Obstructive Sleep Apnoea and Lipid Metabolism: The Summary of Evidence and Future Perspectives in the Pathophysiology of OSA-Associated Dyslipidaemia
Abstract
1. Introduction
2. Overview of Physiological Lipid Metabolism
2.1. The Physiological Role of Chylomicrons
2.2. The Physiological Role of VLDL
2.3. The LDL Metabolism
2.4. The Role of Lipoprotein Lipase
2.5. The Physiological Role of HDL
3. Current Knowledge of The Pathophysiological Lipid Metabolism in OSA
3.1. Animal Models
3.2. Calorie Intake in OSA
3.3. Intestinal Lipid Absorption in OSA
3.4. Impaired Intravascular Lipolysis and Uptake by the Periphery: Lpl Dysfunction in OSA
3.5. Alternative Ways Leading to Decreased Lipid Uptake
3.6. Increased Lipid Production in the Liver
3.7. Abnormal Modifications of LDL in OSA
3.8. HDL Dysfunction in OSA
3.9. Increased Intracellular Lipolysis in Adipose Tissue
4. Further Mechanisms in OSA-Associated Dyslipidaemia
4.1. Adipose Tissue Dysfunction
4.1.1. Inflammation in Adipose Tissue
4.1.2. Role of Adipokines
4.2. Altered Hormone Production
4.3. Sleep Stages
5. Direct Consequences of Dyslipidaemia
5.1. Endothelial Dysfunction
5.2. Systemic Inflammation and Consequential Atherosclerosis
5.3. Insulin Resistance
6. The Effect of OSA Therapy on the Lipid Metabolism
6.1. The Effect of CPAP Therapy
6.2. The Effect of MAD Therapy
6.3. The Effect of Upper Airway Surgery
7. Discussion of Major Findings and Further Research Directions
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gileles-Hillel, A.; Kheirandish-Gozal, L.; Gozal, D. Biological plausibility linking sleep apnoea and metabolic dysfunction. Nat. Rev. Endocrinol. 2016, 12, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk: The Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS). Eur. Heart J. 2019, 41, 111–188. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Savransky, V.; Nanayakkara, A.; Smith, P.L.; O’Donnell, C.P.; Polotsky, V.Y. Hyperlipidemia and lipid peroxidation are dependent on the severity of chronic intermittent hypoxia. J. Appl. Physiol. 2007, 102, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Gündüz, C.; Basoglu, O.K.; Hedner, J.; Zou, D.; Bonsignore, M.R.; Hein, H.; Staats, R.; Pataka, A.; Barbe, F.; Sliwinski, P.; et al. Obstructive sleep apnoea independently predicts lipid levels: Data from the European Sleep Apnea Database. Respirology 2018, 23, 1180–1189. [Google Scholar] [CrossRef]
- Pan, X.; Hussain, M.M. Gut triglyceride production. Biochim. Biophys. Acta. 2012, 1821, 727–735. [Google Scholar] [CrossRef]
- Ramasamy, I. Recent advances in physiological lipoprotein metabolism. Clin. Chem. Lab. Med. 2014, 52, 1695–1727. [Google Scholar] [CrossRef] [PubMed]
- Zilversmit, D.B. Formation and transport of chylomicrons. Fed. Proc. 1967, 26, 1599–1605. [Google Scholar]
- Redgrave, T.G. Chylomicron metabolism. Biochem. Soc. Trans. 2004, 32, 79–82. [Google Scholar] [CrossRef]
- Milger, K.; Herrmann, T.; Becker, C.; Gotthardt, D.; Zickwolf, J.; Ehehalt, R.; Watkins, P.A.; Stremmel, W.; Füllekrug, J. Cellular uptake of fatty acids driven by the ER-localized acyl-CoA synthetase FATP4. J. Cell Sci. 2006, 119, 4678–4688. [Google Scholar] [CrossRef]
- Black, D.D. Development and physiological regulation of intestinal lipid absorption. I. Development of intestinal lipid absorption: Cellular events in chylomicron assembly and secretion. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 293, G519–G524. [Google Scholar] [CrossRef]
- Yen, C.L.; Farese, R.V., Jr. MGAT2, a monoacylglycerol acyltransferase expressed in the small intestine. J. Biol. Chem. 2003, 278, 18532–18537. [Google Scholar] [CrossRef] [PubMed]
- Buhman, K.K.; Smith, S.J.; Stone, S.J.; Repa, J.J.; Wong, J.S.; Knapp, F.F., Jr.; Burri, B.J.; Hamilton, R.L.; Abumrad, N.A.; Farese, R.V., Jr. DGAT1 is not essential for intestinal triacylglycerol absorption or chylomicron synthesis. J. Biol. Chem. 2002, 277, 25474–25479. [Google Scholar] [CrossRef] [PubMed]
- Ko, C.W.; Qu, J.; Black, D.D.; Tso, P. Regulation of intestinal lipid metabolism: Current concepts and relevance to disease. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 169–183. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.M.; Kancha, R.K.; Zhou, Z.; Luchoomun, J.; Zu, H.; Bakillah, A. Chylomicron assembly and catabolism: Role of apolipoproteins and receptors. Biochim. Biophys. Acta 1996, 1300, 151–170. [Google Scholar] [CrossRef]
- Kumar, N.S.; Mansbach, C.M., 2nd. Prechylomicron transport vesicle: Isolation and partial characterization. Am. J. Physiol. 1999, 276, G378–G386. [Google Scholar]
- Siddiqi, S.; Saleem, U.; Abumrad, N.A.; Davidson, N.O.; Storch, J.; Siddiqi, S.A.; Mansbach, C.M., 2nd. A novel multiprotein complex is required to generate the prechylomicron transport vesicle from intestinal ER. J. Lipid Res. 2010, 51, 1918–1928. [Google Scholar] [CrossRef]
- Siddiqi, S.A.; Mahan, J.; Siddiqi, S.; Gorelick, F.S.; Mansbach, C.M., 2nd. Vesicle-associated membrane protein 7 is expressed in intestinal ER. J. Cell Sci. 2006, 119, 943–950. [Google Scholar] [CrossRef]
- Havel, R.J.; Kane, J.P.; Kashyap, M.L. Interchange of apolipoproteins between chylomicrons and high density lipoproteins during alimentary lipemia in man. J. Clin. Investig. 1973, 52, 32–38. [Google Scholar] [CrossRef]
- Willnow, T.E. Mechanisms of hepatic chylomicron remnant clearance. Diabet. Med. 1997, 14 (Suppl. S3), S75–S80. [Google Scholar] [CrossRef]
- Tiwari, S.; Siddiqi, S.A. Intracellular trafficking and secretion of VLDL. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1079–1086. [Google Scholar] [CrossRef]
- Shelness, G.S.; Ingram, M.F.; Huang, X.F.; DeLozier, J.A. Apolipoprotein B in the rough endoplasmic reticulum: Translation, translocation and the initiation of lipoprotein assembly. J. Nutr. 1999, 129, 456s–462s. [Google Scholar] [PubMed]
- Qin, W.; Sundaram, M.; Wang, Y.; Zhou, H.; Zhong, S.; Chang, C.-C.; Manhas, S.; Yao, E.F.; Parks, R.J.; McFie, P.J.; et al. Missense mutation in APOC3 within the C-terminal lipid binding domain of human ApoC-III results in impaired assembly and secretion of triacylglycerol-rich very low density lipoproteins: Evidence that ApoC-III plays a major role in the formation of lipid precursors within the microsomal lumen. J. Biol. Chem. 2011, 286, 27769–27780. [Google Scholar]
- Gusarova, V.; Seo, J.; Sullivan, M.L.; Watkins, S.C.; Brodsky, J.L.; Fisher, E.A. Golgi-associated maturation of very low density lipoproteins involves conformational changes in apolipoprotein B, but is not dependent on apolipoprotein E. J. Biol. Chem. 2007, 282, 19453–19462. [Google Scholar] [CrossRef] [PubMed]
- Soutar, A.K.; Myant, N.B.; Thompson, G.R. The metabolism of very low density and intermediate density lipoproteins in patients with familial hypercholesterolaemia. Atherosclerosis 1982, 43, 217–231. [Google Scholar] [CrossRef]
- Takahashi, S.; Sakai, J.; Fujino, T.; Hattori, H.; Zenimaru, Y.; Suzuki, J.; Miyamori, I.; Yamamoto, T.T. The Very Low-density Lipoprotein (VLDL) Receptor: Characterization and Functions as a Peripheral Lipoprotein Receptor. J. Atheroscler. Thromb. 2004, 11, 200–208. [Google Scholar] [CrossRef]
- Esterbauer, H.; Gebicki, J.; Puhl, H.; Jürgens, G. The role of lipid peroxidation and antioxidants in oxidative modification of LDL. Free Radic. Biol. Med. 1992, 13, 341–390. [Google Scholar] [CrossRef]
- Shelness, G.S.; Sellers, J.A. Very-low-density lipoprotein assembly and secretion. Curr. Opin. Lipidol. 2001, 12, 151–157. [Google Scholar] [CrossRef]
- Orekhov, A.N.; Tertov, V.V.; Mukhin, D.N.; Mikhailenko, I.A. Modification of low density lipoprotein by desialylation causes lipid accumulation in cultured cells: Discovery of desialylated lipoprotein with altered cellular metabolism in the blood of atherosclerotic patients. Biochem. Biophys. Res. Commun. 1989, 162, 206–211. [Google Scholar] [CrossRef]
- Quinn, M.T.; Parthasarathy, S.; Fong, L.G.; Steinberg, D. Oxidatively modified low density lipoproteins: A potential role in recruitment and retention of monocyte/macrophages during atherogenesis. Proc. Natl. Acad. Sci. USA 1987, 84, 2995–2998. [Google Scholar] [CrossRef]
- Sobenin, I.A.; Tertov, V.V.; Orekhov, A.N. Characterization of chemical composition of native and modified low density lipoprotein occurring in the blood of diabetic patients. Int. Angiol. 1994, 13, 78–83. [Google Scholar]
- Kollar, B.; Siarnik, P.; Hluchanova, A.; Klobucnikova, K.; Mucska, I.; Turcani, P.; Paduchova, Z.; Katrencikova, B.; Janubova, M.; Konarikova, K.; et al. The impact of sleep apnea syndrome on the altered lipid metabolism and the redox balance. Lipids Health Dis. 2021, 20, 175. [Google Scholar] [CrossRef] [PubMed]
- Khosravi, M.; Hosseini-Fard, R.; Najafi, M. Circulating low density lipoprotein (LDL). Horm. Mol. Biol. Clin. Investig. 2018, 35. [Google Scholar] [CrossRef] [PubMed]
- McPherson, R.; Gauthier, A. Molecular regulation of SREBP function: The Insig-SCAP connection and isoform-specific modulation of lipid synthesis. Biochem. Cell Biol. 2004, 82, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.S.; Goldstein, J.L. A proteolytic pathway that controls the cholesterol content of membranes, cells, and blood. Proc. Natl. Acad. Sci. USA 1999, 96, 11041–11048. [Google Scholar] [CrossRef]
- Nohturfft, A.; DeBose-Boyd, R.A.; Scheek, S.; Goldstein, J.L.; Brown, M.S. Sterols regulate cycling of SREBP cleavage-activating protein (SCAP) between endoplasmic reticulum and Golgi. Proc. Natl. Acad. Sci. USA 1999, 96, 11235–11240. [Google Scholar] [CrossRef]
- Adams, C.M.; Goldstein, J.L.; Brown, M.S. Cholesterol-induced conformational change in SCAP enhanced by Insig proteins and mimicked by cationic amphiphiles. Proc. Natl. Acad. Sci. USA 2003, 100, 10647–10652. [Google Scholar] [CrossRef]
- Maxwell, K.N.; Breslow, J.L. Adenoviral-mediated expression of Pcsk9 in mice results in a low-density lipoprotein receptor knockout phenotype. Proc. Natl. Acad. Sci. USA 2004, 101, 7100–7105. [Google Scholar] [CrossRef]
- Gorovoy, M.; Gaultier, A.; Campana, W.M.; Firestein, G.S.; Gonias, S.L. Inflammatory mediators promote production of shed LRP1/CD91, which regulates cell signaling and cytokine expression by macrophages. J. Leukoc. Biol. 2010, 88, 769–778. [Google Scholar] [CrossRef]
- Laatsch, A.; Merkel, M.; Talmud, P.J.; Grewal, T.; Beisiegel, U.; Heeren, J. Insulin stimulates hepatic low density lipoprotein receptor-related protein 1 (LRP1) to increase postprandial lipoprotein clearance. Atherosclerosis 2009, 204, 105–111. [Google Scholar] [CrossRef]
- Beisiegel, U.; Weber, W.; Bengtsson-Olivecrona, G. Lipoprotein lipase enhances the binding of chylomicrons to low density lipoprotein receptor-related protein. Proc. Natl. Acad. Sci. USA 1991, 88, 8342–8346. [Google Scholar] [CrossRef]
- Kounnas, M.Z.; Chappell, D.A.; Wong, H.; Argraves, W.S.; Strickland, D.K. The Cellular Internalization and Degradation of Hepatic Lipase Is Mediated by Low Density Lipoprotein Receptor-related Protein and Requires Cell Surface Proteoglycans. J. Biol. Chem. 1995, 270, 9307–9312. [Google Scholar] [CrossRef] [PubMed]
- Laatsch, A.; Panteli, M.; Sornsakrin, M.; Hoffzimmer, B.; Grewal, T.; Heeren, J. Low density lipoprotein receptor-related protein 1 dependent endosomal trapping and recycling of apolipoprotein E. PLoS ONE 2012, 7, e29385. [Google Scholar] [CrossRef] [PubMed]
- Lillis, A.P.; Muratoglu, S.C.; Au, D.T.; Migliorini, M.; Lee, M.J.; Fried, S.K.; Mikhailenko, I.; Strickland, D.K. LDL receptor-related protein-1 (LRP1) regulates cholesterol accumulation in macrophages. PLoS ONE 2015, 10, e0128903. [Google Scholar] [CrossRef]
