Abstract
Introduction: Early onset neonatal sepsis (EONS), particularly in preterm sepsis, is a potentially fatal issue. Evaluation of mean platelet volume (MPV) as an EONS predictor was the goal. Methods: Four databases were used to conduct a systematic evaluation of cohort and case–control studies. Up till the end of October 2022, 137 articles were found utilizing the search method. Following the review, 12 studies were included. Leukocytes, MPV, platelets, gender, birth weight, gestational age, mortality, and C-reactive protein (CRP) were all taken into account while analyzing the prediction of EONS. Inverse-variance methodology and the random-effects model were used. Using GRADE, the evidence’s quality was evaluated. Results: Neonatal patients with sepsis had significantly higher MPV levels than do neonates without sepsis (MD 1.26; 95% CI 0.89–1.63; p < 0.001). An increased MPV during the first 24 h postpartum was associated with high CRP values and high risk of neonatal mortality. In the investigations, the MPV cutoff for sepsis patients was 9.95 (SD 0.843). Overall certainty of the evidence was very low. Conclusions: The increased MPV during the first 24 h postpartum may be predictive of EONS and mortality. Future studies are warranted.
1. Introduction
A potentially fatal issue is EONS. It is responsible for up to 30 to 50 percent of all newborn fatalities in underdeveloped nations [1].
Additionally, neonatal sepsis (NS) is regarded as a significant contributor to newborn morbidity and mortality on a global scale [2]. It generates an economic impact, for example, the annual cost of deaths in newborns that are due to sepsis in the United States is 1.97 billion dollars [3]. EONS develops within the newborn’s first 72 h of life [4]. There is a certain degree of difficulty in its diagnosis due to the non-specific clinical manifestations in this type of patient. [5]. This has led to the search for predictors of sepsis. An example is the mean platelet volume (MPV), which could help diagnose EONS [6].
Likewise, some neonatal and maternal risk factors would lead to the development of sepsis in the newborn, such as, low birth weight, chorioamnionitis, prematurity and premature rupture of membranes [7]. The association between neonatal sepsis and other factors, such as gestational age, mode of delivery, sex, and age of the neonate, is still not well defined [8]. The diagnosis of NS is difficult to establish, mainly due to the non-specific clinical features of the patient, where there is temperature instability, tachypnea, hypotension, hypotonia, and abdominal distention [9]. However, some acute phase reactants and inflammatory mediators are predictors of sepsis, such as procalcitonin (PCT), interleukins 6 and 8, C-reactive protein (CRP), and fibrinogen [10]. Due to their significant negative predictive value, platelets have recently been employed as a biomarker for the exclusion of newborn sepsis [11].
According to studies in recent years, among these mediators is the MPV, which could act as a predictor of EONS [12]. The main mechanisms that platelets offer to identify sepsis are the disturbed platelet production during sepsis and the measurement of MPV as a marker of the mean size of circulating platelets in the whole blood count [13,14]. Therefore, the present study aims to evaluate MPV as a predictor of EONS.
2. Materials and Methods
2.1. Data Sources and Searches
From March to June 2022, we searched PubMed, Embase, Web of Science (WOS), and Scopus. We applied the PRISMA 2020 criteria for reporting this systematic review [15]. The key search terms used were “mean platelet volume” and “neonatal sepsis”. Abstracts from cohort and case–control studies that assessed MPV as a predictor of EONS were included. There was no restriction based on the year of publication.
Reviews, case reports and case series studies, editorials, meta-analysis, and letters were excluded.
2.2. Study Selection
In this study, we employed the P-O criteria, considering the Population (Newborns with an early onset neonatal sepsis diagnosis) and the Outcome (MPV measured by mean and standard deviation). Cohort and case–control studies that evaluated mean platelet volume as a predictor of EONS were included in the study. Case-control studies that matched the first two criteria were also included. We omitted narrative and systematic reviews, experiments on animals, clinical trials, case reports, abstracts from conferences, and letters.
Using Rayyan QCRI (https://rayyan.qcri.org/ (accessed on 20 June 2022)), two writers (C.J.T.H. and C.C.R.) independently reviewed the titles and abstracts in accordance with the inclusion and exclusion criteria [16]. The full texts of chosen pertinent studies were searched for in-depth examination. Conflicts were settled by consensus and, if necessary, input from a third author (J.J.B.). Selected articles were stored using the Endnote 20 program.
