Acute Appendicitis as the Initial Presentation of Kawasaki Disease Shock Syndrome in Children
Abstract
1. Introduction
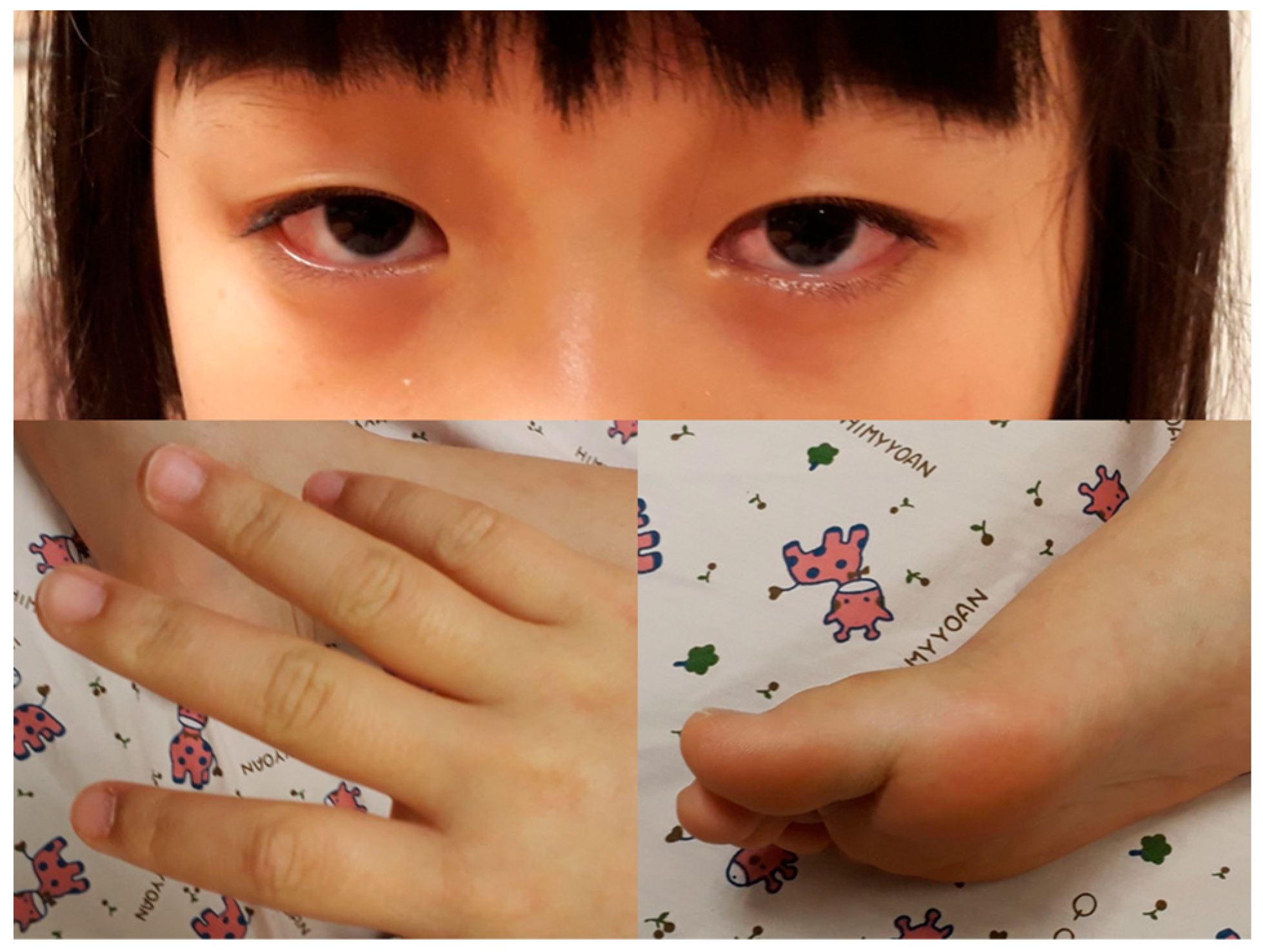
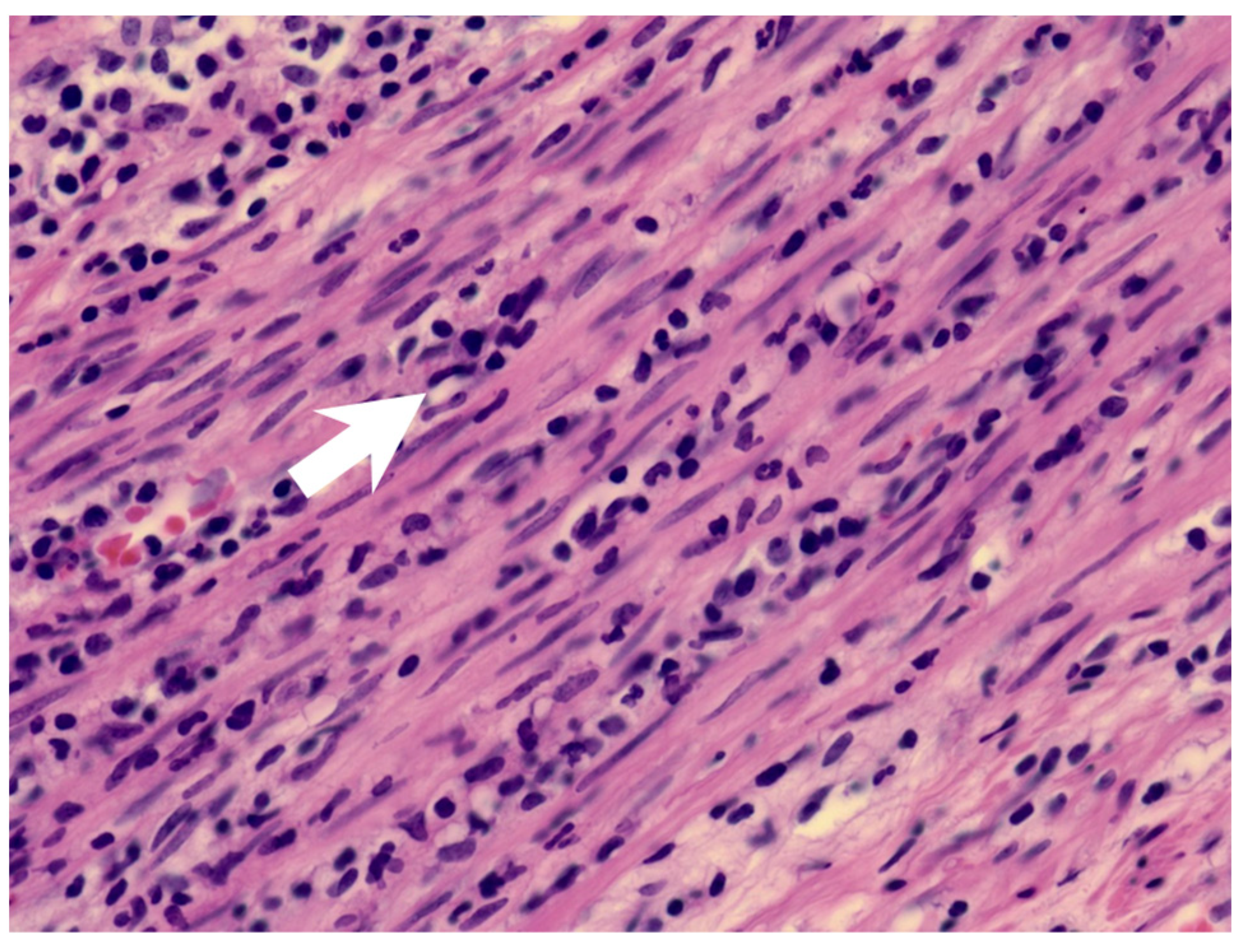
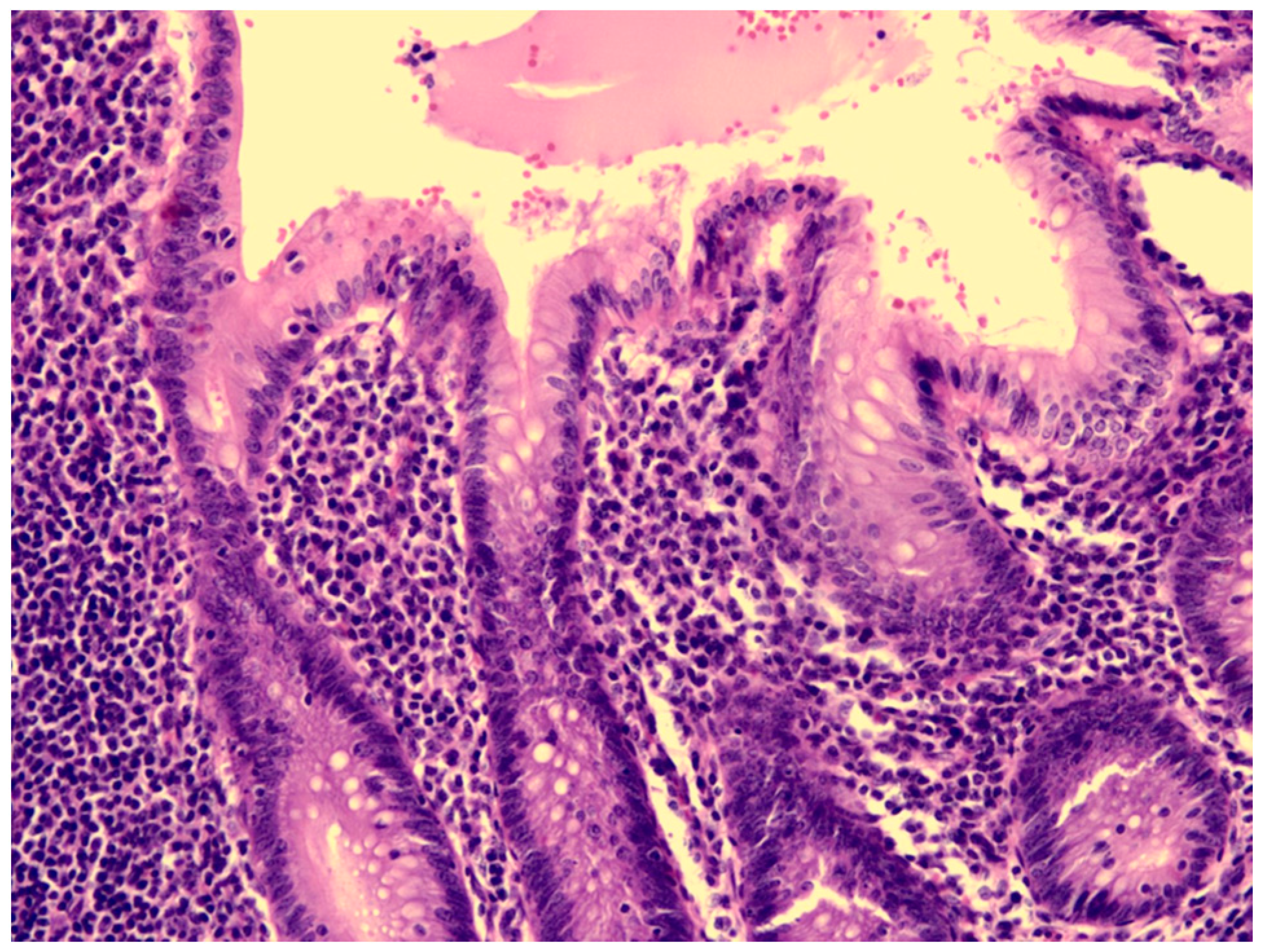
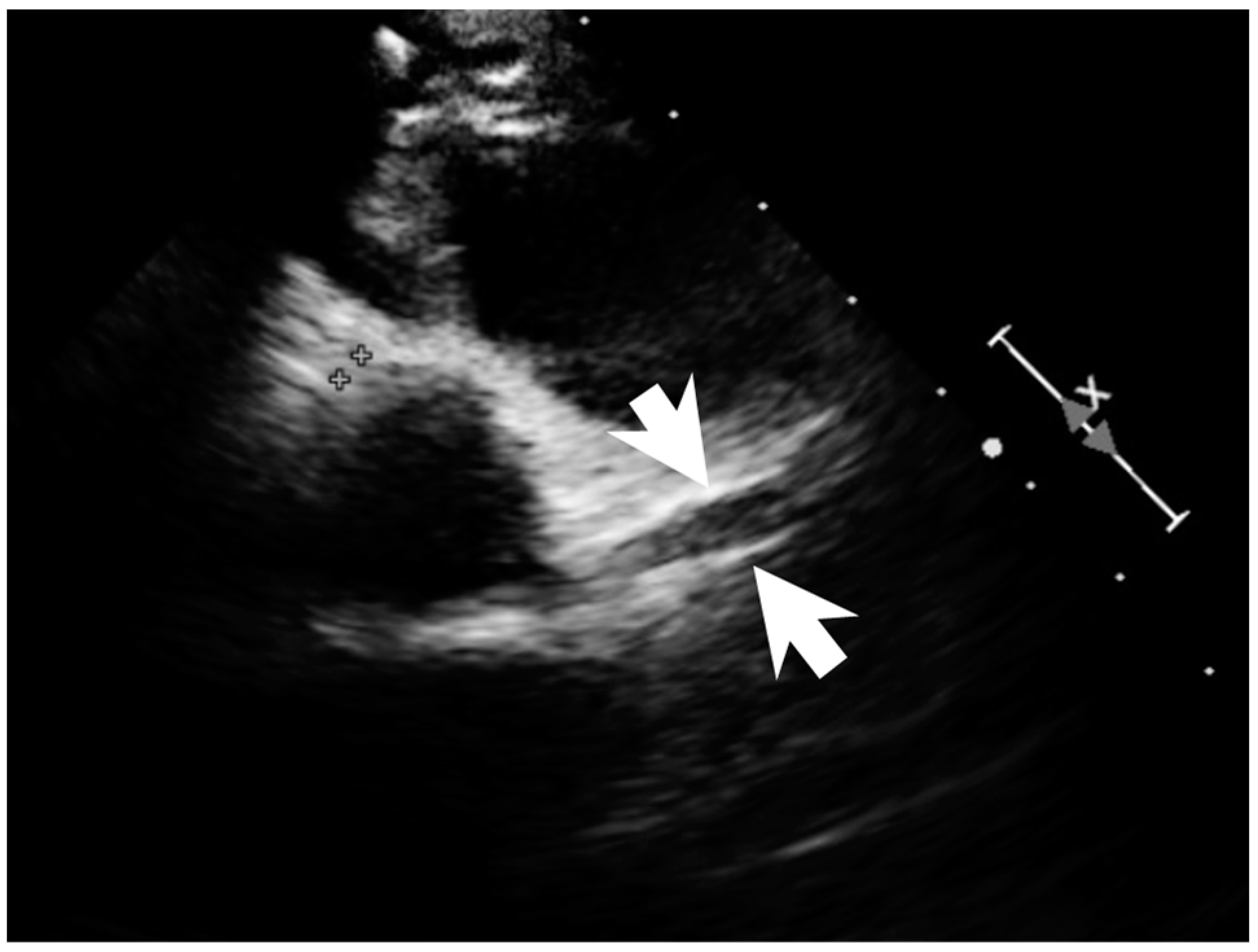
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gamez-Gonzalez, L.B.; Murata, C.; Munoz-Ramirez, M.; Yamazaki-Nakashimada, M. Clinical manifestations associated with Kawasaki disease shock syndrome in Mexican children. Eur. J. Pediatr. 2013, 172, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Newburger, J.W.; Takahashi, M.; Gerber, M.A.; Gewitz, M.H.; Tani, L.Y.; Burns, J.C.; Shulman, S.T.; Bolger, A.F.; Ferrieri, P.; Baltimore, R.S.; et al. Diagnosis, treatment, and long-term management of Kawasaki disease: A statement for health professionals from the Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation 2004, 110, 2747–2771. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Ayusawa, M.; Suzuki, H.; Abe, J.; Ito, S.; Kato, T.; Kamada, M.; Shiono, J.; Suda, K.; Tsuchiya, K.; et al. Revision of diagnostic guidelines for Kawasaki disease (6th revised edition). Pediatr. Int. 2020, 62, 1135–1138. [Google Scholar] [CrossRef] [PubMed]
