Therapeutic Promises of Medicinal Plants in Bangladesh and Their Bioactive Compounds against Ulcers and Inflammatory Diseases
Abstract
1. Introduction
2. Inflammatory Diseases and Ulcers
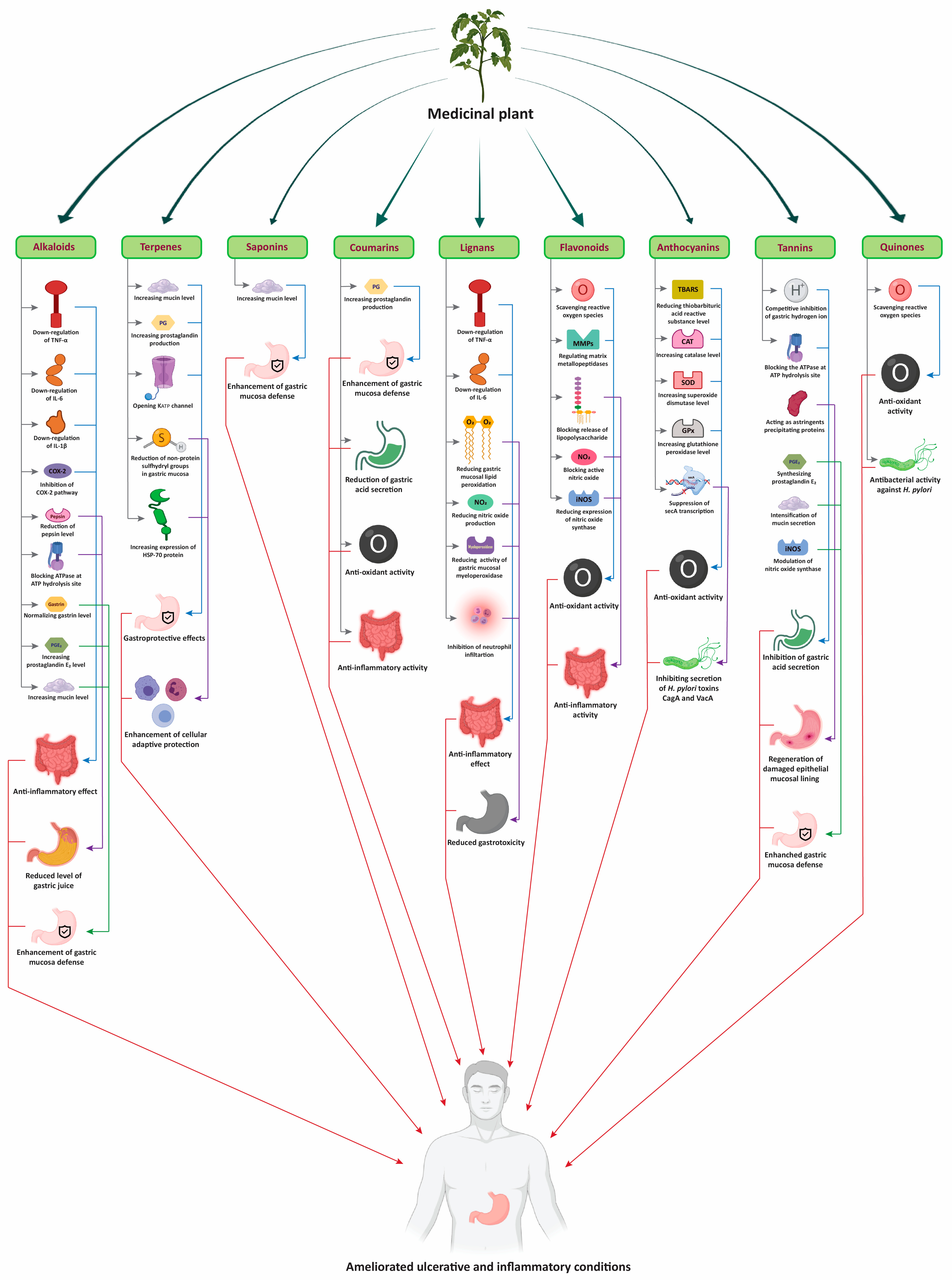
3. Plant Mediated Treatment of Ulcer and Inflammatory Diseases
3.1. Aegle marmelos
3.2. Aloe vera
3.3. Alpinia nigra and A. species
3.4. Annona squamosa
3.5. Anthocephalus cadamba
3.6. Aristolochia indica
3.7. Artocarpus heterophyllus
3.8. Asparagus racemosus
3.9. Beta vulgaris
3.10. Bombax ceiba
3.11. Caesalpinia pulcherrima
3.12. Calendula officinalis
3.13. Calotropis procera
3.14. Camellia sinensis
3.15. Capsicum annuum and C. frutescens
3.16. Carica papaya
3.17. Cinnamomum cassia
3.18. Chromolaena odorata L.
3.19. Curcuma longa L.
3.20. Glycyrrhiza glabra
3.21. Hibiscus rosa-sinensis
3.22. Centella asiatica
3.23. Lagenaria siceraria
3.24. Mangifera indica
3.25. Mimosa pudica
3.26. Momordica charantia
3.27. Moringa oleifera
3.28. Psidium guajava
3.29. Scindapsus officinalis
3.30. Shorea robusta
3.31. Solanum nigrum
3.32. Syzygium aromaticum
3.33. Terminalia chebula
3.34. Tinospora cordifolia
3.35. Zingiber officinale
4. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bishayee, A.; Sethi, G. Bioactive natural products in cancer prevention and therapy: Progress and promise. Semin. Cancer Biol. 2016, 40–41, 1–3. [Google Scholar] [CrossRef]
- Mintah, S.O.; Asafo-Agyei, T.; Archer, M.-A.; Junior, P.A.-A.; Boamah, D.; Kumadoh, D.; Appiah, A.; Ocloo, A.; Boakye, Y.D.; Agyare, C. Medicinal plants for treatment of prevalent diseases. In Pharmacognosy-Medicinal Plants; IntechOpen: London, UK, 2019. [Google Scholar]
- Sumner, J. The Natural History of Medicinal Plants; Timber Press: London, UK, 2000. [Google Scholar]
- Arif, T.; Bhosale, J.D.; Kumar, N.; Mandal, T.K.; Bendre, R.S.; Lavekar, G.S.; Dabur, R. Natural products—Antifungal agents derived from plants. J. Asian Nat. Prod. Res. 2009, 11, 621–638. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta 2013, 1830, 3670–3695. [Google Scholar] [CrossRef]
- Imadi, S.R.; Mahmood, I.; Gul, A. Medicinal Plants Against Cancer. In Plant and Human Health, Volume 1: Ethnobotany and Physiology; Ozturk, M., Hakeem, K.R., Eds.; Springer: Cham, Switzerland, 2018; pp. 139–196. [Google Scholar]
- Laila, U.; Akram, M.; Shariati, M.A.; Hashmi, A.M.; Akhtar, N.; Tahir, I.M.; Ghauri, A.O.; Munir, N.; Riaz, M.; Akhter, N. Role of medicinal plants in HIV/AIDS therapy. Clin. Exp. Pharm. Physiol. 2019, 46, 1063–1073. [Google Scholar] [CrossRef]
- Siddiqui, M.H.; Alamri, S.A.; Al-Whaibi, M.H.; Hussain, Z.; Ali, H.M.; El-Zaidy, M.E. A mini-review of anti-hepatitis B virus activity of medicinal plants. Biotechnol. Biotechnol. Equip. 2017, 31, 9–15. [Google Scholar] [CrossRef]
- Dias, D.A.; Urban, S.; Roessner, U. A historical overview of natural products in drug discovery. Metabolites 2012, 2, 303–336. [Google Scholar] [CrossRef]
- Guaadaoui, A.; Benaicha, S.; Elmajdoub, N.; Bellaoui, M.; Hamal, A. What is a bioactive compound? A combined definition for a preliminary consensus. Int. J. Nutr. Food Sci. 2014, 3, 174–179. [Google Scholar] [CrossRef]
- Mukherjee, P.K. Quality Control of Herbal Drugs: An Approach to Evaluation of Botanicals; Business Horizons: New Delhi, India, 2002. [Google Scholar]
- Jamshidi-Kia, F.; Lorigooini, Z.; Amini-Khoei, H. Medicinal plants: Past history and future perspective. J. Herbmed. Pharm. 2018, 7, 1–7. [Google Scholar] [CrossRef]
- Parveen, A.; Parveen, B.; Parveen, R.; Ahmad, S. Challenges and guidelines for clinical trial of herbal drugs. J. Pharm. Bioallied Sci. 2015, 7, 329–333. [Google Scholar]
- Wakdikar, S. Global health care challenge: Indian experiences and new prescriptions. Electron. J. Biotechnol. 2004, 7, 2–3. [Google Scholar] [CrossRef]
- Lyubchyk, S.; Shapovalova, O.; Lygina, O.; Oliveira, M.C.; Appazov, N.; Lyubchyk, A.; Charmier, A.J.; Lyubchik, S.; Pombeiro, A.J. Integrated Green Chemical Approach to the Medicinal Plant Carpobrotus edulis Processing. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Venkatachalapathi, A.; Sangeeth, T.; Paulsamy, S. Ethnobotanical informations on the species of selected areas in Nilgiri Biosphere Reserve, the Western Ghats, India. J. Res. Biol. 2015, 5, 43–57. [Google Scholar]
- Curtis, P.; Gaylord, S. Safety Issues in the Interaction of Conventional, Complementary, and Alternative Health Care. Complement. Health Pr. Rev. 2005, 10, 3–31. [Google Scholar] [CrossRef]
- Kalra, P.; Sharma, S.; Kumar, S. Antiulcer effect of the methanolic extract of Tamarindus indica seeds in different experimental models. J. Pharm. Bioallied Sci. 2011, 3, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Koo, Y.E.; Song, J.; Bae, S. Use of plant and herb derived medicine for therapeutic usage in cardiology. Medicines 2018, 5, 38. [Google Scholar] [CrossRef]
- Triantafyllidi, A.; Xanthos, T.; Papalois, A.; Triantafillidis, J.K. Herbal and plant therapy in patients with inflammatory bowel disease. Ann. Gastroenterol. 2015, 28, 210–220. [Google Scholar]
- Santana, M.T.; Cercato, L.M.; Oliveira, J.P.; Camargo, E.A. Medicinal plants in the treatment of colitis: Evidence from preclinical studies. Planta Med. 2017, 83, 588–614. [Google Scholar] [CrossRef]
- Wan, P.; Chen, H.; Guo, Y.; Bai, A.-P. Advances in treatment of ulcerative colitis with herbs: From bench to bedside. World J. Gastroenterol. 2014, 20, 14099–14104. [Google Scholar] [CrossRef]
- Oguntibeju, O.O. Medicinal plants with anti-inflammatory activities from selected countries and regions of Africa. J. Inflamm. Res. 2018, 11, 307–317. [Google Scholar] [CrossRef]
- Laveti, D.; Kumar, M.; Hemalatha, R.; Sistla, R.; Gm Naidu, V.; Talla, V.; Verma, V.; Kaur, N.; Nagpal, R. Anti-inflammatory treatments for chronic diseases: A review. Inflamm. Allergy Drug Targets 2013, 12, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Zhu, J.; Cao, F.; Chen, F. Anti-inflammatory properties of extracts from Chimonanthus nitens Oliv. leaf. PLoS ONE 2017, 12, e0181094. [Google Scholar]
- Schett, G.; Neurath, M.F. Resolution of chronic inflammatory disease: Universal and tissue-specific concepts. Nat. Commun. 2018, 9, 1–8. [Google Scholar] [CrossRef]
- Moura, F.A.; Goulart, M.O.F. Inflammatory Bowel Diseases: The Crosslink Between Risk Factors and Antioxidant Therapy. In Gastrointestinal Tissue; Elsevier: London, UK, 2017; pp. 99–112. [Google Scholar]
- Awaad, A.S.; El-Meligy, R.M.; Soliman, G.A. Natural products in treatment of ulcerative colitis and peptic ulcer. J. Saudi Chem. Soc. 2013, 17, 101–124. [Google Scholar] [CrossRef]
- Rau, W.; Hohaus, C.; Jessen, E. A differential approach to form and site of peptic ulcer. Sci. Rep. 2019, 9, 1–21. [Google Scholar]
- Lin, H.-Y.; Weng, S.-F.; Lin, H.-J.; Hsu, C.-C.; Wang, J.-J.; Su, S.-B.; Guo, H.-R.; Huang, C.-C. Peptic ulcer disease in healthcare workers: A nationwide population-based cohort study. PLoS ONE 2015, 10, e0135456. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, M.; Reddy, K.M.; Marsicano, E. Peptic ulcer disease and Helicobacter pylori infection. Mol. Med. 2018, 115, 219–224. [Google Scholar]
- Drini, M. Peptic ulcer disease and non-steroidal anti-inflammatory drugs. Aust. Prescr. 2017, 40, 91–93. [Google Scholar] [CrossRef] [PubMed]
- Sung, J.; Kuipers, E.; El-Serag, H. Systematic review: The global incidence and prevalence of peptic ulcer disease. Aliment. Pharm. Ther. 2009, 29, 938–946. [Google Scholar] [CrossRef]
- Lehours, P.; Ferrero, R.L. Helicobacter: Inflammation, immunology, and vaccines. Helicobacter 2019, 24, e12644. [Google Scholar] [CrossRef]
- Priyanka, V. Some of the medicinal plants with anti-ulcer activity—A review. J. Pharm. Sci. Res. 2015, 7, 772–775. [Google Scholar]
- Gadekar, R.; Singour, P.; Chaurasiya, P.; Pawar, R.; Patil, U. A potential of some medicinal plants as an antiulcer agents. Pharm. Rev. 2010, 4, 136–146. [Google Scholar]
- Goel, R.; Sairam, K. Anti-ulcer drugs from indigenous sources with emphasis on Musa sapientum, tamrahbasma, Asparagus racemosus and Zingiber officinale. Indian J. Pharm. 2002, 34, 100–110. [Google Scholar]
- Verma, V.K.; Singh, N.; Saxena, P.; Singh, R. Anti-ulcer and antioxidant activity of Moringa oleifera (Lam) leaves against aspirin and ethanol induced gastric ulcer in rats. Int. Res. J. Pharm. 2012, 2, 46–57. [Google Scholar]
- Sharifi-Rad, M.; Fokou, P.V.T.; Sharopov, F.; Martorell, M.; Ademiluyi, A.O.; Rajkovic, J.; Salehi, B.; Martins, N.; Iriti, M.; Sharifi-Rad, J. Antiulcer agents: From plant extracts to phytochemicals in healing promotion. Molecules 2018, 23, 1751. [Google Scholar] [CrossRef]
