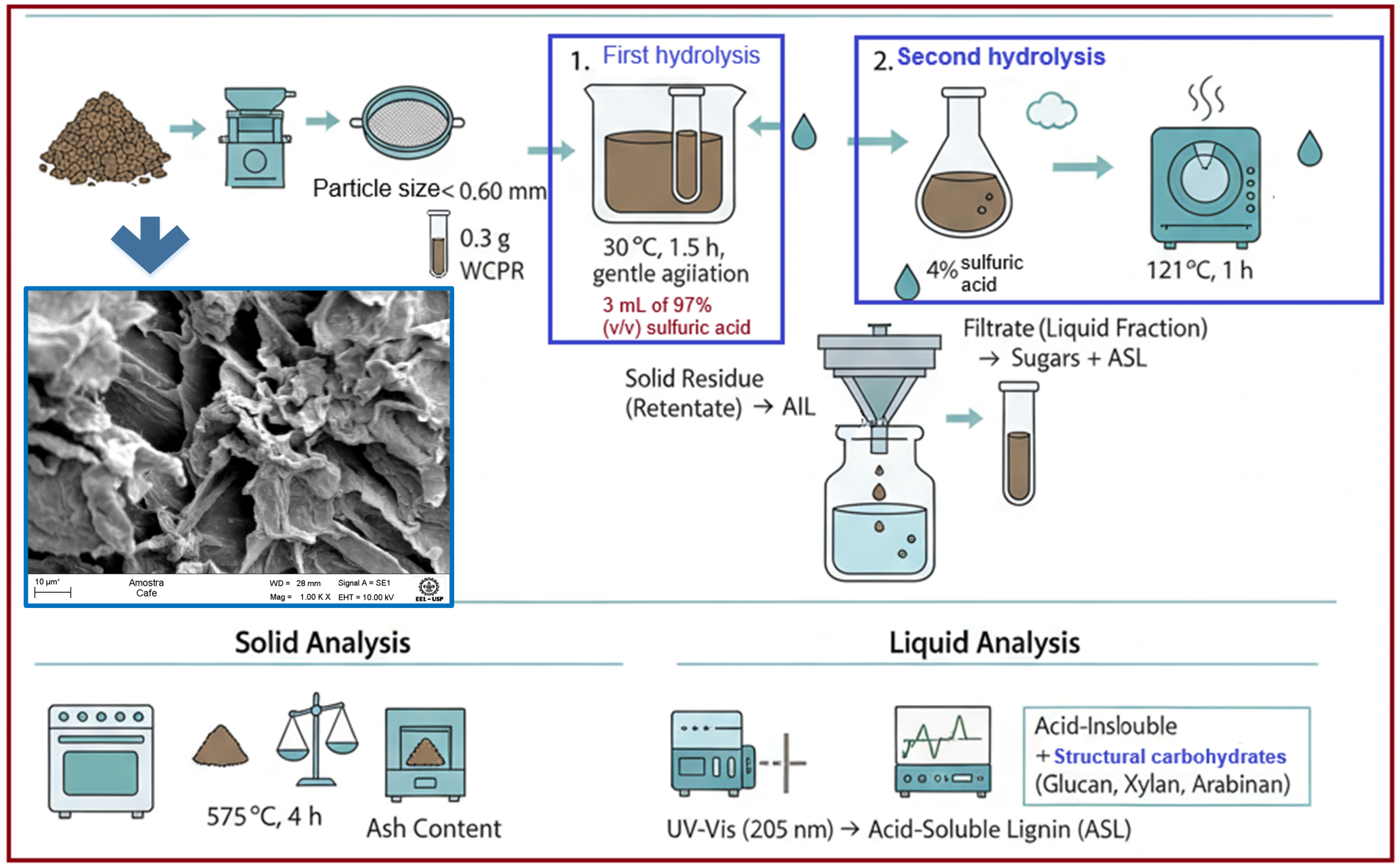
3.2. XOS Production by Hydrothermal Treatment
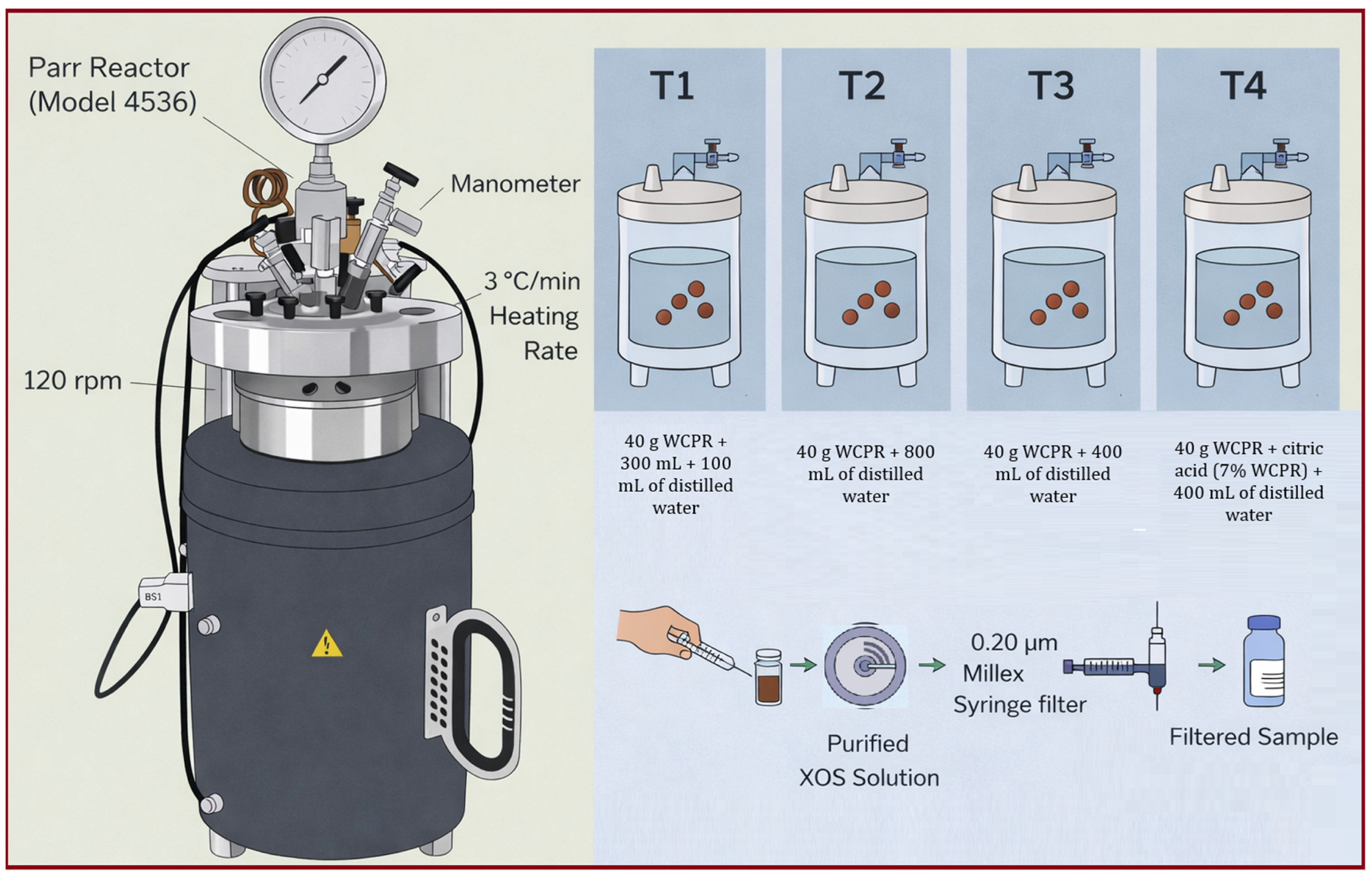
The hydrothermal treatment of wet coffee pulp residues (WCPRs) was carried out to assess the efficiency of structural carbohydrate conversion into xylooligosaccharides (XOSs) and xylose. The effects of dilution (solid-to-liquid ratio) and citric acid addition were examined to determine whether the acid exerted a synergistic catalytic effect on hemicellulose hydrolysis. In treatment T1, water was intentionally added in two sequential steps to ensure adequate mixing, hydration, and homogeneous heat transfer, considering the high water-absorption capacity of wet coffee pulp residues. This strategy resulted in a progressive increase in the effective solid-to-liquid ratio during the reaction, which directly influenced solubilization and product distribution. XOS were detected in the liquid fraction of all treatments and quantified relative to the initial dry mass. As shown in
Figure 5, four hydrothermal treatments (T1–T4) were performed at 180 °C for 15–60 min, monitoring the evolution of xylose (X1), xylobiose (X2), xylotriose (X3), and xylotetraose (X4) by HPLC. Hydrothermal treatment uses pressurized hot water to promote hemicellulose autohydrolysis without the need for mineral catalysts, providing a clean and efficient alternative for lignocellulosic biomass conversion. The addition of citric acid improves selectivity toward XOS through a dual mechanism: as a weak organic acid, it promotes acid-catalyzed hydrolysis, and as a metal chelator, it sequesters trace metals, thereby facilitating selective β–O–4 bond cleavage in lignin-carbohydrate complexes while minimizing inhibitory by-product formation [
21]. In the context of coffee pulp valorization, this approach allows the sustainable recovery of carbohydrate-rich fractions within an integrated biorefinery framework [
3].
