The Dynamics of Flower Development in Castanea sativa Mill.
Abstract
:1. Introduction
2. Results
2.1. Castanea sativa ‘Judia’ Cultivar Flowering Phenology
2.2. Castanea sativa De Novo Transcriptome Analysis
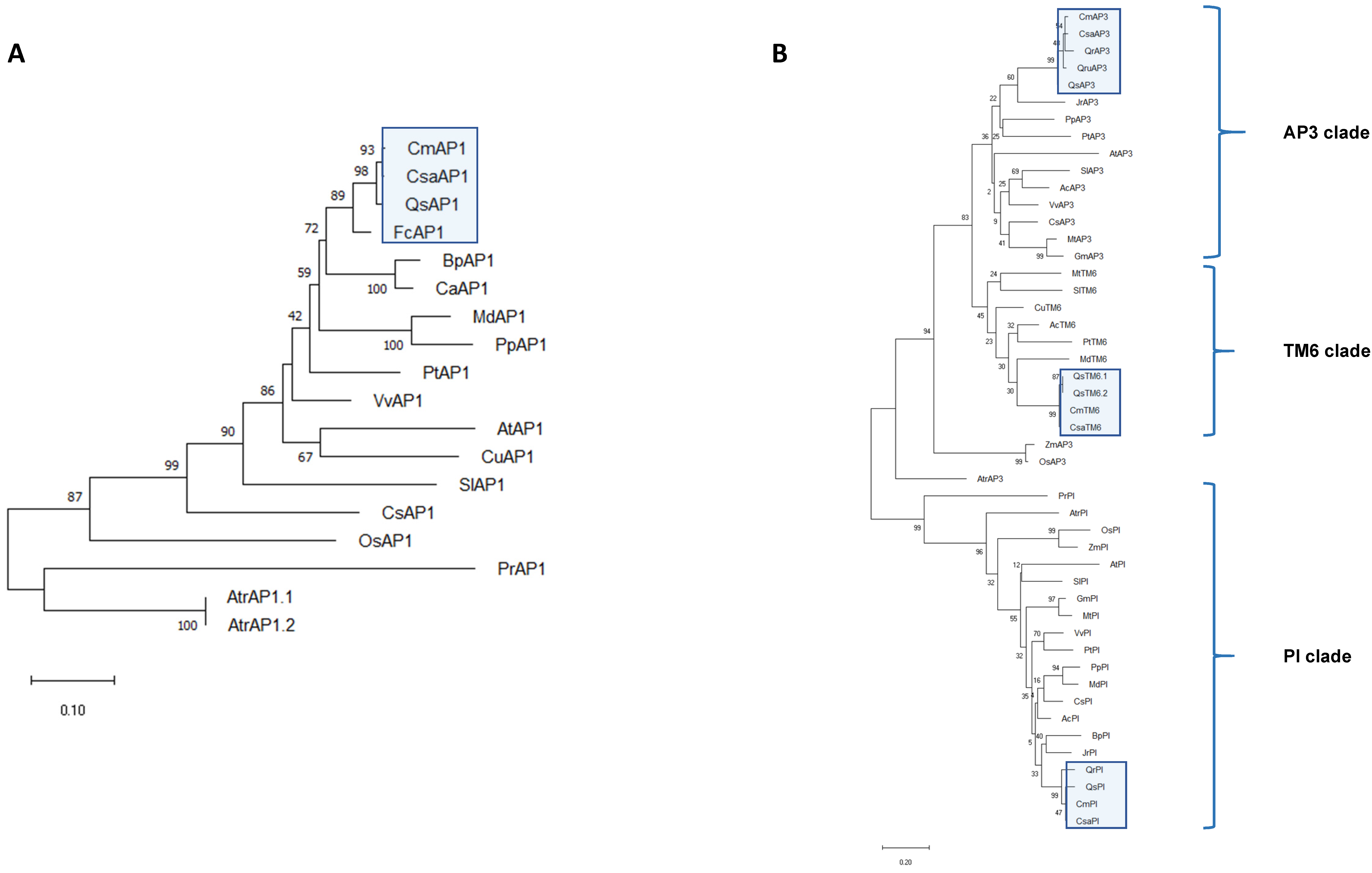
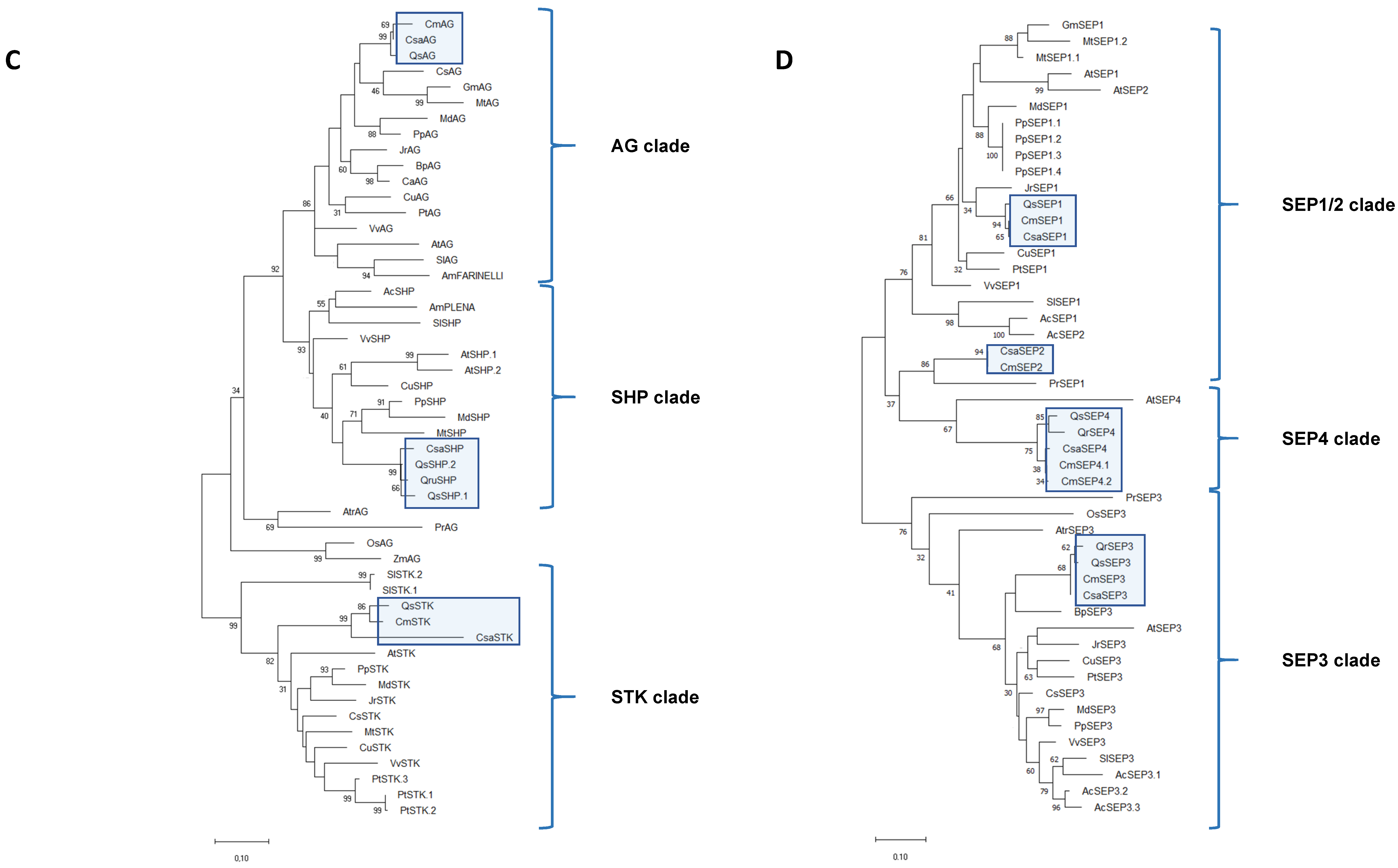
2.3. ABCDE MADS-Box Transcription Factors Were Conserved in Castanea sativa
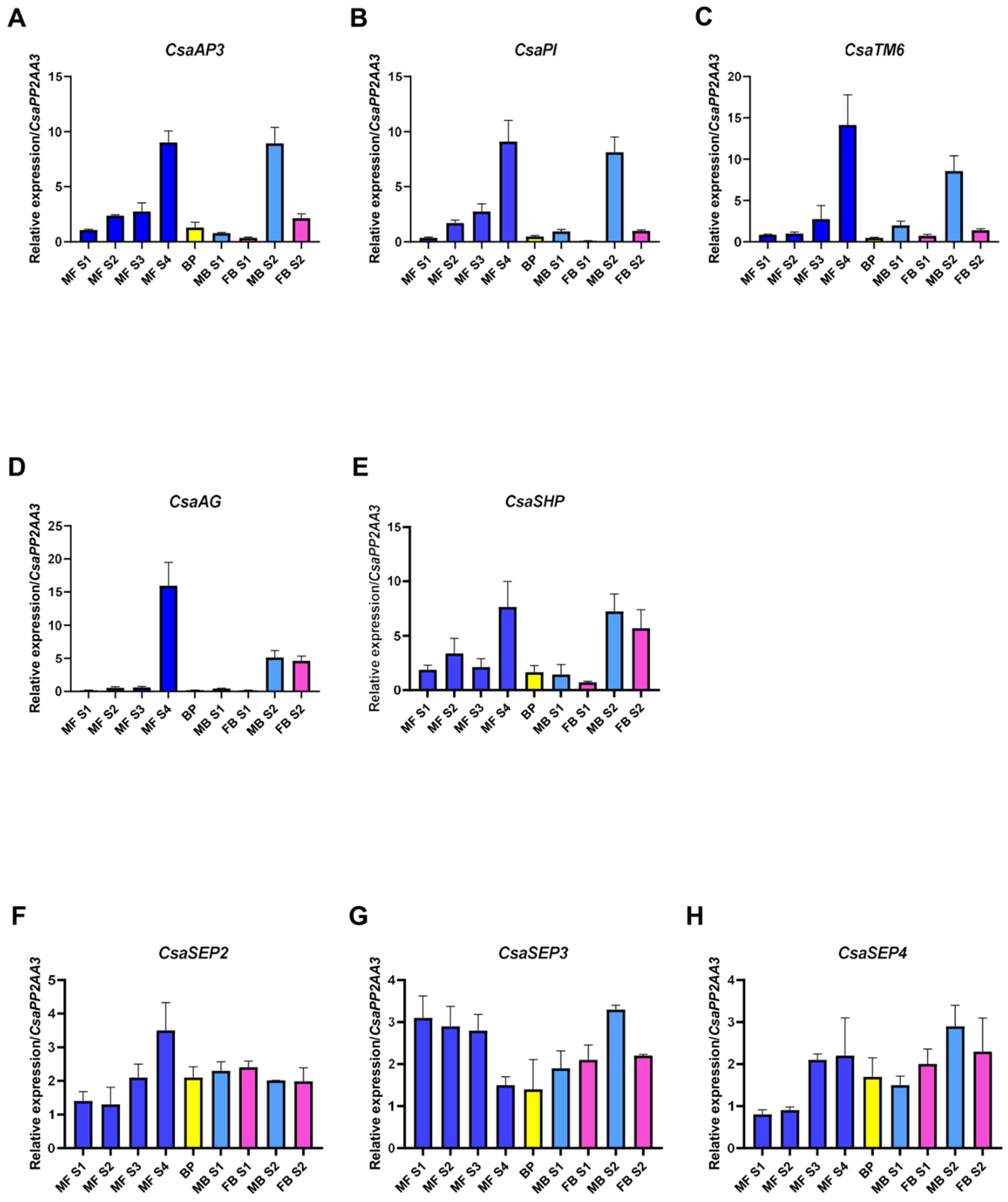
2.4. C. sativa BCE-Like Gene Expression in Unisexual and Bisexual Catkins
2.5. Dimerisation of C. sativa BCE-Like Proteins
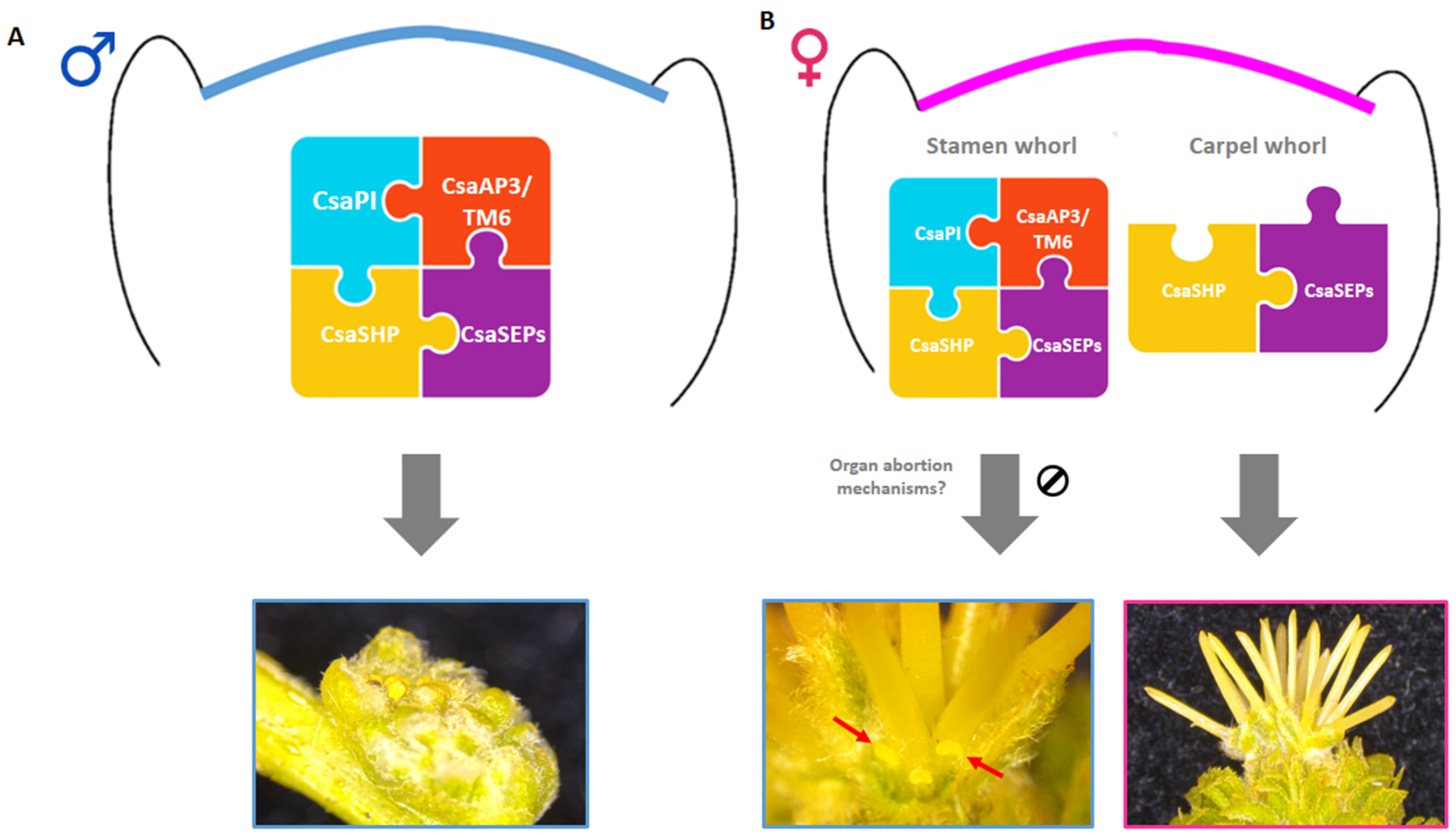
3. Discussion
4. Materials and Methods
4.1. De Novo Transcriptome Assembly
4.1.1. Plant Material
4.1.2. RNA Extraction
4.1.3. Library Preparation and Sequencing
4.1.4. De Novo Assembly of Castanea sativa Transcriptome
4.2. Phylogenetic Analysis
4.3. qRT-PCR Analysis
4.4. Histological Analysis
4.5. Yeast-2-Hybrid Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Charlesworth, B.; Charlesworth, D. A Model for the Evolution of Dioecy and Gynodioecy. Am. Nat. 1978, 112, 975–997. [Google Scholar] [CrossRef]
- Kremer, A.; Abbott, A.G.; Carlson, J.E.; Manos, P.S.; Plomion, C.; Sisco, P.; Staton, M.E.; Ueno, S.; Vendramin, G.G. Genomics of Fagaceae. Tree Genet. Genomes 2012, 8, 583–610. [Google Scholar] [CrossRef] [Green Version]
- Rocheta, M.; Sobral, R.; Magalhães, J.; Amorim, M.I.; Ribeiro, T.; Pinheiro, M.; Egas, C.; Morais-Cecílio, L.; Costa, M.M.R. Comparative transcriptomic analysis of male and female flowers of monoecious Quercus suber. Front. Plant Sci. 2014, 5, 1–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sobral, R.; Costa, M.M.R. Role of floral organ identity genes in the development of unisexual flowers of Quercus suber L. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Sobral, R.; Silva, H.G.; Laranjeira, S.; Magalhães, J.; Andrade, L.; Alhinho, A.T.; Costa, M.M.R. Unisexual flower initiation in the monoecious Quercus suber L.: A molecular approach. Tree Physiol. 2020, 40, 1260–1276. [Google Scholar] [CrossRef] [PubMed]
