Abstract
The performance improvement of mechanical components often relies on heat treatment processes, but these processes inevitably result in oxidation burn-off. The repeated formation and spallation of Fe2O3 rich oxide scales lead to substantial iron depletion and surface deterioration. Consequently, environmentally sustainable and economically viable protective coatings are required to suppress oxidation induced burn off. In this work, a TiO2-MgAl2O4 composite coating was synthesized from magnesium slag and applied to Q235 carbon steel to enhance its performance during prolonged high temperature heat treatment. Oxidation tests conducted at 900 °C for 60 min demonstrated that the coating markedly improved the oxidation resistance of carbon steel, with an enhancement of approximately 87% relative to the uncoated specimens. To elucidate the protective mechanism, SEM-EDS, XRD, TG-DSC, and XPS analyses were employed. Based on Wagner Theory, the formation of interfacial phases such as Mg7.92Al15.31Fe0.66O32, which effectively impeded oxygen ion diffusion and thereby enhanced the oxidation resistance during high-temperature exposure. Furthermore, the synergistic effect of aluminum-, magnesium-, and titanium-containing compounds in the coating contributed to suppressing the diffusion of oxygen and iron ions, thus further improving the protective performance. This study provides a systematic theoretical foundation and practical guidance for addressing material loss during high-temperature processing of mechanical components, as well as for promoting the resource utilization of magnesium slag.
1. Introduction
Heat treatment is one of the core approaches for optimizing the mechanical properties of high-performance mechanical components [1]. Under high-temperature conditions, the internal microstructure of components undergoes transformation, which improves their mechanical performance, but this process is inevitably accompanied by surface oxidation [2,3]. In high-temperature environments, the surface of mechanical components readily forms a composite oxide layer mainly composed of FeO, Fe3O4, and Fe2O3 [4]. Among these oxides, the relatively dense inner FeO/Fe3O4-rich layer can temporarily retard oxygen diffusion and slow further oxidation of the steel substrate, thereby providing limited short-term protection [5]. Nevertheless, most oxide scales are loose and porous, with poor structural stability and a strong tendency to spall [6]. Such non-compact oxide layers not only fail to provide effective protection but also lead to significant material loss of the substrate. More importantly, to meet surface quality and dimensional accuracy requirements, the oxide layer must be removed by sandblasting, pickling, or mechanical machining [3,7]. This not only increases manufacturing steps and production costs, but may also introduce additional dimensional deviations due to uneven removal or tolerance exceedance, and in severe cases, result in component scrap.
Q235 steel is widely used in construction, bridge engineering, and manufacturing because of its balanced combination of strength, ductility, toughness, and weldability [8]. In forging-related applications, it can also be used as a punch material, for which heat treatment is commonly employed to improve service performance. However, during heat treatment, especially above 900 °C, the surface of Q235 is highly susceptible to the formation of a multilayer oxide scale consisting primarily of inner FeO or Fe3O4 and outer Fe2O3 [9]. Although the residual FeO/Fe3O4 layer after Fe2O3 spallation may provide limited corrosion resistance, the predominance of Fe2O3 in the oxide scale leads to substantial iron loss during repeated formation and exfoliation processes [10]. Consequently, pronounced material consumption occurs during high-temperature treatment, reducing material utilization efficiency and compromising dimensional precision. Therefore, developing an effective strategy that can suppress oxidation loss while maintaining a relatively protective surface layer is of considerable engineering significance.
Based on the above considerations, numerous strategies have been explored to mitigate high-temperature oxidation in carbon steel, including reducing its intrinsic oxidation tendency and modifying the surrounding atmospheric conditions [11]. However, these approaches are often associated with high energy consumption and operational complexity, which limit their practical implementation. Protective coatings, in contrast, have been widely recognized as an effective and scalable solution for enhancing the oxidation resistance of steel, and thus have attracted considerable research attention [12,13]. Extensive efforts have focused on the development of coatings via sol–gel techniques, employing materials such as silica (SiO2), alumina (Al2O3), ceria (CeO2), nano-titania (TiO2), zirconia–silica (ZrO2–SiO2), alumina–silica (Al2O3–SiO2), and multi-component systems including SiO2–TiO2–ZrO2 [14,15,16,17,18,19,20]. These coatings have demonstrated effective protection for low-carbon steel, stainless steel, magnesium, aluminum, and their alloys against chemical corrosion and high-temperature oxidation. Although such studies have established a solid theoretical framework and provided important technical support for the development of environmentally friendly anti-oxidation coatings, challenges remain in terms of high material costs and substantial consumption, which constrain large-scale industrial application. The utilization of solid waste resources for the fabrication of anti-oxidation coatings offers a promising pathway toward both environmental sustainability and economic feasibility [21]. Current mainstream approaches involve industrial by-products such as fly ash, waste glass, and scrap metals as raw materials for coating preparation [22,23]. For instance, composite coatings composed of SiO2 and SiC with Al2O3 have exhibited excellent resistance to high-temperature oxidation and thermal corrosion [24], while MgO-based coatings have shown notable protective performance for steel substrates under elevated temperatures [25]. However, previous studies have primarily focused on improving the oxidation resistance of metallic materials, including minimizing the formation of oxide scales and exploring green preparation routes for antioxidant coatings. For punch materials, however, the heat treatment process requires both the formation of a dense oxide layer to provide corrosion protection during service and the control of material loss caused by high-temperature oxidation. A dual-functional coating that simultaneously enables corrosion resistance and reduces oxidation loss has yet to be achieved in existing research.
In this work, a comparative analysis was conducted to evaluate the oxidation resistance of untreated Q235 carbon steel and Q235 carbon steel coated with MgAl2O4 (FM) and TiO2–MgAl2O4 (FTM) ceramic coatings. Advanced characterization techniques, including X-ray diffraction (XRD), field-emission scanning electron microscopy (FE-SEM), energy-dispersive X-ray spectroscopy (EDX), and thermogravimetric–differential scanning calorimetry (TG-DSC), were employed to systematically investigate the physicochemical properties of the ceramic coatings, with particular emphasis on its role in enhancing oxidation resistance. In addition, the potential protective mechanisms of the ceramic coating were comprehensively analyzed, providing scientific support for the sustainable utilization of magnesium slag in anti-oxidation applications.
2. Materials and Methods
The raw materials employed in this study included Light-burned magnesia (MgO, 95 wt.%), titanium dioxide (TiO2, 99 wt.%), all of which were procured from Shanghai Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). Aluminum powder (Al, 99.5 wt.%) was sourced from Merck Biotechnology and Jinzhou Ferroalloy Co., Ltd. (Jinzhou, China). Additionally, N-Methyl-2-pyrrolidone (C5H9NO, 99 wt.%) and polyvinylidene fluoride ((CH2CF2)n, 99 wt.%) were supplied by Shanghai McLean Biochemical Technology Co., Ltd. (Shanghai, China). All the aforementioned reagents were of analytical grade and were used as received without further purification.
2.1. Treatment of Steel Sample
The Q235 carbon steel (Baoshan Iron & Steel Co., Ltd., Shanghai, China), having undergone the normalizing process, was precisely sectioned into cubic specimens measuring 10 mm × 10 mm × 10 mm utilizing a high-speed wire electrical discharge machining apparatus (DK7732, Suzhou Baoma Numerical Control Equipment Co., Ltd., Suzhou, China). The specimens were polished using an automatic grinding machine (Alpha-108, Trojan (Suzhou) Material Technology Co., Ltd., Suzhou, China), ultrasonically cleaned to remove contaminants, and dried at 90 °C. The chemical composition of Q235 carbon steel is presented in Table 1.

Table 1.
Chemical composition of carbon steel.
