Abstract
Background/Objectives: Tumor necrosis factor alpha (TNF-α) is the key inflammatory cytokine involved in the pathogenesis of inflammatory bowel diseases (IBDs). Anti-TNF-α therapy has been successfully used for IBD treatment, although the therapeutic response differs among patients due to the genetic background. The aim of this study was to investigate whether the presence of single nucleotide polymorphisms (SNPs) on TNFA, TNFRSF1A, and TNFRSF1B genes could affect anti-TNF-α treatment effectiveness in IBD patients. Methods: In this prospective cohort study, 83 European IBD patients treated with infliximab or adalimumab (with or without steroid bridge therapy) as first-line therapy were enrolled. Genomic DNA was extracted from peripheral blood, and TNF-α (rs1800629, rs361525, rs1799724), TNFRSF1A (rs767455), and TNFRSF1B (rs1061622, rs1061624, rs3397, rs976881) SNPs were assessed. Steroid-free remission (SFR) (clinical remission together with steroid interruption) and anti-TNF-α therapy persistence after 12 months of follow-up were evaluated. Patients who stopped anti-TNF-α therapy before the end of follow-up, due to side effects or treatment failure, were defined as discontinuers. Results: A higher frequency of the G/G genotype in rs1800629 and the A/A genotype in rs1061624 was observed in the SFR group compared to non-SFR (97.7% vs. 82.8%; p = 0.025 and 32.6% vs. 10.3%; p = 0.029, respectively). Moreover, carriers of the A/A genotype in rs361525 and the C/C genotype in rs767455 had a lower probability of achieving SFR than wild-type patients (OR = 0.14; 95% CI= 0.03–0.69; p = 0.016 and OR = 0.10; 95% CI = 0.02–0.60; p = 0.012, respectively). Furthermore, an increased frequency of rs1800629 A allele was observed in patients who discontinued treatment compared to completers (27.3% vs. 6.9%; p = 0.033), as well as a high risk of interrupting therapy (HR = 6.47; 95% CI = 1.15–36.38). Conclusions: These results suggest that the evaluation of SNPs in TNF-α, TNFR1A, and TNFR1B genes could improve the management of IBD, leading to more effective, individualized treatment plans and a reduction in healthcare costs associated with ineffective therapies and disease complications.
1. Introduction
Inflammatory bowel diseases (IBDs), including Crohn’s disease (CD) and ulcerative colitis (UC), are chronic inflammatory conditions of the gastrointestinal tract, with 3.32 million estimated cases in 1990 and 4.90 million cases in 2019, corresponding to an increase of 47.45% between 1990 and 2019, with a prevalence of 84.3 (79.2–89.9) per 100,000 population [1,2,3,4]. Genetic and environmental factors may influence immune response and related diseases, including CD and UC [5].
In the last decade, the goal of pharmacological treatments has not been limited exclusively to symptom control but has also aimed at “deep remission”, which includes both clinical remission and mucosal healing, in order to prevent disease progression and disability. Anti-inflammatory drugs (aminosalicylates, mesalazine, sulfasalazine, and systemic corticosteroids), antibiotics (amoxicillin, ciprofloxacin, metronidazole, and azithromycin), and immunomodulators (azathioprine, mercaptopurine, and methotrexate) are the main drugs used for IBD treatment and may be prescribed alone or in combination as the mainstay of medical management [6]. Surgery may be necessary when drug therapy is not effective in controlling the disease or in the presence of complications, such as intestinal obstruction, fistulas, or severe bleeding. However, surgery is not curative, and post-operative endoscopic recurrence occurs in more than half of patients within 1 year, especially in CD [7]. In recent years, the use of biotechnological drugs for IBD treatment has increased significantly. The most commonly prescribed biological agents include anti-TNF-α antibodies (infliximab and adalimumab), anti-integrin (vedolizumab), IL-12 and IL-23 inhibitors (ustekinumab), and anti-IL-23 inhibitors (risankizumab and mirikizumab). These drugs have revolutionized the treatment of IBDs by offering effective therapeutic options for patients who do not respond to conventional treatments or who have severe disease activity. In particular, the most widely used biological drugs for the treatment of IBDs are infliximab (a chimeric murine IgG1 monoclonal antibody targeting TNF-α) and adalimumab (a fully humanized IgG1 monoclonal antibody against TNF-α) [8]. However, around 25% of patients experience treatment failure within 1 year of starting a first-line biologic for either UC or CD [9]. A further number of patients who initially respond to treatment eventually lose reactivity, a phenomenon called secondary non-response. Therefore, clinical response may vary from patient to patient, and early identification of treatment failure with anti-TNF-α agents is of significant importance from both a clinical and an economic point of view. Thus, robust data have demonstrated that early treatment of IBDs is critical to prevent complications and, in particular, real-world data have shown that biologic treatment within 2 years of diagnosis was associated with higher rates of remission and mucosal healing compared to late or non-biologic treatment [10].
