Five New Phyllachora Species from Tar Spot Fungi on Poaceae in Sichuan China
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection, Morphological Examination, and Preservation
2.2. DNA Extraction, PCR Amplification, and Sequencing
2.3. Phylogenetic Analyses
3. Results
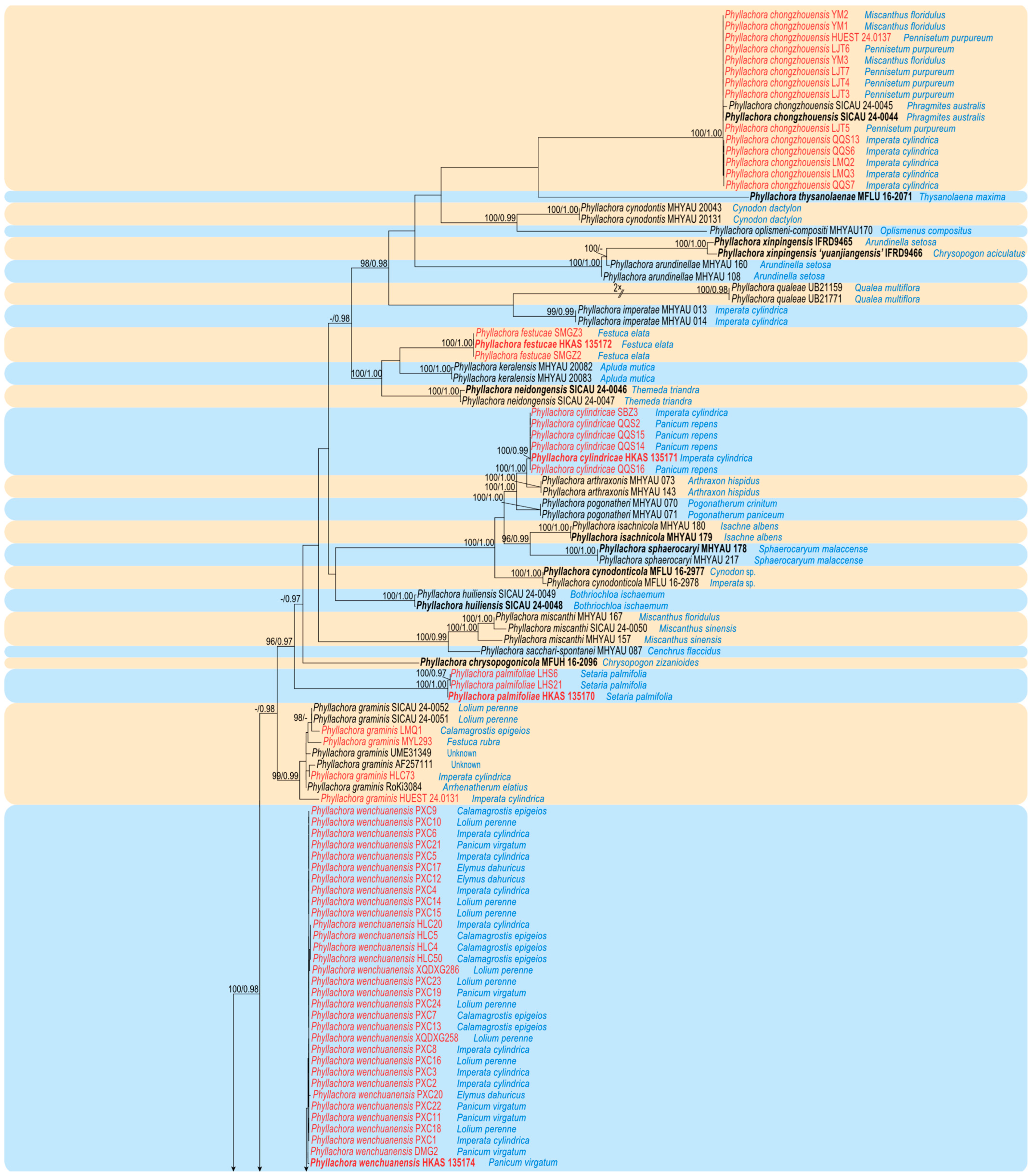
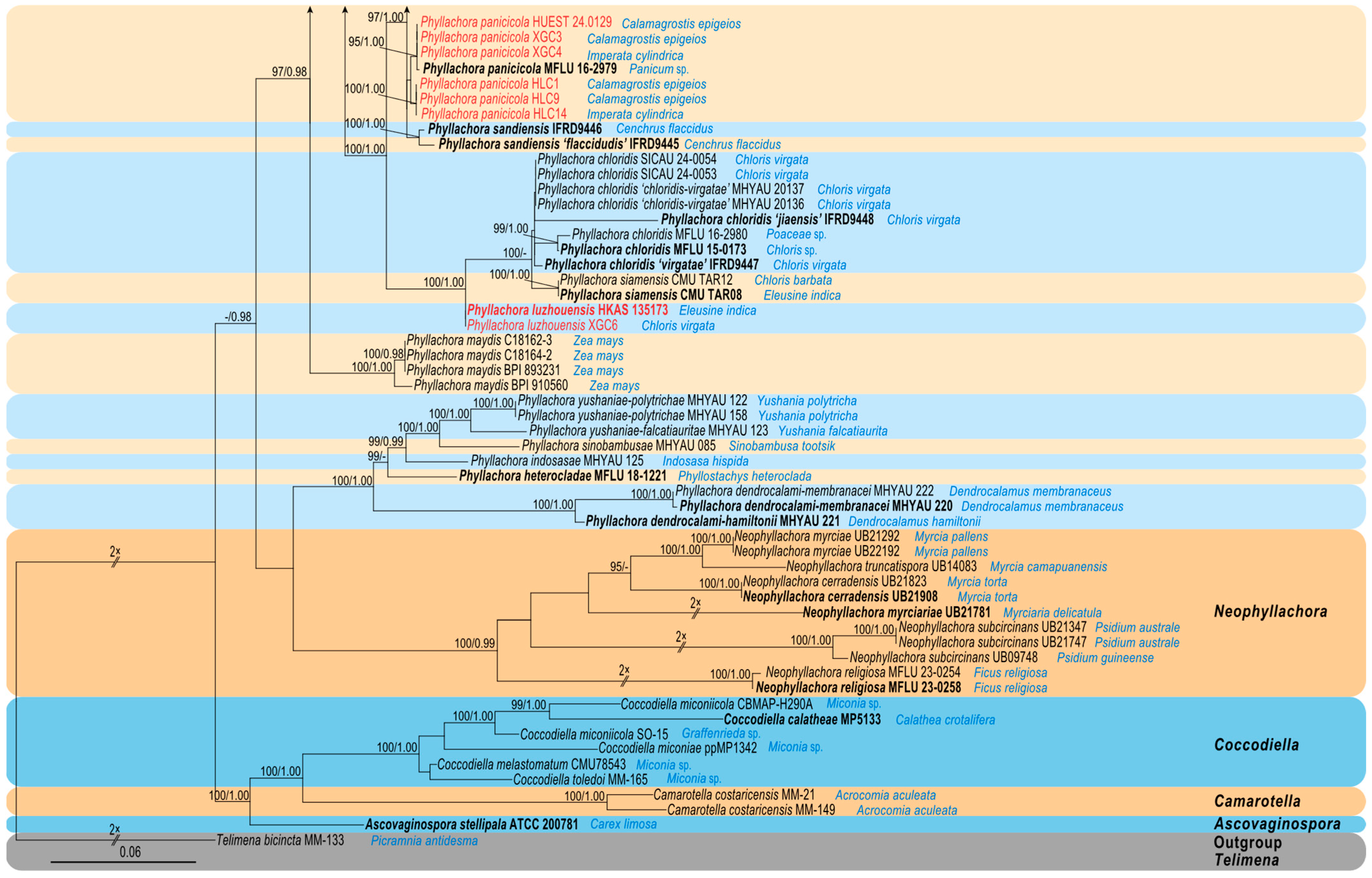
3.1. Molecular Phylogeny
3.2. Taxonomy
3.2.1. Phyllachora chongzhouensis Q.R. Sun, X.L. Xu & C.L. Yang. J. Fungi 10 (No. 588): 8 (2024). Figure 2

3.2.2. Phyllachora cylindricae P.W. Su & Maharachch., sp. Nov. Figure 3

3.2.3. Phyllachora festucae P.W. Su & Maharachch., sp. Nov. Figure 4

3.2.4. Phyllachora graminis (Pers.) Fuckel, Jahrb. Nassauischen Vereins Naturk. 23–24: 216 (1870). Figure 5
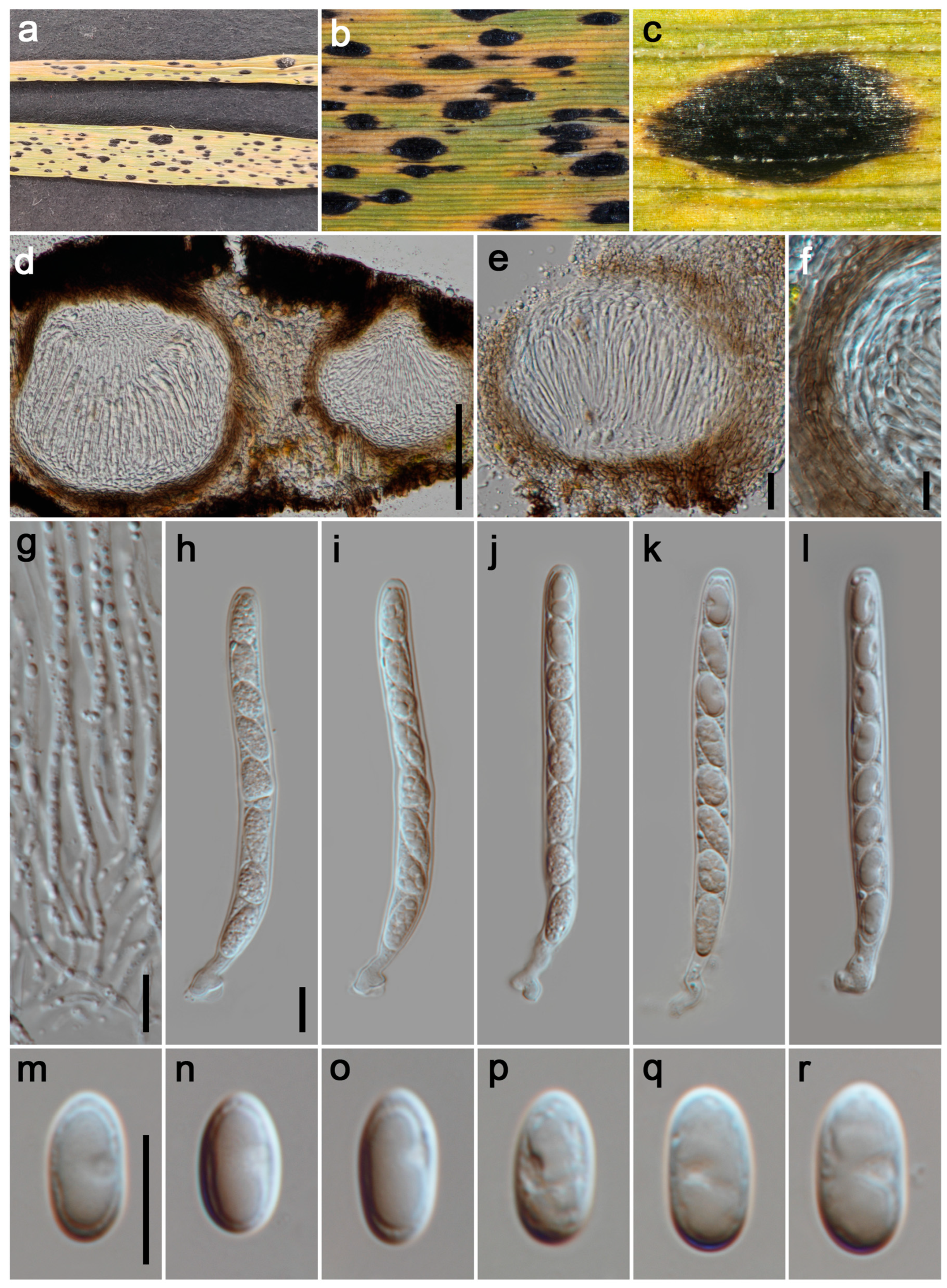
3.2.5. Phyllachora luzhouensis P.W. Su & Maharachch., sp. Nov. Figure 6

3.2.6. Phyllachora palmifoliae P.W. Su & Maharachch., sp. Nov. Figure 7

3.2.7. Phyllachora panicicola Dayar. & K.D. Hyde, Mycosphere 8(10): 1610 (2017). Figure 8

3.2.8. Phyllachora xinpingensis H.X. Wu & J.C. Li, in Li, Wu & Song, Phytotaxa 578(3): 279 (2023)
- = Phyllachora yuanjiangensis H.X. Wu & J.C. Li, in Li, Wu & Song, Phytotaxa 578(3): 282 (2023)
3.2.9. Phyllachora wenchuanensis P.W. Su & Maharachch., sp. Nov. Figure 9

| Species | Voucher | Hosts | Asci (µm) | Ascospores (µm) | Reference | |
|---|---|---|---|---|---|---|
| Size | Shape | |||||
| P. arthraxonis | HMAS 4306 | Arthraxon hispidus (Arthraxon) | 58–83 × 6–16 | 6–14 × 5–7 | ellipsoidal | [14] |
| P. arthraxonis T | - | Arthraxon ciliaris (Arthraxon) | 35–45 × 8–12 | 8–11 × 4–5 | ellipsoidal, ovoid | [38] |
| P. chloridis T | MFLU 15-0173 | Chloris sp. (Poaceae) | 50–72 × 6–8 | 8–12 × 3.5–4.8 | fusiform to oval | [4] |
| P. chongzhouensis T | SICAU 24-0044 | Phragmites australis (Poaceae) | 86–142 × 14–29 | 13–28 × 9–15 | ellipsoidal, ovoid | [16] |
| P. chongzhouensis | HUEST 24.0137 | Pennisetum purpureum (Poaceae) | 92–163 × 17–31 | 17–25 × 10–15 | subglobose to ellipsoidal | This study |