Citric-acid-assisted hydrolysis (T4) yielded the highest xylose concentration (16 g·kg−1 at 30 min), indicating enhanced hemicellulose depolymerization and greater process severity (logR0 ≈ 4.17). In contrast, non-acidic conditions (T3) promoted the selective accumulation of xylobiose (14 g·kg−1) and xylotriose (0.7 g·kg−1), suggesting controlled depolymerization. Higher oligomers (X4) were preserved only under milder conditions (T1–T2), confirming that increased severity leads to complete hemicellulose hydrolysis, while moderate conditions favor XOS selectivity.
As shown in
Figure 5A, the citric-acid treatment (T4) consistently produced the highest xylose concentrations across all reaction times, peaking at 16 g·kg
−1 at 30 min and slightly decreasing to 11 g·kg
−1 at 60 min. This decline reflects secondary degradation of xylose into furfural and organic acids under high-severity conditions. In comparison, T3 reached a maximum of 7 g·kg
−1 at 45 min, T2 maintained moderate values (~4 g·kg
−1) between 45 and 60 min, and T1 showed intermediate results (9 g·kg
−1 at 30 min), which can be directly attributed to the two-step water addition strategy applied in this treatment. The gradual increase in water availability promoted biomass hydration and solubilization while simultaneously introducing a dilution effect, resulting in moderate xylose yields compared to single-step dilution treatments. Statistical analysis revealed significant differences among treatments, though residence time had no major effect. According to Tukey’s test, T1 and T3 formed a homogeneous group, indicating similar depolymerization behavior.
Overall, the addition of citric acid increased process severity, accelerating xylose release and hemicellulose breakdown. Comparable patterns have been observed in beech wood and pineapple residues under hydrothermal conditions [
21,
22]. However, when the severity exceeds log R
0 > 4, the values promote the progressive depolymerization of XOS into xylose monomers through enhanced hydrolysis of glycosidic bonds, secondary reactions intensify, promoting the formation of furfural and organic acids that reduce xylose recovery [
23]. Thus, precise control of pH (~2.5–3.5) and residence time is essential to balance conversion efficiency and minimize degradation.
