Abstract
Medicinal plants have been a traditional remedy for numerous ailments for centuries. However, their usage is limited due to a lack of evidence-based studies elucidating their mechanisms of action. In some countries, they are still considered the first treatment due to their low cost, accessibility, and minor adverse effects. Mexico is in second place, after China, in inventoried plants for medicinal use. It has around 4000 species of medicinal plants; however, pharmacological studies have only been carried out in 5% of its entirety. The species of the Mexican arid zones, particularly in semi-desert areas, exhibit outstanding characteristics, as their adverse growing conditions (e.g., low rainfall and high temperatures) prompt these plants to produce interesting metabolites with diverse biological activities. This review explores medicinal plants belonging to the arid and semi-arid zones of Mexico, focusing on those that have stood out for their bioactive potential, such as Jatropha dioica, Turnera diffusa, Larrea tridentata, Opuntia ficus-indica, Flourensia cernua, Fouquieria splendes, and Prosopis glandulosa. Their extraction conditions, bioactive compounds, mechanisms of action, and biological efficacy are presented, with emphasis on their role in the treatment of respiratory diseases. Additionally, current research, novel applications, and perspectives concerning medicinal plants from these zones are also discussed.
1. Introduction
Throughout the history of humanity, traditional medicine has been the remedy for the cure of many diseases. Thanks to previous generations, humanity has knowledge about the use and effect of various medicinal plants. The most ancient knowledge of herbal medicine is found in Traditional China Medicine, which has more than 5000 years of knowledge of 365 drugs [1]. Over time, different cultures have documented their knowledge of medicinal plants. In Mexico, there is the Codex de la Cruz-Badiano, written by a Nahuatl physician and translated into Latin by Juan Badiano, which describes 227 medicinal plants, most of them of autochthonous origin [2].
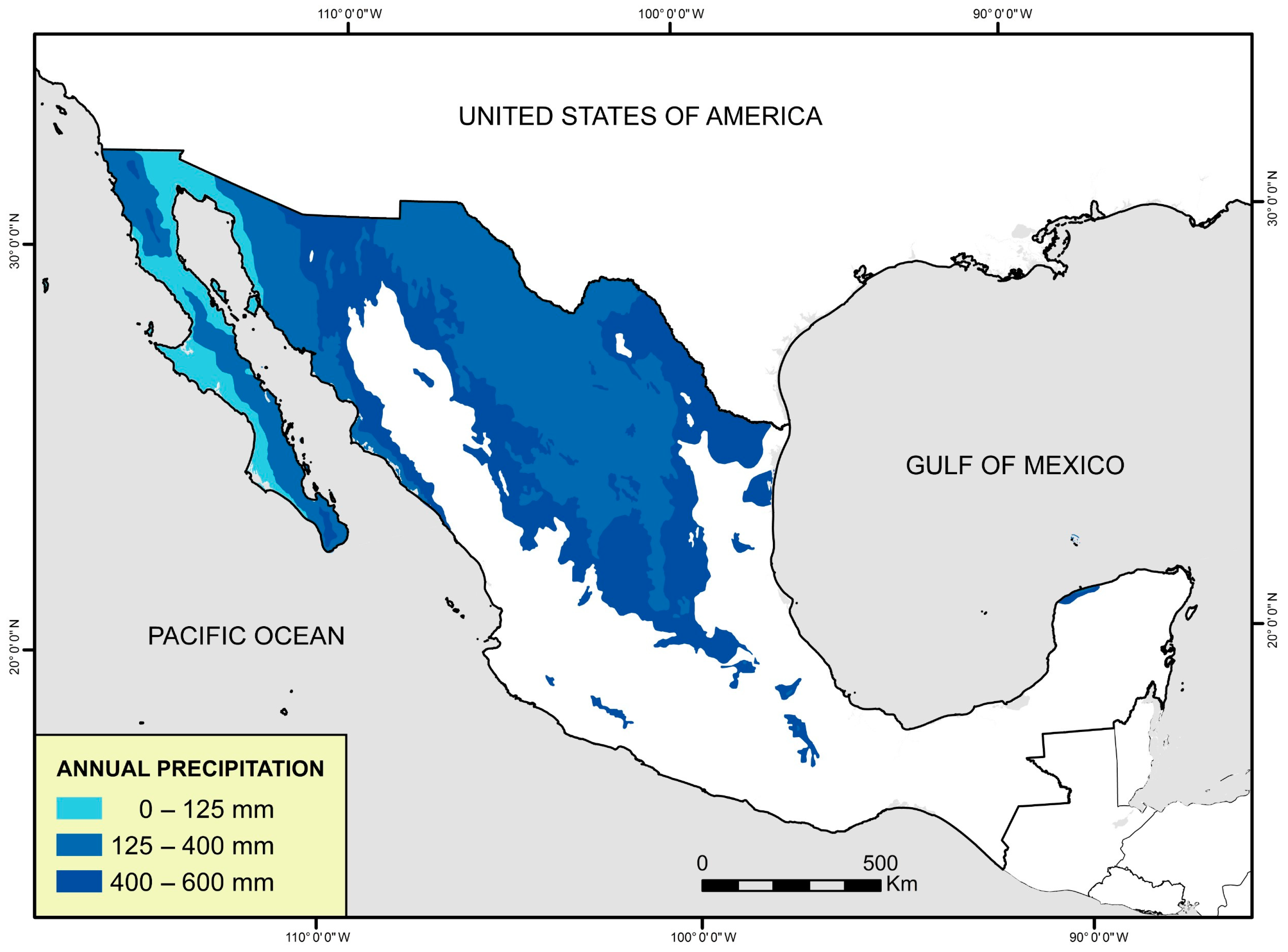
Mexico ranks fifth in the plant diversity and second place in the world in registration of medicinal plants. It has between 21,073 and 23,424 vascular plants that have been classified to date. On the other hand, around 4000 plant species with medicinal attributes are estimated to exist in Mexico, representing 15% of the total Mexican flora [3]. The arid lands cover 60% of its surface. In a semi-arid zone, annual rainfall is between 250 and 500 mm (Figure 1) [4]. The regions with arid and semi-arid zones in Mexico are Chihuahua, Coahuila, Durango, Zacatecas, San Luis Potosí, Nuevo León, Tamaulipas, Baja California, and Sonora [5,6]. These areas have a wide range of flora adapted to hostile environments of low rainfall and high temperatures [7]. Among the arid and semi-arid zones plant species in Mexico, certain ones stand out for their bioactive potential and characteristics: Jatropha dioica, Flourensia cernua, Turnera diffusa, Eucalyptus camaldulensis, Euphorbia antisyphilitica, Larrea tridentata, Lippia graveolens, Opuntia ficus-indica, Agave spp., Prosopis glandulosa, Punica granatum, Eysenhardtia texana, Machaeranthera pinnatifida, Mentha piperita, Rhus microphylla, Notholaena sinuata, Gnaphalium spp., Sambucus nigra, Matricaria chamomilla, and Populus fremontii [8,9,10,11,12].
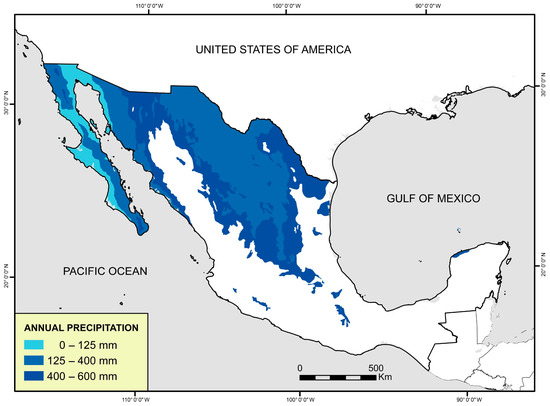
Figure 1.
Arid and semi-arid zones of Mexico. Map of Mexico in blue color shows the very dry, dry, and semi-dry climates of Mexico according to the average annual precipitation.
Medicinal plants are rich in secondary metabolites, and depending on the genera and species, they may contain different chemical classes and concentrations in different parts of the plant. Many plant species in the arid and semi-arid zones of Mexico have been studied for their bioactive compounds, which are produced in response to the climatic conditions that promote the synthesis of secondary metabolites [13,14]. When plants experience stress, such as exposure to certain environmental factors like heat, cold, drought, salinity, or specific pathogens, they often activate various defense mechanisms [14,15,16]. The bioactive compounds present in medicinal plants can vary widely in nature, including polyphenols, flavonoids, terpenoids, alkaloids, glycosides, waxes, vitamins, and fatty acid esters. These compounds may serve different biological functions that benefit the plants, such as defense against pathogen or insect attack, pollinator attraction, resistance to adverse weather conditions, among others [7,17,18].
On the other hand, some of these metabolites have important effects at the cellular and physiological level in the human organism, owing to their therapeutic properties. They have effects on different diseases, including respiratory diseases, neurodegenerative diseases, cardiovascular diseases, diabetes, obesity, among others [18,19,20,21,22,23].
Respiratory tract infections are one of the most common in the world. They mainly include chronic obstructive pulmonary disease (COPD), asthma, pneumonia, pulmonary fibrosis, lung cancer, and, more recently, SARS-CoV-2 (COVID-19). Most of the population suffers from some respiratory disease at least once a year [19,24]. In 2019, the entire world was affected by the SARS-CoV-2, a virus that affects the respiratory system and caused the death of millions of people during the pandemic. One of the alternatives to improve the symptoms caused by COVID-19 is the use of medicinal plants. However, there is limited information on the study of arid and semi-arid zones of Mexico to treat respiratory diseases, which needs further investigation for designing novel herbal formulations [19]. To date, only Opuntia ficus-indica belonging to the semi-arid zones of Mexico has been studied for its interaction between chiral compound astragalin with SARS-CoV-2 Mpro, like a possible protease inhibitor, that is one of the most investigated protein targets of therapeutic strategies for COVID-19 [25].
The plants Jatropha dioica, Turnera diffusa, Larrea tridentata, Opuntia ficus-indica, Flourensia cernua, Fouquieria splendes, and Prosopis glandulosa have long been utilized in traditional medicine within the arid and semi-arid zones of Mexico, including the Chihuahua Desert and the Sonoran Desert. They are employed to alleviate a variety of respiratory ailments such as bronchitis, asthma, colds, and coughs. Therefore, this review discusses the main bioactive compounds of some plants from arid and semi-arid zones of Mexico, their extraction techniques, their effectiveness against certain respiratory diseases due to their scientifically proven pharmacological properties, which can be targeted by therapies for their anti-inflammatory, antimicrobial, and anticancer characteristics.
2. Respiratory Diseases
Respiratory diseases can be caused by microbial infections, environmental pollutants, allergens, and genetic predisposition mainly affecting the lungs, some of the symptoms can cause chest discomfort, wheezing, flu, cough, weakness, and other symptoms. Respiratory diseases can be acute or chronic, untreated can cause further discomfort or be fatal. Infections caused by fungi, viruses or bacteria can affect the respiratory tract and the defense system is activated by sending defense cells such as lymphocytes, monocytes, and neutrophils to the site of infection. Nevertheless, the hyperproduction of neutrophils causes the production of Reactive Oxygen Species (ROS), resulting in oxidative stress and inflammation that can compromise the lung’s defense system [26]. Currently, conventional therapy offers a wide range of treatments for respiratory diseases. However, certain drugs designed to interact with multiple targets have been linked to adverse side effects such as diarrhea, liver dysfunction, nausea, loss of appetite, and vomiting [27]. Conversely, studies have shown that combining conventional treatment with the use of medicinal plants can improve symptoms in patients with respiratory diseases caused by SARS-CoV-2, without adverse reactions [28]. One of the significant challenges identified by the World Health Organization (WHO) is the urgent need to explore alternatives to combat antimicrobial resistance associated with conventional therapies. Without intervention, the WHO estimates that drug-resistant diseases could result in 10 million deaths annually by 2050. Therefore, it is crucial to investigate alternatives such as medicinal plants therapies, either alone or in combination with conventional drugs, to address the impending threat [29]. Antioxidant therapy or antioxidant-rich diet is a good option to counteract lung diseases, including those associated with SARS-CoV-2, to reduce oxidative stress in the respiratory system [20,30]. In this sense, medicinal plants are a form of natural therapy that can counteract or prevent various diseases of the respiratory tract, due to their bioactive compounds, such as polyphenols. Table 1 shows the factors that contribute to respiratory diseases. In addition to the factors mentioned in this table, emotional conditions such as anxiety, stress, and depression can exacerbate respiratory diseases.

Table 1.
Factors involved in respiratory diseases.