Shorter disease duration, isolated colon disease, younger age, and non-smoking status represent the main factors associated with anti-TNF-α treatment success. Conversely, clinical failure may be due to an alternative non-TNF-α-mediated inflammatory pathway, the development of anti-drug antibodies, or the presence of genetic polymorphisms. Improvements in genetic characterization techniques and genome-wide association studies (GWAS) have allowed for the identification of genetic variants that could influence both disease development and response to treatment, as well as the occurrence of adverse events [11,12]. Several genes have been implicated in the pathogenesis of IBDs. However, limited evidence exists regarding the ability to predict response to anti-TNF-α treatment in CD and UC based on genetic data. Some studies have shown that single nucleotide polymorphisms (SNPs) in the genes encoding TNF-α or its receptors, TNFR1A and TNFR1B, can influence the response to anti-TNF-α drugs in patients with IBDs, although some results appear controversial [13,14,15,16]. Therefore, the aim of the present study was to investigate eight SNPs within the genes encoding TNF-α, TNFR1A, and TNFR1B to evaluate whether their presence could influence treatment effectiveness with infliximab or adalimumab in patients with CD and UC in order to allow for efficient, personalized, and cost-effective treatment.
2. Material and Methods
2.1. Patients Recruitment and Data Collection
A prospective cohort study was conducted by enrolling 83 patients from the IBD Unit of the A.O.U. “G. Martino” of Messina in the period between 1 May 2022 and 1 May 2024. Patients affected by UC and CD who started treatment with infliximab or adalimumab were selected and monitored for 12 months after the start of biological therapy. The clinicians involved in the study provided a complete and exhaustive explanation of the protocol at the time of enrollment and collected written informed consent. The study was approved by the Ethics Committee of Messina with the identification code n° 41/22 of 12 April 2022.
The following sociodemographic and clinical characteristics were recorded: gender, smoking, IBD family history, date of diagnosis and start date of biological therapy, type of biological therapy (infliximab or adalimumab), any cause of treatment interruption, concomitant therapies, and comorbidities. Patients were managed according to clinical practice every two months (with close time intervals during the induction of therapy, as per the drug label, or in the case of treatment optimization). At the 12-month follow-up, clinical remission together with the interruption of steroidal bridge therapy were evaluated.
Clinical activity was evaluated by using clinical scores, such as the Harvey Bradshaw Index (HBI) for CD and the partial Mayo Score (pMS) for UC. In particular, steroid-free remission (SFR) was defined as clinical disease activity with a HBI ≤ 5 points for CD and a pMS ≤ 2 without any kind of concomitant steroids.
Patients with SFR were included in the “SFR group”, and all others were included in the “non SFR group”. Patients who discontinued anti-TNF-α treatment due to side effects or treatment failure before the end of follow-up were defined as “discontinuers”.
2.2. SNPs Selection, DNA Extraction, and Genotyping
A PubMed literature search was conducted by using the keywords “tumor necrosis factoralpha”, “anti-TNF-α”, “infliximab”, “adalimumab”, “polymorphism”, “SNP”, “ulcerative colitis”, and “Crohn’s disease” using Boolean operators (AND), (OR), (NOT). Results were restricted to original studies that investigated SNPs and included allele frequencies and genotypes for different groups. SNPs that had biological relevance, those with an expected minor allele frequency ≥ 5%, and those that demonstrated an association with CD or UC and anti-TNF-α treatment were selected. SNPs were excluded if they had been extensively investigated and if there was no prognostic value for the combination of CD or UC and anti-TNF-α treatment response. As a result of this search, the following eight SNPs within 3 genes of TNF-α (rs1800629, rs361525, rs1799724), TNFRSF1A (rs767455), and TNFRSF1B (rs1061622, rs1061624, rs3397, rs976881) were analyzed. Peripheral blood was collected through venipuncture in 3 mL tubes containing EDTA as the anticoagulant. Genomic DNA was extracted from blood samples by using a standard protocol. In summary, cells were lysed in a DNA buffer containing 10 mM Tris-HCl (pH 7.4), 100 mM NaCl, 1 mM EDTA, and 1 mM MgCl2. Following centrifugation, the nuclei were lysed overnight at 37 °C with 10% SDS and proteinase K. Organic solvents, phenol, and chloroform were used to remove cell debris, primarily proteins. DNA was then washed with 70% ethanol and resuspended in an appropriate volume of RNase/DNase-free water. Its concentration was measured spectrophotometrically for each sample and stored at –20 °C until use for genotyping assay [17]. SNP assessment was performed by using TaqMan predesigned genotyping assays (Thermo Fisher Scientific, Waltham, MA, USA). The TaqMan genotyping assays were diluted to a 20× working stock solution with 1× TE buffer (10 mM Tris-HCl, 1 mM EDTA, pH 8.0, in DNase-free, sterile-filtered water), as recommended by the manufacturer. A MicroAmpTM Optical 96-Well Reaction Plate was used, and a total of 25 μL of reaction volume was used (12.50 μL of 2X TaqMan Master Mix, 1.25 μL of 20X assay working stock solution, 1 μL of DNA sample at a concentration of 10 ng/μL, 10.25 of nuclease-free water) for each well; the plate was then sealed and briefly centrifuged to bring the reaction mix to the bottom of the well and remove air bubbles. PCR reactions were performed with the QuantStudio6 Flex system (Applied Biosystems, Foster City, CA, USA), and the following program was used: 95 °C for 10 min, followed by 40 cycles of 95 °C for 15 s and then 60 °C for 1 min. The analysis was performed by using real-time instrument software (Applied Biosystems, Foster City, CA, USA), and each genotype was independently assigned by two investigators. In cases of disagreement, assignment was reached through consensus. Patient carriers of a homozygous major allele for each SNP were considered wild-type (WT).