2.3. Outcomes
The outcome was the Mean platelet volume (fL), measured in mean and standard deviation (SD).
2.4. Data Extraction
Two authors (NCT and JJB) separately extracted the information using the prescribed forms. Consensus was used to resolve disagreements, and a third author was consulted if necessary. The initial author’s name, the year, the study’s design, the participants’ number, the target MPV (fL), the CRP, the mortality, and the study’s results were all gathered. Conflicts were settled with the assistance of a third author.
2.5. Risk of Bias (RoB) Assessment
The RoB was then assessed by two investigators. Finally, the Newcastle Ottawa Scale (NOS) tool was used to examine both cohort and case–control studies independently. This scale was created to evaluate the quality of nonrandomized trials to incorporate quality judgments into meta-analysis interpretation. In the interpretation of meta-analyses, the NOS evaluated quality based on content, design, and simplicity of use. It is made up of eight pieces that are separated into three dimensions (comparison, selection, type of study).
2.6. Statistical Analysis
For meta-analysis, random-effects models and inverse-variance method were applied. For continuous outcomes, the effects of mean platelet volume (MPV) and other characteristics were characterized using mean differences (MD) with 95 percent confidence intervals (95% CI). The I2 statistic was used to look at study heterogeneity: 0–30 percent means low, 30–60 percent means moderate, and >60 percent means high. The primary outcomes were broken down into subgroups based on gestational age (preterm versus term newborns). The R 3.5.1 meta library’s metabin and metacont functions were utilized.
2.7. Quality of Evidence
GRADE was evaluated for the certainty of evidence [17]. A summary results table was created utilizing the results using the GRADEpro software (McMaster University and Evidence Prime Inc., Hamilton, ON, Canada).
3. Results
3.1. Selection of Studies
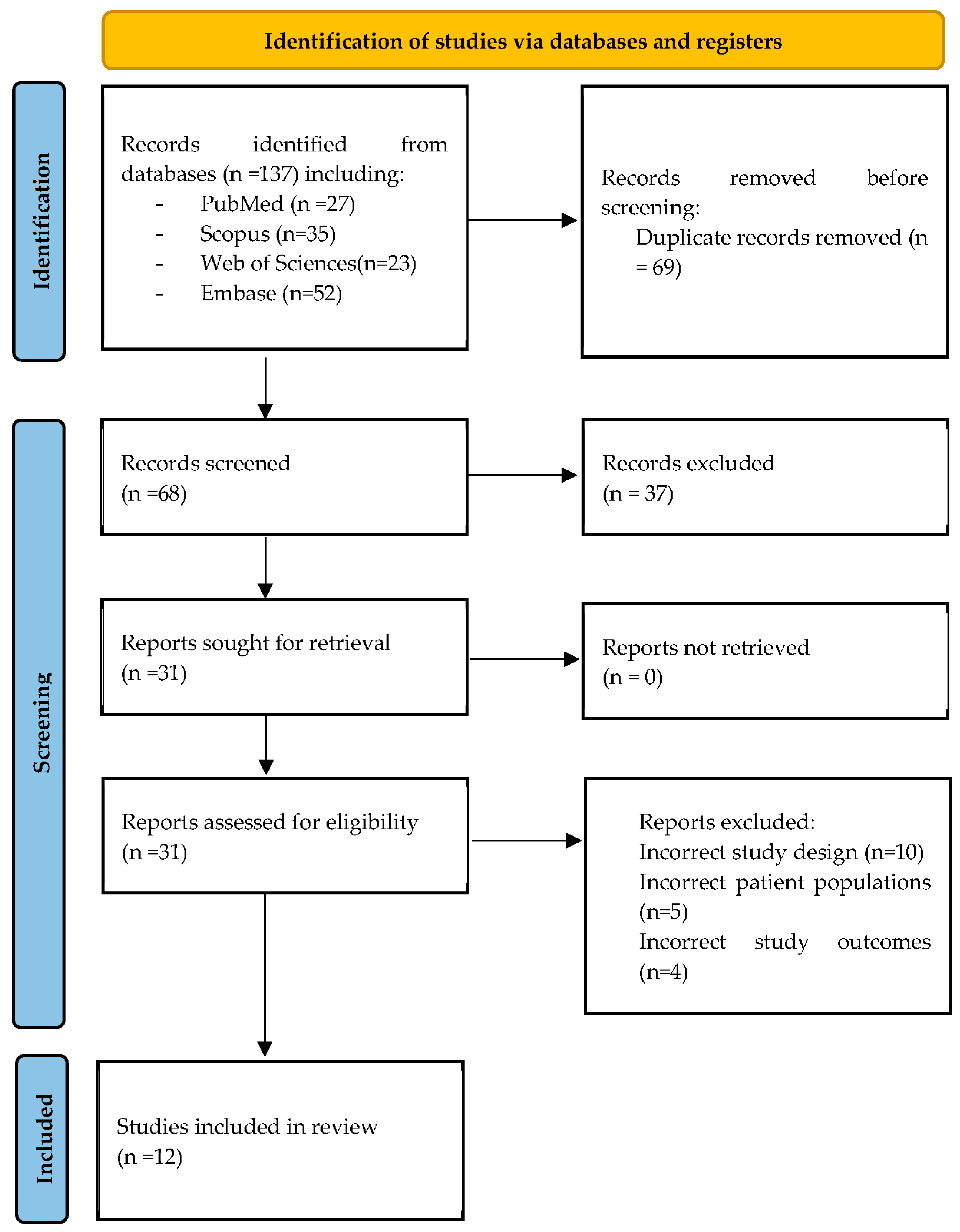
