Functionalization Techniques Empowering Optical Fiber Biosensors in Label-Free Cancer Biomarker Detection
Abstract
1. Introduction
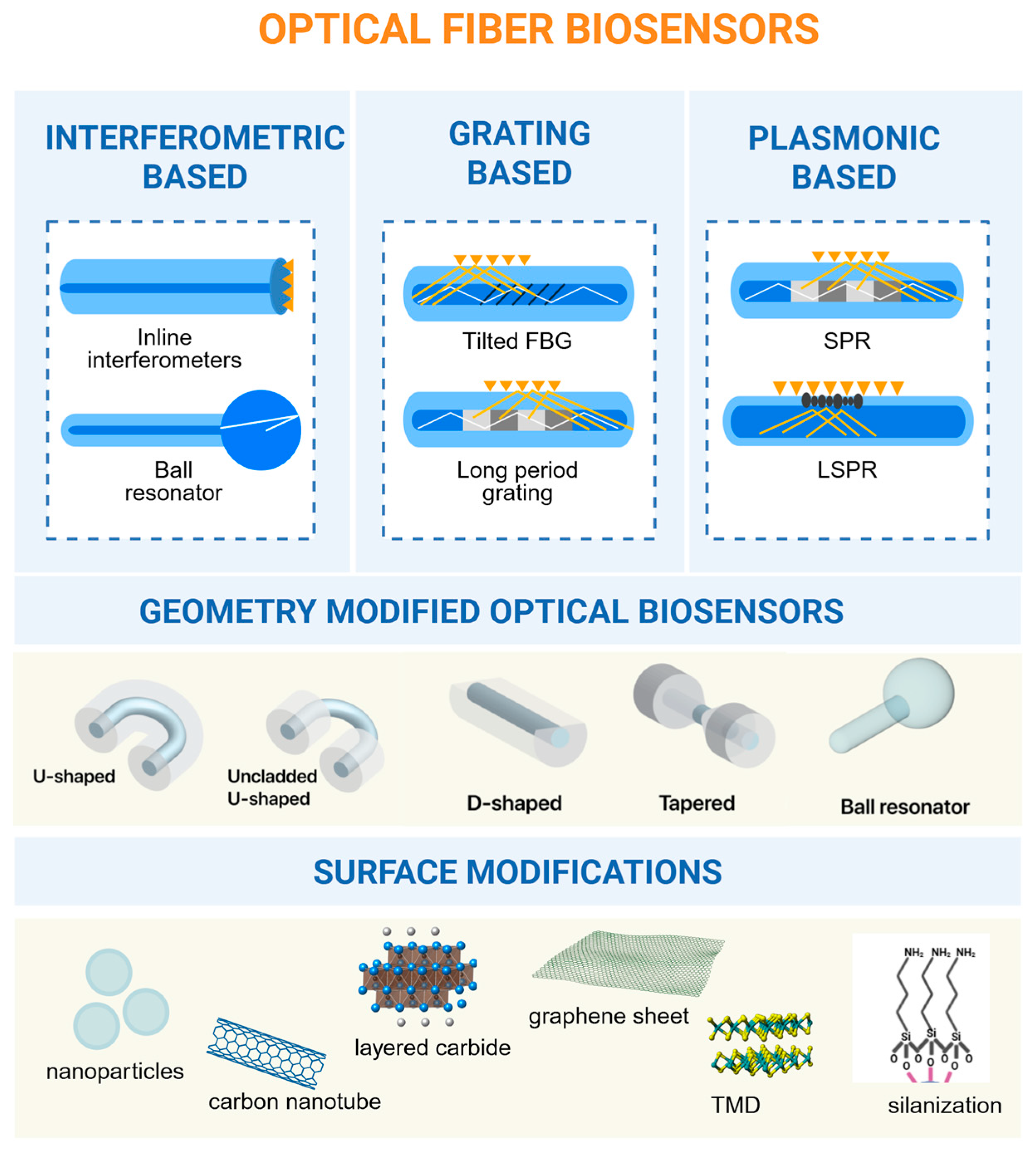
2. Principles and Types of Optical Fiber Biosensors
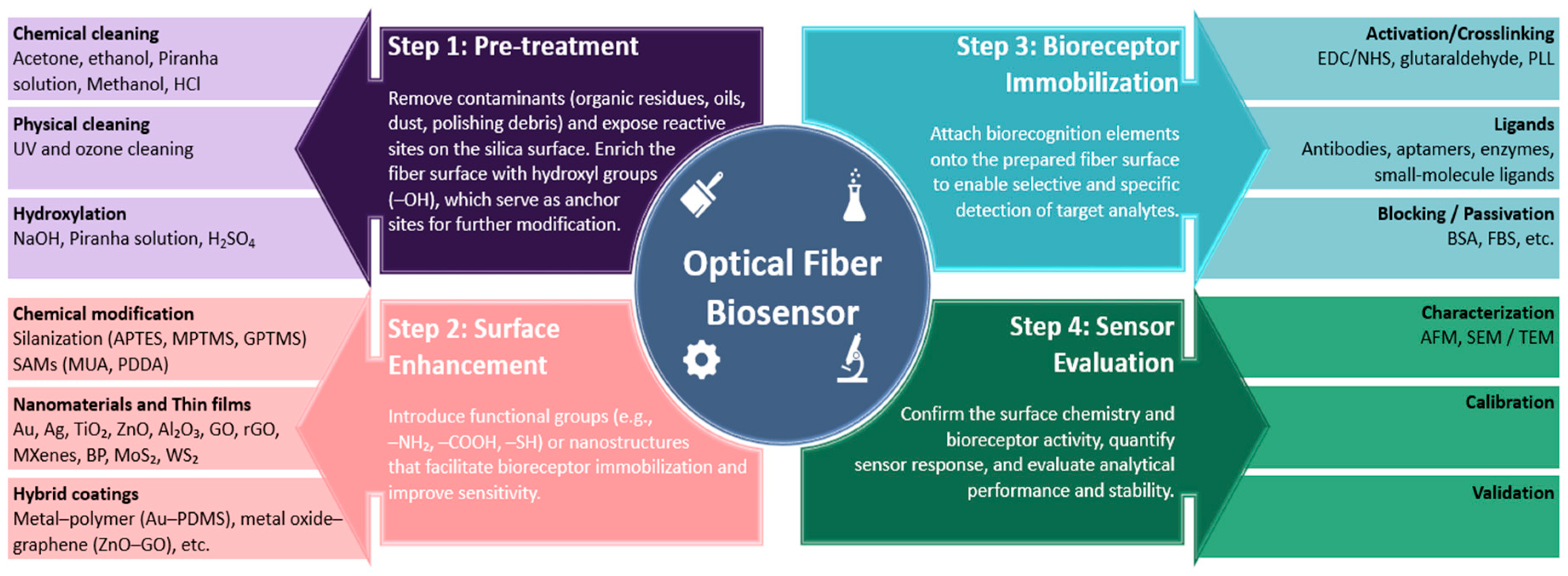
3. Functionalization Techniques for Optical Fiber Biosensors
3.1. Pre-Treatment of Optical Fibers
3.2. Surface Enhancement
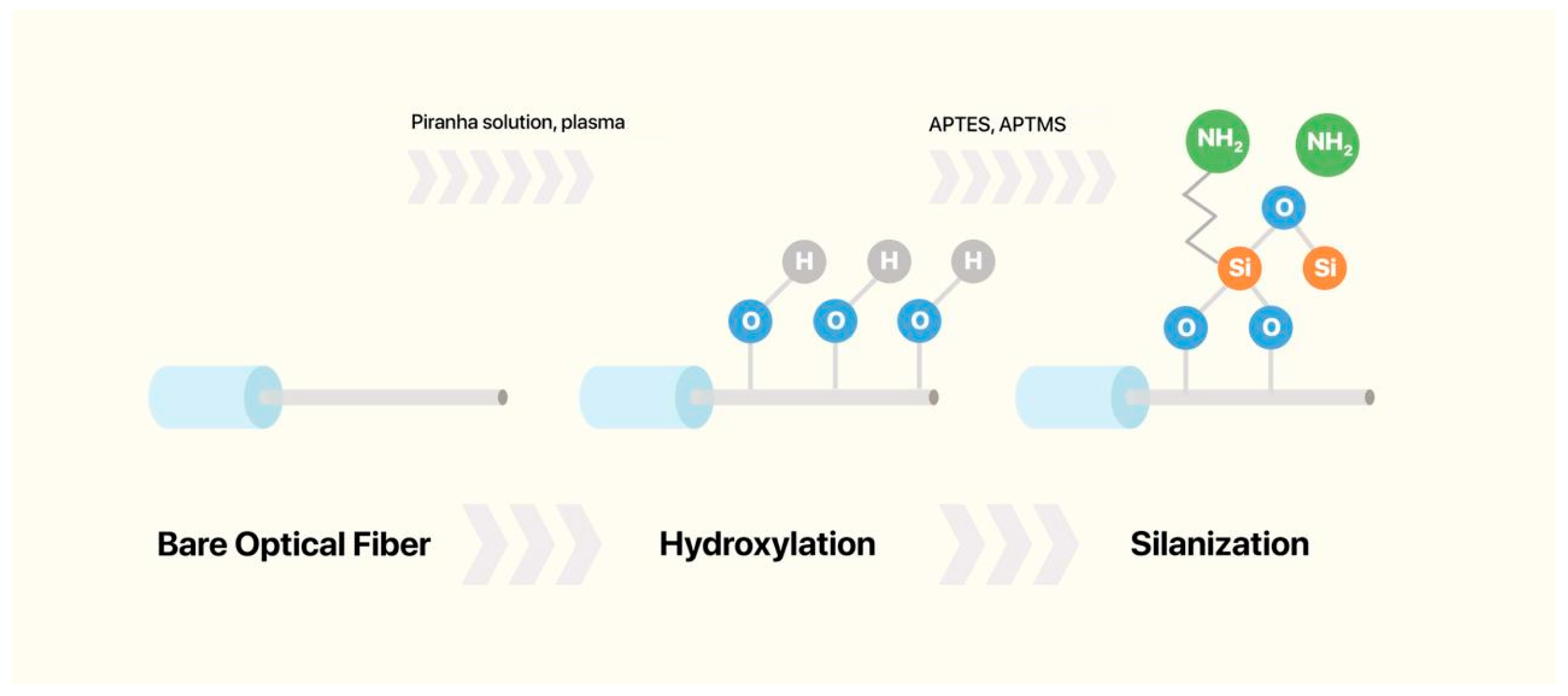
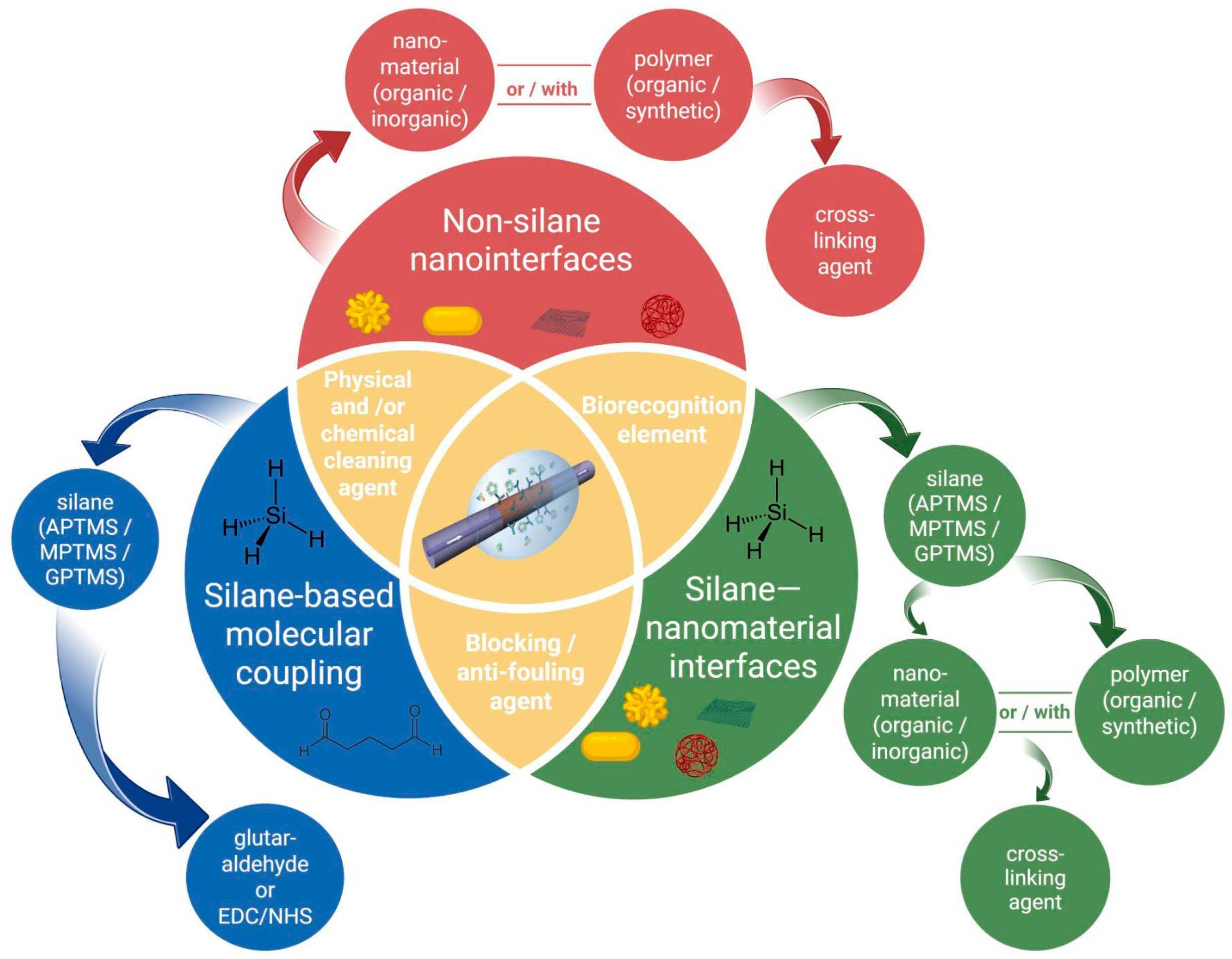
3.2.1. Silanization
3.2.2. Self-Assembled Monolayers (SAMs)
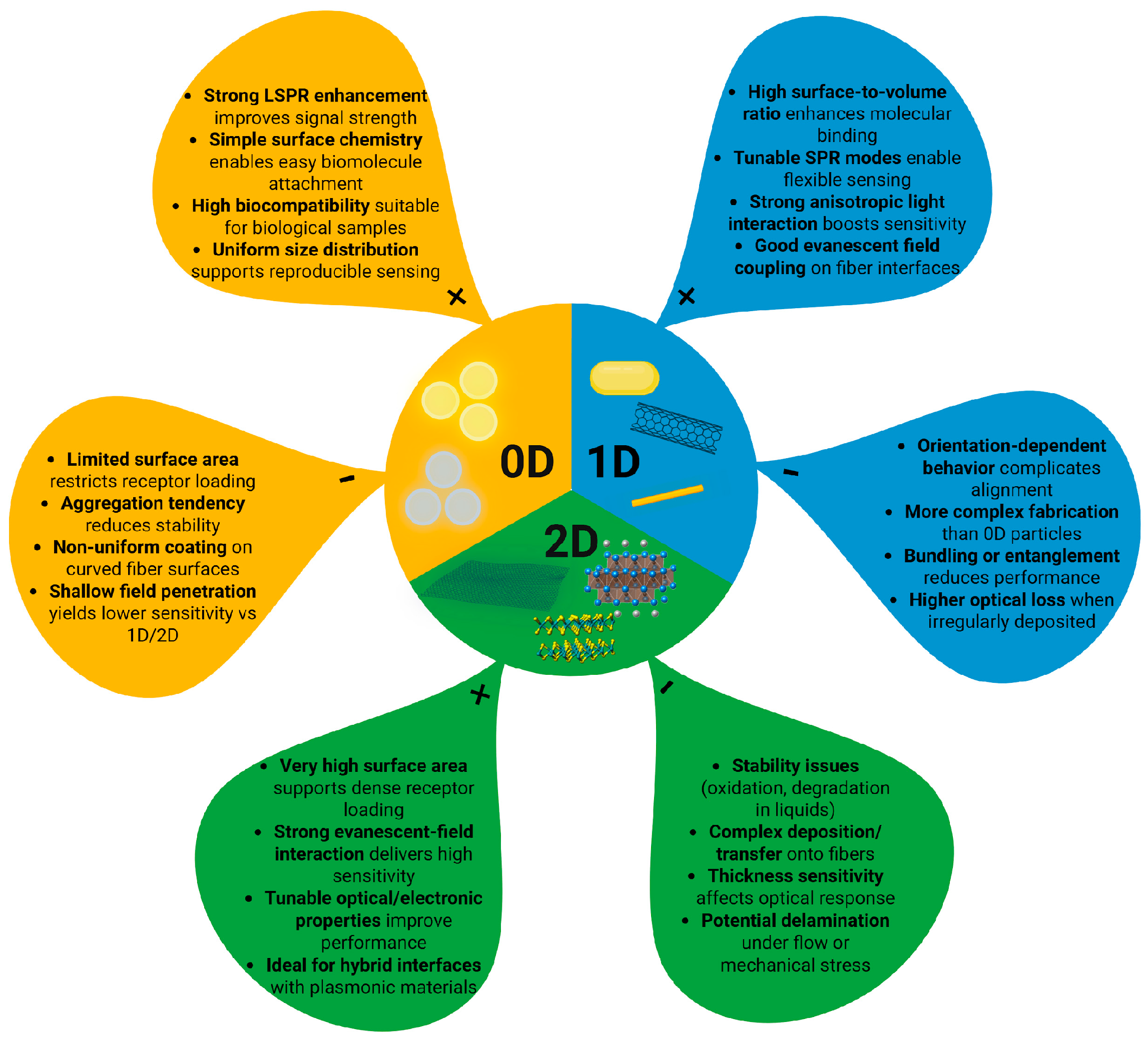
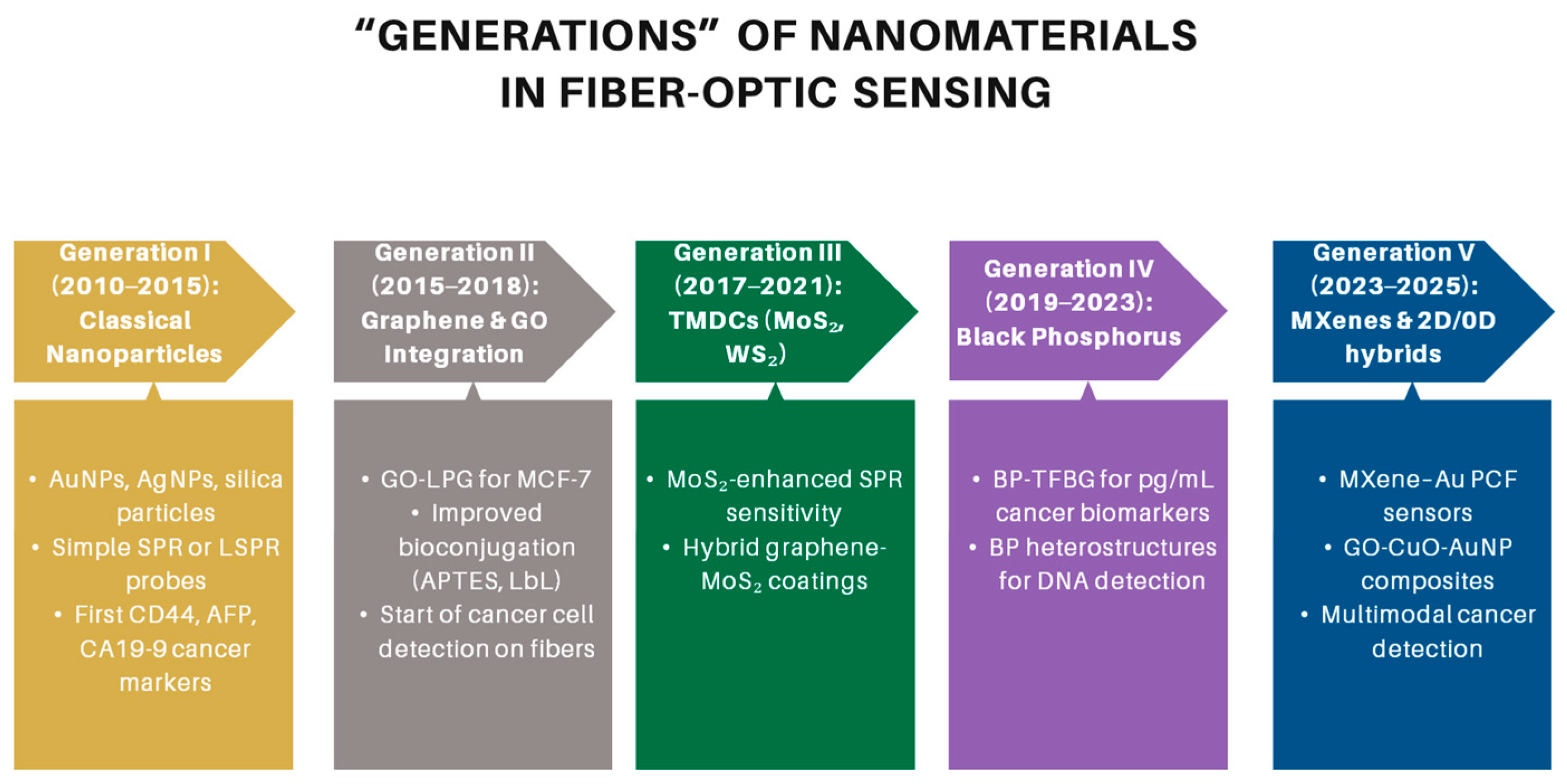