| P. chrysopogonicola T | MFLU 16-2096 | Chrysopogon zizanioides (Poaceae) | 70–122 × 12.5–15 | 20–26 (–30) × 6–8 | unicellular to papillate to turbinate, clavate, | [43] |
| P. cylindrica T | HKAS 135171 | Imperata cylindrica (Poaceae) | 80–128 × 10–16 | 11–15 × 6–8 | tear-shaped, ovoid to ellipsoidal | This study |
| P. festucae T | HKAS 135172 | Festuca elata (Poaceae) | 135–222 × 12–18 | 17–27 × 8–12 | long ellipsoidal to fusiform | This study |
| P. graminis | HUEST 24.0131 | Imperata cylindrica (Poaceae) | 83–105 × 7–9 | 10–13 × 5–7 | ovoid to ellipsoidal | This study |
| P. graminis | SICAU 24-005 | Lolium perenne (Poaceae) | 54–101 × 6–10 | 8–15 × 4–7 | ellipsoidal | [16] |
| P. graminis T | - | Elymus sp. (Poaceae) | 78–80 × 7–8 | 8–12 × 4–5 | oval to ovoid or ovoid | [14,40] |
| P. keralensis | MHYAU 13716 | Apluda mutica (Poaceae) | 48.7–79.1 × 10.7–13.6 | 9.2–12.8 × 5.6–6.9 | ellipsoid or oblong | [44] |
| P. keralensis T | - | Apluda mutica (Poaceae) | 52–68 × 7–9.5 | 10–14 × 5–6.5 | ellipsoid | [39] |
| P. luzhouensis T | HKAS 135173 | Eleusine indica (Poaceae) | 71–132 × 7–10 | 9–14 × 4–7 | ovoid, fusiform to ellipsoidal | This study |
| P. palmifoliae T | HKAS 135170 | Setaria palmifolia (Poaceae) | 110–175 × 9–19 | 12–17 × 5–8 | ovoid, fusiform to ellipsoidal | This study |
| P. panicicola | HUEST 24.0129 | Calamagrostis epigeios (Poaceae) | 86–145 × 7–11 | 10–14 × 5–7 | ovoid to ellipsoidal | This study |
| P. panicicola T | MFLU 16-2979 | Panicum sp. (Poaceae) | 110–130 × 10–14 | 14–16 × 6–8 | ellipsoidal, rounded at the ends | [4] |
| P. flaccidudis T | IFRD9445 | Cenchrus flaccidus (Poaceae) | 80–110 × 7–10 | 11–13 × 4–7 | drop shape, oval to ellipse, rounded at the ends | [13] |
| P. sandiensis T | IFRD9446 | Cenchrus flaccidus (Poaceae) | 92–126 × 7–10 | 10–14 × 6–7 | drop shape, oval to ellipse | [13] |
| P. siamensis T | CMU TAR08 | Eleusine indica (Poaceae) | 62–105 × 7–10 | 10–13 × 5–6 | unicellular to ovate | [42] |
| P. wenchuanensis T | HKAS 135174 | Panicum virgatum (Poaceae) | 75–115 × 7–10 | 10–14 × 5–7 | ovoid or ellipsoidal | This study |
| ITS (%) | P. panicicola MFLU 16-2979 | P. sandiensis IFRD9446 | P. flaccidudis IFRD9445 | P. panicicola HUEST 24.0129 | P. wenchuanensis HKAS 135174 |
