Development of an In Vitro Assessment Method for Chemotherapy-Induced Peripheral Neuropathy (CIPN) by Integrating a Microphysiological System (MPS) with Morphological Deep Learning of Soma and Axonal Images
Abstract
1. Introduction
2. Materials and Methods
2.1. Device Fabrication
2.2. Cell Culture
2.3. Immunocytochemistry
2.4. Deep Learning for Cell Seeding Area Prediction and Neurite Elongation Area Prediction
2.5. Statistical Analysis
2.6. Grad-CAM
3. Results
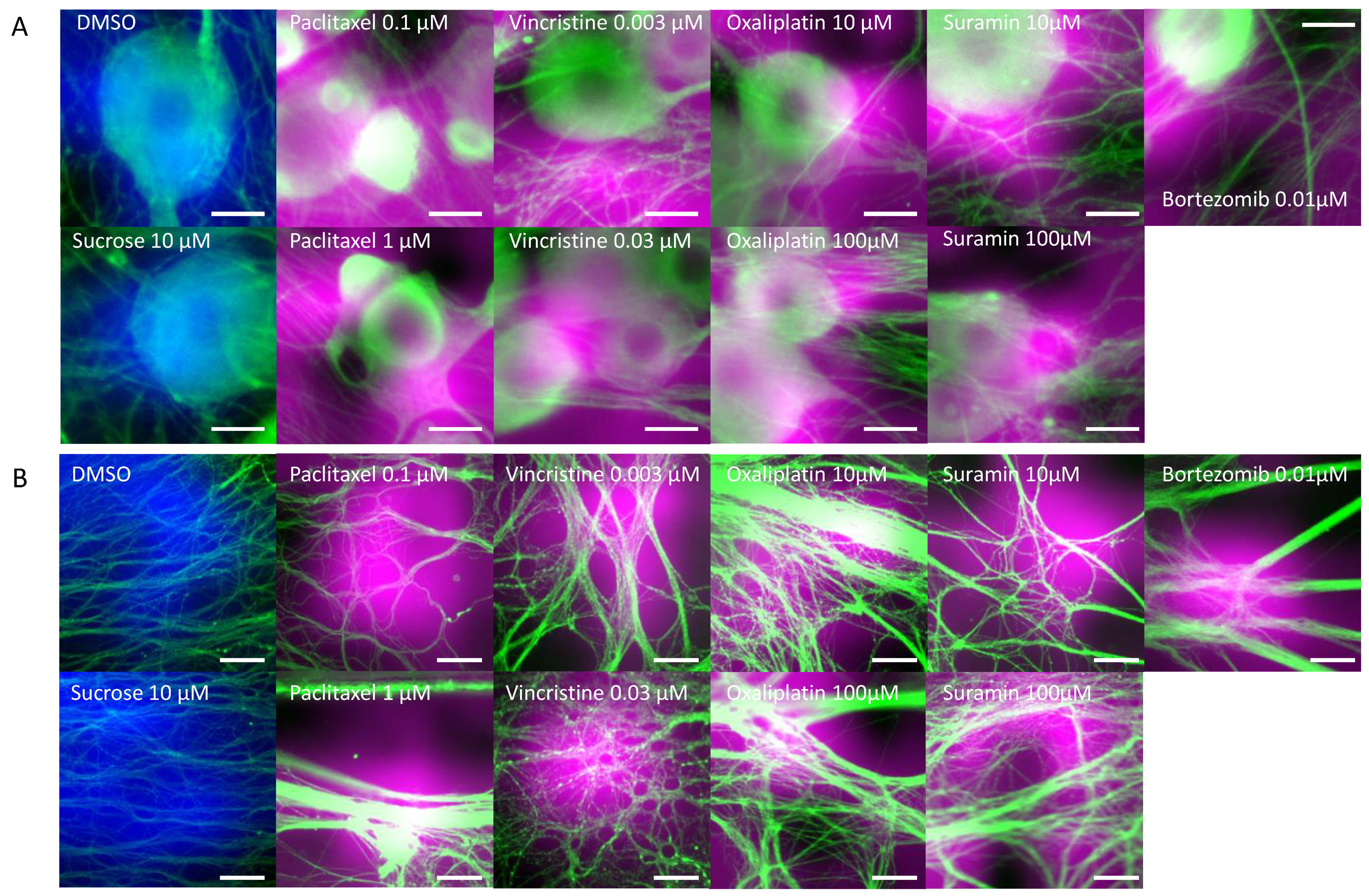
3.1. Morphological Changes in DRG Neurons by CIPN-Inducing Compounds
3.2. Toxicity Prediction Based on Compound-Induced Morphological Changes Using Two AI Models, Soma and Axonal Areas
3.3. Classification of MoA Based on Toxicity Prediction Results from Two AI Models
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pachman, D.R.; Qin, R.; Seisler, D.K.; Smith, E.M.; Beutler, A.S.; Ta, L.E.; Lafky, J.M.; Wagner-Johnston, N.D.; Ruddy, K.J.; Dakhil, S.; et al. Clinical Course of Oxaliplatin-Induced Neuropathy: Results from the Randomized Phase III Trial N08CB (Alliance). J. Clin. Oncol. 2015, 33, 3416–3422. [Google Scholar] [CrossRef]
- Hershman, D.L.; Lacchetti, C.; Dworkin, R.H.; Lavoie Smith, E.M.; Bleeker, J.; Cavaletti, G.; Chauhan, C.; Gavin, P.; Lavino, A.; Lustberg, M.B.; et al. Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. J. Clin. Oncol. 2014, 32, 1941–1967. [Google Scholar] [CrossRef]
- Kelley, M.R.; Fehrenbacher, J.C. Challenges and opportunities identifying therapeutic targets for chemotherapy-induced peripheral neuropathy resulting from oxidative DNA damage. Neural Regen. Res. 2017, 12, 72–74. [Google Scholar] [CrossRef]
- Song, S.J.; Min, J.; Suh, S.Y.; Jung, S.H.; Hahn, H.J.; Im, S.A.; Lee, J.Y. Incidence of taxane-induced peripheral neuropathy receiving treatment and prescription patterns in patients with breast cancer. Support. Care Cancer 2017, 25, 2241–2248. [Google Scholar] [CrossRef]
- Ibrahim, E.Y.; Ehrlich, B.E. Prevention of chemotherapy-induced peripheral neuropathy: A review of recent findings. Crit. Rev. Oncol. Hematol. 2020, 145, 102831. [Google Scholar] [CrossRef]