In first place, a total of 137 records were found in the four databases of which 69 duplicates were eliminated. Second, 68 records were selected by title and abstract, and then just 29 records were evaluated in full text. The 37 excluded did not follow our inclusion or exclusion criteria, or were case reports, letters to editor, narrative review or editorial. Finally, 12 studies were ultimately included in the systematic review [18,19,20,21,22,23,24,25,26,27,28,29]. (Figure 1).
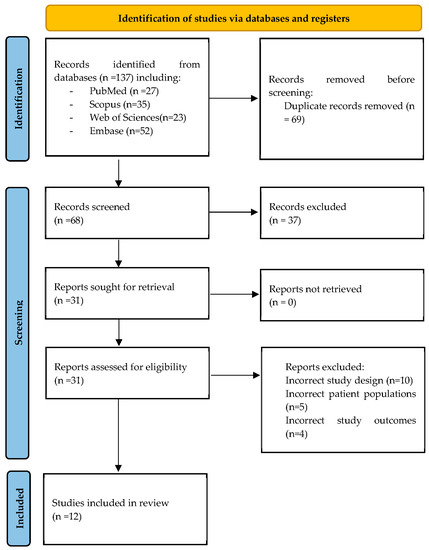
Figure 1.
PRISMA flow chart of the studies selection process.
3.2. Characteristics of Included Studies
Included studies (Table 1) according to the design were nine case–control studies and only one cohort study conducted in countries such as Egypt, Turkey, Iran, and India. The most common concept regarding the definition of sepsis among all studies was: “Proven sepsis” [18,19,20,21,22,23,24,25,26,27], as clinical signs of sepsis with isolation of pathogen in blood, cerebrospinal fluid (CSF), or urine; and, “Probable sepsis” [18,19,20,21,22,23,24,25,26,27], as clinical signs of sepsis without isolated pathogen, with one or more of the following criteria: (a) maternal fever, foul-smelling fluid, prolonged rupture of membranes more than 12 h, gastric polymorph count of more than 6/hpf; (b) positive sepsis screen with 2 of the following 4 parameters: total leukocyte count <5000/mm3, the ratio of band cells to total neutrophils equal to or greater than 0.2, CRP greater than or equal to 0.6 mg/dl, micro Erythrocyte Sedimentation Rate (ESR) greater than or equal to 15 mm at the end of the first hour; and (c) radiological evidence of pneumonia. Most studies agrees that high MPV is predictive of early neonatal sepsis and can function as an adequate biomarker.

Table 1.
Characteristics of Included studies.
3.3. Risk of Bias Analysis
After the evaluation with the Newcastle Ottawa Scale tool, four studies [19,21,23,25] showed a high risk of bias, and six showed a low level of risk of bias according to the assessment.