2.2. Synthesis of Coating
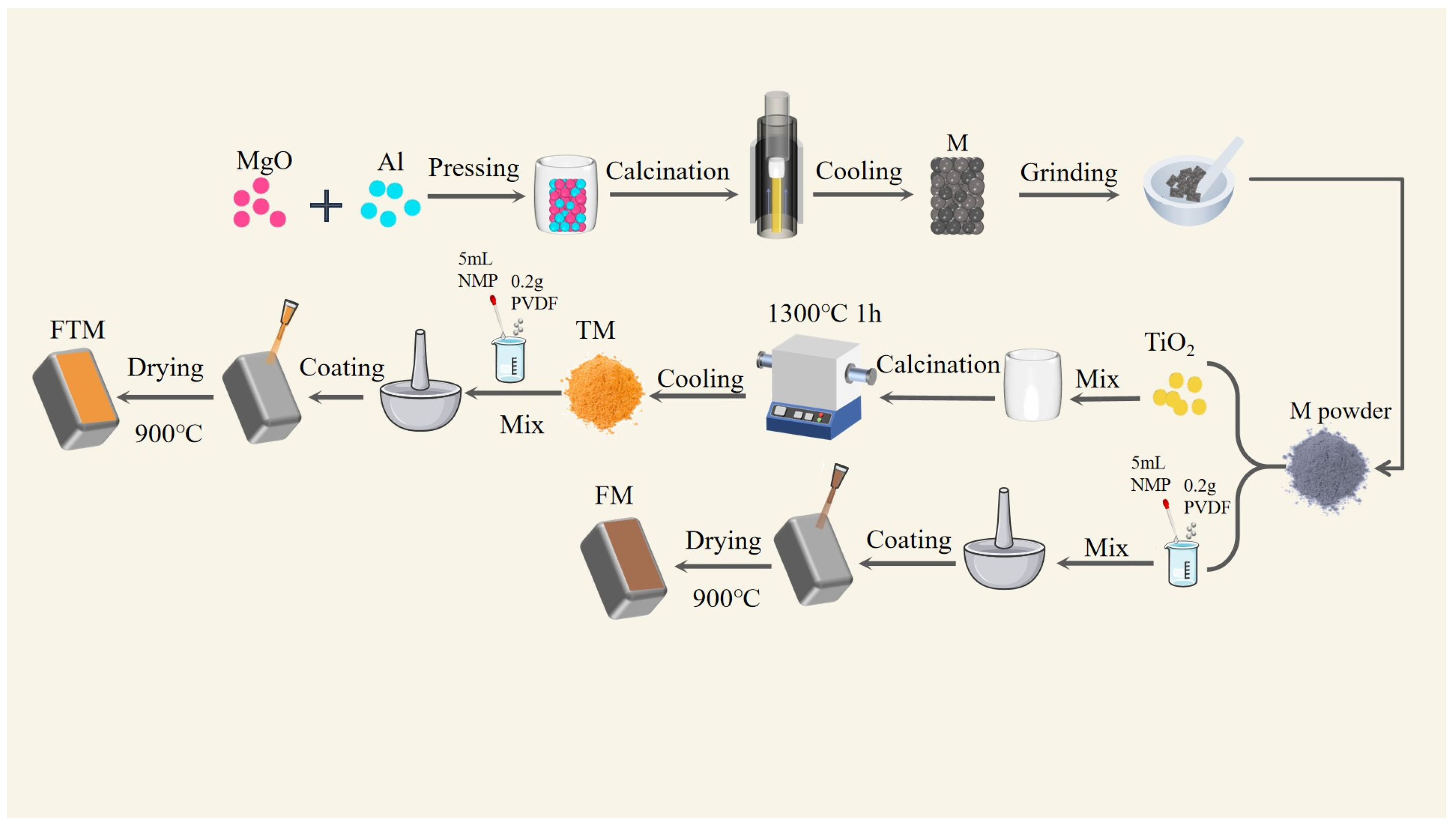
As shown in Figure 1, initially, a mixture of 1.69 mol Light-burned magnesia and 1.19 mol aluminum powder was compressed into tablets under a pressure of 30 MPa. These tablets underwent calcination at 1300 °C in an argon atmosphere flowing at 0.2 m3/h for 1 h, yielding magnesium slag (denoted as M) and magnesium vapor. Magnesium vapor, after condensation and recovery, can be used as crude magnesium for magnesium alloy smelting, while the remaining magnesium slag is a typical solid waste. Subsequently, 0.5 g of the synthesized M was finely ground and combined with titanium dioxide. The mixture was then placed in a corundum crucible, which had an outer diameter of 45 mm and a height of 58 mm. The mixture was subjected to calcination at 1300 °C for 1 h, resulting in the formation of titanium dioxide composites (referred to as TM). Following this, 5 mL of N-methylpyrrolidone (NMP) and 0.2 ± 0.001 g of polyvinylidene fluoride (PVDF) were introduced into a mortar and thoroughly ground to achieve a clear sol. Subsequently, 0.5 ± 0.001 g of TM composite powder or M powder was incorporated into the as-prepared sol under continuous grinding to ensure uniform dispersion, yielding the FTM and FM coating slurries, respectively. This slurry was then applied onto Q235 carbon steel substrates using a low-pressure spray gun (model W-77 C Anest Iwata Corporation, Yokohama, Japan). Finally, both the FTM/FM-coated steel samples and their uncoated counterparts were positioned within a corundum crucible measuring 30 × 20 × 17 mm for further analysis or processing.
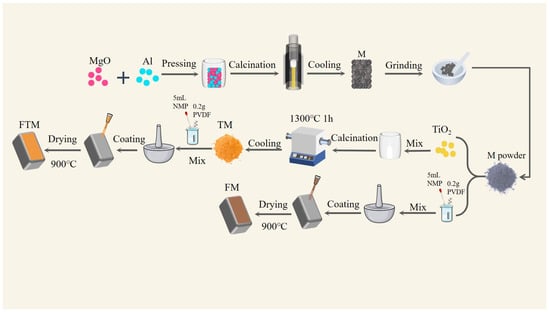
Figure 1.
Flow chart of coating slurry preparation.
2.3. Characterization of the Coating
The oxidation resistance of the coatings was assessed by measuring the weight change in the specimens. MgAl2O4-coated (FM) and TiO2–MgAl2O4-coated (FTM) samples were heated from 25 °C to 900 °C, followed by isothermal exposure for 60 min. After cooling, the oxide scale and coatings were removed, and the specimens were reweighed. To quantify the substrate loss after oxidation, the mass retention rate (α) of each specimen was first calculated using Equation (1), where ma and mb denote the specimen weights before and after oxidation after removing the scale, respectively. The parameter α reflects the residual mass fraction of the steel substrate after heat treatment and was used as an indicator of substrate retention during oxidation. Based on this result, the oxidation resistance efficiency of the coating (E) was further calculated using Equation (2) [26] by comparing the mass retention rates of coated and uncoated specimens. Here, αc and αu represent the mass retention rates of the coated and uncoated specimens, respectively. In this way, Equations (1) and (2) were introduced to quantitatively evaluate the effectiveness of the coatings in reducing oxidation loss of the steel substrate. For each condition, three FM and three FTM specimens were tested, and the average values were reported.
