Diagnostic Performance of a Novel Multiplex PCR Assay for Candidemia among ICU Patients
Abstract
1. Introduction
2. Material and Methods
2.1. Study Design
2.2. Patient Population
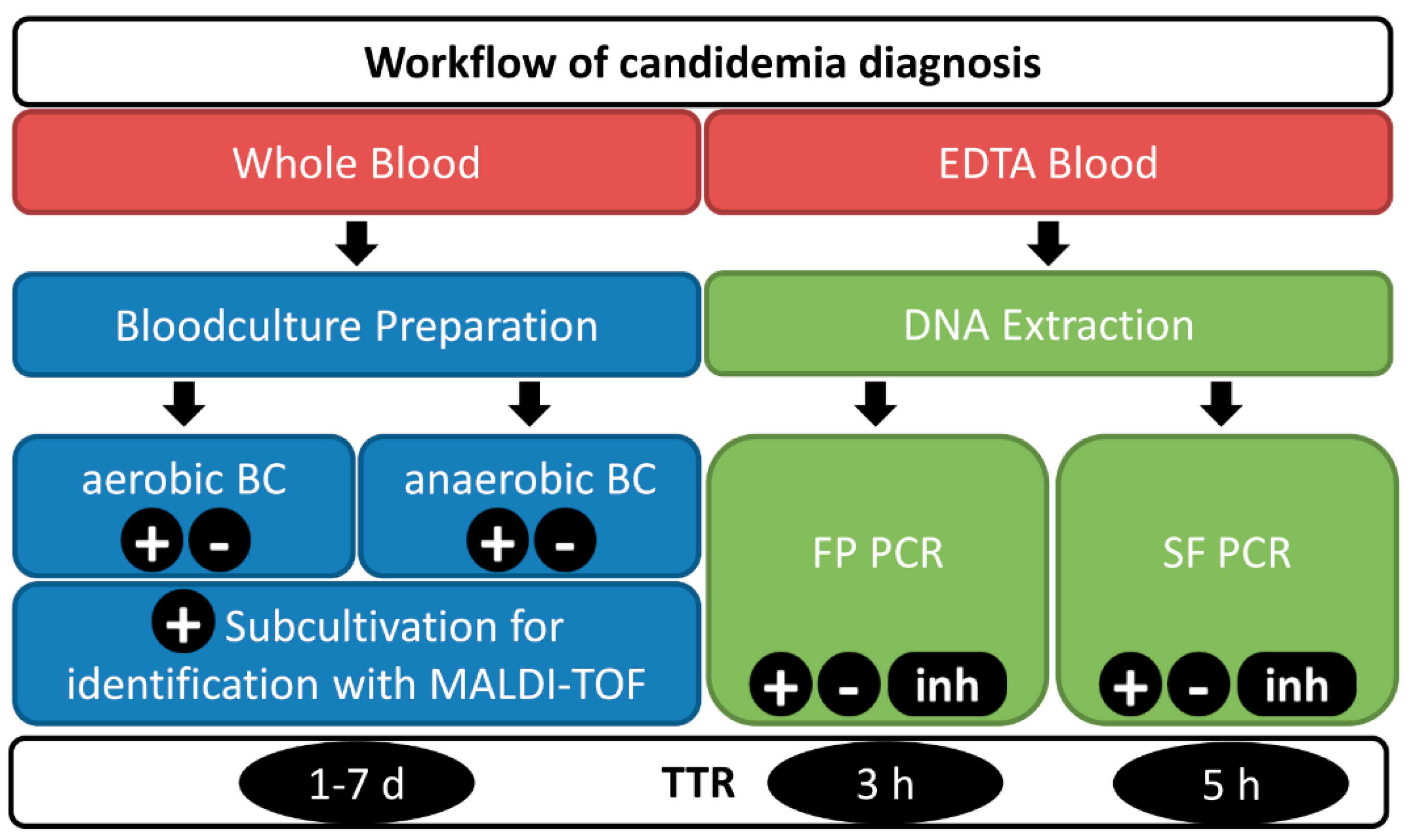
2.3. PCR-Based Assays
2.4. Blood Culture
2.5. Statistical Procedures
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barchiesi, F.; Orsetti, E.; Gesuita, R.; Skrami, E.; Manso, E. Epidemiology, clinical characteristics, and outcome of candidemia in a tertiary referral center in Italy from 2010 to 2014. Infection 2016, 44, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Ng, K.P.; Kuan, C.S.; Kaur, H.; Na, S.L.; Atiya, N.; Velayuthan, R.D. Candida species epidemiology 2000-2013: A laboratory-based report. Trop. Med. Int. Health 2015, 20, 1447–1453. [Google Scholar] [CrossRef] [PubMed]
- Aznar-Marin, P.; Galan-Sanchez, F.; Marin-Casanova, P.; Garcia-Martos, P.; Rodriguez-Iglesias, M. Candida nivariensis as a new emergent agent of vulvovaginal candidiasis: Description of cases and review of published studies. Mycopathologia 2016, 181, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, J.; Fisher, M.C. Global epidemiology of emerging Candida auris. Curr. Opin. Microbiol. 2019, 52, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Lamoth, F.; Lockhart, S.R.; Berkow, E.L.; Calandra, T. Changes in the epidemiological landscape of invasive candidiasis. J. Antimicrob. Chemother 2018, 73, i4–i13. [Google Scholar] [CrossRef] [PubMed]
- Quindos, G.; Marcos-Arias, C.; San-Millan, R.; Mateo, E.; Eraso, E. The continuous changes in the aetiology and epidemiology of invasive candidiasis: From familiar Candida albicans to multiresistant Candida auris. Int. Microbiol. 2018, 21, 107–119. [Google Scholar] [CrossRef]
- Bongomin, F.; Gago, S.; Oladele, R.O.; Denning, D.W. Global and multi-national prevalence of fungal diseases-estimate precision. J. Fungi. (Basel) 2017, 3, 57. [Google Scholar] [CrossRef] [PubMed]
- Kmeid, J.; Jabbour, J.F.; Kanj, S.S. Epidemiology and burden of invasive fungal infections in the countries of the Arab League. J. Infect. Public Health 2019. Epub ahead of print. [Google Scholar] [CrossRef]
- Macedo-Vinas, M.; Denning, D.W. Estimating the burden of serious fungal infections in Uruguay. J. Fungi. (Basel) 2018, 4, 37. [Google Scholar] [CrossRef]
- Ruhnke, M.; Groll, A.H.; Mayser, P.; Ullmann, A.J.; Mendling, W.; Hof, H.; Denning, D.W. Estimated burden of fungal infections in Germany. Mycoses 2015, 58, 22–28. [Google Scholar] [CrossRef]
- Leon, C.; Ruiz-Santana, S.; Saavedra, P.; Castro, C.; Loza, A.; Zakariya, I.; Ubeda, A.; Parra, M.; Macias, D.; Tomas, J.I.; et al. Contribution of candida biomarkers and DNA detection for the diagnosis of invasive candidiasis in ICU patients with severe abdominal conditions. Crit. Care 2016, 20, 149. [Google Scholar] [CrossRef] [PubMed]
- Benedict, K.; Jackson, B.R.; Chiller, T.; Beer, K.D. Estimation of direct healthcare costs of fungal diseases in the United States. Clin. Infect. Dis. 2019, 68, 1791–1797. [Google Scholar] [CrossRef] [PubMed]
