Genetic Diversity of Epichloë Endophytes Associated with Brachypodium and Calamagrostis Host Grass Genera including Two New Species
Abstract
1. Introduction
2. Materials and Methods
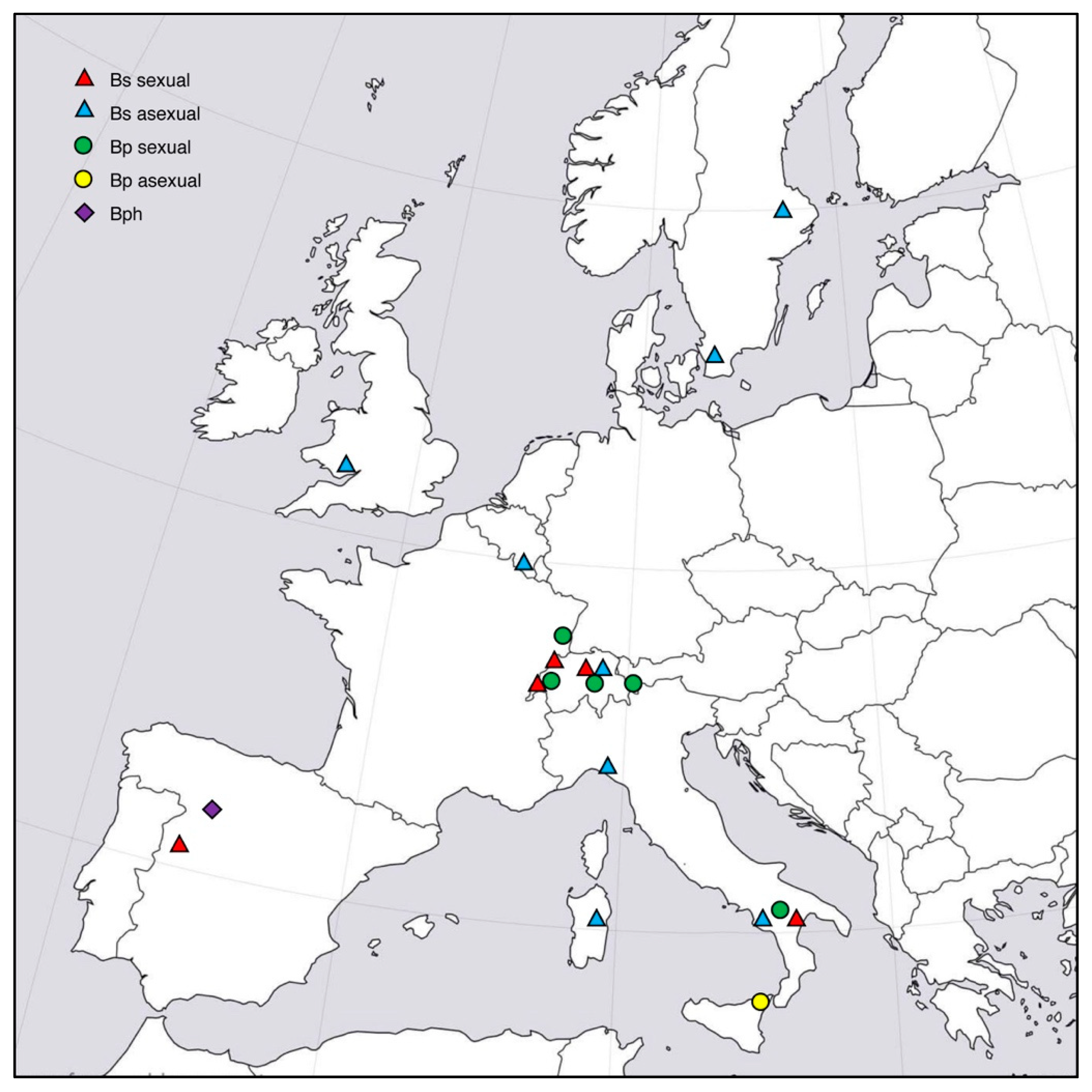
2.1. Host Grass Species
2.2. Endophyte Sampling and Isolation
2.3. Microsatellite Analyses
2.4. DNA Sequencing and Phylogenetic Analyses
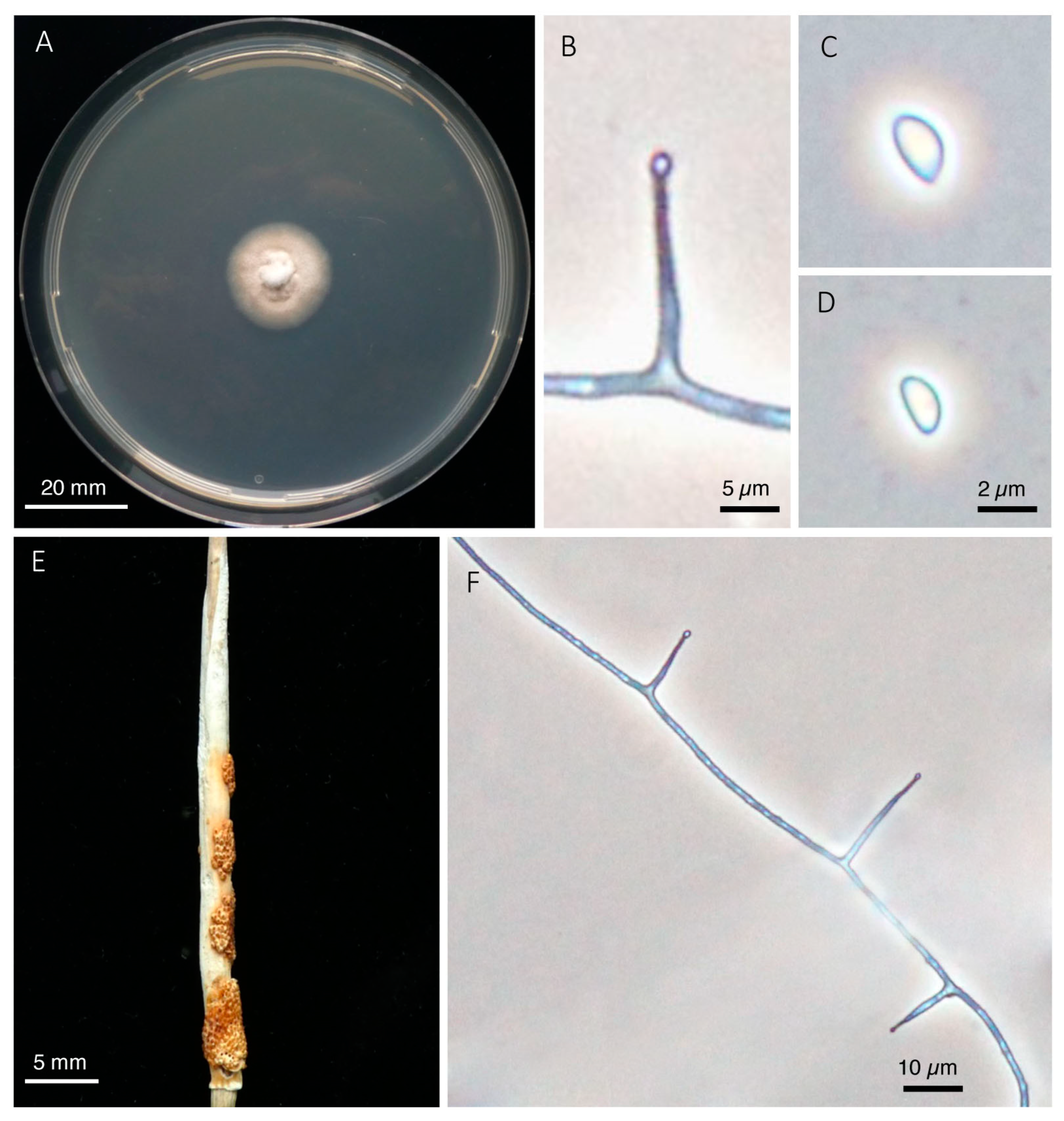
2.5. Morphological Examinations
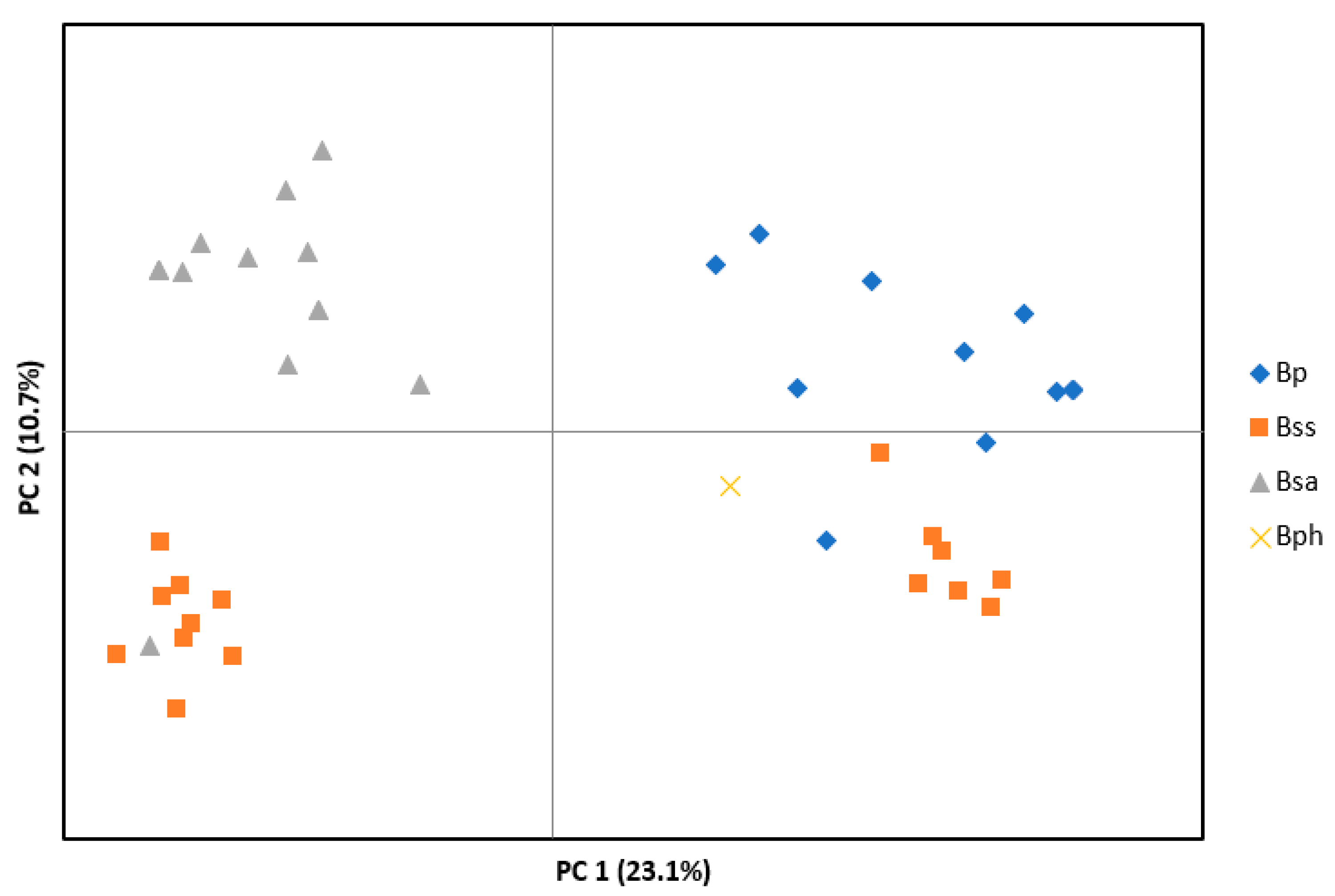
3. Results
3.1. Microsatellite Data
3.2. tubB Phylogeny
3.3. tefA Phylogeny
4. Discussion
5. Taxonomy

Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Giraud, T.; Refrégier, G.; Le Gac, M.; de Vienne, D.M.; Hood, M.E. Speciation in fungi. Fungal Genet. Biol. 2008, 45, 791–802. [Google Scholar] [CrossRef] [PubMed]
- Restrepo, S.; Tabima, J.F.; Mideros, M.F.; Grünwald, N.J.; Matute, D.R. Speciation in fungal and oomycete plant pathogens. Annu. Rev. Phytopathol. 2014, 52, 289–316. [Google Scholar] [CrossRef] [PubMed]
- Stukenbrock, E.H.; McDonald, B.A. The origins of plant pathogens in agro-ecosystems. Annu. Rev. Phytopathol. 2008, 46, 75–100. [Google Scholar] [CrossRef] [PubMed]
- Giraud, T.; Gladieux, P.; Gavrilets, S. Linking the emergence of fungal plant diseases with ecological speciation. Trends Ecol. Evol. 2010, 25, 387–395. [Google Scholar] [CrossRef]
- Schardl, C.L. The epichloae, symbionts of the grass subfamily Poöideae. Ann. Missouri Bot. Gard. 2010, 97, 646–665. [Google Scholar] [CrossRef]
- Leuchtmann, A.; Bacon, C.W.; Schardl, C.L.; White, J.F., Jr.; Tadych, M. Nomenclatural realignment of Neotyphodium species with genus Epichloë. Mycologia 2014, 106, 202–215. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.G.; Nagabhyru, P.; Schardl, C.L. Epichloë festucae endophytic growth in florets, seeds, and seedlings of perennial ryegrass (Lolium perenne). Mycologia 2017, 109, 691–700. [Google Scholar] [CrossRef]
- Zhang, W.; Card, S.D.; Mace, W.J.; Christensen, M.J.; McGill, C.R.; Matthew, C. Defining the pathways of symbiotic Epichloë colonization in grass embryos with confocal microscopy. Mycologia 2017, 109, 153–161. [Google Scholar] [CrossRef]
- White, J.F., Jr. Endophyte-host associations in forage grasses. XI. A proposal concerning origin and evolution. Mycologia 1988, 80, 442–446. [Google Scholar] [CrossRef]
- Bucheli, E.; Leuchtmann, A. Evidence for genetic differentiation between choke-inducing and asymptomatic strains of the Epichloë grass endophyte from Brachypodium sylvaticum. Evolution 1996, 50, 1879–1887. [Google Scholar]
- Meijer, G.; Leuchtmann, A. The effects of genetic and environmental factors on disease expression (stroma formation) and plant growth in Brachypodium sylvaticum infected by Epichloë sylvatica. Oikos 2000, 91, 446–458. [Google Scholar] [CrossRef]
