Abstract
We study the relationship between the results of two qualitatively different semi-empirical models for photoionization cross sections, , of neutral atoms (A) and their cations () centrally encapsulated inside a fullerene anion, , where q represents the negative excess charge on the shell. One of the semi-empirical models, broadly employed in previous studies, assumes a uniform excess negative charge distribution over the entire fullerene cage, by analogy with a charged metallic sphere. The other model, presented here, considers the quantum states of the excess electrons on the shell, determined by specific n and ℓ values of their quantum numbers. Remarkably, both models yield similar photoionization cross sections for the encapsulated species. Consequently, we find that the photoionization of the encapsulated atoms or cations inside the anion is influenced only slightly by the quantum states of the excess electrons on the fullerene cage. Furthermore, we demonstrate that the influence decreases even further as the size of the fullerene cage increases. All this holds true at least under the assumption that the encapsulated atom or cation is compact, i.e., its electron density remains primarily within itself rather than being drawn into the fullerene shell. This remarkable finding results from Hartree–Fock calculations combined with a popular modeling of the fullerene shell which is simulated by an attractive spherical annular potential.
1. Introduction
A frequently used semi-empirical model for a charged fullerene, , assumes that the excess charge q on the fullerene shell/cage is uniformly distributed over the entire outer surface, analogous to a metallic sphere. This model has been applied extensively in theoretical studies of the photoionization of atoms, A, and ions (cations), , encapsulated inside the charged fullerene [1,2,3,4,5,6,7,8,9,10,11,12,13]. Such systems are labeled as , are commonly referred to as charged endohedral fullerenes.
In the present paper, we focus narrowly on further examining this model in relation to the photoionization process of a neutral atom or its cation confined within an endohedral fullerene anion, (), where “A” generally represents either a neutral atom or its cation, as used throughout the paper.
There is a compelling reason for this study. Indeed, the current model neglects to account for the actual quantum states of the attached electrons within the fullerene system, such as the electrons’ principal, n, and orbital, ℓ, quantum numbers. Also, this model assumes that the charge density of the attached electrons remains on the outer surface of the fullerene cage. However, alternative models for [14,15,16,17], where an electron is bound in an ℓ-state by either a Dirac-function-like-potential [14,15], or an annular spherical potential [16,17] (each simulating the cage differently), indicate that the charge density of attached electrons spreads into the hollow interior of the fullerene.
This raises an intriguing question: how would the photoionization cross section of the atom/ion A in , calculated using the former model, change if one accounts for both the quantum states and the partial spread of the charge density of the attached electrons into the fullerene’s interior? Also, would the extensive body of calculated results on the photoionization of atoms encapsulated in fullerene anions, cited above, require re-evaluation from this perspective? To the best of our knowledge, no such comparative study has been conducted to date. This paper addresses this gap by providing new insights obtained from our research.
To address this question, we retain the modeling of the cage as in [1,16,17]. However, instead of uniformly spreading the excess charge over the cage, as in [1], we account for both the quantum states and the spread of the charge density of the attached electrons into the hollow interior of the cage.
Surprisingly, we find that the photoionization of the encapsulated atom or cation is minimally influenced by the quantum states of the attached electrons and the spread of their charge density into the hollow interior of the fullerene shell. Moreover, we demonstrate that this influence decreases as the fullerene size increases, ranging from to the giant . Our findings hold true at least under the assumption that the encapsulated atom is compact, i.e., its electron density remains primarily within the atom itself rather than being drawn into the fullerene shell.
For this case study, we selected the hydrogen atom (H), as well as the helium atom (He) and its ion , to be encapsulated within a fullerene anion.
Atomic units (a.u.) (VKD: Done. Corrected. ), where e and are the electron’s charge and mass, respectively, are used throughout the paper unless specified otherwise.
2. Theory
2.1. Review of the Former Modeling of A@
The quintessence of the model is as follows.
First, a neutral cage is modeled by a spherical annular (rectangular in the radial coordinate) potential of a certain inner radius, , thickness, , and depth, :
Such modeling of the cage was apparently first proposed by Puska and Nieminen [18] and has since been widely used in numerous theoretical studies on photoionization and other elementary processes involving endohedral fullerenes, A@) (see the works cited above, a relatively recent review [19] on these topics, and, more specifically, work [1] to stay closer to the aim of this paper).
Second, in [1], the fullerene anion, , is “built” by uniformly distributing the charge of the excess electrons over the entire outer surface of the cage. This leads to the appearance of a Coulomb spherical potential, , both inside and outside the fullerene cage:
Consequently, the entire model potential for is represented by the sum of the potentials [Equation (1)] and [Equation (2)]:
Next, to address the calculation of the structure and spectra of the A@ atom, the potential is added to the Schrödinger equation (for a single-electron atom), or to the Hartree-Fock, relativistic Dirac-Fock, or other approximation equations.
Specifically, the thus modified non-relativistic Hartree-Fock equation, applicable to A@, is
Here, is the number of electrons in the multielectron atom A, i (and, similar, j) denotes the set of the principal (), orbital (), magnetic () and spin-magnetic () quantum numbers for the i-th atomic electron in A@. (For the continuum spectrum of the i-th electron, both and are replaced by the energy of the continuum spectrum, ). Furthermore, , where is a spherical harmonic and is a radial part of the wavefunction.
We will refer to such model of the fullerene anion as “structureless” model, to underline that it ignores possible quantum states of the excess electrons on the cage.
Next, the dipole photoionization cross section, , of a -subshell of the encapsulated atom is calculated using the standard formula (see, e.g., [20]):
Here, is the photon energy, is the fine structure constant, is the number of electrons in the subshell, is a radial dipole photoionization amplitude,
2.2. Accounting for Quantum States of the Excess Electrons in A@
In our modeling of , the extra electrons in the fullerene anion are bound by the central field which is the sum of the fields of the atom and the neutral cage. Therefore, the states of the attached electrons are characterized by quantum numbers n and ℓ. Let us designate such endohedral fullerene anions as or and so on, depending on how many excess electrons exist and how many different -states they occupy in the fullerene anion. For example, He@ means that the single attached electron resides in a state in the singly-charged fullerene, He@ indicates that one of the two attached electrons in the doubly-charged anion is in a state and the other electron is in a state, and so on.
Obviously, the states of the attached electrons and the electrons in the encapsulated atom in are not independent, as they are interacting parts of the same system. Therefore, the states of all these electrons must be solutions of the same equation, which we choose to be the Hartree-Fock equation:
Here, represents the total number of electrons, where is the number of atomic electrons and is the number of attached electrons. This equation differs from Equation (4) in that the number is replaced by and the potential is replaced by in the former Equation (1). Note that when defining the states of the attached electrons, their possible states, in conjunction with the states of the electrons in the encapsulated atom, must obey the Pauli Exclusion Principle.
We will frequently refer to such model of the fullerene anion as “structured” model in this paper, to underline that it accounts for possible quantum states of the attached electrons in the fullerene anion.