2.3. Sample Size Calculation
A sample size calculation was performed by assuming an expected incidence of non-response equal to 25% after 12 months of anti-TNF-α treatment in IBD patients based on literature data [9] and an expected incidence of 40% for our study group, a probability error = 0.05 and a power level of 80%. Seventy-one patients represented the minimum sample size that guaranteed a powered study. However, 83 patients were enrolled considering possible patient drop-outs.
2.4. Statistical Analysis
A descriptive analysis of the demographic and clinical characteristics of the enrolled patients was performed. Characteristics of the “SFR group” were compared with the “non-SFR group”, and the characteristics of patients who discontinued treatment were compared with patients who completed the follow-up. The results were expressed as the median and the interquartile range (Q1–Q3) for continuous variables and as the absolute frequency and the percentage for categorical variables.
The variables studied did not have a normal distribution (as demonstrated by the Kolmogorov–Smirnov test); therefore, a non-parametric approach was used. Specifically, the Mann–Whitney test was used to compare continuous variables between independent groups, while the Pearson chi-square test was applied for categorical variables.
Univariate logistic regression models were used to identify predictive factors associated with clinical remission of the diseases. All variables identified as predictors were included in a stepwise multivariate logistic regression model (backward procedure, α = 5%), and odds ratios (OR) with their 95% confidence intervals (95% CI) were calculated.
Multivariate Cox regression models were used to assess the risk of biologic treatment discontinuation during the monitoring period, including covariate age, gender, biologic treatment (adalimumab or infliximab), diseases (UC or CD), steroid therapy at baseline, and genotypes. Results were reported as hazard ratios (HRs) with 95% CI.
Values of p < 0.05 were considered statistically significant, and all analyses were performed with SPSS version 23.0 software (IBM Corp., SPSS Statistics, Armonk, NY, USA).
3. Results
3.1. Patients Characteristics
A total of 83 patients were enrolled with a median age of 34 years (27–52 years), and 63.9% (N = 53) were male. In particular, 45 patients (54.4%) were affected by CD, with a median baseline HBI value of 3.0 (1.0–5.5), whereas 38 patients (45.8%) had UC, with a median PMS value of 2.5 (1.0–5.0). Furthermore, 51 patients (61.4%) were treated with infliximab, 32 patients (38.6%) were treated with adalimumab, and 49 (68.1%) also used steroid bridge therapy.
In total, 11 (13.3%) patients discontinued treatment with biologics early during the study period, and 72 patients (86.7%) continued the treatment for at least 12 months.
Among patients who completed the follow-up, 43 (59.7%) were in the SFR group, and 29 (40.3%) were in the non-SFR group. No significant difference was observed between the two groups considering demographic and clinical characteristics (Table 1).

Table 1.
Patients characteristics stratified by response to treatment (SFR vs. non-SFR).
All 83 patients included in the study were genotyped to assess whether the allele frequencies of SNPs TNF-α (rs1800629, rs361525, rs1799724), TNFRSF1A (rs767455), and TNFRSF1B (rs1061624, rs3397, rs976881) were consistent with those normally expected in a European population. Our results showed that the allele frequencies of rs361525 were not in agreement with the Hardy–Weinberg equilibrium, although the allele frequencies of all other SNPs were (Table 2).

Table 2.
Genotype frequency and allele frequency in enrolled patients. 1: Major allele. 2: Minor allele (both in European populations). HWE: Hardy–Weinberg equilibrium.
3.2. SNPs Associated with Clinical Effectiveness
A higher frequency of the G/G genotype in rs1800629 was observed in the SFR group than in the non-SFR group (97.7% vs. 82.8%; p = 0.025). Furthermore, a higher frequency of the occurrence of the A/A genotype in rs1061624 was detected in the SFR group than in the non-SFR group (32.6% vs. 10.3%; p = 0.029 (Table 3).

Table 3.
SNPs genotypes stratified by treatment effectiveness (SFR vs. non-SFR).
The probability of achieving SFR was significantly reduced in patients with a homozygous mutated genotype in rs361525 (OR = 0.14; 95% CI = 0.03–0.69; p = 0.016) and in rs767455 (OR = 0.10; 95% CI = 0.02–0.60; p = 0.012) compared to WT patients for the same SNPs (Table 4).

Table 4.
Identification of predictive factors of effectiveness of anti-TNF-α biological drugs.
