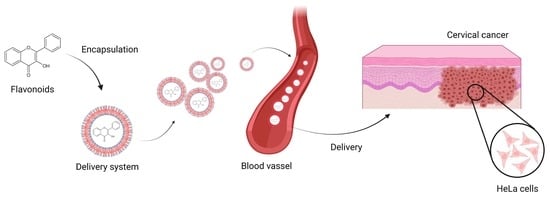
Flavonoids-Based Delivery Systems towards Cancer Therapies
Abstract
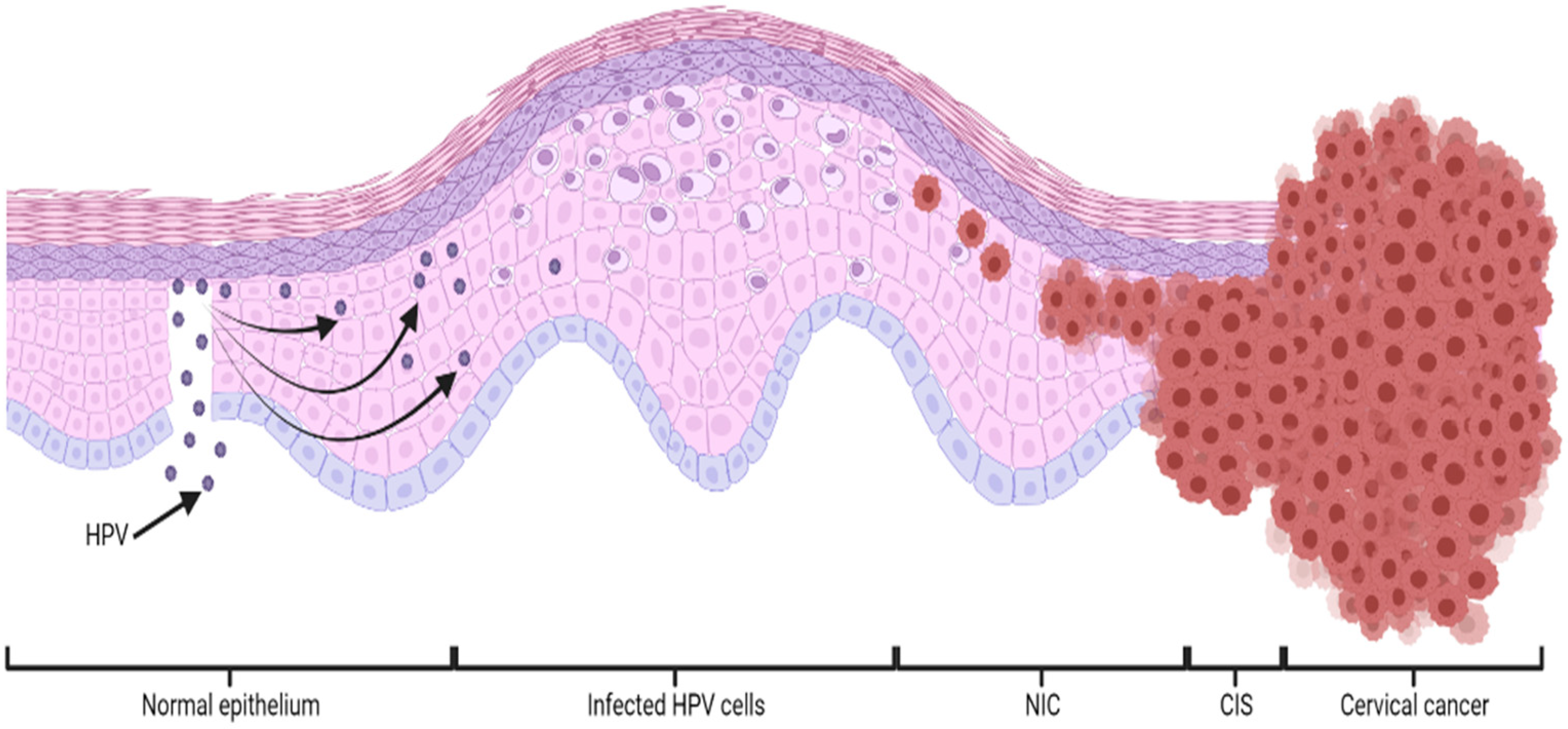
1. Cervical Cancer
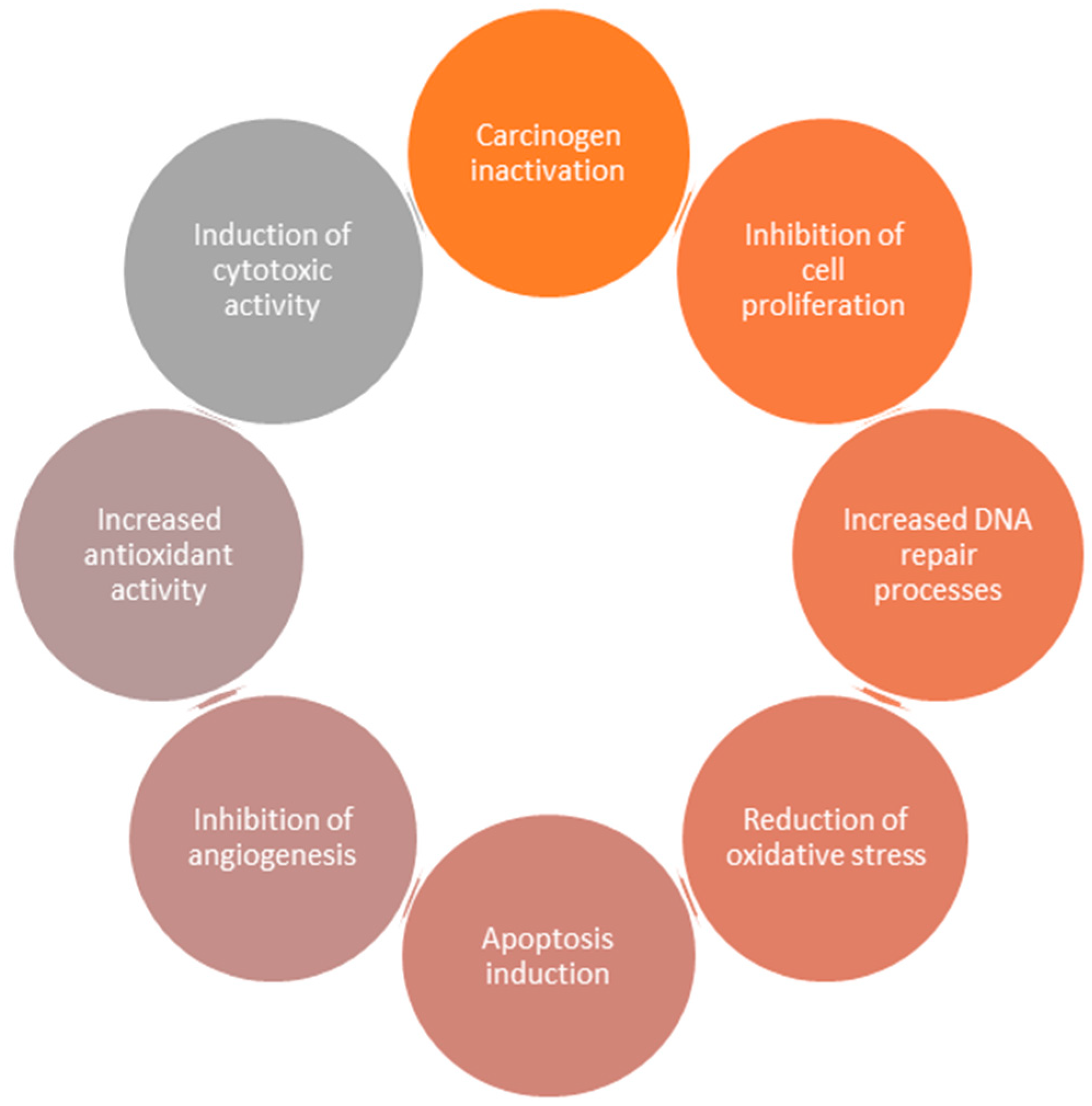
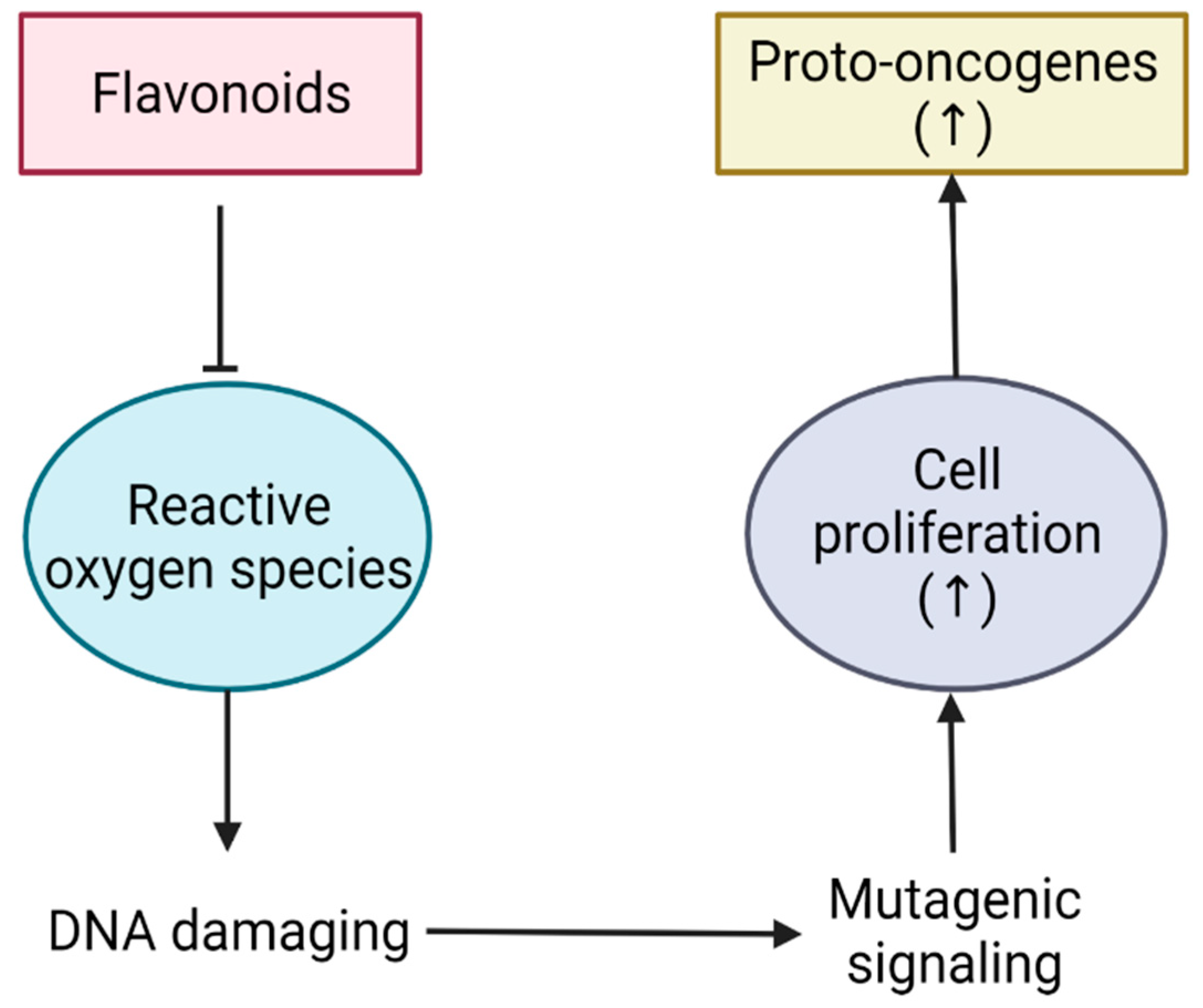
2. Flavonoids
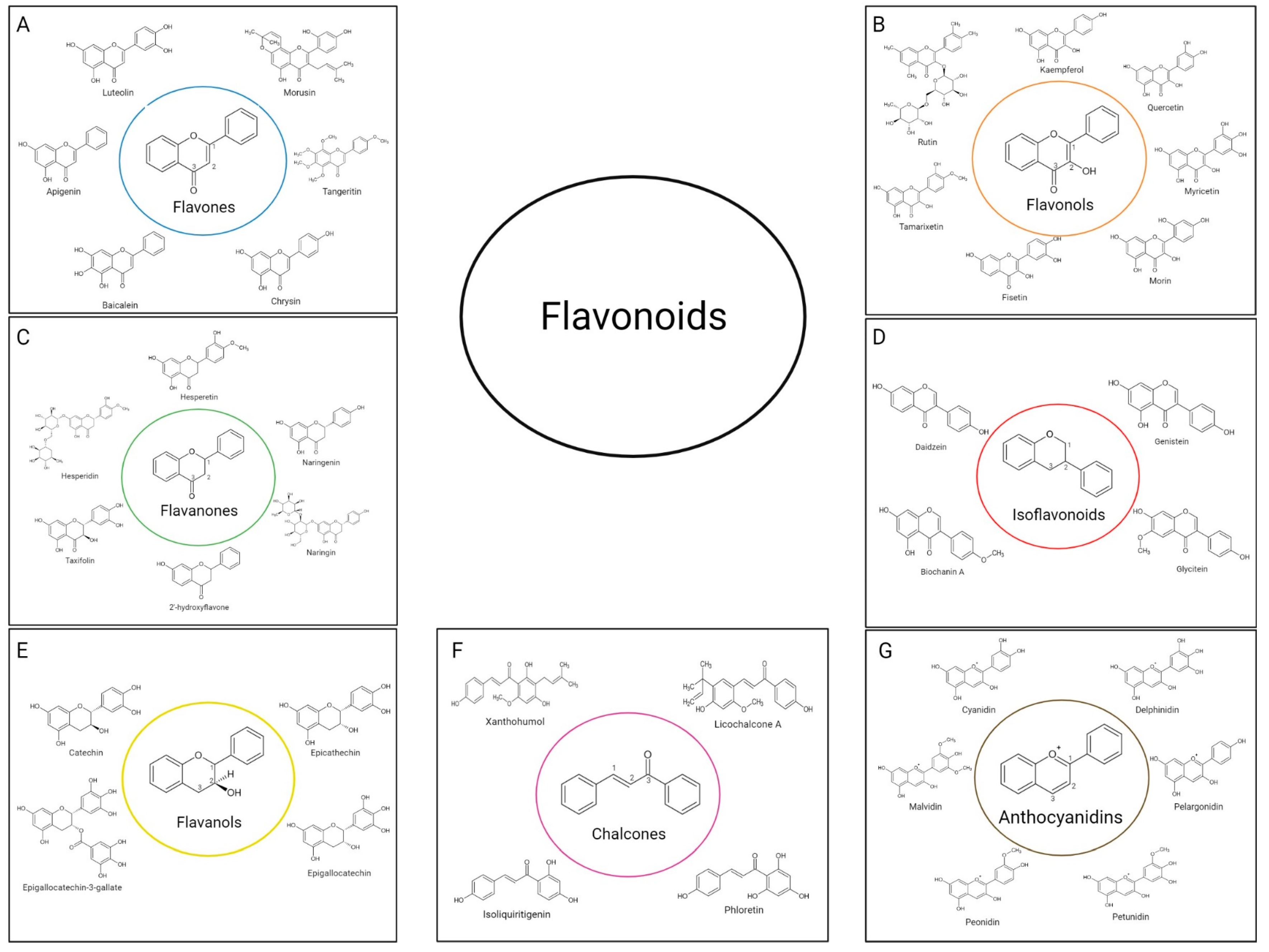
2.1. Flavonoids Subgroups
2.1.1. Flavones
2.1.2. Flavonols
2.1.3. Flavanones
2.1.4. Isoflavonoids
2.1.5. Flavanols (Flavan-3-ols)
2.1.6. Chalcones
2.1.7. Anthocyanidins
2.2. Bioavailability
3. Delivery Systems
3.1. Lipid-Based Delivery System
3.1.1. Liposomes
3.1.2. Lipid-Based Nanoparticles
3.1.3. Emulsions and Nanoemulsions
3.2. Polymer-Based Nanoparticles
3.2.1. Natural Polymers
3.2.2. Synthetic Polymers
3.2.3. Inorganic Polymers
3.3. Micelles
3.4. Inclusion Complexes
3.5. Other Types of Delivery Systems
4. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). GLOBOCAN 2020—The Global Cancer Observatory: Cancer Today—All Cancers; World Health Organization (WHO) International Agency for Research on Cancer: Lion, France, 2020; Available online: https://gco.iarc.fr/today/data/factsheets/cancers/39-All-cancers-fact-sheet.pdf (accessed on 16 December 2021).
