The Effect of Phosphate on the Activity and Sensitivity of Nutritropism toward Ammonium in Rice Roots
Abstract
1. Introduction
2. Results
2.1. Screening of Rice Accessions Showing Nutritropism in the Main Roots
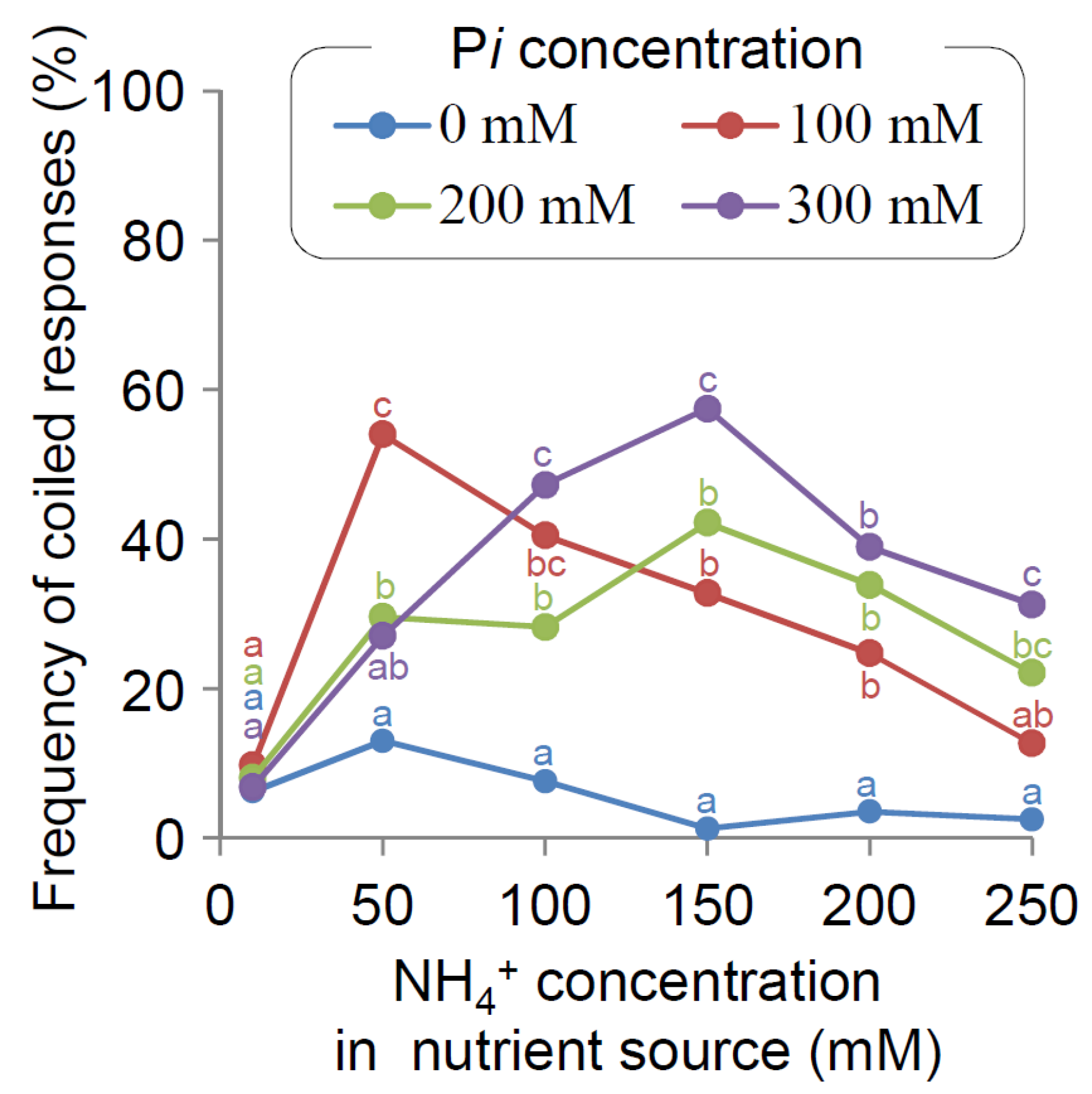
2.2. Major Effects of NH4+ and PO4− on Main Root Nutritropism
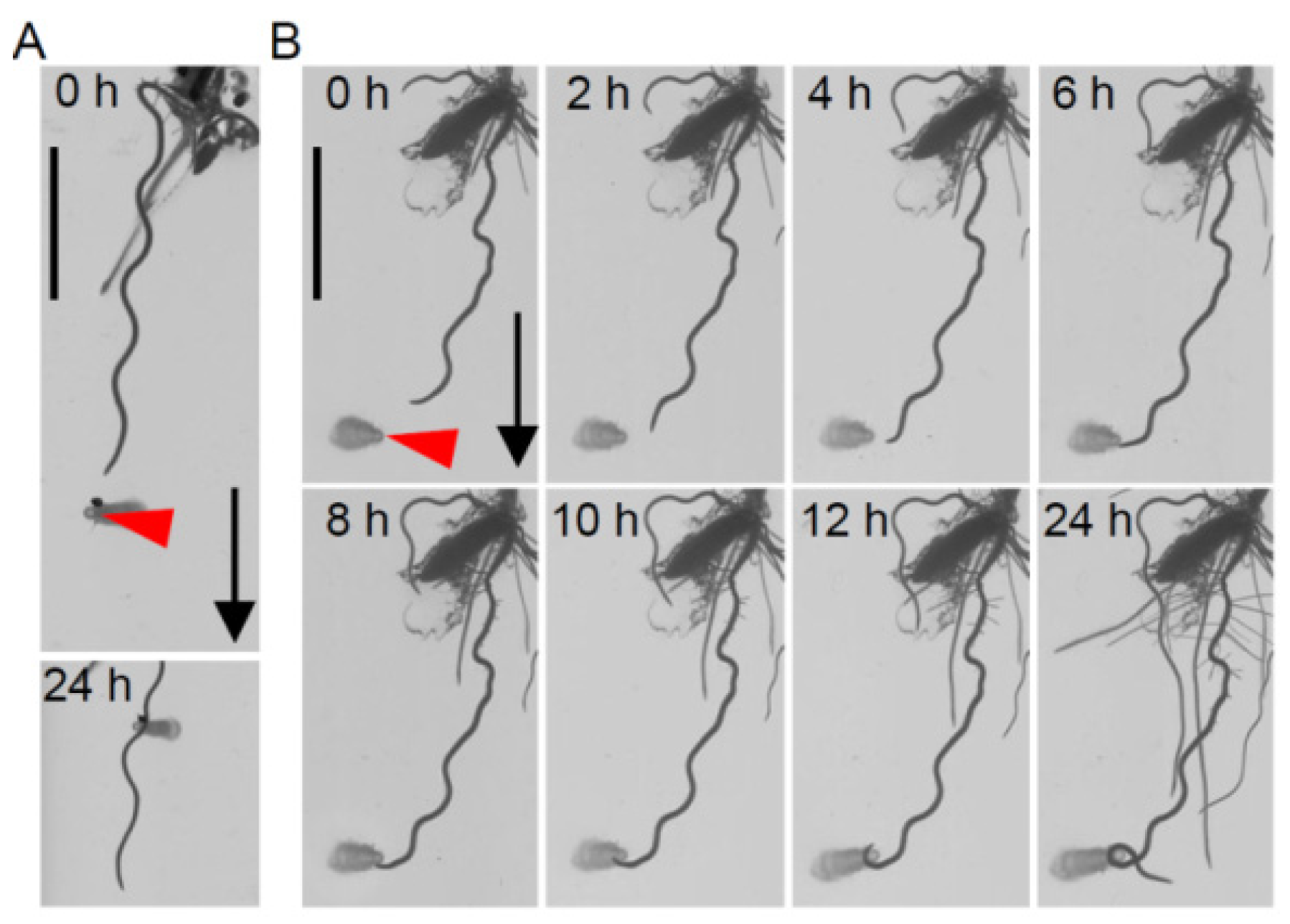
2.3. Activation of Main Root Nutritropism by Non-Gradient Pi
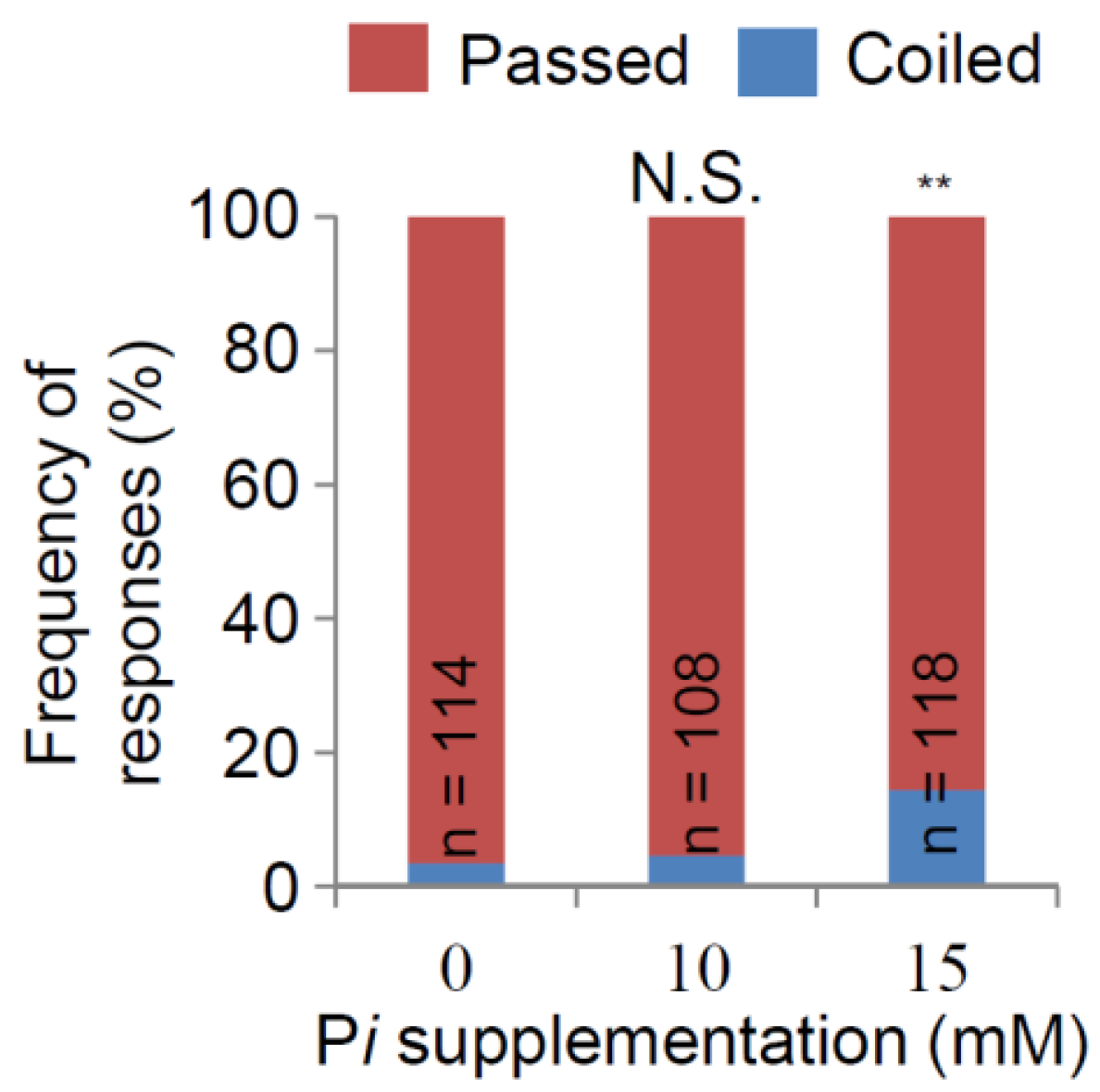
2.4. Pi Desensitization of Nutritropism
2.5. Nutritropic Response in WRC 1
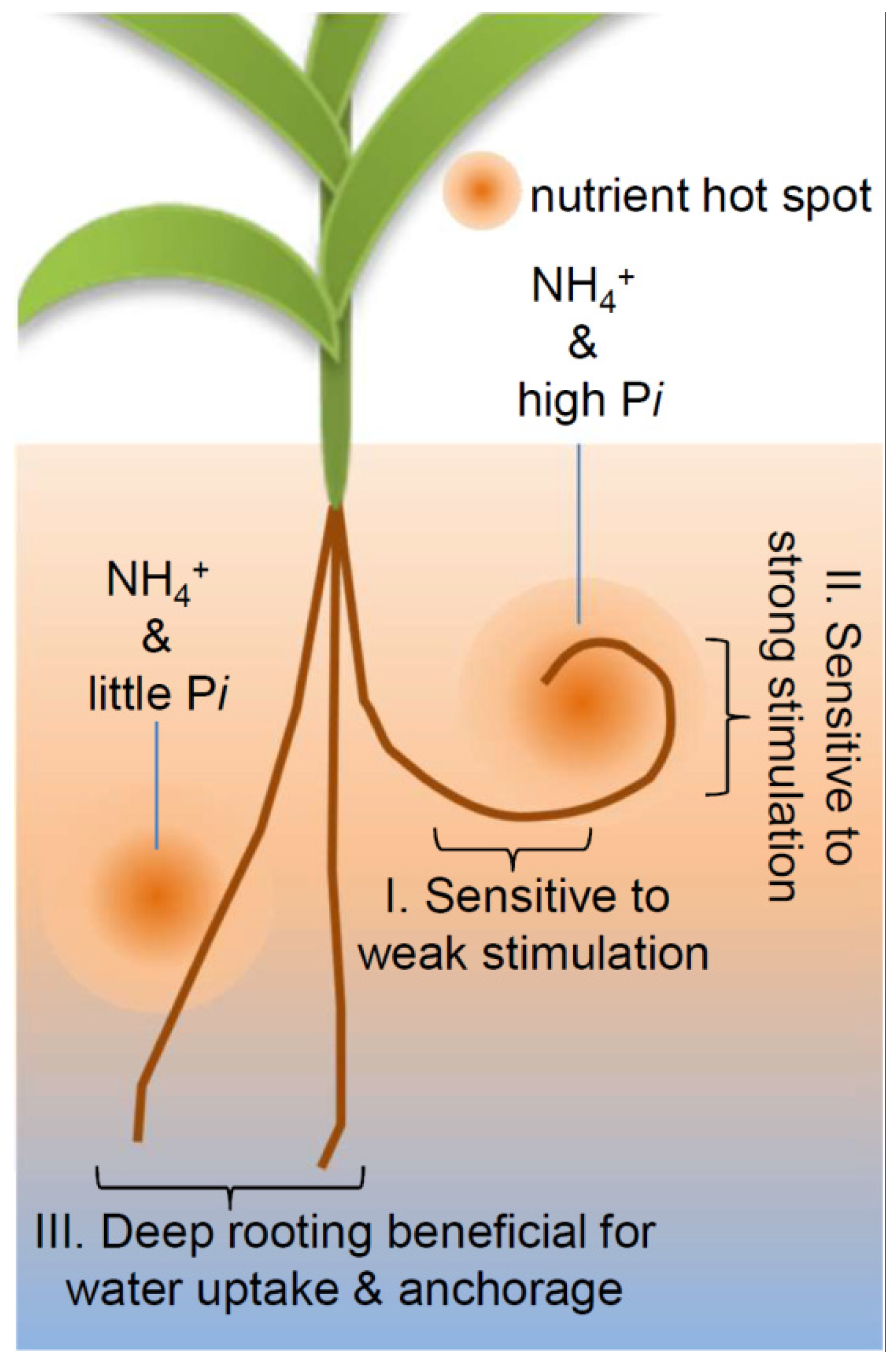
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. Preparation of Nutrient Sources for the Nutritropic Bioassay
4.3. Nutritropic Bioassay
4.4. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Masuda, S. Light detection and signal transduction in the BLUF photoreceptors. Plant Cell Physiol. 2013, 54, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Buckley, T.N. How do stomata respond to water status? New Phytol. 2019, 224, 21–36. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Mao, Y. Research advances in plant–insect molecular interaction. F1000Research 2020, 9, 198. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Rico-Medina, A.; Caño-Delgado, A.I. The physiology of plant responses to drought. Science 2020, 368, 266–269. [Google Scholar] [CrossRef] [PubMed]
- Poorter, H.; Niinemets, Ü.; Ntagkas, N.; Siebenkäs, A.; Mäenpää, M.; Matsubara, S.; Pons, T. A meta-analysis of plant responses to light intensity for 70 traits ranging from molecules to whole plant performance. New Phytol. 2019, 223, 1073–1105. [Google Scholar] [CrossRef] [PubMed]
