The Revival of Aztreonam in Combination with Avibactam against Metallo-β-Lactamase-Producing Gram-Negatives: A Systematic Review of In Vitro Studies and Clinical Cases
Abstract
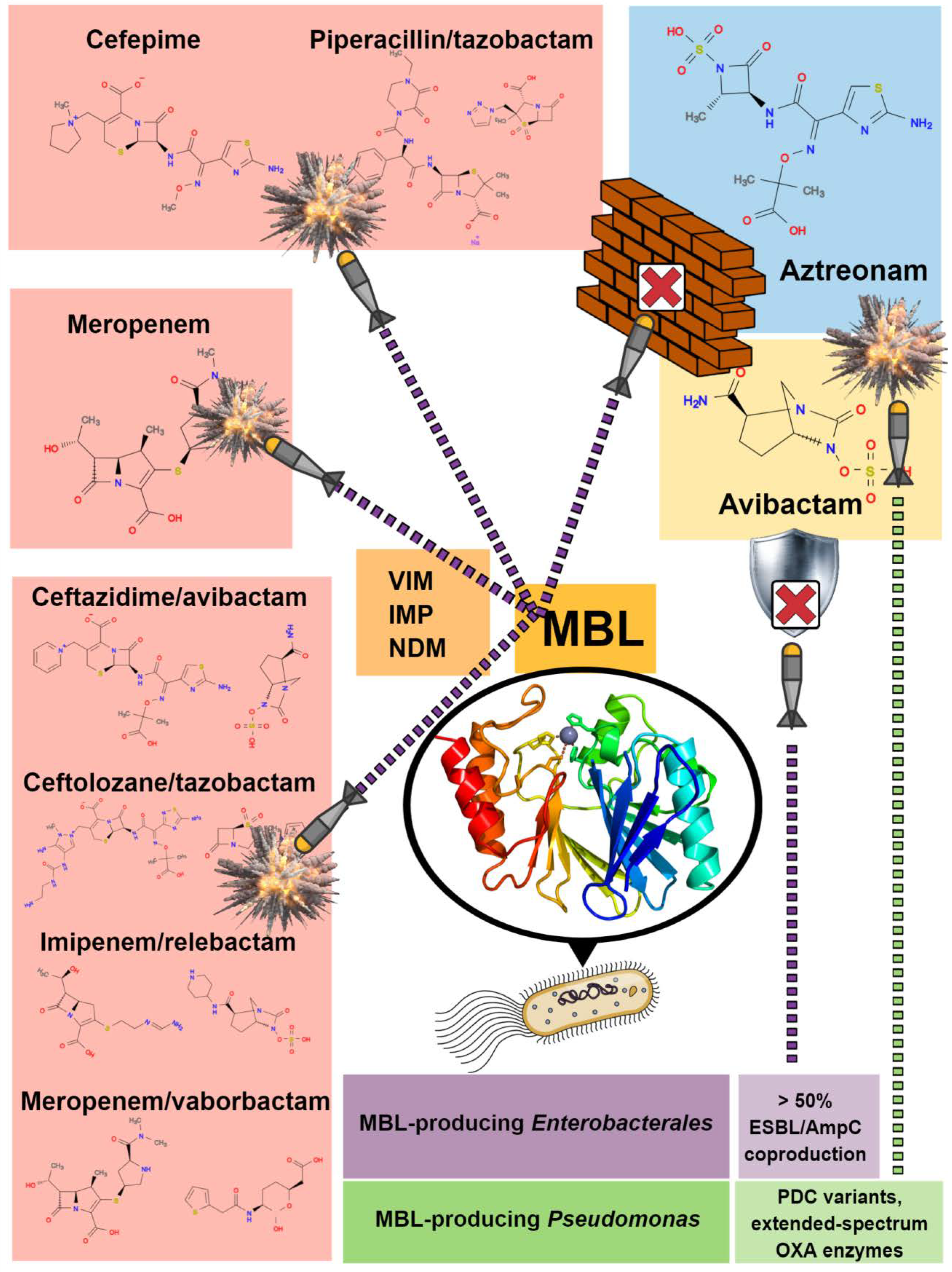
:1. Introduction
2. Results
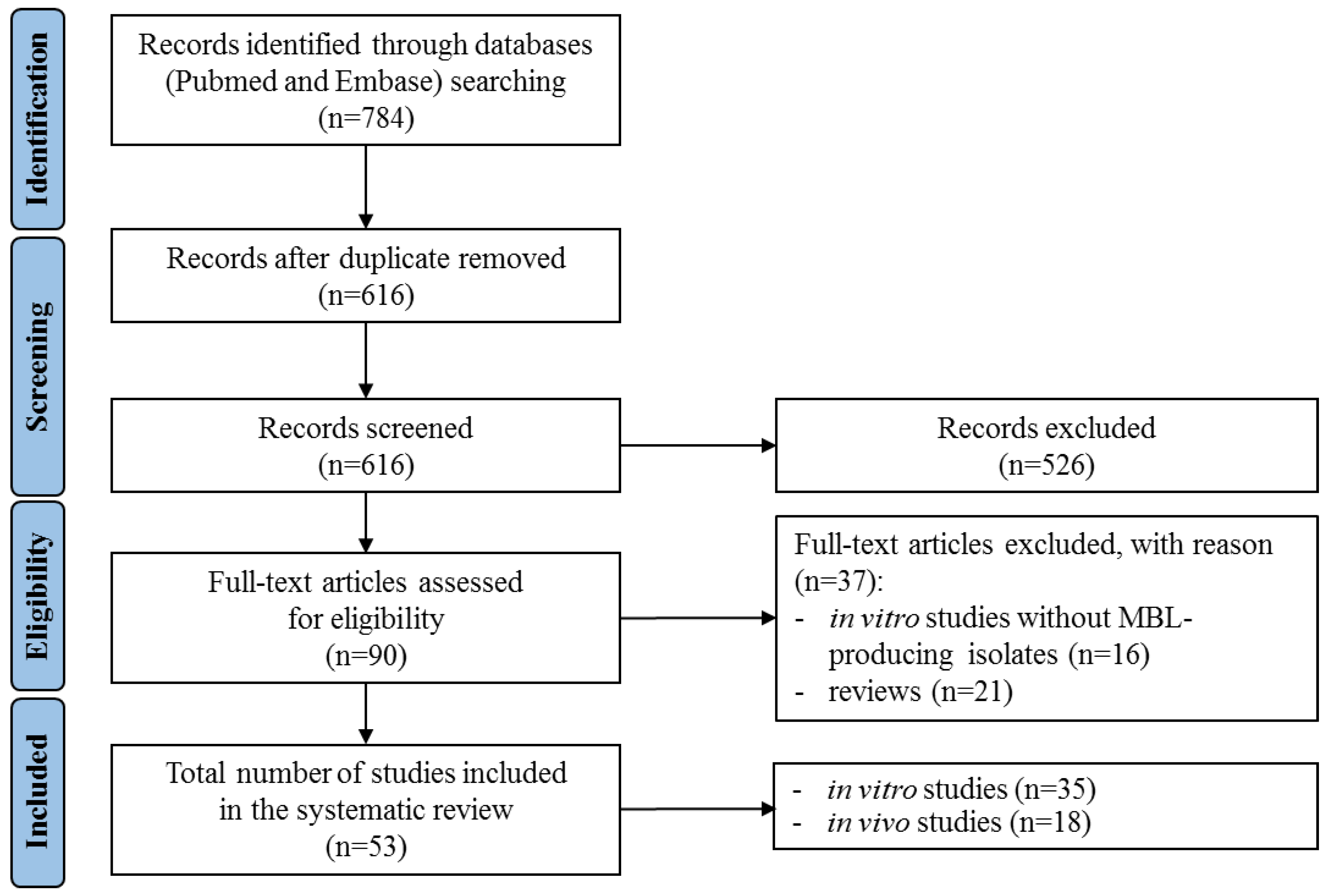
2.1. Literature Search
2.2. General Features and Key Findings
2.2.1. In Vitro Studies
2.2.2. In Vivo Studies
2.3. Quality of Included Studies
2.3.1. In Vitro Studies
2.3.2. In Vivo Studies
3. Discussion
4. Methods
4.1. Protocol and Registration
4.2. Literature Search Strategy
4.3. Study Selection
4.4. Data Extraction
4.5. Strategy for Data Synthesis
4.6. Quality Assessment
4.7. Ethics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tan, X.; Kim, H.S.; Baugh, K.; Huang, Y.; Kadiyala, N.; Wences, M.; Singh, N.; Wenzler, E.; Bulman, Z.P. Therapeutic Options for Metallo-β-Lactamase-Producing Enterobacterales. Infect. Drug Resist. 2021, 14, 125–142. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.U.; Maryam, L.; Zarrilli, R. Structure, genetics and worldwide spread of New Delhi metallo-β-lactamase (NDM): A threat to public health. BMC Microbiol. 2017, 17, 101–112. [Google Scholar] [CrossRef] [Green Version]
- Snyder, B.M.; Montague, B.T.; Anandan, S.; Madabhushi, A.G.; Pragasam, A.K.; Verghese, V.P.; Balaji, V.; Simões, E.A.F. Risk factors and epidemiologic predictors of bloodstream infections with New Delhi metallo-β-lactamase (NDM-1) producing Enterobacteriaceae. Epidemiol. Infect. 2019, 147, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Falcone, M.; Tiseo, G.; Antonelli, A.; Giordano, C.; Di Pilato, V.; Bertolucci, P.; Parisio, E.M.; Leonildi, A.; Aiezza, N.; Baccani, I.; et al. Clinical features and outcomes of bloodstream infections caused by New Delhi metallo-β-lactamase-producing Enterobacterales during a regional outbreak. Open Forum Infect. Dis. 2020, 7, 1–5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spyropoulou, A.; Bartzavali, C.; Vamvakopoulou, S.; Marangos, M.; Anastassiou, E.D.; Spiliopoulou, I.; Christofidou, M. The first NDM metallo-β-lactamase producing Klebsiella pneumoniae isolate in a university hospital of southwestern Greece. J. Chemother. 2016, 28, 350–351. [Google Scholar] [CrossRef] [PubMed]
- Stone, N.R.H.; Woodford, N.; Livermore, D.M.; Howard, J.; Pike, R.; Mushtaq, S.; Perry, C.; Hopkins, S. Breakthrough bacteraemia due to tigecycline-resistant Escherichia coli with New Delhi metallo-β-lactamase (NDM)-1 successfully treated with colistin in a patient with calciphylaxis. J. Antimicrob. Chemother. 2011, 66, 2677–2678. [Google Scholar] [CrossRef] [PubMed]
- Chien, J.M.F.; Koh, T.H.; Chan, K.S.; Chuah, T.H.C.; Tan, T.T. Successful treatment of NDM-1 Klebsiella pneumoniae bacteraemia in a neutropenic patient. Scand. J. Infect. Dis. 2012, 44, 312–314. [Google Scholar] [CrossRef]
- Petersen-Morfin, S.; Bocanegra-Ibarias, P.; Morfin-Otero, R.; Garza-González, E.; Perez-Gomez, H.R.; González-Diaz, E.; Esparza-Ahumada, S.; León-Garnica, G.; Amezcua-Salazar, G.; Rodriguez-Noriega, E. New Delhi metallo-β-lactamase (NDM-1)-producing Klebsiella pneumoniae isolated from a burned patient. Am. J. Case Rep. 2017, 18, 805–809. [Google Scholar] [CrossRef]
- Singh, R.; Kim, A.; Tanudra, M.A.; Harris, J.J.; McLaughlin, R.E.; Patey, S.; O’Donnell, J.P.; Bradford, P.A.; Eakin, A.E. Pharmacokinetics/pharmacodynamics of a β-lactam and β-lactamase inhibitor combination: A novel approach for aztreonam/avibactam. J. Antimicrob. Chemother. 2015, 70, 2618–2626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cornely, O.A.; Cisneros, J.M.; Torre-Cisneros, J.; Rodríguez-Hernández, M.J.; Tallón-Aguilar, L.; Calbo, E.; Horcajada, J.P.; Queckenberg, C.; Zettelmeyer, U.; Arenz, D.; et al. COMBACTE-CARE consortium/REJUVENATE Study Group. Pharmacokinetics and safety of aztreonam/avibactam for the treatment of complicated intra-abdominal infections in hospitalized adults: Results from the REJUVENATE study. J. Antimicrob. Chemother. 2020, 75, 618–627. [Google Scholar] [CrossRef]
- Tsuji, B.T.; Pogue, J.M.; Zavascki, A.P.; Paul, M.; Daikos, G.L.; Forrest, A.; Giacobbe, D.R.; Viscoli, C.; Giamarellou, H.; Karaiskos, I.; et al. International Consensus Guidelines for the Optimal Use of the Polymyxins: Endorsed by the American College of Clinical Pharmacy (ACCP), European Society of Clinical Microbiology and Infectious Diseases (ESCMID), Infectious Diseases Society of America (IDSA), International Society for Anti-infective Pharmacology (ISAP), Society of Critical Care Medicine (SCCM), and Society of Infectious Diseases Pharmacists (SIDP). Pharmacotherapy 2019, 39, 10–39. [Google Scholar] [CrossRef]
- Neu, H.C. Aztreonam activity, pharmacology, and clinical uses. Am. J. Med. 1990, 88, S2–S6, discussion S38–S42. [Google Scholar] [CrossRef]
- Ramsey, C.; MacGowan, A.P. A review of the pharmacokinetics and pharmacodynamics of aztreonam. J. Antimicrob. Chemother. 2016, 71, 2704–2712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamma, P.D.; Aitken, S.L.; Bonomo, R.A.; Mathers, A.J.; van Duin, D.; Clancy, C.J. Infectious Diseases Society of America Guidance on the Treatment of Extended-Spectrum β-lactamase Producing Enterobacterales (ESBL-E), Carbapenem-Resistant Enterobacterales (CRE), and Pseudomonas aeruginosa with Difficult-to-Treat Resistance (DTR-P. aeruginosa). Clin. Infect. Dis. 2021, 72, 1109–1116. [Google Scholar] [CrossRef] [PubMed]
- Livermore, D.M.; Mushtaq, S.; Warner, M.; Zhang, J.; Maharjan, S.; Doumith, M.; Woodford, N. Activities of NXL104 combinations with ceftazidime and aztreonam against carbapenemase-Producing Enterobacteriaceae. Antimicrob. Agents Chemother. 2011, 55, 390–394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Zhang, F.; Zhao, C.; Wang, Z.; Nichols, W.W.; Testa, R.; Li, H.; Chen, H.; He, W.; Wang, Q.; et al. In vitro activities of ceftazidime-avibactam and aztreonam-avibactam against 372 Gram-negative bacilli collected in 2011 and 2012 from 11 teaching hospitals in China. Antimicrob. Agents Chemother. 2014, 58, 1774–1778. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alm, R.A.; Johnstone, M.R.; Lahiri, S.D. Characterization of Escherichia coli NDM isolates with decreased susceptibility to aztreonam/avibactam: Role of a novel insertion in PBP3. J. Antimicrob. Chemother. 2015, 70, 1420–1428. [Google Scholar] [CrossRef] [Green Version]
