Abstract
Precision psychiatry aims to overcome clinical heterogeneity by means of biomarkers that allow predicting the clinical evolution and therapeutic response in psychiatric disorders. This literature review addresses its prognostic role and its potential integration into healthcare practice. The main objective was to compile and synthesize current evidence on prognostic biomarkers in psychiatry, evaluating their usefulness in anticipating clinical evolution, therapeutic response, and risk of relapse. A strategic search was carried out on PubMed, selecting original studies that evaluated blood, genetic, epigenetic, neuroimaging, or electrophysiological biomarkers with prognostic value. We included 30 final studies that met the established inclusion and exclusion criteria and were evaluated according to standardized scales (RoB 2, NOS, AXIS). Inflammatory biomarkers showed potential as clinical modulators. Metabolomic, neuroendocrine, and neurotrophic factors reflected specific biological profiles associated with response to treatment or risk of relapse. Functional connectivity and brain morphometry were useful in the therapeutic prediction and stratification of patients. Finally, genetics and epigenetics are consolidated as tools of sensitivity and pharmacological response. Taken together, the findings reveal specific prognostic utility based on the type of biomarker and the patient’s clinical context. Despite the current methodological limitations and scarce replication of studies, prognostic biomarkers represent a step towards a more personalized psychiatry based on biological mechanisms. The future integration of multimodal models will improve clinical decision-making.
1. Introduction
Prognosis and prediction of therapeutic response in psychiatry remain a complex challenge, with some unresolved questions. Precision psychiatry is a novel therapeutic area that is emerging to provide an answer to these unknowns, trying to be able to discern from early stages of the disease which treatment will be appropriate for each patient, thus optimizing interventions and reducing clinical uncertainty [1,2].
This new branch consists of the search for tools capable of making the diagnosis more precise, indicating prognosis, guiding treatment, and response to it. Precision studies aim to establish subtypes of patients using existing and recorded data, thus being able to predict the prognosis and susceptibility to treatment [3]. The subtypes must be transdiagnostic and cover multiple pathologies, thus finding common patterns that integrate biological and clinical data [4,5].
Traditional systems such as the DSM-5 [6] have limitations when it comes to representing the heterogeneity that characterizes psychiatric conditions, as they exclusively contemplate and categorize clinical symptoms [4,5]. Similarly, there is a disconnect between conventional studies and actual clinical practice, as randomized clinical trials and meta-analyses yield very broad results in broad populations without reflecting particularities [7].
On the other hand, two recent phenomena have increased the amount of information available about the natural history of different psychiatric disorders in specific individuals: important advances in their biological understanding and the widespread use of individual clinical records with comprehensive information on each patient [1]. Such information could lay the foundation for a model capable of pointing out complex nonlinear patterns that associate predictors with outcomes. Thus, software could be developed using artificial intelligence and machine learning that effectively standardizes the analysis of big data and that transfers this data to routine clinical practice and the real patient [5,7].
All of the above underscores the need to find these base tools mentioned above: biomarkers of prognosis and response to treatment. Biomarkers are defined as measurable and objective indicators of a biological condition or state. Applied to precision psychiatry, they may be able to predict the course of the disease and the effectiveness of treatment [3]. However, its inclusion in practice faces other challenges: low sensitivity and replicability, methodological variability, and the absence of a diagnostic gold standard [2,3,5].
When we talk about biomarkers in psychiatry, it is convenient to distinguish diagnostic, prognostic, and treatment-predictive biomarkers. The former have received a lot of attention recently but are limited by the fact that they actually define, instead of a specific diagnosis, a state of severity or disease that is not specific to the disorder. They are sometimes called “trait markers” [8]. The latter refer mainly to those indicators that can indicate a specific state of the disease and guide clinicians on the natural history of the disease, which is why they have also been known as “state markers” [8]. Finally, biomarkers related to treatment response are useful tools to assess adherence or predict drug responses [9,10]. They are generally considered a special type of prognostic biomarker, which could be especially useful in clinical practice.
Despite the optimism that this so-called “precision psychiatry” has been able to generate so far, the truth is that the current evidence available comes mainly from observational studies and small and/or heterogeneous samples, so we must be very cautious in the correct interpretation of its possible practical applications. Therefore, this review aims to synthesize and compile the evidence available in original studies on the existence of prognostic (and treatment-predictive) biomarkers in psychiatry, describing their ability to individualize therapeutic interventions and identify the risk of relapse and clinical course. This research identifies the achievements and the weaknesses present in the field, providing a basis for future research and improvements in clinical practice.
2. Methods
2.1. Study Design
A semi-systematic review of the literature and a summary of the available evidence are provided in the following subsections.
2.2. Inclusion and Exclusion Criteria
The inclusion criteria were based on selecting original studies in the population with psychiatric disorders. These included both experimental studies (clinical trials, intervention trials, etc.) and observational studies (cohorts, case–control, or cross-sectional). The selected psychiatric population includes both adults and children in order to represent both subgroups and the relevance of the results. Exclusion criteria ruled out reviews or meta-analyses, as a primary and direct approach to empirical data is needed. In addition, studies carried out in non-psychiatric areas, studies that did not mention biomarkers in the full text, animal-based studies, and studies on biomarkers that indicated exclusively diagnosis (and therefore no prognostic value) were excluded.
2.3. Search Strategy
The search was carried out using the PubMed database, chosen for its extensive catalog in biomedical literature and relevance in the field of biomarkers. To conduct the search, we used terms based on inclusion and exclusion criteria that could cover the major psychiatric disorders and biomarker groups described in the existing literature on precision psychiatry, in the interest of greater specificity and efficiency in data extraction. Finally, the following search strategy was used:
(“precision psychiatry” OR “personalized psychiatry” OR “psychiatric disorder” OR “schizophrenia” OR “bipolar disorder” OR “depression” OR “Mood disorder” OR “PTSD” OR “Anxiety disorder” OR “psychiatric patients” OR “telepsychiatry”)
AND
(“biomarkers” OR “genetic biomarkers” OR “biological markers” OR “neuroimaging biomarkers” OR “inflammatory biomarkers” OR “EEG biomarkers”)
AND
(“prognosis” OR “prognostic value” OR “outcome prediction” OR “functional outcomes” OR “hospital readmission” OR “relapse” OR “recurrence” OR “relapse prevention”).
After the initial search for studies, we performed a backward citation search against reference lists to ensure that no major studies were included in the review.