- Kanegaye, J.T.; Wilder, M.S.; Molkara, D.; Frazer, J.R.; Pancheri, J.; Tremoulet, A.H.; Watson, V.E.; Best, B.M.; Burns, J.C. Recognition of a Kawasaki disease shock syndrome. Pediatrics 2009, 123, e783–e789. [Google Scholar] [CrossRef]
- Gamez-Gonzalez, L.B.; Moribe-Quintero, I.; Cisneros-Castolo, M.; Varela-Ortiz, J.; Munoz-Ramirez, M.; Garrido-Garcia, M.; Yamazaki-Nakashimada, M. Kawasaki disease shock syndrome: Unique and severe subtype of Kawasaki disease. Pediatr. Int. 2018, 60, 781–790. [Google Scholar] [CrossRef]
- Park, W.Y.; Lee, S.Y.; Kim, G.B.; Song, M.K.; Kwon, H.W.; Bae, E.J.; Choi, E.H.; Park, J.D. Clinical aspects for differential diagnosis of Kawasaki disease shock syndrome: A case control study. BMC Pediatr. 2021, 21, 25. [Google Scholar] [CrossRef]
- Huang, Y.N.; Liu, L.H.; Chen, J.J.; Tai, Y.L.; Duh, Y.C.; Lin, C.Y. Appendicitis as a Leading Manifestation of Kawasaki Disease in Older Children. Children 2022, 9, 193. [Google Scholar] [CrossRef]
- Lin, M.T.; Fu, C.M.; Huang, S.K.; Huang, S.C.; Wu, M.H. Population-based study of Kawasaki disease shock syndrome in Taiwan. Pediatr. Infect. Dis. J. 2013, 32, 1384–1386. [Google Scholar] [CrossRef]