In several countries, herbal remedies offer a cost-effective therapy to treat illnesses at a low cost. While Mexico generally has access to medical services, certain rural areas still prefer natural remedies for diverse ailments, a practice observed throughout the country. In Mexico’s semi-desert regions, specific plants, such as J. dioica, T. diffusa, L. tridentata, F. cernua, among others, are recognized for their bioactive compounds with antiviral, antibacterial, and antioxidant properties, potentially inhibiting the growth of pathogenic microorganism responsible for pneumonia, tuberculosis, and viral flu [46,47,48,49].
3. Scientific Evidence of Semi-Desert Plants Used to Treat Respiratory Diseases
Globally, one of the main causes of concern in hospitals is respiratory tract infections caused by opportunistic bacteria that acquire resistance to antibiotics. Chronic obstructive pulmonary disease and asthma were the two leading causes of death in Mexico, with a total of 21,441 deaths recorded in people over 75 years of age [50]. However, after the pandemic caused by the SARS-CoV-2 virus, which causes respiratory diseases, the number of deaths increased to 334,107 in Mexico [51].
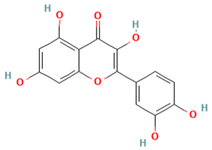
Bioactive compounds are chemical compounds that vary according to each plant genus and species, and various biotic or abiotic factors are involved in their expression and concentration. Among the isolated bioactive compounds of medicinal plants with recognized antiviral action are rutin (known as quercetin-3-rutinoside), a flavonoid glycoside effective against avian influenza virus and parainfluenza -3 virus. Quercetin, an aglycone of rutin, has demonstrated its therapeutic potential against influenza A virus (IFV-A), rhinovirus, dengue virus type 2, poliovirus type 1, adenovirus, Epstein–Barr virus, Mayaro virus, Japanese encephalitis virus, and RSV [21].
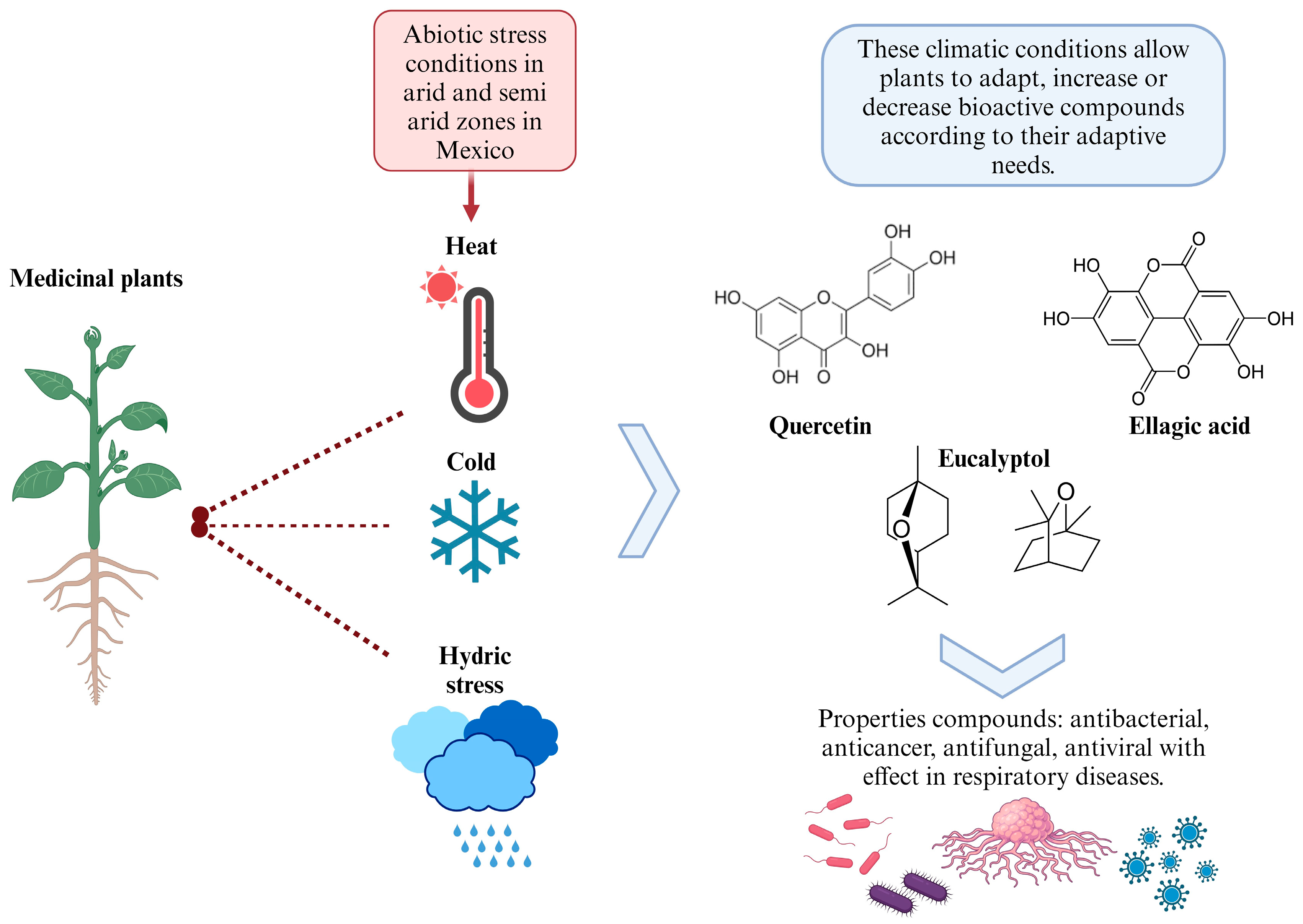
The following section highlights medicinal plants from the arid and semi-arid zones of Mexico known for their role in the treatment of respiratory diseases. Figure 2 provides an overview of these medicinal plants and illustrates how the abiotic stresses, such as extreme temperatures and water scarcity, can influence the synthesis of secondary metabolites. These stressors may either enhance or diminish the production of metabolites, often favoring and increasing compounds with antioxidant properties. This increase in antioxidants can directly impact respiratory diseases, which will be explored in the subsequent sections.
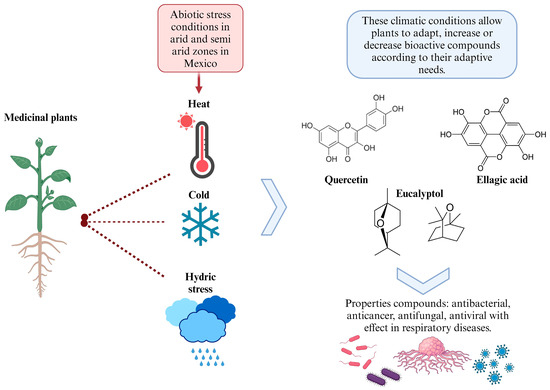
Figure 2.
Effect of plants from arid and semi-arid zones of Mexico against abiotic stress.
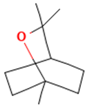
3.1. Jatropha dioica
It is commonly known as “Sangre de Drago”. The habitat of this species is in regions with dry and semi-dry climates. It is in Texas, USA, and the North of Mexico, although it can be found in all the Mexican territory [52]. The genus Jatropha (Euphorbiaceae) has about 200 species of woody trees, shrubs, subshrubs, or herbs. It is a shrub 50 cm to 1.50 m high. It owes its common name to the fact that it has a colorless juice that changes to dark when in contact with air. Its branches are reddish-brown with leaves longer than wide. Its flowers are small and in clusters of pink. The globose fruits are 1.5 cm long and have a seed [53]. Jatropha species are used in traditional medicine to treat many clinical conditions, such as stomach pain, toothache, bloating, inflammation, leprosy, dysentery, dyscrasia, vertigo, anemia, diabetes, as well as to improve human immunodeficiency virus conditions, tumors, ophthalmia, ringworm, ulcers, malaria, skin diseases, bronchitis, asthma, and as an aphrodisiac [54]. Several studies have demonstrated the antimicrobial effectiveness of J. dioica, although it depends on the solvent and the part of the plant used. The methanol extract of J. dioica was effective against Staphylococcus aureus and Klebsiella pneumonia, which presented bioactive compounds such as flavonoids, terpenes/sterols, lactons, and quinons [49]. Another study showed that organic extracts of J. dioica root, prepared with hexane, ethanol, and acetone, evaluated on human pathogens such as Bacillus cereus, Escherichia coli, Salmonella typhi, S. aureus, Enterobacter aerogenes, E. cloacae, S. typhimurium, Cryptococcus neoformans, Candida albicans, C. parapsilosis, and Sorothrix schenckii, had a high antibacterial and antifungal activity that may come from the presence of β-sitosterol (terpene) compounds, which was extracted using hexane [55]. Some of these microorganisms are pathogens that cause respiratory diseases, such as S. aureus, Pseudomona aeruginosa, Haemophylus influenzae, Streptococcus pneumoniae, E. coli [56], among many other strains. In general, different extracts of this plant have shown antimicrobial properties, tested against strains involved in respiratory diseases, proving its specific effectiveness by the bioactive compounds found.
3.2. Turnera diffusa
T. diffusa was one of the most important medicinal plants at the time of the Maya culture. Commonly known as “damiana”, a shrub that grows in the arid and semiarid regions of South America, Mexico, USA, and the West Indies. T. diffusa is a shrub up to 1.5 m high that is very branched and whose stems are slightly reddish. The leaves are small, wrinkled and have teeth on the edge; they give off a strong aroma when squeezed. Its flowers are yellow and look like little stars hidden among the branches and its fruits are capsules. It is one of the most highly appreciated plant aphrodisiacs. In addition, it was also used against cerebral weakness, impotence, and orchitis [47]. It is a medicinal plant studied for its pharmacological action and their properties antioxidant and antimicrobial. The main bioactive compounds of T. diffusa include flavonoids, phenolic acids and derivatives, cyanogenic derivates, fatty acids, alkaloids, terpenoids, and sugars conjugates. In a study reported by Urbizu et al. [57], 21 compounds were identified in essential oils of T. diffusa including Ethanone, 1-(1,3-dimethyl-3-cyclohexen-1-yl) and Eucalyptol. This last is known to have antibacterial properties, thus promoting the elimination of bacteria from tobacco-exposed lungs by attenuating ciliated cell damage and suppressing the expression of the MUC5AC gene, a protein that is related to mucus hypersecretion in the pulmonary tract [58]. Thirty-five bioactive compounds classified within the above compounds were detected in T. diffusa, including five new compounds detected in this plant and one new compound (6) not previously studied: luteolin 8-C-E-propenoic acid (1), luteolin 8-C-β-[6-deoxy-2-O-(α-L-rhamnopyranosyl)-xylo-hexopyranos-3-uloside] (2), apigenin 7-O-(6″-O-p-Z-coumaroyl-β-D-glucopyranoside) (3), apigenin7-O-(4″-O-p-Z-coumaroyl-glucoside) (4), syringetin 3-O-[β-D-glucopyranosyl-(1→6)-β-D-glucopyranoside] (5), and laricitin 3-O-[β-D-glucopyranosyl-(1→6)-β-D-glucopyranoside] (6) [48]. A study showed that T. diffusa has bactericidal, inhibitory, and bacteriostatic action at Minimal Inhibitory Concentration (MIC) of 300 µg/mL doses against S. aureus [59]. This bacterium has multiple virulence factors that give it the ability to penetrate the body’s barriers and produce diseases such as pneumonia [60]. On the other hand, it is known that the medicinal bioactivity and the industrial interest may be affected depending on the locality of collection of this plant, because despite having many bioactive compounds, the presence, or concentrations of these may vary. Despite this, T. diffusa has multiple beneficial properties for human health [57].