- Kazmierczak, K.M.; Rabine, S.; Hackel, M.; McLaughlin, R.E.; Biedenbach, D.J.; Bouchillon, S.K.; Sahm, D.F.; Bradford, P.A. Multiyear, Multinational Survey of the Incidence and Global Distribution of Metallo-β-Lactamase-Producing Enterobacteriaceae and Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2015, 60, 1067–1078. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Estabrook, M.; Jacoby, G.A.; Nichols, W.W.; Testa, R.T.; Bush, K. In vitro susceptibility of characterized β-lactamase-producing strains tested with avibactam combinations. Antimicrob. Agents Chemother. 2015, 59, 1789–1793. [Google Scholar] [CrossRef] [Green Version]
- Vasoo, S.; Cunningham, S.A.; Cole, N.C.; Kohner, P.C.; Menon, S.R.; Krause, K.M.; Harris, K.A.; De, P.P.; Koh, T.H.; Patel, R. In vitro Activities of Ceftazidime-Avibactam, Aztreonam-Avibactam, and a Panel of Older and Contemporary Antimicrobial Agents against Carbapenemase-Producing Gram-Negative Bacilli. Antimicrob. Agents Chemother. 2015, 59, 7842–7846. [Google Scholar] [CrossRef] [Green Version]
- Pillar, C.M.; Stoneburner, A.; Shinabarger, D.L.; Krause, K.M.; Nichols, W.W. The postantibiotic effect and post-β-lactamase-inhibitor effect of ceftazidime, ceftaroline and aztreonam in combination with avibactam against target Gram-negative bacteria. Lett. Appl. Microbiol. 2016, 63, 96–102. [Google Scholar] [CrossRef] [Green Version]
- Thomson, G.K.; Snyder, J.W.; McElheny, C.L.; Thomson, K.S.; Doi, Y. Coproduction of KPC-18 and VIM-1 Carbapenemases by Enterobacter cloacae: Implications for Newer β-Lactam-β-Lactamase Inhibitor Combinations. J. Clin. Microbiol. 2016, 54, 791–794. [Google Scholar] [CrossRef] [Green Version]
- Karlowsky, J.A.; Kazmierczak, K.M.; de Jonge, B.L.M.; Hackel, M.A.; Sahm, D.F.; Bradford, P.A. In vitro Activity of Aztreonam-Avibactam against Enterobacteriaceae and Pseudomonas aeruginosa Isolated by Clinical Laboratories in 40 Countries from 2012 to 2015. Antimicrob. Agents Chemother. 2017, 61, e00472-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marshall, S.; Hujer, A.M.; Rojas, L.J.; Papp-Wallace, K.M.; Humphries, R.M.; Spellberg, B.; Hujer, K.M.; Marshall, E.K.; Rudin, S.D.; Perez, F.; et al. Can Ceftazidime-Avibactam and Aztreonam Overcome β-Lactam Resistance Conferred by Metallo-β-Lactamases in Enterobacteriaceae? Antimicrob. Agents Chemother. 2017, 61, e02243-16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wenzler, E.; Deraedt, M.F.; Harrington, A.T.; Danizger, L.H. Synergistic activity of ceftazidime-avibactam and aztreonam against serine and metallo-β-lactamase-producing gram-negative pathogens. Diagn. Microbiol. Infect. Dis. 2017, 88, 352–354. [Google Scholar] [CrossRef]
- Zhang, Y.; Kashikar, A.; Brown, C.A.; Denys, G.; Bush, K. Unusual Escherichia coli PBP 3 Insertion Sequence Identified from a Collection of Carbapenem-Resistant Enterobacteriaceae Tested In vitro with a Combination of Ceftazidime-, Ceftaroline-, or Aztreonam-Avibactam. Antimicrob. Agents Chemother. 2017, 61, e00389-17. [Google Scholar] [CrossRef] [Green Version]
- Avery, L.M.; Nicolau, D.P. Assessing the in vitro activity of ceftazidime/avibactam and aztreonam among carbapenemase-producing Enterobacteriaceae: Defining the zone of hope. Int. J. Antimicrob. Agents 2018, 52, 688–691. [Google Scholar] [CrossRef] [PubMed]
- Jayol, A.; Nordmann, P.; Poirel, L.; Dubois, V. Ceftazidime/avibactam alone or in combination with aztreonam against colistin-resistant and carbapenemase-producing Klebsiella pneumoniae. J. Antimicrob. Chemother. 2018, 73, 542–544. [Google Scholar] [CrossRef]
- Sader, H.S.; Mendes, R.E.; Pfaller, M.; Shortridge, D.; Flamm, R.K.; Castanheira, M. Antimicrobial activities of aztreonam-avibactam and comparator agents against contemporary (2016) clinical Enterobacteriaceae isolates. Antimicrob. Agents Chemother. 2017, 62, e01856-17. [Google Scholar] [CrossRef] [Green Version]
- Biagi, M.; Wu, T.; Lee, M.; Butler, D.; Wenzler, E. Searching for the optimal treatment for metallo- and serine-β-lactamase Producing Enterobacteriaceae: Aztreonam in combination with ceftazidime-avibactam or meropenem-vaborbactam. Antimicrob. Agents Chemother. 2019, 63, e01426-19. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Xiao, X.; Wang, X.; Xia, M.; Liu, S. In vitro antimicrobial susceptibility differences between carbapenem-resistant KPC-2-producing and NDM-1-producing Klebsiella pneumoniae in a teaching hospital in northeast China. Microb. Drug Resist. 2020, 26, 94–99. [Google Scholar] [CrossRef]
- Mikhail, S.; Singh, N.B.; Kebriaei, R.; Rice, S.A.; Stamper, K.C.; Castanheira, M.; Rybak, M.J. Evaluation of the Synergy of Ceftazidime-Avibactam in Combination with Meropenem, Amikacin, Aztreonam, Colistin, or Fosfomycin against Well-Characterized Multidrug-Resistant Klebsiella pneumoniae and Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2019, 63, e00779-19. [Google Scholar] [CrossRef] [Green Version]
- Pragasam, A.K.; Veeraraghavan, B.; Shankar, B.A.; Bakthavatchalam, Y.D.; Mathuram, A.; George, B.; Chacko, B.; Korula, P.; Anandan, S. Will ceftazidime/avibactam plus aztreonam be effective for NDM and OXA-48-Like producing organisms: Lessons learnt from In vitro study. Indian J. Med. Microbiol. 2019, 37, 34–41. [Google Scholar] [CrossRef]
- Zou, H.; Xiong, S.J.; Lin, Q.X.; Wu, M.L.; Niu, S.Q.; Huang, S.F. CP-CRE/non-CP-CRE Stratification and CRE Resistance Mechanism Determination Help in Better Managing CRE Bacteremia Using Ceftazidime-Avibactam and Aztreonam-Avibactam. Infect. Drug Resist. 2019, 12, 3017–3027. [Google Scholar] [CrossRef] [Green Version]
- Esposito, S.; Stone, G.G.; Papaparaskevas, J. In vitro activity of aztreonam/avibactam against a global collection of Klebsiella pneumoniae collected from defined culture sources in 2016 and 2017. J. Glob. Antimicrob. Resist. 2021, 24, 14–22. [Google Scholar] [CrossRef]
- Kilic, U.; Koroglu, M.; Olmez, M.; Altindis, M. Investigation of the In vitro Effectiveness of Aztreonam/Avibactam, Colistin/Apramycin, and Meropenem/Apramycin Combinations Against Carbapenemase-Producing, Extensively Drug-Resistant Klebsiella pneumoniae Strains. Microb. Drug Resist. 2020, 26, 1291–1297. [Google Scholar] [CrossRef]
- Kim, T.; Lee, S.C.; Bae, M.; Sung, H.; Kim, M.N.; Jung, J.; Kim, M.J.; Kim, S.H.; Lee, S.O.; Choi, S.H.; et al. In vitro Activities and Inoculum Effects of Ceftazidime-Avibactam and Aztreonam-Avibactam against Carbapenem-Resistant Enterobacterales Isolates from South Korea. Antibiotics 2020, 9, 912. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Abbey, T.; Biagi, M.; Wenzler, E. Activity of aztreonam in combination with ceftazidime-avibactam against serine- and metallo-β-lactamase-producing Pseudomonas aeruginosa. Diagn. Microbiol. Infect. Dis. 2021, 99, 115227. [Google Scholar] [CrossRef] [PubMed]