- Taddio, A.; Rossi, E.D.; Monasta, L.; Pastore, S.; Tommasini, A.; Lepore, L.; Bronzetti, G.; Marrani, E.; Mottolese, B.D.; Simonini, G.; et al. Describing Kawasaki shock syndrome: Results from a retrospective study and literature review. Clin. Rheumatol. 2017, 36, 223–228. [Google Scholar] [CrossRef]
- Chen, P.S.; Chi, H.; Huang, F.Y.; Peng, C.C.; Chen, M.R.; Chiu, N.C. Clinical manifestations of Kawasaki disease shock syndrome: A case-control study. J. Microbiol. Immunol. Infect. 2015, 48, 43–50. [Google Scholar] [CrossRef]
- Maddox, R.A.; Person, M.K.; Kennedy, J.L.; Leung, J.; Abrams, J.Y.; Haberling, D.L.; Schonberger, L.B.; Belay, E.D. Kawasaki Disease and Kawasaki Disease Shock Syndrome Hospitalization Rates in the United States, 2006–2018. Pediatr. Infect. Dis. J. 2021, 40, 284–288. [Google Scholar] [CrossRef]
- Zulian, F.; Falcini, F.; Zancan, L.; Martini, G.; Secchieri, S.; Luzzatto, C.; Zacchello, F. Acute surgical abdomen as presenting manifestation of Kawasaki disease. J. Pediatr. 2003, 142, 731–735. [Google Scholar] [CrossRef]
- Kuroda, K.; Stottlemyre, M. Acute Appendicitis Associated With Kawasaki Disease: Case Report and Review of the Literature. Cureus 2021, 13, e18997. [Google Scholar] [CrossRef]
