Abstract
Background: Burkitt lymphoma (BL) is an aggressive non-Hodgkin lymphoma (NHL), subdivided into endemic, sporadic, and immunodeficiency-associated forms. While jaw lesions are common in endemic BL, they are infrequent in sporadic cases, only rarely constituting the first manifestation of the disease. The aim of this study is to present a rare pediatric case of sporadic BL first manifesting as gingival swellings and tooth hypermobility and provide a review of all the published sporadic BL case reports as the first sign of disease. Case report: An 11-year-old Caucasian female was referred for the evaluation of hypermobility of posterior lower teeth, associated with painful gingival swellings of 20 days duration. Clinical examination revealed right facial asymmetry and bilateral prominent swellings of the posterior lower gingiva. A panoramic radiograph revealed ill-defined radiolucent lesions in the posterior mandible bilaterally. On computed tomography, soft-tissue masses were identified along the mandibular ramus extending into the maxillary sinus bilaterally. The histopathologic and immunohistochemical analyses of the lesions led to a diagnosis of Burkitt lymphoma (BL). The patient underwent a full staging work-up, revealing bone marrow involvement and widespread disease. A multi-chemotherapy regimen was initiated with the regression of oral lesions and symptoms within a few weeks and complete disease remission after nine chemotherapy cycles. The patient remains free of disease 11 years later. Conclusions: This case underscores the critical importance of the timely diagnosis and life-saving referral of rapidly growing jaw lesions, which may represent the first sign of an underlying lymphoreticular malignancy with aggressive course, such as BL.
1. Introduction
Non-Hodgkin lymphomas (NHLs) account for about 4% of all malignant diseases in children younger than 15 years old [1]. Burkitt lymphoma (BL) is an aggressive non-Hodgkin B cell lymphoma with the potential to involve multiple organ systems, characterized by the translocation of the MYC oncogene [2].
BL was first described in 1958 by Denis Burkitt as a mandibular malignancy in African children [3,4]. Over time, the recognition of cases worldwide indicated its universal relevance. It is subdivided into three clinical variants with distinct epidemiologic, immunologic, and molecular/cytogenetic characteristics: endemic, sporadic, and immunodeficiency-associated [5].
The most common variant is endemic BL which accounts for 30 to 50% of all childhood cancers in equatorial Africa, with an estimated incidence of 3 to 6 cases per 100,000 children per year [2,6]. It has a peak incidence between ages 6–8 and a male predominance. It appears more frequently in the jaws and Epstein–Barr virus (EBV) is detected in around 95% of the cases [3,7].
Sporadic BL is a rare malignancy among Western populations, with a reported annual incidence of 2–3 cases per million. Still, it accounts for approximately 30–50% of childhood lymphomas, but less than 1% of all NHL in adults [2,8,9]. Sporadic BL appears more frequently in the abdominal region and EBV is detected in about 10–30% of the cases [3].
Immunodeficiency-associated BL is mostly associated with HIV infection, accounting for 20–40% of HIV-associated NHLs [10]. In fact, BL is thought to appear early in HIV infection, before CD4+ cell numbers drop [3,11,12]. This type of BL can also be seen in immunosuppressed allograft recipients and patients with congenital immunodeficiency. It more frequently develops in lymph nodes, bone marrow, and central nervous system [2,5].
BL shows a broad spectrum of clinical manifestations, usually with multi-organ involvement. The African or endemic variant often involves the jaws and other facial bones, while head and neck manifestations in sporadic BL are considered rare [2].
The aim of the present manuscript is to describe a case of sporadic BL with jaw involvement, manifesting with painful gingival swellings and tooth hypermobility as the first signs of the underlying widespread disease. Moreover, the findings of all the published case reports of sporadic BL with oral involvement as first occurrence are summarized in order to enhance the diagnosis of similar cases.
2. Case Presentation
An 11-year-old female was referred for the evaluation of bilateral painful swellings on the gingiva of the lower premolars and molars, associated with tooth hypermobility. The painful symptoms had started 3 weeks before and were initially attributed to erupting second permanent mandibular molars; however, they were progressively worsening, accompanied by difficulty in swallowing, general malaise, and weight loss. Further, a noticeable, right facial asymmetry had also developed over the last 3 days. The patient had been examined by a general dentist and an ENT specialist, who had prescribed antibiotic treatment (amoxicillin for 7 days), analgesics, and antiseptic mouthwashes, without improvement. Following consultation with a periodontist, the child was referred to a specialized Oral Medicine Clinic.
The patient’s medical history was unremarkable and a blood test, performed on the same day, was within normal limits. No fever or lymphadenopathy was present.
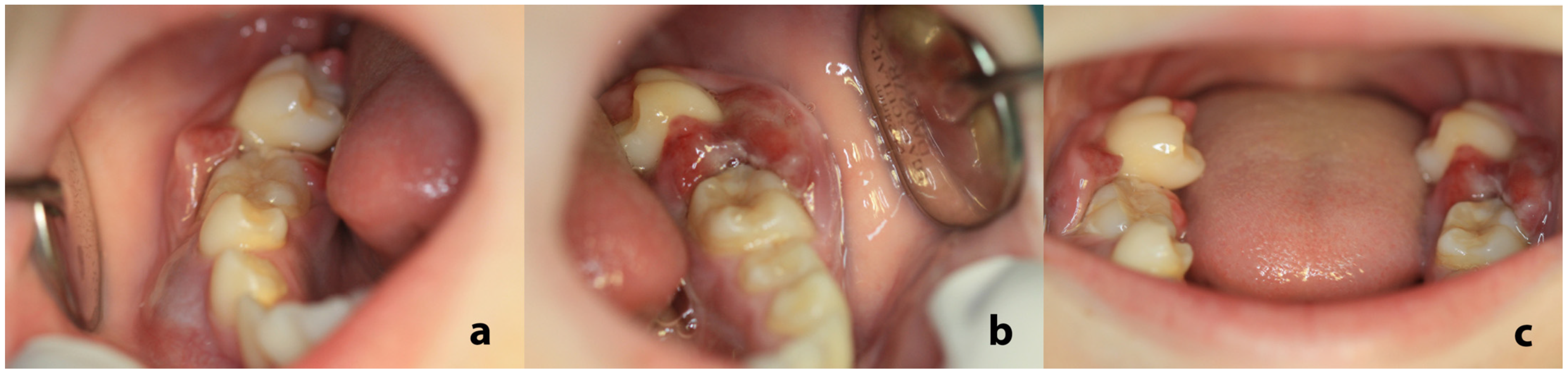
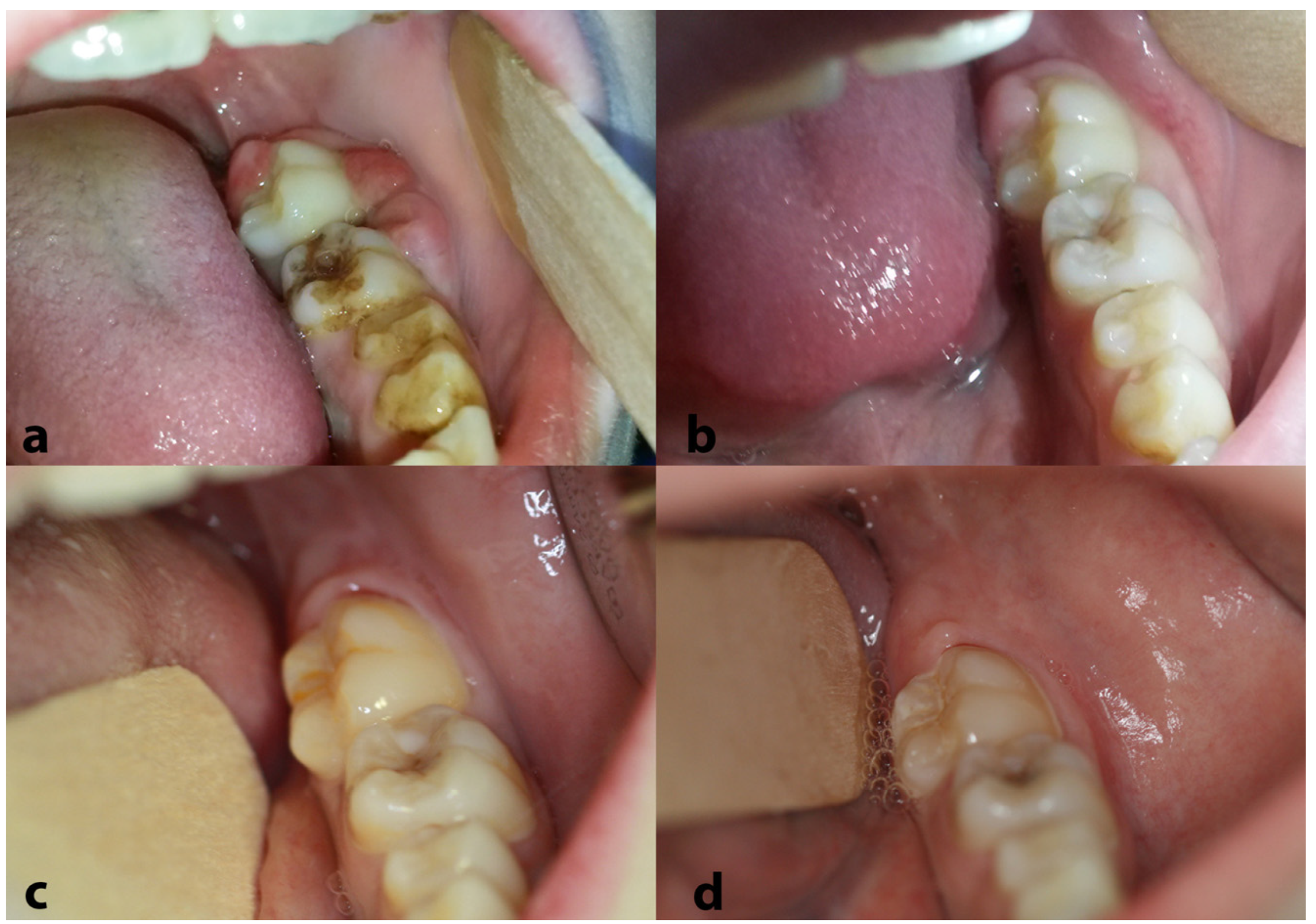
On clinical examination, an extraoral facial swelling along the right posterior mandible was noticeable. Intraorally, bilateral prominent swellings were identified, bilaterally affecting the mandibular premolar and molar gingiva, which were erythematous and focally ulcerated (Figure 1a,b). The aforementioned teeth were extremely hypermobile, some of them displaced and partially extruded (Figure 1c).
