Hydrogel on a Smart Nanomaterial Interface to Carry Therapeutics for Digitalized Glioma Treatment
Abstract
1. Introduction
2. Result and Discussion
2.1. Analysis of Hydrogel as a Drug Carrier for the Treatment of Glioma
2.2. Hydrogel Application for the Blood–Brain Barrier (BBB)
2.3. Stem Cell Application
2.4. Hydrogel Carrier for Gene Therapy
2.5. Pros and Cons of the Hydrogel
2.6. Hydrogel for the Biosensor of Tissue Engineering
3. Conclusions
4. Methods
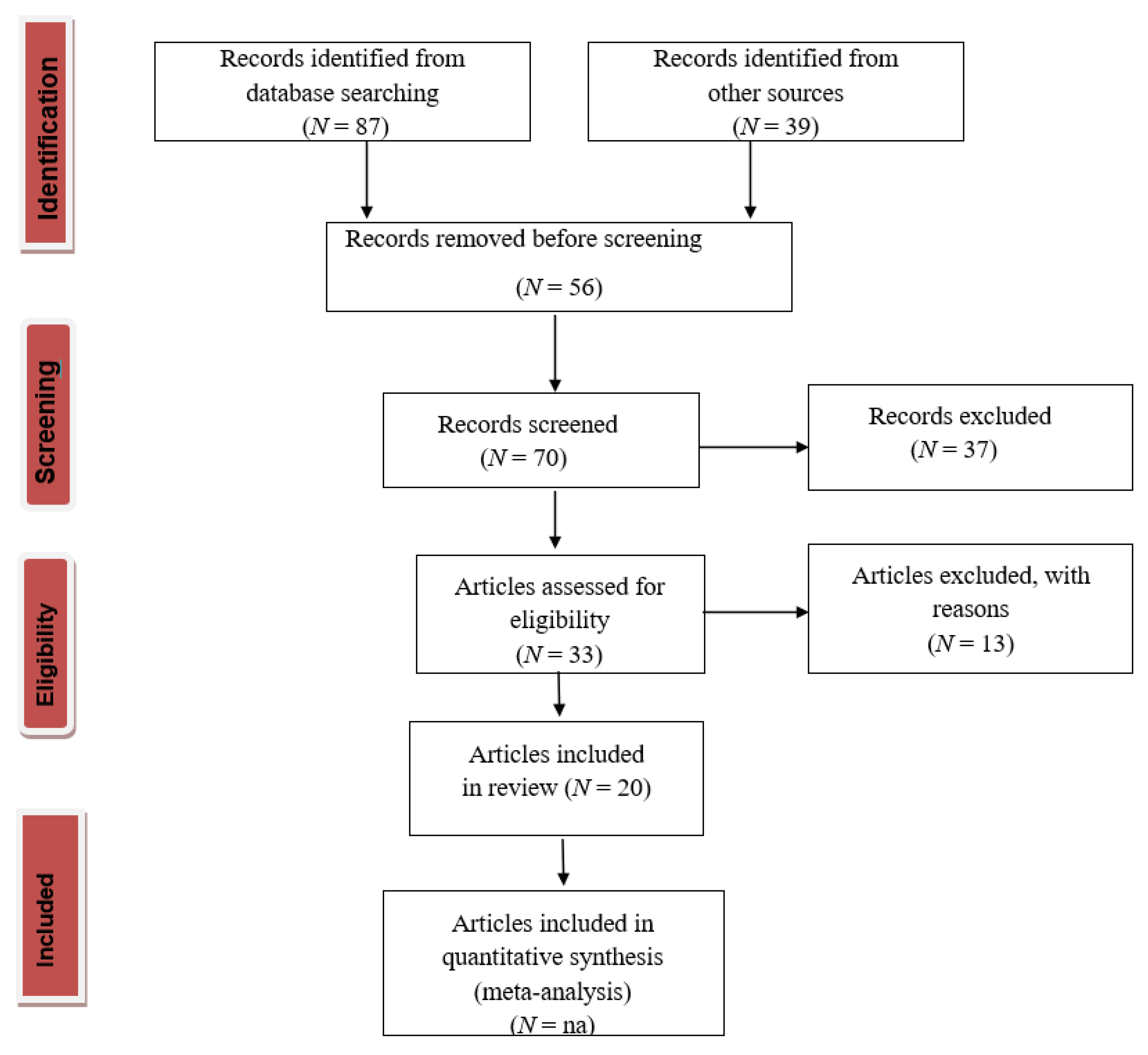
4.1. PRISMA Statement (Preferred Reporting Items for Systematic Reviews and Meta-Analyses)
4.2. Research Process
- i.
- Publication years were between 2020 and 2022.
- ii.
- The keywords ‘‘hydrogel” AND ‘‘glioma cancer” had to appear in the title and abstract.
- iii.
- They had to be scientific indexed papers only.
4.3. Screening
- i.
- “Primary articles” research papers appeared in the peer-reviewed literature and reported original data or results based on observations and experiments.
- ii.