- Zheng, Z.; Huang, Y.; Wang, Z.; Tang, J.; Chen, X.; Li, Y.; Li, M.; Zang, C.; Wang, Y.; Wang, L.; et al. Clinical Features in Children With Kawasaki Disease Shock Syndrome: A Systematic Review and Meta-Analysis. Front. Cardiovasc. Med. 2021, 8, 736352. [Google Scholar] [CrossRef]
- Liang, Y.C.; Chang, C.H.; Lin, M.T.; Kao, F.Y.; Huang, S.K.; Wu, M.H. Shock and unresponsiveness to repeated courses of intravenous immunoglobulin in Kawasaki disease: A nationwide database study. Pediatr. Res. 2020, 87, 961–966. [Google Scholar] [CrossRef]
- Miyamoto, K.; Yamazaki, Y.; Okamoto, K.; Tsuboi, T.; Hirao, J.; Arisaka, O. Kawasaki disease: Relationship between acute surgical abdomen and cytokine profiles. Pediatr. Infect. Dis. J. 2013, 32, 1299. [Google Scholar] [CrossRef]
- Li, Y.; Zheng, Q.; Zou, L.; Wu, J.; Guo, L.; Teng, L.; Zheng, R.; Jung, L.K.L.; Lu, M. Kawasaki disease shock syndrome: Clinical characteristics and possible use of IL-6, IL-10 and IFN-gamma as biomarkers for early recognition. Pediatr. Rheumatol. Online J. 2019, 17, 1. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Nomura, Y.; Masuda, K.; Koriya, C.; Arata, M.; Hazeki, D.; Yanagimoto, K.; Ueno, K.; Eguchi, T.; Kawano, Y. Serum procalcitonin value is useful for predicting severity of Kawasaki disease. Pediatr. Infect. Dis. J. 2012, 31, 523–525. [Google Scholar] [CrossRef]