3.3. SNPs Associated with Anti-TNF-α Treatment Discontinuation
No significant difference was observed between patients who discontinued treatment and patients who completed the follow-up considering the clinical and demographic characteristics. However, the heterozygous genotype in rs1800629 was more frequent in the group of patients who interrupted the treatment compared to patients who did not discontinue (27.3% vs. 6.9%; p = 0.033; Table 5).

Table 5.
SNPs genotypes stratified on the basis of treatment discontinuation (discontinuers vs. completers).
Cox regression analysis reported a significant increased risk of discontinuing biologics in patient carriers of a minor allele in rs1800629 compared to patients with the WT genotype (HR = 5.98; 95% CI = 1.03–34.83; p = 0.047) (Figure 1 and Table 6).
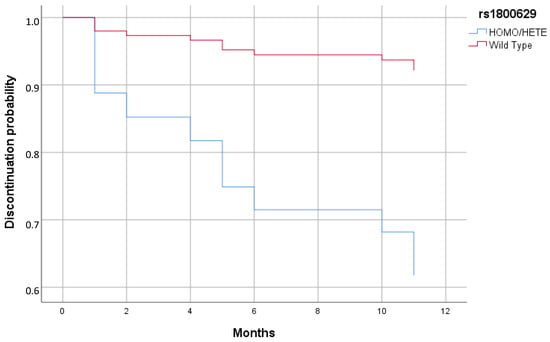
Figure 1.
Cox regression of discontinuation of therapy stratified by rs1800629 minor allele (Homo/Hete) and WT.

Table 6.
Summary of key findings.
4. Discussion
Nowadays, maximizing the therapeutic efficacy of anti-TNF-α biologic drug therapy and minimizing the risk of serious side effects by selecting patients who are likely to have a favorable outcome are of substantial clinical interest. Young age and concomitant immunosuppressive treatment have previously been associated with a short-term beneficial response to infliximab [18], while the development of anti-drug antibodies has been associated with a negative therapeutic response and an increased risk of severe infusion reactions [19,20]. Despite these findings, it has not yet been clarified whether the effectiveness and safety of biologic drugs in IBDs could be effectively predicted before the beginning of therapy. Genetic variation may characterize subgroups of patients with disparate efficacy and safety profiles due to distinct gene expression profiles [21].
To our knowledge, this is the first study that evaluates gene expression profiles in relation to a significant outcome, such as SFR. Previous studies have investigated only clinical response or clinical remission. In this study, eight SNPs in the TNF-α, TNFRSF1A, and TNFRSF1B genes were evaluated, and patients were stratified according to SFR achievement. Interestingly, carriers of the A/A genotype in rs1061624 appeared with a significantly higher frequency in the SFR group compared to the non-SFR group, and the G/G genotype in rs1800629 appeared with a significantly higher frequency in SFR than in the non-SFR group. The results regarding rs1061624 are not consistent with previous studies that demonstrated a correlation between the presence of a G variant in rs1061624 and a long-term response to infliximab [22], as well as an association between the A/T haplotype in rs1061624 and rs3397 and non-response to treatment in 297 Spanish CD patients undergoing infliximab treatment [23]. On the other hand, in accordance with the results of the present study, a previous paper has shown that the presence of a mutated genotype in rs1800629 was correlated with a three-fold increased risk of being a non-responder in a study conducted on 121 Caucasian patients with CD undergoing anti-TNF-α treatment [24]. An increased frequency of the rs1800629 A allele was also observed in non-responders to TNF-α inhibitors compared to responders in a study on 82 Spanish IBD patients [25], thus confirming the results obtained in our study showing that the WT genotype in this SNP appeared with a significantly higher frequency in the SFR group. On the other hand, several studies did not find an association between the presence of rs1800629 and clinical response either in adults or children [26,27,28,29,30], confirming that further validation is needed.
Furthermore, it has been demonstrated that the minor allele of the TNFRSF1A gene in rs767455 is associated with CD in Caucasians [31]. In our study, we aimed at investigating this polymorphism in relation to the effectiveness of anti-TNF-α biologic treatment in IBD patients evaluated with a strong outcome, such as SFR, thus observing that the presence of a homozygous mutated genotype is a predictive factor for non-achievement of SFR. This result was in accordance with previous findings obtained by Matsukura and colleagues in a Japanese population [32] and with the data reported by Pierik et al. that demonstrated an association with non-response evaluated based on serum C Reactive Protein (CRP) levels [33]. Instead, a study conducted on 444 patients with CD found no association between the presence of rs767455 and the clinical response to infliximab [34], just as no association was found in a study on 297 Spanish CD patients who underwent infliximab treatment [23].