- Samouillan, V.; Dandurand, J.; Nasarre, L.; Badimon, L.; Lacabanne, C.; Llorente-Cortés, V. Lipid loading of human vascular smooth muscle cells induces changes in tropoelastin protein levels and physical structure. Biophys. J. 2012, 103, 532–540. [Google Scholar] [CrossRef]
- Ranganathan, S.; Cao, C.; Catania, J.; Migliorini, M.; Zhang, L.; Strickland, D.K. Molecular basis for the interaction of low density lipoprotein receptor-related protein 1 (LRP1) with integrin alphaMbeta2: Identification of binding sites within alphaMbeta2 for LRP1. J. Biol. Chem. 2011, 286, 30535–30541. [Google Scholar] [CrossRef] [PubMed]
- Gaultier, A.; Arandjelovic, S.; Li, X.; Janes, J.; Dragojlovic, N.; Zhou, G.P.; Dolkas, J.; Myers, R.R.; Gonias, S.L.; Campana, W.M. A shed form of LDL receptor-related protein-1 regulates peripheral nerve injury and neuropathic pain in rodents. J. Clin. Investig. 2008, 118, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Beigneux, A.P.; Davies, B.S.J.; Gin, P.; Weinstein, M.M.; Farber, E.; Qiao, X.; Peale, F.; Bunting, S.; Walzem, R.L.; Wong, J.S.; et al. Glycosylphosphatidylinositol-Anchored High-Density Lipoprotein-Binding Protein 1 Plays a Critical Role in the Lipolytic Processing of Chylomicrons. Cell Metab. 2007, 5, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Kersten, S. Physiological regulation of lipoprotein lipase. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2014, 1841, 919–933. [Google Scholar] [CrossRef]
- Doolittle, M.H.; Ben-Zeev, O.; Elovson, J.; Martin, D.; Kirchgessner, T.G. The response of lipoprotein lipase to feeding and fasting. Evidence for posttranslational regulation. J. Biol. Chem. 1990, 265, 4570–4577. [Google Scholar] [CrossRef]
- Borensztajn, J.; Robinson, D.S.T. The effect of fasting on the utilization of chylomicron triglyceride fatty acids in relation to clearing factor lipase (lipoprotein lipase) releasable by heparin in the perfused rat heart. J. Lipid Res. 1970, 11, 111–117. [Google Scholar] [CrossRef]
- Hamilton, M.T.; Etienne, J.; McClure, W.C.; Pavey, B.S.; Holloway, A.K. Role of local contractile activity and muscle fiber type on LPL regulation during exercise. Am. J. Physiol. 1998, 275, E1016–E1022. [Google Scholar] [CrossRef]
- Larsson, M.; Vorrsjö, E.; Talmud, P.; Lookene, A.; Olivecrona, G. Apolipoproteins C-I and C-III Inhibit Lipoprotein Lipase Activity by Displacement of the Enzyme from Lipid Droplets*. J. Biol. Chem. 2013, 288, 33997–34008. [Google Scholar] [CrossRef] [PubMed]
- Quagliarini, F.; Wang, Y.; Kozlitina, J.; Grishin, N.V.; Hyde, R.; Boerwinkle, E.; Valenzuela, D.M.; Murphy, A.J.; Cohen, J.C.; Hobbs, H.H. Atypical angiopoietin-like protein that regulates ANGPTL3. Proc. Natl. Acad. Sci. USA 2012, 109, 19751. [Google Scholar] [CrossRef] [PubMed]
- Mandard, S.; Zandbergen, F.; van Straten, E.; Wahli, W.; Kuipers, F.; Müller, M.; Kersten, S. The Fasting-induced Adipose Factor/Angiopoietin-like Protein 4 Is Physically Associated with Lipoproteins and Governs Plasma Lipid Levels and Adiposity. J. Biol. Chem. 2006, 281, 934–944. [Google Scholar] [CrossRef] [PubMed]
- Tosheska Trajkovska, K.; Topuzovska, S. High-density lipoprotein metabolism and reverse cholesterol transport: Strategies for raising HDL cholesterol. Anatol. J. Cardiol. 2017, 18, 149–154. [Google Scholar] [CrossRef]
- Rached, F.H.; Chapman, M.J.; Kontush, A. HDL particle subpopulations: Focus on biological function. Biofactors 2015, 41, 67–77. [Google Scholar] [CrossRef]
- von Eckardstein, A.; Kardassis, D. High Density Lipoproteins: From Biological Understanding to Clinical Exploitation; Springer: Berlin/Heidelberg, Germany, 2015; p. 694. [Google Scholar]
- Camont, L.; Chapman, M.J.; Kontush, A. Biological activities of HDL subpopulations and their relevance to cardiovascular disease. Trends. Mol. Med. 2011, 17, 594–603. [Google Scholar] [CrossRef]
- Oravec, S.; Dukat, A.; Gavornik, P.; Caprnda, M.; Kucera, M.; Ocadlik, I. Contribution of the atherogenic lipoprotein profile to the development of arterial hypertension. Bratisl Lek Listy 2011, 112, 4–7. [Google Scholar]
- Oravec, S.; Dostal, E.; Dukát, A.; Gavorník, P.; Kucera, M.; Gruber, K. HDL subfractions analysis: A new laboratory diagnostic assay for patients with cardiovascular diseases and dyslipoproteinemia. Neuro. Endocrinol. Lett. 2011, 32, 502–509. [Google Scholar]
- Tailleux, A.; Duriez, P.; Fruchart, J.C.; Clavey, V. Apolipoprotein A-II, HDL metabolism and atherosclerosis. Atherosclerosis 2002, 164, 1–13. [Google Scholar] [CrossRef]
- Klimov, A.N.; Gurevich, V.S.; Nikiforova, A.A.; Shatilina, L.V.; Kuzmin, A.A.; Plavinsky, S.L.; Teryukova, N.P. Antioxidative activity of high density lipoproteins in vivo. Atherosclerosis 1993, 100, 13–18. [Google Scholar] [CrossRef]
- Frank, P.G.; Marcel, Y.L. Apolipoprotein A-I: Structure-function relationships. J. Lipid Res. 2000, 41, 853–872. [Google Scholar] [CrossRef]
- Dominiczak, M.H.; Caslake, M.J. Apolipoproteins: Metabolic role and clinical biochemistry applications. Ann. Clin. Biochem. 2011, 48, 498–515. [Google Scholar] [CrossRef]
- Mulya, A.; Seo, J.; Brown, A.L.; Gebre, A.K.; Boudyguina, E.; Shelness, G.S.; Parks, J.S. Apolipoprotein M expression increases the size of nascent pre beta HDL formed by ATP binding cassette transporter A1. J. Lipid Res. 2010, 51, 514–524. [Google Scholar] [CrossRef] [PubMed]
- Christoffersen, C.; Nielsen, L.B.; Axler, O.; Andersson, A.; Johnsen, A.H.; Dahlbäck, B. Isolation and characterization of human apolipoprotein M-containing lipoproteins. J. Lipid Res. 2006, 47, 1833–1843. [Google Scholar] [CrossRef] [PubMed]
- Christoffersen, C.; Obinata, H.; Kumaraswamy, S.B.; Galvani, S.; Ahnström, J.; Sevvana, M.; Egerer-Sieber, C.; Muller, Y.A.; Hla, T.; Nielsen, L.B.; et al. Endothelium-protective sphingosine-1-phosphate provided by HDL-associated apolipoprotein M. Proc. Natl. Acad. Sci. USA 2011, 108, 9613–9618. [Google Scholar] [CrossRef]
- van der Vorst, E.P.C. High-Density Lipoproteins and Apolipoprotein A1. Subcell Biochem. 2020, 94, 399–420. [Google Scholar]
- Pereira, R.M.; Mekary, R.A.; da Cruz Rodrigues, K.C.; Anaruma, C.P.; Ropelle, E.R.; da Silva, A.S.R.; Cintra, D.E.; Pauli, J.R.; de Moura, L.P. Protective molecular mechanisms of clusterin against apoptosis in cardiomyocytes. Heart Fail. Rev. 2018, 23, 123–129. [Google Scholar] [CrossRef]
- Timmins, J.M.; Lee, J.-Y.; Boudyguina, E.; Kluckman, K.D.; Brunham, L.R.; Mulya, A.; Gebre, A.K.; Coutinho, J.M.; Colvin, P.L.; Smith, T.L.; et al. Targeted inactivation of hepatic Abca1 causes profound hypoalphalipoproteinemia and kidney hypercatabolism of apoA-I. J. Clin. Investig. 2005, 115, 1333–1342. [Google Scholar] [CrossRef]
- Brunham, L.R.; Kruit, J.K.; Iqbal, J.; Fievet, C.; Timmins, J.M.; Pape, T.D.; Coburn, B.A.; Bissada, N.; Staels, B.; Groen, A.K.; et al. Intestinal ABCA1 directly contributes to HDL biogenesis in vivo. J. Clin. Investig. 2006, 116, 1052–1062. [Google Scholar] [CrossRef]
- Langmann, T.; Klucken, J.; Reil, M.; Liebisch, G.; Luciani, M.F.; Chimini, G.; Kaminski, W.E.; Schmitz, G. Molecular cloning of the human ATP-binding cassette transporter 1 (hABC1): Evidence for sterol-dependent regulation in macrophages. Biochem. Biophys. Res. Commun. 1999, 257, 29–33. [Google Scholar] [CrossRef] [PubMed]
- Oram, J.F.; Vaughan, A.M. ABCA1-mediated transport of cellular cholesterol and phospholipids to HDL apolipoproteins. Curr. Opin. Lipidol. 2000, 11, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Bailey, D.; Ruel, I.; Hafiane, A.; Cochrane, H.; Iatan, I.; Jauhiainen, M.; Ehnholm, C.; Krimbou, L.; Genest, J. Analysis of lipid transfer activity between model nascent HDL particles and plasma lipoproteins: Implications for current concepts of nascent HDL maturation and genesis. J. Lipid Res. 2010, 51, 785–797. [Google Scholar] [CrossRef] [PubMed]
- Jauhiainen, M.; Metso, J.; Pahlman, R.; Blomqvist, S.; van Tol, A.; Ehnholm, C. Human plasma phospholipid transfer protein causes high density lipoprotein conversion. J. Biol. Chem. 1993, 268, 4032–4036. [Google Scholar] [CrossRef]
- Acton, S.; Rigotti, A.; Landschulz, K.T.; Xu, S.; Hobbs, H.H.; Krieger, M. Identification of scavenger receptor SR-BI as a high density lipoprotein receptor. Science 1996, 271, 518–520. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Laccotripe, M.; Huang, X.; Rigotti, A.; Zannis, V.I.; Krieger, M. Apolipoproteins of HDL can directly mediate binding to the scavenger receptor SR-BI, an HDL receptor that mediates selective lipid uptake. J. Lipid Res. 1997, 38, 1289–1298. [Google Scholar] [CrossRef]
- Thuren, T. Hepatic lipase and HDL metabolism. Curr. Opin. Lipidol. 2000, 11, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Chapman, M.J.; Le Goff, W.; Guerin, M.; Kontush, A. Cholesteryl ester transfer protein: At the heart of the action of lipid-modulating therapy with statins, fibrates, niacin, and cholesteryl ester transfer protein inhibitors. Eur. Heart J. 2010, 31, 149–164. [Google Scholar] [CrossRef]
- Barter, P.J.; Brewer, H.B., Jr.; Chapman, M.J.; Hennekens, C.H.; Rader, D.J.; Tall, A.R. Cholesteryl ester transfer protein: A novel target for raising HDL and inhibiting atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 160–167. [Google Scholar] [CrossRef]
- Silver, D.L.; Wang, N.; Xiao, X.; Tall, A.R. High density lipoprotein (HDL) particle uptake mediated by scavenger receptor class B type 1 results in selective sorting of HDL cholesterol from protein and polarized cholesterol secretion. J. Biol. Chem. 2001, 276, 25287–25293. [Google Scholar] [CrossRef]
- Pagler, T.A.; Rhode, S.; Neuhofer, A.; Laggner, H.; Strobl, W.; Hinterndorfer, C.; Volf, I.; Pavelka, M.; Eckhardt, E.R.; van der Westhuyzen, D.R.; et al. SR-BI-mediated high density lipoprotein (HDL) endocytosis leads to HDL resecretion facilitating cholesterol efflux. J. Biol. Chem. 2006, 281, 11193–11204. [Google Scholar] [CrossRef] [PubMed]
- Daniil, G.; Phedonos, A.A.; Holleboom, A.G.; Motazacker, M.M.; Argyri, L.; Kuivenhoven, J.A.; Chroni, A. Characterization of antioxidant/anti-inflammatory properties and apoA-I-containing subpopulations of HDL from family subjects with monogenic low HDL disorders. Clin. Chim. Acta. 2011, 412, 1213–1220. [Google Scholar] [CrossRef] [PubMed]
- Kontush, A.; Chantepie, S.; Chapman, M.J. Small, dense HDL particles exert potent protection of atherogenic LDL against oxidative stress. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1881–1888. [Google Scholar] [CrossRef] [PubMed]
- Shuhei, N.; Söderlund, S.; Jauhiainen, M.; Taskinen, M.R. Effect of HDL composition and particle size on the resistance of HDL to the oxidation. Lipids Health Dis. 2010, 9, 104. [Google Scholar] [CrossRef] [PubMed]
- Aviram, M.; Rosenblat, M.; Bisgaier, C.L.; Newton, R.S.; Primo-Parmo, S.L.; La Du, B.N. Paraoxonase inhibits high-density lipoprotein oxidation and preserves its functions. A possible peroxidative role for paraoxonase. J. Clin. Investig. 1998, 101, 1581–1590. [Google Scholar] [CrossRef]
- Nicholls, S.J.; Dusting, G.J.; Cutri, B.; Bao, S.; Drummond, G.R.; Rye, K.A.; Barter, P.J. Reconstituted high-density lipoproteins inhibit the acute pro-oxidant and proinflammatory vascular changes induced by a periarterial collar in normocholesterolemic rabbits. Circulation 2005, 111, 1543–1550. [Google Scholar] [CrossRef]
- Yvan-Charvet, L.; Wang, N.; Tall, A.R. Role of HDL, ABCA1, and ABCG1 Transporters in Cholesterol Efflux and Immune Responses. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 139–143. [Google Scholar] [CrossRef]
- Almendros, I.; Basoglu, Ö.K.; Conde, S.V.; Liguori, C.; Saaresranta, T. Metabolic dysfunction in OSA: Is there something new under the sun? J. Sleep Res. 2022, 31, e13418. [Google Scholar] [CrossRef]