Figure 5B indicates that X2 production was enhanced under moderate, non-citric acidic conditions. Treatment T3 (solid-to-liquid ratio 1:10) achieved the highest X2 yield (14 g·kg
−1 at 60 min), followed by T1 (9 g·kg
−1 at 30 min). In T1, the staged water addition contributed to controlled depolymerization by improving mass transfer and limiting excessive severity at early reaction stages, thereby favoring xylobiose accumulation despite the absence of an added acid catalyst. In contrast, T4 (with citric acid) reached only 7 g·kg
−1 at 15 min, while T2 displayed the lowest values, decreasing from 5 g·kg
−1 at 30 min to 0.55 g·kg
−1 at 60 min. Statistical analysis (letters above bars) shows that treatment significantly affected X2 production, though reaction time had no marked influence. According to Tukey’s test, T1, T2, and T4 formed a homogeneous group, whereas T3 differed statistically, confirming that acid-free conditions favored xylobiose accumulation. Moderate hydrothermal conditions without acid addition promote the formation of low–degree oligomers such as X2, as similarly observed in agricultural residues subjected to mild autohydrolysis [
24]. This trend reflects a controlled partial hydrolysis that preserves the structural integrity of the oligosaccharides, maintaining their prebiotic potential [
25]. Therefore, such conditions are advantageous for bioprocesses aimed at producing functional XOS rather than fermentable sugars.
Figure 5C shows that X3 was detected in treatments T1, T2, and T3, with concentrations below 1 g·kg
−1. The highest value (0.70 g·kg
−1) occurred in T3 at 45 min, followed by T1 (0.60 g·kg
−1 at 60 min) and T2 (0.34 g·kg
−1 at 30–45 min). Both treatment and reaction time significantly influenced X3 production (
p < 0.05). Tukey’s test indicated no significant difference between T1 and T3, suggesting that both provided moderate hydrolysis conditions that favored X3 accumulation while limiting its further conversion to monomers. The detection of X3 in low concentrations supports the formation of intermediate oligomers under controlled hydrolysis conditions prior to their degradation into monosaccharides. This trend agrees with previous studies showing that X3 and X4 appear as transient intermediates during the autohydrolysis of lignocellulosic biomass [
26]. Thus, kinetic control of the hydrothermal treatment plays a crucial role in modulating the molecular weight distribution of XOS, which directly determines their functional and prebiotic properties.
Figure 5D shows that X4 was detected only in treatments T1 and T2, with T1 yielding the highest concentration (3.67 g·kg
−1) at 30 min, while T2 reached only 0.14 g·kg
−1 at the same time. Both treatment and residence time significantly influenced X4 formation (
p < 0.05). These findings indicate that high-degree oligomers such as X4 are maintained only under mild hydrothermal conditions characterized by higher dilution and the absence of acid whereas under more severe conditions (T3 and T4), they are quickly hydrolyzed into smaller XOS or monomeric xylose. This behavior confirms that long-chain oligomers are highly susceptible to degradation under harsh conditions, consistent with reports on rice straw and sugarcane bagasse, where X4–X6 are preserved only during the early stages of hydrolysis [
12]. The selectivity toward longer oligomers is closely linked to temperature intensity and pH, suggesting that careful adjustment of these parameters is essential to optimize the functional value of the final XOS fraction.
Overall, the incorporation of citric acid increased process severity (logR
0 ≈ 4.17), intensifying hemicellulose hydrolysis and favoring the complete conversion of polysaccharides into xylose. Conversely, variations in water availability, particularly the two-step water addition applied in T1, played a measurable role in moderating reaction severity through dilution and improved biomass hydration. This effect was later captured by the multivariate analysis, where the solid-to-liquid ratio emerged as a significant contributor to product distribution. Conversely, moderate acid-free conditions (T3) promoted the selective accumulation of low–degree XOS (X2–X3), demonstrating that reaction medium composition and residence time are key parameters to control product selectivity during hydrothermal treatment. This tunability enables the targeted production of either fermentable sugars or functional XOS from coffee pulp residues within an integrated biorefinery framework. From a broader perspective, these findings highlight hydrothermal treatment as a versatile platform within lignocellulosic biorefineries, capable of generating fermentable sugars for bioenergy applications [
27] or functional oligosaccharides for the food industry and prebiotic potential of short-chain lies in their ability to selectively promote the proliferation of probiotic bacteria, notably Bifidobacterium and Lactobacillus species, in the human gut [
28]. Moreover, the valorization of coffee by-products such as pulp, husk, and silverskin-supports circular economy strategies and strengthens sustainability in coffee-producing regions [
7].
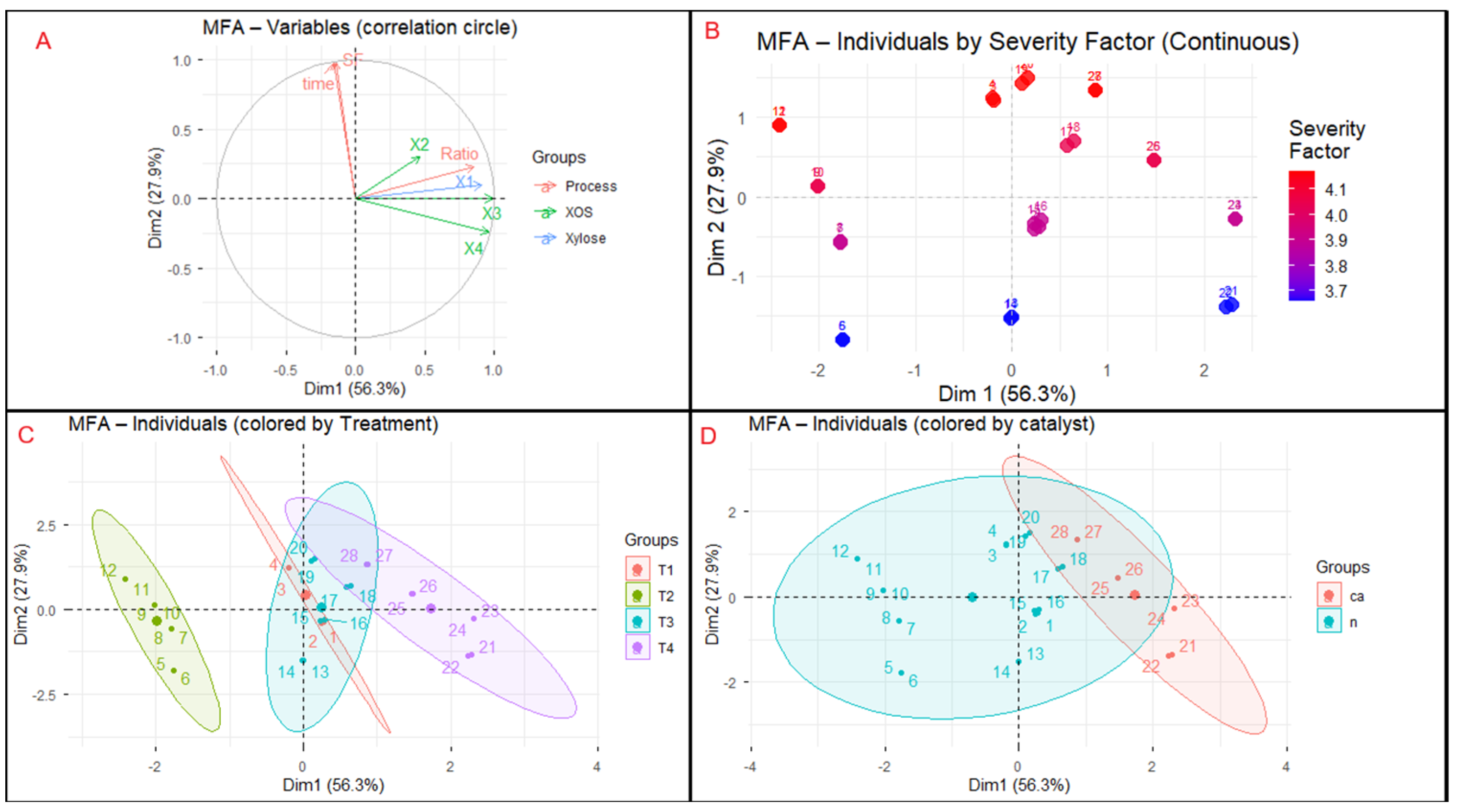
Multiple Factor Analysis (MFA) was applied to integrate and explore correlations among three variable groups: (i) process parameters (Treatment, Catalyst, Time, Ratio, and Severity Factor, SF), (ii) xylose concentration (X1), and (iii) xylooligosaccharides (XOS: X2–X4). The resulting factorial map (