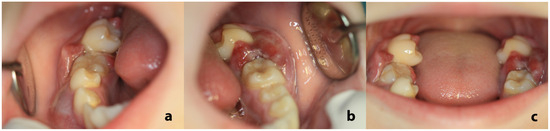
Figure 1.
Intraoral examination: prominent gingival swelling adjacent to the premolars and molars of the right (a) and left (b) mandible; the gingiva were erythematous and focally ulcerated. The aforementioned teeth, especially the second molars, were hypermobile, displaced, and partially extruded (c).
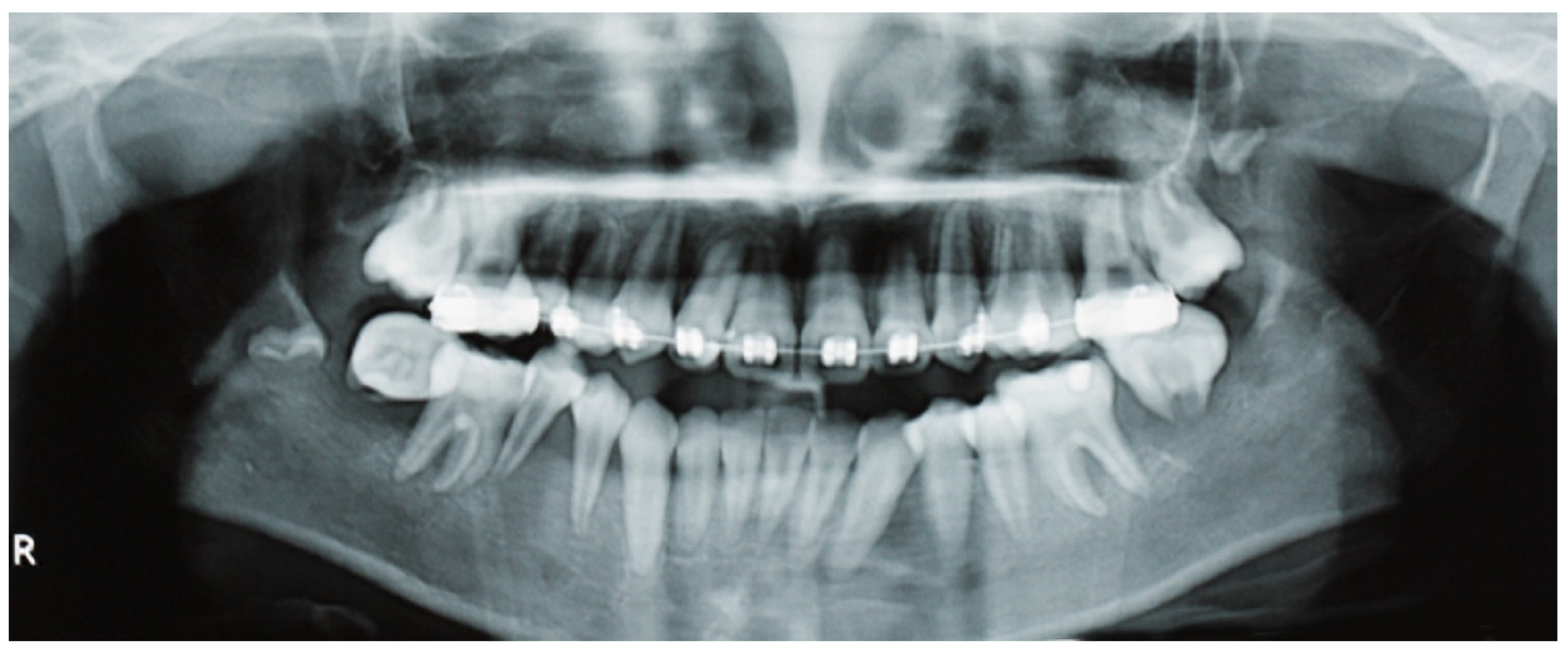
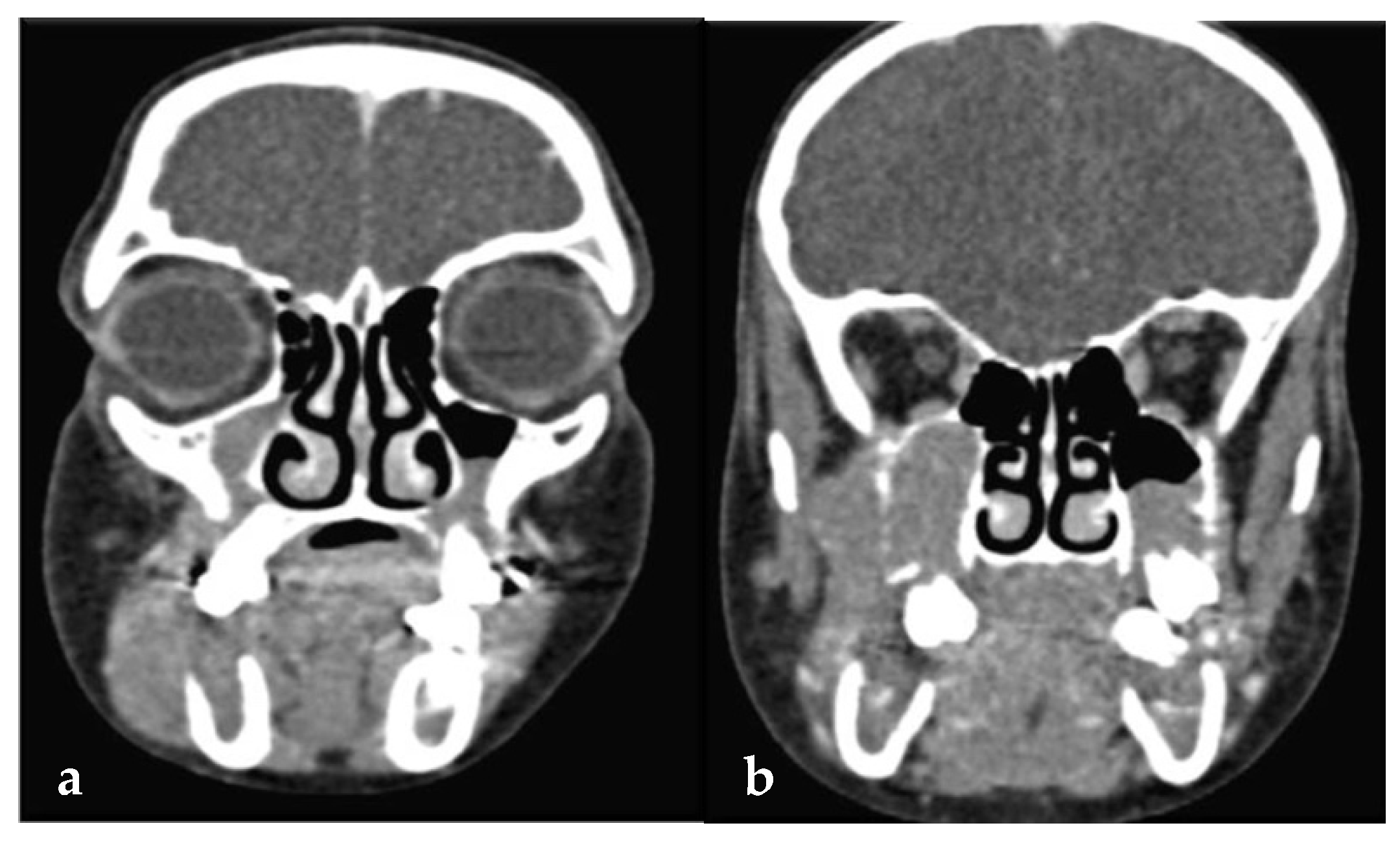
A panoramic radiograph revealed ill-defined radiolucencies in the posterior mandible bilaterally, related to the adjacent permanent teeth, which also showed a ‘floating teeth’ appearance (Figure 2). Moreover, computed tomography showed hypodense (soft tissue) masses along and distal to the mandibular ramus extending upwards into the maxillary sinuses, bilaterally (with dimensions of 1.5 × 3.0 × 7.0 cm and 1.7 × 1.2 × 3.5 cm on the right and left side, respectively), as well as the posterior wall of the right orbit; also, there was a perforation of the right mandibular cortex along the second molar tooth (Figure 3 a,b). Based on the clinical and imaging findings, the differential diagnosis mainly included hematologic neoplastic conditions, such as lymphomas, leukemias, and Langerhans cell disease; the possibility of sarcomas, such as rhabdomyosarcoma or Ewing sarcoma, was also considered, especially considering the patient’s age.