- Farré, R.; Montserrat, J.M.; Gozal, D.; Almendros, I.; Navajas, D. Intermittent Hypoxia Severity in Animal Models of Sleep Apnea. Front. Physiol. 2018, 9, 1556. [Google Scholar] [CrossRef]
- Li, J.; Thorne, L.N.; Punjabi, N.M.; Sun, C.K.; Schwartz, A.R.; Smith, P.L.; Marino, R.L.; Rodriguez, A.; Hubbard, W.C.; O’Donnell, C.P.; et al. Intermittent hypoxia induces hyperlipidemia in lean mice. Circ. Res. 2005, 97, 698–706. [Google Scholar] [CrossRef]
- Li, J.; Bosch-Marce, M.; Nanayakkara, A.; Savransky, V.; Fried, S.K.; Semenza, G.L.; Polotsky, V.Y. Altered metabolic responses to intermittent hypoxia in mice with partial deficiency of hypoxia-inducible factor-1alpha. Physiol. Genom. 2006, 25, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Tabor, D.E.; Kim, J.B.; Spiegelman, B.M.; Edwards, P.A. Identification of conserved cis-elements and transcription factors required for sterol-regulated transcription of stearoyl-CoA desaturase 1 and 2. J. Biol. Chem. 1999, 274, 20603–20610. [Google Scholar] [CrossRef] [PubMed]
- Savransky, V.; Nanayakkara, A.; Li, J.; Bevans, S.; Smith, P.L.; Rodriguez, A.; Polotsky, V.Y. Chronic intermittent hypoxia induces atherosclerosis. Am. J. Respir. Crit. Care Med. 2007, 175, 1290–1297. [Google Scholar] [CrossRef] [PubMed]
- Tamisier, R.; Gilmartin, G.S.; Launois, S.H.; Pépin, J.L.; Nespoulet, H.; Thomas, R.; Lévy, P.; Weiss, J.W. A new model of chronic intermittent hypoxia in humans: Effect on ventilation, sleep, and blood pressure. J. Appl. Physiol. 2009, 107, 17–24. [Google Scholar] [CrossRef]
- Dobrosielski, D.A.; Papandreou, C.; Patil, S.P.; Salas-Salvadó, J. Diet and exercise in the management of obstructive sleep apnoea and cardiovascular disease risk. Eur. Respir. Rev. 2017, 26. [Google Scholar] [CrossRef]
- Giel, K.E.; Bulik, C.M.; Fernandez-Aranda, F.; Hay, P.; Keski-Rahkonen, A.; Schag, K.; Schmidt, U.; Zipfel, S. Binge eating disorder. Nat. Rev. Dis. Primers 2022, 8, 16. [Google Scholar] [CrossRef]
- Matsumoto, T.; Harada, N.; Azuma, M.; Chihara, Y.; Murase, K.; Tachikawa, R.; Minami, T.; Hamada, S.; Tanizawa, K.; Inouchi, M.; et al. Plasma Incretin Levels and Dipeptidyl Peptidase-4 Activity in Patients with Obstructive Sleep Apnea. Ann. Am. Thorac. Soc. 2016, 13, 1378–1387. [Google Scholar] [CrossRef]
- Shobatake, R.; Itaya-Hironaka, A.; Yamauchi, A.; Makino, M.; Sakuramoto-Tsuchida, S.; Uchiyama, T.; Ota, H.; Takahashi, N.; Ueno, S.; Sugie, K.; et al. Intermittent Hypoxia Up-Regulates Gene Expressions of Peptide YY (PYY), Glucagon-like Peptide-1 (GLP-1), and Neurotensin (NTS) in Enteroendocrine Cells. Int. J. Mol. Sci. 2019, 20, 1849. [Google Scholar] [CrossRef]
- Hakim, F.; Wang, Y.; Carreras, A.; Hirotsu, C.; Zhang, J.; Peris, E.; Gozal, D. Chronic sleep fragmentation during the sleep period induces hypothalamic endoplasmic reticulum stress and PTP1b-mediated leptin resistance in male mice. Sleep 2015, 38, 31–40. [Google Scholar] [CrossRef]
- Dash, S.; Xiao, C.; Morgantini, C.; Lewis, G.F. New Insights into the Regulation of Chylomicron Production. Annu. Rev. Nutr. 2015, 35, 265–294. [Google Scholar] [CrossRef]
- Phillips, C.L.; Yee, B.J.; Marshall, N.S.; Liu, P.Y.; Sullivan, D.R.; Grunstein, R.R. Continuous positive airway pressure reduces postprandial lipidemia in obstructive sleep apnea: A randomized, placebo-controlled crossover trial. Am. J. Respir. Crit. Care Med. 2011, 184, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Drager, L.F.; Li, J.; Shin, M.K.; Reinke, C.; Aggarwal, N.R.; Jun, J.C.; Bevans-Fonti, S.; Sztalryd, C.; O’Byrne, S.M.; Kroupa, O.; et al. Intermittent hypoxia inhibits clearance of triglyceride-rich lipoproteins and inactivates adipose lipoprotein lipase in a mouse model of sleep apnoea. Eur. Heart J. 2012, 33, 783–790. [Google Scholar] [CrossRef] [PubMed]
- Morin, R.; Mauger, J.F.; Amaratunga, R.; Imbeault, P. The effect of acute intermittent hypoxia on postprandial triglyceride levels in humans: A randomized crossover trial. J. Transl. Med. 2021, 19, 268. [Google Scholar] [CrossRef] [PubMed]
- Mwaikambo, B.R.; Yang, C.; Chemtob, S.; Hardy, P. Hypoxia up-regulates CD36 expression and function via hypoxia-inducible factor-1- and phosphatidylinosi.itol 3-kinase-dependent mechanisms. J. Biol. Chem. 2009, 284, 26695–26707. [Google Scholar] [CrossRef] [PubMed]
- Flores, J.J.; Klebe, D.; Rolland, W.B.; Lekic, T.; Krafft, P.R.; Zhang, J.H. PPARγ-induced upregulation of CD36 enhances hematoma resolution and attenuates long-term neurological deficits after germinal matrix hemorrhage in neonatal rats. Neurobiol. Dis. 2016, 87, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Rey, E.; Del Pozo-Maroto, E.; Marañón, P.; Beeler, B.; García-García, Y.; Landete, P.; Isaza, S.C.; Farré, R.; García-Monzón, C.; Almendros, I.; et al. Intrahepatic Expression of Fatty Acid Translocase CD36 Is Increased in Obstructive Sleep Apnea. Front. Med. 2020, 7, 450. [Google Scholar] [CrossRef]
- Moon, Y.; Park, B.; Park, H. Hypoxic repression of CYP7A1 through a HIF-1α- and SHP-independent mechanism. BMB Rep. 2016, 49, 173–178. [Google Scholar] [CrossRef]
- Drager, L.F.; Tavoni, T.M.; Silva, V.M.; Santos, R.D.; Pedrosa, R.P.; Bortolotto, L.A.; Vinagre, C.G.; Polotsky, V.Y.; Lorenzi-Filho, G.; Maranhao, R.C. Obstructive sleep apnea and effects of continuous positive airway pressure on triglyceride-rich lipoprotein metabolism. J. Lipid Res. 2018, 59, 1027–1033. [Google Scholar] [CrossRef]
- Mahat, B.; Chassé, É.; Mauger, J.-F.; Imbeault, P. Effects of acute hypoxia on human adipose tissue lipoprotein lipase activity and lipolysis. J. Transl. Med. 2016, 14, 212. [Google Scholar] [CrossRef]
- Iesato, K.; Tatsumi, K.; Saibara, T.; Nakamura, A.; Terada, J.; Tada, Y.; Sakao, S.; Tanabe, N.; Takiguchi, Y.; Kuriyama, T. Decreased lipoprotein lipase in obstructive sleep apnea syndrome. Circ. J. 2007, 71, 1293–1298. [Google Scholar] [CrossRef]
- Yao, Q.; Shin, M.K.; Jun, J.C.; Hernandez, K.L.; Aggarwal, N.R.; Mock, J.R.; Gay, J.; Drager, L.F.; Polotsky, V.Y. Effect of chronic intermittent hypoxia on triglyceride uptake in different tissues. J. Lipid Res. 2013, 54, 1058–1065. [Google Scholar] [CrossRef] [PubMed]
- Jun, J.C.; Shin, M.K.; Yao, Q.; Bevans-Fonti, S.; Poole, J.; Drager, L.F.; Polotsky, V.Y. Acute hypoxia induces hypertriglyceridemia by decreasing plasma triglyceride clearance in mice. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E377–E388. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, Y.; Wang, J.; Feng, P.; Chen, R.; Cao, Y.; Liu, C. Association between serum lipoprotein lipase level and dyslipidemia in patients with obstructive sleep apnea syndrome. Zhonghua Yi Xue Za Zhi 2014, 94, 403–407. [Google Scholar]
- Drager, L.F.; Yao, Q.; Hernandez, K.L.; Shin, M.K.; Bevans-Fonti, S.; Gay, J.; Sussan, T.E.; Jun, J.C.; Myers, A.C.; Olivecrona, G.; et al. Chronic intermittent hypoxia induces atherosclerosis via activation of adipose angiopoietin-like 4. Am. J. Respir. Crit. Care Med. 2013, 188, 240–248. [Google Scholar] [CrossRef]
- Al-Terki, A.; Abu-Farha, M.; AlKhairi, I.; Cherian, P.T.; Sriraman, D.; Shyamsundar, A.; Ali, S.; Almulla, F.; Tuomilehto, J.; Abubaker, J.A. Increased Level of Angiopoietin Like Proteins 4 and 8 in People With Sleep Apnea. Front. Endocrinol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yang, Y.; Jiao, X.; Yu, H.; Du, Y.; Zhang, M.; Hu, C.; Wei, Y.; Qin, Y. The Clinical Role of Angiopoietin-Like Protein 3 in Evaluating Coronary Artery Disease in Patients with Obstructive Sleep Apnea. Cardiovasc. Drugs 2020, 34, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Schoonjans, K.; Peinado-Onsurbe, J.; Lefebvre, A.M.; Heyman, R.A.; Briggs, M.; Deeb, S.; Staels, B.; Auwerx, J. PPARalpha and PPARgamma activators direct a distinct tissue-specific transcriptional response via a PPRE in the lipoprotein lipase gene. Embo J. 1996, 15, 5336–5348. [Google Scholar] [CrossRef]
- Yun, Z.; Maecker, H.L.; Johnson, R.S.; Giaccia, A.J. Inhibition of PPAR gamma 2 gene expression by the HIF-1-regulated gene DEC1/Stra13: A mechanism for regulation of adipogenesis by hypoxia. Dev. Cell 2002, 2, 331–341. [Google Scholar] [CrossRef]
- Mackay, A.G.; Oliver, J.D.; Rogers, M.P. Regulation of lipoprotein lipase activity and mRNA content in rat epididymal adipose tissue in vitro by recombinant tumour necrosis factor. Biochem. J. 1990, 269, 123–126. [Google Scholar] [CrossRef]
- Feingold, K.R.; Marshall, M.; Gulli, R.; Moser, A.H.; Grunfeld, C. Effect of endotoxin and cytokines on lipoprotein lipase activity in mice. Arterioscler. Thromb. 1994, 14, 1866–1872. [Google Scholar] [CrossRef]
- Grunfeld, C.; Gulli, R.; Moser, A.H.; Gavin, L.A.; Feingold, K.R. Effect of tumor necrosis factor administration in vivo on lipoprotein lipase activity in various tissues of the rat. J. Lipid Res. 1989, 30, 579–585. [Google Scholar] [CrossRef]
- Zechner, R.; Newman, T.C.; Sherry, B.; Cerami, A.; Breslow, J.L. Recombinant human cachectin/tumor necrosis factor but not interleukin-1 alpha downregulates lipoprotein lipase gene expression at the transcriptional level in mouse 3T3-L1 adipocytes. Mol. Cell. Biol. 1988, 8, 2394. [Google Scholar] [PubMed]
- Wu, G.; Brouckaert, P.; Olivecrona, T. Rapid downregulation of adipose tissue lipoprotein lipase activity on food deprivation: Evidence that TNF-alpha is involved. Am. J. Physiol. Endocrinol. Metab. 2004, 286, E711–E717. [Google Scholar] [CrossRef] [PubMed]
- Somers, V.K.; Dyken, M.E.; Clary, M.P.; Abboud, F.M. Sympathetic neural mechanisms in obstructive sleep apnea. J. Clin. Investig. 1995, 96, 1897–1904. [Google Scholar] [CrossRef] [PubMed]
- Raynolds, M.V.; Awald, P.D.; Gordon, D.F.; Gutierrez-Hartmann, A.; Rule, D.C.; Wood, W.M.; Eckel, R.H. Lipoprotein Lipase Gene Expression in Rat Adipocytes Is Regulated by Isoproterenol and Insulin through Different Mechanisms. Mol. Endocrinol. 1990, 4, 1416–1422. [Google Scholar] [CrossRef]
- Chiappe de Cingalani, G.E.; Goers, J.W.; Giannotti, M.; Caldiz, C.I. Comparative effects of insulin and isoproterenol on lipoprotein lipase in rat adipose cells. Am. J. Physiol.-Cell Physiol. 1996, 270, C1461–C1467. [Google Scholar] [CrossRef]
- Dijk, W.; Heine, M.; Vergnes, L.; Boon, M.R.; Schaart, G.; Hesselink, M.K.; Reue, K.; van Marken Lichtenbelt, W.D.; Olivecrona, G.; Rensen, P.C.; et al. ANGPTL4 mediates shuttling of lipid fuel to brown adipose tissue during sustained cold exposure. Elife 2015, 4. [Google Scholar] [CrossRef]
- Ong, J.M.; Kirchgessner, T.G.; Schotz, M.C.; Kern, P.A. Insulin increases the synthetic rate and messenger RNA level of lipoprotein lipase in isolated rat adipocytes. J. Biol. Chem. 1988, 263, 12933–12938. [Google Scholar] [CrossRef]
- Inukai, K.; Nakashima, Y.; Watanabe, M.; Kurihara, S.; Awata, T.; Katagiri, H.; Oka, Y.; Katayama, S. ANGPTL3 is increased in both insulin-deficient and -resistant diabetic states. Biochem. Biophys. Res. Commun. 2004, 317, 1075–1079. [Google Scholar] [CrossRef]
- Picard, F.; Richard, D.; Huang, Q.; Deshaies, Y. Effects of leptin adipose tissue lipoprotein lipase in the obese ob/ob mouse. Int. J. Obes. 1998, 22, 1088–1095. [Google Scholar] [CrossRef][Green Version]
- Shimamura, M.; Matsuda, M.; Ando, Y.; Koishi, R.; Yasumo, H.; Furukawa, H.; Shimomura, I. Leptin and insulin down-regulate angiopoietin-like protein 3, a plasma triglyceride-increasing factor. Biochem. Biophys. Res. Commun. 2004, 322, 1080–1085. [Google Scholar] [CrossRef] [PubMed]
- Phillips, B.G.; Kato, M.; Narkiewicz, K.; Choe, I.; Somers, V.K. Increases in leptin levels, sympathetic drive, and weight gain in obstructive sleep apnea. Am. J. Physiol. Heart Circ. Physiol. 2000, 279, H234–H237. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.; Wang, X.; Hu, W.; Wang, L. Association of adiponectin level and obstructive sleep apnea prevalence in obese subjects. Medicine 2017, 96, e7784. [Google Scholar] [CrossRef] [PubMed]