Now, however, a complication arises because such modeled system becomes an open-shell system. To bypass this complication, we employ the term-averaged Hartree-Fock formalism [21] in our study. To avoid diverting the reader’s attention from the main topic of the paper, a review of the term-averaged Hartree–Fock formalism is provided in Appendix A.
Lastly, in our calculations, we use the values for , and for the model potential [Equation (1)], which mimics the / cage, as they are defined and discussed in [22]:
, , a.u., respectively.
3. Results and Discussion
3.1. Versus
Here, we present and discuss calculated results concerning the photoionization of the ground-state hydrogen atom encapsulated within the singly-charged fullerene anion (). The results were obtained in the frameworks of both the structureless [] and structured [] models. As case studies, we arbitrarily choose the and states for the attached electron, as well as and 240.
To help the reader follow the discussion of results more easily, we direct the reader’s attention to the following notations: the function (or the orbital) refers to the radial function of the electron in the hydrogen atom, while the and radial functions (or the and orbitals) correspond to the attached electrons residing in a or state, respectively.
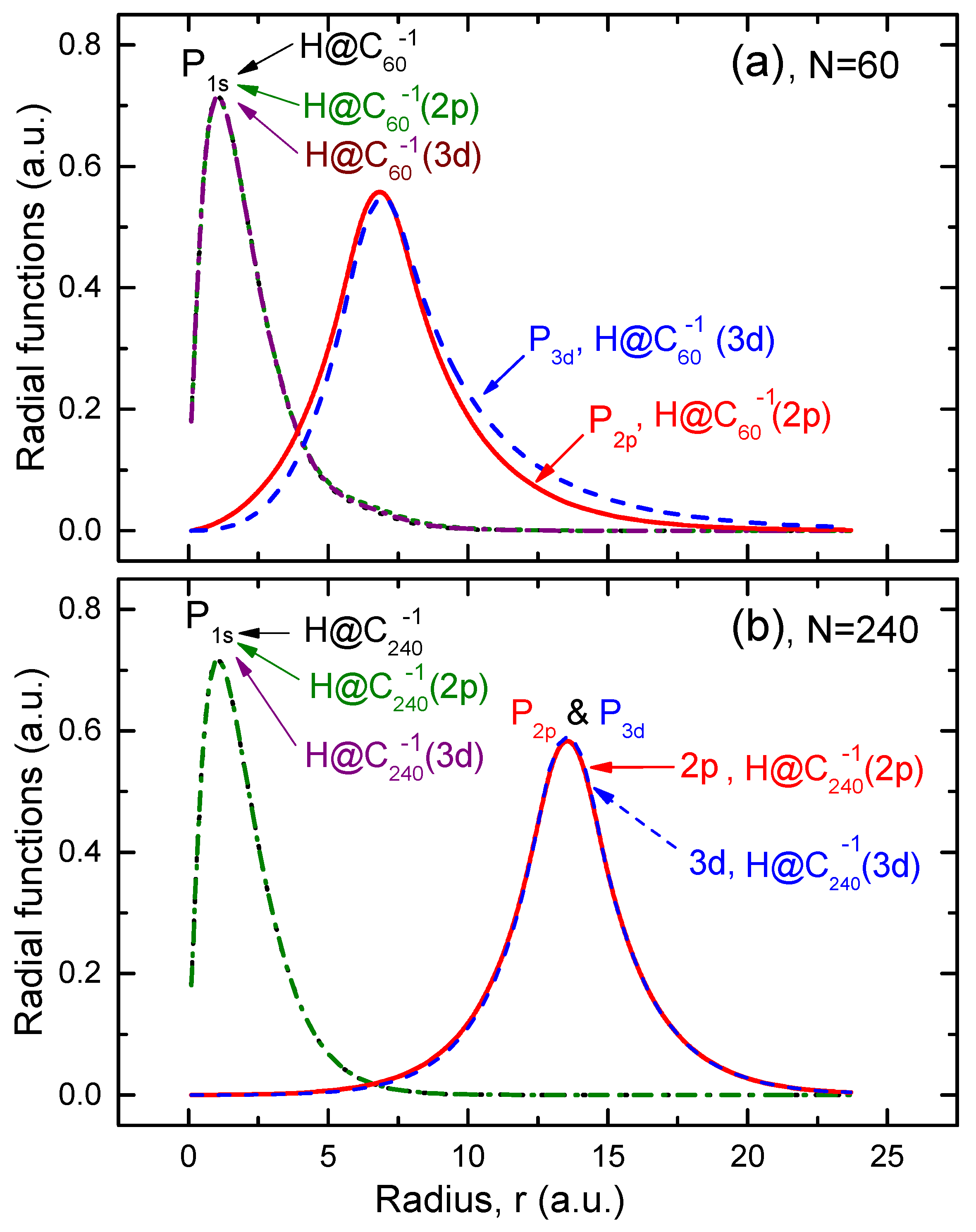
The corresponding , , and functions are depicted in Figure 1.
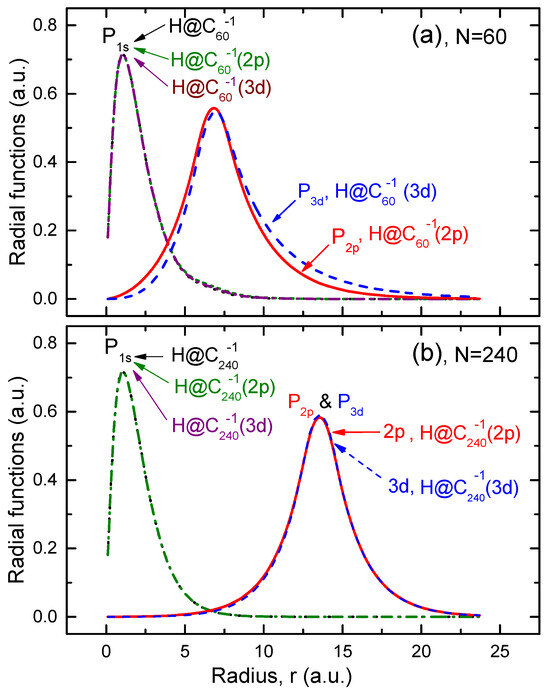
Figure 1.
(a) Calculated radial function of hydrogen in the structureless system, along with the function for hydrogen, and the and radial functions of the attached electrons in the structured systems ( and ), as indicated in the figure. (b) The same as in (a) but for the giant . Note that the function for the encapsulated hydrogen atom is practically the same in both the structureless and structured models of the endohedral fullerene anion.
First, it can be concluded from Figure 1 that the electron density in the encapsulated atom remains largely unaffected by the excess charge on the fullerene cage, especially in the case of the cage.
Second, note how the and functions of the attached electron extend into the hollow interior of the shell, thereby overlapping with the function of the encapsulated H atom. This, obviously, should make the -photoionization cross section, , of the system differ from that of the structureless system. How strong can the difference be? One of the aims of the present study is to answer this question.