- Bowman, J.L.; Smyth, D.R.; Meyerowitz, E.M. Genes directing flower development in Arabidopsis. Plant Cell 1989, 1, 37–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meyerowitz, E.M.; Bowman, J.L.; Brockman, L.L.; Drews, G.N.; Jack, T.; Sieburth, L.E.; Weigel, D. A genetic and molecular model for flower development in Arabidopsis thaliana. Development 1991, 112, 157–167. [Google Scholar] [CrossRef]
- Bowman, J.L.; Alvarez, J.; Weigel, D.; Meyerowitz, E.M.; Smyth, D.R. Control of flower development in Arabidopsis thaliana by APETALA 1 and interacting genes. Development 1993, 119, 721–743. [Google Scholar] [CrossRef]
- Flanagan, C.A.; Hu, Y.; Ma, H. Specific expression of the AGL1 MADS-box gene suggests regulatory functions in Arabidopsis gynoecium and ovule development. Plant J. 1996, 10, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Pelaz, S.; Ditta, G.S.; Baumann, E.; Wisman, E.; Yanofsky, M.F. B and C floral organ identity functions require SEPALLATTA MADS-box genes. Nature 2000, 405, 200–203. [Google Scholar] [CrossRef] [PubMed]
- Liljegren, S.J.; Ditta, G.S.; Eshed, Y.; Savidge, B.; Bowmant, J.L.; Yanofsky, M.F. SHATTERPROOF MADS-box genes control dispersal in Arabidopsis. Nature 2000, 404, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Theißen, G.; Gramzow, L. Structure and Evolution of Plant MADS Domain Transcription Factors; Academic Press: Cambridge, MA, USA, 2016; ISBN 9780128011270. [Google Scholar]
- Theißen, G.; Melzer, R.; Ruümpler, F. MADS-domain transcription factors and the floral quartet model of flower development: Linking plant development and evolution. Development 2016, 143, 3259–3271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaufmann, K.; Melzer, R.; Theißen, G. MIKC-type MADS-domain proteins: Structural modularity, protein interactions and network evolution in land plants. Gene 2005, 347, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Trobner, W.; Ramirez, L.; Motte, P.; Hue, I.; Huijser, P.; Lonnig, W.E.; Saedler, H.; Sommer, H.; Schwarz-Sommer, Z. GLOBOSA: A homeotic gene which interacts with DEFICIENS in the control of Antirrhinum floral organogenesis. EMBO J. 1992, 11, 4693–4704. [Google Scholar] [CrossRef] [PubMed]
- McGonigle, B.; Bouhidel, K.; Irish, V.F. Nuclear localization of the Arabidopsis APETALA3 and PISTILLATA homeotic gene products depends on their simultaneous expression. Genes Dev. 1996, 10, 1812–1821. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riechmann, J.L.; Krizek, B.A.; Meyerowitz, E.M. Dimerization specificity of Arabidopsis MADS domain homeotic proteins APETALA1, APETALA3, PISTILLATA, and AGAMOUS. Proc. Natl. Acad. Sci. USA 1996, 93, 4793–4798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lenser, T.; Theißen, G.; Dittrich, P. Developmental robustness by obligate interaction of class B floral homeotic genes and proteins. PLoS Comput. Biol. 2009, 5. [Google Scholar] [CrossRef] [Green Version]
- Okamoto, M. New Interpretation of the Inflorescence of Fagus Drawn From the Developmental Study of Fagus Crenata, With Description of an Extremely Monstrous Cupule. Am. J. Bot. 1989, 76, 14–22. [Google Scholar] [CrossRef]
- Ducousso, A.; Michaud, H.; Lumaret, R. Reproduction and gene flow in the genus Quercus L. Ann. Des. Sci. For. 1993, 50, 91s–106s. [Google Scholar] [CrossRef] [Green Version]
- Varela, M.C.; Valdiviesso, T. Phenological phases of Quercus suber L. flowering. For. Genet. 1996, 3, 93–102. [Google Scholar]
- Langdon, L.M. Ontogenetic and Anatomical Studies of the Flower and Fruit of the Fagaceae and Juglandaceae. Bot. Gaz. 1939, 101, 301–327. [Google Scholar] [CrossRef]
- Kubitzki, K.; Fagaceae, B.T. Flowering Plants Dicotyledons: Magnoliid, Hamamelid and Caryophyllid Families; Kubitzki, K., Rohwer, J.G., Bittrich, V., Eds.; Springer: Berlin/Heidelberg, Germany, 1993; ISBN 978-3-662-02899-5. [Google Scholar]