- World Health Organization (WHO). GLOBOCAN 2020—The Global Cancer Observatory—Cancer Today Low Income; World Health Organization (WHO) International Agency for Research on Cancer: Lion, France, 2020; Available online: https://gco.iarc.fr/today/data/factsheets/populations/989-low-income-fact-sheets.pdf (accessed on 20 December 2021).
- Almeida, A.M.; Queiroz, J.A.; Sousa, F.; Sousa, Â. Cervical cancer and HPV infection: Ongoing therapeutic research to counteract the action of E6 and E7 oncoproteins. Drug Discov. Today 2019, 24, 2044–2057. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). GLOBOCAN 2020—The Global Observatory: Cancer Today—Cervix Uteri; World Health Organization (WHO) International Agency for Research on Cancer: Lion, France, 2020; Available online: https://gco.iarc.fr/today/data/factsheets/cancers/23-Cervix-uteri-fact-sheet.pdf (accessed on 16 December 2021).
- Lee, S.-J.; Yang, A.; Wu, T.-C.; Hung, C.-F. Immunotherapy for human papillomavirus-associated disease and cervical cancer: Review of clinical and translational research. J. Gynecol. Oncol. 2016, 27, e51. [Google Scholar] [CrossRef]
- Choi, Y.J.; Park, J.S. Clinical significance of human papillomavirus genotyping. J. Gynecol. Oncol. 2016, 27, e21. [Google Scholar] [CrossRef] [PubMed]
- Gomes, D.; Silvestre, S.; Duarte, A.P.; Venuti, A.; Soares, C.P.; Passarinha, L.; Sousa, Â. In silico approaches: A way to unveil novel therapeutic drugs for cervical cancer management. Pharmaceuticals 2021, 14, 741. [Google Scholar] [CrossRef] [PubMed]
- Franconi, R.; Massa, S.; Paolini, F.; Vici, P.; Venuti, A. Plant-derived natural compounds in genetic vaccination and therapy for HPV-associated cancers. Cancers 2020, 12, 3101. [Google Scholar] [CrossRef]
- Clemente-Soto, A.F.; Salas-Vidal, E.; Millan-Pacheco, C.; Sánchez-Carranza, J.N.; Peralta-Zaragoza, O.; González-Maya, L. Quercetin induces G2 phase arrest and apoptosis with the activation of p53 in an E6 expression-independent manner in HPV-positive human cervical cancer-derived cells. Mol. Med. Rep. 2019, 19, 2097–2106. [Google Scholar] [CrossRef]
- Yousefi, M.; Shadnoush, M.; Sohrabvandi, S.; Khorshidian, N.; Mortazavian, A.M. Encapsulation systems for delivery of flavonoids: A review. Biointerface Res. Appl. Chem. 2021, 11, 13934–13951. [Google Scholar] [CrossRef]
- Lesjak, M.; Beara, I.; Simin, N.; Pintać, D.; Majkić, T.; Bekvalac, K.; Orčić, D.; Mimica-Dukić, N. Antioxidant and anti-inflammatory activities of quercetin and its derivatives. J. Funct. Foods 2018, 40, 68–75. [Google Scholar] [CrossRef]
- Aziz, N.; Kim, M.-Y.; Cho, J.Y. Anti-inflammatory effects of luteolin: A review of in vitro, in vivo, and in silico studies. J. Ethnopharmacol. 2018, 225, 342–358. [Google Scholar] [CrossRef]
- Orhan, D.D.; Özçelik, B.; Özgen, S.; Ergun, F. Antibacterial, antifungal, and antiviral activities of some flavonoids. Microbiol. Res. 2010, 165, 496–504. [Google Scholar] [CrossRef]
- Madunić, J.; Madunić, I.V.; Gajski, G.; Popić, J.; Garaj-Vrhovac, V. Apigenin: A dietary flavonoid with diverse anticancer properties. Cancer Lett. 2018, 413, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Dobrzynska, M.; Napierala, M.; Florek, E. Flavonoid nanoparticles: A promising approach for cancer therapy. Biomolecules 2020, 10, 1268. [Google Scholar] [CrossRef]
- Imran, M.; Rauf, A.; Abu-Izneid, T.; Nadeem, M.; Shariati, M.A.; Khan, I.A.; Imran, A.; Orhan, I.E.; Rizwan, M.; Atif, M.; et al. Luteolin, a flavonoid, as an anticancer agent: A review. Biomed. Pharmacother. 2019, 112, 108612. [Google Scholar] [CrossRef] [PubMed]
- Veeramuthu, D.; Raja, W.R.T.; Al-Dhabi, N.A.; Savarimuthu, I. Flavonoids: Anticancer properties. In Flavonoids—From Biosynthesis to Human Health; IntechOpen: London, UK, 2017. [Google Scholar]
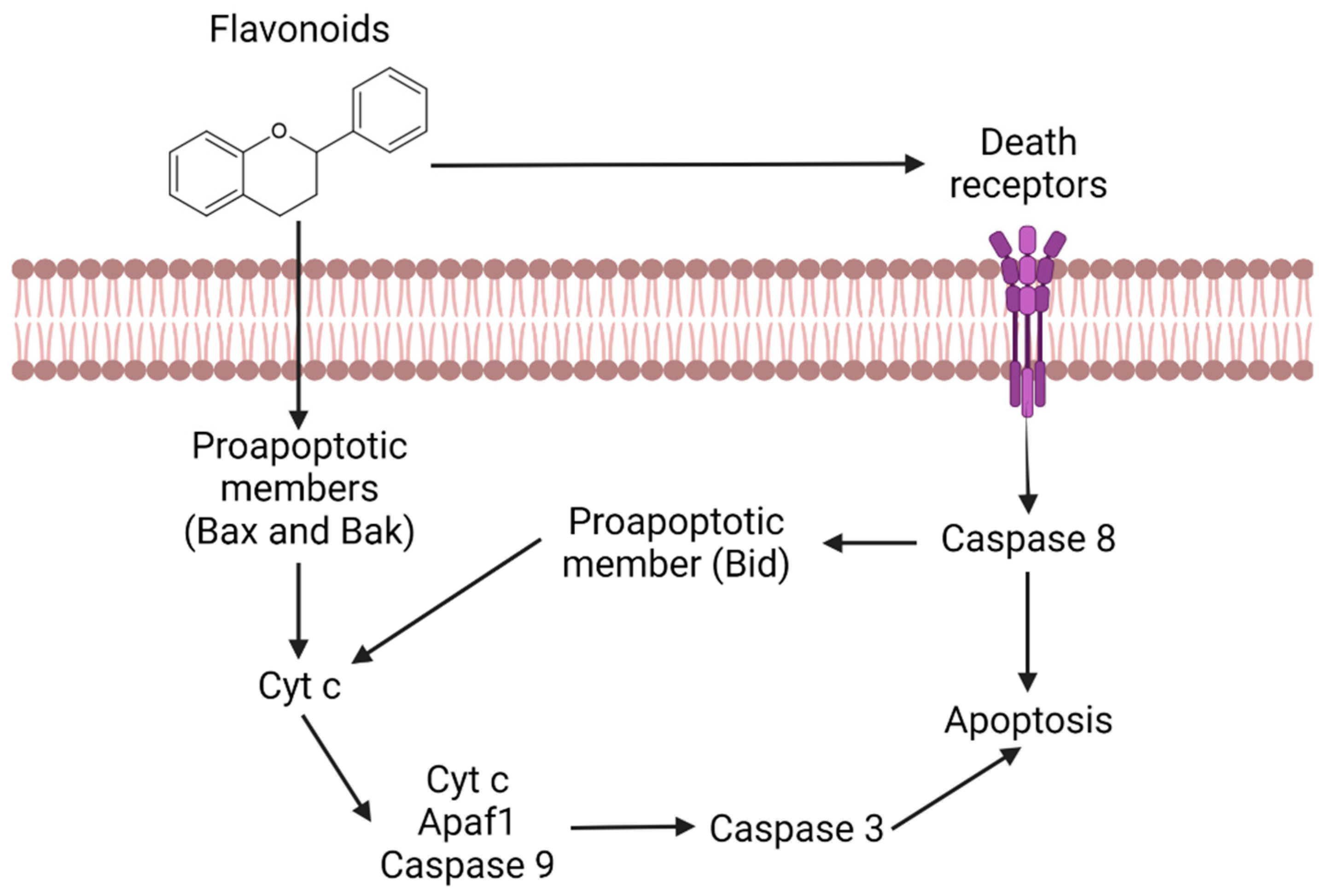
- Ramos, S. Effects of dietary flavonoids on apoptotic pathways related to cancer chemoprevention. J. Nutr. Biochem. 2007, 18, 427–442. [Google Scholar] [CrossRef]
- Cahyana, Y.; Adiyanti, T. Review: Flavonoids as Antidiabetic Agents. Indones. J. Chem. 2021, 21, 512–526. [Google Scholar] [CrossRef]
- Najafi, M.; Tavakol, S.; Zarrabi, A.; Ashrafizadeh, M. Dual role of quercetin in enhancing the efficacy of cisplatin in chemotherapy and protection against its side effects: A review. Arch. Physiol. Biochem. 2020, 1–15. [Google Scholar] [CrossRef]
- Siddiqui, M.; Abdellatif, B.; Zhai, K.; Liskova, A.; Kubatka, P.; Büsselberg, D. Flavonoids alleviate peripheral neuropathy induced by anticancer drugs. Cancers 2021, 13, 1576. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Hortal, M.D.; Varela-López, A.; Romero-Márquez, J.M.; Rivas-García, L.; Speranza, L.; Battino, M.; Quiles, J.L. Role of flavonoids against adriamycin toxicity. Food Chem. Toxicol. 2020, 146, 111820. [Google Scholar] [CrossRef]
- Gibellini, L.; Pinti, M.; Nasi, M.; Montagna, J.P.; De Biasi, S.; Roat, E.; Bertoncelli, L.; Cooper, E.L.; Cossarizza, A. Quercetin and cancer chemoprevention. Evid. Based Complement. Altern. Med. 2011, 2011, 591356. [Google Scholar] [CrossRef]
- Seleem, D.; Pardi, V.; Murata, R.M. Review of flavonoids: A diverse group of natural compounds with anti-Candida albicans activity in vitro. Arch. Oral Biol. 2017, 76, 76–83. [Google Scholar] [CrossRef]
- Liskova, A.; Samec, M.; Koklesova, L.; Brockmueller, A.; Zhai, K.; Abdellatif, B.; Siddiqui, M.; Biringer, K.; Kudela, E.; Pec, M.; et al. Flavonoids as an effective sensitizer for anti-cancer therapy: Insights into multi-faceted mechanisms and applicability towards individualized patient profiles. EPMA J. 2021, 12, 155–176. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed]