Third, one can see that the overlap of the function with the functions for the and attached electrons is insignificant only in the case of the giant . Hence, it is reasonable to expect that any differences in values of calculated, respectively, for the cases of the and the systems will diminish with the increasing size of the fullerene cage.
Fourth, interestingly, the and functions of the attached electron differ only insignificantly from each other, especially in the case of the giant cage. A similar situation was observed and explained for the functions of the -electron attached to the empty fullerene cage [17]. The reason for the noted nearly indistinguishable difference between the functions with different ℓ values (the and functions in our case) arises from the electrons’ primary localization on the cage itself, which has a large radius. Consequently, the centrifugal barrier , which normally ensures the dependence of on ℓ, becomes so small that the difference between the functions with close ℓ values effectively vanishes. The latter is particularly true for the case of the giant cage.
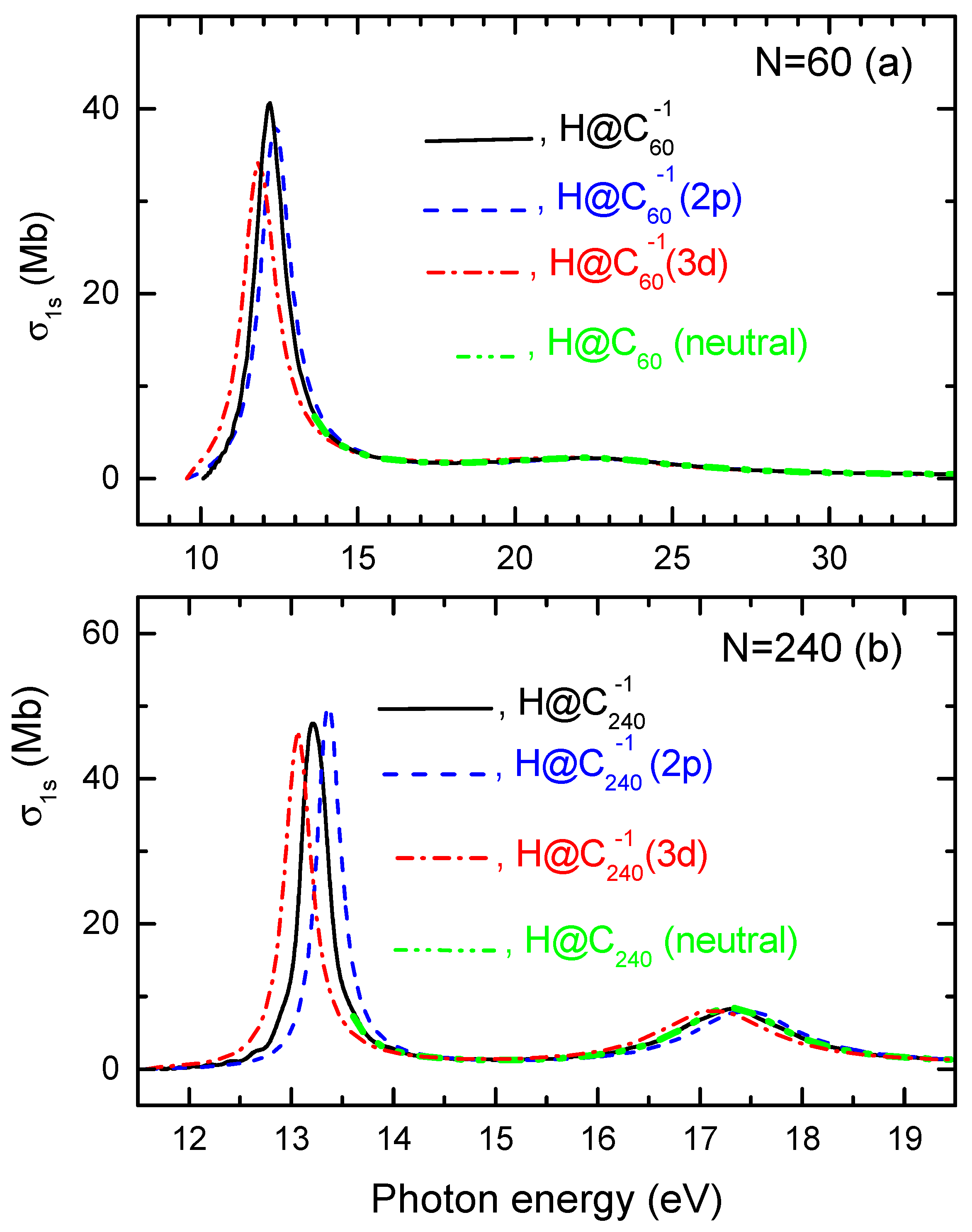
Calculated photoionization cross sections of H@ and H@ with and are depicted in Figure 2.
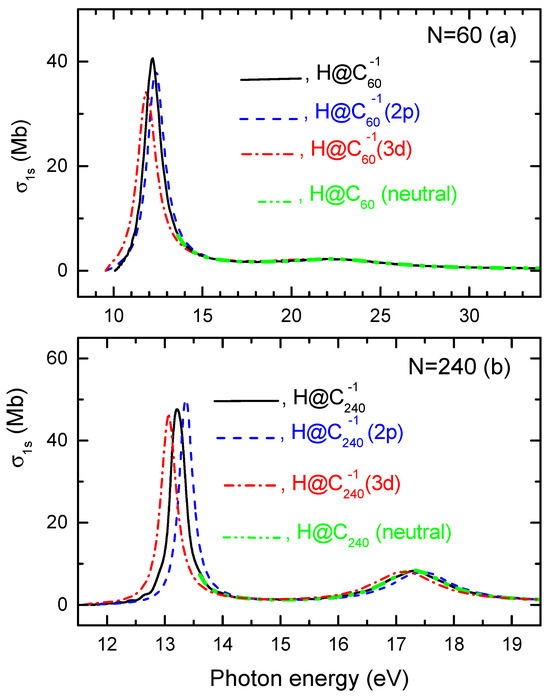
Figure 2.
Calculated photoionization cross sections for the H atom confined inside (a) the structureless H@ and structured H@ and H@ systems, as well as (b) the structureless H@ and structured H@ and H@ giant fullerene anions, as designated in the figure.
One can see that some differences between these cross sections are seen only in the low-energy region. There, the sharp, intense maxima in ’s are only slightly shifted along the energy scale relative to each other without significant changes in their magnitudes. We thus find that the photoionization cross section of the atom encapsulated inside the endohedral fullerene anion depends little on the quantum state of the attached electron in the anion.
And, as expected, the differences between the discussed -photoionization cross sections are diminishing with the increasing size of the fullerene cage. This is obvious from comparing calculated data plotted in Figure 2a (for ) with those plotted in Figure 2b for the case of the giant . Note that a would-be larger shift in energy between the low-energy maxima in the cross sections in Figure 2b than in Figure 2a is deceptive. This is because the energy-scale range is less than 10 eV in Figure 2b, but is much broader, about 30 eV, in Figure 2a.