- Mellano, M.G.; Beccaro, G.L.; Donno, D.; Marinoni, D.T.; Boccacci, P.; Canterino, S.; Cerutti, A.K.; Bounous, G. Castanea spp. biodiversity conservation: Collection and characterization of the genetic diversity of an endangered species. Genet. Resour. Crop Evol. 2012, 59, 1727–1741. [Google Scholar] [CrossRef]
- Bounous, G.; Marinoni, D.T. Chestnut: Botany, Horticulture, and Utilization. Hortic. Rev. (Am. Soc. Hortic. Sci). 2004, 291–347. [Google Scholar]
- Valdiviesso, T. Estudo Sobre a Reprodução Sexuada e Caracterização de Cultivares de Castanea sativa Mill.; Instituto Nacional de Investigação Agrária: Oeiras, Portugal, 1999. [Google Scholar] [CrossRef]
- Botta, R.; Vergano, G.; Me, G.; Vallania, R. Floral biology and embryo development in chestnut (Castanea sativa Mill.). HortScience 1995, 30, 1283–1286. [Google Scholar] [CrossRef] [Green Version]
- Feijó, J.A.; Certal, A.C.; Boavida, L.; Van Nerum, I.; Valdiviesso, T.; Oliveira, M.M.; Broothaerts, W. Advances on the Study of Sexual Reproduction in the Cork-Tree (Quercus suber L.), Chestnut (Castanea sativa Mill.) and in Rosaceae (Apple and Almond). Fertil. High. Plants 1999, 377–396. [Google Scholar] [CrossRef]
- Soylu, A.; Ayfer, M. Floral biology and fruit set of some chestnut cultivars (C. sativa Mill.). Proc. Int. Congr. Chestnut Spoleto Italy 1993, 28, 125–130. [Google Scholar]
- Dinis, L.T.; Ramos, S.; Gomes-Laranjo, J.; Peixoto, F.; Costa, R.; Vallania, R.; Botta, R. Phenology and reproductive biology in cultivar “Judia” (Castanea sativa Mill.). Acta Hortic. 2010, 866, 169–174. [Google Scholar] [CrossRef]
- Fang, G.C.; Blackmon, B.P.; Staton, M.E.; Nelson, C.D.; Kubisiak, T.L.; Olukolu, B.A.; Henry, D.; Zhebentyayeva, T.; Saski, C.A.; Cheng, C.H.; et al. A physical map of the Chinese chestnut (Castanea mollissima) genome and its integration with the genetic map. Tree Genet. Genomes 2013, 9, 525–537. [Google Scholar] [CrossRef] [Green Version]