- von Eynatten, M.; Schneider, J.G.; Humpert, P.M.; Rudofsky, G.; Schmidt, N.; Barosch, P.; Hamann, A.; Morcos, M.; Kreuzer, J.; Bierhaus, A.; et al. Decreased plasma lipoprotein lipase in hypoadiponectinemia: An association independent of systemic inflammation and insulin resistance. Diabetes Care 2004, 27, 2925–2929. [Google Scholar] [CrossRef] [PubMed]
- Costales, P.; Castellano, J.; Revuelta-López, E.; Cal, R.; Aledo, R.; Llampayas, O.; Nasarre, L.; Juarez, C.; Badimon, L.; Llorente-Cortés, V. Lipopolysaccharide downregulates CD91/low-density lipoprotein receptor-related protein 1 expression through SREBP-1 overexpression in human macrophages. Atherosclerosis 2013, 227, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Yin, J.; Huang, K. Free Fatty Acids Increase Intracellular Lipid Accumulation and Oxidative Stress by Modulating PPARα and SREBP-1c in L-02 Cells. Lipids 2016, 51, 797–805. [Google Scholar] [CrossRef]
- Li, J.; Nanayakkara, A.; Jun, J.; Savransky, V.; Polotsky, V.Y. Effect of deficiency in SREBP cleavage-activating protein on lipid metabolism during intermittent hypoxia. Physiol. Genom. 2007, 31, 273–280. [Google Scholar] [CrossRef]
- Guo, Y.X.; He, L.Y.; Zhang, M.; Wang, F.; Liu, F.; Peng, W.X. 1,25-Dihydroxyvitamin D3 regulates expression of LRP1 and RAGE in vitro and in vivo, enhancing Aβ1–40 brain-to-blood efflux and peripheral uptake transport. Neuroscience 2016, 322, 28–38. [Google Scholar] [CrossRef]
- Zhao, Y.; Zeng, C.Y.; Li, X.H.; Yang, T.T.; Kuang, X.; Du, J.R. Klotho overexpression improves amyloid-β clearance and cognition in the APP/PS1 mouse model of Alzheimer’s disease. Aging Cell 2020, 19, e13239. [Google Scholar] [CrossRef]
- Bozkurt, N.C.; Cakal, E.; Sahin, M.; Ozkaya, E.C.; Firat, H.; Delibasi, T. The relation of serum 25-hydroxyvitamin-D levels with severity of obstructive sleep apnea and glucose metabolism abnormalities. Endocrine 2012, 41, 518–525. [Google Scholar] [CrossRef]
- Pákó, J.; Kunos, L.; Mészáros, M.; Tárnoki, D.L.; Tárnoki Á, D.; Horváth, I.; Bikov, A. Decreased Levels of Anti-Aging Klotho in Obstructive Sleep Apnea. Rejuvenation Res. 2020, 23, 256–261. [Google Scholar] [CrossRef] [PubMed]
- de Gonzalo-Calvo, D.; Cenarro, A.; Martínez-Bujidos, M.; Badimon, L.; Bayes-Genis, A.; Ordonez-Llanos, J.; Civeira, F.; Llorente-Cortés, V. Circulating soluble low-density lipoprotein receptor-related protein 1 (sLRP1) concentration is associated with hypercholesterolemia: A new potential biomarker for atherosclerosis. Int. J. Cardiol. 2015, 201, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Meszaros, M.; Kunos, L.; Tarnoki, A.D.; Tarnoki, D.L.; Lazar, Z.; Bikov, A. The Role of Soluble Low-Density Lipoprotein Receptor-Related Protein-1 in Obstructive Sleep Apnoea. J. Clin. Med. 2021, 10, 1494. [Google Scholar] [CrossRef] [PubMed]
- Ntambi, J.M.; Miyazaki, M. Regulation of stearoyl-CoA desaturases and role in metabolism. Prog. Lipid Res. 2004, 43, 91–104. [Google Scholar] [CrossRef]
- Savransky, V.; Jun, J.; Li, J.; Nanayakkara, A.; Fonti, S.; Moser, A.B.; Steele, K.E.; Schweitzer, M.A.; Patil, S.P.; Bhanot, S.; et al. Dyslipidemia and atheroscleros.sis induced by chronic intermittent hypoxia are attenuated by deficiency of stearoyl coenzyme A desaturase. Circ. Res. 2008, 103, 1173–1180. [Google Scholar] [CrossRef]
- Li, J.; Grigoryev, D.N.; Ye, S.Q.; Thorne, L.; Schwartz, A.R.; Smith, P.L.; O’Donnell, C.P.; Polotsky, V.Y. Chronic intermittent hypoxia upregulates genes of lipid biosynthesis in obese mice. J. Appl. Physiol. 2005, 99, 1643–1648. [Google Scholar] [CrossRef]
- Salceda, S.; Caro, J. Hypoxia-inducible factor 1alpha (HIF-1alpha) protein is rapidly degraded by the ubiquitin-proteasome system under normoxic conditions. Its stabilization by hypoxia depends on redox-induced changes. J. Biol. Chem. 1997, 272, 22642–22647. [Google Scholar] [CrossRef]
- Jaakkola, P.; Mole, D.R.; Tian, Y.M.; Wilson, M.I.; Gielbert, J.; Gaskell, S.J.; von Kriegsheim, A.; Hebestreit, H.F.; Mukherji, M.; Schofield, C.J.; et al. Targeting of HIF-alpha to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science 2001, 292, 468–472. [Google Scholar] [CrossRef]
- Goyal, P.; Weissmann, N.; Grimminger, F.; Hegel, C.; Bader, L.; Rose, F.; Fink, L.; Ghofrani, H.A.; Schermuly, R.T.; Schmidt, H.H.; et al. Upregulation of NAD(P)H oxidase 1 in hypoxia activates hypoxia-inducible factor 1 via increase in reactive oxygen species. Free Radic. Biol. Med. 2004, 36, 1279–1288. [Google Scholar] [CrossRef]
- Mesarwi, O.A.; Loomba, R.; Malhotra, A. Obstructive Sleep Apnea, Hypoxia, and Nonalcoholic Fatty Liver Disease. Am. J. Respir. Crit. Care Med. 2019, 199, 830–841. [Google Scholar] [CrossRef]
- Bruinstroop, E.; Pei, L.; Ackermans, M.T.; Foppen, E.; Borgers, A.J.; Kwakkel, J.; Alkemade, A.; Fliers, E.; Kalsbeek, A. Hypothalamic neuropeptide Y (NPY) controls hepatic VLDL-triglyceride secretion in rats via the sympathetic nervous system. Diabetes 2012, 61, 1043–1050. [Google Scholar] [CrossRef]
- Jermendy, G.; Horváth, T.; Littvay, L.; Steinbach, R.; Jermendy, A.L.; Tárnoki, A.D.; Tárnoki, D.L.; Métneki, J.; Osztovits, J. Effect of genetic and environmental influences on cardiometabolic risk factors: A twin study. Cardiovasc. Diabetol. 2011, 10, 96. [Google Scholar] [CrossRef] [PubMed]
- Guilleminault, C.; Partinen, M.; Hollman, K.; Powell, N.; Stoohs, R. Familial aggregates in obstructive sleep apnea syndrome. Chest 1995, 107, 1545–1551. [Google Scholar] [CrossRef] [PubMed]
- Meszaros, M.; Tarnoki, A.D.; Tarnoki, D.L.; Kovacs, D.T.; Forgo, B.; Lee, J.; Sung, J.; Vestbo, J.; Müller, V.; Kunos, L.; et al. Obstructive sleep apnea and hypertriglyceridaemia share common genetic background: Results of a twin study. J. Sleep Res. 2020, 29, e12979. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Zhou, Q.; Zheng, F.; Wu, T.; Tang, Y.D.; Jiang, J. The Causal Effects of Lipid Profiles on Sleep Apnea. Front. Nutr. 2022, 9, 910690. [Google Scholar] [CrossRef]
- Sun, J.; Hu, J.; Tu, C.; Zhong, A.; Xu, H. Obstructive sleep apnea susceptibility genes in Chinese population: A field synopsis and meta-analysis of genetic association studies. PLoS ONE 2015, 10, e0135942. [Google Scholar] [CrossRef]
- Li, Q.; Chen, R.; Bie, L.; Zhao, D.; Huang, C.; Hong, J. Association of the variants in the PPARG gene and serum lipid levels: A meta-analysis of 74 studies. J. Cell Mol. Med. 2015, 19, 198–209. [Google Scholar] [CrossRef]
- Uyrum, E.; Balbay, O.; Annakkaya, A.N.; Gulec Balbay, E.; Silan, F.; Arbak, P. The relationship between obstructive sleep apnea syndrome and apolipoprotein E genetic variants. Respiration 2015, 89, 195–200. [Google Scholar] [CrossRef]
- Fisher, E.A.; Pan, M.; Chen, X.; Wu, X.; Wang, H.; Jamil, H.; Sparks, J.D.; Williams, K.J. The triple threat to nascent apolipoprotein B. Evidence for multiple, distinct degradative pathways. J. Biol. Chem. 2001, 276, 27855–27863. [Google Scholar] [CrossRef]
- Wen, Y.; Leake, D.S. Low Density Lipoprotein Undergoes Oxidation Within Lysosomes in Cells. Circ. Res. 2007, 100, 1337–1343. [Google Scholar] [CrossRef]
- Itabe, H.; Obama, T.; Kato, R. The Dynamics of Oxidized LDL during Atherogenesis. J. Lipids 2011, 2011, 418313. [Google Scholar] [CrossRef] [PubMed]
- Poznyak, A.V.; Nikiforov, N.G.; Markin, A.M.; Kashirskikh, D.A.; Myasoedova, V.A.; Gerasimova, E.V.; Orekhov, A.N. Overview of OxLDL and Its Impact on Cardiovascular Health: Focus on Atherosclerosis. Front. Pharm. 2020, 11, 613780. [Google Scholar] [CrossRef] [PubMed]
- Younis, N.N.; Soran, H.; Pemberton, P.; Charlton-Menys, V.; Elseweidy, M.M.; Durrington, P.N. Small dense LDL is more susceptible to glycation than more buoyant LDL in Type 2 diabetes. Clin. Sci. 2013, 124, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, E.A.; Myasoedova, V.A.; Melnichenko, A.A.; Grechko, A.V.; Orekhov, A.N. Small Dense Low-Density Lipoprotein as Biomarker for Atherosclerotic Diseases. Oxidative Med. Cell Longev. 2017, 2017, 1273042. [Google Scholar] [CrossRef]
- White, C.R.; Anantharamaiah, G.M. Cholesterol reduction and macrophage function: Role of paraoxonases. Curr. Opin. Lipidol. 2017, 28, 397–402. [Google Scholar] [CrossRef] [PubMed]
- Luyster, F.S.; Kip, K.E.; Drumheller, O.J.; Rice, T.B.; Edmundowicz, D.; Matthews, K.; Reis, S.E.; Strollo, P.J., Jr. Sleep apnea is related to the atherogenic phenotype, lipoprotein subclass B. J. Clin. Sleep Med. 2012, 8, 155–161. [Google Scholar] [CrossRef][Green Version]
- Sopkova, Z.; Berneis, K.; Rizzo, M.; Spinas, G.A.; Dorkova, Z.; Tisko, R.; Tkacova, R. Size and Subclasses of Low-Density Lipoproteins in Patients With Obstructive Sleep Apnea. Angiology 2012, 63, 617–621. [Google Scholar] [CrossRef]
- Liu, A.; Cardell, J.; Ariel, D.; Lamendola, C.; Abbasi, F.; Kim, S.H.; Holmes, T.H.; Tomasso, V.; Mojaddidi, H.; Grove, K.; et al. Abnormalities of lipoprotein concentrations in obstructive sleep apnea are related to insulin resistance. Sleep 2015, 38, 793–799. [Google Scholar] [CrossRef]
- Feres, M.C.; Fonseca, F.A.; Cintra, F.D.; Mello-Fujita, L.; de Souza, A.L.; De Martino, M.C.; Tufik, S.; Poyares, D. An assessment of oxidized LDL in the lipid profiles of patients with obstructive sleep apnea and its association with both hypertension and dyslipidemia, and the impact of treatment with CPAP. Atherosclerosis 2015, 241, 342–349. [Google Scholar] [CrossRef]
- Tan, K.C.; Chow, W.S.; Lam, J.C.; Lam, B.; Wong, W.K.; Tam, S.; Ip, M.S. HDL dysfunction in obstructive sleep apnea. Atherosclerosis 2006, 184, 377–382. [Google Scholar] [CrossRef]
- Kizawa, T.; Nakamura, Y.; Takahashi, S.; Sakurai, S.; Yamauchi, K.; Inoue, H. Pathogenic role of angiotensin II and oxidised LDL in obstructive sleep apnoea. Eur. Respir. J. 2009, 34, 1390–1398. [Google Scholar] [CrossRef] [PubMed]
- Tauman, R.; Lavie, L.; Greenfeld, M.; Sivan, Y. Oxidative stress in children with obstructive sleep apnea syndrome. J. Clin. Sleep Med. 2014, 10, 677–681. [Google Scholar] [CrossRef]
- Lee, S.D.; Ju, G.; Choi, J.A.; Kim, J.W.; Yoon, I.Y. The association of oxidative stress with central obesity in obstructive sleep apnea. Sleep Breath 2012, 16, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Svatikova, A.; Wolk, R.; Lerman, L.O.; Juncos, L.A.; Greene, E.L.; McConnell, J.P.; Somers, V.K. Oxidative stress in obstructive sleep apnoea. Eur. Heart J. 2005, 26, 2435–2439. [Google Scholar] [CrossRef] [PubMed]
- Fadaei, R.; Safari-Faramani, R.; Rezaei, M.; Ahmadi, R.; Rostampour, M.; Moradi, N.; Khazaie, H. Circulating levels of oxidized low-density lipoprotein in patients with obstructive sleep apnea: A systematic review and meta-analysis. Sleep Breath 2020, 24, 809–815. [Google Scholar] [CrossRef] [PubMed]
- Undurti, A.; Huang, Y.; Lupica, J.A.; Smith, J.D.; DiDonato, J.A.; Hazen, S.L. Modification of high density lipoprotein by myeloperoxidase generates a pro-inflammatory particle. J. Biol. Chem. 2009, 284, 30825–30835. [Google Scholar] [CrossRef]
- Rosenson, R.S.; Brewer, H.B., Jr.; Ansell, B.J.; Barter, P.; Chapman, M.J.; Heinecke, J.W.; Kontush, A.; Tall, A.R.; Webb, N.R. Dysfunctional HDL and atherosclerotic cardiovascular disease. Nat. Rev. Cardiol. 2016, 13, 48–60. [Google Scholar] [CrossRef]
- Cabana, V.G.; Lukens, J.R.; Rice, K.S.; Hawkins, T.J.; Getz, G.S. HDL content and composition in acute phase response in three species: Triglyceride enrichment of HDL a factor in its decrease. J. Lipid Res. 1996, 37, 2662–2674. [Google Scholar] [CrossRef]
- Song, D.; Fang, G.; Mao, S.Z.; Ye, X.; Liu, G.; Gong, Y.; Liu, S.F. Chronic intermittent hypoxia induces atherosclerosis by NF-κB-dependent mechanisms. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2012, 1822, 1650–1659. [Google Scholar] [CrossRef]