Figure 6) revealed two main dimensions explaining most of the total variance. Dimension 1 (58.3%) represented process intensification, characterized by longer residence times, higher severity, and citric acid catalysis—all positively correlated with xylose release. Dimension 2 (18.9%) captured variations related to the formation and stabilization of longer-chain XOS. Samples treated with citric acid (T4) clustered positively along Dim1, reflecting enhanced hydrolysis efficiency and greater xylose production, while non-acidic treatments (T3) grouped on the negative side, indicating milder depolymerization. X2–X4 loaded strongly and coherently on Dim2, evidencing their shared origin and covariance within the oligomeric fraction. This dimensional separation highlights two complementary mechanisms governing hemicellulose conversion: (i) thermal–catalytic intensification driving depolymerization toward monomeric sugars, and (ii) secondary stabilization dynamics regulating XOS production or degradation. Such behavior aligns with previous reports on hydrothermal treatment and citric-acid-assisted hydrolysis, which modulate polymer distribution within the DP 2–6 range. Overall, MFA provided an integrated multivariate framework to differentiate severity-driven xylose release from oligomer preservation, offering a robust chemometric tool to optimize hydrothermal selectivity for hemicellulose valorization in lignocellulosic biorefineries.
The correlation circle (
Figure 6A) revealed that Time, Severity Factor (SF), and solid-to-liquid Ratio were positively associated with Dim1 and were closely associated with xylose (X1) and xylobiose (X2). This relationship indicates that prolonged residence times and higher severity levels enhance xylose release through progressive hemicellulose depolymerization. In contrast, xylotriose (X3) and xylotetraose (X4) were oriented along Dim2, suggesting that longer-chain XOS are influenced by secondary phenomena such as diffusion constraints or controlled fragmentation rather than direct thermal intensity. The clustering of X2–X4 confirms their shared origin, while the orthogonal position of X1 reflects a trade-off between oligomer preservation and monomer formation. This relationship between process variables and hydrolysate composition is consistent with kinetic models developed for hydrothermal treatment of wood and bagasse, where severity levels (logR ≈ 3.5–4.5) govern the transition from XOS to monomeric xylose [
29]. The orthogonal orientation between XOS and X1 further illustrates the well-known competition between oligomer retention and their degradation to furfural under high-severity conditions [
30].
Figure 6B showed a clear severity-driven gradient of individuals along Dim1, reinforcing the relationships previously observed in the correlation circle (
Figure 6A). Samples processed under higher Severity Factor (SF) values clustered on the positive side of Dim1, where xylose (X1) and short-chain XOS (X2) also exhibited strong positive loadings. This spatial distribution confirms that increasing thermal intensity promotes hemicellulose depolymerization and accelerates the transition from oligomeric fractions toward monomer release, as supported by the kinetic patterns described for hydrothermal treatment of lignocellulosic biomass [
29]. In contrast, samples exposed to milder conditions appeared on the negative side of Dim1, reflecting limited cleavage of hemicellulosic chains and lower solubilization of XOS and xylose. The continuous progression of points along Dim1 also mirrors the gradual shift described earlier between XOS preservation and their subsequent degradation into monomers at higher severities. This behavior aligns with the orthogonality between X1 and higher–degree XOS (X3–X4) observed in
Figure 6A, where the contribution of longer-chain oligomers was predominantly captured by Dim2. Together, these findings illustrate a coordinated severity-dependent mechanism in which moderate SF values favor XOS formation, whereas more intense conditions increase monomeric xylose release. The distribution pattern in
Figure 6B is therefore consistent with the known competitive pathways in hydrothermal systems, where depolymerization, diffusion constraints, and secondary fragmentation collectively define the hydrolysate composition [
30].
Figure 6C revealed a clear grouping of treatments according to their level of process intensification. Treatments T3 and T4, which involved 2 solid-to-liquid ratios and citric acid addition, were positioned in the positive quadrant of Dim1, associated with greater solubilization of xylose and XOS. In contrast, T1 and T2 clustered near the origin or on the negative side of Dim1, reflecting lower hydrolysis efficiency. The compact shape of the ellipses indicated consistent behavior within each treatment, while their progressive displacement from T1 to T4 illustrated a gradual increase in process severity. This clustering pattern confirms the coherence of the experimental design and its accurate reflection in the chemometric space. Comparable trends have been reported in continuous-flow reactor studies, where temperature and residence time directly modulate the XOS/xylose ratio, enabling precise control of process selectivity [
31]. Such gradual behavior supports the applicability of severity-based models as reliable predictors of hydrolysis performance and conversion efficiency.