After removing the oxide scale, the samples were weighed to determine the mass loss due to oxidation, thereby evaluating the oxidation resistance of the coating. The phase composition of the coatings were analyzed using X-ray diffraction (XRD) with Cu Kα radiation (λ = 0.154 nm), scanning from 10° to 90° at a rate of 10°/min. X-ray photoelectron spectroscopy (XPS) was performed using a Thermo Scientific K-Alpha spectrometer (Thermo Scientific K-Alpha XPS System, Thermo Fisher Scientific, East Grinstead, UK) equipped with an Al Kα X-ray source (hν = 1486.6 eV). Survey spectra were collected with a pass energy of 150 eV and a step size of 1 eV, while high-resolution spectra were collected with a pass energy of 50 eV and a step size of 0.1 eV. At least five scanning cycles were accumulated for each high-resolution spectrum depending on the analyzed element. All binding energies were calibrated using the C 1s peak at 284.8 eV, and peak fitting was performed using XPSPEAK software (version 4.1). No sputtering was performed prior to analysis. The surface morphology and micro-area composition of the samples were further characterized using scanning electron microscopy ((SEM, GeminiSEM 300, Carl Zeiss Microscopy Deutschland GmbH, Oberkochen, Germany). Additionally, simultaneous thermal analysis (TG–DSC) was conducted using a simultaneous thermal analyzer (STA 449 F3 Jupiter®, NETZSCH-Gerätebau GmbH, Selb, Germany) to investigate the thermal effects and mass changes of the samples during heating from 30 °C to 900 °C at a heating rate of 10 °C/min.
3. Results
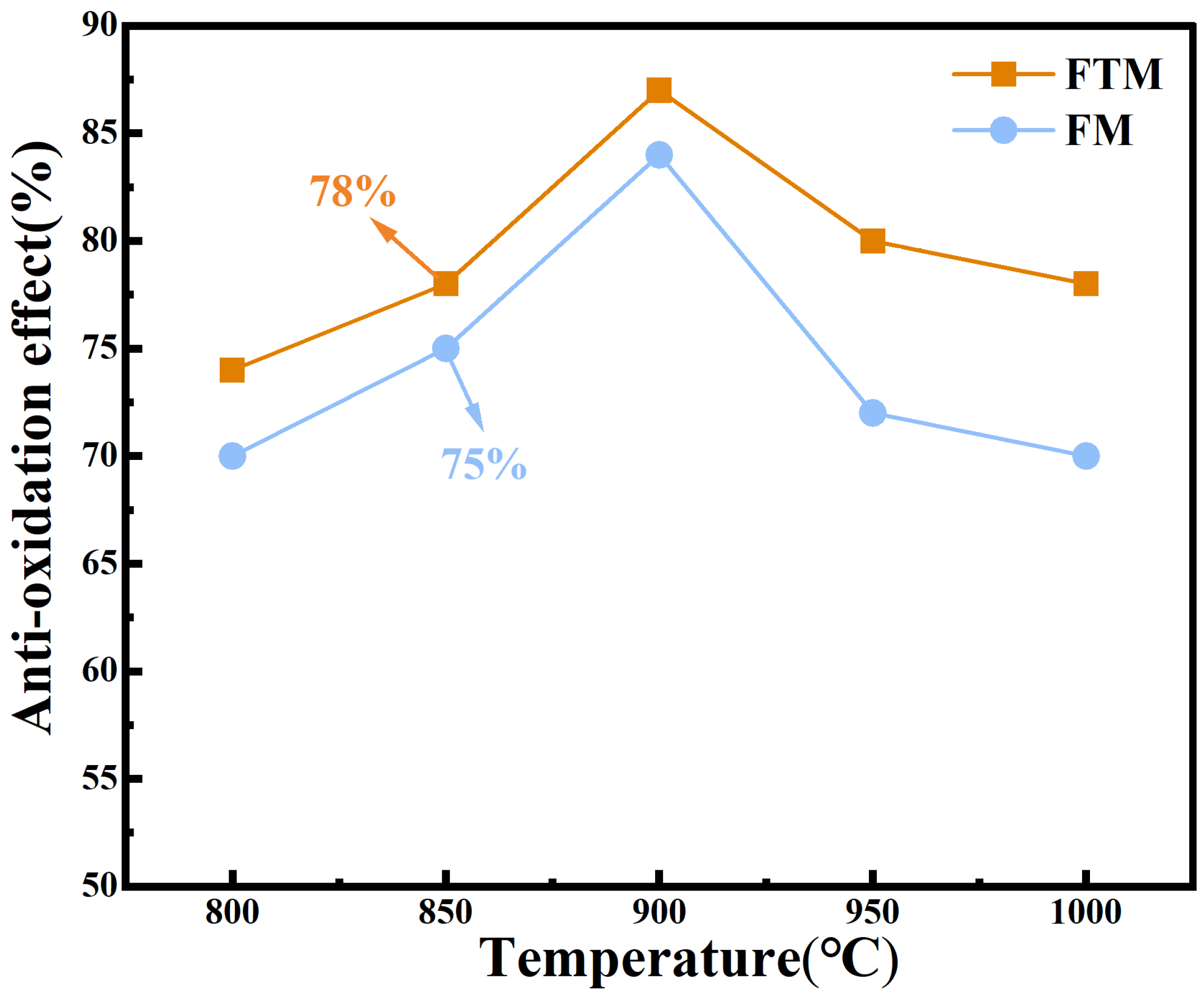
In order to evaluate the overall oxidation resistance of the Q235 steel coating, heat treatment tests were performed on the FM and FTM samples, and the corresponding antioxidant effect are shown in Figure 2. It can be seen that the FTM sample exhibits excellent oxidation resistance, and the E value of FM is lower than that of FTM at all experimental temperatures. In addition, with the increase in temperature, the E value reaches a minimum value of 78% at 850 ° C, but decreases slightly at 900 ° C (87%~79% ). This behavior is attributed to the formation of a dense FexO oxide layer during oxidation, which acts as a barrier to O2 penetration [27]. In this study, the coating generated an even denser protective layer at high temperatures, further restricting the interaction and diffusion of O2 toward the Q235 steel substrate [28]. Whether this dense layer is an intrinsic property of the coating or develops specifically under high-temperature conditions requires further investigation. In this study, the coating formed an even denser protective layer at high temperatures, further preventing the interaction between O2 and the Q235 steel substrate. Whether this high-density protective layer is an intrinsic property of the coating itself or a special characteristic that emerges under high-temperature conditions remains to be further investigated. To validate this hypothesis, thermogravimetric-differential scanning calorimetry (TG-DSC) tests were conducted on the coatings under a constant heating rate (30–900 °C, 10 °C/min) (Figure 3a–c). At low temperatures (below 200 °C), endothermic peaks were observed due to the evaporation of water or organic components in the coating. Subsequent endothermic peaks were attributed to the oxidation of Fe or the further oxidation of its oxides. At 900 °C [29], the mass gains of the FTM and FM samples were 14.96% and 20.26%, respectively, further confirming the superior oxidation resistance of FTM.
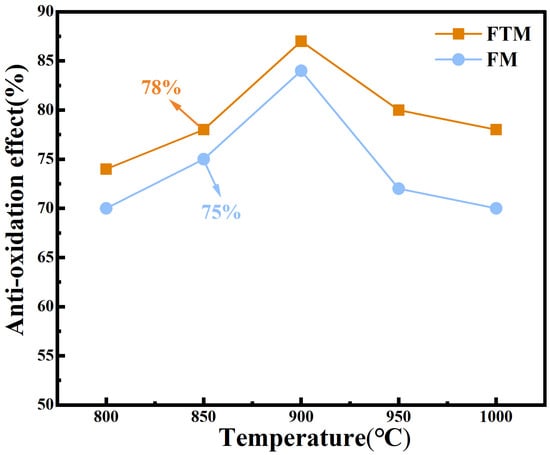
Figure 2.
Oxidation resistance of the coating at 800–1000 °C for 60 min in air.