- Oldroyd, G.E.; Leyser, O. A plant’s diet, surviving in a variable nutrient environment. Science 2020, 368, eaba0196. [Google Scholar] [CrossRef] [PubMed]
- Voss, I.; Sunil, B.; Scheibe, R.; Raghavendra, A. Emerging concept for the role of photorespiration as an important part of abiotic stress response. Plant Biol. 2013, 15, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Fukao, T.; Barrera-Figueroa, B.E.; Juntawong, P.; Peña-Castro, J.M. Submergence and Waterlogging Stress in Plants: A Review Highlighting Research Opportunities and Understudied Aspects. Front. Plant Sci. 2019, 10, 340. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T. Mineral deficiencies of plants. J. Inst. Brew. 1946, 52, 181–187. [Google Scholar] [CrossRef]
- Casal, J.J. Photoreceptor signaling networks in plant responses to shade. Annu. Rev. Plant Biol. 2013, 64, 403–427. [Google Scholar] [CrossRef]
- Giehl, R.F.; Gruber, B.D.; von Wirén, N. It’s time to make changes: Modulation of root system architecture by nutrient signals. J. Exp. Bot. 2014, 65, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Gemenet, D.C.; Villordon, A. Root system architecture and abiotic stress tolerance: Current knowledge in root and tuber crops. Front. Plant Sci. 2016, 7, 1584. [Google Scholar] [CrossRef] [PubMed]
- Darwin, C. The Power of Movement in Plants; D. Appleton and Company: London, UK, 1897. [Google Scholar]
- Liscum, E.; Askinosie, S.K.; Leuchtman, D.L.; Morrow, J.; Willenburg, K.T.; Coats, D.R. Phototropism: Growing towards an understanding of plant movement. Plant Cell 2014, 26, 38–55. [Google Scholar] [CrossRef] [PubMed]