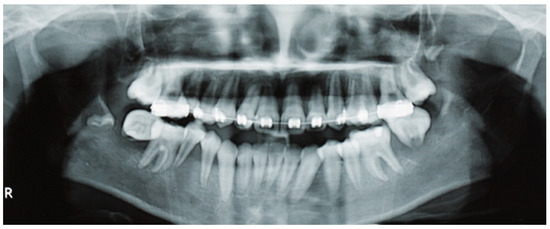
Figure 2.
Panoramic radiography shows ill-defined radiolucencies in the posterior mandible bilaterally, affecting the adjacent permanent teeth that resemble a ‘floating teeth’ appearance. In this particular focal trough, the osteolysis appears to be more diffuse in the right mandible, extending into the premolar region and also causing the displacement of the sperm of the third molar.
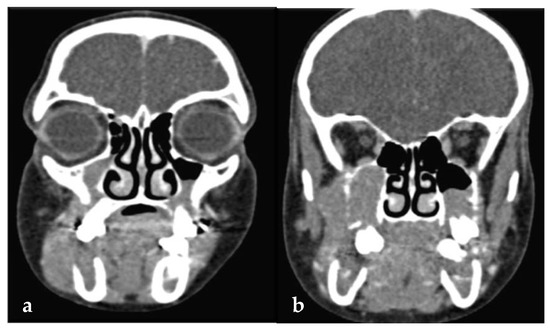
Figure 3.
(a,b): Computed tomography showing hypodense (soft tissue) masses along and distal to the mandibular ramus extending into the maxillary sinus, bilaterally.
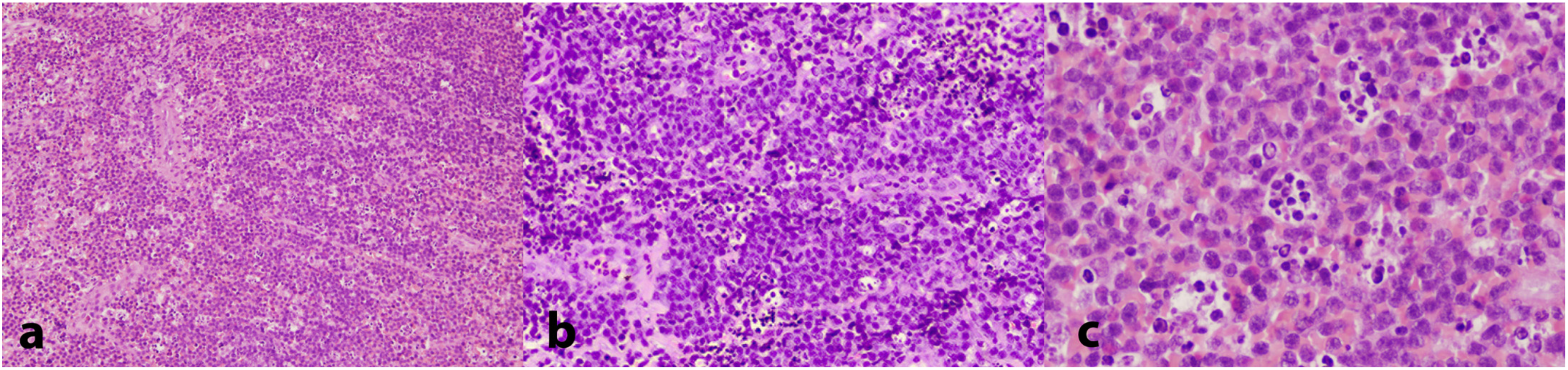
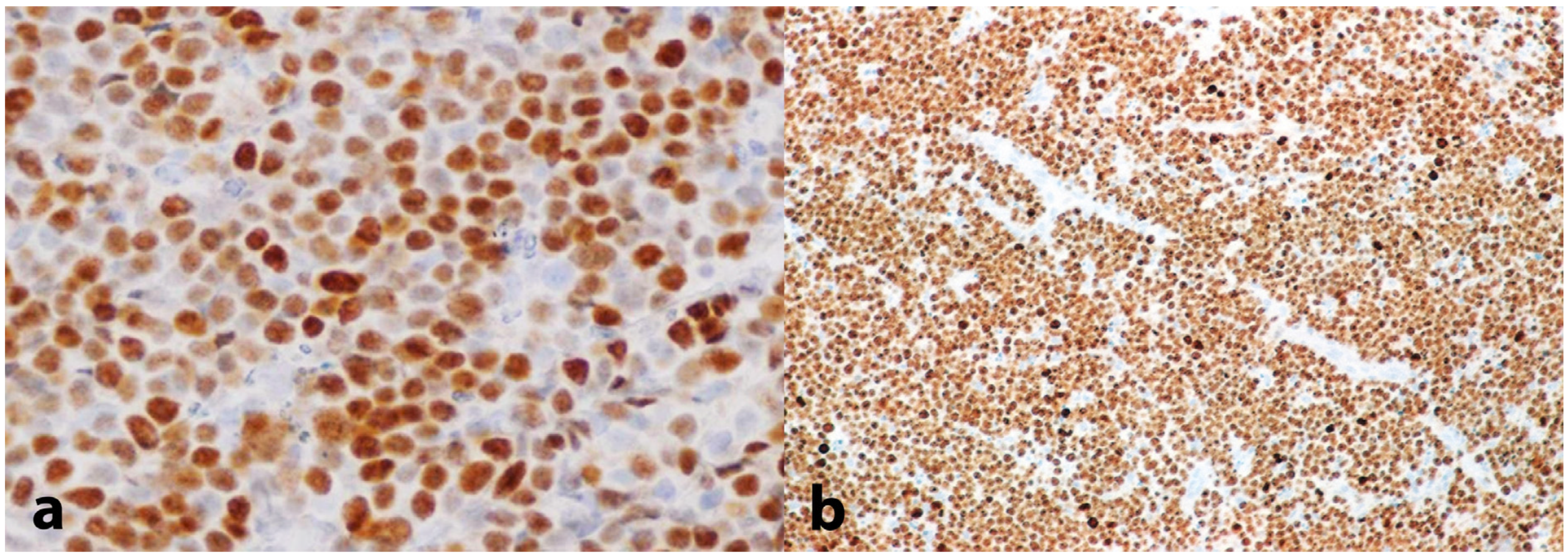
Taking into account the aggressive presentation, a partial biopsy of the affected gingiva in the area of the right second mandibular molar was deemed as an imminent need and was performed on the day of the first visit. Histopathologic findings revealed a dense proliferation of lymphocytes of medium size with minimum cytoplasm and round nuclei with multiple nucleoli, exhibiting a significant mitotic rate and dispersed apoptotic bodies; in addition, abundant indispersed macrophages, generating a ‘starry sky’ appearance, were noticed (Figure 4a–c). An immunohistochemical analysis revealed that the tumor cells were positive for CD20, CD79a, CD10, Bcl-6, and Bmyc (Figure 5a); in contrast, they were negative for Bcl-2, Tdt, Cyclin-D1, MUM1, and myeloperoxidase, while CD3 and CD5 were positive only in a small number of reactive T lymphocytes. In addition, there was kappa light chain restriction, while the Ki-67 cell proliferation index was positive in almost all the neoplastic cells (Figure 5b). Based on the histopathologic and immunohistochemical findings, a final diagnosis of BL, sporadic type, was rendered.
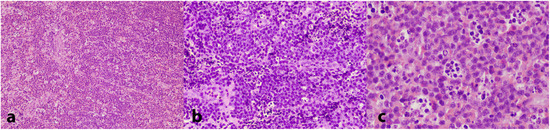
Figure 4.
Histopathologic examination of the biopsy specimen showing a dense infiltrate of lymphocytes of medium size with minimum cytoplasm and round nuclei exhibiting a significant mitotic rate; a ‘starry sky’ pattern, due to abundant indispersed macrophages, is apparent. (H&E stain, (a): 25×; (b): 100×; (c): 200×).
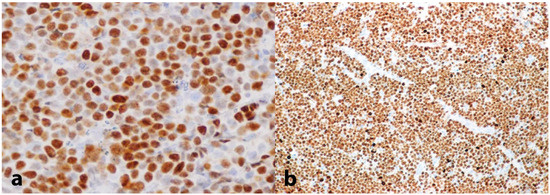
Figure 5.
Immunohistochemical evaluation showing (a) intense positivity for B-myc (200×) and (b) a Ki-67 proliferation index approximating 100% (25×).