- Argyriou, A.A.; Koltzenburg, M.; Polychronopoulos, P.; Papapetropoulos, S.; Kalofonos, H.P. Peripheral nerve damage associated with administration of taxanes in patients with cancer. Crit. Rev. Oncol. Hematol. 2008, 66, 218–228. [Google Scholar] [CrossRef]
- Manasanch, E.E.; Orlowski, R.Z. Proteasome inhibitors in cancer therapy. Nat. Rev. Clin. Oncol. 2017, 14, 417–433. [Google Scholar] [CrossRef]
- Willson, M.L.; Burke, L.; Ferguson, T.; Ghersi, D.; Nowak, A.K.; Wilcken, N. Taxanes for adjuvant treatment of early breast cancer. Cochrane Database Syst. Rev. 2019, 9, Cd004421. [Google Scholar] [CrossRef]
- Section 4: Health Effects. Test No. 424: Neurotoxicity Study in Rodents. Available online: https://www.oecd-ilibrary.org/environment/test-no-424-neurotoxicity-study-in-rodents_9789264071025-en (accessed on 12 September 2023).
- Section 4: Health Effects. Test No. 426: Developmental Neurotoxicity Study. Available online: https://www.oecd-ilibrary.org/environment/test-no-426-developmental-neurotoxicity-study_9789264067394-en (accessed on 12 September 2023).
- S.5002—FDA Modernization Act 2.0. Available online: www.congress.gov/bill/117th-congress/senate-bill/5002 (accessed on 12 September 2023).
- Draft Guidance on the Evaluation of Data from the Developmental Neurotoxicity (DNT) In-Vitro Testing Battery. Available online: https://www.oecd.org/chemicalsafety/testing/draft-guidance-review-documents-monographs.htm (accessed on 12 September 2023).
- Aschner, M.; Ceccatelli, S.; Daneshian, M.; Fritsche, E.; Hasiwa, N.; Hartung, T.; Hogberg, H.T.; Leist, M.; Li, A.; Mundi, W.R.; et al. Reference compounds for alternative test methods to indicate developmental neurotoxicity (DNT) potential of chemicals: Example lists and criteria for their selection and use. ALTEX Altern. Anim. Exp. 2017, 34, 49–74. [Google Scholar] [CrossRef]
- Bal-Price, A.; Hogberg, H.T.; Crofton, K.M.; Daneshian, M.; FitzGerald, R.E.; Fritsche, E.; Heinonen, T.; Hougaard Bennekou, S.; Klima, S.; Piersma, A.H.; et al. Recommendation on test readiness criteria for new approach methods in toxicology: Exemplified for developmental neurotoxicity. ALTEX Altern. Anim. Exp. 2018, 35, 306–352. [Google Scholar] [CrossRef]