Finally, let us comment on the origin and nature of all maxima in the photoionization cross sections displayed in Figure 2. To meet the goal, we also calculated and plotted the photoionization cross sections of the hydrogen atom confined inside a neutral cage, designated as “H (neutral)” in the figure.
First, the calculated values, plotted in Figure 2a or Figure 2b, are nearly identical down to about 14 eV of photon energy. Second, for photon energies in the same energy region, all calculated cross sections exhibit a maximum around 22 eV for (where it is poorly developed, though), as well as around 17 eV for (where the maximum is stronger). Therefore, this higher-energy maximum in for the endohedral fullerene anion has the same origin as the maximum in for the neutral H@. The origin of such maxima in the photoionization cross sections for atoms encapsulated inside neutral endohedral fullerenes have been extensively studied previously; see, e.g., [18,19,23,24,25] (and references therein). These maxima arise from interference between the outgoing photoelectron wave from the encapsulated atom and the photoelectron waves reflected from the boundaries of the fullerene cage. In [24], these resonances were termed. This term has become common in the literature, and we use it in the present paper as well. Thus, the observed higher-energy resonances in for H@, H@, and neutral H@ are confinement resonances.
Next, in the photon energy region below about 14 eV, all calculated values for fullerene anions show a sharp, strong resonance, whereas the for neutral H@ does not extend into this region. The latter is for an obvious reason. Indeed, when there is a negative charge on the fullerene cage, it results in a repulsion between the electrons of the encapsulated atom and the negative charge on the fullerene cage. Consequently, the total energy of the atomic electrons becomes , where is the energy of the atomic electrons when the atom is inside neutral , and is an additional Coulomb energy due to the repulsion by the negative charge on the cage. As is known, the repulsive Coulomb energy, , is positive, whereas is negative. Hence, the ionization potential, , of the atomic electrons inside the fullerene anion decreases compared to that for the atom confined inside neutral . Thus, the graph for for H@ or H@ starts at a lower energy than that for neutral H@. Returning to the discussion of the low-energy maxima in the calculated cross sections for negative fullerene anions, they obviously originate from a different source compared to the confinement resonances in for neutral H@. Such novel resonances in the photoionization cross section of the atom encapsulated within the fullerene anion were predicted, studied, and explained in detail in [1]. It was demonstrated that these resonances arise due to an additional Coulomb potential barrier created by the excess charge on the fullerene cage. In [1], these resonances were termed Coulomb confinement resonances. To date, Coulomb confinement resonances have been explored in various contexts with different levels of detail in numerous theoretical studies (see, e.g., [4,5,8,10,13] and references therein). As the summary on the low-energy sharp resonances in the photoionization cross sections of H@ and H@: they are Coulomb confinement resonances.
In conclusion to this section: (a) we present the first indication that the photoionization cross section of the atom within the endohedral fullerene anion is largely independent of the quantum states of the electrons attached to the fullerene cage; and (b) the most notable, albeit minor, differences in the photoionization cross sections between the calculated values obtained using the structured model for the fullerene anion (H@), and the structureless model (H@), primarily occur in the region of Coulomb confinement resonances.
3.2. , and
As another case study, we examine the photoionization cross sections of the He atom and its ion, , using both the structureless and structured models for the endohedral fullerene anion. Specifically, we arbitrarily choose the singly-charged He@ and He@ as well as the doubly-charged and endohedral fullerene anions ( and 240), as representative species for this study.
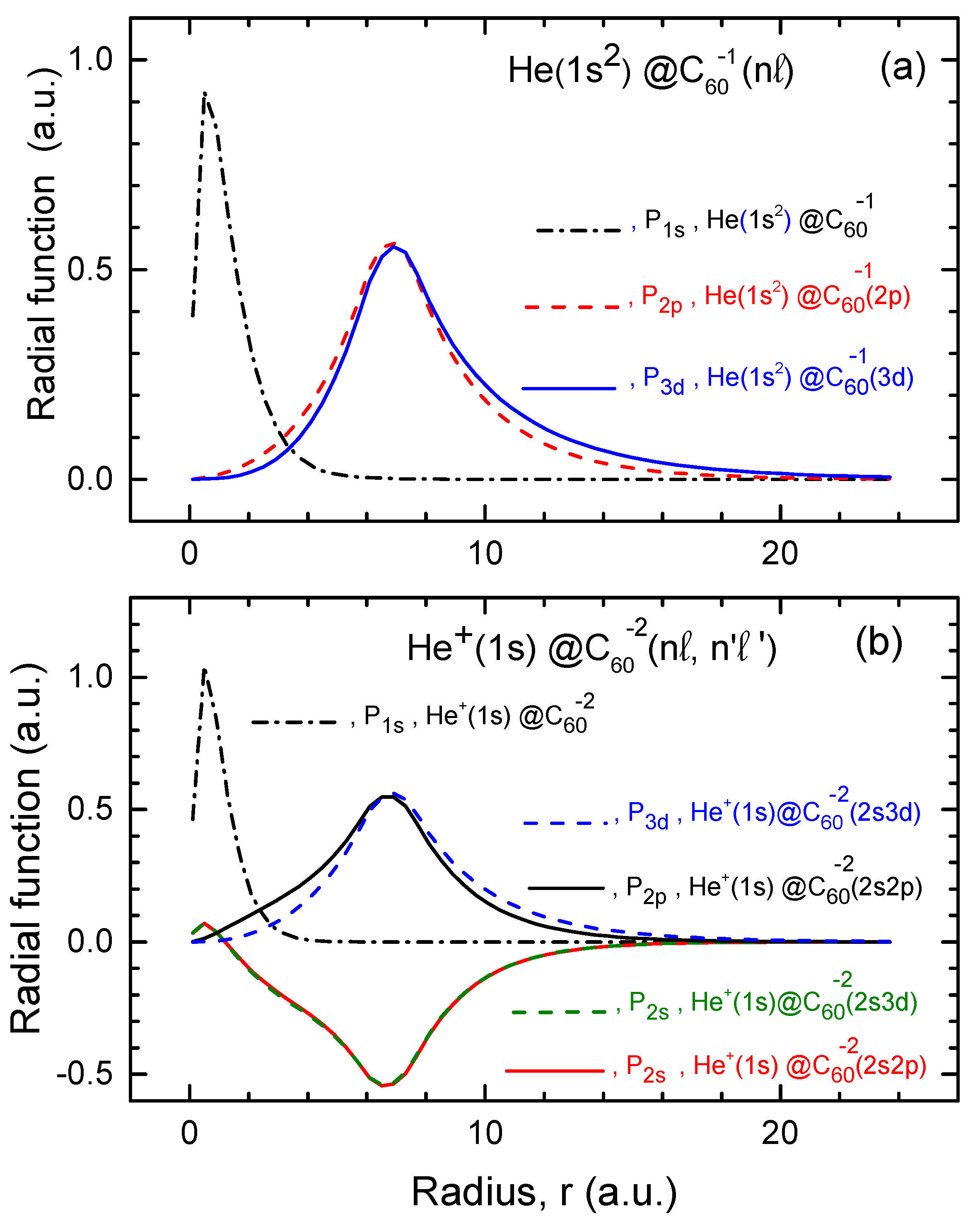
The functions for He and , calculated within both the structureless and structured models, as well as the , and functions of the attached electrons in the corresponding endohedral fullerene anions, are plotted in Figure 3.