|---|---|---|---|---|---|
| P. panicicola MFLU 16-2979 | 100 | ||||
| P. sandiensis IFRD9446 | 97.9 | 100 | |||
| P. flaccidudis IFRD9445 | 96.89 | 99.03 | 100 | ||
| P. panicicola HUEST 24.0129 | 99.81 | 97.7 | 96.57 | 100 | |
| P. wenchuanensis HKAS 135174 | 97.7 | 97.15 | 96.20 | 97.89 | 100 |
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gabel, A.W. Host-parasite relations of Phyllachora species on native grasses. Mycologia 1989, 81, 702–708. [Google Scholar] [CrossRef]
- Sutton, B.C.; Hodges, C.S. Hawaiian forest fungi. III. A new species, Gloeocoryneum hawaiiense, on Acacia koa. Mycologia 1983, 75, 280–284. [Google Scholar] [CrossRef]
- Parbery, D.G. Studies on graminicolous species of Phyllachora Nke. in Fckl. V. A taxonomic monograph. Aust. J. Bot. 1967, 15, 271–375. [Google Scholar] [CrossRef]
- Dayarathne, M.; Maharachchikumbura, S.; Jones, E.; Goonasekara, I.; Bulgakov, T.; Al-Sadi, A. Neophyllachora gen nov. (Phyllachorales), three new species of Phyllachora from Poaceae and resurrection of Polystigmataceae (Xylariales). Mycosphere 2017, 8, 1598–1625. [Google Scholar] [CrossRef]
- MacCready, J.S.; Roggenkamp, E.M.; Gdanetz, K.; Chilvers, M.I. Elucidating the obligate nature and biological capacity of an invasive fungal corn pathogen. Mol. Plant-Microbe Interact. 2023, 36, 411–424. [Google Scholar] [CrossRef] [PubMed]
- Telenko, D.E.P.; Ross, T.J.; Shim, S.; Wang, Q.; Singh, R. Draft genome sequence resource for Phyllachora maydis—An obligate pathogen that causes tar spot of corn with recent economic impacts in the United States. Mol. Plant-Microbe Interact. 2020, 33, 884–887. [Google Scholar] [CrossRef]
- Bissonnette, S. Corn Disease Alert: New Fungal Leaf Disease “Tar Spot” Phyllachora Maydist Identified in 3 Northern Illinois Counties. The Bulletin: Pest Management and Crop Development Information for Illinois. Available online: https://farmdoc.illinois.edu/field-crop-production/diseases/corn-disease-alert-new-fungal-leaf-disease-tar-spot-phyllachora-maydis-identified-in-3-northern-illinois-counties.html (accessed on 16 January 2025).