3.3. Larrea tridentata
Commonly known as “gobernadora” or “creosote bush”, is a bush from the northern region of Mexico and the south of the USA. It is a very branched shrub, evergreen, from 0.6 to 3 m high. Leaves formed by two leaflets joined together at the base to each other at the base. Fruit subglobose to obovoid, 7 mm long, with a long. This vegetal specimen possesses a wide variety of secondary metabolites such as lignans, flavonoids, glycosylated flavonoids, saponins, sterols, tannins, terpenes, and essential oils [61]. Among the most promising and widely reported metabolites from L. tridentata, are as follows: 3′-demethoxy-6-O-demethylisoguaiacin, 3′-O-methyldihydroguaiaretic acid, meso-dihydroguaiaretic acid, and tetra-O-methylnordihydroguaiaretic acid. These have been reported to exhibit antibacterial, antiprotozoal, anthelmintic, antifungal, antiviral, anticancer, and antioxidant activities [62]. Various studies demonstrate the biological effectiveness of this plant in diseases such as: antidiabetic, anti-inflammatory, antibacterial, antituberculosis, antifungal, antiviral activities. Núñez-Mojica [61] showed that chloroform extract of L. tridentata, isolated and characterized two new cyclolignans; one of them proved to have an effect against Mycobacterium tuberculosis. Another study by Favela-Hernández [63] involved the extraction of dried and ground leaves using chloroform, and tested the effectiveness of several bioactive compounds against pathogenic bacteria of the respiratory tract: dihydroguaiaretic acid had activity towards methicillin resistant S. aureus (MRSA) (MIC 50 µg/mL) and multidrug-resistant (MDR) strains of M. tuberculosis (MIC 12.5–50 µg/mL); 4-epi-larreatricin was active against MDR strains of M. tuberculosis (MIC 25 µg /mL); 3′-Demethoxy-6-O-demethylisoguaiacin displayed activity against sensitive and resistant S. aureus (MIC 25 µg/mL) and MDR strains of M. tuberculosis (MIC 12.5 µg /mL). 5,4′-Dihydroxy-3,7,8,3′-tetramethoxyflavone and 5,4′-dihydroxy-3,7,8-trimethoxyflavone were active against M. tuberculosis MDR strains having MIC values of 25 and 25–50 µg/mL, respectively, while 5,4′-dihydroxy-7-methoxyflavone was active against S. aureus (MIC 50 µg/mL). L. tridentata has a great antibacterial potential against respiratory tract diseases, specifically against tuberculosis, but further studies are needed to evaluate possible adverse effects.
3.4. Opuntia spp.
In Mexico, the genus Opuntia has a wide distribution. Wild species of the genus Opuntia are part of the natural ecosystems of semi-arid regions [64]. The regions with the greatest species richness are the central and northern Altiplano, the northwest, the Bajío, and the Tehuacán-Cuicatlán Valley. The subfamily Opuntioideae is characterized by the presence of fugacious, cylindrical, ribless leaves; glochids on the areoles; a hard, generally light-brown aril covering the campylotropic ovule. The arid and desert regions of Mexico are inhabited by the largest number of Opuntia species in the world. There are endemic species of great importance like: O. engelmannii, O. macrocentra, O. durangensis, O. phaeacantha, O. rastrera, and O. microdasys [3].
The endemic species of the north of the country are those that resist frost, usually have thin stalks, which prevents them from dying when the water they contain freezes. Extracts from Opuntia species contain phenolic compounds, other antioxidants such as ascorbate, pigments such as carotenoids and betalains, and other phytochemicals [65]. Elkady et al. (2020) [66] characterized a fraction extracted in ethyl acetate from the prickly pear fruit peel, the authors detected a quercetin 5,4′-dimethyl ether, which exhibited an inhibitory effect against pneumonia pathogens. Opuntia flower extracts also showed in vitro antibacterial activity against P. aeruginosa, S. aureus, B. subtilis, and antifungal activity against C. lipolytica, and Aspergillus niger [65]. Regarding respiratory diseases caused by viruses or bacteria, it is precisely the plants of the Opuntia genus, which have been proven effective against several pathogenic strains that attack the respiratory tract.
3.5. Flourensia cernua
Commonly known as hojasen, it is a shrub that grows in the desert and semi-arid areas of the southern USA and northern Mexico such as Chihuahua, Sonora, Coahuila, Durango, Nuevo León, San Luis Potosi, and Zacatecas. It is a plant known for its use in traditional medicine for the treatment of gastrointestinal diseases such as stomach pain, diarrhea, expectorant, and anti-rheumatic [67,68]. It grows to a height of 1 to 2 m. Branches covered with thick, oval leaves up to 2.5 cm long. The hanging flower heads contain several yellow disk florets. The fruit is a hairy achene up to 1 cm long. Most parts of the plant are highly resinous and have a tarry or hoppy odor with a bitter taste [67]. The people consume this plant in infusion and decoction of leaves and steams [10]. Its bioactive molecules find extensive applications in the agri-food and health industries.
One of most frequent types of cancer in Mexico is lung cancer, more than 7500 new cases and 7100 deaths associated with this neoplasia were registered, being the seventh cancer with the highest incidence and the fourth cause of death from cancer in this country [69]. A study showed that three extracts of F. cernua, F. retinophylla, and F. microphylla, have a high content of total phenolic compounds, and presented antiproliferative effect on A549 cells, which is a lung epithelial cell line derived from human alveolar cell carcinoma. Besides, F. cernua demonstrated the best anti-inflammatory activity, therefore these extracts can be used for the control of lung cancer [18]. A study employing a hexane extract of F. cernua reported a MIC of 50 and 25 μg/mL against sensitive and resistant strains, effectively inhibiting and eradicating M. tuberculosis growth [70]. Therefore, F. cernua can be a target for therapies against lung cancer; case studies are required to verify its effectiveness in different stages of this type of cancer.
3.6. Fouquieria splendes
Commonly known as “ocotillo”, is a medicinal plant endemic to the arid and semi-arid zones of the southwestern United States and northern Mexico, distributed throughout the deserts of Sonora and Chihuahua, and is one of the most representative species of the Fouquieriaceae family (thirteen species) [71,72]. The plant grows as a shrub with thorny stems reaching 2 to 10 m and it is characterized by having leaves after rain. In addition, this plant presents inflorescences made up of flowers that can vary from white to red-orange, usually appearing during spring [73,74]. This plant is used in folk medicine as bark tincture from bark, infusion of the stem, roots, leaves, and flowers, or the direct use of leaves and flowers. These are used against different health conditions, such as cough, congestion, infections, stomach pain, wounds, painful, pelvic circulation, hemorrhoids, prostatic hyperplasia, and menstrual cramps [75,76]. On the other hand, scientific studies have shown that extracts from this plant exhibited antiproliferative, antioxidant, insecticidal, antiparasitic, antifungal, and antimicrobial potential [14,71,73,75,77]. Regarding the antimicrobial effect, some research has been developed. However, this plant has been shown to have an effect against microorganisms associated with respiratory tract infections, such as S. aureus. A study performed by Vega Menchaca et al. [75] demonstrated that a methanol extract of F. splendens leaves affected the growth of S. aureus, showing a MIC value of 25 µg/mL. Similarly, Rodríguez Garza [73] validated that a methanol stem extract inhibited S. aureus growth, showing inhibition halos greater than 20 mm at a concentration of 100 mg/mL. In addition to the above, various investigations have been focused on the antimicrobial analysis of a compound isolated from F. splendens called ocotillo, which is a triterpene, chemically modified with antimicrobial activity against bacteria associated with respiratory infections such as S. aureus, P. aeruginosa, and K. pneumoniae ATCC and resistant clinical isolates [78,79]. On the other hand, it is important to mention that the extract of F. splendens also presents antifungal and antiviral effects against microorganisms not associated with respiratory tract infections [73,80]. The above opens the possibility of analyzing the effect of F. splendens against microorganisms of clinical interest. On the other hand, in addition to triterpenes, other chemical compounds associated with the antimicrobial potential of F. splendens are phenolic compounds, identifying various flavonoids, phenolic acids, and anthocyanins, as well as fatty acids, which have presented antimicrobial effect through in vitro and in vivo tests [14,71,72,74,81]. An important aspect to highlight is that the F. splendens extract demonstrated low cytotoxicity in the in vitro evaluation with cell lines, in addition to showing low in vivo cytotoxicity against Artemia salina when exposed to polar extractions [71,72]. Similar results were reported by Zhou et al. [78] when evaluating the cytotoxicity of ocotillo derivatives using in vitro tests against cell lines. Previous information makes it possible consider this plant source for in vivo systems studies to demonstrate its effectiveness. In this sense, this shrub could be considered a potential antimicrobial source for treating respiratory tract infections.
3.7. Prosopis glandulosa
It is known as “honey mesquite” and is native to southwestern of Mexico and USA, growing mainly in the hyper-arid ecosystems [82]. This plant grows as a spiny shrub or small tree reaching 9 m height and shows bipinnately compound leaves [83]. In addition, its flowers presented a cylindric inflorescences of greenish-yellow color and fruit grow as linear indehiscent pods, straight or slightly curved with seeds and straw color in maturated conditions [83]. One of the main uses of P. glandulosa in local groups is by direct intake as food, using the seeds as floor [84]. Additionally, several ethnic groups use different parts P. glandulosa plant, such as leaves and bark, against different illnesses such as cough, sore throat, infections, inflammation, breast cancer, diabetic mellitus, muscular pain, eye wash, kidney stones, dyspepsia, eruptions, hernias, skin, and umbilical ailments [83,85,86]. On the other hand, scientific studies have demonstrated several biological activities of P. glandulosa extracts/compounds. Biological activities observed included anticancer, antihypertensive, antidiabetic, antimalarial, antihyperlipidemic, antileishmanial, cardioprotective, and immunostimulant; being the most recognize and outstanding, the antimicrobial effect [85,86,87,88,89].
Regarding to the antimicrobial effect of P. glandulosa, several studies have demonstrated its effectiveness against different microorganisms associated with respiratory tract infections. A methanol extract of P. glandulosa exhibited the capacity to inhibit the bacterial growth of P. aeruginosa (45%) and S. aureus (49%) according to the control. Additionally, it was also observed effect against opportunistic fungi cause of respiratory infection, such as A. niger (methanolic extract showed 88% of inhibition growth) and Fusarium solani (methanol and hexane extract showed 92% and 94%, respectively, of growth inhibition) [90]. In a study performed by Moorthy and Kumar [89], ethanol extract from P. glandulosa leaves showed growth inhibition of S. aureus (17.4 mm) and Cryptococcus neoformans (30.6 mm), compared to the control. Similarly, Imam et al. [91] observed that essential oil obtained from P. glandulosa seeds had the capacity to affect the growth of S. aureus (MIC: 3 µg/mL), K. pneumoniae (MIC: 5.5 µg/mL), A. niger (MIC: 34.5 µg/mL), and F. oxysporum (MIC: 20.5 µg/mL). López-Anchondo et al. [92] performed a leave extract from P. glandulosa which showed the capacity to affect the mycelial growth of different opportunistic fungi associated with a respiratory infection, such as F. oxysporum (5% inhibited 65% of mycelial growth), Rhizopus oryzae (5% inhibited 60% of mycelial growth), and R. stolonifer (5% inhibited 57% of mycelial growth). On the other hand, the antiviral effect of P. glandulosa has not been demonstrated; however, other species of the gender showed this effect [93]. The antimicrobial effect of P. glandulosa could be associated with different bioactive compounds identified in the extracts, such as flavonoids, phenolic acids, and alkaloids [94,95,96]. In this regard, Rahman et al. [85] isolated two alkaloids from ethanol extracts from P. glandulosa leaves with antimicrobial potential, which were identified as Δ1,6-juliprosopine and julirposine. Both compounds, showed antibacterial effect against S. aureus (MIC: 10 and 5 µg/mL, respectively), methicillin-resistant S. aureus (MIC: 10 and 5 µg/mL, respectively), and M. intracellulare (MIC: 10 and 2.5 µg/mL, respectively). An effect against C. neoformans was also observed (MIC: 1.25 and 0.63 µg/mL, respectively). In this way, it is important to highlight that both alkaloids showed equal or low MIC values than amphotericin B (MIC: 1.25 µg/mL), one of the most potent antifungals used in clinical practice. Additionally, the alkaloids did not exhibit a cytotoxic effect against VERO cells at the highest tested concentration (23,800 ng/mL). Ashfaq [97] used a root extract from P. glandulosa to isolate an alkaloid, prosopilosidine, which was evaluated against C. neoformans in vivo (murine model). They observed that the intraperitoneal administration (0.0625 mg/kg) reduced the fungal load by 76% compared with amphotericin B (85% at 1.5 mg/kg) after 5 days of the infection. Similarly, the oral administration exhibited a similar result because the tested alkaloid decreased the fungal organism by 82% compared to fluconazole (90% at 1.5 mg/kg) after 5 days of the infection. Additionally, it was observed that the evaluated alkaloid did not exhibit a toxic effect at 50 mg/kg against HepG2 cells. Also, it was observed that a dose of 20 mg/kg did not modify the normal plasma chemistry profile of tested mice. The above suggests that P. glandulosa has an interesting antimicrobial potential against microorganism associated with respiratory infection. Thus, the low cytotoxicity makes this plant an important alternative to develop antimicrobial agents against the mentioned pathogens.