- Fürst, R.; Zündorf, I. Plant-derived anti-inflammatory compounds: Hopes and disappointments regarding the translation of preclinical knowledge into clinical progress. Mediat. Inflamm. 2014, 2014. [Google Scholar] [CrossRef]
- Jantan, I.; Ahmad, W.; Bukhari, S.N. Plant-derived immunomodulators: An insight on their preclinical evaluation and clinical trials. Front. Plant Sci. 2015, 6, 655. [Google Scholar] [CrossRef]
- Triantafillidis, J.; Stanciu, C. Inflammatory Bowel Disease: Etiopathogenesis, Diagnosis, Treatment, 4th ed.; Technogramma Athens: Athens, Greece, 2012. [Google Scholar]
- Harding, S.D.; Sharman, J.L.; Faccenda, E.; Southan, C.; Pawson, A.J.; Ireland, S.; Gray, A.J.G.; Bruce, L.; Alexander, S.P.H.; Anderton, S.; et al. The iuphar/bps guide to pharmacology in 2018: Updates and expansion to encompass the new guide to immunopharmacology. Nucleic Acids Res. 2018, 46, D1091–D1106. [Google Scholar] [CrossRef]
- Yang, M.; He, M.; Zhao, M.; Zou, B.; Liu, J.; Luo, L.-M.; Li, Q.-L.; He, J.-H.; Lei, P.-G. Proton pump inhibitors for preventing non-steroidal anti-inflammatory drug induced gastrointestinal toxicity: A systematic review. Curr. Med. Res. Opin. 2017, 33, 973–980. [Google Scholar] [CrossRef]
- Ghlichloo, I.; Gerriets, V. Nonsteroidal Anti-inflammatory Drugs (NSAIDs). In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Manandhar, B.; Paudel, K.R.; Sharma, B.; Karki, R. Phytochemical profile and pharmacological activity of Aegle marmelos Linn. J. Integr. Med. 2018, 16, 153–163. [Google Scholar] [CrossRef]
- Chakraborty, P.; Banerjee, S.; Kumar, S.; Sadhukhan, S.; Halder, G. Elucidation of ibuprofen uptake capability of raw and steam activated biochar of Aegle marmelos shell: Isotherm, kinetics, thermodynamics and cost estimation. Process. Saf. Environ. Prot. 2018, 118, 10–23. [Google Scholar] [CrossRef]
- Jatav, S.; Pandey, N.; Dwivedi, P.; Bansal, R.; Ahluwalia, V.; Tiwari, V.K.; Mishra, B.B. Isolation of a new flavonoid and waste to wealth recovery of 6-O-Ascorbyl Esters from Seeds of Aegle marmelos (family-Rutaceae). Nat. Prod. Res. 2019, 33, 2236–2242. [Google Scholar] [CrossRef] [PubMed]
- Patil, M.; Patil, R.; Maheshwari, V. Biological activities and identification of bioactive metabolite from endophytic Aspergillus flavus L7 isolated from Aegle marmelos. Curr. Microbiol. 2015, 71, 39–48. [Google Scholar] [CrossRef]
- Daniel, M. Medicinal Plants: Chemistry and Properties; Science Publishers: Enfield, NH, USA, 2006. [Google Scholar]
- Kurian, J. Ethno-medicinal plants of India, Thailand and Vietnam. J. Biodivers. 2012, 3, 61–75. [Google Scholar] [CrossRef]
- Bramhachari, P.; Reddy, Y.; Kotresha, D.; Varaprasad, B. Phytochemical examination, antioxidant and radical scavenging activity of Aegle marmelos (L.) Correa extracts. J. Pharm. Res. 2010, 3, 3023–3025. [Google Scholar]
- Verma, R.S.; Padalia, R.C.; Chauhan, A. Essential oil composition of Aegle marmelos (L.) Correa: Chemotypic and seasonal variations. J. Sci. Food Agric. 2014, 94, 1904–1913. [Google Scholar] [CrossRef] [PubMed]
- Chakthong, S.; Weaaryee, P.; Puangphet, P.; Mahabusarakam, W.; Plodpai, P.; Voravuthikunchai, S.P.; Kanjana-Opas, A. Alkaloid and coumarins from the green fruits of Aegle marmelos. Phytochemistry 2012, 75, 108–113. [Google Scholar] [CrossRef]
- Samarasekera, J.R.; Khambay, B.P.; Hemalal, K.P. A new insecticidal protolimonoid from Aegle marmelos. Nat. Prod. Res. 2004, 18, 117–122. [Google Scholar] [CrossRef]
- Singh, R. Ethno-medicinal and Pharmacological activities of Aegle marmelos (Linn.) Corr: A review. Pharma Innov. J. 2019, 8, 176–181. [Google Scholar]
- Rahman, S.; Quader, M.R.; Khan, M.I. Prevention of peptic ulcer by aqueous extract of Aegle marmelos leaf in rats. Imc J. Med. Sci. 2018, 12, 11–14. [Google Scholar] [CrossRef][Green Version]
- Sharmin Rahman, D.; Quader, M.R.; Sharmin, R.; Momtaz, A.; Sharmin, K.; Eva, E.O.; Mosaddek, A.S.M. Evaluation of Anti Ulcer Activity of Ethanolic Extract of Aegle Marmelos Leaves on Rats. ARC J. Dent. Sci. 2016, 1, 23–26. [Google Scholar]
- Ramakrishna, Y.G.; Savithri, K.; Kist, M.; Devaraj, S.N. Aegle marmelos fruit extract attenuates Helicobacter pylori Lipopolysaccharide induced oxidative stress in Sprague Dawley rats. BMC Complement. Altern. Med. 2015, 15, 375. [Google Scholar] [CrossRef]
- Dhuley, J. Investigation on the gastroprotective and antidiarrhoeal properties of Aegle marmelos unripe fruit extract. Hindustan Antibiot. Bull. 2003, 45, 41–46. [Google Scholar]
- Rahman, S.; Parvin, R. Therapeutic potential of Aegle marmelos (L.)-An overview. Asian Pac. J. Trop. Dis. 2014, 4, 71–77. [Google Scholar] [CrossRef]
- Pathirana, C.K.; Madhujith, T.; Eeswara, J. Bael (Aegle marmelos L. Corrêa), a Medicinal Tree with Immense Economic Potentials. Adv. Agric. 2020, 2020. [Google Scholar] [CrossRef]
- Pynam, H.; Dharmesh, S.M. Antioxidant and anti-inflammatory properties of marmelosin from Bael (Aegle marmelos L.); Inhibition of TNF-α mediated inflammatory/tumor markers. Biomed. Pharm. 2018, 106, 98–108. [Google Scholar] [CrossRef]
- Longo, B.; Sommerfeld, E.P.; Somensi, L.B.; Mariano, L.N.B.; Boeing, T.; de Andrade, S.F.; de Souza, P.; da Silva, L.M. Dual role of eugenol on chronic gastric ulcer in rats: Low-dose healing efficacy and the worsening gastric lesion in high doses. Chemi Biol. Interact. 2021, 333, 109335. [Google Scholar] [CrossRef]
- Chniguir, A.; Pintard, C.; Liu, D.; Dang, P.M.-C.; El-Benna, J.; Bachoual, R. Eugenol prevents fMLF-induced superoxide anion production in human neutrophils by inhibiting ERK1/2 signaling pathway and p47phox phosphorylation. Sci. Rep. 2019, 9, 1–11. [Google Scholar]
- Faruque, M.O.; Feng, G.; Khan, M.N.A.; Barlow, J.W.; Ankhi, U.R.; Hu, S.; Kamaruzzaman, M.; Uddin, S.B.; Hu, X. Qualitative and quantitative ethnobotanical study of the Pangkhua community in Bilaichari Upazilla, Rangamati District, Bangladesh. J. Ethnobiol. Ethnomed. 2019, 15, 8. [Google Scholar] [CrossRef] [PubMed]
- Maan, A.A.; Nazir, A.; Khan, M.K.I.; Ahmad, T.; Zia, R.; Murid, M.; Abrar, M. The therapeutic properties and applications of aloe vera: A review. J. Herb. Med. 2018, 12, 1–10. [Google Scholar] [CrossRef]
- Chowdhury, M.S.H.; Koike, M.; Muhammed, N.; Halim, M.A.; Saha, N.; Kobayashi, H. Use of plants in healthcare: A traditional ethno-medicinal practice in rural areas of southeastern Bangladesh. Int. J. Biodivers. Sci. Manag. 2009, 5, 41–51. [Google Scholar] [CrossRef]
- Suvitayavat, W.; Sumrongkit, C.; Thirawarapan, S.; Bunyapraphatsara, N. Effects of Aloe preparation on the histamine-induced gastric secretion in rats. J. Ethnopharmacol. 2004, 90, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Wani, M.Y.; Hasan, N.; Malik, M.A. Chitosan and Aloe vera: Two gifts of nature. J. Dispers. Sci. Tech. 2010, 31, 799–811. [Google Scholar] [CrossRef]
- El-Batal, A.I.; Ahmed, S.F. Therapeutic effect of Aloe vera and silver nanoparticles on acid-induced oral ulcer in gamma-irradiated mice. Braz. Oral Res. 2018, 32. [Google Scholar] [CrossRef]
- Borra, S.K.; Lagisetty, R.K.; Mallela, G.R. Anti-ulcer effect of Aloe vera in non-steroidal anti-inflammatory drug induced peptic ulcers in rats. Afr. J. Pharm. Pharm. 2011, 5, 1867–1871. [Google Scholar]
- Koo, M. Aloe vera: Antiulcer and antidiabetic effects. Phytother. Res. 1994, 8, 461–464. [Google Scholar] [CrossRef]
- Bahrami, G.; Malekshahi, H.; Miraghaee, S.; Madani, H.; Babaei, A.; Mohammadi, B.; Hatami, R. Protective and Therapeutic Effects of Aloe Vera Gel on Ulcerative Colitis Induced by Acetic Acid in Rats. Clin. Nutr. Res. 2020, 9, 223. [Google Scholar] [CrossRef] [PubMed]
- Foster, M.; Hunter, D.; Samman, S. Evaluation of the nutritional and metabolic effects of Aloe vera. In Herbal Medicine: Biomolecular and Clinical Aspects, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2011. [Google Scholar]
- Gai, L.; Chu, L.; Xia, R.; Chen, Q.; Sun, X. Barbaloin attenuates mucosal damage in experimental models of rat colitis by regulating inflammation and the AMPK signaling pathway. Med. Sci. Monit. Int. Med. J. Eexperi. Clin. Res. 2019, 25, 10045. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Uddin, S.; Wilcock, C. Medicinal plants used by Chakma tribe in Hill Tracts districts of Bangladesh. Indian J. Tradit. Knowl. 2007, 6, 508–517. [Google Scholar]
- Ghosh, S.; Rangan, L. Alpinia: The gold mine of future therapeutics. 3Biotech 2013, 3, 173–185. [Google Scholar] [CrossRef]
- Fan, G.-j.; Kang, Y.-H.; Han, Y.N.; Han, B.H. Platelet-activating factor (PAF) receptor binding antagonists from Alpinia officinarum. Bioorganic Med. Chem. Lett. 2007, 17, 6720–6722. [Google Scholar] [CrossRef]