- Chen, M.; Li, W.; Wang, N.; Zhu, Y.; Wang, X. ROS and NF-kappaB but not LXR mediate IL-1beta signaling for the downregulation of ATP-binding cassette transporter A1. Am. J. Physiol. Cell Physiol. 2007, 292, C1493–C1501. [Google Scholar] [CrossRef]
- Hanikoglu, F.; Huseyinoglu, N.; Ozben, S.; Cort, A.; Ozdem, S.; Ozben, T. Increased plasma soluble tumor necrosis factor receptor-1 and myeloperoxidase activity in patients with obstructive sleep apnea syndrome. Int. J. Neurosci. 2015, 125, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Bergt, C.; Pennathur, S.; Fu, X.; Byun, J.; O’Brien, K.; McDonald, T.O.; Singh, P.; Anantharamaiah, G.M.; Chait, A.; Brunzell, J.; et al. The myeloperoxidase product hypochlorous acid oxidizes HDL in the human artery wall and impairs ABCA1-dependent cholesterol transport. Proc. Natl. Acad. Sci. USA 2004, 101, 13032–13037. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Settle, M.; Brubaker, G.; Schmitt, D.; Hazen, S.L.; Smith, J.D.; Kinter, M. Localization of nitration and chlorination sites on apolipoprotein A-I catalyzed by myeloperoxidase in human atheroma and associated oxidative impairment in ABCA1-dependent cholesterol efflux from macrophages. J. Biol. Chem. 2005, 280, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Ashraf, M.Z.; Zhang, L.; Kar, N.; Byzova, T.V.; Podrez, E.A. Cross-linking modifications of HDL apoproteins by oxidized phospholipids: Structural characterization, in vivo detection, and functional implications. J. Biol. Chem. 2020, 295, 1973–1984. [Google Scholar] [CrossRef]
- Pruzanski, W.; Stefanski, E.; de Beer, F.C.; de Beer, M.C.; Vadas, P.; Ravandi, A.; Kuksis, A. Lipoproteins are substrates for human secretory group IIA phospholipase A2: Preferential hydrolysis of acute phase HDL. J. Lipid Res. 1998, 39, 2150–2160. [Google Scholar] [CrossRef]
- Kar, S.; Patel, M.A.; Tripathy, R.K.; Bajaj, P.; Suvarnakar, U.V.; Pande, A.H. Oxidized phospholipid content destabilizes the structure of reconstituted high density lipoprotein particles and changes their function. Biochim. Biophys. Acta 2012, 1821, 1200–1210. [Google Scholar] [CrossRef]
- Jaouad, L.; Milochevitch, C.; Khalil, A. PON1 paraoxonase activity is reduced during HDL oxidation and is an indicator of HDL antioxidant capacity. Free Radic. Res. 2003, 37, 77–83. [Google Scholar] [CrossRef]
- Lavie, L.; Dyugovskaya, L.; Golan-Shany, O.; Lavie, P. Heat-shock protein 70: Expression in monocytes of patients with sleep apnoea and association with oxidative stress and tumour necrosis factor-alpha. J. Sleep Res. 2010, 19, 139–147. [Google Scholar] [CrossRef]
- Kotani, K.; Kimura, S.; Tsuzaki, K.; Sakane, N.; Komada, I.; Schulze, J.; Gugliucci, A. Reduced paraoxonase 1/arylesterase activity and its post-therapeutic increase in obstructive sleep apnea syndrome: A preliminary study. Clin. Chim. Acta. 2008, 395, 184–185. [Google Scholar] [CrossRef]
- Lavie, L.; Vishnevsky, A.; Lavie, P. Evidence for lipid peroxidation in obstructive sleep apnea. Sleep 2004, 27, 123–128. [Google Scholar]
- Baysal, E.; Taysi, S.; Aksoy, N.; Uyar, M.; Celenk, F.; Karatas, Z.A.; Tarakcioglu, M.; Bilinç, H.; Mumbuç, S.; Kanlikama, M. Serum paraoxonase, arylesterase activity and oxidative status in patients with obstructive sleep apnea syndrome (OSAS). Eur. Rev. Med. Pharm. Sci. 2012, 16, 770–774. [Google Scholar]
- Płóciniczak, A.; Baszczuk, A.; Ludziejewska, A.; Winiarska, H.; Michalak, S.; Kasprzak, G.; Formanowicz, D.; Cofta, S.; Wysocka, E. Paraoxonase 1 gene L55M polymorphism and paraoxonase 1 activity in obstructive sleep apnea patients. Adv. Exp. Med. Biol. 2019, 1150, 17–24. [Google Scholar] [PubMed]
- Shao, B.; Cavigiolio, G.; Brot, N.; Oda, M.N.; Heinecke, J.W. Methionine oxidation impairs reverse cholesterol transport by apolipoprotein A-I. Proc. Natl. Acad. Sci. USA 2008, 105, 12224–12229. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; DiDonato, J.A.; Levison, B.S.; Schmitt, D.; Li, L.; Wu, Y.; Buffa, J.; Kim, T.; Gerstenecker, G.S.; Gu, X.; et al. An abundant dysfunctional apolipoprotein A1 in human atheroma. Nat. Med. 2014, 20, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Svatikova, A.; Wolk, R.; Shamsuzzaman, A.S.; Kara, T.; Olson, E.J.; Somers, V.K. Serum amyloid a in obstructive sleep apnea. Circulation 2003, 108, 1451–1454. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, A.S.; de Beer, M.C.; Steel, D.M.; Rits, M.; Lelias, J.M.; Lane, W.S.; de Beer, F.C. Identification of novel members of the serum amyloid A protein superfamily as constitutive apolipoproteins of high density lipoprotein. J Biol. Chem. 1992, 267, 3862–3867. [Google Scholar] [CrossRef]
- Wroblewski, J.M.; Jahangiri, A.; Ji, A.; de Beer, F.C.; van der Westhuyzen, D.R.; Webb, N.R. Nascent HDL formation by hepatocytes is reduced by the concerted action of serum amyloid A and endothelial lipase. J. Lipid Res. 2011, 52, 2255–2261. [Google Scholar] [CrossRef]
- Nicholls, S.J.; Lundman, P.; Harmer, J.A.; Cutri, B.; Griffiths, K.A.; Rye, K.A.; Barter, P.J.; Celermajer, D.S. Consumption of saturated fat impairs the anti-in.n.nflamma.atory properties of high-density lipoproteins and endothelial function. J. Am. Coll. Cardiol. 2006, 48, 715–720. [Google Scholar] [CrossRef]
- Meszaros, M.; Horvath, P.; Kis, A.; Kunos, L.; Tarnoki, A.D.; Tarnoki, D.L.; Lazar, Z.; Bikov, A. Circulating levels of clusterin and complement factor H in patients with obstructive sleep apnea. Biomark. Med. 2021, 15, 323–330. [Google Scholar] [CrossRef]
- Peng, Y.; Zhou, L.; Cao, Y.; Chen, P.; Chen, Y.; Zong, D.; Ouyang, R. Relation between serum leptin levels, lipid profiles and neurocognitive deficits in Chinese OSAHS patients. Int. J. Neurosci. 2017, 127, 981–987. [Google Scholar] [CrossRef]
- Roche, F.; Sforza, E.; Pichot, V.; Maudoux, D.; Garcin, A.; Celle, S.; Picard-Kossovsky, M.; Gaspoz, J.M.; Barthélémy, J.C. Obstructive sleep apnoea/hypopnea influences high-density lipoprotein cholesterol in the elderly. Sleep Med. 2009, 10, 882–886. [Google Scholar] [CrossRef] [PubMed]
- Drager, L.F.; Lopes, H.F.; Maki-Nunes, C.; Trombetta, I.C.; Toschi-Dias, E.; Alves, M.J.; Fraga, R.F.; Jun, J.C.; Negrão, C.E.; Krieger, E.M.; et al. The impact of obstructive sleep apnea on metabolic and inflammatory markers in consecutive patients with metabolic syndrome. PLoS ONE 2010, 5, e12065. [Google Scholar] [CrossRef] [PubMed]
- Cai, G.; Shi, G.; Xue, S.; Lu, W. The atherogenic index of plasma is a strong and independent predictor for coronary artery disease in the Chinese Han population. Medicine 2017, 96, e8058. [Google Scholar] [CrossRef]
- Shimizu, Y.; Yoshimine, H.; Nagayoshi, M.; Kadota, K.; Takahashi, K.; Izumino, K.; Inoue, K.; Maeda, T. Serum triglyceride levels in relation to high-density lipoprotein cholesterol (TG-HDL) ratios as an efficient tool to estimate the risk of sleep apnea syndrome in non-overweight Japanese men. Environ. Health Prev. Med. 2016, 21, 321–326. [Google Scholar] [CrossRef][Green Version]
- Wu, W.-T.; Tsai, S.-S.; Shih, T.-S.; Lin, M.-H.; Chou, T.-C.; Ting, H.; Wu, T.-N.; Liou, S.-H. The association between obstructive sleep apnea and metabolic markers and lipid profiles. PLoS ONE 2015, 10, e0130279. [Google Scholar] [CrossRef][Green Version]
- Cao, B.; Fan, Z.; Zhang, Y.; Li, T. Independent association of severity of obstructive sleep apnea with lipid metabolism of atherogenic index of plasma (AIP) and apoB/apoAI ratio. Sleep Breath 2020, 24, 1507–1513. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.O.E.; Guimarães, T.M.; Luz, G.P.; Coelho, G.; Badke, L.; Almeida, I.R.; Millani-Carneiro, A.; Tufik, S.; Bittencourt, L.; Togeiro, S.M. Metabolic profile in patients with mild obstructive sleep apnea. Metab. Syndr. Relat. Disord. 2018, 16, 6–12. [Google Scholar] [CrossRef]
- Bikov, A.; Meszaros, M.; Kunos, L.; Negru, A.G.; Frent, S.M.; Mihaicuta, S. Atherogenic Index of Plasma in Obstructive Sleep Apnoea. J. Clin. Med. 2021, 10, 417. [Google Scholar] [CrossRef]
- Bays, H.E.; González-Campoy, J.M.; Bray, G.A.; Kitabchi, A.E.; Bergman, D.A.; Schorr, A.B.; Rodbard, H.W.; Henry, R.R. Pathogenic potential of adipose tissue and metabolic consequences of adipocyte hypertrophy and increased visceral adiposity. Expert Rev. Cardiovasc. Ther. 2008, 6, 343–368. [Google Scholar] [CrossRef]
- Haemmerle, G.; Lass, A.; Zimmermann, R.; Gorkiewicz, G.; Meyer, C.; Rozman, J.; Heldmaier, G.; Maier, R.; Theussl, C.; Eder, S.; et al. Defective lipolysis and altered energy metabolism in mice lacking adipose triglyceride lipase. Science 2006, 312, 734–737. [Google Scholar] [CrossRef]
- Reynisdottir, S.; Dauzats, M.; Thörne, A.; Langin, D. Comparison of hormone-sensitive lipase activity in visceral and subcutaneous human adipose tissue. J. Clin. Endocrinol. Metab. 1997, 82, 4162–4166. [Google Scholar] [CrossRef] [PubMed]
- Fredrikson, G.; Tornqvist, H.; Belfrage, P. Hormone-sensitive lipase and monoacylglycerol lipase are both required for complete degradation of adipocyte triacylglycerol. Biochim. Biophys. Acta 1986, 876, 288–293. [Google Scholar] [CrossRef]
- Chalacheva, P.; Thum, J.; Yokoe, T.; O’Donnell, C.P.; Khoo, M.C. Development of autonomic dysfunction with intermittent hypoxia in a lean murine model. Respir. Physiol. Neurobiol. 2013, 188, 143–151. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Carpéné, C.; Bousquet-Mélou, A.; Galitzky, J.; Berlan, M.; Lafontan, M. Lipolytic effects of beta 1-, beta 2-, and beta 3-adrenergic agonists in white adipose tissue of mammals. Ann. N. Y. Acad. Sci. 1998, 839, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Netchitaïlo, M.; Fabre, F.; Briançon, A.; Belaidi-Corsat, E.; Arnaud, C.; Borel, A.-L.; Lévy, P.; Pépin, J.-L.; Tamisier, R. Two weeks of intermittent hypoxic exposure induce lipolysis at the fat tissue level in healthy human subjects. Eur. Respir. J. 2017, 50, PA2968. [Google Scholar]
- Weiszenstein, M.; Shimoda, L.A.; Koc, M.; Seda, O.; Polak, J. Inhibition of Lipolysis Ameliorates Diabetic Phenotype in a Mouse Model of Obstructive Sleep Apnea. Am. J. Respir. Cell Mol. Biol. 2016, 55, 299–307. [Google Scholar] [CrossRef]
- Musutova, M.; Weiszenstein, M.; Koc, M.; Polak, J. Intermittent hypoxia stimulates lipolysis, but inhibits differentiation and de novo lipogenesis in 3T3-L1 cells. Metab. Syndr. Relat. Disord. 2020, 18, 146–153. [Google Scholar] [CrossRef]
- Krawczyk, S.A.; Haller, J.F.; Ferrante, T.; Zoeller, R.A.; Corkey, B.E. Reactive oxygen species facilitate translocation of hormone sensitive lipase to the lipid droplet during lipolysis in human differentiated adipocytes. PLoS ONE 2012, 7, e34904. [Google Scholar] [CrossRef]
- Wang, H.; Bell, M.; Sreenivasan, U.; Sreenevasan, U.; Hu, H.; Liu, J.; Dalen, K.; Londos, C.; Yamaguchi, T.; Rizzo, M.A.; et al. Unique regulation of adipose triglyceride lipase (ATGL) by perilipin 5, a lipid droplet-associated protein. J. Biol. Chem. 2011, 286, 15707–15715. [Google Scholar] [CrossRef]
- Ryden, M.; Dicker, A.; van Harmelen, V.; Hauner, H.; Brunnberg, M.; Perbeck, L.; Lonnqvist, F.; Arner, P. Mapping of early signaling events in tumor necrosis factor-alpha -mediated lipolysis in human fat cells. J. Biol. Chem. 2002, 277, 1085–1091. [Google Scholar] [CrossRef]
- van Hall, G.; Steensberg, A.; Sacchetti, M.; Fischer, C.; Keller, C.; Schjerling, P.; Hiscock, N.; Møller, K.; Saltin, B.; Febbraio, M.A.; et al. Interleukin-6 stimulates lipolysis and fat oxidation in humans. J. Clin. Endocrinol. Metab. 2003, 88, 3005–3010. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Song, Y.; Ning, P.; Zhang, L.; Wu, S.; Quan, J.; Li, Q. Association between tumor necrosis factor alpha and obstructive sleep apnea in adults: A meta-analysis update. BMC Pulm. Med. 2020, 20, 215. [Google Scholar] [CrossRef] [PubMed]