- Niu, S.; Wei, J.; Zou, C.; Chavda, K.D.; Lv, J.; Zhang, H.; Du, H.; Tang, Y.W.; Pitout, J.D.D.; Bonomo, R.A.; et al. In vitro selection of aztreonam/avibactam resistance in dual-carbapenemase-producing Klebsiella pneumoniae. J. Antimicrob. Chemother. 2020, 75, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Periasamy, H.; Joshi, P.; Palwe, S.; Shrivastava, R.; Bhagwat, S.; Patel, M. High prevalence of Escherichia coli clinical isolates in India harbouring four amino acid inserts in PBP3 adversely impacting activity of aztreonam/avibactam. J. Antimicrob. Chemother. 2020, 75, 1650–1651. [Google Scholar] [CrossRef]
- Wei, J.; Zou, C.; Wang, D.; Huang, A.; Niu, S. Genetic diversity and in vitro activity of ceftazidime/avibactam and aztreonam/avibactam against imipenem-resistant Enterobacteriaceae isolates in Southwest China: A single-centre study. J. Glob. Antimicrob. Resist. 2020, 22, 448–451. [Google Scholar] [CrossRef]
- Yang, T.Y.; Hsieh, Y.J.; Kao, L.T.; Liu, G.H.; Lian, S.H.; Wang, L.C.; Lin, I.L.; Lin, Y.T.; Wang, S.F.; Tseng, S.P.; et al. In vitro and In vivo Evaluations of β-Lactam/β-Lactamase Mono- and Combined Therapies against Carbapenem-Nonsusceptible Enterobacteriaceae in Taiwan. Microorganisms 2020, 8, 1981. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Zhu, Z.; Jia, W.; Qu, F.; Huang, B.; Shan, B.; Yu, H.; Tang, Y.; Chen, L.; Du, H. In vitro activity of aztreonam-avibactam against metallo-β-lactamase-producing Enterobacteriaceae-A multicenter study in China. Int. J. Infect. Dis. 2020, 97, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Zou, C.; Wei, J.; Shan, B.; Chen, X.; Wang, D.; Niu, S. In vitro Activity of Ceftazidime-Avibactam and Aztreonam-Avibactam Against Carbapenem-resistant Enterobacteriaceae Isolates Collected from Three Secondary Hospitals in Southwest China Between 2018 and 2019. Infect. Drug Resist. 2020, 13, 3563–3568. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, A.; Ransom, E.M.; Machado, M.J.; Boyd, S.; Reese, N.; Anderson, K.; Lonsway, D.; Elkins, C.A.; Rasheed, J.K.; Patel, J.B.; et al. Assessing the in vitro impact of ceftazidime on aztreonam/avibactam susceptibility testing for highly resistant MBL-producing Enterobacterales. J. Antimicrob. Chemother. 2021, 76, 979–983. [Google Scholar] [CrossRef]
- Chang, Y.; Zhang, D.; Niu, S.; Chen, Q.; Lin, Q.; Zhang, X. MBLs, Rather Than Efflux Pumps, Led to Carbapenem Resistance in Fosfomycin and Aztreonam/Avibactam Resistant Elizabethkingia anopheles. Infect. Drug Resist. 2021, 14, 315–327. [Google Scholar] [CrossRef]
- Falcone, M.; Daikos, G.L.; Tiseo, G.; Bassoulis, D.; Giordano, C.; Galfo, V.; Leonildi, A.; Tagliaferri, E.; Barnini, S.; Sani, S.; et al. Efficacy of Ceftazidime-avibactam Plus Aztreonam in Patients with Bloodstream Infections Caused by Metallo-β-lactamase-Producing Enterobacterales. Clin. Infect Dis. 2021, 72, 1871–1878. [Google Scholar] [CrossRef]
- Lin, Q.; Zou, H.; Chen, X.; Ma, D.; Yu, H.; Niu, S.; Huang, S. Avibactam potentiated the activity of both ceftazidime and aztreonam against S. maltophilia clinical isolates in vitro. BMC Microbiol. 2021, 21, 60. [Google Scholar] [CrossRef]
- Maraki, S.; Mavromanolaki, V.E.; Moraitis, P.; Stafylaki, D.; Kasimati, A.; Magkafouraki, E.; Scoulica, E. Ceftazidime-avibactam, meropenen-vaborbactam, and imipenem-relebactam in combination with aztreonam against multidrug-resistant, metallo-β-lactamase-producing Klebsiella pneumoniae. Eur. J. Clin. Microbiol. Infect. Dis. 2021, 40, 1755–1759. [Google Scholar] [CrossRef]
- Mojica, M.F.; Ouellette, C.P.; Leber, A.; Becknell, M.B.; Ardura, M.I.; Perez, F.; Shimamura, M.; Bonomo, R.A.; Aitken, S.L.; Shelburne, S.A. Successful Treatment of Bloodstream Infection Due to Metallo-β-Lactamase-Producing Stenotrophomonas maltophilia in a Renal Transplant Patient. Antimicrob. Agents Chemother. 2016, 60, 5130–5134. [Google Scholar] [CrossRef] [Green Version]
- Davido, B.; Fellous, L.; Lawrence, C.; Maxime, V.; Rottman, M.; Dinh, A. Ceftazidime-Avibactam and Aztreonam, an Interesting Strategy to Overcome β-Lactam Resistance Conferred by Metallo-β-Lactamases in Enterobacteriaceae and Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2017, 24, e01008–e01017. [Google Scholar] [CrossRef] [Green Version]
- Mittal, J.; Szymczak, W.A.; Guo, Y.; Levi, M.H.; Chen, L.; Kreiswirth, B.N.; Riska, P.F.; Nori, P. Two for the price of one: Emerging carbapenemases in a returning traveller to New York City. BMJ Case Rep. 2018, 2018. [Google Scholar] [CrossRef]
- Shaw, E.; Rombauts, A.; Tubau, F.; Padullés, A.; Càmara, J.; Lozano, T.; Cobo-Sacristán, S.; Sabe, N.; Grau, I.; Rigo-Bonnin, R.; et al. Clinical outcomes after combination treatment with ceftazidime/avibactam and aztreonam for NDM-1/OXA-48/CTX-M-15-producing Klebsiella pneumoniae infection. J. Antimicrob. Chemother. 2018, 73, 1104–1106. [Google Scholar] [CrossRef]
- Emeraud, C.; Escaut, L.; Boucly, A.; Fortineau, N.; Bonnin, R.A.; Nass, T.; Dortet, L. Aztreonam plus Clavulanate, Tazobactam, or Avibactam for Treatment of Infections Caused by Metallo-β-Lactamase-Producing Gram-Negative Bacteria. Antimicrob. Agents Chemother. 2019, 63, e00010–e00019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hobson, C.A.; Bonacorsi, S.; Fahd, M.; Baruchel, A.; Cointe, A.; Poey, N.; Jacquier, H.; Doit, C.; Monjault, A.; Tenaillon, O.; et al. Successful Treatment of Bacteremia Due to NDM-1-Producing Morganella morganii with Aztreonam and Ceftazidime-Avibactam Combination in a Pediatric Patient with Hematologic Malignancy. Antimicrob. Agents Chemother. 2019, 63, e02463-18. [Google Scholar] [CrossRef] [Green Version]
- Shah, P.J.; Tran, T.; Emelogu, F.; Tariq, F. Aztreonam, Ceftazidime/Avibactam, and Colistin Combination for the Management of Carbapenemase-Producing Klebsiella Pneumoniae Bacteremia: A Case Report. J. Pharm. Pract. 2021, 34, 653–657. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.; Newman, G.S.; Jain, R.; Bryan, A.; Berger, H.; Montenovo, M.; Bakthavatsalam, R.; Kling, C.E.; Sibulesky, L.; Shalhub, S.; et al. Transplant tourism complicated by life-threatening New Delhi metallo-β-lactamase-1 infection. Am. J. Transpl. 2019, 19, 1224–1228. [Google Scholar] [CrossRef] [PubMed]
- Benchetrit, L.; Mathy, V.; Armand-Lefevre, L.; Bouadma, L.; Timsit, J.F. Successful treatment of septic shock due to NDM-1-producing Klebsiella pneumoniae using ceftazidime/avibactam combined with aztreonam in solid organ transplant recipients: Report of two cases. Int. J. Antimicrob. Agents 2020, 55, 105842. [Google Scholar] [CrossRef]
- Alghoribi, M.F.; Alqurashi, M.; Okdah, L.; Alalwan, B.; AlHebaishi, Y.S.; Almalki, A.; Alzayer, M.A.; Alswaji, A.A.; Doumith, M.; Barry, M. Successful treatment of infective endocarditis due to pandrug-resistant Klebsiella pneumoniae with ceftazidime-avibactam and aztreonam. Sci. Rep. 2021, 11, 9684. [Google Scholar] [CrossRef] [PubMed]