- Lee, J.; Kim, K.A.; Jeong, S.; Lee, S.; Park, H.J.; Kim, N.J.; Lim, S. Anti-inflammatory, anti-nociceptive, and anti-psychiatric effects by the rhizomes of Alpinia officinarum on complete Freund’s adjuvant-induced arthritis in rats. J. Ethnopharmacol. 2009, 126, 258–264. [Google Scholar] [CrossRef]
- Das, B.K.; Fatema, U.K.; Hossain, M.S.; Rahman, R.; Fatema, K. Anti-inflammatory and analgesic activities of Alpinia nigra fruit extract in laboratory animals. Eur. J. Med. Plants 2014, 1158–1166. [Google Scholar] [CrossRef]
- Williams, R.J.; Spencer, J.P.; Rice-Evans, C. Flavonoids: Antioxidants or signalling molecules? Free Radic. Biol. Med. 2004, 36, 838–849. [Google Scholar] [CrossRef]
- Khan, E.A.; Khan, T.A. Adsorption of methyl red on activated carbon derived from custard apple (Annona squamosa) fruit shell: Equilibrium isotherm and kinetic studies. J. Mol. Liq. 2018, 249, 1195–1211. [Google Scholar] [CrossRef]
- Luca, S.V.; Macovei, I.; Bujor, A.; Miron, A.; Skalicka-Woźniak, K.; Aprotosoaie, A.C.; Trifan, A. Bioactivity of dietary polyphenols: The role of metabolites. Crit. Rev. Food Sci. Nutr. 2020, 60, 626–659. [Google Scholar] [CrossRef]
- Yang, Y.L.; Chang, F.R.; Wu, Y.C. Annosqualine: A novel alkaloid from the stems of Annona squamosa. Helv. Chim. Acta 2004, 87, 1392–1399. [Google Scholar] [CrossRef]
- Panda, S.; Kar, A. Antidiabetic and antioxidative effects of Annona squamosa leaves are possibly mediated through quercetin-3-O-glucoside. Biofactors 2007, 31, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Zahid, M.; Arif, M.; Rahman, M.A.; Mujahid, M. Hepatoprotective and antioxidant activities of Annona squamosa seed extract against alcohol-induced liver injury in Sprague Dawley rats. Drug Chem. Ttoxicol. 2018, 43, 588–594. [Google Scholar] [CrossRef] [PubMed]
- Chavan, M.; Shinde, D.; Nirmal, S. Major volatile constituents of Annona squamosa L. bark. Nat. Prod. Res. 2006, 20, 754–757. [Google Scholar] [CrossRef]
- Yadav, D.K.; Singh, N.; Dev, K.; Sharma, R.; Sahai, M.; Palit, G.; Maurya, R. Anti-ulcer constituents of Annona squamosa twigs. Fitoterapia 2011, 82, 666–675. [Google Scholar] [CrossRef]
- Singh, D.P.; Mishra, B.; Mishra, R. Anti-nociceptive and anti-inflammatory activity of Annona squamosa L. leaf extract in mice and rats. Res. J. Pharm. Phytochem. 2012, 4, 182–185. [Google Scholar]
- Panda, S.; Kar, A. Annona squamosa seed extract in the regulation of hyperthyroidism and lipid-peroxidation in mice: Possible involvement of quercetin. Phytomedicine 2007, 14, 799–805. [Google Scholar] [CrossRef]
- Kooti, W.; Farokhipour, M.; Asadzadeh, Z.; Ashtary-Larky, D.; Asadi-Samani, M. The role of medicinal plants in the treatment of diabetes: A systematic review. Electron. Physician 2016, 8, 1832–1842. [Google Scholar] [CrossRef]
- Mondal, S.; Bhar, K.; Mahapatra, A.S.; Mukherjee, J.; Mondal, P.; Rahaman, S.T.; Nair, A.P. “Haripriya” god’s favorite: Anthocephalus cadamba (Roxb.) Miq.-At a glance. Pharm. Res. 2020, 12, 1–6. [Google Scholar] [CrossRef]
- Inaparthi, V.K.; Babu, P.N.; Prasad, K.; Nagaraju, B.; Prasanthi, K. Evaluation of anti-ulcer and in-vitro antioxidant activities of aqueous and methanolic extracts of neolamarckia cadamba leaves and bark in wistar albino rats. Int. J. Pharm. Sci. Res. 2014, 5, 1852–1858. [Google Scholar]
- Subhan, N.; Hasan, R.; Hossain, M.; Akter, R.; Majumder, M.M.; Rahman, M.M.; Ahmed, K.; Ghani, A.; Alam, M.A. Antinociceptive and gastro-protective effect of the ethanolic extract of the flowering top of Anthocephalus Cadamba Roxb. Orient. Pharm. Exp. Med. 2009, 9, 326–334. [Google Scholar] [CrossRef]
- Bachhav, R.; Buchake, V.; Saudagar, R. Analgesic and anti-inflammatory activities of Anthocephalus cadamba roxb. leaves in wistar rats. Res. J. Pharm. Technol. 2009, 2, 164–167. [Google Scholar]
- Ambujakshi, H.; Antony, S.; Kanchana, Y.; Patel, R.; Thakkar, H. Analgesic activity of Anthocephalus cadamba leaf extract. J. Pharm. Res. 2009, 2, 1279–1280. [Google Scholar]
- Sumanta, M.; Dash, G.; Suman, A. Analgesic, anti-inflammatory and antipyretic studies of Neolamarckia cadamba barks. J. Pharm. Res. 2009, 2, 1133–1136. [Google Scholar]
- Chandrashekar, K.S.; Prasanna, K.S.; Abinash, B. Anti-inflammatory effect of the methanol extract from Anthocephalus cadamba stem bark in animal models. Int. J. Plant. Biol. 2010, 1, e6. [Google Scholar] [CrossRef]
- Naz, R.; Ayub, H.; Nawaz, S.; Islam, Z.U.; Yasmin, T.; Bano, A.; Wakeel, A.; Zia, S.; Roberts, T.H. Antimicrobial activity, toxicity and anti-inflammatory potential of methanolic extracts of four ethnomedicinal plant species from Punjab, Pakistan. BMC Complement. Altern. Med. 2017, 17, 302. [Google Scholar] [CrossRef]
- Kuo, P.-C.; Li, Y.-C.; Wu, T.-S. Chemical Constituents and Pharmacology of the Aristolochia (馬兜鈴 mădōu ling) species. J. Tradit. Complement. Med. 2012, 2, 249–266. [Google Scholar] [CrossRef]
- Desai, D.C.; Jacob, J.; Almeida, A.; Kshirsagar, R.; Manju, S. Isolation, structural elucidation and anti-inflammatory activity of astragalin,(−) hinokinin, aristolactam I and aristolochic acids (I & II) from Aristolochia indica. Nat. Prod. Res. 2014, 28, 1413–1417. [Google Scholar] [PubMed]
- Mathew, J.E.; Kaitheri, S.K.; DinakaranVachala, S.; Jose, M. Anti-inflammatory, antipruritic and mast cell stabilizing activity of Aristolochia indica. Iran. J. Basic Med. Sci. 2011, 14, 422–427. [Google Scholar] [PubMed]
- Rahmatullah, M.; Rahman, A.; Uddin, F.; Hasan, M.; Khatun, A.; Bashar, A.A.; Ahsan, S.; Mou, S.M.; Begum, R.; Jahan, R. An ethnomedicinal survey conducted amongst folk medicinal practitioners in the two southern districts of Noakhali and Feni, Bangladesh. Adv. Nat. Appl. Sci. 2011, 5, 115–132. [Google Scholar]
- Prakash, O.; Kumar, R.; Chandra, D.; Kumar, A.; Kumar, P. Effect of Artocarpus heterophyllus Lam.(Jackfruit) on Indomethacin-Induced ulcer model in albino rats. Der. Pharm. Lett. 2015, 7, 81–85. [Google Scholar]
- Yao, X.; Wu, D.; Dong, N.; Ouyang, P.; Pu, J.; Hu, Q.; Wang, J.; Lu, W.; Huang, J. Moracin C, a phenolic compound isolated from Artocarpus heterophyllus, suppresses lipopolysaccharide-activated inflammatory responses in murine raw264. 7 macrophages. Int. J. Mol. Sci. 2016, 17, 1199. [Google Scholar] [CrossRef]
- Jashni, H.K.; Jahromi, H.K.; Ranjbary, A.G.; Jahromi, Z.K.; Kherameh, Z.K. Effects of aqueous extract from Asparagus officinalis L. roots on hypothalamic-pituitary-gonadal axis hormone levels and the number of ovarian follicles in adult rats. Int. J. Reprod. Biomed. 2016, 14, 75–80. [Google Scholar]
- Singh, L. Asparagus racemosus: The plant with immense medicinal potential. J. Pharm. Phytochem. 2018, 7, 2199–2203. [Google Scholar]
- Goyal, R.; Singh, J.; Lal, H. Asparagus racemosus-an update. Indian J. Med. Sci. 2003, 57, 408–414. [Google Scholar]
- Joshi, R.K. Asparagus racemosus (Shatawari), phytoconstituents and medicinal importance, future source of economy by cultivation in Uttrakhand: A review. Int. J. Herb. Med. 2016, 4, 18–21. [Google Scholar]
- Thakur, S.; Sharma, D. Review on medicinal plant: Asparagus adscendens Roxb. Int. J. Pharma. Sci. Health Care 2015, 3, 122–137. [Google Scholar]
- Sairam, K.; Priyambada, S.; Aryya, N.; Goel, R. Gastroduodenal ulcer protective activity of Asparagus racemosus: An experimental, biochemical and histological study. J. Ethnopharmacol. 2003, 86, 1–10. [Google Scholar] [CrossRef]
- Bhatnagar, M.; Sisodia, S. Antisecretory and antiulcer activity of Asparagus racemosus Willd. against indomethacin plus pyloric ligation-induced gastric ulcer in rats. J. Herb. Ppharmacother. 2006, 6, 13–20. [Google Scholar] [CrossRef]
- Mashele, S.S. Medicinal Properties of Selected Asparagus Species: A Review. In Phytochemicals in Human Health; IntechOpen: London, UK, 2019. [Google Scholar]
- Kujala, T.S.; Vienola, M.S.; Klika, K.D.; Loponen, J.M.; Pihlaja, K. Betalain and phenolic compositions of four beetroot (Beta vulgaris) cultivars. Eur. Food Res. Technol. 2002, 214, 505–510. [Google Scholar] [CrossRef]
- Samyuktha, K.; Chinnala, K.M.; Prathiba, G.; Rajendhar, D.; Reddy, P.S. Evaluation of anti ulcer activity of ethanolic root extract of Beta vulgaris in rats. Int. J. Basic Clin. Pharm. 2017, 6, 359–364. [Google Scholar] [CrossRef][Green Version]
- Hijazi, H.H.; Rezq, A.A.; Elgazar, A.F. Studying Anti-inflammatory and Anti-Peptic Ulcer Effects of Aqueous Extract of Red Beetroots on Male Rats. Int. J. Pharm. Life Sci. 2018, 9, 5741–5747. [Google Scholar]
- Neha, P.; Sk, J.; Nk, J.; Hk, J. Chemical and functional properties of Beetroot (Beta vulgaris L.) for product development: A review. Int. J. Chem. Stud. 2018, 6, 3190–3194. [Google Scholar]