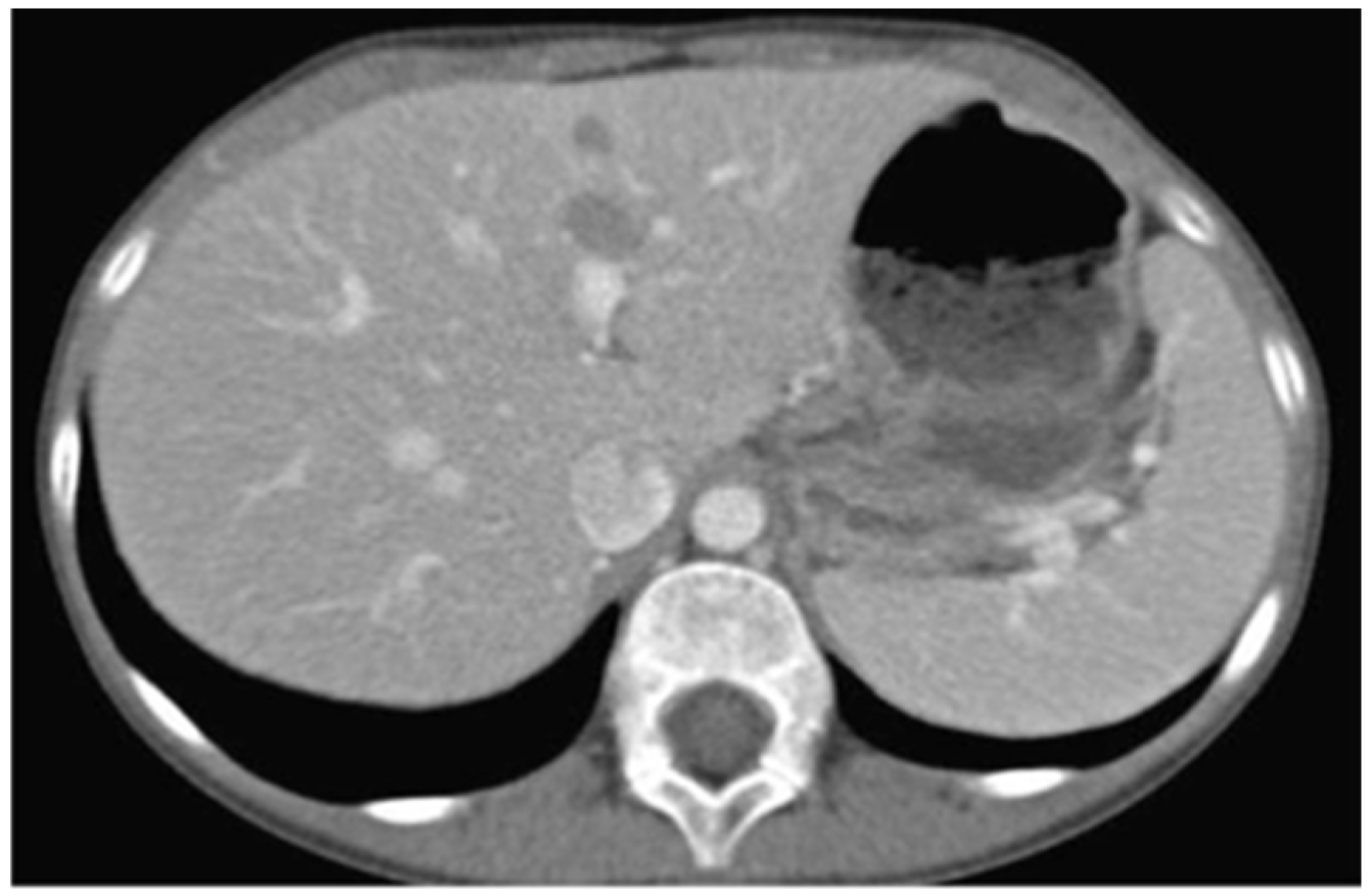
The patient was promptly referred to the Department of Pediatric Oncology of the Children’s Hospital for complete work-up and staging. Full body CT and PET/CT scanning revealed multiple lesions in the skeleton (including the skull, vertebrae, ribs, humerus, femur, and pelvis), liver (Figure 6), and pelvic lymph nodes. Bone marrow aspiration revealed infiltration (15–20%) by neoplastic lymphocytes. The disease was classified as stage IV without CNS involvement.
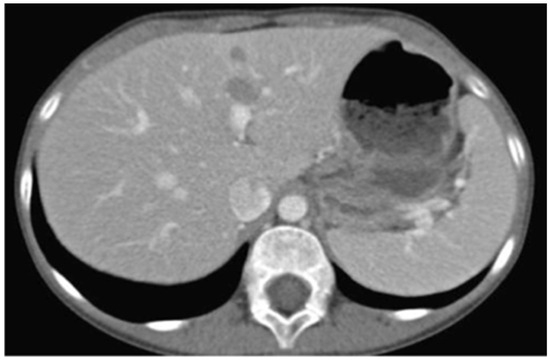
Figure 6.
Abdominal CT showing diffuse tumor infiltration of the liver.
The patient stratified into Group B according to the staging of the disease and received treatment according to the FAB-LMB 96 protocol. Treatment included nine cycles of chemotherapy, with 21-day intervals. Due to inadequate response after the fourth cycle of chemotherapy, the patient was switched to Group C, Arm C1, and the patient achieved Complete Remission at the end of the treatment.
Regarding oral lesions and symptoms, rapid regression was noted right after the initiation of the first chemotherapy cycle (Figure 7a,b). At the end of the chemotherapy, a PET/CT scan confirmed that the patient was free of disease; regular and thorough follow-up, including PET/CT, magnetic resonance imaging (MRI) of the brain and visceral skull, ultrasonography (U/S) of upper and lower abdomen and/or chest X-ray, and appropriate blood tests, confirmed sustained complete remission. Similarly, the oral cavity remained free of lesions during the long-term follow-up (Figure 7c,d). Eleven years later, the patient remains free of disease and totally healthy.
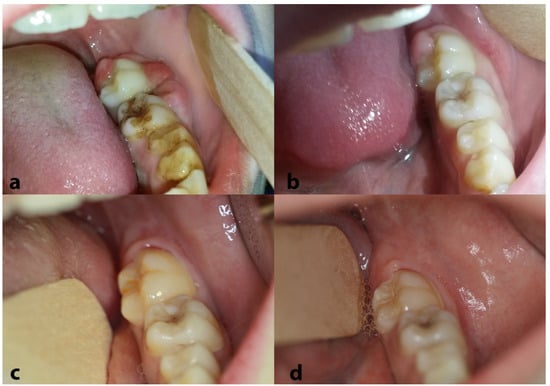
Figure 7.
Clinical appearance of the lesions in the left posterior mandible after 10 days (a) and 15 days (b) from the biopsy; please note the significant regression just a few days following the beginning of chemotherapy. No relapses were noticed during the long-term follow-up [representative clinical views at 9 months (c) and 32 months (d) from the diagnosis].
3. Discussion
BL derives from germinal center B cells and its three subtypes are thought to arise from these cells at different stages of their development [2]. BLs are composed of monomorphic CD19+; CD20+, commonly IgM+ B cells, exhibiting basophilic cytoplasm; numerous mitotic figures; and a Ki67 score of more than 95% [3]. The tumor cells exhibit a high degree of apoptosis, and a ‘starry sky’ pattern is seen at low magnification in H&E-stained slides [13]. The ‘sky’ is formed by round basophilic cells without intercellular stroma and the ‘stars’ correspond to numerous, scattered benign histiocytes [14].
Although the histopathologic appearance is the same, the three BL subtypes vary in clinical features and, especially, age predilection and area of distribution. The endemic form develops in Africa and occurs in early childhood, while the sporadic form shows no geographic predilection and occurs more commonly in children and adolescents. The endemic form mostly involves the jaws, causing tooth mobility and jaw expansion, while the sporadic form is not common in the jaws [15,16,17]. Immunodeficiency-associated BL is mainly seen in HIV patients and only uncommonly affects the jaws [3,5,11,12].
When the jaws are involved, the mandible, especially the posterior part, is more frequently affected [18]; mandibular swelling is the most common clinical feature, as was in our case. Another common clinical symptom is mental nerve neuropathy, also known as ‘numb chin syndrome’, in which the patient complains about chin and lower lip paresthesia [19,20]. Pain and tooth mobility are also frequent, as in our case, and may often lead the clinician to a misdiagnosis of odontogenic infection, causing unnecessary dental interventions, delays in diagnosis and treatment, and worse prognosis [15]. Other clinical signs of jaw involvement include the loosening and extrusion of the molar teeth (primary and permanent), premature shedding of primary molars and eruption of permanent molars, gingival enlargement, maxillary sinus obliteration, and facial asymmetry [21], some of these features being prominent in the present case too.
In cases of jaw involvement, a panoramic radiograph is a helpful initial approach in order to exclude signs of odontogenic infection/inflammation, which is the most prevalent cause of facial swelling in children [18]. The most common radiographic features are non-specific osteolytic lesions with ill-defined borders and, perhaps, an image of ‘floating-in-air’ teeth, like in our case. Other radiographic features of BL may include tooth displacement, lamina dura loss, and periodontal ligament widening [19]. Ultrasonography may be helpful in case of palpable masses in the neck. CT and MRI are valuable to identify bone resorption and adjacent structure involvement and to define the precise extent of the tumor. The appropriate imaging of the chest, abdomen, and pelvis and, especially, PET-CT are useful for tumor staging, as well as for the evaluation of tumor response to therapy [18].
Diagnosis is confirmed via the biopsy of a specific disease site or lymph node. In addition to the aforementioned histopathologic features, the expression of markers typical of germinal center B cells is characteristic [14]. BL expresses monotypic surface IgM, CD19, CD20, CD79a, PAX5, CD43, the plasma cell antigen CD38, and the germinal center antigens CD10 and BCL6, while the Ki-67 proliferative fraction is >95% [14]. Additional tests include bone marrow biopsy, spinal fluid examination, kidney and liver function assessment, and testing for HIV disease [22].
Microscopic differential diagnosis includes hematologic diseases, especially other types of B cell lymphomas, such as diffuse large B cell lymphoma (DLBCL), unclassifiable BL/DLBCL that has an extremely poor prognosis, lymphoblastic lymphoma, blastoid mantle cell lymphoma, and leukemia [6,14].