- Roggenkamp, E.M.; Check, J.C.; Biswal, A.K.; Floyd, C.M.; Miles, L.A.; Nicolli, C.P.; Shim, S.; Salgado-Salazar, C.; Alakonya, A.E.; Malvick, D.K.; et al. Development of a qPCR assay for species-specific detection of the tar spot pathogen Phyllachora maydis. PhytoFrontiers 2023, 4, 61–71. [Google Scholar] [CrossRef]
- Theissen, F.; Sydow, H. Die Dothideales. Ann. Mycol. 1915, 13, 147–746. [Google Scholar]
- Hyde, K.; Noordeloos, M.E.; Savchenko, K. The 2024 Outline of Fungi and fungus-like taxa. Mycosphere 2024, 15, 5146–6239. [Google Scholar] [CrossRef]
- Dos Santos, M.D.M.; de Noronha Fonseca, M.E.; Silva Boiteux, L.; Câmara, P.E.A.; Dianese, J.C. ITS phylogeny and taxonomy of Phyllachora species on native Myrtaceae from the Brazilian Cerrado. Mycologia 2016, 108, 1141–1164. [Google Scholar]
- Kirk, P.M.; Cannon, P.F.; Minter, D.W.; Stalpers, J.A. Dictionary of the Fungi, 10th ed.; CABI Publishing: London, UK, 2008; p. 708. [Google Scholar]
- Li, J.C.; Wu, H.X.; Li, Y.; Li, X.H.; Song, J.Y.; Suwannarach, N.; Wijayawardene, N.N. Taxonomy, phylogenetic and ancestral area reconstruction in Phyllachora, with four novel species from Northwestern China. J. Fungi 2022, 8, 520. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, T. Flora Fungorum Sinicorum. Phyllachora; Science Press: Beijing, China, 2014; Volume 46. [Google Scholar]
- Li, X.L.; Wu, S.R.; Wang, C.L.; Feng, Y.L.; Zhao, C.Y.; Chen, Z.Q.; Yu, J.F.; Luo, R.; Promputtha, I.; Sun, D.F. Two new species of Phyllachora (Phyllachoraceae, Phyllachorales) on bamboo from China. Phytotaxa 2019, 425, 78–86. [Google Scholar] [CrossRef]
- Sun, Q.R.; Xu, X.L.; Zeng, Z.; Deng, Y.; Liu, F.; Gao, L.P.; Wang, F.H.; Yan, Y.Q.; Liu, Y.G.; Yang, C.L. Six species of Phyllachora with three new taxa on grass from Sichuan Province, China. J. Fungi 2024, 10, 588. [Google Scholar] [CrossRef]
- Bundhun, D.; Jeewon, R.; Dayarathne, M.C.; Bulgakov, T.S.; Khramtsov, A.K.; Aluthmuhandiram, J.; Pem, D.; To-Anun, C.; Hyde, K.D. A morpho-molecular re-appraisal of Polystigma fulvum and P. rubrum (Polystigma, Polystigmataceae). Phytotaxa 2019, 422, 209–224. [Google Scholar] [CrossRef]
- Jordi, L.P.; Zhang, F.M.; Ge, S. Plant biodiversity in China: Richly varied, endangered, and in need of conservation. Biodivers. Conserv. 2006, 15, 3983–4026. [Google Scholar]
- Tian, W.H.; Liu, J.W.; Jin, Y.; Chen, Y.P.; Zhou, Y.F.; Wu, K.; Su, P.W.; Guo, X.Y.; Wanasinghe, D.N.; Hyde, K.D.; et al. Morphological and phylogenetic studies of Ascomycota from gymnosperms in Sichuan Province, China. Mycosphere 2024, 15, 1794–1900. [Google Scholar] [CrossRef]
- Yang, C.L.; Xu, X.L.; Liu, Y.G.; Hyde, K.D.; Mckenzie, E.H. A new species of Phyllachora (Phyllachoraceae, Phyllachorales) on Phyllostachys heteroclada from Sichuan, China. Phytotaxa 2019, 392, 186–196. [Google Scholar] [CrossRef]
- Wang, J.; Seyler, B.C.; Ticktin, T.; Zeng, Y.; Ayu, K. An ethnobotanical survey of wild edible plants used by the Yi people of Liangshan Prefecture, Sichuan Province, China. J. Ethnobiol. Ethnomed. 2020, 16, 10. [Google Scholar] [CrossRef]
- Zeng, Q.; Lv, Y.C.; Xu, X.L.; Deng, Y.; Wang, F.H.; Liu, S.Y.; Liu, L.J.; Yang, C.L.; Liu, Y.G. Morpho-molecular characterization of microfungi associated with Phyllostachys (Poaceae) in Sichuan, China. J. Fungi 2022, 8, 702. [Google Scholar] [CrossRef]
- Madhushan, A.; Weerasingha, D.B.; Ilyukhin, E.; Taylor, P.W.; Ratnayake, A.S.; Liu, J.K.; Maharachchikumbura, S.S.N. From Natural Hosts to Agricultural Threats: The Evolutionary Journey of Phytopathogenic Fungi. J. Fungi 2025, 11, 25. [Google Scholar] [CrossRef]
- Toju, H.; Tanabe, A.S.; Yamamoto, S.; Sato, H. High-Coverage ITS primers for the DNA-based identification of Ascomycetes and Basidiomycetes in environmental samples. PLoS ONE 2012, 7, e40863. [Google Scholar] [CrossRef] [PubMed]
- Cubeta, M.; Echandi, E.; Abernethy, T.; Vilgalys, R. Characterization of anastomosis groups of binucleate Rhizoctonia species using restriction analysis of an amplified ribosomal RNA gene. Phytopathology 1991, 81, 1395–1400. [Google Scholar] [CrossRef]
- Vilgalys, R.; Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 1990, 172, 4238–4246. [Google Scholar] [CrossRef] [PubMed]
- Hibbett, D.S. Phylogenetic evidence for horizontal transmission of group I introns in the nuclear ribosomal DNA of mushroom-forming fungi. Mol. Biol. Evol. 1996, 13, 903–917. [Google Scholar] [CrossRef]
- Simon, L.; Lalonde, M.; Bruns, T.D. Specific amplification of 18S fungal ribosomal genes from vesicular-arbuscular endomycorrhizal fungi colonizing roots. Appl. Environ. Microbiol. 1992, 58, 291–295. [Google Scholar] [CrossRef]
- Paradis, E.; Claude, J.; Strimmer, K. APE: Analyses of Phylogenetics and evolution in R language. Bioinform. 2004, 20, 289–290. [Google Scholar] [CrossRef]
- Katoh, K.; Misawa, K.; Kuma, K.i.; Miyata, T. MAFFT: A novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res. 2002, 30, 3059–3066. [Google Scholar] [CrossRef]