- “Review” papers summarized the understanding of hydrogels as carriers and pathways of drug delivery in glioma cancer.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- He, Z.; Charleton, C.; Devine, R.W.; Kelada, M.; Walsh, J.M.D.; Conway, G.E.; Gunes, S.; Mondala, J.R.M.; Tian, F.; Tiwari, B.; et al. Enhanced pyrazolopyrimidinones cytotoxicity against glioblastoma cells activated by ROS-Generating cold atmospheric plasma. Eur. J. Med. Chem. 2021, 224, 113736. [Google Scholar] [CrossRef]
- Brachi, G.; Ruiz-Ramirez, J.; Dogra, P.; Wang, Z.; Cristini, V.; Ciardelli, G.; Rostomily, R.C.; Ferrari, M.; Mikheev, A.M.; Blanco, E.; et al. Intratumoral injection of hydrogel-embedded nanoparticles enhances retention in glioblastoma. Nanoscale 2020, 12, 23838–23850. [Google Scholar] [CrossRef]
- Catarina, F.; Andreia, C.; Ligia, O.; Rita Costa, L.; Paulo, L.; Bruno, C. Claudia, Current Standards of Care in Glioblastoma Therapy. In Glioblastoma; Vleeschouwer, S.D., Ed.; Codon Publications: Singapore, 2017; pp. 197–242. [Google Scholar]
- Basso, J.; Miranda, A.; Nunes, S.; Cova, T.; Sousa, J.; Vitorino, C.; Pais, A. Hydrogel-Based Drug Delivery Nanosystems for the Treatment of Brain Tumors. Gels 2018, 4, 62. [Google Scholar] [CrossRef]
- Pandey, N.; Anastasiadis, P.; Carney, C.P.; Kanvinde, P.P.; Woodworth, G.F.; Winkles, J.A.; Kim, A.J. Nanotherapeutic treatment of the invasive glioblastoma tumor microenvironment. Adv. Drug Deliv. Rev. 2022, 188, 114415. [Google Scholar] [CrossRef]
- Van Solinge, T.S.; Nieland, L.; Chiocca, E.A.; Broekman, M.L.D. Advances in local therapy for glioblastoma-taking the fight to the tumour. Nat. Rev. Neurol. 2022, 18, 221–236. [Google Scholar] [CrossRef]
- Nehra, M.; Uthappa, U.T.; Kumar, V.; Kumar, R.; Dixit, C.; Dilbaghi, N.; Mishra, Y.K.; Kumar, S.; Kaushik, A. Nanobiotechnology-assisted therapies to manage brain cancer in personalized manner. J. Control. Release 2021, 338, 224–243. [Google Scholar] [CrossRef]
- Fang, Y.; Yang, X.; Lin, Y.; Shi, J.; Prominski, A.; Clayton, C.; Ostroff, E.; Tian, B. Dissecting Biological and Synthetic Soft–Hard Interfaces for Tissue-Like Systems. Chem. Rev. 2022, 122, 5233–5276. [Google Scholar] [CrossRef]
- Fang, Y.; Han, E.; Zhang, X.X.; Jiang, Y.; Lin, Y.; Shi, J.; Wu, J.; Meng, L.; Gao, X.; Griffin, P.J.; et al. Dynamic and Programmable Cellular-Scale Granules Enable Tissue-like Materials. Matter 2020, 2, 948–964. [Google Scholar] [CrossRef]
- Fang, Y.; Tian, B. Curving neural nanobioelectronics. Nat. Nanotechnol. 2019, 14, 733–735. [Google Scholar] [CrossRef]
- Christensen, S.C.; Hudecz, D.; Jensen, A.; Christensen, S.; Nielsen, M.S. Basigin Antibodies with Capacity for Drug Delivery Across Brain Endothelial Cells. Mol. Neurobiol. 2021, 58, 4392–4403. [Google Scholar] [CrossRef]