3.2.3. Nanomaterials-Based
3.2.4. Hybrid Structure-Based OFBs
3.2.5. Polymer-Based OFBs
3.3. Biorecognition Elements and Surface Passivation Strategies
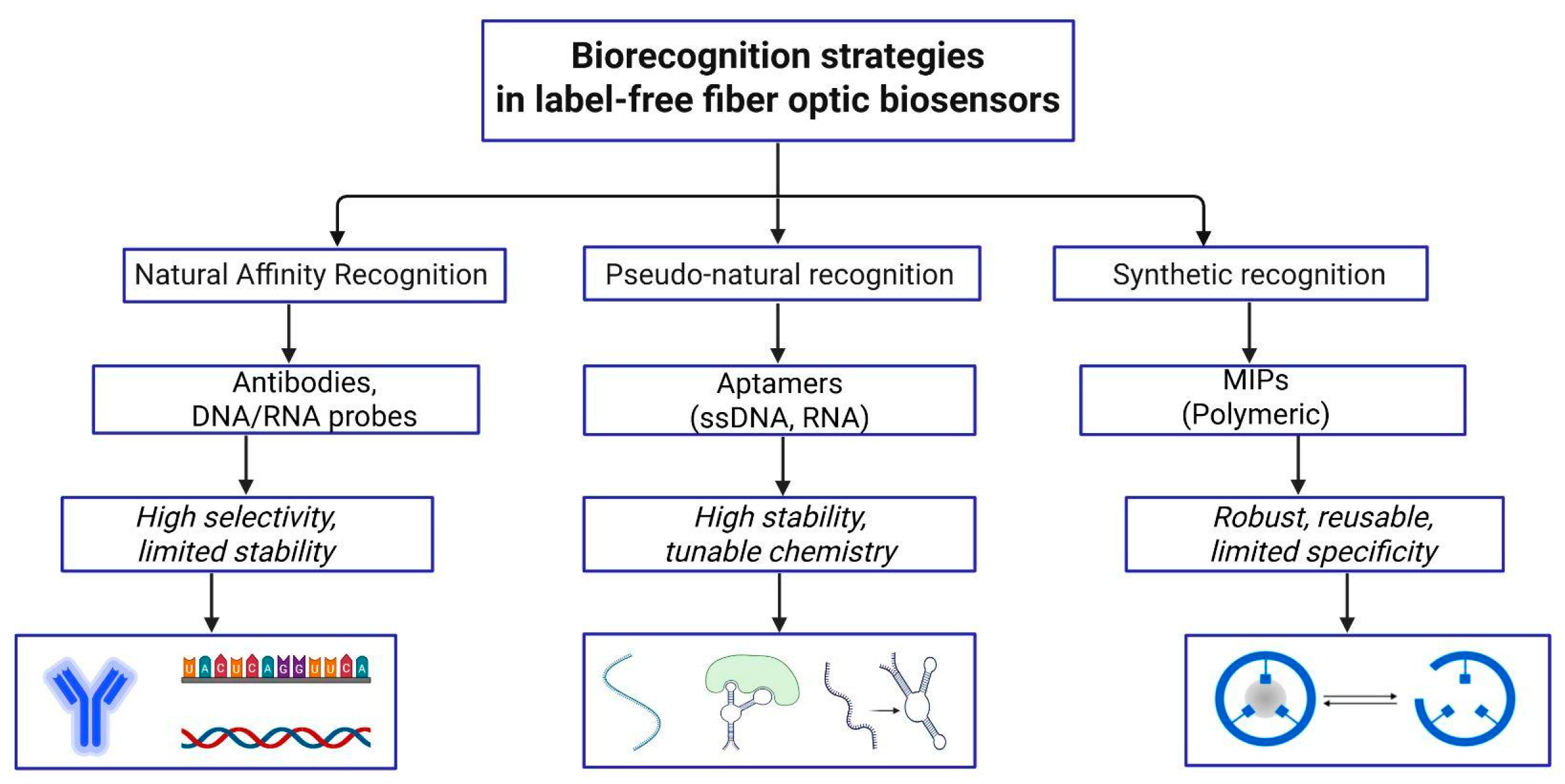
3.3.1. Biorecognition Elements (Antibodies, Aptamers, Nucleic Acids, Metabolic Analogs, Biotin, MIPs)
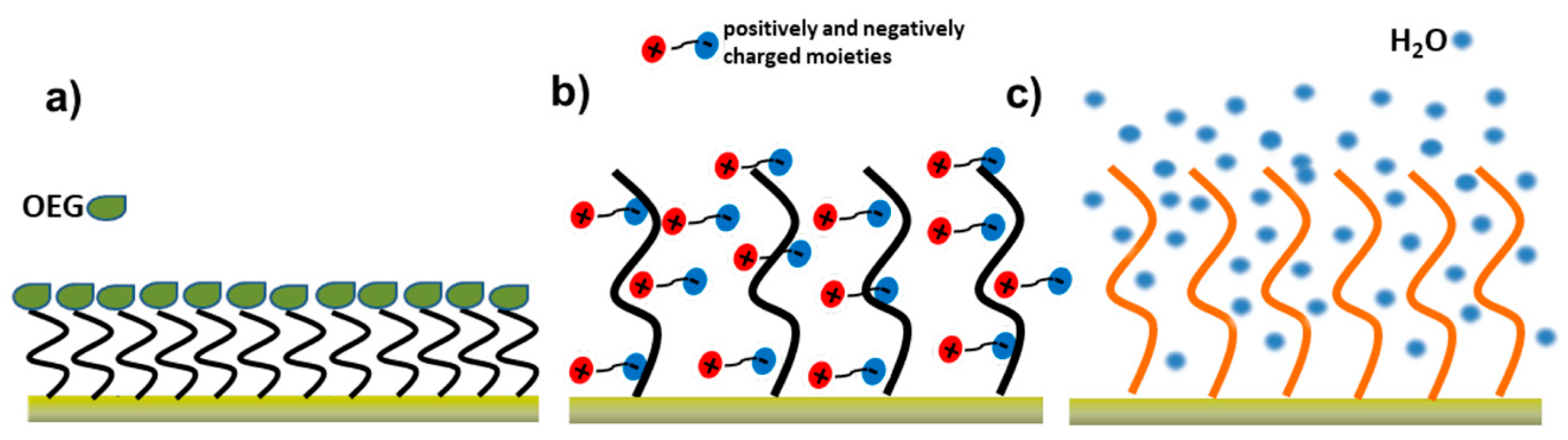
3.3.2. Blocking and Anti-Fouling Strategies
4. Computational Methods in Functionalized Optical Sensing for Oncology
5. Challenges and Future Perspectives
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Cancer. Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 19 January 2023).