Notably, an endothermic peak accompanied by mass loss appeared at 441.35 °C, likely due to interactions between MgAl2O4 and the Q235 steel substrate, leading to the formation of composite compounds and the release of H2O or CO2. As shown in Figure 3d–g, the introduction of TiO2 and the involvement of iron oxides significantly modify the high-temperature phase equilibria of the MgO–Al2O3 system, leading to the formation of new spinel stability regions within specific temperature and compositional ranges. Figure 3d indicates that with increasing TiO2 content, a stable Ti–spinel-related phase field emerges in the intermediate-to-high temperature region, suggesting that Ti4+ can participate in the spinel lattice construction or substitute into crystallographic sites, thereby forming thermodynamically stable titanium-containing spinel phases and broadening the stability range of the spinel structure. Figure 3e–g further demonstrate that in the presence of FeO, Fe2O3, or Fe3O4, “Monoxide + Spinel” or related spinel coexistence regions are observed, indicating that iron, regardless of its oxidation state, possesses a thermodynamic driving force to incorporate into the spinel lattice and form iron spinel phases and corresponding solid solutions.
For the MgAl2O4 spinel coating applied on a steel substrate, iron is not introduced as an external dopant but originates from the outward diffusion and interfacial reactions of the steel matrix during high-temperature oxidation. Therefore, the spinel stability regions revealed in the phase diagrams provide direct thermodynamic guidance for interfacial reactions. Under elevated-temperature service conditions, Fe diffusing from the steel substrate can react with MgO–Al2O3 components in the coating, leading to the in situ formation of iron spinel or composite spinel interfacial layers. Owing to the high thermal stability and low ionic diffusivity of the spinel structure, the formation of such layers contributes to the development of a dense and stable reaction barrier, effectively suppressing inward oxygen diffusion and outward iron migration, thereby enhancing interfacial stability and overall oxidation resistance. From a thermodynamic perspective, the stability regions of Ti-containing and Fe-containing spinel phases provide theoretical support for the high-temperature oxidation performance of MgAl2O4 spinel coatings on steel substrates.
The presence of these newly formed phases was confirmed by the XRD patterns in Figure 3h, which, in addition to Fe2O3, Fe3O4, and MgAl2O4, included MgTi2O5, MgFe2O4, Mg7.92Al15.31Fe0.66O32, and FeTiO5 [28,30,31,32]. This indicates that at high temperatures, the coating (MgAl2O4) reacts with TiO2 and the oxide layer on Q235 steel to form a high-density protective layer with a spinel-type structure.
In theory, a protective layer capable of blocking oxygen typically exhibits a dense microstructure. To verify the density of the coating, the cross-sections of the samples were examined using scanning electron microscopy (SEM) (Figure 4a–c). The uncoated Q235 samples exhibited a clearly loose and porous surface layer, which provides channels for O2 diffusion and thus cannot effectively prevent oxidation. In contrast, after coating with FM and FTM, the original porous structure disappeared, and a dense protective layer formed at the interface. Notably, in the FTM coating, TiO2 migrated toward the outer surface at high temperatures, forming voids. This indicates that TiO2 not only enhances the oxidation resistance of the coating but also promotes its peelable behavior. In conjunction with the previous analysis of oxidation weight-gain behavior, the FTM coating is not only prone to spallation but also results in the formation of an extremely thin oxide layer on the substrate surface. This finding indicates that the FTM coating effectively mitigates material loss caused by high-temperature oxidation while simultaneously establishing a protective layer with certain corrosion resistance on the substrate. These results further confirm the feasibility and effectiveness of the hypothesis proposed in this study.
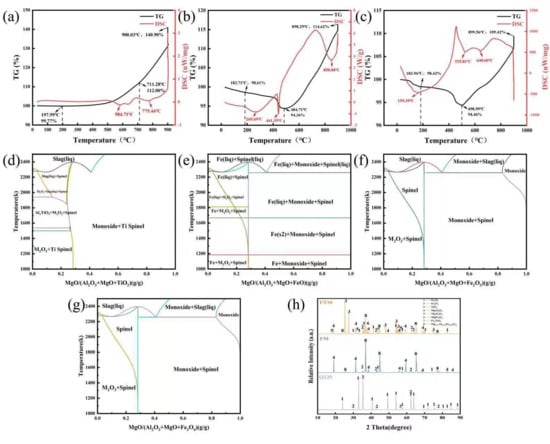
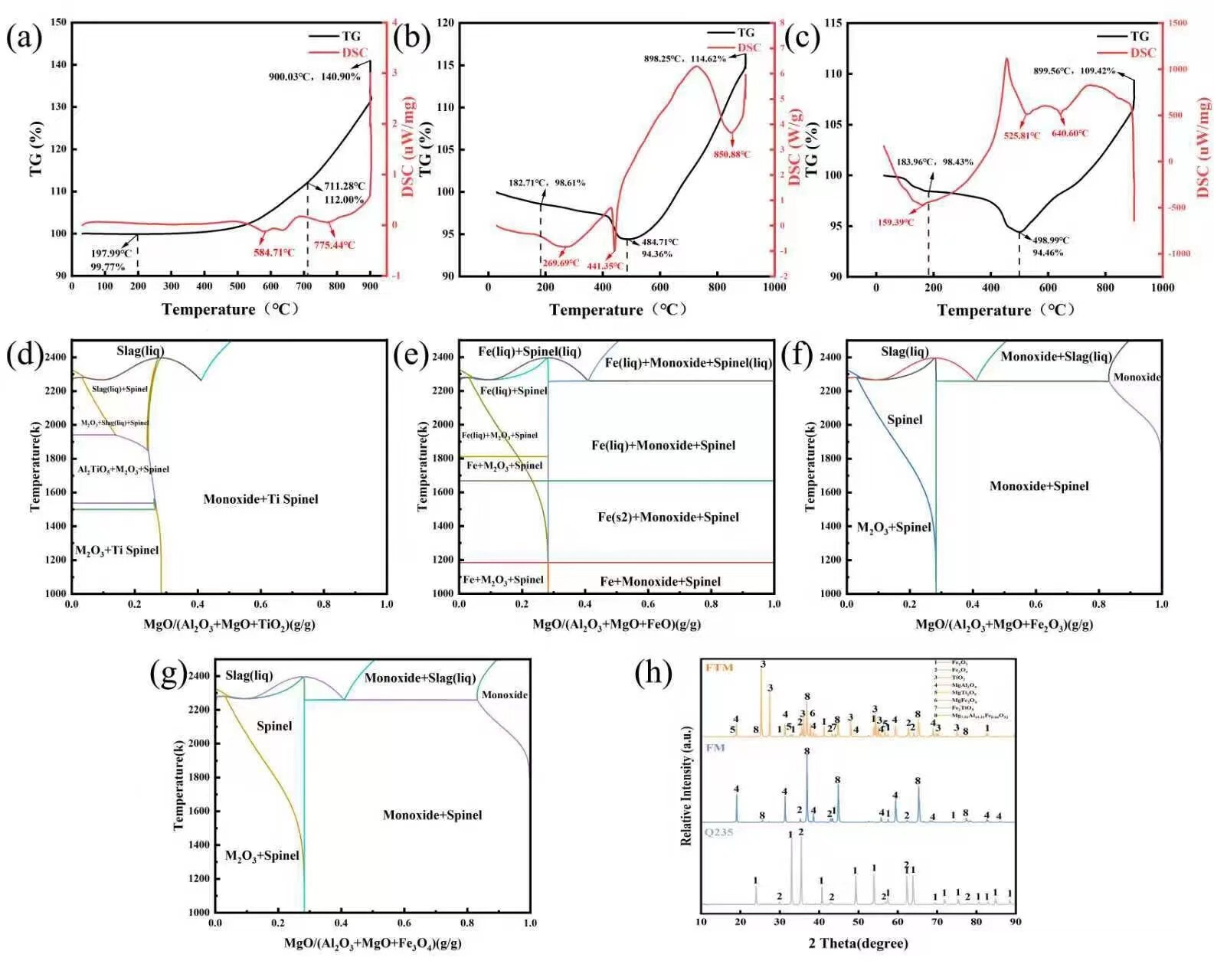



Figure 3.