- Capella-Gutiérrez, S.; Silla-Martínez, J.M.; Gabaldón, T. trimAl: A tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics 2009, 25, 1972–1973. [Google Scholar] [CrossRef]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef]
- Nguyen, L.-T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2014, 32, 268–274. [Google Scholar] [CrossRef]
- Huelsenbeck, J.P.; Ronquist, F. MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef]
- Rambaut, A.; Drummond, A.J.; Xie, D.; Baele, G.; Suchard, M.A. Posterior summarization in Bayesian phylogenetics using Tracer 1.7. Syst. Biol. 2018, 67, 901–904. [Google Scholar] [CrossRef] [PubMed]
- Bouckaert, R.; Vaughan, T.G.; Barido-Sottani, J.; Duchêne, S.; Fourment, M.; Gavryushkina, A.; Heled, J.; Jones, G.; Kühnert, D.; De Maio, N.; et al. BEAST 2.5: An advanced software platform for Bayesian evolutionary analysis. PLoS Comput. Biol. 2019, 15, e1006650. [Google Scholar] [CrossRef] [PubMed]
- Yu, G. Using ggtree to Visualize Data on Tree-Like Structures. Curr. Protoc. Bioinform. 2020, 69, e96. [Google Scholar] [CrossRef]
- Hennings, P. Einige neue Pilze aus Japan. II. Hedwigia 1904, 43, 140–146. [Google Scholar]
- Hosagoudar, V.B.; Manian, S.; Vasuki, S. Miscellaneous fungi from south india iv. J. Econ. Taxon. Bot. 1988, 12, 421–424. [Google Scholar]
- Fuckel, L. Symbolae mycologicae. Jahrbücher Des Nassau. Ver. Für Naturkunde 1870, 23, 216–219. [Google Scholar]
- Li, J.C.; Wu, H.X.; Song, J.Y. Two new Phyllachora species in Southwest China. Phytotaxa 2023, 578, 275–285. [Google Scholar] [CrossRef]
- Bhunjun, C.S.; Niskanen, T.; Suwannarach, N.; Wannathes, N.; Chen, Y.J.; McKenzie, E.H.C.; Maharachchikumbura, S.S.N.; Buyck, B.; Zhao, C.L.; Fan, Y.G.; et al. The numbers of fungi: Are the most speciose genera truly diverse? Fungal Divers. 2022, 114, 387–462. [Google Scholar] [CrossRef]
- Tamakaew, N. Tar spot fungi from Thailand. Mycosphere 2017, 8, 1054–1058. [Google Scholar] [CrossRef]
- Li, X.L.; Zhang, T.; Zhang, Z.Y.; Yang, H.Q.; Teng, X.Q. Four new records of Phyllachora from China. Mycosystema 2010, 29, 918–919. [Google Scholar]
- Li, X.; Yang, Z.; Wang, X.; Wang, Y.; Li, C. Two new species of graminicolous Phyllachora (Phyllachoraceae, Ascomycota). Mycosystema 2018, 37, 1127–1132. [Google Scholar]
- Liu, N.; Li, M. Phyllachora jianfengensis sp. nov. from China. Mycotaxon 2016, 130, 1039–1043. [Google Scholar] [CrossRef]
- Liu, N.; Wang, L.; Huang, G.; Guo, L. Phyllachora hainanensis sp. nov. from China. Mycotaxon 2015, 130, 237–239. [Google Scholar] [CrossRef]
- Mardones, M.; Trampe-Jaschik, T.; Oster, S.; Elliott, M.; Urbina, H.; Schmitt, I.; Piepenbring, M. Phylogeny of the order Phyllachorales (Ascomycota, Sordariomycetes): Among and within order relationships based on five molecular loci. Persoonia 2017, 39, 74–90. [Google Scholar] [CrossRef]
- Ariyawansa, H.A.; Hawksworth, D.L.; Hyde, K.D.; Jones, E.G.; Maharachchikumbura, S.S.N.; Manamgoda, D.S.; Thambugala, K.M.; Udayanga, D.; Camporesi, E.; Daranagama, A.; et al. Epitypification and neotypification: Guidelines with appropriate and inappropriate examples. Fungal Divers. 2014, 69, 57–91. [Google Scholar] [CrossRef]
- Cannon, P.F. A Revision of Phyllachora and Some Similar Genera on the Host Family Leguminosae; Mycol. Papers No.163; CAB International: Wallingford, UK, 1991; p. 302. [Google Scholar]
- Chen, Y.P.; Su, P.W.; Hyde, K.D.; Maharachchikumbura, S.S.N. Phylogenomics and diversification of Sordariomycetes. Mycosphere 2023, 14, 414–451. [Google Scholar] [CrossRef]
| Species | Voucher | Location | Host | Host Family | GenBank Accession Numbers | ||
|---|---|---|---|---|---|---|---|
| LSU | ITS | SSU | |||||
| Ascovaginospora stellipala T | ATCC 200781 | USA | Carex limosa | Cyperaceae | U85088 | - | U85087 |
| Camarotella costaricensis | MM-21 | Panama | Acrocomia aculeata | Arecaceae | KX430490 | KX451900 | KX451851 |
| Ca. costaricensis | MM-149 | Panama | Acrocomia aculeata | Arecaceae | KX430484 | KX451913 | KX451863 |
| Coccodiella calatheae T | MP5133 | Panama | Calathea crotalifera | Marantaceae | MF460370 | MF460366 | MF460376 |
| Co. melastomatum | CMU78543 | Venezuela | Miconia sp. | Melastomataceae | - | - | U78543 |
| Co. miconiae | ppMP1342 | Panama | Miconia sp. | Melastomataceae | KX430506 | MF460365 | KX451871 |
| Co. miconiicola | SO-15 | Ecuador | Graffenrieda sp. | Melastomataceae | MF460374 | MF460369 | MF460380 |
| Co. miconiicola | CBMAP-H290A | Panama | Miconia sp. | Melastomataceae | MF460373 | MF460368 | MF460379 |
| Co. toledoi | MM-165 | Ecuador | Miconia sp. | Melastomataceae | KX430488 | KX451917 | KX451865 |
| Neophyllachora cerradensis T | UB21908 | Brazil | Myrcia torta | Myrtaceae | - | KC683471 | - |
| N. cerradensis | UB21823 | Brazil | Myrcia torta | Myrtaceae | - | KC683470 | - |
| N. myrciae | UB21292 | Brazil | Myrcia pallens | Myrtaceae | - | KC683463 | - |
| N. myrciae | UB22192 | Brazil | Myrcia pallens | Myrtaceae | - | KC683476 | - |
| N. myrciariae T | UB21781 | Brazil | Myrciaria delicatula | Myrtaceae | - | KC683469 | - |
| N. religiosa T | MFLU 23-0258 | Thailand | Ficus religiosa | Moraceae | - | OQ821010 | - |