SNPs in the gene encoding TNF-α have previously been associated with clinical response to anti-TNF-α biologic drugs. Specifically, a polymorphism at position −238 in the promoter region (rs361525) has previously shown conflicting data regarding the response to anti-TNF-α treatment in rheumatoid arthritis, psoriasis, and IBDs [25,35,36]. In our study, a homozygous genotype in rs361525 was correlated with a greater likelihood of being in the non-SFR group after infliximab or adalimumab treatment for 12 months. These results agree with a previous paper that linked this mutation to poor response to anti-TNF-α treatment in Danish patients with IBDs [37]. On the other hand, other previous studies did not find an association between the presence of rs361525 and clinical or biological response to infliximab treatment in CD or UC patients [25,27,30,38].
rs1799724 was also studied in our cohort, and no association was detected with the clinical outcomes, in accordance with the studies conducted by Duricova et al. and Papamichael et al. in adults and children affected by CD [28]. On the contrary, Matsuoka and colleagues demonstrated that the presence of this SNP could be considered a predicting factor for non-remission during infliximab maintenance therapy, at least in Japanese patients [30].
Moreover, in the present study, no correlation was observed with the presence of rs1061622, rs3397, or rs976881 in the TNFRSF1B gene with respect to anti-TNF-α treatment effectiveness, as previously demonstrated in other studies involving CD patients [30,32]. On the other hand, both Medrano et al. and Steenholdt et al. found an association between an increased frequency of the rs1061622 G carrier and disease remission in Caucasian CD patients [20,23], whereas Salvador-Martín et al. showed that rs3397 C allele presence was predictive of a longer time to failure in anti-TNF-α therapy [39]. Additionally, the research group of Steenholdt indicated that the minor allele carriage of rs976881 is associated with a loss of response to infliximab therapy [20].
We also investigated the possible association between the presence of SNPs and discontinuation of anti-TNF-α treatment. A significantly higher risk of treatment discontinuation was observed among heterozygosis carriers of the rs1800629 minor allele, but no result was reported in this study for homozygosis due to the low number of patients enrolled (only one patient in homozygosis). This result is partially at odds with the only other study in the literature regarding the effect of this mutation on treatment discontinuation conducted on psoriasis patients, which showed no significant difference in treatment discontinuation between WT and mutated patients [40].
The identification of SNPs in TNF-α, TNFR1A, and TNFR1B genes as potential predictors of response to infliximab or adalimumab in CD and UC has several important clinical implications. These findings could enhance treatment decision making in real-world settings by contributing to a more personalized approach to anti-TNF-α therapy by optimizing drug selection and treatment initiation. Thus, physicians integrating SNP profiling into routine clinical practice could predict whether a patient is likely to respond to infliximab or adalimumab before initiating treatment. This would allow for the selection of the most effective biologic therapy from the outset, reducing errors in prescribing and minimizing delays in achieving disease control. Moreover, unnecessary exposure to ineffective therapies could be avoided; thus, patients with genetic markers associated with poor response to anti-TNF-α agents could be redirected toward alternative biologic therapies, such as anti-integrins (vedolizumab), anti-interleukins (ustekinumab, risankizumab), or JAK inhibitors (tofacitinib, filgotinib, upadacitinib), earlier in their treatment journey. This would help in preventing prolonged disease activity, reducing adverse effects from ineffective medications, and decreasing the risk of complications. Furthermore, considering that biologic therapies are expensive and non-responders may require multiple treatment switches, leading to increased healthcare costs, the use of genetic tests for relevant SNPs could provide a cost-effective strategy by ensuring that only those likely to benefit receive anti-TNF-α agents, thus optimizing resource utilization.
Nevertheless, the present study presents some limitations. For instance, the relatively small cohort size and the limited ethnic diversity (only Caucasian patients) restrict the generalizability of results. Moreover, we did not evaluate potential confounders (e.g., concomitant medications) that could influence treatment response, and endoscopy was not performed to assess endoscopic activity or mucosal healing at the end of follow-up.
Ultimately, the evaluation of SNPs in TNF-α, TNFR1A, and TNFR1B genes could improve the management of IBD by enabling a precision medicine approach. This would lead to more effective, individualized treatment plans, improved patient outcomes, and a reduction in healthcare costs associated with ineffective therapies and disease complications. However, further validation in larger, multi-center cohorts with diverse patient populations is necessary before widespread clinical implementation.