- Bocanegra-Ibarias, P.; Camacho-Ortiz, A.; Garza-González, E.; Flores-Treviño, S.; Kim, H.; Perez-Alba, E. Aztreonam plus ceftazidime-avibactam as treatment of NDM-1-producing Klebsiella pneumoniae bacteraemia in a neutropenic patient: Last resort therapy? J. Glob. Antimicrob. Resist. 2020, 23, 417–419. [Google Scholar] [CrossRef]
- Cairns, K.A.; Hall, V.; Martin, G.E.; Griffin, D.W.J.; Stewart, J.D.; Khan, S.F.; Abbott, I.J.; Meher-Homji, Z.; Morrissey, C.O.; Sia, C.; et al. Treatment of invasive IMP-4 Enterobacter cloacae infection in transplant recipients using ceftazidime/avibactam with aztreonam: A case series and literature review. Transpl. Infect. Dis. 2021, 23, e13510. [Google Scholar] [CrossRef]
- Cowart, M.C.; Ferguson, C.L. Optimization of Aztreonam in Combination with Ceftazidime/Avibactam in a Cystic Fibrosis Patient with Chronic Stenotrophomonas maltophilia Pneumonia Using Therapeutic Drug Monitoring: A Case Study. Ther. Drug Monit. 2021, 43, 146–149. [Google Scholar] [CrossRef]
- Perrotta, F.; Perrini, M.P. Successful Treatment of Klebsiella pneumoniae NDM Sepsis and Intestinal Decolonization with Ceftazidime/Avibactam Plus Aztreonam Combination in a Patient with TTP Complicated by SARSCoV-2 Nosocomial Infection. Medicina 2021, 57, 424. [Google Scholar] [CrossRef]
- Sieswerda, E.; van den Brand, M.; van den Berg, R.B.; Sträter, J.; Schouls, L.; van Dijk, K.; Budding, A.E. Successful rescue treatment of sepsis due to a pandrug-resistant, NDM-producing Klebsiella pneumoniae using aztreonam powder for nebulizer solution as intravenous therapy in combination with ceftazidime/avibactam. J. Antimicrob. Chemother. 2020, 75, 773–775. [Google Scholar] [CrossRef] [Green Version]
- Yasmin, M.; Fouts, D.E.; Jacobs, M.R.; Haydar, H.; Marshall, S.H.; White, R.; D’Souza, R.; Lodise, T.P.; Rhoads, D.D.; Hujer, A.M.; et al. Monitoring Ceftazidime-Avibactam and Aztreonam Concentrations in the Treatment of a Bloodstream Infection Caused by a Multidrug-Resistant Enterobacter sp. Carrying Both Klebsiella pneumoniae Carbapenemase-4 and New Delhi Metallo-β-Lactamase-1. Clin. Infect. Dis. 2020, 71, 1095–1098. [Google Scholar] [CrossRef]
- Walsh, T.R.; Toleman, M.A.; Poirel, L.; Nordmann, P. Metallo-β-lactamases: The quiet before the storm? Clin. Microbiol. Rev. 2005, 18, 306–325. [Google Scholar] [CrossRef] [Green Version]
- Karakonstantis, S.; Kritsotakis, E.I.; Gikas, A. Treatment options for K. pneumoniae, P. aeruginosa and A. baumannii co-resistant to carbapenems, aminoglycosides, polymyxins and tigecycline: An approach based on the mechanisms of resistance to carbapenems. Infection 2020, 48, 835–851. [Google Scholar] [CrossRef]
- Bassetti, M.; Di Pilato, V.; Giani, T.; Rossolini, G.M.; Marchese, A.; Giacobbe, D.R.; Vena, A. Treatment of severe infections due to metallo-β-lactamases-producing Gram-negative bacteria. Future Microbiol. 2020, 15, 1489–1505. [Google Scholar] [CrossRef]
- Tsivkovski, R.; Totrov, M.; Lomovskaya, O. Biochemical Characterization of QPX7728, a New Ultrabroad-Spectrum β-Lactamase Inhibitor of Serine and Metallo-β-Lactamases. Antimicrob. Agents Chemother. 2020, 64, e00130-20. [Google Scholar] [CrossRef] [Green Version]
- Barnes, M.D.; Taracila, M.A.; Rutter, J.D.; Bethel, C.R.; Galdadas, I.; Hujer, A.M.; Caselli, E.; Prati, F.; Dekker, J.P.; Papp-Wallace, K.M.; et al. Deciphering the evolution of cephalosporin resistance to ceftolozane-tazobactam in Pseudomonas aeruginosa. MBio 2018, 9, e02085-18. [Google Scholar] [CrossRef] [Green Version]
- Vázquez-Ucha, J.C.; Arca-Suárez, J.; Bou, G.; Beceiro, A. New Carbapenemase inhibitors: Clearing the way for the β-lactams. Int. J. Mol. Sci. 2020, 21, 9308. [Google Scholar] [CrossRef]
- Arca-Suárez, J.; Fraile-Ribot, P.; Vázquez-Ucha, J.C.; Cabot, G.; Martínez-Guitián, M.; Lence, E.; González-Bello, C.; Beceiro, A.; Rodríguez-Iglesias, M.; Galán-Sánchez, F.; et al. Challenging Antimicrobial Susceptibility and Evolution of Resistance (OXA-681) during Treatment of a Long-Term Nosocomial Infection Caused by a Pseudomonas aeruginosa ST175 Clone. Antimicrob. Agents Chemother. 2019, 63, e01110-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arca-Suárez, J.; Vázquez-Ucha, J.C. Molecular and biochemical insights into the in vivo evolution of AmpC-mediated resistance to ceftolozane/tazobactam during treatment of an MDR Pseudomonas aeruginosa infection. J. Antimicrob. Chemother. 2020, 75, 3209–3217. [Google Scholar] [CrossRef]
- Fraile-Ribot, P.A.; Mulet, X.; Cabot, G.; Del Barrio-Tofiño, E.; Juan, C.; Pérez, J.L.; Oliver, A. In vivo Emergence of Resistance to Novel Cephalosporin-β-Lactamase Inhibitor Combinations through the Duplication of Amino Acid D149 from OXA-2 β-Lactamase (OXA-539) in Sequence Type 235 Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2017, 61, e01117-17. [Google Scholar] [CrossRef] [Green Version]
- Antunes, N.T.; Fisher, J.F. Acquired Class D β-Lactamases. Antibiotics (Basel) 2014, 3, 398–434. [Google Scholar] [CrossRef] [PubMed]
- Hao, M.; Ma, W.; Dong, X.; Li, X.; Cheng, F.; Wang, X. Comparative genome analysis of multidrug-resistant Pseudomonas aeruginosa JNQH-PA57, a clinically isolated mucoid strain with comprehensive carbapenem resistance mechanisms. BMC Microbiol. 2021, 21, 133. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Feng, Y.; McNally, A.; Zong, Z. Struggle to survive: The choir of target alteration, hydrolyzing enzyme, and plasmid expression as a novel aztreonam-avibactam resistance mechanism. MSystems 2020, 5, e00821-20. [Google Scholar] [CrossRef] [PubMed]
- Lodise, T.P.; Smith, N.M.; O’Donnell, N.; Eakin, A.E.; Holden, P.N.; Boissonneault, K.R.; Zhou, J.; Tao, X.; Bulitta, J.B.; Fowler, V.G.; et al. Determining the optimal dosing of a novel combination regimen of ceftazidime/avibactam with aztreonam against NDM-1-producing Enterobacteriaceae using a hollow-fibre infection model. J. Antimicrob. Chemother. 2020, 75, 2622–2632. [Google Scholar] [CrossRef]
- Falcone, M.; Menichetti, F.; Cattaneo, D.; Tiseo, G.; Baldelli, S.; Galfo, V.; Leonildi, A.; Tagliaferri, E.; Di Paolo, A.; Pai, M.P. Pragmatic options for dose optimization of ceftazidime/avibactam with aztreonam in complex patients. J. Antimicrob. Chemother. 2021, 76, 1025–1031. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, 71. [Google Scholar] [CrossRef]
