Radiomic Analysis as a Powerful Tool for Cytological Images of Benign Thyroid Nodules Treated by Thermal Radiofrequency Ablation
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Data Collection
2.2. Diagnostic and Therapeutic Procedures
2.2.1. Fine-Needle Aspiration Cytology (FNAC)
2.2.2. Thyroid Ultrasound (ECO) Parameters
2.2.3. RFA Procedure—VIVA RF System
2.3. Cytology Image Processing and Feature Extraction
2.3.1. Image Acquisition
2.3.2. Chromatic Analysis of Cytological Images
2.3.3. Radiomics Analysis and Principal Component Analysis (PCA)
2.3.4. Nuclei Shape Analysis
3. Results
3.1. Chromatic Analysis and Nuclei Segmentation
3.2. Volume Reduction Ratio (VRR) and Shape Analysis
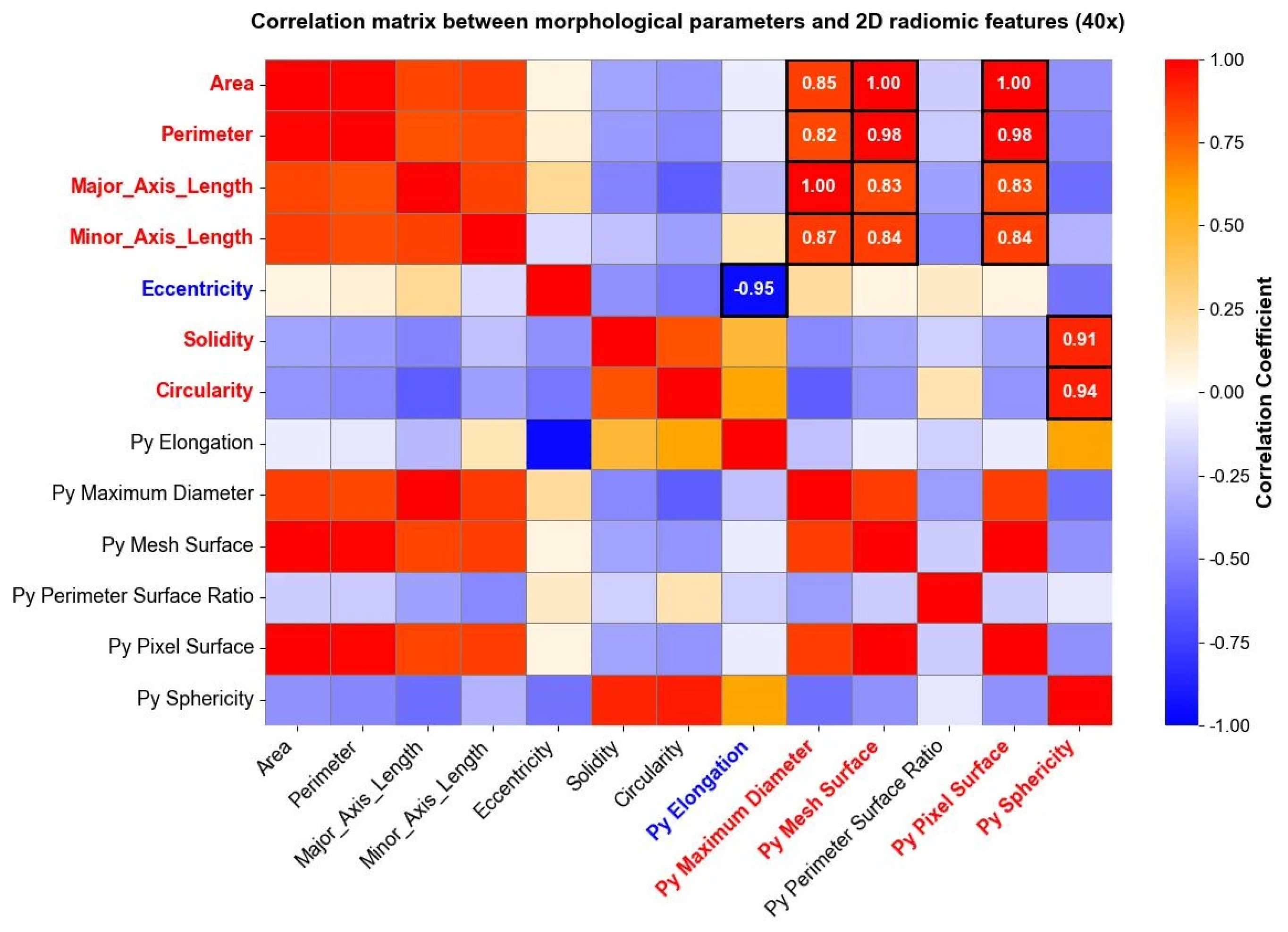
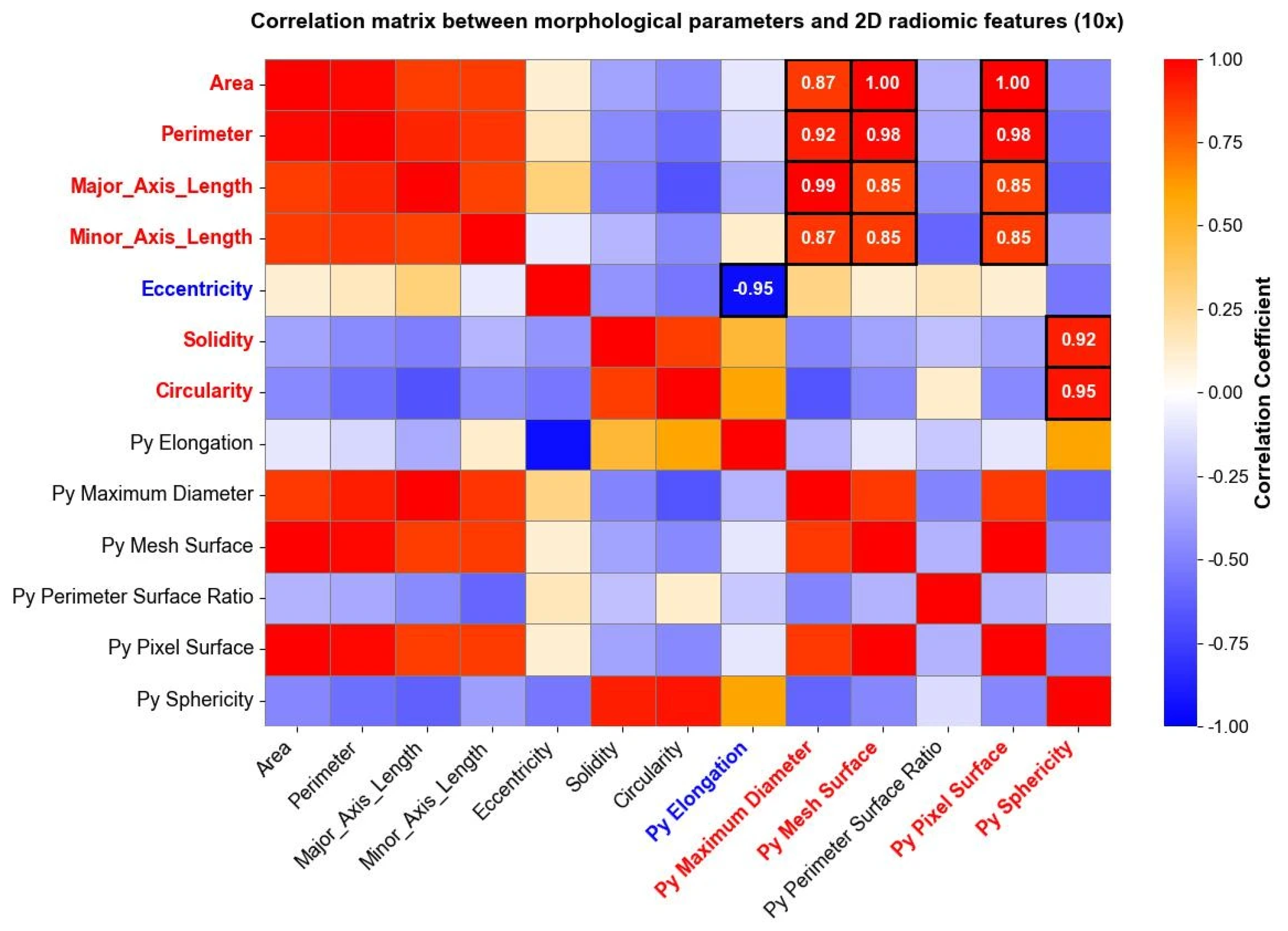
3.3. Radiomics and PCA Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CAD | Computer-Aided Diagnosis |
| CNB | Core Needle Biopsy |
| ECO | Ecography (Thyroid Ultrasound) |
| EOC | Ente Ospedaliero Cantonale |
| FNAC | Fine-Needle Aspiration Cytology |
| FNA | Fine-Needle Aspiration |
| GLCM | Gray-Level Co-occurrence Matrix |
| GLDM | Gray-Level Dependence Matrix |
| GLRLM | Gray-Level Run Length Matrix |
| GLSZM | Gray-Level Size Zone Matrix |
| H&E | Hematoxylin and Eosin |
| HIFU | High-Intensity Focused Ultrasound |
| IBSI | Imaging Biomarker Standardization Initiative |
| IQR | Interquartile Range |
| NGTDM | Neighboring Gray Tone Difference Matrix |
| PCA | Principal Component Analysis |
| Py | Prefix for PyRadiomics (i.e., Py Sphericity, Py Mesh Surface) |
| RFA | Radiofrequency Ablation |
| RF | Radiofrequency |
| ROI | Region of Interest |
| Std | Standard Deviation |
| TBSRTC | The Bethesda System for Reporting Thyroid Cytopathology |
| US | Ultrasound |
| VRR | Volume Reduction Ratio |
| WSI | Whole Slide Image |
References
