMed]
- Tran, L.; Tam, D.N.H.; Elshafay, A.; Dang, T.; Hirayama, K.; Huy, N.T. Quality assessment tools used in systematic reviews of in vitro studies: A systematic review. BMC Med. Res. Methodol. 2021, 21, 101. [Google Scholar] [CrossRef]
- Boon, M.H.; Thomson, H. The effect direction plot revisited: Application of the 2019 Cochrane Handbook guidance on alternative synthesis methods. Res. Synth. Methods 2021, 12, 29–33. [Google Scholar] [CrossRef]
- Prasad, M. Introduction to the GRADE tool for rating certainty in evidence and recommendations. Clin. Epidemiol. Glob. Health 2024, 25, 101484. [Google Scholar]
- Zheng, K.; Feng, G.; Zhang, J.; Xing, J.; Huang, D.; Lian, M.; Zhang, W.; Wu, W.; Hu, Y.; Lu, X.; et al. Basic fibroblast growth factor promotes human dental pulp stem cells cultured in 3D porous chitosan scaffolds to neural differentiation. Int. J. Neurosci. 2021, 131, 625–633. [Google Scholar] [CrossRef]
- Widyastuti, N.H.; Cilmiaty, R.; Prayitno, A.; Wasita, B.; Soetrisno, S. Comparison between Nanochitosane and Calcium Hydroxide on BMP-2 and TGF-β1 Levels in Pulp Capping Treatment (In Vivo Study). J. Med. Chem. Sci. 2024, 7, 681–688. [Google Scholar] [CrossRef]
- Anaya-Sampayo, L.M.; Garcia-Robayo, D.A.; Roa, N.S.; Rodriguez-Lorenzo, L.M.; Martinez-Cardozo, C. Platelet-rich fibrin (PRF) modified nano-hydroxyapatite/chitosan/gelatin/alginate scaffolds increase adhesion and viability of human dental pulp stem cells (DPSC) and osteoblasts derived from DPSC. Int. J. Biol. Macromol. 2024, 273, 133064. [Google Scholar] [CrossRef] [PubMed]
- Wiedermann, W.; Li, X. Direction dependence analysis: A framework to test the direction of effects in linear models with an implementation in SPSS. Behav. Res. Methods 2018, 50, 1581–1601. [Google Scholar] [CrossRef]
- Schuller-Martínez, B.; Meza, N.; Pérez-Bracchiglione, J.; Franco, J.V.A.; Loezar, C.; Madrid, E. Graphical representation of the body of the evidence: The essentials for understanding the evidence gap map approach. Medwave 2021, 21, e8164. [Google Scholar] [CrossRef] [PubMed]
- Cicciù, M.; Fiorillo, L.; Cervino, G. Chitosan use in dentistry: A systematic review of recent clinical studies. Mar. Drugs 2019, 17, 417. [Google Scholar] [CrossRef]
- Crini, G.; Badot, P.-M. Application of chitosan, a natural aminopolysaccharide, for dye removal from aqueous solutions by adsorption processes using batch studies: A review of recent literature. Prog. Polym. Sci. 2008, 33, 399–447. [Google Scholar] [CrossRef]
- Harrison, S.; Jones, H.E.; Martin, R.M.; Lewis, S.J.; Higgins, J.P. The albatross plot: A novel graphical tool for presenting results of diversely reported studies in a systematic review. Res. Synth. Methods 2017, 8, 281–289. [Google Scholar] [CrossRef]




| Author, Year, Study Type | Model/Sample (Species/Cell Type/n) | Intervention (Chitosan-Based: Formulation, Dose/Concentration) | Comparator (Type, Dose) | Outcome Measure (Definition & Unit) | Mean/SD/SE/Event Count | Effect Direction/Trend/Timepoint(s) | Key Findings/Conclusion |
|---|---|---|---|---|---|---|---|
| Widyastuti et al. 2024 [22] In vivo (rats) | Sprague Dawley rats (n = 18) | Chitosan nanoparticles (CN) from red snapper scales; ionic gelation method | Reversible pulpitis alone; Ca(OH)2 application | TGF-β1 expression (ELISA), reparative dentin formation (histopathology) | TGF-β1: p = 0.049; Dentin formation: p = 0.009 | Positive/increased TGF-β1 and dentin formation Not specified | CN enhanced pulp repair and promoted reparative dentin formation compared to control and Ca(OH)2 |
