Fortification of Ground Roasted Coffees with Iron, Zinc, and Calcium Salts: Evaluation of Minerals Recovery in Filtered and Espresso Brews
Abstract
1. Introduction
2. Methods
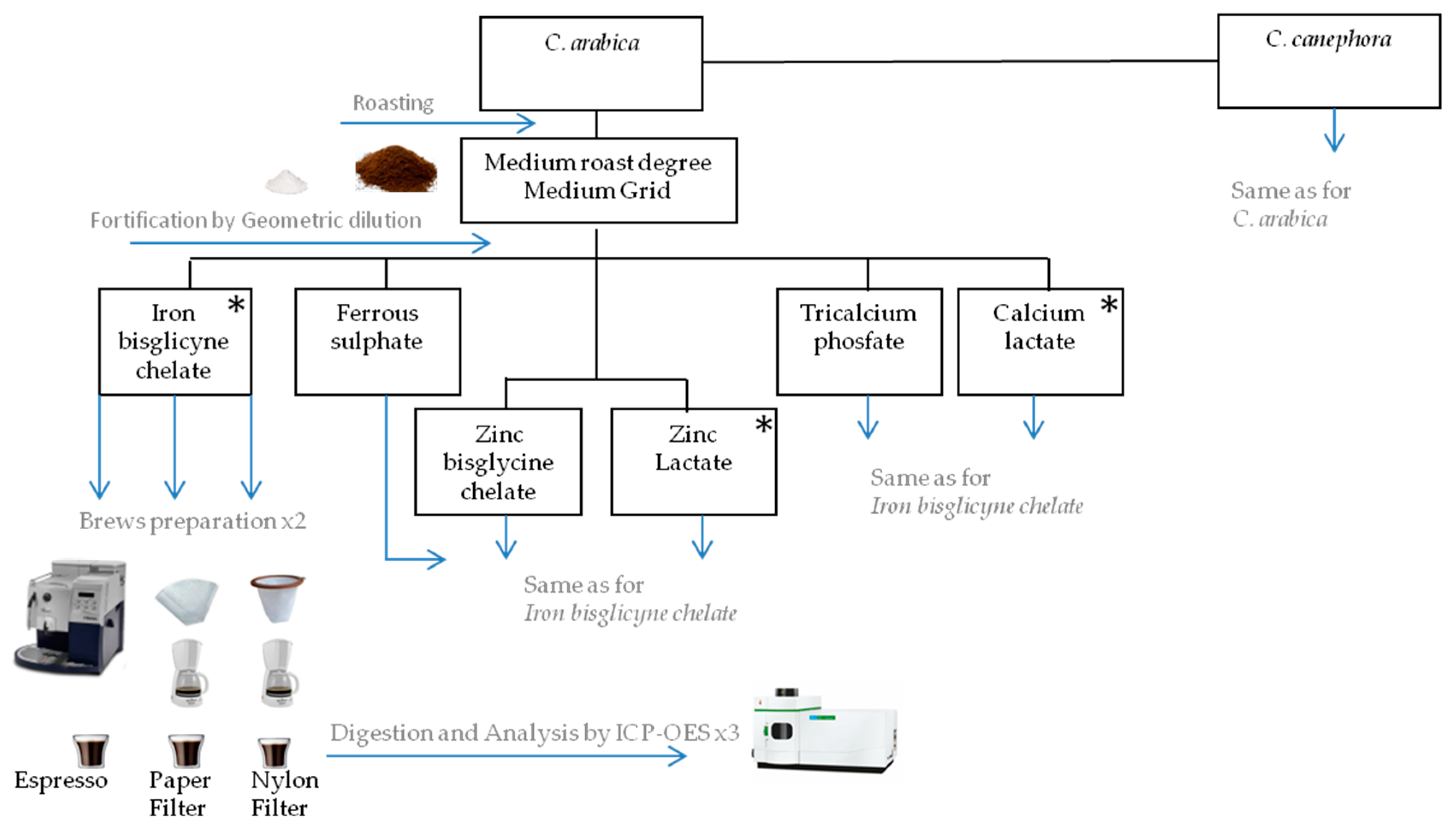
2.1. Preparation of Coffee Matrices
2.2. Mineral Salts
2.3. Fortification of Ground Roasted Coffee
2.4. Preliminary Sensory Test
2.5. Brews Preparation
2.6. Minerals Analyses
2.7. Statistical Analyses
3. Results and Discussion
3.1. Preliminary Sensory Test
3.2. Mineral Contents in Unfortified and Fortified Ground Roasted Coffees
3.2.1. Unfortified (Control) Coffees
3.2.2. Fortified Coffees
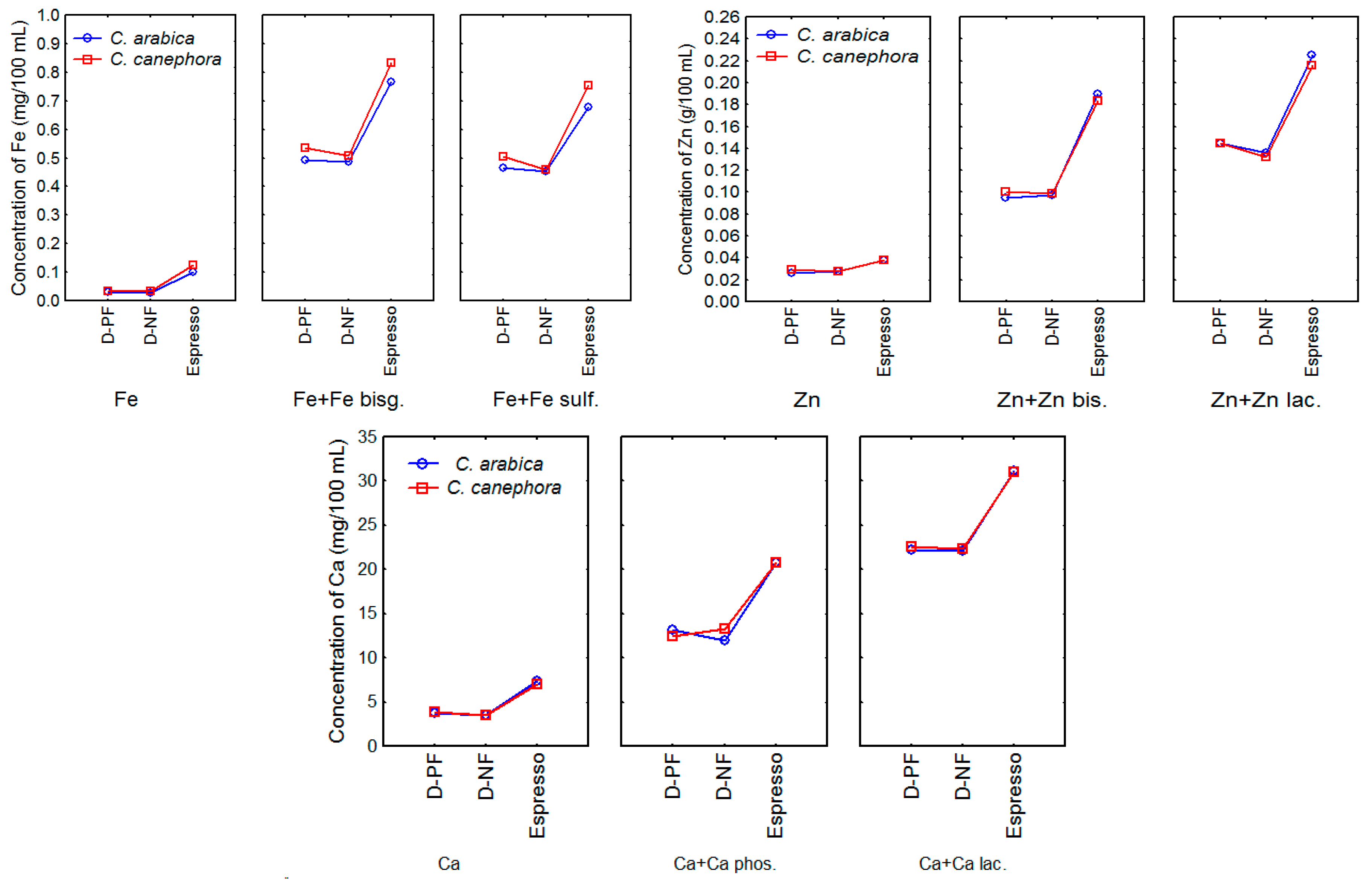
3.3. Mineral Concentrations in Unfortified and Fortified Coffee Brews
3.3.1. Brews from Unfortified Coffee Grounds
3.3.2. Brews from Fortified Ground Coffees
4. Conclusions and Final Considerations
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tulchinsky, T.H. Micronutrient Deficiency conditions: Global health issues. Public Health Rev. 2010, 32, 243–255. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines on Food Fortification with Micronutrients; WHO: Geneva, Switzerland, 2006; Available online: http://www.who.int/nutrition/publications/guide_food_fortification_micronutrients.pdf (accessed on 20 August 2018).