TG-DSC curves of (a) Q235; (b) FM; (c) FTM samples; (d) MgO-Al2O3-TiO2 system phase diagram; (e) MgO-Al2O3-FeO system phase diagram; (f) MgO-Al2O3-Fe2O3 system phase diagram; (g) MgO-Al2O3-Fe3O4 system phase diagram and (h) XRD patterns of samples.
Figure 3.
TG-DSC curves of (a) Q235; (b) FM; (c) FTM samples; (d) MgO-Al2O3-TiO2 system phase diagram; (e) MgO-Al2O3-FeO system phase diagram; (f) MgO-Al2O3-Fe2O3 system phase diagram; (g) MgO-Al2O3-Fe3O4 system phase diagram and (h) XRD patterns of samples.


Figure 4.
SEM images of (a) uncoated Q235 steel, (b) FM-coated Q235 steel, and (c) FTM-coated Q235 steel after high-temperature oxidation treatment.
Figure 4.
SEM images of (a) uncoated Q235 steel, (b) FM-coated Q235 steel, and (c) FTM-coated Q235 steel after high-temperature oxidation treatment.

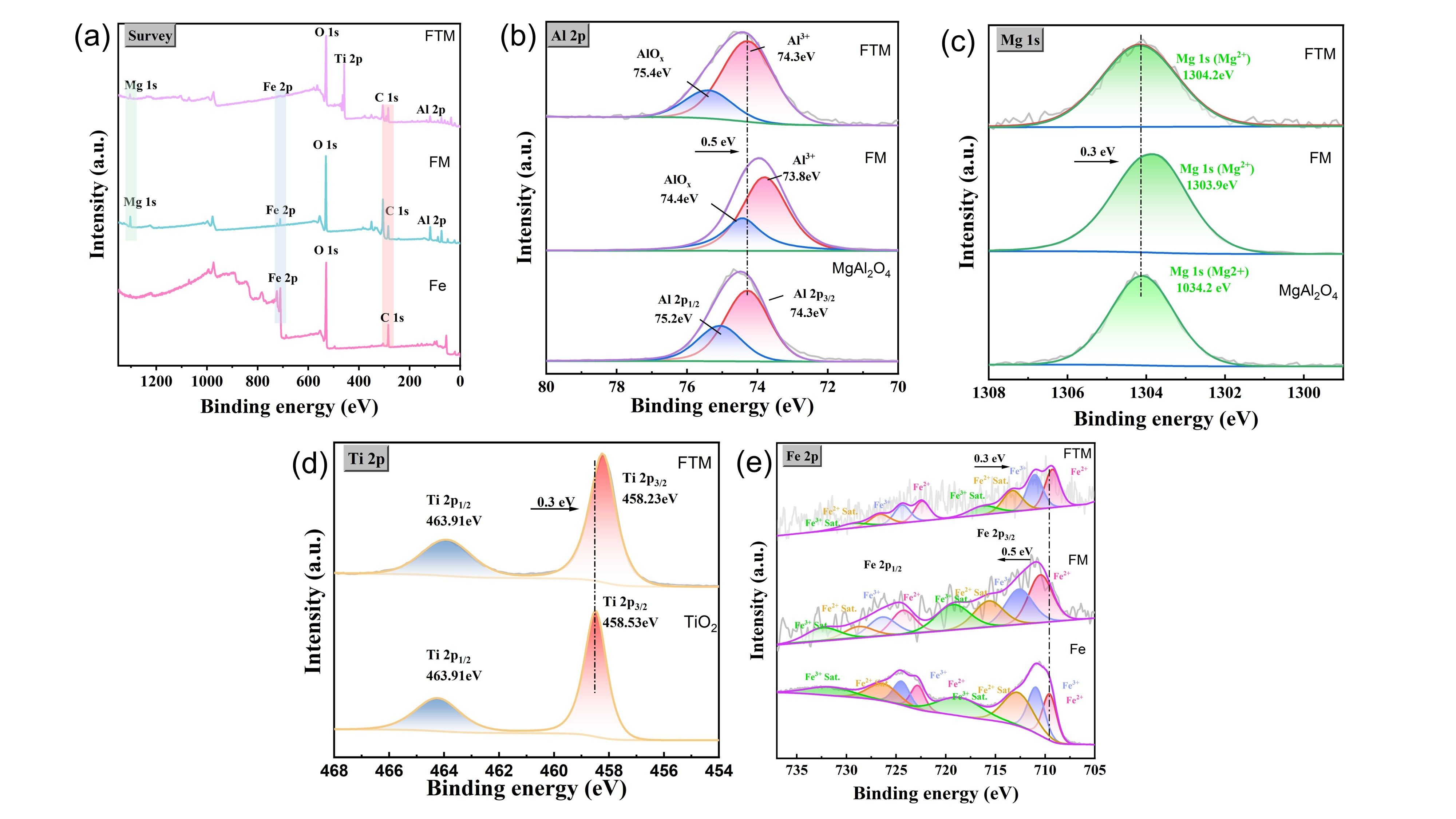
To further investigate the high-temperature properties of the coating, X-ray photoelectron spectroscopy (XPS) was conducted (Figure 5 and Table 2) to elucidate the interactions between MgAl2O4 and Fe and Ti, providing insight into the chemical mechanisms underlying the observed protective behavior. To further determine the composition of the surface oxide film, XPS analysis was conducted, and the results are shown in Figure 5a. Compared with bare Q235 steel, new signals associated with Ti, Mg, and Al were observed in the FTM and FM samples, indicating that these elements were involved in the high-temperature oxidation process and contributed to the formation of new, stable surface phases. For a more detailed analysis, the high-resolution XPS spectra shown in Figure 5b–e confirm the prominent features of the Al 2p peak (74.3 eV) and Mg 1s peak (1304.2 eV) associated with MgAl2O4 in all samples. Notably, the Al 2p (74.3 eV) and Mg 1s (1304.2 eV) peaks in the FTM coating coincide with those of MgAl2O4 [33]. However, in the FM coating, these two peaks shift to lower binding energies to varying degrees (Al 2p at 73.8 eV, Mg 1s at 1303.9 eV). According to Table 1, the binding energy of Mg2+ is 1304.2 eV, while that of metallic Mg is 1303 eV, indicating that the Mg in the FM coating exhibits a valence state between 0 and +2. Similarly, the valence state of Al in the FM coating lies between 0 and +3. Furthermore, the valence state of Ti in the FTM coating changes from +4 to +2, likely due to the presence of oxygen vacancies. These oxygen vacancies cause changes in the electronic structure by introducing defects. By comparing the high-resolution Fe 2p spectra for each sample, In the Q235 sample, the Fe 2p3/2 and Fe 2p1/2 peaks are located in the lower binding energy region (around 710–712 eV), which are characteristic peaks of Fe2+ and Fe3+, accompanied by satellite peaks, indicating the presence of oxides or hydroxyl compounds on the substrate surface [34]. As the MgAl2O4 coating chemically interacts with the Q235 steel surface in the FM sample, some Fe atoms are embedded into the coating or react with oxygen atoms in the coating to form new oxides, resulting in an increased tendency for Fe2+ to convert to Fe3+. This leads to changes in the intensity distribution of Fe2+ and Fe3+, with the peak positions of Fe 2p slightly shifting, an increase in satellite peak intensity, and a slight rise in binding energy (about 0.5 eV). TiO2, with its strong oxidizing nature, promotes the oxidation of Fe, further enhancing this effect in the FTM sample. The introduction of titanium dioxide alters the chemical environment of iron, causing the Fe 2p peaks in the FTM sample to shift further (about 0.3 eV). At the same time, the intensity of the Fe3+ peak increases significantly, while the intensity of Fe2+ decreases, and the energy gap between the satellite peak and the main peak narrows. The introduction of the coating changes the concentration and distribution of surface oxygen, and the binding energy shift reflects the transition of Fe’s chemical environment from metallic to higher oxidation states, which is related to the coordination environment of oxygen atoms in the coating. Magnesium aluminate spinel and titanium dioxide coatings contain oxygen vacancies [31], which lead to a local decrease in electron density and enhance the interaction between Fe and oxygen [35]. These vacancies attract Fe2+ ions to lose electrons and convert to Fe3+ in order to maintain charge neutrality. The addition of titanium dioxide in the FTM coating strengthens the oxidizing environment, leading to an increase in the proportion of Fe3+. During the preparation of FM and FTM coatings, oxygen atoms may partially escape due to high temperatures or a reducing atmosphere, forming oxygen vacancies. These vacancies create local charge imbalances in the lattice. Near the oxygen vacancies, Fe2+ tends to lose electrons to compensate for the charge, converting to Fe3+, which is reflected in the enhancement of the Fe3+ peak and the increase in binding energy. In the FTM coating, titanium dioxide’s strong oxidizing nature reduces the number of oxygen vacancies, but its high oxidation potential further drives the conversion of Fe2+ to Fe3+ [36]. Meanwhile, the intensity and position changes in the satellite peaks reflect the reorganization of the electronic structure. The increased local disorder caused by oxygen vacancies enhances the multi-electron excitation effect, thereby amplifying the satellite peak signal. The binding energy shift in the Fe 2p peaks and the changes in oxidation state ratios are primarily attributed to the chemical interactions between the magnesium aluminate spinel and titanium dioxide coatings and the Q235 steel surface, with oxygen vacancies playing a key role in this process.