| Widyastuti et al. 2024 [15] In vivo (rats) | Sprague-Dawley rats; reversible pulpitis model | Nanochitosan (chitosan nanoparticles) applied as pulp capping | Calcium hydroxide (Ca(OH)2); healthy and untreated pulpitis controls | BMP-2 and TGF-β1 levels measured by ELISA (pg/mL) | Significant difference; One-way ANOVA p < 0.001 (BMP-2), p = 0.016 (TGF-β1) | Positive/Nanochitosan > Ca(OH)2 for BMP-2 and TGF-β1 28 days | Nanochitosan increased BMP-2 and TGF-β1 expression more than Ca(OH)2 in reversible pulpitis rats |
| Sularsih et al. 2024 [14] In vivo (rats) | Male Wistar rats (Rattus norvegicus), n = 60, age: 8–16 weeks, weight: 200–250 g | Direct pulp capping with chitosan + hydroxyapatite paste (CH-HA) | KA: glass ionomer cement; KB: Ca(OH)2; PA: chitosan; PB: hydroxyapatite | VEGF expression (IHC), number of blood vessels, fibroblast cell proliferation (histology) | Reported as mean ± SD/qualitative comparison (p < 0.05) | Positive/increased VEGF, blood vessels, fibroblast proliferation 3, 7, 14 days | CH-HA combination significantly enhanced angiogenesis and fibroblast proliferation compared to all other groups |
| Hoveizi et al. 2023 [11] In vivo (rats) | Male Wistar rats; rat maxillary left first molar pulp (n not specified) | 3D chitosan hydrogel scaffold containing TiO2 nanoparticles + human endometrial stem cells (CS/EnSCs/TiO2) | Control (no treatment), CS alone, CS + stem cells (CS/SCs) | Dentine formation quality and amount (histological evaluation) | CS/EnSCs/TiO2 group showed highest dentine quality and amount; exact numbers not reported | Positive/enhanced dentine formation 8 weeks | Combination of EnSCs + TiO2 NPs + chitosan scaffold accelerates and improves dentine regeneration; suitable for direct pulp capping |
| Zhu et al. 2019 [3] In vitro & in vivo (rats) | Rat model of pulpitis; dental pulp cells in vitro | Silver-doped bioactive glass/chitosan hydrogel (Ag-BG/CS), injectable | Mineral trioxide aggregate (MTA) | Reparative dentin formation, pulp tissue preservation, phosphorylation of p38 and ERK1/2 (MAPK pathway activation) | Reported qualitatively/semi-quantitative | Positive/enhanced reparative dentin formation and pulp preservation Not specified | Ag-BG/CS hydrogel promoted pulp repair and anti-inflammatory effects superior to MTA; enhanced MAPK pathway activation |
| Huang et al. 2025 [5] In vitro | L929 fibroblast cells (in vitro) | ACS-C bioceramic composite (chitosan + C3S powders; AC20-C formulation) | Not reported | Antibacterial efficacy (% inhibition), cell viability (%), biofilm formation inhibition, inhibition zone, compressive strength | Antibacterial > 90%, cell viability > 80%, clear inhibition zone | Positive/effective Not specified | ACS-C materials show strong antibacterial activity, maintain biocompatibility, and have adequate physical properties for pulp capping |
| Anaya-Sampayo et al. 2024 [23] In vitro | Human dental pulp stem cells (DPSC) and OB-DPSC (in vitro) | nHA-CH-GEL-PRF scaffold (nano-hydroxyapatite, chitosan, gelatin, platelet-rich fibrin; lyophilized) | nHA-CH-GEL scaffold without PRF | Cell viability (%), cytotoxicity, growth factor release (PDGF-BB, FGF-B) | Not reported numerically | Positive/improved 24 h (growth factor release), unspecified for cell viability | PRF-supplemented scaffolds increase DPSC and OB-DPSC viability; optimal scaffold properties for bone/pulp tissue regeneration |
| Sornamalar et al. 2024 [16] In vitro | Human dental pulp stem cells (hDPSCs; in vitro) | nPP-CMC scaffold (nano phosphorylated pullulan + carboxymethyl chitosan; 4:5 ratio) | CMC scaffold (group 2), osteogenic medium (group 3) | Cell viability and proliferation (MTT assay, % relative viability) | Significant increase at 21 days vs 7 days (p < 0.05); exact numbers not reported | Positive/improved 0, 7, 14, 21 days | nPP-CMC scaffold shows good bioactivity, biocompatibility, and potential for pulp-dentin regeneration |