- Hirokawa, M.; Auger, M.; Jung, C.K.; Callegari, F.M. Thyroid FNA Cytology: The Eastern versus Western Perspectives. Cancer Cytopathol. 2023, 131, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Ha, E.J.; Baek, J.H.; Che, Y.; Chou, Y.-H.; Fukunari, N.; Kim, J.; Lin, W.-C.; My, L.T.; Na, D.G.; Quek, L.H.H.; et al. Radiofrequency Ablation of Benign Thyroid Nodules: Recommendations from the Asian Conference on Tumor Ablation Task Force. Ultrasonography 2021, 40, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Cibas, E.S.; Ali, S.Z. The 2017 Bethesda System for Reporting Thyroid Cytopathology. Thyroid 2017, 27, 1341–1346. [Google Scholar] [CrossRef]
- Gharib, H. Fine-Needle Aspiration Biopsy of Thyroid Nodules: Advantages, Limitations, and Effect. Mayo Clin. Proc. 1994, 69, 44–49. [Google Scholar] [CrossRef]
- Choi, Y.J.; Baek, J.H.; Hong, M.J.; Lee, J.H. Inter-Observer Variation in Ultrasound Measurement of the Volume and Diameter of Thyroid Nodules. Korean Soc. Radiol. 2015, 16, 560–565. [Google Scholar] [CrossRef]
- Deandrea, M.; Trimboli, P.; Mormile, A.; Cont, A.T.; Milan, L.; Buffet, C.; Giovanella, L.; Limone, P.P.; Poirée, S.; Leenhardt, L.; et al. Determining an Energy Threshold for Optimal Volume Reduction of Benign Thyroid Nodules Treated by Radiofrequency Ablation. Eur. Radiol. 2021, 31, 5189–5197. [Google Scholar] [CrossRef]
- Navin, P.J.; Thompson, S.M.; Kurup, A.N.; Lee, R.A.; Callstrom, M.R.; Castro, M.R.; Stan, M.N.; Welch, B.T.; Schmitz, J.J. Radiofrequency Ablation of Benign and Malignant Thyroid Nodules. Radiographics 2022, 42, 1812–1828. [Google Scholar] [CrossRef]
- Lee, M.; Baek, J.H.; Suh, C.H.; Chung, S.R.; Choi, Y.J.; Lee, J.H.; Ha, E.J.; Na, D.G. Clinical Practice Guidelines for Radiofrequency Ablation of Benign Thyroid Nodules: A Systematic Review. Ultrasonography 2021, 40, 256–264. [Google Scholar] [CrossRef]
- Jung, S.L. Advanced Techniques for Thyroid Nodule Radiofrequency Ablation. Tech. Vasc. Interv. Radiol. 2022, 25, 100820. [Google Scholar] [CrossRef]
- Papini, E.; Monpeyssen, H.; Frasoldati, A.; Hegedüs, L. 2020 European Thyroid Association Clinical Practice Guideline for the Use of Image-Guided Ablation in Benign Thyroid Nodules. Eur. Thyroid J. 2020, 9, 172–185. [Google Scholar] [CrossRef] [PubMed]