- Fritsche, E.; Crofton, K.M.; Hernandez, A.F.; Hougaard Bennekou, S.; Leist, M.; Bal-Price, A.; Reaves, E.; Wilks, M.F.; Terron, A.; Gourmelon, A. OECD/EFSA workshop on developmental neurotoxicity (DNT): The use of non-animal test methods for regulatory purposes. ALTEX Altern. Anim. Exp. 2017, 34, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Masjosthusmann, S.; Blum, J.; Bartmann, K.; Dolde, X.; Holzer, A.-K.; Stürzl, L.-C.; Keßel, E.H.; Förster, N.; Dönmez, A.; Klose, J.; et al. Establishment of an a priori protocol for the implementation and interpretation of an in-vitro testing battery for the assessment of developmental neurotoxicity. EFSA Support. Publ. 2020, 17, 1938E. [Google Scholar] [CrossRef]
- Bartmann, K.; Bendt, F.; Dönmez, A.; Haag, D.; Keßel, E.; Masjosthusmann, S.; Noel, C.; Wu, J.; Zhou, P.; Fritsche, E. A human iPSC-based in vitro neural network formation assay to investigate neurodevelopmental toxicity of pesticides. bioRxiv 2023. [Google Scholar] [CrossRef]
- Ishibashi, Y.; Nagafuku, N.; Kanda, Y.; Suzuki, I. Evaluation of neurotoxicity for pesticide-related compounds in human iPS cell-derived neurons using microelectrode array. Toxicol. In Vitro 2023, 93, 105668. [Google Scholar] [CrossRef]
- Lehmann, H.C.; Staff, N.P.; Hoke, A. Modeling chemotherapy induced peripheral neuropathy (CIPN) in vitro: Prospects and limitations. Exp. Neurol. 2020, 326, 113140. [Google Scholar] [CrossRef]
- Eldridge, S.; Guo, L.; Hamre, J. A Comparative Review of Chemotherapy-Induced Peripheral Neuropathy in In Vivo and In Vitro Models. Toxicol. Pathol. 2020, 48, 190–201. [Google Scholar] [CrossRef]
- Jamieson, S.M.F.; Liu, J.; Connor, B.; McKeage, M.J. Oxaliplatin causes selective atrophy of a subpopulation of dorsal root ganglion neurons without inducing cell loss. Cancer Chemother. Pharmacol. 2005, 56, 391–399. [Google Scholar] [CrossRef]
- Leelawat, K.; Narong, S.; Udomchaiprasertkul, W.; Leelawat, S.; Tungpradubkul, S. Inhibition of PI3K increases oxaliplatin sensitivity in cholangiocarcinoma cells. Cancer Cell Int. 2009, 9, 3. [Google Scholar] [CrossRef]