3.4. Effects of Mean Platelet Volume in Sepsis
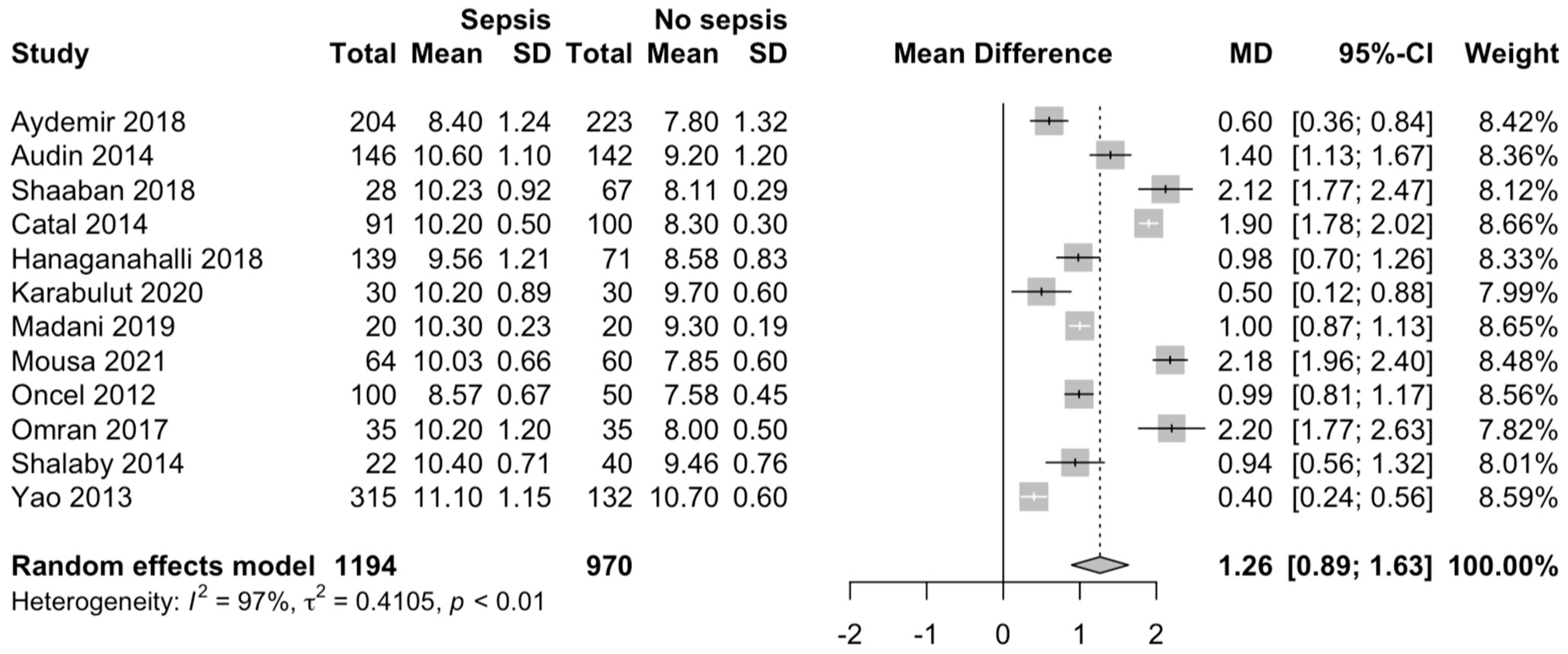
We identified that the MPV is significantly higher in neonates with sepsis (MD 1.26; 95% CI 0.89–1.63; p < 0.001; Figure 2), compared with neonates without sepsis.
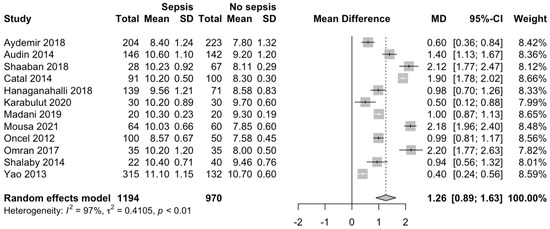
Figure 2.
MPV in neonates with and without EONS [18,19,20,21,22,23,24,25,26,27,28,29].
3.5. Cut-Off and Diagnosis Accuracy between Studies
The cut-off of MPV in patients with sepsis among the studies was 9.95 (SD 0.84). Only four studies had a sensitivity greater than 80% [18,22,25,26]. Similarly, six studies had a specificity greater than 80% [18,21,24,25,27,29]. The Area under curve (AUC) of most studies was greater than 0.6 [18,20,21,22,23,24,25], and two studies [27,28], was less than 0.5 (Table 2).

Table 2.
Diagnosis accuracy between studies.
