Simple Summary
The gut plays a critical role in animal and human health, with inflammatory conditions causing significant welfare and economic concerns. This review examines how the Nrf2 signaling pathway protects the intestine from damage caused by oxidative stress and inflammation. We explore various natural compounds (polyphenols, terpenoids, alkaloids), probiotics, and nutritional interventions that activate Nrf2 to enhance antioxidant defenses, suppress inflammatory responses, strengthen intestinal barriers, and beneficially modulate gut microbiota. The evidence demonstrates that Nrf2 activation represents a promising multi-targeted therapeutic strategy for managing intestinal inflammatory disorders in both clinical and livestock settings.
Abstract
This review synthesizes research on nuclear factor erythroid 2-related factor 2 (Nrf2) in intestinal health across human, livestock, and mouse models. The Nrf2 signaling pathway serves as a master regulator of cellular antioxidant defenses and a key therapeutic target for intestinal inflammatory disorders, including ulcerative colitis and Crohn’s disease. The interplay between oxidative stress, Nrf2 signaling, and NF-κB inflammatory cascades represents a critical axis in the pathogenesis and resolution of intestinal inflammation. Under normal physiological conditions, Nrf2 remains sequestered in the cytoplasm by Kelch-like ECH-associated protein 1 (Keap1), which facilitates its ubiquitination and proteasomal degradation. However, during oxidative stress, reactive oxygen species (ROS) and electrophilic compounds modify critical cysteine residues on Keap1, disrupting the Keap1-Nrf2 interaction and enabling Nrf2 nuclear translocation. Once in the nucleus, Nrf2 binds to antioxidant response elements (ARE) in the promoter regions of genes encoding phase II detoxifying enzymes and antioxidant proteins, including heme oxygenase-1 (HO-1), NAD(P)H quinone oxidoreductase 1 (NQO1), and glutamate-cysteine ligase. This comprehensive review synthesizes current evidence demonstrating that activation of Nrf2 signaling confers protection against intestinal inflammation through multiple interconnected mechanisms: suppression of NF-κB-mediated pro-inflammatory cascades, enhancement of cellular antioxidant capacity, restoration of intestinal barrier integrity, modulation of immune cell function, and favorable alteration of gut microbiota composition. We systematically examine a diverse array of therapeutic agents targeting Nrf2 signaling, including bioactive peptides, natural polyphenols, flavonoids, terpenoids, alkaloids, polysaccharides, probiotics, and synthetic compounds. The mechanistic insights and therapeutic evidence presented underscore the translational potential of Nrf2 pathway modulation as a multi-targeted strategy for managing intestinal inflammatory conditions and restoring mucosal homeostasis.
Keywords:
Nrf2; NF-κB; intestinal health; oxidative stress; inflammation; antioxidants; gut barrier function; livestock 1. Introduction
Inflammatory bowel diseases (IBD), encompassing ulcerative colitis (UC) and Crohn’s disease (CD), represent a group of chronic, relapsing inflammatory disorders of the gastrointestinal tract that afflict millions of individuals worldwide [1,2,3]. These conditions impose a substantial burden on healthcare systems and significantly diminish patients’ quality of life through persistent symptoms including abdominal pain, diarrhea, rectal bleeding, weight loss, and systemic complications [4,5,6,7]. The pathogenesis of IBD is multifactorial, involving complex interactions between genetic susceptibility, environmental triggers, intestinal microbiota dysbiosis, dysregulated immune responses, and impaired mucosal barrier function [8,9]. A hallmark feature of IBD is the generation of excessive reactive oxygen species (ROS) and sustained oxidative stress within the intestinal mucosa, which perpetuates tissue damage, amplifies inflammatory cascades, and compromises epithelial barrier integrity [10,11,12].
The nuclear factor erythroid 2-related factor 2 (Nrf2) serves as the master transcriptional regulator orchestrating cellular antioxidant and cytoprotective responses [13,14,15]. Under homeostatic conditions, Nrf2 activity is tightly regulated by Kelch-like ECH-associated protein 1 (Keap1), which promotes Nrf2 degradation to maintain low basal expression of antioxidant genes [16]. Upon exposure to oxidative stress or electrophilic compounds, Nrf2 stabilizes and translocates to the nucleus, activating transcription of phase II detoxifying enzymes, antioxidant proteins (heme oxygenase-1, superoxide dismutase, catalase, glutathione peroxidase), glutathione biosynthesis enzymes, and numerous other cytoprotective mediators [17,18,19,20]. In intestinal epithelium, Nrf2 activation enhances barrier function, promotes epithelial regeneration, modulates immune responses, and influences microbiota composition, thereby protecting against oxidative damage and inflammation [21,22,23].
The therapeutic potential of Nrf2 pathway modulation in intestinal health has gained considerable attention in recent years [24,25]. Accumulating evidence from preclinical studies demonstrates that pharmacological activation of Nrf2 signaling through diverse natural products, synthetic compounds, and dietary interventions can ameliorate experimental colitis, reduce intestinal inflammation, restore barrier integrity, and modulate gut microbiota composition [26,27]. These beneficial effects are mediated through multiple interconnected mechanisms, including enhanced antioxidant capacity, suppression of pro-inflammatory signaling cascades [particularly the nuclear factor kappa B (NF-κB) pathway], inhibition of inflammasome activation, modulation of immune cell function, promotion of intestinal stem cell activity, and favorable alteration of microbial ecology [28,29]. The cross-talk between Nrf2 and NF-κB pathways represents a particularly important regulatory axis, as activation of Nrf2 can suppress NF-κB-driven inflammatory gene expression through multiple mechanisms, including direct protein–protein interactions, competition for transcriptional co-activators, and indirect effects mediated by Nrf2-induced antioxidant proteins that reduce ROS-dependent NF-κB activation [28,29,30].
These mechanisms underpin their ability to improve gut morphology, modulate microbiota, enhance immune function, and alleviate stress-induced intestinal damage, thereby improving growth performance [31]. Despite these promising physiological and performance-related benefits, current evidence remains limited and heterogeneous across different species and experimental conditions. While their potential in animal husbandry is promising, future research must address gaps in comparative efficacy, synergistic effects, and long-term safety to enable practical applications [32,33,34]. This comprehensive review aims to synthesize current understanding of Nrf2 signaling in intestinal health, with particular emphasis on the molecular mechanisms underlying the protective effects of Nrf2 activation against intestinal inflammation. We systematically examine the intricate interplay between oxidative stress, Nrf2 signaling, and NF-κB inflammatory cascades in the pathogenesis and resolution of intestinal inflammation. With a focus on translational applications across veterinary and clinical settings, we provide a detailed analysis of diverse therapeutic agents that target Nrf2 signaling to ameliorate intestinal inflammatory conditions, including natural bioactive compounds, probiotics, dietary interventions, and synthetic molecules.
2. Literature Search Strategy
A targeted literature search was conducted using PubMed, google scholar, Web of Science, and Scopus databases with the following key terms: Nrf2 signaling, intestinal health, inflammatory bowel disease, intestinal inflammation, colitis, ulcerative colitis, and oxidative stress. Boolean operators (AND, OR) were used to combine search terms. Studies involving livestock, in vitro models, and mouse models were included. Reference lists of relevant reviews were also screened for additional studies. To capture recent advancements in Nrf2 signaling as a therapeutic target for intestinal health, the search was limited to articles published between 2019 and 2025. Only peer-reviewed original research articles and reviews published in English and indexed in science citation index (SCI) journals were considered for inclusion. Conference abstracts, non-peer-reviewed publications, and non-English articles were excluded.
3. The Interplay of Oxidative Stress, Nrf2 Signaling, and NF-κB Pathway in Intestinal Inflammation
3.1. Oxidative Stress in IBD Pathophysiology
Inflammatory bowel diseases exhibit pathologically increased reactive oxygen species (ROS) generation through multiple enzymatic sources, including nicotinamide adenine dinucleotide phosphate (NADPH) oxidases (NOX1, NOX2), mitochondrial electron transport chain dysfunction, xanthine oxidase, and myeloperoxidase released from activated neutrophils [35,36]. This oxidative burden overwhelms endogenous antioxidant defenses, manifesting as elevated lipid peroxidation products (malondialdehyde, 4-hydroxynonenal), protein carbonylation, oxidized glutathione ratios, and DNA oxidation markers (8-hydroxy-2′-deoxyguanosine) [37,38]. The resulting redox dysregulation perpetuates mucosal injury through multiple mechanisms. Oxidative modification of membrane phospholipids compromises epithelial barrier integrity through tight junction protein degradation (occludin, claudins, ZO-1) and cytoskeletal disruption, facilitating paracellular permeability and bacterial translocation [39,40,41]. ROS function as critical second messengers activating redox-sensitive transcription factors, particularly NF-κB, through IκB kinase phosphorylation and subsequent IκBα degradation, thereby amplifying pro-inflammatory gene expression and cytokine production (TNF-α, IL-1β, IL-6) [42].
3.2. Nrf2-Keap1 Regulatory Architecture
The Keap1-Nrf2 axis functions as a molecular rheostat governing cellular redox homeostasis and cytoprotective responses [13,14,15]. Under homeostatic conditions, Keap1 homodimers sequester Nrf2 in the cytoplasm through BTB (broad complex, tramtrack, bric-à-brac) domain-mediated association with the Cullin 3-Rbx1 E3 ubiquitin ligase complex [43]. This molecular architecture maintains constitutive Nrf2 ubiquitination and rapid proteasomal turnover, ensuring low basal expression of ARE-dependent genes [44]. Keap1 contains multiple reactive cysteine residues (Cys151, Cys273, Cys288) functioning as electrophile sensors that undergo post-translational modifications (oxidation, alkylation, nitrosylation) upon encountering oxidative or electrophilic stressors [45]. These modifications disrupt the hinge-and-latch mechanism between Keap1’s double glycine repeat (DGR) domain and Nrf2’s Neh2 domain, stabilizing Nrf2 and promoting nuclear accumulation [46]. Nuclear Nrf2 heterodimerizes with small Maf proteins (MafF, MafG, MafK) and binds antioxidant response elements (ARE) in promoter regions, recruiting transcriptional co-activators (CBP/p300) to orchestrate expression of >200 cytoprotective genes encoding phase II detoxifying enzymes (NQO1, GSTs, UGTs), antioxidant proteins (HO-1, GCLC/M, thioredoxin, peroxiredoxins), and metabolic regulators [46,47].
3.3. Nrf2-NF-κB Crosstalk in Intestinal Inflammation
The Nrf2 and NF-κB signaling pathways engage in extensive bidirectional crosstalk that determines the balance between cytoprotection and inflammation in intestinal tissues [25,28,48]. ROS serve as obligate second messengers driving NF-κB activation through oxidative modification of pathway components, including IκB kinase complex phosphorylation and IκBα degradation [49]. Conversely, Nrf2-mediated induction of antioxidant enzymes attenuates ROS-dependent NF-κB activation, creating a negative feedback loop [50]. Direct protein–protein interactions provide additional regulatory complexity: Keap1 associates with IKKβ modulating its activity, while Nrf2 physically interacts with p65/RelA, either sequestering it in the cytoplasm or competing for limiting transcriptional co-activators (CBP/p300) in the nucleus [51]. Temporal dynamics distinguish these pathways: NF-κB exhibits rapid, transient activation kinetics (minutes) suited for immediate inflammatory responses, whereas Nrf2 demonstrates delayed but sustained activation (hours) appropriate for establishing adaptive cytoprotection [52].
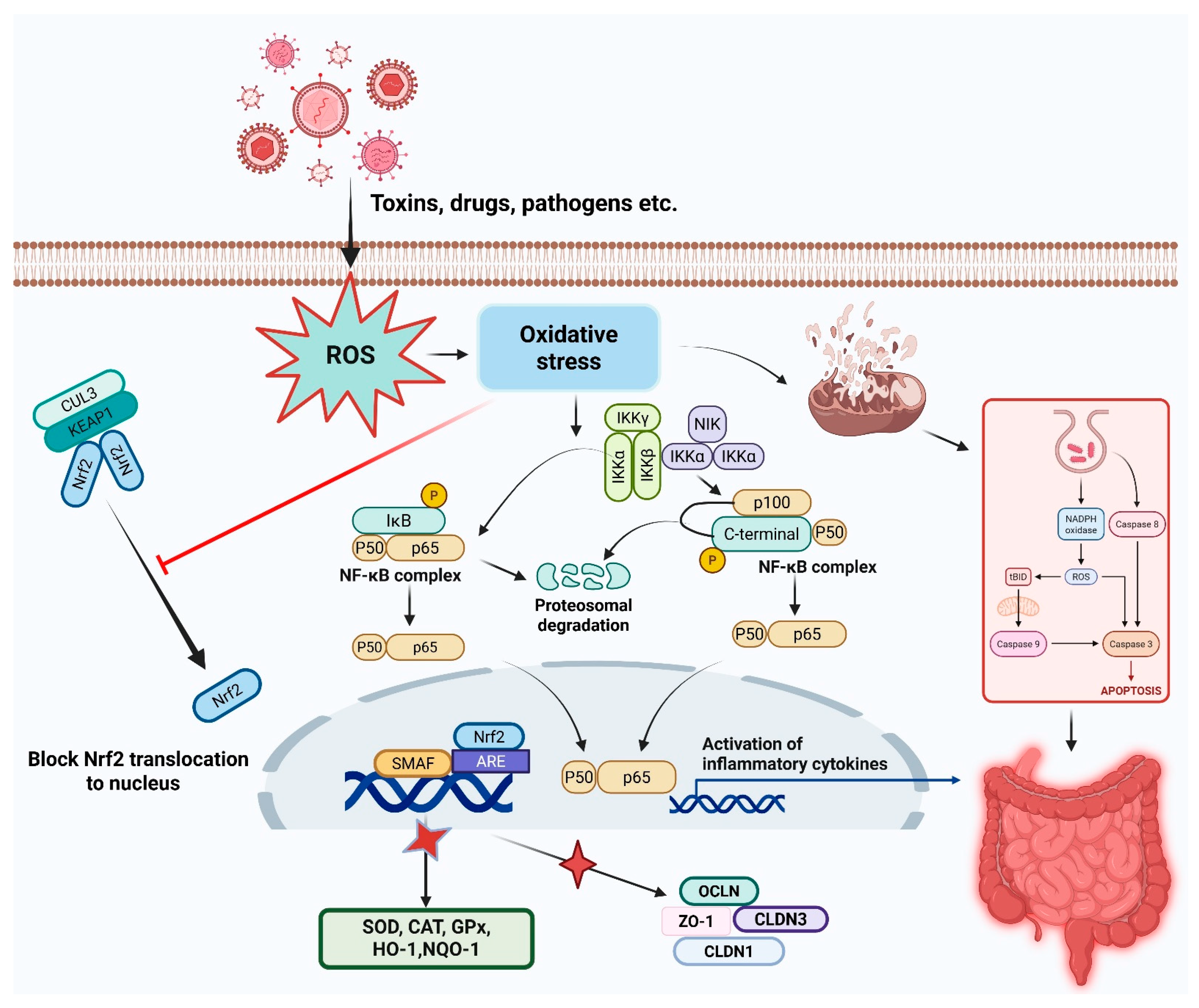
In experimental colitis models, Nrf2−/− mice display heightened disease severity with exacerbated histological damage, increased NF-κB activity, and elevated inflammatory cytokine expression [48,53]. Pharmacological Nrf2 activation confers protection through multi-modal mechanisms: enhanced antioxidant capacity, attenuated pro-inflammatory signaling, preserved epithelial barrier function via tight junction protein upregulation, favorable modulation of immune cell phenotypes (M2 macrophage polarization, regulatory T cell expansion), and beneficial microbiota compositional shifts [53,54,55]. These integrated effects collectively restore intestinal homeostasis and promote mucosal healing in inflammatory conditions. The mechanisms by which oxidative stress and various environmental, dietary, and pathological agents trigger intestinal diseases are summarized in Figure 1.
Figure 1.
Schematic representation of how various environmental, dietary, and pathological agents induce oxidative stress and trigger intestinal diseases including inflammatory bowel diseases. The figure illustrates the pathways leading from oxidative stress inducers to cellular damage, barrier dysfunction, and chronic inflammation in the gastrointestinal tract.
4. Therapeutic Agents Targeting Nrf2 Signaling for Intestinal Health
4.1. Natural Phenolic Compounds
The extensive body of research on natural phenolic compounds as Nrf2 activators underscores their significant therapeutic potential for intestinal inflammatory disorders. The structural diversity of these compounds—from simple phenolic acids to complex flavonoids and stilbenoids—is matched by a consistent ability to enhance antioxidant defenses and suppress pro-inflammatory pathways. Importantly, these compounds have demonstrated efficacy across multiple experimental platforms, including rodent colitis models, porcine and avian livestock systems, and human intestinal cell lines, supporting their translational relevance for both agricultural applications and human inflammatory bowel disease (IBD) therapeutics. The key compounds, their sources, mechanisms, and outcomes discussed in this section are systematically summarized in Table 1.