- Meijer, G.; Leuchtmann, A. Fungal genotype controls mutualism and sex in Brachypodium sylvaticum infected by Epichloë sylvatica. Acta Biol. Hung. 2001, 52, 249–263. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Clarke, B.B.; Funk, C.R. Effect of fertilizer and fungicide applications on choke expression and endophyte transmission in chewings fescue. In Proceedings of the International Symposium on Acremonium/Grass Interactions, Baton Rouge, LA, USA, 3 November 1990; pp. 62–66. [Google Scholar]
- Funk, C.R.; Belanger, F.C.; Murphy, J.A. Role of endophytes in grasses used for turf and soil conservation. In Biotechnology of Endophytic Fungi of Grasses; Bacon, C.W., White, W.F., Jr., Eds.; CRC Press: Boca Raton, FL, USA, 1994; pp. 201–209. [Google Scholar]
- Campbell, M.A.; Tapper, B.A.; Simpson, W.R.; Johnson, R.D.; Mace, W.; Ram, A.; Lukito, Y.; Dupont, P.Y.; Johnson, L.J.; Scott, D.B.; et al. Epichloë hybrida, sp nov., an emerging model system for investigating fungal allopolyploidy. Mycologia 2017, 109, 715–729. [Google Scholar] [CrossRef] [PubMed]
- Shymanovich, T.; Charlton, N.D.; Musso, A.M.; Scheerer, J.; Cech, N.B.; Faeth, S.H.; Young, C.A. Interspecific and intraspecific hybrid Epichloë species symbiotic with the North American native grass Poa alsodes. Mycologia 2017, 109, 459–474. [Google Scholar] [CrossRef] [PubMed]
- Leuchtmann, A.; Young, C.A.; Stewart, A.V.; Simpson, W.R.; Hume, D.E.; Scott, B. Epichloe novae-zelandiae, a new endophyte from the endemic New Zealand grass Poa matthewsii. N. Z. J. Bot. 2019, 57, 271–288. [Google Scholar] [CrossRef]
- Thünen, T.; Becker, Y.; Cox, M.P.; Ashrafi, S. Epichloë scottii sp. nov., a new endophyte isolated from Melica uniflora is the missing ancestor of Epichloë disjuncta. IMA Fungus 2022, 13, 2. [Google Scholar] [CrossRef]
- Tian, P.; Xu, W.B.; Li, C.J.; Song, H.; Wang, M.N.; Schardl, C.L.; Nan, Z.B. Phylogenetic relationship and taxonomy of a hybrid Epichloë species symbiotic with Festuca sinensis. Mycol. Prog. 2020, 19, 1069–1081. [Google Scholar] [CrossRef]
- Moon, C.D.; Craven, K.D.; Leuchtmann, A.; Clement, S.L.; Schardl, C.L. Prevalence of interspecific hybrids amongst asexual fungal endophytes of grasses. Molec. Ecol. 2004, 13, 1455–1467. [Google Scholar] [CrossRef]
- Leuchtmann, A.; Schardl, C.L. Mating compatibility and phylogenetic relationships among two new species of Epichloë and other congeneric European species. Mycol. Res. 1998, 102, 1169–1182. [Google Scholar] [CrossRef]
- Schardl, C.L.; Leuchtmann, A. Three new species of Epichloë symbiotic with North American grasses. Mycologia 1999, 91, 95–107. [Google Scholar] [CrossRef]
- White, J.F., Jr. Endophyte-host associations in grasses. XIX. A systematic study of some sympatric species of Epichloë in England. Mycologia 1993, 85, 444–455. [Google Scholar] [CrossRef]
- Leuchtmann, A.; Schardl, C.L.; Siegel, M.R. Sexual compatibility and taxonomy of a new species of Epichloë symbiotic with fine fescue. Mycologia 1994, 86, 802–812. [Google Scholar] [CrossRef]
- Treindl, A.D.; Leuchtmann, A. Assortative mating in sympatric ascomycete fungi revealed by experimental fertilizations. Fungal Biol. 2019, 123, 676–686. [Google Scholar] [CrossRef] [PubMed]