- Rossi, G.; Petrone, M.C.; Capurso, G.; Albarello, L.; Testoni, S.G.G.; Archibugi, L.; Schiavo Lena, M.; Doglioni, C.; Arcidiacono, P.G. Standardization of a Radiofrequency Ablation Tool in an Ex-Vivo Porcine Liver Model. Gastrointest. Disord. 2020, 2, 300–309. [Google Scholar] [CrossRef]
- Bini, F.; Pica, A.; Marinozzi, F.; Giusti, A.; Leoncini, A.; Trimboli, P. Model-Optimizing Radiofrequency Parameters of 3D Finite Element Analysis for Ablation of Benign Thyroid Nodules. Bioengineering 2023, 10, 1210. [Google Scholar] [CrossRef]
- Bini, F.; Trimboli, P.; Marinozzi, F.; Giovanella, L. Treatment of Benign Thyroid Nodules by High Intensity Focused Ultrasound (HIFU) at Different Acoustic Powers: A Study on in-Silico Phantom. Endocrine 2018, 59, 506–509. [Google Scholar] [CrossRef]
- Bouyssoux, A.; Jarnouen, K.; Lallement, L.; Fezzani, R.; Olivo-Marin, J.-C. Automated Staining Analysis in Digital Cytopathology and Applications. Cytom. Part A 2022, 101, 1068–1083. [Google Scholar] [CrossRef]
- Durkee, M.S.; Abraham, R.; Clark, M.R.; Giger, M.L. Artificial Intelligence and Cellular Segmentation in Tissue Microscopy Images. Am. J. Pathol. 2021, 191, 1693–1701. [Google Scholar] [CrossRef]
- Kan, A. Machine Learning Applications in Cell Image Analysis. Immunol. Cell Biol. 2017, 95, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Voigt, S.P.; Ravikumar, K.; Basu, B.; Kalidindi, S.R. Automated Image Processing Workflow for Morphological Analysis of Fluorescence Microscopy Cell Images. JOM 2021, 73, 2356–2365. [Google Scholar] [CrossRef]
- Chen, S.; Zhao, M.; Wu, G.; Yao, C.; Zhang, J. Recent Advances in Morphological Cell Image Analysis. Comput. Math. Methods Med. 2012, 2012, 101536. [Google Scholar] [CrossRef]
- Burns, B.L.; Rhoads, D.D.; Misra, A. The Use of Machine Learning for Image Analysis Artificial Intelligence in Clinical Microbiology. J. Clin. Microbiol. 2023, 61, e02336-21. [Google Scholar] [CrossRef] [PubMed]