| Gould et al. 2024 [10] In vitro | Human dental pulp cells (hDPCs; in vitro) | Chitosan/alginate (C/A) hydrogel with purified bovine pulp and dentin ECM | Hydrogel without ECM (implied control) | Cell proliferation, cytotoxicity, calcium-ion deposition (Alizarin red S), ALP activity, TGF-β expression, chemoattraction | Not reported numerically | Positive/enhanced Not specified | ECM-loaded C/A hydrogels stimulate dental tissue repair, enhance hDPC proliferation, mineralization, ALP activity, and chemoattraction |
| Kumar et al. 2023 [8] In-vitro | Human dental pulp stem cells (hDPSCs), in vitro | Cobalt-incorporated chitosan (CoCH) scaffold; varying cobalt concentrations, optimal 100 μmol/L in 2% CH, 1:1 ratio | Plain chitosan scaffold | Cytotoxicity (XTT assay), cell adhesion (cell-seeding assay), material characterization (SEM, FTIR, XRD) | Non-cytotoxic; enhanced cell adhesion at optimal Co concentration | Positive/CoCH > CH for cell adhesion Not specified | CoCH scaffold at 100 μmol/L cobalt chloride is biocompatible and enhances hDPSC adhesion, promising for dentin-pulp regeneration |
| Gurucharan et al.2023 [13] In vitro | Human dental pulp stem cells (hDPSCs; in vitro) | CSHA scaffold: nano-hydroxyapatite + carboxymethyl chitosan (1:5 w/w) | Biodentine | Cell viability/proliferation (MTT), biomineralization (ALP, ARS, OPN), odontogenic/angiogenic markers (DSPP, VEGF) | Cell viability: no significant difference vs Biodentine; ALP, ARS, OPN: higher than Biodentine at 14 days; DSPP/VEGF: upregulated at 21 days | Positive/enhanced differentiation & biomineralization 7, 14, 21 days | CSHA scaffold supports hDPSC viability and proliferation comparable to Biodentine and enhances odontogenic differentiation and mineralization |
| Ahmed et al. 2023 [1] In vitro | Human dental pulp stem cells (DPSCs; in vitro) | PCL-nano-chitosan scaffold with synthetic hydroxyapatite (PCL-NC-HA) or Mg-substituted hydroxyapatite (PCL-NC-Mg-HA); bioactive materials: MTA, TheraCal LC, Activa Bioactive | Comparison among scaffolds (PCL-NC-HA vs PCL-NC-Mg-HA) and materials (MTA vs TC vs AB) | Odontogenic differentiation (DSPP gene expression), cell viability, proliferation, morphological attachment (SEM, phase contrast) | DSPP expression significantly higher in PCL-NC-Mg-HA; MTA > TC > AB; exact fold changes reported in paper | Positive/enhanced differentiation with PCL-NC-Mg-HA and MTA Multiple timepoints (not specified) | Scaffold composition and MTA most effectively induce odontogenic differentiation of DPSCs |
| Si Wu et al. 2019 [12] In vitro | Human dental pulp stem cells (DPSCs) | Chitosan/β-glycerophosphate (CS/β-GP) hydrogel delivering VEGF (sustained release) | VEGF alone (without hydrogel) | DPSC proliferation (CCK-8 assay), adhesion, viability, odontogenic differentiation | Reported qualitatively/assay data | Positive/enhanced proliferation and odontogenic differentiation Not specified | CS/β-GP hydrogel allowed sustained VEGF release and enhanced odontogenic differentiation of DPSCs compared to VEGF alone |
| Bordini et al. 2019 [9] In vitro | Human dental pulp cells (HDPCs) | Porous chitosan–calcium–aluminate scaffold (CH-AlCa) + 1 nM 1α,25-dihydroxyvitamin D3 (1α,25VD) | Plain chitosan scaffold (CH); HDPCs alone | Odontoblastic differentiation markers: ALP activity, mineralized matrix deposition, DSPP/DMP1 mRNA expression, cell migration | Qualitative/relative comparison | Positive/increased odontoblastic differentiation & cell migration Not specified | CH-AlCa scaffold enhances HDPC chemotaxis and odontoblastic differentiation; synergistic effect with low-dose 1α,25VD |