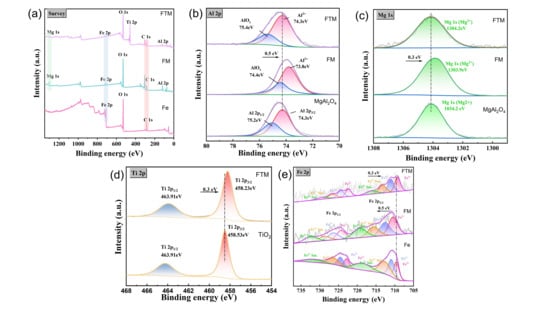
Figure 5.
XPS analysis of samples: (a) survey spectra; (b) Al 2p spectra; (c) Mg 1s spectra; (d) Ti 2p spectra and (e) Fe 2p.

Table 2.
Corresponding binding energies and chemical states of Al and Mg.
4. Protection Mechanism
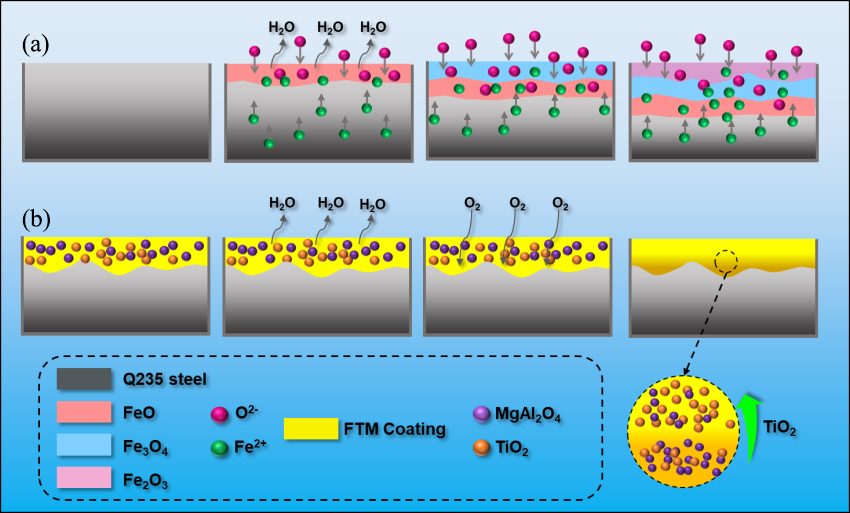
At a high temperature of 900 °C, the oxidation behavior of Q235 steel differs significantly from that at room temperature, primarily due to the occurrence of intense high-temperature oxidation reactions (Equations (3)–(5)), which rapidly lead to the formation of an initial oxide scale on the metal surface [37]. This oxide layer mainly consists of FeO, Fe3O4, and Fe2O3, arranged in a distinct layered structure: Fe2O3, which is loose and porous, forms the outermost layer; Fe3O4 occupies the intermediate layer; and FeO, which is thermodynamically less stable, lies adjacent to the metal substrate (as shown in Figure 6a). Based on the Mott–Cabrera theory, the growth of metal oxide films at low temperatures is predominantly governed by the electric field–driven migration of ions and electrons within the oxide layer [38]. At the early stage of oxidation, oxygen molecules first adsorb on the metal surface and gain electrons to be reduced into O2- ions (Equations (6) and (7)). This reduction leads to charge separation across the oxide film, establishing a strong internal electric field within the layer. Driven by this electric field, Fe2+ ions migrate outward from the metal/oxide interface and react with O2− to form iron oxides. Simultaneously, some O2− ions can also diffuse inward from the gas/oxide interface, facilitating the continued growth of the oxide scale [39]. Notably, Fe3O4 possesses a certain degree of electrical conductivity, allowing for electron transport within the film and thereby promoting further ionic migration and oxidation layer development. In contrast, the oxidation resistance mechanism of the FTM (Functional Multilayer) coating is more intricate (as illustrated in Figure 6b). Based on Wagner’s theory, the high-temperature growth of metal oxide films is predominantly governed by the bulk diffusion of oxygen or metal ions through the oxide scale [40]. Under high-temperature conditions, oxygen molecules first adsorb onto the surface of the coating, particularly on TiO2, and are reduced to O2− ions by acquiring electrons transferred from the metal substrate (Equation (7)). TiO2, being an n-type semiconductor, contains a conduction band rich in free electrons, serving as an efficient electron transport medium and facilitating the reduction in O2. This electron transfer from the substrate through the oxide film to the surface results in a potential difference between the metal/oxide interface and the outer surface, which induces a strong built-in electric field. Under this electric field, O2− ions can migrate inward through the TiO2 and MgAl2O4 layers toward the metal surface. Among these, TiO2 imposes a moderate barrier to O2− migration, while MgAl2O4, with its dense spinel structure and extremely low electrical conductivity, exhibits a strong blocking effect [41]. The combination of the two layers creates an effective gradient diffusion barrier that significantly restricts the inward transport of oxygen ions. Additionally, at elevated temperatures, iron atoms ionize to form Fe2+ (Equation (6)), which diffuse outward under the influence of the electric field and react with O2− to form iron oxides such as FeO (Equation (8)) and Fe3O4, resulting in the formation of an initial oxide film. This initial oxide layer is encapsulated beneath the FTM composite coating, serving as an additional diffusion barrier. Meanwhile, oxygen participates in the formation of multiple interfaces, including TiO2–MgAl2O4 and MgAl2O4–metal, within the multilayered coating. These interfaces introduce compositional discontinuities and diffusion barriers that impede the outward migration of metal ions, thereby significantly suppressing the inward progression of the oxidation reaction.
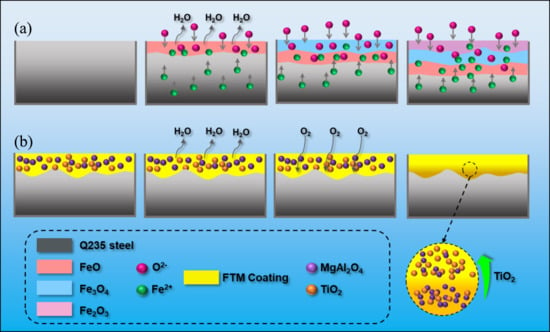
Figure 6.