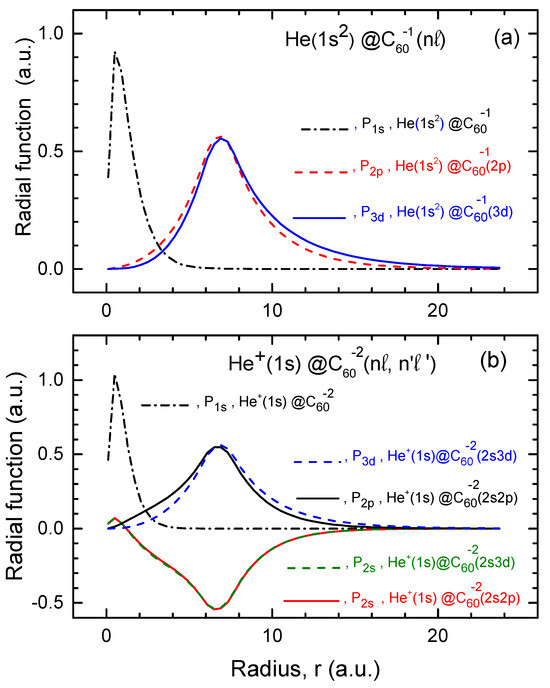
Figure 3.
(a) Calculated of neutral He in He, along with the and functions of the attached electron in the He and He fullerene anions, as indicated in the figure. (b) Calculated of the ion in the doubly-charged , along with the , , and functions of the attached electrons in the doubly-charged and , as shown in the figure. Note: Similar to the case of the H atom, the function of He in He is nearly identical to the corresponding functions of He in He and He. The same applies to the function of in the corresponding doubly-charged fullerene anions. Therefore, is only plotted once in both parts of the figure for clarity.
The behavior of the plotted functions follows similar trends as those observed in the previously discussed case of the H atom encapsulated in the fullerene anion. Thus, we believe that the behavior of these functions is self-explanatory. We leave it to the readers to draw their own conclusions. It is important to emphasize, however, that, as in the case of the H atom, the functions of the encapsulated He atom and ion overlap with those of the attached electrons in the fullerene anions. This overlap is stronger than the overlap between the functions in the case of the H atom. Therefore it could, among other factors, contribute to potentially stronger differences in the photoionization cross sections when comparing calculated results based on the structured and structureless models for fullerene anions.
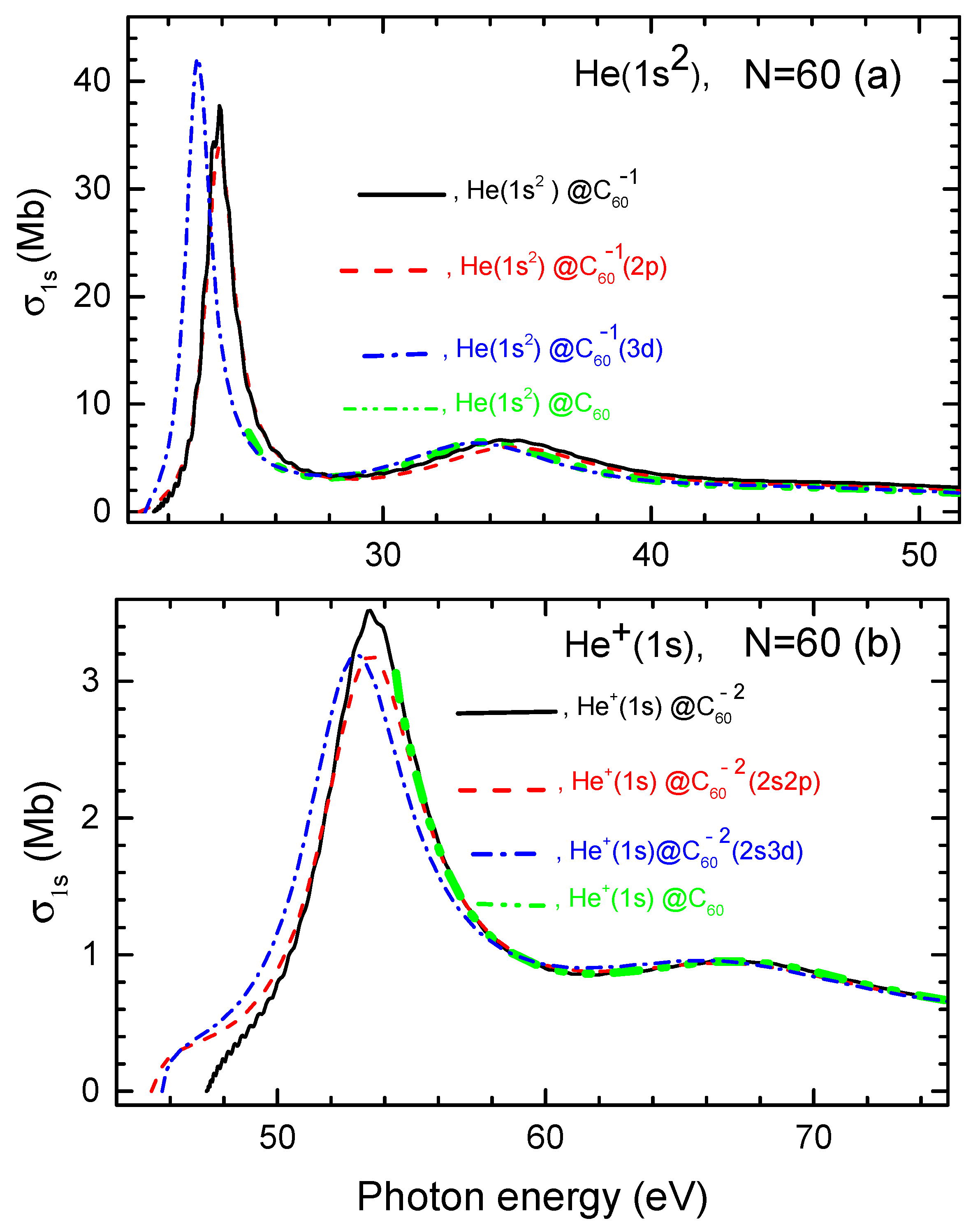
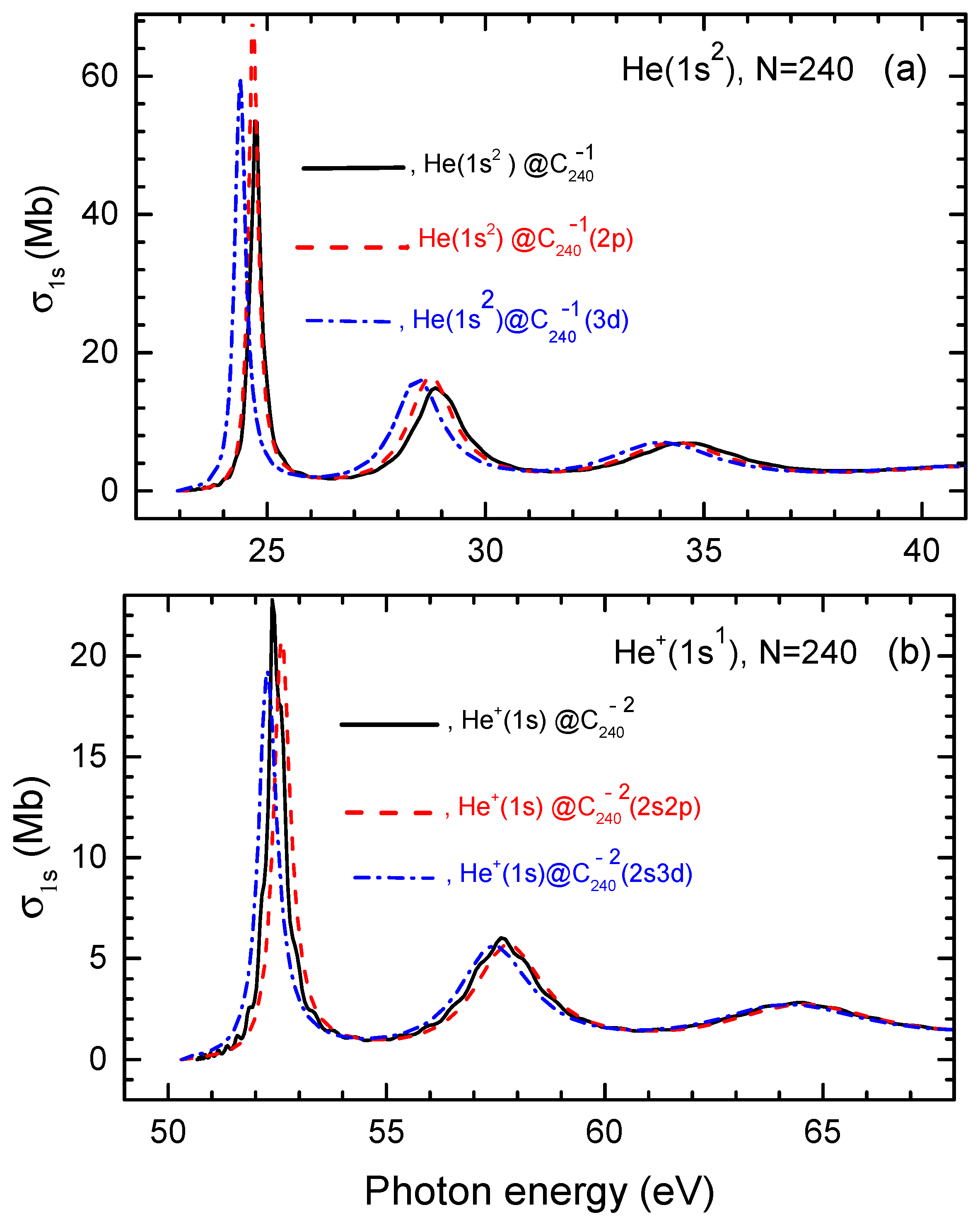
Calculated photoionization cross sections of He, He@ and He@, as well as He, , and are depicted in Figure 4.
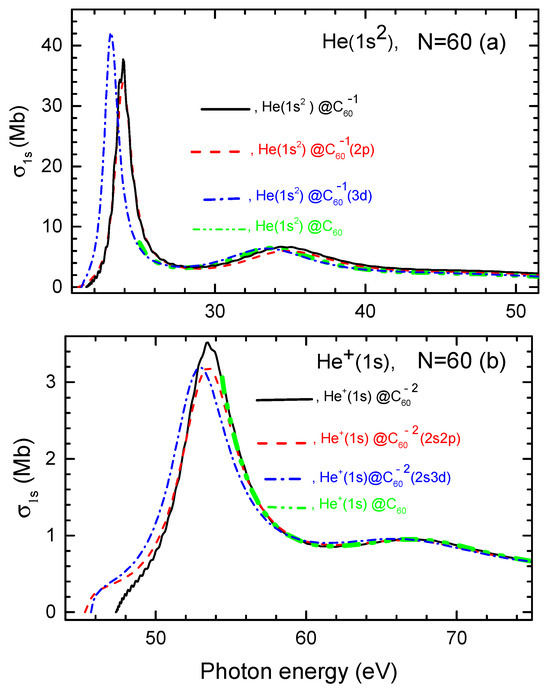
Figure 4.