- Preman, N.K.; Barki, P.R.; Vijayan, A.; Sandesh, G.; Sanjeeva, S.G.; Johnson, R.P. Recent developments in stimuli-responsive polymer nanogels for drug delivery and diagnostics: A review. Eur. J. Pharm. Biopharm. 2020, 157, 121–153. [Google Scholar] [CrossRef] [PubMed]
- Javed, B.; Zhao, X.; Cui, D.; Curtin, J.; Tian, F. Enhanced Anticancer Response of Curcumin- and Piperine-Loaded Lignin-g-p (NIPAM-co-DMAEMA) Gold Nanogels against U-251 MG Glioblastoma Multiforme. Biomedicines 2021, 9, 1516. [Google Scholar] [CrossRef]
- Martinez-Moro, M.; Jenczyk, J.; Giussi, J.M.; Jurga, S.; Moya, S.E. Kinetics of the thermal response of poly(N-isopropylacrylamide co methacrylic acid) hydrogel microparticles under different environmental stimuli: A time-lapse NMR study. J. Colloid Interface Sci. 2020, 580, 439–448. [Google Scholar] [CrossRef]
- Xu, B.; Huo, Z.; Huang, H.; Ji, W.; Bian, Z.; Jiao, J.; Sun, J.; Shao, J. The expression and prognostic value of the epidermal growth factor receptor family in glioma. BMC Cancer 2021, 21, 451. [Google Scholar] [CrossRef] [PubMed]
- Wanjale, M.V.; Sunil-Jaikumar, V.; Sivakumar, K.C.; Ann-Paul, R.; James, J.; Kumar, G. S V. Supramolecular Hydrogel Based Post-Surgical Implant System for Hydrophobic Drug Delivery Against Glioma Recurrence. Int. J. Nanomed. 2022, 17, 2203–2224. [Google Scholar] [CrossRef]
- Li, Q.; Shao, X.; Dai, X.; Guo, Q.; Yuan, B.; Liu, Y.; Jiang, W. Recent trends in the development of hydrogel therapeutics for the treatment of central nervous system disorders. NPG Asia Mat. 2022, 14, 14. [Google Scholar] [CrossRef]
- Kasapidou, P.M.; De-Montulld, E.L.; Dembele, K.P.; Mutel, A.; Desrues, L.; Gubala, V.; Castel, H. Hyaluronic acid-based hydrogels loaded with chemoattractant and anticancer drug-new formulation for attracting and tackling glioma cells. Soft Matter 2021, 17, 10846–10861. [Google Scholar] [CrossRef]
- McCrorie, P.; Vasey, C.E.; Smith, S.J.; Marlow, M.; Alexander, C.; Rahman, R. Biomedical engineering approaches to enhance therapeutic delivery for malignant glioma. J. Control. Release 2020, 328, 917–931. [Google Scholar] [CrossRef]
- Dundar, B.; Markwell, S.M.; Sharma, N.V.; Olson, C.L.; Mukherjee, S.; Brat, D.J. Methods for in vitro modeling of glioma invasion: Choosing tools to meet the need. Glia 2020, 68, 2173–2191. [Google Scholar] [CrossRef] [PubMed]
- Aquib, M.; Juthi, A.Z.; Farooq, M.A.; Ali, M.G.; Janabi, A.H.W.; Bavi, S.; Banerjee, P.; Bhosale, R.; Bavi, R.; Wang, B. Advances in local and systemic drug delivery systems for post-surgical cancer treatment. J. Mater. Chem. B 2020, 8, 8507–8518. [Google Scholar] [CrossRef]