- Muthert, L.W.F.; Izzo, L.G.; Van Zanten, M.; Aronne, G. Root tropisms: Investigations on earth and in space to unravel plant growth direction. Front. Plant Sci. 2020, 10, 1807. [Google Scholar] [CrossRef] [PubMed]
- Gilroy, S. Plant tropisms. Curr. Biol. 2008, 18, R275–R277. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, K.; Ohmori, Y.; Fujiwara, T. A positive tropism of rice roots toward a nutrient source. Plant Cell Physiol. 2020, 61, 546–553. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, J.A.; Rasmussen, A.; Traini, R.; Voß, U.; Sturrock, C.; Mooney, S.J.; Wells, D.M.; Bennett, M.J. Branching out in roots: Uncovering form, function, and regulation. Plant Physiol. 2014, 166, 538–550. [Google Scholar] [CrossRef] [PubMed]
- Hochholdinger, F.; Park, W.J.; Sauer, M.; Woll, K. From weeds to crops: Genetic analysis of root development in cereals. Trends Plant Sci. 2004, 9, 42–48. [Google Scholar] [CrossRef]
- Olatunji, D.; Geelen, D.; Verstraeten, I. Control of endogenous auxin levels in plant root development. Int. J. Mol. Sci. 2017, 18, 2587. [Google Scholar] [CrossRef]
- Giehl, R.F.; von Wirén, N. Root nutrient foraging. Plant Physiol. 2014, 166, 509–517. [Google Scholar] [CrossRef]
- Lima, J.E.; Kojima, S.; Takahashi, H.; von Wirén, N. Ammonium triggers lateral root branching in Arabidopsis in an AMMONIUM TRANSPORTER1; 3-dependent manner. Plant Cell 2010, 22, 3621–3633. [Google Scholar] [CrossRef] [PubMed]
- Linkohr, B.I.; Williamson, L.C.; Fitter, A.H.; Leyser, H.O. Nitrate and phosphate availability and distribution have different effects on root system architecture of Arabidopsis. Plant J. 2002, 29, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Forde, B.G. An Arabidopsis MADS box gene that controls nutrient-induced changes in root architecture. Science 1998, 279, 407–409. [Google Scholar] [CrossRef] [PubMed]