(a) Calculated photoionization cross sections of He as well as He and He along with for the helium atom in neutral He, as marked in the figure. (b) Calculated photoionization cross sections of , and , as well as of inside neutral , as marked in the figure.
Firstly, similar to the encapsulated H atom case discussed earlier, Figure 4 reveals that the photoionization cross section of He and ion exhibits a pronounced, low-energy sharp resonance when photoionization occurs from the corresponding fullerene anions, in contrast to the cases of photoionization of He and in neutral He and He. Thus, as in the case of the encapsulated H atom, these resonances are Coulomb confinement resonances. As photon energy increases, these Coulomb confinement resonances are succeeded by less pronounced resonances in both the charged and neutral endohedral fullerene systems. These latter higher-energy features are, thus, ordinary confinement resonances.
Secondly, a key finding is that the differences in the photoionization cross sections between the structureless and structured models are primarily observed within the corresponding Coulomb confinement resonances. However, these differences are relatively minor, with no significant qualitative or quantitative variations. This, once again, speaks to the fact that the photoionization cross section of the atom or its ion inside the endohedral fullerene anion is minimally influenced by the quantum states of the attached electrons in the system.
Lastly, we move to the discussion of calculated results for the helium photoionization cross sections in the giant endohedral fullerene anions: He, He, He, He, , , , and . Calculated results are depicted in Figure 5.
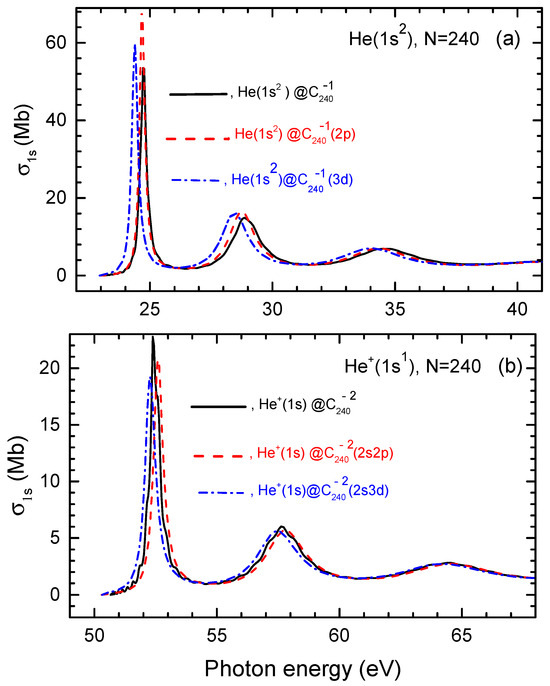
Figure 5.