- Cao, X.; Li, S.; Chen, W.; Lu, H.; Ye, L.; Min, Z.; Sun, S.; Teng, C.; Yin, H.; Zhang, Q.; et al. Multifunctional Hybrid Hydrogel System Enhanced the Therapeutic Efficacy of Treatments for Postoperative Glioma. ACS Appl. Mater. 2022, 14, 27623–27633. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ye, L.; He, W.; Teng, C.; Sun, S.; Lu, H.; Li, S.; Lv, L.; Cao, X.; Yin, H.; et al. In situ targeting nanoparticles-hydrogel hybrid system for combined chemo-immunotherapy of glioma. J. Control. Release 2022, 345, 786–797. [Google Scholar] [CrossRef] [PubMed]
- Parkins, C.C.; McAbee, J.H.; Ruff, L.; Wendler, A.; Mair, R.; Gilbertson, R.J.; Watts, C.; Scherman, O.A. Mechanically matching the rheological properties of brain tissue for drug-delivery in human glioblastoma models. Biomaterials 2021, 276, 120919. [Google Scholar] [CrossRef] [PubMed]
- Erthal, L.C.S.; Gobbo, O.L.; Ruiz-Hernandez, E. Biocompatible copolymer formulations to treat glioblastoma multiforme. Acta Biomater. 2021, 121, 89–102. [Google Scholar] [CrossRef]
- Moore, K.M.; Murthy, A.B.; Graham-Gurysh, E.G.; Hingtgen, S.D.; Bachelder, E.M.; Ainslie, K.M. Polymeric Biomaterial Scaffolds for Tumoricidal Stem Cell Glioblastoma Therapy. ACS Biomater. Sci. Eng. 2020, 6, 3762–3777. [Google Scholar] [CrossRef]
- Cha, J.; Kim, P. Cancer Cell-Sticky Hydrogels to Target the Cell Membrane of Invading Glioblastomas. ACS Appl. Mater. Interfaces 2021, 13, 31371–31378. [Google Scholar] [CrossRef]
- Chen, S.; Qiu, Q.; Wang, D.; She, D.; Yin, B.; Chai, M.; He, H.; Heo, D.N.; Wang, J. Long acting carmustine loaded natural extracellular matrix hydrogel for inhibition of glioblastoma recurrence after tumor resection. Front. Chem. Sci. Eng. 2022, 16, 536–545. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, C.; Li, A.; Jing, W.; Sun, P.; Huang, X.; Liu, Y.; Zhang, S.; Du, W.; Zhang, R.; et al. Immunostimulant hydrogel for the inhibition of malignant glioma relapse post-resection. Nat. Nanotechnol. 2021, 16, 538–548. [Google Scholar] [CrossRef]
- Zhu, Y.; Jia, J.; Zhao, G.; Huang, X.; Wang, L.; Zhang, Y.; Zhang, L.; Konduru, N.; Xie, J.; Yu, R.; et al. Multi-responsive nanofibers composite gel for local drug delivery to inhibit recurrence of glioma after operation. J. Nanobiotechnol. 2021, 19, 198. [Google Scholar] [CrossRef]
- Wiranowska, M.; Singh, R.; Falahat, R.; Williams, E.; Johnson, J.O.; Alcantar, N. Preferential drug delivery to tumor cells than normal cells using a tunable niosome–chitosan double package nanodelivery system: A novel in vitro model. Cancer Nanotechnol. 2020, 11, 3. [Google Scholar] [CrossRef]
- Yao, C.Y.; Qin, Y.; Fan, W.T.; Yan, L.P.; Chen, M.; Liu, Y.L.; Huang, W.H. A three-dimensional electrochemical biosensor integrated with hydrogel for cells culture and lactate release monitoring. J. Electroanal.Chem. 2022, 915, 116338. [Google Scholar] [CrossRef]