- Morohashi, K.; Okamoto, M.; Yamazaki, C.; Fujii, N.; Miyazawa, Y.; Kamada, M.; Kasahara, H.; Osada, I.; Shimazu, T.; Fusejima, Y. Gravitropism interferes with hydrotropism via counteracting auxin dynamics in cucumber roots: Clinorotation and spaceflight experiments. New Phytol. 2017, 215, 1476–1489. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Yamazaki, Y.; Kobayashi, A.; Higashitani, A.; Takahashi, H. Hydrotropism interacts with gravitropism by degrading amyloplasts in seedling roots of Arabidopsis and radish. Plant Physiol. 2003, 132, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Miyazawa, Y.; Fujii, N. Hormonal interactions during root tropic growth: Hydrotropism versus gravitropism. Plant Mol. Biol. 2009, 69, 489–502. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Adamowski, M.; Qi, L.; Alotaibi, S.S.; Friml, J. PIN-mediated polar auxin transport regulations in plant tropic responses. New Phytol. 2021, 232, 510–522. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Liang, W.; Sturrock, C.J.; Pandey, B.K.; Giri, J.; Mairhofer, S.; Wang, D.; Muller, L.; Tan, H.; York, L.M. Rice actin binding protein RMD controls crown root angle in response to external phosphate. Nat. Commun. 2018, 9, 2346. [Google Scholar] [CrossRef] [PubMed]
- Zou, N.; Li, B.; Dong, G.; Kronzucker, H.J.; Shi, W. Ammonium-induced loss of root gravitropism is related to auxin distribution and TRH1 function, and is uncoupled from the inhibition of root elongation in Arabidopsis. J. Exp. Bot. 2012, 63, 3777–3788. [Google Scholar] [CrossRef]