- Jiménez Gaona, Y.; Castillo Malla, D.; Vega Crespo, B.; Vicuña, M.J.; Neira, V.A.; Dávila, S.; Verhoeven, V. Radiomics Diagnostic Tool Based on Deep Learning for Colposcopy Image Classification. Diagnostics 2022, 12, 1694. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Zheng, S.; Li, S.; Huang, Y.; Zhang, W.; Liu, F.; Cao, Q. Machine Learning-Based Pathomics Model Predicts ANGPT2 Expression and Prognosis in Hepatocellular Carcinoma. Am. J. Pathol. 2024, 195, 561–574. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, T.S.; Cho, M. Segmentation of Variants of Nuclei on Whole Slide Images by Using Radiomic Features. Bioengineering 2024, 11, 252. [Google Scholar] [CrossRef]
- Linares, O.C.; Belizario, I.V.; Batah, S.S.; Hamann, B.; Fabro, A.T.; Azevedo-Marques, P.M.; Traina, A.J.M. RadPleura: A radiomics-based framework for lung pleura classification in histology images from interstitial lung diseases. In Proceedings of the 2024 IEEE International Symposium on Biomedical Imaging (ISBI), Athens, Greece, 27–30 May 2024; pp. 1–5. [Google Scholar]
- Baishya, N.K.; Baishya, K. Integration of Radiomics Ultrasound and TIRADS in Diagnosis of Thyroid Nodules: A Narrative Review. Egypt. J. Radiol. Nucl. Med. 2024, 55, 212. [Google Scholar] [CrossRef]
- Cleere, E.F.; Davey, M.G.; O’Neill, S.; Corbett, M.; O’Donnell, J.P.; Hacking, S.; Keogh, I.J.; Lowery, A.J.; Kerin, M.J. Radiomic Detection of Malignancy within Thyroid Nodules Using Ultrasonography-A Systematic Review and Meta-Analysis. Diagnostics 2022, 12, 794. [Google Scholar] [CrossRef]
- Xu, H.; Wang, X.; Guan, C.; Tan, R.; Yang, Q.; Zhang, Q.; Liu, A.; Liu, Q. Value of Whole-Thyroid CT-Based Radiomics in Predicting Benign and Malignant Thyroid Nodules. Front. Oncol. 2022, 12, 828259. [Google Scholar] [CrossRef]
- Lee, J.; Lee, J.; Song, B.-I. A Machine Learning-Based Radiomics Model for the Differential Diagnosis of Benign and Malignant Thyroid Nodules in F-18 FDG PET/CT: External Validation in the Different Scanner. Cancers 2025, 17, 331. [Google Scholar] [CrossRef]