- Xing, Y.; Liu, Y.; Zhang, Q.; Nie, X.; Sun, Y.; Zhang, Z.; Li, H.; Fang, K.; Wang, G.; Huang, H.; et al. Hybrid de novo genome assembly of Chinese chestnut (Castanea mollissima). Gigascience 2019, 8, 1–7. [Google Scholar] [CrossRef]
- Ainsworth, C.; Rahman, A.; Parker, J.; Edwards, G. Intersex inflorescences of Rumex acetosa demonstrate that sex determination is unique to each flower. New Phytol. 2005, 165, 711–720. [Google Scholar] [CrossRef]
- Di Stilio, V.S.; Kramer, E.M.; Baum, D.A. Floral MADS box genes and homeotic gender dimorphism in Thalictrum dioicum (Ranunculaceae)—A new model for the study of dioecy. Plant J. 2005, 41, 755–766. [Google Scholar] [CrossRef] [PubMed]
- Sather, D.N.; Jovanovic, M.; Golenberg, E.M. Functional analysis of B and C class floral organ genes in spinach demonstrates their role in sexual dimorphism. BMC Plant Biol. 2010, 10, 46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coutinho, X.P. Esboço de uma flora lenhosa Portuguesa. Publ. Serv. Florest. Port. 1936, 3, 60–62. [Google Scholar]
- Guerreiro, M.G. Alguns estudos no género Castanea. Publ. Serv. Florest. Port. 1948, 15, 10–15. [Google Scholar]
- Kientzler, L. Cas d’hermaphroditisme chez le châtaignier. Bull. Soc. Bot. France 1959, 106, 211–212. [Google Scholar] [CrossRef]
- Solignat, G. Recherches sur le châtaignier à la St. de Brive. INRA 1952, 3, 15–35. [Google Scholar]
- Solignat, G. Un renouveau de la châtaigneraie fruitière. INRA Cent. Rech. Agron. Bordeaux. Extr. Du Boll. Tech. D’Inf. 1973, 280, 280–281. [Google Scholar]
- Shi, Z.; Stösser, R. Reproductive biology of Chinese chestnut (Castanea mollissima Blume). Eur. J. Hortic. Sci. 2005, 70, 96–103. [Google Scholar]
- Guo, S.J.; Zou, F. Observation on the pistillate differentiation of Chestnut (Castanea) cultivar “Yanshanzaofeng”. J. Chem. Pharm. Res. 2014, 6, 686–690. [Google Scholar]