- Schirrmann, M.K.; Zoller, S.; Fior, S.; Leuchtmann, A. Genetic evidence for reproductive isolation among sympatric Epichloë endophytes as inferred from newly developed microsatellite markers. Microb. Ecol. 2015, 70, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Christensen, M.J.; Leuchtmann, A.; Rowan, D.D.; Tapper, B.A. Taxonomy of Acremonium endophytes of tall fescue (Festuca arundinacea), meadow fescue (Festuca pratensis), and perennial rye-grass (Lolium perenne). Mycol. Res. 1993, 97, 1083–1092. [Google Scholar] [CrossRef]
- van Zijll de Jong, E.; Dobrowolski, M.P.; Bannan, N.R.; Stewart, A.V.; Smith, K.F.; Spangenberg, G.C.; Forster, J.W. Global genetic diversity of the perennial ryegrass fungal endophyte Neotyphodium lolii. Crop Sci. 2008, 48, 1487–1501. [Google Scholar] [CrossRef]
- Ekanayake, P.N.; Rabinovich, M.; Guthridge, K.M.; Spangenberg, G.C.; Forster, J.W.; Sawbridge, T.I. Phylogenomics of fescue grass-derived fungal endophytes based on selected nuclear genes and the mitochondrial gene complement. BMC Evol. Biol. 2013, 13, 270. [Google Scholar] [CrossRef]
- Clay, K.; Leuchtmann, A. Infection of woodland grasses by fungal endophytes. Mycologia 1989, 81, 805–811. [Google Scholar] [CrossRef]
- Oberhofer, M.; Leuchtmann, A. Genetic diversity in epichloid endophytes of Hordelymus europaeus suggests repeated host jumps and interspecific hybridizations. Molec. Ecol. 2012, 21, 2713–2726. [Google Scholar] [CrossRef]
- Afkhami, M.E. Fungal endophyte-grass symbioses are rare in the California floristic province and other regions with Mediterranean-influenced climates. Fungal Ecol. 2012, 5, 345–352. [Google Scholar] [CrossRef]
- Song, H.; Li, X.; Bao, G.; Song, Q.; Tian, P.; Nan, Z. Phylogeny of Neotyphodium endophyte from western Chinese Elymus species based on act sequences. Acta Microbiol. Sin. 2015, 55, 273–281. [Google Scholar]
- Davies, M.S.; Long, G.L. Performance of two contrasting morphs of Brachypodium sylvaticum transplanted into shaded and unshaded sites. J. Ecol. 1991, 79, 505–517. [Google Scholar] [CrossRef]
- Schippmann, U. Revision der europäischen Arten der Gattung Brachypodium Palisot de Beauvois (Poaceae); Conservatoire et jardin botaniques de Genève: Pregny-Chambesy, Switzerland, 1991; pp. 1–250. [Google Scholar]
- Eckblad, F.; Torkelsen, A. Epichloë typhina in Norway. Opera Bot. 1989, 100, 51–57. [Google Scholar]
- Raynal, G. Présence dans les graminées spontanées et prairiales de champignons endophytes du genre Acremonium. In Proceedings of the ANPP—Quatrième Conférence Internationale sur les Maladies des Plantes, Bordeaux, France, 6–8 December 1994; pp. 1125–1130. [Google Scholar]
- Enomoto, T.; Tsukiboshi, M.; Shimanuki, T. The gramineous plants in which Neotyphodium endophyes were found. J. Weed Sci. Technol. 1998, 43, 76–77. (In Japanese) [Google Scholar]