- Konca, C.; Elhan, A.H. Unveiling the Accuracy of Ultrasonographic Assessment of Thyroid Volume: A Comparative Analysis of Ultrasonographic Measurements and Specimen Volumes. J. Clin. Med. 2023, 12, 6619. [Google Scholar] [CrossRef]
- Lin, W.C.; Wang, C.K.; Wang, W.H.; Kuo, C.Y.; Chiang, P.L.; Lin, A.N.; Baek, J.H.; Wu, M.H.; Cheng, K.L. Multicenter Study of Benign Thyroid Nodules with Radiofrequency Ablation: Results of 762 Cases over 4 Years in Taiwan. J. Pers. Med. 2022, 12, 63. [Google Scholar] [CrossRef] [PubMed]
- Hussein, Z.; Mahameed, A.; Fadhil, J. Distinguishing of Different Tissue Types Using K-Means Clustering of Color Segmentation. East. Eur. J. Enterp. Technol. 2021, 5, 22–28. [Google Scholar] [CrossRef]
- Stringer, C.; Wang, T.; Michaelos, M.; Pachitariu, M. Cellpose: A Generalist Algorithm for Cellular Segmentation. Nat. Methods 2021, 18, 100–106. [Google Scholar] [CrossRef]
- Van Griethuysen, J.J.M.; Fedorov, A.; Parmar, C.; Hosny, A.; Aucoin, N.; Narayan, V.; Beets-Tan, R.G.H.; Fillon-Robin, J.C.; Pieper, S.; Aerts, H.J.W.L. Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res. 2017, 77, e104–e107. [Google Scholar] [CrossRef]
- Zwanenburg, A.; Vallières, M.; Abdalah, M.A.; Aerts, H.J.W.L.; Andrearczyk, V.; Apte, A.; Ashrafinia, S.; Bakas, S.; Beukinga, R.J.; Boellaard, R.; et al. The Image Biomarker Standardization Initiative: Standardized Quantitative Radiomics for High-Throughput Image-Based Phenotyping. Radiology 2020, 295, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Wirth, M.A. Shape Analysis and Measurement; University of Guelph: Guelph, ON, Canada, 2004; pp. 1–49. [Google Scholar]
- Choi, S.; Wang, W.; Ribeiro, A.J.; Kalinowski, A.; Gregg, S.Q.; Opresko, P.L.; Niedernhofer, L.J.; Rohde, G.K.; Dahl, K.N. Computational Image Analysis of Nuclear Morphology Associated with Various Nuclear-Specific Aging Disorders. Nucleus 2011, 2, 570–579. [Google Scholar] [CrossRef]