- El-Beltagi, H.S.; Mohamed, H.I.; Megahed, B.M.; Gamal, M.; Safwat, G. Evaluation of some chemical constituents, antioxidant, antibacterial and anticancer activities of Beta vulgaris L. root. Fresenius Environ. Bull. 2018, 27, 6369–6378. [Google Scholar]
- Ceclu, L.; Nistor, O. Red Beetroot: Composition and Health Effects—A Review. J. Nutr. Med. Diet. Care 2020, 6, 43. [Google Scholar]
- Nadipelly, J.; Kothapalli, J. Antiulcer activity of petroleum ether extract of Beta vulgaris (L.). Int. J. Res.Dev. Pharm. Life Sci. 2016, 5, 2414–2416. [Google Scholar]
- Jain, S.; Garg, V.K.; Sharma, P.K. Anti-inflammatory activity of aqueous extract of Beta vulgaris L. J. Basic Clin. Pharm. 2011, 2, 83–86. [Google Scholar] [PubMed]
- Chaudhary, P.H.; Tawar, M.G. Pharmacognostic and Phytopharmacological Overview on Bombax ceiba. Syst. Rev. Pharm. 2019, 10, 20–25. [Google Scholar] [CrossRef]
- Panda, D. Ethnobotanical study of medicinal plants in Jajpur district of Odisha, India. J. Pharm. Phytochem. 2018, 7, 1508–1512. [Google Scholar]
- Navdeep Ranjan, S.K.S.a.C.K. Role of medicinal plants in traditional medicine system in bihar—A review. World J. Pharm. Res. 2018, 7, 1687–1701. [Google Scholar]
- Dar, A.; Faizi, S.; Naqvi, S.; Roome, T.; Zikr-ur-Rehman, S.; Ali, M.; Firdous, S.; Moin, S.T. Analgesic and antioxidant activity of mangiferin and its derivatives: The structure activity relationship. Biol. Pharm. Bull. 2005, 28, 596–600. [Google Scholar] [CrossRef]
- Jagtap, A.; Niphadkar, P.; Phadke, A. Protective effect of aqueous extract of Bombax malabaricum DC on experimental models of inflammatory bowel disease in rats and mice. Indian J. Exp. Biol. 2011, 49, 343–351. [Google Scholar]
- Hussain, L.; Akash, M.; Naseem, S.; Rehman, K.; Ahmed, K.Z. Anti-ulcerogenic effects of Salmalia malabarica in gastric ulceration-pilot study. Adv. Clin. Exp. Med. 2015, 24, 595–605. [Google Scholar] [CrossRef]
- Barakat, M.; El-Boghdady, N.; Farrag, E.; Said, A.; Shaker, S. Protective and curative effects of Bombax ceiba flower and Ziziphus spina christi fruit extracts on gastric ulcer. J. Biol. Sci. 2019, 19, 161–172. [Google Scholar]
- Srinivas, T.L.; Lakshmi, S.M.; Shama, S.N.; Reddy, G.K.; Prasanna, K. Medicinal plants as anti-ulcer agents. J. Pharm. Phytochem. 2013, 2, 91–97. [Google Scholar]
- Jaikumar, K.; Sheik Noor Mohamed, M.; John Wyson, W.; Deventhiran, M.; Anand, D.; Saravanan, P. Phytochemical analysis of Caesalpinia pulcherrima (L.) Sw. Leaf extract using GC-MS analysis. Int. J. Pharm. Sci. Drug Res. 2017, 9, 90–93. [Google Scholar] [CrossRef]
- Gilani, S.M.U.; Ahmed, S.; Baig, S.G.; Hasan, M.M. Ethnopharmacognosy, phytochemistry and pharmacology of genus Caesalpinia: A review. J. Pharm. Phytochem. 2019, 8, 2222–2229. [Google Scholar]
- Marques, F.d.C.J.; da Silva Pantoja, P.; Matos, V.E.A.; Silva, R.O.; Damasceno, S.R.B.; Franco, Á.X.; Alves, R.C.; Justino, P.F.C.; de Souza, M.H.L.P.; Feitosa, J.P.A. Galactomannan from the seeds of Caesalpinia pulcherrima prevents indomethacin-induced gastrointestinal damage via neutrophil migration. Int. J. Biol. Macromol. 2019, 141, 68–75. [Google Scholar] [CrossRef]
- Sharma, V.; Rajani, G. Evaluation of Caesalpinia pulcherrima Linn. for anti-inflammatory and antiulcer activities. Indian J. Pharm. 2011, 43, 168–171. [Google Scholar] [CrossRef]
- Khan, M.A.; Islam, M.K.; Siraj, M.A.; Saha, S.; Barman, A.K.; Awang, K.; Rahman, M.M.; Shilpi, J.A.; Jahan, R.; Islam, E. Ethnomedicinal survey of various communities residing in Garo Hills of Durgapur, Bangladesh. J. Ethnobiol. Ethnomed. 2015, 11, 44. [Google Scholar] [CrossRef]
- Chandra, P.; Kishore, K.; Ghosh, A.K. Evaluation of antacid capacity and antiulcer activity of Calendula officinalis L. in experimental rats. Orient. Pharm. Exp. Med. 2015, 15, 277–285. [Google Scholar] [CrossRef]
- Yadav, A.K.; Pushpesh, K.; Jain, P.; Chandana, V.; Tiwari, S.; Singh, V. Investigation of Calendula officinalis whole plant as a gastroprotective and antioxidant in peptic ulcer. Braz. J. Med. Health Res. 2016, 3, 67–76. [Google Scholar]
- Preethi, K.C.; Kuttan, G.; Kuttan, R. Anti-inflammatory activity of flower extract of Calendula officinalis Linn. and its possible mechanism of action. Indian J. Exp. Biol. 2009, 47, 113–120. [Google Scholar]
- Mali, R.P.; Rao, P.S.; Jadhav, R. A Review on Pharmacological Activities of Calotropis Procera. J. Drug Deliv. Thera. 2019, 9, 947–951. [Google Scholar]
- Shamim, S.A.; Fatima, L. Pharmacological actions and therapeutic uses of Aak (Calotropis procera): A Review. Pharma Innov. J. 2019, 8, 40–47. [Google Scholar]
- Mandaville, J.P. Bedouin Ethnobotany: Plant Concepts and Uses in a Desert Pastoral World; University of Arizona Press: Tucson, AZ, USA, 2019. [Google Scholar]
- Meena, A.K.; Yadav, A.; Rao, M. Ayurvedic uses and pharmacological activities of Calotropis procera Linn. Asian J. Tradit. Med. 2011, 6, 45–53. [Google Scholar]
- Tour, N.S.; Talele, G.S. Gastric antiulcer and antiinflammatory activities of Calotropis procera stem bark. Rev. Bras. Farmacogn. 2011, 21, 1118–1126. [Google Scholar] [CrossRef]
- Al-Taweel, A.M.; Perveen, S.; Fawzy, G.A.; Rehman, A.U.; Khan, A.; Mehmood, R.; Fadda, L.M. Evaluation of antiulcer and cytotoxic potential of the leaf, flower, and fruit extracts of Calotropis procera and isolation of a new lignan glycoside. Evid. Based Complement. Altern. Med. 2017, 2017. [Google Scholar] [CrossRef]
- Kumar, V.L.; Pandey, A.; Verma, S.; Das, P. Protection afforded by methanol extract of Calotropis procera latex in experimental model of colitis is mediated through inhibition of oxidative stress and pro-inflammatory signaling. Biomed. Pharm. 2019, 109, 1602–1609. [Google Scholar] [CrossRef]
- Naveed, M.; BiBi, J.; Kamboh, A.A.; Suheryani, I.; Kakar, I.; Fazlani, S.A.; FangFang, X.; Yunjuan, L.; Kakar, M.U.; El-Hack, M.E.A. Pharmacological values and therapeutic properties of black tea (Camellia sinensis): A comprehensive overview. Biomed. Pharm. 2018, 100, 521–531. [Google Scholar] [CrossRef]
- Koch, W.; Zagórska, J.; Marzec, Z.; Kukula-Koch, W. Applications of Tea (Camellia sinensis) and Its Active Constituents in Cosmetics. Molecules 2019, 24, 4277. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ho, C.T.; Zhou, J.; Santos, J.S.; Armstrong, L.; Granato, D. Chemistry and biological activities of processed Camellia sinensis teas: A comprehensive review. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1474–1495. [Google Scholar] [CrossRef]
- Aboulwafa, M.M.; Youssef, F.S.; Gad, H.A.; Altyar, A.E.; Al-Azizi, M.M.; Ashour, M.L. A comprehensive insight on the health benefits and phytoconstituents of Camellia sinensis and recent approaches for its quality control. Antioxidants 2019, 8, 455. [Google Scholar] [CrossRef] [PubMed]
- Shivashankara, A.R.; Rao, S.; George, T.; Abraham, S.; Colin, M.D.; Palatty, P.L.; Baliga, M.S. Tea (Camellia sinensis L. Kuntze) as Hepatoprotective Agent: A Revisit. In Dietary Interventions in Liver Disease; Elsevier: San Diego, CA, USA, 2019; pp. 183–192. [Google Scholar]
- Scoparo, C.T.; Souza, L.M.; Dartora, N.; Sassaki, G.L.; Santana-Filho, A.P.; Werner, M.F.P.; Borato, D.G.; Baggio, C.H.; Iacomini, M. Chemical characterization of heteropolysaccharides from green and black teas (Camellia sinensis) and their anti-ulcer effect. Int. J. Biol. Macromol. 2016, 86, 772–781. [Google Scholar] [CrossRef] [PubMed]
- Adhikary, B.; Yadav, S.K.; Roy, K.; Bandyopadhyay, S.K.; Chattopadhyay, S. Black tea and theaflavins assist healing of indomethacin-induced gastric ulceration in mice by antioxidative action. Evid. Based Complement. Altern. Med. 2011, 2011. [Google Scholar] [CrossRef]
- Adhikary, B.; Yadav, S.K.; Chand, S.; Bandyopadhyay, S.K.; Chattopadhyay, S. Black tea and theaflavins suppress various inflammatory modulators and i-NOS mediated nitric oxide synthesis during gastric ulcer healing. Free Radic. Res. 2011, 45, 767–778. [Google Scholar] [CrossRef]
- Bae, J.; Kim, N.; Shin, Y.; Kim, S.-Y.; Kim, Y.-J. Activity of catechins and their applications. Biomed. Derm. 2020, 4, 1–10. [Google Scholar] [CrossRef]
- Navarro-Martínez, M.D.; Navarro-Perán, E.; Cabezas-Herrera, J.; Ruiz-Gómez, J.; García-Cánovas, F.; Rodríguez-López, J.N. Antifolate activity of epigallocatechin gallate against Stenotrophomonas maltophilia. Antimicrob. Agents Chemother. 2005, 49, 2914–2920. [Google Scholar] [CrossRef]
- Adcocks, C.; Collin, P.; Buttle, D.J. Catechins from green tea (Camellia sinensis) inhibit bovine and human cartilage proteoglycan and type II collagen degradation in vitro. J. Nutr. 2002, 132, 341–346. [Google Scholar] [CrossRef]