- Zabalgogeazcoa, I.; Vázquez de Aldana, B.R.; García Criado, B.; Gracía Ciudad, A. Fungal endophytes in natural grasslands of western Spain. In Book of Abstracts, Proceedings of the Grassland Conference, 4th International Neotyphodium/Grass Interactions Symposium, Soest, Germany, 27–29 September 2000; University of Paderborn: Soest, Germany, 2000; p. 126. [Google Scholar]
- Meijer, G.; Leuchtmann, A. Multistrain infections of the grass Brachypodium sylvaticum by its fungal endophyte Epichloë sylvatica. New Phytol. 1999, 141, 355–368. [Google Scholar] [CrossRef]
- Zabalgogeazcoa, I.; García Ciudad, G.; Leuchtmann, A.; Vázques de Aldana, B.R.; García Criado, B. Effects of choke disease in the grass Brachypodium phoenicoides. Plant Pathol. 2008, 57, 467–472. [Google Scholar] [CrossRef]
- Hess, H.E.; Landolt, E.; Hirzel, R. Flora der Schweiz und angrenzender Gebiete, 2nd ed.; Birkäuser: Basel, Switzerland, 1976; Volume 1, pp. 1–858. [Google Scholar]
- Iannone, L.J.; Novas, M.V.; Young, C.A.; De Battista, J.P.; Schardl, C.L. Endophytes of native grasses from South America: Biodiversity and ecology. Fungal Ecol. 2012, 5, 357–363. [Google Scholar] [CrossRef]
- White, J.F., Jr.; Halisky, P.M.; Sun, S.; Morgan-Jones, G.; Funk, C.R., Jr. Endophyte-host associations in grasses. XVI. Patterns of endophyte distribution in species of the tribe Agrostideae. Am. J. Bot. 1992, 79, 472–477. [Google Scholar] [CrossRef]
- White, J.F., Jr. Endophyte-host associations in grasses. XX. Structural and reproductive studies of Epichloë amarillans sp. nov. and comparisons to E. typhina. Mycologia 1994, 86, 571–580. [Google Scholar] [CrossRef]
- Craven, K.D.; Hsiau, P.T.W.; Leuchtmann, A.; Hollin, W.; Schardl, C.L. Multigene phylogeny of Epichloë species, fungal symbionts of grasses. Ann. Mo. Bot. Gard. 2001, 88, 14–34. [Google Scholar] [CrossRef]
- Ji, Y.L.; Zhan, L.H.; Kang, Y.; Sun, X.H.; Yu, H.S.; Wang, Z.W. A new stromata-producing Neotyphodium species symbiotic with clonal grass Calamagrostis epigeios (L.) Roth. grown in China. Mycologia 2009, 101, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Soreng, R.J.; Peterson, P.M.; Romaschenko, K.; Davidse, G.; Zuloaga, F.O.; Judziewicz, E.J.; Filgueiras, T.S.; Davis, J.I.; Morrone, O. A worldwide phylogenetic classification of the Poaceae (Gramineae). J. Syst. Evol. 2015, 53, 117–137. [Google Scholar] [CrossRef]
- Roy, B.A.; Coulson, T.; Blaser, W.; Policha, T.; Stewart, J.L.; Blaisdell, G.K.; Güsewell, S. Population regulation by enemies of the grass Brachypodium sylvaticum: Demography in native and invaded ranges. Ecology 2011, 92, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Väre, H.; Itämies, J. Phorbia phrenione (Séguy) (Diptera: Anthomyiidae) in Finland. Sahlbergia 1995, 2, 119–124. [Google Scholar]
- Leuchtmann, A. Isozyme relationships of Acremonium endophytes from twelve Festuca species. Mycol. Res. 1994, 98, 25–33. [Google Scholar] [CrossRef]
- White, J.F., Jr.; Cole, G.T. Endophyte-host assiciations in forage grasses. II. Taxonomic observations on the endophyte of Fesutca arundinacea. Mycologia 1985, 77, 483–486. [Google Scholar] [CrossRef]