Author Contributions
Conceptualization, A.V. and G.P.; data curation, C.B. and L.P.; formal analysis, M.R.; investigation, I.P., D.A.G., F.M. and R.C.; supervision, G.P.; validation, V.A., G.C. and S.P.; writing—original draft, M.R., I.P. and G.P.; writing—review and editing, N.I., W.F., A.V. and G.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of Messina with the protocol code n° 41/22 on 12 April 2022.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors upon request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Ungaro, R.; Mehandru, S.; Allen, P.B.; Peyrin-Biroulet, L.; Colombel, J.-F. Ulcerative Colitis. Lancet 2017, 389, 1756–1770. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.; Mehandru, S.; Colombel, J.-F.; Peyrin-Biroulet, L. Crohn’s Disease. Lancet 2017, 389, 1741–1755. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Li, Z.; Liu, S.; Zhang, D. Global, Regional and National Burden of Inflammatory Bowel Disease in 204 Countries and Territories from 1990 to 2019: A Systematic Analysis Based on the Global Burden of Disease Study 2019. BMJ Open 2023, 13, e065186. [Google Scholar] [CrossRef] [PubMed]
- Alatab, S.; Sepanlou, S.G.; Ikuta, K.; Vahedi, H.; Bisignano, C.; Safiri, S.; Sadeghi, A.; Nixon, M.R.; Abdoli, A.; Abolhassani, H.; et al. The Global, Regional, and National Burden of Inflammatory Bowel Disease in 195 Countries and Territories, 1990–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2020, 5, 17–30. [Google Scholar] [CrossRef]
- Agrawal, M.; Jess, T. Implications of the Changing Epidemiology of Inflammatory Bowel Disease in a Changing World. United Eur. Gastroenterol. J. 2022, 10, 1113–1120. [Google Scholar] [CrossRef]
- Pallio, G.; Bitto, A.; Pizzino, G.; Galfo, F.; Irrera, N.; Squadrito, F.; Squadrito, G.; Pallio, S.; Anastasi, G.P.; Cutroneo, G.; et al. Adenosine Receptor Stimulation by Polydeoxyribonucleotide Improves Tissue Repair and Symptomology in Experimental Colitis. Front. Pharmacol. 2016, 7, 273. [Google Scholar] [CrossRef]
- Kozuch, P.L.; Hanauer, S.B. Treatment of Inflammatory Bowel Disease: A Review of Medical Therapy. World J. Gastroenterol. 2008, 14, 354. [Google Scholar] [CrossRef]
- Rutgeerts, P.; Geboes, K.; Vantrappen, G.; Beyls, J.; Kerremans, R.; Hiele, M. Predictability of the Postoperative Course of Crohn’s Disease. Gastroenterology 1990, 99, 956–963. [Google Scholar] [CrossRef]
- Kapizioni, C.; Desoki, R.; Lam, D.; Balendran, K.; Al-Sulais, E.; Subramanian, S.; Rimmer, J.E.; De La Revilla Negro, J.; Pavey, H.; Pele, L.; et al. Biologic Therapy for Inflammatory Bowel Disease: Real-World Comparative Effectiveness and Impact of Drug Sequencing in 13 222 Patients within the UK IBD BioResource. J. Crohn’s Colitis 2024, 18, 790–800. [Google Scholar] [CrossRef]
- Ungaro, R.C.; Aggarwal, S.; Topaloglu, O.; Lee, W.; Clark, R.; Colombel, J. Systematic Review and Meta-analysis: Efficacy and Safety of Early Biologic Treatment in Adult and Paediatric Patients with Crohn’s Disease. Aliment. Pharmacol. Ther. 2020, 51, 831–842. [Google Scholar] [CrossRef]
- Witte, J.S. Rare Genetic Variants and Treatment Response: Sample Size and Analysis Issues. Stat. Med. 2012, 31, 3041–3050. [Google Scholar] [CrossRef]
- McInnes, G.; Yee, S.W.; Pershad, Y.; Altman, R.B. Genomewide Association Studies in Pharmacogenomics. Clin. Pharmacol. Ther. 2021, 110, 637–648. [Google Scholar] [CrossRef] [PubMed]
- Qasem, A.; Ramesh, S.; Naser, S.A. Genetic Polymorphisms in Tumour Necrosis Factor Receptors (TNFRSF1A/1B) Illustrate Differential Treatment Response to TNFα Inhibitors in Patients with Crohn’s Disease. BMJ Open Gastroenterol. 2019, 6, e000246. [Google Scholar] [CrossRef]
- Prieto-Pérez, R.; Almoguera, B.; Cabaleiro, T.; Hakonarson, H.; Abad-Santos, F. Association between Genetic Polymorphisms and Response to Anti-TNFs in Patients with Inflammatory Bowel Disease. Int. J. Mol. Sci. 2016, 17, 225. [Google Scholar] [CrossRef]