| N. religiosa | MFLU 23-0254 | Thailand | Ficus religiosa | Moraceae | - | OQ821006 | - |
| N. subcircinans | UB09748 | Brazil | Myrtaceae | Myrtaceae | - | KC683441 | - |
| N. subcircinans | UB21347 | Brazil | Myrtaceae | Myrtaceae | - | KC683466 | - |
| N. subcircinans | UB21747 | Brazil | Myrtaceae | Myrtaceae | - | KC683467 | KC902622 |
| N. truncatispora | UB14083 | Brazil | Myrcia camapuanensis | Myrtaceae | - | KC683448 | KC902614 |
| Phyllachora arthraxonis | MHYAU 073 | China | Arthraxon hispidus | Poaceae | MG269804 | MG269750 | - |
| P. arthraxonis | MHYAU 143 | China | Arthraxon hispidus | Poaceae | MG269805 | MG269751 | MH992445 |
| P. arundinellae | MHYAU 108 | China | Arundinella setosa | Poaceae | MG269815 | MG269761 | MH992450 |
| P. arundinellae | MHYAU 160 | China | Arundinella setosa | Poaceae | MG269816 | MG269762 | - |
| P. chloridis T | MFLU 15-0173 | Thailand | Chloris sp. | Poaceae | MF197499 | KY594026 | MF197505 |
| P. chloridis | MFLU 16-2980 | Thailand | Poaceae sp. | Poaceae | MF197500 | KY594027 | MF197506 |
| P. chloridis | SICAU 24-0053 | China | Chloris virgata | Poaceae | PP785310 | PP785299 | PP785321 |
| P. chloridis | SICAU 24-0054 | China | Chloris virgata | Poaceae | PP785311 | PP785300 | PP785322 |
| P. chloridis-virgatae | MHYAU 20136 | China | Chloris virgata | Poaceae | MG356685 | KY498122 | - |
| P. chloridis-virgatae | MHYAU 20137 | China | Chloris virgata | Poaceae | MG356686 | KY498092 | - |
| P. chongzhouensis T | SICAU 24-0044 | China | Phragmites australis | Poaceae | PP785312 | PP785301 | PP785323 |
| P. chongzhouensis | SICAU 24-0045 | China | Phragmites australis | Poaceae | PP785313 | PP785302 | PP785324 |
| P. chongzhouensis | HUEST 24.0137 | China | Pennisetum purpureum | Poaceae | PP464570 | PP472693 | PP464728 |
| P. chongzhouensis | LJT3 | China | Pennisetum purpureum | Poaceae | PP464571 | PP472694 | PP464729 |
| P. chongzhouensis | LJT4 | China | Pennisetum purpureum | Poaceae | PP464572 | PP472695 | PP464730 |
| P. chongzhouensis | LJT5 | China | Pennisetum purpureum | Poaceae | PP464573 | PP472696 | PP464731 |
| P. chongzhouensis | LJT6 | China | Pennisetum purpureum | Poaceae | PP464574 | PP472697 | PP464732 |
| P. chongzhouensis | LJT7 | China | Pennisetum purpureum | Poaceae | PP464575 | PP472698 | PP464733 |
| P. chongzhouensis | LMQ2 | China | Imperata cylindrica | Poaceae | PP464577 | PP472700 | PP464735 |
| P. chongzhouensis | LMQ3 | China | Imperata cylindrica | Poaceae | PP464578 | PP472701 | PP464736 |
| P. chongzhouensis | QQS6 | China | Imperata cylindrica | Poaceae | PP464613 | PP472736 | PP464771 |
| P. chongzhouensis | QQS7 | China | Imperata cylindrica | Poaceae | PP464614 | PP472737 | PP464772 |
| P. chongzhouensis | QQS13 | China | Imperata cylindrica | Poaceae | PP464608 | PP472731 | PP464766 |
| P. chongzhouensis | YM1 | China | Miscanthus floridulus | Poaceae | PP464624 | PP472747 | PP464782 |
| P. chongzhouensis | YM2 | China | Miscanthus floridulus | Poaceae | PP464625 | PP472748 | PP464783 |
| P. chongzhouensis | YM3 | China | Miscanthus floridulus | Poaceae | PP464626 | PP472749 | PP464784 |
| P. chrysopogonicola T | MFUH 16-2096 | Thailand | Chrysopogon zizanioides | Poaceae | MF372146 | MF372145 | - |
| P. cylindricae T | HKAS 135171 | China | Imperata cylindrica | Poaceae | PP464615 | PP472738 | PP464773 |
| P. cylindricae | SBZ3 | China | Imperata cylindrica | Poaceae | PP464616 | PP472739 | PP464774 |
| P. cylindricae | QQS2 | China | Panicum repens | Poaceae | PP464612 | PP472735 | PP464770 |
| P. cylindricae | QQS14 | China | Panicum repens | Poaceae | PP464609 | PP472732 | PP464767 |
| P. cylindricae | QQS15 | China | Panicum repens | Poaceae | PP464610 | PP472733 | PP464768 |
| P. cylindricae | QQS16 | China | Panicum repens | Poaceae | PP464611 | PP472734 | PP464769 |
| P. cynodonticola T | MFLU 16-2977 | Thailand | Cynodon sp. | Poaceae | MF197501 | KY594024 | MF197507 |
| P. cynodonticola | MFLU 16-2978 | Thailand | Imperata sp. | Poaceae | MF197502 | KY594025 | MF197508 |
| P. cynodontis | MHYAU 20043 | China | Cynodon dactylon | Poaceae | KY498081 | KY471329 | MH992435 |
| P. cynodontis | MHYAU 20131 | China | Cynodon dactylon | Poaceae | KY498079 | KY471327 | - |
| P. dendrocalami-hamiltonii T | MHYAU 221 | China | Dendrocalamus hamiltonii | Poaceae | MK614118 | - | - |
| P. dendrocalami-membranacei T | MHYAU 220 | China | Dendrocalamus membranaceus | Poaceae | MK614117 | MK614102 | - |
| P. dendrocalami-membranacei | MHYAU 222 | China | Dendrocalamus membranaceus | Poaceae | MK614119 | MK614103 | - |
| P. festucae T | HKAS 135172 | China | Festuca elata | Poaceae | PP464617 | PP472740 | PP464775 |
| P. festucae | SMGZ2 | China | Festuca elata | Poaceae | PP464618 | PP472741 | PP464776 |
| P. festucae | SMGZ3 | China | Festuca elata | Poaceae | PP464619 | PP472742 | PP464777 |
| P. flaccidudis T | IFRD9445 | China | Cenchrus flaccidus | Poaceae | ON072101 | ON075524 | ON072097 |
| P. graminis | HUEST 24.0131 | China | Imperata cylindrica | Poaceae | PP464623 | PP472746 | PP464781 |