3.6. Quality of Evidence
We assess the quality of evidence using the GRADE tool (Table 3). Overall certainty of the evidence was very low. The risk of bias decreases by two levels because there are more than two studies with a high risk of bias. Additionally, the inconsistency was decreased by two levels because heterogeneity is higher than 60%. The indirectness and impression were not serious.

Table 3.
GRADE summary of findings table.
4. Discussion
Our study found an increase in MPV in the first 24 h in neonates with sepsis compared to without sepsis group. Therefore, elevated MPV values are predictive of EONS. In addition, we noted an increase of mortality and increased CRP values in patients with neonatal sepsis.
Blood culture is the gold standard for diagnosing newborn sepsis, which has some inefficiencies, such as a high false positive rate (due to contamination) and possible difficulty in obtaining results within 48 to 72 h [30]. Due to the deficiencies above, it was decided to look for alternatives. Here, the possibility of using inflammatory mediators appears, as in the case of MPV, which allows the diagnosis, follow-up, and prediction of the severity of sepsis in neonates [31].
In the pathophysiology of sepsis, there will be an alteration in the coagulation cascade, which will allow the release of multiple pro-and anti-inflammatory cytokines, leading to thrombus formation [32]. This will cause fibrinolytic and fibrinogenic substances to be depleted, leading to further platelet destruction. Finally, the bone marrow will increase the production of young platelets, which are larger and functionally more active, expressing themselves in an increase in vascular endothelial growth factor (VEGF) [33]. The bone marrow will then express itself in an increase in MPV, thus predicting the onset of sepsis.
Platelets have a significant role in sepsis-induced coagulopathy in septic newborns. P-selectin, which is expressed on the surface of platelets during systemic inflammation, enhances platelet adherence to leukocytes and platelet aggregation in addition to tissue factor expression on monocytes [34]. In septic infants, platelet consumption and thrombus development through active endothelium cause thrombocytopenia [35]. C-reactive protein reaches its peak after fifty hours and helps complement bind to foreign or damaged cells in response to inflammation. In addition to clinical evidence, CRP, a non-specific illness response, can aid in the diagnosis of septicemia [36].
It should be mentioned that no previous systematic reviews were found with which this work can be compared. On the other hand, we searched four databases, after which we included ten studies, and, in addition, we assessed the risk of bias using the NOS tool. Regarding the statistical analysis, our study analyzed the continuous variables with the mean difference and standard deviation according to the homogeneity of the measurement factor. Additionally, the random-effects model was applied.
The Importance of our findings lies in the fact that MPV is routinely reported as part of the complete blood count. However, a midpoint for EONS prediction could not be found because the studies reported MPV values with different measurement techniques.
Most of the studies evaluated reported values above the established cut-off, which may be associated with the high sensitivity and specificity reported among the studies. With the values established as an average, it is possible to have an estimated cut-off for evaluating patients with a potential diagnosis of neonatal sepsis [37]. Other studies also reported cut-off values in other population settings in sepsis [38,39,40].
Likewise, the low cost and easy availability makes the MPV a marker with greater significance. Another critical point to highlight is the homogeneity of the studies, nine of which were of the case–control type, allowing greater homogeneity in the groups studied. Associated factors, such as prematurity or low birth weight, were considered in very few studies, so their true significance could not be determined.
The study has some limitations. First, observational studies are subject to confounding variables, thus increasing statistical heterogeneity between studies, and may influence the true overall effect. Second, there is a very low certainty of the evidence, which limits an adequate interpretation despite the significant effect. However, this certainty of the evidence is not related to interventions but as a predictor.
5. Conclusions
In conclusion, with the results of this study, we can affirm that increased MPV may be used as a predictor of EONS, which would potentially allow us to avoid excessive antibiotic use and unnecessary hospitalizations.