- Imani, M.M.; Sadeghi, M.; Khazaie, H.; Emami, M.; Sadeghi Bahmani, D.; Brand, S. Evaluation of Serum and Plasma Interleukin-6 Levels in Obstructive Sleep Apnea Syndrome: A Meta-Analysis and Meta-Regression. Front. Immunol. 2020, 11, 1343. [Google Scholar] [CrossRef] [PubMed]
- Briançon-Marjollet, A.; Monneret, D.; Henri, M.; Hazane-Puch, F.; Pepin, J.L.; Faure, P.; Godin-Ribuot, D. Endothelin regulates intermittent hypoxia-induced lipolytic remodelling of adipose tissue and phosphorylation of hormone-sensitive lipase. J. Physiol. 2016, 594, 1727–1740. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.J.; Sridhar, K.; Bernlohr, D.A.; Kraemer, F.B. Interaction of rat hormone-sensitive lipase with adipocyte lipid-binding protein. Proc. Natl. Acad. Sci. USA 1999, 96, 5528–5532. [Google Scholar] [CrossRef]
- Català, R.; Cabré, A.; Hernández-Flix, S.; Ferré, R.; Sangenís, S.; Plana, N.; Texidó, A.; Masana, L. Circulating FABP4 and FABP5 levels are differently linked to OSA severity and treatment. Sleep 2013, 36, 1831–1837. [Google Scholar] [CrossRef]
- Lam, D.C.; Xu, A.; Lam, K.S.; Lam, B.; Lam, J.C.; Lui, M.M.; Ip, M.S. Serum adipocyte-fatty acid binding protein level is elevated in severe OSA and correlates with insulin resistance. Eur. Respir. J. 2009, 33, 346–351. [Google Scholar] [CrossRef]
- Balci, M.M.; Arslan, U.; Firat, H.; Kocaoğlu, I.; Vural, M.G.; Balci, K.G.; Maden, O.; Gürbüz, O.A.; Ardiç, S.; Yeter, E. Serum levels of adipocyte fatty acid-binding protein are independently associated with left ventricular mass and myocardial performance index in obstructive sleep apnea syndrome. J. Investig. Med. 2012, 60, 1020–1026. [Google Scholar] [CrossRef]
- Hofer, P.; Boeszoermenyi, A.; Jaeger, D.; Feiler, U.; Arthanari, H.; Mayer, N.; Zehender, F.; Rechberger, G.; Oberer, M.; Zimmermann, R.; et al. Fatty Acid-binding Proteins Interact with Comparative Gene Identification-58 Linking Lipolysis with Lipid Ligand Shuttling *. J. Biol. Chem. 2015, 290, 18438–18453. [Google Scholar] [CrossRef]
- Gordon, E.S. Non-Esterified Fatty Acids in the Blood of Obese and Lean Subjects. Am. J. Clin. Nutr. 1960, 8, 740–747. [Google Scholar] [CrossRef]
- Frühbeck, G.; Gómez-Ambrosi, J.; Salvador, J. Leptin-induced lipolysis opposes the tonic inhibition of endogenous adenosine in white adipocytes. FASEB J. 2001, 15, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Wedellová, Z.; Dietrich, J.; Siklová-Vítková, M.; Kološtová, K.; Kováčiková, M.; Dušková, M.; Brož, J.; Vedral, T.; Stich, V.; Polák, J. Adiponectin inhibits spontaneous and catecholamine-induced lipolysis in human adipocytes of non-obese subjects through AMPK-dependent mechanisms. Physiol. Res. 2011, 60, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Kapsimalis, F.; Varouchakis, G.; Manousaki, A.; Daskas, S.; Nikita, D.; Kryger, M.; Gourgoulianis, K. Association of sleep apnea severity and obesity with insulin resistance, C-reactive protein, and leptin levels in male patients with obstructive sleep apnea. Lung 2008, 186, 209–217. [Google Scholar] [CrossRef]
- Tokuda, F.; Sando, Y.; Matsui, H.; Koike, H.; Yokoyama, T. Serum levels of adipocytokines, adiponectin and leptin, in patients with obstructive sleep apnea syndrome. Intern. Med. 2008, 47, 1843–1849. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ormazabal, V.; Nair, S.; Elfeky, O.; Aguayo, C.; Salomon, C.; Zuñiga, F.A. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc. Diabetol. 2018, 17, 122. [Google Scholar] [CrossRef]
- Pasarica, M.; Rood, J.; Ravussin, E.; Schwarz, J.-M.; Smith, S.R.; Redman, L.M. Reduced oxygenation in human obese adipose tissue is associated with impaired insulin suppression of lipolysis. J. Clin. Endocrinol. Metab. 2010, 95, 4052–4055. [Google Scholar] [CrossRef]
- Rooney, K.; Trayhurn, P. Lactate and the GPR81 receptor in metabolic regulation: Implications for adipose tissue function and fatty acid utilisation by muscle during exercise. Br. J. Nutr. 2011, 106, 1310–1316. [Google Scholar] [CrossRef] [PubMed]
- Späth-Schwalbe, E.; Gofferje, M.; Kern, W.; Born, J.; Fehm, H.L. Sleep disruption alters nocturnal ACTH and cortisol secretory patterns. Biol. Psychiatry 1991, 29, 575–584. [Google Scholar] [CrossRef]
- Brindley, D.N.; McCann, B.S.; Niaura, R.; Stoney, C.M.; Suarez, E.C. Stress and lipoprotein metabolism: M odulators and mechanisms. Metabolism 1993, 42, 3–15. [Google Scholar] [CrossRef]
- Young, T.; Palta, M.; Dempsey, J.; Skatrud, J.; Weber, S.; Badr, S. The occurrence of sleep-disordered breathing among middle-aged adults. N. Engl. J. Med. 1993, 328, 1230–1235. [Google Scholar] [CrossRef]
- Peppard, P.E.; Young, T.; Palta, M.; Dempsey, J.; Skatrud, J. Longitudinal study of moderate weight change and sleep-disordered breathing. Jama 2000, 284, 3015–3021. [Google Scholar] [CrossRef] [PubMed]
- Ryan, S.; Arnaud, C.; Fitzpatrick, S.F.; Gaucher, J.; Tamisier, R.; Pépin, J.-L. Adipose tissue as a key player in obstructive sleep apnoea. Eur. Respir. Rev. 2019, 28, 190006. [Google Scholar] [CrossRef] [PubMed]
- Jo, J.; Gavrilova, O.; Pack, S.; Jou, W.; Mullen, S.; Sumner, A.E.; Cushman, S.W.; Periwal, V. Hypertrophy and/or hyperplasia: Dynamics of adipose tissue growth. PLoS Comput. Biol. 2009, 5, e1000324. [Google Scholar] [CrossRef] [PubMed]
- Cildir, G.; Akıncılar, S.C.; Tergaonkar, V. Chronic adipose tissue inflammation: All immune cells on the stage. Trends Mol. Med. 2013, 19, 487–500. [Google Scholar] [CrossRef]
- Ryan, S. Adipose tissue inflammation by intermittent hypoxia: Mechanistic link between obstructive sleep apnoea and metabolic dysfunction. J. Physiol. 2017, 595, 2423–2430. [Google Scholar] [CrossRef]
- Poulain, L.; Thomas, A.; Rieusset, J.; Casteilla, L.; Levy, P.; Arnaud, C.; Dematteis, M. Visceral white fat remodelling contributes to intermittent hypoxia-induced atherogenesis. Eur. Respir. J. 2014, 43, 513. [Google Scholar] [CrossRef]
- Poulain, L.; Richard, V.; Lévy, P.; Dematteis, M.; Arnaud, C. Toll-like receptor-4 mediated inflammation is involved in the cardiometabolic alterations induced by intermittent hypoxia. Mediat. Inflamm. 2015, 2015, 620258. [Google Scholar] [CrossRef]
- Briançon-Marjollet, A.; Pépin, J.L.; Weiss, J.W.; Lévy, P.; Tamisier, R. Intermittent hypoxia upregulates serum VEGF. Sleep Med. 2014, 15, 1425–1426. [Google Scholar] [CrossRef]
- Murphy, A.M.; Thomas, A.; Crinion, S.J.; Kent, B.D.; Tambuwala, M.M.; Fabre, A.; Pepin, J.L.; Roche, H.M.; Arnaud, C.; Ryan, S. Intermittent hypoxia in obstructive sleep apnoea mediates insulin resistance through adipose tissue inflammation. Eur. Respir. J. 2017, 49, 1601731. [Google Scholar] [CrossRef]
- Kanda, H.; Tateya, S.; Tamori, Y.; Kotani, K.; Hiasa, K.; Kitazawa, R.; Kitazawa, S.; Miyachi, H.; Maeda, S.; Egashira, K.; et al. MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J. Clin. Investig. 2006, 116, 1494–1505. [Google Scholar] [CrossRef]
- Kim, J.; Lee, C.H.; Park, C.S.; Kim, B.G.; Kim, S.W.; Cho, J.H. Plasma levels of MCP-1 and adiponectin in obstructive sleep apnea syndrome. Arch. Otolaryngol. Head Neck Surg. 2010, 136, 896–899. [Google Scholar] [CrossRef][Green Version]
- Fanfulla, F.; Rotondi, M.; Morrone, E.; Coperchini, F.; Lodigiani, S.; Trentin, R.; Maccabruni, V.; Chiovato, L. Sleep hypoxia and not obesity is the main determinant of the increasing monocyte chemoattractant protein-1 (MCP-1) in patients with obstructive sleep apnoea. ERJ Open Res. 2017, 3, P70. [Google Scholar]
- Taylor, C.T.; Kent, B.D.; Crinion, S.J.; McNicholas, W.T.; Ryan, S. Human adipocytes are highly sensitive to intermittent hypoxia induced NF-kappaB activity and subsequent inflammatory gene expression. Biochem. Biophys Res. Commun. 2014, 447, 660–665. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Harris, T.B.; Hue, T.; Miljkovic, I.; Satterfield, S.; de Rekeneire, N.; Mehta, M.; Sahyoun, N.R. Hyperleptinemia, adiposity, and risk of metabolic syndrome in older adults. J. Nutr. Metab. 2013, 2013, 327079. [Google Scholar] [CrossRef] [PubMed]
- Welsh, P.; Murray, H.M.; Buckley, B.M.; de Craen, A.J.M.; Ford, I.; Jukema, J.W.; Macfarlane, P.W.; Packard, C.J.; Stott, D.J.; Westendorp, R.G.J.; et al. Leptin predicts diabetes but not cardiovascular disease: Results from a large prospective study in an elderly population. Diabetes Care 2009, 32, 308–310. [Google Scholar] [CrossRef] [PubMed]
- Sierra-Johnson, J.; Romero-Corral, A.; Lopez-Jimenez, F.; Gami, A.S.; Sert Kuniyoshi, F.H.; Wolk, R.; Somers, V.K. Relation of increased leptin concentrations to history of myocardial infarction and stroke in the United States population. Am. J. Cardiol. 2007, 100, 234–239. [Google Scholar] [CrossRef]
- Ku, I.A.; Farzaneh-Far, R.; Vittinghoff, E.; Zhang, M.H.; Na, B.; Whooley, M.A. Association of low leptin with cardiovascular events and mortality in patients with stable coronary artery disease: The Heart and Soul Study. Atherosclerosis 2011, 217, 503–508. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schäfer, H.; Pauleit, D.; Sudhop, T.; Gouni-Berthold, I.; Ewig, S.; Berthold, H.K. Body fat distribution, serum leptin, and cardiovascular risk factors in men with obstructive sleep apnea. Chest 2002, 122, 829–839. [Google Scholar] [CrossRef]
- Ozturk, L.; Unal, M.; Tamer, L.; Celikoglu, F. The association of the severity of obstructive sleep apnea with plasma leptin levels. Arch. Otolaryngol. Head Neck Surg. 2003, 129, 538–540. [Google Scholar] [CrossRef]
- Tatsumi, K.; Kasahara, Y.; Kurosu, K.; Tanabe, N.; Takiguchi, Y.; Kuriyama, T. Sleep oxygen desaturation and circulating leptin in obstructive sleep apnea-hyp.p.p.popnea syndrome. Chest 2005, 127, 716–721. [Google Scholar] [CrossRef]
- McArdle, N.; Hillman, D.; Beilin, L.; Watts, G. Metabolic risk factors for vascular disease in obstructive sleep apnea: A matched controlled study. Am. J. Respir. Crit. Care Med. 2007, 175, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Hirotsu, C.; Albuquerque, R.G.; Nogueira, H.; Hachul, H.; Bittencourt, L.; Tufik, S.; Andersen, M.L. The relationship between sleep apnea, metabolic dysfunction and inflammation: The gender influence. Brain Behav. Immun. 2017, 59, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Arnardottir, E.S.; Maislin, G.; Jackson, N.; Schwab, R.J.; Benediktsdottir, B.; Teff, K.; Juliusson, S.; Pack, A.I.; Gislason, T. The role of obesity, different fat compartments and sleep apnea severity in circulating leptin levels: The Icelandic Sleep Apnea Cohort study. Int. J. Obes. 2013, 37, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Knight, Z.A.; Hannan, K.S.; Greenberg, M.L.; Friedman, J.M. Hyperleptinemia is required for the development of leptin resistance. PLoS ONE 2010, 5, e11376. [Google Scholar] [CrossRef]
- Berger, S.; Polotsky, V.Y. Leptin and leptin resistance in the pathogenesis of obstructive sleep apnea: A possible link to oxidative stress and cardiovascular complications. Oxidative Med. Cell Longev. 2018, 2018, 5137947. [Google Scholar] [CrossRef]
- Ciriello, J.; Moreau, J.M.; Caverson, M.M.; Moranis, R. Leptin: A Potential Link Between Obstructive Sleep Apnea and Obesity. Front. Physiol. 2022, 12, 767318. [Google Scholar] [CrossRef]
- Pan, W.; Kastin, A.J. Leptin: A biomarker for sleep disorders? Sleep Med. Rev. 2014, 18, 283–290. [Google Scholar] [CrossRef]
- Gonnissen, H.K.; Hursel, R.; Rutters, F.; Martens, E.A.; Westerterp-Plantenga, M.S. Effects of sleep fragmentation on appetite and related hormone concentrations over 24 h in healthy men. Br. J. Nutr. 2013, 109, 748–756. [Google Scholar] [CrossRef]