2.4. Selection of Studies
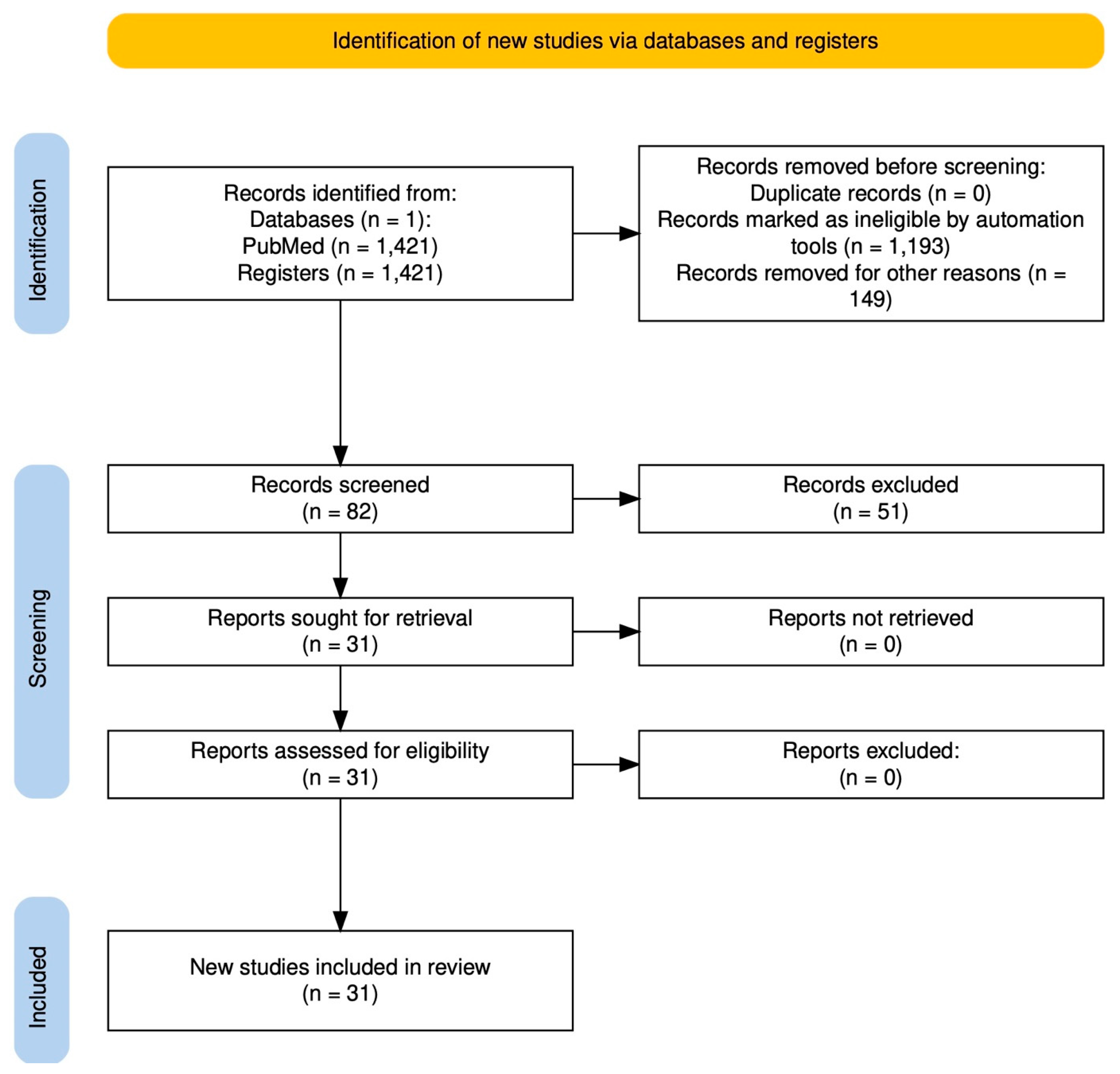
The studies identified from the search strategy were 1421. Automatic tools based on study type filters removed 1193 items. Duplicate studies totaled 0, while those eliminated for other reasons (not focusing on psychiatry) totaled 149. Rayyan web-based software (Rayyan Systems Inc., Cambridge, MA, USA) was used to review the 82 studies resulting from the search. Manual screening based on exclusion and inclusion criteria led to the selection of 30 studies that met these criteria. The complete flowchart is represented in Figure 1.
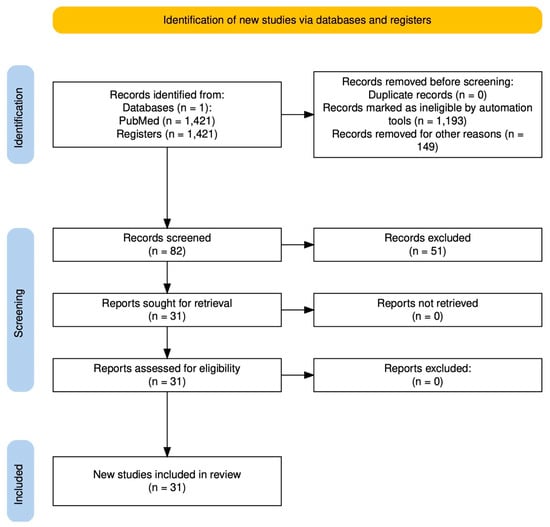
Figure 1.
Flowchart.
2.5. Data Extraction
Data extraction is found in Table 1. The author, year, and study design; the characteristics of the population studied and the sample size; the specific biomarkers evaluated; the method of evaluation and its type; level of evidence and risk of bias; and finally, the main results and objectives of the studies have been considered.

Table 1.
Revised studies.
2.6. Assessment of Quality and Risk of Bias
Various assessment scales were applied manually depending on the design of each study. For randomized clinical trials, RoB 2 (Cochrane Risk of Bias 2) and NOS (Newcastle-Ottawa Scale) were used for observational cohort or case–control studies, and the AXIS Tool was used for cross-sectional studies. In the case of exploratory genetic, proteomic, or neuroimaging studies, in the absence of specific standardized tools, adapted criteria (internal validity, methodological transparency, replicability, and sample size) were applied.
The result of this assessment is reflected in Table 1, classified as low, moderate, or high risk of bias and high, moderate, or low level of evidence. In each entry, this categorization is justified.
3. Results
The main findings of the review can be found in Table 1. Of the 30 studies that were finally included for review, 20 were longitudinal observational studies, 3 were cross-sectional studies, and 7 were experimental studies. The cumulative total n of all studies was 7363. In terms of diagnoses, 11 studies referred to the population with schizophrenia or related disorders, 16 to major depression, 2 to bipolar disorder, and 1 to generalized anxiety disorder. Regarding the methodological quality of revised studies, low levels of bias predominated, while high levels prevailed in the evidence. Of the 30 studies analyzed, 15 had a low level of bias, 14 had a moderate level, and 1 had a high level. The articles with the lowest risk of bias were mostly randomized clinical trials, with clear randomization, well-defined control groups, use of objective measurements, and robust statistical analysis.
Regarding the level of evidence, 16 studies presented high, 13 moderate, and 1 low levels. The strongest evidence came from studies with longitudinal follow-up, large samples, cross-validation, and standardized quantitative techniques.
In conclusion, the methodological evaluation offers a significantly robust empirical base, with mostly well-designed studies and no risk of critical bias.
Below are the main groups of biomarkers studied.
3.1. Immune Biomarkers
Several studies included in this review agree that inflammatory biomarkers (e.g., IL-6, IL-1β, TNF-α, and CRP) have a significant correlation with the severity of depressive symptoms, specifically in their somatic and neurovegetative dimensions. However, they do not consistently predict overall therapeutic response or the risk of relapse [11].
On the other hand, another study indicates that there are biomarkers related to subjective cognitive impairment. Plasma levels of basic FGF, INF-γ, IL-1β, MCP-1, M-CSF, and SCF were higher in patients with this clinical dimension, while IL-9, RANTES, and PDGF-BB were lower in this group. In addition, high accuracy was found in a predictive model composed of SCF and PDGF-BB combined with the initial PDQ-D (Perceived Deficits Questionnaire for Depression) assessment after antidepressant treatment [12].
In relation to remission in depression, it was found that elevated levels of IL-6 and CRP at the beginning of treatment were related to a lower probability. It was only seen in patients treated with CBT, not those treated with antidepressants [13].
Regarding the transition to psychosis, two proteomic profiling studies identified systemic inflammatory markers (complement pathways, coagulation cascade, or alpha-2-macroglobulin) with moderate predictive capacity in adolescents at high clinical risk [14,15].
Finally, it was observed that high monocyte and basophil counts were related to the risk of psychotic relapse in patients with schizophrenia in remission. In contrast to a high PRL, it behaved as a protective factor [16].
3.2. Metabolomic Biomarkers
In response to antidepressant treatment, higher baseline concentrations of phosphatidylcholine C38:1 were found to be associated with a lower reduction in depressive symptoms after treatment, suggesting an association with antidepressant resistance. In contrast, a ratio of hydroxylated sphingomyelins over non-hydroxylated sphingomyelins predicted a better response. This finding reflects a better neuronal plasticity profile [17].
Regarding schizophrenia, a proteomic signature was identified with reduced levels of leptin and proinsulin, related to an increased risk of relapse in the short term. Early insulin, leptin, and C-peptide response during the first few weeks of treatment was associated with future relapse [15].
Elevated levels of unconjugated bilirubin (UCB) were associated with greater severity of psychotic symptoms, prolonged length of hospitalization, and aggressive behaviors during relapse. This was accentuated in patients with schizoaffective disorder. In remission, a negative correlation was identified between UCB and psychomotor retardation [18].
Finally, regarding the adverse effects of antipsychotics, it was observed that lactate levels in arterial blood rose after 90 days of antipsychotic treatment. Specifically, patients who presented extrapyramidal symptoms (dystonia and Parkinsonism) had even higher values [19].
3.3. Neuroendocrine Biomarkers
An attenuated response to the dexamethasone suppression test (Dex/CRH) was associated with a lower probability of remission in patients with major depression. The presence of hypercortisolemia and poor suppression of the HPA axis during the first weeks of treatment resulted in a worse clinical prognosis. However, a progressive improvement in the regulation of the shaft after the first few weeks meant the opposite [20].
In adolescents, a higher cortisol/CRP ratio in morning saliva was shown to double the risk of developing a depressive episode in the following two years. In contrast, both variables separately were not able to predict the onset of the pathology [21].