EBV was identified in 1964 in endemic (African) type BL. The prevalence of EBV infection varies greatly in the different BL subtypes and there are still unresolved questions concerning the contribution of EBV to BL oncogenesis. Endemic BL cases are considered at least 95% EBV-associated, with supportive evidence, including the presence of EBV-DNA clonally integrated into tumor cells and epidemiologic associations with serum EBV antibodies, indicating that EBV infection precedes the malignant transformation [3,23]. On the other hand, only one-third of non-endemic BL is EBV-positive [5]. The variable EBV association in the three BL variants has prompted many to contemplate the possibility of the virus being a passenger in the neoplastic process and not the initiating factor. It is believed that EBV, analogous to malaria, leads to polyclonal B cell activation and permits the poorly controlled proliferation of EBV-positive B cells, further leading to a greater risk of c-myc rearrangement and lymphomagenesis [24]. More recent studies propose a hit-and-run mechanism for the EBV-negative BL. It is suggested that EBV plays an initiating role in oncogenesis, but the viral genome is lost [3].
In all variants, irrespective of EBV status, the constitutive activation of the c-myc (c-MYC) oncogene (human genes are to be written in capital italicized letters I think) is clearly the oncogenic key factor through its translocation, involving the long arm of chromosome 8 and the Ig heavy chain gene on chromosome 14 (>85% of cases) or the Ig light chain genes, [25] leading to the expression of MYC. The MYC/Ig translocation may not be highly detected by common cytogenetics compared to fluorescence in situ hybridization or polymerase chain reaction that increases the chance of positive verification [26]. Interestingly, the position of chromosomal breakpoint relative to the human c-MYC gene differs between endemic and sporadic BL, suggesting different genetic mechanisms involved [27].
The stage of non-Hodgkin lymphoma is determined according to the number of groups of lymph nodes affected and their distribution in the body, as well as the involvement of other organs, such as bone marrow or liver. The system that is still used for BL classification is the Ann Arbor or the St Jude/Murphy staging system, extending from I to IV [28]. Frequently, patients are presented with late disease stages III–IV (70% of the cases). Similarly, in the present case, the patient was categorized as stage III after a thorough work-up.
BL is fatal if left untreated. Prognosis depends on the extent of the disease, the patient’s age, and the timing of diagnosis, described as excellent for the early/moderate stages (survival rate reaching 97–98%), while in the advanced stages (III or IV) it drops to 87.3% [29]. The features associated with an adverse outcome include older age, advanced stage, poor performance status, bulky disease, high lactate dehydrogenase (LDH), and central nervous system (CNS) or bone marrow involvement [30]. In fact, in a recent study, four clinical factors were identified as independently prognostic for patients’ outcomes: age ≥ 40 years, LDH > 3× normal, Eastern Cooperative Oncology Group Performance Status (ECOG PS) ≥ 2, and CNS involvement [31].
The current treatment is chemotherapy with multi-agent regimes, including doxorubicin, vincristine, alkylators, and etoposide, for either short or longer duration [2]. More recently, monoclonal antibodies have been used as adjuvant therapy in BL. In this respect, the anti-CD20 monoclonal antibody rituximab has been used in combination with chemotherapy for the treatment of BL especially for advanced stages, where it has shown improvement in survival by over 95% [2,29]. Radiotherapy is reserved for overt CNS disease resistant to chemotherapy and is reported to be useful in certain emergencies, such as airway obstruction. Hematopoietic stem cell transplantation remains experimental in adults and could be an option in younger patients with refractory/resistant or relapsed disease [32]. The surgical management of BL is limited to emergency cases, necessitating the removal of parts of the intestine that are blocked, bleeding, or have ruptured [33]. Due to the rapidly growing knowledge about the molecular biology of the disease, novel treatment options are in early development, including epigenetic regulators like histone deacetylase inhibitors and DNA methyltransferases inhibitors, as well as small peptide nucleic acids to target oncogenes [32].
Close monitoring during treatment and surveillance following its completion are mandatory; follow-up is recommended every 2–3 months with thorough clinical examination, imaging studies, and blood investigation [2]. The frequency of surveillance decreases over the years since more relapses occur in the first year after treatment completion [29].
In the case presented here, the most clinically significant aspect from a diagnostic standpoint was the fact that the first manifestations were in the mandible, presenting with clinical signs and symptoms that mimicked common innocuous dental inflammatory conditions. Following initial empirical attempts to alleviate the patient’s symptoms on the presumption that they experienced secondary tooth eruption and associated pericoronitis, appropriate referral to a specialized clinic and immediate diagnostic work-up, both imaging and microscopic, was virtually life-saving; it ensured establishing the correct diagnosis and initiating the appropriate chemotherapy in a timely fashion in the face of a very aggressive and rapidly progressing neoplasm.
Accordingly, we performed a review of all the published sporadic BL cases in the English language literature, focusing on well-documented reports with oral and maxillofacial involvement as the first manifestation of the disease. The cases were divided depending on the age of the patients, i.e., children (0–17 years) or adults (≥18 years). The results, including age, sex, presenting sign/symptom, clinical characteristics, imaging features, and outcome, are presented in Table 1 [34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67] and Table 2 [19,21,43,61,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98], respectively. In summary, there were 44 [15,18,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67] cases of sporadic BL in children, including our case. The age distribution ranged between 3 and 16 years, with a mean age of 7.9 years, while the male to female ratio was 2.1:1. In adults, there were 38 cases of sporadic BL; the age distribution ranged between 18 and 84 years, with a mean age of 43 years, while the male to female ratio was 2.8:1.

Table 1.
Cases of sporadic Burkitt lymphoma in children (0–17 years) with oral and maxillofacial involvement as the first manifestation of the disease.

Table 2.
Cases of sporadic Burkitt lymphoma in adults (≥18 years) with oral and maxillofacial involvement as the first manifestation of the disease.
In children, the most common presenting oral and maxillofacial signs and symptoms, corresponding to the first manifestations of the disease, were swelling (in 68.2% of the cases, more frequently localized in the mandible in 34.1%) and pain (34.1%, frequently presenting as toothache); tooth hypermobility or loosening was a chief complaint in 18.2% of the cases, while paresthesia was rarely reported. In adults, swelling (52.6%, more frequently in the mandible in 28.9%) and pain (42.1%, sometimes in the form of toothache) were also common, similar to children; on the other hand, paresthesia, especially numbness of the chin and/or lower lip, was much more common (50%), while tooth hypermobility was only rarely a chief complaint (5.3%) in adults. It should be mentioned that in many cases, lesions were misdiagnosed as dentoalveolar infections and clinicians proceeded with root canal treatment or the extraction of teeth in the lesional area, followed by an antibiotic regimen, with only the deterioration of the symptoms and progression of the disease.
The main finding of clinical examination, in both children and adults, included a swelling and/or mass (84.1% and 71.1%, respectively); th edetection of palpable lymph nodes was comparable among children and adults (27.3% and 21.1%, respectively). In contrast, tooth mobility and/or displacement were much more common in children (61.4% vs. 18.4% in adults), while neurological signs (paresthesia/hypoesthesia/anesthesia) were more frequent in adults (28.9% vs. 4.5% in children). In several cases, other remote sites of involvement were noticed following further investigation at the time of diagnosis.
Various imaging modalities, including panoramic and periapical X-rays, CBCT, CT, MRI, and PET/CT, aided in the diagnosis and allowed the identification of the exact localization of the involved areas and the extent of the disease into adjacent structures. In most cases, single or multiple lytic/destructive bone lesions (93.2% in children and 47.4% in adults) and/or space-occupying lesions (29.5% in children and 18.4% in adults) were noticed. Other imaging findings related to tooth structures included the loss of lamina dura (29.5% in children and 13.2% in adults), teeth displacement (25% in children, but not reported in adults), root resorption (18.2% in children and 7.9% in adults), ‘floating-in-air’ teeth appearance (15.9% in children and 2.6% in adults) and periodontal ligament widening (2.3% in children and 5.3% in adults). Noteworthy is that the overall incidence of the aforementioned imaging findings was almost universally more common in children compared to adults, although the use of different imaging modalities does not allow a direct comparison.
On follow-up (1–192 months for children and 1–60 months for adults), patients were reported to remain free of disease in 50% of children (between 4 and 192 months, mean 59 months) and in 57.9% of adults (between 3 and 60 months, mean 23.2 months). A fatal outcome was reported in 29.5% of children (in 3–10 months following diagnosis) and 26.3% (in 1–11 months following diagnosis) of adults.
4. Conclusions
BL is an aggressive B cell lymphoma, but promptly diagnosed, it rapidly responds to treatment and exhibits a satisfactory long-term prognosis. Jaw lesions demonstrating rapid osseous destruction and aggressive clinical course, especially in the absence of an obvious odontogenic etiology, may constitute the early signs of a more serious condition. The timely detection and appropriate referral of such a rapidly growing jaw lesion, similar to the case presented here, may be life-saving, since it can lead to prompt diagnosis and treatment, ultimately contributing critically to a favorable outcome. Regular long-term follow-up, including a thorough dental and oral and maxillofacial examination, in order to detect possible relapses is also indispensable.
Author Contributions
Conceptualization, E.P. (Erofili Papadopoulou), M.K. and N.G.N.; methodology, E.P. (Erofili Papadopoulou), M.G., M.K. and N.G.N.; formal analysis, E.P. (Erofili Papadopoulou), A.A. and E.P. (Evangelia Piperi); data curation, A.V., G.A., A.K. and N.G.N.; writing—original draft preparation, E.P. (Erofili Papadopoulou), M.G., M.K. and A.A.; writing—review and editing, E.P. (Evangelia Piperi) D.V., S.D., K.D., I.K.K., A.K. and N.G.N.; visualization, A.V., G.A., A.K. and N.G.N.; supervision, A.K. and N.G.N. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study protocol was approved by the Research Ethics Committee of the School of Dentistry of National and Kapodistrian University of Athens (with approval code: 659/08.08.2024, approval date: 30 October 2024) and was performed in accordance with the Declaration of Helsinki.