- Piccoli, N.B.; Grede, N.; Pee, S.; Singhkumarwong, A.; Roks, E.; Moench-Pfanner, R.; Bloem, M.W. Rice fortification: Its potential for improving micronutrient intake and steps required for implementation at scale. Food Nutr. Bull. 2012, 33, S360–S372. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The Double Burden of Malnutrition—Policy Brief; WHO: Geneva, Switzerland, 2016; Available online: http://apps.who.int/iris/bitstream/handle/10665/255413/WHO-NMH-NHD-17.3-eng.pdf?ua=1 (accessed on 5 August 2018).
- Metas Mundiales de Nutritión 2025: Documento Normativo sobre Anemia; WHO: Geneva, Switzerland, 2017. Available online: https://www.who.int/nutrition/publications/globaltargets2025_policybrief_anaemia/es/ (accessed on 29 July 2018).
- McLean, E.; Cogswel, M.; Egli, I.; Wojdyla, D.; Benoist, B. Worldwide prevalence of anaemia, WHO Vitamin and Mineral Nutrition Information System, 1993–2005. Public Health Nutr. 2009, 12, 444–454. [Google Scholar] [CrossRef] [PubMed]
- Mirza, F.G.; Kadir, R.A.; Breymann, C.; Fraser, I.S.; Taher, I. Impact and management of iron deficiency and iron deficiency anemia in women’s health. Expert Rev. Hematol. 2018, 11, 727–736. [Google Scholar] [CrossRef]
- Choi, S.; Liu, X.; Pan, Z. Zinc deficiency and cellular oxidative stress: Prognostic implications in cardiovascular diseases. Acta Pharmacol. Sin. 2018, 39, 1120–1132. [Google Scholar] [CrossRef] [PubMed]
- Chasapis, C.C.; Loutsidou, A.S.; Chara, S.M. Zinc and human health: An update. Arch. Toxicol. 2012, 86, 521–534. [Google Scholar] [CrossRef] [PubMed]
- Maxfield, L.; Crane, J.S. Zinc Deficiency. StatPearls Publishing LLC: Florida, FL, USA, 2018. [Google Scholar]
- Ross, C.; Taylor, C.L.; Yaktine, A.L.; Del Valle, H.B. Dietary Reference Intakes for Calcium and Vitamin D; Institute of Medicine (US) Committee; National Academies Press (US): Washington, DC, USA, 2011. [Google Scholar]
- Prentice, A. Nutritional rickets around the world. J. Steroid Biochem. Mol. Biol. 2013, 136, 201–206. [Google Scholar] [CrossRef]
- US Department of Health & Human Services. National Institutes of Health, 2018. Available online: https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional/ (accessed on 16 October 2018).
- Marques, M.F.; Marques, M.M.; Xavier, E.R.; Gregório, E.L. Fortificação de alimentos: Uma alternativa para suprir as necessidades de micronutrientes no mundo contemporâneo. HU Revista 2012, 38, 79–86. [Google Scholar]
- De Pee, S. Proposing Nutrients and Nutrient Levels for Rice Fortification; Nutrition Advisory Office, World Food Programme: Rome, Italy, 2014. [Google Scholar]
- International Coffee Organization (ICO). Coffee Market Report; International Coffee Organization: London, UK, 2017. Available online: http://www.ico.org/prices/new-consumption-table.pdf (accessed on 15 November 2018).