4.1.1. Stilbenoids
Resveratrol demonstrates multifaceted Nrf2-mediated intestinal protection beyond antioxidant effects. In DSS models, it activates Nrf2/HO-1 signaling, enhancing transepithelial electrical resistance and upregulating tight junction proteins ZO-1 and Occludin [54]. Resveratrol simultaneously suppresses NF-κB p65 translocation while activating Nrf2/HO-1, addressing both oxidative and inflammatory injury components in zearalenone-induced damage [48]. Network pharmacology confirms its effects operate primarily through Nrf2-dependent oxidative stress modulation [54]. Notably, resveratrol counteracts ferroptosis through Nrf2/FTH1/GPX4 axis activation in exercise-induced intestinal damage [56], positioning it as a prototype for understanding multi-pathway protection through upstream Nrf2 activation. Collectively, stilbenoids, particularly resveratrol, represent well-characterized Nrf2 activators with robust preclinical evidence supporting their development as therapeutic agents for oxidative stress-related intestinal pathologies in both veterinary and human medicine.
4.1.2. Phenolic Acids
Phenolic acids demonstrate mechanistic precision in Nrf2 activation while revealing gut microbiota’s critical role in therapeutic efficacy. Caffeic acid dietary administration in piglets enhanced growth and gut health, with in vitro H2O2 protection completely abrogated by Nrf2 inhibitor ML385, establishing unequivocal pathway dependency [21]. Ellagic acid exhibits bidirectional microbiota interactions, with intestinal biotransformation into bioavailable urolithin metabolites that activate Nrf2 while inhibiting NF-κB [57]. Ellagic acid also modulates microbiome composition, reducing oxidative stress via Nrf2/ROS/ NOD-like receptor pyrin domain-containing protein 3 (NLRP3) axis [58], and mitigates paraquat-induced stress through Nrf2 translocation and HO-1/NQO1 upregulation [59].
Paralleling these mechanistic insights, chlorogenic acid exemplifies dual-pathway regulation, inhibiting TLR4/NF-κB while activating Nrf2-mediated antioxidant responses. In piglets, supplementation elevated systemic antioxidant capacity and modulated ileal microbiota [49]. Its protective effects extend to the gut–brain axis, protecting against sleep deprivation-induced cognitive deficits through Nrf2/PPAR activation while enhancing barrier function [60]. The phenolic acid family demonstrates consistent dose-dependent Nrf2/HO-1 activation: ferulic and quinic acids reduce inflammation in acetic acid-induced colitis [61], while coumaric and syringic acids alleviate colitis with significant TNF-α and IL-1β reductions [62]. Tannic acid engages the p62-Keap1-Nrf2 pathway while inhibiting TLR4-NF-κB-NLRP3 signaling against ETEC K88-induced diarrhea [63], suggesting autophagy-dependent Nrf2 activation mechanisms. Taken together, phenolic acids constitute a versatile class of Nrf2 activators whose microbiota-mediated biotransformation and dual antioxidant-anti-inflammatory actions establish them as promising candidates for translational development across agricultural and clinical settings.
4.1.3. Coumarins
Coumarins combine Nrf2-mediated protection with direct antimicrobial activity. For example, Esculetin exhibits antibacterial activity against multidrug-resistant Salmonella typhimurium through downregulation of the T3SS-1 virulence system, preventing bacterial invasion of intestinal cells, and in infected mice, esculetin ameliorated cecal injury and corrected microbial dysbiosis through Nrf2 pathway activation [64]. This dual antimicrobial and cytoprotective mechanism is complemented by scopoletin, which demonstrates the characteristic dual-pathway mechanism by suppressing NF-κB while activating Nrf2 to upregulate HO-1 and NQO1, with restoration of tight junction proteins confirming barrier-protective effects [65]. The dual antimicrobial-cytoprotective properties of coumarins position them as particularly valuable therapeutic candidates in the context of rising antimicrobial resistance. Their ability to simultaneously target bacterial virulence mechanisms and reinforce host intestinal defenses through Nrf2 activation represents a novel therapeutic strategy applicable to both food animal production—where reducing antibiotic usage is a priority—and human gastrointestinal infections complicated by dysbiosis. Thus, coumarins represent a distinct subclass of phenolic compounds whose combined antimicrobial and Nrf2-activating properties offer unique advantages for managing infectious enteritis across species.
4.1.4. Flavonoids
Flavonoids represent the most extensively studied polyphenol class for intestinal Nrf2 modulation. Morin reverses deoxynivalenol-induced damage through direct Keap1 inhibition, activating Nrf2 and enhancing intestinal stem cell proliferation [24]. Morin hydrate functions as a dual Nrf2 and PPARγ agonist with stable molecular interactions confirmed by docking studies [66]. Complementing this pharmacological approach, Apigenin pretreatment alleviates ischemia/reperfusion injury by upregulating Nrf2, HO-1, and tight junction proteins, with Nrf2 knockdown abolishing protection [26].
Building on these findings, naringenin activates Nrf2 components (HO-1, NQO1) while suppressing NF-κB, reducing TNF-α and IL-1β in UC models [67]. Naringin modulates stress and inflammation through coordinated TLR4/p38 MAPK/NF-κB inhibition and Nrf2 activation, affecting tight junctions, mucin 2, and permeability markers [68]. Quercetin provides compelling Nrf2-dependent barrier protection, with effects requiring Nrf2 expression [69]. Dose-dependent activation reverses DON-induced claudin-4 reduction, completely blocked by brusatol [70]. Quercetin also protects against combined zearalenone and lipopolysaccharide (LPS) toxicity through upregulation of HO-1, SOD2, and NQO1 [71], and against radiation-induced injury through ROS curtailment [72].
Extending beyond conventional flavonoids, Genistein reverses H2O2-induced Nrf2 suppression, protecting barrier integrity [46], and alleviates IBD through dual mechanisms: enriching Akkermansia muciniphila while activating GPR30-Nrf2 axis to suppress NLRP3 inflammasome [73]. Paralleling this multifaceted approach, baicalin attenuates barrier damage through AMPK/Nrf2 modulation, with AMPK knockdown abolishing protection [74]. In addition, baicalein suppresses ferroptosis by upregulating GPX4 and FTH1 via Nrf2 while modulating gut microbiota [75]. Moreover, rutin improves barrier integrity through Nrf2/Keap1 activation with concurrent cecal microbiota modulation [76], and taxifolin demonstrates consistent protection across multiple stress models [40,77]. Expanding this therapeutic repertoire, additional flavonoids with documented Nrf2-mediated protection include pectolinarigenin [78], wogonin [79], and Eucommia ulmoides flavonoids [50]. Overall, flavonoids constitute the most extensively validated class of natural Nrf2 activators, with mechanistically diverse compounds offering multiple therapeutic entry points for intestinal inflammatory disorders across veterinary and human medical applications.
4.1.5. Specialized Polyphenols
Curcumin represents the most extensively characterized Nrf2 activator. Dietary curcumin mitigates AFB1 toxicity by upregulating Nrf2, HO-1, and NQO-1 systemically [43]. Mechanistically, it activates SIRT1/NRF2 while inhibiting TLR4, reducing NLRP3-mediated pyroptosis [80]. Notably, nanoparticle formulations (Theracurmin) by targeting NRF2 signaling, significantly attenuate DSS-induced colitis while inducing regulatory T cells [81]. Complementing these findings, epigallocatechin gallate (EGCG) alleviates cisplatin-induced injury through Nrf2/Keap1 activation with microbiota and autophagy modulation, with ML385 completely reversing protection [54]. Extending this mechanistic diversity, carthamin yellow activates Nrf2/GPX4 axis to counter ferroptosis in UC, with effects abolished in Nrf2-knockout mice and with ML385 treatment [82]. Moreover, hydroxytyrosol concurrently activates PI3K/Akt-Nrf2 signaling and promotes mitophagy, with disruption of either pathway compromising protection [83], revealing synergistic mechanisms whereby Nrf2 clears ROS through antioxidant upregulation while mitophagy eliminates damaged mitochondria. These findings demonstrate that specialized polyphenols exemplify the successful translation of Nrf2-targeting compounds from preclinical models to clinical application, with advanced formulation strategies and emerging mechanistic insights continuing to expand their therapeutic potential.

Table 1.
Natural Phenolic Compounds Targeting Nrf2 Signaling for Intestinal Health.
Table 1.
Natural Phenolic Compounds Targeting Nrf2 Signaling for Intestinal Health.
| Model Category | Compound | Category | Model System | Key Pathways | Ref. |
|---|---|---|---|---|---|
| Rodent Models | Resveratrol | Stilbenoid | DSS-induced colitis mice; ZEA-injured mice; High-intensity exercise mice | Nrf2/HO-1, NF-κB; Nrf2/FTH1/GPX4 | [6,11,48,56] |
| Resveratrol Analog C33 | Stilbenoid | Mouse colitis model; Nrf2-KO mice | Nrf2 | [84] | |
| Sericic Acid | Triterpenoid | DSS-induced colitis mice | NF-κB, Nrf2 | [85] | |
| Dehydrocostus Lactone | Sesquiterpene | DSS-induced colitis | NF-κB, Keap1/Nrf2 | [86] | |
| SLRF | Sesquiterpene | DSS-induced UC | Nrf2-Hmox-1, NF-κB/MAPK | [87] | |
| Epoxymicheliolide | Sesquiterpene | DSS-induced colitis | TAK1-NF-κB, Keap1/Nrf2 | [88] | |
| Gingerenone A | Phenolic | DSS-induced colitis mice | Nrf2-Gpx4 | [89] | |
| Carnosic Acid | Diterpene | DSS-induced colitis mice | Microbiota → Nrf2 | [90] | |
| Sesamin | Lignan | DSS-induced murine colitis | AKT/ERK → Nrf2 | [91] | |
| Ellagic Acid | Phenolic Acid | DSS-induced colitis mice | Nrf2, NF-κB, ROS/ NLRP3 | [57,58] | |
| Chlorogenic Acid | Phenolic Acid | Sleep-deprived mice | Nrf2/PPAR | [60] | |
| Ferulic/Quinic Acid | Phenolic Acid | Acetic acid-induced colitis rats | Nrf2/HO-1 | [61] | |
| Coumaric/Syringic Acid | Phenolic Acid | Colitis model | Nrf2/HO-1 | [62] | |
| Esculetin | Coumarin | Salmonella-infected mice | Nrf2 | [64] | |
| Scopoletin | Coumarin | DSS-induced UC mice | Nrf2/HO-1/NQO1, NF-κB | [65] | |
| Umbelliferone | Coumarin | Acetic acid-induced UC rats | TLR4/NF-κB, SIRT1/PPARγ | [92] | |
| Apigenin | Flavonoid | Rat I/R model | Nrf2/HO-1 | [26] | |
| Naringenin | Flavonoid | DSS-induced UC mice | Nrf2/HO-1/NQO1, NF-κB | [67] | |
| Naringin | Flavonoid | LPS-challenged mice | Nrf2, TLR4/p38 MAPK/NF-κB | [68] | |
| Quercetin | Flavonoid | Radiation-injured mice | Nrf2 | [72] | |
| Diosmin | Flavonoid | DSS-induced colitis mice | Nrf2, NF-κB | [93] | |
| Curcumin (Theracurmin) | Polyphenol | DSS-induced colitis mice | Nrf2/regulatory T cells | [81] | |
| EGCG | Polyphenol | Cisplatin-injured rats | Nrf2/Keap1 | [54] | |
| Carthamin Yellow | Polyphenol | DSS-induced colitis mice | Nrf2/GPX4 | [82] | |
| Moringin | Polyphenol | DSS-induced colitis mice | Nrf2/NF-κB, PI3K/AKT/mTOR | [94] | |
| Geniposide | Polyphenol | DSS-induced colitis mice | Nrf2/ARE, NF-κB | [72] | |
| Grape Seed Anthocyanins | Polyphenol | DSS-induced colitis mice | Nrf2, TLR4/NF-κB | [34] | |
| Loganic Acid | Iridoid | DSS-induced colitis mice | TLR4/NF-κB, SIRT1/Nrf2 | [95] | |
| Cell Line Models | Resveratrol | Stilbenoid | IPEC-J2 cells; H2O2-induced | PI3K/Akt/Nrf2 | [96] |
| Resveratrol | Stilbenoid | DSS-induced IEC barrier dysfunction | Nrf2/HO-1 | [97] | |
| Resveratrol | Stilbenoid | TNF-α-challenged Caco-2 | Nrf2/IL-1β/IL-11 | [98] | |
| Sesamin | Lignan | Caco-2 cells | AKT/ERK → Nrf2 | [94] | |
| Epoxymicheliolide | Sesquiterpene | Macrophages | TAK1-NF-κB, Keap1/Nrf2 | [91] | |
| Apigenin | Flavonoid | H/R cells | Nrf2/HO-1 | [26] | |
| Genistein | Flavonoid | H2O2-induced IPEC-J2 cells | Nrf2; GPR30-Nrf2 | [46,47,73] | |
| Baicalin | Flavonoid | H2O2-induced IPEC-J2 cells | AMPK/Nrf2 | [74,75] | |
| Taxifolin | Flavonoid | Diquat/DON-induced IPEC-J2 | Nrf2 | [40,77] | |
| Astaxanthin | Polyphenol | AFB1-exposed IPEC-J2 cells | Nrf2/HO-1/NQO1/SOD2 | [99] | |
| Gardenin A | Polyphenol | Alcohol-damaged HepG2/Caco2 | AMPK/Nrf2 | [39] | |
| Carthamin Yellow | Polyphenol | LPS-stimulated Caco-2 | Nrf2/GPX4 | [82] | |
| Moringin | Polyphenol | LPS-stimulated Caco-2 | Nrf2/NF-κB | [97] | |
| Geniposide | Polyphenol | LPS-stimulated Caco-2 | Nrf2/ARE, NF-κB | [72] | |
| Domestic Animals | Pterostilbene | Stilbenoid | Livestock models | Nrf2/HO-1, NF-κB | [6,11] |
| Caffeic Acid | Phenolic Acid | Weaned piglets; IPEC-J2 | Nrf2 | [21] | |
| Chlorogenic Acid | Phenolic Acid | Weaned piglets | Nrf2, TLR4/NF-κB | [49] | |
| Tannic Acid | Phenolic Acid | ETEC K88-challenged piglets | p62-Keap1-Nrf2, TLR4-NF-κB | [63] | |
| Morin | Flavonoid | DON-induced damage (livestock) | Keap1/Nrf2 | [24,25] | |
| Quercetin | Flavonoid | DON-challenged piglets | Nrf2 | [70,71] | |
| Rutin | Flavonoid | Weaned piglets | Nrf2/Keap1 | [76] | |
| EUF | Flavonoid | DON-challenged piglets | Nrf2/Keap1 | [50] | |
| Curcumin | Polyphenol | AFB1-exposed broilers | Nrf2/HO-1/NQO-1 | [43] | |
| Hydroxytyrosol | Polyphenol | Diquat-induced pig model | PI3K/Akt-Nrf2, Mitophagy | [83] | |
| Oat Bran Polyphenols | Polyphenol | Livestock applications | ROS/Akt/Nrf2 | [100] |
4.2. Bioactive Nutrients and Microbial Agents
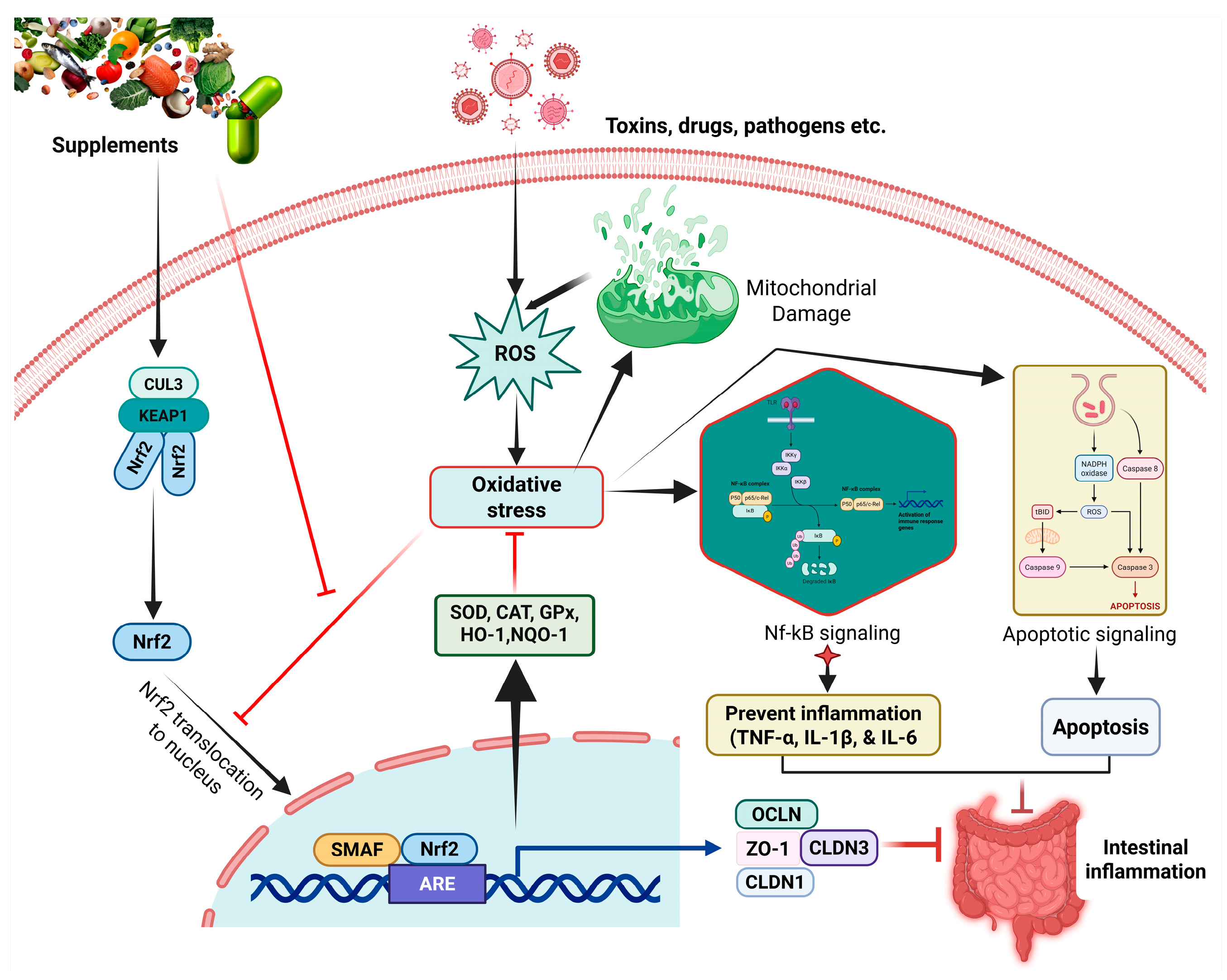
Beyond phenolic compounds, a wide array of bioactive nutrients, microbial agents, and their metabolites also exert protective effects on intestinal health primarily through the modulation of the Nrf2 pathway. These agents, including polysaccharides, peptides, probiotics, and short-chain fatty acids, often function through unique mechanisms involving gut microbiota-mediated biotransformation. Critically, these compounds are particularly amenable to dietary supplementation strategies in both livestock production and human nutrition, facilitating their practical implementation across diverse settings. The evidence for these diverse Nrf2-targeting nutrients and microbial agents is consolidated in Table 2, and their mechanisms are shown in Figure 2.