- Fan, X.; Yuan, D.; Tian, X.; Zhu, Z.; Liu, M.; Cao, H. Comprehensive Transcriptome Analysis of Phytohormone Biosynthesis and Signaling Genes in the Flowers of Chinese Chinquapin (Castanea henryi). J. Agric. Food Chem. 2017, 65, 10332–10349. [Google Scholar] [CrossRef] [PubMed]
- Ditta, G.; Pinyopich, A.; Robles, P.; Pelaz, S.; Yanofsky, M.F. The SEP4 Gene of Arabidopsis thaliana Functions in Floral Organ and Meristem Identity. Curr. Biol. 2004, 14, 1935–1940. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brunner, A.M.; Rottmann, W.H.; Sheppard, L.A.; Krutovskii, K.; DiFazio, S.P.; Leonardi, S.; Strauss, S.H. Structure and expression of duplicate AGAMOUS orthologues in poplar. Plant Mol. Biol. 2000, 44, 619–634. [Google Scholar] [CrossRef]
- Lemmetyinen, J.; Hassinen, M.; Elo, A.; Porali, I.; Keinonen, K.; Mäkelä, H.; Sopanen, T. Functional characterization of SEPALLATA3 and AGAMOUS orthologues in silver birch. Physiol. Plant. 2004, 121, 149–162. [Google Scholar] [CrossRef]
- Zhou, Y.; Xu, Z.; Yong, X.; Ahmad, S.; Yang, W.; Cheng, T.; Wang, J.; Zhang, Q. SEP-class genes in Prunus mume and their likely role in floral organ development. BMC Plant Biol. 2017, 17, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Favaro, R.; Pinyopich, A.; Battaglia, R.; Kooiker, M.; Borghi, L.; Ditta, G.; Yanofsky, M.F.; Kater, M.M.; Colombo, L. MADS-Box Protein Complexes Control Carpel and Ovule Development in Arabidopsis. Plant Cell 2003, 15, 2603–2611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, Z.; Zhuo, S.; Liu, X.; Che, G.; Wang, Z.; Gu, R.; Shen, J.; Song, W.; Zhou, Z.; Han, D.; et al. The MADS-Box Gene CsSHP Participates in Fruit Maturation and Floral Organ Development in Cucumber. Front. Plant Sci. 2020, 10, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fourquin, C.; Ferrándiz, C. Functional analyses of AGAMOUS family members in Nicotiana benthamiana clarify the evolution of early and late roles of C-function genes in eudicots. Plant J. 2012, 71, 