- Crosby, D.; Bhatia, S.; Brindle, K.M.; Coussens, L.M.; Dive, C.; Emberton, M.; Esener, S.; Fitzgerald, R.C.; Gambhir, S.S.; Kuhn, P.; et al. Early detection of cancer. Science 2022, 375, eaay9040. [Google Scholar] [CrossRef]
- Soares, M.S.; Vidal, M.; Santos, N.F.; Costa, F.M.; Marques, C.; Pereira, S.O.; Leitão, C. Immunosensing based on optical fiber technology: Recent advances. Biosensors 2021, 11, 305. [Google Scholar] [CrossRef]
- Fitzgerald, R.C.; Antoniou, A.C.; Fruk, L.; Rosenfeld, N. The future of early cancer detection. Nat. Med. 2022, 28, 666–677. [Google Scholar] [CrossRef]
- Leitão, C.; Pereira, S.O.; Marques, C.; Cennamo, N.; Zeni, L.; Shaimerdenova, M.; Ayupova, T.; Tosi, D. Cost-Effective Fiber Optic Solutions for Biosensing. Biosensors 2022, 12, 575. [Google Scholar] [CrossRef] [PubMed]
- Zubair, H.; Begum, M.; Moradi, F.; Rahman, A.K.M.M.; Mahdiraji, G.A.; Oresegun, A.; Louay, G.T.; Omar, N.Y.; Khandaker, M.U.; Adikan, F.R.M.; et al. Recent Advances in Silica Glass Optical Fiber for Dosimetry Applications. IEEE Photonics J. 2020, 12, 6801525. [Google Scholar] [CrossRef]
- Zubia, J.; Arrue, J. Plastic Optical Fibers: An Introduction to Their Technological Processes and Applications. Opt. Fiber Technol. 2001, 7, 101–140. [Google Scholar] [CrossRef]
- Peters, K. Polymer optical fiber sensors—A review. Smart Mater. Struct. 2010, 20, 013002. [Google Scholar] [CrossRef]
- Wandermur, G.; Rodrigues, D.; Allil, R.; Queiroz, V.; Peixoto, R.; Werneck, M.; Miguel, M. Plastic optical fiber-based biosensor platform for rapid cell detection. Biosens. Bioelectron. 2014, 54, 661–666. [Google Scholar] [CrossRef]
- Kaur, B.; Kumar, S.; Kaushik, B.K. Recent advancements in optical biosensors for cancer detection. Biosens. Bioelectron. 2022, 197, 113805. [Google Scholar] [CrossRef] [PubMed]
- Azab, M.Y.; Hameed, M.F.O.; Obayya, S.S.A. Overview of Optical Biosensors for Early Cancer Detection: Fundamentals, Applications and Future Perspectives. Biology 2023, 12, 232. [Google Scholar] [CrossRef]
- Wallace, G.Q.; Masson, J.-F. From single cells to complex tissues in applications of surface-enhanced Raman scattering. Analyst 2020, 145, 7162–7185. [Google Scholar] [CrossRef]
- Lin, C.; Li, Y.; Peng, Y.; Zhao, S.; Xu, M.; Zhang, L.; Huang, Z.; Shi, J.; Yang, Y. Recent development of surface-enhanced Raman scattering for biosensing. J. Nanobiotechnol. 2023, 21, 149. [Google Scholar] [CrossRef] [PubMed]
- Home. Nanobiosensors and Fluorescence Based Biosensors: An Overview. 2018. Available online: https://www.SID.ir (accessed on 12 August 2025).
- Fan, X.; White, I.M.; Shopova, S.I.; Zhu, H.; Suter, J.D.; Sun, Y. Sensitive optical biosensors for unlabeled targets: A review. Anal. Chim. Acta 2008, 620, 8–26. [Google Scholar] [CrossRef] [PubMed]
- Jin, F.; Xu, Z.; Cao, D.; Ran, Y.; Guan, B.-O. Fiber-Optic Biosensors for Cancer Theranostics: From in Vitro to in Vivo. Photonic Sens. 2024, 14, 240415. [Google Scholar] [CrossRef]
- Bureau, B.; Zhang, X.H.; Smektala, F.; Adam, J.-L.; Troles, J.; Ma, H.-L.; Boussard-Plèdel, C.; Lucas, J.; Lucas, P.; Le Coq, D.; et al. Recent advances in chalcogenide glasses. J. Non-Cryst. Solids 2004, 345–346, 276–283. [Google Scholar] [CrossRef]
- Bureau, B.; Boussard, C.; Cui, S.; Chahal, R.; Anne, M.L.; Nazabal, V.; Sire, O.; Loréal, O.; Lucas, P.; Monbet, V.; et al. Chalcogenide optical fibers for mid-infrared sensing. Opt. Eng. 2014, 53, 027101. [Google Scholar] [CrossRef]
- Lucas, P.; Riley, M.R.; Boussard-Plédel, C.; Bureau, B. Advances in chalcogenide fiber evanescent wave biochemical sensing. Anal. Biochem. 2006, 351, 1–10. [Google Scholar] [CrossRef]
- Sanghera, J.S.; Shaw, L.; Aggarwal, I.D. Applications of chalcogenide glass optical fibers. C. R. Chim. 2002, 5, 873–883. [Google Scholar] [CrossRef]
- Lucas, P.; Coleman, G.J.; Jiang, S.; Luo, T.; Yang, Z. Chalcogenide glass fibers: Optical window tailoring and suitability for bio-chemical sensing. Opt. Mater. 2015, 47, 530–536. [Google Scholar] [CrossRef]
- Li, X.; Chen, N.; Zhou, X.; Zhang, Y.; Zhao, Y.; Nguyen, L.V.; Ebendorff-Heidepriem, H.; Warren-Smith, S.C. In-situ DNA detection with an interferometric-type optical sensor based on tapered exposed core microstructured optical fiber. Sens. Actuators B Chem. 2022, 351, 130942. [Google Scholar] [CrossRef]
- Barozzi, M.; Manicardi, A.; Vannucci, A.; Candiani, A.; Sozzi, M.; Konstantaki, M.; Pissadakis, S.; Corradini, R.; Selleri, S.; Cucinotta, A. Optical Fiber Sensors for Label-Free DNA Detection. J. Light. Technol. 2016, 35, 3461–3472. [Google Scholar] [CrossRef]
- Lyu, S.; Wu, Z.; Shi, X.; Wu, Q. Optical Fiber Biosensors for Protein Detection: A Review. Photonics 2022, 9, 987. [Google Scholar] [CrossRef]
- Lukose, J.; Chidangil, S.; George, S.D. Optical technologies for the detection of viruses like COVID-19: Progress and prospects. Biosens. Bioelectron. 2021, 178, 113004. [Google Scholar] [CrossRef] [PubMed]
- Vajhadin, F.; Mazloum-Ardakani, M.; Sanati, A.; Haghniaz, R.; Travas-Sejdic, J. Optical cytosensors for the detection of circulating tumour cells. J. Mater. Chem. B 2022, 10, 990–1004. [Google Scholar] [CrossRef]
- Zu, L.; Chen, Y.; Xie, J.; Liu, W.; Feng, Y.; Zhang, Z.; Zhao, X.; Ma, Y.; Fang, Q.; Li, K.; et al. In situ tumor cells detection using nanotube-functionalized & microfluidic-controlling multiresonance optical fiber. Sens. Actuators B Chem. 2023, 393, 134176. [Google Scholar] [CrossRef]
- Soler, M.; Lechuga, L.M. Biochemistry strategies for label-free optical sensor biofunctionalization: Advances towards real applicability. Anal. Bioanal. Chem. 2021, 414, 5071–5085. [Google Scholar] [CrossRef]
- Lin, P.-H.; Li, B.-R. Antifouling strategies in advanced electrochemical sensors and biosensors. Analyst 2019, 145, 1110–1120. [Google Scholar] [CrossRef]
- Ramola, A.; Shakya, A.K.; Bergman, A. Comprehensive Analysis of Advancement in Optical Biosensing Techniques for Early Detection of Cancerous Cells. Biosensors 2025, 15, 292. [Google Scholar] [CrossRef]
- Dabagh, S.; Singh, R.; Borri, C.; Chiavaioli, F. Functional Nanomaterial Coatings on Optical Fibers: Toward Enhanced Biosensing Performance. IEEE Sens. Rev. 2025, 2, 157–169. [Google Scholar] [CrossRef]
- Szunerits, S.; Spadavecchia, J.; Boukherroub, R. Surface plasmon resonance: Signal amplification using colloidal gold nanoparticles for enhanced sensitivity. Rev. Anal. Chem. 2014, 33, 153–164. [Google Scholar] [CrossRef]
- Topor, C.-V.; Puiu, M.; Bala, C. Strategies for Surface Design in Surface Plasmon Resonance (SPR) Sensing. Biosensors 2023, 13, 465. [Google Scholar] [CrossRef]
- Gade, A.; Sharma, A.; Srivastava, N.; Flora, S. Surface plasmon resonance: A promising approach for label-free early cancer diagnosis. Clin. Chim. Acta 2022, 527, 79–88. [Google Scholar] [CrossRef]
- Lobry, M.; Loyez, M.; Hassan, E.M.; Chah, K.; DeRosa, M.C.; Goormaghtigh, E.; Wattiez, R.; Caucheteur, C. Multimodal plasmonic optical fiber grating aptasensor. Opt. Express 2020, 28, 7539–7551. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.; Shankar, P.M.; Mutharasan, R. A review of fiber-optic biosensors. Sens. Actuators B Chem. 2007, 125, 688–703. [Google Scholar] [CrossRef]
- Wu, Q.; Qu, Y.; Liu, J.; Yuan, J.; Wan, S.-P.; Wu, T.; He, X.-D.; Liub, B.; Liuc, D.; Ma, Y.; et al. Singlemode-Multimode-Singlemode Fiber Structures for Sensing Applications—A Review. IEEE Sens. J. 2020, 21, 12734–12751. [Google Scholar] [CrossRef]
- Wu, X.; Wang, Y.; Zhang, J.; Zhang, Y.; Rao, X.; Chen, C.; Liu, H.; Deng, Y.; Liao, C.; Smietana, M.J.; et al. A D-Shaped Polymer Optical Fiber Surface Plasmon Resonance Biosensor for Breast Cancer Detection Applications. Biosensors 2023, 14, 15. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Kumar, S.; Liu, F.-Z.; Shuang, C.; Zhang, B.; Jha, R.; Kaushik, B.K. Etched multicore fiber sensor using copper oxide and gold nanoparticles decorated graphene oxide structure for cancer cells detection. Biosens. Bioelectron. 2020, 168, 112557. [Google Scholar] [CrossRef]
- Guo, T.; González-Vila, Á.; Loyez, M.; Caucheteur, C. Plasmonic optical fiber-grating immunosensing: A review. Sensors 2017, 17, 2732. [Google Scholar] [CrossRef]
- Zhong, X.; Xie, Q.; Liu, Y.; He, Y.; Zhong, N.; Zhang, Z.; Karimi-Maleh, H.; Peng, X.; Lichtfouse, E. Recent advances in optical fiber grating sensors for detection of organic substances. Chem. Eng. J. 2024, 492, 152260. [Google Scholar] [CrossRef]
- Lee, B.H.; Kim, Y.H.; Park, K.S.; Eom, J.B.; Kim, M.J.; Rho, B.S.; Choi, H.Y. Interferometric fiber optic sensors. Sensors 2012, 12, 2467–2486. [Google Scholar] [CrossRef]
- Elsherif, M.; Salih, A.E.; Muñoz, M.G.; Alam, F.; AlQattan, B.; Antonysamy, D.S.; Zaki, M.F.; Yetisen, A.K.; Park, S.; Wilkinson, T.D.; et al. Optical Fiber Sensors: Working Principle, Applications, and Limitations. Adv. Photonics Res. 2022, 3, 2100371. [Google Scholar] [CrossRef]
- Shadab, A.; Raghuwanshi, S.K.; Kumar, S. Advances in Micro-Fabricated Fiber Bragg Grating for Detection of Physical, Chemical, and Biological Parameters—A Review. IEEE Sens. J. 2022, 22, 15650–15660. [Google Scholar] [CrossRef]