Figure 2.
Nrf2 signaling pathway as a therapeutic target for intestinal inflammation. The figure depicts the Keap1-Nrf2 regulatory axis, showing how Nrf2 activation leads to transcription of antioxidant response element (ARE)-dependent genes including HO-1, NQO1, and glutathione synthesis enzymes, ultimately providing protection against oxidative stress and inflammation in intestinal epithelial cells.
4.2.1. Polysaccharides
Complex polysaccharides demonstrate Nrf2-activating mechanisms involving gut microbiota-mediated biotransformation and prebiotic effects. For example, brown algae polysaccharides (BAPs) alleviate oxidative stress through Nrf2/ARE activation, increasing nuclear Nrf2 translocation and upregulating HO-1, SOD, and CAT [101]. Epimedium polysaccharide alleviates UC by activating Keap1/Nrf2 while promoting protective autophagy via AMPK/mTOR, with Nrf2-dependent efficacy demonstrated in knockout mice. Efficacy was demonstrated to be Nrf2-dependent using knockout mice, establishing the hierarchy of pathway activation [102]. Similarly, Artemisia annua polysaccharide improves growth performance in E. coli-challenged broilers while enhancing antioxidant capacity through Nrf2 activation and TLR4/MyD88/NF-κB inhibition [103]. Furthermore, Mussel polysaccharide activates Nrf2-Keap1 pathway with HO-1 and antioxidant enzyme upregulation, protecting against cyclophosphamide-induced oxidative injury [104].
Extending beyond direct Nrf2 activation, Lycium barbarum polysaccharides activate Nrf2 signaling while enriching beneficial microbiota (Ruminococcaceae, Lactobacillus, Akkermansia) and enhancing short-chain fatty acid production [105]. Paralleling this integrated approach, polysaccharide suppresses NF-κB and NLRP3 inflammasome while activating Nrf2/HO-1 and mediating anti-ferroptosis effects [106,107]. Additional mechanistically characterized compounds include Melientha longistaminea polysaccharide [108], Floccularia luteovirens polysaccharide [109,110], Artemisia capillaris polysaccharide [111], yeast β-glucan (activating both Nrf2 and AHR) [112], and Camellia oleifera polysaccharide [113] as shown in Table 2. Accordingly, polysaccharides represent multifunctional Nrf2 activators whose combined antioxidant, anti-inflammatory, and prebiotic properties position them as ideal candidates for dietary-based intestinal health interventions across species.
4.2.2. Peptides and Amino Acids
Bioactive peptides provide intestinal protection through Nrf2-mediated antioxidant responses combined with immunomodulation. Peptide-induced Nrf2 activation upregulates antioxidant enzymes, reducing ROS and subsequently inhibiting ROS-sensitive NF-κB [17]. For example, activates Keap1-Nrf2 pathway, with LC-MS identifying four specific bioactive sequences [114]. Complementing these findings, rice protein peptides have been found to ctivate Keap1-Nrf2 signaling while modulating gut microbiota, increasing Firmicutes/Bacteroidetes ratio and promoting beneficial bacteria [115].
In addition to bioactive peptides, individual amino acids also demonstrate Nrf2-activating capacity beyond nutritional roles. For example, proline upregulates Nrf2/HO-1, suppressing NF-κB p65 phosphorylation to alleviate hibernation-induced barrier dysfunction [116]. While, taurine produces dose-dependent improvements in antioxidant status through enhanced Nrf2, HO-1, GPX1, and SOD1 expression, with optimal doses (0.3–0.4%) favorably modulating gut microbiota [117]. In addition, L-theanine has been shown to promote Nrf2 nuclear translocation while decreasing Keap1, with ML385 completely abolishing protection [118]. Extending this mechanistic diversity, glutamine prevents 5-FU-induced mucositis by inhibiting TLR4/NF-κB while activating Nrf2/HO-1 and increasing mTOR, with concurrent microbial diversity enhancement [119]. Furthermore, betaine activates Nrf2/Keap1 while inhibiting TLR4-NF-κB/MAPK, with effects transmissible through maternal supplementation [120], demonstrating transgenerational protective mechanisms. From a practical standpoint, amino acids and bioactive peptides constitute an immediately translatable class of Nrf2 activators, with established clinical and agricultural applications that can be enhanced through mechanistic understanding of their Nrf2-dependent protective actions.
4.2.3. Fatty Acids and Lipids
Dietary lipids demonstrate Nrf2-activating properties contributing to intestinal health benefits. α-Linolenic acid activates NRF2 signaling dose-dependently, with 600 mg/kg identified as optimal [121]. Similarly, Deer oil suppresses NF-κB phosphorylation while upregulating Nrf2/HO-1, with parallel microbiota modulation increasing Odoribacter, Blautia, and short-chain fatty acids [30]. Moreover, Coix seed oil activates Keap1/Nrf2 in kidney while reinforcing intestinal barrier integrity through tight junction upregulation [41]. Expanding this therapeutic repertoire, α-Lipoic acid activates Keap1-Nrf2 while inhibiting both ferroptosis and endoplasmic reticulum stress [122]. Furthermore, icosapent ethyl activates SIRT1/Nrf2/HO-1 and PI3K/Akt signaling while suppressing NF-κB, with benefits abolished by SIRT1 inhibition [123]. Hence, dietary lipids represent an accessible class of Nrf2 activators whose protective intestinal effects complement their established systemic health benefits, supporting integrated dietary strategies for gut-systemic health optimization.
4.2.4. Organic Acids
Simple organic acids demonstrate Nrf2-activating capacity useful in feed additives and functional foods. Glyceryl lactate combined with lactic acid activates Nrf2 while modulating cecal microbiota to increase SCFA-producing bacteria, proving most effective in combination [124]. Building on this synergistic principle, encapsulated benzoic acid outperforms unencapsulated forms in suppressing TLR4/NF-κB and activating NRF2, with NRF2 inhibition abolishing benefits [108]. Complementing these findings, itaconic acid activates Keap1/Nrf2/HO-1 while reversing gut dysbiosis induced by perfluorooctanoic acid (PFOA) exposure, reducing harmful bacteria while promoting Lactobacillus [121]. Notably, organic acids offer practical, cost-effective Nrf2 activation strategies particularly suited to large-scale livestock production, with potential extension to human functional food development.
4.2.5. Probiotics
Probiotic organisms activate Nrf2 through microbial metabolite production, pattern recognition receptor engagement, or direct host cell interaction. Bacteroides thetaiotaomicron increases tryptophan metabolites including indole, enhancing barrier integrity and reducing inflammation via AHR-Nrf2 signaling [22]. Building on this framework, Lactobacillus acidophilus activates p62-Keap1-Nrf2 pathway while increasing Akkermansia and restoring short-chain fatty acids [31]. Similarly, Clostridium dalinum inhibits NF-κB while activating Keap1-Nrf2-ARE, with metabolomics identifying elevated anti-inflammatory metabolites and butanoate metabolism activation [32]. Expanding this mechanistic repertoire, Lacticaseibacillus paracasei CCFM1222 inhibits TLR4/MyD88/NF-κB while activating Nrf2, elevating antioxidant enzymes and shifting microbiota [29]. Moreover, Clostridium butyricum enhances intestinal health by activating Nrf2 and suppressing NF-κB while improving digestive enzyme activity [105], whereas Bacillus amyloliquefaciens mediates protection through Keap1/Nrf2 signaling, increasing Nrf2 while decreasing Keap1 [45]. As such, probiotics exemplify the successful integration of Nrf2-targeting strategies with microbiome modulation, offering established therapeutic platforms for continued development in both veterinary and human intestinal health applications.
4.2.6. Microbial Metabolites
Microbiota-derived metabolites represent an emerging therapeutic class mediating many probiotic and prebiotic effects. Indole-3-lactic acid, produced by Lactiplantibacillus plantarum DPUL-S164 from tryptophan metabolism, strengthens intestinal barrier via coordinated AhR and Nrf2 activation, with both dependent on AhR expression [125]. Paralleling these findings, sodium butyrate alleviates DSS-induced colitis by activating COX-2/Nrf2/HO-1 while suppressing NF-κB/NLRP3 inflammasome and promoting mitophagy via Pink1/Parkin [126]. Furthermore, butyric acid upregulates Nrf2 in vitro, with fecal microbiota transplantation from treated mice replicating therapeutic effects and Nrf2 activation [127], demonstrating that microbiota-derived butyrate is sufficient to activate host Nrf2 with functional consequences. Consequently, microbial metabolites constitute an emerging class of Nrf2 activators that bridge dietary, microbiome, and pharmaceutical intervention strategies, offering precise molecular tools for modulating intestinal oxidative stress responses.

Table 2.
Bioactive nutrients and microbial agents targeting Nrf2 for intestinal health.
Table 2.
Bioactive nutrients and microbial agents targeting Nrf2 for intestinal health.
| Model Category | Compound | Category | Model System | Key Pathways | Ref. |
|---|---|---|---|---|---|
| Rodent Models | Spermidine | Polyamine | Experimental IBD; colitis models | AhR-Nrf2, AhR-STAT3 | [128] |
| Carbocisteine | Mucoregulator | Acetic acid-induced UC rats | Nrf2/HO-1, NF-κB | [129] | |
| Myristicin | Phenylpropanoid | Acetic acid-induced UC rats | ERS, Nrf2/HO-1, NF-κB | [130] | |
| Sulforaphane | Isothiocyanate | DSS-induced UC mice | Nrf2, STAT3 | [131] | |
| MLS Polysaccharide | Polysaccharide | CTX-induced immunosuppression mice | NF-κB, Nrf2 | [108] | |
| FLP1 Polysaccharide | Polysaccharide | Mouse immunosuppression model | MAPK, Nrf2/Keap1 | [109,110] | |
| APS Polysaccharide | Polysaccharide | ANIT-induced cholestasis mice | Nrf2 (via butyric acid) | [111] | |
| Mussel Polysaccharide | Polysaccharide | Cyclophosphamide-injured mice | Nrf2-Keap1/HO-1 | [104] | |
| EPS Polysaccharide | Polysaccharide | DSS-induced UC mice; Nrf2 KO | Keap1/Nrf2, AMPK/mTOR | [102] | |
| LBP Polysaccharide | Polysaccharide | DSS-induced chronic UC mice | Nrf2 | [105] | |
| Wheat Peptide | Peptide | DSS-induced colitis mice | Keap1-Nrf2 | [114] | |
| Glutamine | Amino Acid | 5-FU-induced mucositis mice | Nrf2/HO-1, TLR4/NF-κB | [119] | |
| Deer Oil | Lipid | DSS-induced UC mice | Nrf2/HO-1, NF-κB | [30] | |
| Coix Seed Oil | Lipid | Hyperuricemia mice | Keap1/Nrf2, PI3K/AKT/mTOR | [41] | |
| α-Lipoic Acid | Fatty Acid | DSS-induced UC mice | Keap1-Nrf2 | [122] | |
| Icosapent Ethyl | Fatty Acid | Acetic acid-induced UC rats | SIRT1/Nrf2/HO-1, NF-κB | [123] | |
| TDCA | Bile Acid | Diquat-induced mice | Nrf2 | [132] | |
| B. thetaiotaomicron | Probiotic | Cold-stressed mice | AHR-Nrf2 | [22] | |
| L. acidophilus | Probiotic | Salmonella-infected mice | p62-Keap1-Nrf2 | [31] | |
| L. paracasei CCFM1222 | Probiotic | Murine colitis model | Nrf2, TLR4/MyD88/NF-κB | [29] | |
| L. casei + VIP | Probiotic + Peptide | DSS-induced UC mice | Nrf2, NF-κB | [133] | |
| Sodium Butyrate | Metabolite | DSS-induced murine colitis | Nrf2/HO-1, NF-κB/NLRP3 | [126] | |
| Cell Line Models | Encapsulated Benzoic Acid | Organic Acid | LPS-challenged IPEC-J2 | NRF2, TLR4/NF-κB | [36] |
| Clostridium dalinum | Probiotic | Caco-2 cells | Keap1-Nrf2-ARE, NF-κB | [32] | |
| B. amyloliquefaciens | Probiotic | LPS-challenged Caco-2 | Keap1/Nrf2 | [45] | |
| Indole-3-lactic Acid | Metabolite | LPS-damaged HT-29 cells | AhR-Nrf2 | [125] | |
| α-Lipoic Acid | Fatty Acid | Erastin-induced cells | Keap1-Nrf2 | [122] | |
| Domestic Animals | Selenomethionine | Selenium | DON-induced damage; ISC (piglets) | Keap1/Nrf2 | [134] |
| BAO (Benzoic + EO) | Organic Acid/EO | LPS-challenged weaned piglets | Nrf2, TLR4/NF-κB/MAPK | [127] | |
| BAPs | Polysaccharide | Diquat-challenged piglets | Nrf2/ARE | [101] | |
| AAP Polysaccharide | Polysaccharide | E. coli-challenged broilers | Nrf2, TLR4/MyD88/NF-κB | [103] | |
| Proline | Amino Acid | Turtles’ post-hibernation | Nrf2/HO-1, NF-κB | [116] | |
| Taurine | Amino Acid | Early-weaned piglets | Nrf2/HO-1/GPX1/SOD1 | [117] | |
| L-theanine | Amino Acid | Weaned piglets; IPEC-J2 | Nrf2 | [118] | |
| Betaine | Amino Acid | Sows/offspring piglets | Nrf2/Keap1, TLR4-NF-κB | [120] | |
| α-Linolenic Acid | Fatty Acid | Broilers (42-day) | NRF2/HO-1 | [35] | |
| Itaconic Acid | Organic Acid | perfluorooctanoic acid (PFOA)-exposed laying hens | Keap1/Nrf2/HO-1, NF-κB | [121] | |
| LA/GL | Organic Acid | 28-day piglet study | Nrf2 | [124] | |
| C. butyricum | Probiotic | Broilers | Nrf2, NF-κB | [135] | |
| B. amyloliquefaciens | Probiotic | Broilers | Keap1/Nrf2 | [45] |
4.3. Terpenoids, Alkaloids, Plant Extracts and Traditional Medicines in Intestinal Health
The therapeutic repertoire of Nrf2 activators extends further into structurally complex terpenoids, potent alkaloids, and multi-component plant extracts and traditional formulas. These compounds often exhibit high-affinity interactions with the Keap1-Nrf2 complex and engage in sophisticated multi-pathway regulation. Notably, many of these agents derive from traditional medicine systems with centuries of empirical validation in treating gastrointestinal disorders, providing a strong rationale for their contemporary mechanistic investigation and clinical development. The broad spectrum of these plant-derived therapeutics and their documented efficacy in experimental models of intestinal inflammation are cataloged in Table 3.