Author Contributions
Conceptualization, C.C.-R., C.Q.-V., D.A.L.-F., C.J.T.-H., N.C.-T., J.J.B. and J.A.C.-A.; methodology, C.C.-R., C.Q.-V., D.A.L.-F., C.J.T.-H., N.C.-T., J.J.B. and J.A.C.-A.; software, C.C.-R.; validation, C.C.-R., C.Q.-V., D.A.L.-F., C.J.T.-H., N.C.-T., J.J.B. and J.A.C.-A.; formal analysis, C.C.-R., C.Q.-V., D.A.L.-F., C.J.T.-H., N.C.-T., J.J.B. and J.A.C.-A.; investigation, C.C.-R., C.Q.-V., D.A.L.-F., C.J.T.-H., N.C.-T., J.J.B. and J.A.C.-A.; resources, C.C.-R., C.Q.-V., D.A.L.-F., C.J.T.-H., N.C.-T., J.J.B. and J.A.C.-A.; data curation, C.C.-R., C.Q.-V., D.A.L.-F., C.J.T.-H., N.C.-T., J.J.B. and J.A.C.-A.; writing—original draft preparation, C.C.-R., C.Q.-V., D.A.L.-F., C.J.T.-H., N.C.-T., J.J.B. and J.A.C.-A.; writing—review and editing, C.C.-R., C.Q.-V., D.A.L.-F., C.J.T.-H., N.C.-T., J.J.B. and J.A.C.-A. visualization, C.C.-R., C.Q.-V., D.A.L.-F., C.J.T.-H., J.J.B. and J.A.C.-A.; supervision, C.C.-R. and J.J.B.; project administration, C.C.-R., C.Q.-V., D.A.L.-F., C.J.T.-H., J.J.B. and J.A.C.-A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Dündar, B.; Çakmak, B.D.; Özgen, G.; Taşöz, F.N.; Güçlü, T.; Ocakoglu, G. The association of platelet indices and adverse neonatal outcomes in pregnancies complicated with preterm premature rupture of membranes. J. Turk. Ger. Gynecol. Assoc. 2016, 17, S228. [Google Scholar]
- Rajaratnam, J.K.; Marcus, J.R.; Flaxman, A.D.; Wang, H.; Levin-Rector, A.; Dwyer, L.; Costa, M.; Lopez, A.D.; Murray, C.J. Neonatal, postneonatal, childhood, and under-5 mortality for 187 countries, 1970–2010: A systematic analysis of progress towards Millennium Development Goal 4. Lancet 2010, 375, 1988–2008. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Farahbakhsh, N.; Shastri, S.; Sharma, P. Biomarkers for diagnosis of neonatal sepsis: A literature review. J. Matern. Fetal Neonatal. Med. 2018, 31, 1646–1659. [Google Scholar] [CrossRef] [PubMed]
- Koroglu, M.; Terzi, H.A.; Gurol, G.; Demiray, T.; Ozbek, A.; Hafizoglu, T. Early detection of sepsis in newborn clinic, is it possible? Clin. Biochem. 2014, 47, 781. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Z.; Zhang, M.; Lou, Z.; Deng, J.; Li, Q. Diagnostic value of mean platelet volume for neonatal sepsis: A systematic review and meta-analysis. Medicine 2020, 99, e21649. [Google Scholar] [CrossRef]
- Pamudji, K.M.; Kardana, I.M. Diagnostic value of mean platelet volume in neonatal sepsis. Paediatr. Indones. 2019, 59, 289–293. [Google Scholar] [CrossRef]
- Cekmez, Y.; Güleçoğlu, M.D.; Özcan, C.; Karadeniz, L.; Kiran, G. The utility of maternal mean platelet volume levels for early onset neonatal sepsis prediction of term infants. Ginekol. Pol. 2017, 88, 312–314. [Google Scholar] [CrossRef]
- Gray, A.; Sharara, F. Global and regional sepsis and infectious syndrome mortality in 2019: A systematic analysis. Lancet Glob. Health 2022, 10 (Suppl. 1), S2. [Google Scholar] [CrossRef]
- Eichberger, J.; Resch, E.; Resch, B. Diagnosis of Neonatal Sepsis: The Role of Inflammatory Markers. Front. Pediatr. 2022, 10, 840288. [Google Scholar] [CrossRef]
- Mari, J.; Kajlik, R.; Gajda, A.; Kiss, J.; Gyurkovits, Z. Use of antibiotics in suspected early-onset neonatal sepsis. Orv. Hetil. 2022, 163, 431–437. [Google Scholar] [CrossRef]
- Iroh Tam, P.Y.; Bendel, C.M. Diagnostics for neonatal sepsis: Current approaches and future directions. Pediatr. Res. 2017, 82, 574–583. [Google Scholar] [CrossRef] [PubMed]