Figure 6D revealed a clear separation between citric-acid-catalyzed (ca) and non-catalyzed (n) samples. Reactions performed with citric acid were positioned toward positive values on Dim1, associated with higher concentrations of xylose (X1) and short-chain XOS (X2–X4), indicating greater hydrolysis efficiency. In contrast, non-catalyzed treatments were projected toward the negative side of Dim1, reflecting milder depolymerization and lower product release. Citric acid acted as a weak organic catalyst that enhanced hemicellulose solubilization and accelerated hydrolysis reactions, consistent with previous observations in microwave-assisted and autohydrolysis systems, where organic acidity improved xylan conversion efficiency without compromising the structural integrity of the remaining material [
21,
32]. This distinction underscores the role of citric acid as a key parameter for process intensification in hydrothermal treatments.
Supplementary Figure S1 provides additional MFA outputs that reinforce the interpretation of the main results. The group representation (
Figure S1A) shows that the Process group dominates the variance along Dim1, while XOS and Xylose contribute orthogonally, confirming that they describe complementary aspects of hydrolysate composition. The partial projections (
Figure S1B) highlight the coordinated influence of process variables on monosaccharide and oligomer release, consistent with the patterns observed in
Figure 6A–D. The variable contributions (
Figure S1C) indicate that xylose and low–degree XOS are the main drivers of Dim1, supporting the interpretation of this axis as a conversion gradient shaped by severity. Finally, the time-grouped distribution (
Figure S1D) illustrates the expected progression from oligomer-rich to monomer-rich profiles as reaction time increases. Together, these supplementary analyses validate the mechanistic trends identified in the main MFA without overloading the primary figures.
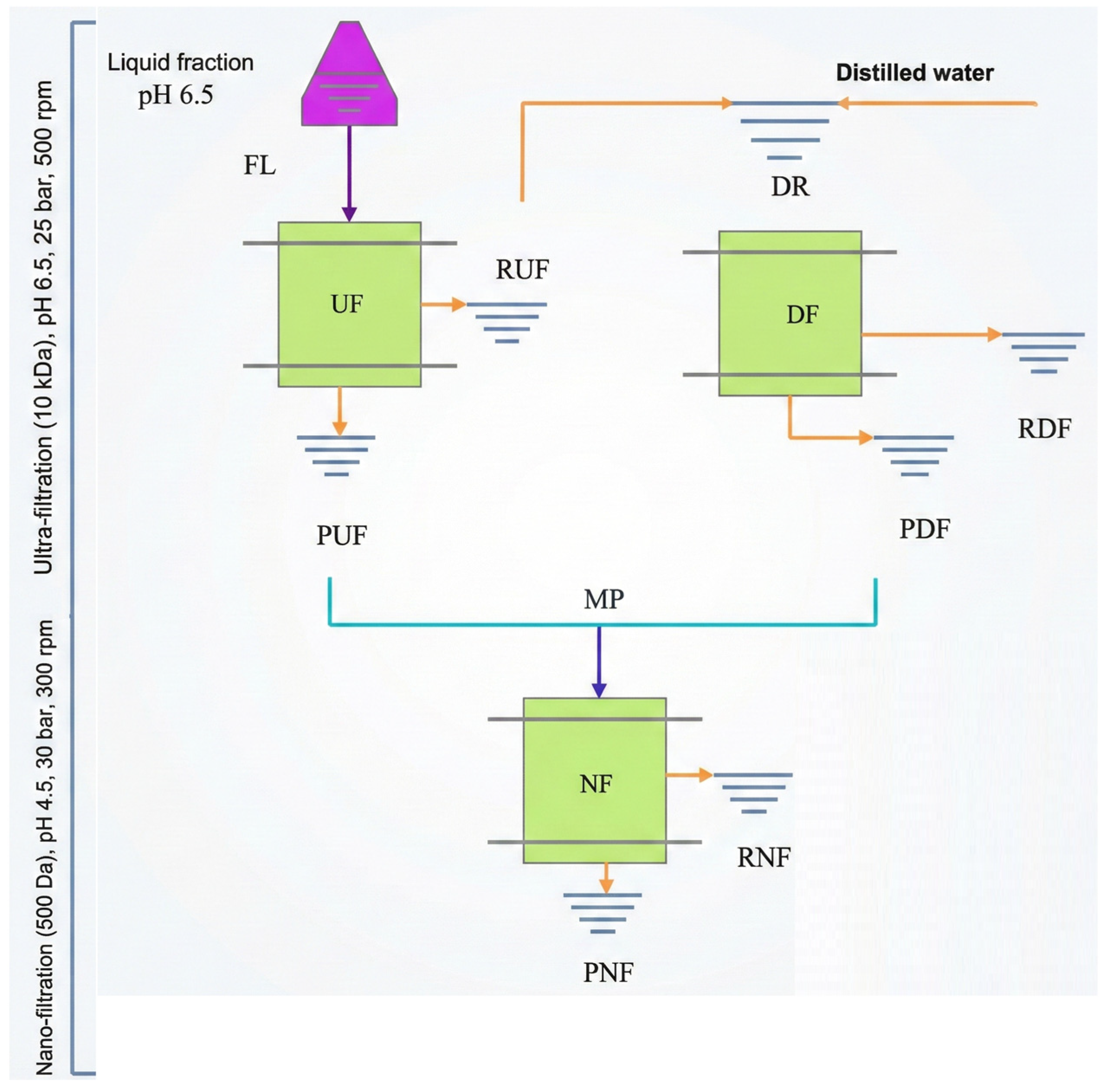
3.3. Membrane Concentration off the Liquid Fraction for XOS Production
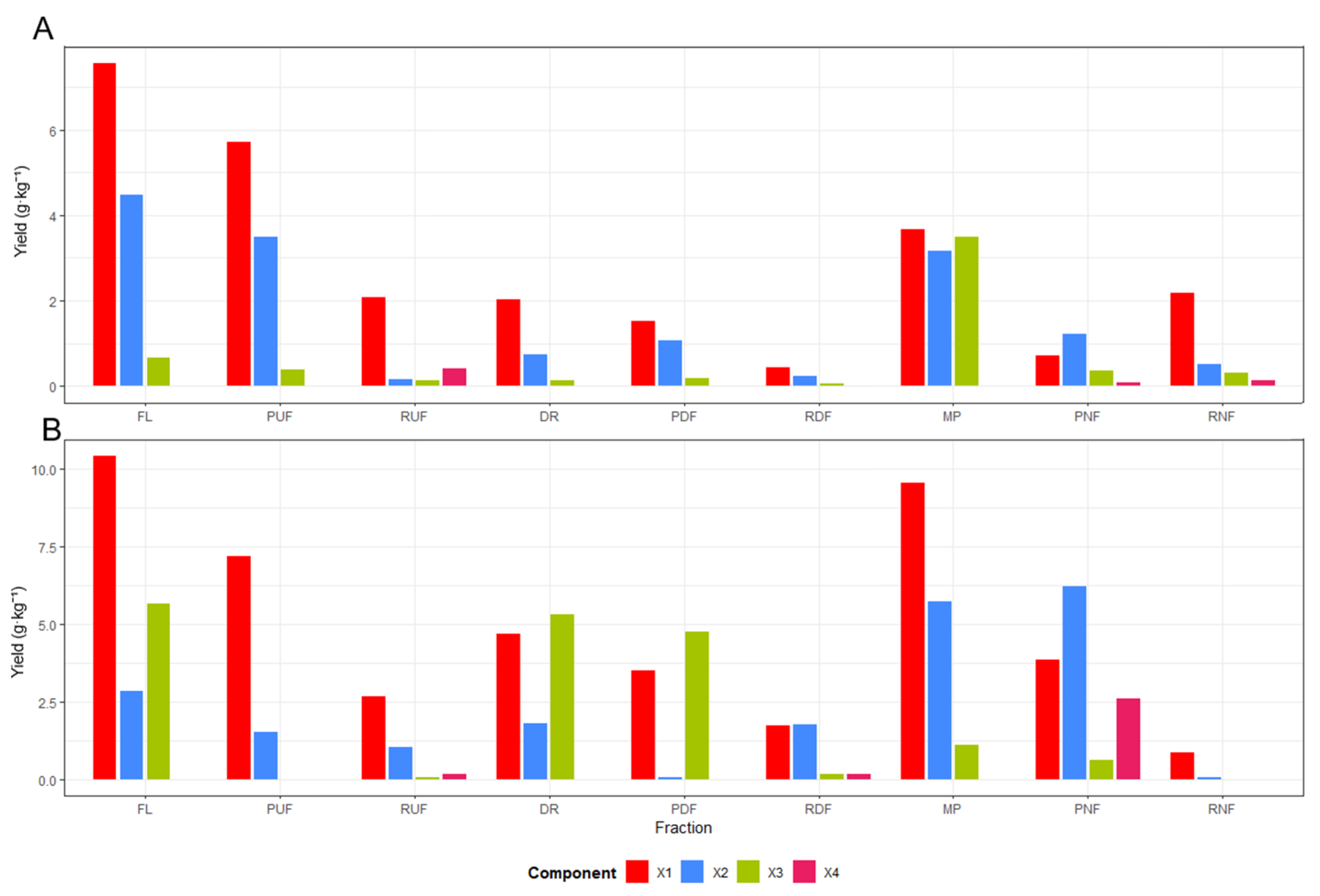
The sequential membrane system comprising ultrafiltration (UF), diafiltration (DF), and nanofiltration (NF) enabled efficient molecular-weight-based separation and enrichment of xylooligosaccharides (XOS), expressed as biomass-normalized yields (g·kg