On the other hand, alterations in nocturnal secretion of growth hormone (GH) during adolescence were associated with an increased risk of developing depression and suicidal risk in adulthood. The patterns identified consisted of early GH elevation after sleep onset and persistently high levels in the early hours of the night [22].
3.4. Neurotrophic Factors
The studies included in this review support the neurotrophic hypothesis, showing that alteration of brain-derived neurotrophic factor (BDNF) has been associated with the pathophysiology of depression and response to antidepressant treatment. BDNF levels are lower in patients with untreated depression compared to healthy controls. Similarly, higher levels are related to greater clinical remission, especially in response to antidepressants (especially SSRIs) [23].
Another study noted that an early elevation of BDNF during the first 2 weeks of antidepressant treatment is associated with a higher likelihood of remission at 8 weeks, regardless of immediate clinical improvement. Early improvement in executive functions (inhibitory control and cognitive flexibility) was also found to be positively associated with remission [24].
3.5. Genetic and Epigenetic Biomarkers
In depression, a combined predictive model of clinical biomarkers, mRNA (STMN1 and PPP1R9B), and microRNA (miR-3688 and miR-5695) expression was identified, capable of predicting with great accuracy the worsening of suicidal ideation during treatment with duloxetine [25].
Regarding bipolar disorder, one study identified four SNPs on chromosome 21 (rs79663003, rs78015114, rs74795342, and rs75222709) related to the response to lithium and located in lncRNAs AL157359.3 and AL157359.4. Similarly, in an independent cohort, it was found that carriers of these alleles had a lower risk of relapse at two years [26].
In schizophrenia, a GWAS was performed in which five genetic loci were found in relation to the general response to antipsychotics (MEGF10, SLC1A, PCDH7, CNTNAP5, and TNIK). In parallel, specific associations were identified between specific SNPs and certain drugs, such as rs2239063 in CACNA1C with response to olanzapine, rs16921385 in SLC1A1 with risperidone, and rs17022006 in CNTN4 with aripiprazole [27].
Also, miRNAs showed predictive power in antipsychotic treatment. Levels of miR-30e, miR-181b, miR-34a, miR-346, and miR-7 were found to be significantly increased in patients with schizophrenia prior to treatment, and after six weeks, levels of miR-132 and miR-432 were markedly reduced in patients who improved clinically [28].
Finally, in generalized anxiety disorder (GAD), it was found that the combination of haplotype 5-HTTLPR/rs25531 (SLC6A4) and SNP rs7997012 (HTR2A) was able to modulate the response to venlafaxine [29].
3.6. Neuroimmaging
3.6.1. Functional Connectivity (fMRI)
In young people with depression, weaker inhibitory modulation between the ventrolateral prefrontal cortex (vlPFC) and the amygdala during emotional regulation tasks was associated with greater symptom severity [30]. On the other hand, reduced excitatory connectivity between the ventromedial prefrontal cortex (vmPFC) and the amygdala after treatment with CBT and fluoxetine was associated with a higher probability of clinical remission. This means that lower prefrontal-limbic connectivity may be associated with better emotional regulation or a higher likelihood of remission [31].
In line with these findings, another study identified that greater connectivity between the amygdala and the insula was associated with a worse therapeutic response. However, in contrast to the previous paragraph, it was also found that lower baseline prefrontal-limbic connectivity was associated with a lower probability of remission. These two described patterns differentially predicted response to CBT or pharmacotherapy [13].
On the other hand, deactivation of the primary somatosensory cortex during dysphoric mood induction tasks was associated with a higher risk of long-term relapse, while lower activity in the left LPFC after treatment was associated with a lower risk of relapse [31].
In schizophrenia, 91 functional connections of the striatum with prognostic potential were identified by resting fMRI. Greater connectivity in posterior regions was associated with better clinical outcomes, as opposed to lower connectivity in frontal regions, which was associated with a better response to antipsychotic treatment. Similarly, an increased striatal connectivity index was correlated with longer duration of hospitalization in acute psychosis [32].
Finally, another study using resting fMRI identified two brain biotypes in patients with schizophrenia, based on differences in cognitive, social, and symptom performance, without depending on the DSM diagnosis. The pattern that was altered pointed to regions associated with mentalization and the network of mirror neurons [33].
3.6.2. Brain Morphometry and Structure (sMRI and DTI)
Lower fractional anisotropy (FA) in white matter tracts (anterior cingulate and stria terminalis) predicted remission by up to 64% before antidepressant treatment, increasing to 74% when adding age as a moderating variable [34].
The application of machine learning (VMS) on structural magnetic resonance imaging (sMRI) is useful in classifying response to antidepressant treatment. It allowed patients to be differentiated into refractory and non-refractory with great precision. White matter was more predictive in non-refractory patients (84.65%), while gray matter was more predictive in refractory patients (67.39%) [35].
In melancholic depression, it was found by sMRI that increased CSF volume in the left Sylvian fissure was associated with poorer response to treatment and longer time to remission. Similarly, it was found that the increase in global cortical volume of CSF was correlated with a 7.8-fold increased risk of relapse or recurrence during the two years of follow-up [36].
Moving on to juvenile bipolar disorder, brain morphometry was used before and after one week of treatment with lithium and quetiapine. The baseline structural characteristics and their early changes demonstrated great accuracy in predicting the response (75%), which increased for both quetiapine (83.2%) and lithium (83.5%) when both moments were combined. In fact, cortical thickness and surface area were used for quetiapine, while hippocampal volume and cortical area were used for lithium [37].
3.6.3. Receptors and Functional Neuroimaging (SPECT)
SPECT with 123I-IBZM and striatal/occipital (S/O) ratio were used to study patients with previously untreated schizophrenia by measuring the density of D2 dopaminergic receptors (D2R) in the striatum. It was found that a higher density of D2R in the striatum, as well as a higher S/O ratio were associated with worse clinical outcomes. In parallel, this higher striatal dopaminergic density was related to a worse premorbid fit (assessed by the PAS scale) [38].
3.7. Electroencephalography (EEG, ERPs)
Quantitative electroencephalography (qEEG) was applied to assess the brain activity of patients with depression in the early stages of treatment. An early reduction in theta concordance in the middle and right frontal region was found to correlate with a higher likelihood of remission at one week [39].
In patients with schizophrenia in remission and treated with antipsychotics, visual evoked potentials (ERPs) were used during discrimination tasks. Significantly higher latency in the NA component, as well as alterations in the N2 and P3 components, were associated with a higher likelihood of relapse in the following two years. The delay in NA latency had a sensitivity of 90% [40].