- Strollo, S.; Lionakis, M.S.; Adjemian, J.; Steiner, C.A.; Prevots, D.R. Epidemiology of hospitalizations associated with invasive candidiasis, United States, 2002-2012. Emerg. Infect. Dis. 2016, 23, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Phoompoung, P.; Chayakulkeeree, M. Recent progress in the diagnosis of pathogenic Candida species in blood culture. Mycopathologia 2016, 181, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Posch, W.; Heimdorfer, D.; Wilflingseder, D.; Lass-Florl, C. Invasive candidiasis: Future directions in non-culture based diagnosis. Expert. Rev. Anti. Infect. Ther 2017, 15, 829–838. [Google Scholar] [CrossRef] [PubMed]
- O'Leary, R.A.; Einav, S.; Leone, M.; Madach, K.; Martin, C.; Martin-Loeches, I. Management of invasive candidiasis and candidaemia in critically ill adults: Expert opinion of the european society of anaesthesia intensive care scientific subcommittee. J. Hosp. Infect. 2018, 98, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Patch, M.E.; Weisz, E.; Cubillos, A.; Estrada, S.J.; Pfaller, M.A. Impact of rapid, culture-independent diagnosis of candidaemia and invasive candidiasis in a community health system. J. Antimicrob. Chemother. 2018, 73, iv27–iv30. [Google Scholar] [CrossRef] [PubMed]
- Avni, T.; Leibovici, L.; Paul, M. Pcr diagnosis of invasive candidiasis: Systematic review and meta-analysis. J. Clin. Microbiol. 2011, 49, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Clancy, C.J.; Nguyen, M.H. Diagnosing invasive candidiasis. J. Clin. Microbiol. 2018, 56, e01909-17. [Google Scholar] [CrossRef] [PubMed]
- Schabereiter-Gurtner, C.; Selitsch, B.; Rotter, M.L.; Hirschl, A.M.; Willinger, B. Development of novel real-time PCR assays for detection and differentiation of eleven medically important Aspergillus and Candida species in clinical specimens. J. Clin. Microbiol. 2007, 45, 906–914. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dark, P.; Blackwood, B.; Gates, S.; McAuley, D.; Perkins, G.D.; McMullan, R.; Wilson, C.; Graham, D.; Timms, K.; Warhurst, G. Accuracy of lightcycler((R)) septifast for the detection and identification of pathogens in the blood of patients with suspected sepsis: A systematic review and Meta-analysis. Intensive Care Med. 2015, 41, 21–33. [Google Scholar] [CrossRef]
- Knabl, L.; Mutschlechner, W.; Orth-Holler, D. Evaluation of a multiplex onspot primer-extension PCR assay in the diagnosis of sepsis. J Microbiol. Methods 2016, 120, 91–93. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Andersen, J.S.; Holten, M.K.; Krarup, K.B.; Reiter, N.; Schierbeck, J.; Helleberg, M. Diagnostic performance of T2Candida among ICU patients with risk factors for invasive candidiasis. Open Forum Infect. Dis 2019, 6, ofz136. [Google Scholar] [CrossRef] [PubMed]
- Opota, O.; Jaton, K.; Greub, G. Microbial diagnosis of bloodstream infection: Towards molecular diagnosis directly from blood. Clin. Microbiol. Infect. 2015, 21, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Pilecky, M.; Schildberger, A.; Orth-Holler, D.; Weber, V. Pathogen enrichment from human whole blood for the diagnosis of bloodstream infection: Prospects and limitations. Diagn Microbiol. Infect. Dis. 2019, 94, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Elges, S.; Arnold, R.; Liesenfeld, O.; Kofla, G.; Mikolajewska, A.; Schwartz, S.; Uharek, L.; Ruhnke, M. Prospective evaluation of the septifast multiplex real-time pcr assay for surveillance and diagnosis of infections in haematological patients after allogeneic stem cell transplantation compared to routine microbiological assays and an in-house real-time PCR method. Mycoses 2017, 60, 781–788. [Google Scholar] [PubMed]