−1 dry biomass), from hydrolysates obtained in treatments T3 and T4 after 60 min of hydrothermal treatment (
Figure 7). The process began with UF (10 kDa), which removed suspended solids and lignin-rich macromolecules, followed by DF, which enhanced washing and recovery of soluble carbohydrates through selective dilution with distilled water (1:1
v/
v). The final NF step (150 Da) concentrated low-molecular-weight oligomers (X2–X3) in the NF retentate (RNF), yielding purified XOS fractions with reduced levels of furfural and organic acids. In the case of T3, the increase observed in the RNF fraction mainly reflects redistribution during nanofiltration rather than an actual enhancement in overall XOS recovery. The acid-assisted treatment (T4) produced higher short-chain XOS yields, with xylobiose (X2) reaching ≈2.0 g·kg
−1 (dry biomass basis) in the DF retentate (RDF), while nanofiltration mainly redistributed mono- and oligosaccharides between PNF and RNF, confirming the synergistic effect of citric acid catalysis and membrane selectivity. Overall, the integrated UF–DF–NF configuration provided a selective, solvent-free strategy for purifying and concentrating XOS from lignocellulosic hydrolysates, representing an efficient approach for hemicellulose valorization within sustainable biorefinery frameworks [
33,
34]. Altogether, this membrane sequence demonstrated high efficiency for valorizing hemicellulose-derived hydrolysates produced by hydrothermal treatment.
In treatment T3, the liquid fraction (FL) exhibited a heterogeneous composition predominantly composed of xylose (X1) and xylobiose (X2), while higher oligomers (X3 and X4) were detected only in trace amounts. During ultrafiltration (UF), most of the low–molecular–weight sugars (X1–X2) permeated through the 10 kDa membrane, confirming their small molecular size, whereas the retentate (RUF) retained a minor portion of larger XOS. The subsequent diafiltration (DF) step effectively washed and recovered soluble carbohydrates, as evidenced by the decreased X1–X2 content in the retentate (RDF) and their increased presence in the permeate (PDF).