- Kojima, Y.; Ebana, K.; Fukuoka, S.; Nagamine, T.; Kawase, M. Development of an RFLP-based rice diversity research set of germplasm. Breed. Sci. 2005, 55, 431–440. [Google Scholar] [CrossRef]
- Jia, L.; Xie, Y.; Wang, Z.; Luo, L.; Zhang, C.; Pélissier, P.; Parizot, B.; Qi, W.; Zhang, J.; Hu, Z. Rice plants respond to ammonium stress by adopting a helical root growth pattern. Plant J. 2020, 104, 1023–1037. [Google Scholar] [CrossRef] [PubMed]
- Hanserud, O.S.; Lyng, K.; Vries, J.W.D.; Øgaard, A.F.; Brattebø, H. Redistributing phosphorus in animal manure from a livestock-intensive region to an arable region: Exploration of environmental consequences. Sustainability 2017, 9, 595. [Google Scholar] [CrossRef]
- Wang, T.; Montell, C. Phototransduction and retinal degeneration in Drosophila. Pflügers Arch. Eur. J. Physiol. 2007, 454, 821–847. [Google Scholar] [CrossRef] [PubMed]
- Everett, M. Dose-response curves for radish seedling phototropism. Plant Physiol. 1974, 54, 222–225. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Heathcote, D.; Bircher, B. Enhancement of phototropic response to a range of light doses in Triticum aestivum coleoptiles in clinostat-simulated microgravity. Planta 1987, 170, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Barbez, E.; Dünser, K.; Gaidora, A.; Lendl, T.; Busch, W. Auxin steers root cell expansion via apoplastic pH regulation in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2017, 114, E4884–E4893. [Google Scholar] [CrossRef] [PubMed]