- Framnes, S.N.; Arble, D.M. The Bidirectional Relationship Between Obstructive Sleep Apnea and Metabolic Disease. Front. Endocrinol. 2018, 9, 440. [Google Scholar] [CrossRef]
- Grosfeld, A.; Andre, J.; Hauguel-De Mouzon, S.; Berra, E.; Pouyssegur, J.; Guerre-Millo, M. Hypoxia-inducible factor 1 transactivates the human leptin gene promoter. J. Biol. Chem. 2002, 277, 42953–42957. [Google Scholar] [CrossRef]
- Gallardo, N.; Bonzón-Kulichenko, E.; Fernández-Agulló, T.; Moltó, E.; Gómez-Alonso, S.; Blanco, P.; Carrascosa, J.M.; Ros, M.; Andrés, A. Tissue-specific effects of central leptin on the expression of genes involved in lipid metabolism in liver and white adipose tissue. Endocrinology 2007, 148, 5604–5610. [Google Scholar] [CrossRef] [PubMed]
- Brabant, G.; Müller, G.; Horn, R.; Anderwald, C.; Roden, M.; Nave, H. Hepatic leptin signaling in obesity. FASEB J. 2005, 19, 1048–1050. [Google Scholar] [CrossRef] [PubMed]
- Sertogullarindan, B.; Komuroglu, A.U.; Ucler, R.; Gunbatar, H.; Sunnetcioglu, A.; Cokluk, E. Betatrophin association with serum triglyceride levels in obstructive sleep apnea patients. Ann. Thorac. Med. 2019, 14, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Morawietz, H.; Bornstein, S.R. Leptin, Endothelin, NADPH Oxidase, and Heart Failure. Hypertension 2006, 47, e20–e21. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pilz, S.; Horejsi, R.; Möller, R.; Almer, G.; Scharnagl, H.; Stojakovic, T.; Dimitrova, R.; Weihrauch, G.; Borkenstein, M.; Maerz, W.; et al. Early atherosclerosis in obese juveniles is associated with low serum levels of adiponectin. J. Clin. Endocrinol. Metab. 2005, 90, 4792–4796. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Kim, C.; Ding, E.L.; Townsend, M.K.; Lipsitz, L.A. Adiponectin levels and the risk of hypertension: A systematic review and meta-analysis. Hypertension 2013, 62, 27–32. [Google Scholar] [CrossRef]
- Lu, M.; Fang, F.; Wang, Z.; Wei, P.; Hu, C.; Wei, Y. Association between serum/plasma levels of adiponectin and obstructive sleep apnea hypopnea syndrome: A meta-analysis. Lipids Health Dis. 2019, 18, 30. [Google Scholar] [CrossRef]
- Domagała-Kulawik, J.; Osińska, I.; Piechuta, A.; Bielicki, P.; Skirecki, T. T, B, and NKT Cells in Systemic Inflammation in Obstructive Sleep Apnoea. Mediat. Inflamm. 2015, 2015, 161579. [Google Scholar] [CrossRef]
- Zhang, D.-M.; Pang, X.-L.; Huang, R.; Gong, F.-Y.; Zhong, X.; Xiao, Y. Adiponectin, Omentin, Ghrelin, and Visfatin Levels in Obese Patients with Severe Obstructive Sleep Apnea. Biomed. Res. Int. 2018, 2018, 3410135. [Google Scholar] [CrossRef]
- Gonzalez, F.J.; Xie, C.; Jiang, C. The role of hypoxia-inducible factors in metabolic diseases. Nat. Rev. Endocrinol. 2018, 15, 21–32. [Google Scholar] [CrossRef]
- Oku, H.; Matsuura, F.; Koseki, M.; Sandoval, J.C.; Yuasa-Kawase, M.; Tsubakio-Yamamoto, K.; Masuda, D.; Maeda, N.; Ohama, T.; Ishigami, M.; et al. Adiponectin deficiency suppresses ABCA1 expression and ApoA-I synthesis in the liver. FEBS Lett. 2007, 581, 5029–5033. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, F.; Oku, H.; Koseki, M.; Sandoval, J.C.; Yuasa-Kawase, M.; Tsubakio-Yamamoto, K.; Masuda, D.; Maeda, N.; Tsujii, K.; Ishigami, M.; et al. Adiponectin accelerates reverse cholesterol transport by increasing high density lipoprotein assembly in the liver. Biochem. Biophys. Res. Commun. 2007, 358, 1091–1095. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, M.; Maruoka, S.; Katayose, S. Decreased plasma adiponectin concentrations in women with dyslipidemia. J. Clin. Endocrinol. Metab. 2002, 87, 2764–2769. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, J.; Kusunoki, M.; Murase, Y.; Kawashiri, M.; Higashikata, T.; Miwa, K.; Katsuda, S.; Takata, M.; Asano, A.; Nohara, A.; et al. Relationship of lipoprotein lipase and hepatic triacylglycerol lipase activity to serum adiponectin levels in Japanese hyperlipidemic men. Horm. Metab. Res. 2005, 37, 505–509. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.; Zou, C.; van der Westhuyzen, D.R.; Shao, J. Adiponectin reduces plasma triglyceride by increasing VLDL triglyceride catabolism. Diabetes 2008, 57, 1824–1833. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, H.; Hirowatari, Y.; Kurosawa, H.; Tada, N. Implications of decreased serum adiponectin for type IIb hyperlipidaemia and increased cholesterol levels of very-low-density lipoprotein in type II diabetic patients. Clin. Sci. 2005, 109, 297–302. [Google Scholar] [CrossRef]
- Møller, N.; Gjedsted, J.; Gormsen, L.; Fuglsang, J.; Djurhuus, C. Effects of growth hormone on lipid metabolism in humans. Growth Horm. IGF Res. 2003, 13 (Suppl. SA), S18–S21. [Google Scholar] [CrossRef]
- Brindley, D.N. Role of glucocorticoids and fatty acids in the impairment of lipid metabolism observed in the metabolic syndrome. Int. J. Obes. Relat. Metab. Disord. 1995, 19 (Suppl. S1), S69–S75. [Google Scholar]
- Williamson, D.H. Role of insulin in the integration of lipid metabolism in mammalian tissues. Biochem. Soc. Trans. 1989, 17, 37–40. [Google Scholar] [CrossRef]
- Christ, E.R.; Cummings, M.H.; Russell-Jones, D.L. Dyslipidaemia in Adult Growth Hormone (GH) Deficiency and the Effect of GH Replacement Therapy: A Review. Trends Endocrinol. Metab. 1998, 9, 200–206. [Google Scholar] [CrossRef]
- Lanfranco, F.; Motta, G.; Minetto, M.A.; Ghigo, E.; Maccario, M. Growth hormone/insulin-like growth factor-I axis in obstructive sleep apnea syndrome: An update. J. Endocrinol. Investig. 2010, 33, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Whitworth, J.A.; Williamson, P.M.; Mangos, G.; Kelly, J.J. Cardiovascular consequences of cortisol excess. Vasc. Health Risk Manag. 2005, 1, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Kritikou, I.; Basta, M.; Vgontzas, A.N.; Pejovic, S.; Fernandez-Mendoza, J.; Liao, D.; Bixler, E.O.; Gaines, J.; Chrousos, G.P. Sleep apnoea and the hypothalamic–pituitary–adrenal axis in men and women: Effects of continuous positive airway pressure. Eur. Respir. J. 2016, 47, 531. [Google Scholar] [CrossRef] [PubMed]
- Somers, V.K.; Dyken, M.E.; Mark, A.L.; Abboud, F.M. Sympathetic-nerve activity during sleep in normal subjects. N. Engl. J. Med. 1993, 328, 303–307. [Google Scholar] [CrossRef]
- Spiegel, K.; Leproult, R.; Van Cauter, E. Impact of sleep debt on metabolic and endocrine function. Lancet 1999, 354, 1435–1439. [Google Scholar] [CrossRef]
- Takahashi, Y.; Kipnis, D.M.; Daughaday, W.H. Growth hormone secretion during sleep. J. Clin. Investig. 1968, 47, 2079–2090. [Google Scholar] [CrossRef]
- Alzoubaidi, M.; Mokhlesi, B.O.O. Obstructive sleep apnea during rapid eye movement sleep: Clinical relevance and therapeutic implications. Curr. Opin. Pulm. Med. 2016, 22, 545–554. [Google Scholar] [CrossRef]
- Bikov, A.; Lazar, Z.; Horvath, P.; Tarnoki, D.L.; Tarnoki, A.D.; Fesus, L.; Horvath, M.; Meszaros, M.; Losonczy, G.; Kunos, L. Association Between Serum Lipid Profile and Obstructive Respiratory Events During REM and Non-REM Sleep. Lung 2019, 197, 443–450. [Google Scholar] [CrossRef]
- Uchida, T.; Nishimura, A.; Kasai, T.; Kikuno, S.; Nagasawa, K.; Okubo, M.; Narui, K.; Mori, Y. Relationship between obstructive sleep apnoea during rapid eye movement sleep and metabolic syndrome parameters in patients with type 2 diabetes mellitus. Sleep Breath 2021, 25, 309–314. [Google Scholar] [CrossRef]
- Xu, H.; Xia, Y.; Li, X.; Qian, Y.; Zou, J.; Fang, F.; Yi, H.; Wu, H.; Guan, J.; Yin, S. Association between obstructive sleep apnea and lipid metabolism during REM and NREM sleep. J. Clin. Sleep Med. 2020, 16, 475–482. [Google Scholar] [CrossRef]
- Martínez-Cerón, E.; Casitas, R.; Galera, R.; Sánchez-Sánchez, B.; Zamarrón, E.; Garcia-Sanchez, A.; Jaureguizar, A.; Cubillos-Zapata, C.; Garcia-Rio, F. Contribution of sleep characteristics to the association between obstructive sleep apnea and dyslipidemia. Sleep Med. 2021, 84, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Coulet, F.; Nadaud, S.; Agrapart, M.; Soubrier, F. Identification of hypoxia-response element in the human endothelial nitric-oxide synthase gene promoter. J. Biol. Chem. 2003, 278, 46230–46240. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Yan, B.; Song, D.; Ye, X.; Liu, S.F. Chronic intermittent hypoxia down-regulates endothelial nitric oxide synthase expression by an NF-κB-dependent mechanism. Sleep Med. 2013, 14, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Kuzkaya, N.; Weissmann, N.; Harrison, D.G.; Dikalov, S. Interactions of peroxynitrite, tetrahydrobiopterin, ascorbic acid, and thiols: Implications for uncoupling endothelial nitric-oxide synthase. J. Biol. Chem. 2003, 278, 22546–22554. [Google Scholar] [CrossRef]
- Vásquez-Vivar, J.; Kalyanaraman, B.; Martásek, P.; Hogg, N.; Masters, B.S.; Karoui, H.; Tordo, P.; Pritchard, K.A. Superoxide generation by endothelial nitric oxide synthase: The influence of cofactors. Proc. Natl. Acad. Sci. USA 1998, 95, 9220–9225. [Google Scholar] [CrossRef]
- Jelic, S.; Padeletti, M.; Kawut, S.S.S.S.S.M.; Higgins, C.; Canfield, S.M.; Onat, D.; Colombo, P.C.; Basner, R.C.; Factor, P.; LeJemtel, T.H. Inflammation, oxidative stress, and repair capacity of the vascular endothelium in obstructive sleep apnea. Circulation 2008, 117, 2270–2278. [Google Scholar] [CrossRef]
- Lee, W.J.; Ou, H.C.; Hsu, W.C.; Chou, M.M.; Tseng, J.J.; Hsu, S.L.; Tsai, K.L.; Sheu, W.H. Ellagic acid inhibits oxidized LDL-mediated LOX-1 expression, ROS generation, and inflammation in human endothelial cells. J. Vasc. Surg. 2010, 52, 1290–1300. [Google Scholar] [CrossRef]
- Wang, W.; Hein, T.W.; Zhang, C.; Zawieja, D.C.; Liao, J.C.; Kuo, L. Oxidized low-density lipoprotein inhibits nitric oxide-mediated coronary arteriolar dilation by up-regulating endothelial arginase I. Microcirculation 2011, 18, 36–45. [Google Scholar] [CrossRef]
- Kattoor, A.J.; Pothineni, N.V.K.; Palagiri, D.; Mehta, J.L. Oxidative Stress in Atherosclerosis. Curr. Atheroscler. Rep. 2017, 19, 42. [Google Scholar] [CrossRef]
- Williams, A.; Scharf, S.M. Obstructive sleep apnea, cardiovascular disease, and inflammation--is NF-kappaB the key? Sleep Breath 2007, 11, 69–76. [Google Scholar] [CrossRef]
- Horváth, P.; Lázár, Z.; Gálffy, G.; Puskás, R.; Kunos, L.; Losonczy, G.; Mészáros, M.; Tárnoki Á, D.; Tárnoki, D.L.; Bikov, A. Circulating P-Selectin Glycoprotein Ligand 1 and P-Selectin Levels in Obstructive Sleep Apnea Patients. Lung 2020, 198, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Pak, V.M.; Keenan, B.T.; Jackson, N.; Grandner, M.A.; Maislin, G.; Teff, K.; Schwab, R.J.; Arnardottir, E.S.; Júlíusson, S.; Benediktsdottir, B.; et al. Adhesion molecule increases in sleep apnea: Beneficial effect of positive airway pressure and moderation by obesity. Int. J. Obes. 2015, 39, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Galkina, E.; Ley, K. Leukocyte influx in atherosclerosis. Curr. Drug Targets 2007, 8, 1239–1248. [Google Scholar] [CrossRef]
- Kattoor, A.J.; Goel, A.; Mehta, J.L. LOX-1: Regulation, Signaling and Its Role in Atherosclerosis. Antioxidants 2019, 8, 218. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Riek, A.E.; Weng, S.; Petty, M.; Kim, D.; Colonna, M.; Cella, M.; Bernal-Mizrachi, C. Endoplasmic reticulum stress controls M2 macrophage differentiation and foam cell formation. J. Biol. Chem. 2012, 287, 11629–11641. [Google Scholar] [CrossRef] [PubMed]
- Tontonoz, P.; Nagy, L.; Alvarez, J.G.; Thomazy, V.A.; Evans, R.M. PPARgamma promotes monocyte/macrophage differentiation and uptake of oxidized LDL. Cell 1998, 93, 241–252. [Google Scholar] [CrossRef]
- Tall, A.R.; Yvan-Charvet, L. Cholesterol, inflammation and innate immunity. Nat. Rev. Immunol. 2015, 15, 104–116. [Google Scholar] [CrossRef]