4. Mechanisms of Action of Bioactive Compounds and/or Crude Extracts against the Disease
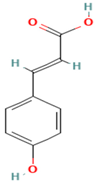
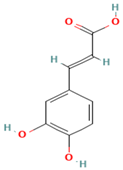
Plants contain chemical substances known as bioactive compounds, which can have a medicinal effect on the body due to their pharmacological activity. The main chemical groups of active drug components under broader heads are heterosides (e.g., anthraquinones, cardiac glycosides, cyanogenics, coumarins, phenols, flavonics, ranunculosides, saponosides, sulfurides), polyphenols (e.g., phenolic acids, cumarins, flavonoids, lignans, tannins, quinones), terpenoids (e.g., essential oils, iridoids, lactones, diterpones, saponins), and alkaloids (atropine, cocaine, daturin, hiosciamin, lysergic acid, nicotine, quinine) [98]. Mucilage and gums are heterogeneous polysaccharides, formed by different sugars, in general, they contain uronic acids. Other relevant active constituents in plants, such as vitamins, minerals, amino acids, carbohydrates and fibers, some sugars, organic acids, lipids, and antibiotics, are essential nutrients [44]. Flavonoids are a group of bioactive compounds that have antioxidant, antibacterial, antiviral, anti-inflammatory, antiangionic, analgesic, and antiallergic properties. On the other hand, they have also been found to have mutagenic activities and/or prooxidant effects. The human body contains numerous proteins with diverse functions. It has been studied that proteins Cytochromes P450 (CYPs) are a group of proteins that are distributed in many organs of the body and interact with flavonoids [45]. These compounds induce or modulate their metabolic activity. Flavonoids present in food are considered non-absorbable since they are attached to saccharides as beta-glucosides. However, the intestinal flora is responsible for hydrolyzing them into free flavonoids (aglycones), in this chemical form is easier to pass through the intestinal wall. However, quercetin, an onion glycoside absorbs even better than pure aglycone. The mechanism of action of flavonoids against microbial growth involves altering the plasma membrane, inducing mitochondrial dysfunction, and inhibiting key cellular process including cell wall formation, cell division, RNA, and protein synthesis, as well as efflux-mediated pumping system [99]. Furthermore, studies have shown that certain bioactive compounds, such as Narcissin, can suppress neutrophil infiltration and the activity of immune cells (CD3+/CD4+, CD3+/CD8+, and Gr-1+/CD11b) in bronchoalveolar lavage fluid (BALF) and lungs [100,101]. Ellagic acid has been found to stimulate enzyme responses like SOD and CAT, thereby attenuating pulmonary emphysema by inhibiting ROS, reducing lipid peroxidation, and enhancing antioxidant activity [14,102,103,104]. Carvacrol disrupts bacterial cell membranes, increases membrane permeability, and decreases cytoplasmic pH, thereby affecting the respiratory system [104,105,106,107]. Besides, additional compounds are listed in Table 2.

Table 2.
Efficacy of bioactive compounds derived from plants in arid and semi-desert regions of Mexico against respiratory diseases.
The plants discussed in this review contain a large amount of these compounds that have antioxidant function and intervene in various ailments including respiratory diseases. Table 2 shows several bioactive compounds that have been shown to have effects on respiratory system diseases, these compounds have been detected in plants from arid and semi-arid zones of Mexico. To date, many metabolic pathways that explain the action of the active compounds isolated from the plants of the Mexican semi-desert in different respiratory diseases are unknown.
In Table 3 is presented both endemic plants and others with development in the arid and semi-arid zones of Mexico, and their effectiveness against respiratory diseases, doses, and some mechanisms of action.

Table 3.
Endemic and locally adapted plants from arid and semi-desert regions of Mexico, their effectiveness against respiratory diseases, doses, and mechanisms of action.
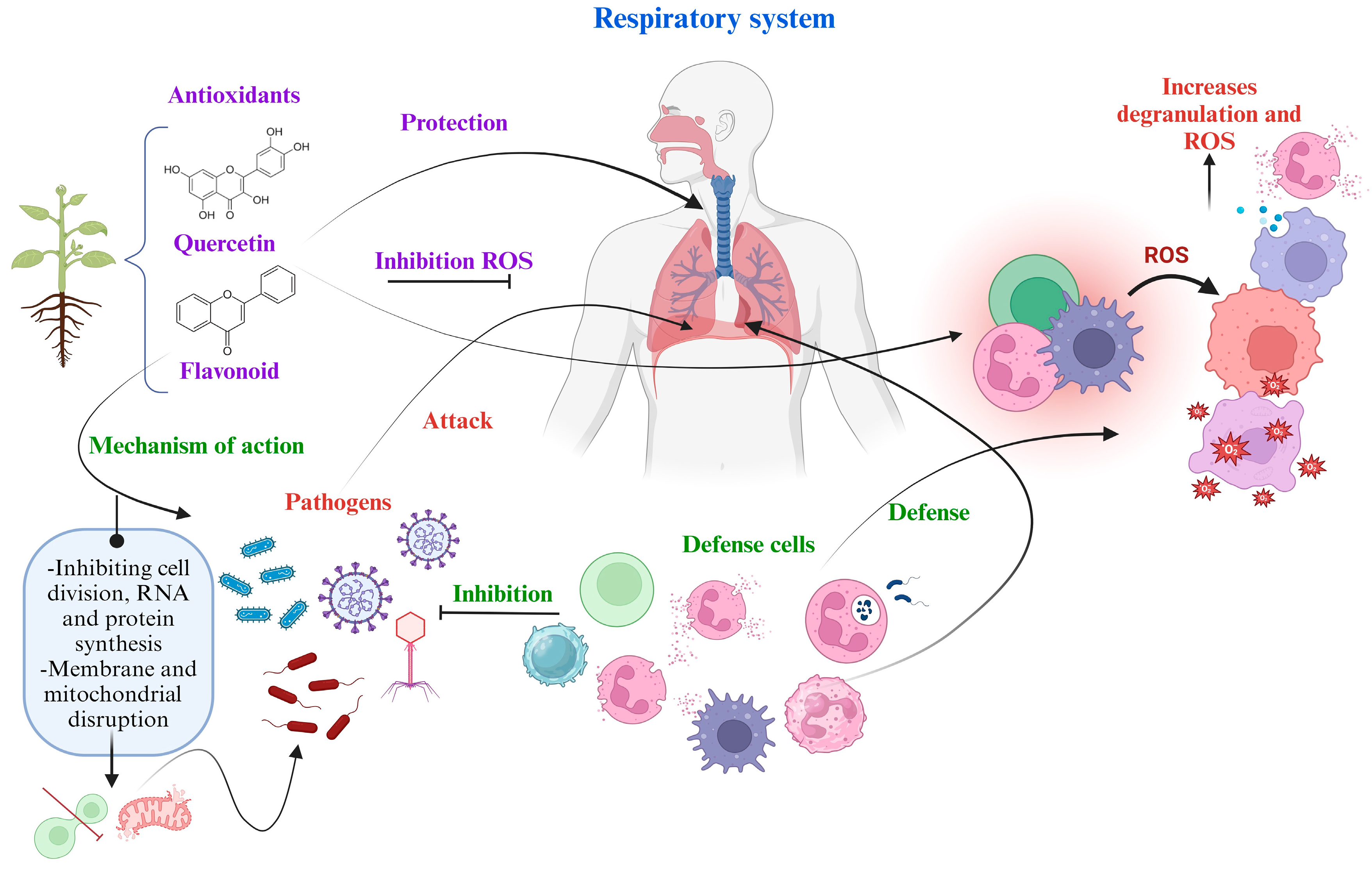
The human can avoid diseases due to the protection provided by the immune system. In a process of microbial infection or oxidative stress, defense cells are activated, also the synthesis of pro-inflammatory cells such as macrophages, interleukins, neutrophils from the MAPK metabolic pathway, which helps to inhibit pathogens. However, exposure of these cells to ROS leads to their lysis and degranulation, resulting in an increase in ROS level [131]. At this point, an important factor of protection is antioxidants from medicinal plants, which can inhibit ROS. In plants from arid and semi-arid zones of Mexico, the climatic factor can favor the expression of secondary metabolites, the effectiveness of their compounds and the description of some metabolic pathways on diseases of the respiratory system, which have been mentioned in Table 2 and Table 3. In the Figure 3, the general defense mechanisms available to humans against respiratory system diseases are presented. Bioactive compounds, such as antioxidants, exert their mechanism of action on bacteria, viruses, and fungi by inhibiting their development through the disruptions in cell division, mitochondria, protein synthesis, RNA, and ROS.
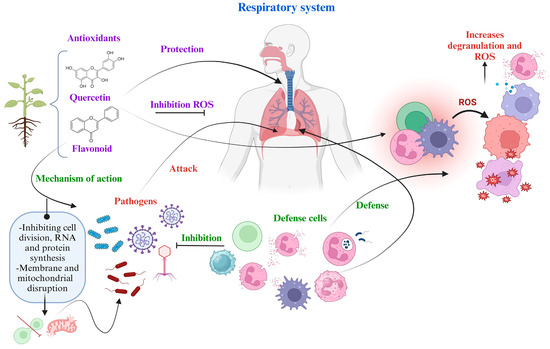
Figure 3.
General mechanism of action of medicinal plants and cell defense against pathogens and ROS.
5. Ethnopharmacology of Plants from Arid and Semi-Arid Zones of Mexico
Traditional medicine continues to be a good alternative for healthcare in Mexico, particularly in rural regions where access to conventional medical services and medications is limited. Its utilization not only addressed gaps in healthcare accessibility but also contributes to reducing the reliance on antibiotics and other allopathic treatments [132]. An ethnopharmacological study carried out in Mexico revealed that 54% of healthcare professionals and 49% of physicians have integrated medicinal plants into their alternative practices [132].
Ethnopharmacology serves as an important link between cultures preserving ancestral knowledge of medicinal plants as healing practices and pharmacology as a science field that standardizes and regulates these remedies. The interdisciplinary approach facilitates the evaluation of the biological efficiency of traditional medicines and enables the development of new therapeutic interventions. Table 4 presents a comprehensive overview of native plants endemic to the arid and semi-arid regions of Mexico, outlining their traditional uses and effectiveness in treating respiratory ailments.

Table 4.
Ethnopharmacology of plants from arid and semiarid zones in Mexico.
6. Commercially Available Important Plants in Mexico
In Mexico, there is a variety of commercially available herbal remedies, although only a limited number have received sanitary registration approval from the Federal Commission for Protection against Health Risks (COFEPRIS). As of 2023, one such approved herbal remedy is Aloe vera, commonly grown throughout the country and commercially marketed as Restaude [140]. Available in gel form, this pharmaceutical product is primarily designed to treat cosmetic skin concerns like acne scars and is also recognized for its efficacy in treating burns. In addition, beyond its known applications, A. vera has undergone research for additional therapeutic uses. Molecular docking studies have identified feralolide, also known as or ligand 6, as the most promising candidate among ten isolated compounds from A. vera, exhibiting significant reactivity with COVID-19 main protease (Mpro) [141].
Another notable example is the homeopathic medicine Simplex-Jet-Lag, approved by COFEPRIS in 2020) [140]. Formulated with Passiflora, Valeriana Arnica montana, this product is indicated for managing stress-related conditions and insomnia. While these plants are distributed in the South United States, Central America, and South America, their therapeutic effects extend beyond their common use. Despite its common use to treat anxiety and insomnia, Passiflora has a compound named Vitexin (apigenin-8-C-β-D-glucopyranoside), a flavone glycoside, which has demonstrated efficacy in reducing pulmonary edema and protein concentration in the alveoli [142]. Valerian extract has been found to modulate LL-37 gene and protein expression in lung cells, thus enhancing the immune system’s response to respiratory infections, including COVID-19 [143]. Additionally, Arnica exhibits bronchodilator activity like salbutamol, a well-known antiasthmatic medication [144].
7. Final Remarks
Respiratory diseases pose a significant global health challenge, affecting millions of individuals and causing substantial morbidity and mortality, particularly in underserved regions lacking adequate healthcare infrastructure. Among these, the emergence of the SARS-CoV-2 virus has underscored the urgent need for effective treatments, as it continues to claim countless lives worldwide. With no specific pharmaceutical intervention available for this novel virus, the exploration of alternative therapies has garnered increasing attention.
Medicinal plants, deeply rooted in the traditions of ancestors and indigenous communities, offer promising opportunities for the management of respiratory diseases alongside conventional medicine. In Mexico’s arid and semi-arid zones, numerous plant species have been scientifically documented for their pharmacological efficacy against respiratory ailments. These plants not only demonstrate potent anti-inflammatory, antimicrobial, and anticancer properties but also harbor specific bioactive compounds with proven therapeutic benefits.