- Dekkers, O.M.; Egger, M.; Altman, D.G.; Vandenbroucke, J.P. Distinguishing case series from cohort studies. Ann. Intern. Med. 2012, 156, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, B.R.; Juni, P. Systematic reviews and meta-analyses of randomized trials: Principles and pitfalls. Eur. Heart J. 2014, 35, 3336–3345. [Google Scholar] [CrossRef] [PubMed]
- Murad, M.H.; Sultan, S.; Haffar, S.; Bazerbachi, F. Methodological quality and synthesis of case series and case reports. BMJ Evid. Based Med. 2018, 23, 60–63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Author and Publication Year (ref) | Region/Country or Type of Collection, Collection Period | MBL Isolates (n.) | MBL Determinants | ATM MIC (or MIC Range) (mg/L) | No. (%) of Isolates with MIC ≤4 mg/L for ATM in Combination with AVI | MIC range (mg/L) MIC50 (mg/L) MIC90 (mg/L) for ATM in Combination with AVI | Methods Used to Evaluate Interactions | Other Resistance Determinants | Antibiotic Combination | Notes |
|---|---|---|---|---|---|---|---|---|---|---|
| Livermore, 2011 [15] | NA | Enterobacterales (35) | NDM-1 (17)IMP-type (13)VIM-type (5) | 0.06- >256 0.12- >256 0.25- >256 | 35 (100%) | All strains: ≤0.03–4; MIC50: 0.25; MIC90: 2; NDM: ≤0.03–4; IMP: 0.06–4; VIM: 0.12–0.5 | CB | NA | ATM+AVI | - |
| Wang, 2014 [16] | China 2011–2012 | Enterobacterales (14) | IMP-4 (10) NDM-1 (3) IMP-8 (1) | ≤0.064- >128 MIC50: 128 MIC90: >128 (4 IMP-4-producing isolates were susceptible) | 14 (100%) | ≤0.064–0.5 MIC50: 0.125 MIC90: 0.5 | CB | Most isolates co-produced various resistance determinants | ATM+AVI | - |
Alm, 2015 [17] | USA, Philippines and India; collection period NA | E. coli (15) K. pneumoniae (12) E. cloacae (4) | NDM-type (15) NDM-type (12) NDM-type (4) | 8- >512 32–512 256–512 | 13/15 (86.7%) 12/12 (100%) 4/4 (100%) | 0.125–16 0.03–0.25 0.25–1 | CB | All isolates co-produced a CMY-type enzyme; 13/15 isolates also produced ESBL determinants (mostly CTX-M-15) | ATM+AVI | Fourteen E. coli isolates presented a PBP3 insertion (MIC values of ATM in combination ranged from 4 to 16 mg/L. The last one E. coli (without insertion in PBP3) presented an ATM MIC value of 0.125 mg/L |
| Kazmierczak, 2015 [18] | Multiple sites around the world (40 countries) 2012–2014 | Enterobacterales (163) P. aeruginosa (308) | NDM-type (72) VIM-type (64) IMP-type (27) VIM-type (270) IMP-type (35) NDM-type (3) | NDM ≤0.015- >128 MIC50: 128 MIC90: >128 VIM ≤0.015- >128 MIC50: 8 MIC90: 128 IMP 0.06- >128 MIC50: 64 MIC90: >128 VIM 0.5- >128 MIC50: 16 MIC90: 64 IMP 0.5- >128 MIC50: 16 MIC90: 64 NDM >128 | NA (cumulative data only) | NDM ≤0.015- 8 MIC50: 0.12 MIC90: 0.5 VIM ≤0.015–2 MIC50: 0.12 MIC90: 1IMP 0.03–4 MIC50: 0.25 MIC90: 1 VIM 0.25- >128 MIC50: 16 MIC90: 32 IMP 2–128 MIC50: 32 MIC90: 64NDM 16- >128 (MIC range only) | CB | Most isolates also produced various resistance determinants | ATM+AVI | Mostly synergistic (cumulative data only, specific MIC values NA) Mostly not synergistic (cumulative data only, specific MIC values NA) |
| Li, 2015 [19] | Strains from not specified collection; collection period NA | E. coli (1) E. cloacae (1) | VIM-1 (2) | >64 | 0 (0%) 1 (100%) | 8 0.12 | CB | Both isolates co-produced TEM-1; E. coli co-produced KPC-3 variant | ATM+AVI | - |
| Vasoo, 2015 [20] | USA, Singapore and 1 strain from NCTC UK reference isolate NA | Enterobacterales (43) P. aeruginosa (4) | NDM-type (32) IMP-type (11) VIM-type (4) | 0.12 to >512 ≤0.06 to >512 8 | 30 (93.8%) 11 (100%) 1 (25%) | NDM: ≤0.06 to 16; MIC50: 0.12;MIC90: 1; IMP: ≤0.06 to 1; MIC50: 0.25; MIC90: 1; VIM: 4 to 8; MIC50: 8; MIC90: 8 | AD | NA | ATM+AVI | Two NDM-producing E. coli with ATM in combinations with AVI with MIC values at 8 (NDM-1) and 16 (NDM-7) mg/L |
| Pillar, 2016 [21] | NA | E. coli (1) | NDM-1 | 16 | 1 (100%) | 0.12 | CB | NA | ATM+AVI | - |
| Thomson, 2016 [22] | Kentucky (USA); collection period NA | E. cloacae (1) | VIM-1 | >64 | 1 (100%) | 0.5 | CB | The isolate co-produced a KPC-18 variant | ATM+AVI | Synergistic interaction |
| Karlowsky, 2017 [23] | Worldwide (40 countries) 2012–2015 | Enterobacterales (267) P. aeruginosa (452) | NDM-type (142) VIM-type (96) IMP-type (29) NA | ≤0.015- >128 MIC50: 64 MIC90: >128 0.25- >128 MIC50: 16 MIC90: 64 | 265 (99.3%) 47 (10.4%) | ≤0.015–8 MIC50: 0.12 MIC90: 1 0.25- >128 MIC50: 16 MIC90: 32 | CB | Most isolates also produced various resistance determinants | ATM+AVI | - |
| Marshall, 2017 [24] | Not specified collection; Collection period NA | Enterobacterales (21) | NDM-1 (20) IMP-type (1) | 0.0625->512 (17/21 isolates were resistant) | 0 (0%) | 8–64 | AD with ATM fixed at 8, 16, 32 and 64 | Most isolates also produced various resistance determinants | ATM+CZA | Mostly synergistic interactions (20/21) with ATM fixed at 8, 16, 32, and 64 mg/L |
| Wenzler, 2017 [25] | NA | E. coli (1) P. aeruginosa (1) C. freundii (1) | NDM-type (1) IMP-type (1) VIM-type (1) | >256 32 8 | NA | NA | GDS | NA | ATM+CZA | Synergistic interactions for E. coli and C. freundii (FIC index ≤ 0.5), indifferent interaction for P. aeruginosa (0.5 < FIC index < 1) |
| Zhang, 2017 [26] | Indiana (USA) 2010–2013 | E. cloacae (3) K. pneumoniae (1) | VIM-1 (3) NDM-1 (1) | NA | 4 (100%) | 0.12–0.25 | CB | All isolates co-produced KPC-3 variant | ATM+AVI | - |
| Avery, 2018 [27] | AR Bank collection; collection period NA | E. coli (5) K. pneumoniae (5) | NDM-1 (2) NDM-5 (2) NDM-6 (1) NDM-1 (3) VIM-1 (1) IMP-4 (1) | 64- >256 | 10 (100%) | NA | GDS | All isolates co-produce at least one serine-β-lactamase | ATM+CZA | E. coli: 4 isolates showed synergistic interactions (FIC index ≤ 0.5), 1 isolate showed additive interaction (0.5 < FIC index < 1) |
| Jayol, 2018 [28] | France, Colombia and Turkey; collection period NA | K. pneumoniae (15) | NDM-type (15) | 0.19- >256 MIC50: >256 MIC90: >256 | 15 (100%) | 0.094–2 MIC50: 0.38 MIC90: 1.5 | GDS | Six isolates co-produced OXA-48 enzymes; 1 isolate co-produced OXA-181 enzyme; all strains were resistant to colistin | ATM+CZA | - |
| Sader, 2018 [29] | Worldwide, 2016 | Enterobacterales (61) | NDM-1 (41)NDM-type (10) VIM-1 (7) IMP-type (3) | NA | 61 (100%) | ≤0.03 to 4 MIC50: 0.12MIC90: 0.5 | CB | NA | ATM+AVI | - |
| Biagi, 2019 [30] | NA | E. coli (4) K. pneumoniae (4) | NDM-type (8) | 0.25- ≥256 MIC50: 128 MIC90: ≥256 | 7 (87.5%) | ≤0.03–16 MIC50: 0.25 MIC90: 16 | CB, TK | All isolates co-produce at least one serine-β-lactamase | ATM+CZA | Time kill: synergistic for seven strains, except for the ATM-susceptible E. coli |
| Lin, 2019 [31] | China 2015–2016 | K. pneumoniae (23) | NDM-1 (23) | MIC values NA; all strains were resistant | 21 (91.3%) | ≤0.25–8 MIC50: 0.5 MIC90: 2 | CB | All isolates co-produced TEM-1 and SHV-12; most strains also have other resistance determinants | ATM+AVI | - |