- Meier, M.; Liu, Y.; Lay-Pruitt, K.S.; Takahashi, H.; von Wirén, N. Auxin-mediated root branching is determined by the form of available nitrogen. Nat. Plants 2020, 6, 1136–1145. [Google Scholar] [CrossRef] [PubMed]
- Blancaflor, E.; Fasano, J.; Gilroy, S. Laser ablation of root cap cells: Implications for models of graviperception. Adv. Space Res. 1999, 24, 731–738. [Google Scholar] [CrossRef]
- Fisher, R.A. Statistical methods for research workers. oliver and boyd, edinburgh (1925). Sci. Rep. 2020, 10, 21739. [Google Scholar]
- Holm, S. A simple sequentially rejective multiple test procedure. Scand. J. Stat. 1979, 6, 65–70. [Google Scholar]
- Hervé, M.; Hervé, M.M. Package ‘RVAideMemoire’. 2020. Available online: https://cran.r-project.org/web/packages/RVAideMemoire/index.html (accessed on 11 February 2022).

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamazaki, K.; Fujiwara, T. The Effect of Phosphate on the Activity and Sensitivity of Nutritropism toward Ammonium in Rice Roots. Plants 2022, 11, 733. https://doi.org/10.3390/plants11060733
Yamazaki K, Fujiwara T. The Effect of Phosphate on the Activity and Sensitivity of Nutritropism toward Ammonium in Rice Roots. Plants. 2022; 11(6):733. https://doi.org/10.3390/plants11060733
Chicago/Turabian StyleYamazaki, Kiyoshi, and Toru Fujiwara. 2022. "The Effect of Phosphate on the Activity and Sensitivity of Nutritropism toward Ammonium in Rice Roots" Plants 11, no. 6: 733. https://doi.org/10.3390/plants11060733
APA StyleYamazaki, K., & Fujiwara, T. (2022). The Effect of Phosphate on the Activity and Sensitivity of Nutritropism toward Ammonium in Rice Roots. Plants, 11(6), 733. https://doi.org/10.3390/plants11060733