990–1001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, Y.; Liu, W.; Chen, X.; Xu, Y.; Lu, W.; Hou, J.; Ni, J.; Wang, Y.; Wu, L. Flower development and sex determination between male and female flowers in Vernicia fordii. Front. Plant Sci. 2017, 8, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, M.; Li, W.; Zhao, G.; Fan, X.; Long, H.; Fan, Y.; Shi, M.; Tan, X.; Zhang, L. New Insights of Salicylic Acid into Stamen Abortion of Female Flowers in Tung Tree (Vernicia fordii). Front. Genet. 2019, 10, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Xu, Z.; Wang, Y.; Chen, Y.; Yin, H.; Wu, L.; Zhao, Y.; Wang, M.; Gao, M. A model of hormonal regulation of stamen abortion during pre-meiosis of Litsea cubeba. Genes 2020, 11, 48. [Google Scholar] [CrossRef] [Green Version]
- Ooi, S.E.; Sarpan, N.; Aziz, N.A.; Nuraziyan, A.; Nor-Azwani, A.B.; Ong-Abdullah, M. Transcriptomics of microdissected staminodes and early developing carpels from female inflorescences of Elaeis guineensis. J. Oil Palm Res. 2020, 32, 559–568. [Google Scholar] [CrossRef]
- Ferreira-cardoso, J.; Portela, E.; Abreu, C.G. Castanheiros; Universidade de Trás-os-Montes e Alto Douro: Vila Real, Portugal, 2007; ISBN 9789726698449. [Google Scholar]
- Chang, S.; Puryear, J.; Cairney, J. A simple and efficient method for isolating RNA from pine trees. Plant Mol. Biol. Rep. 1993, 11, 113–116. [Google Scholar] [CrossRef]
- Le Provost, G.; Herrera, R.; Paiva, J.A.; Chaumeil, P.; Salin, F.; Plomion, C. A micromethod for high throughput RNA extraction in forest trees. Biol. Res. 2007, 40, 291–297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serrazina, S.; Santos, C.; Machado, H.; Pesquita, C.; Vicentini, R.; Pais, M.S.; Sebastiana, M.; Costa, R. Castanea root transcriptome in response to Phytophthora cinnamomi challenge. Tree Genet. Genomes 2015, 11, 6. [Google Scholar] [CrossRef]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. Available online: http://www.bioinform.babraham.ac.uk/proj./Fastqc/2010 (accessed on 26 July 2021).