(a) Calculated photoionization cross sections of He as well as He and He along with of neutral He, as designated in the figure. (b) Calculated photoionization cross sections of , and , as well as of in neutral , as marked in the figure.
The trends observed in the calculated photoionization cross sections mirror those just discussed above for He and in the cases of the fullerene cages with . It is worth noting, however, that the differences of interest in the corresponding photoionization cross sections are significantly smaller in the case of the giant endohedral fullerene anions than in the other case, which was anticipated.
Thus, we see, once again, that the photoionization cross sections of both a neutral atom and its ion in the corresponding endohedral fullerene anion are affected only little by the quantum states of the attached electrons in the endohedral system.
4. Conclusions
In this study, we investigated the photoionization cross sections of H and He atoms, as well as the ion, when centrally encapsulated within a fullerene anion cage. These encapsulated species are considered compact, as their electron densities remain largely unaffected by the fullerene cage.
The results and discussion presented in this paper suggest that the photoionization cross section of a centrally encapsulated atom or cation within an endohedral fullerene anion is minimally influenced by the quantum states of the attached electrons, provided the electron density of the encapsulated atom or ion remains primarily self-contained. This conclusion likely extends to other centrally confined compact atoms, as no contrary indication has been found.
The most notable differences in the calculated photoionization cross sections, arising from the consideration of the quantum states of the attached electrons, occur primarily between the Coulomb confinement resonances. However, these differences are relatively minor and diminish significantly with increasing fullerene cage size. Consequently, both the structureless and structured models of the endohedral fullerene anion yield closely aligned photoionization results, particularly for giant fullerene anions. Thus, either semi-empirical model effectively captures the photoionization cross sections of atoms encapsulated within fullerene anions, provided the encapsulated atom’s electron density remains largely self-contained.
For atoms that significantly contribute their electron density to the fullerene cage, we anticipate that the photoionization cross sections of the inner electrons will similarly exhibit minimal dependence on the quantum structure of the excess electrons on the fullerene cage. However, studying such cases requires a separate, focused investigation beyond the scope of this paper.
We advise some caution regarding semi-empirical model studies of the photoionization of positive ions confined within a fullerene anion, as presented here for the photoionization of and . Indeed, there is potential for the attached electrons to transition into the ion’s open orbital, leading to processes such as Auger decay. However, given the narrow focus of this study on understanding how quantum states of electrons attached to a fullerene anion might influence atomic photoionization calculations, the omission of such processes is considered permissible here, as the intent is not to predict outcomes for a specific system.
Lastly, we note that numerous aspects of the structure and spectra of atoms under various types of confinement—impenetrable, open boundaries, spheroidal, conoidal, Debye, -like, etc.—have been intensely scrutinized worldwide [26,27,28,29,30] (and references therein). Confinement significantly alters atomic behavior, providing insights into diverse interdisciplinary problems. Therefore, this study’s relevance extends beyond endohedral fullerene anions, contributing valuable knowledge to the general field of confined quantum systems.
Author Contributions
V.K.D.: Conceptualization, methodology, formal analysis, calculations, validation, preparation, writing the original draft, review and editing. L.V.C.: software, calculation, editing. V.G.Y.: formal analysis, writing the original manuscript, review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding, and the APC was funded by the MDPI itself.
Data Availability Statement
Data is contained within the article.
Conflicts of Interest
The author declares no conflicts of interest.
Appendix A. Term-Averaged Hartree–Fock Approximation
Below, we outline the key steps that lead to the term-averaged methodology for the Hartree–Fock formalism [21] (also with its extention to the calculation of many-body Feynman diagrams) used in the present study.
In the calculation of atomic wavefunctions using the Hartree–Fock method, the combined direct and exchange Coulomb interelectron interaction energy, , in the atom is given by:
Here, the sum runs over all atomic subshells and all values of and that satisfy the triangular rule , and , provided the sum of the angular moments in brackets is even.
The Coulomb integral, expressed in terms of the radial parts of the atomic wavefunctions, is given by the standard formula:
Here, () is the larger (smaller) of the two radial parameters r and .
The weight factors for the direct Coulomb interaction for open atomic shells are tabulated for all terms in [31]. The weight factors for the direct Coulomb interaction as well as the weight factors for the ‘exchange Coulomb interaction that are presented in (A1) depend on the values of and for the interacting shells k and p as well as on the total values of the angular and spin moments [32].
For the closed-shell atom with electrons in the shell, the weight factors before the Coulomb integrals are determined as [32]:
whereas
To avoid accounting for the exact angular-momentum-coupling scheme in our study, for the sake of simplicity, in the present work we use the average-term approximation, the essence of which is based on the introduction of the average direct Coulomb and exchange weight factors into the interelectron interaction potential, as follows.
Since the expression for is proportional to the number of interacting pairs of electrons in the closed shell of electrons, it is natural to expect that the same is true for the coefficients as well. Thus, for the N-electron open shells, we write Equations (A3) and (A4) as follows:
Equation (A5) for is exact for unfilled shells.
The exact weight factors for depend on the values of L, S, and additional quantum numbers. These factors are tabulated in [31] for all possible terms. The value equals the value of averaged over all terms of the configuration using their statistical weights.
Next, for the interaction of two closed shells, and , the equations for the weight factors are as follows:
For closed shells, for .
Equation (A7) indicates that the intershell interaction is proportional to the numbers of electrons in the interacting shells. This allows us to write the average weighs factors for a similar interaction between the open shells as follows
and
In our study of the structured open-shell fullerene anions, these weight factors were substituted into the Equation (A1) for the interelectron interaction potential to calculate the term-averaged energies and radial parts of the wavefunctions of electrons.