- Newland, B.; Starke, J.; Bastiancich, C.; Gonçalves, D.P.N.; Bray, L.J.; Wang, W.; Werner, C. Well-Defined Polyethylene Glycol Microscale Hydrogel Blocks Containing Gold Nanorods for Dual Photothermal and Chemotherapeutic Therapy. Pharmaceutics 2022, 14, 551. [Google Scholar] [CrossRef]
- Babaei, M.; Davoodi, J.; Dehghan, R.; Zahiri, M.; Abnous, K.; Taghdisi, S.M.; Ramezani, M.; Alibolandi, M. Thermosensitive composite hydrogel incorporated with curcumin-loaded nanopolymersomes for prolonged and localized treatment of glioma. J. Drug Deliv. Sci. Technol. 2020, 59, 101885. [Google Scholar] [CrossRef]
- Costagliola-Di-Polidoro, A.; Zambito, G.; Haeck, J.; Mezzanotte, L.; Lamfers, M.; Netti, P.A.; Torino, E. Theranostic Design of Angiopep-2 Conjugated Hyaluronic Acid Nanoparticles (Thera-ANG-cHANPs) for Dual Targeting and Boosted Imaging of Glioma Cells. Cancers 2021, 13, 503. [Google Scholar] [CrossRef]
- Bastiancich, C.; Bianco, J.; Vanvarenberg, K.; Ucakar, B.; Joudiou, N.; Gallez, B.; Bastiat, G.; Lagarce, F.; Preat, V.; Danhier, F. Injectable nanomedicine hydrogel for local chemotherapy of glioblastoma after surgical resection. J. Control. Release 2017, 264, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Semmler-Behnke, M.; Kreyling, W.G.; Lipka, J.; Fertsch, S.; Wenk, A.; Takenaka, S.; Schmid, G.; Brandau, W. Biodistribution of 1.4- and 18-nm gold particles in rats. Small 2008, 4, 2108–2111. [Google Scholar] [CrossRef] [PubMed]
- Connolly, N.P.; Galisteo, R.; Xu, S.; Bar, E.E.; Peng, S.; Tran, N.L.; Ames, H.M.; Kim, A.J.; Woodworth, G.F.; Winkles, J.A. Elevated fibroblast growth factor-inducible 14 expression transforms proneural-like gliomas into more aggressive and lethal brain cancer. Glia 2021, 69, 2199–2214. [Google Scholar] [CrossRef] [PubMed]
- Scully, S.; Francescone, R.; Faibish, M.; Bentley, B.; Taylor, S.L.; Oh, D.; Schapiro, R.; Moral, L.; Yan, W.; Shao, R. Transdifferentiation of glioblastoma stem-like cells into mural cells drives vasculogenic mimicry in glioblastomas. J. Neurosci. 2012, 32, 12950–12960. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, S.; Tian, F.; Stoeger, T.; Kreyling, W.; De-La-Fuente, J.M.; Grazu, V.; Borm, P.; Estrada, G.; Ntziachristos, V.; Razansky, D. Multifunctional Nanocarriers for diagnostics, drug delivery and targeted treatment across blood-brain barrier: Perspectives on tracking and neuroimaging. Part. Fibre Toxicol. 2010, 7, 3. [Google Scholar] [CrossRef]
- Li, Z.; Li, M.; Xia, P.; Wang, L.; Lu, Z. Targeting long non-coding RNA PVT1/TGF-β/Smad by p53 prevents glioma progression. Cancer Biol. Ther. 2022, 23, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.S.; Harford, J.B.; Moghe, M.; Slaughter, T.; Doherty, C.; Chang, E.H. A tumor-targeting nanomedicine carrying the p53 gene crosses the blood–brain barrier and enhances anti-PD-1 immunotherapy in mouse models of glioblastoma. Int. J. Cancer 2019, 145, 2535–2546. [Google Scholar] [CrossRef]