- Peakall, R.; Smouse, P.E. GenAlEx 6.5: Genetic analysis in Excel. Population genetic software for teaching and research-an update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef]
- Swofford, D.L. PAUP: Phylogenetic Analysis Using Parsimony and Other Methods; Sinauer Associates: Sunderland, MA, USA, 2003. [Google Scholar]
- Hewitt, G. The genetic legacy of the Quaternary ice ages. Nature 2000, 405, 907–913. [Google Scholar] [CrossRef]
- Afkhami, M.E.; Rudgers, J.A. Symbiosis lost: Imperfect vertical transmission of fungal endophytes in grasses. Am. Nat. 2008, 172, 405–416. [Google Scholar] [CrossRef]
- Siegel, M.R.; Bush, L.P. Defensive chemicals in grass-fungal endophyte associations. Recent Adv. Phytochem. 1996, 30, 81–119. [Google Scholar]
- Schardl, C.L.; Florea, S.; Pan, J.; Nagabhyru, P.; Bec, S.; Calie, P.J. The epichloae: Alkaloid diversity and roles in symbiosis with grasses. Curr. Opin. Plant Biol. 2013, 16, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Leuchtmann, A.; Schmidt, D.; Bush, L.P. Different levels of protective alkaloids in grasses with stroma-forming and seed-transmitted Epichloë/Neotyphodium endophytes. J. Chem. Ecol. 2000, 26, 1025–1036. [Google Scholar] [CrossRef]
- Berry, D.; Mace, W.; Grage, K.; Wesche, F.; Gore, S.; Schardl, C.L.; Young, C.A.; Dijkwel, P.P.; Leuchtmann, A.; Bode, H.B.; et al. Efficient nonenzymatic cyclization and domain shuffling drive pyrrolopyrazine diversity from truncated variants of a fungal NRPS. Proc. Natl. Acad. Sci. USA 2019, 116, 25614–25623. [Google Scholar] [CrossRef] [PubMed]
- Brem, D.; Leuchtmann, A. Epichloë grass endophytes increase herbivore resistance in the woodland grass Brachypodium sylvaticum. Oecologia 2001, 126, 522–530. [Google Scholar] [CrossRef]
- Schardl, C.L.; Young, C.A.; Hesse, U.; Amyotte, S.G.; Andreeva, K.; Calie, P.J.; Fleetwood, D.J.; Haws, D.C.; Moore, N.; Oeser, B.; et al. Plant-symbiotic fungi as chemical engineers: Multi-genome analysis of the Clavicipitaceae reveals dynamics of alkaloid loci. PLoS Genet. 2013, 9, e1003323. [Google Scholar] [CrossRef]
- Ambrose, K.V.; Koppenhoefer, A.M.; Belanger, F.C. Horizontal gene transfer of a bacterial insect toxin gene into the Epichloë fungal symbionts of grasses. Sci. Rep. 2014, 4, 5562. [Google Scholar] [CrossRef]
- Clay, K. Effects of fungal endophytes on the seed and seedling biology of Lolium perenne and Festuca arundinacea. Oecologia 1987, 73, 358–362. [Google Scholar] [CrossRef]
- Clay, K. The impact of parasitic and mutualistic fungi on competitive interactions among plants. In Perspectives on Plant Competition; Grace, J.B., Tilman, D., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 391–412. [Google Scholar]
- Brem, D.; Leuchtmann, A. Intraspecific competition of endophyte infected vs uninfected plants of two woodland grass species. Oikos 2002, 96, 281–290. [Google Scholar] [CrossRef]
- Wang, H.; Sun, S.; Ge, W.; Zhao, L.; Hou, B.; Wang, K.; Lyu, Z.; Chen, L.; Xu, S.; Guo, J.; et al. Horizontal gene transfer of Fhb7 from fungus underlies Fusarium head blight resistance in wheat. Science 2020, 368, eaba5435. [Google Scholar] [CrossRef]