- Grosse-Onnebrink, J.; Stehling, F.; Tschiedel, E.; Olivier, M.; Mellies, U.; Schmidt, R.; Buer, J.; Rath, P.M.; Steinmann, J. Bacteraemia and fungaemia in cystic fibrosis patients with febrile pulmonary exacerbation: A prospective observational study. BMC. Pulm. Med. 2017, 17, 96. [Google Scholar] [CrossRef]
- Warhurst, G.; Maddi, S.; Dunn, G.; Ghrew, M.; Chadwick, P.; Alexander, P.; Bentley, A.; Moore, J.; Sharman, M.; Carlson, G.L.; et al. Diagnostic accuracy of septifast multi-pathogen real-time PCR in the setting of suspected healthcare-associated bloodstream infection. Intensive Care Med 2015, 41, 86–93. [Google Scholar] [CrossRef]
- Lackner, M.; Lass-Florl, C. Commercial molecular tests for fungal diagnosis from a practical point of view. Methods Mol. Biol. 2017, 1508, 85–105. [Google Scholar]
- Baratloo, A.; Hosseini, M.; Negida, A.; El, A.G. Part 1: Simple definition and calculation of accuracy, sensitivity and specificity. Emergency 2015, 3, 48–49. [Google Scholar]
- Munoz, P.; Vena, A.; Machado, M.; Gioia, F.; Martinez-Jimenez, M.C.; Gomez, E.; Origuen, J.; Orellana, M.A.; Lopez-Medrano, F.; Fernandez-Ruiz, M.; et al. T2Candida MR as a predictor of outcome in patients with suspected invasive candidiasis starting empirical antifungal treatment: A prospective pilot study. J. Antimicrob. Chemother. 2018, 73, iv6–iv12. [Google Scholar] [CrossRef] [PubMed]
- Mikulska, M.; Calandra, T.; Sanguinetti, M.; Poulain, D.; Viscoli, C. The use of mannan antigen and anti-mannan antibodies in the diagnosis of invasive candidiasis: Recommendations from the third European conference on infections in leukemia. Crit Care 2010, 14, R222. [Google Scholar] [CrossRef] [PubMed]
- Makristathis, A.; Harrison, N.; Ratzinger, F.; Kussmann, M.; Selitsch, B.; Forstner, C.; Hirschl, A.M.; Burgmann, H. Substantial diagnostic impact of blood culture independent molecular methods in bloodstream infections: Superior performance of PCR/ESI-MS. Sci Rep 2018, 8, 16024. [Google Scholar] [CrossRef] [PubMed]
- Mikulska, M.; Furfaro, E.; De, C.E.; Drago, E.; Pulzato, I.; Borghesi, M.L.; Zappulo, E.; Raiola, A.M.; Grazia, C.D.; Del, B.V.; et al. Use of Aspergillus fumigatus real-time PCR in bronchoalveolar lavage samples (BAL) for diagnosis of invasive aspergillosis, including azole-resistant cases, in high risk haematology patients: The need for a combined use with galactomannan. Med. Mycol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Schrader, C.; Schielke, A.; Ellerbroek, L.; Johne, R. PCR inhibitors-occurrence, properties and removal. J. Appl. Microbiol. 2012, 113, 1014–1026. [Google Scholar] [CrossRef]