4.3.1. Terpenoids
Terpenoids demonstrate Nrf2-activating mechanisms often involving direct protein-target interactions amenable to structural characterization. Cardamonin functions through hierarchical AhR activation that subsequently promotes Nrf2 nuclear translocation and NLRP3 inflammasome inhibition, with both CH223191 (AhR inhibitor) and ML385 (Nrf2 inhibitor) abolishing anti-inflammatory effects [33]. Moreover, efficacy extends to heat stress conditions, activating Nrf2 components (Nrf2, HO-1, NQO1) while modulating gut microbiota [136]. Building upon these hierarchical activation mechanisms, neferine competitively binds Keap1 at Cys-288, promoting Nrf2 nuclear translocation and activating Nrf2/FPN and Nrf2/xCT/GPX4 axes. This dual-axis activation inhibits ferroptosis in severe acute pancreatitis, with effects reversed by ML385 and erastin [27].
Extending beyond single-pathway modulation, α-mangostin activates Nrf2/HO-1 while suppressing NF-κB/NLRP3/caspase-1, reducing oxidative stress and promoting autophagy. Treatment remodeled gut microbiota, increasing Ligilactobacillus and Muribaculum that correlated with elevated propionic and butyric acid [137]. Similarly, sclareol activates Nrf2 while inhibiting NF-κB/MLCK pathway, preserving tight junctions and reducing inflammation and bacterial translocation [138]. Furthermore, limonin demonstrates direct Nrf2/ARE interaction via molecular docking, with ML385 reversing protection against NSAID-induced injury [139]. Complementing these findings, oleanolic acid 28-O-β-D-glucopyranoside (OAG) alleviates UC by inhibiting ferroptosis through Nrf2/x-CT/GPX4. Multi-omics analysis confirmed OAG directly binds and modulates GPX4, NRF2, HO-1, and x-CT [140]. Additional terpenoids include panaxadiol (inhibiting MAPK/NF-κB while activating AMPK/NRF2/NQO1) [141], columbianadin (potentiating Nrf2 while suppressing TLR4/NF-κB) [99,142], and melianodiol (activating Nrf2 while suppressing JAK/STAT and NF-κB) [143]. Together, these data indicate that terpenoids represent a structurally diverse class of Nrf2 activators whose well-defined molecular interactions facilitate rational drug design and clinical translation for intestinal inflammatory disorders.
4.3.2. Alkaloids
Alkaloids demonstrate potent Nrf2-activating capacity with well-characterized molecular mechanisms. Berberine produces marked Nfe2l2 upregulation with enhanced SOD, CAT, GPx, and GR activities in experimental colitis [144]. In ETEC-infected piglets, berberine downregulates TLR4/MyD88/NF-κB while upregulating Nrf2, optimizing gut microbiota by increasing beneficial bacteria [145]. Notably, berberine also regulates gut microbiota-associated tryptophan metabolites through AhR activation, restoring gut barrier function through this microbiota-metabolite-receptor axis [146]. Complementing these findings, sinomenine activates Nrf2/HO-1 while suppressing NF-κB to reduce TNF-α, IL-6, and IL-1β. It modulates cell death processes (necroptosis, pyroptosis) and remodels gut microbiota by correcting Firmicutes and Bacteroidetes imbalances [147]. Similarly, cepharanthine activates AMPK-α1/AKT/GSK-3β signaling, driving NRF2-dependent antioxidant genes (HO-1, NQO-1) while inhibiting MAPK and NF-κB p65. Effects were entirely abrogated in NRF2-knockout mice [148]. Isoquinoline alkaloids from Macleaya cordata co-regulate TLR4/MyD88/NF-κB and Nrf2 pathways, restoring gut microbiota and barrier integrity [149]. Oxyberberine, a microbiota-derived berberine metabolite with superior efficacy, activates Keap1/Nrf2 by promoting Nrf2 nuclear translocation while suppressing NF-κB through IκBα phosphorylation inhibition [150]. Additional alkaloids include dictamnine (activating Nrf2-Gpx4 to inhibit ferroptosis) [151], corynoline (with ML385-abolished protection) [152], and cinnamaldehyde (activating PI3K/Akt/Nrf2 to restore barrier integrity) [47]. Based on these findings, alkaloids constitute potent, clinically validated Nrf2 activators with established safety profiles and regulatory precedent, positioning them as leading candidates for continued therapeutic development in intestinal inflammatory disorders.
4.3.3. Plant Extracts
Complex plant extracts demonstrate therapeutic effects potentially through synergistic multi-target engagement or enhanced bioavailability. For instance, Hericium erinaceus extract activates Nrf2 and upregulates HO-1 while enhancing microbiome diversity, providing gastroprotection against ethanol-induced and acetic acid-induced injury [153]. Similarly, Rosa roxburghii fermentation broths activate Nrf2/HO-1/NQO1 through Nrf2 nuclear translocation, attenuating pulmonary fibrosis while modulating gut microbiota. Correlation analysis linked microbial changes (reduced Proteus, increased Ileibacterium and Dubosiella) to oxidative stress inhibition [154]. In a comparable manner, Glycine tabacina ethanol extract suppresses NF-κB and MAPK/JNK while activating Nrf2, coordinately reducing pro-inflammatory cytokines, matrix metalloproteinases, and oxidative stress [28]. Furthermore, Viola yedoensis Makino inhibits NF-κB/NLRP3 while activating Nrf2/MAPK, reducing mitochondrial damage and apoptosis while modulating microbiota [155]. Correspondingly, Forsythia suspensa extract activates Nrf2-NLRP3 coordination, with Nrf2 silencing abolishing anti-pyroptotic effects. Metabolomic analysis revealed reversal of colitis-associated dysfunction in glutathione metabolism, aminoacyl-tRNA biosynthesis, and linoleic acid metabolism [156]. Additional plant extracts with characterized Nrf2-mediated protection include Aurantii Fructus extract (activating Nrf2/HO-1 while suppressing NF-κB and restoring microbiota) [157], Mesua Assamica extract (suppressing NF-κB/STAT3 while activating HO-1/Nrf2/SIRT1) [158], apple polyphenols (exhibiting Nrf2-dependent barrier protection reduced by ML385) [159], grape seed anthocyanins encapsulated in chitosan-phytic acid gel (inhibiting TLR4/NF-κB while activating Nrf2) [34], and maggot extracts (increasing Nrf2 while preventing Keap1-mediated degradation, abolished by ML385) [160]. Therefore, plant extracts represent pragmatic Nrf2-targeting interventions whose multi-component nature may confer therapeutic advantages through synergistic pathway modulation, supporting their development as functional foods and nutraceuticals for intestinal health.
4.3.4. Traditional Herbal Formulas
Classical herbal formulations represent empirically validated multi-component therapeutics converging on Nrf2 modulation. Lizhong decoction (LZD) alleviates UC by inhibiting ferroptosis through Nrf2 activation, upregulating SLC7A11 and GPX4 while suppressing oxidative stress and iron overload [161]. Building upon these observations, licorice and its principal active component glycyrrhetinic acid activate Nrf2/PINK1 to regulate mitophagy, reducing inflammation and oxidative stress while repairing mitochondrial damage and enhancing barrier proteins. Gene silencing abolished protective effects [162,163]. Similarly, Gegen Qinlian decoction (GQ) activates Nrf2/ARE to combat oxidative stress, with puerarin, berberine, and liquiritin identified as primary drivers [164].
Certain formulations achieve effects through microbiota-mediated mechanisms converging on Nrf2 signaling. In contrast to the direct pathway activation described above, Yinchen Linggui Zhugan decoction modifies gut microbiota to enrich SCFA-producing bacteria and elevate butyric acid, which subsequently activates hepatic SIRT1/Nrf2 to upregulate HO-1 and NQO1 [136]. Ginseng root extract (GRE) suppresses MAPK/NF-κB while activating p62-Nrf2-Keap1 axis to enhance antioxidant defense and mitigate mitochondrial dysfunction. GRE also induces cytoprotective autophagy via Akt-mTOR pathway [107]. Ultimately, traditional herbal formulas represent a rich source of validated Nrf2-targeting therapeutics whose integration of empirical clinical evidence with contemporary molecular mechanistic understanding offers a robust foundation for evidence-based application in modern intestinal disease management.

Table 3.
Terpenoids, alkaloids, plant extracts and traditional medicines.
Table 3.
Terpenoids, alkaloids, plant extracts and traditional medicines.
| Model Category | Compound | Category | Model System | Key Pathways | Ref. |
|---|---|---|---|---|---|
| Rodent Models | Ruscogenin | Saponin | TNBS-induced Crohn’s-like colitis | Nrf2/NQO1/HO-1 | [165] |
| Bryostatin-1 | Macrolide | Intestinal I/R injury mice | Nrf2/HO-1 | [166] | |
| Curculigoside | Glycoside | Murine UC models | Keap1/Nrf2 → autophagy | [167] | |
| Cannabidiol | Cannabinoid | DSS-induced colitis | Nrf2/HO-1, NF-κB, TGF-β | [168] | |
| Mushroom Extract (AP + FV) | Extract | DSS-induced UC with liver injury | TLR4/NF-κB, Keap1/Nrf2 | [169] | |
| Andrographolide | Diterpenoid | DSS-induced mice | Nrf2/HO-1 | [170] | |
| Coix Seed | Functional Food | DSS-induced UC mice | Src/JNK MAPK, Nrf2/PPARγ | [171] | |
| Neferine | Terpenoid | Severe acute pancreatitis mice | Nrf2/FPN, Nrf2/xCT/GPX4 | [27] | |
| α-Mangostin | Terpenoid | Alcohol-induced gastric ulcers rats | Nrf2/HO-1, NF-κB/NLRP3 | [172] | |
| Sclareol | Terpenoid | TNBS-induced CD mice | Nrf2, NF-κB/MLCK | [138] | |
| Limonin | Terpenoid | Indomethacin-induced injury rats | Nrf2/ARE | [139] | |
| Zingerone | Terpenoid | HFD-induced duodenal injury rats | Nrf2, NF-κB | [173] | |
| Melatonin | Terpenoid-like | BPA-exposed colon | SIRT1/PGC-1α/Nrf2 | [174] | |
| Ginsenoside Rg1 | Terpenoid | DSS-induced UC mice | Nrf-2/HO-1/NF-κB | [151] | |
| Berberine | Alkaloid | ETEC-infected piglets | Nrf2, TLR4/MyD88/NF-κB | [145] | |
| Oxyberberine | Alkaloid | TNBS-induced colitis rats | Keap1/Nrf2, NF-κB | [150] | |
| Sinomenine | Alkaloid | DSS-induced UC rats | Nrf2/HO-1, NF-κB | [147] | |
| Cepharanthine | Alkaloid | Experimental colitis; Nrf2-KO | AMPK/AKT/GSK-3β → NRF2 | [148] | |
| Columbianadin | Alkaloid | DSS-induced UC rats | Nrf2, TLR4/NF-κB | [142] | |
| Hernandezine | Alkaloid | DSS-induced colitis | AMPK/NRF2 | [175] | |
| Oleracein E | Alkaloid | TNBS-induced rats | Nrf2/HO-1 | [40] | |
| GTE | Plant Extract | DSS colitis mice | Nrf2, NF-κB, MAPK/JNK | [28] | |
| Hericium erinaceus Extract | Plant Extract | Ethanol/acetic acid-injured rats | Nrf2/HO-1 | [153] | |
| RRFBs | Plant Extract | Bleomycin-induced fibrosis mice | Nrf2/HO-1/NQO1 | [154] | |
| SP Extract | Plant Extract | DSS-induced UC mice | Nrf2/Keap1/HO-1/NQO1 | [176] | |
| PHE | Plant Extract | DSS-induced colitis | KEAP1 → Nrf2 | [177] | |
| Lizhong Decoction | Herbal Formula | DSS-induced UC mice | Nrf2/SLC7A11/GPX4 | [161] | |
| Licorice | Herbal Formula | DSS-induced UC mice | Nrf2/PINK1 | [162,163] | |
| Gegen Qinlian Decoction | Herbal Formula | DSS-induced UC rats | Nrf2/ARE | [164] | |
| OAG | Triterpene | TNBS-induced UC rats | Nrf2/x-CT/GPX4 | [140] | |
| Lithospermic Acid | Plant Compound | Murine colitis | Nrf2 + NF-κB inhibition | [178] | |
| Cell Line Models | Ruscogenin | Saponin | LPS-stimulated organoids | Nrf2/NQO1/HO-1 | [165] |
| Bryostatin-1 | Macrolide | Cellular models | Nrf2/HO-1 | [166] | |
| Curculigoside | Glycoside | Organoid, cellular UC models | Keap1/Nrf2 → autophagy | [167] | |
| Andrographolide | Diterpenoid | LPS-induced cells | Nrf2/HO-1 | [170] | |
| α-Mangostin | Terpenoid | GSE-1/RAW264.7 cells | Nrf2/HO-1, NF-κB/NLRP3 | [172] | |
| Sclareol | Terpenoid | TNF-α-induced organoids | Nrf2, NF-κB/MLCK | [138] | |
| Cinnamaldehyde | Terpenoid | P. gingivalis-infected IEC-6 | PI3K/Akt/Nrf2 | [47] | |
| Oleracein E | Alkaloid | LPS-stimulated cells | Nrf2/HO-1 | [40] | |
| Lizhong Decoction | Herbal Formula | RSL3-induced Caco-2 | Nrf2/SLC7A11/GPX4 | [161] | |
| Licorice | Herbal Formula | LPS-induced Caco2 | Nrf2/PINK1 | [163] | |
| Gegen Qinlian Decoction | Herbal Formula | TNF-α-stimulated Caco-2 | Nrf2/ARE | [164] | |
| Lithospermic Acid | Plant Compound | NCM460 cells | Nrf2 + NF-κB inhibition | [178] | |
| Domestic Animals | Cardamonin | Terpenoid | LPS-challenged piglets; Heat-stressed chickens | AhR/Nrf2/NLRP3 | [33] |
| Isoquinoline Alkaloids | Alkaloid | LPS-challenged broilers | Nrf2, TLR4/MyD88/NF-κB | [149] | |
| Viola yedoensis Makino | Plant Extract | LPS-challenged broilers | Nrf2/MAPK, NF-κB/NLRP3 | [155] |
4.4. Therapeutic Potential and Translational Outlook
The extensive body of evidence reviewed in this section establishes the Nrf2 signaling pathway as a validated therapeutic target for intestinal inflammatory disorders across multiple compound classes and experimental platforms. Across structurally diverse compound classes—ranging from phenolics and terpenoids to microbial metabolites—convergent protective mechanisms have been consistently observed, including antioxidant enzyme induction, NF-κB suppression, ferroptosis inhibition, and gut microbiota modulation. This mechanistic convergence underscores the fundamental role of Nrf2 signaling in maintaining intestinal homeostasis and highlights its therapeutic relevance across multiple disease contexts. The demonstrated efficacy in rodent disease models, livestock production systems, and human intestinal cell lines collectively establishes a robust preclinical foundation for clinical development, while concurrent agricultural applications present viable alternatives to antimicrobial growth promoters with attendant benefits for animal welfare and food safety.
Among the phenolic compounds, resveratrol and phenolic acids occupy a distinctive translational niche owing to their abundance in common dietary sources and well-established safety profiles. Their efficacy in weaned piglet models, which closely approximate human intestinal physiology, supports dual applications as livestock feed additives and nutraceutical interventions for inflammatory bowel disease management. Flavonoids, particularly quercetin and curcumin, exhibit exceptional translational potential through extensive validation across DSS-induced murine colitis, deoxynivalenol-challenged livestock, and human intestinal cell lines, with both compounds having advanced to human clinical trials as adjunctive therapies. Curcumin and epigallocatechin gallate represent the most clinically mature Nrf2 activators, demonstrating efficacy in multiple human ulcerative colitis trials while advanced delivery systems, including nanoparticles and liposomes, have successfully addressed historical bioavailability limitations. The emerging recognition of anti-ferroptotic mechanisms further expands their therapeutic scope to encompass chemotherapy-induced intestinal mucositis, thereby addressing a significant unmet clinical need.
Endogenous and dietary metabolites demonstrate similarly robust translational applicability. Amino acids and bioactive peptides, including glutamine, taurine, and betaine, offer immediate clinical relevance given their established utilization in clinical nutrition and animal feed formulations; the elucidation of Nrf2-mediated mechanisms now provides a rational basis for optimizing dosing regimens and developing targeted combination therapies. Lipid-based activators, particularly omega-3 polyunsaturated fatty acids, benefit from well-characterized dietary incorporation pathways and integrate intestinal barrier protection with established cardiovascular and metabolic benefits. Polysaccharides present exceptional advantages through their prebiotic properties and compatibility with existing dietary supplementation infrastructure, whereas organic acids currently employed as antimicrobial feed preservatives now possess mechanistic justification for their observed gut health benefits through Nrf2 pathway activation.