- Zhang, F.; Stephan, S.B.; Ene, C.I.; Smith, T.T.; Holland, E.C.; Stephan, M.T. Nanoparticles That Reshape the Tumor Milieu Create a Therapeutic Window for Effective T-cell Therapy in Solid Malignancies. Cancer Res. 2018, 78, 3718–3730. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, G.; Landrain, T.E.; Jaramillo, A. De novo automated design of small RNA circuits for engineering synthetic riboregulation in living cells. Comput. Biol. Chem. 2012, 109, 15271–15276. [Google Scholar] [CrossRef] [PubMed]
- Scioli, M.G.; Terriaca, S.; Fiorelli, E.; Storti, G.; Fabbri, G.; Cervelli, V.; Orlandi, A. Extracellular Vesicles and Cancer Stem Cells in Tumor Progression: New Therapeutic Perspectives. Int. J. Mol. Sci. 2021, 22, 10572. [Google Scholar] [CrossRef]
- Lovrak, M.; Hendriksen, W.E.J.; Maity, C.; Mytnyk, S.; Steijn, V.V.; Eelkema, R.; Esch, J.H.V. Free-standing supramolecular hydrogel objects by reaction-diffusion. Nat. Commun. 2017, 8, 15317. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Mishchenko, A.; Carvalho, A.; Neto, A.H.C. 2D materials and van der Waals heterostructures. Science 2016, 353, 9439. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Gao, W.; Yin, Z. Materials, devices and systems of soft bioelectronics for precision therapy. Adv. Healthc. Mater. 2017, 6, 1700017. [Google Scholar] [CrossRef] [PubMed]
- Mehrali, M.; Bagherifard, S.; Akbari, M.; Thakur, A.; Mirani, B.; Mehrali, M.; Hasany, M.; Orive, G.; Das, P.; Emneus, J. Blending electronics with the human body: A pathway toward a cybernetic future. Adv. Sci. 2018, 5, 1700931. [Google Scholar] [CrossRef] [PubMed]
- Someya, T.; Bao, Z.; Malliaras, G.G. The rise of plastic bioelectronics. Nature 2016, 540, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.D.; Melosh, N.A. Electronic and ionic materials for neurointerfaces. Adv. Funct. Mater. 2018, 28, 1704335. [Google Scholar] [CrossRef]
- Rebelo, R.; Barbosa, A.I.; Correlo, V.M.; Reis, R.L. An outlook on implantable biosensors for personalized medicine. Engineering 2021, 7, 1696–1699. [Google Scholar] [CrossRef]
- Ullah, F.; Othman, M.B.H.; Javed, F.; Ahmad, Z.; Akil, H.M. Classification, processing and application of hydrogels: A review. Mater. Sci. Eng. C 2015, 57, 414–433. [Google Scholar] [CrossRef]
- Liu, J.; Lin, S.; Li, W.; Zhao, Y.; Liu, D.; He, Z.; Wang, D.; Lei, M.; Hong, B.; Wu, H. Ten-Hour Stable Noninvasive Brain-Computer Interface Realized by Semidry Hydrogel-Based Electrodes. Research 2022, 10, 9830457. [Google Scholar] [CrossRef] [PubMed]
- Prince, E.; Kumacheva, E. Design and applications of man-made biomimetic fibrillar hydrogels. Nat. Rev. Mater. 2019, 4, 99–115. [Google Scholar] [CrossRef]