- Chiavaioli, F.; Baldini, F.; Tombelli, S.; Trono, C.; Giannetti, A. Biosensing with optical fiber gratings. Nanophotonics 2017, 6, 663–679. [Google Scholar] [CrossRef]
- Sun, D.; Ran, Y.; Wang, G. Label-free detection of cancer biomarkers using an in-line taper fiber-optic interferometer and a fiber bragg grating. Sensors 2017, 17, 2559. [Google Scholar] [CrossRef]
- Quero, G.; Consales, M.; Severino, R.; Vaiano, P.; Boniello, A.; Sandomenico, A.; Ruvo, M.; Borriello, A.; Diodato, L.; Zuppolini, S.; et al. Long period fiber grating nano-optrode for cancer biomarker detection. Biosens. Bioelectron. 2016, 80, 590–600. [Google Scholar] [CrossRef] [PubMed]
- Loyez, M.; Lobry, M.; Wattiez, R.; Caucheteur, C. Optical fiber gratings immunoassays. Sensors 2019, 19, 2595. [Google Scholar] [CrossRef]
- Chiang, C.-C.; Yeh, Y.-T.; Wang, T.-E.; Hsu, H.-C.; Wen, H.-Y. A pathway for detection of gastric cancer biomarkers via using a layer-by-layer coated D-shaped grinding long-period fiber grating sensor. Anal. Chim. Acta 2024, 1318, 342927. [Google Scholar] [CrossRef]
- Homola, J. Surface Plasmon Resonance Sensors for Detection of Chemical and Biological Species. Chem. Rev. 2008, 108, 462–493. [Google Scholar] [CrossRef] [PubMed]
- Bekmurzayeva, A.; Nurlankyzy, M.; Abdossova, A.; Myrkhiyeva, Z.; Tosi, D. All-fiber label-free optical fiber biosensors: From modern technologies to current applications [Invited]. Biomed. Opt. Express 2024, 15, 1453–1473. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Wei, Y.; Wang, Y.; Li, K.; Yu, H.; Wu, Y. Ultrasensitive interferometers based on zigzag-shaped tapered optical microfibers operating at the dispersion turning point. Opt. Express 2021, 29, 36926–36935. [Google Scholar] [CrossRef]
- Sun, D.; Fu, Y.; Yang, Y. Label-free detection of breast cancer biomarker using silica microfiber interferometry. Opt. Commun. 2020, 463, 125375. [Google Scholar] [CrossRef]
- Lobry, M.; Fasseaux, H.; Loyez, M.; Chah, K.; Goormaghtigh, E.; Wattiez, R.; Chiavaioli, F.; Caucheteur, C. Plasmonic Fiber Grating Biosensors Demodulated through Spectral Envelopes Intersection. J. Light. Technol. 2021, 39, 7288–7295. [Google Scholar] [CrossRef]
- Rivero, P.J.; Goicoechea, J.; Arregui, F.J. Layer-by-Layer Nano-assembly: A Powerful Tool for Optical Fiber Sensing Applications. Sensors 2019, 19, 683. [Google Scholar] [CrossRef] [PubMed]
- Bekmurzayeva, A.; Ashikbayeva, Z.; Myrkhiyeva, Z.; Nugmanova, A.; Shaimerdenova, M.; Ayupova, T.; Tosi, D. Label-free fiber-optic spherical tip biosensor to enable picomolar-level detection of CD44 protein. Sci. Rep. 2021, 11, 19583. [Google Scholar] [CrossRef]
- Bekmurzayeva, A.; Ashikbayeva, Z.; Assylbekova, N.; Myrkhiyeva, Z.; Dauletova, A.; Ayupova, T.; Shaimerdenova, M.; Tosi, D. Ultra-wide, attomolar-level limit detection of CD44 biomarker with a silanized optical fiber biosensor. Biosens. Bioelectron. 2022, 208, 114217. [Google Scholar] [CrossRef]
- Nedoma, J.; Krizan, D.; Stipal, J.; Pereira, L.; Bekmurzayeva, A.; Tosi, D.; Costa, M.B.; Leal-Junior, A.; Martinek, R.; Marques, C. Decade of advancements in light–matter interaction-based optical fiber biosensing: Innovations, challenges, and future directions. Adv. Photonics 2025, 8, 14004. [Google Scholar] [CrossRef]
- Shen, C.; Chen, X.; Huang, Z.; Wang, Z.; Liu, J.; Deng, H.; Liu, D.; Shu, F. High sensitivity and fast response optical fiber nucleic acid sensor. Opt. Laser Technol. 2022, 154, 108271. [Google Scholar] [CrossRef]
- Spence, J. Chemical Safety-Special Hazards Safe Work Procedure (SWP-009), Piranha Solution, Occupational Health, Safety and Environment; University of Victoria: Victoria, BC, Canada, 2022. [Google Scholar]
- Schmidt, H.G. Safe Piranhas: A Review of Methods and Protocols. ACS Chem. Health Saf. 2021, 29, 54–61. [Google Scholar] [CrossRef]
- Kumar, S.; Kaushik, B.K.; Singh, R.; Chen, N.-K.; Yang, Q.S.; Zhang, X.; Wang, W.; Zhang, B. LSPR-based cholesterol biosensor using a tapered optical fiber structure. Biomed. Opt. Express 2019, 10, 2150–2160. [Google Scholar] [CrossRef]
- Zhou, L.; Liu, C.; Sun, Z.; Mao, H.; Zhang, L.; Yu, X.; Zhao, J.; Chen, X. Black phosphorus based fiber optic biosensor for ultrasensitive cancer diagnosis. Biosens. Bioelectron. 2019, 137, 140–147. [Google Scholar] [CrossRef]
- Paltusheva, Z.U.; Ashikbayeva, Z.; Tosi, D.; Gritsenko, L.V. Highly Sensitive Zinc Oxide Fiber-Optic Biosensor for the Detection of CD44 Protein. Biosensors 2022, 12, 1015. [Google Scholar] [CrossRef]
- Loyez, M.; Lobry, M.; Hassan, E.M.; DeRosa, M.C.; Caucheteur, C.; Wattiez, R. HER2 breast cancer biomarker detection using a sandwich optical fiber assay. Talanta 2021, 221, 121452. [Google Scholar] [CrossRef]
- Rostami, S.; Zibaii, M.I.; Babakhani-Fard, M.-M.; Layeghi, A.; Latifi, H. Sensitivity enhancement and thermal compensation of LSPR-based optical fibre refractive index sensor using annealing of Au nanoparticles. Sens. Actuators A Phys. 2025, 395, 117015. [Google Scholar] [CrossRef]
- Verding, P.; Joy, R.M.; Reenaers, D.; Kumar, R.S.N.; Rouzbahani, R.; Jeunen, E.; Thomas, S.; Desta, D.; Boyen, H.-G.; Pobedinskas, P.; et al. The Influence of UV–Ozone, O2 Plasma, and CF4 Plasma Treatment on the Droplet-Based Deposition of Diamond Nanoparticles. ACS Appl. Mater. Interfaces 2023, 16, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; O’Hare, D.; Ladame, S. Surface immobilization strategies for the development of electrochemical nucleic acid sensors. Biosens. Bioelectron. 2023, 237, 115440. [Google Scholar] [CrossRef] [PubMed]
- Nurlankyzy, M.; Kantoreyeva, K.; Myrkhiyeva, Z.; Ashikbayeva, Z.; Baiken, Y.; Kanayeva, D.; Tosi, D.; Bekmurzayeva, A. Label-free optical fiber biosensor for the detection of CD44-expressing breast cancer cells. Sens. BioSens. Res. 2024, 44, 100661. [Google Scholar] [CrossRef]
- Sypabekova, M.; Amantayeva, A.; Vangelista, L.; González-Vila, Á.; Caucheteur, C.; Tosi, D. Ultralow Limit Detection of Soluble HER2 Biomarker in Serum with a Fiber-Optic Ball-Tip Resonator Assisted by a Tilted FBG. ACS Meas. Sci. Au 2022, 2, 309–316. [Google Scholar] [CrossRef]
- Murugan, D.; Tintelott, M.; Amiri, H.; Kasavetov, M.; Besedin, D.; Ingebrandt, S.; Vu, X.T.; Pachauri, V. Comparative Study of Surface Activation Steps for Thermally Grown Oxide Interface and Optimal Silanization. Phys. Status Solidi (A) 2023, 220, 2300294. [Google Scholar] [CrossRef]
- Arghir, I.; Spasic, D.; Verlinden, B.E.; Delport, F.; Lammertyn, J. Improved surface plasmon resonance biosensing using silanized optical fibers. Sens. Actuators B Chem. 2015, 216, 518–526. [Google Scholar] [CrossRef]
- Kim, H.-M.; Uh, M.; Jeong, D.H.; Lee, H.-Y.; Park, J.-H.; Lee, S.-K. Localized surface plasmon resonance biosensor using nanopatterned gold particles on the surface of an optical fiber. Sens. Actuators B Chem. 2019, 280, 183–191. [Google Scholar] [CrossRef]
- Li, X.; Wang, Z.; Hu, L.; Xia, J.; Li, S.; Kuai, Y.; Fu, W.; Cao, Z.; Wang, M.; Yu, B.; et al. Rapid and Sensitive Detection of Liver Cancer Markers Based on a Microtapered Long-Period Grating Sensor Coated with Gold Nanorods. IEEE Sens. J. 2025, 25, 26608–26615. [Google Scholar] [CrossRef]
- Boccafoschi, F.; Fusaro, L.; Cannas, M. Immobilization of peptides on cardiovascular stent. In Functionalised Cardiovascular Stents; Elsevier: UK, Cambridge, 2018; pp. 305–318. [Google Scholar]
- Qu, W.; Chen, Y.; Liu, S.; Luo, L. Advances and Prospects of Nanomaterial Coatings in Optical Fiber Sensors. Coatings 2025, 15, 1008. [Google Scholar] [CrossRef]
- Singh, M.; Kaur, N.; Comini, E. The role of self-assembled monolayers in electronic devices. J. Mater. Chem. C 2020, 8, 3938–3955. [Google Scholar] [CrossRef]
- Zhu, S.; Xie, Z.; Chen, Y.; Liu, S.; Kwan, Y.-W.; Zeng, S.; Yuan, W.; Ho, H.-P. Real-Time Detection of Circulating Tumor Cells in Bloodstream Using Plasmonic Fiber Sensors. Biosensors 2022, 12, 968. [Google Scholar] [CrossRef] [PubMed]
- Mekuye, B.; Abera, B. Nanomaterials: An overview of synthesis, classification, characterization, and applications. Nano Sel. 2023, 4, 486–501. [Google Scholar] [CrossRef]
- Adul-Rasool, A.A.; Athair, D.M.; Zaidan, H.K.; Rheima, A.M.; Al-Sharify, Z.T.; Mohammed, S.H.; Kianfar, E. 0,1,2,3D nanostructures, types of bulk nanostructured materials, and drug nanocrystals: An overview. Cancer Treat. Res. Commun. 2024, 40, 100834. [Google Scholar] [CrossRef] [PubMed]
- Baig, N.; Kammakakam, I.; Falath, W. Nanomaterials: A review of synthesis methods, properties, recent progress, and challenges. Mater. Adv. 2021, 2, 1821–1871. [Google Scholar] [CrossRef]
- Gupta, R.; Prakash, N.; Paul, D.; Mukherji, S. Anti-nucleolin aptamer mediated specific detection of cancer cells by Localized Surface Plasmon Resonance-based U-bent optical fiber. Biosens. Bioelectron. X 2023, 13, 100318. [Google Scholar] [CrossRef]
- Liang, G.; Zhao, Z.; Wei, Y.; Liu, K.; Hou, W.; Duan, Y. Plasma enhanced label-free immunoassay for alpha-fetoprotein based on a U-bend fiber-optic LSPR biosensor. RSC Adv. 2015, 5, 23990–23998. [Google Scholar] [CrossRef]