Schematic illustration of the oxidation mechanism of Q235 steel after exposure at 900 °C for 60 min (a) Uncoated Q235; (b) FTM coated Q235.
5. Conclusions
For the heat treatment of steel, particularly during prolonged high-temperature heat treatment, surface oxidation poses a critical challenge. To address this issue, a TiO2–MgAl2O4 coating was proposed to enhance the oxidation resistance of steel and to mitigate the material loss caused by heat treatment. Based on this study, the main conclusions are as follows:
- The TiO2–MgAl2O4 (FTM) ceramic coating significantly enhanced the oxidation resistance of Q235 carbon steel during heat treatment at 900 °C. After 60 min of oxidation, the oxidation weight gain was reduced by 87% compared with that of the uncoated substrate, indicating that the FTM coating can effectively suppress high-temperature oxidation and reduce substrate loss. In addition, the coating exhibited favorable removability after oxidation, which is beneficial for practical post-treatment application.
- The improved oxidation resistance was closely related to the formation of dense interfacial reaction products during high-temperature exposure. XRD analysis showed that the interaction among MgAl2O4, TiO2, and iron oxides led to the formation of MgTi2O5, MgFe2O4, Mg7.92Al15.31Fe0.66O32, and FeTiO5, which contributed to the establishment of a stable protective barrier and effectively suppressed oxygen inward diffusion and iron outward migration.
- In the TiO2–MgAl2O4 ceramic coating, oxygen-vacancy engineering induced by lattice defects modulates the electronic structure, thereby facilitating the generation of Al-, Mg-, and Ti-oxide phases that function as protective layers, which are critical to the enhanced oxidation resistance of carbon steel.
- Based on the findings, this study offers critical insights into the rational design of oxidation resistant coatings for environmental and energy related applications, while providing a conceptual framework and practical guidance for the efficient and value oriented utilization of magnesium slag in coating development.
Author Contributions
Conceptualization, Z.D.; Methodology, Z.D. and T.Z.; Software, Y.L.; Validation, Z.D.; formal analysis, Z.D. and Y.L.; Investigation, Y.L.; Resources, Z.D. and T.Z.; data curation, Y.L. and Z.D.; writing—original draft preparation, Y.L.; writing—review and editing, Z.D. and T.Z.; Visualization, Z.D.; Supervision, T.Z.; project administration, Z.D. and T.Z.; funding acquisition, Z.D. and T.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the China Postdoctoral Science Foundation (No. 2023T160088) and the Youth Fund of the National Natural Science Foundation of China (No. 52304324) and the National Natural Science Foundation of China (U1908225, U1702253) and the Special Funds for Basic Research Operations of Central Universities (N182515007, N170908001, N2025004) and the Liaoning Revitalization Talents Program (XLYC2402029).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Abbreviations
The following abbreviations are used in this manuscript:
| FM | Q235 carbon steel coated with MgAl2O4 |
| FTM | Q235 carbon steel coated with TiO2–MgAl2O4 |
| XRD | X-ray diffraction |
| FE-SEM | Field-emission scanning electron microscopy |
| TG-DSC | Thermogravimetric–differential scanning calorimetry |
| EDX | Energy-dispersive X-ray spectroscopy |
References
- Laleh, M.; Sadeghi, E.; Revilla, R.I.; Chao, Q.; Haghdadi, N.; Hughes, A.E.; Xu, W.; De Graeve, I.; Qian, M.; Gibson, I.; et al. Heat Treatment for Metal Additive Manufacturing. Prog. Mater. Sci. 2023, 133, 101051. [Google Scholar] [CrossRef]
- Di Schino, A.; Testani, C. Heat Treatment of Steels. Metals 2021, 11, 1168. [Google Scholar] [CrossRef]
- An, J.; Chen, Y.; Liu, Z.; Tian, Y. Surface Oxidation Behavior and Wear Performance of a Fe-21.3Cr-3.5Al-0.5Ti-0.4Zr Steel. Metals 2020, 10, 1032. [Google Scholar] [CrossRef]
- Parkinson, G.S. Iron Oxide Surfaces. Surf. Sci. Rep. 2016, 71, 272–365. [Google Scholar] [CrossRef]
- Kang, J.; Chen, Z.; Zhu, X.; Zhou, S.; Zhou, L.; Wang, Z.; Wang, J.; Khater, G.A.; Yue, Y. Effect of Replacement of Na2O by Fe2O3 on the Crystallization Behavior and Acid Resistance of MgO Al2O3SiO2 Glass-Ceramics. J. Non-Cryst. Solids 2019, 503–504, 1–6. [Google Scholar] [CrossRef]
- Sun, B.; Cheng, L.; Du, C.-Y.; Zhang, J.-K.; He, Y.-Q.; Cao, G.-M. Effect of Oxide Scale Microstructure on Atmospheric Corrosion Behavior of Hot Rolled Steel Strip. Coatings 2021, 11, 517. [Google Scholar] [CrossRef]
- de Andrade, L.M.; Paternoster, C.; Chevallier, P.; Gambaro, S.; Mengucci, P.; Mantovani, D. Surface Processing for Iron-Based Degradable Alloys: A Preliminary Study on the Importance of Acid Pickling. Bioact. Mater. 2022, 11, 166–180. [Google Scholar] [CrossRef]
- Su, F.; Wen, Z. Experimental Study on Oxidation Behavior of Q235B Steel in Oxygen-Enriched Combustion Atmosphere. Oxid. Met. 2018, 89, 641–650. [Google Scholar] [CrossRef]
- Zambrano, O.A.; Coronado, J.J.; Rodríguez, S.A. Mechanical Properties and Phases Determination of Low Carbon Steel Oxide Scales Formed at 1200 °C in Air. Surf. Coat. Technol. 2015, 282, 155–162. [Google Scholar] [CrossRef]
- Gleeson, B.; David, J. Young: High Temperature Corrosion and Oxidation of Metals. Oxid. Met. 2011, 75, 121–123. [Google Scholar] [CrossRef]
- Fu, G.Y.; Wei, L.Q.; Zhang, X.M.; Cui, Y.B.; Lv, C.C.; Ding, J.; Yu, B.; Ye, S.F. A High-Silicon Anti-Oxidation Coating for Carbon Steel at High Temperature. Surf. Coat. Technol. 2017, 310, 166–172. [Google Scholar] [CrossRef]
- Pint, B.; Monceau, D. High-Temperature Protective Coatings. Oxid. Met. 2017, 88, 71. [Google Scholar] [CrossRef]
- Yatsenko, E.A.; Li, W.; Izvarin, A.I.; Goltsman, B.M.; Ryabova, A.V.; Kurdashov, V.M. Review of Protective Coatings for Pipelines. Int. J. Hydrogen Energy 2024, 110, 656–663. [Google Scholar] [CrossRef]