- Bushnell, B. BBMap: A Fast, Accurate, Splice-Aware Aligner. In Proceedings of the 9th Annual Genomics of Energy & Environment Meeting, Walnut Creek, CA, USA, 17–20 March 2014. [Google Scholar]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef] [Green Version]
- Haas, B.J.; Papanicolaou, A.; Yassour, M.; Grabherr, M.; Blood, P.D.; Bowden, J.; Couger, M.B.; Eccles, D.; Li, B.; Lieber, M.; et al. De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat. Protoc. 2013, 8, 1494–1512. [Google Scholar] [CrossRef] [PubMed]
- Smith-Unna, R.; Boursnell, C.; Patro, R.; Hibberd, J.M.; Kelly, S. TransRate: Reference-free quality assessment of de novo transcriptome assemblies. Genome Res. 2016, 26, 1134–1144. [Google Scholar] [CrossRef] [Green Version]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [Green Version]
- Fu, L.; Niu, B.; Zhu, Z.; Wu, S.; Li, W. CD-HIT: Accelerated for clustering the next-generation sequencing data. Bioinformatics 2012, 28, 3150–3152. [Google Scholar] [CrossRef]
- Simao, F.A.; Waterhouse, R.M.; Ioannidis, P.; Kriventseva, E.V.; Zdobnov, E.M. BUSCO online supplementary information: Assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 2015, 31, 3210–3212. [Google Scholar] [CrossRef] [Green Version]
- Götz, S.; García-Gómez, J.M.; Terol, J.; Williams, T.D.; Nagaraj, S.H.; Nueda, M.J.; Robles, M.; Talón, M.; Dopazo, J.; Conesa, A. High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic Acids Res. 2008, 36, 3420–3435. [Google Scholar] [CrossRef]
- Camacho, C.; Coulouris, G.; Avagyan, V.; Ma, N.; Papadopoulos, J.; Bealer, K.; Madden, T.L. BLAST+: Architecture and applications. BMC Bioinform. 2009, 10, 421. [Google Scholar] [CrossRef] [Green Version]
- Sievers, F.; Wilm, A.; Dineen, D.; Gibson, T.J.; Karplus, K.; Li, W.; Lopez, R.; McWilliam, H.; Remmert, M.; Söding, J.; et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 2011, 7, 539. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Marum, L.; Miguel, A.; Ricardo, C.P.; Miguel, C. Reference gene selection for quantitative real-time PCR normalization in Quercus suber. PLoS ONE 2012, 7, e35113. [Google Scholar] [CrossRef]
- Coen, E.S.; Romero, J.M.; Doyle, S.; Elliott, R.; Murphy, G.; Carpenter, R. floricaula: A homeotic gene required for flower development in antirrhinum majus. Cell 1990, 63, 1311–1322. [Google Scholar] [CrossRef]
- Viejo, M.; Rodríguez, R.; Valledor, L.; Pérez, M.; Cañal, M.J.; Hasbún, R. DNA methylation during sexual embryogenesis and implications on the induction of somatic embryogenesis in Castanea sativa Miller. Sex Plant Reprod. 2010, 23, 315–323. [Google Scholar] [CrossRef]
- Causier, B.; Davies, B. Analysing protein-protein interactions with the yeast two-hybrid system. Plant Mol. Biol. 2002, 50, 855–870. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alhinho, A.T.; Ramos, M.J.N.; Alves, S.; Rocheta, M.; Morais-Cecílio, L.; Gomes-Laranjo, J.; Sobral, R.; Costa, M.M.R. The Dynamics of Flower Development in Castanea sativa Mill. Plants 2021, 10, 1538. https://doi.org/10.3390/plants10081538
Alhinho AT, Ramos MJN, Alves S, Rocheta M, Morais-Cecílio L, Gomes-Laranjo J, Sobral R, Costa MMR. The Dynamics of Flower Development in Castanea sativa Mill. Plants. 2021; 10(8):1538. https://doi.org/10.3390/plants10081538
Chicago/Turabian StyleAlhinho, Ana Teresa, Miguel Jesus Nunes Ramos, Sofia Alves, Margarida Rocheta, Leonor Morais-Cecílio, José Gomes-Laranjo, Rómulo Sobral, and Maria Manuela Ribeiro Costa. 2021. "The Dynamics of Flower Development in Castanea sativa Mill." Plants 10, no. 8: 1538. https://doi.org/10.3390/plants10081538
APA StyleAlhinho, A. T., Ramos, M. J. N., Alves, S., Rocheta, M., Morais-Cecílio, L., Gomes-Laranjo, J., Sobral, R., & Costa, M. M. R. (2021). The Dynamics of Flower Development in Castanea sativa Mill. Plants, 10(8), 1538. https://doi.org/10.3390/plants10081538