- Yan, L.; Zhang, M.; Li, X.; Li, Y.; Luo, Y. A Nomogram to Predict Regrowth After Ultrasound-Guided Radiofrequency Ablation for Benign Thyroid Nodules. Front. Endocrinol. 2022, 12, 774228. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.H.; Lin, W.C.; Luo, S.D.; Chiang, P.L.; Chen, Y.S.; Chen, W.C.; Lin, A.N.; Wang, C.K.; Baek, J.H.; Chen, H.L. Residual, Regrowth, and New Growth of Radiofrequency Ablation for Benign Thyroid Nodules of Different Volumes: Two-Year Follow-up Results. Int. J. Hyperth. 2022, 39, 1172–1178. [Google Scholar] [CrossRef]
- Sim, J.S.; Baek, J.H. Long-Term Outcomes of Thermal Ablation for Benign Thyroid Nodules: The Issue of Regrowth. Int. J. Endocrinol. 2021, 59, 9922509. [Google Scholar] [CrossRef]
- Cattell, R.B. The Scree Test for the Number of Factors. Multivar. Behav. Res. 1966, 1, 245–276. [Google Scholar] [CrossRef]
- Jolliffe, I.T. Principal Component Analysis, 2nd ed.; Springer Series in Statistics; Springer: New York, NY, USA, 2002; ISBN 978-0-387-95442-4. [Google Scholar]
- Fiorentino, V.; Pizzimenti, C.; Franchina, M.; Micali, M.G.; Russotto, F.; Pepe, L.; Militi, G.B.; Tralongo, P.; Pierconti, F.; Ieni, A.; et al. The Minefield of Indeterminate Thyroid Nodules: Could Artificial Intelligence Be a Suitable Diagnostic Tool? Diagn. Histopathol. 2023, 29, 396–401. [Google Scholar] [CrossRef]
- Patel, J.; Klopper, J.; Cottrill, E.E. Molecular Diagnostics in the Evaluation of Thyroid Nodules: Current Use and Prospective Opportunities. Front. Endocrinol. 2023, 14, 1101410. [Google Scholar] [CrossRef]
- Russotto, F.; Fiorentino, V.; Pizzimenti, C.; Micali, M.G.; Franchina, M.; Pepe, L.; Riganati, G.; Giordano, W.; Magliolo, E.; Ristagno, S.; et al. Histologic Evaluation of Thyroid Nodules Treated with Thermal Ablation: An Institutional Experience. Int. J. Mol. Sci. 2024, 25, 10182. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Wang, Z.; Lai, S.; Meng, L.; Lu, Y.; Lu, X.; Lin, L.; Tang, Y.; Zhang, S.; Mo, S.; et al. Comparative Study of Radiomics, Tumor Morphology, and Clinicopathological Factors in Predicting Overall Survival of Patients with Rectal Cancer before Surgery. Transl. Oncol. 2022, 18, 101352. [Google Scholar] [CrossRef] [PubMed]