4. Discussion
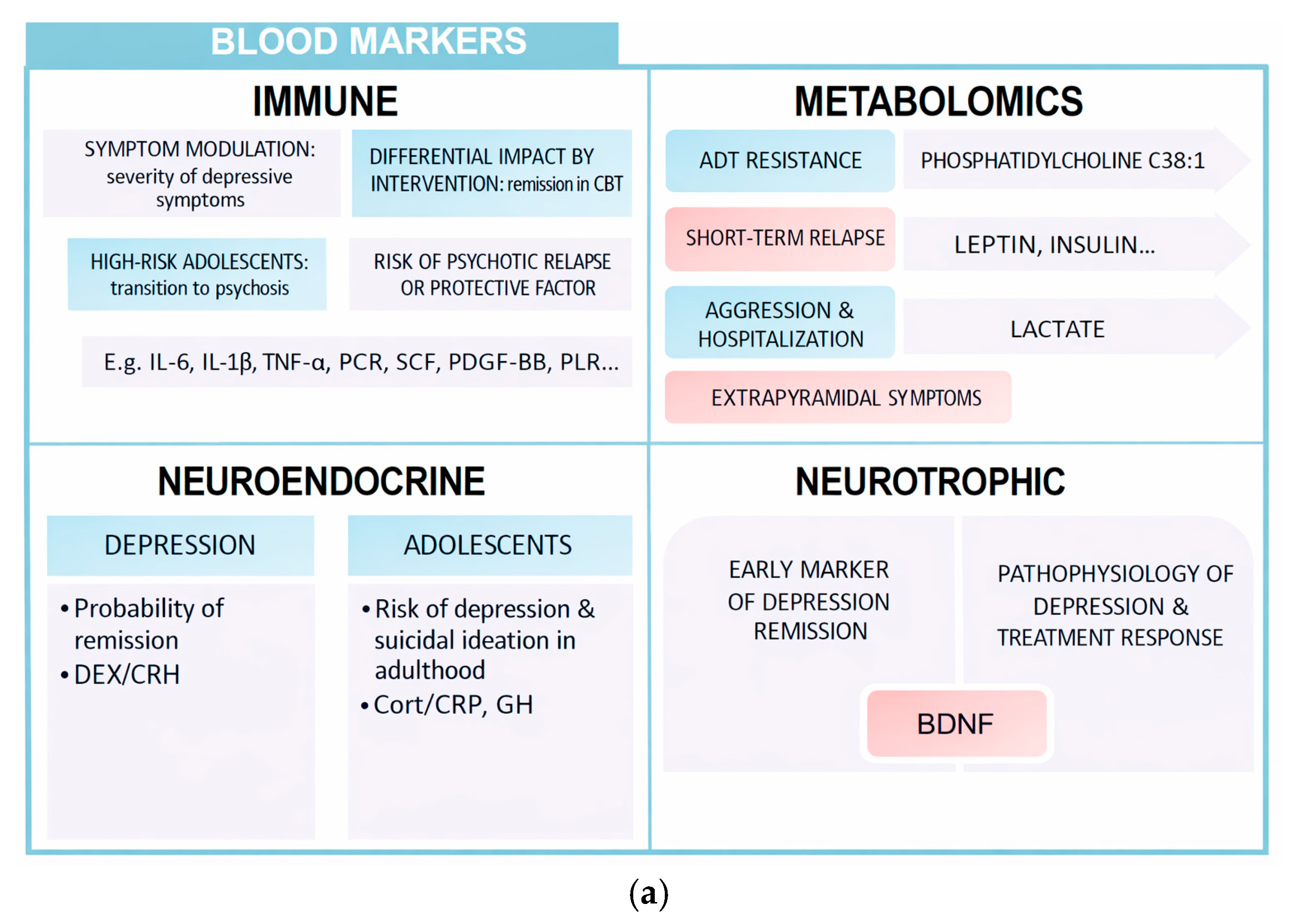
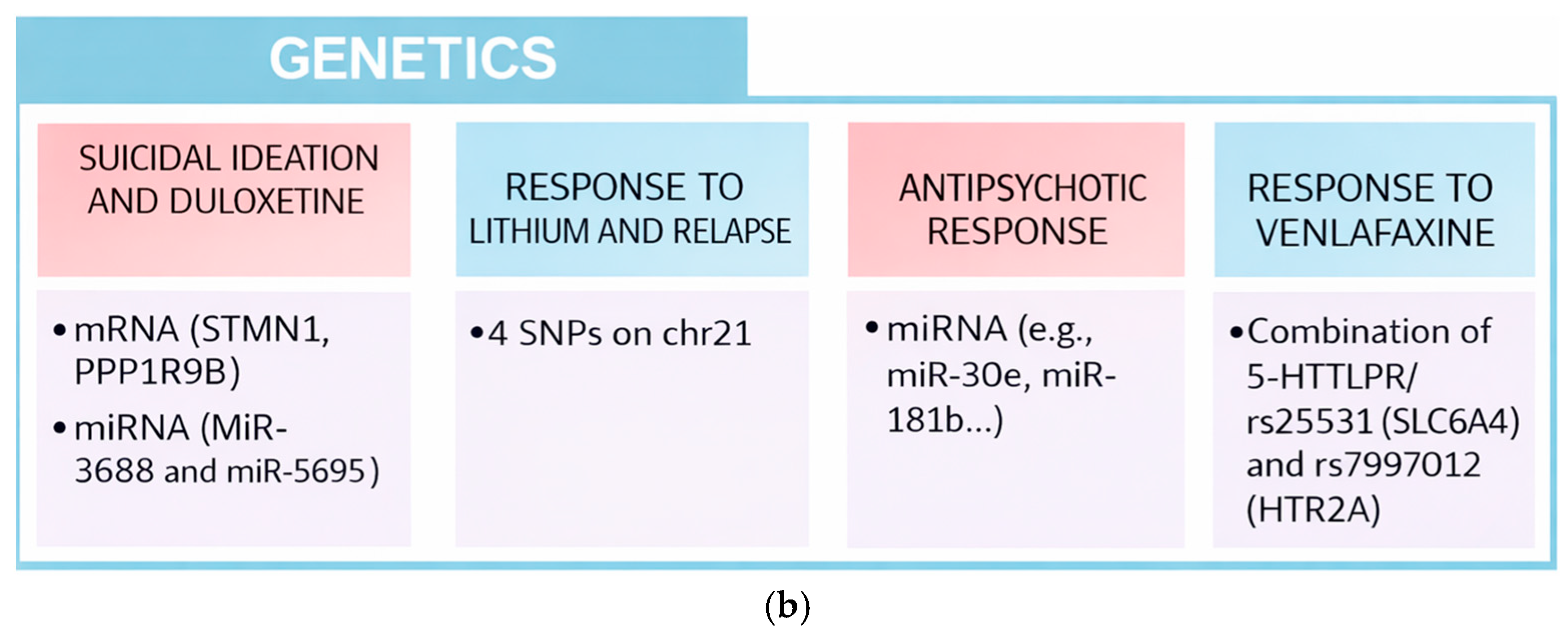
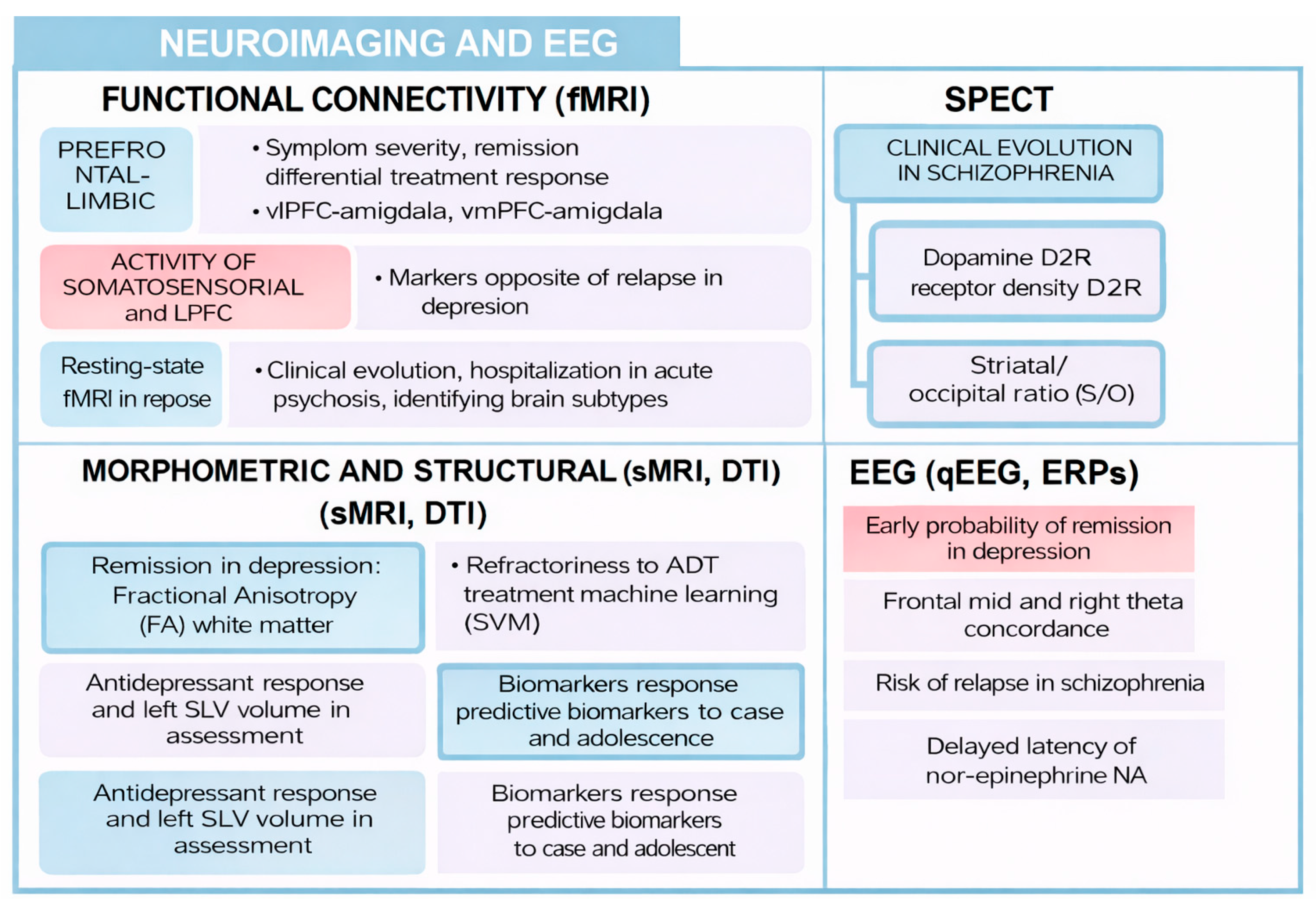
This review has collected different types of biomarkers with potential prognostic capacity in psychiatry (Figure 2 and Figure 3). Its integrated analysis has made it possible to identify which biomarkers are related to remission, relapse, or therapeutic response in various disorders. Next, its usefulness, methodological soundness, and real clinical implications in the context of precision psychiatry are discussed.