- Guo, L.; Hamre, J., III; Eldridge, S.; Behrsing, H.P.; Cutuli, F.M.; Mussio, J.; Davis, M. Editor’s Highlight: Multiparametric Image Analysis of Rat Dorsal Root Ganglion Cultures to Evaluate Peripheral Neuropathy-Inducing Chemotherapeutics. Toxicol. Sci. 2017, 156, 275–288. [Google Scholar] [CrossRef]
- Livni, L.; Lees, J.G.; Barkl-Luke, M.E.; Goldstein, D.; Moalem-Taylor, G. Dorsal root ganglion explants derived from chemotherapy-treated mice have reduced neurite outgrowth in culture. Neurosci. Lett. 2019, 694, 14–19. [Google Scholar] [CrossRef]
- Imai, S.; Koyanagi, M.; Azimi, Z.; Nakazato, Y.; Matsumoto, M.; Ogihara, T.; Yonezawa, A.; Omura, T.; Nakagawa, S.; Wakatsuki, S.; et al. Taxanes and platinum derivatives impair Schwann cells via distinct mechanisms. Sci. Rep. 2017, 7, 5947. [Google Scholar] [CrossRef] [PubMed]
- Kusumoto, D.; Seki, T.; Sawada, H.; Kunitomi, A.; Katsuki, T.; Kimura, M.; Ito, S.; Komuro, J.; Hashimoto, H.; Fukuda, K.; et al. Anti-senescent drug screening by deep learning-based morphology senescence scoring. Nat. Commun. 2021, 12, 257. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Matsuda, N.; Matsuda, K.; Yamanaka, M.; Suzuki, I. An in vitro microfluidic culture device for peripheral neurotoxicity prediction at low concentrations based on deep learning. Fundam. Toxicol. Sci. 2022, 9, 203–209. [Google Scholar] [CrossRef]
- Yamanaka, M.; Wen, X.; Imamura, S.; Sakai, R.; Terada, S.; Kamei, K.I. Cyclo olefin polymer-based solvent-free mass-productive microphysiological systems. Biomed. Mater. 2021, 16, 35009. [Google Scholar] [CrossRef]
- Ta, L.E.; Espeset, L.; Podratz, J.; Windebank, A.J. Neurotoxicity of oxaliplatin and cisplatin for dorsal root ganglion neurons correlates with platinum–DNA binding. NeuroToxicology 2006, 27, 992–1002. [Google Scholar] [CrossRef]
- National Research Council. Guide for the Care and Use of Laboratory Animals: Eighth Edition; The National Academies Press: Washington, DC, USA, 2011. [Google Scholar] [CrossRef]
- Selvaraju, R.R.; Cogswell, M.; Das, A.; Vedantam, R.; Parikh, D.; Batra, D. Grad-cam: Visual explanations from deep networks via gradient-based localization. In Proceedings of the IEEE International Conference on Computer Vision, Venice, Italy, 22–29 October 2017; pp. 618–626. [Google Scholar]