After combining the permeates (MP) and performing nanofiltration (NF), a molecular separation was observed in T3, with xylose (X1) and xylobiose (X2) preferentially recovered in the permeate (PNF), while the retentate (RNF) contained only minor amounts of oligomeric species. This distribution indicates molecular redistribution rather than effective enrichment of xylooligosaccharides under non-acidic conditions (T3).
This fractionation behavior confirms that 10 kDa UF membranes function as an effective clarification step, allowing free passage of mono and disaccharides, whereas the 150 Da NF membrane preferentially retains oligomers with a degree of polymerization (DP ≥ 3). Similar trends were reported for rice-straw hydrolysates, where the UF–NF configuration yielded XOS fractions with purities above 77% [
14]. The concentration increase observed in the RNF highlights the ability of this integrated membrane process to selectively concentrate functional XOS without chemical additives, making it highly suitable for food and biotechnological applications [
35].
For the citric-acid-assisted hydrolysate (T4), a markedly different compositional behavior was observed compared to the non-acidic treatment. The liquid fraction (FL) and the UF retentate (RUF) exhibited similar proportions of xylose (X1) and xylobiose (X2), whereas the diafiltration step (PDF–RDF) substantially enhanced the recovery of medium-chain oligomers particularly in the RDF fraction, where xylobiose (X2) reached ≈2.0 g·kg−1 (dry biomass basis). This trend suggests that mild acid catalysis promoted partial and selective depolymerization of hemicellulose, generating oligomers with a degree of polymerization between 2 and 3 that were efficiently retained and concentrated during DF.
In the final nanofiltration (NF) stage, monomeric sugars were preferentially recovered in the permeate (PNF), while the RNF showed only limited retention of oligomeric species, confirming that nanofiltration mainly contributed to the separation of mono- and oligosaccharides rather than to XOS enrichment. Compared to treatment T3, T4 showed a clearer enrichment of short-chain XOS during diafiltration, highlighting the influence of citric acid on both hydrolysate composition and membrane selectivity. The presence of citric acid in T4 thus enabled a controlled hydrolysis regime, producing fractions rich in short-chain oligomers (DP 2–3) that were efficiently concentrated by DF and NF. Similar behavior has been reported in organic acid–assisted hydrolysis systems, where moderate severity levels (logR
0 ≈ 4) allow the production of high-purity XOS with minimal monomer content [
36,
37]. The enhanced enrichment of X2–X3 in the RDF also suggests a synergistic interaction between acid catalysis and membrane selectivity, consistent with observations in sugarcane and coffee byproduct hemicelluloses [
34].
The overall findings confirm that the sequential membrane process effectively fractionated the hydrolysates according to molecular weight, achieving both purification and enrichment of xylooligosaccharides (XOSs). Under non-acidic conditions (T3), the membrane system mainly promoted carbohydrate redistribution without significant XOS enrichment, whereas acid-assisted processing (T4) favored the recovery of short-chain XOS, with diafiltration identified as the key step governing XOS enrichment. These outcomes demonstrate that coupling hydrothermal treatment with membrane fractionation provides a selective, solvent-free, and environmentally benign strategy for recovering XOS from coffee pulp hydrolysates, suitable for use as prebiotic ingredients or fermentation substrates. Collectively, these results validate the efficiency of integrating hydrothermal and membrane technologies for hemicellulose valorization. This hybrid approach enables the production of high-purity XOS with proven prebiotic functionality, comparable to those obtained from other lignocellulosic sources such as rice straw and sugarcane bagasse [
38]. Furthermore, the use of membrane-based separations minimizes energy demand and eliminates the need for solvents or ion-exchange resins, reinforcing its applicability within sustainable biorefinery frameworks [
28]. From a broader perspective, this strategy aligns with the “hemicellulose-first” paradigm, prioritizing the selective recovery of XOS before cellulose and lignin conversion, thus maximizing the comprehensive utilization of agroindustrial coffee residues.
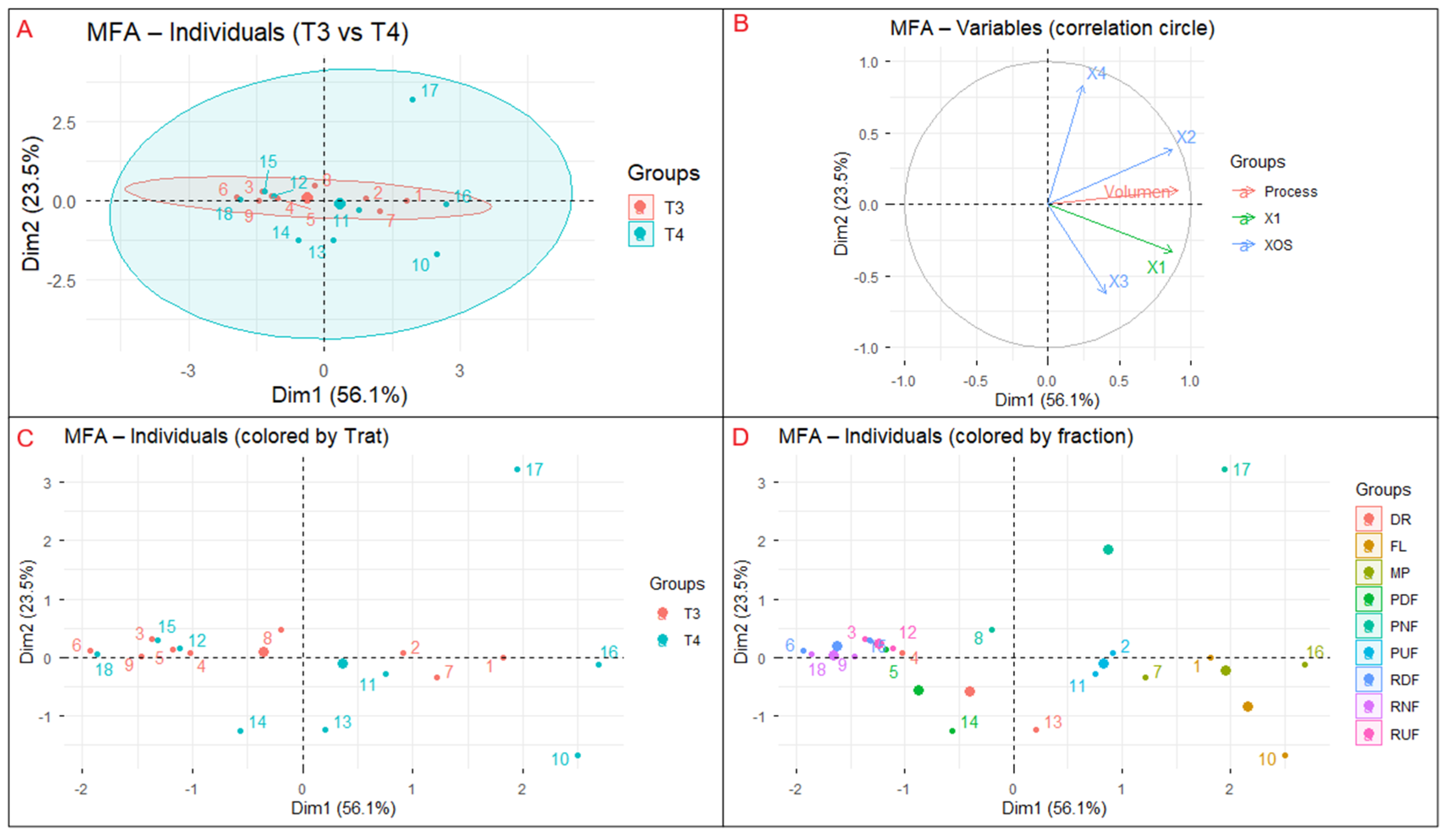