- Lutolf, M.P.; Hubbell, J.A. Synthetic biomaterials as instructive extracellular microenvironments for morphogenesis in tissue engineering. Nat. Biotechnol. 2005, 23, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, K.; Pan, Z.; Guan, E.; Ge, S.R.; Liu, Y.J.; Nakamura, T.; Ren, X.D.; Rafailovich, M.; Clark, R.A.F. Cell adaptation to a physiologically relevant ECM mimic with different viscoelastic properties. Biomaterials 2007, 28, 671–679. [Google Scholar] [CrossRef] [PubMed]
- Ridley, A.J.; Schwartz, M.A.; Burridge, K.; Firtel, R.A.; Ginsberg, M.H.; Borisy, G.; Parsons, J.T.; Horwitz, A.R. Cell migration: Integrating signals from front to back. Science 2003, 302, 1704–1709. [Google Scholar] [CrossRef] [PubMed]
- Friedl, P.; Gilmour, D. Collective cell migration in morphogenesis, regeneration and cancer. Nat. Rev. Mol. Cell Bio. 2009, 10, 445–457. [Google Scholar] [CrossRef]
- Kim, S.; Park, S.; Na, B.; Yoon, S. Spiking-yolo: Spiking neural network for energyefficient object detection. In Proceedings of the AAAI Conference on Artificial Intelligence, New York, NY, USA, 7 February 2020. [Google Scholar]
- Yang, J.J.; Strukov, D.B.; Stewart, D.R. Memristive devices for computing. Nat. Nanotechnol. 2013, 8, 13–24. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
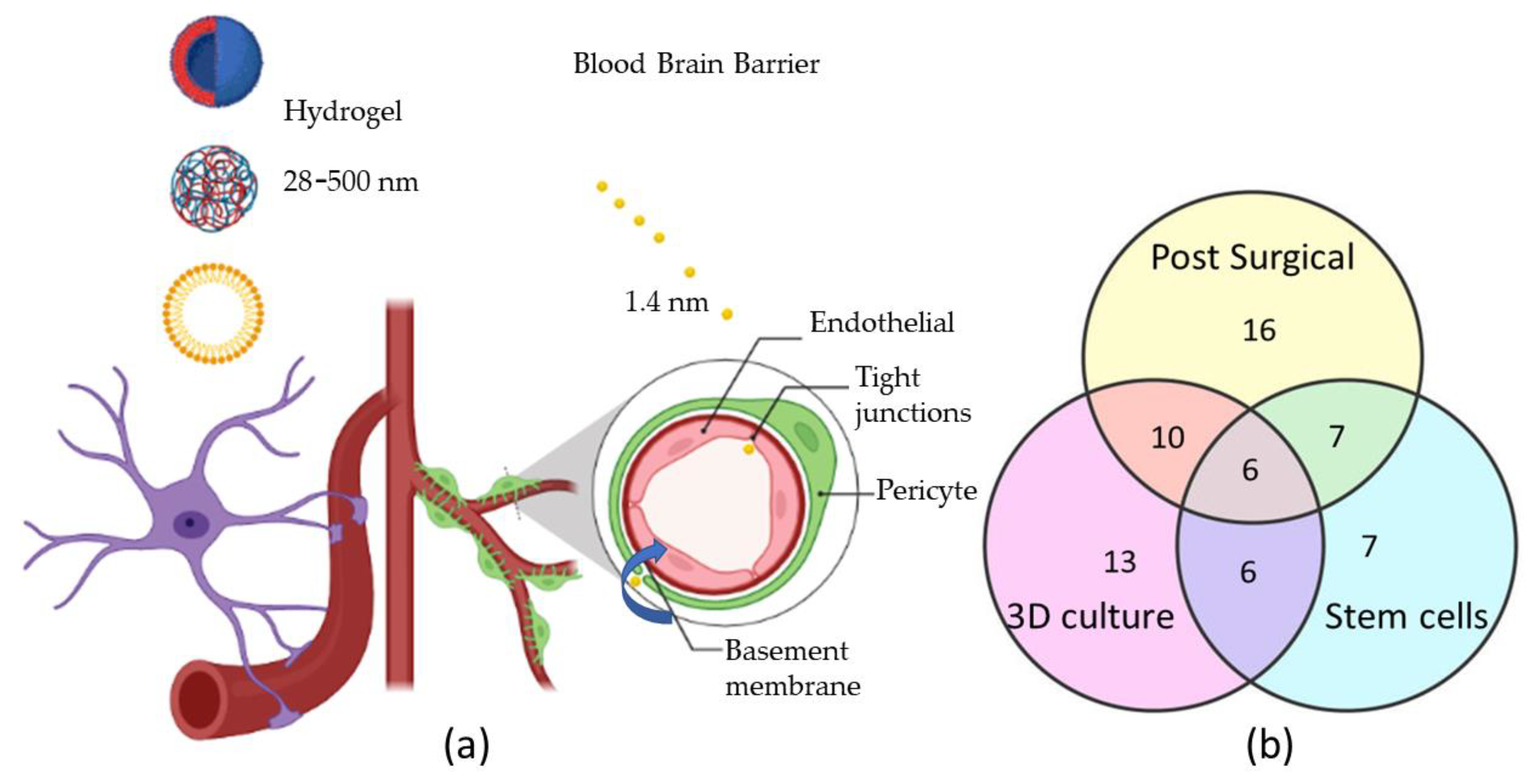
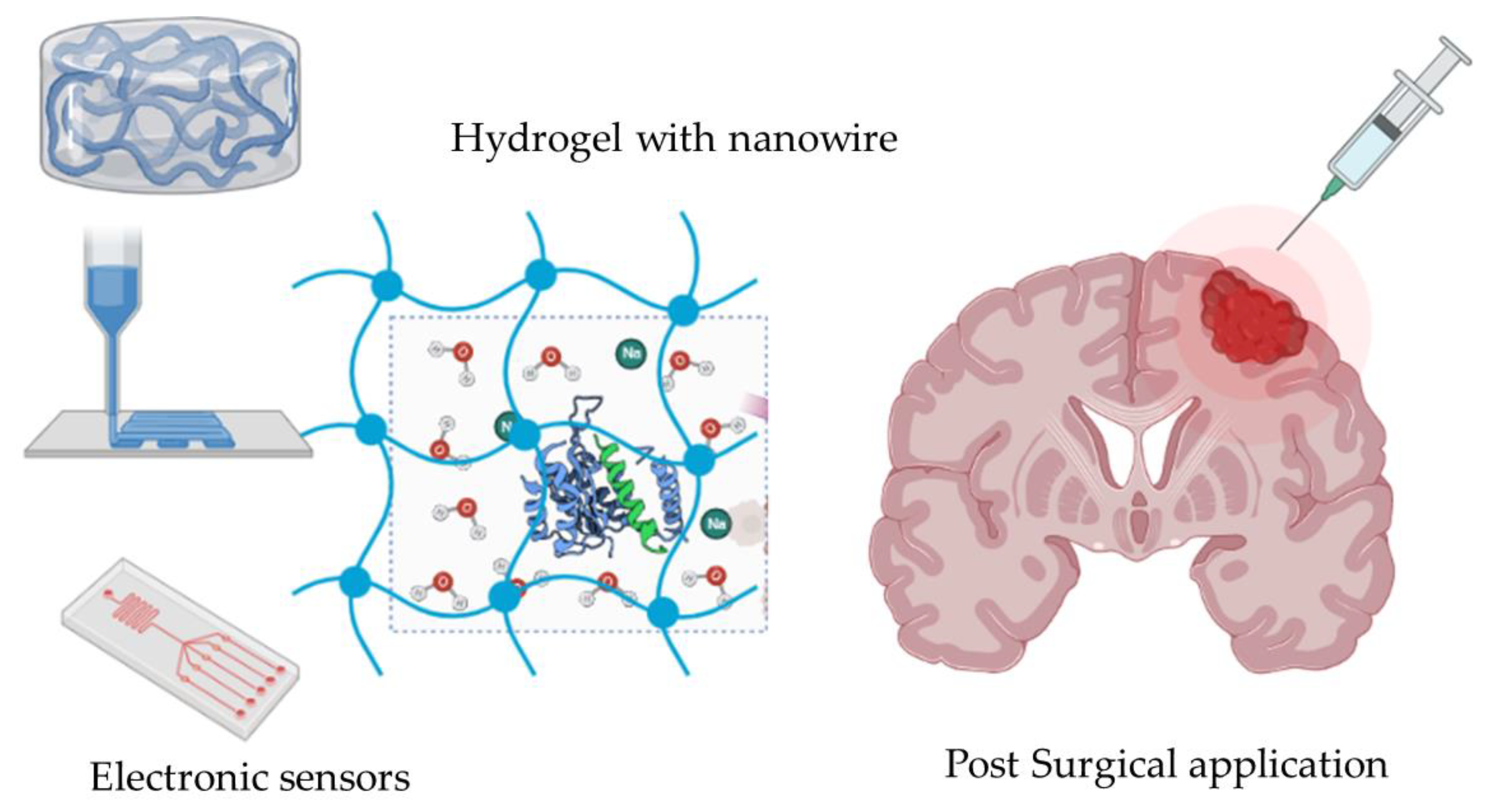
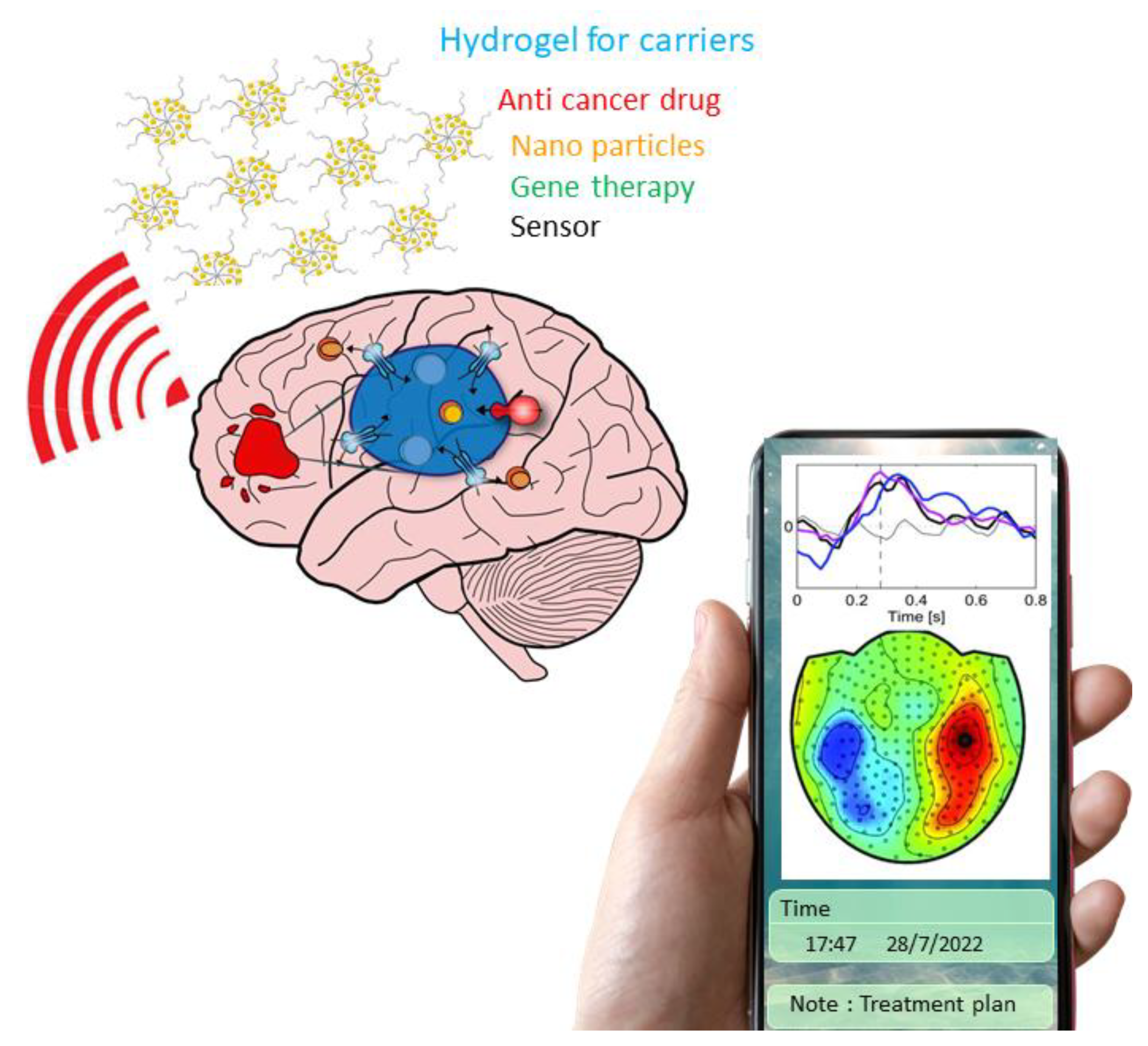
| Hydrogel Type | Post-Surgical | Cell Culture | Glioma Stem Cell | Drug Delivered | Particle Size (nm) | Sensor | Pros | Cons | Reference |
|---|---|---|---|---|---|---|---|---|---|
| PCL-PEG | Yes | 3D | Yes | Carmustine, | 597 | No | Easy synthesis, safe effective treatment | N/A | [15] |
| Hyaluronic acid/sodium alginate | Yes | 3D | Yes | Histamine | N/A | No | Sufficient filling of irregular damage sites, provision of 3D cell growth environment | N/A | [16] |
| PTX PLGA-NPs-loaded | Yes | 3D | Yes | Paclitaxel | N/A | No | N/A | N/A | [17] |
| PLGA-PEG-PLGA | Yes | 3D | Yes | Temozolomide | 35 | No | Enhance cellular internalization in GBM cells, improve low biological stability and drug’s efficacy | Undesirable monocyte migration, localization to systemic tissue | [18] |
| Hyaluronic acid | Yes | 3D | Yes | No | N/A | No | Ease, low cost, and rapid setup | Difficult real time monitor and genetic manipulations | [19] |
| PVA | Yes | 3D | Yes | Gemcitabine | N/A | No | Controlled and constant drug release, enhanced dosage at the targeted site | N/A | [20] |
| CP and CL@ RNP PTXb | Yes | 3D | No | Luminol | 80 | No | N/A | N/A | [21] |
| PNPPTX and MNP CpG | Yes | 3D | No | Paclitaxel | 127 | No | Quickly cross-linked to form a hydrogel, cellular targeted nanoparticles reach the lesion directly | N/A | [22] |