- Einöther, S.J.; Giesbrecht, T. Caffeine as an attention enhancer: Reviewing existing assumptions. Psychopharmacology 2013, 225, 251–274. [Google Scholar] [CrossRef]
- Nehlig, A. Is caffeine a cognitive enhancer? J. Alzheimers Dis. 2010, 20, S85–S94. [Google Scholar] [CrossRef]
- Borota, D.; Murray, E.; Keceli, G.; Chang, A.; Watabe, J.M.; Ly, M.; Toscano, J.P.; Yassa, M.A. Post-study caffeine administration enhances memory consolidation in humans. Nat. Neurosci. 2014, 17, 201–203. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.; Sutherland, D.; Christopher, G. Effects of repeated doses of caffeine on mood and performance of alert and fatigued volunteers. J. Psychopharmacol. 2005, 19, 620–626. [Google Scholar] [CrossRef] [PubMed]
- Olson, C.A.; Thornton, J.A.; Adam, G.E.; Lieberman, H.R. Effects of 2 adenosine antagonists, quercetin and caffeine, on vigilance and mood. J. Clin. Psychopharmacol. 2010, 30, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Arab, L.; Khan, F.; Lam, H. Epidemiologic evidence of a relationship between tea, coffee, or caffeine consumption and cognitive decline. Adv. Nutr. 2013, 4, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, K.; Carrière, I.; De Mendonça, A.; Portet, F.; Dartigues, J.F.; Rouaud, O.; Barberger-Gateau, P.; Ancelin, M.L. The neuroprotective effects of caffeine: A prospective population study (the three city study). Neurology 2007, 69, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Corley, J.; Jia, X.; Kyle, J.A.; Gow, A.J.; Brett, C.E.; Starr, J.M.; Mcneill, G.; Deary, I.J. Caffeine consumption and cognitive function at age 70: The Lothian Birth Cohort 1936 Study. Psychosom. Med. 2010, 72, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, E.R.; Ziegenfuss, T.; Kalman, D.; Kreider, R.; Campbell, B.; Wilborn, C.; Taylor, L.; Willoughby, D.; Stout, J.; Graves, B.S.; et al. International society of sports nutrition position stand: Caffeine and performance. J. Int. Soc. Sports Nutr. 2010, 7, 5. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef]
- Liang, N.; Kitts, D.D. Role of chlorogenic acids in controlling oxidative and inflammatory stress conditions. Nutrients 2015, 8, 16. [Google Scholar] [CrossRef]
- Figueiredo, R.C. Enriquecimento de Café Solúvel com Ferro, Zinco e Ácido Fólico: Avaliação da Biodisponibilidade em Mulheres Adultas [Dissertação de Mestrado]; Universidade Federal do Rio de Janeiro: Rio de Janeiro, Brazil, 2007. [Google Scholar]
- Giorgini, E.; Fisberg, M.; De Paula, R.A.C.; Ferreira, A.M.A.; Valle, J.; Braga, J.A.P. The use of sweet rolls fortified with iron bis-glycinate chelate in the prevention of iron deficiency anemia in preschool children. Archivos Latinoamericanos de Nutrición 2001, 1, 48–53. [Google Scholar]
- Rodrigues, J.E.F.G.; Pineda, O.; Name, J.J.; Sanchez, J.G. Effectiveness of iron bis-glycine chelate in chocolate drink in the control of iron deficiency in preschool children. Nutrire 2006, 31, 43–52. [Google Scholar]
- International Coffee Organization (ICO). Annual Review, 2011/2012. London, UK. Available online: http://www.ico.org/documents/cy2012-13/annual-review-2011-12e.pdf (accessed on 20 September 2018).
- Associação Brasileira da Indústria de Café. Tendências do Mercado de Café. 2016. Rio de Janeiro, Brasil. Available online: http://abic.com.br/src/uploads/2018/05/2016.pdf (accessed on 8 October 2018).