- Hasan, M.M.; Hossain, S.A.; Ali, M.A.; Alamgir, A. Medicinal plant diversity in Chittagong, Bangladesh: A database of 100 medicinal plants. J. Sci. Innov. Res. 2014, 3, 500–514. [Google Scholar]
- Rahman, M.M.; Masum, G.Z.H.; Sharkar, P.; Sima, S.N. Medicinal plant usage by traditional medical practitioners of rural villages in Chuadanga district, Bangladesh. Int. J. Biodivers. Sci. Ecost. Serv. Manag. 2013, 9, 330–338. [Google Scholar] [CrossRef]
- Omar, O.A.S.; Bukhari, H.M.; ElSawy, N.A.; Header, E.A. Efficacy of capsicum frutescens in curing the peptic ulcer. Int. J. Pure Appl. Sci. Technol. 2013, 15, 43–54. [Google Scholar]
- Mendivil, E.J.; Sandoval-Rodriguez, A.; Meza-Ríos, A.; Zuñiga-Ramos, L.; Dominguez-Rosales, A.; Vazquez-Del Mercado, M.; Sanchez-Orozco, L.; Santos-Garcia, A.; Armendariz-Borunda, J. Capsaicin induces a protective effect on gastric mucosa along with decreased expression of inflammatory molecules in a gastritis model. J. Func. Foods 2019, 59, 345–351. [Google Scholar] [CrossRef]
- Santos, P.L.; Santos, L.N.S.; Ventura, S.P.M.; de Souza, R.L.; Coutinho, J.A.P.; Soares, C.M.F.; Lima, Á.S. Recovery of capsaicin from Capsicum frutescens by applying aqueous two-phase systems based on acetonitrile and cholinium-based ionic liquids. Chem. Eng. Res. Des. 2016, 112, 103–112. [Google Scholar] [CrossRef]
- Macalood, J.S.; Vicente, H.J.; Boniao, R.D.; Gorospe, J.G.; Roa, E.C. Chemical analysis of Carica papaya L. Crude latex. Am. J. Plant. Sci. 2013, 4, 1941–1948. [Google Scholar] [CrossRef]
- Nakhate, Y.D.; Talekar, K.S.; Giri, S.V.; Vasekar, R.D.; Mankar, H.C.; Tiwari, P.R. Pharmacological and chemical composition of Carica papaya: On overview. World J. Pharm. Res. 2019, 8, 811–821. [Google Scholar]
- Santana, L.F.; Inada, A.C.; Santo, E.; Filiú, W.F.; Pott, A.; Alves, F.M.; Guimarães, R.d.C.A.; Freitas, K.d.C.; Hiane, P.A. Nutraceutical potential of Carica papaya in metabolic syndrome. Nutrients 2019, 11, 1608. [Google Scholar] [CrossRef] [PubMed]
- Vij, T.; Prashar, Y. A review on medicinal properties of Carica papaya Linn. Asian Pac. J. Trop. Dis. 2015, 5, 1–6. [Google Scholar] [CrossRef]
- Rahmani, A.H.; Aldebasi, Y.H. Potential role of carica papaya and their active constituents in the prevention and treatment of diseases. Int. J. Pharm. Pharm. Sci. 2016, 8, 11–15. [Google Scholar]
- Kaur, M.; Talniya, N.C.; Sahrawat, S.; Kumar, A.; Stashenko, E.E. Ethnomedicinal Uses, Phytochemistry and Pharmacology of Carica papaya Plant: A Compendious Review. Mini Rev. Org. Chem. 2019, 16, 463–480. [Google Scholar] [CrossRef]
- Zhang, C.; Fan, L.; Fan, S.; Wang, J.; Luo, T.; Tang, Y.; Chen, Z.; Yu, L. Cinnamomum cassia Presl: A review of its traditional uses, phytochemistry, pharmacology and toxicology. Molecules 2019, 24, 3473. [Google Scholar] [CrossRef]
- Rao, P.V.; Gan, S.H. Cinnamon: A multifaceted medicinal plant. Evid. Based Complement. Altern. Med. 2014, 2014. [Google Scholar] [CrossRef]
- Gupta, C.; Kumari, A.; Garg, A.P.; Catanzaro, R.; Marotta, F. Comparative study of cinnamon oil and clove oil on some oral microbiota. Acta Biomed. 2011, 82, 197–199. [Google Scholar]
- Muhammad, J.S.; Zaidi, S.F.; Shaharyar, S.; Refaat, A.; Usmanghani, K.; Saiki, I.; Sugiyama, T. Anti-inflammatory effect of cinnamaldehyde in Helicobacter pylori induced gastric inflammation. Biol. Pharm. Bull. 2015, 38, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraghavan, K.; Rajkumar, J.; Bukhari, S.N.A.; Al-Sayed, B.; Seyed, M.A. Chromolaena odorata: A neglected weed with a wide spectrum of pharmacological activities. Mol. Med. Rep. 2017, 15, 1007–1016. [Google Scholar] [CrossRef]
- Dulla, O.; Jahan, F.I. Ethnopharmacological survey on traditional medicinal plants at Kalaroa Upazila, Satkhira district, Khulna Division, Bangladesh. J. Intercul. Ethnopharmacol. 2017, 6, 316–325. [Google Scholar] [CrossRef]
- Paul, T.S.; Das, B.B.; Ingale, S.P.; Killedar, N.; Apte, K.G. Oral intake of polyphenols of Chromolaena odorata: A perspective in peptic ulcer, thrombocytopenia, and heparin-induced bleeding diathesis in rodent model. Pharm. Res. 2018, 10, 426–431. [Google Scholar] [CrossRef]
- Verma, R.K.; Kumari, P.; Maurya, R.K.; Kumar, V.; Verma, R.; Singh, R.K. Medicinal properties of turmeric (Curcuma longa L.): A review. Int. J. Chem. Stud. 2018, 6, 1354–1357. [Google Scholar]
- Li, S.; Yuan, W.; Deng, G.; Wang, P.; Yang, P.; Aggarwal, B. Chemical composition and product quality control of turmeric (Curcuma longa L.). Pharm. Crop. 2011, 5, 28–54. [Google Scholar] [CrossRef]
- Ayati, Z.; Ramezani, M.; Amiri, M.S.; Moghadam, A.T.; Rahimi, H.; Abdollahzade, A.; Sahebkar, A.; Emami, S.A. Ethnobotany, phytochemistry and traditional uses of Curcuma spp. and pharmacological profile of two important species (C. longa and C. zedoaria): A review. Curr. Pharm. Des. 2019, 25, 871–935. [Google Scholar] [CrossRef]
- Rajkumari, S.; Sanatombi, K. Nutritional value, phytochemical composition, and biological activities of edible Curcuma species: A review. Int. J. Food Prop. 2017, 20, S2668–S2687. [Google Scholar] [CrossRef]
- Omosa, L.; Midiwo, J.; Kuete, V. Curcuma longa. In Medicinal Spices and Vegetables from Africa; Elsevier: Amsterdam, The Netherlands, 2017; pp. 425–435. [Google Scholar]
- Rahmani, A.H.; Alsahli, M.A.; Aly, S.M.; Khan, M.A.; Aldebasi, Y.H. Role of curcumin in disease prevention and treatment. Adv. Biomed. Res. 2018, 7. [Google Scholar] [CrossRef]
- Bachmeier, B.E.; Killian, P.H.; Melchart, D. The role of curcumin in prevention and management of metastatic disease. Int. J. Mol. Sci. 2018, 19, 1716. [Google Scholar] [CrossRef] [PubMed]
- Vallée, A.; Lecarpentier, Y. Curcumin and Endometriosis. Int. J. Mol. Sci. 2020, 21, 2440. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Yue, Y.; Zheng, X.; Zhang, K.; Chen, S.; Du, Z. Curcumin, inflammation, and chronic diseases: How are they linked? Molecules 2015, 20, 9183–9213. [Google Scholar] [CrossRef] [PubMed]
- Arshad, L.; Haque, M.A.; Abbas Bukhari, S.N.; Jantan, I. An overview of structure–activity relationship studies of curcumin analogs as antioxidant and anti-inflammatory agents. Future Med. Chem. 2017, 9, 605–626. [Google Scholar] [CrossRef]
- Yang, H.; Du, Z.; Wang, W.; Song, M.; Sanidad, K.; Sukamtoh, E.; Zheng, J.; Tian, L.; Xiao, H.; Liu, Z. Structure–activity relationship of curcumin: Role of the methoxy group in anti-inflammatory and anticolitis effects of curcumin. J. Agric. Food Chem. 2017, 65, 4509–4515. [Google Scholar] [CrossRef]
- Teng, C.-F.; Yu, C.-H.; Chang, H.-Y.; Hsieh, W.-C.; Wu, T.-H.; Lin, J.-H.; Wu, H.-C.; Jeng, L.-B.; Su, I.-J. Chemopreventive effect of phytosomal curcumin on hepatitis b virus-related hepatocellular carcinoma in a transgenic mouse model. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef]
- Singh, A.G.; Kumar, A.; Tewari, D.D. An ethnobotanical survey of medicinal plants used in Terai forest of western Nepal. J. Ethnobiol. Ethnomed. 2012, 8, 19. [Google Scholar] [CrossRef]
- Razafindraibe, M.; Kuhlman, A.R.; Rabarison, H.; Rakotoarimanana, V.; Rajeriarison, C.; Rakotoarivelo, N.; Randrianarivony, T.; Rakotoarivony, F.; Ludovic, R.; Randrianasolo, A. Medicinal plants used by women from Agnalazaha littoral forest (Southeastern Madagascar). J. Ethnobiol. Ethnomed. 2013, 9, 73. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Yuan, B.-C.; Ma, Y.-S.; Zhou, S.; Liu, Y. The anti-inflammatory activity of licorice, a widely used Chinese herb. Pharm. Biol. 2017, 55, 5–18. [Google Scholar] [CrossRef]
- Chowdhary, Z.; Alamgir, A.; Alauddin, M.; Islan, M.; Chakma, K.; Hogue, M.; Kabir, M. Traditional knowledge related to medicinal and aromatic plants in tribal societies and the quantitative study of alkaloids in medicinal plants of the hill tracts in Bangladesh. Pharm. Mag. 2008, 4, 137–144. [Google Scholar]
- Jalilzadeh-Amin, G.; Najarnezhad, V.; Anassori, E.; Mostafavi, M.; Keshipour, H. Antiulcer properties of Glycyrrhiza glabra L. extract on experimental models of gastric ulcer in mice. Iran. J. Pharm. Res. 2015, 14, 1163–1170. [Google Scholar]
- Duke, J.A. Handbook of Medicinal Herbs; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Zhu, Z.; Tao, W.; Li, J.; Guo, S.; Qian, D.; Shang, E.; Su, S.; Duan, J.A. Rapid determination of flavonoids in licorice and comparison of three licorice species. J. Sep. Sci. 2016, 39, 473–482. [Google Scholar] [CrossRef]
- Fukai, T.; Marumo, A.; Kaitou, K.; Kanda, T.; Terada, S.; Nomura, T. Anti-Helicobacter pylori flavonoids from licorice extract. Life Sci. 2002, 71, 1449–1463. [Google Scholar] [CrossRef]
- Wittschier, N.; Faller, G.; Hensel, A. Aqueous extracts and polysaccharides from liquorice roots (Glycyrrhiza glabra L.) inhibit adhesion of Helicobacter pylori to human gastric mucosa. J. Ethnopharmacol. 2009, 125, 218–223. [Google Scholar] [CrossRef]