| P. graminis | MYL293 | China | Festuca rubra | Poaceae | PP464622 | PP472745 | PP464780 |
| P. graminis | HLC73 | China | Imperata cylindrica | Poaceae | PP464565 | PP472688 | PP464723 |
| P. graminis | LMQ1 | China | Calamagrostis epigeios | Poaceae | PP464576 | PP472699 | PP464734 |
| P. graminis | AF257111 | Unknown | Unknown | Poaceae | - | AF257111 | - |
| P. graminis | RoKi3084 | Germany | Arrhenatherum elatius | Poaceae | - | - | KX451872 |
| P. graminis | UME31349 | Sweden | Unknown | Poaceae | - | - | AF064051 |
| P. graminis | SICAU 24-0051 | China | Lolium perenne | Poaceae | PP785306 | PP785295 | PP785317 |
| P. graminis | SICAU 24-0052 | China | Lolium perenne | Poaceae | PP785307 | PP785296 | PP785318 |
| P. heterocladae T | MFLU 18-1221 | China | Phyllostachys heteroclada | Poaceae | MK296472 | MK305902 | MK296468 |
| P. huiliensis T | SICAU 24-0048 | China | Bothriochloa ischaemum | Poaceae | PP785308 | PP785297 | PP785319 |
| P. huiliensis | SICAU 24-0049 | China | Bothriochloa ischaemum | Poaceae | PP785309 | PP785298 | PP785320 |
| P. imperatae | MHYAU 013 | China | Imperata cylindrica | Poaceae | MG269799 | MG269745 | - |
| P. imperatae | MHYAU 014 | China | Imperata cylindrica | Poaceae | MG269800 | MG269746 | - |
| P. indosasae | MHYAU 125 | China | Indosasa hispida | Poaceae | MG195662 | MG195637 | - |
| P. isachnicola T | MHYAU 179 | China | Isachne albens | Poaceae | MH018563 | MH018561 | - |
| P. isachnicola | MHYAU 180 | China | Isachne albens | Poaceae | MH018564 | MH018562 | - |
| P. jiaensis T | IFRD9448 | China | Chloris virgata | Poaceae | ON075440 | ON075527 | ON072100 |
| P. keralensis | MHYAU 20082 | China | Apluda mutica | Poaceae | MG269792 | KY498106 | MH992447 |
| P. keralensis | MHYAU 20083 | China | Apluda mutica | Poaceae | MG269793 | KY498088 | - |
| P. luzhouensis T | HKAS 135173 | China | Eleusine indica | Poaceae | PP464582 | PP472705 | PP464740 |
| P. luzhouensis | XGC6 | China | Chloris virgata | Poaceae | PP464583 | PP472706 | PP464741 |
| P. maydis | BPI 893231 | USA | Zea mays | Poaceae | - | KU184459 | - |
| P. maydis | BPI 910560 | Wisconsin | Zea mays | Poaceae | - | MG881846 | - |
| P. maydis | C18162-3 | USA | Zea mays | Poaceae | OL314408 | OL342794 | - |
| P. maydis | C18164-2 | USA | Zea mays | Poaceae | OL314409 | OL342796 | - |
| P. miscanthi | MHYAU 157 | China | Miscanthus sinensis | Poaceae | MG195668 | MG195643 | - |
| P. miscanthi | MHYAU 167 | China | Miscanthus sinensis | Poaceae | MG195669 | MG195644 | - |
| P. miscanthi | SICAU 24-0050 | China | Miscanthus floridulus | Poaceae | PP785305 | PP785294 | PP785316 |
| P. neidongensis | SICAU 24-0046 | China | Themeda triandra | Poaceae | PP785314 | PP785303 | PP785325 |
| P. neidongensis | SICAU 24-0047 | China | Themeda triandra | Poaceae | PP785315 | PP785304 | PP785326 |
| P. oplismeni-compositi | MHYAU 170 | China | Oplismenus compositus | Poaceae | MG195673 | MG195648 | - |
| P. palmifoliae T | HKAS 135170 | China | Setaria palmifolia | Poaceae | PP464568 | PP472691 | PP464726 |
| P. palmifoliae | LHS6 | China | Setaria palmifolia | Poaceae | PP464567 | PP472690 | PP464725 |
| P. palmifoliae | LHS21 | China | Setaria palmifolia | Poaceae | PP464569 | PP472692 | PP464727 |
| P. panicicola T | MFLU 16-2979 | China | Panicum sp. | Poaceae | MF197503 | KY594028 | MF197504 |
| P. panicicola | HUEST 24.0129 | China | Calamagrostis epigeios | Poaceae | PP464579 | PP472702 | PP464737 |
| P. panicicola | XGC3 | China | Calamagrostis epigeios | Poaceae | PP464580 | PP472703 | PP464738 |
| P. panicicola | XGC4 | China | Imperata cylindrica | Poaceae | PP464581 | PP472704 | PP464739 |
| P. panicicola | HLC1 | China | Calamagrostis epigeios | Poaceae | PP464559 | PP472682 | PP464717 |
| P. panicicola | HLC9 | China | Calamagrostis epigeios | Poaceae | PP464566 | PP472689 | PP464724 |
| P. panicicola | HLC14 | China | Imperata cylindrica | Poaceae | PP464560 | PP472683 | PP464718 |
| P. pogonatheri | MHYAU 070 | China | Pogonatherum crinitum | Poaceae | MG269801 | MG269747 | - |
| P. pogonatheri | MHYAU 071 | China | Pogonatherum paniceum | Poaceae | MG269802 | MG269748 | - |
| P. qualeae | UB21159 | Brazil | Qualea multiflora | Vochysiaceae | - | KU682781 | - |
| P. qualeae | UB21771 | Brazil | Qualea multiflora | Vochysiaceae | - | KU682780 | - |
| P. sacchari-spontanei | MHYAU 087 | China | Saccharum spontaneum | Poaceae | MG195670 | MG195645 | - |
| P. sandiensis T | IFRD9446 | China | Cenchrus flaccidus | Poaceae | ON075528 | ON075525 | ON072098 |
| P. siamensisT | CMU TAR08 | Thailand | Eleusine indica | Poaceae | MZ749659 | MZ749653 | MZ749661 |
| P. siamensis | CMU TAR12 | Thailand | Chloris barbata | Poaceae | MZ749660 | MZ749654 | MZ749662 |
| P. sinobambusae | MHYAU 085 | China | Sinobambusa tootsik | Poaceae | MG195655 | MG195630 | - |
| P. sphaerocaryi T | MHYAU 178 | China | Sphaerocaryum malaccense | Poaceae | - | MH018560 | - |
| P. sphaerocaryi | MHYAU 217 | China | Sphaerocaryum malaccense | Poaceae | MK614114 | MK614100 | - |