- Kim, H.-M.; Yang, S.-C.; Park, J.-H.; Lee, S.-K. Fabrication of Top–Down-Based Optical Fiber Nanoprobes and Their Diagnostic Application for Pancreatic Cancer. IEEE Sens. J. 2024, 24, 11966–11973. [Google Scholar] [CrossRef]
- Chen, X.; Xu, P.; Lin, W.; Jiang, J.; Qu, H.; Hu, X.; Sun, J.; Cui, Y. Label-free detection of breast cancer cells using a functionalized tilted fiber grating. Biomed. Opt. Express 2022, 13, 2117–2129. [Google Scholar] [CrossRef]
- Zamri, A.; Mustafa, M.; Awang, N.; Zalkepali, N.; Mahmud, N.; Muhammad, N. Fiber-laser based on D-shaped fiber biosensor for prostate cancer biomarker detection. Mater. Today Proc. 2024, in press. [Google Scholar] [CrossRef]
- Liang, M.; Li, X.; Chen, Y. SPR-based fiber optic biosensor for the detection of HER2. In Proceedings of the 2024 International Conference on Optoelectronic Information and Optical Engineering (OIOE 2024), Kunming, China, 8–10 March 2024; pp. 307–313. [Google Scholar]
- Yildizhan, Y.; Driessens, K.; Tsao, H.S.K.; Boiy, R.; Thomas, D.; Geukens, N.; Hendrix, A.; Lammertyn, J.; Spasic, D. Detection of Breast Cancer-Specific Extracellular Vesicles with Fiber-Optic SPR Biosensor. Int. J. Mol. Sci. 2023, 24, 3764. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Cheng, K.; Chen, S.; Wei, S.; Ding, L.; Yang, L.; Che, T.; Wang, N. Early Detection and Screening of Bladder Cancer Based on a Novel Optical Fiber Surface Plasmon Resonance Sensor. IEEE Sens. J. 2025, 25, 24030–24037. [Google Scholar] [CrossRef]
- Tosi, D.; Ashikbayeva, Z.; Bekmurzayeva, A.; Myrkhiyeva, Z.; Rakhimbekova, A.; Ayupova, T.; Shaimerdenova, M. Optical fiber ball resonator sensor spectral interrogation through undersampled klt: Application to refractive index sensing and cancer biomarker biosensing. Sensors 2021, 21, 6721. [Google Scholar] [CrossRef]
- Lobry, M.; Loyez, M.; Debliquy, M.; Chah, K.; Goormaghtigh, E.; Caucheteur, C. Electro-plasmonic-assisted biosensing of proteins and cells at the surface of optical fiber. Biosens. Bioelectron. 2022, 220, 114867. [Google Scholar] [CrossRef]
- Ashikbayeva, Z.; Bekmurzayeva, A.; Myrkhiyeva, Z.; Assylbekova, N.; Atabaev, T.S.; Tosi, D. Green-synthesized gold nanoparticle-based optical fiber ball resonator biosensor for cancer biomarker detection. Opt. Laser Technol. 2023, 161, 109136. [Google Scholar] [CrossRef]
- Li, J.; Wang, H.; Li, Z.; Su, Z.; Zhu, Y. Preparation and application of metal nanoparticals elaborated fiber sensors. Sensors 2020, 20, 5155. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Kim, K.Y.; Lee, S.H.; Kim, K.K.; Lee, K.; Lee, W.; Jeon, H.; Ko, S.H. Recent Advances in 1D Nanomaterial-Based Bioelectronics for Healthcare Applications. Adv. NanoBiomed Res. 2021, 2, 2100111. [Google Scholar] [CrossRef]
- Liang, H.; Zhou, L.; Chen, P.; Zheng, J.; Huang, Y.; Liang, J.; Zhong, J.; Huang, Y.; Yu, M.; Guan, B.-O. Optical Microfiber with a Gold Nanorods–Black Phosphorous Nanointerface: An Ultrasensitive Biosensor and Nanotherapy Platform. Anal. Chem. 2022, 94, 8058–8065. [Google Scholar] [CrossRef]
- Lutomia, D.; Poria, R.; Kala, D.; Garg, P.; Nagraik, R.; Kaushal, A.; Gupta, S.; Kumar, D. 2D nanomaterials in biosensing: Synthesis, characterization, integration in biosensors and their applications. Biosens. Bioelectron. X 2025, 24, 100615. [Google Scholar] [CrossRef]
- Yang, W.; Jiang, M.; Jiang, S.; Du, L.; Cheng, Y.; Li, P.; Wang, C. Design and fabrication of Gr/Ag-coated tilted grating sensor for ultra-sensitive detection of DNA hybridization. Sens. Actuators B Chem. 2022, 359, 131587. [Google Scholar] [CrossRef]
- Qiu, H.; Yao, Y.; Dong, Y.; Tian, J. Fiber-optic immunosensor based on a Fabry–Perot interferometer for single-molecule detection of biomarkers. Biosens. Bioelectron. 2024, 255, 116265. [Google Scholar] [CrossRef]
- Sun, J.; Jiang, H.; Chavan, K.J.; Coutts, A.S.; Chen, X. Graphene Oxide-Functionalized Optical Sensor for Label-Free Detection of Breast Cancer Cells. ACS Appl. Nano Mater. 2025, 8, 16770–16778. [Google Scholar] [CrossRef]
- Deng, X.; Peng, Y.; Long, R. Microfiber biosensor for detection of ultra-low concentration alpha-fetoprotein based on graphene oxide. Opt. Fiber Technol. 2025, 93, 104229. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, L.; Qiao, D.; Liu, M.; Yang, H.; Meng, C.; Miao, T.; Xue, J.; Yao, Y. Progress on Optical Fiber Biochemical Sensors Based on Graphene. Micromachines 2022, 13, 348. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, J.; Kim, S.; Min, D.-H. Biosensors based on graphene oxide and its biomedical application. Adv. Drug Deliv. Rev. 2016, 105, 275–287. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.; Zhang, J.; Li, S. Molybdenum Disulfide as Tunable Electrochemical and Optical Biosensing Platforms for Cancer Biomarker Detection: A Review. Biosensors 2023, 13, 848. [Google Scholar] [CrossRef]
- Ye, L.; Gan, X.; Schirhagl, R. Two-Dimensional MoS2-Based Photodetectors. Sustainability 2024, 16, 10137. [Google Scholar] [CrossRef]
- Sinha, A.; Dhanjai; Tan, B.; Huang, Y.; Zhao, H.; Dang, X.; Chen, J.; Jain, R. MoS2 nanostructures for electrochemical sensing of multidisciplinary targets: A review. TrAC Trends Anal. Chem. 2018, 102, 75–90. [Google Scholar] [CrossRef]
- Kaur, B.; Kumar, S.; Kaushik, B.K. 2D Materials-Based Fiber Optic SPR Biosensor for Cancer Detection at 1550 nm. IEEE Sens. J. 2021, 21, 23957–23964. [Google Scholar] [CrossRef]
- Huraiya, A.; Shoshi, M.S.; Chakrabarti, K.; Tabata, H.; Ramaraj, S.G.; Razzak, S.M.A.; Eid, M.M.A.; Rashed, A.N.Z. Ultra-Sensitive Au-based Circular Photonic Fibers Based Surface Plasmonic Resonance Biosensor for Various Cancer Level Diagnostics and Detection. Plasmonics 2025, 20, 8879–8892. [Google Scholar] [CrossRef]
- Li, H.; Huang, T.; Yuan, H.; Lu, L.; Cao, Z.; Zhang, L.; Yang, Y.; Yu, B.; Wang, H. Combined Ultrasensitive Detection of Renal Cancer Proteins and Cells Using an Optical Microfiber Functionalized with Ti3C2 MXene and Gold Nanorod-Nanosensitized Interfaces. Anal. Chem. 2023, 95, 5142–5150. [Google Scholar] [CrossRef]
- Kumar, A.; Verma, P.; Jindal, P. Surface plasmon resonance sensor based on MXene coated PCF for detecting the cancer cells with machine learning approach. Microelectron. Eng. 2022, 267–268, 111897. [Google Scholar] [CrossRef]
- Lu, M.; He, Y.; Xi, S.; Zhong, P.; Zhang, Y.; Tian, H.; Wang, Y.; Lu, H.; Hu, J.; Tang, J. High-Sensitivity MXene-Functionalized Photonic Crystal Fiber Surface Plasmon Resonance Sensor with Dual Rectangular Grooves for Cancer Detection. Sensors 2025, 25, 5705. [Google Scholar] [CrossRef]
- Thawany, P.; Khanna, A.; Tiwari, U.K.; Deep, A. L-cysteine/MoS2 modified robust surface plasmon resonance optical fiber sensor for sensing of Ferritin and IgG. Sci. Rep. 2023, 13, 5297. [Google Scholar] [CrossRef] [PubMed]
- Cennamo, N.; Pasquardini, L.; Arcadio, F.; Vanzetti, L.E.; Bossi, A.M.; Zeni, L. D-shaped plastic optical fibre aptasensor for fast thrombin detection in nanomolar range. Sci. Rep. 2019, 9, 18740. [Google Scholar] [CrossRef]
- Li, X.; Gong, P.; Zhao, Q.; Zhou, X.; Zhang, Y.; Zhao, Y. Plug-in optical fiber SPR biosensor for lung cancer gene detection with temperature and pH compensation. Sens. Actuators B Chem. 2022, 359, 131596. [Google Scholar] [CrossRef]
- Kong, X.; Li, M.; Xiao, W.; Li, Y.; Luo, Z.; Shen, J.-W.; Duan, Y. Ω-Shaped fiber optic LSPR coated with hybridized nanolayers for tumor cell sensing and photothermal treatment. Talanta 2024, 278, 126381. [Google Scholar] [CrossRef]
- Wei, Y.; Zhou, W.; Wu, Y.; Zhu, H. High Sensitivity Label-Free Quantitative Method for Detecting Tumor Biomarkers in Human Serum by Optical Microfiber Couplers. ACS Sens. 2021, 6, 4304–4314. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liu, X.; Sun, H.; Xi, J.; Chang, C.; Deng, L.; Yang, Y.; Li, X. Optical fiber sensing probe for detecting a carcinoembryonic antigen using a composite sensitive film of PAN nanofiber membrane and gold nanomembrane. Opt. Express 2024, 32, 20024–20034. [Google Scholar] [CrossRef]
- Xiao, A.; Huang, Y.; Zheng, J.; Chen, P.; Guan, B.-O. An Optical Microfiber Biosensor for CEACAM5 Detection in Serum: Sensitization by a Nanosphere Interface. ACS Appl. Mater. Interfaces 2019, 12, 1799–1805. [Google Scholar] [CrossRef]
- Hu, J.; He, P.; Zhao, F.; Lin, W.; Xue, C.; Chen, J.; Yu, Z.; Ran, Y.; Meng, Y.; Hong, X.; et al. Magnetic microspheres enhanced peanut structure cascaded lasso shaped fiber laser biosensor for cancer marker-CEACAM5 detection in serum. Talanta 2024, 271, 125625. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, J.; Han, A.; Zhang, X.; Yang, M.; Li, H.; Yu, B.; Zhang, G.; Zhou, S. Ultrasensitive cancer cell sensing based on tapered optical fiber operating near the dispersion turning point. Sens. Actuators B Chem. 2024, 407, 135473. [Google Scholar] [CrossRef]