- Hudon, R.E.; Leung, V.H.; Petit, S.; Banach, D.B. Hospital Readmissions Among Infants Diagnosed with Early-Onset Neonatal Sepsis in Connecticut. J. Pediatr. Infect. Dis. Soc. 2022, 11, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Budak, Y.U.; Polat, M.; Huysal, K. The use of platelet indices, plateletcrit, mean platelet volume and platelet distribution width in emergency non-traumatic abdominal surgery: A systematic review. Biochem. Med. 2016, 26, 178–193. [Google Scholar] [CrossRef] [PubMed]
- Eicher, J.D.; Lettre, G.; Johnson, A.D. The genetics of platelet count and volume in humans. Platelets 2018, 29, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: 343 an updated guideline for reporting systematic reviews. BMJ 2021, 372. [Google Scholar] [CrossRef]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan-a web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef]
- Aguayo-Albasini, J.L.; Flores-Pastor, B.; Soria-Aledo, V. Sistema GRADE: Clasificación de la calidad de la evidencia 347 y graduación de la fuerza de la recomendación. Cir. Esp. 2014, 92, 82–88. [Google Scholar] [CrossRef]
- Shaaban, H.A.; Safwat, N. Mean platelet volume in preterm: A predictor of early onset neonatal sepsis. J. Matern. Fetal Neonatal. Med. 2018, 33, 206–211. [Google Scholar] [CrossRef]
- Oncel, M.Y.; Ozdemir, R.; Yurttutan, S.; Canpolat, F.E.; Erdeve, O.; Oguz, S.S.; Uras, N.; Dilmen, U. Mean Platelet Volume in Neonatal Sepsis. J. Clin. Lab. Anal. 2012, 26, 493–496. [Google Scholar] [CrossRef]
- Omran, A.; Maaroof, A.; Saleh, M.H.; Abdelwahab, A. Salivary C-reactive protein, mean platelet volume and neutrophillymphocyte ratio as diagnostic markers for neonatal sepsis. J. Pediatr. (Rio J.) 2018, 94, 82–87. [Google Scholar] [CrossRef]
- Mousa, S.O.; Moustafa, A.N.; Aly, H.M. Prognostic value of red cell distribution width, platelet parameters, and the hematological scoring system in neonatal sepsis. Egypt J. Haematol. 2019, 44, 183–189. [Google Scholar] [CrossRef]
- Karabulut, B.; Alatas, S.O. Diagnostic Value of Neutrophil to Lymphocyte Ratio and Mean Platelet Volume on Early Onset Neonatal Sepsis on Term Neonate. J. Pediatr. Intensive Care 2021, 10, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Madani, S.H.; Amiri, S.; Khazaei, S.; Erfan, M.B.K.; Rostami-Far, Z.; Tarlan, M.; Akya, A.; Azizi, A.; Izadi, B.; Shaveisi-Zadeh, J. Platelet indices as useful indicators of neonatal sepsis. J. Evol. Med. Dent. Sci. 2019, 8, 1612–1617. [Google Scholar] [CrossRef]
- Hanaganahalli, S.B.; Sreeram, S.; Bompada, M.; Kuppannagari, S.K.; Suresh, P.K.; Philipose, C.S. Is MPV a Predictive Marker for Neonatal Sepsis? A Pilot Study. J. Pediatr. Hematol. Oncol. 2018, 40, 548–552. [Google Scholar] [CrossRef]
- Catal, F.; Tayman, C.; Tonbul, A.; Akça, H.; Kara, S.; Tatli, M.M.; Oztekin, O.; Bilici, M. Mean platelet volume (MPV) may simply predict the severity of sepsis in preterm infants. Clin. Lab. 2014, 60, 1193–1200. [Google Scholar] [CrossRef]
- Aydın, B.; Dilli, D.; Zenciroğlu, A.; Karadag, N.; Beken, S.; Okumuş, N. Mean Platelet Volume and Uric Acid Levels in Neonatal Sepsis. Indian J. Pediatr. 2014, 81, 1342–1346. [Google Scholar] [CrossRef]
- Aydemir, C.; Aydemir, H.; Kokturk, F.; Kulah, C.; Mungan, A.G. The cut-off levels of procalcitonin and C-reactive protein and the kinetics of mean platelet volume in preterm neonates with sepsis. BMC Pediatr. 2018, 18, 253. [Google Scholar] [CrossRef]