- Schirrmann, M.K.; Leuchtmann, A. The role of host-specificity in the reproductive isolation of Epichloë endophytes revealed by reciprocal infections. Fungal Ecol. 2015, 15, 29–38. [Google Scholar] [CrossRef]
- Leuchtmann, A.; Michelsen, V. Biology and evolution of the Epichloë-associated Botanophila species found in Europe (Diptera: Anthomyiidae). Insect Syst. Evol. 2016, 47, 1–14. [Google Scholar] [CrossRef]
- Bultman, T.L.; Leuchtmann, A. Biology of the Epichloë-Botanophila interaction: An intriguing association between fungi and insects. Fungal Biol. Rev. 2008, 22, 131–138. [Google Scholar] [CrossRef]


| Isolate No. | Host Plant | Collection Site | Country | Reproduction | Isolation | Micro-Satellites | tubB | tefA |
|---|---|---|---|---|---|---|---|---|
| 8927/2 | Brachypodium sylvaticum | Albisgüetli, Zürich | Switzerland | asexual | culm | + | – | – |
| 8928/2 | Brachypodium sylvaticum | Uetliberg, Zürich | Switzerland | sexual | ascospores | + | KC296736 | KC296740 |
| 9028/3 | Brachypodium sylvaticum | Albisgüetli, Zürich | Switzerland | sexual | ascospores | + | MW283338 | MW283379 |
| 9127/1 | Brachypodium sylvaticum | Weissenstein, Solothurn | Switzerland | sexual | culm (stroma) | + | MW283339 | MW283380 |
| 9251/2 | Brachypodium sylvaticum | Sihlwald, Zürich | Switzerland | sexual | ascospores | + | MW283340 | MW283381 |
| 9301/1 | Brachypodium sylvaticum | Sihlwald, Zürich | Switzerland | asexual | seed | + | L78291 | AF231219 |
| 9344 | Brachypodium sylvaticum | Uetliberg, Zürich | Switzerland | sexual | culm | + | MW283341 | MW283382 |
| 9504 | Brachypodium sylvaticum | Nishinasuno, Tochigi Pref. | Japan | sexual | culm (stroma) | + | L78278 | AF231218 |
| 9701 a | Brachypodium sylvaticum | Albisgüetli, Zürich | Switzerland | sexual | culm | + | – | – |
| 9702 a | Brachypodium sylvaticum | Uetliberg, Zürich | Switzerland | sexual | culm | + | – | – |
| 9703 a | Brachypodium sylvaticum | Sihlwald, Zürich | Switzerland | sexual | culm | + | – | – |
| 9704 a | Brachypodium sylvaticum | Sihlwald, Zürich | Switzerland | sexual | culm | + | – | – |
| 9705 a | Brachypodium sylvaticum | Sihlwald, Zürich | Switzerland | asexual | culm | + | – | – |
| 9806/4 | Brachypodium sylvaticum | Zollikerberg, Zürich | Switzerland | sexual | ascospores | + | MW283342 | MW283383 |
| 9808/1 | Brachypodium sylvaticum | Parheng, Lund | Sweden | asexual | seed | + | MW283343 | MW283384 |
| 9809/1 | Brachypodium sylvaticum | Bächkrälen, Lund | Sweden | asexual | seed | + | MW283344 | – |
| 9810/1 | Brachypodium sylvaticum | Bächriks, Lund | Sweden | asexual | seed | + | MW283345 | – |
| 9811/1 | Brachypodium sylvaticum | Stokkem, Zuid-Limburg | Netherlands | asexual | seed | + | MW283346 | MW283385 |
| 9812/1 | Brachypodium sylvaticum | Schiepersberg, Zuid-Limburg | Netherlands | asexual | seed | + | MW283347 | – |
| 9813/1 | Brachypodium sylvaticum | Portofino, Liguria | Italy | asexual | seed | + | JF718489 | JF718542 |