- Shaimerdenova, M.; Ayupova, T.; Ashikbayeva, Z.; Bekmurzayeva, A.; Blanc, W.; Tosi, D. Reflector-Less Shallow-Tapered Optical Fiber Biosensors for Rapid Detection of Cancer Biomarkers. J. Light. Technol. 2023, 41, 4114–4122. [Google Scholar] [CrossRef]
- Myrkhiyeva, Z.; Kantoreyeva, K.; Bekmurzayeva, A.; Gomez, A.W.; Ashikbayeva, Z.; Tilegen, M.; Pham, T.T.; Tosi, D. Dynamic Measurement of a Cancer Biomarker: Towards In Situ Application of a Fiber-Optic Ball Resonator Biosensor in CD44 Protein Detection. Sensors 2024, 24, 1991. [Google Scholar] [CrossRef]
- Yegizbay, Z.; Fatima, M.; Bekmurzayeva, A.; Ashikbayeva, Z.; Tosi, D.; Blanc, W. Label-Free and Protein G-Enhanced Optical Fiber Biosensor for Detection of ALDH1A1 Cancer Biomarker. Fibers 2025, 13, 131. [Google Scholar] [CrossRef]
- Morales, M.A.; Halpern, J.M. Guide to Selecting a Biorecognition Element for Biosensors. Bioconjugate Chem. 2018, 29, 3231–3239. [Google Scholar] [CrossRef]
- Barbosa, O.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Rodrigues, R.C.; Fernandez-Lafuente, R. Glutaraldehyde in bio-catalysts design: A useful crosslinker and a versatile tool in enzyme immobilization. RSC Adv. 2014, 4, 1583–1600. [Google Scholar] [CrossRef]
- Yang, C. Enhanced physicochemical properties of collagen by using EDC/NHS-crosslinking. Bull. Mater. Sci. 2012, 35, 913–918. [Google Scholar] [CrossRef]
- Sequeira-Antunes, B.; Ferreira, H.A. Nucleic Acid Aptamer-Based Biosensors: A Review. Biomedicines 2023, 11, 3201. [Google Scholar] [CrossRef]
- Zhao, Y.; Xin, H.; Wang, C. Biomarker Multiplexing with Rational Design of Nucleic Acid Probe Complex. Anal. Sens. 2024, 4, e202400009. [Google Scholar] [CrossRef]
- Liberti, M.V.; Locasale, J.W. The Warburg Effect: How Does it Benefit Cancer Cells? Trends Biochem. Sci. 2016, 41, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Pajak, B.; Siwiak, E.; Sołtyka, M.; Priebe, A.; Zieliński, R.; Fokt, I.; Ziemniak, M.; Jaśkiewicz, A.; Borowski, R.; Domoradzki, T.; et al. 2-Deoxy-D-Glucose and its analogs: From diagnostic to therapeutic agents. Int. J. Mol. Sci. 2019, 21, 234. [Google Scholar] [CrossRef]
- Cesewski, E.; Johnson, B.N. Electrochemical biosensors for pathogen detection. Biosens. Bioelectron. 2020, 159, 112214. [Google Scholar] [CrossRef] [PubMed]
- Leibl, N.; Haupt, K.; Gonzato, C.; Duma, L. Molecularly Imprinted Polymers for Chemical Sensing: A Tutorial Review. Chemosensors 2021, 9, 123. [Google Scholar] [CrossRef]
- Yang, W.; Ma, Y.; Sun, H.; Huang, C.; Shen, X. Molecularly imprinted polymers based optical fiber sensors: A review. TrAC Trends Anal. Chem. 2022, 152, 116608. [Google Scholar] [CrossRef]
- Chen, X.; Noy, A. Antifouling strategies for protecting bioelectronic devices. APL Mater. 2021, 9, 020701. [Google Scholar] [CrossRef]
- Gundagatti, S.; Srivastava, S. An optimization of blocking agents for designing reliable electrochemical biosensors for ovarian cancer. Mater. Today Proc. 2023, in press. [Google Scholar] [CrossRef]
- Almeida, L.C.; Frade, T.; Correia, R.D.; Niu, Y.; Jin, G.; Correia, J.P.; Viana, A.S. Electrosynthesis of polydopamine-ethanolamine films for the development of immunosensing interfaces. Sci. Rep. 2021, 11, 2237. [Google Scholar] [CrossRef]
- Chiang, C.-Y.; Chen, C.-H.; Wu, C.-W. Fiber Optic Localized Surface Plasmon Resonance Sensor Based on Carboxymethylated Dextran Modified Gold Nanoparticles Surface for High Mobility Group Box 1 (HMGB1) Analysis. Biosensors 2023, 13, 522. [Google Scholar] [CrossRef]
- Ucci, S.; Spaziani, S.; Quero, G.; Vaiano, P.; Principe, M.; Micco, A.; Sandomenico, A.; Ruvo, M.; Consales, M.; Cusano, A. Advanced Lab-on-Fiber Optrodes Assisted by Oriented Antibody Immobilization Strategy. Biosensors 2022, 12, 1040. [Google Scholar] [CrossRef]
- Hasler, R.; Vísová, I.; Vrabcová, M.; Houska, M.; Spasovová, M.; Lísalová, H.; Dostálek, J. Fiber optic probe with antifouling polymer brush biointerface for bi-modal biosensing in complex liquid samples. In Biophotonics in Point-of-Care II; Canva, M.T., Giannetti, A., Altug, H., Moreau, J., Eds.; SPIE: Bellingham, WA, USA, 2022; pp. 31–41. [Google Scholar]
- Qian, H.; Huang, Y.; Duan, X.; Wei, X.; Fan, Y.; Gan, D.; Yue, S.; Cheng, W.; Chen, T. Fiber optic surface plasmon resonance biosensor for detection of PDGF-BB in serum based on self-assembled aptamer and antifouling peptide monolayer. Biosens. Bioelectron. 2019, 140, 90–95. [Google Scholar] [CrossRef]
- D’agata, R.; Bellassai, N.; Jungbluth, V.; Spoto, G. Recent Advances in Antifouling Materials for Surface Plasmon Resonance Biosensing in Clinical Diagnostics and Food Safety. Polymers 2021, 13, 1929. [Google Scholar] [CrossRef]
- Völlmecke, K.; Afroz, R.; Bierbach, S.; Brenker, L.J.; Frücht, S.; Glass, A.; Giebelhaus, R.; Hoppe, A.; Kanemaru, K.; Lazarek, M.; et al. Hydrogel-Based Biosensors. Gels 2022, 8, 768. [Google Scholar] [CrossRef] [PubMed]
- Sitinjak, N.A.; Huang, C.-W.; Yang, T.-Y.; Chau, L.-K.; Wang, C.-H. Synthesis of Carboxymethyl Dextran-Coated Gold Nanoparticles as Stable and Storable Optical Labels for Ultrasensitive Plasmonic Nanoparticle-Linked Sorbent Assay. Sensors 2025, 25, 7156. [Google Scholar] [CrossRef]
- Mortazavi, S.; Makouei, S.; Abbasian, K.; Danishvar, S. Emerging Trends in Optical Fiber Biosensing for Non-Invasive Biomedical Analysis. Photonics 2025, 12, 1202. [Google Scholar] [CrossRef]
- Malakar, T.; Amina, M.N.N.; Nijhum, Z.T.; Lalin, N.S. Optimized Single-Core PCF-Based SPR Biosensor for High-Performance Early-Stage Multi-Cancer Detection. arXiv 2025, arXiv:2510.17283. [Google Scholar]
- Chaudhary, V.S.; Kumar, D.; Kumar, S. Au-TiO2 Coated Photonic Crystal Fiber Based SPR Refractometric Sensor for Detection of Cancerous Cells. IEEE Trans. Nanobiosci. 2022, 22, 562–569. [Google Scholar] [CrossRef]
- Hu, J.; Song, E.; Liu, Y.; Yang, Q.; Sun, J.; Chen, J.; Meng, Y.; Jia, Y.; Yu, Z.; Ran, Y.; et al. Fiber Laser-Based Lasso-Shaped Biosensor for High Precision Detection of Cancer Biomarker-CEACAM5 in Serum. Biosensors 2023, 13, 674. [Google Scholar] [CrossRef] [PubMed]
- Khani, S.; Hayati, M. Optical biosensors using plasmonic and photonic crystal band-gap structures for the detection of basal cell cancer. Sci. Rep. 2022, 12, 5246. [Google Scholar] [CrossRef] [PubMed]
- Chaity, A.C. Highly Sensitive Photonic Crystal Fiber Biosensor Based on Surface Plasmon Resonance for Six Distinct Types of Cancer Detection. Plasmonics 2023, 19, 1891–1902. [Google Scholar] [CrossRef]
- Dashtmian, K.; Reza, M.; Fallahi, V.; Seifouri, M.; Olyaee, S. Gold Nanowire-Enhanced SPR-PCF Biosensor for High-Throughput Cancer Cell Detection in Near-Infrared. Plasmonics 2025, 20, 11853–11865. [Google Scholar] [CrossRef]
- Hossain, B.; Akib, T.B.A.; Abdulrazak, L.F.; Rana, M. Numerical modeling of graphene-coated fiber optic surface plasmon resonance biosensor for BRCA1 and BRCA2 genetic breast cancer detection. Opt. Eng. 2019, 58, 037104. [Google Scholar] [CrossRef]
- Verma, P.; Kumar, A.; Jindal, P. Machine Learning Approach for SPR based Photonic Crystal Fiber Sensor for Breast Cancer Cells Detection. In Proceedings of the 2022 IEEE 7th Forum on Research and Technologies for Society and Industry Innovation (RTSI), Paris, France, 24–26 August 2022; Institute of Electrical and Electronics Engineers Inc.: New York, NY, USA, 2022; pp. 7–12. [Google Scholar]
- Sobur, T.R.; Hasan, M.; Khatun, M.R.; Mia, M.; Islam, W.; Iqbal, S.; Hossain, M.M. Machine learning-enhanced SPR-based optical biosensor for cancer cell detection. Opt. Laser Technol. 2025, 192, 113796. [Google Scholar] [CrossRef]
- Wekalao, J.; Li, M.; Zhang, F.; Zhang, X.; Liu, W. Machine learning-enhanced Kretschmann configuration plasmonic biosensor for real-time brain tumor biomarker detection. Surf. Interfaces 2025, 75, 107774. [Google Scholar] [CrossRef]
- Ashrafian, M.; Olyaee, S.; Seifouri, M. Highly sensitive cancer detection using an open D-channel PCF-based SPR biosensor. Sci. Rep. 2025, 15, 10168. [Google Scholar] [CrossRef]
- Zhang, Y.; Miao, T.; Mu, Q.; Zhou, L.; Meng, C.; Xue, J.; Yao, Y. A Novel High-Sensitivity Terahertz Microstructure Fiber Biosensor for Detecting Cancer Cells. Photonics 2022, 9, 639. [Google Scholar] [CrossRef]
- Lv, J.; Wang, J.; Yang, L.; Liu, W.; Fu, H.; Chu, P.K.; Liu, C. Recent advances of optical fiber biosensors based on surface plasmon resonance: Sensing principles, structures, and prospects. Sens. Diagn. 2024, 3, 1369–1391. [Google Scholar] [CrossRef]
- Bissen, A.; Yunussova, N.; Myrkhiyeva, Z.; Salken, A.; Tosi, D.; Bekmurzayeva, A. Unpacking the packaged optical fiber bio-sensors: Understanding the obstacle for biomedical application. Front. Bioeng. Biotechnol. 2024, 12, 1401613. [Google Scholar] [CrossRef]
- SDS Optic S.A. Starts Collaboration with Accrea Medical Robotics sp. Zoo and MedApp S.A. Available online: https://www.sdsoptic.pl/ (accessed on 23 December 2025).
- Detection of Breast Cancer by Fluorescence Using an Optical Fiber Needle. Available online: https://www.sedi-ati.com/business_cases/detection-of-breast-cancer-by-fluorescence-using-an-optical-fiber-needle/ (accessed on 23 December 2025).