- Scuteri, A.; Nicolini, G.; Miloso, M.; Bossi, M.; Cavaletti, G.; Windebank, A.J.; Tredici, G. Paclitaxel toxicity in post-mitotic dorsal root ganglion (DRG) cells. Anticancer Res. 2006, 26, 1065–1070. [Google Scholar]
- Monici, M. Cell and tissue autofluorescence research and diagnostic applications. In Biotechnology Annual Review; Elsevier: Amsterdam, The Netherlands, 2005; Volume 11, pp. 227–256. [Google Scholar]
- Gill, J.S.; Windebank, A.J. Suramin induced ceramide accumulation leads to apoptotic cell death in dorsal root ganglion neurons. Cell Death Differ. 1998, 5, 876–883. [Google Scholar] [CrossRef]
- Geisler, S.; Doan, R.A.; Cheng, G.C.; Cetinkaya-Fisgin, A.; Huang, S.X.; Höke, A.; Milbrandt, J.; DiAntonio, A. Vincristine and bortezomib use distinct upstream mechanisms to activate a common SARM1-dependent axon degeneration program. JCI Insight 2019, 4, 17:1–17:17. [Google Scholar] [CrossRef]
- Wang, M.-S.; Wu, Y.; Culver, D.G.; Glass, J.D. Pathogenesis of Axonal Degeneration: Parallels between Wallerian Degeneration and Vincristine Neuropathy. J. Neuropathol. Exp. Neurol. 2000, 59, 599–606. [Google Scholar] [CrossRef]
- Gu, H.; Wang, C.; Li, J.; Yang, Y.; Sun, W.; Jiang, C.; Li, Y.; Ni, M.; Liu, W.-T.; Cheng, Z.; et al. High mobility group box-1-toll-like receptor 4-phosphatidylinositol 3-kinase/protein kinase B-mediated generation of matrix metalloproteinase-9 in the dorsal root ganglion promotes chemotherapy-induced peripheral neuropathy. Int. J. Cancer 2020, 146, 2810–2821. [Google Scholar] [CrossRef]
- Nishida, K.; Kuchiiwa, S.; Oiso, S.; Futagawa, T.; Masuda, S.; Takeda, Y.; Yamada, K. Up-regulation of matrix metalloproteinase-3 in the dorsal root ganglion of rats with paclitaxel-induced neuropathy. Cancer Sci. 2008, 99, 1618–1625. [Google Scholar] [CrossRef] [PubMed]
- Russell, J.W.; Windebank, A.J.; Podratz, J.L. Role of nerve growth factor in suramin neurotoxicity studied in vitro. Ann. Neurol. 1994, 36, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Hertz, D.L.; Kidwell, K.M.; Vangipuram, K.; Li, F.; Pai, M.P.; Burness, M.; Griggs, J.J.; Schott, A.F.; Van Poznak, C.; Hayes, D.F.; et al. Paclitaxel Plasma Concentration after the First Infusion Predicts Treatment-Limiting Peripheral Neuropathy. Clin. Cancer Res. 2018, 24, 3602–3610. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Haacke, E.M.; Li, J. Peripheral nerve magnetic resonance imaging. F1000Research 2019, 8, 1803. [Google Scholar] [CrossRef]