- Monteiro, M.A.; Minim, V.P.; Silva, A.F.; Chaves, J.B. Influência da torra sobre a aceitação da bebida café. Rev. Ceres 2010, 57, 145–150. [Google Scholar] [CrossRef]
- Umbelino, D.C.; Rossi, E.A.; Cardello, H.M.; Lepera, J.S. Aspectos tecnológicos e sensoriais do “iogurte” de soja enriquecido com cálcio. Ciência Tecnologia Alimentos 2001, 21, 276–280. [Google Scholar] [CrossRef]
- Tuma, R.B.; Yuyama, L.K.; Aguiar, J.; Marques, H. Impacto da farinha de mandioca fortificada com ferro aminoácido quelato no nível de hemoglobina de pré-escolares. Revista Nutrição 2003, 16, 29–39. [Google Scholar] [CrossRef]
- Casé, F.; Deliza, R.; Rosenthal, A.; Mantovani, D.; Felberg, I. Produção de “leite” de soja enriquecido com cálcio. Ciência Tecnologia Alimentos 2005, 25, 86–91. [Google Scholar] [CrossRef]
- Gonçalves, A.A.; Leindecker, T.; Biedrzycki, A. Suco de uva em pó fortificado com ferro. Alimentos Nutrição Araraquara 2008, 19, 177–181. [Google Scholar]
- Costa, L. Café Torrado e Moído Fortificado: Avaliação da Eficiência de Extração dos Minerais Adicionados e Análise Sensorial do Produto Desenvolvido [Dissertação de Mestrado]; Universidade Federal do Rio de Janeiro: Rio de Janeiro, Brazil, 2010. [Google Scholar]
- Lima, E.C.; Cardoso, M.H. Bebida de soja (Glycine Max) e acerola (Malpighia Punicifolia) enriquecida com cálcio. Alim. Nutr Araraquara 2012, 23, 549–553. [Google Scholar]
- Brasil Ministério da Saúde. Agência Nacional De Vigilância Sanitária. Resolução RDC nº 269, 22 de setembro de 2005; Regulamento técnico sobre a ingestão diária recomendada (IDR) de proteína, vitaminas e minerais; Ministério da Saúde: Brasília (DF), 2005.
- Alyami, H.; Dahmash, E.; Bowen, J.; Mohammed, A.R. An investigation into the effects of excipient particle size, blending techniques and processing parameters on the homogeneity and content uniformity of a blend containing low-dose model drug. PLoS ONE 2017, 12, e0178772. [Google Scholar] [CrossRef] [PubMed]
- Meilgaard, M.C.; Civille, G.V.; Carr, B.T. Sensory Evaluation Techniques, 3rd ed.; CRC Press: Boca Raton, FL, USA, 1999; 387p. [Google Scholar]
- Choi, S.E. Sensory Evaluation. In Food Science: An Ecological Approach, 1st ed.; Edelstein, S., Ed.; Jones and Bartlett Learning LLC: Burlington, MA, USA, 2014. [Google Scholar]
- Wrobel, K. Determination of total aluminum, chromium, copper, iron, manganese, and nickel and their fractions leached to the infusions of black tea, green tea, Hibiscus sabdariffa, and Ilex paraguariensis (mate) by ETA-AAS. Biol. Trace Elem. Res. 2000, 78, 271–280. [Google Scholar] [CrossRef]
- Mehansho, H. Iron fortification technology development: New approaches. J. Nutr. 2006, 136, 1059–1063. [Google Scholar] [CrossRef] [PubMed]
- Sampaio, C.R.; Ferreira, S.M.; Canniatti-Brazaca, S.G. Perfil sensorial e aceitabilidade de barras de cereais fortificadas com ferro. Alim. Nutr. 2009, 20, 95–106. [Google Scholar]
- Sousa, C.; Fernandes, B.C.; Fernandes, P.H. Characterization of lactic drink pasteurized with added iron. Rev. Teccen 2015, 8, 1–32. [Google Scholar]
- Donangelo, C.M. Minerals. In Coffee: Production, Quality and Chemistry; Farah, A., Ed.; Royal Society of Chemistry: London, UK, 2019; pp. 505–516. [Google Scholar]
- Gogoasa, L.; Pirvu, A.; Alda, M.; Ariana, V.; Maria, R.; Maria, B.D.; Diana, M.; Simion, B.; Gergen, L. The mineral contentofdifferentcoffeebrands. J. HFB 2013, 17, 68–71. [Google Scholar]
- Farnezi, M.M.; Silva, E.B.; Guimarães, P.T. Nutritional diagnosis of coffee plantations in the Upper Jequitinhonha Valley, Minas Gerais State, Brazil: DRIs norms and critical nutrient ranges. Revista Brasileira de Ciência do Solo 2009, 33, 969–978. [Google Scholar] [CrossRef]
- Farah, A. Coffee Constituents. In Coffee: Emerging Health Effects and Disease Prevention, 1st ed.; Chu, Y., Ed.; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2012; pp. 21–57. [Google Scholar]
- TACO—Tabela Brasileira de Composição de Alimentos/NEPA—UNICAMP, 4th ed.NEPA-UNICAMP: Campinas, Brazil, 2011.