Further studies are needed to comprehensively understand various aspects: efficacy, optimal doses, bioactive compounds’ mechanisms against respiratory diseases, drug interactions, combining these plants with conventional medicine, potential side effects, action mechanisms against pathogens, and health regulations. While research suggests potential in treating respiratory diseases using plants from Mexico’s arid and semi-arid zones, more studies are warranted in the aforementioned areas. Their proven bioactive compounds’ antimicrobial and antiproliferative effects, coupled with traditional use, highlight them as a promising area for future medical research.
Author Contributions
Writing-original draft preparation, methodology, I.E.D.-R.; conceptualization; writing-original draft preparation, supervision, A.V.C.-R. and J.C.L.-R.; writing-review and editing, project administration, supervision, M.L.F.-L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
Irma E. Dávila-Rangel thanks Consejo Nacional de Humanidades, Ciencias y Tecnologías (CONAHCYT, Mexico) for Postdoctoral fellowship support (CONAHCYT grant number 3054517). The authors would also like to thank to Eng. Luis Fernando Camacho Guerra of CIICyT (Centro de Investigación e Innovación Científica y Tecnológica, Universidad Autónoma de Coahuila, Mexico) for his assistance during this study.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Petrovska, B.B. Historical Review of Medicinal Plants’ Usage. Pharmacogn. Rev. 2012, 6, 1–5. [Google Scholar] [CrossRef]
- INAH 2023 Códice de La Cruz-Badiano. Available online: https://www.mediateca.inah.gob.mx/repositorio/islandora/object/codice:851#page/1/mode/2up (accessed on 30 May 2023).
- Comisión Nacional Para El Conocimiento y Uso de La Biodiversidad|Gobierno|Gob.Mx. Available online: https://www.gob.mx/conabio (accessed on 9 May 2023).
- Bigurra-Alzati, C.A.; Ortiz-Gómez, R.; Vázquez-Rodríguez, G.A.; López-León, L.D.; Lizárraga-Mendiola, L. Water Conservation and Green Infrastructure Adaptations to Reduce Water Scarcity for Residential Areas with Semi-Arid Climate: Mineral de La Reforma, Mexico. Water 2021, 13, 45. [Google Scholar] [CrossRef]
- Hernández-Magaña, R.; Hernández-Oria, J.G.; Chávez, R. Floristic Conservation Data Based on the Geographical Extent of the Species in the Semidesert Queretano, Mexico. Acta Bot. Mex. 2012, 99, 105–140. [Google Scholar] [CrossRef]
- Carlos-Hernández, S.; Carrillo-Parra, A.; Díaz-Jiménez, L.; Salas-Cruz, L.R.; Rosales-Serna, R.; Ngangyo-Heya, M. Transformation Processes for Energy Production Alternatives from Different Biomass Sources in the Highlands and Semi-Desert Areas of Mexico. Resources 2023, 12, 103. [Google Scholar] [CrossRef]
- Estrada-Castillón, E.; Villarreal-Quintanilla, J.Á.; Encina-Domínguez, J.A.; Jurado-Ybarra, E.; Cuéllar-Rodríguez, L.G.; Garza-Zambrano, P.; Arévalo-Sierra, J.R.; Cantú-Ayala, C.M.; Himmelsbach, W.; Salinas-Rodríguez, M.M.; et al. Ethnobotanical Biocultural Diversity by Rural Communities in the Cuatrociénegas Valley, Coahuila; Mexico. J. Ethnobiol. Ethnomed. 2021, 17, 21. [Google Scholar] [CrossRef] [PubMed]
- Ascacio-Valdés, J.A.; Aguilera-Carbó, A.; Rodríguez-Herrera, R.; Aguilar-González, C. Análisis de Ácido Elágico En Algunas Plantas Del Semidesierto Mexicano. Rev. Mex. Cienc. Farm. 2013, 44, 36–40. [Google Scholar]
- Armijo-Nájera, G.M.; Moreno-Reséndez, A.; Blanco-Contreras, E.; Borroel-García, J.V.; Reyes-Carrillo, L.J. Mesquite Pod (Prosopis Spp.) Food for Goats in the Semi-Desert. Rev. Mex. Cienc. Agric. 2019, 10, 113–122. [Google Scholar]
- Torres-León, C.; Rebolledo Ramírez, F.; Aguirre-Joya, J.A.; Ramírez-Moreno, A.; Chávez-González, M.L.; Aguillón-Gutierrez, D.R.; Camacho-Guerra, L.; Ramírez-Guzmán, N.; Hernández Vélez, S.; Aguilar, C.N. Medicinal Plants Used by Rural Communities in the Arid Zone of Viesca and Parras Coahuila in Northeast Mexico. Saudi Pharm. J. 2023, 31, 21–28. [Google Scholar] [CrossRef] [PubMed]
- González Elizondo, M.; López Enriquez, I.L.; González Elizondo, M.S.; Tena Flores, J.A. Plantas Medicinales Del Estado de Durango y Zonas Aledañas; CIIDIR Durango, Ed.; Instituto Politécnico Nacional: Durango, México, 2004. [Google Scholar]
- Dimayuga, R.E.; Virgen, M.; Ochoa, N. Antimicrobial Activity of Medicinal Plants from Baja California Sur (Mexico). Pharm. Biol. 1998, 36, 33–43. [Google Scholar] [CrossRef]
- Zahedi, S.M.; Karimi, M.; Venditti, A. Plants Adapted to Arid Areas: Specialized Metabolites. Nat. Prod. Res. 2021, 35, 3314–3331. [Google Scholar] [CrossRef] [PubMed]
- López-Romero, J.C.; Torres-Moreno, H.; Ireta-Paredes, A.d.R.; Charles-Rodríguez, A.V.; Flores-López, M.L. Chemical and Bioactive Compounds from Mexican Desertic Medicinal Plants. In Aromatic and Medicinal Plants of Drylands and Deserts: Ecology, Ethnobiology, and Potential Uses; CRC Press: Boca Raton, FL, USA, 2023; pp. 189–218. [Google Scholar]
- Zhang, Y.; Xu, J.; Li, R.; Ge, Y.; Li, Y.; Li, R. Plants’ Response to Abiotic Stress: Mechanisms and Strategies. Int. J. Mol. Sci. 2023, 24, 10915. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, L.F.; Figueroa, G. Fitoquímica; UNAM; FES Zaragoza: Ciudad de México, Mexico, 2022; Volume 1, ISBN 978-607-30-6019-6. [Google Scholar]
- Guía-García, J.L.; Charles-Rodríguez, A.V.; Reyes-Valdés, M.H.; Ramírez-Godina, F.; Robledo-Olivo, A.; García-Osuna, H.T.; Cerqueira, M.A.; Flores-López, M.L. Micro and Nanoencapsulation of Bioactive Compounds for Agri-Food Applications: A Review. Ind. Crops Prod. 2022, 186, 115198. [Google Scholar] [CrossRef]
- Jasso de Rodríguez, D.; Torres-Moreno, H.; López-Romero, J.C.; Vidal-Gutiérrez, M.; Villarreal-Quintanilla, J.A.; Carrillo-Lomelí, D.A.; Robles-Zepeda, R.E.; Vilegas, W. Antioxidant, Anti-Inflammatory, and Antiproliferative Activities of Flourensia spp. Biocatal. Agric. Biotechnol. 2023, 47, 102552. [Google Scholar] [CrossRef]
- Pranskuniene, Z.; Balciunaite, R.; Simaitiene, Z.; Bernatoniene, J. Herbal Medicine Uses for Respiratory System Disorders and Possible Trends in New Herbal Medicinal Recipes during COVID-19 in Pasvalys District, Lithuania. Int. J. Environ. Res. Public Health 2022, 19, 8905. [Google Scholar] [CrossRef]
- Lammi, C.; Arnoldi, A. Food-Derived Antioxidants and COVID-19. J. Food Biochem. 2021, 45, e13557. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Yarmolinsky, L.; Porat, D.; Dahan, A. Antiviral Effect of Phytochemicals from Medicinal Plants: Applications and Drug Delivery Strategies. Drug Deliv. Transl. Res. 2020, 10, 354–367. [Google Scholar] [CrossRef]
- Pagliaro, B.; Santolamazza, C.; Simonelli, F.; Rubattu, S. Phytochemical Compounds and Protection from Cardiovascular Diseases: A State of the Art. BioMed Res. Int. 2015, 2015, 918069. [Google Scholar] [CrossRef]
- Guan, R.; Van Le, Q.; Yang, H.; Zhang, D.; Gu, H.; Yang, Y.; Sonne, C.; Lam, S.S.; Zhong, J.; Jianguang, Z.; et al. A Review of Dietary Phytochemicals and Their Relation to Oxidative Stress and Human Diseases. Chemosphere 2021, 271, 129499. [Google Scholar] [CrossRef]
- Aminian, A.R.; Mohebbati, R.; Boskabady, M.H. The Effect of Ocimum basilicum L. and Its Main Ingredients on Respiratory Disorders: An Experimental, Preclinical, and Clinical Review. Front. Pharmacol. 2022, 12, 805391. [Google Scholar] [CrossRef] [PubMed]
- Vicidomini, C.; Roviello, V.; Roviello, G.N. In Silico Investigation on the Interaction of Chiral Phytochemicals from Opuntia Ficus-Indica with SARS-CoV-2 Mpro. Symmetry 2021, 13, 1041. [Google Scholar] [CrossRef]
- Vijakumaran, U.; Goh, N.-Y.; Razali, R.A.; Abdullah, N.A.H.; Yazid, M.D.; Sulaiman, N. Role of Olive Bioactive Compounds in Respiratory Diseases. Antioxidants 2023, 12, 1140. [Google Scholar] [CrossRef]
- Yumura, M.; Nagano, T.; Nishimura, Y. Novel Multitarget Therapies for Lung Cancer and Respiratory Disease. Molecules 2020, 25, 3987. [Google Scholar] [CrossRef]
- Zong, X.; Liang, N.; Wang, J.; Li, H.; Wang, D.; Chen, Y.; Zhang, H.; Jiao, L.; Li, A.; Wu, G.; et al. Treatment Effect of Qingfei Paidu Decoction Combined With Conventional Treatment on COVID-19 Patients and Other Respiratory Diseases: A Multi-Center Retrospective Case Series. Front. Pharmacol. 2022, 13, 849598. [Google Scholar] [CrossRef]
- WHO. Un Nuevo Informe Insta a Actuar Con Urgencia Para Prevenir Una Crisis Causada Por La Resistencia a Los Antimicrobianos. Available online: https://www.who.int/es/news/item/29-04-2019-new-report-calls-for-urgent-action-to-avert-antimicrobial-resistance-crisis (accessed on 21 January 2024).