- Sałat, K. Chemotherapy-induced peripheral neuropathy—Part 2: Focus on the prevention of oxaliplatin-induced neurotoxicity. Pharmacol. Rep. 2020, 72, 508–527. [Google Scholar] [CrossRef]
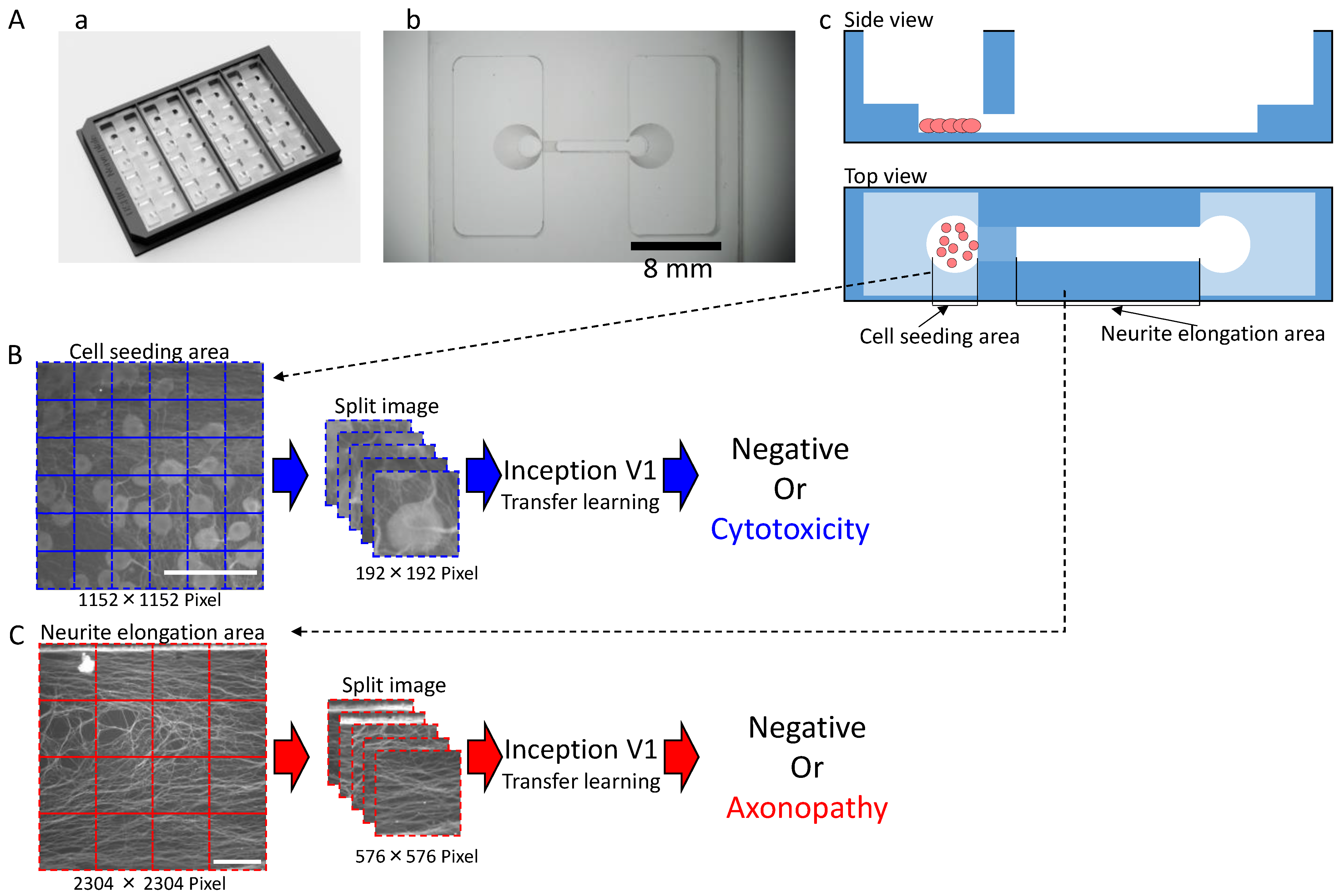
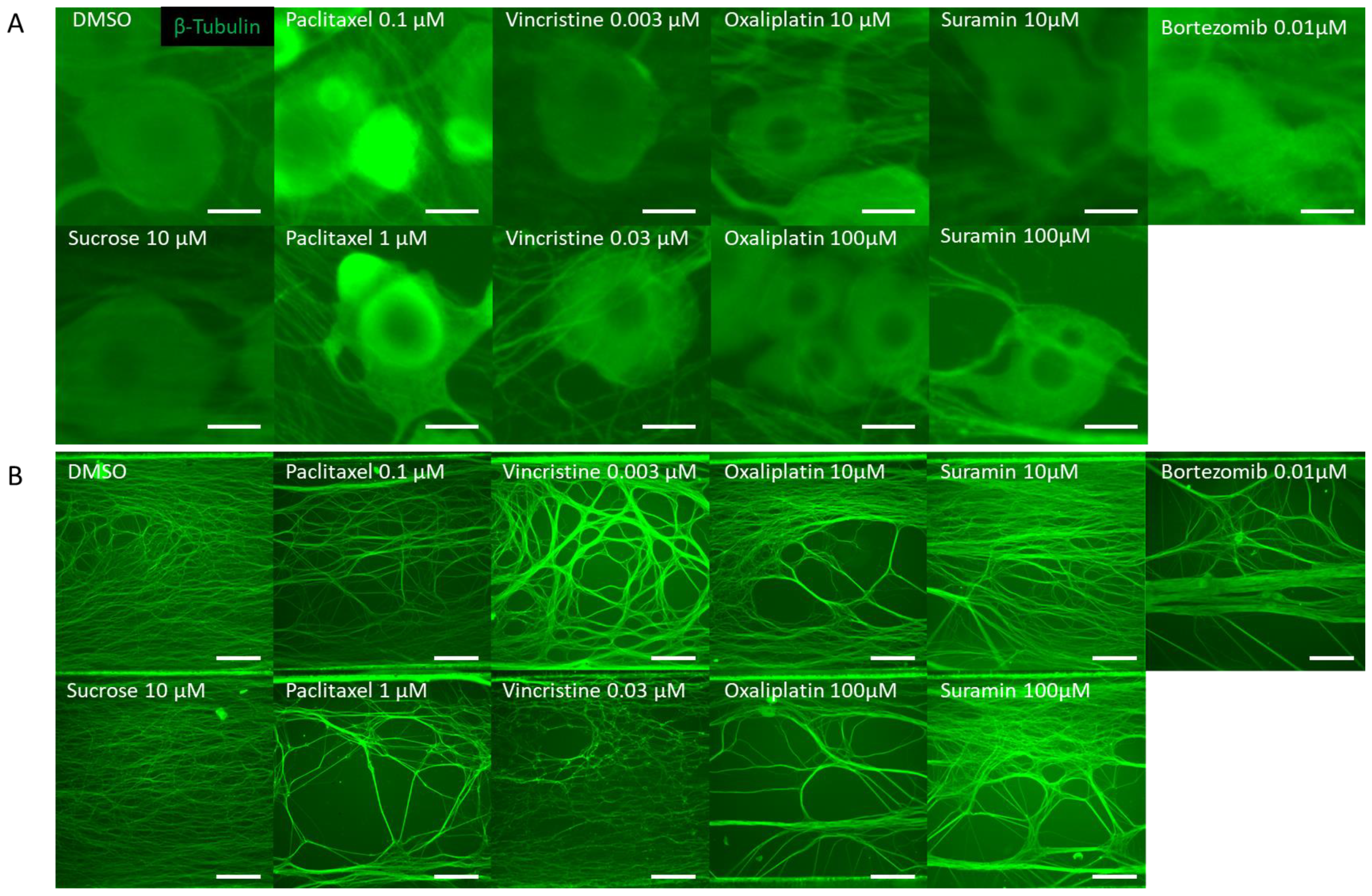
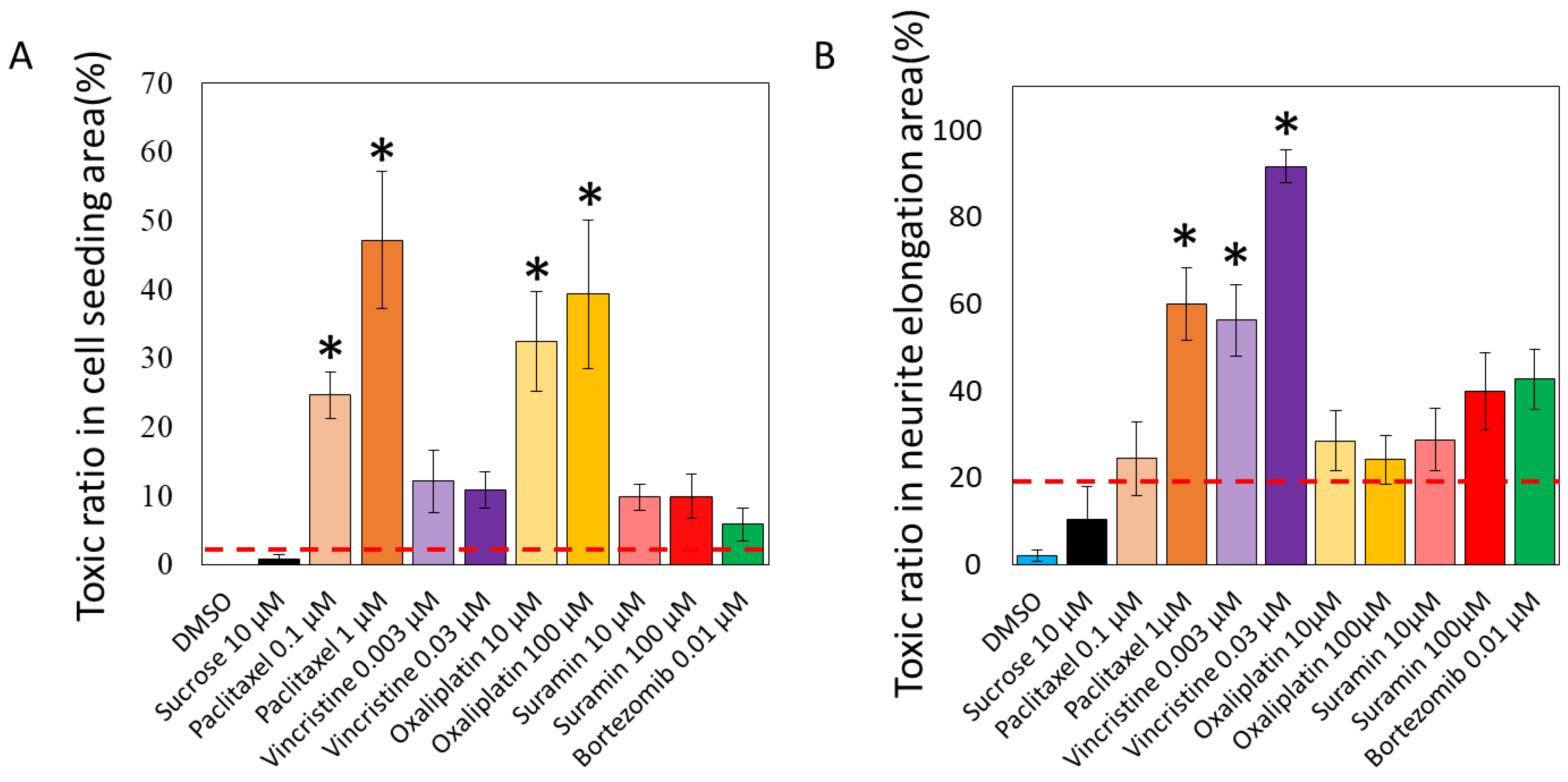
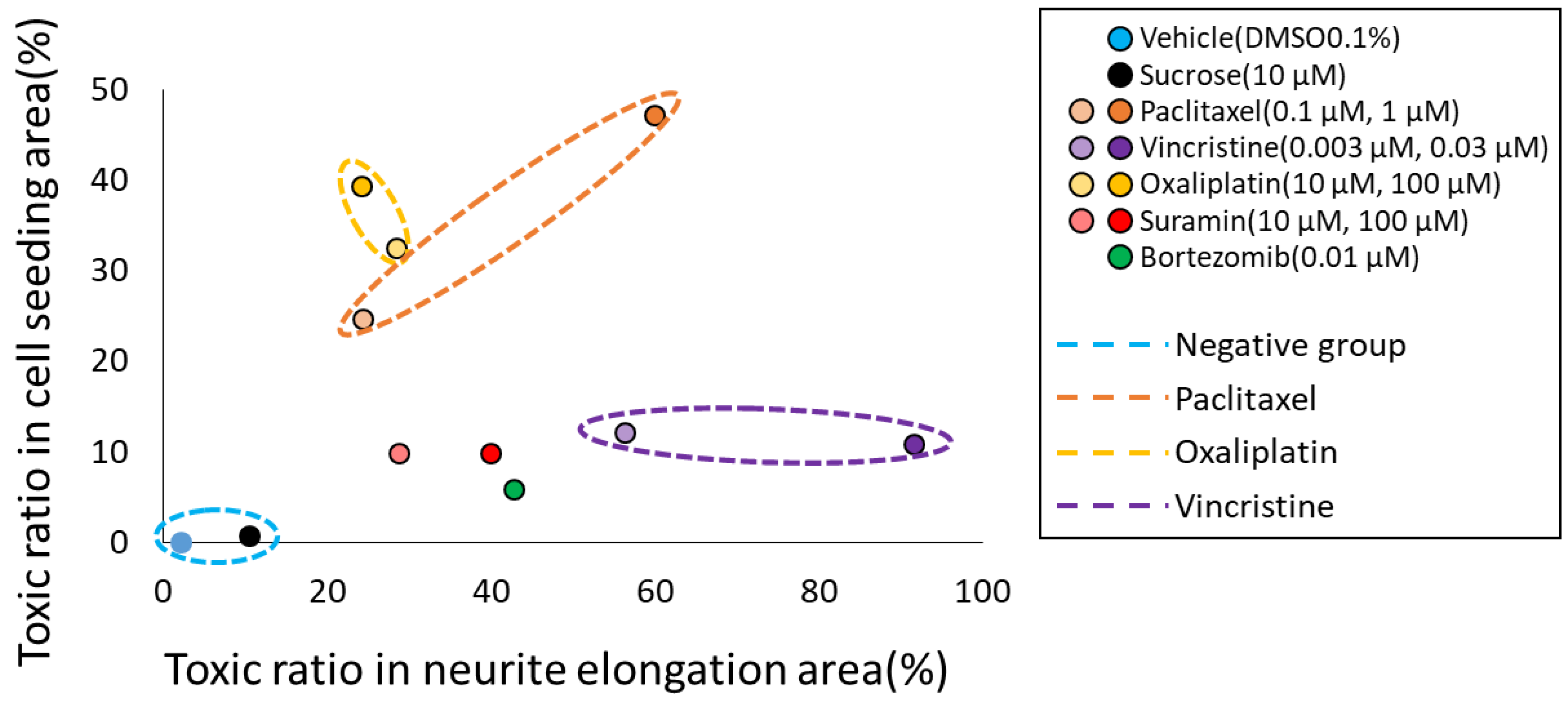
| Training Dataset | Testing Dataset | |||
|---|---|---|---|---|
| Compounds | Concentration (µM) | n (Images) | Concentration (µM) | n (Images) |
| DMSO | 0.10% | 24 | 0.10% | 8 |
| Sucrose | 10 | 24 | 10 | 12 |
| Oxaliplatin | 10 | 12 | 10 | 8 |
| 100 | 8 | 100 | 8 | |
| Paclitaxel | - | - | 0.1 | 38 |
| - | - | 1 | 10 | |
| Vincristine | - | - | 0.003 | 13 |
| - | - | 0.03 | 24 | |
| Suramin | - | - | 10 | 20 |
| - | - | 100 | 16 | |
| Bortezomib | - | - | 0.01 | 8 |
| Training Dataset | Testing Dataset | |||
|---|---|---|---|---|
| Compounds | Concentration (µM) | n (Images) | Concentration (µM) | n (Images) |
| DMSO | 0.10% | 10 | 0.10% | 6 |
| Sucrose | 10 | 7 | 10 | 3 |
| Vincristine | 0.003 | 8 | 0.003 | 5 |
| 0.03 | 9 | 0.03 | 6 | |
| Paclitaxel | - | - | 0.1 | 10 |
| - | - | 1 | 10 | |
| Oxaliplatin | - | - | 10 | 16 |
| - | - | 100 | 15 | |
| Suramin | - | - | 10 | 10 |
| - | - | 100 | 13 | |
| Bortezomib | - | - | 0.01 | 6 |
| Compounds | Concentration (µM) | vs. DMSO | vs. Sucrose | vs. Paclitaxel | vs. Vincristine | vs. Oxaliplatin | vs. Suramin | vs. Bortezomib | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.10% | 10 | 0.1 | 1 | 0.003 | 0.03 | 10 | 100 | 10 | 100 | 0.01 | ||
| DMSO | 0.10% | ― | ** p = 2.66 × 10−6 | ** p = 1.48 × 10−21 | ** p = 4.89 × 10−40 | ** p = 7.18 × 10−43 | ** p = 1.61 × 10−136 | ** p = 3.77 × 10−29 | ** p = 8.27 × 10−23 | ** p = 1.53 × 10−24 | ** p = 1.54 × 10−19 | ** p = 6.81 × 10−58 |
| Sucrose | 10 | ** p = 2.66 × 10−06 | ― | ** p = 4.51 × 10−09 | ** p = 3.91 × 10−21 | ** p = 3.71 × 10−21 | ** p = 1.56 × 10−87 | ** p = 4.59 × 10−15 | ** p = 2.24 × 10−10 | ** p = 1.36 × 10−09 | ** p = 7.86 × 10−08 | ** p = 8.91 × 10−24 |