| Sadeghinia et al. 2019 [7] In vitro | Human dental pulp stem cells (hDPSCs; in vitro) | CS–G/nHA scaffold treated with a-PRP + Fibrin Glue (FG); also FG alone, a-PRP alone, CS–G/nHA alone | CS–G/nHA scaffold alone | Cell adhesion/viability (MTT), osteogenic differentiation (Alizarin red staining, BGLAP, BMP2, RUNX2 expression) | a-PRP–FG/CS–G/nHA group: significantly higher adhesion, mineralization, and osteogenic gene expression; exact numbers not reported | Positive/enhanced 7, 14, 21 days | Composite scaffold treated with a-PRP + FG enhanced adhesion, mineralization, and osteogenic differentiation of h-DPSCs |
| Hashemi-Beni et al. 2018 [4] In vitro | Stem cells from human exfoliated deciduous teeth (SHED; in vitro; n not specified) | PHB/chitosan/nano-bioglass (nBG) scaffold; also PHB, PHB/chitosan, PHB/chitosan/nBG + MTA | PHB scaffold, PHB/chitosan scaffold, MTA alone | Cell viability/proliferation (MTT assay) | PHB/chitosan/nBG scaffold and PHB/chitosan/nBG + MTA showed significantly higher viability at day 7; exact numbers not reported | Positive/enhanced viability 3, 5, 7 days | Scaffolds containing nBG nanoparticles are more biocompatible and promote SHED proliferation better than other scaffolds |
| Study | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | Overall |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Widyastuti et al. 2024 [22] | U | L | U | U | H | U | H | L | L | U | H |
| Widyastuti et al. 2024 [15] | U | L | U | U | H | U | U | L | L | U | U |
| Sularsih et al. 2024 [17] | U | L | U | U | H | U | U | L | L | U | U |
| Hoveizi et al. 2023 [11] | U | L | U | U | H | U | U | L | L | U | U |
| Zhu et al. 2019 [3] | U | U | U | U | U | U | U | L | L | U | H |
| Study (Author, Year) | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | Overall |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sornamalar et al. 2024 [16] | L | L | L | U | H | L | L | L | L | U | L |
| Gurucharan et al. 2022 [13] | L | L | L | U | U | L | L | L | L | U | M |
| Sadeghinia et al. 2019 [7] | L | L | L | U | H | L | L | L | L | U | L |
| Wu et al. 2019 [12] | L | L | L | U | H | L | L | L | L | U | L |
| Hashemi et al. 2018 [4] | L | L | L | U | H | L | L | L | L | U | L |
| Study (Author, Year) | Model Type | Outcome Domain(s) | Comparator Group | Effect Direction (+1 = Positive, 0 = Neutral, –1 = Negative) | Evidence Weight (2 = Mean ± SD; 1 = p-Only; 0.5 = Qualitative) | Primary Finding Summary | Rationale for Code |
|---|---|---|---|---|---|---|---|
| Widyastuti et al. 2024 [15] | In vivo | BMP-2, TGF-β1 (pg/mL) | Ca(OH)2 | 1 | 2 | Nanochitosan ↑ BMP-2 & TGF-β1 vs Ca(OH)2 (p < 0.001) | Numeric ELISA data with ANOVA significance |
| Widyastuti et al. 2024 [22] | In vivo | TGF-β1 (pg/mL), Reparative dentin | Ca(OH)2/control | 1 | 2 | CN ↑ TGF-β1 expression and dentin formation (p < 0.05) | ELISA numeric data provided |
| Sularsih et al. 2024 [14] | In vivo | VEGF (IHC), Fibroblast proliferation | Ca(OH)2/GIC/HA alone | 1 | 1.5 | CH-HA ↑ angiogenesis & fibroblast count (p < 0.05) | Means ± SD reported; semi-quantitative IHC |
| Hoveizi et al. 2023 [11] | In vivo | Dentin formation (histologic score) | CS alone/no treatment | 1 | 1 | CS/EnSCs/TiO2 ↑ dentin quality and amount | Descriptive with qualitative scoring |