- Qu, Y.; Zong, L.; Xu, M.; Dong, Y.; Lu, L. Effects of 18α-glycyrrhizin on TGF-β1/Smad signaling pathway in rats with carbon tetrachloride-induced liver fibrosis. Int. J. Clin. Exp. Pathol. 2015, 8, 1292. [Google Scholar]
- Chatterjee, A.; Bandyopadhyay, S.K. Herbal remedy: An alternate therapy of nonsteroidal anti-Inflammatory drug induced gastric ulcer healing. Ulcers 2014, 2014. [Google Scholar] [CrossRef]
- Zargari-Samadnejad, A.; Mehrvarz, S.; Allizadeh-Naeini, S.; Tanideh, N. Healing effect of Licorice extract in acetic acid-induced ulcerative colitis in rat. Res. Pharm. Sci. 2012, 7, 837. [Google Scholar]
- Al-Snafi, A.E. Chemical constituents, pharmacological effects and therapeutic importance of Hibiscus rosa-sinensis—A review. J. Pharm. 2018, 8, 101–119. [Google Scholar]
- Phani Kumar, K.; Annapurna, A.; Ramya, G.; Sheba, D. Gastroprotective effect of flower extracts of Hibiscus rosa sinensis against acute gastric lesion models in rodents. J. Pharm. Phytochem. 2014, 3, 137–145. [Google Scholar]
- Sharma, D.; Bhatt, S. Comprehensive review on ulcer healing potential of medicinal plants. Int. J. Pharm. Pharm. Sci. 2014, 6, 3–11. [Google Scholar]
- Bhaskar, A.; Nithya, V. Evaluation of the wound-healing activity of Hibiscus rosa sinensis L. (Malvaceae) in Wistar albino rats. Indian J. Pharm. 2012, 44, 694–698. [Google Scholar] [CrossRef]
- Harun, N.H.; Septama, A.W.; Ahmad, W.; Nizam, W.A.; Suppian, R. The Potential of Centella asiatica (Linn.) Urban as an Anti-Microbial and Immunomodulator Agent: A Review. Nat. Prod. Sci. 2019, 25, 92–102. [Google Scholar] [CrossRef]
- Sun, B.; Wu, L.; Wu, Y.; Zhang, C.; Qin, L.; Hayashi, M.; Kudo, M.; Gao, M.; Liu, T. Therapeutic potential of Centella asiatica and its triterpenes: A review. Front. Pharm. 2020, 11, 1373. [Google Scholar] [CrossRef]
- Kasote, D.; Ahmad, A.; Viljoen, A. Proangiogenic potential of medicinal plants in wound healing. In Evidence-Based Validation of Herbal Medicine; Elsevier: Amsterdam, The Netherlands, 2015; pp. 149–164. [Google Scholar]
- Cheng, C.L.; Guo, J.S.; Luk, J.; Koo, M.W.L. The healing effects of Centella extract and asiaticoside on acetic acid induced gastric ulcers in rats. Life Sci. 2004, 74, 2237–2249. [Google Scholar] [CrossRef]
- Abdulla, M.; Al-Bayaty, F.; Younis, L.; Abu Hassan, M. Anti-ulcer activity of Centella asiatica leaf extract against ethanol-induced gastric mucosal injury in rats. J. Med. Plant. Res. 2010, 4, 1253–1259. [Google Scholar]
- Bhowmik, R.; Saha, M.R.; Rahman, M.A.; Islam, M.A.U. Ethnomedicinal survey of plants in the Southern District Noakhali, Bangladesh. Bangladesh Pharm. J. 2014, 17, 205–214. [Google Scholar] [CrossRef]
- Mokganya, M.; Tshisikhawe, M. Medicinal uses of selected wild edible vegetables consumed by Vhavenda of the Vhembe District Municipality, South Africa. S. Afr. J. Bot. 2019, 122, 184–188. [Google Scholar] [CrossRef]
- Shah, B.; Seth, A.; Desai, R. Phytopharmacological profile of Lagenaria siceraria: A review. Asian J. Plant. Sci. 2010, 9, 152–157. [Google Scholar] [CrossRef]
- Manchala, P. Evaluation of Anti-ulcer activity of Lagenaria siceraria chloroform extracts in pylorus ligated rats. Electron. J. Biol. 2019, 15, 27–37. [Google Scholar]
- Shendge, P.N.; Belemkar, S. Acute and 28-day oral toxicity studies of methanolic extract of Lagenaria siceraria (Cucurbitaceae) fruit in rats. Drug Chem. Toxicol. 2019, 1–9. [Google Scholar] [CrossRef]
- Batool, N.; Ilyas, N.; Shabir, S.; Saeed, M.; Mazhar, R.; Amjid, M.W. A mini-review of therapeutic potential of Mangifera indica L. Pak. J. Pharm. Sci. 2018, 31, 1441–1448. [Google Scholar] [PubMed]
- Neelima, N.; Sudhakar, M.; Patil, M.B.; Lakshmi, B. Anti-ulcer activity and HPTLC analysis of Mangifera indica L. leaves. Int. J. Pharm. Phytopharm. Res. 2012, 1, 146–155. [Google Scholar]
- Tumpa, S.I.; Hossain, M.I.; Ishika, T. Ethnomedicinal uses of herbs by indigenous medicine practitioners of Jhenaidah district, Bangladesh. J. Pharm. Phytochem. 2014, 3, 23–33. [Google Scholar]
- Stohs, S.; Swaroop, A.; Moriyama, H.; Bagchi, M.; Ahmad, T.; Bagchi, D. A Review on Antioxidant, Anti-Inflammatory and Gastroprotective Abilities of Mango (Magnifera indica) Leaf Extract and Mangiferin. J. Nutr. Health Sci. 2018, 5, 303. [Google Scholar]
- Chand, R.; Devi, S.; Seeristi, S.; Kumar, S.; Goundar, N.; Naranyan, N.; Chandra, P. Traditional use of Medicinal plants among selected Villages in Fiji Islands: A Review Perspective. Pac. Med. Stud. J. 2018, 2018, 10–20. [Google Scholar]
- Rahman, A. Ethnomedicinal survey of angiosperm plants used by Santal tribe of Joypurhat District, Bangladesh. Int. J. Adv. Res. Dev. 2015, 3, 990–1001. [Google Scholar]
- Vinothapooshan, G.; Sundar, K. Anti-ulcer activity of Mimosa pudica leaves against gastric ulcer in rats. Res. J. Pharm. Biol. Chem. Sci. 2010, 1, 606–614. [Google Scholar]
- Zaware, B.; Gilhotra, R.; Chaudhari, S.R. Potential of Mimosa pudica leaf in the treatment of ulcerative colitis in rat. Bangladesh J. Pharm. 2018, 13, 241–247. [Google Scholar] [CrossRef]
- Shahadat, H.; Mostofa, M.; Mamum, M.; Hoque, M.; Awal, M. Comparative efficacy of korolla (Momordica charantia) extract and Ivermec® pour on with their effects on certain blood parameters and body weight gain in indigenous chicken infected with Ascaridia galli. Bangladesh J. Vet. Med. 2008, 6, 153–158. [Google Scholar] [CrossRef]
- Rahmatullah, M.; Khairuzzaman, M.; Saleem, S.M.; Sattar, F.; Rahman, I.l.; Yesmin, M.S.; Malek, I.; Bashar, A.B.M.A. Documentation of some folk medicinal practices in Sylhet &Moulavibazar districts, Bangladesh. World J. Pharm. Pharm. Sci. 2015, 4, 176–186. [Google Scholar]
- Gill, N.; Rani, P.; Arora, R.; Dhawan, V.; Bali, M. Evaluation of antioxidant, antiinflammatory and antiulcer potential of Momordica charantia methanolic seed extract. Res. J. Phytochem. 2012, 6, 96–104. [Google Scholar] [CrossRef][Green Version]
- Shahrokhi, N.; Keshavarzi, Z.; Khaksari, M. Ulcer healing activity of Mumijo aqueous extract against acetic acid induced gastric ulcer in rats. J. Pharm. Bioallied Sci. 2015, 7, 56–59. [Google Scholar]
- Alam, S.; Asad, M.; Asdaq, S.M.B.; Prasad, V.S. Antiulcer activity of methanolic extract of Momordica charantia L. in rats. J. Ethnopharmacol. 2009, 123, 464–469. [Google Scholar] [CrossRef]
- Rajendran, A.; Sureshkumar, S. Phytonutrients: Moringa oleifera leaf extracts anincredible health super food supplement. Pharm. Innov. J. 2019, 8, 29–33. [Google Scholar]
- Vimala, G.; Gricilda Shoba, F. A review on antiulcer activity of few Indian medicinal plants. Int. J. Microbiol. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Kleemann, R.; Verschuren, L.; Morrison, M.; Zadelaar, S.; van Erk, M.J.; Wielinga, P.Y.; Kooistra, T. Anti-inflammatory, anti-proliferative and anti-atherosclerotic effects of quercetin in human in vitro and in vivo models. Atherosclerosis 2011, 218, 44–52. [Google Scholar] [CrossRef]
- Choudhary, M.K.; Bodakhe, S.H.; Gupta, S.K. Assessment of the antiulcer potential of Moringa oleifera root-bark extract in rats. J. Acupunct. Meridian Stud. 2013, 6, 214–220. [Google Scholar] [CrossRef]
- Devaraj, V.; Asad, M.; Prasad, S. Effect of Leaves and Fruits of Moringa oleifera. on Gastric and Duodenal Ulcers. Pharm. Biol. 2007, 45, 332–338. [Google Scholar] [CrossRef]
- Parvez, G.M.; Shakib, U.; Khokon, M.; Sanzia, M. A short review on a nutritional fruit: Guava. Toxicol. Res. 2018, 1, 1–8. [Google Scholar]
- Rahman, A.; Kumar, A. Investigation of medicinal plants at Katakhali Pouroshova of Rajshahi District, Bangladesh and their conservation management. Appl. Ecol. Environ. Sci. 2015, 3, 184–192. [Google Scholar]
- Tuhin, M.I.H.; Asaduzzaman, M.; Islam, E.; Khatun, Z.; Rahmatullah, M. Medicinal plants used by folk medicinal herbalists in seven villages of Bhola district, Bangladesh. Am. Eur. J. Sustain. Agric. 2013, 7, 210–218. [Google Scholar]
- Choudhary, A.; Singh, A.; Duggal, N.; Kumar, B. Fruits of Scindapsus officinalis attenuates pylorus ligation induced ulcer in rats. Int. J. Pharm. Sci. Res. 2014, 5, 4351–4358. [Google Scholar]
- Darekar, S.M.; Jayakumari, S. A review on wound healing activity of different extracts and formulations of Shorea robusta resin. Drug Invent. Today 2018, 10, 59–61. [Google Scholar]
- Santhoshkumar, M.; Anusuya, N.; Bhuvaneswari, P. Antiulcerogenic effect of resin from Shorea robusta Gaertn. f. on experimentally induced ulcer models. Int. J. Pharm. Pharm. Sci 2012, 5, 269–272. [Google Scholar]