| P. thysanolaenae T | MFLU 16-2071 | Thailand | Thysanolaena maxima | Poaceae | - | - | MF372147 |
| P. virgatae T | IFRD9447 | China | Chloris virgata | Poaceae | ON075439 | ON075526 | ON072099 |
| P. wenchuanensis T | HKAS 135174 | China | Panicum virgatum | Poaceae | PP464557 | PP472680 | PP464715 |
| P. wenchuanensis | DMG2 | China | Panicum virgatum | Poaceae | PP464558 | PP472681 | PP464716 |
| P. wenchuanensis | XQDXG258 | China | Lolium perenne | Poaceae | PP464620 | PP472743 | PP464778 |
| P. wenchuanensis | XQDXG286 | China | Lolium perenne | Poaceae | PP464621 | PP472744 | PP464779 |
| P. wenchuanensis | HLC4 | China | Calamagrostis epigeios | Poaceae | PP464562 | PP472685 | PP464720 |
| P. wenchuanensis | HLC5 | China | Calamagrostis epigeios | Poaceae | PP464563 | PP472686 | PP464721 |
| P. wenchuanensis | HLC20 | China | Imperata cylindrica | Poaceae | PP464561 | PP472684 | PP464719 |
| P. wenchuanensis | HLC50 | China | Calamagrostis epigeios | Poaceae | PP464564 | PP472687 | PP464722 |
| P. wenchuanensis | PXC1 | China | Imperata cylindrica | Poaceae | PP464584 | PP472707 | PP464742 |
| P. wenchuanensis | PXC2 | China | Imperata cylindrica | Poaceae | PP464595 | PP472718 | PP464753 |
| P. wenchuanensis | PXC3 | China | Imperata cylindrica | Poaceae | PP464601 | PP472724 | PP464759 |
| P. wenchuanensis | PXC4 | China | Imperata cylindrica | Poaceae | PP464602 | PP472725 | PP464760 |
| P. wenchuanensis | PXC5 | China | Imperata cylindrica | Poaceae | PP464603 | PP472726 | PP464761 |
| P. wenchuanensis | PXC6 | China | Imperata cylindrica | Poaceae | PP464604 | PP472727 | PP464762 |
| P. wenchuanensis | PXC7 | China | Calamagrostis epigeios | Poaceae | PP464605 | PP472728 | PP464763 |
| P. wenchuanensis | PXC8 | China | Imperata cylindrica | Poaceae | PP464606 | PP472729 | PP464764 |
| P. wenchuanensis | PXC9 | China | Calamagrostis epigeios | Poaceae | PP464607 | PP472730 | PP464765 |
| P. wenchuanensis | PXC10 | China | Lolium perenne | Poaceae | PP464585 | PP472708 | PP464743 |
| P. wenchuanensis | PXC11 | China | Panicum virgatum | Poaceae | PP464586 | PP472709 | PP464744 |
| P. wenchuanensis | PXC12 | China | Elymus dahuricus | Poaceae | PP464587 | PP472710 | PP464745 |
| P. wenchuanensis | PXC13 | China | Calamagrostis epigeios | Poaceae | PP464588 | PP472711 | PP464746 |
| P. wenchuanensis | PXC14 | China | Lolium perenne | Poaceae | PP464589 | PP472712 | PP464747 |
| P. wenchuanensis | PXC15 | China | Lolium perenne | Poaceae | PP464590 | PP472713 | PP464748 |
| P. wenchuanensis | PXC16 | China | Lolium perenne | Poaceae | PP464591 | PP472714 | PP464749 |
| P. wenchuanensis | PXC17 | China | Elymus dahuricus | Poaceae | PP464592 | PP472715 | PP464750 |
| P. wenchuanensis | PXC18 | China | Lolium perenne | Poaceae | PP464593 | PP472716 | PP464751 |
| P. wenchuanensis | PXC19 | China | Panicum virgatum | Poaceae | PP464594 | PP472717 | PP464752 |
| P. wenchuanensis | PXC20 | China | Elymus dahuricus | Poaceae | PP464596 | PP472719 | PP464754 |
| P. wenchuanensis | PXC21 | China | Panicum virgatum | Poaceae | PP464597 | PP472720 | PP464755 |
| P. wenchuanensis | PXC22 | China | Panicum virgatum | Poaceae | PP464598 | PP472721 | PP464756 |
| P. wenchuanensis | PXC23 | China | Lolium perenne | Poaceae | PP464599 | PP472722 | PP464757 |
| P. wenchuanensis | PXC24 | China | Lolium perenne | Poaceae | PP464600 | PP472723 | PP464758 |
| P. xinpingensis | IFRD9465 | China | Chrysopogon aciculatus | Poaceae | OP359416 | OP359398 | - |
| P. yuanjiangensis | IFRD9466 | China | Arundinella setosa | Poaceae | OP359417 | OP359399 | OP359400 |
| P. yushaniae-falcatiauritae | MHYAU 123 | China | Yushania falcatiaurita | Poaceae | MG195656 | MG195631 | - |
| P. yushaniae-polytrichae | MHYAU 122 | China | Yushania polytricha | Poaceae | MG195657 | MG195632 | - |
| P. yushaniae-polytrichae | MHYAU 158 | China | Yushania polytricha | Poaceae | MG195658 | MG195633 | - |
| Telimena bicincta | MM-133 | Costa Rica | Picramnia antidesma | Picramniaceae | KX430478 | KX451910 | KX451861 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Su, P.; Liao, Y.; Jin, Y.; Chen, Y.; Madhushan, A.; Liu, J.-K.; Maharachchikumbura, S.S.N. Five New Phyllachora Species from Tar Spot Fungi on Poaceae in Sichuan China. J. Fungi 2025, 11, 208. https://doi.org/10.3390/jof11030208
Su P, Liao Y, Jin Y, Chen Y, Madhushan A, Liu J-K, Maharachchikumbura SSN. Five New Phyllachora Species from Tar Spot Fungi on Poaceae in Sichuan China. Journal of Fungi. 2025; 11(3):208. https://doi.org/10.3390/jof11030208
Chicago/Turabian StyleSu, Pengwei, Yuechi Liao, Yan Jin, Yanpeng Chen, Asanka Madhushan, Jian-Kui Liu, and Sajeewa S. N. Maharachchikumbura. 2025. "Five New Phyllachora Species from Tar Spot Fungi on Poaceae in Sichuan China" Journal of Fungi 11, no. 3: 208. https://doi.org/10.3390/jof11030208
APA StyleSu, P., Liao, Y., Jin, Y., Chen, Y., Madhushan, A., Liu, J.-K., & Maharachchikumbura, S. S. N. (2025). Five New Phyllachora Species from Tar Spot Fungi on Poaceae in Sichuan China. Journal of Fungi, 11(3), 208. https://doi.org/10.3390/jof11030208