| Zhu et al. 2019 [3] | In vivo + in vitro | Reparative dentin, Inflammation/MAPK activation | MTA | 1 | 1 | Ag-BG/CS > MTA for pulp repair & anti-inflammatory effect | Qualitative histology and western blot |
| Sornamalar et al. 2024 [16] | In vitro | Cell viability (MTT %) | CMC scaffold | 1 | 2 | nPP–CMC ↑ relative viability vs CMC (p < 0.05) | Table data numeric and significant |
| Gurucharan et al. 2022 [13] | In vitro | Cell viability (MTT OD 570 nm), ALP, OPN expression | Biodentine | 1 | 2 | CSHA scaffold ↑ differentiation & biomineralization (p <0.05) | Multiple markers improved vs comparator |
| Wu et al. 2019 [12] | In vitro | Cell proliferation (CCK-8 OD 450 nm), VEGF release | NC (no hydrogel) | 1 | 2 | CS/β-GP hydrogel ↑ proliferation and VEGF sustained release | Quantitative mean ± SD visible on figure |
| Sadeghinia et al. 2019 [7] | In vitro | Cell viability (MTT %)/Mineralization | Control | 1 | 2 | a-PRP–FG/CS–G/nHA ↑ adhesion & viability (p < 0.0001) | Clear quantitative difference; replicates = 3 |
| Hashemi-Beni et al. 2018 [4] | In vitro | Cell viability (MTT OD 540 nm) | PHB scaffold/control | 1 | 2 | PHB/CH/nBG + MTA ↑ cell viability vs control (p < 0.05) | Quantitative mean ± SD reported; consistent significance |
| Certainty-Assessment Criteria | Evaluation | ||
|---|---|---|---|
| Number of studies | 5 animal RCTs (≈253 samples) + 11 in vitro studies | ||
| Study design | Controlled preclinical experiments (no human data) | ||
| Risk of bias | Serious—randomization and blinding rarely reported | ||
| Inconsistency | Serious—variation in formulations, models, and assays | ||
| Indirectness | Very serious—animal and in vitro evidence only; surrogate endpoints | ||
| Imprecision | Serious—small sample sizes and incomplete variance data | ||
| Other considerations | Publication bias likely—all included studies reported positive effects | ||
| Outcome | Comparator | Relative Effect (95% CI) | Absolute Effect (95% CI) |
| Pulp healing and reparative dentin formation with chitosan-based nanomaterials | Conventional pulp-capping agents (Ca(OH)2, MTA, GIC) | Not estimable—no pooled quantitative data | Descriptive synthesis only |
| Overall certainty of evidence (GRADE-Preclinical) | ⨁⨁◯◯ Low to Very Low | ||
| Importance | Biologically promising preclinical results but limited translational certainty for human use | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alqahtani, S.A.; Alamri, M.; Alwadai, G.; Abogazalah, N.N.; Mathew, V.B.; Joseph, B. Chitosan-Based Nanoparticles and Biomaterials for Pulp Capping and Regeneration: A Systematic Review with Quantitative and Evidence-Mapping Synthesis. Biomimetics 2025, 10, 822. https://doi.org/10.3390/biomimetics10120822
Alqahtani SA, Alamri M, Alwadai G, Abogazalah NN, Mathew VB, Joseph B. Chitosan-Based Nanoparticles and Biomaterials for Pulp Capping and Regeneration: A Systematic Review with Quantitative and Evidence-Mapping Synthesis. Biomimetics. 2025; 10(12):822. https://doi.org/10.3390/biomimetics10120822
Chicago/Turabian StyleAlqahtani, Saleh Ali, Mohammad Alamri, Ghadeer Alwadai, Naif N. Abogazalah, Vinod Babu Mathew, and Betsy Joseph. 2025. "Chitosan-Based Nanoparticles and Biomaterials for Pulp Capping and Regeneration: A Systematic Review with Quantitative and Evidence-Mapping Synthesis" Biomimetics 10, no. 12: 822. https://doi.org/10.3390/biomimetics10120822
APA StyleAlqahtani, S. A., Alamri, M., Alwadai, G., Abogazalah, N. N., Mathew, V. B., & Joseph, B. (2025). Chitosan-Based Nanoparticles and Biomaterials for Pulp Capping and Regeneration: A Systematic Review with Quantitative and Evidence-Mapping Synthesis. Biomimetics, 10(12), 822. https://doi.org/10.3390/biomimetics10120822