- Baksi, R.; Singh, D.P.; Borse, S.P.; Rana, R.; Sharma, V.; Nivsarkar, M. In vitro and in vivo anticancer efficacy potential of Quercetin loaded polymeric nanoparticles. Biomed. Pharmacother. 2018, 106, 1513–1526. [Google Scholar] [CrossRef]
- Zhao, J.; Fang, Z.; Zha, Z.; Sun, Q.; Wang, H.; Sun, M.; Qiao, B. Quercetin inhibits cell viability, migration and invasion by regulating miR-16/HOXA10 axis in oral cancer. Eur. J. Pharmacol. 2019, 847, 11–18. [Google Scholar] [CrossRef]
- Hsiao, Y.-C.; Kuo, W.-H.; Chen, P.-N.; Chang, H.-R.; Lin, T.-H.; Yang, W.-E.; Hsieh, Y.-S.; Chu, S.-C. Flavanone and 2′-OH flavanone inhibit metastasis of lung cancer cells via down-regulation of proteinases activities and MAPK pathway. Chem. Biol. Interact. 2007, 167, 193–206. [Google Scholar] [CrossRef]
- Cayetano-Salazar, L.; Olea-Flores, M.; Zuñiga-Eulogio, M.D.; Weinstein-Oppenheimer, C.; Fernández-Tilapa, G.; Mendoza-Catalán, M.A.; Zacapala-Gómez, A.E.; Ortiz-Ortiz, J.; Ortuño-Pineda, C.; Navarro-Tito, N. Natural isoflavonoids in invasive cancer therapy: From bench to bedside. Phytother. Res. 2021, 35, 4092–4110. [Google Scholar] [CrossRef]
- Miadoková, E. Isoflavonoids—An overview of their biological activities and potential health benefits. Interdiscip. Toxicol. 2009, 2, 211–218. [Google Scholar] [CrossRef]
- Heiss, C.; Keen, C.L.; Kelm, M. Flavanols and cardiovascular disease prevention. Eur. Heart J. 2010, 31, 2583–2592. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.K.A.; Fontenelle, R.O.S.; Magalhães, F.E.A.; Bandeira, P.N.; De Menezes, J.E.S.A.; dos Santos, H.S. Chalcones pharmacological potential: A brief review. Rev. Virtual Quim. 2018, 10, 1455–1473. [Google Scholar] [CrossRef]
- Kuber Banoth, R.; Thatikonda, A. A review on natural chalcones: An update. Int. J. Pharm. Sci. Res. 2020, 11, 546. [Google Scholar] [CrossRef]
- Kopustinskiene, D.M.; Jakstas, V.; Savickas, A.; Bernatoniene, J. Flavonoids as anticancer agents. Nutrients 2020, 12, 457. [Google Scholar] [CrossRef] [PubMed]
- Sinopoli, A.; Calogero, G.; Bartolotta, A. Computational aspects of anthocyanidins and anthocyanins: A review. Food Chem. 2019, 297, 124898. [Google Scholar] [CrossRef] [PubMed]
- Lima, B.D.S.; Shanmugam, S.; de Souza Siqueira Quintans, J.; Quintans-Júnior, L.J.; de Souza Araújo, A.A. Inclusion complex with cyclodextrins enhances the bioavailability of flavonoid compounds: A systematic review. Phytochem. Rev. 2019, 18, 1337–1359. [Google Scholar] [CrossRef]
- Thilakarathna, S.H.; Vasantha Rupasinghe, H.P. Flavonoid bioavailability and attempts for bioavailability enhancement. Nutrients 2013, 5, 3367–3387. [Google Scholar] [CrossRef]
- Ding, Y.; Tong, Z.; Jin, L.; Ye, B.; Zhou, J.; Sun, Z.; Yang, H.; Hong, L.; Huang, F.; Wang, W.; et al. An NIR discrete metallacycle constructed from perylene bisimide and tetraphenylethylene fluorophores for imaging-guided cancer radio-chemotherapy. Adv. Mater. 2022, 34, 2106388. [Google Scholar] [CrossRef]
- Zhou, J.; Yu, G.; Yang, J.; Shi, B.; Ye, B.; Wang, M.; Huang, F.; Stang, P.J. Polymeric nanoparticles integrated from discrete organoplatinum(II) metallacycle by stepwise post-assembly polymerization for synergistic cancer therapy. Chem. Mater. 2020, 32, 4564–4573. [Google Scholar] [CrossRef]
- Zhou, J.; Rao, L.; Yu, G.; Cook, T.R.; Chen, X.; Huang, F. Supramolecular cancer nanotheranostics. Chem. Soc. Rev. 2021, 50, 2839–2891. [Google Scholar] [CrossRef]
- Khan, H.; Ullah, H.; Martorell, M.; Valdes, S.E.; Belwal, T.; Tejada, S.; Sureda, A.; Kamal, M.A. Flavonoids nanoparticles in cancer: Treatment, prevention and clinical prospects. Semin. Cancer Biol. 2021, 69, 200–211. [Google Scholar] [CrossRef]
- Li, J.; Shi, M.; Ma, B.; Niu, R.; Zhang, H.; Kun, L. Antitumor activity and safety evaluation of nanaparticle-based delivery of quercetin through intravenous administration in mice. Mater. Sci. Eng. C 2017, 77, 803–810. [Google Scholar] [CrossRef]
- Li, J.; Li, Z.; Gao, Y.; Liu, S.; Li, K.; Wang, S.; Gao, L.; Shi, M.; Liu, Z.; Han, Z.; et al. Effect of a drug delivery system made of quercetin formulated into PEGylation liposomes on cervical carcinoma in vitro and in vivo. J. Nanomater. 2021, 2021, 9389934. [Google Scholar] [CrossRef]
- Saraswat, A.L.; Maher, T.J. Development and optimization of stealth liposomal system for enhanced in vitro cytotoxic effect of quercetin. J. Drug Deliv. Sci. Technol. 2020, 55, 101477. [Google Scholar] [CrossRef]
- Ding, B.; Chen, P.; Kong, Y.; Zhai, Y.; Pang, X.; Dou, J.; Zhai, G. Preparation and evaluation of folate-modified lipid nanocapsules for quercetin delivery. J. Drug Target. 2014, 22, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Zhang, H.; Gao, L.; Zhai, Y.; Shi, M.; Li, J.; Xiu, C.; Cao, J.; Cheng, S.; Jiang, L.; et al. Preparation and characterization of baicalein-loaded nanoliposomes for antitumor therapy. J. Nanomater. 2016, 2016, 2861915. [Google Scholar] [CrossRef]
- Ni, S.; Hu, C.; Sun, R.; Zhao, G.; Xia, Q. Nanoemulsions-based delivery systems for encapsulation of Quercetin: Preparation, characterization, and cytotoxicity studies. J. Food Process Eng. 2017, 40, e12374. [Google Scholar] [CrossRef]
- Rahimi, S.; Khoee, S.; Ghandi, M. Preparation and characterization of rod-like chitosan–quinoline nanoparticles as pH-responsive nanocarriers for quercetin delivery. Int. J. Biol. Macromol. 2019, 128, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Kunjiappan, S.; Panneerselvam, T.; Somasundaram, B.; Sankaranarayanan, M.; Chowdhury, R.; Chowdhury, A. Design, in silico modeling, biodistribution study of rutin and quercetin loaded stable human hair keratin nanoparticles intended for anticancer drug delivery. Biomed. Phys. Eng. Express 2018, 4, 025019. [Google Scholar] [CrossRef]