The Multiple Factor Analysis (MFA) was applied to integrate and explore global relationships among process and compositional variables derived from hydrothermal treatments T3 and T4, providing a multivariate perspective on membrane separation behavior. The analysis encompassed three variable groups: (i) process parameter (reaction volume), (ii) compositional variable (X1), and (iii) xylooligosaccharide responses (X2–X4). The first two dimensions (Dim1 = 56.1% and Dim2 = 23.5%) together explained 79.6% of the total variance, indicating a robust factorial structure that effectively captured the system’s multivariate variability. The application of MFA enabled the identification of nonlinear interrelationships between process parameters and compositional responses, overcoming the limitations of univariate analysis. This approach is particularly valuable for complex separation systems such as membrane-based processes, where hydrodynamic conditions, treatment severity, and molecular composition interact simultaneously. Recent studies highlight the relevance of multivariate techniques for optimizing hemicellulose fractionation in biorefineries, demonstrating that over 70% of process variability can be attributed to the synergistic influence of operational parameters and oligomer distribution [
8,
28].
The individual factor map (
Figure 8A) distinguishes treatments T3 and T4, with T4 samples predominantly associated with positive Dim1 values, whereas T3 samples cluster closer to the origin or toward negative Dim1 values. This distribution indicates that acid-assisted hydrothermal treatment promotes higher solute mobilization and transport through the membrane system, while non-acidic conditions result in lower overall carbohydrate displacement. The separation between treatments reflects the influence of reaction severity on hemicellulose depolymerization and solubilization, in agreement with previous studies showing that controlled hydrothermal severity favors oligomer release prior to extensive monomer formation [
39].
The correlation circle (
Figure 8B) shows that reaction volume and xylose (X1) are strongly aligned with Dim1, indicating that this axis primarily represents transport intensity and monosaccharide mobility within the membrane system. In contrast, xylooligosaccharides (X2–X4) load preferentially along Dim2, defining a second, independent axis associated with molecular-weight-driven fractionation. This pattern suggests that while xylose transport is governed mainly by process-related factors such as dilution and permeate flow, XOS behavior is controlled by selective retention mechanisms linked to oligomer size. Similar decoupling between monosaccharide transport and oligomer fractionation has been reported in membrane-assisted XOS purification systems [
37].
The treatment projection (
Figure 8C) confirms that T4 samples exhibit higher scores along both Dim1 and Dim2, reflecting enhanced carbohydrate mobilization together with improved short-chain XOS recovery. This behavior is consistent with kinetic trends observed in hydrothermal hemicellulose conversion, where moderate severity conditions favor oligomeric products over monomeric sugars [
23]. In contrast, T3 samples show limited displacement along Dim2, supporting the observation that non-acidic treatment primarily induces carbohydrate redistribution rather than effective XOS enrichment.
Finally, the fraction distribution map (
Figure 8D) reveals clear differentiation among membrane streams. Low–molecular–weight fractions (FL, PNF, PDF) are mainly located along Dim1, reflecting transport-dominated behavior, whereas retentate fractions (RUF, RDF, RNF) tend to project toward positive Dim2 values, consistent with molecular-weight-dependent retention. Notably, RDF samples are more strongly associated with Dim2 than RNF, indicating that diafiltration plays a more relevant role than nanofiltration in XOS enrichment under the conditions evaluated. This separation reflects classical membrane sieving effects and is consistent with ultrafiltration studies of hemicellulose-derived oligosaccharides, where chain length governs retention and permeation behavior [
34].
Supplementary Figure S2 provides additional insight into the mechanistic interpretation of the MFA results. The partial individual projections (
Figure S2A) show that process variables primarily drive xylose-related displacement, while XOS variables follow a distinct trajectory dominated by Dim2. The contribution map (
Figure S2B) confirms that Dim1 is mainly driven by reaction volume and X1, whereas Dim2 is dominated by XOS variables, particularly X3 and X4, highlighting the role of molecular size in governing separation behavior. The group representation (
Figure S2C) illustrates the near-orthogonality between Process, X1, and XOS groups, reinforcing that membrane performance is governed by two complementary but largely independent mechanisms.
Overall, the MFA indicates that membrane performance is driven by two principal axes: a transport-related axis (Dim1) associated with reaction volume and xylose mobility, and a molecular-weight axis (Dim2) governing selective XOS fractionation. These trends confirm that hydrothermal treatment coupled with membrane separation enables effective molecular differentiation of hemicellulose-derived streams, supporting the design of integrated, hemicellulose-first biorefinery processes [
10].