- Eskandari, M.; Assadi, M.; Shirzadian, S.; Mehregan, I. Ethnobotanical Study and Distribution of the Solanum Section Solanum Species (Solanaceae) in Iran. J. Med. Plant. 2019, 3, 85–98. [Google Scholar] [CrossRef]
- Mayilsamy, M.; Rajendran, A. Ethnomedicinal plants used by paliyar tribals in Dindigul district of Tamil Nadu, India. Int. J. Sci. Innov. Dis. 2013, 3, 146–152. [Google Scholar]
- Mittal, M.; Gupta, N.; Parashar, P.; Mehra, V.; Khatri, M. Phytochemical evaluation and pharmacological activity of Syzygium aromaticum: A comprehensive review. Int. J. Pharm. Pharm. Sci. 2014, 6, 67–72. [Google Scholar]
- Bhowmik, D.; Kumar, K.S.; Yadav, A.; Srivastava, S.; Paswan, S.; Dutta, A.S. Recent trends in Indian traditional herbs Syzygium aromaticum and its health benefits. J. Pharm. Phytochem. 2012, 1, 13–23. [Google Scholar]
- Winnie, G.M. A study on medicinal plants used in ‘Karkidaka Kanji’: The ayurvedic medicine. Int. J. Sci. Res. Sci. Eng. Technol. 2018, 4, 303–309. [Google Scholar]
- Batiha, G.E.-S.; Alkazmi, L.M.; Wasef, L.G.; Beshbishy, A.M.; Nadwa, E.H.; Rashwan, E.K. Syzygium aromaticum L. (Myrtaceae): Traditional uses, bioactive chemical constituents, pharmacological and toxicological activities. Biomolecules 2020, 10, 352. [Google Scholar] [CrossRef]
- Shaheen, E.K.; Syef, A.; Saha, S.S.; Islam, S.; Al Hossain, D.; Sujan, A.I.; Rahmatullah, M. Medicinal plants used by the folk and tribal medicinal practitioners in two villages of Khakiachora and Khasia Palli in Sylhet district, Bangladesh. Adv. Nat. Appl. Sci. 2011, 5, 100–111. [Google Scholar]
- Roy, S.; Uddin, M.Z.; Hassan, M.A.; Rahman, M.M. Medico-botanical report on the Chakma community of Bangladesh. Bangladesh J. Plant. Taxon. 2008, 15, 67–72. [Google Scholar] [CrossRef]
- Mishra, V.; Agrawal, M.; Onasanwo, S.A.; Madhur, G.; Rastogi, P.; Pandey, H.P.; Palit, G.; Narender, T. Anti-secretory and cyto-protective effects of chebulinic acid isolated from the fruits of Terminalia chebula on gastric ulcers. Phytomedicine 2013, 20, 506–511. [Google Scholar] [CrossRef]
- Saha, S.; Ghosh, S. Tinospora cordifolia: One plant, many roles. Anc. Sci. Life 2012, 31, 151–159. [Google Scholar]
- Kaur, M.; Singh, A.; Kumar, B. Comparative antidiarrheal and antiulcer effect of the aqueous and ethanolic stem bark extracts of Tinospora cordifolia in rats. J. Adv. Pharm. Technol. Res. 2014, 5, 122–128. [Google Scholar]
- Upadhyay, A.K.; Kumar, K.; Kumar, A.; Mishra, H.S. Tinospora cordifolia (Willd.) Hook. f. and Thoms.(Guduchi)–validation of the Ayurvedic pharmacology through experimental and clinical studies. Int. J. Ayurveda Res. 2010, 1, 112. [Google Scholar] [CrossRef] [PubMed]
- Toda, K.; Hitoe, S.; Takeda, S.; Shimoda, H. Black ginger extract increases physical fitness performance and muscular endurance by improving inflammation and energy metabolism. Heliyon 2016, 2, e00115. [Google Scholar] [CrossRef] [PubMed]
- Shahrajabian, M.H.; Sun, W.; Cheng, Q. Clinical aspects and health benefits of ginger (Zingiber officinale) in both traditional Chinese medicine and modern industry. Acta Agric. Scand. B Soil Plant. Sci. 2019, 69, 546–556. [Google Scholar] [CrossRef]
- Asnaashari, S.; Dastmalchi, S.; Javadzadeh, Y. Gastroprotective effects of herbal medicines (roots). Int. J. Food Prop. 2018, 21, 902–920. [Google Scholar] [CrossRef]
- Al-Howiriny, T.; Alsheikh, A.; Alqasoumi, S.; Al-Yahya, M.; ElTahir, K.; Rafatullah, S. Gastric antiulcer, antisecretory and cytoprotective properties of celery (Apium graveolens) in rats. Pharm. Biol. 2010, 48, 786–793. [Google Scholar] [CrossRef]
- Singh, D.P.; Borse, S.P.; Nivsarkar, M. A novel model for NSAID induced gastroenteropathy in rats. J. Pharm. Toxicol. Methods 2016, 78, 66–75. [Google Scholar] [CrossRef]
- Ahmad, B.; Rehman, M.U.; Amin, I.; Arif, A.; Rasool, S.; Bhat, S.A.; Afzal, I.; Hussain, I.; Bilal, S. A review on pharmacological properties of zingerone (4-(4-Hydroxy-3-methoxyphenyl)-2-butanone). Sci. World J. 2015, 2015, 816364. [Google Scholar] [CrossRef]
- Sistani Karampour, N.; Arzi, A.; Rezaie, A.; Pashmforoosh, M.; Kordi, F. Gastroprotective effect of zingerone on ethanol-induced gastric ulcers in rats. Medicina 2019, 55, 64. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, F.; Zhao, Y.; Wang, P.; Sang, S. Gastroprotective [6]-gingerol aspirinate as a novel chemopreventive prodrug of aspirin for colon cancer. Sci. Rep. 2017, 7, 40119. [Google Scholar] [CrossRef]
- Khedr, A. Antiulcer protective activity of gum Arabic (Acacia Senegal) in adult rats. Bull. Natl. Nutr. Inst. Arab Repub. Egypt 2017, 49, 1–28. [Google Scholar] [CrossRef]
- Sohel, M.; Kawsar, M.; Sumon, M.; Sultana, T. Ethnomedicinal studies of Lalmohan Thana in Bhola district, Bangladesh. Altern. Integr. Med. 2016, 5, 2. [Google Scholar]
- Azam, M.N.K.; Ahmed, M.N.; Rahman, M.M.; Rahmatullah, M. Ethnomedicines used by the Oraon and Gor tribes of Sylhet district, Bangladesh. Am. Eur. J. Sustain. Agric. 2013, 7, 391–402. [Google Scholar]
- Maury, P.K.; Jain, S.; Lal, N.; Alok, S. A review on antiulcer activity. Int. J. Pharm. Sci. Res. 2012, 3, 2487–2493. [Google Scholar]
- Dey, A. Achyranthes aspera L: Phytochemical and pharmacological aspects. Int. J. Pharm. Sci. Rev. Res. 2011, 9, 72–82. [Google Scholar]
- Agrawal, T. Allophylus serratus; A pharmacologically important plant. World J. Pharm. Res. 2018, 7, 247–251. [Google Scholar]
- Vanita, K.; Deepali, M. Evaluation of antipyretic and antiulcer activity of ethanolic extract of leaves of Alstonia scholaris l. In albino wistar rats. Asian J. Pharm. Clin. Res. 2019, 12, 203–208. [Google Scholar] [CrossRef]
- Reyad-ul-Ferdous, M.; Shamim Shahjahan, D.; Sharif, T.; Mohsina, M. Present biological status of potential medicinal plant of amaranthus viridis: A comprehensive review. Am. J. Clin. Exp. Med. 2015, 3, 12–17. [Google Scholar] [CrossRef]
- Wong, V.; Yu, L.; Cho, C. Protective effect of polysaccharides from Angelica sinensis on ulcerative colitis in rats. Inflammopharmacology 2008, 16, 162–167. [Google Scholar] [CrossRef]
- Rahman, M.S.; Rahman, M.Z.; Uddin, A.A.; Rashid, M.A. Steriod and Triterpenoid from Anogeissus latifolia. Dhaka Univ. J. Pharm. Sci. 2007, 6, 47–50. [Google Scholar] [CrossRef]
- Veitch, G.E.; Beckmann, E.; Burke, B.J.; Boyer, A.; Maslen, S.L.; Ley, S.V. Synthesis of azadirachtin: A long but successful journey. Angew. Chem. Int. Ed. 2007, 46, 7629–7632. [Google Scholar] [CrossRef] [PubMed]
- Anbari, K.; Hasanvand, A.; Andevari, A.N.; Abbaszadeh, S. Concise overview: A review on natural antioxidants and important herbal plants on gastrointestinal System. Res. J. Pharm. Technol. 2019, 12, 841–847. [Google Scholar] [CrossRef]
- Azad, A.; Mahmud, M.R.; Parvin, A.; Chakrabortty, A.; Akter, F.; Moury, S.I.; Anny, I.P.; Rehnoma, S.; Tarannom, S.K.J.; Chowdhury, S.Y. Medicinal plants of a folk medicinal healer of Rangpur district, Bangladesh. J. Med. Plants 2014, 2, 46–50. [Google Scholar]
- Singh, A.; Rani, R.; Sharma, M. Medicinal herbs of Punjab (India). Int. J. 2018, 10, 10–27. [Google Scholar]
- Deng, R. Therapeutic effects of guggul and its constituent guggulsterone: Cardiovascular benefits. Cardiovasc. Drug Rev. 2007, 25, 375–390. [Google Scholar] [CrossRef] [PubMed]
- Al-Snafi, A.E. Arabian medicinal plants possessed gastroprotective effects-plant based review (part 1). IOSR J. Pharm. 2018, 8, 77–95. [Google Scholar]
- Ahmed, M.; Azam, K.; Nur, M. Traditional knowledge and formulations of medicinal plants used by the traditional medical practitioners of Bangladesh to treat schizophrenia like psychosis. Schizophr. Res. Treat. 2014, 2014. [Google Scholar] [CrossRef]
- Arefin, M.; Hossain, M.; Hossain, M.A. Plant diversity of Sonadia Island An ecologically critical area of South-East Bangladesh. Bangladesh J. Plant. Taxon. 2017, 24, 107–116. [Google Scholar] [CrossRef][Green Version]
- Gregory, M.; Divya, B.; Mary, R.A.; Viji, M.H.; Kalaichelvan, V.; Palanivel, V. Anti–ulcer activity of Ficus religiosa leaf ethanolic extract. Asian Pac. J. Trop. Biomed. 2013, 3, 554–556. [Google Scholar] [CrossRef]
- Rahman, A.; Khanom, A. Taxonomic and Ethno-Medicinal Study of Species from Moraceae (Mulberry) Family in Bangladesh Flora. Res. Plant. Sci. 2013, 1, 53–57. [Google Scholar]
- Zhou, Y.-H.; Yu, J.-P.; Liu, Y.-F.; Teng, X.-J.; Ming, M.; Lv, P.; An, P.; Liu, S.-Q.; Yu, H.-G. Effects of Ginkgo biloba extract on inflammatory mediators (SOD, MDA, TNF-α, NF-κBp65, IL-6) in TNBS-induced colitis in rats. Mediat. Inflamm. 2006, 2006, 1–9. [Google Scholar] [CrossRef]
- Rahman, A.M.; Akter, M. Taxonomy and traditional medicinal uses of Apocynaceae (Dogbane) family of Rajshahi district, Bangladesh. Res. Rev. J. Bot. Sci. 2015, 4, 1–12. [Google Scholar]