Microbiome-based interventions constitute particularly promising therapeutic strategies warranting focused development. Probiotics represent the most clinically advanced approach, with Nrf2 activation identified as a conserved protective mechanism across taxonomically diverse species, thereby providing a rational framework for strain selection and combination therapy design. The emergence of next-generation probiotics, including Akkermansia muciniphila and specific Clostridium species, opens additional therapeutic avenues. Microbial metabolites, particularly short-chain fatty acids and tryptophan derivatives, function as the mechanistic interface between dietary intake, microbiome composition, and host intestinal health. Butyrate supplementation is already employed clinically for ulcerative colitis, while postbiotic formulations comprising defined metabolite mixtures are under active investigation.
Plant-derived terpenoids and alkaloids benefit from detailed structural characterization that enables systematic structure-activity relationship optimization. Ginsenosides are progressing through Western clinical trials following extensive historical application in traditional Chinese medicine, establishing precedent for related terpenoid compounds. Berberine represents one of the most clinically advanced natural Nrf2 activators, with demonstrated efficacy in human metabolic and gastrointestinal disorders and regulatory approval in several countries for diarrheal diseases. Its established efficacy in enterotoxigenic Escherichia coli-infected piglet models further supports therapeutic development for both agricultural applications and pediatric gastroenteritis. Traditional herbal formulations offer the distinctive advantage of centuries of empirical clinical validation, and mechanistic elucidation of their Nrf2-dependent actions facilitates international regulatory acceptance while enabling the standardization essential for contemporary pharmaceutical development.
Future research priorities should include standardization of optimal dosing regimens across species, development of advanced delivery systems to overcome bioavailability constraints, and identification of patient subpopulations most likely to derive benefit from Nrf2-targeted interventions. Integration of omics-based biomarkers for monitoring pathway activation status will facilitate personalized therapeutic approaches and accelerate clinical translation of these promising natural therapeutics.
5. Conclusions
This review demonstrates that the Nrf2/Keap1 signaling pathway serves as a central therapeutic target for intestinal inflammatory disorders. Diverse agents—including polyphenols (resveratrol, quercetin, curcumin), terpenoids, alkaloids, polysaccharides, and probiotics—activate NRF2 through distinct mechanisms: direct Keap1 cysteine modification, upstream kinase signaling, and gut microbiota-derived metabolites. These compounds exhibit coordinated dual-pathway regulation, simultaneously enhancing Nrf2-mediated antioxidant defenses while suppressing NF-κB-driven inflammation. Beyond classical antioxidant induction, Nrf2 activation promotes multiple protective mechanisms including ferroptosis inhibition, tight junction reinforcement, and inflammasome suppression. The gut microbiota critically modulates therapeutic efficacy through metabolite production and compound biotransformation. These findings establish Nrf2 pathway modulation as a promising multi-targeted strategy for managing IBD in both clinical and livestock settings. However, several knowledge gaps and challenges remain. First, most studies rely on rodent models, and translational validation in larger animals and human clinical trials is needed. Second, the optimal dosing regimens, delivery systems, and long-term safety profiles of Nrf2 activators require further investigation. Third, the interplay between Nrf2 activation and gut microbiota composition warrants mechanistic studies. Future research should focus on developing species-specific formulations, investigating synergistic effects of combined Nrf2 activators, and conducting comparative efficacy studies across different livestock species.
Author Contributions
M.Z.K., Q.M. and C.W.: Conceptualization, writing—original draft; M.Z.K., S.L., Y.L., A.U., M.A., F.M.A., K.F.A., K.J.A., C.W. and Q.M.: writing—review and editing, literature search, proofreading, data curation, software, M.Z.K., Q.M. and C.W.: supervision, validation, resources and funding. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by National Key R&D Program of China (grant numbers 2023YFD1302004), Livestock and Poultry Breeding Industry Project of the Ministry of Agriculture and Rural Affairs (grant number 19211162), the Horizontal Scientific Research Project of Liaocheng University (K25LD167), the Liaocheng University scientific research fund (318052339), the Shandong Province Agricultural Major Technology Collaborative Promotion Plan (SDNYXTTG-2024-13), and the Deanship of Research and Graduate Studies at King Khalid University for funding this work through Large Research Project under grant number RGP.2/731/46, the Liaocheng Municipal Bureau of Science and Technology, High-talented Foreign Expert Introduction Program (GDWZ202401).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Patel, R.; Mohammed, Y.S.; Akula, S.B.; Aramin, M.A.; Naradasu, P.R.; Patel, S.; Desai, H.D. Inflammatory bowel disease in youth under 20 from 1990–2019: A global perspective on regional and national variations from the gbd 2019 analysis. Inflamm. Bowel Dis. 2024, 30, S71–S72. [Google Scholar] [CrossRef]
- Gorospe, J.; Windsor, J.; Hracs, L.; Coward, S.; Buie, M.; Quan, J.; Caplan, L.; Markovinovic, A.; Cummings, M.; Goddard, Q.; et al. Trends in inflammatory bowel disease incidence and prevalence across epidemiologic stages: A global systematic review with meta-analysis. Gastroenterology 2024, 166, S56–S57. [Google Scholar] [CrossRef]
- Wang, S.; Dong, Z.; Wan, X. Global, regional, and national burden of inflammatory bowel disease and its associated anemia, 1990 to 2019 and predictions to 2050: An analysis of the global burden of disease study 2019. Autoimmun. Rev. 2024, 23, 103498. [Google Scholar] [CrossRef]
- Muro, P.; Zhang, L.; Li, S.; Zhao, Z.; Jin, T.; Mao, F.; Mao, Z. The emerging role of oxidative stress in inflammatory bowel disease. Front. Endocrinol. 2024, 15, 1390351. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Shen, L.; Yu, X.; Zhang, L.; Xu, K.; Xia, Y.; Zha, L.; Wu, J.; Luo, H. The role of Nrf2 in the pathogenesis and treatment of ulcerative colitis. Front. Immunol. 2023, 14, 1200111. [Google Scholar] [CrossRef]
- Yuan, L.; Wang, Y.; Li, N.; Yang, X.; Sun, X.; Tian, H.; Zhang, Y. Mechanism of action and therapeutic implications of Nrf2/HO-1 in inflammatory bowel disease. Antioxidants 2024, 13, 1012. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.-T.; Liu, Z.-d.; Wang, P.; Zeng, C.-Y.; Chen, Y.-X. Lipopolysaccharide-regulated RNF31/NRF2 axis in colonic epithelial cells mediates homeostasis of the intestinal barrier in ulcerative colitis. Cell Signal. 2024, 124, 111480. [Google Scholar] [CrossRef]
- Yang, Q.-H.; Zhang, C.-N. Comparative study on the pathogenesis of Crohn’s disease and ulcerative colitis. World J. Gastroenterol. 2025, 31, 106406. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Yang, Y.; Li, Y.; Zhang, Q.; Zhang, W.; Jia, H.; Han, Z.; Wang, J. Th17/Treg imbalance in inflammatory bowel disease: Immunological mechanisms and microbiota-driven regulation. Front. Immunol. 2025, 16, 1651063. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Ma, Q.; Li, Y.; Wei, L.; Zhang, Z.; Khan, A.; Khan, M.Z.; Wang, C. Butyrate supplementation improves intestinal health and growth performance in livestock: A review. Biomolecules 2025, 15, 85. [Google Scholar] [CrossRef] [PubMed]
- Meng, T.; Wen, Z.; Cheng, X.; Li, C.; Zhang, P.; Xiao, D.; Xu, Y. Unlocking Gut Health: The Potent Role of Stilbenoids in Intestinal Homeostasis. Animals 2025, 15, 417. [Google Scholar] [CrossRef]
- Wang, P.; Li, T.; Niu, C.; Sun, S.; Liu, D. ROS-activated MAPK/ERK pathway regulates crosstalk between Nrf2 and Hif-1α to promote IL-17D expression protecting the intestinal epithelial barrier under hyperoxia. Int. Immunopharmacol. 2023, 116, 109763. [Google Scholar] [CrossRef]
- Khan, M.Z.; Li, L.; Zhan, Y.; Binjiang, H.; Liu, X.; Kou, X.; Khan, A.; Qadeer, A.; Ullah, Q.; Alzahrani, K.J.; et al. Targeting Nrf2/KEAP1 signaling pathway using bioactive compounds to combat mastitis. Front. Immunol. 2025, 16, 1425901. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.Z.; Khan, A.; Chen, W.; Chai, W.; Wang, C. Advancements in genetic biomarkers and exogenous antioxidant supplementation for safeguarding mammalian cells against heat-induced oxidative stress and apoptosis. Antioxidants 2024, 13, 258. [Google Scholar] [CrossRef] [PubMed]
- Thiruvengadam, M.; Venkidasamy, B.; Subramanian, U.; Samynathan, R.; Ali Shariati, M.; Rebezov, M.; Girish, S.; Thangavel, S.; Dhanapal, A.R.; Fedoseeva, N.; et al. Bioactive compounds in oxidative stress-mediated diseases: Targeting the NRF2/ARE signaling pathway and epigenetic regulation. Antioxidants 2021, 10, 1859. [Google Scholar] [CrossRef] [PubMed]
- Di Stasi, L.C. Natural coumarin derivatives activating Nrf2 signaling pathway as lead compounds for the design and synthesis of intestinal anti-inflammatory drugs. Pharmaceuticals 2023, 16, 511. [Google Scholar] [CrossRef]
- Chu, Z.; Zhu, L.; Zhou, Y.; Yang, F.; Hu, Z.; Luo, Y.; Li, W.; Luo, F. Targeting Nrf2 by bioactive peptides alleviate inflammation: Expanding the role of gut microbiota and metabolites. Crit. Rev. Food Sci. Nutr. 2025, 65, 3314–3333. [Google Scholar] [CrossRef]
- Khan, M.Z.; Khan, A.; Huang, B.; Wei, R.; Kou, X.; Wang, X.; Chen, W.; Li, L.; Zahoor, M.; Wang, C. Bioactive compounds protect mammalian reproductive cells from xenobiotics and heat stress-induced oxidative distress via Nrf2 signaling activation: A narrative review. Antioxidants 2024, 13, 597. [Google Scholar] [CrossRef]
- Khan, M.Z.; Chen, W.; Liu, X.; Kou, X.; Khan, A.; Khan, R.U.; Zahoor, M.; Wang, C. An Overview of Bioactive Compounds’ Role in Modulating the Nrf2/Keap1/NF-κB Pathway to Alleviate Lipopolysaccharide-Induced Endometritis. Int. J. Mol. Sci. 2024, 25, 10319. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, X.; Podio, N.S.; Wang, X.; Xu, S.; Jiang, S.; Wei, X.; Han, Y.; Cai, Y.; Chen, X.; et al. Research progress on the regulation of oxidative stress by phenolics: The role of gut microbiota and Nrf2 signaling pathway. J. Sci. Food Agric. 2024, 104, 1861–1873. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Qiao, T.; Huang, Z.; Jia, G.; Zhao, H.; Chen, X. Caffeic acid improves intestinal barrier function integrity through activation of Nrf2 signaling pathway in weaned piglets and H2O2 induced IPEC-J2 cells. J. Nutr. Biochem. 2025, 143, 109952. [Google Scholar] [CrossRef]
- Luo, Y.; Lan, C.; Ren, W.; Wu, A.; Yu, B.; He, J.; Chen, D. Bacteroides thetaiotaomicron: A symbiotic ally against diarrhea along with modulation of gut microbial ecological networks via tryptophan metabolism and AHR-Nrf2 signaling. J. Adv. Res. 2025. [Google Scholar] [CrossRef] [PubMed]
- Geertsema, S.; Bourgonje, A.R.; Fagundes, R.R.; Gacesa, R.; Weersma, R.K.; van Goor, H.; Mann, G.E.; Dijkstra, G.; Faber, K.N. The NRF2/Keap1 pathway as a therapeutic target in inflammatory bowel disease. Trends Mol. Med. 2023, 29, 830–842. [Google Scholar] [CrossRef] [PubMed]
- Zan, G.; He, H.; Wang, X.; Zhou, J.; Wang, X.; Yan, H. Morin reactivates Nrf2 by targeting inhibition of keap1 to alleviate deoxynivalenol-induced intestinal oxidative damage. Int. J. Mol. Sci. 2025, 26, 1086. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Liu, J.; Liu, J.; Fatima, M.; Yang, L.; Qin, Y.; Li, W.; Sun, Z.; Yang, B. Exercise antagonizes cadmium-caused liver and intestinal injury in mice via Nrf2 and TLR2/NF-κB signalling pathway. Ecotoxicol. Environ. Saf. 2025, 294, 118100. [Google Scholar] [CrossRef]
- Xu, B.; Zhuang, Y.; Zhang, Y.; Liu, S.; Fan, R.; Jiang, W. Apigenin Alleviates Intestinal Ischemia/Reperfusion Injury via Upregulating Nrf2-Mediated Tight Junction Integrity. Mol. Nutr. Food Res. 2025, 69, e70043. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, K.; Jiang, W.; Ding, P.; Wang, W.; Zhao, K.; Chen, C. Neferine Ameliorates Severe Acute Pancreatitis-Associated Intestinal Injury by Promoting NRF2-mediated Ferroptosis. Int. J. Biol. Sci. 2025, 21, 3247. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Miao, Z.; Chen, J.; Tan, L.; Zhao, Y.; Cui, X.; Zhong, J.; Zhong, R.; Yue, W.; Qiu, B.; et al. Glycine tabacina extract alleviates inflammatory bowel disease via NF-κB, JNK and Nrf2 signaling pathways. J. Ethnopharmacol. 2025, 347, 119744. [Google Scholar]
- Guo, S.; Chu, H.; Peng, B.; Niu, J.; Yang, X.; Guo, Y.; Wang, Z.; Zhang, W. Effects of Lactic Acid and Glyceryl Lactate on Growth Performance, Antioxidant Capacity, and Intestinal Health of Piglets. Antioxidants 2025, 14, 391. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Han, L.; Zong, Y.; Feng, R.; Chen, W.; Geng, J.; Li, J.; Zhao, Y.; Wang, Y.; He, Z.; et al. Deer oil improves ulcerative colitis induced by DSS in mice by regulating the intestinal microbiota and SCFAs metabolism and modulating NF-κB and Nrf2 signaling pathways. J. Sci. Food Agric. 2025, 105, 382–393. [Google Scholar] [CrossRef]
- Li, H.; Ma, X.; Shang, Z.; Liu, X.; Qiao, J. Lactobacillus acidophilus alleviate Salmonella enterica Serovar Typhimurium-induced murine inflammatory/oxidative responses via the p62-Keap1-Nrf2 signaling pathway and cecal microbiota. Front. Microbiol. 2025, 15, 1483705. [Google Scholar] [CrossRef]