- López-Serrano, P.; Pérez-Calle, J.L.; Pérez-Fernández, M.T.; Fernández-Font, J.M.; Boixeda de Miguel, D.; Fernández-Rodríguez, C.M. Environmental Risk Factors in Inflammatory Bowel Diseases. Investigating the Hygiene Hypothesis: A Spanish Case–Control Study. Scand. J. Gastroenterol. 2010, 45, 1464–1471. [Google Scholar] [CrossRef] [PubMed]
- Muro, M. Immunogenetic Biomarkers in Inflammatory Bowel Diseases: Role of the IBD3 Region. World J. Gastroenterol. 2014, 20, 15037. [Google Scholar] [CrossRef] [PubMed]
- Pallio, G.; Irrera, N.; Bitto, A.; Mannino, F.; Minutoli, L.; Rottura, M.; Pallio, S.; Altavilla, D.; Alibrandi, A.; Marciano, M.C.; et al. Failure of Achieving Tacrolimus Target Blood Concentration Might Be Avoided by a Wide Genotyping of Transplanted Patients: Evidence from a Retrospective Study. J. Pers. Med. 2020, 10, 47. [Google Scholar] [CrossRef]
- Vermeire, S.; Louis, E.; Carbonez, A.; Assche, G.; Noman, M.; Belaiche, J.; Vos, M.; Gossum, A.; Pescatore, P.; Fiasse, R.; et al. Demographic and Clinical Parameters Influencing the Short-Term Outcome of Anti-Tumor Necrosis Factor (Infliximab) Treatment in Crohn’s Disease. Am. J. Gastroenterol. 2002, 97, 2357–2363. [Google Scholar] [CrossRef]
- Steenholdt, C.; Svenson, M.; Bendtzen, K.; Thomsen, O.Ø.; Brynskov, J.; Ainsworth, M.A. Severe Infusion Reactions to Infliximab: Aetiology, Immunogenicity and Risk Factors in Patients with Inflammatory Bowel Disease. Aliment. Pharmacol. Ther. 2011, 34, 51–58. [Google Scholar] [CrossRef]
- Steenholdt, C.; Bendtzen, K.; Brynskov, J.; Thomsen, O.Ø.; Ainsworth, M.A. Cut-off Levels and Diagnostic Accuracy of Infliximab Trough Levels and Anti-Infliximab Antibodies in Crohn’s Disease. Scand. J. Gastroenterol. 2011, 46, 310–318. [Google Scholar] [CrossRef]
- Arijs, I.; Quintens, R.; Van Lommel, L.; Van Steen, K.; De Hertogh, G.; Lemaire, K.; Schraenen, A.; Perrier, C.; Van Assche, G.; Vermeire, S.; et al. Predictive Value of Epithelial Gene Expression Profiles for Response to Infliximab in Crohn’s Disease. Inflamm. Bowel. Dis. 2010, 16, 2090–2098. [Google Scholar] [CrossRef] [PubMed]
- Salvador-Martín, S.; López-Cauce, B.; Nuñez, O.; Laserna-Mendieta, E.J.; García, M.I.; Lobato, E.; Abarca-Zabalía, J.; Sanjurjo-Saez, M.; Lucendo, A.J.; Marín-Jiménez, I.; et al. Genetic Predictors of Long-Term Response and Trough Levels of Infliximab in Crohn’s Disease. Pharmacol. Res. 2019, 149, 104478. [Google Scholar] [CrossRef] [PubMed]
- Medrano, L.M.; Taxonera, C.; Márquez, A.; Barreiro-de Acosta, M.; Gómez-García, M.; González-Artacho, C.; Pérez-Calle, J.L.; Bermejo, F.; Lopez-Sanromán, A.; Martín Arranz, M.D.; et al. Role of TNFRSF1B Polymorphisms in the Response of Crohn’s Disease Patients to Infliximab. Hum. Immunol. 2014, 75, 71–75. [Google Scholar] [CrossRef]
- Netz, U.; Carter, J.V.; Eichenberger, M.R.; Dryden, G.W.; Pan, J.; Rai, S.N.; Galandiuk, S. Genetic Polymorphisms Predict Response to Anti-Tumor Necrosis Factor Treatment in Crohn’s Disease. World J. Gastroenterol. 2017, 23, 4958. [Google Scholar] [CrossRef]
- López-Hernández, R.; Valdés, M.; Campillo, J.A.; Martínez-Garcia, P.; Salama, H.; Salgado, G.; Boix, F.; Moya-Quiles, M.R.; Minguela, A.; Sánchez-Torres, A.; et al. Genetic Polymorphisms of Tumour Necrosis Factor Alpha (TNF-α) Promoter Gene and Response to TNF-α Inhibitors in Spanish Patients with Inflammatory Bowel Disease. Int. J. Immunogenet. 2014, 41, 63–68. [Google Scholar] [CrossRef]
- Curci, D.; Lucafò, M.; Cifù, A.; Fabris, M.; Bramuzzo, M.; Martelossi, S.; Franca, R.; Decorti, G.; Stocco, G. Pharmacogenetic Variants of Infliximab Response in Young Patients with Inflammatory Bowel Disease. Clin. Transl. Sci. 2021, 14, 2184–2192. [Google Scholar] [CrossRef] [PubMed]
- Papamichael, K.; Gazouli, M.; Karakoidas, C.; Panayotou, I.; Roma-Giannikou, E.; Mantzaris, G.J. Association of TNF and FcγRΙΙΙA Gene Polymorphisms with Differential Response to Infliximab in a Greek Cohort of Crohn’s Disease Patients. Ann. Gastroenterol. 2011, 24, 35–40. [Google Scholar]
- Duricova, D.; Pedersen, N.; Lenicek, M.; Hradsky, O.; Bronsky, J.; Adamcova, M.; Elkjaer, M.; Andersen, P.S.; Vitek, L.; Larsen, K.; et al. Infliximab Dependency in Children with Crohn’s Disease. Aliment. Pharmacol. Ther. 2009, 29, 792–799. [Google Scholar] [CrossRef]