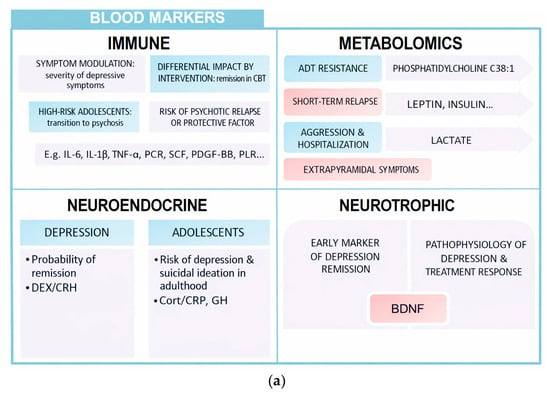
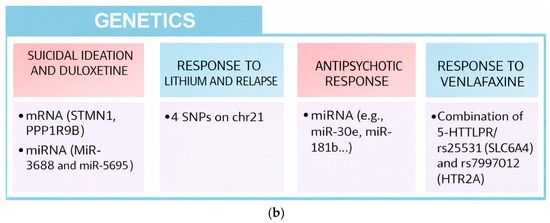
Figure 2.
(a) Blood markers. (b) Genetic markers.
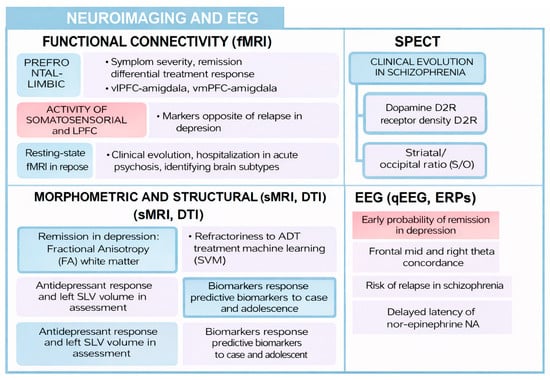
Figure 3.
Neuroimaging and EEG biomarkers.
Peripheral markers allow an approximation to the biological substrate of many mental illnesses, with high prognostic value. This summary table shows some of the most important aspects that have been the subject of this review: immune, metabolomic, neuroendocrine, neurotrophic, and genetic.
Both modern neuroimaging techniques and electroencephalography are indirect methods of visualizing brain structure and functioning. In this review, several markers have been discussed that could have a role in certain prognostic variables associated with highly prevalent mental disorders.
4.1. Interpretation of the Results
The biomarkers collected in this review should not be understood as universal and stable markers, but as objective and dynamic indicators that acquire predictive value depending on the clinical and therapeutic context. In other words, they do not generally correspond to absolute values; they must be interpreted according to the relationship with the type of intervention we use, the time of the natural history of the disorder, and, of course, the patient’s profile.
Inflammatory biomarkers such as IL-6, IL-1β, TNF-α, and CRP emerged as symptomatic modulators of depression, not as indicators of global response to treatment or relapse. They were related to symptomatic severity in somatic and neurovegetative dimensions and subjective cognitive impairment. This means that they should be understood as modulators of the clinical profile [11,12]. Similarly, their ability to predict remission was closely related to the type of intervention provided, which was higher in patients treated with CBT. This reinforces the differential and contextual impact of these biomarkers [13].
Also worthy of mention in this subtype of biomarkers are the increased counts of monocytes and basophils at risk of relapse into schizophrenia. The relationship with monocytes is consistent with previous studies on the activation of microglia and dysfunction of the immune system in psychosis, while that of basophils is novel. Moreover, PLR as a protective factor implies that the balance between innate and adaptive immunity would regulate the risk of relapse [16].
Regarding metabolomic biomarkers, an interesting debate has been opened. The fact that phosphatidylcholine C38:1 is associated with a worse antidepressant response suggests that the patient’s baseline biochemistry tells us how he or she will respond. This would therefore imply that resistance to treatment would be present in the lipid profile. On the other hand, the relationship between sphingomyelin ratios and the antidepressant response reinforces the idea that neuronal plasticity is clearly linked to brain lipid metabolism [17].
We must not forget hormonal markers and their potential as predictors of relapse in schizophrenia. Leptin, C-peptide, proinsulin, and insulin are altered early during the first weeks of treatment, showing predictive potential to warn us of decompensation before the mind [15]. Furthermore, the relationship of UCB with aggressiveness and prolonged hospitalization in psychosis indicates structural vulnerability (especially in the schizoaffective phenotype) and is not an evolutionary marker [18]. Finally, regarding the adverse effects of antipsychotics, the increased lactate levels in patients who develop dystonia or Parkinsonism underline the importance of peripheral mitochondrial metabolism. This is because it could alert us to neurological toxicity before it manifests itself clinically [19].
When talking about neuroendocrine markers, we must keep in mind that the HHA axis not only tells us about stress but also about physiological rigidity. This means that a neuroendocrine system that does not react regardless of the treatment we provide or the environment is associated with a worse prognosis in depression [20]. If we add to this that the cortisol/CRP ratio in adolescents was not able to predict separately, it indicates that the problem is not at a single level. In other words, there is a loss of synchrony between systems (in this case, neuroendocrine and immune) [21]. Finally, the alteration of nocturnal GH secretion in adolescents suggests that circadian rhythms at this stage would be a useful marker of future vulnerability [22].
Moving on to neurotrophic biomarkers, BDNF again demonstrates that it is not a state marker but rather reflects global plasticity. It would indicate a worse adaptation of the nervous system of depressive patients and, by extension, guide which patients will respond and respond to treatment [23]. At the same time, this biomarker shows potential as a bridge between affective and cognitive symptomatology. That is, given its role in neurogenesis and memory, it could be linked to the improvement of cognitive functions [24].
Genetic biomarkers should be understood as predictors of sensitivity, i.e., they are useful for identifying which patients are more vulnerable to the outside and their likelihood of responding to treatment (e.g., polymorphisms or miRNAs related to response to antipsychotics) [27,28]. Above all, in this category, the importance lies in the gene-environment interaction and in how genes are expressed through epigenetics, since they are genes responsible for neuroplasticity, inflammation, and emotional response. In conclusion, it is not enough to look for a single depression gene but to integrate it into multivariate models that integrate the clinical phenotype [25,26,29]. Similarly, we must also highlight the preliminary usefulness they have shown to anticipate the response to various antidepressant and antipsychotic drugs, laying the foundations of pharmacogenetics. Functional connectivity studies indicate that there are alterations in the cross-communication of a set of regions involved in various brain processes, identifying failures in executive, perceptual, and affective circuits. However, discordance about the probability of remission and prefrontal–limbic connectivity could indicate that they are indeed context-dependent dynamic biomarkers [13,30,31]. Similarly, they are biomarkers that show potential in the prediction of the therapeutic response with differential impact, as we observed in immune markers (CBT and pharmacotherapy) [13]. Somatosensory and lateral prefrontal cortex (LPFC) deactivation are considered to be opposite predictors of depression relapse. In other words, while the former was associated with greater vulnerability in the long term, the latter paradoxically did so with a lower probability of relapse. This indicates that patients regulate their emotions in a more automatic and less forced way. Not only is it important which areas are activated, but also how much energy is used [31].
In schizophrenia, the use of resting fMRI is useful in anticipating response and clinical evolution. Specifically, the connectivity of the differential striatum with posterior and frontal regions could indicate that the more disconnected from frontal control it is, the more effective the drugs are. This could be interpreted as meaning that in these patients, the disorder would be more focused on the classic dopaminergic pathway and, therefore, they are more sensitive to the blockade of antipsychotics. In parallel, an increased global index of striatal connectivity suggests greater functional disorganization in acute phases, thus being related to the duration of hospitalization [32]. Finally, resting fMRI also allows differentiating two biotypes based on connectivity in mentalization networks and mirror neuron systems (cognitive differences, social functionality, and symptomatic load), contributing to patient stratification [33].