| Mikhail, 2019 [32] | NA | P. aeruginosa (2) | IMP-48 (2) | 64 | 0 (0%) | 64 | CB | OprD loss, OXA-10, MexCD-OprN overexpression (5X; only one isolate) | ATM+CZA | Not synergistic interactions |
| Pragasam, 2019 [33] | India, NA | K. pneumoniae (9) | NDM-type (9) | 128- >1024 | 9 (100%) | ≤0.12–0.25; MIC50: ≤0.12; MIC90: ≤0.12 | CB | Six isolates co-harbored OXA-48-type enzymes | ATM+CZA | - |
| Zou, 2019 [34] | China 2012–2018 | Enterobacterales (13) | NDM-5 (5) NDM-1 (4) IMP-4 (3) IMP-8 (1) | NA | 12 (92.3%) | <=0.125–8 MIC50: 0.5 MIC90: 4 | CB | All isolates co-produced ESBLs and presented porin (OMP) loss; the IMP-4-producing isolate co-produced a KPC-2 variant | ATM+AVI | - |
| Esposito, 2020 [35] | Africa/Middle east, Asia-Pacific, Europe and Latin America 2016–2017 | K. pneumoniae (114) | NA | 0.06- ≥256 MIC50: ≥256 MIC90: ≥256 | 114 (100%) | 0.03–0.5 MIC50: 0.12 MIC90: 0.25 | CB | NA | ATM+AVI | - |
| Kilic, 2020 [36] | Turkey; collection period NA | K. pneumoniae (17) | NDM-1 (13) NDM-1 + OXA-48 (4) | ≥64 | 17 (100%) | <4; MIC50 and MIC90 values were NA | CB | NA | ATM+AVI | Synergistic for all isolates (FIC index ≤0.5) |
| Kim, 2020 [37] | Korea 201 4–2018 | K. pneumoniae (11) | NDM-type | 256- >512 MIC50: ≥512 MIC90: ≥512 | 11 (100%) | 0.06–2 MIC50: 0.25 MIC90: 1 | CB | NA | ATM+AVI | Two isolates showed MIC values of 64 mg/L for ATM in combination with AVI when tested at high bacterial inoculum (1 × 107 CFU/mL) |
| Lee, 2020 [38] | FDA-CDC Bank collection, collection period NA | P. aeruginosa (5) | VIM-2 (3) VIM-4 (1) IMP-14 (1) | 16- >128 | 0 (0%) | 16–64 MIC50: 32 MIC90: 64 | CB, TK | All isolates co-produce at least one serine-β-lactamase | ATM+CZA | Mostly synergistic interactions (4/5) with MIC values >4 mg/L for ATM in combination |
| Niu, 2020 [39] | HMH-CDI collection, NA | K. pneumoniae (68) | Single MBLs (55): NDM-type (43) VIM-type (11) IMP-type (1) Dual MBLs (12) Triple MBLs (1) | ≤0.5 to >128 (65/68 strains were resistant) | 66 (97.1%) | All strains: ≤0.25 to 8; MIC50: ≤0.25; MIC90: 1; Single MBL: ≤0.25 to 1; MIC50: ≤0.25; MIC90: 0.5; Dual/triple CPE: ≤0.25 to 8; MIC50: 0.5; MIC90: 8 | CB | The two NDM-1-producing isolates with ATM MIC value of 8 mg/L produced NDM-1, OXA-48, CTX-M-15, CMY-16, SHV-1, TEM-1, OXA-10, SCO-1 associated with outer membrane protein defects (OmpK35, OmpK36) | ATM+AVI | Synergistic interactions |
| Periasamy, 2020 [40] | India 2016–2018 | E. coli (23) | NDM-type (23) | >32 | 10 (43.5%) | 2–16 | CB | All isolates presented insertions in PBP3 | ATM+AVI | - |
| Wei, 2020 [41] | Southwest China 2018–2019 | Enterobacterales (26) | NDM-5 (14) NDM-1 (11) IMP-4 (1) | 2–512 | 26 (100%) | 0.125–2; MIC50: 0.125 MIC90: 2 | CB | Four NDM-1-producing isolates co-harbored KPC genes | ATM+AVI | - |
| Yang, 2020 [42] | Taiwan 2012–2015 | K. pneumoniae (14) E. coli (9) | K. pneumoniae: IMP-type (9) VIM-type (5) E. coli: NDM-type (5) VIM-type (3) IMP-type (1) | K. pneumoniae: 0.125- >32 (11/14 were resistant) E. coli: 2- >32 (8/9 were resistant) | K. pneumoniae: 13 (92.9%) E. coli: 9 (100%) | K. pneumoniae: <0.06- >32 MIC50: 0.125 MIC90: 0.5 E. coli: <0.06- 2 MIC50: 0.125 MIC90: 2 | AD | All isolates also produced ESBL genes | ATM+AVI | - |
| Zhang, 2020 [43] | China October 2016–September 2017 | Enterobacterales (161) | NDM-type (151) IMP-type (13) VIM-type (2) | ≤1- ≥64 MIC50: ≥64 MIC90: ≥64 | 161 (100%) | ≤0.125–4 MIC50: ≤0.125 MIC90: 1 | CB | Most isolates also produced various resistance determinants | ATM+AVI | Five isolates co-harbored two MBLs |
| Zou, 2020 [44] | Southwest China, 2018–2019 | Enterobacterales (29) | NDM-type (24) IMP-4 (1) NDM-1 (3) NDM-1+VIM-4 (1) | NA | 29 (100%) | ≤0.125 to 4 MIC50: ≤0.125 MIC90:1 (Cumulative data, not related only to MBL isolates) | CB | Three NDM-1-producing isolates co-harbored a KPC-2 gene | ATM+AVI | - |
| Bhatnagar, 2021 [45] | CDC & FDA Antibiotic Resistance Isolate Bank or CDC’s internal collection; collection period NA | Enterobacterales (37) | NDM-type (2) NDM-1 (24) NDM-5 (2) NDM-6 (1) NDM-7 (1) VIM-1 (2) VIM-27 (2) IMP-4 (2) IMP-8 (1) | 8- >64 | 32 (80%) | ≤0.5–8 MIC50: ≤0.5 MIC90: 8 | CB | Five NDM-producing K. pneumoniae and one NDM-producing E. coli also co-produced OXA-232 and OXA-181, respectively | ATM+AVI | Five NDM-producing E. coli showed MIC values of 8 mg/L for ATM in combination with AVI |
| Chang, 2021 [46] | China 2015–2019 | Elizabethkingia anophelis (37) | GOB and BlaB (basal enzymes) | >256 | 0 (0%) | >256 | CB | 35 isolates co-produced CME determinant | ATM+AVI | - |
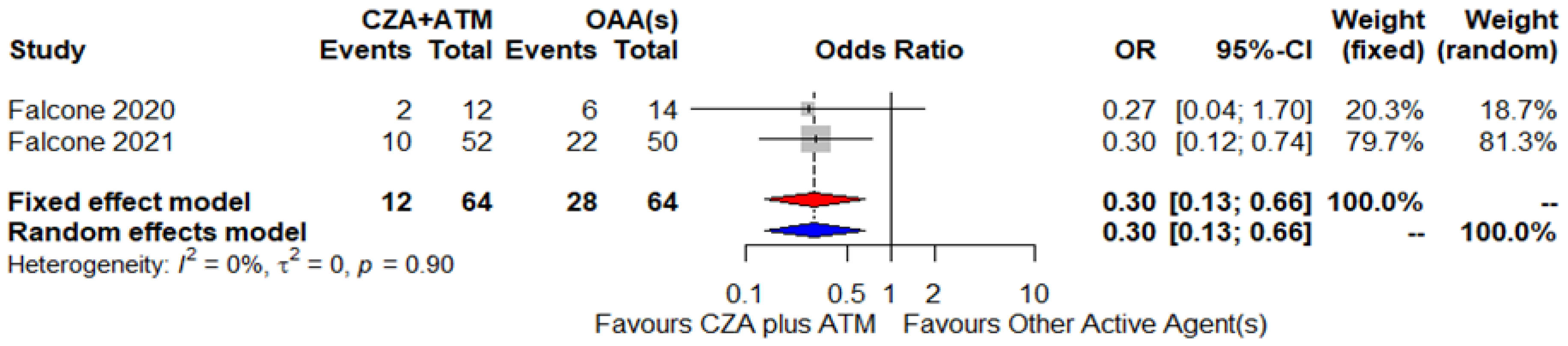
| Falcone, 2021 [47] | Italy and Greece, November 2018–December 2019 | Enterobacterales (52) | NDM-type (47) VIM-type (5) | NA | NA | NA | DD GDS | NA | ATM+CZA | Synergistic interactions |
| Lin, 2021 [48] | China 2011–2018 | Stenotrophomonas maltophilia (76) | L1 (basal enzymes) | 1–1024 | 65 (85.5%) | 0.06–64 MIC50: 2 MIC90: 8 | CB | All isolates co-produced L2 determinant (basal enzyme) | ATM+AVI | - |
| Maraki, 2021 [49] | Greece 2016–2020 | K. pneumoniae (40) | NDM-type (35) VIM-type (2) NDM-type+VIMP-type (3) | 24- >256 | 40 (100%) | 0.06–0.56 MIC50: 0.31 MIC90: 0.37 | GDS | KPC-type enzymes co-produced by one NDM-producing and two VIM-producing isolates | ATM+CZA | - |
| Author and Publication Year (ref) | Number and Species (%) of Strains with MIC >4 mg/L for ATM+AVI | MBL Determinant | Presumptive Determinants Involved in High MIC Values for ATM in Combination | MICs of ATM in Combination with AVI and Supplemental Notes | Antibiotic Combination |
|---|---|---|---|---|---|
| Alm, 2015 [17] | 2/31 (6.5%); E. coli | NDM-1 | Both E. coli isolates presented a PBP3 (YRIK) insertion and co-production of other resistance determinants: 1) CTX-M-15, CMY-42, TEM-1, OXA-1; 2) CTX-M-15, CMY-4, OXA-1 | 14 E. coli isolates presented a PBP3 insertion but only two with MIC values >4 mg/L for ATM in combination with AVI | ATM+AVI |
| Kazmierczak, 2015 * [18] | 308 (100%) P. aeruginosa | VIM-type (n = 270) IMP-type (n = 35) NDM-type (n = 3) | Co-production of various (not specified) resistance determinants (cumulative data) | Mostly not synergistic; MIC50 ≥16 mg/L MIC90 ≥32 mg/L (cumulative data) | ATM+AVI |
| Li, 2015 [19] | 1/2 (50%) E. coli | VIM-1 | Loss of an outer membrane protein; co-production of KPC-3 and TEM-1 | MIC 8 mg/L | ATM+AVI |
| Vasoo, 2015 [20] | 5/47 (10.6%) isolates: 2/32 (6.3%) E. coli; 3/4 (75%) P. aeruginosa | NDM-1 and NDM-7 (n = 2; E. coli) VIM-type (n = 3; P. aeruginosa) | Altered PBP3 affinity (E. coli); porin loss and overexpression of efflux pump (P. aeruginosa) | E. coli isolates with MIC values for ATM in combination with AVI MIC values of 8 (NDM-1) and 16 (NDM-7) mg/L, while P. aeruginosa isolates with MIC value of 8 mg/L | ATM+AVI |