- Ono, R.; Shimizu, M.; Yamamoto, K.; Umehara, N.; Manabe, A. Kawasaki disease shock syndrome: Case report and cytokine profiling. Pediatr. Int. 2019, 61, 620–622. [Google Scholar] [CrossRef]
- Kobayashi, T.; Saji, T.; Otani, T.; Takeuchi, K.; Nakamura, T.; Arakawa, H.; Kato, T.; Hara, T.; Hamaoka, K.; Ogawa, S.; et al. Efficacy of immunoglobulin plus prednisolone for prevention of coronary artery abnormalities in severe Kawasaki disease (RAISE study): A randomised, open-label, blinded-endpoints trial. Lancet 2012, 379, 1613–1620. [Google Scholar] [CrossRef]
- Chen, S.; Dong, Y.; Yin, Y.; Krucoff, M.W. Intravenous immunoglobulin plus corticosteroid to prevent coronary artery abnormalities in Kawasaki disease: A meta-analysis. Heart 2013, 99, 76–82. [Google Scholar] [CrossRef] [PubMed]
| Reference Study (Publication Year) | Surgical Intervention | Postoperative Findings | Pathological Confirmation of Appendicitis | KD Type |
|---|---|---|---|---|
| Our patient | Yes | Hyperemic and engorged appendix with fibrin coating | Yes | Complete KD |