| 9814/1 | Brachypodium sylvaticum | Tongwynlais, Cardiff | U.K. | asexual | seed | + | MW283348 | MW283386 |
| 9815/1 | Brachypodium sylvaticum | Chéserex, Vaud | Switzerland | sexual | culm (stroma) | + | MW283349 | MW283387 |
| 9817/1 | Brachypodium sylvaticum | Chéserex, Vaud | Switzerland | sexual | culm | + | – | – |
| 9818/2 | Brachypodium sylvaticum | Kirchspiel Hållnäs, Uppsala | Sweden | asexual | seed | + | MW283350 | – |
| 9819/1 | Brachypodium sylvaticum | Kirchspiel Hållnäs, Uppsala | Sweden | asexual | seed | + | MW283351 | MW283388 |
| 0731/1 | Brachypodium sylvaticum | Hinter Guldenen, Zürich | Switzerland | sexual | stroma | + | KC296737 | KC296741 |
| 1301/1 | Brachypodium sylvaticum | Valle di Oddeone, Sardinia | Italy | asexual | culm | + | MW283334 | MW283375 |
| 1753/2 | Brachypodium sylvaticum | Pisciotta, Campania | Italy | asexual | culm | + | MW283335 | MW283376 |
| 1809/1 | Brachypodium sylvaticum | Montemayor, Salamanca | Spain | sexual | stroma | + | MW283336 | MW283377 |
| 1902 | Brachypodium sylvaticum | Policoro, Basilicata | Italy | sexual | stroma | + | MW283337 | MW283378 |
| 9410 | Brachypodium pinnatum | Perroudaz, Vaud | Switzerland | sexual | culm | + | L78292 | AF231223 |
| 9435/1 | Brachypodium pinnatum | Zumikon, Zürich | Switzerland | sexual | ascospores | + | MW283331 | MW283372 |
| 9612 | Brachypodium pinnatum | Zumikon, Zürich | Switzerland | sexual | culm | + | – | – |
| 9613 | Brachypodium pinnatum | Zumikon, Zürich | Switzerland | sexual | culm | + | – | – |
| 9639 | Brachypodium pinnatum | Zumikon, Zürich | Switzerland | sexual | culm | + | – | – |
| 9640/1 | Brachypodium pinnatum | Zumikon, Zürich | Switzerland | sexual | culm | + | MW283332 | MW283373 |
| 0728 | Brachypodium pinnatum | Eschikon, Zürich | Switzerland | sexual | culm | + | – | – |
| 0901/1 | Brachypodium pinnatum | Hitzfelden, Alsace | France | sexual | stroma | + | JF718488 | JF718541 |
| 1407/1 | Brachypodium pinnatum | Linguaglossa, Sicily | Italy | asexual | culm | + | MW283328 | MW283369 |
| 1604/2 | Brachypodium pinnatum | La Rippe, Vaud | Switzerland | sexual | stroma | + | – | – |
| 1901/1 | Brachypodium pinnatum | Marsicovetere, Basilicata | Italy | sexual | stroma | + | MW283329 | MW283370 |
| 2010/1 | Brachypodium pinnatum | Ftan, Grisons | Switzerland | sexual | stroma | – | MW283330 | MW283371 |
| 2001 | Brachypodium phoenicoides | Torres del Carrizal, Zamora | Spain | sexual | culm | + | MW283333 | MW283374 |
| 1614/1 | Calamagrostis arundinacea | Ftan, Grisons | Switzerland | sexual | stroma | + | MW283352 | MW283389 |
| 1614/2 | Calamagrostis arundinacea | Ftan, Grisons | Switzerland | sexual | stroma | – | MW283353 | MW283390 |
| 2015/1 | Calamagrostis arundinacea | Ftan, Grisons | Switzerland | sexual | stroma | – | MW283354 | MW283391 |
| 0908/1 | Calamagrostis purpurea | Paltamo, Oulu | Finland | sexual | stroma | – | MW283355 | MW283392 |