| Karlowsky, 2017 [23] | 2/267 (0.7%) Enterobacterales 405/452 (89.6%) P. aeruginosa | NA (cumulative data) | NA | Enterobacterales: MICs 8 mg/L P. aeruginosa (cumulative data): MIC50 16 mg/L MIC90 32 mg/L | ATM+AVI |
| Marshall, 2017 [24] | 4/21 (19%) isolates: E. coli (n = 2), K. pneumoniae, P. rettgeri | NDM-1 (n = 4) | Other co-produced determinants: E. coli—CTX-M-15, CMY-2, TEM-type; K. pneumoniae—CTX-M-15, CMY-2, DHA-type, SHV-type, TEM-type; P. rettgeri—CMY-2, DHA-type | All four isolates had MICs ≥ 16 mg/L. One E. coli showed synergy with ATM fixed at 32 mg/L, in combination with AVI; one E. coli and K. pneumoniae showed synergy with ATM fixed at 64 mg/L. P. rettgeri was not evaluated with ATM fixed at 32 and 64 mg/L | ATM+CZA |
| Wenzler, 2017 [25] | 1/3 (33.3%) P. aeruginosa | IMP-type | NA | NA | ATM+CZA |
| Biagi, 2019 [30] | 1/8 (12.5%) Enterobacterales (E. coli) | NDM-type, | Co-production of CMY-2/FOX-type, CTX-M-1, TEM-type | MIC 16 mg/L | ATM+CZA |
| Lin, 2019 [31] | 2/23 (8.7%) K. pneumoniae | NDM-1 | Co-production of TEM-1 and SHV-12 | MICs 8 mg/L | ATM+AVI |
| Mikhail, 2019 [32] | 2/2 (100%) P. aeruginosa | IMP-48 | OprD loss, OXA-10, MexCD-OprN overexpression (5X; only one isolate) | MICs 64 mg/L | ATM+CZA |
| Zou, 2019 [34] | 1/12 (8.3%) Enterobacterales | NDM-5 | Loss of an outer membrane protein and co-production of ESBL determinant | MIC 8 mg/L | ATM+AVI |
| Lee, 2021 [38] | 5/5 (100%) P. aeruginosa | IMP-14; VIM-4; VIM-2; VIM-2; VIM-2 | Co-production of: OXA-10, OXA-488, VEB-9, PDC-2; OXA-396, PDC-3; OXA-488, PDC-2; OXA-488, PDC-3; PDC-8 | MIC range 16–64 mg/L | ATM+AVI |
| Niu, 2020 [39] | 2/68 (2.9%) isolates: K. pneumoniae | NDM-1 | NDM-1, OXA-48, CTX-M-15, CMY-16, SHV-1, TEM-1, OXA-10, SCO-1 associated with outer membrane protein defects (OmpK35, OmpK36) | MICs 8 mg/L | ATM+AVI |
| Periasamy, 2020 [40] | 13/23 (56.5%) E. coli | NDM-type | Insertions in PBP3 (YRIN and YRIK) | MICs 8–16 mg/L | ATM+AVI |
| Yang, 2020 [42] | 1/14 (7.1%) K. pneumoniae | NA | NA | MIC 32 mg/L | ATM+AVI |
| Bhatnagar, 2021 [45] | 5/37 (20%) E. coli | NDM-1 (3), NDM-5 (1), NDM-6 (1) | NA | MICs 8 mg/L | ATM+AVI |
| Chang, 2021 [46] | 37/37 (100%) Elizabethkingia anophelis | GOB and BlaB (constitutionally produced enzymes) | GOB, BlaB and CME determinants | MICs >256 mg/L | ATM+AVI |
| Lin, 2021 [48] | 11/76 (15.5%) Stenotrophomonas maltophilia | L1 (constitutionally produced enzyme) | NA | MIC range 8–64 mg/L | ATM+AVI |
| Authors and Publication Year (ref) | Study Design | Country and Time Span | Number of Patients (and Age in Years, Mean) a | Sex (M, F, n) | Types of Infection (n) | Microorganism (n) | Mechanism of Resistance | AST Profile | Therapeutic Regimen (n) | Type and Line b of Therapy (n) | Outcome | Notes |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mojica, 2016 [50] | Case report | United States, NR | 1 (19) | 1F | CLABSI (1) | Stenotrophomonas maltophilia (1) | L1 and L2 β-lactamases | Resistance to CAZ, CZA, ATM, IMI | CZA 2.5 g tid + ATM 2 g tid for 48 days (1) | Targeted (1) – Salvage (1) | Clinical resolution without AE and no recurrence after ≥90 days of follow-up | Renal transplant patient. CZA + ATM started after 63 days of previous antibiotics |
| Davido, 2017 [51] | Cases report | United States, NR | 2 (61) | 1M, 1F | CLABSI (1), pneumonia with lung abscess (1) | Klebsiella pneumoniae (1), Pseudomonas aeruginosa (1) | CLABSI→OXA-48, NDM-1 Pneumonia→NDM-1, AmpC-hyperproducing | Both strains→Resistance to ATM, CZA (susceptibility only to COL and AMK) | CZA 2.5 g tid + ATM 2 g tid for 10 days (1, CLABSI); CZA 2.5 g bid + ATM 2 g bid for 42 days (1, pneumonia) | Targeted (2)–First line (1, pneumonia) Salvage (1, CLABSI) | CLABSI→resolution of infection, later death due to heart failure; Pneumonia→clinical resolution | In CLABSI patient, CZA + ATM started after 26 days of previous antibiotics |
| Mittal, 2018 [52] | Case report | United States, 2017 | 1 (42) | 1M | BJI (1) | Klebsiella pneumoniae (1) | NDM-1 and OXA-181 | Resistance to ATM, CZA, MER (susceptibility only to COL) | CZA 2.5 g tid + ATM 2 g tid for 48 days (1) | Targeted (1) – Salvage (1) | Microbiological cure and wound healing (functional limitation of the arm) | Right elbow osteomyelitis in a returning traveler from Bangladesh. Concurrent osteomyelitis from Rhizopus |
| Shaw, 2018 [53] | Case series | Spain, January 2016–June 2017 | 10 (68.5) | 7M, 3F | BSI (4), HAP (2), cUTI (2), CLABSI (1), mediastinitis (1) | Klebsiella pneumoniae (10) | NDM-1 and OXA-48 as well as CTX-M-15 | Resistance to all β-lactams, intermediate to TIG; in 40% of case resistance also to COL | CZA 2.5 g tid + ATM 3 g od (4); CZA 2.5 g tid + ATM 1 g tid (2); CZA 2.5 g tid + ATM 2 g tid (1); CZA 1.5 g tid + ATM 3 g od (1); CZA 1.25 g od + ATM 2 g od (1); CZA 940 mg bid + ATM 2 g od (1) | Targeted (10)–First line (5) Salvage (5) | At day 30, success rate 60%, 3 deaths and 1 recurrence (cUTI); among the 6 successful cases, 2 recurrences at day 90 (1 BSI, 1 cUTI). No AE and no attributable deaths related to infections. | Hospital outbreak started in late 2015 by an XDR strain (KP-HUB-ST147). Therapy duration: 3–28 days |
| Emeraud, 2019 [54] | Case report | France, NR | 1 (70) | 1M | cUTI (1) | Escherichia coli (1) | NDM-1 and OXA-48 as well as CTX-M-15 | Extremely drug-resistant strain | CZA 2.5 g tid + ATM 2 g tid for 10 days (1) | Targeted (1) – Salvage (1) | Clinical resolution without AE and no recurrence over 2 months | Medical history of recent travel in India |
| Hobson, 2019 [55] | Case report | France, NR | 1 (3) | 1F | BSI (1) | Morganella morganii (1) | NDM-1 | Resistance to CAZ, CZA, ATM, IMI | CZA 100 mg/Kg/day (as for ceftazidime component) + ATM 150 mg/Kg/die for 10 days (1) | Targeted (1) – First line (1) | Clinical resolution without AE and no recurrence over 6 months days of follow-up | Pediatric patient with lymphoblastic acute leukemia |
| Shah, 2019 [56] | Case report | United States, NR | 1 (80) | 1M | BSI (1) | Klebsiella pneumoniae (1) | Suspected but not confirmed carbapenemase (molecular testing not performed) | Susceptibility only to COL and TIG | CZA 0.94 g bid + ATM 1 g tid for 10 days (1) | Empiric (1)– Salvage (1) | Only partial improvement | BSI from urinary source. Therapy included POLB as third agent. Previous exposure to COL in India |