- Louis, E.; Vermeire, S.; Rutgeerts, P.; De Vos, M.; Van Gossum, A.; Pescatore, P.; Fiasse, R.; Pelckmans, P.; Reynaert, H.; D’Haens, G.; et al. A Positive Response to Infliximab in Crohn Disease: Association with a Higher Systemic Inflammation before Treatment but Not with -308 TNF Gene Polymorphism. Scand. J. Gastroenterol. 2002, 37, 818–824. [Google Scholar] [CrossRef]
- Matsuoka, K.; Hamada, S.; Shimizu, M.; Nanki, K.; Mizuno, S.; Kiyohara, H.; Arai, M.; Sugimoto, S.; Iwao, Y.; Ogata, H.; et al. Factors Predicting the Therapeutic Response to Infliximab during Maintenance Therapy in Japanese Patients with Crohn’s Disease. PLoS ONE 2018, 13, e0204632. [Google Scholar] [CrossRef]
- Larabi, A.; Barnich, N.; Nguyen, H.T.T. New Insights into the Interplay between Autophagy, Gut Microbiota and Inflammatory Responses in IBD. Autophagy 2020, 16, 38–51. [Google Scholar] [CrossRef] [PubMed]
- Matsukura, H.; Ikeda, S.; Yoshimura, N.; Takazoe, M.; Muramatsu, M. Genetic Polymorphisms of Tumour Necrosis Factor Receptor Superfamily 1A and 1B Affect Responses to Infliximab in Japanese Patients with Crohn’s Disease. Aliment. Pharmacol. Ther. 2008, 27, 765–770. [Google Scholar] [CrossRef]
- Pierik, M.; Vermeire, S.; Steen, K.V.; Joossens, S.; Claessens, G.; Vlietinck, R.; Rutgeerts, P. Tumour Necrosis Factor-α Receptor 1 and 2 Polymorphisms in Inflammatory Bowel Disease and Their Association with Response to Infliximab. Aliment. Pharmacol. Ther. 2004, 20, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Mascheretti, S.; Hampe, J.; Kühbacher, T.; Herfarth, H.; Krawczak, M.; Fölsch, U.R.; Schreiber, S. Pharmacogenetic Investigation of the TNF/TNF-Receptor System in Patients with Chronic Active Crohn’s Disease Treated with Infliximab. Pharmacogenom. J. 2002, 2, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, J.R.; Potter, C.; Hyrich, K.L.; Barton, A.; Worthington, J.; Isaacs, J.D.; Morgan, A.W.; Wilson, A.G. Association of the Tumour Necrosis Factor-308 Variant with Differential Response to Anti-TNF Agents in the Treatment of Rheumatoid Arthritis. Hum. Mol. Genet. 2008, 17, 3532–3538. [Google Scholar] [CrossRef]
- Lee, Y.H.; Ji, J.D.; Bae, S.; Song, G.G. Associations Between Tumor Necrosis Factor-α (TNF-α) −308 and −238 G/A Polymorphisms and Shared Epitope Status and Responsiveness to TNF-α Blockers in Rheumatoid Arthritis: A Metaanalysis Update. J. Rheumatol. 2010, 37, 740–746. [Google Scholar] [CrossRef]
- Bank, S.; Andersen, P.S.; Burisch, J.; Pedersen, N.; Roug, S.; Galsgaard, J.; Turino, S.Y.; Brodersen, J.B.; Rashid, S.; Rasmussen, B.K.; et al. Associations between Functional Polymorphisms in the NFκB Signaling Pathway and Response to Anti-TNF Treatment in Danish Patients with Inflammatory Bowel Disease. Pharmacogenom. J. 2014, 14, 526–534. [Google Scholar] [CrossRef]
- Dideberg, V.; Théâtre, E.; Farnir, F.; Vermeire, S.; Rutgeerts, P.; De Vos, M.; Belaiche, J.; Franchimont, D.; Van Gossum, A.; Louis, E.; et al. The TNF/ADAM 17 System: Implication of an ADAM 17 Haplotype in the Clinical Response to Infliximab in Crohn’s Disease. Pharmacogenet. Genom. 2006, 16, 727–734. [Google Scholar] [CrossRef]
- Salvador-Martín, S.; Bossacoma, F.; Pujol-Muncunill, G.; Navas-López, V.M.; Gallego-Fernández, C.; Viada, J.; Muñoz-Codoceo, R.; Magallares, L.; Martínez-Ojinaga, E.; Moreno-Álvarez, A.; et al. Genetic Predictors of Long-term Response to Antitumor Necrosis Factor Agents in Pediatric Inflammatory Bowel Disease. J. Pediatr. Gastroenterol. Nutr. 2020, 71, 508–515. [Google Scholar] [CrossRef]
- Membrive-Jiménez, C.; Pérez-Ramírez, C.; Arias-Santiago, S.; Richetta, A.G.; Ottini, L.; Pineda-Lancheros, L.E.; Ramírez-Tortosa, M.d.C.; Jiménez-Morales, A. Impact of Functional Polymorphisms on Drug Survival of Biological Therapies in Patients with Moderate-to-Severe Psoriasis. Int. J. Mol. Sci. 2023, 24, 8703. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).