- Shalaby, M.M.; Sobeih, A.A.; Abdulghany, W.E.; Behiry, E.G.; Ismail, Y.M.; Abd-El-Aziz, M.A. Mean platelet volume and serum uric acid in neonatal sepsis: A case-control study. Ann. Med. Surg. 2017, 20, 97–102. [Google Scholar] [CrossRef]
- Yao, Y.; Tu, Y.; Lu, Q. Values of C-reactive protein, percentage of neutrophils and mean platelet volume in early diagnosis of neonatal sepsis. Zhongguo Dang Dai Er Ke Za Zhi 2015, 17, 425–429. (In Chinese) [Google Scholar]
- Gialamprinou, D.; Mitsiakos, G.; Katsaras, G.N.; Kontovazainitis, C.G.; Karagianni, P.; Roilides, E.; Kenet, G. Neonatal Sepsis and Hemostasis. Diagnostics 2022, 12, 261. [Google Scholar] [CrossRef]
- Burgunder, L.; Heyrend, C.; Olson, J.; Stidham, C.; Lane, R.D.; Workman, J.K.; Larsen, G.Y. Medication and Fluid Management of Pediatric Sepsis and Septic Shock. Paediatr. Drugs 2022, 24, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Akhmaltdinova, L.; Kolesnichenko, S.; Lavrinenko, A.; Kadyrova, I.; Avdienko, O.; Panibratec, L. Influence of Pathogen Type on Neonatal Sepsis Biomarkers. Int. J. Inflam. 2021, 2021, 1009231. [Google Scholar] [CrossRef] [PubMed]
- Siavashi, V.; Asadian, S.; Taheri-Asl, M.; Keshavarz, S.; Zamani-Ahmadmahmudi, M.; Nassiri, S.M. Endothelial Progenitor Cell Mobilization in Preterm Infants with Sepsis Is Associated with Improved Survival. J. Cell. Biochem. 2017, 118, 3299–3307. [Google Scholar] [CrossRef] [PubMed]
- Tunjungputri, R.N.; van de Heijden, W.; Urbanus, R.T.; de Groot, P.G.; van der Ven, A.; de Mast, Q. Higher platelet reactivity and platelet-monocyte complex formation in Gram-positive sepsis compared to Gram-negative sepsis. Platelets 2017, 28, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Iba, T.; Levy, J. Inflammation and thrombosis: Roles of neutrophils, platelets and endothelial cells and their interactions in thrombus formation during sepsis. J. Thromb. Haemost. 2018, 16, 231–241. Available online: https://pubmed.ncbi.nlm.nih.gov/29193703/ (accessed on 20 June 2022). [CrossRef]
- Mair, R.J.; Joy, A.H.; Finney, H.; Weaver, A. Creating an algorithm for requesting C-reactive protein in the emergency department. Eur. J. Emerg. Med. 2010, 17, 125–126. [Google Scholar] [CrossRef]
- Cai, N.; Chen, Z.Q.; Tao, M.; Fan, W.T.; Liao, W. Mean platelet volume and red blood cell distribution width is associated with prognosis in premature neonates with sepsis. Open Med. (Wars) 2021, 16, 1175–1181. [Google Scholar] [CrossRef]
- Vélez-Paez, J.L.; Velarde-Montero, C.; Irigoyen-Mogro, E.; Vélez-Páez, P.; Cifuentes-López, P.; Vélez, J.W.; Flores, J.L.R.A.; Barboza, J.J. Mean platelet volume as a predictor of mortality in patients with sepsis: Systematic review and meta-analysis. Infectio 2020, 24, 162–168. [Google Scholar] [CrossRef][Green Version]
- Vélez-Páez, J.L.; Armendáriz-Carvajal, A.C.; Rueda-Barragán, F.E.; Castro-Reyes, E.E.; Escola-Brito, A.C.; Chambergo-Michilot, D.; Castillo-Sancho, S.A.; Hilario-Gomez, M.M.; Barboza, J.J. Utility of cytometric parameters and indices as predictors of mortality in patients with sepsis. Infectio 2021, 25, 153–158. [Google Scholar] [CrossRef]
- Toro-Huamanchumo, C.J.; Barboza, J.J.; Pinedo-Castillo, L.; Barros-Sevillano, S.; Gronerth-Silva, J.K.; Gálvez-Díaz, N.d. Maternal factors associated with prematurity in pregnant women from a public hospital in Trujillo, Peru. Rev. Del Cuerpo Med. Hosp. Nac. Almanzor Aguinaga Asenjo 2021, 14, 287–290. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).