- Finti, A.; Stefano, A.; Pasini, G.; Russo, G.; Marinozzi, F.; Bini, F. Preliminary radiomics-based classification of tumor nuclei in melanoma histopathology. In Proceedings of the Image Analysis and Processing—ICIAP 2025 Workshops, Rome, Italy, 15–19 September 2025; Springer Nature: Cham, Switzerland, 2026; pp. 166–174. [Google Scholar]


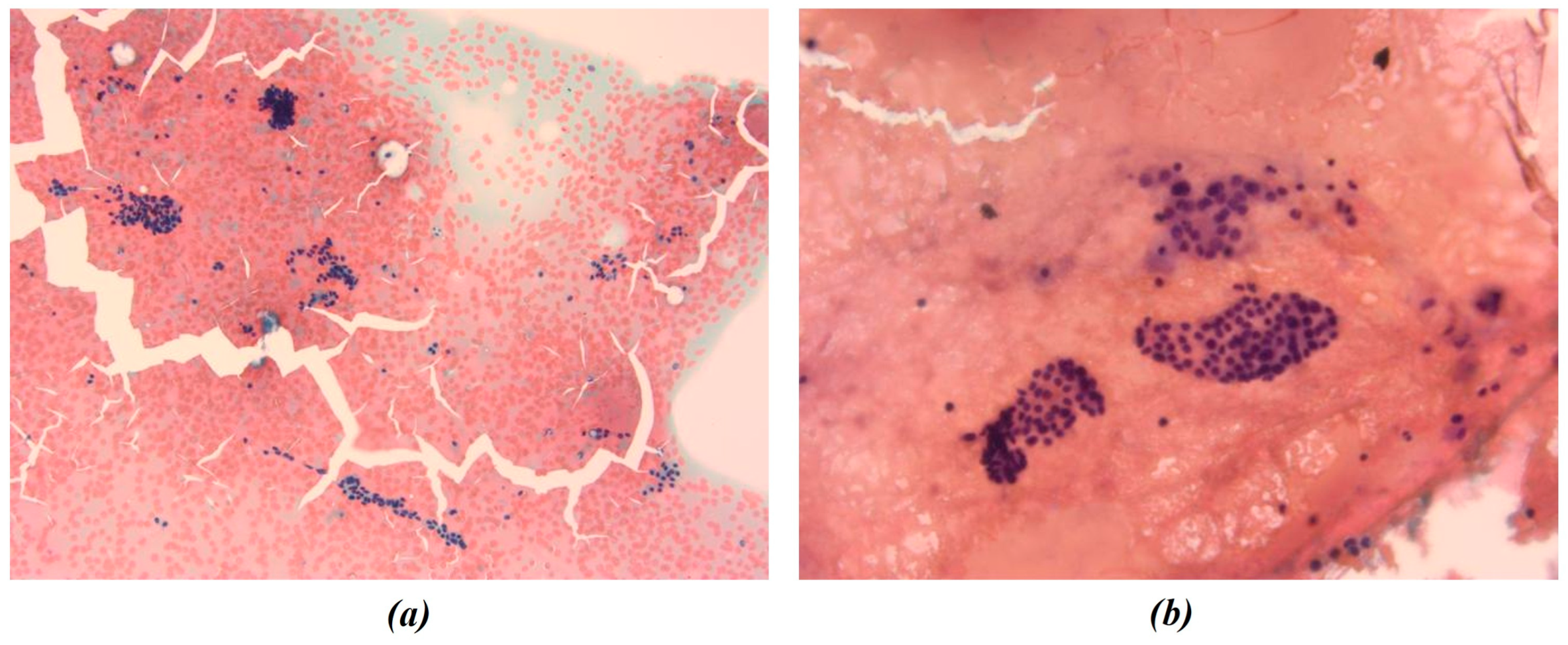
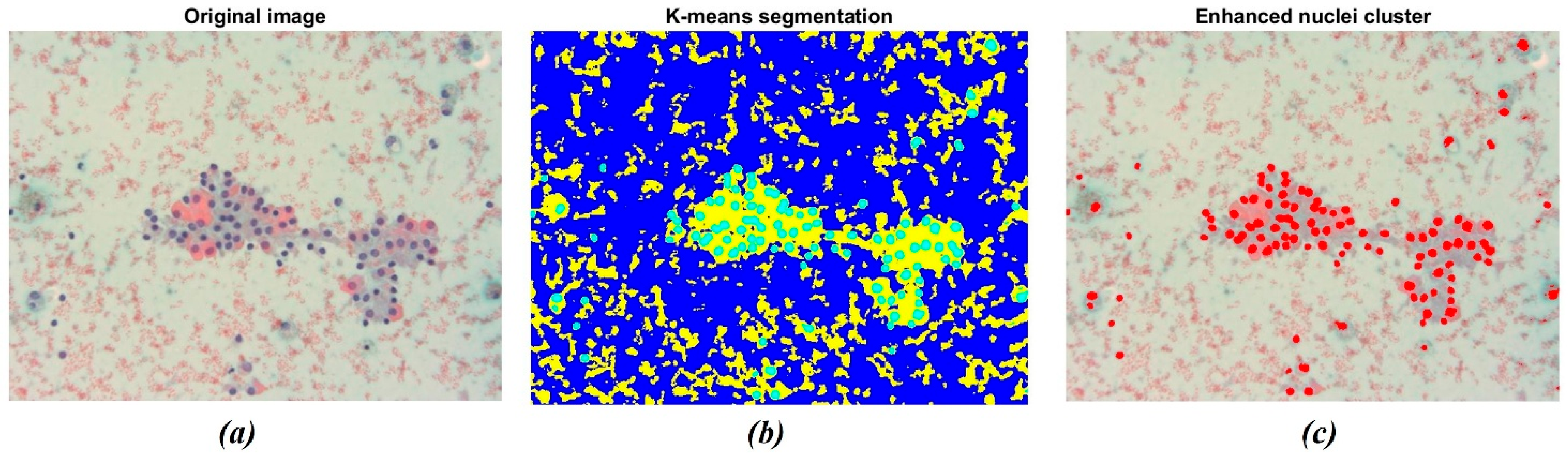

| Patient | Initial Volume | Volume One Month After RFA Treatment | Volume Six Months After RFA Treatment | VRR (1st Follow-Up) | VRR (2nd Follow-Up) |
|---|---|---|---|---|---|
| A | 5.01 mL | 3.56 mL | 2.80 mL | 29% | 44% |
| B | 7.45 mL | 5.22 mL | 5.70 mL | 30% | 23% |
| C | 3.05 mL | 1.76 mL | n/a | 42% | n/a |
| Patient A | Patient B | Patient C | ||||
|---|---|---|---|---|---|---|
| Average | Std | Average | Std | Average | Std | |
| Circularity [0, 1] | 0.84 | 0.08 | 0.72 | 0.14 | 0.77 | 0.19 |
| Eccentricity [0, 1] | 0.56 | 0.16 | 0.77 | 0.14 | 0.68 | 0.18 |
| Solidity [0, 1] | 0.95 | 0.02 | 0.91 | 0.06 | 0.90 | 0.07 |
| PC (40×) | Features (95% of Maximum Loading) | Explained Variance (%) | PC (10×) | Features (95% of Maximum Loading) | Explained Variance (%) |
|---|---|---|---|---|---|
| 1 | original_firstorder_Entropy original_firstorder_Range | 34.33% | 1 | original_firstorder_Entropy. original_firstorder_MeanAbsoluteDeviation original_firstorder_Range original_glcm_JointEntropy original_glcm_SumEntropy original_glrlm_GrayLevelVariance original_glrlm_HighGrayLevelRunEmphasis original_glszm_HighGrayLevelZoneEmphasis | 36.45% |
| 2 | original_glcm_Contrast original_glcm_DifferenceAverage original_glcm_Id original_glcm_Idm original_glcm_Idn original_glcm_InverseVariance | 17.10% | 2 | original_gldm_LargeDependenceEmphasis original_glrlm_RunPercentage | 21.13% |
| 3 | original_glrlm_GrayLevelNonUniformity original_glrlm_RunLengthNonUniformity original_glszm_GrayLevelNonUniformity original_glszm_SizeZoneNonUniformity | 10.16% | 3 | original_shape2D_MeshSurface original_shape2D_PixelSurface original_glrlm_GrayLevelNonUniformity original_glrlm_RunLengthNonUniformity | 8.5% |