- Poornima, N.; Umarajan, K.; Babu, K. Studies on anatomical and phytochemical analysis of Oxystelma esculentum (Lf) R. br. Ex Schltes. Bot. Res. Int. 2009, 2, 239–243. [Google Scholar]
- Urmistha, S.; Ankit, S.; Mrityunjoy, M. Anti-ulcer activity of hydroalcoholic extract of Piper betle leaf on experimental animals. Asian J. Pharm. Clin. Res. 2019, 12, 226–229. [Google Scholar] [CrossRef]
- Tanna, A.; Nair, R.; Chanda, S. Assessment of anti-inflammatory and hepatoprotective potency of Polyalthia longifolia var. pendula leaf in Wistar albino rats. J. Nat. Med. 2009, 63, 80–85. [Google Scholar] [CrossRef]
- Mahmud, S.; Mahmud, S.; Hasan, M.K.; Rahman, S.; Kar, A.; Shathy, E.J.; Mohiuddin, A. A survey on medicinal plants usage by folk medicinal practitioners in different villages of Jhenaigati Upazila, Sherpur district, Bangladesh. J. Pharm. Phytochem. 2016, 5, 167–180. [Google Scholar]
- Bhalke, R.D.; Giri, M.A.; Anarthe, S.J.; Pal, S.C. Antiulcer activity of the ethanol extract of leaves of Sesbania grandiflora (Linn.). Int. J. Pharm. Pharm. Sci. 2010, 2, 206–208. [Google Scholar]
- Uddin, M.Z.; Hassan, M.A. Plant diversity of Dhaka university campus, Bangladesh. J. Asiat. Soc. Bangladesh Sci. 2016, 42, 49–68. [Google Scholar] [CrossRef]
- Sen, S.; Chakraborty, R.; Debnath, B. Challenges and opportunities in the advancement of herbal medicine: India’s position and role in a global context. J. Herb. Med. 2011, 1, 67–75. [Google Scholar] [CrossRef]
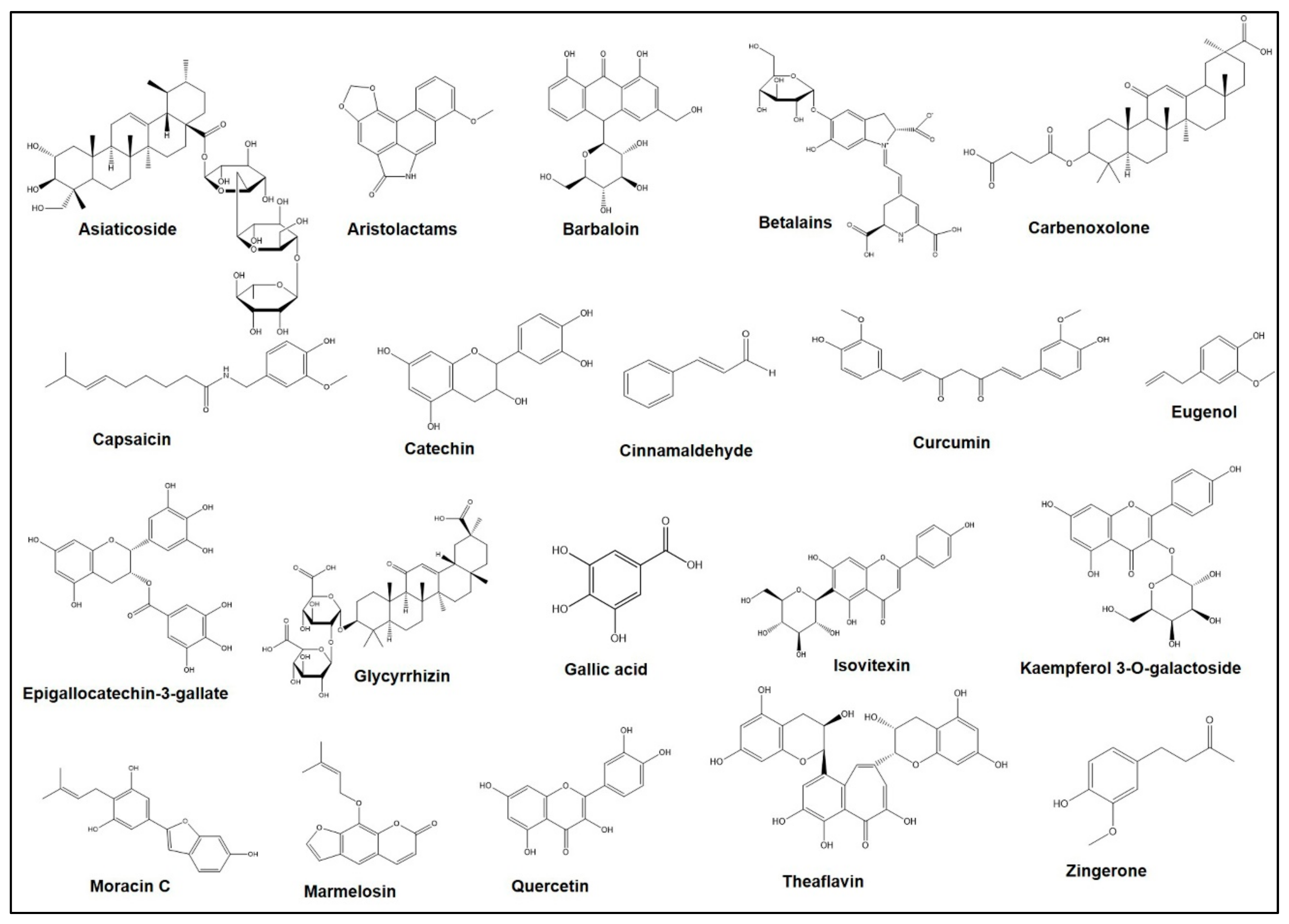
| Name (Family) | Local Name in Bangladesh | Commonly Used Medicinal Parts | Therapeutic Uses | References |
|---|---|---|---|---|
| Acacia senegal (Leguminosae) | Babla | Roots, bark, wood leaves, flowers, gums, and seeds | Used to treat ulcers, inflammation, to cure stomach and throat pain | [228,258] |
| Acacia farnesiana (Leguminosae) | Belatibabul | Leaves, bark, and flowers | Anti-inflammatory, anti-hepatotoxic, anti-pyritic and anti-ulcerogenic | [259,260] |
| Achyranthes aspera (Amaranthaseae) | Apang | Whole plant, roots, and seeds | Used to treat ulcers and inflammation | [261,262] |
| Albizia procera (Mimosaceae) | Silkorai | Bark and leaves | Used to treat ulcers, threadworms, skin scabies, and tooth pain | [233] |
| Allophylus serratus (Sapindaceae) | Tippani | Leaves, roots, flowers and seeds | Used to treat ulcers, inflammation, and gastrointestinal disorders | [130,263] |
| Alstonia scholaris (Apocynaceae) | Chatim | Bark and latex | Used to treat ulcer, dysentery, and rheumatism | [36,264] |
| Amaranthus viridis (Amaranthaceae) | Noteyshak | Leaves and seeds | Used to treat ulcer and inflammation | [265] |
| Amberboa moschata (Asteraceae) | Jam | Roots | Used to treat ulcers, malignancy, and menstrual disorders | [66] |
| Angelica sinensis (Apiaceae) | Not known | Aerial parts | Used to treat ulcerative colitis | [266] |
| Anogeissus latifolia (Combretacea) | Dhai | Roots, bark, leaves and fruits | Used to treat inflammation, ulcer, dysentery, hemorrhoids, and liver diseases | [261,267] |
| Azadirachta indica (Meliaceae) | Neem | Leaves, roots, seeds, and bark | Anti-inflammatory, anti-pyretic, anti-ulcer, anti-arrhythmic, anti-protozoal, and gastrointestinal disease | [268,269,270] |
| Basella alba Linn (Basellaceae) | Puishak | Leaves | Used to treat ulcers and constipation | [259] |
| Butea frondosa (Leguminosae) | Palash | Flower, roots, gums, stem, bark and seeds | Anti-ulcerogenic, anti-hemorrhagic activity, and septic sore throats | [36] |
| Carissa congesta (Apocynaceae) | Karamcha | Roots and leaves | Used to treat diabetes and ulcers | [157] |
| Cissus quadrangularis (Vitaceae) | Harjora or Harbhanga | Stems and rhizomes | Used to treat stomach trouble and ulcers | [135] |
| Colocasia esculenta (Araceae) | Mukhikachu | Whole plant | Used to treat tumor, ulcers, cancer, constipation, and indigestion | [157,219,233,271] |
| Commiphora mukul (Burseraceae) | Guggul | Guggul gum | Used to treat ulcers, atherosclerosis, rheumatism, and hypercholesterolemia | [228,272] |
| Desmostachya bipinnata (Gramineae) | Kush or Durva | Roots | Used to treat ulcers, cancers, and diarrheal disease | [130,273] |
| Dyospyros perigrina (Ebenaceae) | Gaab | Fruits and seeds | Used to treat ulcer, diarrhea, dysentery, and wounds | [233] |
| Dyospyros philippensis (Ebenaceae) | Boniok | Fruits and seeds | Used to treat ulcer, diarrhea, dysentery, and wounds | [233] |
| Euphorbia hirta (Euphorbiaceae) | Bara or Dudhia | Whole plant | Used to treat ulcers, inflammation, and bronchitis | [219,233,273] |
| Euphorbia neriifolia (Eurphorbiaceae) | Mansaij or Patasij | Leaves, flowers, fruits, and seeds | Used to treat ulcers, scabies, and schizophrenia | [228,274] |
| Excoecaria agallocha (Euphorbiaceae) | Gewa | Leaves, bark, roots | Used to treat microbial infections, cancers, wound and ulcers. | [130,275] |
| Ficus religiosa (Urticaceae) | Aswatha or Panbot | Leaves, stem bark, seeds, and roots | Used to treat ulcers, asthma, epilepsy, and inflammatory gastric problems | [276,277] |
| Ginkgo biloba (Ginkgoaceae) | Adel or Ginkgo | Root | Applied to cure colitis | [278] |
| Heliotropium indicum (Boraginaceae) | Hatisur | Whole plant | Used to treat ulcers, sores, and rheumatism | [233,244] |
| Lannea coromandelica (Anacardiaceae) | Jiga | Bark | Anti-eruptions, anti-leprous, anti-ulcer, and ulcerative dyspepsia | [158,233] |
| Lawsonia inermis (Lythraceae) | Mehedi | Leaves | Cures wounds and ulcers | [228,244] |
| Lens esculenta (Leguminosae) | Masur | Seeds | Used to treat ulcers | [233] |
| Lippia nodiflora (Verbenaceae) | Bhui Okar | Leaves and whole plant | Used to treat bronchial problem and ulcers | [259] |
| Ludwigia adscendens (Onagraceae) | Keshardam | Whole plant | Used to treat ulcers | [233] |
| Mikania micrantha (Asteraceae) | Toopainna Lata | Leaves | Used to treat gastric ulcers and discomfort of digestive tract | [66] |
| Nerium indicum (Apocynaceae) | Karabi | Roots, leaves, and whole plant | Anti-ulcer, diuretic, and reduce swellings | [279] |
| Oxystelma esculentum (Asclepiadaceae) | Dudhi or Dudhia Lata | Leaves, petiole, stem, roots, and rhizomes | Anti-ulcer, diuretic, and anti-bronchitis activity | [261,280] |
| Piper betel (Piperaceae) | Paan | Leaves | Used as digestive aid, anti-oxidant, anti-inflammatory, and analgesic property | [130,281] |
| Plumeria alba (Apocynaceae) | Kathgolap | Bark, leaves, flower buds, and latex | Used to treat syphilitic ulcers | [279] |
| Polyalthia longifolia (Annonaceae) | Debdaru | Whole plant | Anti-ulcerogenic, hepatoprotective, anti-inflammatory, blood pressure, fever, and moisturizing activity | [282,283] |
| Scoparia dulcis (Scrophulariaceae) | Michri Dana | Leaves | Used in the treatment of inflammation and nerve problems | [244] |
| Sesbania grandiflora (Leguminosae) | Bokful | Leaves | Used to treat ulcers | [284,285] |
| Smilax ovalifolia ( Smilacaceae) | Kumarilata | Leaves and stems | Used to treat ulcers | [66] |
| Vernonia patula (Asteraceae) | Kuksim | Aerial parts | Anti-ulcer, anti-inflammatory, and anti-dropsy | [219] |
| Xanthium indicum (Asteraceae) | Ghagra | Stems, roots, fruits, leaves, and whole plant | Used to treat ulcers, tumors, and smallpox | [219] |
| Zizyphus mauritiana (Rhamnaceae) | Ber or Kool | Roots and bark | Used to treat wounds, fever, ulcers, and diarrhea | [233] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmed, S.R.; Rabbee, M.F.; Roy, A.; Chowdhury, R.; Banik, A.; Kubra, K.; Hassan Chowdhury, M.M.; Baek, K.-H. Therapeutic Promises of Medicinal Plants in Bangladesh and Their Bioactive Compounds against Ulcers and Inflammatory Diseases. Plants 2021, 10, 1348. https://doi.org/10.3390/plants10071348
Ahmed SR, Rabbee MF, Roy A, Chowdhury R, Banik A, Kubra K, Hassan Chowdhury MM, Baek K-H. Therapeutic Promises of Medicinal Plants in Bangladesh and Their Bioactive Compounds against Ulcers and Inflammatory Diseases. Plants. 2021; 10(7):1348. https://doi.org/10.3390/plants10071348
Chicago/Turabian StyleAhmed, Sheikh Rashel, Muhammad Fazle Rabbee, Anindita Roy, Rocky Chowdhury, Anik Banik, Khadizatul Kubra, Mohammed Mehadi Hassan Chowdhury, and Kwang-Hyun Baek. 2021. "Therapeutic Promises of Medicinal Plants in Bangladesh and Their Bioactive Compounds against Ulcers and Inflammatory Diseases" Plants 10, no. 7: 1348. https://doi.org/10.3390/plants10071348
APA StyleAhmed, S. R., Rabbee, M. F., Roy, A., Chowdhury, R., Banik, A., Kubra, K., Hassan Chowdhury, M. M., & Baek, K.-H. (2021). Therapeutic Promises of Medicinal Plants in Bangladesh and Their Bioactive Compounds against Ulcers and Inflammatory Diseases. Plants, 10(7), 1348. https://doi.org/10.3390/plants10071348