- Kobiyama, K.; Ley, K. Atherosclerosis: A Chronic Inflammatory Disease with an Autoimmune Component. Circ. Res. 2018, 123, 1118–1120. [Google Scholar] [CrossRef]
- Nofer, J.-R.; van der Giet, M.; Tölle, M.; Wolinska, I.; von Wnuck Lipinski, K.; Baba, H.A.; Tietge, U.J.; Gödecke, A.; Ishii, I.; Kleuser, B.; et al. HDL induces NO-dependent vasorelaxation via the lysophospholipid receptor S1P3. J. Clin. Investig. 2004, 113, 569–581. [Google Scholar] [CrossRef]
- Kimura, T.; Sato, K.; Kuwabara, A.; Tomura, H.; Ishiwara, M.; Kobayashi, I.; Ui, M.; Okajima, F. Sphingosine 1-phosphate may be a major component of plasma lipoproteins responsible for the cytoprotective actions in human umbilical vein endothelial cells. J. Biol. Chem. 2001, 276, 31780–31785. [Google Scholar] [CrossRef]
- Kontush, A.; Therond, P.; Zerrad, A.; Couturier, M.; Négre-Salvayre, A.; de Souza, J.A.; Chantepie, S.; Chapman, M.J. Preferential Sphingosine-1-Phosphate Enrichment and Sphingomyelin Depletion Are Key Features of Small Dense HDL3 Particles. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1843–1849. [Google Scholar] [CrossRef] [PubMed]
- Li, X.A.; Titlow, W.B.; Jackson, B.A.; Giltiay, N.; Nikolova-Karakashian, M.; Uittenbogaard, A.; Smart, E.J. High density lipoprotein binding to scavenger receptor, Class B, type I activates endothelial nitric-oxide synthase in a ceramide-dependent manner. J. Biol. Chem. 2002, 277, 11058–11063. [Google Scholar] [CrossRef] [PubMed]
- Nofer, J.R.; Brodde, M.F.; Kehrel, B.E. High-density lipoproteins, platelets and the pathogenesis of atherosclerosis. Clin. Exp. Pharm. Physiol. 2010, 37, 726–735. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Fillmore, J.J.; Chen, Y.Y.; Yu, C.; Moore, I.K.; Pypaert, M.; Lutz, E.P.; Kako, Y.; Velez-Carrasco, W.; Goldberg, I.J.; et al. Tissue-specific overexpression of lipoprotein lipase causes tissue-specific insulin resistance. Proc. Natl. Acad. Sci. USA 2001, 98, 7522–7527. [Google Scholar] [CrossRef] [PubMed]
- Boden, G. Obesity, insulin resistance and free fatty acids. Curr. Opin. Endocrinol. Diabetes Obes. 2011, 18, 139–143. [Google Scholar] [CrossRef]
- Patil Susheel, P.; Ayappa Indu, A.; Caples Sean, M.; Kimoff, R.J.; Patel Sanjay, R.; Harrod Christopher, G. Treatment of Adult Obstructive Sleep Apnea with Positive Airway Pressure: An American Academy of Sleep Medicine Clinical Practice Guideline. J. Clin. Sleep Med. 2019, 15, 335–343. [Google Scholar] [CrossRef]
- Ip, M.S.; Lam, K.S.; Ho, C.; Tsang, K.W.; Lam, W. Serum leptin and vascular risk factors in obstructive sleep apnea. Chest 2000, 118, 580–586. [Google Scholar] [CrossRef]
- Robinson, G.V.; Pepperell, J.C.; Segal, H.C.; Davies, R.J.; Stradling, J.R. Circulating cardiovascular risk factors in obstructive sleep apnoea: Data from randomised controlled trials. Thorax 2004, 59, 777–782. [Google Scholar] [CrossRef]
- Börgel, J.; Sanner, B.M.; Bittlinsky, A.; Keskin, F.; Bartels, N.K.; Buechner, N.; Huesing, A.; Rump, L.C.; Mügge, A. Obstructive sleep apnoea and its therapy influence high-density lipoprotein cholesterol serum levels. Eur. Respir. J. 2006, 27, 121–127. [Google Scholar] [CrossRef]
- Dorkova, Z.; Petrasova, D.; Molcanyiova, A.; Popovnakova, M.; Tkacova, R. Effects of Continuous Positive Airway Pressure on Cardiovascular Risk Profile in Patients With Severe Obstructive Sleep Apnea and Metabolic Syndrome. Chest 2008, 134, 686–692. [Google Scholar] [CrossRef]
- Barceló, A.; Barbé, F.; de la Peña, M.; Martinez, P.; Soriano, J.B.; Piérola, J.; Agustí, A.G. Insulin resistance and daytime sleepiness in patients with sleep apnoea. Thorax 2008, 63, 946–950. [Google Scholar] [CrossRef] [PubMed]
- Coughlin, S.R.; Mawdsley, L.; Mugarza, J.A.; Wilding, J.P.; Calverley, P.M. Cardiovascular and metabolic effects of CPAP in obese males with OSA. Eur. Respir. J. 2007, 29, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Lattimore, J.L.; Wilcox, I.; Skilton, M.; Langenfeld, M.; Celermajer, D.S. Treatment of obstructive sleep apnoea leads to improved microvascular endothelial func.c.ction in the systemic circulation. Thorax 2006, 61, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Kitahara, Y.; Hattori, N.; Yokoyama, A.; Nakajima, M.; Kohno, N. Effect of CPAP on brachial-ankle pulse wave velocity in patients with OSAHS: An open-labelled study. Respir. Med. 2006, 100, 2160–2169. [Google Scholar] [CrossRef]
- Drager, L.F.; Bortolotto, L.A.; Figueiredo, A.C.; Krieger, E.M.; Lorenzi, G.F. Effects of continuous positive airway pressure on early signs of atherosclerosis in obstructive sleep apnea. Am. J. Respir. Crit. Care Med. 2007, 176, 706–712. [Google Scholar] [CrossRef]
- Nadeem, R.; Singh, M.; Nida, M.; Kwon, S.; Sajid, H.; Witkowski, J.; Pahomov, E.; Shah, K.; Park, W.; Champeau, D. Effect of CPAP treatment for obstructive sleep apnea hypopnea syndrome on lipid profile: A meta-regression analysis. J. Clin. Sleep Med. 2014, 10, 1295–1302. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Yi, H.; Guan, J.; Yin, S. Effect of continuous positive airway pressure on lipid profile in patients with obstructive sleep apnea syndrome: A meta-analysis of randomized controlled trials. Atherosclerosis 2014, 234, 446–453. [Google Scholar] [CrossRef]
- Lin, M.T.; Lin, H.H.; Lee, P.L.; Weng, P.H.; Lee, C.C.; Lai, T.C.; Liu, W.; Chen, C.L. Beneficial effect of continuous positive airway pressure on lipid profiles in obstructive sleep apnea: A meta-analysis. Sleep Breath 2015, 19, 809–817. [Google Scholar] [CrossRef]
- Chen, B.; Guo, M.; Peker, Y.; Salord, N.; Drager, L.F.; Lorenzi-Filho, G.; Tang, X.; Li, Y. Effect of Continuous Positive Airway Pressure on Lipid Profiles in Obstructive Sleep Apnea: A Meta-Analysis. J. Clin. Med. 2022, 11, 596. [Google Scholar] [CrossRef]
- Cholidou, K.G.; Kostakis, I.D.; Manali, E.D.; Perrea, D.; Margeli, A.; Vougas, K.; Markozannes, E.; Koulouris, N.; Alchanatis, M. Calprotectin: A protein related to cardiovascular risk in adult patients with obstructive sleep apnea. Cytokine 2013, 61, 917–923. [Google Scholar] [CrossRef]
- Chen, B.; Somers, V.K.; Tang, X.; Li, Y. Moderating Effect of BMI on the Relationship Between Sympathetic Activation and Blood Pressure in Males with Obstructive Sleep Apnea. Nat. Sci. Sleep 2021, 13, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Schmoller, A.; Eberhardt, F.; Jauch-Chara, K.; Schweiger, U.; Zabel, P.; Peters, A.; Schultes, B.; Oltmanns, K.M. Continuous positive airway pressure therapy decreases evening cortisol concentrations in patients with severe obstructive sleep apnea. Metabolism 2009, 58, 848–853. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.; Zhang, Y.; Wang, G.; Han, D. Benefits of continuous positive airway pressure on glycaemic control and insulin resistance in patients with type 2 diabetes and obstructive sleep apnoea: A meta-analysis. Diabetes Obes. Metab. 2021, 23, 540–548. [Google Scholar] [CrossRef] [PubMed]
- Koenig, A.M.; Koehler, U.; Hildebrandt, O.; Schwarzbach, H.; Hannemann, L.; Boneberg, R.; Heverhagen, J.T.; Mahnken, A.H.; Keller, M.; Kann, P.H.; et al. The Effect of Obstructive Sleep Apnea and Continuous Positive Airway Pressure Therapy on Skeletal Muscle Lipid Content in Obese and Nonobese Men. J. Endocr. Soc. 2021, 5, bvab082. [Google Scholar] [CrossRef] [PubMed]
- Chopra, S.; Rathore, A.; Younas, H.; Pham, L.V.; Gu, C.; Beselman, A.; Kim, I.Y.; Wolfe, R.R.; Perin, J.; Polotsky, V.Y.; et al. Obstructive Sleep Apnea Dynamically Increases Nocturnal Plasma Free Fatty Acids, Glucose, and Cortisol During Sleep. J. Clin. Endocrinol. Metab. 2017, 102, 3172–3181. [Google Scholar] [CrossRef]
- Fadaei, R.; Koushki, M.; Sharafkhaneh, A.; Moradi, N.; Ahmadi, R.; Rostampour, M.; Khazaie, H. The impact of continuous positive airway pressure therapy on circulating levels of malondialdehyde: A systematic review and meta-analysis. Sleep Med. 2020, 75, 27–36. [Google Scholar] [CrossRef]
- Akinnusi, M.E.; Laporta, R.; El-Solh, A.A. Lectin-like oxidized low-density lipoprotein receptor-1 modulates endothelial apoptosis in obstructive sleep apnea. Chest 2011, 140, 1503–1510. [Google Scholar] [CrossRef]
- Chirinos, J.A.; Gurubhagavatula, I.; Teff, K.; Rader, D.J.; Wadden, T.A.; Townsend, R.; Foster, G.D.; Maislin, G.; Saif, H.; Broderick, P.; et al. CPAP, weight loss, or both for obstructive sleep apnea. N. Engl. J. Med. 2014, 370, 2265–2275. [Google Scholar] [CrossRef]
- Ramar, K.; Dort Leslie, C.; Katz Sheri, G.; Lettieri Christopher, J.; Harrod Christopher, G.; Thomas Sherene, M.; Chervin Ronald, D. Clinical Practice Guideline for the Treatment of Obstructive Sleep Apnea and Snoring with Oral Appliance Therapy: An Update for 2015. J. Clin. Sleep Med. 2015, 11, 773–827. [Google Scholar] [CrossRef]
- Recoquillon, S.; Pépin, J.L.; Vielle, B.; Andriantsitohaina, R.; Bironneau, V.; Chouet-Girard, F.; Fleury, B.; Goupil, F.; Launois, S.; Martinez, M.C.; et al. Effect of mandibular advancement therapy on inflammatory and metabolic biomarkers in patients with severe obstructive sleep apnoea: A randomised controlled trial. Thorax 2019, 74, 496–499. [Google Scholar] [CrossRef]
- Venema, J.; Vries, G.E.K.; van Goor, H.; Westra, J.; Hoekema, A.; Wijkstra, P.J. Cardiovascular and metabolic effects of a mandibular advancement device and continuous positive airway pressure in moderate obstructive sleep apnea: A randomized controlled trial. J. Clin. Sleep Med. 2022, 18, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.; Guimarães, T.M.; Pontes, G.; Coelho, G.; Badke, L.; Fabbro, C.D.; Tufik, S.; Bittencourt, L.; Togeiro, S. The effects of continuous positive airway pressure and mandibular advancement therapy on metabolic outcomes of patients with mild obstructive sleep apnea: A randomized controlled study. Sleep Breath 2021, 25, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhan, X.; Wang, N.; Pinto, J.M.; Ge, X.; Wang, C.; Tian, J.; Wei, Y. Does airway surgery lower serum lipid levels in obstructive sleep apnea patients? A retrospective case review. Med. Sci. Monit. 2014, 20, 2651–2657. [Google Scholar] [PubMed]
- She, W.; Wang, J.; Qian, X.; Hang, M. Long-term follow-up of patients with obstructive sleep apnea syndrome treated with uvulopalatopharyngoplasty. Zhonghua Er Bi Yan Hou Ke Za Zhi 2001, 36, 227–230. [Google Scholar] [PubMed]
- Bikov, A.; Frent, S.; Reisz, D.; Negru, A.; Gaita, L.; Breban Schwarzkopf, D.; Mihaicuta, S. Comparison of Composite Lipid Indices in Patients with Obstructive Sleep Apnoea. Nat. Sci. Sleep 2022, 14, 1333–1340. [Google Scholar] [CrossRef]

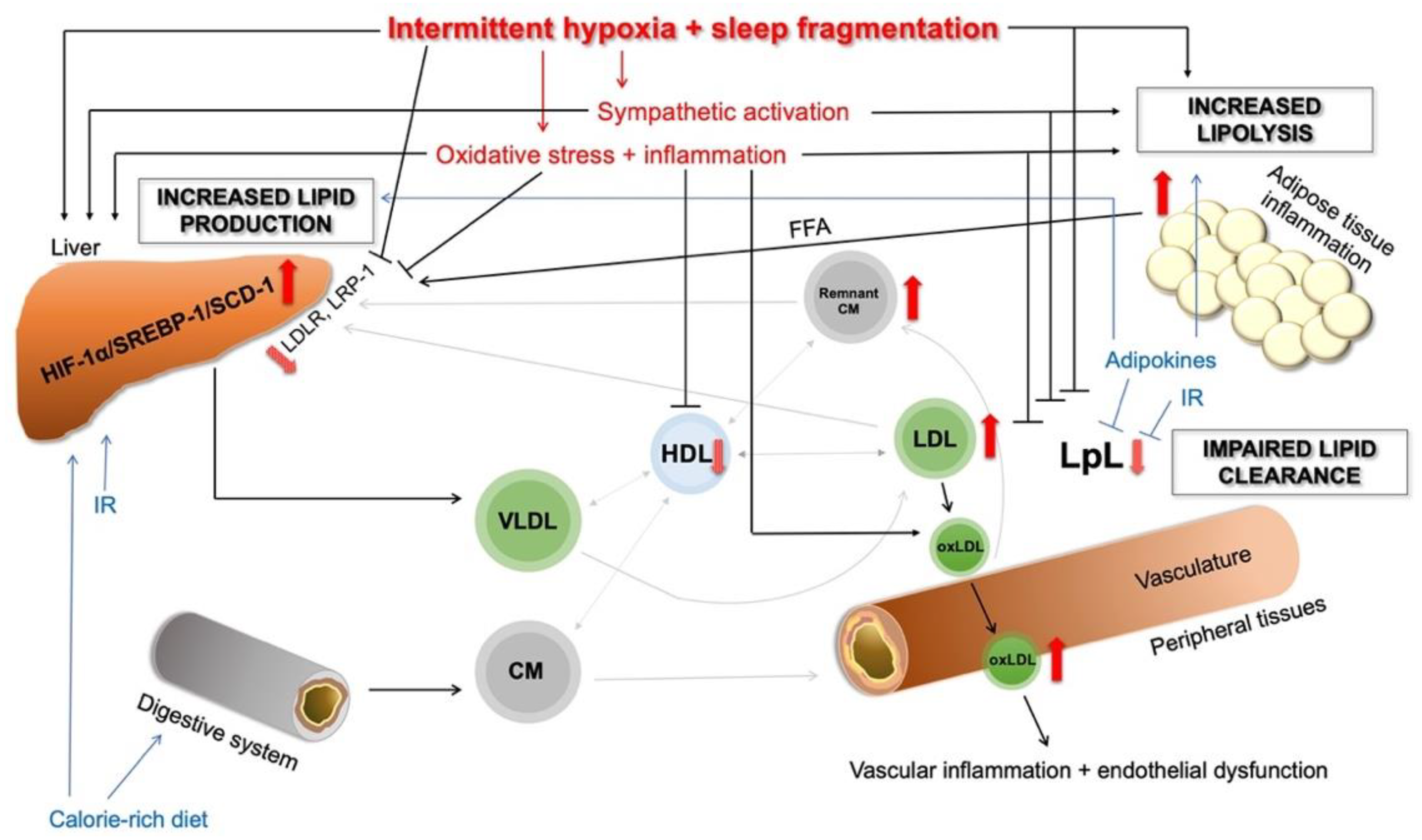
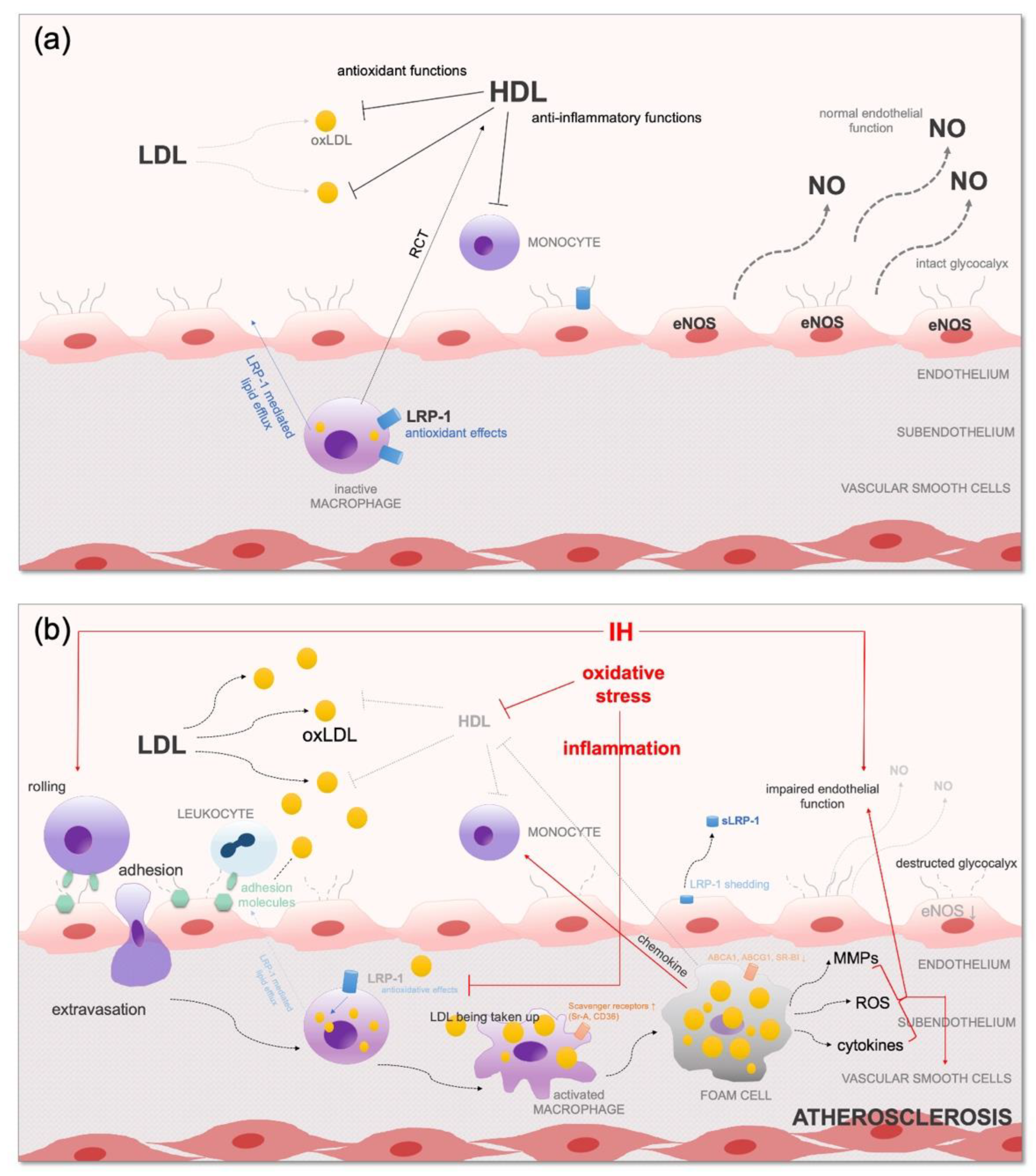
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meszaros, M.; Bikov, A. Obstructive Sleep Apnoea and Lipid Metabolism: The Summary of Evidence and Future Perspectives in the Pathophysiology of OSA-Associated Dyslipidaemia. Biomedicines 2022, 10, 2754. https://doi.org/10.3390/biomedicines10112754
Meszaros M, Bikov A. Obstructive Sleep Apnoea and Lipid Metabolism: The Summary of Evidence and Future Perspectives in the Pathophysiology of OSA-Associated Dyslipidaemia. Biomedicines. 2022; 10(11):2754. https://doi.org/10.3390/biomedicines10112754
Chicago/Turabian StyleMeszaros, Martina, and Andras Bikov. 2022. "Obstructive Sleep Apnoea and Lipid Metabolism: The Summary of Evidence and Future Perspectives in the Pathophysiology of OSA-Associated Dyslipidaemia" Biomedicines 10, no. 11: 2754. https://doi.org/10.3390/biomedicines10112754
APA StyleMeszaros, M., & Bikov, A. (2022). Obstructive Sleep Apnoea and Lipid Metabolism: The Summary of Evidence and Future Perspectives in the Pathophysiology of OSA-Associated Dyslipidaemia. Biomedicines, 10(11), 2754. https://doi.org/10.3390/biomedicines10112754