- Zhu, G.; Zhang, M.; Lu, L.; Lou, X.; Dong, M.; Zhu, L. Metal-organic framework/enzyme coated optical fibers as waveguide-based biosensors. Sens. Actuators B Chem. 2019, 288, 12–19. [Google Scholar] [CrossRef]
- Karimian, S.; Ali, M.M.; McAfee, M.; Saleem, W.; Duraibabu, D.; Memon, S.F.; Lewis, E. Challenges in Adapting Fibre Optic Sensors for Biomedical Applications. Biosensors 2025, 15, 312. [Google Scholar] [CrossRef] [PubMed]
| # | Fiber Type | Main Mechanism | Material | Biorecognition Element | Cancer Cell Type (s) | Sensitivity | LoD | Ref. |
|---|---|---|---|---|---|---|---|---|
| 1 | TFBG + SMF | Multiresonance light scattering sensing | Halloysite nanotubes (HNTs) | Physical cell capture via HNT slit-like nanostructures (no molecular affinity receptor) | Breast cancer | Not given | 10 cells·mL−1 | [27] |
| 2 | D-shaped polymer OF | SPR | Gold nanofilm + CYTOP polymer | HER2-specific aptamer | Breast cancer | 28,100 nm/RIU | ~5.28 nM | [38] |
| 3 | MCF+SMF | SPR | GO/AuNPs/CuO nanoflowers | 2-deoxy-D-glucose (2-DG), targeting GLUT receptors | Liver, lung, breast cancer | Not given | 3, 2, 2, 2, 4, 10 cells/mL | [39] |
| 4 | SMF ball resonator | Refractometric | Gold thin film | Anti-CD44 antibody | Breast, colon, gastric, lung, ovarian, cervical cancers | 95.76 dB·RIU−1 | 17 pM | [56] |
| 5 | SMF ball resonator | Refractometric | APTMS + Glutaraldehyde | Anti-CD44 antibody | Breast cancer | −85 to −120 dB·RIU−1 | 4.68 aM | [57] |
| 6 | SMF | Refractometric | APTES + glutaraldehyde | DNA probe | Not cancer cell-specific | 0.175 dB·nM−1 | 1 nM | [59] |
| 7 | TFG | Refractometric | Black phosphorus (BP) | Anti-neuron-specific enolase (anti-NSE) antibodies | Lung cancer | NSE | 1.0 pg/mL | [63] |
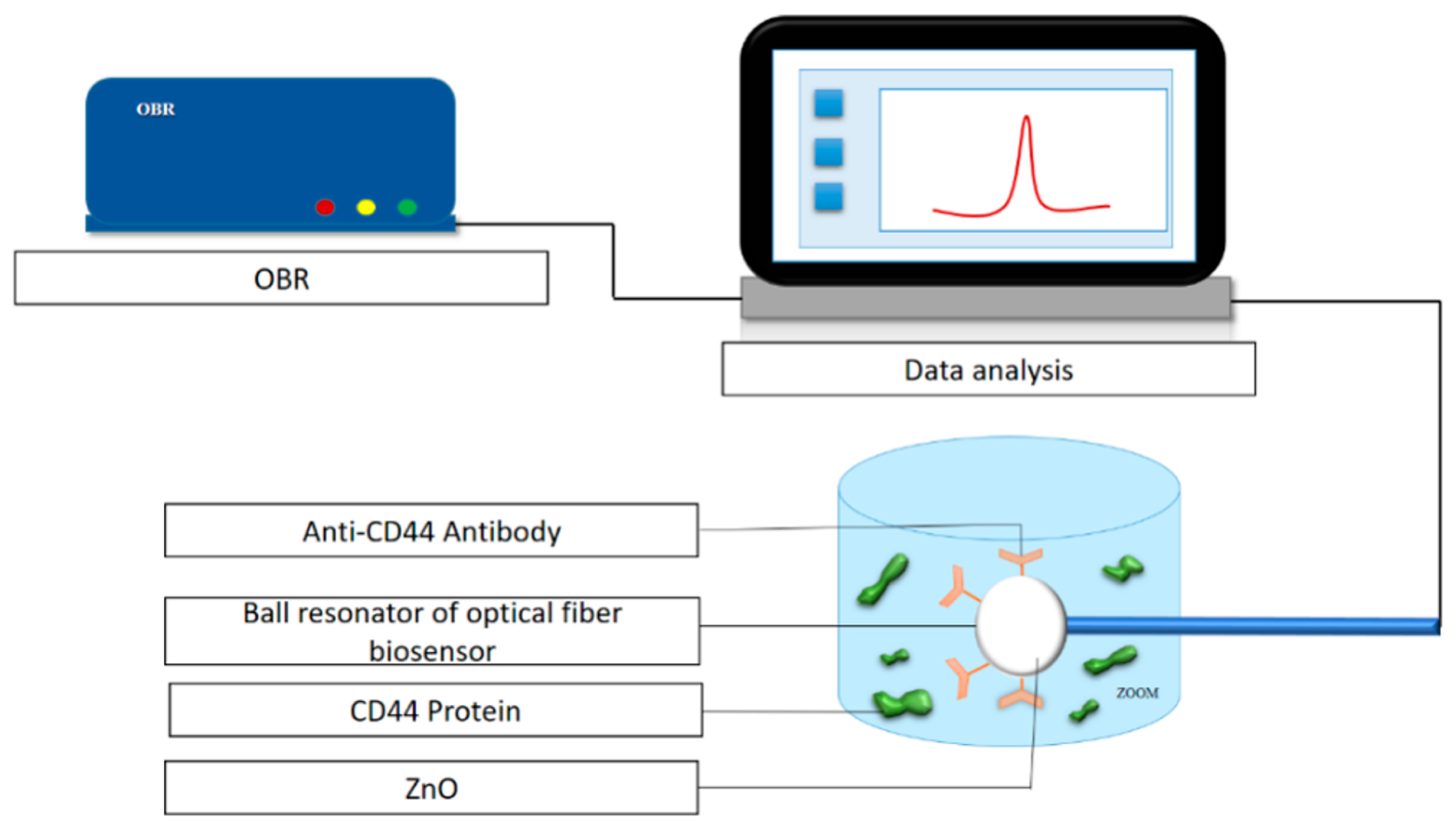
| 8 | SMF ball resonator | Refractometric | Zinc oxide | Anti-CD44 antibody | Breast cancer | −80.056 dB/RIU | 0.8 fM | [64] |
| 9 | Unclad MMF | SPR | Gold thin film | HER2 antibody | Breast cancer | Not given | 6.6 × 10−7 g/mL | [65] |
| 10 | SMF ball resonator | Refractometric | APTMS + Glutaraldehyde | Anti-CD44 antibody | Breast cancer | −92.1 dB·RIU−1 | 335 cells·mL−1 | [69] |
| 11 | TFBG + ball resonator | Refractometric | APTMS, glutaraldehyde | Anti-HER2 antibody | Breast cancer | 4034 dB/RIU | In buffer and in a 1/10 diluted serum of 151.5 ag/mL and 3.7 pg/mL, respectively | [70] |
| 12 | Microfiber | Refractometric | APTES + Glutaraldehyde | Anti-HER2 antibody | Breast cancer | 1867 nm·RIU−1 | Down to 2 ng mL−1 in serum | [53] |
| 13 | MMF | SPR | Gold nanodisk particles | Anti-PSA antibody | Prostate cancer | 5700 RIU−1 | 1.3 pg·mL−1 | [73] |
| 14 | mTLPG | SPR | Gold nanorods | Anti-AFP antibodies | Liver cancer | 3 pm/(ng/mL) | 17.6 ng/mL | [74] |
| 15 | MMF+SMF | SPR | Gold thin film | Anti-EpCAM antibody | Breast cancer | 1933.4 nm·RIU−1 | 1.4 cells·µL−1 | [78] |
| 16 | U-bent MMF | SPR | Gold nanoparticles | Anti-nucleolin DNA aptamer | Retinoblastoma, meningioma, breast, cervical, and colon cancer | Not given | 500 cells·mL−1 (bench); 50 cells·mL−1 (portable μSens system) | [82] |
| 17 | Multimode U-bend fiber | SPR | Gold nanoparticles | AFP antibody | Hepatocellular carcinoma | Not given | 0.85 and 3.3 ng·mL−1 for PBS nad human serum, respectively | [83] |
| 18 | MMF | SPR | Bottom-up gold nanospheres | Anti-CA19-9 antibody | Pancreatic cancer | Not given | 0.25 U/mL | [84] |
| 19 | TFBG | SPR | Gold thin film | GRP-30 antibody | Breast cancer | Not given | Single-cell level | [85] |
| 20 | D-shaped fiber | SPR | Gold thin film | Anti-hCG antibody | Prostate cancer | 0.2171 nm/(µg/mL) | Not given | [86] |
| 21 | Hetero-core fiber | SPR | Gold thin film | Anti-HER2 antibody | Breast cancer | 0.20666 nm/(μg/mL) | Not given | [87] |
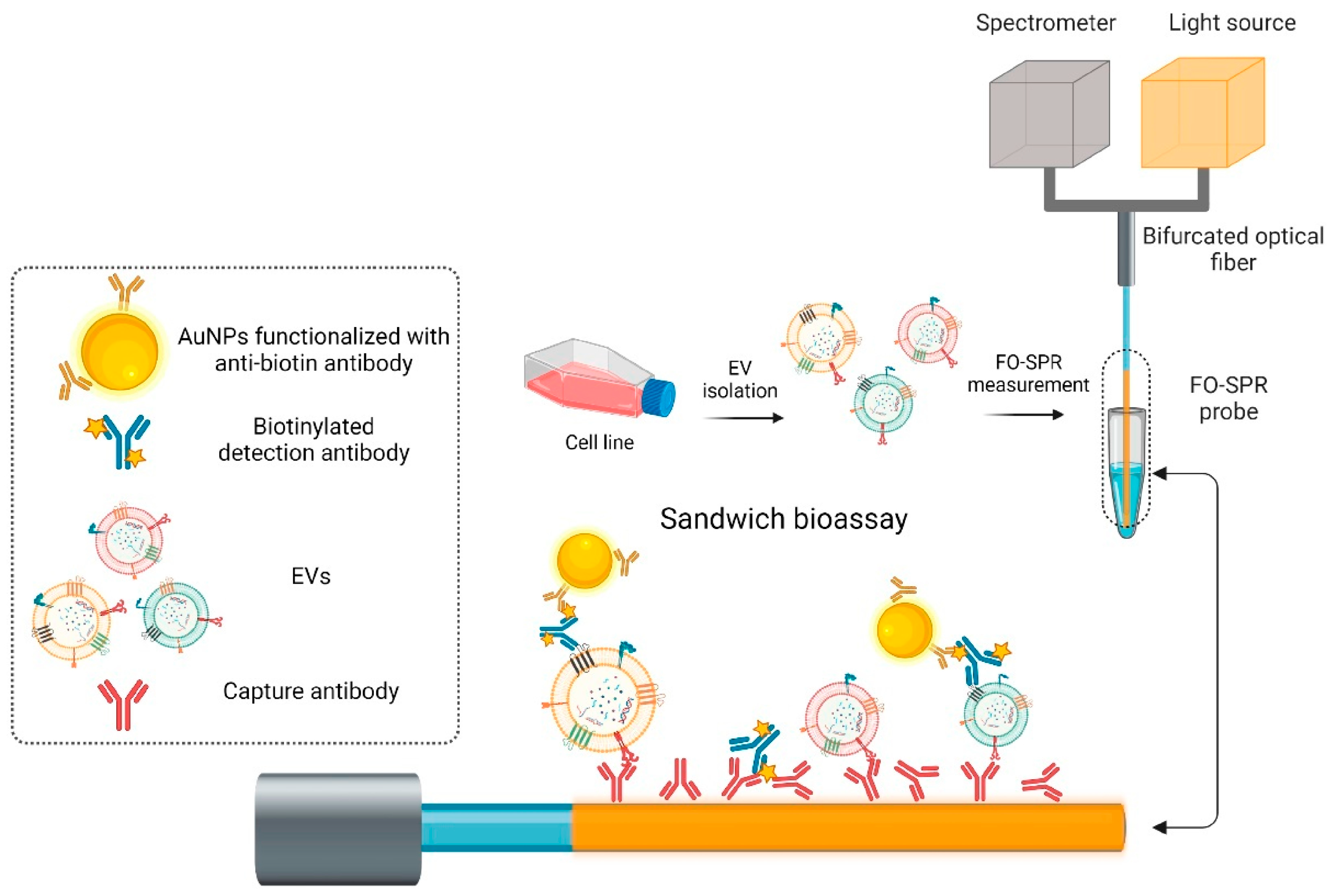
| 22 | MMF | SPR | Gold thin film | Capture antibodies: Anti-HER, anti-EpCAM; Detection antibodies: biotinylated anti-CD9/CD63/CD81 | Breast cancer | Not given | 2.1 × 107 particles/mL (SK-BR-3 EVs, buffer); 7 × 108 particles/mL (SK-BR-3 EVs, plasma); 1.1 × 108 particles/mL (MCF7 EVs, plasma) | [88] |
| 23 | Plastic-clad quartz optical fiber (HCPCF) | SPR | Au thin film | Anti-NMP22 antibody | Bladder cancer | 0.092 nm·(ng mL−1)−1 | 0.092 ng mL−1 | [89] |
| 24 | SMF ball resonator | Refractometric | Gold thin film | Anti-CD44 antibody | Breast cancer | 1594 RIU−1 | 19.7 pM | [90] |
| 25 | SMF + TFBG | SPR | Au thin film | Anti-HER2 aptamer | Breast cancer | ~0.8 nm·(µg mL−1)−1 (from ~800 pm shift at 10−6 g mL−1) | 10−12 g mL−1 (8.36 fM) | [91] |
| 26 | SMF ball resonator | Refractometric | Green-synthesized gold nanoparticles | Anti-CD44 antibody | Breast cancer | 1.52 dB per 10× concentration increase | 0.111 pM | [92] |
| 27 | Microfiber | SPR | Gold nanorods + Black Phosphorus | HER2 antibody | Breast cancer | 0.66 aM in buffer solution and 0.77 aM in 10% serum | Not given | [95] |
| 28 | D-shaped HCPCF | SPR | AuNP@GO | Anti-ProGRP antibody | Lung cancer | 583,000 nm/RIU | 17.1 ag/mL | [98] |
| 29 | LPG | Refractometric | Graphene oxide | Not applied | Breast cancer | Not given | 270 cells/mL | [99] |
| 30 | Microfiber | Refractometric | Graphene oxide | Anti-AFP antibody | Hepatocellular carcinoma | 1.11582 nm/lg (mol/L) | 78 zg/mL | [100] |
| 31 | Microfiber | SPR | Ti3C2 MXene/AuNR hybrid | CAIX-specific DNA aptamer | Renal cancer | Not given | 13.8 zM in pure buffer solution and 0.19 aM in 30% serum solution for CAIXproteins, 180 cells/mL for living cancer cells | [108] |
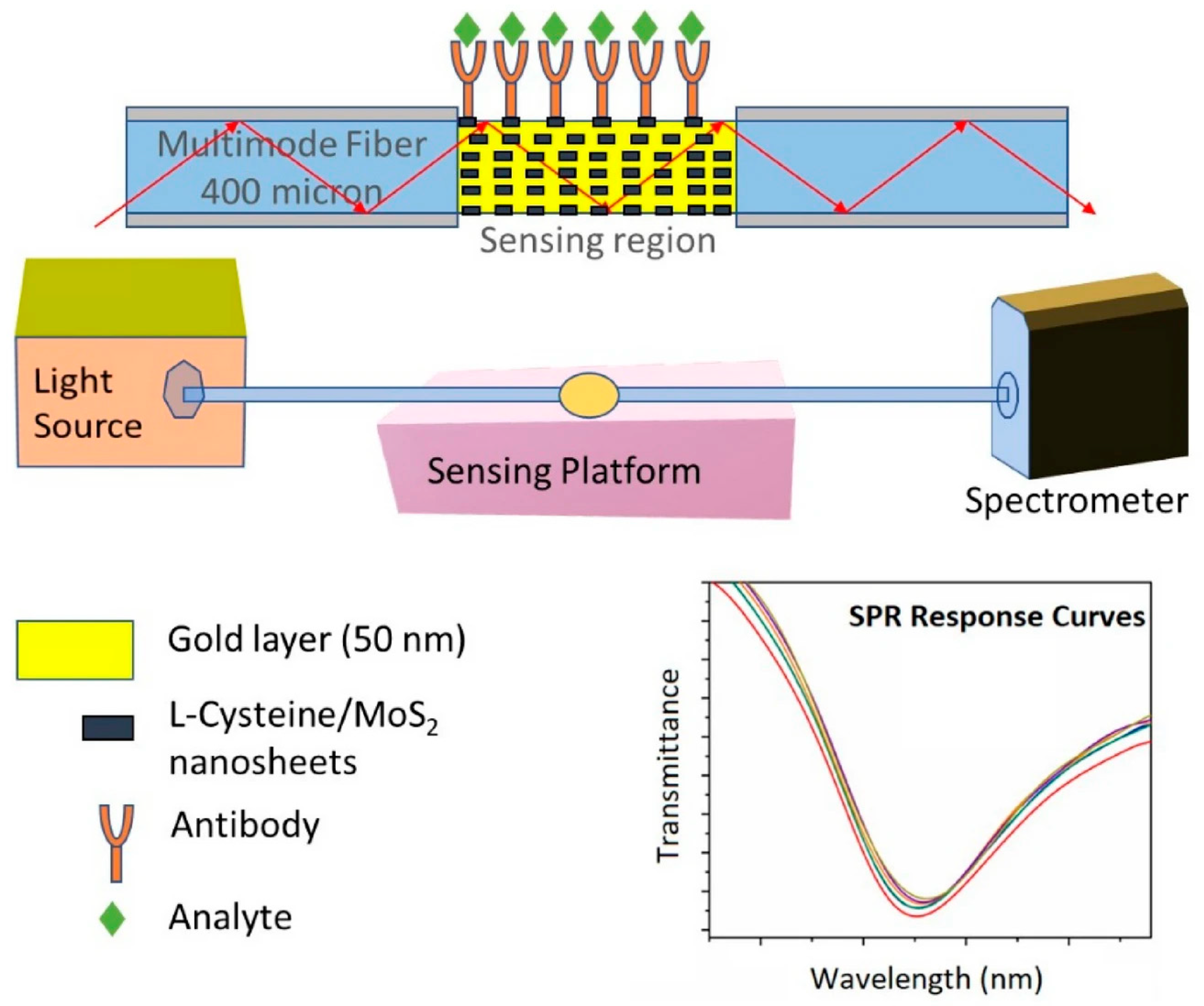
| 32 | D-shaped fiber | SPR | Au + L-cys/MoS2 | Anti-Ferritin antibody; Anti-IgG antibody | Not cancer-cell specific | 0.04 nm per µg/mL and 0.024 for IgG and ferritin, respectively | 12 ng/mL and 7.2 µg/mL for ferritin and IgG, respectively | [111] |
| 33 | FBG fused to MMF | SPR | Au + PAA/CS multilayernanofilm | Probe DNA | Lung cancer | 0.04 nm/nM | 13.5 nM | [113] |
| 34 | Ω-shaped fiber | SPR | AuNPs/AuNRs@PDA | MUC1 DNA aptamer | Breast cancer | 37.59 a.u/RIU | Not given | [114] |
| 35 | Microfiber | Refractometric | PDDA/PAA polyelectrolyte multilayer | Anti-CEA antibody | Colorectal, breast, and lung cancers | Up to ~10–12 nm·(pg mL−1)−1 in the ultralow range (200 fg mL−1–1 pg mL−1) | 34.6 fg mL−1 (0.475 fM) | [115] |
| 36 | TFBG | SPR | PAN + Gold | Anti-CEA antibody | Pancreatic, gastric, breast, and lung cancers | 0.46 dB/(µg/mL) | 505.4 ng/mL | [116] |
| 37 | Tapered microfiber | Refractometric | PS@Au nanospheres | Anti-CEACAM5 antibody | Gastrointestinal, colorectal, and pancreatic, lung cancers | Not given | 3.54 × 10−17 M and 5.27 × 10−16 M for pure and serum solutions, respectively | [117] |
| 38 | Peanut structure cascaded lasso shaped fiber | Refractometric | Fe3O4 microspheres | Anti-CEACAM5 antibody | Gastrointestinal, colorectal, and pancreatic, lung cancers | 1747 nm/RIU | 0.11 ng/mL in buffer solution | [118] |
| 39 | U-shaped thin-core fiber | Refractometric | APTES | Biotin | Lung and breast cancer | 18,390 nm·RIU−1 | 49 cells·mL−1 | [119] |