- Chen, T.; Bai, L.; Wen, K. Preparation and Molten Salt Corrosion Behaviors of TiO2/Al2O3/Incone1625 Composite Coatings on the Surface of Q235 Alloy at 900 °C. IOP Conf. Ser. Mater. Sci. Eng. 2019, 631, 022059. [Google Scholar] [CrossRef]
- Ruhi, G.; Modi, O.P.; Singh, I.B.; Jha, A.K.; Yegneswaran, A.H. Wear and Electrochemical Characterization of Sol-Gel Alumina Coating on Chemically Pre-Treated Mild Steel Substrate. Surf. Coat. Technol. 2006, 201, 1866–1872. [Google Scholar] [CrossRef]
- Hübert, T.; Svoboda, S.; Oertel, B. Wear Resistant Alumina Coatings Produced by a Sol–Gel Process. Surf. Coat. Technol. 2006, 201, 487–491. [Google Scholar] [CrossRef]
- Lgamri, A.; Guenbour, A.; Ben Bachir, A.; El Hajjaji, S.; Aries, L. Characterisation of Electrolytically Deposited Alumina and Yttrium Modified Alumina Coatings on Steel. Surf. Coat. Technol. 2003, 162, 154–160. [Google Scholar] [CrossRef]
- Wilson, S.; Hawthorne, H.M.; Yang, Q.; Troczynski, T. Sliding and Abrasive Wear of Composite Sol–Gel Alumina Coated Al Alloys. Surf. Coat. Technol. 2000, 133–134, 389–396. [Google Scholar] [CrossRef]
- Tan, A.L.K.; Soutar, A.M.; Annergren, I.F.; Liu, Y.N. Multilayer Sol–Gel Coatings for Corrosion Protection of Magnesium. Surf. Coat. Technol. 2005, 198, 478–482. [Google Scholar] [CrossRef]
- Li, H.; Liang, K.; Mei, L.; Gu, S.; Wang, S. Oxidation Protection of Mild Steel by Zirconia Sol–Gel Coatings. Mater. Lett. 2001, 51, 320–324. [Google Scholar] [CrossRef]
- Gutierrez-Velasquez, E.I.; Monteiro, S.N.; Colorado, H.A. Characterization of Expanded Polystyrene Waste as Binder and Coating Material. Case Stud. Constr. Mater. 2022, 16, e00804. [Google Scholar] [CrossRef]
- Biondi, L.; Perry, M.; Vlachakis, C.; Wu, Z.; Hamilton, A.; McAlorum, J. Ambient Cured Fly Ash Geopolymer Coatings for Concrete. Materials 2019, 12, 923. [Google Scholar] [CrossRef] [PubMed]
- Mourou, C.; Martín-Morales, M.; Zamorano, M.; Ruiz, D.P. Light Reflectance Characterization of Waste Glass Coating for Tiles. Appl. Sci. 2022, 12, 1537. [Google Scholar] [CrossRef]
- Fu, G.; Wei, L.; Zhang, X.; Cui, Y.; Wang, Y.; Yu, B.; Lv, C.; Ye, S. A MgO–SiO2–Al2O3–ZnO Ceramic-Glass Coating to Improve the Anti-Oxidation of Carbon Steel at High Temperature. ISIJ Int. 2018, 58, 929–935. [Google Scholar] [CrossRef]
- Azarian, N.; Mousavi Khoei, S.M. Characteristics of a Multi-Component MgO-Based Bioceramic Coating Synthesized in-Situ by Plasma Electrolytic Oxidation. J. Magnes. Alloys 2021, 9, 1595–1608. [Google Scholar] [CrossRef]
- Fu, G.; Wei, L.; Shan, X.; Zhang, X.; Ding, J.; Lv, C.; Liu, Y.; Ye, S. Influence of a Cr2O3 Glass Coating on Enhancing the Oxidation Resistance of 20MnSiNb Structural Steel. Surf. Coat. Technol. 2016, 294, 8–14. [Google Scholar] [CrossRef]
- Balinski, K.; Jahns, K.; Schroth, M.; Bursich, S.; Salime, J.; Impey, S.; Chalk, C.; Wübbelmann, J.; Nicholls, J.R.; Krupp, U. Oxidation Behaviour of Synthetic Stainless Steel Interdiffusion Layers. Mater. High Temp. 2018, 35, 89–96. [Google Scholar] [CrossRef]
- Xie, D.; Hong, H.; Duo, S.; Li, Q. Effect of Kerosene Combustion Atmosphere on the Mild Steel Oxide Layer. Sci. Rep. 2022, 12, 379. [Google Scholar] [CrossRef]
- Krishna, V.; Padmapreetha, R.; Chandrasekhar, S.B.; Murugan, K.; Johnson, R. Oxidation Resistant TiO2–SiO2 Coatings on Mild Steel by Sol–Gel. Surf. Coat. Technol. 2019, 378, 125041. [Google Scholar] [CrossRef]
- Han, D.; Zhang, J.; Liu, P.; Wang, S. Effect of Polymorphism of Al2O3 on the Sintering and Microstructure of Transparent MgAl 2O4 Ceramics. Opt. Mater. 2017, 71, 62–65. [Google Scholar] [CrossRef]
- Nakagoshi, Y.; Sato, J.; Morimoto, M.; Suzuki, Y. Near-Zero Volume-Shrinkage in Reactive Sintering of Porous MgTi2O5 with Pseudobrookite-Type Structure. Ceram. Int. 2016, 42, 9139–9144. [Google Scholar] [CrossRef]
- Akbari, S.; Masoudpanah, S.M.; Mirkazemi, S.M.; Aliyan, N. PVA Assisted Coprecipitation Synthesis and Characterization of MgFe2O4 Nanoparticles. Ceram. Int. 2017, 43, 6263–6267. [Google Scholar] [CrossRef]
- Das, S.; Rahman, M.L.; Mondal, P.P.; Mahapatra, P.L.; Saha, D. Screen-Printed MgAl2O4 Semi-Thick Film Based Highly Sensitive and Stable Capacitive Humidity Sensor. Ceram. Int. 2021, 47, 33515–33524. [Google Scholar] [CrossRef]
- Yamashita, T.; Hayes, P. Analysis of XPS Spectra of Fe2+ and Fe3+ Ions in Oxide Materials. Appl. Surf. Sci. 2008, 254, 2441–2449. [Google Scholar] [CrossRef]
- Fan, W.; Wang, H.; Wang, C.; Liu, Z.; Wang, Z.; Liu, S.; Zhu, Y. Oxygen Vacancy Modified α-Fe2O3 Nanorods Provide an Environment-Friendly and Efficient Anti-Corrosion Passive Film into Polymer Coating. Corros. Sci. 2023, 215, 111045. [Google Scholar] [CrossRef]
- Xu, Q.-Q.; Cheng, X.-L.; Zhang, B.-Y.; Zhang, F.; Wang, X.; Li, S.-S.; Zhang, Y.-X. Fe-Doping Induced Surface Fe2+/Fe3+ Cycle and Activated Redox-Inert TiO2 for Enhanced Hg(II) Electrochemical Sensing: An Efficient Strategy to Strengthen the Redox Activity. Anal. Chim. Acta 2022, 1232, 340472. [Google Scholar] [CrossRef]
- Hayashi, S.; Yamanouchi, Y.; Hayashi, K.; Hidaka, Y.; Sato, M. Stress Measurement in the Iron Oxide Scale Formed on Pure Fe during Isothermal Transformation by in Situ High-Temperature X-Ray Diffraction. Corros. Sci. 2021, 187, 109482. [Google Scholar] [CrossRef]
- Bailey, J.M.; Ritchie, I.M. Metal Oxidation: An Electrochemical Perspective. Oxid. Met. 1988, 30, 405–418. [Google Scholar] [CrossRef]
- Robbennolt, S.; Nicolenco, A.; Mercier Fernandez, P.; Auffret, S.; Baltz, V.; Pellicer, E.; Menéndez, E.; Sort, J. Electric Field Control of Magnetism in Iron Oxide Nanoporous Thin Films. ACS Appl. Mater. Interfaces 2019, 11, 37338–37346. [Google Scholar] [CrossRef]
- Cheng, T.-L.; Wen, Y.-H.; Hawk, J.A. Diffuse-Interface Modeling and Multiscale-Relay Simulation of Metal Oxidation Kinetics—With Revisit on Wagner’s Theory. J. Phys. Chem. C 2014, 118, 1269–1284. [Google Scholar] [CrossRef]
- Hao, Y.; Wang, S.; Zhang, K. Effect of Y3+ on the Photoluminescence of MgAl2O4:Cr3+ Nanopowders. Mater. Chem. Phys. 2020, 253, 123323. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.