- Li, Z.-Q.; Li, R.; Deng, W.; An, R.; Ma, P.; Li, J.; She, R.; Yang, X.-Y.; Yang, X.; Xiao, W. Clostridium dalinum: A novel lactic acid bacterium with excellent antioxidant and anti-inflammatory functions through NF-κB, Keap1-Nrf2-ARE signaling pathways. Probiotics Antimicrob. Proteins 2025, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Fang, Y.; Zhang, Y.; Deng, T.; Ji, M.; Yang, W.; Shi, L.; Xun, W. Cardamonin alleviates intestinal inflammation in LPS-induced weaned piglets through the AhR/Nrf2/NLRP3 signaling pathway. Int. Immunopharmacol. 2025, 162, 115144. [Google Scholar] [CrossRef] [PubMed]
- Niu, B.; Gao, W.; Li, F.; Pei, Z.; Wang, H.; Tian, F.; Zhao, J.; Lu, W. Enhancing colonic health with encapsulated grape seed anthocyanins: Oral capsule for Colon-targeted delivery. Food Chem. 2025, 469, 142544. [Google Scholar] [CrossRef] [PubMed]
- Kang, A.; Ni, J.; Cheng, X.; Wu, S.; Liu, Y.; Ma, W.; Wang, D. Influence of α-Linolenic Acid on the Intestinal Barrier Integrity and Intestinal Antioxidant Status in Broilers. Food Sci. Nutr. 2025, 13, e70271. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, T.; He, Q.; Abouelfetouh, M.M.; Fu, Q.; Yao, S.; Ju, S.; Zhou, Z.; Li, Q. Encapsulated benzoic acid alleviated LPS-induced intestinal damage via NRF2 activation. Food Biosci. 2025, 68, 106704. [Google Scholar] [CrossRef]
- Wang, H.; Wang, J.; Yang, G.; Li, Y.; Chen, W.; Yu, J.; Han, X. Human umbilical cord mesenchymal stem cell-derived exosomes mitigate acute radiation-induced intestinal oxidative damage via the Nrf2/HO-1/NQO1 signaling pathway. PLoS ONE 2025, 20, e0324238. [Google Scholar] [CrossRef]
- Li, Y.; Ma, Q.; Li, M.; Liu, W.; Liu, Y.; Wang, M.; Wang, C.; Khan, M.Z. Non-bovine milk as functional foods with focus on their antioxidant and anti-inflammatory bioactivities. Antioxidants 2025, 14, 801. [Google Scholar] [CrossRef]
- Chadha, P.; Aghara, H.; Johnson, D.; Sharma, D.; Odedara, M.; Patel, M.; Kumar, H.; Thiruvenkatam, V.; Mandal, P. Gardenin A alleviates alcohol-induced oxidative stress and inflammation in HepG2 and Caco2 cells via AMPK/Nrf2 pathway. Bioorganic Chem. 2025, 161, 108543. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Li, Y.; Zhang, Y.; Zhu, M. Alleviation effect of taxifolin on diquat-induced damage to porcine intestinal epithelial cells. Biochem. Biophys. Res. Commun. 2025, 748, 151318. [Google Scholar] [CrossRef]
- Wu, G.; Wang, X.; Dong, H.; Yu, J.; Li, T.; Wang, X. Coix Seed Oil Alleviates Hyperuricemia in Mice by Ameliorating Oxidative Stress and Intestinal Microbial Composition. Nutrients 2025, 17, 1679. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.Z.; Li, L.; Wang, T.; Liu, X.; Chen, W.; Ma, Q.; Zahoor, M.; Wang, C. Bioactive compounds and probiotics mitigate mastitis by targeting NF-κB signaling pathway. Biomolecules 2024, 14, 1011. [Google Scholar] [CrossRef]
- Zhang, J.; Sun, X.; Chai, X.; Jiao, Y.; Sun, J.; Wang, S.; Yu, H.; Feng, X. Curcumin mitigates oxidative damage in broiler liver and ileum caused by aflatoxin B1-contaminated feed through Nrf2 signaling pathway. Animals 2024, 14, 409. [Google Scholar] [CrossRef]
- Ma, H.; Mueed, A.; Liu, D.; Ali, A.; Wang, T.; Ibrahim, M.; Su, L.; Wang, Q. Polysaccharides of Floccularia luteovirens regulate intestinal immune response, and oxidative stress activity through MAPK/Nrf2/Keap1 signaling pathway in immunosuppressive mice. Int. J. Biol. Macromol. 2024, 277, 134140. [Google Scholar] [CrossRef]
- Chen, X.; Zheng, A.; Li, S.; Wang, Z.; Chen, Z.; Chen, J.; Zou, Z.; Liang, H.; Liu, G. Bacillus amyloliquefaciens regulates the Keap1/Nrf2 signaling pathway to improve the intestinal (Caco-2 cells and chicken jejunum) oxidative stress response induced by lipopolysaccharide (LPS). Antioxidants 2024, 13, 1550. [Google Scholar] [CrossRef]
- Li, Y.; Cai, L.; Bi, Q.; Sun, W.; Pi, Y.; Jiang, X.; Li, X. Genistein Alleviates Intestinal Oxidative Stress by Activating the Nrf2 Signaling Pathway in IPEC-J2 Cells. Vet. Sci. 2024, 11, 154. [Google Scholar] [CrossRef] [PubMed]
- Sampath, C.; Chukkapalli, S.S.; Raju, A.V.; Alluri, L.S.C.; Srisai, D.; Gangula, P.R. Cinnamaldehyde protects against P. gingivalis induced intestinal epithelial barrier dysfunction in IEC-6 cells via the PI3K/Akt-mediated NO/Nrf2 signaling pathway. Int. J. Mol. Sci. 2024, 25, 4734. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Yan, C.; Gu, J.; Yuan, Y.; Zou, H.; Liu, Z.; Bian, J. Resveratrol alleviates zearalenone-induced intestinal dysfunction in mice through the NF-κB/Nrf2/HO-1 signalling pathway. Foods 2024, 13, 1217. [Google Scholar] [CrossRef]
- Zhang, B.; Tian, M.; Wu, J.; Qiu, Y.; Xu, X.; Tian, C.; Hou, J.; Wang, L.; Gao, K.; Yang, X.; et al. Chlorogenic Acid Enhances the Intestinal Health of Weaned Piglets by Inhibiting the TLR4/NF-κB Pathway and Activating the Nrf2 Pathway. Int. J. Mol. Sci. 2024, 25, 9954. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Tan, B.; Jiang, Q.; Chen, F.; Liu, K.; Liao, P. Eucommia ulmoides flavonoids alleviate intestinal oxidative stress damage in weaned piglets by regulating the Nrf2/Keap1 signaling pathway. Ecotoxicol. Environ. Saf. 2024, 288, 117373. [Google Scholar] [CrossRef]
- Yang, Y.; Pang, F.; Zhou, M.; Guo, X.; Yang, Y.; Qiu, W.; Liao, C.; Chen, Y.; Tang, C. Electroacupuncture reduces inflammatory bowel disease in obese mice by activating the Nrf2/HO-1 signaling pathways and repairing the intestinal barrier. Diabetes Metab. Syndr. Obes. 2024, 17, 435–452. [Google Scholar] [CrossRef]
- Yu, X.; Wang, Y.; Xu, Y.; Li, X.; Zhang, J.; Su, Y.; Guo, L. Resveratrol attenuates intestinal epithelial barrier dysfunction via Nrf2/HO-1 pathway in dextran sulfate sodium-induced Caco-2 cells. Immun. Inflamm. Dis. 2024, 12, e1193. [Google Scholar] [CrossRef] [PubMed]
- Piechota-Polanczyk, A.; Mariwani, Z.; Fichna, J.; Polanczyk, A.; Jozkowicz, A. Chemical inhibition of NRF2 transcriptional activity influences colon function and oestrogen receptor expression in mice at different ages. Int. J. Mol. Sci. 2024, 25, 13647. [Google Scholar] [CrossRef] [PubMed]
- Xu, E.; Sun, Y.; Yu, Z.; Zheng, J. Epigallocatechin Gallate Alleviates Cisplatin Induced Intestinal Injury in Rats via Inhibiting NRF2/Keap1 Signaling Pathway and Regulating Gut Microbiota and Metabolites. Mol. Nutr. Food Res. 2025, 69, e202400784. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.-C.; Jin, C.-L.; Hu, T.-C.; Zhou, J.-Y.; Wang, X.-F.; Wang, X.-Q.; Kong, X.-F.; Yan, H.-C. Early Weaning Inhibits Intestinal Stem Cell Expansion to Disrupt the Intestinal Integrity of Duroc Piglets via Regulating the Keap1/Nrf2 Signaling. Antioxidants 2024, 13, 1188. [Google Scholar] [CrossRef]
- Xu, Z.; Sun, X.; Ding, B.; Zi, M.; Ma, Y. Resveratrol attenuated high intensity exercise training-induced inflammation and ferroptosis via Nrf2/FTH1/GPX4 pathway in intestine of mice. Turk. J. Med. Sci. 2023, 53, 446–454. [Google Scholar] [CrossRef]
- Leng, P.; Wang, Y.; Xie, M. Ellagic acid and gut microbiota: Interactions, and implications for health. Food Sci. Nutr. 2025, 13, e70133. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Cheng, Z.; Zhang, Y.; Liu, T.; Wan, Z.; Xia, C.; Zhou, B.; Shan, C.; Song, D.; Miao, F. Ellagic acid alleviates DSS–induced ulcerative colitis by inhibiting ROS/NLRP3 pathway activation and modulating gut microbiota in mice. Eur. J. Nutr. 2025, 64, 64. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Huang, R.; Wang, N.; Deng, Y.; Tan, B.; Yin, Y.; Qi, M.; Wang, J. Ellagic acid alleviates oxidative stress by mediating Nrf2 signaling pathways and protects against paraquat-induced intestinal injury in piglets. Antioxidants 2022, 11, 252. [Google Scholar] [CrossRef]
- Zhu, H.; Shen, F.; Wang, X.; Qian, H.; Liu, Y. Chlorogenic acid improves the cognitive deficits of sleep-deprived mice via regulation of immunity function and intestinal flora. Phytomedicine 2024, 123, 155194. [Google Scholar] [CrossRef]
- Ekhtiar, M.; Ghasemi-Dehnoo, M.; Azadegan-Dehkordi, F.; Bagheri, N. Evaluation of Anti-Inflammatory and Antioxidant Effects of Ferulic Acid and Quinic Acid on Acetic Acid-Induced Ulcerative Colitis in Rats. J. Biochem. Mol. Toxicol. 2025, 39, e70169. [Google Scholar] [CrossRef]
- Ekhtiar, M.; Ghasemi-Dehnoo, M.; Mirzaei, Y.; Azadegan-Dehkordi, F.; Amini-Khoei, H.; Lorigooini, Z.; Samiei-Sefat, A.; Bagheri, N. The coumaric acid and syringic acid ameliorate acetic acid-induced ulcerative colitis in rats via modulator of Nrf2/HO-1 and pro-inflammatory cytokines. Int. Immunopharmacol. 2023, 120, 110309. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Guo, K. Tannic acid alleviates ETEC K88-induced intestinal damage through regulating the p62-keap1-Nrf2 and TLR4-NF-κB-NLRP3 pathway in IPEC-J2 cells. J. Sci. Food Agric. 2024, 104, 5186–5196. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Ding, W.; Jia, L.; Zhu, K.; Luo, Q. Esculetin combats multidrug-resistant Salmonella infection and ameliorates intestinal dysfunction via the Nrf2 pathway. Antioxidants 2024, 13, 1170. [Google Scholar] [CrossRef]
- Gowtham, A.; Mishra, T.; Gungha, T.; Kaundal, R.K. Scopoletin Mitigates DSS-Induced Ulcerative Colitis by Attenuating NF-κB/MMP-9 Mediated Inflammation and Activating the Nrf2 Pathway to Preserve Colonic Barrier Integrity. Drug Dev. Res. 2025, 86, e70165. [Google Scholar] [CrossRef]
- Touny, A.A.; Venkataraman, B.; Almarzooqi, S.; Patil, R.B.; Bhongade, B.A.; Harjaček, M.; Rizvi, T.A.; Ojha, S.; Harihar Gowdru, S.N.; Subramanya, S.B. Molecular Docking, Dynamics, and Preclinical Studies Reveal Morin Hydrate as a Potent PPARγ and Nrf2 Agonist That Mitigates Colon Inflammation. Pharmacol. Res. Perspect. 2025, 13, e70132. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hua, L.; Hu, M.; Zhu, N.; Dong, S.; Jing, X.; Zhu, Z.; Liu, Y.; Zhou, Y. The protective effect of naringenin on ulcerative colitis in mice through increasing Nrf2 pathway activity: Naringenin protects mice with ulcerative colitis by increasing Nrf2 pathway activity. Acta Biochim. Biophys. Sin. 2025, 57, 1068. [Google Scholar] [CrossRef] [PubMed]
- Luo, D.; Huang, Z.; Jia, G.; Zhao, H.; Liu, G.; Chen, X. Naringin mitigates LPS-induced intestinal barrier injury in mice. Food Funct. 2023, 14, 1617–1626. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Lei, J.; Zhang, B. Effects of dietary quercetin on the antioxidative status and cecal microbiota in broiler chickens fed with oxidized oil. Poult. Sci. 2020, 99, 4892–4903. [Google Scholar] [CrossRef]
- Li, E.; Li, C.; Horn, N.; Ajuwon, K.M. Quercetin attenuates deoxynivalenol-induced intestinal barrier dysfunction by activation of Nrf2 signaling pathway in IPEC-J2 cells and weaned piglets. Curr. Res. Toxicol. 2023, 5, 100122. [Google Scholar] [CrossRef]
- Wang, C.; Fu, Y.; Wang, R.; Wang, Q.; Yu, H.; Zhang, J. Quercetin Attenuates the Combined Effects of Zearalenone and Lipopolysaccharide on IPEC-J2 Cell Injury through Activating the Nrf2 Signaling Pathway. Toxins 2023, 15, 679. [Google Scholar] [CrossRef]
- Zhu, X.; Li, Y.; Yue, L.; Zhou, X.; Li, J.; Zhang, Y.; Yu, Z.; Liu, Y.; Xu, Y.; Wu, L.; et al. Quercetin mitigates radiation-induced intestinal injury and promotes intestinal regeneration via Nrf2-mediated antioxidant pathway. Radiat. Res. 2023, 199, 252–262. [Google Scholar] [CrossRef]
- Cao, J.; Shi, D.; Cui, Y.; Zhu, H.; Liang, H.; Wei, Q.; Huang, J. Genistein maintains intestinal homeostasis in colitis mice via activating GPR30-Nrf2 signaling pathway. J. Nutr. Biochem. 2025, 145, 110036. [Google Scholar] [CrossRef]
- Liang, J.; Zhou, Y.; Cheng, X.; Chen, J.; Cao, H.; Guo, X.; Zhang, C.; Zhuang, Y.; Hu, G. Baicalin attenuates H2O2-induced oxidative stress by regulating the AMPK/Nrf2 signaling pathway in IPEC-J2 cells. Int. J. Mol. Sci. 2023, 24, 9435. [Google Scholar] [CrossRef]
- Lv, W.; Han, S.; Li, K.; Yan, A.; Wang, W.; Lu, W.; Han, J.; Zhang, C. Baicalein ameliorates DSS-induced ulcerative colitis in mice by inhibiting ferroptosis and regulating gut microbiota. Front. Pharmacol. 2025, 16, 1564783. [Google Scholar] [CrossRef]
- Ma, L.; Zhou, B.; Liu, H.; Chen, S.; Zhang, J.; Wang, T.; Wang, C. Dietary rutin improves the antidiarrheal capacity of weaned piglets by improving intestinal barrier function, antioxidant capacity and cecal microbiota composition. J. Sci. Food Agric. 2024, 104, 6262–6275. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Fang, Y.; Cheng, Y.; Xu, E.; Zhang, Y.; Zhai, Z. The Alleviating effect of taxifolin on deoxynivalenol-induced damage in porcine intestinal epithelial cells. Vet. Sci. 2024, 11, 156. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Bhandari, R.; Li, C.; Shu, P.; Shaikh, I.I. Pectolinarigenin suppresses LPS-induced inflammatory response in macrophages and attenuates DSS-induced colitis by modulating the NF-κB/Nrf2 signaling pathway. Inflammation 2022, 45, 2529–2543. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Dou, F.; Song, H.; Liu, T. Anti-ulcerative effects of wogonin on ulcerative colitis induced by dextran sulfate sodium via Nrf2/TLR4/NF-κB signaling pathway in BALB/c mice. Environ. Toxicol. 2022, 37, 954–963. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Wu, X.; Peng, B.; Zou, H.; Li, S.; Wang, J.; Cao, J. Curcumin improves necrotising microscopic colitis and cell pyroptosis by activating SIRT1/NRF2 and inhibiting the TLR4 signalling pathway in newborn rats. Innate Immun. 2020, 26, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Ohno, M.; Nishida, A.; Sugitani, Y.; Nishino, K.; Inatomi, O.; Sugimoto, M.; Kawahara, M.; Andoh, A. Nanoparticle curcumin ameliorates experimental colitis via modulation of gut microbiota and induction of regulatory T cells. PLoS ONE 2017, 12, e0185999. [Google Scholar] [CrossRef]
- Bian, W.; Wei, L.; Wang, K. Carthamin yellow alleviates dextran sodium sulfate-induced ulcerative colitis by repairing the intestinal barrier and activating the Nrf2/GPX4 axis. Int. Immunopharmacol. 2024, 141, 113020. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Tang, S.; Wan, F.; Zhong, R.; Chen, L.; Zhang, H. The PI3K/Akt-Nrf2 signaling pathway and mitophagy synergistically mediate hydroxytyrosol to alleviate intestinal oxidative damage. Int. J. Biol. Sci. 2024, 20, 4258. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zheng, Z.; Li, C.; Pan, Y.; Tang, X.; Wang, X.J. Synthetic imine resveratrol analog 2-methoxyl-3, 6-dihydroxyl-IRA ameliorates colitis by activating protective Nrf2 pathway and inhibiting NLRP3 expression. Oxidative Med. Cell. Longev. 2019, 2019, 7180284. [Google Scholar] [CrossRef]