- Loonen, A.J.; Bos, M.P.; van Meerbergen, B.; Neerken, S.; Catsburg, A.; Dobbelaer, I.; Penterman, R.; Maertens, G.; van de Wiel, P.; Savelkoul, P.; et al. Comparison of pathogen DNA isolation methods from large volumes of whole blood to improve molecular diagnosis of bloodstream infections. PLoS ONE 2013, 8, e72349. [Google Scholar] [CrossRef]
- Westh, H.; Lisby, G.; Breysse, F.; Boddinghaus, B.; Chomarat, M.; Gant, V.; Goglio, A.; Raglio, A.; Schuster, H.; Stuber, F.; et al. Multiplex real-time PCR and blood culture for identification of bloodstream pathogens in patients with suspected sepsis. Clin. Microbiol. Infect. 2009, 15, 544–551. [Google Scholar] [CrossRef]
- Psifidi, A.; Dovas, C.I.; Bramis, G.; Lazou, T.; Russel, C.L.; Arsenos, G.; Banos, G. Comparison of eleven methods for genomic DNA extraction suitable for large-scale whole-genome genotyping and long-term DNA banking using blood samples. PLoS ONE 2015, 10, e0115960. [Google Scholar] [CrossRef]
- Sanguinetti, M.; Posteraro, B.; Lass-Flörl, C. Antifungal drug resistance among Candida species: Mechanisms and clinical impact. Mycoses 2015, 58, 2–13. [Google Scholar] [CrossRef]
- Whaley, S.G.; Berkow, E.L.; Rybak, J.M.; Nishimoto, A.T.; Barker, K.S.; Rogers, P.D. Azole antifungal resistance in Candida albicans and emerging non-albicans Candida species. Front. Microbiol. 2016, 7, 2173. [Google Scholar] [CrossRef]
| FP PCR | SF PCR | |
|---|---|---|
| Candida krusei | Candida krusei | |
| Candida glabrata | Candida glabrata | |
| Candida spp. incl. | C. albicans | Candida albicans |
| C. parapsilosis | Candida parapsilosis | |
| C. tropicalis | Candida tropicalis | |
| C. dubliniensis | ||
| Result (Candida) | BC | FP | SF | ||
|---|---|---|---|---|---|
| POS | 5 | 8 | Cq | 5 | Cq |
| Candida albicans | Candida spp. | 32.59 | Candida albicans | 30.69 | |
| Candida albicans | Candida spp. | 33.42 | Candida albicans | 31.04 | |
| Candida glabrata | Candida glabrata | 35.31 | Candida glabrata | 33.19 | |
| Candida dubliniense | Candida spp. | 36.30 | neg | n/a | |
| Candida guillermondii | Candida spp. | 38.38 | neg | n/a | |
| neg | Candida spp. | 37.17 | Candida albicans | 33.82 | |
| neg | Candida spp. | 39.11 | Candida albicans | 33.64 | |
| neg | Candida glabrata | 34.19 | neg | n/a | |
| NEG | 53 | 48 | 48 | ||
| inh (PCR) | n/a | 2 | 5 | ||
| total | n = 58 | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fuchs, S.; Lass-Flörl, C.; Posch, W. Diagnostic Performance of a Novel Multiplex PCR Assay for Candidemia among ICU Patients. J. Fungi 2019, 5, 86. https://doi.org/10.3390/jof5030086
Fuchs S, Lass-Flörl C, Posch W. Diagnostic Performance of a Novel Multiplex PCR Assay for Candidemia among ICU Patients. Journal of Fungi. 2019; 5(3):86. https://doi.org/10.3390/jof5030086
Chicago/Turabian StyleFuchs, Stefan, Cornelia Lass-Flörl, and Wilfried Posch. 2019. "Diagnostic Performance of a Novel Multiplex PCR Assay for Candidemia among ICU Patients" Journal of Fungi 5, no. 3: 86. https://doi.org/10.3390/jof5030086
APA StyleFuchs, S., Lass-Flörl, C., & Posch, W. (2019). Diagnostic Performance of a Novel Multiplex PCR Assay for Candidemia among ICU Patients. Journal of Fungi, 5(3), 86. https://doi.org/10.3390/jof5030086