- Malik, J.; Szakova, J.; Drabek, O.; Balik, J.; Kokoska, L. Determination of certain micro and macro elements in plant stimulants and their infusions. Food Chem. 2008, 111, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Gillies, M.E.; Birkbeck, J.A. Tea and coffee as sources of some minerals in the New Zealand diet. Am. J. Clin. Nutr. 1983, 38, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Flaten, T.P. Aluminium in tea—Concentrations, speciation and bioavailability. Coord. Chem. Rev. 2002, 228, 385–395. [Google Scholar] [CrossRef]
- Food and Agriculture Organization. Ferrous glycinate (processed with citric acid). Chem. Tech. Assess. 2004. [Google Scholar]
- Drago, S.R.; Valencia, M.E. Effect of fermentation on iron, zinc, and calcium availability from iron-fortified dairy products. J. Food Sci. 2002, 67, 3130–3134. [Google Scholar] [CrossRef]
- Osman, A.K.; Al-Othaimeen, A. Experience with ferrous bis-glycinechelate as an iron fortificant in milk. Int. J. Nutr. Res. 2002, 72, 257–263. [Google Scholar] [CrossRef]
- Marchi, R.P.; Szarfarc, S.C.; Rodrigues, J.E. Consumption of fortified rice in prophylaxis of iron deficiency. Nutrire 2004, 28, 53–64. [Google Scholar]
- Cecafé, Brazilian Coffee Exporters Council. Available online: Https://www.cecafe.com.br/en/about-coffee/consumption/ (accessed on 9 October 2018).
- USDA Nutrient Database for Standard Reference. US Department of Agriculture, Agricultural Research Service. Nutrient Data Laboratory Home Page. Available online: http://www.nal.usda. gov/fnic/foodcomp (accessed on 19 November 2018).
- Mazariegos, M.; Hambidge, K.M.; Krebs, N.F.; Westcott, J.E.; Lei, S.; Grunwald, G.K. Zinc absorption in Guatemalan school children fed normal or low-phytate maize. Am. J. Clin. Nutr. 2006, 83, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Brune, M.; Rossander, L.; Hallberg, L. Iron absorption and phenolic compounds: Importance of different phenolic structures. Eur. J. Clin. Nutr. 1989, 43, 547–557. [Google Scholar] [PubMed]
- Lohmann, P. Minerais na alimentação. Food Ingred. Brasil 2008, 4, 48–65. [Google Scholar]
- Brown, K.H.; Hambidge, M.; Ranum, P. Zinc fortification of cereal flours: Current recommendations and research needs. Food Nutr. Bull. 2010, 31, S62–S74. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.B. Desenvolvimento de Produtos Alimentares Adicionados de ferro, Cálcio, Zinco e Carotenoides (Alfacaroteno e Betacaroteno) Como Proposta de Alimentos Enriquecidos ou Fontes Destes Nutrientes [Dissertação de Mestrado]; Universidade Federal Rural do Rio de Janeiro: Rio de Janeiro, Brazil, 2008. [Google Scholar]
- Araujo, M.C.; Bezerra, I.N.; Barbosa, F.S.; Junger, W.L.; Yokoo, E.M.; Pereira, R.A.; Sichieri, R. Macronutrient consumption and inadequate micronutrient intake in adults. Revista Saúde Pública 2013, 47, 177s–189s. [Google Scholar] [CrossRef]
- World Health Organization. World Health Report; WHO: Geneva, Switzerland, 2013. Available online: https://www.who.int/gho/publications/world_health_statistics/EN_WHS2013_Full.pdf (accessed on 10 November 2018).