| 40 | Shallow-tapered SMF | Refractometric | APTMS + Glutaraldehyde | Anti-CD44 antibody | Breast cancer | Up to 1.33 nm/RIU | 16.4 pM | [120] |
| 41 | SMF ball resonator | Refractometric | APTMS + Glutaraldehyde | Anti-CD44 antibody | Breast cancer | −85 dB/RIU (in the dynamic conditions) | Femtomolar range | [121] |
| 42 | SMF + EBF (Enhanced Backscattering Fiber) | Refractometric | APTMS + Glutaraldehyde | Anti-ALDH1A1 | Breast, lung, colorectal, prostate cancers and lymphoma | Up to 92.4 dB·RIU−1 | 172 fM | [122] |
| # | Fiber Sensor Type | Cancer Cell Type | Surface Functionalization | Modeling/Simulation Method | Simulated Outputs | Ref. |
|---|---|---|---|---|---|---|
| 1 | D-shaped polymer optical fiber (POF) SPR biosensor | HER2 protein | Gold + HER2 aptamer | FEM + multilayer Transfer Matrix Method (TMM) | Transmission spectra shifts for gold thicknesses, resonant wavelength redshift as gold thickness increases, sensitivity, FWHM (Full Width at Half Maximum) variations for each thickness, Peak FOM | [38] |
| 2 | PCF-SPR sensor | Basal, HeLa, PC-12, MDA-MB-231, MCF-7 | Hybrid Au/Ti3C2Tx (MXene) thin-film coating | FEM + Machine Learning | Resonance wavelength shift, wavelength sensitivity, confinement loss, effective refractive index (Neff), resolution, FOM, ML-predicted sensitivity and Neff | [109] |
| 3 | PCF-SPR biosensor | Basal skin, HeLa, Jurkat, PC-12, MDA-MB-231, MCF-7 | Au + MXene | FEM (Finite Element Method) with PML | Effective index of core & SPP mode, Confinement loss, Mode field distributions, Resonance wavelength (RW) shift, Sensitivity, FOM (Figure of Merit), Resolution | [110] |
| 4 | Single-core PCF-based SPR biosensor with U-shaped analyte channel | Basal (skin), Jurkat (blood), PC12/adrenocortical (adrenal gland) cancer cells | Gold (Au) plasmonic layer with V2O5 adhesion nanolayer | FEM | Resonance wavelength shift, confinement loss, wavelength sensitivity, resolution, FOM | [144] |
| 5 | Terahertz porous-core MSF (microstructure fiber) biosensor | Breast, skin, gastric cancer cells | Geometric functionalization (porous-core structure, Zeonex polymer matrix) | Full-vector FEM (Finite Element Method) with PML (Perfectly Matched Layer) boundary conditions | Relative sensitivity, Effective Material Loss, Confinement Loss, Numerical Aperture, Effective Mode Area, mode-field distribution | [155] |
| 6 | PCF (Photonic crystal fiber) SPR (Surface Plasmon Resonance) (PCF-SPR) biosensor | MDAMB-231, MCF-7, PC12, HeLa, Jurkat cells | TiO2 adhesion layer + Au plasmonic thin film | Full-vector FEM with PML | Confinement loss, resonance wavelength shift, effective index matching, amplitude sensitivity, refractive index resolution | [145] |
| 7 | Lasso-shaped SMF (single mode) fiber-laser biosensor | CEACAM5 proteins | Silanization + Anti-CEACAM5 antibody | Full-vector BPM (Beam Propagation Method) with PML conditions | Optical field distribution in straight vs. bent single-mode fiber (SMF), mode coupling between core and cladding, formation of cladding modes and Multimode Interference (MMI), evanescent field depth, sensitivity | [146] |
| 8 | Hybrid plasmonic-photonic crystal (MIM + 1D PC PBG) biosensor | Basal cell carcinoma | Ag/GaAs/air (no bioreceptors) | FDTD (Finite-Difference Time-Domain) simulation; TLM validation | Transmission spectrum, PBG wavelength shift, sensitivity, FOM | [147] |
| 9 | PCF-SPR biosensor | Basal skin, HeLa, Jurkat, PC-12, MDA-MB-231, MCF-7 | Gold + TiO2 | FEM (Finite Element Method) with PML | Confinement loss, Mode field distributions, Resonance wavelength (RW) shift, Sensitivity, FOM, Resolution | [148] |
| 10 | PCF-SPR biosensor | Basal, HeLa, Jurkat, PC-12, MDA-MB-231, MCF-7 | Gold nanowire | FEM (Finite Element Method), Drude-Lorentz model | CL spectra, resonance wavelength, WS (wavelength sensitivity), AS (amplitude sensitivity), resolution | [149] |
| 11 | LSPR (Localized Surface Plasmon Resonance) biosensor | BRCA-1 and BRCA-2 genetic 12breast cancer cells | Au film + graphene layers + immobilized probe DNA | Analytical multilayer Fresnel transfer matrix modeling; SPR angle & SRF simulations | SPR angle, Surface Resonance Frequency shift reflectance curves, effect of graphene layer number | [150] |
| 12 | SPR biosensor | Jurkat, HeLa, PC12, MDA-MB-231, MCF7 cells | Perfluorinated polymer + Ag/MoS2/polymer/graphene | TMM, Multilayer optical model | SPR angle, power loss spectrum, FWHM, sensitivity, FOM, LoD | [106] |
| 13 | PCF-SPR biosensor | MCF-7, MDA-MB-231 cells | Au + TiO2 | FEM with PML & Machine Learning analysis | Neff(core), Neff (SPP), confinement loss, sensitivity, resonance wavelength shift | [151] |
| 14 | Circular shaped HCF—SPR biosensor | A549, HepG2, MCF-7, basal cells | Graphene + MoS2 + Gold | FEM (Finite Element Method) + Machine Leaning | SPR wavelength shift, Confinement loss, wavelength sensitivity, ML-predicted optics | [107] |
| 15 | PCF-SPR biosensor | Basal cancer, MDA-MB-231, MCF-7, Jurkat, PC12, HeLa cells | Gold (Au) | FEM (Finite Element Method) + Machine Learning | Resonance wavelength shift; confinement loss (dB/cm); effective refractive index (Neff); wavelength sensitivity | [152] |
| 16 | Kretschmann-configuration plasmonic biosensor (prism-coupled SPR) | Brain tumor biomarkers | Graphene/Ag/WS2 multilayer | FEM (Finite Element Method) + transfer matrix modeling + Machine Learning | Angular reflectance spectra, resonance angle shift, sensitivity, detection limit, figure of merit, ML-simulation correlation | [153] |
| 17 | Open D-channel PCF-SPR sensor | Basal, Jurkat, HeLa, PC12, MDA-MB-231, MCF7 cells | Au/TiO2 thin-film bilayer | FEM with PML | Resonance wavelength shift; wavelength sensitivity; amplitude sensitivity; resolution; FOM | [154] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Omirzakova, A.; Mukhangaliyeva, L.; Katrenova, Z.; Aituganova, A.; Bekmurzayeva, A.; Tosi, D.; Ashikbayeva, Z. Functionalization Techniques Empowering Optical Fiber Biosensors in Label-Free Cancer Biomarker Detection. Biosensors 2026, 16, 25. https://doi.org/10.3390/bios16010025
Omirzakova A, Mukhangaliyeva L, Katrenova Z, Aituganova A, Bekmurzayeva A, Tosi D, Ashikbayeva Z. Functionalization Techniques Empowering Optical Fiber Biosensors in Label-Free Cancer Biomarker Detection. Biosensors. 2026; 16(1):25. https://doi.org/10.3390/bios16010025
Chicago/Turabian StyleOmirzakova, Aigerim, Lyazzat Mukhangaliyeva, Zhanerke Katrenova, Aida Aituganova, Aliya Bekmurzayeva, Daniele Tosi, and Zhannat Ashikbayeva. 2026. "Functionalization Techniques Empowering Optical Fiber Biosensors in Label-Free Cancer Biomarker Detection" Biosensors 16, no. 1: 25. https://doi.org/10.3390/bios16010025
APA StyleOmirzakova, A., Mukhangaliyeva, L., Katrenova, Z., Aituganova, A., Bekmurzayeva, A., Tosi, D., & Ashikbayeva, Z. (2026). Functionalization Techniques Empowering Optical Fiber Biosensors in Label-Free Cancer Biomarker Detection. Biosensors, 16(1), 25. https://doi.org/10.3390/bios16010025