- Deepika, M.S.; Thangam, R.; Sheena, T.S.; Sasirekha, R.; Sivasubramanian, S.; Babu, M.D.; Jeganathan, K.; Thirumurugan, R. A novel rutin-fucoidan complex based phytotherapy for cervical cancer through achieving enhanced bioavailability and cancer cell apoptosis. Biomed. Pharmacother. 2019, 109, 1181–1195. [Google Scholar] [CrossRef]
- Fuster, M.G.; Carissimi, G.; Montalbán, M.G.; Víllora, G. Improving anticancer therapy with naringenin-loaded silk fibroin nanoparticles. Nanomaterials 2020, 10, 718. [Google Scholar] [CrossRef]
- Filho, I.K.; Machado, C.S.; Diedrich, C.; Karam, T.K.; Nakamura, C.V.; Khalil, N.M.; Mainardes, R.M. Optimized chitosan-coated gliadin nanoparticles improved the hesperidin cytotoxicity over tumor cells. Braz. Arch. Biol. Technol. 2021, 64, 200795. [Google Scholar] [CrossRef]
- Mei, L.; Zhang, H.; Zeng, X.; Huang, L.; Wang, Z.; Liu, G.; Wu, Y.; Yang, C. Fabrication of genistein-loaded biodegradable TPGS-b-PCL nanoparticles for improved therapeutic effects in cervical cancer cells. Int. J. Nanomed. 2015, 10, 2461–2473. [Google Scholar] [CrossRef]
- Van Thoai, D.; Nguyen, D.T.; Dang, L.H.; Nguyen, N.H.; Nguyen, V.T.; Doan, P.; Nguyen, B.T.; Van Thu, L.; Tung, N.N.; Quyen, T.N. Lipophilic effect of various pluronic-grafted gelatin copolymers on the quercetin delivery efficiency in these self-assembly nanogels. J. Polym. Res. 2020, 27, 369. [Google Scholar] [CrossRef]
- Elgogary, R.I.; Rubio, N.; Wang, J.T.-W.; Al-Jamal, W.T.; Bourgognon, M.; Kafa, H.; Naeem, M.; Klippstein, R.; Abbate, V.; Leroux, F.; et al. Polyethylene glycol conjugated polymeric nanocapsules for targeted delivery of quercetin to folate-expressing cancer cells in vitro and in vivo. ACS Nano 2014, 8, 1384–1401. [Google Scholar] [CrossRef] [PubMed]
- Ghafelehbashi, R.; Yaraki, M.T.; Saremi, L.H.; Lajevardi, A.; Haratian, M.; Astinchap, B.; Rashidi, A.M.; Moradian, R. A pH-responsive citric-acid/α-cyclodextrin-functionalized Fe3O4 nanoparticles as a nanocarrier for quercetin: An experimental and DFT study. Mater. Sci. Eng. C 2020, 109, 110597. [Google Scholar] [CrossRef] [PubMed]
- Payne, J.N.; Badwaik, V.D.; Waghwani, H.K.; Moolani, H.V.; Tockstein, S.; Thompson, D.H.; Dakshinamurthy, R. Development of dihydrochalcone-functionalized gold nanoparticles for augmented antineoplastic activity. Int. J. Nanomed. 2018, 13, 1917–1926. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Zhu, J.; Pan, Y.; Chen, Z.; Liang, H.; Liu, H.; Wang, H. Synthesis, cytotoxic activity, and DNA binding properties of copper (II) complexes with hesperetin, naringenin, and apigenin. Bioinorg. Chem. Appl. 2009, 2009, 347872. [Google Scholar] [CrossRef] [PubMed]
- Simon, A.T.; Dutta, D.; Chattopadhyay, A.; Ghosh, S.S. Quercetin-loaded luminescent hydroxyapatite nanoparticles for theranostic application in monolayer and spheroid cultures of cervical cancer cell line in vitro. ACS Appl. Bio Mater. 2021, 4, 4495–4506. [Google Scholar] [CrossRef] [PubMed]
- Mashhadi Malekzadeh, A.; Ramazani, A.; Tabatabaei Rezaei, S.J.; Niknejad, H. Design and construction of multifunctional hyperbranched polymers coated magnetite nanoparticles for both targeting magnetic resonance imaging and cancer therapy. J. Colloid Interface Sci. 2017, 490, 64–73. [Google Scholar] [CrossRef]
- Yamina, A.M.; Fizir, M.; Itatahine, A.; He, H.; Dramou, P. Preparation of multifunctional PEG-graft-halloysite nanotubes for controlled drug release, tumor cell targeting, and bio-imaging. Colloids Surf. B Biointerfaces 2018, 170, 322–329. [Google Scholar] [CrossRef]
- Akal, Z.; Alpsoy, L.; Baykal, A. Biomedical applications of SPION@APTES@PEG-folic acid@carboxylated quercetin nanodrug on various cancer cells. Appl. Surf. Sci. 2016, 378, 572–581. [Google Scholar] [CrossRef]
- Alpsoy, L.; Baykal, A.; Kurtan, U.; Ülker, Z. Synthesis and characterization of carboxylated luteolin (CL)-functionalized SPION. J. Supercond. Nov. Magn. 2017, 30, 2797–2804. [Google Scholar] [CrossRef]
- Yu, C.; Gao, C.; Lü, S.; Chen, C.; Huang, Y.; Liu, M. Redox-responsive shell-sheddable micelles self-assembled from amphiphilic chondroitin sulfate-cholesterol conjugates for triggered intracellular drug release. Chem. Eng. J. 2013, 228, 290–299. [Google Scholar] [CrossRef]
- Jeong, D.; Choi, J.M.; Choi, Y.; Jeong, K.; Cho, E.; Jung, S. Complexation of fisetin with novel cyclosophoroase dimer to improve solubility and bioavailability. Carbohydr. Polym. 2013, 97, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Sundararajan, M.; Thomas, P.A.; Venkadeswaran, K.; Jeganathan, K.; Geraldine, P. Synthesis and characterization of chrysin-loaded β-cyclodextrin-based nanosponges to enhance in-vitro solubility, photostability, drug release, antioxidant effects and antitumorous efficacy. J. Nanosci. Nanotechnol. 2017, 17, 8742–8751. [Google Scholar] [CrossRef]
- Lv, T.; Yu, T.; Fang, Y.; Zhang, S.; Jiang, M.; Zhang, H.; Zhang, Y.; Li, Z.; Chen, H.; Gao, Y. Role of generation on folic acid-modified poly(amidoamine) dendrimers for targeted delivery of baicalin to cancer cells. Mater. Sci. Eng. C 2017, 75, 182–190. [Google Scholar] [CrossRef]
- Wang, W.; Sun, C.; Mao, L.; Ma, P.; Liu, F.; Yang, J.; Gao, Y. The biological activities, chemical stability, metabolism and delivery systems of quercetin: A review. Trends Food Sci. Technol. 2016, 56, 21–38. [Google Scholar] [CrossRef]
- Caballero, S.; Li, Y.O.; McClements, D.J.; Davidov-Pardo, G. Encapsulation and delivery of bioactive citrus pomace polyphenols: A review. Crit. Rev. Food Sci. Nutr. 2021, 1–17. [Google Scholar] [CrossRef]
- Katopodi, A.; Detsi, A. Solid lipid nanoparticles and nanostructured lipid carriers of natural products as promising systems for their bioactivity enhancement: The case of essential oils and flavonoids. Colloids Surf. A Physicochem. Eng. Asp. 2021, 630, 127529. [Google Scholar] [CrossRef]