Informed Consent Statement
Informed consent was obtained from all the subjects involved in the study.
Data Availability Statement
Department of Oral Medicine & Pathology and Hospital Dentistry, School of Dentistry, National and Kapodistrian University of Athens, Greece.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Alexander, D.D.; Mink, P.J.; Adami, H.O.; Chang, E.T.; Cole, P.; Mandel, J.S.; Trichopoulos, D. The non-Hodgkin lymphomas: A review of the epidemiologic literature. Int. J. Cancer 2007, 120, 1–39. [Google Scholar] [CrossRef]
- Kalisz, K.; Alessandrino, F.; Beck, R.; Smith, D.; Kikano, E.; Ramaiya, N.H.; Tirumani, S.H. An update on Burkitt lymphoma: A review of pathogenesis and multimodality imaging assessment of disease presentation, treatment response, and recurrence. Insights Imaging 2019, 10, 56. [Google Scholar] [CrossRef]
- Rochford, R. Reframing Burkitt lymphoma: Virology not epidemiology defines clinical variants. Ann. Lymphoma 2021, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Burkitt, D. A sarcoma involving the jaws in African children. Br. J. Surg. 1958, 46, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Ferry, J.A. Burkitt’s lymphoma: Clinicopathologic features and differential diagnosis. Oncologist 2006, 11, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Magrath, I. Epidemiology: Clues to the pathogenesis of Burkitt lymphoma. Br. J. Haematol. 2012, 156, 744–756. [Google Scholar] [CrossRef] [PubMed]
- Shannon-Lowe, C.; Rickinson, A.B.; Bell, A.I. Epstein-Barr virus-associated lymphomas. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2017, 372, 20160271. [Google Scholar] [CrossRef] [PubMed]
- Mbulaiteye, S.M.; Anderson, W.F.; Ferlay, J.; Bhatia, K.; Chang, C.; Rosenberg, P.S.; Devesa, S.S.; Parkin, D.M. Pediatric, elderly, and emerging adult-onset peaks in Burkitt’s lymphoma incidence diagnosed in four continents, excluding Africa. Am. J. Hematol. 2012, 87, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Morton, L.M.; Wang, S.S.; Devesa, S.S.; Hartge, P.; Weisenburger, D.D.; Linet, M.S. Lymphoma incidence patterns by WHO subtype in the United States, 1992–2001. Blood 2006, 107, 265–276. [Google Scholar] [CrossRef]
- Komatsu, N.; Kawase-Koga, Y.; Mori, Y.; Kamikubo, Y.; Kurokawa, M.; Takato, T. HIV-associated Burkitt lymphoma in a Japanese patient with early submandibular swelling. BMC Res. Notes 2013, 6, 557. [Google Scholar] [CrossRef]
- Guech-Ongey, M.; Simard, E.P.; Anderson, W.F.; Engels, E.A.; Bhatia, K.; Devesa, S.S.; Mbulaiteye, S.M. AIDS-related Burkitt lymphoma in the United States: What do age and CD4 lymphocyte patterns tell us about etiology and/or biology? Blood 2010, 116, 5600–5604. [Google Scholar] [CrossRef]
- Alderuccio, J.P.; Olszewski, A.J.; Evens, A.M.; Collins, G.P.; Danilov, A.V.; Bower, M.; Jagadeesh, D.; Zhu, C.; Sperling, A.; Kim, S.H.; et al. HIV-associated Burkitt lymphoma: Outcomes from a US-UK collaborative analysis. Blood Adv. 2021, 5, 2852–2862. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.S.; Ye, H.; Du, M.Q.; Lu, C.L.; Dogan, A.; Hsieh, P.P.; Huang, W.T.; Jung, Y.C. Histopathology and immunohistochemistry in distinguishing Burkitt lymphoma from diffuse large B-cell lymphoma with very high proliferation index and with or without a starry-sky pattern: A comparative study with EBER and FISH. Am. J. Clin. Pathol. 2007, 128, 558–564. [Google Scholar] [CrossRef]
- Dozzo, M.; Carobolante, F.; Donisi, P.M.; Scattolin, A.; Maino, E.; Sancetta, R.; Viero, P.; Bassan, R. Burkitt lymphoma in adolescents and young adults: Management challenges. Adolesc. Health Med. Ther. 2016, 8, 11–29. [Google Scholar] [CrossRef] [PubMed]
- Cho, B.H.; Shin, D.H.; Jung, Y.H.; Park, H.R. Widely disseminated sporadic Burkitt lymphoma initially presented as oral manifestations in a 6-year-old boy. J. Oral Biol. Craniofacial Res. 2018, 8, 140–142. [Google Scholar] [CrossRef] [PubMed]
- Molyneux, E.M.; Rochford, R.; Griffin, B.; Newton, R.; Jackson, G.; Menon, G.; Harrison, C.J.; Israels, T.; Bailey, S. Burkitt’s lymphoma. Lancet 2012, 379, 1234–1244. [Google Scholar] [CrossRef]
- Lee, D.H.; Yu, M.S.; Lee, B.J. Primary Burkitt’s Lymphoma in the Nasal Cavity and Paranasal Sinuses. Clin. Exp. Otorhinolaryngol. 2013, 6, 184–186. [Google Scholar] [CrossRef] [PubMed]
- De Coninck, W.; Govaerts, D.; Bila, M.; Vansteenkiste, G.; Uyttebroeck, A.; Tousseyn, T.; Politis, C. Burkitt lymphoma in children causing an osteolytic lesion in the mandible: A case report. Clin. Case Rep. 2020, 9, 938–943. [Google Scholar] [CrossRef]
- Azimi, N.; Razmara, F.; Derakhshan, S.; Kardouni Khoozestani, N. Mandibular sporadic Burkitt lymphoma in an adult patient: A case report and review of the literature. Clin. Case Rep. 2021, 9, e04535. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.D.; Ferreira, C.B.; Leite, G.B.; de Menezes Pontes, J.R.; Antunes, H.S. Oral manifestations of lymphoma: A systematic review. E Cancer Med. Sci. 2016, 10, 665. [Google Scholar] [CrossRef]
- Landesberg, R.; Yee, H.; Datikashvili, M.; Ahmed, A.N. Unilateral mandibular lip anesthesia as the sole presenting symptom of Burkitt’s lymphoma: Case report and review of literature. J. Oral Maxillofac. Surg. 2001, 59, 322–326. [Google Scholar] [CrossRef]
- Blum, K.A.; Lozanski, G.; Byrd, J.C. Adult Burkitt leukemia and lymphoma. Blood 2004, 104, 3009–3020. [Google Scholar] [CrossRef] [PubMed]
- Neri, A.; Barriga, F.; Inghirami, G.; Knowles, D.M.; Neequave, J.; Magrath, I.T.; Dalla-Favera, R. Epstein-Barr virus infection precedes clonal expansion in Burkitt’s and acquired immunodeficiency syndrome-associated lymphoma. Blood 1991, 77, 1092–1095. [Google Scholar] [CrossRef] [PubMed]
- Grywalska, E.; Markowicz, J.; Grabarczyk, P.; Pasiarski, M.; Rolinski, J. Epstein-Barr virus-associated lymphoproliferative disorders. Postepy Hig. Med. Dosw. (Online) 2013, 67, 481–490. [Google Scholar] [CrossRef]
- Kimura, H.; Kawada, J.; Ito, Y. Epstein-Barr virus-associated lymphoid malignancies: The expanding spectrum of hematopoietic neoplasms. Nagoya J. Med. Sci. 2013, 75, 169–179. [Google Scholar]
- Burmeister, T.; Schwartz, S.; Horst, H.A.; Rieder, H.; Gokbuget, N.; Hoelzer, D.; Thiel, E. Molecular heterogeneity of sporadic adult Burkitt-type leukemia/lymphoma as revealed by PCR and cytogenetics: Correlation with morphology, immunology and clinical features. Leukemia 2005, 19, 1391–1398. [Google Scholar] [CrossRef]
- Brady, G.; MacArthur, G.J.; Farrell, P.J. Epstein-Barr virus and Burkitt lymphoma. J. Clin. Pathol. 2007, 60, 1397–1402. [Google Scholar] [CrossRef]
- Sandlund, J.T. Burkitt lymphoma: Staging and response evaluation. Br. J. Haematol. 2012, 156, 761–765. [Google Scholar] [CrossRef]
- Minard-Colin, V.; Aupérin, A.; Pillon, M.; Burke, G.A.A.; Barkauskas, D.A.; Wheatley, K.; Delgado, R.F.; Alexander, S.; Uttebroeck, A.; Bollard, C.M.; et al. European Intergroup for Childhood Non-Hodgkin Lymphoma; Children’s Oncology Group. Rituximab for High-Risk, Mature B-Cell Non-Hodgkin’s Lymphoma in Children. N. Engl. J. Med. 2020, 382, 2207–2219. [Google Scholar] [CrossRef] [PubMed]