References
- Dolmatov, V.K.; Manson, S.T. Photoionization of atoms encapsulated in endohedral ions A@C60. Phys. Rev. A 2006, 73, 013201. [Google Scholar] [CrossRef]
- Dolmatov, V.K.; Craven, G.T.; Guler, E.; Keating, D. Revivification of confinement resonances in the photoionization of A@C60 endohedral atoms far above thresholds. Phys. Rev. A 2009, 80, 035401. [Google Scholar] [CrossRef]
- Ludlow, J.A.; Lee, T.-G.; Pindzola, M.S. Double photoionization of atoms and ions confined in charged fullerenes. J. Phys. B 2010, 43, 235202. [Google Scholar] [CrossRef]
- Kumar, A.; Varma, H.R.; Deshmukh, P.C.; Manson, S.T.; Dolmatov, V.K.; Kheifets, A.S. Effect of coulomb confinement resonances on time delay in . J. Phys. Conf. Ser. 2015, 635, 092042. [Google Scholar] [CrossRef]
- Kumar, A.; Varma, H.R.; Deshmukh, P.C.; Manson, S.T.; Dolmatov, V.K.; Kheifets, A. Wigner photoemission time delay from endohedral anions. Phys. Rev. A 2016, 94, 043401. [Google Scholar] [CrossRef]
- Chaudhuri, S.K.; Mukherjee, P.K.; Fricke, B. Spectroscopy of low lying transitions of He confined in a fullerene cage. Eur. Phys. J. D 2016, 70, 196. [Google Scholar] [CrossRef]
- Chaudhuri, S.K.; Chaudhuri, R.K.; Mukherjee, P.K.; Chattopadhyay, S. A confinement induced spectroscopic study of noble gas atoms using equation of motion architecture: Encapsulation within fullerene’s voids. J. Chem. Phys. 2017, 147, 034111. [Google Scholar] [CrossRef]
- Thuppilakkadan, A.; Jose, J.; Varma, H.R. Systematic study of Coulomb confinement resonances of atoms trapped inside charged fullerenes. Phys. Rev. A 2020, 102, 062826. [Google Scholar] [CrossRef]
- Thuppilakkadan, A.; Jose, J.; Varma, H.R. Confinement enhanced spin-orbit interchannel coupling effect on the atomic photoionization. J. Phys. B 2021, 54, 145001. [Google Scholar] [CrossRef]
- Thuppilakkadan, A.; Jose, J.; Varma, H.R. Photoionization dynamics of Ar trapped in a fullerene anion: Coulomb confinement resonances in 2s subshell and its impact on the 3s subshell. Phys. Scr. 2021, 96, 104004. [Google Scholar] [CrossRef]
- Dubey, K.A.; Jose, J. Elastic electron scattering from Ar@: Dirac partial-wave analysis. J. Phys. B 2021, 54, 115204. [Google Scholar] [CrossRef]
- Chaudhuri, S.K. Spectroscopy of He atom encapsulated in C60 cage: Low lying magnetic dipolar (M1) and magnetic quadrupolar (M2) transitions. Can. J. Phys. 2022, 100, 362. [Google Scholar] [CrossRef]
- Thuppilakkadan, A.; Banerjee, S.; Varma, H.R. Modifications in the angular photoemission time delay in Ar@: Coulomb confinement resonance as an amplifier of the spin-orbit-interaction-activated interchannel coupling effect. Phys. Rev. A 2023, 107, 052804. [Google Scholar] [CrossRef]
- Lohr, L.L.; Blinder, S.M. Electron photodetachment from a Dirac bubble potential. A model for the fullerene negative ion . Chem. Phys. Lett. 1992, 198, 100. [Google Scholar] [CrossRef]
- Amusia, M.Y.; Baltenkov, A.S.; Krakov, B.G. Photodetachment of negative C60- ions. Phys. Lett. A 1998, 243, 99. [Google Scholar] [CrossRef]
- Dolmatov, V.K. Spin-Polarized Photoelectron Fluxes from Fullerene Anions. Atoms 2020, 8, 65. [Google Scholar] [CrossRef]
- Dolmatov, V.K.; Manson, S.T. A glimpse into photodetachment spectra of giant and nested fullerene anions. Atoms 2022, 10, 99. [Google Scholar] [CrossRef]
- Puska, M.J.; Nieminen, R.M. Photoabsorption of atoms inside C60. Phys. Rev. A 1993, 47, 1181. [Google Scholar] [CrossRef]
- Deshmukh, P.C.; Jose, J.; Varma, H.R.; Manson, S.T. Electronic structure and dynamics of confined atoms. Eur. Phys. J. D 2021, 75, 166. [Google Scholar] [CrossRef]
- Amusia, M.Y.; Chernysheva, L.V. Computation of Atomic and Molecular Processes: Introducing the ATOM-M Software Suite; Springer: Berlin/Heidelberg, Germany, 2021; Volume 117, ISBN 978-3-030-85142-2. [Google Scholar] [CrossRef]
- Chernysheva, L.V.; Yarzhemsky, V.G. Photoionization cross-sections of valence ns and np (n = 2, 3) atomic shells in soft x-ray region. At. Data Nucl. Data Tables 2024, 155, 101621. [Google Scholar] [CrossRef]
- Dolmatov, V.K.; Brewer, P.; Manson, S.T. Photoionization of atoms confined in giant single-walled and multiwalled fullerenes. Phys. Rev. A 2008, 78, 013415. [Google Scholar] [CrossRef]
- Baltenkov, A.S. Resonances in photoionization cross sections of inner subshells of atoms inside the fullerene cage. J. Phys. B 1999, 32, 2745. [Google Scholar] [CrossRef]
- Connerade, J.P.; Dolmatov, V.K.; Manson, S.T. On the nature and origin of confinement resonances. J. Phys. B 1999, 33, 2279. [Google Scholar] [CrossRef]
- Dolmatov, V.K. Photoionization of atoms encaged in spherical fullerenes. In Theory of Confined Quantum Systems. Part 2 (Advances in Quantum Chemistry); Sabin, J.R., Brändas, E., Eds.; Academic: New York, NY, USA, 2009; Volume 58, pp. 13–68. [Google Scholar]
- Sabin, J.R.; Brändas, E.; Cruz, S.A. (Eds.) Theory of Confined Quantum Systems. Part 1, (Advances in Quantum Chemistry Volume 57); Academic: New York, NY, USA, 2009; 297p. [Google Scholar]
- Sabin, J.R.; Brändas, E.; Cruz, S.A. (Eds.) Theory of Confined Quantum Systems. Part 2, Advances in Quantum Chemistry Volume 58); Academic: New York, NY, USA, 2009; 334p. [Google Scholar]
- Sen, K.D. (Ed.) Electronic Structure of Quantum Confined Atoms and Molecules; Springer: Cham, Switzerland, 2014. [Google Scholar]
- Ley-Koo, E. Recent progress in confined atoms and molecules: Superintegrability and symmetry breakings. Rev. Mex. Fis. 2018, 64, 326. [Google Scholar] [CrossRef]
- Chakraborty, H.S.; Varma, H.R. Photon and Particle Impact Spectroscopy and Dynamics of Atoms, Molecules, and Clusters. Atoms 2023, 11, 156. [Google Scholar] [CrossRef]
- Nielson, C.W.; Koster, G.F. Spectroscopic Coefficients for the pn, dn, and fn Onfigurations; The MIT Press: Cambridge, MA, USA, 1963. [Google Scholar]
- Sobelman, I.I. Atomic Spectra and Radiative Transitions; Springer: Berlin/Heidelberg, Germany, 1992; ISBN 978-3-540-54518-7. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).