- Patel, G.; Thakur, N.S.; Kushwah, V.; Patil, M.D.; Nile, S.H.; Jain, S.; Banerjee, U.C.; Kai, G. Liposomal delivery of mycophenolic acid with quercetin for improved breast cancer therapy in SD rats. Front. Bioeng. Biotechnol. 2020, 8, 631. [Google Scholar] [CrossRef]
- Hu, J.; Wang, J.; Wang, G.; Yao, Z.; Dang, X. Pharmacokinetics and antitumor efficacy of DSPE-PEG2000 polymeric liposomes loaded with quercetin and temozolomide: Analysis of their effectiveness in enhancing the chemosensitization of drug-resistant glioma cells. Int. J. Mol. Med. 2016, 37, 690–702. [Google Scholar] [CrossRef]
- Rezaei-Sadabady, R.; Eidi, A.; Zarghami, N.; Barzegar, A. Intracellular ROS protection efficiency and free radical-scavenging activity of quercetin and quercetin-encapsulated liposomes. Artif. Cells Nanomed. Biotechnol. 2016, 44, 128–134. [Google Scholar] [CrossRef]
- Bazylińska, U.; Pucek, A.; Sowa, M.; Matczak-Jon, E.; Wilk, K.A. Engineering of phosphatidylcholine-based solid lipid nanocarriers for flavonoids delivery. Colloids Surf. A Physicochem. Eng. Asp. 2014, 460, 483–493. [Google Scholar] [CrossRef]
- Fathi, M.; Varshosaz, J.; Mohebbi, M.; Shahidi, F. Hesperetin-loaded solid lipid nanoparticles and nanostructure lipid carriers for food fortification: Preparation, characterization, and modeling. Food Bioprocess Technol. 2013, 6, 1464–1475. [Google Scholar] [CrossRef]
- Bose, S.; Du, Y.; Takhistov, P.; Michniak-Kohn, B. Formulation optimization and topical delivery of quercetin from solid lipid based nanosystems. Int. J. Pharm. 2013, 441, 56–66. [Google Scholar] [CrossRef]
- Scalia, S.; Haghi, M.; Losi, V.; Trotta, V.; Young, P.M.; Traini, D. Quercetin solid lipid microparticles: A flavonoid for inhalation lung delivery. Eur. J. Pharm. Sci. 2013, 49, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Nie, S.; Pan, X.; Zhang, R.; Fan, Z.; Wang, S. Quercetin-nanostructured lipid carriers: Characteristics and anti-breast cancer activities in vitro. Colloids Surf. B Biointerfaces 2014, 113, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, H.; Cui, H.; Zhang, F.; Zhao, L.; Liu, Y.; Meng, Q. Combined and targeted drugs delivery system for colorectal cancer treatment: Conatumumab decorated, reactive oxygen species sensitive irinotecan prodrug and quercetin co-loaded nanostructured lipid carriers. Drug Deliv. 2022, 29, 342–350. [Google Scholar] [CrossRef]
- Firoozeh, N.; Mahmoud, O.; Layasadat, K.; Mohammadreza, A.; Esrafil, M.; Ali, K. Effects of quercetin-loaded nanoparticles on MCF-7 human breast cancer cells. Medicina 2019, 55, 114. [Google Scholar]
- Yostawonkul, J.; Surassmo, S.; Iempridee, T.; Pimtong, W.; Suktham, K.; Sajomsang, W.; Gonil, P.; Ruktanonchai, U.R. Surface modification of nanostructure lipid carrier (NLC) by oleoyl-quaternized-chitosan as a mucoadhesive nanocarrier. Colloids Surf. B Biointerfaces 2017, 149, 301–311. [Google Scholar] [CrossRef]
- Dora, C.L.; Silva, L.F.C.; Mazzarino, L.; Siqueira, J.M.; Fernandes, D.; Pacheco, L.K.; Maioral, M.F.; Santos-Silva, M.C.; Baischl, A.L.M.; Assreuy, J.; et al. Oral delivery of a high quercetin payload nanosized emulsion: In vitro and in vivo activity against B16-F10 melanoma. J. Nanosci. Nanotechnol. 2016, 16, 1275–1281. [Google Scholar] [CrossRef]
- Qian, C.; McClements, D.J. Formation of nanoemulsions stabilized by model food-grade emulsifiers using high-pressure homogenization: Factors affecting particle size. Food Hydrocoll. 2011, 25, 1000–1008. [Google Scholar] [CrossRef]
- Ting, Y.; Chiou, Y.-S.; Pan, M.-H.; Ho, C.-T.; Huang, Q. In vitro and in vivo anti-cancer activity of tangeretin against colorectal cancer was enhanced by emulsion-based delivery system. J. Funct. Foods 2015, 15, 264–273. [Google Scholar] [CrossRef]
- Ragelle, H.; Crauste-Manciet, S.; Seguin, J.; Brossard, D.; Scherman, D.; Arnaud, P.; Chabot, G.G. Nanoemulsion formulation of fisetin improves bioavailability and antitumour activity in mice. Int. J. Pharm. 2012, 427, 452–459. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Bao, Y.; Ahire, J.; Chao, Y. Co-encapsulation of biodegradable nanoparticles with silicon quantum dots and quercetin for monitored delivery. Adv. Healthcare Mater. 2013, 2, 459–466. [Google Scholar] [CrossRef]
- Kim, E.S.; Kim, D.Y.; Lee, J.-S.; Lee, H.G. Quercetin delivery characteristics of chitosan nanoparticles prepared with different molecular weight polyanion cross-linkers. Carbohydr. Polym. 2021, 267, 118157. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Dutta, P.K.; Kumar, H.; Kureel, A.K.; Rai, A.K. Synthesis of chitin-glucan-aldehyde-quercetin conjugate and evaluation of anticancer and antioxidant activities. Carbohydr. Polym. 2018, 193, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Pedro, R.D.O.; Hoffmann, S.; Pereira, S.; Goycoolea, F.M.; Schmitt, C.C.; Neumann, M.G. Self-assembled amphiphilic chitosan nanoparticles for quercetin delivery to breast cancer cells. Eur. J. Pharm. Biopharm. 2018, 131, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.; Goycoolea, F.M. Chitosan/cyclodextrin/TPP nanoparticles loaded with quercetin as novel bacterial quorum sensing inhibitors. Molecules 2017, 22, 1975. [Google Scholar] [CrossRef]
- Krauland, A.H.; Alonso, M.J. Chitosan/cyclodextrin nanoparticles as macromolecular drug delivery system. Int. J. Pharm. 2007, 340, 134–142. [Google Scholar] [CrossRef]
- Fülöp, Z.; Saokham, P.; Loftsson, T. Sulfobutylether-β-cyclodextrin/chitosan nano- and microparticles and their physicochemical characteristics. Int. J. Pharm. 2014, 472, 282–287. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, M.; Zhang, Z.; Hu, X.; Hao, L.; Lin, Q.; Wang, S.; Jiang, W. Preparation and characterization of magnetic fluorescent microspheres for delivery of kaempferol. Mater. Technol. 2017, 32, 125–130. [Google Scholar] [CrossRef]