- Derouiche, S. Oxidative Stress Associated with SARS-CoV-2 (COVID-19) Increases the Severity of the Lung Disease—A Systematic Review. J. Infect. Dis. Epidemiol. 2020, 6, 121–126. [Google Scholar] [CrossRef]
- Koya, T.; Hasegawa, T. Aggravation of Asthma by Cold, Fatigue, Stress, or Discontinuation of Medicines: What Should We Measures and Prevents Worse of Asthma Control Induced by the Aggravation of the Environmental Hygiene and/or the Stopping Medicine? In Disaster and Respiratory Diseases; Fujimoto, K., Ed.; Series: Diagnostic Tools and Disease Managements; Springer: Singapore, 2019; pp. 67–78. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, S.; Chen, Z.; Zu, B.; Zhao, Y. Effects of Variations in Meteorological Factors on Daily Hospital Visits for Asthma: A Time-Series Study. Environ. Res. 2020, 182, 109115. [Google Scholar] [CrossRef]
- Kobayashi, S. Exacerbation of COPD by Air Pollution, Cold Temperatures, or Discontinuation of Medicine: What Should Be Measured to Help Prevent It? In Disaster and Respiratory Diseases; Fujimoto, K., Ed.; Respiratory Disease Series: Diagnostic Tools and Disease Managements; Springer: Singapore, 2019; pp. 79–90. [Google Scholar] [CrossRef]
- Ostridge, K.; Gove, K.; Paas, K.H.W.; Burke, H.; Freeman, A.; Harden, S.; Kirby, M.; Peterson, S.; Sieren, J.; McCrae, C.; et al. Using Novel Computed Tomography Analysis to Describe the Contribution and Distribution of Emphysema and Small Airways Disease in Chronic Obstructive Pulmonary Disease. Ann. Am. Thorac. Soc. 2019, 16, 990–997. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.J.; Sethi, S.; Murphy, T.; Nariya, S.; Boushey, H.A.; Lynch, S.V. Airway Microbiome Dynamics in Exacerbations of Chronic Obstructive Pulmonary Disease. J. Clin. Microbiol. 2014, 52, 2813–2823. [Google Scholar] [CrossRef] [PubMed]
- Schabath, M.B.; Cote, M.L. Cancer Progress and Priorities: Lung Cancer. Cancer Epidemiol. Biomark. Prev. 2019, 28, 1563–1579. [Google Scholar] [CrossRef]
- Aredo, J.V.; Luo, S.J.; Gardner, R.M.; Sanyal, N.; Choi, E.; Hickey, T.P.; Riley, T.L.; Huang, W.Y.; Kurian, A.W.; Leung, A.N.; et al. Tobacco Smoking and Risk of Second Primary Lung Cancer. J. Thorac. Oncol. 2021, 16, 968–979. [Google Scholar] [CrossRef]
- Ho, L.J.; Yang, H.Y.; Chung, C.H.; Chang, W.C.; Yang, S.S.; Sun, C.A.; Chien, W.C.; Su, R.Y. Increased Risk of Secondary Lung Cancer in Patients with Tuberculosis: A Nationwide, Population-Based Cohort Study. PLoS ONE 2021, 16, e0250531. [Google Scholar] [CrossRef]
- Fujimoto, D.; Yomota, M.; Sekine, A.; Morita, M.; Morimoto, T.; Hosomi, Y.; Ogura, T.; Tomioka, H.; Tomii, K. Nivolumab for Advanced Non-Small Cell Lung Cancer Patients with Mild Idiopathic Interstitial Pneumonia: A Multicenter, Open-Label Single-Arm Phase II Trial. Lung Cancer 2019, 134, 274–278. [Google Scholar] [CrossRef]
- Herrera-Lara, S.; Fernández-Fabrellas, E.; Cervera-Juan, Á.; Blanquer-Olivas, R. Do Seasonal Changes and Climate Influence the Etiology of Community Acquired Pneumonia? Arch. Bronconeumol. 2013, 49, 140–145. [Google Scholar] [CrossRef]
- Kharwadkar, S.; Attanayake, V.; Duncan, J.; Navaratne, N.; Benson, J. The Impact of Climate Change on the Risk Factors for Tuberculosis: A Systematic Review. Environ. Res. 2022, 212, 113436. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Garcia, M.L.; Gonzalez-Carrasco, E.; Bracamonte, T.; Molinero, M.; Pozo, F.; Casas, I.; Calvo, C. Impact of Prematurity and Severe Viral Bronchiolitis on Asthma Development at 6–9 Years. J. Asthma Allergy 2020, 13, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Leimanis, M.L.; Adams, M.; Bachmann, A.S.; Uhl, K.L.; Bupp, C.P.; Hartog, N.L.; Kort, E.J.; Olivero, R.; Comstock, S.S.; et al. Balancing Precision versus Cohort Transcriptomic Analysis of Acute and Recovery Phase of Viral Bronchiolitis. Am. J. Physiol. Lung Cell Mol. Physiol. 2021, 320, L1147–L1157. [Google Scholar] [CrossRef]
- Nowicki, J.; Murray, M.T. Bronchitis and Pneumonia. In Textbook of Natural Medicine; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1196–1201. [Google Scholar] [CrossRef]
- Passos, S.D.; Gazeta, R.E.; Felgueiras, A.P.; Beneli, P.C.; Coelho, M.D.S.Z.S. Do Pollution and Climate Influence Respiratory Tract Infections in Children? Rev. Assoc. Med. Bras. 2014, 60, 276–283. [Google Scholar] [CrossRef][Green Version]
- Bocanegra-García, V.; Del Rayo Camacho-Corona, M.; Ramírez-Cabrera, M.; Rivera, G.; Garza-Gonzlez, E. The Bioactivity of Plant Extracts against Representative Bacterial Pathogens of the Lower Respiratory Tract. BMC Res. Notes 2009, 2, 95. [Google Scholar] [CrossRef]
- Szewczyk, K.; Zidorn, C. Ethnobotany, Phytochemistry, and Bioactivity of the Genus Turnera (Passifloraceae) with a Focus on Damiana—Turnera diffusa. J. Ethnopharmacol. 2014, 152, 424–443. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Pawar, R.S.; Ali, Z.; Khan, I.A. Phytochemical Investigation of Turnera diffusa. J. Nat. Prod. 2007, 70, 289–292. [Google Scholar] [CrossRef]
- Serrano-Gallardo, L.-B.; Castillo-Maldonado, I.; Borjón-Ríos, C.-G.; Rivera-Guillén, M.-A.; Morán-Martínez, J.; Téllez-López, M.-A.; García-Salcedo, J.-J.; Pedroza-Escobar, D.; Vega-Menchaca, M. del C. Antimicrobial Activity and Toxicity of Plants from Northern Mexico; NISCAIR-CSIR: New Delhi, India, 2017; Volume 16. [Google Scholar]
- WHO. 2020 Respiratory Diseases. Available online: https://platform.who.int/mortality/themes/theme-details/topics/topic-details/MDB/respiratory-diseases (accessed on 27 May 2023).
- Datos Abiertos Dirección General de Epidemiología|Secretaría de Salud|Gobierno|Gob.Mx. Available online: https://www.gob.mx/salud/documentos/datos-abiertos-152127 (accessed on 27 May 2023).
- Ramírez-Moreno, A.; Delgadillo-Guzmán, D.; Bautista-Robles, V.E.; Marszalek, J.; Keita, H.; Kourouma, A.; Ramírez-García, S.A.; Rodríguez Amado, J.R.; Tavares-Carvalho, J.C. Jatropha Dioica, an Aztec Plant with Promising Pharmacological Properties: A Systematic Review. Afr. J. Pharm. Pharmacol. 2020, 14, 169–178. [Google Scholar] [CrossRef]
- UNAM. Biblioteca Digital de La Medicina Tradicional Mexicana. 2010. Available online: http://www.medicinatradicionalmexicana.unam.mx/apmtm/termino.php?l=3&t=jatropha-dioica (accessed on 28 May 2023).
- Basilio Heredia, J.; Gutiérrez-Grijalva, E.P.; Angulo-Escalante, M.A.; Soto-Landeros, F. Recent Studies on Jatropha Research; Plant Science Research and Practices; Nova Science Publishers: Hauppauge, NY, USA, 2021; ISBN 9781536194944. [Google Scholar]
- Silva-Belmares, Y.; Rivas-Morales, C.; Viveros-Valdez, E.; de la Cruz-Galicia, M.G.; Carranza-Rosales, P. Antimicrobial and Cytotoxic Activities from Jatropha dioica Roots. Pak. J. Biol. Sci. 2014, 17, 748–750. [Google Scholar] [CrossRef] [PubMed]
- Guzmán, L.M.; Albarado, I.L.; Betancourt, J.; Medina, B. Bacterias Patógenas En Infecciones Del Tracto Respiratorio: Servicio Autónomo Hospital Universitario “Antonio Patricio de Alcalá”. Cumaná, Estado Sucre. Kasmera 2005, 33, 16–26. [Google Scholar]
- Urbizu-González, A.L.; Castillo-Ruiz, O.; Martínez-Ávila, G.C.G.; Torres-Castillo, J.A. Natural Variability of Essential Oil and Antioxidants in the Medicinal Plant Turnera diffusa. Asian Pac. J. Trop. Med. 2017, 10, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Yu, N.; Sun, Y.T.; Su, X.M.; He, M.; Dai, B.; Kang, J. Eucalyptol Protects Lungs against Bacterial Invasion through Attenuating Ciliated Cell Damage and Suppressing MUC5AC Expression. J. Cell Physiol. 2019, 234, 5842–5850. [Google Scholar] [CrossRef]
- Snowden, R.; Harrington, H.; Morrill, K.; Jeane, L.D.; Garrity, J.; Orian, M.; Lopez, E.; Rezaie, S.; Hassberger, K.; Familoni, D.; et al. A Comparison of the Anti-Staphylococcus aureus Activity of Extracts from Commonly Used Medicinal Plants. J. Altern. Complement. Med. 2014, 20, 375–382. [Google Scholar] [CrossRef]
- Pivard, M.; Moreau, K.; Vandenesch, F. Staphylococcus aureus Arsenal to Conquer the Lower Respiratory Tract. mSphere 2021, 6, 10–1128. [Google Scholar] [CrossRef]
- Núñez-Mojica, G.; Vázquez-Ramírez, A.L.; García, A.; Rivas-Galindo, V.M.; Garza-González, E.; Cuevas González-Bravo, G.E.; Toscano, R.A.; Moo-Puc, R.E.; Villanueva-Toledo, J.R.; Marchand, P.; et al. New Cyclolignans of Larrea Tridentata and Their Antibacterial and Cytotoxic Activities. Phytochem. Lett. 2021, 43, 212–218. [Google Scholar] [CrossRef]
- Reyes-Melo, K.Y.; Galván-Rodrigo, A.A.; Martínez-Olivo, I.E.; Núñez-Mojica, G.; Ávalos-Alanís, F.G.; García, A.; del Rayo Camacho-Corona, M. Larrea tridentata and Its Biological Activities. Curr. Top. Med. Chem. 2021, 21, 2352–2364. [Google Scholar] [CrossRef]
- Favela-Hernández, J.M.J.; García, A.; Garza-González, E.; Rivas-Galindo, V.M.; Camacho-Corona, M.R. Antibacterial and Antimycobacterial Lignans and Flavonoids from Larrea Tridentata. Phytother. Res. 2012, 26, 1957–1960. [Google Scholar] [CrossRef]
- Pimienta-Barrios, E. Prickly Pear (Opuntia spp.): A Valuable Fruit Crop for the Semi-Arid Lands of Mexico. J. Arid. Environ. 1994, 28, 1–11. [Google Scholar] [CrossRef]
- Aruwa, C.E.; Amoo, S.O.; Kudanga, T. Opuntia (Cactaceae) Plant Compounds, Biological Activities and Prospects—A Comprehensive Review. Food Res. Int. 2018, 112, 328–344. [Google Scholar] [CrossRef] [PubMed]
- Elkady, W.M.; Bishr, M.M.; Abdel-Aziz, M.M.; Salama, O.M. Identification and Isolation of Anti-Pneumonia Bioactive Compounds from: Opuntia ficus-indica Fruit Waste Peels. Food Funct. 2020, 11, 5275–5283. [Google Scholar] [CrossRef] [PubMed]
- Aranda-Ledesma, N.E.; González-Hernández, M.D.; Rojas, R.; Paz-González, A.D.; Rivera, G.; Luna-Sosa, B.; Martínez-Ávila, G.C.G. Essential Oil and Polyphenolic Compounds of Flourensia Cernua Leaves: Chemical Profiling and Functional Properties. Agronomy 2022, 12, 2274. [Google Scholar] [CrossRef]
- Alvarez-Pérez, O.B.; Ventura-Sobrevilla, J.M.; Ascacio-Valdés, J.A.; Rojas, R.; Verma, D.K.; Aguilar, C.N. Valorization of Flourensia cernua DC as Source of Antioxidants and Antifungal Bioactives. Ind. Crops Prod. 2020, 152, 112422. [Google Scholar] [CrossRef]
- GLOBOCAN 2020—Oncologia.Mx. Available online: https://oncologia.mx/tag/globocan-2020/ (accessed on 5 April 2023).