- Gerstner, G. How to fortify beverages with calcium. Food Market. Technol. 2003, 10, 16–19. [Google Scholar]
- Elian, M.; Srianta, I.; Trisnawati, C.; Arisasmita, J.H. Effects of calcium fortification (calcium lactate gluconate) on the physicochemical and sensory properties of soy-corn milk. Int. J. Food Nutr. Public Health 2012, 5, 91–104. [Google Scholar]
- Haro, J.F.; Martínez, C.; Ros, G.; Vidal, M.L. Stability of calcium bioaccessibility and sensory parameters during the storage of fortified juices. Food Sci. Technol. Int. 2006, 12, 281–285. [Google Scholar] [CrossRef]
- Singh, G.; Mukumarappan, K. Influence of calcium fortification on sensory, physical and rheological characteristics of fruit yogurt. Food Sci. Technol. 2008, 41, 1145–1152. [Google Scholar] [CrossRef]
- Viñas, B.R.; Barba, L.R.; Ngo, J.; Gurinovic, M.; Novakovic, R.; Cavelaars, A.; de Groot, L.C.P.G.M.; van’t Veer, P.; Matthys, C.; Majem, L.S. Prevalence of Inadequate Nutrient Intakes in Europe. Ann. Nutr. Metab. 2011, 59, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Nawrot, P.; Jordan, S.; Eastwood, J.; Rotstein, J.; Hugenholtz, A.; Feeley, M. Effects of caffeine on human health. Food Addit. Contam. 2003, 20, 1–30. [Google Scholar] [CrossRef] [PubMed]
- De Paula-Lima, J.; Farah, A. Potential Negative Effects of Caffeine Consumption on Health. In Coffee: Consumption and Health Implications; Farah, A., Ed.; Royal Society of Chemistry: London, UK, 2019; pp. 493–512. [Google Scholar]
- Otten, J.; Hellwig, J.P.; Meyers, L.D. (Eds.) The National Academy of Sciences, Dietary Reference Intakes: The Essential Guide to Nutrient Requirements; National Academy of Sciences: Washington, D.C., WA, USA, 2006. [Google Scholar]
- Padovani, R.M.; Amaya-Farfán, J.; Colugnati, F.A.; Domene, S.M. Dietary reference intakes: Application of tables in nutritional studies. Rev. Nutr. 2006, 19, 741–760. [Google Scholar] [CrossRef]

| SALT & Molecular Formula | Ranking | Attributes in Aqueous Solution | Attributes in Coffee Brew | Status |
|---|---|---|---|---|
| Ferrous | 1 | Slight metallic taste | Metallic taste | Passed |
| bisglycinate | ||||
| chelate | ||||
| C4H8FeN2O4 | ||||
| Ferrous sulfate | 2 | Intense metallic taste | Intense metallic taste | Passed |
| FeSO4 | ||||
| Ferrous fumarate | 3 | Intense metallic taste | Intense metallic taste | Eliminated |
| C4H2FeO4 | Astringency | |||
| 1 | Slight astringency | Slight astringency | Passed | |
| Zinc lactate | Neutral | |||
| C6H10O6Zn | ||||
| 2 | Very salty taste | Slightly salty | Passed | |
| Zinc bisglycinate | Slight residual | Residual astringency | ||
| chelate | astringency | |||
| C4H8N2O4Zn | Neutral | |||
| Zinc sulfate | 3 | Unpleasant metallic | Intense metallic taste | Eliminated |
| Heptahydrate | taste | |||
| ZnSO4·7H2O | Bitter taste | |||
| Calcium lactate | 1 | Neutral taste | Slight | Passed |
| C6H10CaO6 | Paper taste | calcareous taste | ||
| Slight astringency | ||||
| Tricalcium fosfate | 2 | Neutral taste | Slight residual astringency | Passed |
| Ca3(PO4)2 | Slight residual astringency | |||
| Calcium citrate | 3 | Intense astringency | Intense astringency | Eliminated |
| Ca3(C6H5O7)2 | Significant residual astringency | Intense residual astringency |
| Mineral Content (mg/100 g) | ||||||
|---|---|---|---|---|---|---|
| Unfortified Ground Roasted Coffees (Controls) | Fe | Zn | Ca | |||
| C. arabica | 5.60 ± 0.01 a | 0.83 ± 0.02 a | 132.82 ± 0.03 b | |||
| C. canephora | 5.75 ± 0.02 b | 0.82 ± 0.03 a | 130.66 ± 0.02 a | |||