- Zhang, J.; Li, X.; Zhu, Y.; Wang, Y.; Liu, D. Sericic Acid Ameliorates DSS-induced Ulcerative Colitis in Mice by Modulating the NF-ΚB and Nrf2 Pathways. Curr. Mol. Pharmacol. 2023, 16, 759–770. [Google Scholar]
- Yuan, Y.; Hu, Q.; Liu, L.; Xie, F.; Yang, L.; Li, Y.; Zhang, C.; Chen, H.; Tang, J.; Shen, X. Dehydrocostus lactone suppresses dextran sulfate sodium-induced colitis by targeting the IKKα/β-NF-κB and Keap1-Nrf2 signalling pathways. Front. Pharmacol. 2022, 13, 817596. [Google Scholar] [CrossRef]
- Chen, Y.; Miao, Z.; Sheng, X.; Li, X.; Ma, J.; Xu, X.; Li, H.; Kang, A. Sesquiterpene lactones-rich fraction from Aucklandia lappa Decne. alleviates dextran sulfate sodium induced ulcerative colitis through co-regulating MAPK and Nrf2/Hmox-1 signaling pathway. J. Ethnopharmacol. 2022, 295, 115401. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Liu, L.; Liu, X.; Chen, H.; Liu, K.; Huang, N.; Wang, Y. Epoxymicheliolide prevents dextran sulfate sodium-induced colitis in mice by inhibiting TAK1-NF-κB pathway and activating Keap1-NRF2 signaling in macrophages. Int. Immunopharmacol. 2022, 113, 109404. [Google Scholar] [CrossRef]
- Chen, Y.; Zhu, S.; Chen, Z.; Liu, Y.; Pei, C.; Huang, H.; Hou, S.; Ning, W.; Liang, J. Gingerenone A alleviates ferroptosis in secondary liver injury in colitis mice via activating Nrf2–Gpx4 signaling pathway. J. Agric. Food Chem. 2022, 70, 12525–12534. [Google Scholar] [CrossRef]
- Xiaohan, W.; Feng, T. P1335 Dietary Carnosic Acid Eases Colitis by Activating NRF2 Signal via Regulating Gut Microbiota and Metabolites. J. Crohn’s Colitis 2025, 19, i2405–i2406. [Google Scholar] [CrossRef]
- Bai, X.; Gou, X.; Cai, P.; Xu, C.; Cao, L.; Zhao, Z.; Huang, M.; Jin, J. Sesamin enhances Nrf2-mediated protective defense against oxidative stress and inflammation in colitis via AKT and ERK activation. Oxidative Med. Cell. Longev. 2019, 2019, 2432416. [Google Scholar] [CrossRef]
- Abdel-Wahab, B.A.; Alkahtani, S.A.; Alqahtani, A.A.; Hassanein, E.H. Umbelliferone ameliorates ulcerative colitis induced by acetic acid via modulation of TLR4/NF-κB-p65/iNOS and SIRT1/PPARγ signaling pathways in rats. Environ. Sci. Pollut. Res. 2022, 29, 37644–37659. [Google Scholar] [CrossRef]
- Salem, M.B.; El-Lakkany, N.M.; El-Din, S.H.S.; Hammam, O.A.; Samir, S. Diosmin alleviates ulcerative colitis in mice by increasing Akkermansia muciniphila abundance, improving intestinal barrier function, and modulating the NF-κB and Nrf2 pathways. Heliyon 2024, 10, e27527. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhao, L.; Xu, M.; Jiang, P.; Zhang, K. Moringin alleviates DSS-induced ulcerative colitis in mice by regulating Nrf2/NF-κB pathway and PI3K/AKT/mTOR pathway. Int. Immunopharmacol. 2024, 134, 112241. [Google Scholar] [CrossRef]
- Prakash, A.N.; Prasad, N.; Puppala, E.R.; Panda, S.R.; Jain, S.; Ravichandiran, V.; Singh, M.; Naidu, V. Loganic acid protects against ulcerative colitis by inhibiting TLR4/NF-κB mediated inflammation and activating the SIRT1/Nrf2 anti-oxidant responses in-vitro and in-vivo. Int. Immunopharmacol. 2023, 122, 110585. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Y.; Wu, H.; Wang, X.; He, J.; He, S.; Yin, Y. Resveratrol attenuates oxidative stress-induced intestinal barrier injury through PI3K/Akt-mediated Nrf2 signaling pathway. Oxidative Med. Cell. Longev. 2019, 2019, 7591840. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Li, X.; Xu, Y.; Li, Y.; Zhou, Y.; Zhang, J.; Guo, L. Resveratrol ameliorates ulcerative colitis by upregulating Nrf2/HO-1 pathway activity: Integrating animal experiments and network pharmacology. Mol. Med. Rep. 2024, 29, 77. [Google Scholar] [CrossRef] [PubMed]
- Sabzevary-Ghahfarokhi, M.; Soltani, A.; Luzza, F.; Larussa, T.; Rahimian, G.; Shirzad, H.; Bagheri, N. The protective effects of resveratrol on ulcerative colitis via changing the profile of Nrf2 and IL-1β protein. Mol. Biol. Rep. 2020, 47, 6941–6947. [Google Scholar] [CrossRef]
- Tian, Y.; Che, H.; Yang, J.; Jin, Y.; Yu, H.; Wang, C.; Fu, Y.; Li, N.; Zhang, J. Astaxanthin alleviates aflatoxin B1-induced oxidative stress and apoptosis in IPEC-J2 cells via the Nrf2 signaling pathway. Toxins 2023, 15, 232. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Bai, B.; Huang, K.; Li, S.; Cao, H.; Guan, X. Bound polyphenols of oat bran released by gut microbiota mitigate high fat diet-induced oxidative stress and strengthen the gut barrier via the colonic ROS/Akt/Nrf2 pathway. J. Agric. Food Chem. 2024, 72, 13099–13110. [Google Scholar] [CrossRef] [PubMed]
- Hou, C.; Yu, Z.; Shi, C.; Huang, Y.; Liu, H. Brown Algae Polysaccharides Alleviate Diquat-Induced Oxidative Stress in Piglets and IPEC-J2 Cells via Nrf2/ARE Signaling Pathway. Animal 2025, 15, 559. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Jiang, T.; Zhang, Y.; Shen, Z. Epimedium polysaccharides ameliorate ulcerative colitis by inhibiting oxidative stress and regulating autophagy. J. Sci. Food Agric. 2025, 105, 2655–2670. [Google Scholar] [CrossRef]
- Guo, S.; Shi, B.; Xing, Y.; Xu, Y.; Jin, X.; Hong, L.; Zhang, S.; Qiao, M.; Yan, S. Artemisia annua L. polysaccharide improves the growth performance and intestinal barrier function of broilers challenged with Escherichia coli. Front. Microbiol. 2024, 15, 1390815. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.L.; Xu, X.G.; Chang, Y.C.; Xu, Y.P.; Zhou, X.Q.; Su, H.L.; Cui, X.H.; Wan, X.Q.; Mao, G.X. Protective effect of mussel polysaccharide on cyclophosphamide-induced intestinal oxidative stress injury via Nrf2-Keap1 signaling pathway. Food Sci. Nutr. 2023, 11, 4233–4245. [Google Scholar] [CrossRef]
- Li, Z.-Y.; Lin, L.-H.; Liang, H.-J.; Li, Y.-Q.; Zhao, F.-Q.; Sun, T.-Y.; Liu, Z.-Y.; Zhu, J.-Y.; Gu, F.; Xu, J.-N.; et al. Lycium barbarum polysaccharide alleviates DSS-induced chronic ulcerative colitis by restoring intestinal barrier function and modulating gut microbiota. Ann. Med. 2023, 55, 2290213. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wu, P.; Fan, Z.; He, X.; Liu, J.; Li, M.; Chen, F. Dandelion polysaccharide treatment protects against dextran sodium sulfate-induced colitis by suppressing NF-κB/NLRP3 inflammasome-mediated inflammation and activating Nrf2 in mouse colon. Food Sci. Nutr. 2023, 11, 7271–7282. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.; Yin, L.; Dong, R. Inhibition of IEC-6 cell proliferation and the mechanism of ulcerative colitis in C57BL/6 mice by dandelion root polysaccharides. Foods 2023, 12, 3800. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, H.; Ge, Q.; Shi, J.; Zhang, H.; Gao, J.; Han, J. Polysaccharides of Melientha longistaminea regulates immune function and gut microbiota in cyclophosphamide (CTX)-induced immunosuppressed mice. Int. Immunopharmacol. 2025, 156, 114702. [Google Scholar] [CrossRef]
- Liu, S.Y.; Li, D.L.; Zhu, R.; Meng, S.T.; Wang, Y.T.; Li, L.; Yang, Z.Y.; Wu, L.F. Mechanisms underlying mitochondrial dysfunction and intestinal damage induced by ingestion of microplastics in Leuciscus waleckii: The role of the NF-κB/Nrf2 signaling pathway. Chemosphere 2024, 367, 143676. [Google Scholar] [CrossRef]
- Ma, H.; Mueed, A.; Ma, Y.; Ibrahim, M.; Su, L.; Wang, Q. Fecal microbiota transplantation activity of Floccularia luteovirens polysaccharides and their protective effect on cyclophosphamide-induced immunosuppression and intestinal injury in mice. Foods 2024, 13, 3881. [Google Scholar] [CrossRef]
- Cai, J.; Zhu, Z.; Li, Y.; Li, Q.; Tian, T.; Meng, Q.; Wang, T.; Ma, Y.; Wu, J. Artemisia capillaris Thunb. Polysaccharide alleviates cholestatic liver injury through gut microbiota modulation and Nrf2 signaling pathway activation in mice. J. Ethnopharmacol. 2024, 327, 118009. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; He, D.; Gao, X.; Wang, A.; Yu, J.; Wang, S.; Cui, B.; Mu, G.; Ma, C.; Tuo, Y. β-Glucan Extracted from Pichia kudriavzevii DPUL-51–6Y, Kluyveromyces marxianus DPUL-F15, and Saccharomyces cerevisiae DPUL-C6 Shows Ameliorating Effects on DSS-Induced Ulcerative Colitis on BALB/c Mice. J. Agric. Food Chem. 2025, 73, 10265–10278. [Google Scholar] [CrossRef] [PubMed]
- Xin, J.; Wang, T.; Ou, Y.; Qiu, X.; Li, M.; Zhang, Y.; Lu, S.; Xu, L.; Guan, L.; Yao, H. Structural Characterization and Protective Effects of Camellia oleifera Fruit Shell Polysaccharides against DSS-Induced Ulcerative Colitis in Mice. J. Agric. Food Chem. 2025, 73, 16860–16878. [Google Scholar] [CrossRef]
- Xie, W.-Y.; Ji, Z.-H.; Ren, W.-Z.; Zhao, P.-S.; Wei, F.-H.; Hu, J.; Yuan, B.; Gao, W. Wheat peptide alleviates DSS-induced colitis by activating the Keap1–Nrf2 signaling pathway and maintaining the integrity of the gut barrier. Food Funct. 2024, 15, 5466–5484. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Huang, Z.; Xiong, H.; Wang, J.; Zhang, H.; Guo, F.; Wang, C.; Sun, Y. Rice protein peptides alleviate dextran sulfate sodium-induced colitis via the Keap1–Nrf2 signaling pathway and regulating gut microbiota. J. Agric. Food Chem. 2022, 70, 12469–12483. [Google Scholar] [CrossRef]
- Luo, J.; Pan, J.; Song, H.; Cao, G.; Zhang, X.; Wang, W.; Qian, G.; Zhang, H.; Li, C. Proline improves intestinal damage induced by hibernation stress through Nrf2/ho-1/p65 signaling pathway in Pelodiscus sinensis. Aquaculture 2026, 610, 742891. [Google Scholar] [CrossRef]
- Wang, L.; Jiang, L.; Chu, Y.; Feng, F.; Tang, W.; Chen, C.; Qiu, Y.; Hu, Z.; Diao, H.; Tang, Z. Dietary taurine improves growth performance and intestine health via the GSH/GSSG antioxidant system and Nrf2/ARE signaling pathway in weaned piglets. Antioxidants 2023, 12, 1852. [Google Scholar] [CrossRef]
- Li, Z.; Chen, L.; Huang, Z.; Jia, G.; Zhao, H.; Liu, G.; Chen, X. Supplementation with L-theanine promotes intestinal antioxidant ability via Nrf2 signaling pathway in weaning piglets and H2O2-induced IPEC-J2 cells. J. Funct. Foods 2024, 121, 106433. [Google Scholar] [CrossRef]
- Kuo, Y.R.; Lin, C.H.; Lin, W.S.; Pan, M.H. L-Glutamine Substantially Improves 5-Fluorouracil-Induced Intestinal Mucositis by Modulating Gut Microbiota and Maintaining the Integrity of the Gut Barrier in Mice. Mol. Nutr. Food Res. 2024, 68, 2300704. [Google Scholar] [CrossRef]
- Xiong, L.; Wang, K.; Song, M.; Azad, M.A.K.; Zhu, Q.; Kong, X. Dietary betaine supplementation enhances colonic barrier function through the Nrf2/Keap1 and TLR4-NF-κB/MAPK signaling pathways and alters colonic microbiota in Bama mini-pigs. Antioxidants 2023, 12, 1926. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Hu, Z.; Luo, X.; Ge, C.; Lv, Y.; Zhan, S.; Huang, W.; Shen, X.; Yu, D.; Liu, B. Itaconic acid alleviates perfluorooctanoic acid-induced oxidative stress and intestinal damage by regulating the Keap1/Nrf2/Ho-1 pathway and reshaping the gut microbiota. Int. J. Mol. Sci. 2024, 25, 9826. [Google Scholar] [CrossRef]
- Jiang, P.; Zhai, Z.; Zhao, L.; Zhang, K.; Duan, L. α-Lipoic acid alleviates dextran sulfate sodium salt-induced ulcerative colitis via modulating the Keap1–Nrf2 signaling pathway and inhibiting ferroptosis. J. Sci. Food Agric. 2024, 104, 1679–1690. [Google Scholar] [CrossRef] [PubMed]
- Abdelsameea, A.A.; Alsemeh, A.E.; Alabassery, N.; Samy, W.; Fawzy, A.; Abbas, N.A. Icosapent ethyl alleviates acetic acid-induced ulcerative colitis via modulation of SIRT1 signaling pathway in rats. Int. Immunopharmacol. 2023, 115, 109621. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Tang, X.; Zhang, Q.; Xiong, F.; Yan, Y.; Zhao, J.; Mao, B.; Zhang, H.; Cui, S. Lacticaseibacillus paracasei CCFM1222 ameliorated the intestinal barrier and regulated gut microbiota in mice with dextran sulfate sodium-induced colitis. Probiotics Antimicrob. Proteins 2025, 17, 1001–1013. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Guan, C.; Wang, T.; Mu, G.; Tuo, Y. Indole-3-lactic acid, a tryptophan metabolite of Lactiplantibacillus plantarum DPUL-S164, improved intestinal barrier damage by activating AhR and Nrf2 signaling pathways. J. Agric. Food Chem. 2023, 71, 18792–18801. [Google Scholar] [CrossRef] [PubMed]
- Bian, Z.; Zhang, Q.; Qin, Y.; Sun, X.; Liu, L.; Liu, H.; Mao, L.; Yan, Y.; Liao, W.; Zha, L.; et al. Sodium butyrate inhibits oxidative stress and NF-κB/NLRP3 activation in dextran sulfate sodium salt-induced colitis in mice with involvement of the Nrf2 signaling pathway and mitophagy. Dig. Dis. Sci. 2023, 68, 2981–2996. [Google Scholar] [CrossRef]
- Cui, C.; Wei, Y.; Wang, Y.; Ma, W.; Zheng, X.; Wang, J.; Ma, Z.; Wu, C.; Chu, L.; Zhang, S.; et al. Dietary supplementation of benzoic acid and essential oils combination enhances intestinal resilience against LPS stimulation in weaned piglets. J. Anim. Sci. Biotechnol. 2024, 15, 4. [Google Scholar] [CrossRef]
- Yan, B.; Mao, X.; Hu, S.; Wang, S.; Liu, X.; Sun, J. Spermidine protects intestinal mucosal barrier function in mice colitis via the AhR/Nrf2 and AhR/STAT3 signaling pathways. Int. Immunopharmacol. 2023, 119, 110166. [Google Scholar] [CrossRef] [PubMed]
- Abdelhamid, A.M.; Youssef, M.E.; Cavalu, S.; Mostafa-Hedeab, G.; Youssef, A.; Elazab, S.T.; Ibrahim, S.; Allam, S.; Elgharabawy, R.M.; El-Ahwany, E. Carbocisteine as a modulator of Nrf2/HO-1 and NFκB interplay in rats: New inspiration for the revival of an old drug for treating ulcerative colitis. Front. Pharmacol. 2022, 13, 887233. [Google Scholar] [CrossRef]
- El-Fadl, H.M.I.A.; Mohamed, M.F. Targeting endoplasmic reticulum stress, Nrf-2/HO-1, and NF-κB by myristicin and its role in attenuation of ulcerative colitis in rats. Life Sci. 2022, 311, 121187. [Google Scholar] [CrossRef]