- Molina-Salinas, G.M.; Ramos-Guerra, M.C.; Vargas-Villarreal, J.; Mata-Cárdenas, B.D.; Becerril-Montes, P.; Said-Fernández, S. Bactericidal Activity of Organic Extracts from Flourensia Cernua DC against Strains of Mycobacterium tuberculosis. Arch. Med. Res. 2006, 37, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Nevárez-Prado, L.O.; Rocha-Gutiérrez, B.A.; Neder-Suárez, D.; Cordova-Lozoya, M.T.; Ayala-Soto, J.G.; Salazar-Balderrama, M.I.; de Ruiz-Anchondo, T.J.; Hernández-Ochoa, L.R. The Genus Fouquieria: Description and Review of Ethnobotanical, Phytochemical, and Biotechnological Aspects. Tecnociencia Chihuah. 2021, 15, 186–220. [Google Scholar] [CrossRef]
- López-Romero, J.C.; Torres-Moreno, H.; Rodríguez-Martínez, K.L.; Ramírez-Audelo, V.; Vidal-Gutiérrez, M.; Hernández, J.; Robles-Zepeda, R.E.; Ayala-Zavala, J.F.; González-Ríos, H.; Valenzuela-Melendres, M. Fouquieria splendens: A Source of Phenolic Compounds with Antioxidant and Antiproliferative Potential. Eur. J. Integr. Med. 2022, 49, 102084. [Google Scholar] [CrossRef]
- Rodríguez Garza, R.G. Tamizaje Fitoquímico y Actividad Biológica de Fouquieria splendens (Engelmann), Ariocarpus retusus (Scheidweiler) y Ariocarpus kotschoubeyanus (Lemaire). Ph.D. Thesis, Universidad Autónoma de Nuevo León, Nuevo León, México, 2010. [Google Scholar]
- Monreal-García, H.M.; Almaraz-Abarca, N.; Ávila-Reyes, J.A.; Torres-Ricario, R.; González-Elizondo, M.S.; Herrera-Arrieta, Y.; Gutiérrez-Velázquez, M.V. Phytochemical Variation among Populations of Fouquieria splendens (Fouquieriaceae). Bot. Sci. 2019, 97, 398–412. [Google Scholar] [CrossRef]
- Vega Menchaca, M.D.C.; Rivas Morales, C.; Verde Star, J.; Oranday, C.A.; Rubio Morales, M.E.; Núñez González, M.A.; Serrano Gallardo, L.B. Antimicrobial Activity of Five Plants from Northern Mexico on Medically Important Bacteria. Afr. J. Microbiol. Res. 2013, 7, 5011–5017. [Google Scholar] [CrossRef]
- Nevárez-Prado, L.O.; Amaya-Olivas, N.; Sustaita-Rodriguez, A.; Rodríguez-Zapién, J.; Zúñiga-Rodríguez, E.; Cordova-Lozoya, M.; García-Triana, A.; Sandoval-Salas, F.; Hernández-Ochoa, L. Chemical Composition and Toxicity of Extracts of Fouquieria splendens. AIMS Agric. Food 2022, 7, 357–369. [Google Scholar] [CrossRef]
- Orozco Meléndez, L.R.; García Muñoz, S.A.; Leyva Chávez, A.N.; González Aldana, R.A.; Villalobos Pérez, E.; Yáñez Muñoz, R.M. Insecticidal Properties of Secondary Metabolites of Fouquieria splendens Engelm (Ocotillo). Biol. Agropecu. Tuxpan 2017, 6, 1763–1774. [Google Scholar]
- Zhou, Z.; Ma, C.; Zhang, H.; Bi, Y.; Chen, X.; Tian, H.; Xie, X.; Meng, Q.; Lewis, P.J.; Xu, J. Synthesis and Biological Evaluation of Novel Ocotillol-Type Triterpenoid Derivatives as Antibacterial Agents. Eur. J. Med. Chem. 2013, 68, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.; Ma, C.; Zhou, Z.; Zhang, T.; Zhang, H.; Zhang, X.; Lu, J.; Meng, Q.; Lewis, P.J.; Xu, J. Synthesis and Antibacterial Evaluation of Novel Hydrophilic Ocotillol-Type Triterpenoid Derivatives from 20(S)-Protopanaxadiol. Rec. Nat. Prod. 2015, 9, 356–368. [Google Scholar]
- Akihisa, T.; Tokuda, H.; Ukiya, M.; Suzuki, T.; Enjo, F.; Koike, K.; Nikaido, T.; Nishino, H. 3-Epicabraleahydroxylactone and Other Triterpenoids from Camellia Oil and Their Inhibitory Effects on Epstein–Barr Virus Activation. Chem. Pharm. Bull. 2004, 52, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Morales-Cepeda, A.; Macclesh del Pino-Pérez, L.A.; Marmolejo, M.; Rivera-Armenta, J.L.; Peraza-Vázquez, H. Isolation of Ocotillol/Ocotillone from Fouquieria splendens (Ocote) Using a Batch Reactor. Prep. Biochem. Biotechnol. 2022, 52, 540–548. [Google Scholar] [CrossRef]
- Abdelmoteleb, A.; Valdez-Salas, B.; Ceceña-Duran, C.; Tzintzun-Camacho, O.; Gutiérrez-Miceli, F.; Grimaldo-Juarez, O.; González-Mendoza, D. Silver Nanoparticles from Prosopis glandulosa and Their Potential Application as Biocontrol of Acinetobacter Calcoaceticus and Bacillus Cereus. Chem. Speciat. Bioavailab. 2017, 29, 1–5. [Google Scholar] [CrossRef]
- SEINet Portal Network—Prosopis Glandulosa. Available online: https://swbiodiversity.org (accessed on 20 November 2023).
- Felker, P.; Takeoka, G.; Dao, L. Pod Mesocarp Flour of North and South American Species of Leguminous Tree Prosopis (Mesquite): Composition and Food Applications. Food Rev. Int. 2013, 29, 49–66. [Google Scholar] [CrossRef]
- Rahman, A.A.; Samoylenko, V.; Jacob, M.R.; Sahu, R.; Jain, S.K.; Khan, S.I.; Tekwani, B.L.; Muhammad, I. Antiparasitic and Antimicrobial Indolizidines from the Leaves of Prosopis glandulosa Var glandulosa. Planta Med. 2011, 77, 1639–1643. [Google Scholar] [CrossRef]
- Kumar Raju, S.; Shridharshini, K.; Mohanapriya, K.; Praveen, S.; Maruthamuthu, M.; Anajana, E.; Mythili, A. An Updated Review on Phytochemical Composition and Pharmacological Activities of Prosopis Glandulosa Torr.: An Invasive Exotic Plant. Indian J. Nat. Sci. 2022, 13, 46100–46110. [Google Scholar]
- Samoylenko, V.; Ashfaq, M.K.; Jacob, M.R.; Tekwani, B.L.; Khan, S.I.; Manly, S.P.; Joshi, V.C.; Walker, L.A.; Muhammad, I. Indolizidine, Antiinfective and Antiparasitic Compounds from Prosopis glandulosa Torr. Var. glandulosa. Planta Med. 2009, 75, 399–457. [Google Scholar] [CrossRef]
- Kumar, R.S.; Rajkapoor, B.; Perumal, P.; Dhanasekaran, T.; Jose, M.A.; Jothimanivannan, C. Antitumor Activity of Prosopis Glandulosa Torr. on Ehrlich Ascites Carcinoma (EAC) Tumor Bearing Mice. Iran. J. Pharm. Res. 2011, 10, 505. [Google Scholar]
- Moorthy, K.; Kumar, R.S. Phytochemical and Antimicrobial Studies of Leaf Extract of Prosopis glandulosa. J. Ecotoxicol. Environ. Monit. 2011, 21, 143. [Google Scholar]
- Ali, M.S.; Azhar, I.; Ahmad, F.; Ahmad, V.U.; Usmanghani, K. Antimicrobial Screening of Mimoaceous Plants. Pharm. Biol. 2001, 39, 43–46. [Google Scholar] [CrossRef]
- Imam, R.; Rafiq, M.; Sheng, Z.; Naqvi, S.H.A.; Talpur, F.N.; Abdelkhalek, A.; Jokhio, M.A. Evaluation of Physicochemical Properties and Antimicrobial Activity of Essential Oils from Seeds of Prosopis juliflora, P. Glandulosa and P. Cineraria. J. Essent. Oil Bear. Plants 2019, 22, 554–562. [Google Scholar] [CrossRef]
- López-Anchondo, A.N.; López-de la Cruz, D.; Gutiérrez-Reyes, E.; Castañeda-Ramírez, J.C.; De la Fuente-Salcido, N.M. Antifungal Activity In Vitro and In Vivo of Mesquite Extract (Prosopis glandulosa) against Phytopathogenic Fungi. Indian J. Microbiol. 2021, 61, 85–90. [Google Scholar] [CrossRef]
- Gupta, A.; Chaphalkar, S.R. Virucidal Potential of Prosopis spicigera and Mangifera indica on Human Peripheral Blood Mononuclear Cells. J. HerbMed Pharmacol. 2016, 5, 162–165. [Google Scholar]
- Patel, N.; Sharath Kumar, L.; Gajera, J.; Jadhav, A.; Muguli, G.; Babu, U. Isolation and Characterization of Flavonoid C-Glycosides from Prosopis glandulosa Torr. Leaves. Pharmacogn. Mag. 2018, 14, 451–454. [Google Scholar] [CrossRef]
- Odoh, U.E.; Uzor, P.F.; Eze, C.L.; Akunne, T.C.; Onyegbulam, C.M.; Osadebe, P.O. Medicinal Plants Used by the People of Nsukka Local Government Area, South-Eastern Nigeria for the Treatment of Malaria: An Ethnobotanical Survey. J. Ethnopharmacol. 2018, 218, 1–15. [Google Scholar] [CrossRef]
- Samoylenko, V.; Chuck Dunbar, D.; Jacob, M.R.; Joshi, V.C.; Ashfaq, M.K.; Muhammad, I. Two New Alkylated Piperidine Alkaloids from Western Honey Mesquite: Prosopis Glandulosa Torr. Var. Torreyana. Nat. Prod. Commun. 2008, 3, 35–40. [Google Scholar] [CrossRef]
- Ashfaq, M.K.; Abdel-Bakky, M.S.; Maqbool, M.T.; Samoylenko, V.; Rahman, A.A.; Muhammad, I. Efficacy of Prosopilosidine from Prosopis Glandulosa Var. Glandulosa against Cryptococcus Neoformans Infection in a Murine Model. Molecules 2018, 23, 1674. [Google Scholar] [CrossRef] [PubMed]
- Alamgir, A.N.M. Phytoconstituents—Active and Inert Constituents, Metabolic Pathways, Chemistry and Application of Phytoconstituents, Primary Metabolic Products, and Bioactive Compounds of Primary Metabolic Origin. In Therapeutic Use of Medicinal Plants and their Extracts: Volume 2. Progress in Drug Research; Springer: Cham, Switzerland, 2018; Volume 74, pp. 25–164. [Google Scholar] [CrossRef]
- Al Aboody, M.S.; Mickymaray, S. Anti-Fungal Efficacy and Mechanisms of Flavonoids. Antibiotics 2020, 9, 45. [Google Scholar] [CrossRef]
- Lee, Y.S.; Yang, W.K.; Park, Y.R.; Park, Y.C.; Park, I.J.; Lee, G.J.; Kang, H.S.; Kim, B.K.; Kim, S.H. Opuntia ficus-indica Alleviates Particulate Matter 10 Plus Diesel Exhaust Particles (PM10D)—Induced Airway Inflammation by Suppressing the Expression of Inflammatory Cytokines and Chemokines. Plants 2022, 11, 520. [Google Scholar] [CrossRef]
- Yoo, G.; Oh, Y.; Yang, H.; Kim, T.; Sung, S.; Kim, S. Efficient Preparation of Narcissin from Opuntia ficus-indica Fruits by Combination of Response Surface Methodology and High-Speed Countercurrent Chromatography. Pharmacogn. Mag. 2018, 14, 338–343. [Google Scholar] [CrossRef]
- Linares-Braham, A.; Palomo-Ligas, L.; Nery-Flores, S.D. Bioactive Compounds and Pharmacological Potential of Hojasen (Flourensia cernua): A Mini Review. Plant Sci. Today 2023, 10, 304–312. [Google Scholar] [CrossRef]
- Mansouri, Z.; Dianat, M.; Radan, M.; Badavi, M. Ellagic Acid Ameliorates Lung Inflammation and Heart Oxidative Stress in Elastase-Induced Emphysema Model in Rat. Inflammation 2020, 43, 1143–1156. [Google Scholar] [CrossRef] [PubMed]
- Morales-Ubaldo, A.L.; Rivero-Perez, N.; Valladares-Carranza, B.; Madariaga-Navarrete, A.; Higuera-Piedrahita, R.I.; Delgadillo-Ruiz, L.; Bañuelos-Valenzuela, R.; Zaragoza-Bastida, A. Phytochemical Compounds and Pharmacological Properties of Larrea Tridentata. Molecules 2022, 27, 5393. [Google Scholar] [CrossRef]
- Marin-Tinoco, R.I.; Ortega-Ramírez, A.T.; Esteban-Mendez, M.; Silva-Marrufo, O.; Barragan-Ledesma, L.E.; Valenzuela-Núñez, L.M.; Briceño-Contreras, E.A.; Sariñana-Navarrete, M.A.; Camacho-Luis, A.; Navarrete-Molina, C. Antioxidant and Antibacterial Activity of Mexican Oregano Essential Oil, Extracted from Plants Occurring Naturally in Semiarid Areas and Cultivated in the Field and Greenhouse in Northern Mexico. Molecules 2023, 28, 6547. [Google Scholar] [CrossRef] [PubMed]