| Taddio, A. et al. (2017) [9] | Yes | Thickening of the ileal loops, consistent with mesenteric vasculitis | No | NA |
| Gamez-Gonzalez, L.B. et al. (2013) [1] | Yes | Positive for peritoneal cavity effusion | No | Incomplete KD |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, Y.-H.; Lin, C.-Y.; Liu, L.-H.; Huang, F.-H.; Cheng, Y.-J. Acute Appendicitis as the Initial Presentation of Kawasaki Disease Shock Syndrome in Children. Children 2022, 9, 1819. https://doi.org/10.3390/children9121819
Chang Y-H, Lin C-Y, Liu L-H, Huang F-H, Cheng Y-J. Acute Appendicitis as the Initial Presentation of Kawasaki Disease Shock Syndrome in Children. Children. 2022; 9(12):1819. https://doi.org/10.3390/children9121819
Chicago/Turabian StyleChang, Yuan-Hao, Chien-Yu Lin, Lu-Hang Liu, Fu-Huan Huang, and Yu-Jyun Cheng. 2022. "Acute Appendicitis as the Initial Presentation of Kawasaki Disease Shock Syndrome in Children" Children 9, no. 12: 1819. https://doi.org/10.3390/children9121819
APA StyleChang, Y.-H., Lin, C.-Y., Liu, L.-H., Huang, F.-H., & Cheng, Y.-J. (2022). Acute Appendicitis as the Initial Presentation of Kawasaki Disease Shock Syndrome in Children. Children, 9(12), 1819. https://doi.org/10.3390/children9121819