- Qureshi, W.A.; Zhao, R.; Wang, H.; Ji, T.; Ding, Y.; Ihsan, A.; Mujeeb, A.; Nie, G.; Zhao, Y. Co-delivery of doxorubicin and quercetin via mPEG–PLGA copolymer assembly for synergistic anti-tumor efficacy and reducing cardio-toxicity. Sci. Bull. 2016, 61, 1689–1698. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, C.; Tang, C.; Yin, C. Dual stimulus-responsive chitosan-based nanoparticles co-delivering doxorubicin and quercetin for cancer therapy. Mater. Lett. 2021, 305, 130826. [Google Scholar] [CrossRef]
- Konecsni, K.; Low, N.H.; Nickerson, M.T. Chitosan–tripolyphosphate submicron particles as the carrier of entrapped rutin. Food Chem. 2012, 134, 1775–1779. [Google Scholar] [CrossRef]
- Bi, F.; Yong, H.; Liu, J.; Zhang, X.; Shu, Y.; Liu, J. Development and characterization of chitosan and D-α-tocopheryl polyethylene glycol 1000 succinate composite films containing different flavones. Food Packag. Shelf Life 2020, 25, 100531. [Google Scholar] [CrossRef]
- Saha, C.; Kaushik, A.; Das, A.; Pal, S.; Majumder, D. Anthracycline drugs on modified surface of quercetin-loaded polymer nanoparticles: A dual drug delivery model for cancer treatment. PLoS ONE 2016, 11, e0155710. [Google Scholar] [CrossRef]
- Karthick, V.; Panda, S.; Kumar, V.G.; Kumar, D.; Shrestha, L.K.; Ariga, K.; Vasanth, K.; Chinnathambi, S.; Dhas, T.S.; Suganya, K.S.U. Quercetin loaded PLGA microspheres induce apoptosis in breast cancer cells. Appl. Surf. Sci. 2019, 487, 211–217. [Google Scholar] [CrossRef]
- Sunoqrot, S.; Abujamous, L. pH-sensitive polymeric nanoparticles of quercetin as a potential colon cancer-targeted nanomedicine. J. Drug Deliv. Sci. Technol. 2019, 52, 670–676. [Google Scholar] [CrossRef]
- Pandey, S.K.; Patel, D.K.; Thakur, R.; Mishra, D.P.; Maiti, P.; Haldar, C. Anti-cancer evaluation of quercetin embedded PLA nanoparticles synthesized by emulsified nanoprecipitation. Int. J. Biol. Macromol. 2015, 75, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Sheu, M.-T.; Chen, L.-C.; Chen, Y.-C.; Su, C.-Y.; Hong, C.-S.; Ho, H.-O. Development and characterization of self-assembling lecithin-based mixed polymeric micelles containing quercetin in cancer treatment and an in vivo pharmacokinetic study. Int. J. Nanomed. 2016, 11, 1557–1566. [Google Scholar] [CrossRef]
- Patra, A.; Satpathy, S.; Shenoy, A.K.; Bush, J.A.; Kazi, M.; Hussain, M.D. Formulation and evaluation of mixed polymeric micelles of quercetin for treatment of breast, ovarian, and multidrug resistant cancers. Int. J. Nanomed. 2018, 13, 2869–2881. [Google Scholar] [CrossRef]
- Cote, B.; Carlson, L.J.; Rao, D.A.; Alani, A.W.G. Combinatorial resveratrol and quercetin polymeric micelles mitigate doxorubicin induced cardiotoxicity in vitro and in vivo. J. Control. Release 2015, 213, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.-H.; Yuan, L.; Meng, L.-Y.; Qiu, J.-L.; Wang, C.-B. Quercetin-loaded mixed micelles exhibit enhanced cytotoxic efficacy in non-small cell lung cancer in vitro. Exp. Ther. Med. 2017, 14, 5503–5508. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Khonkarn, R.; Mankhetkorn, S.; Hennink, W.E.; Okonogi, S. PEG-OCL micelles for quercetin solubilization and inhibition of cancer cell growth. Eur. J. Pharm. Biopharm. 2011, 79, 268–275. [Google Scholar] [CrossRef]
- Gao, X.; Huang, N.; Shi, H.; Ren, L.; Xu, G.; Gou, H.; Gong, D. Enhancing the anti-colon cancer activity of quercetin by self-assembled micelles. Int. J. Nanomed. 2015, 10, 2051–2063. [Google Scholar] [CrossRef]
- Trapani, A.; Lopedota, A.; Franco, M.; Cioffi, N.; Ieva, E.; Garcia-Fuentes, M.; Alonso, M.J. A comparative study of chitosan and chitosan/cyclodextrin nanoparticles as potential carriers for the oral delivery of small peptides. Eur. J. Pharm. Biopharm. 2010, 75, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Trapani, A.; Garcia-Fuentes, M.; Alonso, M.J. Novel drug nanocarriers combining hydrophilic cyclodextrins and chitosan. Nanotechnology 2008, 19, 185101. [Google Scholar] [CrossRef]
- Zhu, P.; Chen, L.; Zhao, Y.; Gao, C.; Yang, J.; Liao, X.; Liu, D.; Yang, B. A novel host-guest complex based on biotin functionalized polyamine-β-cyclodextrin for tumor targeted delivery of luteolin. J. Mol. Struct. 2021, 1237, 130339. [Google Scholar] [CrossRef]
- Liu, B.; Li, W.; Zhao, J.; Liu, Y.; Zhu, X.; Liang, G. Physicochemical characterisation of the supramolecular structure of luteolin/cyclodextrin inclusion complex. Food Chem. 2013, 141, 900–906. [Google Scholar] [CrossRef]
- D’Onofre Couto, B.; Novaes da Costa, R.; Castro Laurindo, W.; Moraes da Silva, H.; Rocha da Silva, C.; Sélia dos Reis Coimbra, J.; Barbosa Mageste, A.; de Cássia Dias, S.; José Boggione Santos, I. Characterization, techno-functional properties, and encapsulation efficiency of self-assembled β-lactoglobulin nanostructures. Food Chem. 2021, 356, 129719. [Google Scholar] [CrossRef]
- Zu, Y.; Wu, W.; Zhao, X.; Li, Y.; Zhong, C.; Zhang, Y. The high-water solubility of inclusion complex of taxifolin-γ-CD prepared and characterized by the emulsion solvent evaporation and the freeze drying combination method. Int. J. Pharm. 2014, 477, 148–158. [Google Scholar] [CrossRef]
- Liu, M.; Liao, R.; Zhao, Y.; Yang, B. Host—Guest Inclusion System of Luteolin with Polyamine-β-cyclodextrin: Preparation, Characterisation, Anti-oxidant and Anti-cancer Activity. Aust. J. Chem. 2015, 69, 174–182. [Google Scholar]