Finally, to elaborate on the neuroimaging and electrophysiological biomarkers, the included study using SPECT shows potential to indirectly analyze dopaminergic pathways in naïve schizophrenia. This means that a higher density of D2 receptors or S/O ratio would help us to anticipate a worse clinical outcome [38]. On the other hand, the most relevant thing about EEG is that it reflects emotional reactivity and not only basal activation, which gives it great potential for the evaluation of the antidepressant therapeutic response and the personalization of strategies at an early stage [39]. In addition, the delay of NA latency in schizophrenia showed great sensitivity as an independent predictor of relapse, as it was related to an alteration in early perceptual processing [40].
4.2. Limitations of the Included Studies
Despite the great potential shown in the previous section, the studies collected in this review have important methodological limitations that restrict their current clinical applicability. The small sample size in a large part of the studies is particularly noteworthy, especially in those of functional connectivity and some inflammatory studies.
If we focus on studies focused on machine learning, for example, we find that many of these analyses are based on small samples and limited validation strategies. So, these findings should be framed as exploratory or proof-of-concept rather than as evidence of robust predictive biomarkers. Greater reproducibility and external validation of these findings are therefore needed. Another limitation to note is the low level of external replication in many studies that propose predictive models using machine learning or multivariate analysis. Methodological heterogeneity is also a problem, as well as the use of different types of neuroimaging tests, analytical protocols, clinical classifications, statistical thresholds, and inclusion criteria. This makes it difficult to compare between studies and draw conclusions. Publication bias is also relevant, as few studies report negative results or apply corrections for multiple comparisons. Finally, in some subtypes of biomarkers (such as metabolomic or structural), the observational design and exploratory nature of the studies do not allow causal relationships to be established. In conclusion, these discrepancies explain the current low integration of biomarkers in healthcare psychiatry. This means that, despite the promising advances of this new biological psychiatry, the existence of clinical decision rules based on biomarkers that can be generalized to a multitude of different disorders may be a reality that is, in fact, not as close to being achieved as we would like.
4.3. Strengths and Limitations of the Review
This review presents a series of strengths that support the validity of the findings described. Firstly, it is comprehensive and compiles different types of blood, genetic, neuroimaging, and electrophysiological biomarkers in an integrative manner, encompassing different methodologies and prognostic perspectives. This has allowed a comparative interpretation to be made between clinical dimensions and biological systems involved. In addition, both the adult, child, and adolescent populations have been contemplated with the aim of increasing representativeness. The clinical perspective, focused on the prognostic value (clinical evolution, response to treatment, and relapse), favors applicability to real clinical practice.
Another relevant strength is that the methodology has followed a logical sequence of search and selection of studies, which has ensured a transparent and replicable structure. Clear inclusion and exclusion criteria were defined, a methodical screening process was carried out, and the findings were organized according to biomarker subtype. The selection of final studies is rigorous, as it includes only original studies and excludes other reviews, meta-analyses, and preclinical studies. Regarding the table of results, it is extensive and complete, collecting key information from each study and validated in each entry against the original article. Similarly, a meticulous assessment of the level of evidence and the risk of bias of the studies has been carried out, using standardized tools adapted to each design (RoB 2, NOS, AXIS).
However, this review is not without limitations. The main one is that it is not a systematic review in the strict sense, as peer screening has not been carried out, and there is no search protocol registered. This may lead to some selection biases that limit the validity of the conclusions. Similarly, despite various psychiatric disorders, there is a clear predominance of depression and schizophrenia, leaving aside other disorders. Something similar occurs with the predominance of studies focused on adults, with the child and adolescent population being underrepresented. Likewise, the articles included were limited to the English language.
On the other hand, the possibility of generalizing results has been limited due to the great clinical and methodological heterogeneity between studies. For this reason, this is a qualitative and descriptive review, without joint statistical analysis (meta-analysis). Despite having carried out an evaluation of methodological quality, the design of some of the studies is observational and exploratory, with little replication and decreased sample sizes. In addition to all this, there is a significant dispersion of results (especially in functional connectivity and inflammation) that limits the extraction of consistent results.
4.4. Clinical Implications and Future Research
Biomarkers show great potential to be the next step in the evolution of a more individualized, objective, and biology-based psychiatry. This review has highlighted its ability to predict clinical evolution, therapeutic response, and risk of relapse. However, their actual applicability remains limited, and the integration of biomarkers into healthcare practice remains in the preliminary phase.
Currently, its most reasonable clinical value is not diagnostic but prognostic and stratifying. Its use would make it possible to personalize prevention and follow-up strategies, with the aim of identifying patients at greater risk of relapse or chronification. In this sense, the distinction between static (morphometric or genetic) and dynamic (inflammatory, EEG, or functional connectivity) biomarkers will be of great importance in devising tools adapted to the different phases of the clinical course.
Future research should be directed towards the design of multimodal predictive models that integrate biomarker subtypes. This convergence will allow us to reflect the biological complexity of psychiatric disorders and design clinically useful predictors. Therefore, the development of these models must be supported by artificial intelligence capable of integrating heterogeneous data and guiding clinical decision-making. Similarly, adopting transdiagnostic approaches that go beyond traditional categories would allow us to identify common vulnerability mechanisms and profiles.
By delving into each subtype of biomarker and its specific applications, inflammatory biomarkers allow the identification of clinical profiles with somatic or neurovegetative symptoms or subjective cognitive impairment. Its most promising application would be as modulators of clinical phenotype and as intervention guides (e.g., use of CBT in proinflammatory profiles). Metabolomic biomarkers reflect the patient’s baseline biochemistry, which is useful for predicting therapeutic resistance or vulnerability to adverse effects. Neuroendocrine biomarkers inform us about the reactivity of the HPA axis and its interaction with other systems (e.g., immune), indicating physiological styles of adaptation and vulnerability to stress. As for neurotrophic drugs, they allow us to analyze brain plasticity and can be used to monitor the biological response to treatment.
Functional connectivity shows great potential to more accurately predict therapeutic response and risk of relapse and to define transdiagnostic functional biotypes for the individualization of treatments. Structural neuroimaging acts as a trait biomarker, helping to identify resistance, chronicity, or low plasticity. On the other hand, SPECT allows us to evaluate dopaminergic activity in vivo and apply it in the stratification of subtypes sensitive to antipsychotics. Finally, EEG, being so accessible and sensitive to change, shows potential to be integrated as a monitoring and therapeutic anticipation tool, especially in the early stages.
The gradual introduction of these biomarkers in the coming years will not only depend on technical advances but also on the ability of the system to guarantee their accessibility, standardization, and ethical use. Biomarkers will be really useful in diverse healthcare settings, not only in highly specialized environments. This implies moving towards cost-effective techniques, homogeneous protocols, and common interpretative criteria. Above all, its incorporation must prioritize equity in access, data protection, and the ethical impact of the use of biomarkers in mental health.
5. Conclusions
This work highlights the potential of biomarkers as complementary tools in psychiatry, with specific applications in the prediction of the clinical course, therapeutic response, and risk of relapse. Far from replacing the clinic, these tools should be understood as a natural extension of psychiatry. In this sense, they do not come to resolve the debate between the objective and the subjective but to offer one more way of approaching the complexity of the human being.
Throughout this review, different subtypes of biomarkers have been explored, and consistent patterns have been identified. Although the healthcare integration of these indicators is still limited, this review shows that there are already solid findings that expand the path of a more precise and less reductionist psychiatry. However, the results obtained should be interpreted with caution since the included studies have some relevant limitations, such as small sample size, heterogeneity of designs, low external replication, or publication bias.
Looking ahead, the development of validated and replicable multimodal predictive models that relate biological data to clinical trajectories is needed. This will involve incorporating contextual variables such as the psychopathological profile, the therapeutic history, and the psychosocial environment of the patient. Similarly, it will be necessary to implement criteria of clinical applicability and equitable accessibility to guarantee its ethical and effective use. The standardization of protocols, the training of health personnel, and the design of affordable and interpretable tools will be essential steps for their gradual implementation.
The future of psychiatry could lie in overcoming the dichotomy of the biological and psychosocial [41]. Biomarkers do not represent a biological dogma but an integrating possibility by recognizing the biological uniqueness of each patient and, at the same time, their human experience. A truly integrative psychiatry will be able to combine empathic listening with biological reading. So, biomarkers are an inevitable step in the progress of psychiatry towards a more humane, precise, and fair medicine.