| 9039 | Calamagrostis villosa | Lavin, Grisons | Switzerland | sexual | culm (stroma) | – | L78270 | AF231196 |
| 9618/1 | Calamagrostis villosa | Bever, Grisons | Switzerland | sexual | culm (stroma) | – | MW283357 | MW283394 |
| 0430/1 | Calamagrostis villosa | Albula, Grisons | Switzerland | sexual | stroma | – | MW283356 | MW283393 |
| Multiplex | Locus | Dye Label | Repeat Motif | Primer Sequence (5′-3′) | Size Range (bp) |
|---|---|---|---|---|---|
| M1 | E8 | 6-FAM | (AC)14 | F: CATGGACCAAGTTGTGAGACC | 216–266 |
| R: AGCAAGTCTCGTAACGGTCTG | |||||
| E39 | 6-FAM | (GTTTC)12 | F: GTAGCACATGCATCGAATCAG | 408–554 | |
| R: ACCCACTAAAGACGGATGACA | |||||
| E29 | VIC | (AGC)9 | F: TTCCAGCAGCTCTTCAATACC | 123–182 | |
| R: ACAGTGGTTCCTGAGGTTTGA | |||||
| E50 | VIC | (TTG)12 | F: TCGTCTTGGACTTTGCCTTT | 312–369 | |
| R: TTGAGGTTGTCGAGATACACG | |||||
| M2 | E13 | 6-FAM | (GA)11 | F: GTTCTCCAAGGCTTCCAATTT | 464–508 |
| R: GAGAAACGATATTCGCATTGG | |||||
| E47 | VIC | (CTCA)9 | F: GCCTGTTGAGAAAGACGTGAT | 284–320 | |
| R: GATCGAAACACGGGATCATAC | |||||
| E32 | NED | (CAG)11 | F: AGATGAATGGTCAGCAGTTCC | 316–343 | |
| R: GGACCATACTTCGTCAACGTC | |||||
| E45 | NED | (GT)15 | F: TTGACGTCGGGAGGTAGTAGA | 392–432 | |
| R: CTGGTTACGGAAAGCGAGATA | |||||
| M3 | E4 | 6-FAM | (AG)9 | F: ATTGACCTGTAGCGCGAGTAG | 120–134 |
| R: CAGAACCAATTCGAATCCATC | |||||
| E33 | 6-FAM | (TCG)11 | F: TGCCAGATGTTTCAATGACTG | 326–334 | |
| R: AACCCATACTCAGCTTTGCAG | |||||
| E36 | VIC | (TGC)7 | F: ATTCGAGAATGGATGACCTGA | 402–414 | |
| R: AAGAAAGGAATGGGATTGCTC | |||||
| E41 | VIC | (TG)11 | F: ATTGCCCTGCAGAAGTTGTTA | 304–350 | |
| R: TGAGTCGATCGAGAACAAAGA | |||||
| M4 | E22 | 6-FAM | (TGGA)10 | F: GCAAGGATTGGTTGGTGATAA | 127–171 |
| R: GCGGATCACTCTGTAGGCTAA | |||||
| E11 | 6-FAM | (CT)11 | F: GTCAGAGGGCAGTAGTGACG | 264–280 | |
| R: ATGTAATGCTCTGCCTGCTTC | |||||
| E27 | VIC | (GA)8 | F: TATAAATGACGCTGGGCTTGT | 365–391 | |
| R: TGCACTTGAAGAAGCCATGTA | |||||
| E46 | NED | (AG)9 | F: TCGTGACACCTTCTTCGGTAT | 376–392 | |
| R: AGAGGTTGTCGTGAGCATCAT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leuchtmann, A.; Schardl, C.L. Genetic Diversity of Epichloë Endophytes Associated with Brachypodium and Calamagrostis Host Grass Genera including Two New Species. J. Fungi 2022, 8, 1086. https://doi.org/10.3390/jof8101086
Leuchtmann A, Schardl CL. Genetic Diversity of Epichloë Endophytes Associated with Brachypodium and Calamagrostis Host Grass Genera including Two New Species. Journal of Fungi. 2022; 8(10):1086. https://doi.org/10.3390/jof8101086
Chicago/Turabian StyleLeuchtmann, Adrian, and Christopher L. Schardl. 2022. "Genetic Diversity of Epichloë Endophytes Associated with Brachypodium and Calamagrostis Host Grass Genera including Two New Species" Journal of Fungi 8, no. 10: 1086. https://doi.org/10.3390/jof8101086
APA StyleLeuchtmann, A., & Schardl, C. L. (2022). Genetic Diversity of Epichloë Endophytes Associated with Brachypodium and Calamagrostis Host Grass Genera including Two New Species. Journal of Fungi, 8(10), 1086. https://doi.org/10.3390/jof8101086