| Paclitaxel | 0.1 | ** p = 1.48 × 10−21 | ** p = 4.51 × 10−09 | ― | ** p = 4.01 × 10−42 | ** p = 1.54 × 10−23 | ** p = 2.24 × 10−124 | ** p = 4.18 × 10−04 | ** p = 9.88 × 10−09 | ** p = 3.02 × 10−21 | ** p = 6.33 × 10−26 | ** p = 5.81 × 10−14 |
| 1 | ** p = 4.89 × 10−40 | ** p = 3.91 × 10−21 | ** p = 4.01 × 10−42 | ― | ** p = 3.89 × 10−14 | ** p = 3.51 × 10−48 | ** p = 8.12 × 10−18 | ** p = 2.56 × 10−23 | ** p = 9.51 × 10−55 | ** p = 7.30 × 10−41 | ** p = 2.43 × 10−18 | |
| Vincristine | 0.003 | ** p = 7.18 × 10−43 | ** p = 3.71 × 10−21 | ** p = 1.54 × 10−23 | ** p = 3.89 × 10−14 | ― | ** p = 4.18 × 10−50 | ** p = 6.00 × 10−20 | ** p = 6.80 × 10−26 | ** p = 1.78 × 10−17 | ** p = 1.55 × 10−04 | ** p = 2.08 × 10−07 |
| 0.03 | ** p = 1.61 × 10−136 | ** p = 1.56 × 10−87 | ** p = 2.24 × 10−124 | ** p = 3.51 × 10−48 | ** p = 4.18 × 10−50 | ― | ** p = 4.96 × 10−81 | ** p = 2.18 × 10−103 | ** p = 1.96 × 10−106 | ** p = 1.19 × 10−56 | ** p = 5.01 × 10−85 | |
| Oxaliplatin | 10 | ** p = 3.77 × 10−29 | ** p = 4.59 × 10−15 | ** p = 4.18 × 10−04 | ** p = 8.12 × 10−18 | ** p = 6.00 × 10−20 | ** p = 4.96 × 10−81 | ― | * p = 0.040 | ** p = 1.50 × 10−38 | ** p = 6.17 × 10−32 | ** p = 2.41 × 10−19 |
| 100 | ** p = 8.27 × 10−23 | ** p = 2.24 × 10−10 | ** p = 9.88 × 10−09 | ** p = 2.56 × 10−23 | ** p = 6.80 × 10−26 | ** p = 2.18 × 10−103 | * p = 0.040 | ― | ** p = 7.09 × 10−33 | ** p = 6.22 × 10−32 | ** p = 1.64 × 10−17 | |
| Suramin | 10 | ** p = 1.53 × 10−24 | ** p = 1.36 × 10−09 | ** p = 3.02 × 10−21 | ** p = 9.51 × 10−55 | ** p = 1.78 × 10−17 | ** p = 1.96 × 10−106 | ** p = 1.50 × 10−38 | ** p = 7.09 × 10−33 | ― | ** p = 1.97 × 10−04 | ** p = 1.20 × 10−06 |
| 100 | ** p = 1.54 × 10−19 | ** p = 7.86 × 10−08 | ** p = 6.33 × 10−26 | ** p = 7.30 × 10−41 | ** p = 1.55 × 10−04 | ** p = 1.19 × 10−56 | ** p = 6.17 × 10−32 | ** p = 6.22 × 10−32 | ** p = 1.97 × 10−04 | ― | p = 0.067 | |
| Bortezomib | 0.01 | ** p = 6.81 × 10−58 | ** p = 8.91 × 10−24 | ** p = 5.81 × 10−14 | ** p = 2.43 × 10−18 | ** p = 2.08 × 10−07 | ** p = 5.01 × 10−85 | ** p = 2.41 × 10−19 | ** p = 1.64 × 10−17 | ** p = 1.20 × 10−06 | p = 0.067 | ― |