| Fortified ground roasted coffees | Fe + ferrous Sulfate | Fe + ferrous bisglycinate | Zn + zinc bisglycinate | Zn + zinc Lactate | Ca +tricalcium phosphate | Ca + calcium lactate |
| C. arabica | 7.72 ± 0.03 a | 7.73 ± 0.03 a | 1.85 ± 0.02 a | 1.84 ± 0.01 a | 282.51 ± 0.0 5 a | 283.27 ± 0.05 a |
| C. canephora | 7.76 ± 0.02 a | 7.77 ± 0.01 a | 1.82 ± 0.09 a | 1.83 ± 0.03 a | 281.82 ± 0.05 b | 282.15 ± 0.04 b |
| Mineral Concentrations in Brews (Unfortified Coffees) | Mineral Concentrations in Brews from Fortified Coffees | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| (C. arabica) | (C. arabica) | ||||||||
| Brewing method | Fe | Zn | Ca | Fe + ferrous bisglycinate | Fe + ferrous sulfate | Zn + zinc bisglycinate | Zn + zinc Lactate | Ca + tricalcium Phosphate | Ca + calcium lactate |
| Electric dripper (paper filter) | 0.032 ± 0.001 a | 0.026 ± 0.001 a | 3.719 ± 0.002 a | 0.492 ± 0.001 a | 0.463 ± 0.001 a | 0.095 ± 0.001 a | 0.145 ± 0.002 a | 13.159 ± 0.001 a | 22.200 ± 0.002 a |
| Electric dripper (nylon filter) | 0.027 ± 0.001 b | 0.028 ± 0.000 a | 3.513 ± 0.000 b | 0.484 ± 0.000 b | 0.452 ± 0.000 b | 0.097 ± 0.000 a | 0.136 ± 0.000 b | 11.970 ± 0.000 b | 22.100 ± 0.000 b |
| Espresso | 0.101 ± 0.002 c | 0.038 ± 0.000 b | 7.407 ± 0.001 c | 0.765 ± 0.001 c | 0.679 ± 0.001 c | 0.190 ± 0.001 b | 0.225 ± 0.001 b | 20.790 ± 0.000 c | 31.200 ± 0.001 c |
| (C. canephora) | (C. canephora) | ||||||||
| Brewing method | Fe | Zn | Ca | Fe + ferrous bisglycinate | Fe + ferrous sulfate | Zn + zinc bisglycinate | Zn + zinc lactate | Ca + tricalcium phosphate | Ca + calcium lactate |
| Electric dripper (paper filter) | 0.035 ± 0.003 a | 0.029 ± 0.001 a | 3.849 ± 0.002 a | 0.535 ± 0.001 a | 0.504 ± 0.001 a | 0.100 ± 0.001 a | 0.145 ± 0.000 a | 12.419 ± 0.000 a | 22.549 ± 0.001 a |
| Electric dripper (nylon filter) | 0.035 ± 0.000 a | 0.028 ± 0.001 b | 3.506 ± 0.002 b | 0.507 ± 0.000 b | 0.459 ± 0.000 b | 0.098 ± 0.000 a | 0.131 ± 0.000 b | 13.260 ± 0.000 b | 22.300 ± 0.000 b |
| Espresso | 0.125 ± 0.002 b | 0.038 ± 0.002 c | 6.990 ± 0.000 c | 0.831 ± 0.000 c | 0.753 ± 0.001 c | 0.183 ± 0.002 b | 0.216 ± 0.000 c | 20.780 ± 0.000 c | 31.050 ± 0.000 c |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soares, A.; Barros, N.M.; D Saint’Pierre, T.; De P. Lima, J.; Calado, V.; Donangelo, C.M.; Farah, A. Fortification of Ground Roasted Coffees with Iron, Zinc, and Calcium Salts: Evaluation of Minerals Recovery in Filtered and Espresso Brews. Beverages 2019, 5, 4. https://doi.org/10.3390/beverages5010004
Soares A, Barros NM, D Saint’Pierre T, De P. Lima J, Calado V, Donangelo CM, Farah A. Fortification of Ground Roasted Coffees with Iron, Zinc, and Calcium Salts: Evaluation of Minerals Recovery in Filtered and Espresso Brews. Beverages. 2019; 5(1):4. https://doi.org/10.3390/beverages5010004
Chicago/Turabian StyleSoares, Angela, Nathalia M. Barros, Tatiana D Saint’Pierre, Juliana De P. Lima, Verônica Calado, Carmen. M Donangelo, and Adriana Farah. 2019. "Fortification of Ground Roasted Coffees with Iron, Zinc, and Calcium Salts: Evaluation of Minerals Recovery in Filtered and Espresso Brews" Beverages 5, no. 1: 4. https://doi.org/10.3390/beverages5010004
APA StyleSoares, A., Barros, N. M., D Saint’Pierre, T., De P. Lima, J., Calado, V., Donangelo, C. M., & Farah, A. (2019). Fortification of Ground Roasted Coffees with Iron, Zinc, and Calcium Salts: Evaluation of Minerals Recovery in Filtered and Espresso Brews. Beverages, 5(1), 4. https://doi.org/10.3390/beverages5010004