| 4 | original_ngtdm_Coarseness | 5.76% | 4 | original_gldm_LargeDependenceLowGrayLevelEmphasis | 5.24% |
| 5 | original_firstorder_90Percentile original_firstorder_Mean original_firstorder_RootMeanSquared | 4.94% | 5 | original_glszm_SizeZoneNonUniformityNormalized original_ngtdm_Contrast original_ngtdm_Strength | 3.75% |
| 6 | original_gldm_LargeDependenceLowGrayLevelEmphasis original_gldm_LowGrayLevelEmphasis | 4.68% | 6 | original_firstorder_90Percentile | 3.34% |
| 7 | original_gldm_DependenceVariance | 3.01% | 7 | original_firstorder_Kurtosis original_firstorder_Maximum | 2.97% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Finti, A.; Marinozzi, F.; Franzò, M.; Federici, F.; Bolognese, M.; Giusti, A.; Leoncini, A.; Bini, F. Radiomic Analysis as a Powerful Tool for Cytological Images of Benign Thyroid Nodules Treated by Thermal Radiofrequency Ablation. Bioengineering 2026, 13, 171. https://doi.org/10.3390/bioengineering13020171
Finti A, Marinozzi F, Franzò M, Federici F, Bolognese M, Giusti A, Leoncini A, Bini F. Radiomic Analysis as a Powerful Tool for Cytological Images of Benign Thyroid Nodules Treated by Thermal Radiofrequency Ablation. Bioengineering. 2026; 13(2):171. https://doi.org/10.3390/bioengineering13020171
Chicago/Turabian StyleFinti, Alessia, Franco Marinozzi, Michela Franzò, Flavia Federici, Matteo Bolognese, Alessandro Giusti, Andrea Leoncini, and Fabiano Bini. 2026. "Radiomic Analysis as a Powerful Tool for Cytological Images of Benign Thyroid Nodules Treated by Thermal Radiofrequency Ablation" Bioengineering 13, no. 2: 171. https://doi.org/10.3390/bioengineering13020171
APA StyleFinti, A., Marinozzi, F., Franzò, M., Federici, F., Bolognese, M., Giusti, A., Leoncini, A., & Bini, F. (2026). Radiomic Analysis as a Powerful Tool for Cytological Images of Benign Thyroid Nodules Treated by Thermal Radiofrequency Ablation. Bioengineering, 13(2), 171. https://doi.org/10.3390/bioengineering13020171