- He, C.; Gao, M.; Zhang, X.; Lei, P.; Yang, H.; Qing, Y.; Zhang, L. The protective effect of sulforaphane on dextran sulfate sodium-induced colitis depends on gut microbial and Nrf2-related mechanism. Front. Nutr. 2022, 9, 893344. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, Y.; Song, M.; Guo, X.; Fan, J.; Tao, S. Taurodeoxycholic acid alleviates diquat-induced intestinal barrier function injury in mice through the upregulation of Nrf2-mediated signaling pathway. Anim. Dis. 2024, 4, 34. [Google Scholar] [CrossRef]
- Li, Z.; Long, L.; Jin, X.; Li, Y.; Wu, Q.; Chen, X.; Geng, Z.; Zhang, C. Effects of Clostridium butyricum on growth performance, meat quality, and intestinal health of broilers. Front. Vet. Sci. 2023, 10, 1107798. [Google Scholar] [CrossRef]
- Zhu, C.; Liang, S.; Zan, G.; Wang, X.; Gao, C.; Yan, H.; Wang, X.; Zhou, J. Selenomethionine alleviates DON-induced oxidative stress via modulating Keap1/Nrf2 signaling in the small intestinal epithelium. J. Agric. Food Chem. 2022, 71, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Qiao, L.; Dou, X.; Song, X.; Chang, J.; Zeng, X.; Xu, C. Lactobacillus casei ATCC 393 combined with vasoactive intestinal peptide alleviates dextran sodium sulfate-induced ulcerative colitis in C57BL/6 mice via NF-κB and Nrf2 signaling pathways. Biomed. Pharmacother. 2023, 165, 115033. [Google Scholar] [CrossRef]
- Jiang, H.; Mao, T.; Sun, Z.; Shi, L.; Han, X.; Zhang, Y.; Zhang, X.; Wang, J.; Hu, J.; Zhang, L.; et al. Yinchen Linggui Zhugan decoction ameliorates high fat diet-induced nonalcoholic fatty liver disease by modulation of SIRT1/Nrf2 signaling pathway and gut microbiota. Front. Microbiol. 2022, 13, 1001778. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Liu, G.; Xia, X.; Gan, D.; Xiang, S.; Xiang, M. α-Mangostin suppresses ethanol-induced gastric ulceration by regulating the Nrf2/HO-1 and NF-κB/NLRP3/caspase-1 signaling pathways and gut microbiota. Heliyon 2024, 10, e24339. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Song, X.; Zhou, Y.; Xia, Y.; Yang, Z.; Chen, X.; Shi, R.; Geng, Z.; Zhang, X.; Wang, Y.; et al. Sclareol protected against intestinal barrier dysfunction ameliorating Crohn’s disease-like colitis via Nrf2/NF-B/MLCK signalling. Int. Immunopharmacol. 2024, 133, 112140. [Google Scholar] [CrossRef]
- Jia, B.; Zhao, L.; Liu, P.; Li, M.; Tian, Z. Limonin ameliorates indomethacin-induced intestinal damage and ulcers through Nrf2/ARE pathway. Immun. Inflamm. Dis. 2023, 11, e787. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Li, Z.; Liu, L.; Geng, X.; Liu, H.; Wang, C.; Wu, Y.; Lin, H.; Liu, J. Oleanolic acid 28-O-β-D-glucopyranoside alleviates TNBS-induced ulcerative colitis in rat by regulating Nrf2/x-CT/GPX4-mediated ferroptosis. J. Ethnopharmacol. 2025, 353, 120369. [Google Scholar] [CrossRef]
- Qin, Y.; Zhang, R.-Y.; Zhang, Y.; Zhao, Y.-Q.; Hao, H.-F.; Wang, J.-P. Network pharmacology and in vivo study: Unraveling the therapeutic mechanisms of Panax ginseng in potentially treating ulcerative colitis. World J. Gastroenterol. 2025, 31, 100271. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Yuan, R.; Fan, Q.; Zhang, C.; Han, S.; Li, J.; Xu, Z.; Sun, K.; Xu, Q.; Yao, C.; et al. Ginsenoside Rb1 exerts therapeutic effects on ulcerative colitis through regulating the Nrf2/PIP2/NLRP3 inflammasome signaling pathway. J. Funct. Foods 2023, 102, 105475. [Google Scholar] [CrossRef]
- Shen, J.; Ma, X.; He, Y.; Wang, Y.; Zhong, T.; Zhang, Y. Anti-inflammatory and anti-oxidant properties of Melianodiol on DSS-induced ulcerative colitis in mice. PeerJ 2022, 10, e14209. [Google Scholar] [CrossRef]
- Jia, L.; Xue, K.; Liu, J.; Habotta, O.A.; Hu, L.; Abdel Moneim, A.E. Anticolitic effect of berberine in rat experimental model: Impact of PGE2/p38 MAPK pathways. Mediat. Inflamm. 2020, 2020, 9419085. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, Z.; Du, M.; Ji, X.; Liu, X.; Zhao, C.; Pang, X.; Jin, E.; Wen, A.; Li, S.; et al. Berberine alleviates ETEC-induced intestinal inflammation and oxidative stress damage by optimizing intestinal microbial composition in a weaned piglet model. Front. Immunol. 2024, 15, 1460127. [Google Scholar] [CrossRef]
- Xu, X.; Li, W.; Yu, Z.; Zhang, L.; Duo, T.; Zhao, Y.; Qin, W.; Yang, W.; Ma, L. Berberine ameliorates dextran sulfate sodium-induced ulcerative colitis and inhibits the secretion of gut lysozyme via promoting autophagy. Metabolites 2022, 12, 676. [Google Scholar] [CrossRef] [PubMed]
- Niu, Z.; Li, X.; Yang, X.; Sun, Z. Protective effects of sinomenine against dextran sulfate sodium-induced ulcerative colitis in rats via alteration of HO-1/Nrf2 and inflammatory pathway. Inflammopharmacology 2024, 32, 2007–2022. [Google Scholar] [CrossRef]
- Chen, G.; Wen, D.; Shen, L.; Feng, Y.; Xiong, Q.; Li, P.; Zhao, Z. Cepharanthine exerts antioxidant and anti-inflammatory effects in lipopolysaccharide (LPS)-induced macrophages and DSS-induced colitis mice. Molecules 2023, 28, 6070. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Han, K.; Liu, H.; Jia, G.; Comer, L.; Wang, G.; Pan, Z.; Zhao, Y.; Jiang, S.; Jiao, N.; et al. Macleaya cordata isoquinoline alkaloids attenuate Escherichia coli lipopolysaccharide-induced intestinal epithelium injury in broiler chickens by co-regulating the TLR4/MyD88/NF-κB and Nrf2 signaling pathways. Front. Immunol. 2024, 14, 1335359. [Google Scholar] [CrossRef]
- Li, C.; Liu, M.; Deng, L.; Luo, D.; Ma, R.; Lu, Q. Oxyberberine ameliorates TNBS-induced colitis in rats through suppressing inflammation and oxidative stress via Keap1/Nrf2/NF-κB signaling pathways. Phytomedicine 2023, 116, 154899. [Google Scholar] [CrossRef]
- Yuan, J.; Su, J.; Zhong, S.; Yuan, X.; Zhu, J.; Lu, S.; Zhang, D.; Li, G.; Xue, H.; Yan, M.; et al. Dictamnine alleviates DSS-induced colitis mice by inhibiting ferroptosis of enterocytes via activating Nrf2-Gpx4 signaling pathway. Eur. J. Pharmacol. 2025, 997, 177464. [Google Scholar] [CrossRef]
- Zhang, H.; Lang, W.; Li, S.; Xu, C.; Wang, X.; Li, Y.; Zhang, Z.; Wu, T.; Feng, M. Corynoline ameliorates dextran sulfate sodium-induced colitis in mice by modulating Nrf2/NF-κB pathway. Immunopharmacol. Immunotoxicol. 2023, 45, 26–34. [Google Scholar] [CrossRef]
- Zhu, F.; Pan, H.; Li, B.; Mao, X.; Chen, Z.; Ye, G.; Gao, J.; Shen, Y.; Hu, J.; Lou, Z. Hericium erinaceus extract attenuates gastric mucosal injury via Nrf2/HO-1 pathway activation and gut microbiota modulation in rats. J. Funct. Foods 2025, 128, 106842. [Google Scholar] [CrossRef]
- Zhou, H.; Zheng, X.; Huang, S.; Wang, X.; Zhou, T.; Zhang, S.; Ling, Y.; Wang, W.; Li, X.; Li, S.; et al. Rosa roxburghii Fermentation Broths Attenuate Bleomycin-Induced Pulmonary Fibrosis by Activating the Nrf2/HO-1/NQO1 Signaling Pathway and Modulating Gut Microbiota. Food Sci. Nutr. 2025, 13, e70105. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Huang, Y.; Yang, W.; Huang, C.; Ou, Z.; He, J.; Yang, M.; Wu, J.; Yao, H.; Yang, Y.; et al. Viola yedoensis Makino alleviates lipopolysaccharide-induced intestinal oxidative stress and inflammatory response by regulating the gut microbiota and NF-κB-NLRP3/Nrf2-MAPK signaling pathway in broiler. Ecotoxicol. Environ. Saf. 2024, 282, 116692. [Google Scholar] [CrossRef]
- Chao, L.; Lin, J.; Zhou, J.; Du, H.; Chen, X.; Liu, M.; Qu, Q.; Lv, W.; Guo, S. Polyphenol rich Forsythia suspensa extract alleviates DSS-induced ulcerative colitis in mice through the Nrf2-NLRP3 pathway. Antioxidants 2022, 11, 475. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Mei, W.; Zhu, J.; Wang, Z.; Liu, X.; Zhou, R.; Liu, X. Aurantii Fructus extract alleviates DSS-induced colitis in mice via regulating NF-κB and Nrf2/HO-1 signaling pathways and modulating intestinal microbiota. Front. Nutr. 2025, 12, 1661040. [Google Scholar] [CrossRef] [PubMed]
- Puppala, E.R.; Yalamarthi, S.S.; Aochenlar, S.L.; Prasad, N.; Syamprasad, N.; Singh, M.; Nanjappan, S.K.; Ravichandiran, V.; Tripathi, D.M.; Gangasani, J.K. Mesua assamica (King&Prain) kosterm. Bark ethanolic extract attenuates chronic restraint stress aggravated DSS-induced ulcerative colitis in mice via inhibition of NF-κB/STAT3 and activation of HO-1/Nrf2/SIRT1 signaling pathways. J. Ethnopharmacol. 2023, 301, 115765. [Google Scholar] [PubMed]
- Huang, T.; Che, Q.; Chen, X.; Chen, D.; Yu, B.; He, J.; Chen, H.; Yan, H.; Zheng, P.; Luo, Y.; et al. Apple polyphenols improve intestinal antioxidant capacity and barrier function by activating the Nrf2/Keap1 signaling pathway in a pig model. J. Agric. Food Chem. 2022, 70, 7576–7585. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Luo, Y.; Lu, Y.; Wang, D.; Wang, T.; Pu, W.; Wang, Y. Maggot extracts alleviate inflammation and oxidative stress in acute experimental colitis via the activation of Nrf2. Oxidative Med. Cell. Longev. 2019, 2019, 4703253. [Google Scholar] [CrossRef]
- Li, W.; Wang, Y.; Zhang, Y.; Fan, Y.; Liu, J.; Zhu, K.; Jiang, S.; Duan, J. Lizhong decoction ameliorates ulcerative colitis by inhibiting ferroptosis of enterocytes via the Nrf2/SLC7A11/GPX4 pathway. J. Ethnopharmacol. 2024, 326, 117966. [Google Scholar] [CrossRef]
- Kong, J.; Xiang, Q.; Ge, W.; Wang, Y.; Xu, F.; Shi, G. Network pharmacology mechanisms and experimental verification of licorice in the treatment of ulcerative colitis. J. Ethnopharmacol. 2024, 324, 117691. [Google Scholar] [CrossRef] [PubMed]
- Kong, J.; Xiang, Q.; Shi, G.; Xu, Z.; Ma, X.; Wang, Y.; Xuan, Z.; Xu, F. Licorice protects against ulcerative colitis via the Nrf2/PINK1-mediated mitochondrial autophagy. Immun. Inflamm. Dis. 2023, 11, e757. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Zhou, Z.; Zhang, L.; Wang, H.; Lu, J.; Wang, X.; An, R. Gegen qinlian decoction relieves ulcerative colitis via adjusting dysregulated Nrf2/ARE signaling. Evid.-Based Complement. Altern. Med. 2022, 2022, 2934552. [Google Scholar] [CrossRef]
- Wen, H.; Zhang, X.; Li, Q.; Huang, J.; Liu, G.; Zhao, J.; Liu, Y.; Shen, L.; Li, Y.; Yang, K.; et al. Ruscogenins improve CD-like enteritis by inhibiting apoptosis of intestinal epithelial cells and activating Nrf2/NQO1 pathway. Oxidative Med. Cell. Longev. 2022, 2022, 4877275. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Wen, H.; Zuo, L.; Song, X.; Geng, Z.; Ge, S.; Ge, Y.; Wu, R.; Chen, S.; Yu, C. Bryostatin-1 attenuates intestinal ischemia/reperfusion-induced intestinal barrier dysfunction, inflammation, and oxidative stress via activation of Nrf2/HO-1 signaling. FASEB J. 2023, 37, e22948. [Google Scholar] [CrossRef]
- Li, F.; Huang, H.; Zhao, P.; Jiang, J.; Ding, X.; Lu, D.; Ji, L. Curculigoside mitigates dextran sulfate sodium-induced colitis by activation of KEAP1-NRF2 interaction to inhibit oxidative damage and autophagy of intestinal epithelium barrier. Int. J. Mol. Med. 2023, 52, 107. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ji, X.; Wang, X.; Sun, M.; Li, C.; Wu, D. Cannabidiol Alleviates Intestinal Fibrosis in Mice with Ulcerative Colitis by Regulating Transforming Growth Factor Signaling Pathway. J. Inflamm. Res. 2025, 18, 1–15. [Google Scholar] [CrossRef]
- Tsopmejio, I.S.N.; Yuan, J.; Diao, Z.; Fan, W.; Wei, J.; Zhao, C.; Li, Y.; Song, H. Auricularia polytricha and Flammulina velutipes reduce liver injury in DSS-induced Inflammatory Bowel Disease by improving inflammation, oxidative stress, and apoptosis through the regulation of TLR4/NF-κB signaling pathways. J. Nutr. Biochem. 2023, 111, 109190. [Google Scholar] [CrossRef] [PubMed]
- Shu, L.; Fu, H.; Pi, A.; Feng, Y.; Dong, H.; Si, C.; Li, S.; Zhu, F.; Zheng, P.; Zhu, Q. Protective effect of andrographolide against ulcerative colitis by activating Nrf2/HO-1 mediated antioxidant response. Front. Pharmacol. 2024, 15, 1424219. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Tong, M.; Sun, J.; Wu, Q.; Li, Z.; Lv, T.; Yin, X.; Zhang, M.; Li, L. Integrated lipid metabolomics and network pharmacology to investigate the potential mechanism of Coix seed in alleviating ulcerative colitis. J. Funct. Foods 2025, 127, 106744. [Google Scholar] [CrossRef]
- Yang, S.; Li, F.; Lu, S.; Ren, L.; Bian, S.; Liu, M.; Zhao, D.; Wang, S.; Wang, J. Ginseng root extract attenuates inflammation by inhibiting the MAPK/NF-κB signaling pathway and activating autophagy and p62-Nrf2-Keap1 signaling in vitro and in vivo. J. Ethnopharmacol. 2022, 283, 114739. [Google Scholar] [CrossRef]
- BinMowyna, M.N. Zingerone attenuates intestinal injury and colitis caused by a high-fat diet through Nrf2 signaling regulation. Saudi J. Biol. Sci. 2023, 30, 103775. [Google Scholar] [CrossRef]
- Yao, Y.; Chen, T.; Wu, H.; Yang, N.; Xu, S. Melatonin attenuates bisphenol A-induced colon injury by dual targeting mitochondrial dynamics and Nrf2 antioxidant system via activation of SIRT1/PGC-1α signaling pathway. Free Radic. Biol. Med. 2023, 195, 13–22. [Google Scholar] [CrossRef]
- Yu, T.; Zhang, J.; Zhai, F.; Yu, D.; An, L.; Hu, Q.; Song, C.; Li, J. Hernandezine alleviates inflammatory bowel disease by enhancing gut barrier integrity via the AMPK/NRF2 pathway and regulating immune-metabolic balance. Int. Immunopharmacol. 2025, 163, 115230. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.-S.; Chen, Q.-L.; Wu, G.-P.; Yao, Y.-W.; Fan, Y.-X.; Linghu, K.-G.; Chen, J.-M.; Xiong, W.; Yu, H. Sigesbeckia pubescens makino alleviates ulcerative colitis in mice by modulating the Nrf2/Keap1 pathway. Front. Pharmacol. 2025, 16, 1588525. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Liu, J.; Yan, H.; Yuan, Q.; Zhan, H.; Ai, Y.; Wang, X.; Chen, Y.; Shi, L.; Zhang, Y. Polygonum hydropiper L. ethanol extract attenuates ferroptosis and ulcerative colitis by modulating glutathione metabolism through inhibition of KEAP1-Nrf2 interaction. Phytomedicine 2025, 146, 157096. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Tian, X.; Yan, Y.; Tian, S.; Liu, D.; Xu, J. Lithospermic acid alleviates oxidative stress and inflammation in DSS-induced colitis through Nrf2. Eur. J. Pharmacol. 2025, 995, 177390. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.