- Patte, C.; Auperin, A.; Michon, J.; Behrendt, H.; Leverger, G.; Frappaz, D.; Lutz, P.; Coze, C.; Perel, Y.; Raphael, M.; et al. Société Française d’Oncologie Pédiatrique. The Société Française d’Oncologie Pédiatrique LMB89 protocol: Highly effective multiagent chemotherapy tailored to the tumor burden and initial response in 561 unselected children with B-cell lymphomas and L3 leukemia. Blood 2001, 97, 3370–3379. [Google Scholar]
- Evens, A.M.; Danilov, A.; Jagadeesh, D.; Sperling, A.; Kim, S.H.; Vaca, R.; Wei, C.; Rector, D.; Sundaram, S.; Reddy, N.; et al. Burkitt lymphoma in the modern era: Real-world outcomes and prognostication across 30 US cancer centers. Blood 2021, 137, 374–386. [Google Scholar] [CrossRef] [PubMed]
- Pagano, L.; Caira, M.; Valentini, C.G.; Fianchi, L. Clinical aspects and therapy of sporadic burkitt lymphoma. Mediterr. J. Hematol. Infect. Dis. 2009, 1, e2009030. [Google Scholar] [CrossRef] [PubMed]
- Kasamon, Y.L.; Swinnen, L.J. Treatment advances in adult Burkitt lymphoma and leukemia. Curr. Opin. Oncol. 2004, 16, 429–435. [Google Scholar] [CrossRef]
- Stewart, D.J.; Whitlock, R.I. Burkitt’s lymphoma. A case in Ireland. Br. Dent. J. 1972, 133, 255–257. [Google Scholar] [CrossRef] [PubMed]
- Joncas, J.H.; Rioux, E. Burkitt’s lymphoma of the jaw in a Canadian child. Can. Med. Assoc. J. 1977, 117, 367–368. [Google Scholar]
- Terrill, D.G.; Lee, A.; LeDonne, M.A.; Nusbaum, T.G. American Burkitt’s lymphoma in Pittsburgh, Pennsylvania. Oral Surg. Oral Med. Oral Pathol. 1977, 44, 411–418. [Google Scholar] [CrossRef]
- Moore, S.A.; Israel, H.; Johnson, G.F.; Bullock, J.D.; Stout, R.D. Clinical and radiographic oral changes in a case of American Burkitt’s lymphoma. Pediatr. Dent. 1983, 5, 145–148. [Google Scholar] [PubMed]
- Boraz, R.A. American Burkitt’s lymphoma: Report of a case with involvement of the jaws. Pediatr. Dent. 1983, 5, 273–275. [Google Scholar]
- Zachariades, N.; Papanicolaou, S. Non-endemic Burkitt’s lymphoma. Int. J. Oral Maxillofac. Surg. 1986, 15, 88–92. [Google Scholar] [CrossRef]
- Kearns, D.B.; Smith, R.J.; Pitcock, J.K. Burkitt’s lymphoma. Int. J. Pediatr. Otorhinolaryngol. 1986, 12, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Alaluusua, S.; Donner, U.; Rapola, J. Nonendemic Burkitt’s lymphoma with jaw involvement: Case report. Pediatr. Dent. 1987, 9, 158–162. [Google Scholar] [PubMed]
- Svoboda, W.E.; Aaron, G.R.; Albano, E.A. North American Burkitt’s lymphoma presenting with intraoral symptoms. Pediatr. Dent. 1991, 13, 52–58. [Google Scholar]
- Wang, M.B.; Strasnick, B.; Zimmerman, M.C. Extranodal American Burkitt’s lymphoma of the head and neck. Arch. Otolaryngol. Head. Neck Surg. 1992, 118, 193–199. [Google Scholar] [CrossRef]
- Ardekian, L.; Peleg, M.; Samet, N.; Givol, N.; Taicher, S. Burkitt’s lymphoma mimicking an acute dentoalveolar abscess. J. Endod. 1996, 22, 697–698. [Google Scholar] [CrossRef] [PubMed]
- Lund, D.I.; Rodd, H.; Craig, G.T. Burkitt’s lymphoma presenting with jaw lesions in a young white girl. Br. J. Oral Maxillofac. Surg. 1997, 35, 438–441. [Google Scholar] [CrossRef]
- Alpaslan, C.; Cetiner, S.; Emek, D.; Oygur, T. Mandibular soft tissue mass as the initial presentation of Burkitt’s lymphoma. J. Clin. Pediatr. Dent. 1997, 21, 333–335. [Google Scholar]
- Hanazawa, T.; Kimura, Y.; Sakamaki, H.; Yamaguchi, A.; Nagumo, M.; Okano, T. Burkitt’s lymphoma involving the mandible: Report of a case and review of Japanese cases. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1998, 85, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Mitsudo, K.; Tohnai, I.; Hayashi, Y.; Ueda, M.; Yambe, M.; Hirose, Y. A case of Burkitt’s lymphoma that presented initially with resorption of alveolar bone. Oral Dis. 2000, 6, 256–258. [Google Scholar] [CrossRef]
- Tsui, S.H.; Wong, M.H.; Lam, W.Y. Burkitt’s lymphoma presenting as mandibular swelling--report of a case and review of publications. Br. J. Oral Maxillofac. Surg. 2000, 38, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.S.; Liu, H.C.; Bu, J.Q.; Dong, S.N. Burkitt’s lymphoma presenting with jaw lesions. J. Periodontol. 2000, 71, 646–649. [Google Scholar] [CrossRef]
- Durmuş, E.; Oz, G.; Güler, N.; Avunduk, M.; Calişkan, U.; Blanchaert, R.H., Jr. Intraosseous mandibular lesion. J. Oral Maxillofac. Surg. 2003, 61, 246–249. [Google Scholar] [CrossRef]
- Comfort, A.O. Burkitt’s lymphoma of the jaws: Role of dental practitioner in management. Pac. Health Dialog 2004, 11, 89–93. [Google Scholar] [PubMed]
- Jan, A.; Vora, K.; Sándor, G.K. Sporadic Burkitt’s lymphoma of the jaws: The essentials of prompt life-saving referral and management. J. Can. Dent. Assoc. 2005, 71, 165–168. [Google Scholar] [PubMed]
- Ugar, D.A.; Bozkaya, S.; Karaca, I.; Tokman, B.; Pinarli, F.G. Childhood craniofacial Burkitt’s lymphoma presenting as maxillary swelling: Report of a case and review of literature. J. Dent. Child. 2006, 73, 45–50. [Google Scholar]
- Patil, K.; Mahima, V.G.; Jayanth, B.S.; Ambika, L. Burkitt’s lymphoma in an Indian girl: A case report. J. Indian Soc. Pedod. Prev. Dent. 2007, 25, 194–199. [Google Scholar] [CrossRef]
- Freitas, R.d.A.; Veras Barros, S.S.; Quinderé, L.B. Oral Burkitt’s lymphoma--case report. Braz. J. Otorhinolaryngol. 2008, 74, 458–461. [Google Scholar] [CrossRef]
- Pereira, C.M.; Lopes, A.P.; Meneghini, A.J.; Silva, G.B.; Monteiro, M.C.; Botelho, T.D.L. Burkitt’s lymphoma in a young Brazilian boy. Malays. J. Pathol. 2010, 32, 59–64. [Google Scholar] [PubMed]
- Valenzuela-Salas, B.; Dean-Ferrer, A.; Alamillos-Granados, F.J. Burkitt’s lymphoma: A child’s case presenting in the maxilla. Clinical and radiological aspects. Med. Oral Patol. Oral Cir. Bucal 2010, 15, e479-82. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vasudevan, V.; Mohandas, U.; Manjunath, V. Burkitt’s lymphoma in leukemic phase in an Indian boy. Indian J. Dent. Res. 2011, 22, 340–344. [Google Scholar] [CrossRef] [PubMed]
- Chbicheb, S.; Hakkou, F.; El Wady, W. Lymphome de Burkitt buccal chez une enfant immunocompétente [Oral Burkitt lymphoma in an immunocompetent patient]. Arch. Pediatr. 2012, 19, 288–2892. [Google Scholar] [CrossRef]
- Rebelo-Pontes, H.A.; Abreu, M.C.; Guimarães, D.M.; Fonseca, F.P.; Andrade, B.A.; Almeida, O.P.; Pinto, D.D., Jr.; Corrêa-Pontes, F.S. Burkitt’s lymphoma of the jaws in the Amazon region of Brazil. Med. Oral Patol. Oral Cir. Bucal 2014, 19, e32–e38. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cabras, M.; Arduino, P.G.; Chiusa, L.; Broccoletti, R.; Carbone, M. Case Report: Sporadic Burkitt lymphoma misdiagnosed as dental abscess in a 15-year-old girl. F1000Res. 2018, 7, 1567. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, B.; Somay, E.; Hasbay, B. A rare case of Burkitt lymphoma in a 13-year-old girl. J. Pediatr. Res. 2021, 8, 202–205. [Google Scholar] [CrossRef]
- Kulczyk, T.; Daktera-Micker, A.; Biedziak, B.; Wziatek, A.; Bednarek-Rajewska, K. The Primary Outbreaks of Burkitt Lymphoma in the Oral Cavity. A Report of Two Cases, Review of the Literature and Dental Implications. Diagnostics 2021, 11, 2358. [Google Scholar] [CrossRef] [PubMed]
- Riaz, N.; Saeed, T.; Nadeem, M. Burkitt’s lymphoma of mandible in a young Pakistani boy: A case report. J. Pak. Med. Assoc. 2021, 71, 2265–2267. [Google Scholar] [PubMed]
- De Freitas Filho, S.A.; Moura, L.L.; de Souza, M.C.; Rubira, C.M.; Oliveira, D.T. Bilateral jaws involvement of Burkitt’s lymphoma in a pediatric patient. J. Clin. Exp. Dent. 2021, 13, e323–e327. [Google Scholar] [CrossRef] [PubMed]