| Hyaluronic acid/Cucurbit | Yes | 3D | No | Doxorubicin | 100 | No | A higher survival rate, improved drug bioavailability | N/A | [23] |
| PCLA-PEG-PCLA | Yes | 3D | No | Paclitaxel | 500 | No | Solubilization of hydrophobic drugs, implanted after surgical resection of a tumor | Difficult synchronized release of components, lack safety data | [24] |
| Gelatin | Yes | 2D | Yes | Thymidine kinase | 283 | No | High glioma stem cell loading capacity, more shielded from hostile resection | Increased intracranial pressure, toxic degradation byproducts | [25] |
| CSH: TCEP-immobilized agarose | Yes | 2D | No | TCEP | N/A | No | Improved efficacy and targetability of soluble biomolecules | low therapeutic efficiency, inevitable drug resistance | [26] |
| Pig diemel, N,N-cyclohexyl carbide diimine, N-hydroxybutylphthalimine | Yes | 2D | No | Carmustine | 275 | No | Readily adopted by body system, slow release, continuously released by hydrogel | N/A | [27] |
| Fmoc-F–FF-DOPA | Yes | 2D | No | CXCL10 | 164 | No | Low IDO1 expression, high IDO1 protein levels in tumor tissues | N/A | [28] |
| Triglycerol monostearate/PPS60 | Yes | 2D | No | Temozolomide | N/A | No | Good biocompatibility, broad cancer treatment application | N/A | [29] |
| Chitosan-b-glycerophosphate | Yes | 2D | No | Paclitaxel | 100 | No | Protects niosomes against external tonicity fluctuation, prevents uncontrollable release of paclitaxel | N/A | [30] |
| Collagen | No | 3D | No | No | N/A | Yes * | High sensitivity, nondestructive, real-time monitor of reactive oxygen species from microglial cells | N/A | [31] |
| PEGDA | No | 3D | No | Doxorubicin | 28 | No | Raise local dose drug at tumor site, will not pass blood–brain barrier | N/A | [32] |
| PCLA-PEG-PCLA | No | 3D | No | Curcumin | 189 | No | Increased patient compliance, decreased side effects | N/A | [33] |
| Hyaluronic acid | No | 2D | No | Irinotecan | 107 | No | N/A | N/A | [34] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, X.; Javed, B.; Tian, F.; Liu, K. Hydrogel on a Smart Nanomaterial Interface to Carry Therapeutics for Digitalized Glioma Treatment. Gels 2022, 8, 664. https://doi.org/10.3390/gels8100664
Zhao X, Javed B, Tian F, Liu K. Hydrogel on a Smart Nanomaterial Interface to Carry Therapeutics for Digitalized Glioma Treatment. Gels. 2022; 8(10):664. https://doi.org/10.3390/gels8100664
Chicago/Turabian StyleZhao, Xinyi, Bilal Javed, Furong Tian, and Kangze Liu. 2022. "Hydrogel on a Smart Nanomaterial Interface to Carry Therapeutics for Digitalized Glioma Treatment" Gels 8, no. 10: 664. https://doi.org/10.3390/gels8100664
APA StyleZhao, X., Javed, B., Tian, F., & Liu, K. (2022). Hydrogel on a Smart Nanomaterial Interface to Carry Therapeutics for Digitalized Glioma Treatment. Gels, 8(10), 664. https://doi.org/10.3390/gels8100664