| Stewart, 2019 [57] | Case report | United States, NR | 1 (61) | 1M | Necrotizing kidney allograft infection (1) | Enterobacter cloacae (1) | NDM-1 | Susceptibility only to COL | CZA + ATM for 46 days (1) | Targeted (1) – Salvage (1) | Infection resolution but patient dialysis- dependent at 4-month follow-up | Surgical source control was carried out. Concurrent fungal infection of the kidney with Rhizopus |
| Benchetrit, 2020 [58] | Cases report | France, NR | 2 (55.5) | 2F | BSI (1), VAP (1) | Klebsiella pneumoniae (2) | NDM-1 | Susceptibility only to COL | CZA 0.94 g od + ATM 2 g bid for 14 days (1, BSI); CZA 2.5 g od + ATM 2 g bid for 14 days (1, VAP) | Targeted (2) – Salvage (2) | Resolution of infection, but in both cases recurrence within 30 days, treated with CZA+ATM; the patient affected by BSI survived, the other one died due to chronic lung transplant rejection | The cases described involved 2 solid organ transplant recipients (kidney and lung). In the case of VAP, TIG was added |
| Falcone, 2020 [4] | Retrospective cohort | Italy, November 2018–May 2019 | NAAG | NAAG | BSI (12) | Klebsiella pneumoniae (NAAG), Escherichia coli (NAAG) | NDM-1 and NDM-5 | NAAG | CZA 2.5 g tid + ATM 2 g tid | Targeted (12) – NAAG | Two patients died out of 12 | Original cohort made up of 40 patients, 70% males, median age 70.5 (IQR 55.3–77.8), susceptibility rate of 91.4% to COL (out of 35 available strains). In the whole cohort, Charlson comorbidity index score and age were factors independently associated with 30-day mortality at Cox regression analysis |
| Alghoribi, 2021 [59] | Case report | Saudi Arabia, February 2019 | 1 (40) | 1F | CIED/IE (1) | Klebsiella pneumoniae (1) | NDM-1, OXA-48, CTX-M-14b, SHV-28 and OXA-1 | Pan-resistance | CZA 2.5 g tid + ATM 2 g tid for 46 days (1) | Targeted (1)– Salvage (1) | Infection resolution and no recurrence at 6-month follow-up | IE affecting the tricuspid valve along with CIED. Source control obtained through pacer device and lead tip replacement |
| Bocanegra-Ibarias, 2021 [60] | Case report | Mexico, NR | 1 (35) | 1M | BSI (1) | Klebsiella pneumoniae (1) | NDM-1 | Susceptibility only to COL | CZA 2.5 g tid + ATM 2 g tid for 10 days (1) | Targeted (1)– Salvage (1) | Infection resolution and no signs or symptoms of infection after 80 days of follow-up, but subsequent death related to underlying disease | The patient underwent HSCT to treat a severe form of aplastic anemia |
| Cairns, 2021 [61] | Case series | Australia, NR | 4 (59) | 4M | BJI (1), CLABSI (1), cUTI (2) | Enterobacter cloacae (4) | IMP-4 | Resistance to all β-lactams | CZA 2.5 g tid + ATM 2 g tid (3); CZA 0.94 g bid +ATM 1.5 g tid (1) | Targeted (4)– Salvage (4) | Infection resolution in all cases; one recurrence of cUTI at 4-month follow-up and one death not related to CLABSI beyond 4 weeks of follow-up | Outbreak in a single institution involving three SOT patients and one HSCT subject. Therapy duration: 24–49 days. |
| Cowart, 2021 [62] | Case report | United States | 1 (11) | 1F | Pneumonia (1) | Stenotrophomonas maltophilia (1) | L1 and L2 β-lactamases | Susceptibility only to MIN and TMP/SMX | CZA 150–200 mg/Kg tid (as for ceftazidime component) + ATM 50 mg/Kg qid for 11 days (1) | Targeted (1)– First-line (1) | Infection resolution | Association with minocycline. Implementation of a continuous infusion strategy with TDM |
| Falcone, 2021 [47] | Prospective cohort | Italy and Greece, November 2018–May 2019 | 52 (69) | 39M, 13F | BSI (52) | Klebsiella pneumoniae (NAAG), Escherichia coli (NAAG), Enterobacter species (NAAG), and Morganella morganii (NAAG), | NDM and VIM | NAAG | CZA 2.5 g tid + ATM 2 g tid (1) | Targeted (52)– NAAG | Ten patients died out of 52 | Original cohort made up of 102 patients, 69% males, median age 70, susceptibility rate of 88 % to COL. Median duration of antibiotic therapy 10 days (IQR 7–14) |
| Perrotta, 2021 [63] | Case report | Italy, December 2020 | 1 (57) | 1M | BSI (1) | Klebsiella pneumoniae (1) | NDM | Susceptibility only to COL | CZA 1.25 g tid + ATM 1 g tid for 10 days (1) | Targeted (1)– First-line (1) | Clinical resolution and microbiological cure | Patient affected by TTP and COVID-19, with ICU admission due to severe interstitial pneumonia and with known rectal colonization by MBL-producing strain. At the end of therapy, the patient’s rectal swab tested negative, showing achievement of decolonization |
| Sieswerda, 2021 [64] | Case report | Netherlands, NR | 1 (around 60 years) | 1F | cUTI (1) | Klebsiella pneumoniae (1) | NDM-1 | Pan-resistance | CZA 2.5 g tid + ATM 2 g tid for 14 days (1) | Targeted (1)– First-line (1) | After clinical resolution, the patient experienced a pyelonephritis by the same strain, recovered with an identical treatment | A case of BSI from urinary source in a kidney transplant recipient. Treatment commenced as empiric covering a previous bacteriuria by a carbapenemase-producing K. pneumoniae strain, then targeted according to blood culture results |
| Yasmin, 2021 [65] | Case report | United States, NR | 1 (4) | 1M | BSI (1) | Enterobacter hormaechei subsp. hoffmannii (1) | NDM-1 and KPC-4 | Susceptibility only to COL | CZA 50 mg/Kg tid (as for ceftazidime component) + ATM 50 mg/Kg tid for 14 days (1) | Targeted (1) – Salvage (1) | Clinical and microbiological cure without relapse in 30-day follow-up | Pediatric patient with B cell acute lymphoblastic leukemia developing infection after stem cell infusion |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mauri, C.; Maraolo, A.E.; Di Bella, S.; Luzzaro, F.; Principe, L. The Revival of Aztreonam in Combination with Avibactam against Metallo-β-Lactamase-Producing Gram-Negatives: A Systematic Review of In Vitro Studies and Clinical Cases. Antibiotics 2021, 10, 1012. https://doi.org/10.3390/antibiotics10081012
Mauri C, Maraolo AE, Di Bella S, Luzzaro F, Principe L. The Revival of Aztreonam in Combination with Avibactam against Metallo-β-Lactamase-Producing Gram-Negatives: A Systematic Review of In Vitro Studies and Clinical Cases. Antibiotics. 2021; 10(8):1012. https://doi.org/10.3390/antibiotics10081012
Chicago/Turabian StyleMauri, Carola, Alberto Enrico Maraolo, Stefano Di Bella, Francesco Luzzaro, and Luigi Principe. 2021. "The Revival of Aztreonam in Combination with Avibactam against Metallo-β-Lactamase-Producing Gram-Negatives: A Systematic Review of In Vitro Studies and Clinical Cases" Antibiotics 10, no. 8: 1012. https://doi.org/10.3390/antibiotics10081012
APA StyleMauri, C., Maraolo, A. E., Di Bella, S., Luzzaro, F., & Principe, L. (2021). The Revival of Aztreonam in Combination with Avibactam against Metallo-β-Lactamase-Producing Gram-Negatives: A Systematic Review of In Vitro Studies and Clinical Cases. Antibiotics, 10(8), 1012. https://doi.org/10.3390/antibiotics10081012