- Chait, F.; Bahlouli, N.; Laasri, K.; Sfar, K.; Lamalmi, N.; Allali, N.; El Haddad, S.; Chat, L. A Dental Extraction Revealing a Multisystem Burkitt’s Lymphoma: A Case Report. Glob. Pediatr. Health 2024, 11, 2333794X241227704. [Google Scholar] [CrossRef]
- Baden, E.; Carter, R. Intraoral presentation of American Burkitt’s lymphoma after extraction of a mandibular left third molar. J. Oral Maxillofac. Surg. 1987, 45, 689–693. [Google Scholar] [CrossRef] [PubMed]
- Lynch, T.J.; Harris, N.L. Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Case 27-1994. A 41-year-old woman with neurologic abnormalities and an osteolytic lesion in the mandible. N. Engl. J. Med. 1994, 331, 107–113. [Google Scholar]
- Yoskovitch, A.; Hier, M.P.; Bégin, L.R.; Okrainec, A.; Nachtigal, D.; Trudel, M.A.; Black, M.J. Dorsal tongue mass. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2000, 90, 5–8. [Google Scholar] [CrossRef]
- Manolopoulos, L.; Nikolopoulos, T.P.; Yiotakis, J.; Karapatsas, J.; Maris, A.; Ferekidis, E. Burkitt’s lymphoma in the base of the tongue: Differential diagnosis and management. ORL J. Otorhinolaryngol. Relat. Spec. 2003, 65, 226–229. [Google Scholar] [CrossRef]
- Lau, J.J.; Okada, C.Y.; Trobe, J.D. Galloping ophthalmoplegia and numb chin in Burkitt lymphoma. J. Neuro-Ophthalmol. 2004, 24, 130–134. [Google Scholar]
- Cascarini, L.; Brown, A.E. Burkitt’s lymphoma: An unusual cause of lip numbness. Dent. Update 2005, 32, 97–98+100. [Google Scholar] [CrossRef] [PubMed]
- Nissenbaum, M.; Kaban, L.B.; Troulis, M.J. Toothache, paresthesia, and Horner syndrome: An unusual presentation of disseminated Burkitt’s lymphoma. J. Oral Maxillofac. Surg. 2007, 65, 1395–1401. [Google Scholar] [CrossRef] [PubMed]
- Feinberg, S.M.; Ou, S.H.; Gu, M.; Shibuya, T.Y. Burkitt’s lymphoma of the base of the tongue: A case report and review of the literature. Ear Nose Throat J. 2007, 86, 356–360. [Google Scholar] [CrossRef]
- Balasubramaniam, R.; Goradia, A.; Turner, L.N.; Stoopler, E.T.; Alawi, F.; Frank, D.M.; Greenberg, M.S. Burkitt lymphoma of the oral cavity: An atypical presentation. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2009, 107, 240–245. [Google Scholar] [CrossRef]
- Martos-Díaz, P.; Bances-del-Castillo, R.; Vidal-Laso, R.; Mancha-de-la-Plata, M.; Cho-Lee, G.Y.; Naval-Gias, L. Bilateral mental nerve neuropathy as the sole presenting symptom of Burkitt’s Lymphoma. Med. Oral Patol. Oral Cir. Bucal 2009, 14, e408–e410. [Google Scholar]
- Nikgoo, A.; Mirafshariyeh, S.A.; Kazeminajad, B.; Eshkevari, P.S.; Fatemitabar, S.A. Burkitt’s Lymphoma of maxillary sinuses: Review of literature and report of bilateral case. J. Oral Maxillofac. Surg. 2009, 67, 1755–1763. [Google Scholar] [CrossRef]
- Kita, K.; Koura, T.; Ejiri, H.; Kobayashi, N.; Kuroiwa, M.; Miyazono, T.; Yamashiro, S. Numb Chin Syndrome as a Primary Symptom of Burkitt Lymphoma. Gen. Med. 2010, 11, 35–38. [Google Scholar] [CrossRef]
- Sudhakara Reddy, R.; Saimadhavi, N.; Govindraj kumar, N.; Ramesh, T.; Vijaya laxni, N.; Ramya, K. Burkitt’s lymphoma-An unusual presentation of a case. Int. J. Dent. Case Rep. 2011, 1, 49–53. [Google Scholar]
- Faltas, B.; Phatak, P.; Sham, R. Mental nerve neuropathy: Frequently overlooked clinical sign of hematologic malignancies. Am. J. Med. 2011, 124, e1–e2. [Google Scholar] [CrossRef]
- García-Álvarez, S.M.; Olondo-Zulueta, L.; Pericás, J.M.; Colomo, L.; Bosch, X. Numb chin syndrome with vagal and hypoglossal paralysis: An initial sign of an uncommon diagnosis. Am. J. Med. Sci. 2012, 344, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, K.; Inoue, H.; Miyazaki, Y.; Ide, F.; Matsuki, E.; Shigematu, H.; Okamoto, S.; Sakashita, H.; Kusama, K. Adult sporadic burkitt lymphoma of the oral cavity: A case report and literature review. J. Oral Maxillofac. Surg. 2012, 70, 2936–2943. [Google Scholar] [CrossRef] [PubMed]
- Dennis Chun Yu, H.; Bey-Rong, G.; Chia-Yuen, C.; Sey-En, L.; Benny Chih-Yuan, F.; Charles Kuan-Chou, L. Burkitt’s Lymphomas of the Gingiva A Case Report. J. Oral Maxillofac. Surg. 2013, 24, 180–193. [Google Scholar]
- Galvao Barboza, C.A.; Ginani, F.; Souza Medeiros Lima, H.C.D.; Orsini Machado de Sousa, S.; Hitoshi Shinohara, E. Burkitt’s lymphoma presenting as a maxillary swelling in a HIV-negative adult. Acta Stomatol. Croat. 2013, 47, 336–341. [Google Scholar] [CrossRef]
- Boffano, P.; Gallesio, C.; Benech, R.; Berrone, S. Bilateral oral non-endemic burkitt lymphoma. J. Craniofacial Surg. 2013, 24, 1057–1058. [Google Scholar] [CrossRef] [PubMed]
- Manne, R.K.; Madu, C.S.; Talla, H.V. Maxillary sporadic Burkitt’s lymphoma associated with neuro-orbital involvement in an Indian male. Contemp. Clin. Dent. 2014, 5, 231–235. [Google Scholar] [CrossRef]
- Patankar, S.; Venkatraman, P.; Sridharan, G.; Kane, S. Burkitt’s lymphoma of maxillary gingiva: A case report. World J. Clin. Cases 2015, 3, 1011–1016. [Google Scholar] [CrossRef] [PubMed]
- Sethi, N.; Patankar, S.; Jain, R.; Mehta, A. An unusual case of Burkitt’s lymphoma presenting as a gingival enlargement. J. Indian Soc. Periodontol. 2015, 19, 573–577. [Google Scholar]
- Goto, M.; Saito, T.; Kuroyanagi, N.; Sato, H.; Watanabe, H.; Kamiya, N.; Kurita, K.; Shimozato, K. Intraosseous lymphoma of the oral and maxillofacial regions: Report of our experiences, involving some difficult cases to be diagnosed. J. Oral Maxillofac. Surg. Med. Pathol. 2016, 28, 41–46. [Google Scholar] [CrossRef]
- Garcia, N.G.; Rodrigues, M.T.V.; Aleixo, R.Q.; Oliveira, D.T. Burkitt Lymphoma in Adult with Atypical Clinical Presentation Primarily Involving the Oral Soft Tissue. J. Craniofacial Surg. 2017, 28, e795–e797. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.S.; Wu, Y.H.; Sun, A.; Chiang, C.P. Burkitt’s lymphoma of the mandible. J. Dent. Sci. 2017, 12, 421–423. [Google Scholar] [CrossRef] [PubMed]
- Pedraza, R.M.; Arboleda, L.P.A.; Sánchez-Romero, C.; Quiñones, J.A.A.; Tovar, C.J.M.; Henao, J.R.; de Almeida, O.P. Intraoral EBV-positive sporadic Burkitt lymphoma in an elderly patient with bilateral presentation. Autops. Case Rep. 2019, 9, e2019117. [Google Scholar] [CrossRef]
- Tseng, C.H.; Wang, W.C.; Chen, C.Y.; Hsu, H.J.; Chen, Y.K. Clinical manifestations of oral lymphomas—Retrospective study of 15 cases in a Taiwanese population and a review of 592 cases from the literature. J. Formos. Med. Assoc. 2021, 120 Pt 2, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Parker, W.D.; Jones, K. Burkitt’s lymphoma: An unexpected cause of dental pain. J. Surg. Case Rep. 2021, 2021, rjaa557. [Google Scholar] [CrossRef] [PubMed]
- Stanbouly, D.; Clark, M.; Philipone, E. Burkitt Lymphoma Masquerading as Osteomyelitis: An Interesting Road to Diagnosis. J. Craniofacial Surg. 2022, 33, e236–e238. [Google Scholar] [CrossRef]
- Sodnom-Ish, B.; Seo, M.H.; Huh, K.H.; Myoung, H.; Kim, S.M. A Life-Saving Early Diagnosis of Burkitt Lymphoma: The Role of a Dentist. J. Craniofacial Surg. 2022, 33, e326–e329. [Google Scholar] [CrossRef] [PubMed]
- Tereshko, Y.; Hector Ercole, B.; Christian, L.; Belgrado, E.; Dal Bello, S.; Giovanni, M.; Luigi Gigli, G.; Valente, M. Botulinum toxin type A improves pain in numb chin syndrome. Toxicon 2024, 238, 107565. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).