- Luong, D.; Kesharwani, P.; Deshmukh, R.; Mohd Amin, M.C.I.; Gupta, U.; Greish, K.; Iyer, A.K. PEGylated PAMAM dendrimers: Enhancing efficacy and mitigating toxicity for effective anticancer drug and gene delivery. Acta Biomater. 2016, 43, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, A.S. Dendrimers for drug delivery. Molecules 2018, 23, 938. [Google Scholar] [CrossRef] [PubMed]

| Type of Delivery System | Flavonoid | Constitution | Characteristics | Type of Study | Experimental Studies | References |
|---|---|---|---|---|---|---|
| Liposomes | Quercetin | Soybean phosphatidylcholine and cholesterol | Size: 143.1 nm EE 1: 96.96% | In vitro and in vivo | IC502: 10–50 µM. DR 3: 26.5% after 12 h. Tumor decreases by about 50%. | [43] |
| PEG 4, cholesterol and soybean phosphatidylcholine | Size: 171.3 nm EE: 81.25% | In vitro and in vivo | Toxicity: 10% IC50: 3.033 µM after 48 h. Tumor decreases from 1500 mm3 to 500 mm3. | [44] | ||
| Egg-phosphatidylcholine, cholesterol and 2-distearoyl-sn-glycero-3-phosphoethanolamine-PEG 2000 | Size: 109.79 nm. EE: superior to 80% | In vitro | IC50: 185 µM, 40 µM and 14 µM were established after 24, 48 and 72 h, respectively. | [45] | ||
| Triglycerides, lecithin, PEG and acid folic | EE: 96.01% | In vitro | IC50: 13 µM. | [46] | ||
| Baicalein | Soybean phosphatidylcholine and cholesterol | Size: between 166.9 and 194.6 nm ZP 5: between −18.23 and −30.73 mV EE: 44.3% | In vitro | Inhibition rate of 66.34%. | [47] | |
| Nanoemulsion | Quercetin | Polyglyceryl-10 laurate, polyglycerol-6 monostearate and sucrose esters-11 | Size: between 93 nm and 233 nm EE: 84.7% | In vitro | VR 6 to 90%. | [48] |
| Biopolymer | Quercetin | Chitosan and quinoline | Size: 174.8 nm EE: 77.2% | In vitro | IC50: 10–14 ug/mL after 48 h. DR: 69.3%–78.4% after 8 h. | [49] |
| Rutin and quercetin | Keratin and sodium dodecyl sulphate | Size: 55 nm ZP: −28.09 mV EE: 86.5% | In vitro | 85% was released within 30 h. VR up to 80%. | [50] | |
| Rutin | Fucoidan | Size: 221 nm | In vitro | IC50: 20 µg/mL | [51] | |
| Naringenin | Silk fibroin | Size: between 148.4 and 180.1 nm ZP: between −30.5 and −39.1 mV EE: 21.81% | In vitro | IC50: 250 µg/mL. | [52] | |
| Hesperidin | Gliadin coated with chitosan | Size: between 226.5 and 321.40 nm ZP: between −2.91 and 21.40 mV EE: between 73.10 and 80.11% | In vitro | IC50: 16 ug/mL For blank nanoparticles IC50 of 159.33 µg/mL | [53] | |
| Synthetic polymer | Genistein | Poly e-caprolactone and PEG 1000 succinate | Size: 181.83 nm ZP: −14.70 mV EE: 95.56% | In vitro and in vivo | IC50: 24.3 ug/mL, 13.6 µg/mL and 5 µg/mL after 24, 48 and 72 h, respectively. In vivo studies showed a reduction of tumour weight by about 4 times. | [54] |
| Quercetin | Gelatin modified pluronics | Size: between 79.52 and 152.51 nm EE: 93.02% | In vitro | IC50: 45.83 µM. | [55] | |
| PEG and poly lactide-co-glycolide | Size: between 143.1 and 153 nm EE: between 97.8% and 99% | In vitro | IC50: 10 µM. | [56] | ||
| Inorganic polymer | Quercetin | Oxide nanoparticles functionalized with citric acid and α-cyclodextrin | Size: between 22.35 and 59.9 nm ZP: between −15.4 and 35.6 mV EE: higher than 75% | In vitro | VR: almost zero for nanoparticles with a drug concentration of 100 µg/mL. | [57] |
| Phloretin | Gold nanoparticles | Size: 8 and 15 nm ZP: between −31.7 and −38.2 mV | In vitro | VR: 12.5% with a concentration of 4 mg/mL. | [58] | |
| Hesperetin, naringenin and apigenin | Copper complexes | In vitro | Inhibitory rate between 20 and 30%. | [59] | ||
| Inorganic polymer/biopolymer | Quercetin | Copper nanoclusters with hydroxyapatite | Size: 36.2 nm ZP: −19.3 mV EE: 72% | In vitro | IC50: 500 µM. | [60] |
| Inorganic/synthetic polymer | Quercetin | Magnetic nanoparticles coated with poly citric acid and functionalized with folic acid and PEG | Size: between 10 and 49 nm EE: 80.3% | In vitro | VR: 25% with 100 µg/mL of quercetin. Toxicity: 0% | [61] |
| Halloysites nanotubes functionalized with PEG | ZP: 37.44 mV | In vitro | VR: 30% for a drug concentration of 50 µg/mL. Toxicity: 10%. | [62] | ||
| Quercetin and luteolin | Magnetic iron oxide nanoparticles modified with 3-aminopropyl triethoxysilane, folic acid and PEG | Size: between 8 and 20 nm | In vitro | VR: 20% and 40% with 100 µg/mL of quercetin and luteolin, respectively. | [63,64] | |
| Micelles | Quercetin | Chondroitin sulfate and cholesterol | Size: between 124 and 237 nm EE: 30.6% | In vitro | VR: near to 80% for a drug concentration of 200 µg/mL. | [65] |
| Inclusion complex | Fisetin | Cyclosophoraose dimers | Showed 2.4 times more solubility of fisetin than β-cyclodextrin | In vitro | VR: 29% after an incubation of 24 h with 100 µM of drug. | [66] |
| Chrysin | β-cyclodextrin | Size: 458 nm ZP: −38.4 mV EE: 59.1% | In vitro | VR: 11.5% after 48 h with 100 µM of drug. | [67] | |
| Dendrimers | Baicalin | Poly amidoamine dendrimers modified with folic acid | Size: between 174.4 and 258.8 nm ZP: between −2.9 mV and −9.3 mV EE: between 53.5 and 91.9% | In vitro | VR: 40% after 48 h with 25 µg/mL of baicalin. | [68] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira, M.; Costa, D.; Sousa, Â. Flavonoids-Based Delivery Systems towards Cancer Therapies. Bioengineering 2022, 9, 197. https://doi.org/10.3390/bioengineering9050197
Ferreira M, Costa D, Sousa Â. Flavonoids-Based Delivery Systems towards Cancer Therapies. Bioengineering. 2022; 9(5):197. https://doi.org/10.3390/bioengineering9050197
Chicago/Turabian StyleFerreira, Miguel, Diana Costa, and Ângela Sousa. 2022. "Flavonoids-Based Delivery Systems towards Cancer Therapies" Bioengineering 9, no. 5: 197. https://doi.org/10.3390/bioengineering9050197
APA StyleFerreira, M., Costa, D., & Sousa, Â. (2022). Flavonoids-Based Delivery Systems towards Cancer Therapies. Bioengineering, 9(5), 197. https://doi.org/10.3390/bioengineering9050197