- Man, A.; Santacroce, L.; Jacob, R.; Mare, A.; Man, L. Antimicrobial Activity of Six Essential Oils against a Group of Human Pathogens: A Comparative Study. Pathogens 2019, 8, 15. [Google Scholar] [CrossRef]
- Hernández, T.; Canales, M.; Duran, A.; María García, A.; Guillermo Avila, J.; Hernández-Portilla, L.; Alvarado, M.; Romero, M.; Terán, B.; Dávila, P.; et al. Variation in the Hexanic Extract Composition of Lippia graveolens in an Arid Zone from Mexico: Environmental Influence or True Chemotypes? Open Plant Sci. J. 2009, 3, 29–34. [Google Scholar] [CrossRef]
- Lee, S.Y.; Bae, C.S.; Choi, Y.H.; Seo, N.S.; Na, C.S.; Yoo, J.C.; Cho, S.S.; Park, D.H. Opuntia Humifusa Modulates Morphological Changes Characteristic of Asthma via Il-4 and Il-13 in an Asthma Murine Model. Food Nutr. Res. 2017, 61, 1393307. [Google Scholar] [CrossRef]
- Martins, S.; Teixeira, J.A.; Mussatto, S.I. Solid-State Fermentation as a Strategy to Improve the Bioactive Compounds Recovery from Larrea Tridentata Leaves. Appl. Biochem. Biotechnol. 2013, 171, 1227–1239. [Google Scholar] [CrossRef]
- Loukili, E.H.; Bouchal, B.; Bouhrim, M.; Abrigach, F.; Genva, M.; Zidi, K.; Bnouham, M.; Bellaoui, M.; Hammouti, B.; Addi, M.; et al. Chemical Composition, Antibacterial, Antifungal and Antidiabetic Activities of Ethanolic Extracts of Opuntia dillenii Fruits Collected from Morocco. J. Food Qual. 2022, 2022, 9471239. [Google Scholar] [CrossRef]
- Wong-Paz, J.E.; Contreras-Esquivel, J.C.; Rodríguez-Herrera, R.; Carrillo-Inungaray, M.L.; López, L.I.; Nevárez-Moorillón, G.V.; Aguilar, C.N. Total Phenolic Content, in Vitro Antioxidant Activity and Chemical Composition of Plant Extracts from Semiarid Mexican Region. Asian Pac. J. Trop. Med. 2015, 8, 104–111. [Google Scholar] [CrossRef]
- Jakovljević, M.; Jokić, S.; Molnar, M.; Jašić, M.; Babić, J.; Jukić, H.; Banjari, I. Bioactive Profile of Various Salvia officinalis L. Preparations. Plants 2019, 8, 55. [Google Scholar] [CrossRef]
- Rahman, F.; Tabrez, S.; Ali, R.; Alqahtani, A.S.; Ahmed, M.Z.; Rub, A. Molecular Docking Analysis of Rutin Reveals Possible Inhibition of SARS-CoV-2 Vital Proteins. J. Tradit. Complement. Med. 2021, 11, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Mata, R.; Figueroa, M.; Navarrete, A.; Rivero-Cruz, I. Chemistry and Biology of Selected Mexican Medicinal Plants. In Progress in the Chemistry of Organic Natural Products; Springer: Cham, Switzerland, 2019; Volume 108, pp. 1–142. [Google Scholar]
- García-Andrade, M.; González-Laredo, R.F.; Rocha-Guzmán, N.E.; Gallegos-Infante, J.A.; Rosales-Castro, M.; Medina-Torres, L. Mesquite Leaves (Prosopis laevigata), a Natural Resource with Antioxidant Capacity and Cardioprotection Potential. Ind. Crops Prod. 2013, 44, 336–342. [Google Scholar] [CrossRef]
- González-Mendoza, D.; Troncoso-Rojas, R.; Gonzalez-Soto, T.; Grimaldo-Juarez, O.; Ceceña-Duran, C.; Duran-Hernandez, D.; Gutierrez-Miceli, F. Changes in the Phenylalanine Ammonia Lyase Activity, Total Phenolic Compounds, and Flavonoids in Prosopis glandulosa Treated with Cadmium and Copper. An. Acad. Bras. Cienc. 2018, 90, 1465–1472. [Google Scholar] [CrossRef] [PubMed]
- Arteaga, S.; Andrade-Cetto, A.; Cárdenas, R. Larrea Tridentata (Creosote Bush), an Abundant Plant of Mexican and US-American Deserts and Its Metabolite Nordihydroguaiaretic Acid. J. Ethnopharmacol. 2005, 98, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Govea-Salas, M.; Morlett-Chávez, J.; Rodriguez-Herrera, R.; Ascacio-Valdés, J. Some Mexican Plants Used in Traditional Medicine. In Aromatic and Medicinal Plants—Back to Nature; InTech: Houston, TX, USA, 2017. [Google Scholar]
- Favela-Hernández, J.M.J.; Clemente-Soto, A.F.; Balderas-Rentería, I.; Garza-González, E.; Camacho-Corona, M.D.R. Potential Mechanism of Action of 3′-Demethoxy-6-O-Demethylisoguaiacin on Methicillin Resistant Staphylococcus aureus. Molecules 2015, 20, 12450–12458. [Google Scholar] [CrossRef] [PubMed]
- Porras, G.; Chassagne, F.; Lyles, J.T.; Marquez, L.; Dettweiler, M.; Salam, A.M.; Samarakoon, T.; Shabih, S.; Farrokhi, D.R.; Quave, C.L. Ethnobotany and the Role of Plant Natural Products in Antibiotic Drug Discovery. Chem. Rev. 2021, 121, 3495–3560. [Google Scholar] [CrossRef] [PubMed]
- Seol, G.H.; Kim, K.Y. Eucalyptol and Its Role in Chronic Diseases. In Advances in Experimental Medicine and Biology; Springer: Cham, Switzerland, 2016; Volume 929, pp. 389–398. [Google Scholar] [CrossRef]
- Rivero-Cruz, I.; Duarte, G.; Navarrete, A.; Bye, R.; Linares, E.; Mata, R. Chemical Composition and Antimicrobial and Spasmolytic Properties of Poliomintha longiflora and Lippia graveolens Essential Oils. J. Food Sci. 2011, 76, C309–C317. [Google Scholar] [CrossRef] [PubMed]
- Kheiry, M.; Dianat, M.; Badavi, M.; Mard, S.A.; Bayati, V. P-Coumaric Acid Attenuates Lipopolysaccharide-Induced Lung Inflammation in Rats by Scavenging ROS Production: An In Vivo and In Vitro Study. Inflammation 2019, 42, 1939–1950. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Li, S.; Liu, X.; An, H.; Kang, X.; Guo, S. Caffeic Acid, an Active Ingredient in Coffee, Combines with DOX for Multitarget Combination Therapy of Lung Cancer. J. Agric. Food Chem. 2022, 70, 8326–8337. [Google Scholar] [CrossRef] [PubMed]
- Pino, A.; Hernandez, J.I.; Roncal, E. Comparison of Isolation Procedures for Mexican Oregano Oil. Food/Nahr. 1990, 34, 825–830. [Google Scholar] [CrossRef]
- Liu, M.; Niu, W.; Ou, L. β-Caryophyllene Ameliorates the Mycoplasmal Pneumonia through the Inhibition of NF-ΚB Signal Transduction in Mice. Saudi J. Biol. Sci. 2021, 28, 4240–4246. [Google Scholar] [CrossRef] [PubMed]
- Estell, R.E.; Havstad, K.M.; Fredrickson, E.L.; Gardea-Torresdey, J.L. Secondary Chemistry of the Leaf Surface of Flourensia cernua. Biochem. Syst. Ecol. 1994, 22, 73–77. [Google Scholar] [CrossRef]
- Perez Gutierrez, R.M.; Mota Flores, J.M. Petiveria Alliacea Suppresses Airway Inflammation and Allergen-Specific Th2 Responses in Ovalbumin-Sensitized Murine Model of Asthma. Chin. J. Integr. Med. 2018, 24, 912–919. [Google Scholar] [CrossRef]
- Molina-Salinas, G.M.; Pérez-López, A.; Becerril-Montes, P.; Salazar-Aranda, R.; Said-Fernández, S.; Torres, N.W. de Evaluation of the Flora of Northern Mexico for in Vitro Antimicrobial and Antituberculosis Activity. J. Ethnopharmacol. 2007, 109, 435–441. [Google Scholar] [CrossRef]
- Wächter, G.A.; Hoffmann, J.J.; Furbacher, T.; Blake, M.E.; Timmermann, B.N. Antibacterial and Antifungal Flavanones from Eysenhardtia texana. Phytochemistry 1999, 52, 1469–1471. [Google Scholar] [CrossRef]
- Riaz, B.; Sohn, S. Neutrophils in Inflammatory Diseases: Unraveling the Impact of Their Derived Molecules and Heterogeneity. Cells 2023, 12, 2621. [Google Scholar] [CrossRef]
- Alonso-Castro, A.J.; Zapata-Morales, J.R.; Ruiz-Padilla, A.J.; Solorio-Alvarado, C.R.; Rangel-Velázquez, J.E.; Cruz-Jiménez, G.; Orozco-Castellanos, L.M.; Domínguez, F.; Maldonado-Miranda, J.J.; Carranza-Álvarez, C.; et al. Use of Medicinal Plants by Health Professionals in Mexico. J. Ethnopharmacol. 2017, 198, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Abarca-Vargas, R.; Petricevich, V.L. Bougainvillea Genus: A Review on Phytochemistry, Pharmacology, and Toxicology. Evid.-Based Complement. Altern. Med. 2018, 2018, 9070927. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Lu, P.; Wu, H.; Liu, Z.; Sharifi-Rad, J.; Setzer, W.N.; Suleria, H.A.R. Current Insights into Phytochemistry, Nutritional, and Pharmacological Properties of Prosopis Plants. Evid.-Based Complement. Altern. Med. 2022, 2022, 2218029. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Franco, C.; Maldonado Aguirre, L.J. Overview of Past, Current and Potential Uses of Mesquite in Mexico. In Prosopis; Semiarid Fuelwood and Forage Tree Building Consensus for the Disenfranchised; U.S. National Academy of Sciences Building: Washington, DC, USA, 1996; Volume 13. [Google Scholar]
- Kaushik, V.; Niketan, S.; Sachdeva, S.; Saini, V. A Review on Phytochemical and Pharmacological Potential of Prosopis cineraria. Int. J. Ethnobiol. Ethnomed. 2020, 1, 1–4. [Google Scholar]
- Ammar, I.; Ennouri, M.; Bouaziz, M.; Ben Amira, A.; Attia, H. Phenolic Profiles, Phytchemicals and Mineral Content of Decoction and Infusion of Opuntia ficus-indica Flowers. Plant Foods Hum. Nutr. 2015, 70, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Fackler, C. Three Ethnobotanically Important Plants of Texas: Southern Prickly Ash, Ocotillo, and Jimson Weed. American Botanical Council. HerbalEGram 2016, 13. [Google Scholar]
- Villagomez-Ibarra, J.R.; Sánchez, M.; Espejo, O.; Zuniga-Estrada, A.; Torres-Valencia, J.M.; Joseph-Nathan, P. Antimicrobial Activity of Three Mexican Gnaphalium Species. Fitoterapia 2001, 72, 692–694. [Google Scholar] [CrossRef]
- Listados de Registros Sanitarios de Medicamentos|Comisión Federal Para La Protección Contra Riesgos Sanitarios|Gobierno|Gob.Mx. Available online: https://www.gob.mx/cofepris/documentos/registros-sanitarios-medicamentos (accessed on 17 January 2024).
- Mpiana, P.T.; Ngbolua, K.-T.; Tshibangu, D.S.; Kilembe, J.T.; Gbolo, B.Z.; Mwanangombo, D.T.; Inkoto, C.L.; Lengbiye, E.M.; Mbadiko, C.M.; Matondo, A.; et al. Identification of Potential Inhibitors of SARS-CoV-2 Main Protease from Aloe Vera Compounds: A Molecular Docking Study. Chem. Phys. Lett. 2020, 754, 137751. [Google Scholar] [CrossRef]
- Timalsina, D.; Pokhrel, K.P.; Bhusal, D. Pharmacologic Activities of Plant-Derived Natural Products on Respiratory Diseases and Inflammations. BioMed Res. Int. 2021, 2021, 1636816. [Google Scholar] [CrossRef]
- Mohammadi, Z.; Pishkar, L.; Eftekhari, Z.; Barzin, G.; Babaeekhou, L. The Human Host Defense Peptide LL-37 Overexpressed in Lung Cell Lines by Methanolic Extract of Valeriana officinalis. Braz. J. Pharm. Sci. 2023, 59, e21025. [Google Scholar] [CrossRef]
- Šutovská, M.; Capek, P.; Kočmalová, M.; Pawlaczyk, I.; Zaczyńska, E.; Czarny, A.; Uhliariková, I.; Gancarz, R.; Fraňová, S. Characterization and Pharmacodynamic Properties of Arnica Montana Complex. Int. J. Biol. Macromol. 2014, 69, 214–221. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).