Author Contributions
Conceptualization, I.M.B.-B., J.H.-I. and I.R.; methodology, I.M.B.-B., J.H.-I. and I.R.; software, I.M.B.-B.; validation, L.G.-R. and S.J.-F.; formal analysis, I.M.B.-B.; investigation, I.M.B.-B., B.I.-R. and G.P.-G.; resources, J.H.-I. and I.R.; data curation, I.M.B.-B., J.H.-I. and I.R.; writing—original draft preparation, I.M.B.-B., G.P.-G. and B.I.-R.; writing—review and editing, J.H.-I., I.R., L.G.-R. and S.J.-F.; visualization I.M.B.-B., J.H.-I. and I.R.; supervision, J.H.-I. and I.R. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki. As it is a review work, this study did not need to be approved by any institutional ethics committee.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Acknowledgments
The authors thank their co-workers from the research groups IBIMA-C-03 and CTS-549, as well as the Departments of Psychiatry of the Universities of Malaga and Granada, for their support during this research.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AXIS | Appraisal Tool for Cross-Sectional Studies |
| BDNF | Brain-Derived Neurotrophic Factor |
| CRH | Corticotropin-Releasing Hormone |
| CRP | C-Reactive Protein |
| DSM | Diagnostic and Statistical Manual of Mental Disorders |
| DTI | Diffusor Tensor Imaging |
| EEG | Electroencephalogram |
| FA | Fractional Anisotropy |
| GH | Growth Hormone |
| GWAS | Genome-Wide Association Study |
| HPA | Hypothalamic–Pituitary–Adrenal Axis |
| IL | Interleukin |
| INF | Interferon |
| SSRI | Selective Serotonin Reuptake Inhibitors |
| CSF | Cerebrospinal Fluid |
| LPFC | Lateral Prefrontal Cortex |
| mRNA | Messenger RNA |
| miRNA | MicroRNA |
| MRI | Magnetic Resonance Imaging (fMRI: functional) |
| NA | Negative Afterpotential (potencial visual tardío) |
| NOS | Newcastle-Ottawa Scale |
| PLR | Platelet-to-Lymphocyte Ratio |
| SAF | Schizoaffective Disorder |
| SCF | Stem Cell Factor |
| SVM | Support Vector Machines |
| GAD | Generalized Anxiety Disorder |
| CBT | Cognitive-Behavioral Therapy |
| TNF | Tumor Necrosis Factor |
| UCB | Unconjugated Bilirubin |
References
- Serretti, A. Precision Psychiatry. Braz. J. Psychiatry 2022, 44, 115–116. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, B.S.; Quevedo, J.; Zhao, Z. Fostering Precision Psychiatry through Bioinformatics. Braz. J. Psychiatry 2022, 44, 119–120. [Google Scholar] [CrossRef]
- Van Dellen, E. Precision Psychiatry: Predicting Predictability. Psychol. Med. 2024, 54, 1500–1509. [Google Scholar] [CrossRef]
- Williams, L.M.; Carpenter, W.T.; Carretta, C.; Papanastasiou, E.; Vaidyanathan, U. Precision Psychiatry and Research Domain Criteria: Implications for Clinical Trials and Future Practice. CNS Spectr. 2024, 29, 26–39. [Google Scholar] [CrossRef]
- Lin, E.; Lin, C.H.; Lane, H.Y. Precision Psychiatry Applications with Pharmacogenomics: Artificial Intelligence and Machine Learning Approaches. Int. J. Mol. Sci. 2020, 21, 969. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; (DSM-5); CBS Publishers & Distributors Pvt. Ltd.: Delhi, India, 2013; ISBN 0890425558. [Google Scholar]
- Passos, I.C.; Ballester, P.; Rabelo-da-Ponte, F.D.; Kapczinski, F. Precision Psychiatry: The Future Is Now. Can. J. Psychiatry 2022, 67, 21–25. [Google Scholar] [CrossRef]
- Chen, Y.; Cinnamon Bidwell, L.; Norton, D. Trait vs. State Markers for Schizophrenia: Identification and Characterization through Visual Processes. Curr. Psychiatry Rev. 2006, 2, 431–438. [Google Scholar] [CrossRef]
- Herrera-Imbroda, J.; Flores-López, M.; Requena-Ocaña, N.; Araos, P.; Ropero, J.; García-Marchena, N.; Bordallo, A.; Suarez, J.; Pavón-Morón, F.J.; Serrano, A.; et al. Antipsychotic Medication Influences the Discriminative Value of Acylethanolamides as Biomarkers of Substance Use Disorder. Int. J. Mol. Sci. 2023, 24, 9371. [Google Scholar] [CrossRef]
- Herrera-Imbroda, J.; Flores-López, M.; Requena-Ocaña, N.; Araos, P.; García-Marchena, N.; Ropero, J.; Bordallo, A.; Suarez, J.; Pavón-Morón, F.J.; Serrano, A.; et al. Antidepressant Medication Does Not Contribute to the Elevated Circulating Concentrations of Acylethanolamides Found in Substance Use Disorder Patients. Int. J. Mol. Sci. 2023, 24, 14788. [Google Scholar] [CrossRef]
- Kofod, J.; Elfving, B.; Nielsen, E.H.; Mors, O.; Köhler-Forsberg, O. Depression and Inflammation: Correlation between Changes in Inflammatory Markers with Antidepressant Response and Long-Term Prognosis. Eur. Neuropsychopharmacol. 2022, 54, 116–125. [Google Scholar] [CrossRef]
- Wang, M.; Wei, Z.; Huang, Q.; Yang, W.; Wu, C.; Cao, T.; Zhao, J.; Lyu, D.; Wang, F.; Zhou, N.; et al. Prognostic Prediction of Subjective Cognitive Decline in Major Depressive Disorder Based on Immune Biomarkers: A Prospective Observational Study. BMC Psychiatry 2023, 23, 54. [Google Scholar] [CrossRef] [PubMed]
- Dunlop, B.W.; Binder, E.B.; Cubells, J.F.; Goodman, M.M.; Kelley, M.E.; Kinkead, B.; Kutner, M.; Nemeroff, C.B.; Newport, D.J.; Owens, M.J.; et al. Predictors of Remission in Depression to Individual and Combined Treatments (PReDICT): Study Protocol for a Randomized Controlled Trial. Trials 2012, 13, 106. [Google Scholar] [CrossRef] [PubMed]
- Mongan, D.; Föcking, M.; Healy, C.; Susai, S.R.; Heurich, M.; Wynne, K.; Nelson, B.; McGorry, P.D.; Amminger, G.P.; Nordentoft, M.; et al. Development of Proteomic Prediction Models for Transition to Psychotic Disorder in the Clinical High-Risk State and Psychotic Experiences in Adolescence. JAMA Psychiatry 2021, 78, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, E.; Guest, P.C.; Steiner, J.; Bogerts, B.; Bahn, S. Identification of Blood-Based Molecular Signatures for Prediction of Response and Relapse in Schizophrenia Patients. Transl. Psychiatry 2012, 2, e82. [Google Scholar] [CrossRef]
- Llorca-Bofí, V.; Madero, S.; Amoretti, S.; Cuesta, M.J.; Moreno, C.; González-Pinto, A.; Bergé, D.; Rodriguez-Jimenez, R.; Roldán, A.; García-León, M.Á.; et al. Inflammatory Blood Cells and Ratios at Remission for Psychosis Relapse Prediction: A Three-Year Follow-up of a Cohort of First Episodes of Schizophrenia. Schizophr. Res. 2024, 267, 24–31. [Google Scholar] [CrossRef]
- Czysz, A.H.; South, C.; Gadad, B.S.; Arning, E.; Soyombo, A.; Bottiglieri, T.; Trivedi, M.H. Can Targeted Metabolomics Predict Depression Recovery? Results from the CO-MED Trial. Transl. Psychiatry 2019, 9, 11. [Google Scholar] [CrossRef]
- Gama Marques, J.; Ouakinin, S. Clinical Profile in Schizophrenia and Schizoaffective Spectrum: Relation with Unconjugated Bilirubin in a Prospective and Controlled Study with Psychopathological and Psychosocial Variables. CNS Spectr. 2020, 25, 782–789. [Google Scholar] [CrossRef]