| Toxicity Detection Concentration (µM) | Toxicity Detection (This Work) | Reference | |||||
|---|---|---|---|---|---|---|---|
| Compound | Concentration Tested (µM) | Cell | Axon | Cell | Axon | MANOVA | |
| Previous Report | Previous Report | ||||||
| Paclitaxel | 0.1 | over 0.1 | Over 0.01 | ◎ | ○ | ◎ | [31] |
| 1 | ◎ | ◎ | ◎ | ||||
| Vincristine | 0.003 | 24 h no effect | 24 h Over 0.001 | ○ | ◎ | ◎ | [23] |
| 0.03 | 72 h 0.1 | ○ | ◎ | ◎ | |||
| Oxaliplatin | 10 | Over 10 | Over 3.3 | ◎ | ○ | ◎ | [29] |
| 100 | ◎ | ○ | ◎ | ||||
| Suramin | 10 | 48 h 300 | 48 h 200 | ○ | ○ | ◎ | [33,38] |
| 100 | 8 day Over 100 | ○ | ○ | ◎ | |||
| Bortezomib | 0.01 | 24 h no effect | 24 h 1 | ○ | ○ | ◎ | [23] |
| 72 h 0.012 | 72 h 0.03 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsuda, K.; Han, X.; Matsuda, N.; Yamanaka, M.; Suzuki, I. Development of an In Vitro Assessment Method for Chemotherapy-Induced Peripheral Neuropathy (CIPN) by Integrating a Microphysiological System (MPS) with Morphological Deep Learning of Soma and Axonal Images. Toxics 2023, 11, 848. https://doi.org/10.3390/toxics11100848
Matsuda K, Han X, Matsuda N, Yamanaka M, Suzuki I. Development of an In Vitro Assessment Method for Chemotherapy-Induced Peripheral Neuropathy (CIPN) by Integrating a Microphysiological System (MPS) with Morphological Deep Learning of Soma and Axonal Images. Toxics. 2023; 11(10):848. https://doi.org/10.3390/toxics11100848
Chicago/Turabian StyleMatsuda, Kazuki, Xiaobo Han, Naoki Matsuda, Makoto Yamanaka, and Ikuro Suzuki. 2023. "Development of an In Vitro Assessment Method for Chemotherapy-Induced Peripheral Neuropathy (CIPN) by Integrating a Microphysiological System (MPS) with Morphological Deep Learning of Soma and Axonal Images" Toxics 11, no. 10: 848. https://doi.org/10.3390/toxics11100848
APA StyleMatsuda, K., Han, X., Matsuda, N., Yamanaka, M., & Suzuki, I. (2023). Development of an In Vitro Assessment Method for Chemotherapy-Induced Peripheral Neuropathy (CIPN) by Integrating a Microphysiological System (MPS) with Morphological Deep Learning of Soma and Axonal Images. Toxics, 11(10), 848. https://doi.org/10.3390/toxics11100848