- Elmorsy, E.; Shahda, M.; Mahmoud, E.H.M.; Rakha, S.A.; Shoaib, M. Blood Lactate Levels as a Biomarker of Antipsychotic Side Effects in Patients with Schizophrenia. J. Psychopharmacol. 2016, 30, 63–68. [Google Scholar] [CrossRef]
- Ising, M.; Horstmann, S.; Kloiber, S.; Lucae, S.; Binder, E.B.; Kern, N.; Künzel, H.E.; Pfennig, A.; Uhr, M.; Holsboer, F. Combined Dexamethasone/Corticotropin Releasing Hormone Test Predicts Treatment Response in Major Depression—A Potential Biomarker? Biol. Psychiatry 2007, 62, 47–54. [Google Scholar] [CrossRef]
- Landau, E.R.; Raniti, M.B.; Blake, M.; Waloszek, J.M.; Blake, L.; Simmons, J.G.; Schwartz, O.; Murray, G.; Trinder, J.; Allen, N.B.; et al. The Ratio of Morning Cortisol to CRP Prospectively Predicts First-Onset Depression in at-Risk Adolescents. Soc. Sci. Med. 2021, 281, 114098. [Google Scholar] [CrossRef]
- Coplan, J.D.; Wolk, S.I.; Goetz, R.R.; Ryan, N.D.; Dahl, R.E.; Mann, J.J.; Weissman, M.M. Nocturnal Growth Hormone Secretion Studies in Adolescents with or without Major Depression Re-Examined: Integration of Adult Clinical Follow-up Data. Biol. Psychiatry 2000, 47, 594–604. [Google Scholar] [CrossRef]
- Molendijk, M.L.; Bus, B.A.A.; Spinhoven, P.; Penninx, B.W.J.H.; Kenis, G.; Prickaerts, J.; Voshaar, R.C.O.; Elzinga, B.M. Serum Levels of Brain-Derived Neurotrophic Factor in Major Depressive Disorder: State-Trait Issues, Clinical Features and Pharmacological Treatment. Mol. Psychiatry 2011, 16, 1088–1095. [Google Scholar] [CrossRef] [PubMed]
- Tadić, A.; Wagner, S.; Gorbulev, S.; Dahmen, N.; Hiemke, C.; Braus, D.F.; Lieb, K. Peripheral Blood and Neuropsychological Markers for the Onset of Action of Antidepressant Drugs in Patients with Major Depressive Disorder. BMC Psychiatry 2011, 11, 16. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Belzeaux, R.; Fiori, L.M.; Lopez, J.P.; Boucekine, M.; Boyer, L.; Blier, P.; Farzan, F.; Frey, B.N.; Giacobbe, P.; Lam, R.W.; et al. Predicting Worsening Suicidal Ideation with Clinical Features and Peripheral Expression of Messenger RNA and MicroRNA During Antidepressant Treatment. J. Clin. Psychiatry 2019, 80, 18m12556. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.; Heilbronner, U.; Degenhardt, F.; Adli, M.; Akiyama, K.; Akula, N.; Ardau, R.; Arias, B.; Backlund, L.; Banzato, C.E.M.; et al. Genetic Variants Associated with Response to Lithium Treatment in Bipolar Disorder: A Genome-Wide Association Study. Lancet 2016, 387, 1085. [Google Scholar] [CrossRef]
- Yu, H.; Yan, H.; Wang, L.; Li, J.; Tan, L.; Deng, W.; Chen, Q.; Yang, G.; Zhang, F.; Lu, T.; et al. Five Novel Loci Associated with Antipsychotic Treatment Response in Patients with Schizophrenia: A Genome-Wide Association Study. Lancet Psychiatry 2018, 5, 327–338. [Google Scholar] [CrossRef]
- Sun, X.Y.; Zhang, J.; Niu, W.; Guo, W.; Song, H.T.; Li, H.Y.; Fan, H.M.; Zhao, L.; Zhong, A.F.; Dai, Y.H.; et al. A Preliminary Analysis of MicroRNA as Potential Clinical Biomarker for Schizophrenia. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2015, 168B, 170–178. [Google Scholar] [CrossRef]
- Lohoff, F.W.; Narasimhan, S.; Rickels, K. Interaction between Polymorphisms in Serotonin Transporter (SLC6A4) and Serotonin Receptor 2A (HTR2A) Genes Predict Treatment Response to Venlafaxine XR in Generalized Anxiety Disorder. Pharmacogenom. J. 2013, 13, 464–469. [Google Scholar] [CrossRef]
- Kung, P.H.; Davey, C.G.; Harrison, B.J.; Jamieson, A.J.; Felmingham, K.L.; Steward, T. Frontoamygdalar Effective Connectivity in Youth Depression and Treatment Response. Biol. Psychiatry 2023, 94, 959–968. [Google Scholar] [CrossRef]
- Farb, N.A.S.; Desormeau, P.; Anderson, A.K.; Segal, Z.V. Static and Treatment-Responsive Brain Biomarkers of Depression Relapse Vulnerability Following Prophylactic Psychotherapy: Evidence from a Randomized Control Trial. Neuroimage Clin. 2022, 34, 102969. [Google Scholar] [CrossRef]
- Sarpal, D.K.; Argyelan, M.; Robinson, D.G.; Szeszko, P.R.; Karlsgodt, K.H.; John, M.; Weissman, N.; Gallego, J.A.; Kane, J.M.; Lencz, T.; et al. Baseline Striatal Functional Connectivity as a Predictor of Response to Antipsychotic Drug Treatment. Am. J. Psychiatry 2016, 173, 69–77. [Google Scholar] [CrossRef]
- Viviano, J.D.; Buchanan, R.W.; Calarco, N.; Gold, J.M.; Foussias, G.; Bhagwat, N.; Stefanik, L.; Hawco, C.; DeRosse, P.; Argyelan, M.; et al. Resting-State Connectivity Biomarkers of Cognitive Performance and Social Function in Individuals with Schizophrenia Spectrum Disorder and Healthy Control Subjects. Biol. Psychiatry 2018, 84, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Korgaonkar, M.S.; Williams, L.M.; Ju Song, Y.; Usherwood, T.; Grieve, S.M. Diffusion Tensor Imaging Predictors of Treatment Outcomes in Major Depressive Disorder. Br. J. Psychiatry 2014, 205, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Gong, Q.; Wu, Q.; Scarpazza, C.; Lui, S.; Jia, Z.; Marquand, A.; Huang, X.; McGuire, P.; Mechelli, A. Prognostic Prediction of Therapeutic Response in Depression Using High-Field MR Imaging. Neuroimage 2011, 55, 1497–1503. [Google Scholar] [CrossRef] [PubMed]
- Cardoner, N.; Pujol, J.; Vallejo, J.; Urretavizcaya, M.; Deus, J.; López-Sala, A.; Benlloch, L.; Menchón, J.M. Enlargement of Brain Cerebrospinal Fluid Spaces as a Predictor of Poor Clinical Outcome in Melancholia. J. Clin. Psychiatry 2003, 64, 691–697. [Google Scholar] [CrossRef]
- Lei, D.; Qin, K.; Li, W.; Pinaya, W.H.L.; Tallman, M.J.; Patino, L.R.; Strawn, J.R.; Fleck, D.; Klein, C.C.; Lui, S.; et al. Brain Morphometric Features Predict Medication Response in Youth with Bipolar Disorder: A Prospective Randomized Clinical Trial. Psychol. Med. 2023, 53, 4083–4093. [Google Scholar] [CrossRef]
- Pérez, V.; Catafau, A.M.; Corripio, I.; Martín, J.C.; Alvarez, E. Preliminary Evidence of Striatal D2 Receptor Density as a Possible Biological Marker of Prognosis in Naive Schizophrenic Patients. Prog. Neuropsychopharmacol. Biol. Psychiatry 2003, 27, 767–770, Erratum in Prog. Neuropsychopharmacol. Biol. Psychiatry 2003, 30, 1118. https://doi.org/10.1016/S0278-5846(03)00106-4. [Google Scholar] [CrossRef]
- Cook, I.A.; Hunter, A.M.; Abrams, M.; Siegman, B.; Leuchter, A.F. Midline and Right Frontal Brain Function as a Physiologic Biomarker of Remission in Major Depression. Psychiatry Res. 2009, 174, 152–157. [Google Scholar] [CrossRef]
- Matsuoka, H.; Matsumoto, K.; Yamazaki, H.; Yoshida, S.; Numachi, Y.; Saito, H.; Ueno, T.; Sato, M. Delayed Visual NA Potential in Remitted Schizophrenia: A New Vulnerability Marker for Psychotic Relapse under Low-Dose Medication. Biol. Psychiatry 1999, 45, 107–115. [Google Scholar] [CrossRef]
- Huda, A.S. The Medical Model and Its Application in Mental Health. Int. Rev. Psychiatry 2021, 33, 463–470. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.